Anticoagulants are drugs that delay or prevent the clotting of blood. In a perioperative setting, patients receive anticoagulation for cardiovascular procedures, thromboprophylaxis, or for cardiovascular disease and/or atrial fibrillation. The therapeutic potential of anticoagulation must be considered against risks for increased bleeding. Many agents are also used in the perioperative setting that may not be routinely monitored, including drugs such as low-molecular-weight heparin, new oral anticoagulants that include direct thrombin inhibitors (dabigatran [Pradaxa] and factor Xa inhibitors rivaroxaban [Xarelto] and apixaban), or newer platelet inhibitors. This chapter will review the different anticoagulation agents, including antiplatelet agents, and considerations for their use in the perioperative use. The agents most commonly used will be considered in detail. Guidelines for management are published about every 4 years by the American College of Chest Physicians (ACCP) and should be referred to for more detail.1–9
Heparin
Unfractionated heparin (UFH) is an extract of porcine intestine or bovine lung, where heparin is stored in the mast cells. It is a mixture of highly sulfated glycosaminoglycans with molecular weights ranging from 3,000 to 30,000 daltons that produce their anticoagulant effects by binding to antithrombin (AT) (previously known as antithrombin III), a circulating serine protease. Heparin acts as an anticoagulant by binding to AT, enhancing the rate of thrombin–AT complex formation by 1,000 to 10,000 times. Other factors in the clotting cascade, including factor Xa but also XII, XI, and IX are also inhibited by AT.10Anticoagulation thus depends on the presence of adequate amounts of circulating AT as shown in the Figure 30-1.

Standardization of heparin potency is based on in vitro comparison with a known standard. A unit of heparin is defined as the volume of heparin-containing solution that will prevent 1 mL of citrated sheep blood from clotting for 1 hour after the addition of 0.2 mL of 1:100 calcium chloride. Heparin must contain at least 120 United States Pharmacopeia (USP) units per milliliter. Because the potency of different commercial preparations of heparin may vary greatly, the heparin dosing should always be prescribed in units, and most heparin is porcine in origin.
Pharmacokinetics
Heparin is a highly charged acidic molecule administered by intravenous (IV) or subcutaneous (SC) injection. The pharmacokinetics of heparin are based on measurements of its biologic activity using an anti-Xa assay. Over the range of heparin concentrations used clinically, the dose-response relationship is not linear for multiple reasons, including the need for AT to potentiate its effect, the effects of temperature, its highly charged nature that causes protein binding, and the variability of anticoagulation responses. The precise pathway of heparin elimination is uncertain, and the influence of renal and hepatic disease on its pharmacokinetics is less than with other anticoagulants. Heparin binds to many different proteins, which can affect its anticoagulant activity and contributes to heparin resistance.11
Laboratory Evaluation of Coagulation
The anticoagulant response to heparin varies widely especially in critically ill patients with alterations in AT and other plasma proteins. Different tests are used to monitor UFH and other anticoagulants as follows.2
Activated Partial Thromboplastin Time
Heparin treatment is usually monitored to maintain the ratio of the activated partial thromboplastin time (aPTT) within a defined range of approximately 1.5 to 2.5 times normal values, typically 30 to 35 seconds. An excessively prolonged aPTT (>120 seconds) is readily shortened by omitting a dose because heparin has a brief elimination half-time. When low-dose heparin is used, laboratory tests may not be required to monitor treatment because the dosage and schedule are well known. However, some hospital laboratories have changed to anti-Xa assays instead of aPTT monitoring because of the variability of responses, with low-dose regimens targeting levels of 0.3 to 0.5 unit/mL and high-dose regimens targeting levels 0.5 to 0.8 unit/mL.
Activated Clotting Time
At higher heparin concentrations like those typically used during cardiopulmonary bypass, the activated clotting time is used to monitor anticoagulation. The activated clotting time (ACT) is performed by mixing whole blood with an activating substance that has a large surface area, such as celite (diatomaceous earth—silicon dioxide) or kaolin (clay—aluminum silicate). This is a contact activation through the classic intrinsic pathway where factor XII initiates activation of the clotting cascade. The activator speeds up the clotting time to normal values of approximately 100 to 150 seconds, depending on the device. Several commercially available timing systems used clinically to measure the ACT are based on detecting the onset of clot formation. Nevertheless, results between different commercial devices to measure the ACT may not be interchangeable, especially if the type of activator (celite or kaolin) is different.
Heparin effect and its antagonism by protamine are commonly monitored in patients undergoing cardiovascular procedures by measuring the ACT. Because the ACT is easy to use and reliable for high heparin concentrations (>1.0 unit/mL), it has become the mainstay of heparin anticoagulation monitoring in perioperative management and for cardiac catheterization. In addition to the presence of a heparin effect, the ACT may be influenced by hypothermia, thrombocytopenia, presence of contact activation inhibitors (aprotinin), and preexisting coagulation deficiencies (fibrinogen, factor XII, factor VII). With aprotinin therapy, the recommendation is to use kaolin-ACT rather than a celite-ACT determination as kaolin binds to aprotinin to minimize its effect.
For cardiac surgery, a baseline value for the ACT is determined (a) before the IV administration of heparin, (b) 3 to 5 minutes after administration, and (c) at 30-minute intervals, thereafter. The ACT response to heparin is not linear for multiple reasons, including the need for AT for its effectiveness and because of several other factors that affect ACT. During cardiopulmonary bypass, the target ACT value is still controversial but often considered adequate if the ACT is longer than 350 seconds, although most cardiac surgical centers target an ACT of longer than 400 seconds. The need to measure ACT repeatedly is emphasized by the fourfold variation in heparin sensitivity between patients and the threefold variation in the rate at which heparin is metabolized. Furthermore, ACT values can be misleading during cardiopulmonary bypass with respect to heparin-induced anticoagulation because of the effects of hypothermia and hemodilution on the measurement system.12
Clinical Uses
Heparin is used extensively for multiple purposes including the prevention and treatment of venous thrombosis and pulmonary embolism, for acute coronary syndromes, and for perioperative anticoagulation for extracorporeal circulation and hemodialysis. When administered intravenously, heparin has an immediate onset of action, whereas SC administration results in variable bioavailability with an onset of action in 1 to 2 hours.
