Arvind Bhimaraj, Celeste T. Williams, and David O. Taylor
Though major advances in the therapeutic armamentarium have provided improvements in mortality and morbidity for heart failure patients, outcomes are far from optimal. The 1-year survival of patients with New York Heart Association (NYHA) functional class IV heart failure on optimal medical therapy averages 50% to 80%.1 More than 100,000 individuals are estimated to be in end-stage heart failure, while only around 2,000 transplants have been performed annually in the United States for the last many years.2 In contrast to the dismal prognosis of end-stage heart failure patients, the 1-year survival after transplantation averages 85% to 90% at most US centers.2 Hence, heart transplants (and ventricular assist devices) are viable last resort options for heart failure patients to improve mortality and morbidity. Knowledge of the selection criteria and contraindications for heart transplant and timing for consideration of advanced therapies is necessary to provide the best care for heart failure patients. Many heart transplant patients continue to seek care from nontransplant cardiologists, and hence, basic concepts of rejection and immunomodulating pharmacology are necessary in order to suspect rejection and avoid effects of drug interactions. Recent recognition for the need of standardization of care for the cardiac transplant patients has led to development of guidelines by the International Society of Heart and Lung Transplantation (ISHLT).3 The guidelines use categorization of recommendations similar to the American College of Cardiology (ACC)/American Heart Association (AHA) guidelines and are referred to for certain topics thought to be relevant for a general cardiologist.
ORGAN ALLOCATION
There were 16,070 deceased organ donors in the United States during 2007 and 2008, and 28% of the time, a heart was recovered and transplanted.4 The United Network for Organ Sharing (UNOS) has the government contract for the procurement and distribution of cadaveric organs in the United States. UNOS operates through 11 geographic regions of the country with each region further divided into 58 organ procurement organizations (OPOs). The OPO’s main responsibility is to communicate with local hospitals, identify potential donors, and coordinate the transplant process.
THE DONOR
The local OPO is responsible for the initial identification and screening of the potential donor. Once consent for donation is obtained, the primary screening consists of confirming brain death, age, body size, and ABO blood type of the potential donor. The initial evaluation also includes obtaining routine laboratory tests, serologic tests (hepatitis B and C, HIV), identifying the presence of active malignancies, clinical course, and the etiology of death. Cardiac screening includes an electrocardiogram, chest x-ray, transthoracic echocardiogram (if inadequate, transesophageal echocardiogram), and determination of prolonged hypotension or cardiopulmonary resuscitation. A cardiac catheterization may be performed, depending on the age and the presence of coronary artery disease risk factors. Generally, if the potential donor is male and >45 years of age or female and >50 years of age, a cardiac catheterization is recommended.
Once the screening is completed, the donor is entered into the UNOS database and a “rank list” is obtained for each organ. The OPO contacts the recipient transplant centers in order of rank on the list. Candidates are ranked by a variety of factors including ABO blood group compatibility, geographic proximity between donor and transplanting hospitals, priority status of the potential recipient (as detailed below in the recipient section), and length of time spent on the waiting list. In general, the allocation of hearts is first within the local OPO and then outside the OPO in 500-mile concentric rings with the donor hospital in the center of the rings.
The final screen is completed by the harvesting surgeon and involves a review of the locally obtained data as well as a visual inspection of the donor heart. The surgeon looks for contusions, palpates for any obvious atherosclerotic lesions in the epicardial vessels, and reassesses cardiac function.
After brain death has been established and up to the time the organ is procured, the goal of medical management is to maintain hemodynamic stability of the potential donor. Brain death is associated with a high adrenergic state causing fluctuation in blood pressure, arterial vasoconstriction, and end-organ under-perfusion. Therefore, continuous monitoring of arterial pressure, central venous pressure, and urinary output is essential. The targeted systolic blood pressure is >100 mm Hg, central venous pressure between 8 and 12 mm Hg, urinary output >100 mL/h but 300 mL/h, and hematocrit >30%. Close attention must be made to maintaining normal electrolytes, acid–base balance, and oxygenation. The goal is to optimize cardiac output of the donor heart to achieve blood flow that promotes organ function with the least amount of vasoactive drug support. However, inotropic or vasopressor agents (dopamine, dobutamine, and epinephrine) may be needed. Recommendations on donor heart selection have been recently published by the ISHLT, the details of which are beyond the scope of this chapter.3
THE RECIPIENT
Due to disparity between the need and supply of donor hearts, the process of patient selection and allocation must be examined closely. It is crucial that transplant centers allocate organs to patients with the greatest need and the greatest chance to derive maximal benefit.
The current indications for cardiac transplantation are listed in Table 17.1. Potential recipients must undergo a thorough evaluation (Table 17.2) in an attempt to identify any condition that could potentially adversely affect the recipient’s survival or quality of life after transplantation. Initial evaluation involves identifying comorbidities that might increase mortality and morbidity independent of heart failure and hence preclude from consideration for transplant.
TABLE
17.1 Selection Criteria for Heart Transplant Listing

TABLE
17.2 Recommended Evaluation Prior to Transplantation
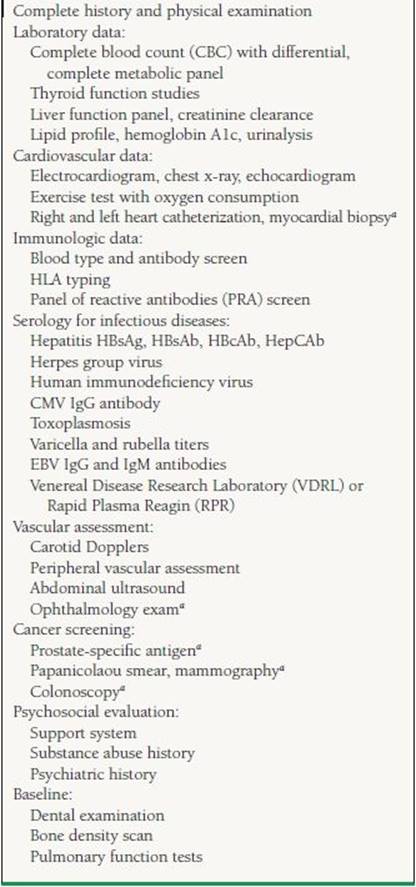
aIf appropriate.
The patient also needs immunologic testing including ABO blood typing, tissue typing for determination of human leukocyte antigens (HLAs), and screening for existing anti-HLA antibodies (Panel Reactive Antigen test). It is imperative that a careful psychosocial evaluation be performed to identify patients with substance abuse, noncompliance, or any behavioral trait that would lead to adverse posttransplant outcomes.
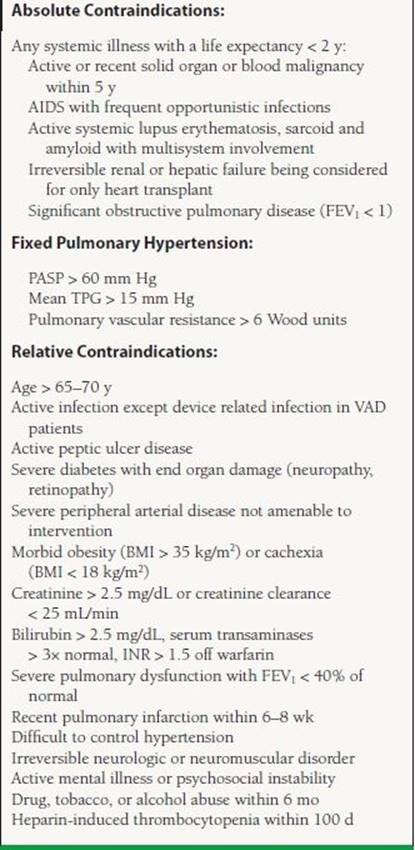
The list of contraindications for transplant listing is evolving constantly. Mancini and Lietz5 published a general list in 2010 and is presented in Table 17.3. There is no consensus for certain relative contraindications and hence the list varies based on individual institution experience and preference.
TABLE
17.3 Exclusion Criteria for Cardiac Transplantation
A patient who qualifies for a cardiac transplant gets a priority based on the severity of illness. Hence, the patients on the transplant list are divided into Status 1A (highest priority), defined as patients limited to the intensive care units who are dependent on mechanical circulatory support devices (mechanical assist device, intra-aortic balloon pump, extracorporeal membrane oxygenator) or high-dose intravenous inotropes plus Swan–Ganz catheter. Patients who are mechanically ventilated or have ventricular-assist device-related complications such as a thromboembolism or a device infection and those with a mechanical assist device for a 30 day period are also listed as Status 1A. Status 1B includes patients on continuous intravenous inotropes or patients with ventricular-assist devices once their 30 day 1A time has expired. A patient who does not meet criteria for Status 1A or 1B is listed as Status 2. Status 7 patients are those who are considered temporarily unsuitable to receive a transplant (Table 17.4).
