Christopher M. Tarnay, MD
PELVIC FLOOR DISORDERS
Pelvic floor disorders (PFDs) include urinary incontinence, pelvic organ prolapse, fecal incontinence, and other sensory and emptying abnormalities of the lower urinary and gastrointestinal tracts. Nearly one-quarter of all women and more than one-third of older women reported symptoms of at least 1 pelvic floor disorder. The prevalence of PFDs increase as women age. Advances in modern medicine during the last 80 years have increased the life expectancy of women well into the eighth and ninth decades. We are caring for patients longer and better than ever, effectively managing chronic medical problems such as hypertension, cardiovascular disease, and diabetes, enabling women to enjoy longer and more productive lives. Using US Census data projections, by 2030, more than one-fifth of women will be 65 years of age or older. This will result in a large population of women living up to one-third of their life after menopause, thereby introducing a whole host of medical issues and health concerns.
A prime example of this is the problem of urinary incontinence, which has become more prevalent as the population of aging women grows. Urinary incontinence affects millions adult women in the United States. It is estimated to affect 50% of American women during their lifetime and results in substantial medical, social, and economic burdens. Despite its prevalence and estimated costs in excess of $19.5 billion annually, up to two-thirds of women do not seek help for incontinence, primarily because of social embarrassment or because they are unaware that help is available. Because of increasing awareness by both patients and physicians, the societal concept that incontinence is part of the “normal” aging process is no longer acceptable.
ESSENTIALS OF DIAGNOSIS
![]() The symptoms of urinary incontinence involve involuntary leakage of urine.
The symptoms of urinary incontinence involve involuntary leakage of urine.
![]() History and clinical examination can often effectively diagnose the correct condition.
History and clinical examination can often effectively diagnose the correct condition.
![]() Two most common types are stress incontinence (loss of urine with physical exertion) and urinary urge incontinence (sudden urge to urinate and losing urine before toileting).
Two most common types are stress incontinence (loss of urine with physical exertion) and urinary urge incontinence (sudden urge to urinate and losing urine before toileting).
![]() The term overactive bladder (OAB) is often used to describe the most common symptoms of urinary urgency, usually accompanied by frequency and nocturia, with or without urgency urinary incontinence.
The term overactive bladder (OAB) is often used to describe the most common symptoms of urinary urgency, usually accompanied by frequency and nocturia, with or without urgency urinary incontinence.
![]() The use of pads to protect soiling undergarments is the most common coping mechanism for women.
The use of pads to protect soiling undergarments is the most common coping mechanism for women.
![]() Behavioral methods such as fluid restriction, avoidance of dietary triggers, and pelvic floor muscle strengthening can be helpful to reduce symptoms.
Behavioral methods such as fluid restriction, avoidance of dietary triggers, and pelvic floor muscle strengthening can be helpful to reduce symptoms.
![]() Surgery, such as a midurethral sling, can be effective to cure stress urinary incontinence.
Surgery, such as a midurethral sling, can be effective to cure stress urinary incontinence.
![]() Medications or neuromodulation can be helpful for women with urinary urge incontinence who do not respond to behavioral methods.
Medications or neuromodulation can be helpful for women with urinary urge incontinence who do not respond to behavioral methods.
ANATOMY
The urinary and reproductive tracts are intimately associated during embryologic development. The lower urinary tract can be divided into 3 parts: the bladder, the vesical neck, and the urethra (Fig. 42–1). The bladder is a hollow muscular organ lined with transitional epithelium designed for urine storage. The bladder musculature consists of layers of smooth muscle, which are densely intertwined and constitute the detrusor muscle. The bladder stays relaxed to facilitate urine storage and contracts periodically to completely evacuate its contents when appropriate and acceptable. At the bladder base is the trigone, which is embryologically distinct from the bladder.

Figure 42–1. Interrelationships and approximate location of paraurethral structures. Levator ani muscles are shown as light lines running deep to the pelvic viscera. AT, arcus tendineus fasciae pelvis; BC, bulbocavernosus muscle; CU, compressor urethrae; D, detrusor loop; IC, ischiocavernosus muscle; LA, levator ani muscles; MAT, muscular attachment of the urethral supports; PVL, pubovesical ligament (muscle); US, urethral sphincter; UVS, urethrovaginal sphincter.
The 2 ureteral orifices and the internal urethral meatus form the boundaries of the trigone. The trigone has 2 distinct muscular layers: superficial and deep. The deep layer shares a similar cholinergic autonomic innervation as the detrusor muscle, whereas the superficial layer is densely innervated by noradrenergic nerves. This distinct difference in receptor distribution is important, as it provides opportunities to target more specific sites for pharmacotherapeutic intervention. The superficial detrusor layer extends muscular fibers that contribute to the distal urethra and posterior to the proximal urethra. The urethral “sphincter” itself is not a well-delineated structure; rather, it is a complex and intricate meshwork of intertwining smooth and striated muscle fibers that functionally responds neurophysiologically to variable degrees of vesicle pressures and facilitates urine storage and voiding.
The female urethra is approximately 3–4 cm long. The composition and support of the urethra and bladder neck play key roles in the function and maintenance of urinary continence. Together the striated urethral and periurethral muscles compose the extrinsic urethral sphincter mechanism. The urethral sphincter, along with the levator ani, function in the reflex contraction. The urethra is surrounded by dense vasculature that contributes to the urethral mucosal seal and urethral closure pressure. An abundance of submucosal glands are found along the dorsal surface. Most of the urethral diverticula arise from this area. The uroepithelium is stratified squamous (Fig. 42–2).

Figure 42–2. Urethral anatomy. The submucosal vascular plexus matures after puberty but undergoes great changes after menopause. The amount of smooth and especially striated muscles decreases with age, and the striated components become almost rudimentary. (Reproduced, with permission, from Rud T, Asmussen M. Neurophysiology of the lower urinary tract as measured by simultaneous urethral cystometry. In Ostergard DR, Bent AE (eds): Urogynecology and Urodynamics: Theory and Practice. 4th ed. Baltimore, MD: Williams & Wilkins; 1996, p. 55.)
Support of the urethra and distal vaginal wall are closely linked. For much of its length, the urethra is fused with the vaginal wall, and the structures that determine urethral position and distal anterior vaginal wall position are the same. The anterior vaginal wall and urethral support system provide a supportive layer on which the proximal urethra and mid urethra rest. The major components of this supportive structure are the vaginal wall, the endopelvic fascia, the arcus tendineus fasciae pelvis, and the levator ani muscles (Fig. 42–3). The endopelvic fascia is a dense, fibrous connective tissue layer that surrounds the vagina and attaches it to each arcus tendineus fascia pelvis laterally. Each arcus tendineus fascia pelvis in turn is attached to the pubic bone ventrally and to the ischial spine dorsally. They act like the cable of a suspension bridge that is supported at each end to the pelvis and provides attachment points along the vaginal length, providing the support of the anterior vaginal wall. Although it is well defined as a fibrous band near its origin at the pubic bone, the arcus tendineus fascia pelvis appears as a sheet of fascia as it fuses with the endopelvic fascia, where it merges with the levator ani muscles.

Figure 42–3. Lateral view of the components of the urethral support system. Note how the levator ani muscles support the rectum, vagina, and urethrovesical neck. Also note how the endopelvic fascia beside the urethra attaches to the levator ani muscle; contraction of the levator muscle leads to elevation of the urethrovesical neck. Puborectalis muscle is removed for clarity. (Reproduced, with permission, from Ashton-Miller J, DeLancey JOL. Functional anatomy of the female pelvic floor. Ann NY Acad Sci 2007;1101:266–296.)
The principal components of the basin-like pelvic floor are the pelvic bones (including the coccyx), the endopelvic fascia, and the levator and perineal muscles. These structures normally support and maintain the position of the pelvic viscera despite great increments in intraabdominal pressure that occur with straining, coughing, and heavy lifting when the patient is in the erect position. The urogenital hiatus (“anterior levator muscle gap”), which permits the urethra, vagina, and anus to emerge from the pelvis, is a site of potential weakness. Attenuation of the pubococcygeal and puborectal portions of the levator muscles, whether as the result of a traumatic delivery or of involutional changes, widens the levator gap and converts this potential weakness to an actual defect. If there has been a concomitant injury or attenuation of the endopelvic fascia (uterosacral and cardinal ligaments, rectovaginal and pubocervical fascia), heightened intraabdominal pressure gradually leads to uterine prolapse, along with anterior vaginal prolapse, rectocele, and enterocele. If the integrity of the endopelvic fascia and its condensations has been maintained, the incompetency of the genital hiatus and levator muscles may be associated only with elongation of the cervix.
Neuroanatomy
Neuronal innervation of the lower urinary tract is considered part of the autonomic and somatic nervous systems. The autonomic system (ie, the para-sympathetic and sympathetic components) receives visceral sensation and regulates smooth muscle actively during conscious and involuntary lower urinary tract functions. The autonomic nervous system constitutes the bulk of neural control of the lower urinary tract. Sympathetic contributions from T1–L2 and parasympathetic contributions from S2–4 compose the neuronal control system (Fig. 42–4). Voluntary control of micturition is controlled by the central nervous system. Cortical control of the detrusor muscle rests in the supramedial portion of the frontal lobes and in the genu of the corpus callosum. Receiving both sensory afferent and modulating motor efferent nerves, the net effect is that the brain provides tonic inhibition of detrusor contraction. Lesions in the frontal lobe chiefly cause loss of voluntary control of micturition and thus loss of suppression of the detrusor reflex, resulting in uncontrolled voiding or urge urinary incontinence. The pons and mesencephalic reticular formation in the brainstem constitute the micturition center. A reflex activation in the central brainstem and peripheral spinal cord mediate a coordinated series of events, consisting of relaxation of the striated urethral musculature and detrusor contraction that result in opening of the bladder neck and urethra. Lesions that interrupt these pathways have various effects depending on the level of interruption, essentially resulting abnormal detrusor function.
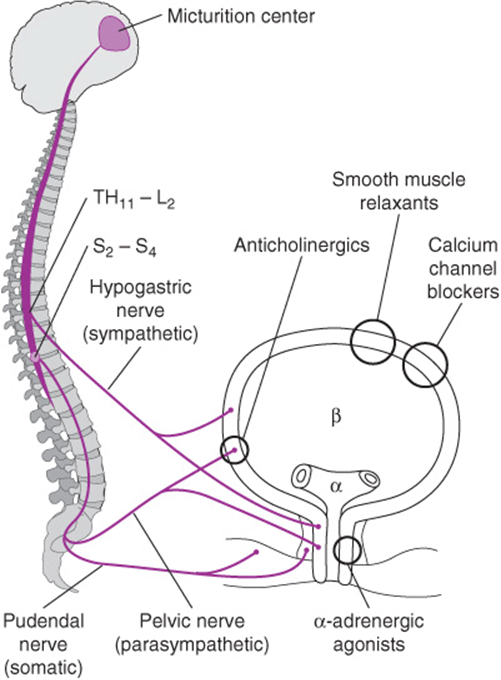
Figure 42–4. Schematic neuroanatomy of the lower urinary tract, with major sites of drug action. (Reproduced, with permission, from Sourander LB. Treatment of urinary incontinence. Gerontology1990;36(Suppl 2):19. Copyright Karger S.)
URINARY INCONTINENCE
Definition
Urinary incontinence as defined by the Consensus Committee on Pelvic Floor Disorders is the complaint of any involuntary leakage of urine. Incontinence can be a sign, a symptom (patient complaint), or a condition diagnosed by an examiner. There are many types and causes of urinary incontinence (Table 42–1). The reported incidence of urinary incontinence varies widely, ranging from 10–70% of women living in a community setting to more than 50% of women living in a nursing home. Incontinence becomes more common as women age, particularly after menopause.
Table 42–1. Differential diagnosis.