Heparin-Induced Thrombocytopenia
Thrombocytopenia due to UFH is common and can begin within hours in patients exposed to heparin. However, a more severe and even life-threatening syndrome develops in 0.5% to 6.0% of patients, manifesting as severe thrombocytopenia (50% drop in platelet count or <100,000 cells/mm3), that can be associated with thrombotic events (heparin-induced thrombocytopenia with thrombosis). This severe response typically develops after 4 to 5 days of heparin therapy and is caused by heparin-dependent antibodies to platelet factor IV that trigger platelet aggregation and result in thrombocytopenia (see the more detailed discussion in Physiology of Hemostasis, Chapter 27Physiology of Hemostasis).13
Allergic Reactions
Heparin can cause allergic reactions, but these are rare and present in a manner typical of other hypersensitivity reactions. In patients that do experience immediate reactions, heparin-induced thrombocytopenia (HIT) should also be suspected due to the presence of preformed antibodies. Rapid IV infusion of large doses of heparin usually causes minimal hemodynamic changes.13
Reversal of Heparin-Induced Anticoagulation with Protamine
Protamine is one of the few agents available for reversing anticoagulation. Protamine is a strongly alkaline (nearly two-thirds of the amino acid composition is arginine), polycationic, low-molecular-weight protein found in salmon sperm. The positively charged alkaline protamine combines with the negatively charged acidic heparin to form a stable complex that is devoid of anticoagulant activity. These heparin–protamine complexes are removed by the reticuloendothelial system. Clearance of protamine by the reticuloendothelial system (within 20 minutes) is more rapid than heparin clearance and that may explain, in part, the phenomenon of heparin rebound. The dose of protamine required to antagonize heparin is typically 1 mg for every 100 units of circulating heparin activity. A more specific dose of protamine is calculated by heparin-protamine titration. Most clinicians give too much protamine because they reverse based on the total dose or heparin administered without accounting for heparin elimination prior to the administration of protamine. Heparin has a half life of approximately one hour, so determinations of protamine dosing should include considerations of the circulating heparin level for reversal. (See also “Protamine” in Chapter 29, Procoagulants.)
Low-Molecular-Weight Heparins
Enoxaparin and dalteparin are two commonly administered low-molecular-weight heparins (LMWHs) derived from standard commercial-grade UFH by chemical depolymerization to yield fragments with a mean molecular weight of 4,000 to 5,000 daltons. Depolymerization of heparin results in a change in its anticoagulant profile, pharmacokinetics, and effects on platelet function. Compared with heparin, which has an anti-Xa to anti-IIa activity of about 1:1, enoxaparin has a corresponding ratio that varies between 4:1 and 2:1.14 The pharmacokinetics of enoxaparin and dalteparin between patients are more consistent than heparin because these drugs bind less avidly to proteins than heparin. This contributes to better bioavailability at low doses. Although protection against venous thromboembolism (VTE) in high-risk medical and surgical patients is often thought to be better with LMWH than with heparin, LMWH’s effect is greatly prolonged with renal failure and anticoagulants such as UFH should be used in this population. Therefore, care should be taken to delay surgery for 12 hours after the last dose of LMWH in patients with normal renal function and longer with renal dysfunction. Protamine does not neutralize LMWH.2,14
Spinal and Epidural Hematomas
The risk of spontaneous hematoma formation may be increased in the presence of LMWH and indwelling epidural catheters for administration of postoperative analgesia and by concomitant use of other drugs that affect hemostasis (nonsteroidal antiinflammatory drugs, platelet inhibitors) and by traumatic or repeated attempts to accomplish entry into the epidural or subarachnoid space. This increased risk of hematoma formation is a consideration when selecting regional anesthesia in patients being treated with LMWH preparations. Recommendations for management of patients for regional anesthesia in the patient receiving antithrombotic or thrombolytic therapy are reported in American Society of Regional Anesthesia and Pain Medicine Evidence-Based Guidelines (Third Edition).15
Fondaparinux
Fondaparinux is a synthetic anticoagulant composed of the five saccharide units that make up the active site of heparin that binds AT, such as LMWH, to inhibit factor Xa but has no direct activity against thrombin. Administered subcutaneously, fondaparinux is rapidly absorbed and has an elimination half-time of 15 hours, allowing for once daily administration. Metabolism does not occur and the drug is eliminated by the kidneys and should not be used in patients with renal failure. Clinical uses of fondaparinux include prevention of deep vein thrombosis (DVT) and pulmonary embolism and as an alternate anticoagulant in patients with HIT. Because of its long duration of action, it is used primarily in patients with HIT or concerns about sensitization.16
Danaparoid
Danaparoid is a glycosaminoglycuronan that is derived from porcine intestinal mucosa and consists of a mixture mostly of dermatan sulfate, and chondroitin sulfate. This low-molecular-weight heparinoid compound attenuates fibrin formation principally by binding AT. Elimination of danaparoid is predominately through the kidneys. Danaparoid is effective in decreasing the incidence of DVT following total hip arthroplasty and was used for the treatment of HIT; this agent is no longer available in the United States (it was removed from the U.S. market in 2002 due to a shortage in drug substance) but is still in use in other countries.
Prophylaxis against Venous Thromboembolism
Surgical procedures have been associated with a 20-fold increase in risk for VTE, which is understandable considering that the majority of surgical patients have one or more risk factors for developing VTE.17The incidence of DVT is 10% to 40% among general surgery patients, and higher still in high-risk surgery patient populations (e.g., orthopedic, thoracic, cardiac, and vascular surgery).2,3 Fortunately, thromboprophylaxis is known to effectively reduce VTE in a cost-effective manner.2 However, despite their effectiveness, there are specific challenges regarding the use of currently recommended anticoagulants.7
To prevent VTE, patients are treated with anticoagulants. Although SC heparin and LMWH are commonly used, multiple novel agents are also approved for different indications including fondaparinux, rivaroxaban, and dabigatran with different indications depending on the country. Enoxaparin and dalteparin are commonly used LMWHs. Before the availability of LMWH, low-dose heparin, 5,000 units subcutaneously every 8 to 12 hours, was a common regimen. In those with renal failure or renal dysfunction, heparin and warfarin are the only drugs minimally affected because of nonrenal clearance.