TABLE
17.4 UNOS Status Definitions

Inpatients in need of a transplant are more clinically obvious than ambulatory patients. Determining the appropriate timing for consideration for transplant listing for ambulatory patients can be complicated. A patient should be considered for transplant when the expected survival without transplantation is lower than after transplantation. Variables like ejection fraction, NYHA class, and etiology of heart failure do not predict outcomes in heart failure consistently. In ambulatory patients, one of the best predictors of survival is peak oxygen uptake (VO2) measured by a cardiopulmonary exercise testing, which is an objective measure of functional status. The patient should achieve maximal exercise, represented by respiratory exchange ratio (RER) > 1.05 in order for the VO2 to accurately predict outcomes. In cases where a maximal threshold is not achieved, the carbon dioxide ventilator equivalent ratio (VE/VCO2) >35 can be used as a cutoff to refer for transplant listing. The ISHLT guidelines recommend repeating the test every 6 to 12 months to objectively reassess the need to remain on the transplant list.3 The goal is to list a patient for transplantation after all medical and surgical options have been exhausted, but before the patient becomes debilitated with end-organ damage that may compromise posttransplant survival. Figure 17.1 provides an algorithm for patient selection for transplant listing.

FIGURE 17.1 Steps in considering a patient for transplantation in the setting of acute decomposition (A) and in relatively stable (B) clinical situations. DT, destination therapy; BTT, bridge to transplant.
POSTTRANSPLANT MANAGEMENT ISSUES
Management of heart transplant patients broadly comprises monitoring for and management of the following: (a) allograft rejection, (b) complications and drug interactions of immunosuppressive agents, (c) infections, and (d) malignancies.
Rejection
The transplanted heart is identified as foreign by the recipients' immune system and is subject to a constant attempt at immune destruction. Cellular identity of self and nonself for an organism is mediated primarily through a group of antigens called major histocompatibility complex (MHC) and expressed on cell surfaces. In humans, this group of proteins is named human leukocyte antigens (as they were first identified on leukocytes).
Rejection can involve both cellular and humoral (antibody-mediated) immune injury to the allograft and is often classified into four major types: hyperacute, acute cellular, antibody-mediated (humoral), and chronic (cardiac allograft vasculopathy [CAV]).
Hyperacute rejection is an antibody-mediated event, which occurs minutes to hours after transplantation and is caused by preexisting recipient antibodies against the donor’s HLA present on the vascular endothelial cells. The histologic hallmark of hyperacute rejection is leaky capillaries, endothelial swelling, microthrombosis, polymononuclear infiltrate, and, subsequently, tissue necrosis. Immunohistochemical studies show deposition of immunoglobulin and complement within the vessel walls. Clinically, there is profound hemodynamic compromise and graft failure. Even with aggressive treatment, hyperacute rejection almost always leads to rapid graft loss. The catastrophic effects of hyperacute rejection can generally be prevented by PRA screening and by donor-recipient HLA and ABO blood- group cross-matching prior to transplantation. It is rarely seen in the current era.
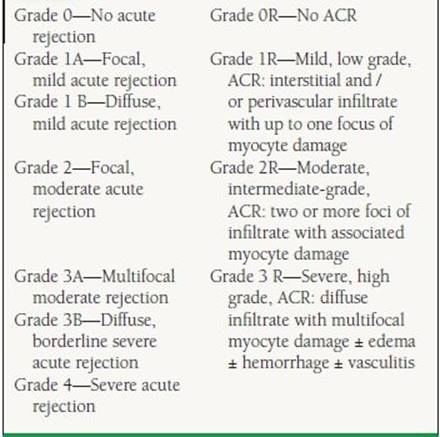
Unlike hyperacute rejection, acute cellular rejection (ACR) is primarily a T-lymphocyte–mediated process, which can occur from the first week after transplantation up to many years out. Twenty to forty percent of transplant patients experience at least one episode of ACR in the first year posttransplantation.6 A histologic grading system was developed in 1990 and updated in 2004 by the ISHLT. This grading classification is based on the amount of inflammatory infiltrate and presence or absence of myocyte necrosis7 (Table 17.5) and helps guide immunosuppressive therapy. The consensus of most transplant centers is that rejection is considered significant when the biopsy is graded at least 2R or if there is any evidence of hemodynamic compromise regardless of grade. Many advanced grades of rejection may be present for prolonged periods (weeks) prior to the development of allograft dysfunction, thus the rationale for “surveillance” biopsies to detect rejection prior to the progression to significant allograft compromise.
TABLE
17.5 The 1990 and Revised 2004 ISHLT Pathologic Grading of ACR
Antibody-mediated rejection, also known as humoral rejection, is initiated by alloantibodies directed against donor HLAs on endothelial cells.8 Antibody-mediated rejection is much less common than ACR. The biopsy reveals arteriolar, venular, and capillary endothelial swelling, nuclear enlargement, and infiltration of macrophages with or without lymphocytes (with B-cell predominance) early in the process. Once complement activation is initiated, there is recruitment of neutrophils, interstitial edema, and intravascular thrombosis with cell injury. Immunoflouresence microscopy shows complement components C3d, C4d, and C1q within the vessel walls. Episodes of antibody-mediated rejection are more severe than ACR and are usually associated with greater hemodynamic compromise, increased incidence of accelerated coronary artery vasculopathy at 1 year, and graft failure, with an overall poorer prognosis. Both AMR and ACR can coexist in 25% of acute rejection episodes.9 Patients at the highest risk for developing humoral rejection include women, patients with high panel reactive antibodies and/or a positive donor-recipient cross-match, cytomegalovirus (CMV) seropositivity, and patients with sensitization to OKT3.10 The 2004 ISHLT pathologic criteria classify AMR into present as AMR1 and absent as AMR 0. The criteria for AMR are (a) evidence of histologic features: myocardial capillary injury with endothelial swelling and intravascular macrophage accumulation with possible interstitial edema and presence of neutrophils and (b) positive immunofluorescence or positive immunoperoxidase staining for AMR (+CD68, C4d). Serologic evidence of donorspecific alloantibodies can be used as a supportive finding but is not required. Though immunoperoxidase staining can be used, immunofluorescence seems to be more sensitive.11 With advancement in laboratory techniques, the concepts, diagnosis, and therapeutics of AMR are evolving.
Monitoring for rejection primarily involves surveillance endomyocardial biopsies (EMBs). Because most rejection episodes occur within the first 3 to 6 months after transplantation, the frequency of biopsies is greater early on. A typical schedule might be weekly for 1 month, every other week for next month, every 3 to 4 weeks for next 1 to 2 months, every 4 to 6 weeks for 1 to 2 months, every 6 to 8 weeks until 1 year after transplant, and then every 3 to 6 months for the next 1 to 3 years while immunosuppression is being altered. Many programs stop routine surveillance biopsies after 3 to 5 years if the patient is stable on maintenance immunosuppression. It is reasonable to consider EMB for unexplained acute graft dysfunction regardless of time posttransplant or if major changes in the immunosuppressive regimen are required. Biopsies for the first 6 to 12 postoperative months and for patients at high risk for rejection beyond 12 months are considered reasonable. The utility of EMBs beyond 5 years is unclear. The current ISHLT guidelines3 do not consider it necessary to perform routine immunostaining techniques screening for AMR, unless there is a suspicion on microscopy. Several centers, however, perform routine immunostaining.
Many studies have evaluated the utility of noninvasive tests such as ECG, signal-averaged ECG, heart rate variability, QT dispersion, echocardiogram, and MRI, but none have shown convincing strength to predict rejection. The only noninvasive technique currently utilized in allograft rejection surveillance is gene expression profiling (GEP). The Allomap test, which utilizes GEP, was approved by the FDA in 2008 for use after 2 months posttransplant. The GEP technique was validated in the Cardiac Allograft Gene Expression Observational (CARGO) study in which peripheral blood mononuclear cells were analyzed for upregulation of certain target genes (which are upregulated during allo- reaction).12 A group of 11 discriminator genes were identified and validated to biopsy evidence of significant rejection (Grade 2R). A scoring system from 0 to 40 is used with the threshold suggesting no rejection varying with the duration from transplant: 3 to 6 months (<20), 6 to 9 months (<30), and >12 months (<34).
The Allomap testing tool can be used for its high negative predictive value in individuals at a low risk of rejection. It is not indicated to be used in those who are <2 months posttransplant, are still on high-dose steroids (≥20 mg prednisone or high dose of IV steroids), have undergone myeloablative therapy in the last 21 days, received blood products or hematopoietic growth factors in the last month, are pregnant, or are <15 years old. The IMAGE trial was undertaken to establish the utility of a clinical strategy of GEP-based rejection surveillance as a noninferior technique compared to the standard of surveillance EMBs.13 In patients >6 months posttransplant, the trial showed that at 1 year, the use of GEP along with clinical and echocardiographic assessment was noninferior (HR 1.04, CI 0.67–1.68) compared with routine EBMs and decreased the number of biopsies per patient. A key question this study raised was the utility of surveillance, irrespective of the type of test, as rejection episodes that were diagnosed prior to clinical graft dysfunction in either arm late after transplant was extremely low.