Pathogenesis
PFDs are most assuredly caused by multiple factors. The multifactorial model is the clearest one to explain the incidence of PFD and variety of anatomic findings despite shared risk factors.
For urinary incontinence, numerous factors play a role in maintaining urinary continence; therefore, the development of incontinence is frequently not attributable to any single cause. Sex, age, hormonal status, birthing trauma, and genetic differences in connective tissue all contribute to the development of incontinence. Urinary incontinence is 2–3 times more common in women than in men because of women’s shorter urethral length and the risk of connective tissue, muscle, and nerve injury associated with childbirth. Observational studies have consistently noted a high incidence of incontinence in the elderly population, with 1 study finding a 30% higher prevalence for each 5-year increase in age. The association of childbirth with urinary incontinence has long been suspected and has generated new interest in identifying the causes. In 1 study of more than 15,000 women, the risk of developing urinary incontinence was 2.3 times higher in women who had a vaginal delivery compared with nulliparous women. Damage to the pelvic floor neuromusculature during vaginal delivery may lead to loss of pelvic muscle strength and nerve function, resulting in both stress urinary incontinence (SUI) and pelvic floor support defects. Although muscle strength may be regained over time or with the help of pelvic floor muscle exercises, dysfunction may be permanent.
Aging and incontinence are closely associated. The prevalence of incontinence increases as women age, but the specific cause is unclear. Global decrease in the storage capacity, reduced receptor response, general loss in muscle tone, or latent manifestation from denervation during parturition may all be important factors. The state of hypoestrogenism as a woman transitions to menopause may also contribute to urinary incontinence. Although estrogen reduces urinary urgency, results from studies specifically examining menopausal status have been equivocal, with some studies showing a positive association and others showing no association. Abnormalities in the muscular components and innervation of the pelvic floor and the connective tissue to this region likely contribute to the multifactorial etiology of incontinence. Initial observations that the prevalence of abdominal hernias, lower leg varices, and uterine prolapse was higher in women with SUI suggested that connective tissue weakness might identify women at risk for developing incontinence. Subsequent studies have supported a connection between relative collagen deficiencies in the connective tissues of incontinent patients versus continent controls.
Incontinence affects a woman’s quality of life, and it is an uncomfortable and embarrassing problem. The psychosocial impact on the patient as well as her family is enormous. Women with urinary incontinence are reported to be more depressed, to have lower self-esteem, and to be ashamed about their appearance and the odor. Urinary incontinence affects sexual desire and reduces sexual activity. This can curb social interactions to the point where individuals become isolated and even entirely homebound.
Prevention
One of the first attempts at the prevention of urinary incontinence was described by Dr. Arnold Kegel in the 1950s. To reduce the risk of postpartum urinary loss, women were taught to contract and thereby strengthen the levator ani muscles in what is now commonly known as a “Kegel” exercise. Other attempts at mitigating the potential detrimental impact of pregnancy and vaginal delivery on the subsequent development of urinary incontinence have led to the increasing prevalence of maternal choice caesarean delivery as a method of perineal preservation. To date there are no high-quality data supporting either empiric pelvic floor strengthening or the avoidance of vaginal delivery as a protective measure against future urinary incontinence.
For urge-related urinary loss, the avoidance of caffeinated beverages, alcoholic drinks, or other potentially irritative foods or beverages can be suggested as a measure to reduce urgency and frequency.
Clinical Findings
The first step in evaluating an incontinent patient is a thorough history. The nature and extent of the patient’s lower urinary tract symptoms (LUTS) should be elucidated. Knowledge of the duration, frequency, and severity of the urinary incontinence is essential to understanding the social implications and its impact on the patient’s life and aids the clinician in determining the direction and extent of diagnostic and therapeutic measures (Table 42–2). A multitude of diagnostic and imaging studies are available, but taking a thorough but focused urogynecologic history can isolate many of the easily reversible causes of incontinence (Table 42–3). Knowledge of the use of protective items, such as sanitary napkins, panty liners, absorbent pads, or adult diapers, is useful in quantitating urinary loss. Including questions about menopausal status and use of hormone treatment, history of urinary tract infections, previous surgery to remedy incontinence, and the patient’s mental and functional status are essential.
Table 42–2. Lower urinary tract symptoms.

Table 42–3. Helpful questions when taking history of incontinence.

A. Patient Questionnaires
Survey instruments can be valuable in helping to identify and determine the severity of patient symptoms. Although initially designed for clinical research, short forms of longer questionnaires exist and can be used for clinical care. Surveys such as the Urinary Distress Inventory (UDI-6) and Incontinence Impact Questionnaire (IIQ-7) can be easily filled out by a patient to facilitate diagnosis and to follow treatment interventions.
B. Voiding Diary
A voiding diary, or urolog, that quantitates frequency and volume is a helpful tool. For a 24- to 48-hour period, the patient records all fluid intake and measures and records all urine output, including frequency and episodes of leakage (Fig. 42–5). Numerous studies have validated the voiding diary as a reliable tool in the diagnosis and management of urinary urgency or urge incontinence. These data are beneficial to the physician because they clarify home voiding patterns, particularly in the elderly. They are often useful to patients as well because they provide a focus on the problem and can serve as a baseline for treatment interventions such as behavioral training, bladder drills, and pharmacologic management.