Among surgical patients, those undergoing total hip replacement are at unique risk for developing DVT and many of the studies for approval of new anticoagulants have focused on this group and other orthopedic patients. The risk of DVT is more protracted after hip surgery than after general surgery, when it usually develops during the first few postoperative days. The surgical technique for hip surgery, which kinks the femoral vein, seems to stimulate proximal DVT in the operated leg, whereas calf vein thrombosis is more likely to develop in either leg. Another effect unique to hip surgery is impairment of venous hemodynamics, which may last several weeks in the operated leg. Indeed, there are significantly fewer venous thromboembolic complications in patients undergoing elective hip replacement when prophylaxis with LMWH is given for 1 month rather than only during the hospitalization. VTE is also a common, life-threatening complication of major trauma. Pulmonary embolism has been observed to occur in 2% to 22% of patients with major trauma, and fatal pulmonary embolism is the third most common cause of death in patients who survive the first 24 hours.7,17
Direct Thrombin Inhibitors: Parenteral Agents
An important class of anticoagulants that high-risk surgery patients at risk for HIT may receive are the direct thrombin inhibitors, including bivalirudin, argatroban, lepirudin, and desirudin (Table 30-1). Bivalirudin is also commonly used for cardiac interventional procedures. The direct thrombin inhibitors also vary in their binding affinities for thrombin. Desirudin, lepirudin, and bivalirudin bond in a bivalent manner to thrombin by interacting with both the catalytic site and fibrinogen-binding site. Bivalent direct thrombin inhibitors show higher affinity and specificity for thrombin compared with univalent direct thrombin inhibitors, which bind to the catalytic site only. Direct thrombin inhibitors vary substantially in their pharmacokinetic properties in terms of half life and metabolism. There are also differences in immunogenicity between the direct thrombin inhibitors and with 40% to 70% of patients developing antihirudin antibodies after 4 or more days of treatment.18

Bivalirudin
Bivalirudin, a synthetic analog of hirudin with a half-life of 25 minutes, has been widely studied in patients with and without acute coronary syndromes undergoing percutaneous coronary intervention (PCI). This agent is indicated for use in patients with unstable angina undergoing percutaneous transluminal coronary angioplasty (PTCA); in patients with or at risk for HIT or HIT with thrombosis syndrome (HITTS) undergoing PCI; and with provisional use of glycoprotein (GP) IIb/IIIa inhibitors in patients undergoing PCI. Although it is a polypeptide, bivalirudin is considered a safe anticoagulant in patients with HIT. In patients with HIT antibodies undergoing cardiopulmonary bypass, bivalirudin provided safe and effective anticoagulation, with a 94% success rate for the procedures.19 Further, multiple studies have demonstrated its application as a heparin replacement in patients who are HIT positive and require on or off pump cardiac surgery, although this is an off-label use for the drug.19–21
Argatroban
Argatroban is an injectable, synthetic, univalent direct thrombin inhibitor indicated for prophylaxis or treatment of thrombosis in patients with or at risk of HIT undergoing PCI. It has a relatively short half-life of 40 to 50 minutes, and anticoagulation returns to baseline when stopping it after approximatrly 4 hours.22 Patients with HIT are likely to have renal dysfunction and most of the agents used for HIT are all primarily renally eliminated. Argatroban is hepatically eliminated, thus no dose adjustments are required in patients with renal impairment. As lepirudin is renally eliminated and bivalirudin is partially (~20%) renally eliminated, their use may require dose adjustment in renally impaired patients to avoid accumulation. Antibodies to argatroban have not been detected after prolonged or repeated use due to its low molecular weight.23
Lepirudin and Desirudin
Lepirudin and desirudin are recombinant hirudins, synthetic analogs of hirudin, the direct thrombin inhibitor first isolated from leeches as Hirudo medicinalis is the name of the leech. These proteins are manufactured by recombinant methods. Lepirudin is approved for use in patients with HIT and associated thromboembolic disease to prevent further thromboembolic complications. Lepirudin was initially reported for cardiac surgical patients; however, bleeding was a major problem due to its ability to irreversibly inhibit thrombin. HIT patients receiving lepirudin generate antibodies and require close monitoring (using aPTT) to avoid bleeding complications. In patients with renal dysfunction the drug may have a prolonged half-life.16,23
Desirudin (another recombinant hirudin) is approved for use in Europe and now in the United States for the prevention of VTE after total hip or knee replacement surgery and has been studied extensively in patients with stable angina or acute coronary syndromes undergoing PTCA. Antigenicity and anaphylaxis are also reported, although the risk of hypersensitivity to desirudin appears relatively low. Because desirudin is primarily eliminated by the kidneys, patients with renal impairment require monitoring and the aPTT can be used.16
Oral Anticoagulants
Vitamin K Antagonists—Warfarin
Oral anticoagulants are derivatives of 4-hydroxycoumarin (coumarin). Warfarin is the most frequently used anticoagulant because of its predictable onset and duration of action and its excellent bioavailability after oral administration (Table 30-2). Treatment usually begins with an oral warfarin dose of 5 to 10 mg, and the average maintenance dose is 5 mg; however, the dose varies widely among individuals due to pharmacogenetic differences. Warfarin has been the only oral agent available until the recent approval of new agents that are described in the sections that follow. Disadvantages of warfarin include delayed onset of action, the need for regular laboratory monitoring, and difficulty in reversal should a surgical procedure create concern about bleeding.3

Mechanism of Action
Warfarin inhibits vitamin K epoxide reductase that converts the vitamin K–dependent coagulation proteins (factors II [prothrombin], VII, IX, and X) to their active form, a posttranslational modification. The anticoagulant effect of oral or IV warfarin is delayed for 8 to 12 hours, reflecting the onset of inhibition of clotting factor synthesis and the elimination half-time of previously formed clotting factors that are not altered by the oral anticoagulant. Peak effects of warfarin do not occur for 36 to 72 hours.3
Pharmacokinetics
Warfarin is rapidly and completely absorbed, with peak concentrations occurring within 1 hour after ingestion. It is 97% bound to albumin, and this contributes to its negligible renal excretion and long elimination half-time of 24 to 36 hours after oral administration. Warfarin, however, does cross the placenta and produces exaggerated effects in the fetus, who has limited ability to synthesize clotting factors. Warfarin is metabolized to inactive metabolites that are conjugated with glucuronic acid and ultimately excreted in bile (enterohepatic circulation) and urine.