CAV, often called chronic rejection, remains a major limiting factor to long-term survival following cardiac transplantation. The incidence of CAV after transplant increases with the time from transplant: 8% at 1 year, 20% at 3 years, 30% at 5 years, and >50% at 10 years. It is an aggressive form of coronary artery disease that occurs months to years after transplantation and involves predominantly the entire length of the arterial vasculature with occasional involvement of the veins. Often the small intramyocardial vessels are severely involved. The histologic characteristics show concentric intimal thickening comprising of proliferative smooth muscle cells and extracellular matrix (ECM) with the vascular media and adventitia relatively unaffected. CAV is elicited by endothelial injury with a response from both cellular and humoral immune systems. Immune recognition to endothelial antigens leads to recruitment of inflammatory cells, of which the major effector cells are macrophages. These cells secrete proinflammatory cytokines and chemokines that influence proliferation of smooth muscle cells and deposition of ECM protein, causing luminal occlusion (5). Three stages of evolution of CAV have been suggested: (a) nonspecific endothelial injury (e.g., ischemia, trauma, and infection), (b) allo-response—recruitment of predominantly monocyte—macrophages and T-lymphocytes, and (c) arteriopathy—smooth muscle—like cell proliferation and ECM deposition.
ISHLT GUIDELINES3: REJECTION SURVEILLENCE:
Class IIa:
1. The standard of care for adult heart transplant recipients is to perform periodic endomyocardial biopsy (EMB) during the first 6 to 12 post operative months for surveillance of rejection. (Level of Evidence: C)
2. After the first postoperative year, biopsy surveillance for an extended period of time is recommended in patients at higher risk for late acute rejection, to reduce the risk for rejection with hemodynamic compromise and the risk of death in Africa-American recipients (who are at higher risk of rejection). (Level of Evidence: C)
3. Gene expression profiling can be used to rule out the presence of acute cellular rejection (ACR) of grade 2R or greater in appropriate low-risk patients, between 6 months and 5 years HT. (Level of Evidence: B)
Class IIb:
1. The use of routine EMB later than 5 years after HT is optional, depending on clinical judgment and the risk of for late allograft rejection. (Level of Evidence: C)
Class III:
1. The routine use of ECG parameters for acute allograft rejection monitoring is not recommended. (Level of Evidence: C)
2. The use of echocardiography as an alternative to EMB for rejection monitoring is not recommended (Level of Evidence: C)
3. The routine clinical use of MRI for acute allograft rejection monitoring is not recommended. (Level of Evidence: C)
4. The use of B-type natriuretic peptide (BNP), troponin I or T, C-reactive protein (CRP) levels for acute allograft rejection monitoring is not recommended. (Level of Evidence: C)
5. The use of systemic inflammatory markers for acute heart allograft rejection monitoring is not recommended. (Level of Evidence: C)
Both immune and nonimmune factors contribute to the pathogenesis of CAV The 2010 ISHLT report3 looked at 5,677 transplants performed between July 1997 and June 2001 to determine that the risk of developing CAV within 8 years of transplant was influenced by donor and recipient age, donor diabetes/hypertension, donor body size, and donor/recipient characteristics. Early CAV risk factors include pretransplant coronary artery disease, increase in donor age, donor body mass index (BMI), and a history of donor hypertension, while lesser risks for developing early CAV were seen in female donors and recipients. Late CAV risk factors include donor hypertension, hospitalization during the first year posttransplant for rejection, pretransplant coronary artery disease, HLA-DR mismatch, decreasing recipient age, and increasing donor age.
Unfortunately, the clinical diagnosis of CAV is usually made after the disease is advanced. Many times, the first clinical manifestation of CAV is ventricular arrhythmias, congestive heart failure, or sudden death. The surgical denervation of the heart prevents the pain associated with myocardial ischemia or infarction, particularly in the first 5 to 10 years after transplant. Because of the absence of symptoms, annual angiograms are often performed to detect CAV Angiograms are somewhat insensitive because of the poor visualization of the concentric lesions that affect distal and small vessels before they become apparent in the main epicardial vessels. Coronary angiograms have been shown to underestimate the presence of disease as demonstrated by histopathol- ogy studies and intracoronary ultrasound (IVUS). Studies have shown IVUS to be a more sensitive tool in detecting and following the progression of CAV by identifying maximal intimal thickness (an increase in ≥0.5 mm on serial IVUS examinations is considered significant); however, its increased cost, invasiveness, and lack of universal availability limit its use. An ISHLT consensus document proposes a new prognostically relevant nomenclature of CAV14
Angioplasty of CAV lesions, if discrete, may provide short-term palliation; however, restenosis rates are high and retrospective studies have shown DES to be better than BMS. Coronary artery bypass has limited use because CAV usually involves distal vessels and thus provides poor targets for bypassing. Retransplantation is an option, but the risk is higher than at the first transplant.
The mainstay of therapy for CAV is prevention and modification of coronary artery disease risk factors, including weight loss, lipid reduction, and controlling hypertension, and diabetes. These modifiable risk factors are thought to contribute to endothelial injury and the proliferation of smooth muscle cells and thus progression of CAV. Along with risk-factor modification, certain therapeutic modalities have been shown to be of some benefit in the prevention and progression of CAV. Statins not only lower cholesterol but also downregulate cytokine expression, lower plasma levels of C-reactive protein, and improve endothelial function and hence potentially decrease the onset of CAV. Calcium channel blockers have also been found to stabilize the endothelium and decrease platelet aggregation with a decrease in release of platelet-derived growth factor. Single-center studies have suggested that supplementation with vitamin C and E may retard early progression of transplant-associated arteriosclerosis. Two newer immunosuppressive agents, Sirolimus and Everolimus, have potent antiproliferative and antimigratory actions on vascular smooth muscle cells and have been shown in two multicenter prospective studies to decrease IVUS parameters of coronary vasculopathy. Furthermore, small single-center studies by Mancini et al.15 and Segovia et al.16 (RAPASTAT study) have suggested the benefit of Sirolimus in reducing adverse clinical outcomes and progression of IVUS parameters, respectively, in patients diagnosed with CAV. The clinical use of these proliferation signal inhibitors is limited by poor tolerability related to serositis and infections.
ISHLT GUIDELINES3: SCREENING FOR CAV
Class I:
1. Annual or biannual coronary angiography should be considered to assess the development of cardiac allograft vasculopathy (CAV). Patients free of CAV at 3 to 5 years after transplant, especially those with renal insufficiency may undergo less frequent invasive evaluation. (Level of Evidence: C)
2. Follow-up coronary angiography is recommended at 6 months after a PCI (percutaneous coronary intervention) because of high restenosis rates in transplant recipients. (Level of Evidence: C)
Class II a:
1. Evaluation of Coronary Flow Reserve in conjunction with coronary angiography may be useful for the detection of small vessel coronary artery disease (CAD), which is a manifestation of CAV. (Level of Evidence: C)
2. Treadmill or dobutamine stress echo and myocardial perfusion imaging may be useful for the detection of CAV in heart transplant recipients unable to undergo invasive evaluation. (Level of Evidence: B)
ISHLT GUIDELINES3: CAV MANAGEMENT
Class I:
1. Primary prevention of CAV in heart transplant recipients should include strict control of cardiovascular risk factors as well as strategies for the prevention of cytomegalovirus (CMV) infection. (Level of Evidence: C)
2. In heart transplant recipients, statin therapy has been shown to reduce CAV and improve long-term outcomes regardless of lipid levels and should be considered for all HT recipients. (Level of Evidence: A)
Class IIa:
1. In HT recipients with established CAV, the substitution of mycophenolate mofetil (MMF) or azathioprine (AZA) with a PSI can be considered. (Level of Evidence: B)
2. PCI with drug eluting stents is recommended in both adults and children with CAV and offers short term palliation for appropriate discrete lesions. (Level of Evidence : C)
3. Surgical revascularization can be considered in appropriate individuals (Level of Evidence C)
4. Cardiac transplantation may be considered in patients with severe CAV and absence of contraindications. (Level of Evidence C)
Immunosuppressive Strategies
Historically, during evolution of immunosupression for organ transplantation, a concept of induction was developed as a strategy to develop tolerance in the recipient to the donor organ. By attempting to wipe out the immune system during the pretransplant period it was thought that the donor antigen exposure and hence immune memory can be diminished leading to decreased chance of rejection on the long term. Though true tolerance is far from reality, such induction immunosuppressive strategies have proved beneficial in kidney transplants. Similar benefits were not realized in orthotopic heart transplant recipients when used routinely. Due to multiple reports of higher risk of infection and long-term risk of malignancy in patients receiving induction immunosuppressive therapy, some centers are reserving immune depletion agents to two clinical scenarios: (a) individuals at high risk of rejection and (b) in individuals with abnormal renal function as alternative potent immunosuppressive agents to delay initiation of calcineurin inhibitors giving time for the kidney function to recover from perioperative insult. Agents used are either lymphocyte depleting (antithymocyte globulins [ATG], Alemtuzumab, and Muromonab) or nonlymphocyte depleting (Daclizumab and Basiliximab).