Figure 42–5. Urinary diary (urolog).
C. Urinalysis
Examination of the urine is an essential part of the workup of urinary incontinence for any patient with LUTS. Infection is a common cause of urinary complaints, including frequency, urgency, and incontinence. A clean-catch voided specimen is suitable for routine urinalysis; however, a sterile “in and out” catheterized specimen is appropriate for patients unable to correctly perform collection or if urine culture has been previously equivocal because of skin flora contamination.
Urinary protein, glucose, ketones, hemoglobin, casts, and nitrates can indicate primary renal disease or injury. Microscopic evaluation of the urinary sediment may indicate renal tubular damage with the presence of casts or indicate infection by the presence of leukocytes and red blood cells. More than 6–8 white blood cells per high-power field along with the presence of bacteria are very suggestive of urinary tract infection.
D. Physical Examination
A general gynecologic and neurologic examination should be performed on all patients, with a focus on the vaginal walls and pelvic floor. The patient should come to the clinic with a comfortably full bladder for spontaneous uroflowmetry and postvoid residual assessment. An examination should be performed with the patient in the lithotomy position. The examination should begin with an assessment of the vulvar area. In postmenopausal patients, atrophy and change in labial architecture may be due to estrogen deficiency. Vulvar dermatoses may be coexistent with vulvar complaints ascribed to incontinence. The presence of inflammation or irritation from chronic moisture or pad usage should be noted. The presence of discharge should be noted because this may mimic urinary incontinence. Examination of the urethra with palpation of the anterior vaginal wall under the urethra for fluctuance, masses, or discharge may reveal signs of urethral diverticulum, infection of the urethra, or rarely carcinoma. Tenderness may point to urethral pain syndrome, a condition marked by episodic urethral pain usually with voiding, and by daytime frequency and nocturia.
Vaginal wall integrity must be assessed. Vaginal rugae, or the folds in the epithelium, are normal and tend to be absent if the underlying supportive endopelvic fascia is detached. The presence of anterior wall defects (cystoceles), posterior vaginal wall defects (rectoceles), and apical defects (uterine prolapse or enteroceles) can be quantified. The uterocervical position, or, if the woman has had a hysterectomy, the cuff position and its descent should be recorded. The position of the vaginal walls should be noted in the lithotomy position at rest and with Valsalva’s/straining maneuver. A Sims’ speculum or the lower blade of a Graves’ speculum allows easy visualization of either the anterior or posterior vaginal wall. The severity of vaginal laxity, which may be masked in the supine position, can often best be elicited by repeating the examination in the standing position while the patient places 1 foot on the step of the examination table or on a small portable step.
Mobility at the level of the bladder neck is often seen in women with urinary incontinence. Urethral hypermobility must be interpreted with caution because it may be present in women without incontinence. In the absence of mobility, the physician must question the diagnosis of stress incontinence and entertain the possibility of a fixed and damaged urethral sphincter (intrinsic sphincteric deficiency) to explain stress-related urinary loss.
E. Urinary Cough Stress Test
Having the patient perform Valsalva’s maneuver or to cough forcefully multiple times to reproduce urine loss at the beginning of the examination may reveal the presence of incontinence. Observation of urine lost immediately with the cough or Valsalva’s maneuver may obviate the need for more complex urodynamic testing if the complaint is minor. If no urine loss is exhibited, the patient is asked to stand with legs shoulder width apart and asked to cough. Immediate loss of urine suggests a diagnosis of SUI.
Bimanual examination to evaluate the uterine size, position, and descent within the vaginal canal and palpation of the ovaries should be performed. A rectovaginal examination permits adequate assessment of the posterior vaginal wall. Anal sphincter tone can be assessed at rest and with anal tightening. The presence of fecal impaction must be ruled out because this condition has been shown to be a contributing factor to urinary incontinence, particularly in the elderly population.
F. Neurologic Examination
The control of micturition is complex and multitiered, with both autonomic and voluntary control. In addition to a complete history and screening for neurologic symptoms, a thorough physical examination is important because many neurologic diseases may present with voiding dysfunction in the absence of overt neurologic findings.
Mental status, motor strength, sensory function, deep tendon reflexes, and sacral spinal cord integrity should all be assessed. Testing the patient’s orientation to place and time and assessing speech and comprehension skills will help to ascertain her mental status. Motor control may be diminished in focal brain or cord lesions, most commonly Parkinson’s disease, multiple sclerosis, and cerebrovascular accident. Motor strength is tested in the lower extremities by assessing hip, knee, and ankle flexion, as well as ankle eversion and inversion. Deep tendon reflexes are tested at the patella, ankle, and foot planus. Sensation can be tested at the dermatomes using light touch and pin-prick over the perineum and thigh area. Deficits should be noted, but it should be kept in mind that there is considerable overlap in sensory innervation in the sensory nerve roots. The sacral spinal cord nerve roots 2–4 contain vital neurons controlling micturition. The anal wink reflex and the bulbocavernosus reflex can confirm integrity of neurovisceral and urethral reflex functions. These reflexes can be evoked by stroking the perianal area and looking for an external anal sphincter contraction, and by tapping or gently squeezing the clitoris and watching for contraction of the bulbocavernosus muscle, respectively. These reflexes are often easier to elicit at the beginning of the examination, but their absence is not always indicative of neurologic deficit. Clinically observed neurologic deficits should lead to a neurologic consultation.
G. Urodynamics
A urodynamic study is any test that provides objective dynamic information about lower urinary tract function. Many methods and tests are available (Table 42–4). Some methods are simple, such as diaries that track frequency and volume of urination, and some methods are more complex, requiring special equipment and training. A cystometrogram can aid in the discovery of an unstable bladder, overflow incontinence, reduced bladder capacity, or abnormalities of bladder sensation. A cystometrogram can be performed using water manometry or more advanced methods. Complex urodynamic testing increases the diagnostic accuracy and may often identify the reason for failure of previous therapy. Uroflowmetry can be performed to measure detrusor pressure and flow rate to evaluate for voiding dysfunction. If a poorly functioning urethra, such as in intrinsic sphincter deficiency (ISD), is suspected, urethral pressure profile (UPP) or abdominal leak point pressure (ALPP) can be measured to evaluate urethral closure pressures. Such testing is particularly helpful in difficult or complex cases.
Table 42–4. Urodynamic testing methods.

The indications for more complex testing in the form of multichannel urodynamics are not standardized, and each patient must be assessed individually (Table 42–5). However, some basic criteria, if met, indicate a need for urodynamic evaluation, which can aid in more accurate diagnosis and thus appropriate medical or surgical management.
Table 42–5. Indications for multichannel urodynamic testing.

H. Cystourethroscopy
Endoscopic evaluation is an invaluable adjunct for the diagnosis and management of the urogynecologic patient. It is a simple office procedure that can yield important data when performed by experienced operators. Cystourethroscopy is indicated for hematuria and irritative voiding symptoms, particularly in the presence of previous continence surgery, obstructive voiding, suspicion of diverticula or fistula, or persistent incontinence and as a preoperative evaluation before reconstructive pelvic surgery.
I. Imaging Tests
Radiologic studies can be an integral component of the evaluation of lower urinary tract dysfunction and abnormalities. However, these modalities are of limited use in the evaluation of all but the most complex of incontinent patients. Ultrasound has become an increasingly frequent adjunct investigation for pelvic floor disorders both in the office and in the urodynamic laboratory. Magnetic resonance imaging (MRI) has become more extensively used in patients with PFDs and prolapse. As the technique becomes less costly, applications for the uses of MRI to aid in the urogynecologic workup will expand.
Differential Diagnosis
See Table 42–1.
STRESS URINARY INCONTINENCE
Stress urinary incontinence (SUI) is defined as the complaint of involuntary leakage on effort or physical exertion or on coughing or sneezing. Normally, at rest the intraurethral pressure is greater than the intravesical pressure. The pressure difference between the bladder and the urethra is known as the urethral closure pressure. If intraabdominal pressure increases, as it does with a cough, sneeze, or strain, and if this pressure is not equally transmitted to the urethra, then continence is not maintained and leakage of urine occurs. What is thought to cause this inequity of pressure transmission is not universally accepted; however, surgical therapy directed at stabilization of the suburethral support appears to be the mechanism for long-term correction.
Treatment
A. Nonsurgical Measures
For most patients with SUI, consideration of the simplest, least invasive, and least costly interventions is appropriate (Table 42–6). Dietary measures can be instituted, with identification of items that can be modified. Reduction in consumption of caffeinated beverages and alcoholic drinks should be encouraged. Fluid restriction in patients without chronic medical problems, such as cardiovascular, renal, or endocrinologic disease, can be attempted. Timed voiding to prevent filling the bladder to a capacity that causes urine loss should be undertaken with the use of a urine diary. The diary can also facilitate discussion between patient and clinician as therapy progresses.
Table 42–6. Nonsurgical management of urinary incontinence.

Pelvic floor muscle exercises or Kegel exercises have been found to be extremely helpful in patients with mild to moderate forms of incontinence. Focused repetitive voluntary contractions of the levator ani muscles (pubococcygeus, coccygeus, and iliococcygeus) created by having the patient contract or “squeeze” the muscle as if to prevent the passage of rectal gas is an effective therapy. The contractions exert a closing force on the urethra and increase muscle support to the pelvic organs. The patient should be provided written and verbal instructions on performing the exercises. Repetitions, with each contraction held for 3–5 seconds alternated with periods of relaxation, should be begun at 45–100 repetitions daily. In settings in which the patient is motivated and has individual instruction and thorough follow-up and support, results for cure or improvement of bladder control (reduction in urine loss) can be up to 75%.
1. Biofeedback—Biofeedback is an adjunct to pelvic floor exercises that is used to facilitate the patient’s comprehension of the proper muscles to contract. By using a pressure catheter and myographic monitoring, a visual or auditory signal of the physiologic response can be provided to the patient to help refine exercise skills. Using surface electro-myography on the perineum to measure levator contraction and a pressure monitor in the vagina or rectum to indicate abdominal pressure, the patient can be instructed to preferentially contract the pelvic floor without concomitant abdominal contraction. Studies using a variety of techniques demonstrate a 54–95% cure rate or improvement in SUI. The efficacy of this modality is highly dependent on patient motivation and compliance. Pelvic floor muscle exercises with or without biofeedback require continued implementation and practice or effectiveness will wane.
2. Electrical stimulation—As an alternative to active patient contraction of the levator muscles, electrical stimulation of the muscles via small electrical currents can be used to help both SUI and mixed incontinence. Using intravaginal or transrectal electrodes with stimulators, the pelvic muscles automatically contract and are thereby artificially “trained.” When used long term, weakened muscles are strengthened and innervation re-established during activation. Experiences with the devices are variable, but they generally show a positive impact on incontinence and acceptable patient tolerance.
3. Pessaries—Intravaginal devices or pessaries to correct the anatomic deficits associated with stress incontinence have long been used to address this vexing problem. Many devices have been proffered, but long-term solutions to incontinence have yet to be proven in the general population. Pessaries, traditionally used for treatment of genital prolapse, have also been shown to have a potential role in supporting the bladder neck and urethra and preventing stress incontinence. Many pessary devices designed to fit within the vagina and elevate the bladder neck are available. Continence can often be achieved because many devices adequately obstruct the bladder neck and urethra. As with all intravaginal devices, maintenance is essential to avoid urinary obstruction and vaginal erosion if the pessary is too compressive.
B. Surgical Management
Surgical treatment may be offered for moderate to severe incontinence. Urinary incontinence is not a life-threatening condition, and the decision to operate must be based on the patient’s symptoms and the impact on daily life. Many patients are able to tolerate slight urine loss, and what often provokes a desire for treatment is an increase in loss above a tolerable threshold. If medical management to improve bladder control is possible and symptoms are reduced to below this threshold, then medical management is most desirable. If not, surgery should be considered.
At least 130 operative procedures have been described for treatment of female urinary stress incontinence. It is therefore not surprising that many of these procedures have not resulted in long-term success. For patients who desire surgical correction, the options can be categorized by method of surgical approach (Table 42–7). Common to most surgical procedures is restoration of bladder neck support by elevation of the urethrovesical junction. Some procedures reconstruct bladder neck supports and provide a stable suburethral layer.
Table 42–7. Surgical treatment of stress urinary incontinence.