Laboratory Evaluation
Treatment with oral anticoagulants is best guided by measurement of the prothrombin time. The prothrombin time is particularly sensitive to three of the four vitamin K–dependent clotting factors (prothrombin and factors VII and X). Commercial prothrombin time reagents vary markedly in their responsiveness to warfarin-induced decreases in clotting factors. Therefore, prothrombin time results obtained with different reagents are not interchangeable between laboratories. This problem of variability in the responsiveness of prothrombin time reagents has been overcome by the introduction of a standardized system of reporting known as the international normalized ratio (INR). Each manufacturer assigns a specific value that indicates how a particular batch of tissue factor compares to an international reference tissue. The INR is the ratio of a patient’s prothrombin time to a normal (control) sample, adjusted by the factor assigned by the manufacturer for the batch of factor being used in the assay. For most indications, a moderate anticoagulant effect with a targeted INR of 2.0 to 3.0 is appropriate, including prosthetic valve prophylaxis. An excessively prolonged prothrombin time is not readily shortened by omitting a dose because of the long elimination half-time of oral anticoagulants. Likewise, an inadequate therapeutic effect is not readily corrected by increasing the dose because of the delayed onset of therapeutic effect.
Unexpected fluctuations in the dose response to warfarin may reflect changes in diet, undisclosed drug use, poor patient compliance, surreptitious self-medication, or intermittent alcohol consumption. Concomitant medication with over-the-counter and prescription drugs can augment or inhibit the anticoagulant effect of coumarin drugs on hemostasis or interfere with platelet function. Patients receiving coumarin drugs are sensitive to fluctuating levels of dietary vitamin K, which is obtained predominantly from leafy green vegetables. The effect of coumarin can be potentiated in sick patients with poor vitamin K intake, particularly if they are treated with antibiotics and IV fluids without vitamin K supplementation. Preexisting liver disease and advanced age are associated with enhanced effects of oral anticoagulants.3,24
Clinical Uses
Vitamin K antagonists (VKAs) are effective in the prevention of VTE, the prevention of systemic embolization and resultant stroke in patients with prosthetic heart valves or atrial fibrillation, and for treatment of patients with thrombophilia who are hypercoagulable. Because of the extensive new range of oral anticoagulants for perioperative VTE prophylaxis, these agents are used less and less for this indication.
Management before Elective Surgery
In patients receiving a VKA, the INR should be checked preoperatively. Although minor surgical procedures can be safely performed in patients receiving oral anticoagulants, for major surgery, discontinuation of oral anticoagulants 1 to 3 days preoperatively is recommended to permit the prothrombin time to return to within 20% of its normal range. This approach, followed by reinstitution of the oral anticoagulant regimen 1 to 7 days postoperatively, is not accompanied by an increased incidence of thromboembolic complications in vulnerable patients. However, patients at high risk, such as those with prosthetic heart valves, may require bridging with UFH.3
Bleeding is the main complication of any anticoagulant therapy, including the VKAs. The risk of bleeding is influenced by the intensity of the anticoagulant therapy, the patient’s underlying disorder, and the concomitant use of aspirin. Bleeding that occurs when the INR is less than 3.0 are frequently associated with an obvious underlying cause (neoplasm, peptic ulcer). These drugs may increase the incidence of intracranial hemorrhage after a cerebrovascular accident. Compression neuropathy has been observed in treated patients after brachial artery puncture to obtain a sample for blood gas analysis. Treatment of bleeding depends on the severity and underlying patient and location. In emergency situations, oral or IV administration of vitamin K is used but will not immediately reverse the anticoagulant effect. If immediate reversal is needed, for performance of high-risk surgical procedures such as craniotomy, administration of prothrombin complex concentrates (PCCs) is needed if available or other reversal strategies as defined in Chapter 29 (Procoagulants).3,25 Guidelines for perioperative management are available.1
New Oral Agents
For many years, warfarin has been the only oral anticoagulant available but has variabilities regarding dosing and effects and requires frequent monitoring and may take up to 5 days before therapeutic levels can be obtained. The newer therapeutic agents have a rapid onset with therapeutic anticoagulation within hours of administration and do not need routine monitoring. Dabigatran is an oral direct thrombin inhibitor, and rivaroxaban is a direct factor Xa inhibitor similar to LMWH.16 Both of the newer agents require dose adjustments for renal failure and will be considered separately, along with agents still under investigation.9,16 (Table 30-2)
Direct Factor Xa Inhibitors
Rivaroxaban (Xarelto)
Rivaroxaban is an oral, direct factor Xa inhibitor with >10,000-fold greater selectivity for factor Xa than for other related serine proteases. In contrast to LMWH and similar agents, rivaroxaban does not require AT as a cofactor. Direct factor Xa inhibitors, including rivaroxaban, can inhibit free factor Xa, clot-bound factor Xa, and factor Xa bound to the prothrombinase complex unlike indirect factor Xa inhibitors, such as fondaparinux, which are unable to inhibit factor Xa within the prothrombinase complex. Rivaroxaban is also a non-heparin-like molecule that may be suitable for the management of patients with HIT. Rivaroxaban has been approved for the reduction of risk of stroke and systemic embolism in nonvalvular atrial fibrillation, prophylaxis of deep vein thrombosis following hip or knee replacement surgery, treatment of DVTs, treatment of pulmonary embolism (PE), and reducing the risk of recurrence of DVTs and PE.9,16
When used with neuraxial anesthesia, an epidural catheter should not be removed earlier than 18 hours after the last administration of rivaroxaban, and the next rivaroxaban dose should be administered no earlier than 6 hours after the removal of the catheter and as noted in the manufacturer’s package insert.