While maintenance immunosuppressive agents are used to prevent significant rejection episodes, acute rejection episodes are treated with uptitration of the maintenance doses in either low-grade rejection, high-dose steroids in high grade cellular rejection, or combination of lymphocytedepleting or nondepleting agents with or without IVIG and plasmapheresis in antibody-mediated rejection.
The understanding of the pharmacodynamics of immunosuppressant agents is closely related to basic immunology of T-cell and B-cell activation and mounting a rejection episode. Figure 17.2 depicts the sites of action of immunosuppressant agents in relation to T-cell activation.17

FIGURE 17.2 Schematic of mechanisms of action of immunospuressive drugs. (Reprinted from Halloran PF, Gourishankar S. Principles and overview of immunosuppresion. In: Norman DJ, Turka LA, eds. Primer on Transplantation. Mt Laurel, NJ: American Society of Transplantation; 2001:87–98, with permission.)
Individual Immunosuppresant Agents
Polyclonal Antibodies
Antilymphocyte antibodies are used immediately posttransplantation to treat rejection episodes refractory to high-dose steroids or in patients with rejection associated with hemodynamic compromise. Polyclonal antithymocyte antibodies are antibodies derived from horse (ATGAM) or rabbit (Thymoglobulin). These antibodies are directed against multiple hematopoietic cell surface antigens and bind to the surface antigens, leading to cell depletion. T-cell surface antigens (CD2, CD3, CD4, and CD8) and B-cell surface antigens (CD19, CD20, and CD21) are predominantly affected leading to depletion of these cells in both peripheral circulation and lymphoid organs. These agents are considered efficacious when the CD3/CD2 counts are reduced to <10% of the pretreatment values. Complications can include febrile reactions, which usually occur during the initial infusion, and rarely, serum sickness. A higher incidence of serum sickness in the horse-derived antibodies has made it a less-favorable choice. The development of leukopenia or thrombocytopenia may require a reduction in dose or termination of therapy. Studies have shown an increased incidence in viral infections (CMV) and perhaps posttransplant lymphoproliferative disease (PTLD) associated with polyclonal antibody use.
Monoclonal Antibodies
Muromonab-CD3 (OKT3) is used for induction therapy in recipients with greater risk of rejection and for episodes of steroid-resistant rejection. OKT3 is an anti-CD3 monoclonal antibody that prevents the activation of the CD3-TCR receptor, which is required to generate the intracellular signals for T-cell activation. As the antibody remains bound to the receptor, further activation and proliferation of T cells are inhibited. However, OKT3 binding to the CD3-TCR complex on the surface of the T cell by itself can activate the T cell leading to cytokine release. Fever, chills, wheezing, chest pain, and hypotension characterize the cytokine release syndrome, which can be potentially life threatening. Symptoms are minimized by premedication with acetaminophen, intravenous steroids, and antihistamines. CD3+ T cells are generally undetectable during OKT3 therapy; however, within 12 to 24 hours after cessation of OKT3, CD3+ T cells reappear in circulation, unlike after treatment with ATG preparations, with which the lymphocyte depletion is present for weeks. There is an increased risk of developing HSV and CMV infections, especially in patients who are CMV donor positive and recipient negative (Table 17.6). Also, latent Epstein-Barr virus (EBV) may reactivate, leading to lymphoproliferative disorders including malignant monoclonal B-cell lymphomas. OKT3 is occasionally associated with aseptic meningitis or encephalopathy. Clinical studies have shown a significantly higher incidence of pulmonary edema, hypotension, and lymphomas compared to ATGAM. Due to these reasons, OKT3 has fallen out of favor as an agent for induction therapy.
TABLE
17.6 Risk of CMV Clinical Disease
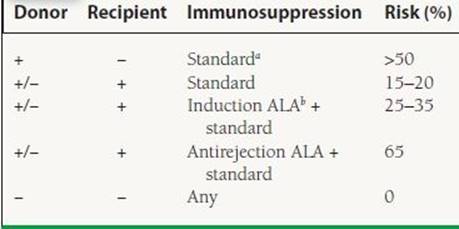
aCyclosporine or FK 506, azathioprine or mycophenolate-mofetil, and prednisone.
bAntilymphocyte antibody.
The interleukin-2 (IL-2) receptor (CD25) antibodies Basiliximab (Simulect) and Daclizumab (Zenapax) selectively inhibit T-cell proliferation by binding to the IL-2 receptor of activated T cells, preventing clonal expansion and activation of T cells. In heart transplant recipients, IL-2 receptor antagonists reduce the risk of rejection without increasing the incidence of infections during the early postoperative period. These monoclonal antibodies have been mostly used during induction to delay the introduction of calcineurin inhibitors. This approach creates a window to improve renal dysfunction exacerbated by ischemic-reperfusion injury. Basiliximab and Daclizumab are considered to be nondepleting induction agents because they do not affect resting lymphocytes. Basiliximab is a chimeric antibody (30% murine, 70% human protein), whereas Dacliximab is a humanized antibody (10% murine, 90% human protein), both designed to be less immunogenic than a fully murine monoclonal antibody. The IL-2 receptor antibodies appear to offer some advantage in heart transplant recipients, including the lack of the cytokine release syndrome and no reported increased risk of infections or malignancies. In 2009, most patients undergoing induction therapy received IL2R antagonist.2
A newer agent Alemtuzumab, a humanized murine monoclonal antibody against CD52 (cell surface protein expressed on lymphocytes, NK cells, monocytes, and thymocytes), is being studied in heart transplant recipients as an induction agent.
ISHLT GUIDELINES3: INDUCTION AGENTS
Class II a:
1. Immunosuppressive induction with polyclonal antibody preparations may be beneficial in patients at high risk of renal dysfunction when used with the intent to delay or avoid the use of a CNI (calcineurin inhibitor). (Level of Evidence: B)
Class II b:
1. Routine use of immunosuppressive induction in all patients has not been shown to be superior to immunosuppressive regimens that do not employ such therapy. (Level of Evidence: B)
2. classClass II b: Immunosuppressive induction with antithymocyte globulin (ATG) may be beneficial in patients at high risk for acute rejection. (Level of Evidence: C)
Calcineurin Inhibitors
In the early 1980s, cyclosporine (CsA), a lipophilic endacapeptide calcineurin inhibitor derived from a plant fungus, was first introduced as an immunosuppressant. Its use resulted in a dramatic reduction in the incidence of acute rejection in heart transplant recipients. Today, the calcineurin inhibitors are the cornerstone of therapy after heart transplantation, in conjunction with an antiproliferative agent and corticosteroids (CSs) (the so-called triple-drug therapy).
Calcineurin, a calcium-dependent serine-threoninephosphatase, is a vital enzyme in the transcription of IL-2 and other cytokine genes. The interaction of IL-2 with its receptor on activated T cells induces T-cell proliferation, which triggers the emergence of effector cells responsible for tissue destruction, resulting in clinical acute rejection. The T-cell receptor (TCR) is activated in response to alloantigens, causing an increase in intracellular calcium, which in turn activates the cytosolic protein, calmodulin. Ca2+-calmodulin interacts with calcineurin, activating its phosphatase moiety. Calcineurin is then able to dephosphorylate the nuclear factor of activated T cells (NFAT). NFAT translocates into the nucleus and causes transcription of T-cell–dependent lymphokines, such as IL-2 (and its receptor), interferon-γ, and tumor necrosis factor-α.
The calcineurin inhibitors (cyclosporine and tacrolimus) exert their effects by binding to cytosolic proteins called immunophilins upon entry into the T cell. Cyclosporin binds to cyclophilin and tacrolimus binds to FK-binding protein-12 (FKBP-12). Binding of cyclosporine and tacrolimus to its respective immunophilin enhances the immunophilins affinity to calcineurin. The immunophilin–drug complex inhibits the phosphatase activity of calcineurin, thereby preventing translocation of NFAT into the nucleus and therefore preventing the transcription of IL-2 and other cytokine genes.
The early preparation of cyclosporine (Sandimmune) was oil based and its bioavailability was unpredictable due to variations in absorption and metabolism. Neoral, the new microemulsion formulation of cyclosporine, has demonstrated greater bioavailability and more predictable pharmacokinetics than Sandimmune and hence translated into less rejection episodes, lower required dose, and less treatment failures.18 Cyclosporine is absorbed in the upper GI tract and the majority is eliminated through metabolism in the cytochrome P450 system. It also inhibits CYP3A4 enzymes altering metabolism of medications being processed through this enzyme.
The side effects and toxicities of cyclosporine include nephrotoxicity, hypertension, gingival hyperplasia, hirsutism, neuropathy, hyperlipidemia, and hyperkalemia. Drug-level monitoring is helpful in lessening the risk of toxicity while maintaining antirejection efficacy. Target CsA levels are measured trough levels. Because rejection is more prevalent early posttransplantation, higher levels of CsA are generally targeted (Table 17.7).