Assessment of the cure rate of any surgical treatment for genuine stress incontinence must take into account the selection of patients, accuracy of the preoperative diagnosis, length of postoperative follow-up, and criteria for cure. Reported cure rates for procedures range from 60–100%, with 75–90% being the generally accepted rate. Most failures appear to result from incorrect preoperative diagnosis, poor surgical technique, and healing failures.
1. Abdominal Retropubic Colpopexy—The Marshall-Marchetti-Krantz (MMK) and Burch’s colposuspension are the 2 classic retropubic surgeries for incontinence. They share the same mechanism of correction. First, both suspend the periurethral and paravaginal tissue at the level of the urethrovesical junction, and second, both use a firm point of attachment for fixation of these suspension sutures. In the MMK procedure, the sutures are fixed to the periosteum of the pubic bone, and in Burch’s procedure, the iliopectineal ligament (Cooper’s ligament) (Fig. 42–6). Burch’s colposuspension has been a longstanding treatment of patients with hypermobility of the bladder neck and genuine SUI. In both longitudinal studies and randomized comparative trials against other procedures, Burch’s procedure maintains high objective and subjective cure rates of 80% after 5 years and 68% after 10 years of follow-up.

Figure 42–6. Abdominal surgical procedure to correct stress incontinence. A: Anterior vaginal wall has been mobilized. Two sutures have been placed on either side and far lateral from the midline. Distal sutures are opposite the midurethra. Proximal sutures are at the end of the vesicourethral junction. Sutures are attached to Cooper’s ligament. B: Cross-section shows urethra free in the retropubic space, with anterior vaginal wall lifting and supporting it. (Reproduced, with permission, from Tanagho EA. Colpocystourethropexy. J Urol 1876;116:751. Copyright 1976 by Williams & Wilkins.)
A laparoscopic approach to Burch’s colposuspension offers the benefit of minimally invasive surgery with the same level of efficacy.
2. Suburethral Slings—The suburethral sling was one of the original surgical procedures developed for correction of SUI. The concept of restoring continence by encircling the urethra with supportive tissue, either from the patient or foreign material, was introduced at the beginning of the 20th century. Contemporary techniques have used a patient’s own fascia harvested from the leg or rectus fascia, or donor fascia in the form of cadaveric fascia lata. Cure rates of suburethral sling procedures for genuine stress incontinence vary from 70–95%. Reported rates vary because of the heterogeneity of patients, and many are previous surgical failures. Variations in sling material and technique have made cure rates among sling techniques difficult to interpret. Furthermore, most studies vary in the definition of cure and may not distinguish between cure and improvement.
In a large prospective trial comparing suburethral fascial slings versus Burch’s procedure, at 24 months, success rates were higher for women who underwent the sling procedure than for those who underwent Burch’s procedure (66% vs. 49%, respectively). These efficacy results are notable; in a review study summarizing cure rates of surgical treatments for SUI, 16 studies comparing sling procedures with colpo-suspension were reviewed. Of the 4 that were randomized controlled trials comprising 150 patients, none reported a difference in cure.
3. Midurethral Slings—This recent modification of the sling is the use of tension-free vaginal mesh made of polypropylene placed at the level of the midurethra. This technique, developed in Sweden, was introduced to the United States in the late 1990s. Use of tension-free vaginal tape (TVT) (Fig. 42–7) was developed as a minimally invasive technique for surgical correction of genuine SUI. The initial study had an 84% cure rate in 75 women with 2-year follow-up. The success rates of this procedure that have been reported in trials range from 86% to 99%, with up to 10-year follow-up. Because these results have been reproduced, most clinicians consider the midurethral sling as the gold standard for continence surgery.
Because of the success of TVT midurethral sling, numerous other devices using the same principles and technique are available. All use a polypropylene mesh but have different designs of delivery needle/trocar, mesh construction, and sheath type. Comparative data between devices are scanty.

Figure 42–7. A: Placement of midurethral sling in the sagittal view. B: Retropubic passage of midurethral sling. C: Transobturator passage of midurethral sling.
Because of the success of TVT midurethral sling, numerous other devices using the same principles and technique are available. All use a polypropylene mesh but have different designs of delivery needle/trocar, mesh construction, and sheath type. Comparative data between devices are scanty.
An alternate to retropubic passage is the transobturator route, in which the sling is passed through the obturator foramen laterally. This creates a more lateral point of fixation. The purported advantage is reduction in bladder, bowel, or major vascular injury because this method avoids the space of Retzius and does not traverse the peritoneal space. Studies suggest 2-year cure rates comparable to those of retropubic passage (94%). There are reservations regarding the transobturator approach as it relates to correcting SUI due to a poorly functioning urethra and in complications related to groin pain, particularly in thin patients (Fig. 42–7).
A more recent introduction is the single-incision slings, in which instead of passing externally through the obturator foramen, the mesh sling is anchored internally into the fascia/muscle of the obturator.
C. Periurethral and Transurethral Injection
Periurethral or transurethral injection of a bulking agent into the submucosal space of the bladder neck causes narrowing or coaptation of the proximal urethra and bladder neck opening. This increases urethral resistance to involuntary urine loss without changing resting urethral closure pressure. Currently glutaraldehyde cross-linked bovine collagen and calcium hydroxyl apatite are the most commonly used materials. This procedure is generally reserved for genuine SUI caused by intrinsic sphincteric deficiency. The injections can be performed with the patient under sedation with local anesthetic in an outpatient or office setting. These materials are biologic and resorb over time. Improvement and cure rates are 53–66% in the short term, and complications are minimal.
D. Artificial Sphincters
The artificial urethral sphincter is an effective option for patients with incontinence not amenable to standard surgical treatment because of urethral scarring or atony. The artificial urinary sphincter is best used in patients with incontinence due to poor urethral sphincter function. The sphincter obstructs the urethra by compressing the bladder neck via a pressure-regulated balloon and releases the compression when the patient desires to void. Reported success rates are up to 91%, but complication rates are high, with 21% of patients requiring surgical replacement of parts or the entire sphincter.
URGE URINARY INCONTINENCE
Urge urinary incontinence is the complaint of involuntary leakage accompanied by urgency. Urge urinary incontinence may be associated with involuntary contractions of the bladder or detrusor contractions; however, most often it is attributable to increased sensation with strong urge. The term overactive bladder (OAB) is often used to describe the most common symptoms of urinary urgency, usually accompanied by frequency and nocturia, with or without urgency urinary incontinence, in the absence of urinary tract infection or other obvious pathology. Not all patients with OAB have detrusor overactivity, and neither will all patients with detrusor overactivity have OAB. OAB is a term that lends itself to encompassing all conditions related to bladder urgency and frequency with and without incontinence. OAB has become a preferred clinical term because it comprises the spectrum of related symptoms.
Pathogenesis
The incidence of OAB varies depending on the population studied and the definition applied. Consequently, the reported prevalence varies widely from 8–50% in the general population, and in women older than 65 years, it is estimated to be at least 38%. An important concept is that involuntary detrusor contractions for bladder emptying are normally overridden by cortical inhibition of reflex bladder activity. In the majority of cases the cause of OAB symptoms is unknown. Patients with underlying neurologic disease may manifest with urinary incontinence. Although neurologic disease is not a common cause of OAB, multiple sclerosis, cerebrovascular disease, Parkinson’s disease, and Alzheimer’s disease are most often associated with involuntary bladder contractions.
Clinical Findings
OAB is suggested by urinary frequency often associated with a strong urge or a sense of impending urine loss. Incontinence often occurs before reaching the toilet. Physical or environmental stimuli, such as running water, cold weather, or hand washing, may elicit an urge. Patients often describe “key in lock” syndrome. This is typically characterized by an uncontrollable urge to void when unlocking the door after returning from a trip out of the house. The first thing done upon return is to immediately rush to the toilet or risk losing urine.
Treatment
Adequate therapy depends greatly on accuracy of diagnosis of OAB. History is most often suggestive, and the diagnosis can be supported by urinary diary or confirmed with office cystometry or more precisely with multichannel urodynamics.
Patients with OAB first should be offered simple treatments. Behavioral modifications and medical treatment are the standard first-line therapy for urge urinary incontinence.
A. Behavioral Therapy
Behavioral therapy includes bladder training, timed voiding, and pelvic floor muscle exercises. Bladder training is an educational program that combines written and verbal instruction to educate patients about the mechanisms of normal bladder control with the teaching of relaxation and distraction skills to resist premature signals to urinate. Creating a voiding schedule for which the patient urinates at preset intervals while attempting to ignore the urge to urinate may progressively lead to re-establishment of cortical voluntary control over the micturition reflex.
Timed voiding is a form of bladder retraining that again mandates regularly scheduled voiding and attempts to match the person’s natural voiding schedule. No effort is made to motivate the patient to delay voiding by resisting the urge. This method is geared more toward elderly patients with more challenging problems who have skilled help available.
Pelvic floor exercises may aid in the treatment of OAB. Evidence supports the utility of this modality in all types of incontinence. Particularly when augmented with biofeedback, pelvic floor exercises can greatly reduce symptoms of urinary frequency and urge incontinence, by up to 54–85%.
B. Pharmacologic Therapy
One of the most effective and popular treatments for urge urinary incontinence and OAB is drug therapy. Numerous agents for the treatment of these patients have been tried over the years, but only a few have demonstrated substantial impact on reduction of symptoms in controlled trials. One of the main difficulties in treating OAB is that the cause of OAB is still under investigation. The drugs available can be divided into classes by mechanism of action (Table 42–8).
Table 42–8. Pharmacologic treatment of urge incontinence.