Apixaban
Apixaban is another oral, direct factor Xa inhibitor administered twice daily. Like rivaroxaban, apixiban is approved for the reduction of risk of stroke and systemic embolism in nonvalvular atrial fibrillation, prophylaxis of deep vein thrombosis following hip or knee replacement surgery, treatment of DVTs, treatment of pulmonary embolism (PE), and reducing the risk of recurrence of DVTs and PE.
Other Direct Factor Xa Inhibitors under Investigation
Several other direct factor Xa inhibitors are under investigation and are approved in certain countries. Edoxaban is being evaluated for atrial fibrillation and DVT prophylaxis.
Direct Thrombin Inhibitors
Ximelagatran
Ximelagatran is an oral, direct thrombin inhibitor that was approved in Europe for the VTE prophylaxis but later withdrawn from the market in 2006 due to concerns over potential liver toxicity. However, ximelagatran provided proof of principle that oral agents that act via direct inhibition of thrombin were an effective mode of action for new anticoagulants.16
Dabigatran Etexilate (Pradaxa)
Dabigatran etexilate is an oral, direct thrombin inhibitor approved to reduce the risk of stroke and systemic embolism in patients with non-valvular atrial fibrillation, for the treatment of DVT and PE in patients who have been treated with a parenteral anticoagulant for 5-10 days, and to reduce the risk of recurrence of DVT and PE in patients who have been previously treated. Dabigatran’s effects can be measured best by thrombin times and also by aPTT values, although thrombin times are preferred.9 Administration of the first dose should occur a minimum of 2 hours after the catheter is removed, and patients should be observed for neurologic signs and symptoms.9,16 Dosing should also be adjusted for patients with renal dysfunction.
Perioperative Management of the New Oral Anticoagulants
The newer therapeutic agents have a rapid onset with therapeutic anticoagulation within hours of administration and do not need routine monitoring. Dabigatran is an oral direct thrombin inhibitor, and rivaroxaban is a direct factor Xa inhibitor similar to LMWH.16 Both of the newer agents require dose adjustments for renal failure.16 One of the new challenges with use of these agents is how to manage patients perioperatively. In the United States, warfarin is still a problem for clinicians because the PCCs effective for immediate INR reversal are not available.25 Vitamin K takes days to work; use of 4 units of fresh frozen plasma (FFP) is often used, but associated with transfusion risk and volume overload, and FFP never restores the INR to baseline, but usually to approximately 1.4 to 1.6 which is the baseline INR for FFP.25
The French Study Group on thrombosis and hemostasis has proposed perioperative management strategies regarding the risk of bleeding and thrombosis for the new agents. The newer oral anticoagulants may increase surgical bleeding, they have no validated antagonists, they cannot be monitored by simple standardized laboratory assays, and their pharmacokinetics vary significantly between patients.26 For procedures with low hemorrhagic risk, a therapeutic window of 48 hours (last administration 24 hours before surgery, restart 24 hours after) is proposed. For procedures with medium or high hemorrhagic risk, they suggest stopping therapy 5 days before surgery to ensure complete elimination in all patients. Treatment should be resumed only when the risk of bleeding subsides. In patients at high thrombotic risk (e.g., those in atrial fibrillation with a history of stroke), bridging with heparin (LMWH, or UFH, if the former is contraindicated) is proposed. In an emergency, the procedure should be postponed for as long as possible (minimum 1 to 2 half-lives) and nonspecific antihemorrhagic agents, such as recombinant human activated factor VIIa or PCCs should not be given for prophylactic reversal due to their uncertain benefit-risk.26
Although routine monitoring of the new anticoagulants is not standard, if needed they are best evaluated with specialized tests. For dabigatran, thrombin clotting time (TT), ecarin clotting time (ECT), and activated partial thromboplastin time (aPTT) can measure its effects.27 Prothrombin time (INR) is not recommended. The aPTT, a standard test, can provide a useful qualitative assessment of anticoagulant activity but is less sensitive at supratherapeutic dabigatran levels and limited data exists for activated clotting time (ACT). Overall, the aPTT and TT are the most accessible qualitative methods for determining the presence or absence of anticoagulant effect. Although there is no specific antidote to antagonize the anticoagulant effect of dabigatran, because of its short duration of effect, drug discontinuation should be considered as previously noted. With overdose, dabigatran can also be dialyzed in patients with renal impairment. In instances of life-threatening bleeding, where conventional measures have failed or are unavailable, other prohemostatic agents such as recombinant activated factor VII and PCCs can be considered.27 For rivaroxaban, prolongation of most standard hemostatic tests are too variable and specialized tests evaluating anti-Xa are required.28 Recent data also suggests PCCs completely reverse the anticoagulant effect of rivaroxaban in healthy subjects but have minimal effects on dabigatran at the PCC doses of 50 IU/kg used in the study.29
In summary, for those who require urgent surgery, managing patients who receive dabigatran, rivaroxaban, and other novel oral anticoagulants is still not well defined. Risk versus benefit should be considered in logical decision making. It is important to note the therapies for reversal are off-label uses from the literature as referenced but provide important perspective for perioperative management for the clinician faced with managing patients receiving these agents.