TABLE
17.7 Drug Therapy in Cardiac Transplantation

Tacrolimus (Tac), formerly called FK506, is a highly immunosuppressive calcineurin inhibitor. Tac is about 100 times more potent than CsA. Like CsA, it is metabolized via the cytochrome p450 3A-4 system, and its intravenous dose is one-fourth to one-fifth of its oral dose. Toxicities include nephrotoxicity, neurotoxicity, hyperuricemia, hypomagnesemia, gastrointestinal (GI) symptoms, diabetes, hyperkalemia, hyperlipidemia, and alopecia. Drug monitoring is very important to lessen the toxic effects while maintaining efficacy (see Table 17.7).
Clinical trials have shown mixed results when the performance of Tac and conventional CsA was compared among heart transplant patients in conjunction to receiving azathioprine, mycophenolate mofetil (MMF), and steroids. Early studies showed no significant differences in outcomes between the two calcineurin inhibitors, while later studies revealed significantly lower 6 month rates of any treated rejection and ISHLT cellular grade 3A rejection episodes in the tacrolimus group.19 Monotherapy with tacrolimus was recently evaluated in the TICTAC (Tacrolimus in Combination, Tacrolimus alone compared)20 trial showing comparable rejection rates in comparison to using a combination of Tac/MMF. Several studies have also shown the effectiveness of replacing CsA with Tac in cases of refractory rejection, gingival hyperplasia, or hirsutism. Patients treated with CsA have higher cholesterol and triglyceride levels, more hypertension, cholelithiasis, gingival hyperplasia, and hirusitism compared to patients on Tac, while the latter had more diabetes mellitus, tremor, and anemia.19,21,22 The calcineurin inhibitor chosen is often dependent on the patient, the side- effect profile, and the institutional experience.
In view of the side effect profile of the CNIs and suggestive benefit of antiproliferative agents in regard to allograft coronary vasculopathy, newer trials are evaluating CNI sparing regimens.
ISHLT GUIDELINES3: CNI USE
Class I:
1. Lower levels of CNIs in HT should be sought when CNIs are used in conjunction with MMF because with this combination lower levels are safe and associated with lower rejection rates as well as improved renal function. (Level of Evidence: B)
Class IIa:
1. Calcineurin inhibitor-based therapy remains the standard in immunosuppressive protocols used after heart transplant. (Level of Evidence: B)
Class IIb:
1. The results of clinical trials suggest that TAC-based regimens may be associated with lower rejection rate but not superior survival after HT than cyclosporine based regimens. (Level of Evidence: B)
2. CNI monotherapy with early CS withdrawal may be considered in highly selected individuals. (Level of Evidence: B)
Antiproliferative Agents
Azathioprine (Imuran) is a purine analog that impairs DNA synthesis and acts as an antiproliferative agent. It suppresses both T- and B-cell synthesis. It is well absorbed in the upper GI tract and metabolized in the liver. Some of its metabolites are broken down via xanthine oxidase. Hence, a xanthine oxidase inhibitor such as allopurinol can increase the azathioprine levels up to four times. The usual dose of azathioprine is 1 to 3 mg/kg/d, with the aim of keeping the white blood cell count >3,000 and the platelet count >100,000. Myelosuppression is the major toxicity, and it is generally dose dependent. Withdrawal of azathioprine usually reverses myelosuppression within 7 to 10 days. Other side effects include hepatotoxicity and pancreatitis. Malignancies, especially cutaneous malignancies, may be more common when compared to other, newer agents. Due to its side effect profile and available alternatives, the use of azathioprine has declined.2 Randomized trials have also shown that MMF, evorolimus, and sirolimus have lower rates of CAV when compared to azathioprine.23–25
MMF (Cellcept), also an antiproliferative agent, blocks the de novo pathway of purine synthesis in T and B lymphocytes that lack a robust salvage pathway. Mycophesnolic acid (MPA), a product of a Penicillin fungus, is the active metabolite of MMF. It is readily absorbed across the GI tract; however, the absorption of MPA is decreased in the presence of antacids containing magnesium and aluminum hydroxides. Toxicities of MMF include GI symptoms (nausea, vomiting, and diarrhea) and myelosuppression. The incidence of these adverse events is higher in patients receiving >3 g/d. Most symptoms will resolve with reduction of dose. Though many centers have MPA serum levels available, the relationship between MPA levels and rejection remains unclear. Hence, the current guidelines recommend against routine monitoring of drug level to adjust dosing3 (see Table 17.7). In specific situations where there is a suspicion of low-drug exposure leading to rejection, a trough level can be used to tailor dose adjustment. The serum levels of MPA are higher when this drug is administered with Tac compared to CsA, thus it is advisable to empirically reduce the dosage of MMF when switching from CsA to Tac. Comparing MMF to azathioprine on background of CsA and prednisone, there appears to be a 3-year survival advantage and a reduction of graft loss to rejection with MMF.23 However, one trial showed more opportunistic infections, diarrhea, and esophagitis in the MMF group compared to AZA-treated patients. MMF has become the dominant antiproliferative agent used in clinical practice.
TOR Inhibitors
Rapamycin (Sirolimus) is a macrolide antibiotic with a similar structure to tacrolimus. It is in the class of immunosuppressants called target of rapamycin (TOR) inhibitors. The TOR enzyme is a cytoplasmic protein responsible for connecting signals from the surface of the T cell to the nucleus for stimulation of growth and proliferation of the T lymphocytes. Rapamycin binds to TOR and inhibits cell proliferation stimulated by growth factors. It is known to inhibit platelet-derived growth factor and basic fibroblast growth factors in the arterial smooth muscle cells and endothelial cells, respectively. Studies have shown a decrease in the incidence of coronary allograft vasculopathy in heart transplant recipients receiving this immunosuppressant.25 Common side effects include hyperlipidemia and thrombocytopenia. When rapamycin is used alone there appears to be no adverse effects on kidney function; however, when it is used in combination with calcineurin inhibitors, there is a potentiation of the calcineurin inhibitor–induced nephrotoxicity. Therefore, the dose and target levels of the calcineurin inhibitor must be reduced substantially.
Everolimus (RAD: Certican) is a derivative of sirolimus with an identical mechanism of action. RAD, like rapamycin, inhibits clonal expansion of T cells but does not inhibit T-cell activation. It exerts its affects by forming a complex with FKBP-12 to inhibit the cyclin-dependent kinases termed the TOR. This leads to G1 S-phase cell cycle arrest. When compared to sirolimus, everolimus has a shorter half-life (30 hours compared to rapamycin at 60 hours) as well as a relatively higher bioavailability. Like the calcineurin inhibitors, RAD and rapamycin are biotransformed through the cytochrome P450, 3A-4 system. Side effect profile is similar to Sirolimus and has also been associated with a significant reduction in allograft vasculopathy measured by IVUS at 1 year (7).
Corticosteroids
CSs are important for induction, maintenance, and treatment of rejection in heart transplant recipients. CSs have immunosuppressive and anti-inflammatory effects and affect the number, distribution, and function of T cells, B cells, macrophages, as well as endothelial cells. The usual treatment for moderate rejection (grade 2R) without hemodynamic compromise is pulse-dose steroids (250 to 1,000 mg solumedrol intravenously daily for 3 days). Most rejection episodes respond to initial therapy.
ISHLT GUIDELINES3: ANTIPROLIFERATIVE AGENTS
Class IIa:
1. MMF, EVL or SRL as tolerated should be included in contemporary immunosuppressive regimens because therapies including these drugs have been shown to reduce onset and progression of CAV (assessed with IVUS). (Level of Evidence: B)
ISHLT GUIDELINES3: CORTICOSTEROID USE
Class I:
1. CS withdrawal can be successfully achieved 3 to 6 months after HT in many low risk patients. (Level of Evidence: B)
Class IIa:
1. Corticosteroid avoidance, early CS weaning or very low dose maintenance CS therapy are all acceptable therapeutic approaches. (Level of Evidence: B)
2. If used, CS weaning should be attempted if there are significant CS side effects and no recent rejection episodes. (Level of Evidence: C)
Steroids are associated with many side effects including cataracts, diabetes, myopathy, osteopenia, growth retardation in children, aseptic necrosis, hirsutism, cushingoid appearance, and dermatologic problems. They also exacerbate hypertension and hyperlipidemia, and cause adrenal insufficiency. Thus, it is important to give stress doses of hydrocortisone when indicated (illness, surgical procedures) to patients on chronic CSs. Current practice favors withdrawal of steroids to minimal dosage or none at 6 months to 1 year posttransplant, provided there are no rejection episodes. Yet, data from the ISHLT registry between 2007 and 2009 shows that 89% of patients are still on steroids at the end of 1 year and 52% at 5 years.2 Typically, serial EBMs are performed to monitor for rejection while CS are withdrawn.