Antimuscarinics, or anticholinergics, have become the mainstay of drug treatment of OAB. Acetylcholine is the primary neurotransmitter involved with bladder contraction. The detrusor muscle of the bladder is heavily populated with cholinergic receptors. Anticholinergic activity, therefore, is a property of most drugs used to treat OAB. The mainstays of drug therapy for OAB include oxybutynin chloride and tolterodine. Oxybutynin chloride has been shown in randomized placebo-controlled trials to be effective in increasing bladder capacity, decreasing the frequency of detrusor contractions, and improving symptoms of urinary urgency in approximately 70% of patients. It is effective for both idiopathic and neuropathic etiologies of detrusor instability.
Tolterodine is a medication designed specifically for OAB. It also has anticholinergic activity with specificity for the bladder, and it acts through muscarinic receptors as well as smooth muscle relaxation. In a multicenter randomized controlled trial, the medication compared favorably with oxybutynin in terms of reducing the number of micturitions in 24 hours and the number of incontinent episodes. Because of its bladder specificity, tolterodine has a more favorable side effect profile than oxybutynin. It is also dosed less frequently and improves patient compliance. Both are available in immediate-release and long-acting formulations. Oxybutynin is also available for delivery in a transdermal patch.
A large randomized comparative trial evaluating the performance of the long-acting formulations of oxybutynin and tolterodine demonstrated similar efficacy. Adverse events were similar, but the occurrence of dry mouth was higher in the oxybutynin group.
Other antimuscarinics are available. All significantly improve OAB symptoms compared with placebo. Evidence suggests that medications such as darifenacin, solifenacin, trospium chloride, and fesoterodine have side effects similar to or lower than those of traditional antimuscarinics. Comparative trials exist essentially demonstrating comparable efficacy and adverse event profiles. Most of the clinical effectiveness of antimuscarinics, however, is limited by side effects. Long-term compliance of these medications appears to be imperfect at best.
C. Sacral Neuromodulation
Electrical stimulation to the nerves that control the bladder have been used in people with OAB. An electrode is placed via the sacral foramen alongside a sacral nerve (usually S3). In a second procedure, the electrode is connected by cables under the skin to an implanted programmable pulse generator that provides stimulation within set stimulation parameters. Implantation of the pulse generator is usually carried out only after a successful trial. The technology has been used for patients with OAB, urgency incontinence, and voiding (retention of urine) difficulties, and for some patients with defecation problems. It has also been used in the management of chronic pelvic pain, and it is approved for fecal incontinence, but this indication is not included in this review.
In patients with urge urinary incontinence caused by detrusor overactivity, sacral neuromodulation seems to act primarily by activation of nonmuscular afferent sacral nerve fibers that inhibit parasympathetic motor neurones in the cord through interneurons. It appears that any change induced by implanted electrodes persists only while the stimulator is turned on, returning to baseline when the stimulation is stopped. A longer lasting benefit has, however, been reported for stimulation via vaginal or anal plug electrodes, persisting after active treatment stops. These devices are expensive, the surgery is invasive, and many people need another operation.
It is not clear how best to use these devices. However, implantable stimulators that provide continuous electrical stimulation to the nerves or nerve roots supplying the bladder and pelvis, or to the peripheral nerves that share the same nerve roots, can benefit selected patients with difficult-to-control urinary problems.
MIXED INCONTINENCE
Mixed incontinence occurs when both stress incontinence and urge incontinence occur simultaneously. Patients may present with symptoms of both types of incontinence. These patients present both a diagnostic and therapeutic dilemma. The prevalence of mixed incontinence is more common than most practitioners realize. A detailed history will reveal symptoms of SUI with urine loss associated with cough, sneeze, or other increase in Valsalva’s pressure, as well as urinary urgency, frequency, and concomitant incontinence. The coexistence of these 2 conditions may be brought about by many causes. Patients with SUI often preemptively urinate to avoid a full bladder and subsequent urine loss, thereby conditioning the bladder to habituate to a low functional capacity. This may promote premature signaling of bladder fullness and result in frequent urge symptoms.
Treatment
For mixed incontinence, treatment should be based on the patient’s worst symptoms. Often patients can prioritize their symptoms, stating that one component impacts their life more than the other. By having the patient separate the symptoms, a practical management plan with realistic expectations can be devised. A great disservice can be done by operating on a patient to restore bladder neck support and remove stress symptoms when the patient’s main concern is daily urge incontinence while she is at work. Conservative measures should be tried first, and if symptoms do not improve, surgical measures can be entertained to target alleviation of the stress component; however, there is a 50–60% chance that urge symptoms may resolve after a midurethral sling is performed.
OVERFLOW INCONTINENCE
Overflow incontinence is the involuntary loss of urine associated with bladder overdistention in the absence of detrusor contraction.
Pathogenesis
This condition classically occurs in men who have outlet obstruction secondary to prostatic enlargement that progresses to urinary retention. In women this is a relatively uncommon cause of urinary incontinence. When it does occur, it can be from increased outlet resistance from advanced vaginal prolapse causing a “kink” in the urethra or after an anti-incontinence procedure that has overcorrected the problem. Additionally, it can result from bladder hyporeflexia from a variety of neurologic causes (Table 42–9).
Table 42–9. Causes of overflow incontinence.