Platelet Inhibitors
Aspirin
Antiplatelet agents are the mainstay therapy for patients with atherosclerotic vascular disease and coronary artery disease, therapy consistent with the role of platelets in atherosclerosis.30 Treatment with aspirin reduces the incidence of occlusive arterial vascular events. Aspirin irreversibly acetylates cyclooxygenase and thereby prevents formation of thromboxane A2. Despite rapid clearance from the body, the effects of aspirin on platelets are irreversible and last for the life of the platelet, 7 to 10 days. The ACCP’s widely quoted guidelines suggest in patients who require temporary interruption of aspirin- or clopidogrel-containing drugs before surgery or a procedure, stopping this treatment 7 to 10 days before the procedure is recommended over stopping this treatment closer to surgery. In patients who have had temporary interruption of aspirin therapy because of surgery or a procedure, resuming aspirin approximately 24 hours (or the next morning) after surgery when there is adequate hemostasis is recommended instead of resuming aspirin closer to surgery.1
Thienopyridines: Clopidogrel, Prasugrel, and Ticagrelor
Currently approved thienopyridines include clopidogrel (Plavix), prasugrel (Effient), and ticagrelor. The first two agents are prodrugs requiring in vivo metabolism each to an active metabolite as shown in Table 30-3. Ticlopidine is now rarely used clinically and will not be considered. Thienopyridines irreversibly bind to P2Y12 receptors thereby blocking adenosine diphosphate (ADP) binding. This P2Y12receptor antagonism inhibits ADP-mediated platelet activation and aggregation due to the critical role ADP plays in platelet function. When ADP is secreted from internal stores, it amplifies platelet responses induced by other platelet agonists to increase activation, an internal to external signaling mechanism.31 The ADP-induced signal is again mediated by P2Y receptors, which are G-coupled 7-membrane-spanning proteins that are present in many different cells.31 There are multiple other P2Y receptor subgroups, but the Gi-coupled P2Y12 receptor mediates inhibition of adenylyl cyclase and amplifies the platelet aggregation response.31
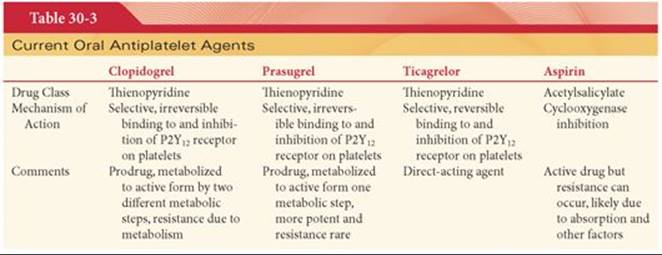
Dual antiplatelet therapy—a thienopyridine (ADP P2Y12 receptor antagonist) coadministered with aspirin— is commonly used for improving clinical outcomes in patients with acute coronary syndrome and undergoing percutaneous intervention. New agents include prasugrel and ticagrelor. Ticagrelor is a reversible, direct-acting P2Y12 receptor antagonist. The P2Y12 is a Gi-coupled platelet receptor for ADP that plays a central role in platelet function. Drugs that inhibit P2Y12 are potent antithrombotic drugs. Clopidogrel is the most widely used agent, but resistance, as defined as its inability to inhibit adequately P2Y12-dependent platelet function, occurs in 20% to 30% of patients. Prasugrel and ticagrelor appear to be more effective than clopidogrel in preventing thrombosis, although they increase the incidence of major bleeding, a problem with the efficacy of all anticoagulants.
Current recommendations are to discontinue thienopyridines 7 days before elective surgery and to avoid regional anesthesia until the effects of these drugs have dissipated. Guidelines for management of patients with coronary stents on antiplatelet agents have been proposed and are often elaborate (Table 30-4). The guidelines take in to consideration the type of coronary stent used and the interval since the stent was placed as well as the urgency of need for surgery in decision making.

Dipyridamole
Dipyridamole is an agent that increases cyclic adenosine monophosphate in platelets to inhibit their function. This agent was also used for cardiac stress testing because of its coronary vasodilatory effects (dipyridamole-thallium stress test). Currently, it is most frequently administered in combination with aspirin to prevent stroke in patients who cannot take a thienopyridine. It can increase bleeding and should be stopped preoperatively, but the aspirin component has a longer half life than dipyridamole.
Dextran
Dextran-70 (70,000 daltons) binds to platelets and inhibits their function. This agent was used clinically to reduce thrombosis after carotid surgery and a few other indications but is now rarely used for this indication.
Platelet Glycoprotein IIb/IIIa Antagonists
An important advance in managing ischemic cardiovascular disease was the development of platelet GP IIb/IIIa receptor inhibitors, although these agents are now often replaced with newer therapies. The IIb/IIIa receptor antagonists (abciximab, tirofiban, eptifibatide) either bind or competitively inhibit the corresponding fibrinogen receptor that is important for platelet aggregation. These drugs block fibrinogen binding to platelet GP IIb/IIIa receptors that are a common pathway of platelet aggregation. In multiple clinical trials, they have provided proof of concept on the critical role that platelet inhibition has in reducing ischemic events associated with acute coronary syndrome and PCIs. In recent years, thienopyridines and direct thrombin inhibitor (bivalirudin) in addition to PCIs including stenting have had a significant impact; nonetheless, inhibiting platelet function has been critical to prevent platelet responses to vascular injury and clot formation. These agents prevent thrombus formation initiated by platelets is in the pathogenesis of acute coronary syndrome (unstable angina, myocardial infarction), angioplasty failure, and stent thrombosis.32,33 A summary of these agents is given in Table 30-5.

Various antagonists of GP IIb/IIIa are available. The first of these agents, the monoclonal antibody abciximab (ReoPro), was been approved for use in PCI. Tirofiban (Aggrastat), a nonpeptide, for treatment of acute coronary syndromes (unstable angina or non–Q-wave myocardial infarction) and eptifibatide (Integrelin), a peptide, for use both in PCI and acute coronary syndromes. New nonpeptide oral antagonists of GP IIb/IIIa intended for long-term use are in various stages of clinical development and may find application in a broad spectrum of atherothrombotic disease. Although GP IIb/IIIa antagonists are indicated for the acute coronary syndrome and in patients undergoing interventional cardiology procedures, thienopyridines have largely replaced these agents due to cost and increasing clinical data favoring the thienopyridines. Abciximab has the longest half-life of all these agents as a monoclonal antibody, whereas the other agents have shorter half-lives. All of three agents can cause thrombocytopenia.33
Perioperative Management of Patients on Platelet Inhibitors
Perioperative management of patients on various platelet inhibitors is complex and requires careful coordinated care with multiple specialties. The risks and benefits of discontinuing antiplatelet therapy must be carefully considered for each individual patient, especially prior to elective surgery. The most recent guidelines by Fleisher et al.34 are listed in Table 30-4.