Combination Regimen
Most transplant centers initiate triple therapy with a CNI, an antiproliferative agent, and a CS. Figure 17.2 reflects the idea of using medications acting on different pathways of T-cell activation in order to achieve effective immunosuppression. In general, the various clinical trials have not shown significant mortality benefit of one combination over the other. Tacrolimus, mycophenolate, and prednisone are currently the dominant immunosuppressive choices. The mTOR inhibitors are not used widely due to their side effect profile. Sirolimus and evorolimus are used in 11% of patients at 1 year and 23% at 5 years posttransplant.2
ISHLT GUIDELINES3: CHOICE OF IMMUNOSUPRESSION AGENTS
Class IIB:
1. The adverse events of immunosuppressive drugs observed in randomized clinical trials underscore the need for individualization of immunosuppressant according to the characteristics and risks of the individual recipient. (Level of Evidence: C)
POSTTRANSPLANT COMPLICATIONS
Infection
Preventing allograft rejection with immunosuppressive agents increases the risk of infection posttransplantation. Infections continue to be one of the leading causes of death after cardiac transplantation. Knowing the timetable of common infections following solid-organ transplant will aid in formulating a differential diagnosis and determining the timing of the various preventative strategies.
A pretransplant infectious disease evaluation is used to identify any condition that would disqualify a potential recipient for transplantation, update immunizations, identify and treat active infections, and define the risk of infection in order to determine the strategy for preventing posttransplant infections.
During the first 30 days after transplantation, there are generally three types of infections that occur: (a) active infection transmitted with the allograft, (b) untreated pre-transplant infection in the recipient, and (c) nosocomial infections, which are commonly related to surgical wounds or indwelling catheters. More than 95% of the nosocomial infections during this period are bacterial or fungal (Candida species). In contrast, late infections that occur 1 to 6 months following transplantation are generally caused by opportunistic organisms such as Pneumocystis carinii (PCP), Aspergillosis species, Nocardia asteroids, and Listeria monocytogenes and viral infections such as CMV or EBV, which are by far the most common. After 6 months posttransplantation, most patients require decreasing levels of immunosuppression and thus their infectious disease risks become similar to those of the general population. The majority of patients require antiviral, antibacterial, and antifungal prophylaxis for 6 to 12 months posttransplantation. Approximately 5% to 10% of transplant recipients experience recurrent rejection episodes and thus are still at risk of developing opportunistic infections secondary to increased immunosuppressive therapy.
There are no specific guidelines for endocarditis prophylaxis in heart transplant patients. Although the incidence of endocarditis is low in this population, the mortality rate is approximately 80%. Individuals who develop valvular heart disease have a higher risk than those who do not. The use of antibiotic prophylaxis for dental procedures in heart transplant patients is considered reasonable.
Viral Infections
CMV remains the most important infection affecting the morbidity and mortality of heart transplant recipients. The serologic (presence of antibody to CMV) status of the donor and the recipient is a predominant predictor of posttransplant CMV disease events. Donor seropositive (D+)/recipient seronegative (R-) bears the greatest risk of developing CMV clinical disease, which can present as leukocytopenia, pneumonia, colitis, gastritis, esophagitis, hepatitis, or myocarditis. With D+/R- status, there is an increased incidence of tissue-invasive CMV, recurrent CMV, ganciclovir-resistant CMV, and CAV. Patients at highest risk receive prophylaxis with oral valgancyclovir with or without CMV hyperimmune globulin (CMV–IVIG, Cryptogam). Active CMV disease must be treated with intravenous gancyclovir with or without CMV–IVIG, depending on whether or not invasive CMV is present.
Fungal Infections
Candida species and Aspergillosis are the most common fungal infections after transplantation. Oral clotrimazole or nystatin is used during the first 3 to 6 months or during periods of enhanced immunosuppression, when there is an increased risk of opportunistic infection.
Protozoal Infections
Trimethoprim-sulfamethoxazole (TMP-SMX) is highly effective against PCP and Nocardia infections. Prior to the institution of PCP prophylaxis, approximately 10% of cardiac transplant recipients developed PCP, with a mortality rate up to 40%. Nowadays, with TMP-SMX prophylaxis, PCP is exceedingly rare. Toxoplasmosis is also a concern in heart transplant recipients. A Toxoplasma-seronegative recipient of a Toxoplasma-seropositive donor is at highest risk of developing toxoplasmosis posttransplant. Prophylaxis with TMP- SMX is also effective posttransplantation and during episodes of increased immunosuppression therapy (steroid-resistant rejection). Active toxoplasmosis can present as myocarditis and is treated with pyrimethamine and sulfonamide.
Malignancies
Cutaneous malignant lesions are the most common tumors after cardiac transplantation and account for nearly 40% of de novo cancers.26 Posttransplantation, the incidence of squamous cell carcinoma and basal cell carcinoma is increased, with basal cell carcinoma being the most common type (unlike the general population, in which squamous cell carcinoma is the most common type of skin cancer).
PTLD is a unique type of lymphoma that occurs in approximately 3.4% of all heart transplant recipients.27 Approximately 90% of all PTLDs are associated with EBV These tumors are B-cell in origin and range from a benign polyclonal process to a highly malignant monoclonal lymphoma. Typically the tumor arises 12 to 18 months following transplant and is most commonly located intraabdominally. The patient may have a mononucleosis-like presentation. Risk factors for developing PTLD include EBV-seropositive donor to EBV-seronegative recipient, type of organ transplanted (lung and heart have the highest incidence), preceding CMV infection, and the level and type of immunosuppression used posttransplantation. PTLDs have variable prognoses, with treatment strategies geared toward drastically decreasing background immunosuppressant drug therapy. This tactic may lead to a regression of PTLD in 23% to 50% of the patients.27 Malignant lymphomas (even if EBV initiated) usually require cytotoxic chemotherapy as well, and despite aggressive therapy, have poor response rates (<50%).
Chronic immunosuppression and induction therapy have been implicated as risk factors for malignancy. A strategy of minimization of immunosuppression with acceptance of lower CNI levels and/or decreased MMF dose is important upon diagnosis of a lymphoid malignancy. Cancer screening recommendations are similar to the general population. Standard therapy is recommended for individual cancers. Certain cancers have a more malignant course in transplant patients compared to the general population.
Chronic Renal Dysfunction
Renal dysfunction remains an important complication. About 20% of patients have some degree of renal dysfunction 1 year following cardiac transplantation. By year 7, approximately 10% of survivors have a creatinine >2.5 mg/dL, while 4% are on chronic dialysis.2
Risk factors for late renal dysfunction include chronic administration of calcineurin inhibitors (cyclosporine and tacrolimus), preexisting renal dysfunction, diabetes, hypertension, and generalized atherosclerosis. The renal toxicity associated with calcineurin inhibitors includes early functional nephrotoxicity and late structural nephrotoxicity. The early form of nephrotoxicity occurs when calcineurin inhibitors are administered for the first time. The calcineurin inhibitors cause vasoconstriction of the afferent arterioles, resulting in a decrease in renal blood flow and a decrease in glomerular filtration rate. The late form of renal dysfunction is thought to be caused by a combination of the acute reno-vascular effects plus direct toxic effects on renal tubular epithelial cells. Cyclosporine has been shown experimentally to cause apoptosis in tubular and interstitial cells, potentially inducing tubular atrophy and subsequent fibrosis. Both early and late renal effects are dose related. The management of chronic renal insufficiency is to minimize the dosage of calcineurin inhibitors, which may or may not halt the progression of the renal dysfunction. Another alternative is to switch to a sirolimus-based regimen, which may have renalsparing effects if initiated before renal dysfunction is progressive. Kidney transplantation should be considered for end-stage renal dysfunction.
Hypertension
The majority of adult cardiac transplant recipients are diagnosed with arterial hypertension. The use of cyclosporine is linked directly to the development of posttransplant hypertension. Three proposed mechanisms are direct sympathetic activation, increased responsiveness to circulating neurohormones, and direct vascular effects. A common endpoint of these proposed mechanisms is vasoconstriction of the renal vasculature, leading to sodium retention and an elevated plasma volume. Hypertension has been found to be less common in patients receiving tacrolimus than in those receiving cyclosporine. Steroids also play a role in the development of hypertension posttransplant. The mineralocorticoid activity causes sodium retention and also contributes to the increase in plasma volume. The denervated transplanted heart may not respond well to the increased after load, and persistent hypertension may lead to left ventricular hypertrophy and subsequent left ventricular systolic and diastolic dysfunction. Initial nonpharmacologic therapy should include sodium restriction. First-line pharmacologic agents include calcium channel blockers and angiotensin-converting enzyme (ACE) inhibitors. Calcium channel blockers are the most commonly used class of medications followed by ACE inhibitor. Diltiazem has the advantage of increasing the cyclosporine level by competing with cytochrome P450, thereby decreasing the required cyclosporine dose. Monotherapy is effective in <50% of patients posttransplantation and multiple agents are often needed to achieve adequate blood pressure control. Historically, beta-blockers have been avoided, secondary to concerns about excessive bradycardia or exercise intolerance in the denervated heart; however, if they are needed, they are effective antihypertensive agents.