Overflow incontinence most often occurs due to postoperative obstruction if the bladder neck is overcorrected or with a hyporeflexic bladder due to neurologic disease or spinal cord injury. The normal act of voiding is controlled centrally by sacral and pontine micturition centers. Impaired emptying can be the result of disruption of either central or peripheral neurons mediating detrusor function. Failure to identify the cause early may lead to permanent dysfunction and may lead to injury to the detrusor muscle or compromise in the parasympathetic ganglia in the bladder wall.
Clinical Findings
Usually symptoms are loss of urine without awareness or intermittent dribbling and constant wetness. Suprapubic pressure or pain may be associated. Patients will often note a sensation of a full bladder and the need to strain in order to empty or apply suprapubic pressure to void. Patients are at risk for urinary tract infection secondary to persistent residual urine in the bladder, which acts as a medium for bacterial growth. It is commonly seen after a bladder neck suspension. Complaints of poor urinary stream and sense of incomplete emptying combined with having to strain or apply hand pressure to void are likely.
Evaluation should always include a postvoid residual and, if the diagnosis is questionable, voiding pressure flow studies. An imaging study of the upper urinary tract to evaluate the ureters and kidney should follow, because persistent high-volume retention can lead to reflux and hydroureter or hydronephrosis and renal injury if left unchecked.
Treatment
Bladder drainage to relieve retention is the first priority. Self-intermittent or prolonged catheterization may be necessary, depending on resolution of the inciting cause. In cases of postoperative urinary retention, bladder function can be evaluated by serial postvoid residual urine determinations. Although no normal volume for residual urine is universally accepted, less than 100 mL is generally considered to be within normal limits and greater than 150 mL is considered abnormal. More than 1 value is needed because persistently high residual volumes will require prolonged catheterization.
When urinary retention occurs in the setting of neurologic disease, diabetes, or stroke, correction of the underlying cause is often impossible; therefore, the goal is to prevent injury or damage to the upper urinary tract. Intermittent self-catheterization is preferable to an indwelling catheter, which may predispose to infection, bladder spasms, or erosion.
Medical therapy may assist in the care of these patients. Acetylcholine agonists can stimulate detrusor contractions in patients who have vesical areflexia. α-Adrenergic blockers can facilitate bladder emptying by relaxing tone at the bladder neck.
Behavior modification in the form of timed voiding on a preset schedule to empty regardless of urge will prevent accumulation of excess urine. Usually a voiding pattern of every 2–3 hours is preferable. In bladder areflexia, manual pressure or abdominal splinting may facilitate emptying.
BYPASS INCONTINENCE
Urinary loss due to abnormal anatomic variations is uncommon but extremely important to consider in the evaluation of the incontinent woman. Bypass incontinence may often mimic other forms of urinary incontinence but usually presents as constant dribbling or dampness. Patients may complain of positional loss of urine without urge or forewarning. Diagnosing this type of incontinence requires a high level of suspicion and an understanding of the underlying anatomic deviation in the lower urinary tract. Genitourinary fistulas (vesicovaginal or ureterovaginal) can be a debilitating cause of incontinence and are formed because of poor wound healing after a traumatic insult (eg, obstetric laceration, pelvic surgery, perineal trauma, or radiation exposure). Leakage due to fistulas is generally continuous, although it may be elicited by position change or stress-inducing activities. Evaluation should include a careful examination of the vaginal walls for fistulas. This can be facilitated by filling the bladder with milk or dilute indigo carmine dye and looking for pooling in the vaginal canal. Pad testing can be performed by having the patient ingest 200 mg of oral phenazopyridine hydro-chloride (Pyridium) several hours before a subsequent examination. By placing a tampon in the vagina and on the perineum, the diagnosis may be confirmed by inspection of the pads after a period of time. Further imaging (intravenous urography) and cystoscopy can identify the exact location of the aberrant communication. If diagnosis is made early, the fistulous tract may heal with prolonged catheterization. However, if this procedure is unsuccessful or if diagnosis is made late, surgical correction is generally the only hope for cure.
Urethral Diverticula
Another important but uncommon cause of involuntary urine loss is urethral diverticula. Diverticula are essentially weaknesses or “hernias” in the supportive fascial layer of the bladder or urethra. Urethral diverticulum is most likely to cause symptoms of urinary loss. It has an incidence of 0.3–3% in women and is thought to be largely an acquired condition resulting from obstruction and expansion of the paraurethral Skene’s glands. The symptoms of constant small amounts of leakage or urethral discharge are often described. A suburethral mass is visible and palpable on physical examination. Urine or discharge may often be “milked” by palpation of the suburethral mass. Treatment is usually surgical excision of the diverticulum.
FUNCTIONAL AND TRANSIENT INCONTINENCE
Incontinence may be caused by factors outside the lower urinary tract and is particularly significant in the geriatric population, because often a multitude of special circumstances affect the health of the elderly. Physical impairment, cognitive function, medication, systemic illness, and bowel function are all factors that may contribute to incontinence. Many immobile patients are incontinent because of the inability to toilet. Cognitive disturbances limit a patient’s ability to respond normally to the sensation to void. Numerous medications have effects on the bladder that may reduce capacity, inhibit bladder function, increase diuresis and bladder load, or relax the urinary sphincter. Additionally, stool impaction and constipation both have been associated with increased prevalence of urinary incontinence. Treatments should first identify the etiologic factors of the incontinence and then reduce or remove the cause.
PROGNOSIS
Urinary incontinence is fundamentally a condition that affects quality of life. The natural history of incontinence is generally that of stability or progression. Excluding transient causes, spontaneous resolution is not a feature of this condition. Most women learn coping skills (pad use, frequent toileting, timed voiding) and adaptive measures (fluid restriction or toilet mapping) to handle the detriment in quality of life. Although bothersome and even socially debilitating, the extent of treatment is dependent on patient direction. One example of true health risk with incontinence is in the instance of outlet obstruction and urinary retention with resultant overflow incontinence. Chronic urinary retention may lead to upper urinary tract dilation and ultimately lead to reduction in urinary function, therefore intervention in this setting is warranted.
Albo M, Richter HE, Brubaker L, et al. Burch colposuspension versus fascial sling to reduce urinary stress incontinence. N Engl J Med 2007;356:2143–2155. PMID: 17517855.
American Urogynecologic Society. www.augs.org. Accessed March 13, 2012.
Ashton-Miller J, DeLancey JOL. Functional anatomy of the female pelvic floor. Ann NY Acad Sci 2007;1101:266–296. PMID: 17416924.
Brubaker L, Nygaard I, Richter HE, et al. Two-year outcomes after sacrocolpopexy with and without burch to prevent stress urinary incontinence. Obstet Gynecol 2008;112:49–55. PMID: 18591307.
Burgio KL, Goode PS, Richter HE, et al. Combined behavioral and individualized drug therapy versus individualized drug therapy alone for urge urinary incontinence in women. J Urol 2010;184:598–603. PMID: 20639023.
Diokno A, Sampselle CM, Herzog AR, et al. Prevention of urinary incontinence by behavioral modification program: A randomized, controlled trial among older women in the community. J Urol2004;171:1165–1171. PMID: 14767293.
Diokno AC, Appell RA, Sand PK, et al. OPERA Study Group. Prospective, randomized, double-blind study of the efficacy and tolerability of the extended-release formulations of oxybutynin and tolterodine for overactive bladder: Results of the OPERA trial. Mayo Clin Proc 2003;78:687–695. PMID: 12934777.
Elkelini MS, Abuzgaya A, Hassouna MM. Mechanisms of sacral neuromodulation. Int Urogynecol J 2010;21(Suppl 2):S439–S446. PMID: 20972548.
Holmgren C, Nilsson S, Lanner L, Hellberg D. Long-term results with tension-free vaginal tape on mixed and stress urinary incontinence. Obstet Gynecol 2005;106:38–43. PMID: 15994615.
Holroyd-Leduc JM, Straus SE. Management of urinary incontinence in women: Scientific review. JAMA 2004;291:986–995. PMID: 14982915.
National Association for Continence. www.nafc.org. Accessed March 13, 2012.
Novara G, Galfano A, Secco S, D’Elia C, Cavalleri S, Ficarra V, Artibani W. A systematic review and meta-analysis of randomized controlled trials with antimuscarinic drugs for overactive bladder. Eur Urol2008;54:740–763. PMID: 18632201.
Ogah J, Cody JD, Rogerson L. Minimally invasive synthetic suburethral sling operations for stress urinary incontinence in women. Cochrane Database Syst Rev 2009;CD006375. PMID: 19821363.
Richter H, Albo ME, Zyczynski HM, et al. Retropubic versus transobturator midurethral slings for stress incontinence. N Engl J Med 2010;362:2066–2076. PMID: 20479459.
Rortveit G, Daltveit AK, Hannestad YS, Hunskaar S. Norwegian EPINCONT Study. Urinary incontinence after vaginal delivery or cesarean section. N Engl J Med 2003;348:900–907. PMID: 12621134.
Ulmsten U. An introduction to tension-free vaginal tape (TVT)—A new surgical procedure for treatment of female urinary incontinence. Int Urogynecol J Pelvic Floor Dysfunct 2001;12(Suppl 2):S3–S4. PMID: 11450978.
Voices for PFD. www.mypelvichealth.org. Accessed March 13, 2012.
Ward K, Hilton P; United Kingdom and Ireland Tension Free Vaginal Tape Trial Group. Prospective multicentre randomised trial of tension-free vaginal tape and colposuspension as primary treatment for stress incontinence. BMJ 2002;325:67. PMID: 12114234.
PELVIC ORGAN PROLAPSE
Pelvic organ prolapse (POP), including anterior vaginal and posterior vaginal prolapse, uterine prolapse, and enteroceles, is a common group of clinical conditions affecting women. The prevalence rates increase with age, and POP currently affects millions of women. In the United States, POP is responsible for more than 200,000 surgeries per year. The lifetime risk that a woman will undergo surgery for prolapse or urinary incontinence is 11%, with a third of surgeries representing repeat procedures. The risk of requiring a repeat procedure for POP may be as high as 29%. As our population ages, quality-of-life–altering conditions such as POP will demand more attention from our health care services. Prolapse can be asymptomatic or manifest with severe debility and associated bladder, bowel, or sexual dysfunction. The ability to screen, diagnose, and treat these entities will become increasingly important for clinicians.
Defects in the pelvic supporting structures result in a variety of clinically evident pelvic relaxation abnormalities. Pelvic support defects can be classified by their anatomic location.
Anterior Vaginal Wall Defects
ESSENTIALS OF DIAGNOSIS
![]() Anterior vaginal prolapse describes an anterior vaginal wall defect in which the bladder is associated with the prolapse. It is also known as a cystocele (Fig. 42–8).
Anterior vaginal prolapse describes an anterior vaginal wall defect in which the bladder is associated with the prolapse. It is also known as a cystocele (Fig. 42–8).
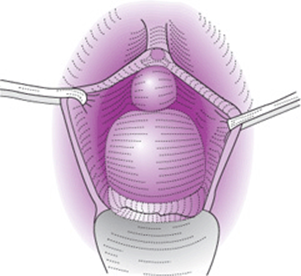
Figure 42–8. Anterior vaginal prolapse, known as a cystocele.
![]() Paravaginal/midline/transverse prolapse are terms used to indicate the location of anterior vaginal wall defects (Fig. 42–9).
Paravaginal/midline/transverse prolapse are terms used to indicate the location of anterior vaginal wall defects (Fig. 42–9).
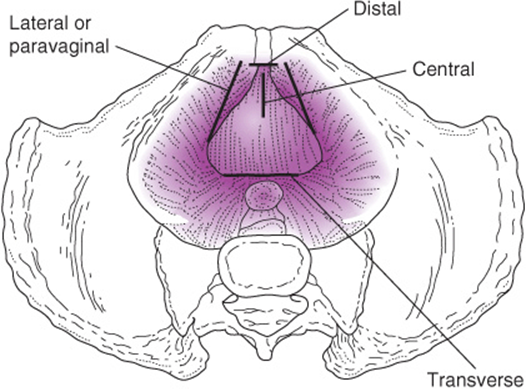
Figure 42–9. Four areas in which pubocervical fascia can break or separate—4 defects.
Apical Prolapse
ESSENTIALS OF DIAGNOSIS
![]() Uterine prolapse is shown in Figure 42–10.
Uterine prolapse is shown in Figure 42–10.

Figure 42–10. Prolapse of the uterus.
![]() Vaginal vault prolapse (posthysterectomy)
Vaginal vault prolapse (posthysterectomy)
![]() Enterocele describes an apical vaginal wall defect in which bowel is contained within the prolapsed segment (Fig. 42–11). Generally occurs in posthysterectomy women, but can occur with the uterus in situ.
Enterocele describes an apical vaginal wall defect in which bowel is contained within the prolapsed segment (Fig. 42–11). Generally occurs in posthysterectomy women, but can occur with the uterus in situ.

Figure 42–11. Enterocele and prolapsed uterus.
Posterior Vaginal Wall Prolapse
ESSENTIALS OF DIAGNOSIS
![]() Posterior vaginal wall prolapse describes a posterior vaginal wall defect. It is also known as a rectocele (Figs. 42–12 and 42–13).
Posterior vaginal wall prolapse describes a posterior vaginal wall defect. It is also known as a rectocele (Figs. 42–12 and 42–13).

Figure 42–12. Posterior vaginal prolapse.