Thrombolytic Drugs
Pharmacologic thrombolysis is produced by drugs that act as plasminogen activators to convert the endogenous proenzyme plasminogen to the fibrinolytic enzyme plasmin that lysis clot and other proteins. The goal of thrombolytic therapy is to restore circulation through a previously occluded artery or vein, most often a coronary artery. Fibrinolytic therapy was used previously in the treatment of acute coronary syndrome but current American College of Cardiology and American Heart Association published evidence-based guidelines for the management of patients depend on whether a conservative (i.e., noninvasive) approach or an invasive strategy (i.e., PCI with possible angioplasty or coronary artery bypass graft [CABG] surgery) is possible, specifically whether cardiac catheterization is available.
Acute interventions with fibrinolytic agents can be lifesaving in patients with pulmonary emboli,35 ischemic stroke (e.g., middle cerebral arterial occlusion),6,36 and in patients suffering acute myocardial infraction without immediate access to PCIs.37 Bleeding complications (5% to 30%) may occur whether fibrinolytics are injected systemically or directly into the affected arterial lesion.38 Currently available fibrinolytics include streptokinase, urokinase, and tissue plasminogen activator (tPA). These agents activate plasminogen to plasmin, the major enzyme responsible for clot breakdown. Plasmin is a serine protease that degrades fibrin (ogen) and factors V and VIII. In clinical practice, tPA is most commonly used because of its localized catalytic effect on plasminogen activation in the presence of fibrin.39 Blood flow to the thrombus is vital for the delivery of tPA, and thus localized activation of fibrinolysis via catheter-directed drug delivery is theoretically more favorable than systemic administration.
Thrombolytic agents have an associated risk of bleeding (particularly intracranial hemorrhage) and hemorrhagic complications occur more often in trauma, surgery, or following invasive diagnostic procedures. Intracranial hemorrhage occurs in 1.7% to 8.0% of treated patients.40 Following lytic therapy, hemorrhagic transformation of ischemic infarcts can occur. The recommended treatment of intracranial or serious systemic bleeding after thrombolytic therapy is administration of cryoprecipitate and platelets, although evidence-based guidelines for such an approach are lacking.40 Angioedema occurs in 1% to 5% of patients receiving IV rt-PA, and the use of angiotensin-converting enzyme inhibitors is strongly associated with this complication.40
References
1. Douketis JD, Spyropoulos AC, Spencer FA, et al; American College of Chest Physicians. Perioperative management of antithrombotic therapy: Antithrombotic Therapy and Prevention of Thrombosis, 9th ed: American College of Chest Physicians Evidence-Based Clinical Practice Guidelines. Chest. 2012;141(2)(suppl):e326S–e350S.
2. Gould MK, Garcia DA, Wren SM, et al; American College of Chest Physicians. Prevention of VTE in nonorthopedic surgical patients: Antithrombotic Therapy and Prevention of Thrombosis, 9th ed: American College of Chest Physicians Evidence-Based Clinical Practice Guidelines. Chest. 2012;141(2)(suppl):e227S–e277S.
3. Weitz JI, Eikelboom JW, Samama MM; American College of Chest Physicians. New antithrombotic drugs: Antithrombotic Therapy and Prevention of Thrombosis, 9th ed: American College of Chest Physicians Evidence-Based Clinical Practice Guidelines. Chest. 2012;141(2)(suppl):e120S–e151S.
4. Whitlock RP, Sun JC, Fremes SE, et al; American College of Chest Physicians. Antithrombotic and thrombolytic therapy for valvular disease: Antithrombotic Therapy and Prevention of Thrombosis, 9th ed: American College of Chest Physicians Evidence-Based Clinical Practice Guidelines. Chest. 2012;141(2)(suppl):e576S–e600S.
5. Linkins LA, Dans AL, Moores LK, et al; American College of Chest Physicians. Treatment and prevention of heparin-induced thrombocytopenia: Antithrombotic Therapy and Prevention of Thrombosis, 9th ed: American College of Chest Physicians Evidence-Based Clinical Practice Guidelines. Chest. 2012;141(2)(suppl):e495S–e530S.
6. Ageno W, Gallus AS, Wittkowsky A, et al; American College of Chest Physicians. Oral anticoagulant therapy: Antithrombotic Therapy and Prevention of Thrombosis, 9th ed: American College of Chest Physicians Evidence-Based Clinical Practice Guidelines. Chest. 2012;141(2)(suppl):e44S–e88S.
7. Kearon C, Akl EA, Comerota AJ, et al; American College of Chest Physicians. Antithrombotic therapy for VTE disease: Antithrombotic Therapy and Prevention of Thrombosis, 9th ed: American College of Chest Physicians Evidence-Based Clinical Practice Guidelines. Chest. 2012;141(2)(suppl):e419S–e494S.
8. Eikelboom JW, Hirsh J, Spencer FA, et al. Antiplatelet drugs: Antithrombotic Therapy and Prevention of Thrombosis, 9th ed: American College of Chest Physicians Evidence-Based Clinical Practice Guidelines. Chest. 2012;141(2)(suppl):e89S–e119S.
9. Garcia DA, Baglin TP, Weitz JI, et al; American College of Chest Physicians. Parenteral anticoagulants: Antithrombotic Therapy and Prevention of Thrombosis, 9th ed: American College of Chest Physicians Evidence-Based Clinical Practice Guidelines. Chest. 2012;141(2)(suppl):e24S–e43S.
10. Levy JH. Novel intravenous antithrombins. Am Heart J. 2001;141:1043–1047.
11. Hirsh J. Heparin. N Engl J Med. 1991;324:1565–1574.
12. Despotis GJ, Gravlee G, Filos K, et al. Anticoagulation monitoring during cardiac surgery: a review of current and emerging techniques. Anesthesiology. 1999;91:1122–1151.
13. Levy JH, Tanaka KA, Hursting MJ. Reducing thrombotic complications in the perioperative setting: an update on heparin-induced thrombocytopenia. Anesth Analg. 2007;105:570–582.