Hyperlipidemia
Approximately 1 month after heart transplantation, recipients frequently demonstrate an increase in total cholesterol, low-density lipoprotein (LDL) cholesterol, apolipoprotein B, and triglyceride levels. The etiology of dyslipidemia in heart transplant recipients is multifactorial, including genetic predisposition, high-fat diets, and immunosuppressive agents. In particular, CSs and cyclosporine have been found to be important immunomodulating drugs that contribute to the development of hyperlipidemia posttransplant. Cyclosporine decreases bile acid synthesis from cholesterol and thus increases serum cholesterol levels. CSs increase acetyl coenzyme A (CoA) carboxylase activity and free fatty acid synthesis, which is a precursor to cholesterol synthesis. Statins have been found to reduce LDL cholesterol levels in heart transplant recipients. In particular, pravastatin has been shown to cause not only a significant reduction in cholesterol levels at 3, 6, 9, and 12 months posttransplantation but also a reduction in rejection episodes associated with hemodynamic compromise, a decrease in the incidence of CAV, and an increased overall 1-year survival.28 Current recommendations are to prescribe a low-dose statins to all transplant recipients early after transplantation, regardless of their lipid levels, as long as liver function tests are normal. A critical interaction between the statins and cyclosporine or tacrolimus (via cytochrome P450–3A inhibition) increases the risk of myositis and rhabdomyolysis. Thus, statins are started at lower doses in transplant patients and titrated up carefully.
Tricuspid Regurgitation
Tricuspid regurgitation (TR) is not uncommon in the transplanted heart. Mild TR is present in virtually all transplanted hearts, and moderate–severe TR is present in up to 50% of transplant patients who live >5 years. Mechanical torsion on the tricuspid annulus due to the biatrial anastamosis accounts for at least mild–moderate regurgitation. The more anatomically correct, bicaval anastamotic technique has decreased the incidence but not prevented it. Recipients with preexisting pulmonary artery hypertension often experience acute right ventricular dysfunction and subsequent chronic dilation, which contributes to the regurgitation. More importantly, repeated EMBs with damage to the valve, chordal apparatus, and papillary muscles account for the majority of severe cases of TR. Generally, even severe TR is well tolerated, but in a small minority of patients with progressive right heart failure due to TR, tricuspid replacement is needed (<3% of all heart transplants). Heart transplant patients with TR are at higher risk of endocarditis than those with no valvular pathology.
Osteoporosis
Osteoporosis remains a common problem in heart transplant recipients, contributing to fracture-associated immobility that may compromise quality of life posttransplantation. The risk factors for osteoporosis may begin well before transplantation. Patients awaiting heart transplantation have a mean average reduction in bone mineral density (BMD) up to 10% compared to age-matched healthy individuals. Contributing factors to bone loss in severe heart failure patients include reduced exercise or immobilization, cardiac cachexia, smoking, alcohol, low calcium intake, renal failure, hypogonadism, heparin, and loop diuretic administration.
Bone loss is most rapid during the first 6 months following transplant, which coincides with the period of aggressive immunosuppression. Lumbar spine and femoral neck BMD declines 3% to 10% and 6% to 11%, respectively, in the first 6 months and stabilizes thereafter. Glucocorticoids are known to cause accelerated bone loss and are associated with a higher-than-normal incidence of vertebral fractures. Glucocorticoids reduce bone density by direct inhibition of osteoblast function and impairment of collagen and new bone formation. Shane et al.29 showed that at 2 years after cardiac transplant, severe osteoporosis was detected in the lumbar spine in 28% of patients and within the femoral neck in 20% of patients. It is common to perform routine screening of transplant candidates with baseline BMD study prior to transplantation to identify and correct any secondary causes of bone loss. A regimen of elemental calcium, vitamin D supplementation, with or without bisphosphonates, and proper exercise training is indicated pretransplantation, and it is recommended to begin bisphosphonates for all posttransplant patients receiving CSs immediately after transplant and continue at least through the first year post-transplant. The American College of Rheumatology guidelines recommend calcium and vitamin D supplementation for all patients receiving CSs to prevent osteoporosis.30 With decreased doses of steroids, recent studies have shown a decrease in bone loss in posttransplant patients when compared to the past.
SURVIVAL
In the current era, the 1-year survival after cardiac transplantation exceeds 85% and approaches 90% at most institutions.2 The greatest mortality occurs in the first year posttransplantation and the temporal improvement in heart transplant survival over the last decade is due to improvements in outcomes during this time. After year 1, the annual mortality rate is approximately 3.6%, such that the 5- and 10-year survival rates are approximately 70% to 75% and 50% to 60%, respectively.2 The survival for retransplantation has improved drastically if the patient is undergoing retransplantation at least 12 months after the initial transplant. Currently, the 1-year survival in this group of patients is approximately 84%.2
Data from the ISHLT registries have shown that graft failure is the primary cause of death during the first 30 days posttransplantation, accounting for 41% of the deaths, followed by non-CMV infections (14%) and multiorgan failure (13%). After the first month and up until day 365, non-CMV infections account for almost 35% of deaths, followed by graft failure (19%) and acute rejection (12%). After 5 years posttransplantation, CAV and graft failure combined account for 30% of deaths, while malignancies account for 24% and non-CMV infections for 10%.2
REFERENCES
1. Roger VL, et al. Trends in heart failure incidence and survival in a community-based population. JAMA. 2004;292(3):344–350.
2. Stehlik J, et al. The Registry of the International Society for Heart and Lung Transplantation: twenty-seventh official adult heart transplant report—2010. J Heart Lung Transplant. 2010;29(10):1089–1103.
3. Costanzo MR, et al. The International Society of Heart and Lung Transplantation Guidelines for the care of heart transplant recipients. J Heart Lung Transplant. 2010;29(8):914–956.
4. U.S. Department of Health and Human Services, Health Resources and Services Administration, and H.S. Bureau. The 2009 Annual Report of the OPTN and SRTR: Transplant Data 1999–2008.Rockville, MD, 2010.
5. Mancini D, Lietz K. Selection of cardiac transplantation candidates in 2010. Circulation. 2010;122(2):173–183.
6. Patel JK, Kobashigawa JA. Should we be doing routine biopsy after heart transplantation in a new era of anti-rejection? Curr Opin Cardiol. 2006;21(2):127–131.
7. Stewart S, et al. Revision of the 1990 working formulation for the standardization of nomenclature in the diagnosis of heart rejection. J Heart Lung Transplant. 2005;24(11): 1710–1720.
8. Hammond EH, et al. Vascular (humoral) rejection in heart transplantation: pathologic observations and clinical implications. J Heart Transplant. 1989;8(6):430–443.
9. Taylor DO, et al. Allograft coronary artery disease: clinical correlations with circulating anti-HLA antibodies and the immunohistopathologic pattern of vascular rejection. J Heart Lung Transplant. 2000;19(6):518–521.
10. Micheals PJ, et al. Humoral rejection in cardiac transplantation: risk factors, hemodynamic consequences and relationship to transplant coronary artery disease. J Heart Lung Transplant. 2003;22(1):58–69.
11. Chantranuwat C, et al. Immunoperoxidase staining for C4d on paraffin-embedded tissue in cardiac allograft endomyocardial biopsies: comparison to frozen tissue immunofluorescence. Appl Immunohistochem Mol Morphol. 2004;12(2):166–171.
12. Deng MC, et al. Noninvasive discrimination of rejection in cardiac allograft recipients using gene expression profiling. Am J Transplant. 2006;6(1):150–160.
13. Pham MX, et al. Molecular testing for long-term rejection surveillance in heart transplant recipients: design of the Invasive Monitoring Attenuation Through Gene Expression (IMAGE) trial. J Heart Lung Transplant. 2007;26(8):808–814.
14. Mehra MR, et al. International Society for Heart and Lung Transplantation working formulation of a standardized nomenclature for cardiac allograft vasculopathy-2010. J Heart Lung Transplant. 2010;29(7):717–727.
15. Mancini D, et al. Use of rapamycin slows progression of cardiac transplantation vasculopathy. Circulation. 2003;108(1):48–53.
16. Segovia J, Gomez-Bueno M, Alonso-Pulpon L. Treatment of al- lograft vasculopathy in heart transplantation. Expert Opin Phar- macother. 2006;7(17):2369–2383.
17. Halloran PF, Gourishankar S. Principles and overview of immunosuppresion. In: Norman DJ, Turka LA, eds. Primer on Transplantation. Mt Laurel, NJ: American Socity of Transplantation; 2001:87–98.
18. Eisen HJ, et al. Safety, tolerability, and efficacy of cyclosporine microemulsion in heart transplant recipients: a randomized, multicenter, double-blind comparison with the oil-based formulation of cyclosporine—results at 24 months after transplantation. Transplantation.2001;71(1):70–78.
19. Reichart B, et al. European Multicenter Tacrolimus (FK506) Heart Pilot Study: one-year results—European Tacrolimus Multicenter Heart Study Group. J Heart Lung Transplant. 1998;17(8):775–781.
20. Baran DA, et al. Randomized trial of tacrolimus monotherapy: tacrolimus in combination, tacrolimus alone compared (the TICTAC trial). J Heart Lung Transplant. 2007;26(10):992–997.