Figure 42–13. Posterior vaginal prolapse, known as rectocele.
Description and Staging of Pelvic Organ Prolapse
Two general classifications are used to describe and document the severity of pelvic organ prolapse. The most current system employs objective measurements from fixed anatomic points. The Pelvic Organ Prolapse Quantification (POP-Q) system standardizes terminology of female pelvic organ prolapse. This is accepted as the most objective method for quantifying prolapse as it provides a more precise description of the anatomy. This descriptive system contains a series of site-specific measurements of vaginal and perineal anatomy. Prolapse in each segment is evaluated and measured relative to the hymen, which is a fixed anatomic landmark that can be consistently identified. The anatomic position of the 6 defined points for measurement should be in centimeters above the hymen (negative number) or centimeters beyond the hymen (positive number). The plane at the level of the hymen is defined as 0 (Fig. 42–14 and Table 42–10). Stages are assigned according to the most severe portion of the prolapse when the full extent of the protrusion has been demonstrated. An ordinal system is used for measurements of different points along the vaginal canal that facilitates communication among clinicians and enables objective tracking of surgical results. The POP-Q system has generally replaced the “1/2 way” system designed by Baden and Walker.

Figure 42–14. Six sites (points Aa, Ba, C, D, Bp, and Ap), genital hiatus (gh), perineal body (pb), and total vaginal length (TVL) used for pelvic organ quantitation. (Reproduced with permission from Bump RC, Mattiasson A, Bø K, et al. The standardization of terminology of female pelvic organ prolapse and pelvic floor dysfunction. Am J Obstet Gynecol 1996;175:10–17.)
Table 42–10. Staging of pelvic organ prolapse.

A better understanding of the pathophysiology of the pelvic supportive defects, their causes, and clinical presentations allows the individualization of the therapy most likely to successfully affect long-term outcome for each patient. Because POP is a disease impacting the quality of life, obtaining a detailed symptom history is an essential starting point.
General Considerations
Anterior and posterior vaginal relaxation, as well as incompetence of the perineum, often accompanies prolapse of the uterus. Large anterior vaginal prolapse is more common than posterior vaginal prolapse because the bladder is more easily carried downward than is the rectum. Before menopause, the prolapsed uterus hypertrophies and is engorged and flaccid. After the menopause, the uterus atrophies. In procidentia, the vaginal mucosa thickens and cornifies, coming to resemble skin.
ESSENTIALS OF DIAGNOSIS
The symptoms of POP are in general not unique to any particular vaginal defect. Often the symptoms are a reflection of only the most prominent point of prolapse. Most women become symptomatic only when the prolapse nears the vaginal opening. A critical concept is that the functional complaints may not always relate to the anatomic findings.
Symptoms of POP include:
![]() Sensation of vaginal fullness, pressure, heaviness, “something falling out”
Sensation of vaginal fullness, pressure, heaviness, “something falling out”
![]() Sensation of “sitting on a ball”
Sensation of “sitting on a ball”
![]() Discomfort in the vaginal area
Discomfort in the vaginal area
![]() Presence of a soft, reducible mass bulging into the vagina and distending through vaginal introitus
Presence of a soft, reducible mass bulging into the vagina and distending through vaginal introitus
![]() With straining or coughing, there is increased bulging and descent of the vaginal wall.
With straining or coughing, there is increased bulging and descent of the vaginal wall.
![]() Back pain and pelvic pain are often also associated with POP. It is important in women with these complaints to investigate other causes, as a direct link in mild to moderate prolapse is unproven.
Back pain and pelvic pain are often also associated with POP. It is important in women with these complaints to investigate other causes, as a direct link in mild to moderate prolapse is unproven.
![]() Urinary symptoms are also common:
Urinary symptoms are also common:
• Feeling of incomplete emptying of the bladder
• Stress incontinence
• Urinary frequency
• Urinary hesitancy
• Perhaps a need to push the bladder up in order to void (splinting)
• Patients with advanced prolapse may have “potential” stress urinary incontinence. A condition in which underlying urinary incontinence is masked by kinking of the urethra and causing functional continence.
Defecatory symptoms may also occur, more commonly in posterior vaginal prolapse. The sense is one of incomplete emptying, need to strain, or manually splint in the vagina or on the perineal body (space between vagina and anus) in order to defecate. The history may include prolonged, excessive use of laxatives or frequent enemas. Other nonspecific symptoms such as low back pain, dyspareunia, or even fecal and gas incontinence may be reported.
Symptoms of sexual function may also be elicited. Coital laxity or a sense of feeling “loose” may be reported. Avoiding intercourse as a consequence of embarrassment may occur. Attention to this aspect of a woman’s symptoms is especially critical if any surgical intervention is considered.
Pathogenesis
For pelvic organ prolapse, proven risk factors include age, increasing parity, obesity, and history of pelvic surgery, specifically hysterectomy. Additionally, certain lifestyle or disease conditions can promote the development of POP. Chronic coughing from lung disease and straining from chronic constipation, for example, may increase the pressures on the pelvic floor. Acting as a constant piston, driving forces exerted onto the pelvic support tissues can cause herniation of the vaginal walls. In a similar manner, occupational activity requiring repetitive heavy lifting (eg, environmental service workers or care providers of the elderly) may promote the development of POP with this daily insult of frequent pelvic pressure.
Furthermore, menopausal status, physical debilitation, and even neurologic decline can contribute to the development of POP. Yet even with a multitude of risk factors, certain women are predisposed to developing POP. As prolapse has been demonstrated in women with no identifiable risk factors, the inherent quality of a woman’s connective tissue plays a large role in the susceptibility to the development of prolapse and related conditions. Investigating the genotype, consistency, and composition of the endopelvic “fascial” tissues and the interplay of enzymatic remodeling is an area of intense interest and current research.
Parity has long been recognized as a prime risk factor for the development of POP. Not surprisingly, it is also strongly associated with anal and urinary incontinence as well. Parity is clearly associated with POP, as case-controlled studies show vaginal parity as an independent risk factor with a 3-fold increased risk for POP among parous women compared with nullipara controls. This risk increases up to 4.5-fold with more than 2 vaginal deliveries. The question of whether it is the pregnancy, the size of the baby, or the mode of delivery that plays the largest role in the development of POP is still not clear. During labor, as the vertex descends through the vagina, the physical forces on the pelvic tissues can be severe. The muscles, viscera, connective tissue, and nerves are all potentially susceptible to injury. Forces of compression and stretching combine to injure pelvic-floor nerves, leading to ischemia and neurapraxia. Myofascial fibers can be disrupted or torn because of distention of the fetal head and body. When tissues are injured, the body will repair them. Factors impairing adequate tissue repair and wound healing may also play an as yet undetermined role in the development of POP.
Clinical Findings
A. Physical Examination
Examination for pelvic organ prolapse should begin in the dorsal lithotomy position. Inspection of the vulva and perineum should focus on evaluation of vulvar architecture and the presence of pressure ulceration or erosions or other skin lesions. Epithelial skin lesions, particularly in the elderly, should be biopsied.
At first, with the patient at rest, the labia should be separated and any prolapse noted (Figs. 42–8 and 42–13). Examination of the patient with vaginal prolapse reveals a relaxed and open genital hiatus with a thin-walled, rather smooth, bulging mass. Vaginal rugae are normally present. A loss of rugation denotes disruption of the connective tissue attachment below the epithelium.
During evaluation for urinary incontinence, a stress test is performed at this initial portion of the examination. The patient should be asked to cough forcefully, and any loss of urine is noted.
For prolapse assessment, when using the POP-Q system, the genital hiatus, perineal body, and vaginal length can be recorded. (Use of a wooden PAP spatula and tape measure can be helpful.) Vaginal support can then be assessed with strain (cough or Valsalva’s maneuver), and the point of maximal protrusion should be noted in centimeters relative to the hymen and recorded. A speculum can also be used to “usher” the prolapse out during straining. This is also the most effective way to evaluate uterocervical support. In posthysterectomy patients, the cuff can often be visualized by the presence of “dimples” in the vaginal epithelium at the apex. Discriminate examination of the vaginal walls using the posterior blade of a Graves’ speculum or Sims’ retractor should then be used to evaluate the anterior and posterior walls separately, again noting the point of maximal prolapse during strain. For evaluation of the anterior wall, compress the posterior wall and have the patient strain. For evaluation of the posterior wall, elevate the anterior wall and have the patient strain. Complete examination should also include a rectovaginal palpation. In this way, one can evaluate for the presence of concurrent enterocele in addition to a rectocele. The septal defect may involve only the lower third of the posterior vaginal wall, but it often happens that the entire length of the rectovaginal septum is thinned out. The finger in the rectum confirms sacculation into the vagina. A deep pocket into the perineal body may be noted, so that on apposition of the finger in the rectum and the thumb on the outside, the perineal body seems to consist of nothing but skin and rectal wall.
Assessment of anal sphincter tone should also be performed both at rest and with squeeze contraction. The presence of perianal lesions or hemorrhoids should be noted.
If during examination the prolapse is not able to be reproduced based on symptoms, examination with the woman in the standing position should be performed. With the patient facing the seated examiner, knees slightly bent, and with strain, prolapse not demonstrable in the supine position because of poor Valsalva’s maneuver can often be confirmed in the upright position.
Assessment of the pelvic floor strength is accomplished by vaginal or rectovaginal palpation of the levator ani musculature. Within 2 to 3 cm from the hymen, the bulk of the pubococcygeus component of the levator ani muscle can be palpated. The patient should be asked to contract the muscle, and the tone, symmetry, and duration of contraction should be recorded. This portion of the examination is often a valuable time to provide feedback to the patient about the volitional ability to contract the pelvic floor muscles. If the patient’s ability to identify and contract the muscles is inadequate, the examiner may facilitate isolation of the proper muscles using verbal cues and manual feedback.
Evaluation of urinary function is also important in patients with POP. This is most germane in patients with large anterior vaginal defects. With prolapse of the anterior vagina, the bladder and urethra may herniate into the vagina. The urethra can bend and kink as it is fixed distally at the level of the pubourethral ligament. This “kinking” can alter normal voiding function in 2 fundamental ways. First, it will increase outflow resistance and impair normal emptying. After voiding, simple catheterization or ultrasonographic measure should be performed and the residual volume measured. Although not standardized, postvoid residual volumes greater than 100 mL are considered elevated and may indicate abnormal voiding and require referral for more sophisticated testing.
The second way urethral kinking can impact voiding is by masking underlying stress urinary incontinence. With increased outflow resistance, functional continence is created. Reduction of the prolapse during examination can be performed (elevation of anterior segment with a pessary, ring forceps, or speculum). The patient strains/coughs and the presence of urinary loss confirms the condition of SUI. This is termed stress incontinence on prolapse reduction and may be addressed with an anti-incontinence procedure at the same time if surgery is offered for POP.
B. Imaging Studies
In general, a complete discriminative gynecologic examination is all that is necessary to accurately assess pelvic organ prolapse. In certain cases, further diagnostic studies can be used.
Recent advances in radiologic medicine have allowed assessment of the pelvic floor with sonography and MRI. Despite newer techniques, intravenous pyelogram or computed tomography urogram still hold great value, as they are simple and safe methods to visualize the urinary tract. They can be used to evaluate the bladder and ureters. The course of the ureters can be identified preoperatively if obstruction caused by pelvic mass or scarring is suspected. These imaging tests can be used to evaluate for fistulae, congenital anomalies, or suspected damage as a result of operative injury. However, they lack sensitivity in imaging the pelvic floor and its associated defects, do not yield much information regarding vaginal support and pelvic floor musculature, and lack dynamic capabilities.
Ultrasound techniques can be an important tool to the urogynecologist. Compared with other radiologic techniques, ultrasound is noninvasive and inexpensive and does not require contrast media. Its main disadvantage is that the quality of the study depends heavily on the skill of the operator. When performed transabdominally, transvaginally, or transperineally and combined with Doppler or endoluminal transducers, the bladder, urethra, and surrounding structures can be visualized with detail.
1. Videocystourethrography (VCUG)—VCUG combines a fluoroscopic voiding cystourethrogram with simultaneous recording of intravesical, intraurethral, and intraabdominal pressure and urine flow rate. The contrast in the bladder allows dynamic evaluation of the bladder and bladder support.
2. Magnetic Resonance Imaging—MRI has evolved into an important tool for the evaluation of the pelvic floor. It is an ideal modality because its resolution of soft tissues is superior to that of other radiologic techniques. The capability to image in multiple planes is also an advantage, particularly when visualizing the complex 3-dimensional relationships of the pelvic floor. Dynamic straining can be used to demonstrate prolapse under pressure and is often useful in surgical planning. As this modality becomes less costly and techniques evolve to allow evaluation of patients in the upright position, the information provided by MRI will be invaluable in increasing our knowledge and understanding of functional pelvic support.
Differential Diagnosis
Prolapse of the vagina is generally a straightforward diagnosis. However, less common disease entities may present as bulges in the vagina. Tumors of the urethra and bladder are often more indurated and fixed than is anterior vaginal prolapse.
A large urethral diverticulum may look and feel like an anterior vaginal prolapse but usually is more focal and may be painful. With urethral diverticulum, compression may express some purulent material from the urethral meatus. Anterolateral defects can represent embryologic remnants such as a Gartner’s duct cyst.
Skene’s and Bartholin’s glands can become obstructed and enlarge to form cysts or abscesses. Rarely, hemangiomas will present as vaginal bulging, although they will often have characteristic purple discoloration on the overlying epithelium.
Soft tumors (lipoma, leiomyoma, sarcoma, myofibroblastoma) of the vagina are more fixed and are nonreducible.
Cervical tumors—as well as endometrial tumors (pedunculated myoma or endometrial polyps)—if prolapsed through a dilated cervix and presenting in the lower third of the vagina, may be confused with mild or moderate uterine prolapse. Myomas or polyps may coexist with prolapse of the uterus and cause unusual symptoms.
Despite the variety of possibilities, the history and physical findings in vaginal or uterine prolapse are so characteristic that diagnosis is usually not a challenge.
Prevention
Prevention of genital prolapse is the focus of much debate. Antepartum, intrapartum, and postpartum exercises, especially those designed to strengthen the levator and perineal muscle groups (Kegel), often help improve or maintain pelvic support. Obesity, chronic cough, straining, and traumatic deliveries must be corrected or avoided. Estrogen therapy after menopause may help to maintain the tone and vitality of pelvic musculofascial tissues; however, evidence is lacking to support its benefit to prevent or postpone the appearance of anterior vaginal prolapse and other forms of relaxation.
Treatment
Pelvic organ prolapse, except in rare situations, is a condition that impacts only the quality of life. Consequently, the extent and type of treatment should reflect and be commensurate with the degree of impact on the quality of life the patient experiences. Patient perception is also a critical component, and self-image and conceptual discomfort are relevant to any discussion of therapy. Common reasons to intervene are when function is impaired because of the prolapse. Anterior prolapse can contribute to urinary incontinence or, when severe, urinary obstruction. Bulging vaginal epithelium can come into contact with undergarments and clothing and over time develop pressure sores and erosions. A posterior vaginal defect can become so large that fecal evacuation is difficult, or the patient finds it necessary to manually reduce the posterior vaginal wall into the vagina to expedite expulsion of feces. Mobility can be impaired by a large prolapse. All of the preceding complaints are reasons to discuss surgical repair.
Chronic decubitus ulceration of the vaginal epithelium may develop in procidentia. Urinary tract infection may occur with prolapse because of anterior vaginal prolapse, and partial ureteral obstruction with hydronephrosis may occur in procidentia. Hemorrhoids result from straining to overcome constipation. Small-bowel obstruction from a deep enterocele is rare.
A. Conservative Measures
The patient with a small or moderate-sized POP requires reassurance that the pressure symptoms are not the result of a serious condition and that, in the absence of urinary retention or severe skin pressure ulceration, no serious illness will result. The natural history of POP is such that it either will stay the same or progress. There is some evidence that a small subset of patients may experience regression of the prolapse after menopause or postpartum if the prolapse is noted shortly after delivery. Reassurance and observation of prolapse should be encouraged in the absence of symptoms.
If prolapse presents in the reproductive years, surgical correction of POP is rarely indicated in women who are not family complete. If the young woman does present with significant symptoms related to POP or with a disturbing degree of urinary incontinence, then temporary medical measures may provide adequate relief until she has completed childbearing, whereupon a definitive operative procedure can be accomplished.
1. Pessary—Pessary use in selected patients may provide adequate relief of symptoms. There are a variety of available pessary types and sizes that allow for individualization of therapy (Fig. 42–15). For the most common type of POP of the anterior or apical segment, a ring pessary is usually a sensible starting point for treatment. For the patient with complicating medical factors who is a poor operative risk, the temporary use of a vaginal pessary may provide relief of symptoms until her general condition has improved.