14. Weitz JI. Low-molecular-weight heparins. N Engl J Med. 1997;337:688–698.
15. Horlocker TT, Wedel DJ, Rowlingson JC, et al. Executive summary: regional anesthesia in the patient receiving antithrombotic or thrombolytic therapy: American Society of Regional Anesthesia and Pain Medicine Evidence-Based Guidelines (Third Edition). Reg Anesth Pain Med. 2010;35:102–105.
16. Levy JH, Key NS, Azran MS. Novel oral anticoagulants: implications in the perioperative setting. Anesthesiology. 2010;113:726–745.
17. Geerts WH, Bergqvist D, Pineo GF, et al. Prevention of venous thromboembolism: American College of Chest Physicians Evidence-Based Clinical Practice Guidelines (8th Edition). Chest. 2008;133:381S–453S.
18. Di Nisio M, Middeldorp S, Buller HR. Direct thrombin inhibitors. N Engl J Med. 2005;353:1028–1040.
19. Koster A, Dyke CM, Aldea G, et al. Bivalirudin during cardiopulmonary bypass in patients with previous or acute heparin-induced thrombocytopenia and heparin antibodies: results of the CHOOSE-ON trial. Ann Thorac Surg. 2007;83:572–577.
20. Koster A, Spiess B, Jurmann M, et al. Bivalirudin provides rapid, effective, and reliable anticoagulation during off-pump coronary revascularization: results of the “EVOLUTION OFF” trial. Anesth Analg. 2006;103:540–544.
21. Merry AF, Raudkivi PJ, Middleton NG, et al. Bivalirudin versus heparin and protamine in off-pump coronary artery bypass surgery. Ann Thorac Surg. 2004;77:925–931; discussion 931.
22. McKeage K, Plosker GL. Argatroban. Drugs. 2001;61:515–522; discussion 523–524.
23. Levy JH, Hursting MJ. Heparin-induced thrombocytopenia, a prothrombotic disease. Hematol Oncol Clin North Am. 2007;21:65–88.
24. Vermeer C, Schurgers LJ. A comprehensive review of vitamin K and vitamin K antagonists. Hematol Oncol Clin North Am. 2000;14:339–353.
25. Levy JH, Tanaka KA, Dietrich W. Perioperative hemostatic management of patients treated with vitamin K antagonists. Anesthesiology. 2008;109:918–926.
26. Sie P, Samama CM, Godier A, et al. Surgery and invasive procedures in patients on long-term treatment with direct oral anticoagulants: thrombin or factor-Xa inhibitors. Recommendations of the Working Group on perioperative haemostasis and the French Study Group on thrombosis and haemostasis. Arch Cardiovasc Dis. 2011;104:669–676.
27. van Ryn J, Stangier J, Haertter S, et al. Dabigatran etexilate—a novel, reversible, oral direct thrombin inhibitor: interpretation of coagulation assays and reversal of anticoagulant activity. Thromb Haemost. 2010;103:1116–1127.
28. Samama MM, Contant G, Spiro TE, et al. Evaluation of the anti-factor Xa chromogenic assay for the measurement of rivaroxaban plasma concentrations using calibrators and controls. Thromb Haemost. 2011;107:379–387.
29. Eerenberg ES, Kamphuisen PW, Sijpkens MK, et al. Reversal of rivaroxaban and dabigatran by prothrombin complex concentrate: a randomized, placebo-controlled, crossover study in healthy subjects. Circulation. 2011;124:1573–1579.
30. Schneider DJ, Sobel BE. Conundrums in the combined use of anticoagulants and antiplatelet drugs. Circulation. 2007;116:305–315.
31. Cattaneo M. The platelet P2Y receptor for adenosine diphosphate: congenital and drug-induced defects. Blood. 2010;117:2102–2112.
32. Levy JH, Smith PK. Platelet inhibitors and cardiac surgery. Ann Thoracic Surg. 2000;70:S1–S2.
33. Atwater BD, Roe MT, Mahaffey KW. Platelet glycoprotein IIb/IIIa receptor antagonists in non-ST segment elevation acute coronary syndromes: a review and guide to patient selection. Drugs. 2005;65:313–324.
34. Fleisher LA, Beckman JA, Brown KA, et al. ACC/AHA 2007 Guidelines on perioperative cardiovascular evaluation and care for noncardiac surgery: executive summary: a report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines (Writing Committee to Revise the 2002 Guidelines on Perioperative Cardiovascular Evaluation for Noncardiac Surgery): developed in collaboration with the American Society of Echocardiography, American Society of Nuclear Cardiology, Heart Rhythm Society, Society of Cardiovascular Anesthesiologists, Society for Cardiovascular Angiography and Interventions, Society for Vascular Medicine and Biology, and Society for Vascular Surgery. Circulation. 2007;116:1971–1996.
35. Hefer DVF, Munir A, Khouli H. Low-dose tenecteplase during cardiopulmonary resuscitation due to massive pulmonary embolism: a case report and review of previously reported cases. Blood Coag Fibrin. 2007;18:691–694.
36. Albers GW, Amarenco P, Easton JD, et al. Antithrombotic and thrombolytic therapy for ischemic stroke: the Seventh ACCP Conference on Antithrombotic and Thrombolytic Therapy. Chest. 2004;126:483S–512S.
37. Singh KP, Harrington RA. Primary percutaneous coronary intervention in acute myocardial infarction. Med Clin North Am. 2007;91:639–655; x–xi.
38. Alesh I, Kayali F, Stein PD. Catheter-directed thrombolysis (intrathrombus injection) in treatment of deep venous thrombosis: a systematic review. Catheter Cardiovasc Interv. 2007;70:143–148.
39. Hoylaerts M, Rijken DC, Lijnen HR, et al. Kinetics of the activation of plasminogen by human tissue plasminogen activator. Role of fibrin. J Bio Chem. 1982;257:2912–2919.
40. Wechsler LR. Intravenous thrombolytic therapy for acute ischemic stroke. N Engl J Med. 2011;364:2138–2146.