21. Taylor DO, et al. A randomized, multicenter comparison of tacrolimus and cyclosporine immunosuppressive regimens in cardiac transplantation: decreased hyperlipidemia and hypertension with tacrolimus. J Heart Lung Transplant. 1999;18(4):336–453.
22. Grimm M, et al. Superior prevention of acute rejection by tacrolimus vs. cyclosporine in heart transplant recipients—a large European trial. Am J Transplant. 2006;6(6):1387–1397.
23. Kobashigawa J, et al. A randomized active-controlled trial of mycophenolate mofetil in heart transplant recipients. Mycophenolate Mofetil Investigators. Transplantation. 1998;66(4):507–515.
24. Eisen HJ, et al. Everolimus for the prevention of allograft rejection and vasculopathy in cardiac-transplant recipients. N Engl J Med. 2003;349(9):847–858.
25. Keogh A, et al. Sirolimus in de novo heart transplant recipients reduces acute rejection and prevents coronary artery disease at 2 years: a randomized clinical trial. Circulation. 2004;110(17):2694–2700.
26. Taylor DO, et al. The Registry of the International Society for Heart and Lung Transplantation: twenty-first official adult heart transplant report—2004. J Heart Lung Transplant. 2004;23(7):796–803.
27. Cockfield SM. Identifying the patient at risk for post-transplant lymphoproliferative disorder. Transpl Infect Dis. 2001;3(2):70–78.
28. Kobashigawa JA, et al. Effect of pravastatin on outcomes after cardiac transplantation. N Engl J Med. 1995;333(10):621–627.
29. Shane E, et al. Osteoporosis after cardiac transplantation. Am J Med. 1993;94(3):257–264.
30. Recommendations for the prevention and treatment of glucocorticoid-induced osteoporosis: 2001 update. American College of Rheumatology Ad Hoc Committee on Glucocorticoid-Induced Osteoporosis. Arthritis Rheum. 2001;44(7):1496–1503.
QUESTIONS AND ANSWERS
Questions
1. A 28-year-old man underwent heart transplantation 5 years ago for a presumed postviral dilated cardiomyopathy. He is seen at an urgent care clinic with complaints of a nonproductive cough, sore throat, and low-grade fever. He is diagnosed with an upper respiratory tract infection and prescribed clarithromycin, 500 mg twice daily for 10 days. Two weeks later, after completing antibiotics, he returns to the transplant clinic with complaints of generalized fatigue, shortness of breath, and a persistent nonproductive cough. His current immunosuppressive regimen includes prednisone, 10 mg daily; mycophenolate mofetil (MMF), 750 mg twice daily; and tacrolimus, 2 mg twice daily.
Physical examination: Sick-appearing man. Blood pressure is 142/92 mm Hg, heart rate is 98 beats per minute and regular. Oropharynx is clear. Chest with bibasilar crackles. His abdomen shows no organomegaly or ascites. There is 3+ peripheral edema. ECG: Normal sinus rhythm, biatrial enlargement, and incomplete right bundle branch block. Laboratory studies: Hematocrit 36%, white cell count 4,100/µL, BUN 60 mg/dL, creatinine 5.3 mg/dL (baseline 1.3), tacrolimus level 29 ng/mL, and MMF 4.5 ng/mL.
Which of the following is the most likely explanation of the patient’s current condition?
a. Humoral rejection with hemodynamic compromise
b. Interaction of tacrolimus with MMF, causing cellular rejection and thus acute renal failure
c. Immunocompromised patient with a viral syndrome
d. Interaction of tacrolimus with clarithromycin, resulting in acute renal failure
e. Noncompliance
2. A 23-year-old woman is seen for routine monthly posttransplant follow-up. She underwent an orthotopic cardiac transplantation 9 months ago for familial dilated cardiomyopathy. She has returned to college and reports that she saw an internist 3 weeks ago and was taken off of diltiazem, 60 mg twice daily, secondary to severe ankle swelling. She reports that her ankle swelling has improved and she is able to walk an hour a day without becoming fatigued or dyspneic. Current medications include prednisone, 10 mg daily; MMF, 500 mg twice daily; cyclosporine, 75 mg twice daily; and bactrim, 1 tablet daily.
Physical examination: Blood pressure 110/78 mm Hg, heart rate 95 beats per minute, and RR 18 per minute. Chest is clear, PMI nondisplaced, normal S1 and S2. Extremities: no edema.
ECG: Normal sinus rhythm at 95, with nonspecific ST-T abnormalities.
Laboratory: Hematocrit 37%, white blood cell count 8,400/μL, platelets 220,000/μL, BUN 30 mg/dL, creatinine 1.1 mg/dL, cyclosporine 45 ng/mL, and MMF level 2.2 ng/mL. Right ventricular biopsy shows prominent lymphocytic infiltrate with associated areas of myocyte necrosis with ISHLT Grade 3R (3A) rejection.
Which of the following statements is false?
a. Cyclosporine dose should be increased to achieve adequate trough levels.
b. This rejection episode is most likely secondary to the discontinuation of diltiazem.
c. There is no need to treat this rejection episode; the patient feels great and is hemodynamically stable.
d. Patients should report all changes in medications to the transplant clinic.
3. All of the following are risk factors for posttransplant mortality except:
a. Diabetes with end-organ damage
b. Reversible pulmonary hypertension
c. Active smoking
d. Left ventricular assist device <30 days
e. Active infection
4. What is the most sensitive tool in detecting cardiac allograft vasculopathy (CAV) posttransplantation?
a. Serial echocardiography
b. Intracoronary vascular ultrasound
c. Positron emission scanning
d. RV endomyocardial biopsy
e. Coronary angiogram
5. Which of the following is true regarding posttransplant lymphoproliferative disease (PTLD)?
a. Treatment includes reduction in immunosuppression therapy.
b. It occurs in approximately 90% of all cardiac transplant patients.
c. The highest-risk group for developing PTLD is recipients who are Epstein-Barr virus (EBV) seronegative who receive an EBV-seronegative heart.
d. The lymphomas that arise in PTLD are usually T cell in origin.
e. The tumor usually arises 1 month after transplantation, with the cervical lymph nodes being the most common site.
Answers
1. Answer D: The patient’s clinical picture is most likely secondary to tacrolimus (Tac) toxicity resulting in acute renal failure. Clarithromycin inhibits the cytochrome P450 system, causing increased levels of calcineurin inhibitors (tacrolimus and cyclosporine). The target trough level of tacrolimus >12 months posttransplant is 8 to 10 ng/mL. The patient should discontinue tacrolimus until target trough levels are obtained. This clinical scenario could represent a rejection episode; however, with the given history, tacrolimus toxicity is most likely the culprit. Tacrolimus taken with MMF is a common immunosuppression regimen. There is no increased incidence of renal failure or episodes of cellular rejection with this particular regimen. When taking Tac with MMF, the MMF dose should be decreased to lessen the likelihood of developing MMF toxicity (myelosuppression). The patient is definitely compliant, secondary to elevated trough levels of immunosuppressants.
2. Answer C: The patient’s episode of rejection is most likely secondary to discontinuing diltiazem 3 weeks earlier. Diltiazem inhibits the cytochrome P450 system, causing increased levels of cyclosporine and therefore requiring lesser dosages to achieve trough levels. Once diltiazem was discontinued, the dose of cyclosporine should have been increased. Almost all rejection episodes Grade 3R (3A) and higher and all rejection episodes that show hemodynamically instability (regardless of grade) are treated with augmented immunosuppression. It is important for transplant recipients to communicate any changes in medications to the transplant clinic, so these complications can be avoided.
3. Answer B: Risk factors for posttransplant mortality include but are not limited to short-term ventricular assist device use (<30 days), irreversible pulmonary vascular resistance (PVR > 4 Wood units), mechanical ventilation at the time of transplant, active infection, active smoking, diabetes particularly with end-organ damage, and hepatic or renal insufficiency.
4. Answer B: Serial echocardiograms are important for following graft function posttransplant; however, they are not very sensitive for detecting early CAV. Periodic right ventricular biopsy is the standard method of surveillance for cellular rejection, but adds little to the diagnosis of CAV. Positron emission scans are used to detect ischemia, and scarred and hibernating myocardium; however, they suffer from poor sensitivity in detecting CAV. Studies have shown intracoronary ultrasound (IVUS) to be the most sensitive tool in detecting and following the progression of CAV, compared to coronary angiograms.
5. Answer A: PTLD is a unique polyclonal B-cell lymphoma that occurs in approximately 3.4% of all heart transplant recipients. Ninety percent of PTLDs are associated with EBV; with EBV D+/R- being a high-risk group for developing PTLD. These tumors usually arise 12 to 18 months following transplant, after a mononucleosislike illness (fever, sore throat, myalgias, and lymphadenopathy) and commonly are located intra-abdominally. Treatment includes decreasing the level of immunosu- pression, surgical debulking, cytotoxic chemotherapy, and radiation therapy if indicated; however, the response rate of advanced disease to treatment is poor (<50%).