Figure 42–15. Types of pessaries.
Prolonged use of pessaries, if improperly managed, may lead to pressure necrosis and vaginal ulceration. The vaginal pessary is a prosthesis of ancient lineage, now made of rubber, plastics, and silicone-based material, often with a metal band or spring frame. Many types have been devised, but fewer than a dozen are basically unique and specifically helpful.
Pessaries are principally used to support the uterus and vaginal walls. They are effective because they reduce vaginal prolapse and increase the tautness of the pelvic floor structures. Little or no leverage is involved. Either by placement behind the pubic bone and perineal body or by filling the vaginal vault, pessaries remain in place to hold up the prolapsing vaginal walls or uterus. In most cases, adequate support anteriorly and a reasonably good perineal body are required; otherwise, the pessary may slip from behind the symphysis and extrude from the vagina.
Pessaries are contraindicated in acute genital tract infections and in adherent retroposition of the uterus.
Several pessary types are available:
• A ring pessary with or without support provides relief of uterine prolapse or anterior vaginal prolapse.
• Gellhorn pessaries are uniquely shaped like a collar button and provide a ringlike platform for the cervix or apex. The pessary is stabilized by a stem that rests on the perineum. These pessaries are used to correct marked prolapse when the perineal body is reasonably adequate.
• The doughnut is made of soft rubber or silicone, and this type of pessary provides support for severe uterine prolapse or vault prolapse.
• The Gehrung pessary resembles 2 firm letter Us attached by crossbars. It rests in the vagina with the cervix cradled between the long arms; this arches the anterior or posterior vaginal wall and helps reduce the vaginal prolapse.
• The Hodge pessary (Smith-Hodge, or Smith and other variations) is an elongated, curved ovoid. One end is placed behind the symphysis and the other in the posterior vaginal fornix. The anterior bow is curved to avoid the urethra; the cervix rests within the larger, posterior bow. This type of pessary is used to hold the uterus in place after it has been repositioned.
• The inflatable pessary functions much like a doughnut pessary. The ball valve is moved up and down; when the ball is in the down position, air inflates the pessary; when in the up position, the air is sealed in and inflation is maintained.
• The cube is a flexible rubber cube with suction cups on each of its 6 sides that adhere to the vaginal walls. This is useful in women with severe prolapse. However, vaginal erosions are common and can be severe. Frequent monitoring initially to identify pressure ulcers is critical.
A. FITTING OF PESSARIES—Medicine is known as both an art and a science. Pessary fitting (Fig. 42–16) falls into the art category. Pessaries that are too large cause irritation and ulceration; those that are too small may not stay in place and may protrude.

Figure 42–16. Insertion of Hodge-type pessary.
In general, fitting a pessary is very much a trial-and-error endeavor. Once a type is selected based on the defects in the vaginal anatomy and on symptoms, sizing is best done with an office sizing set. This task is somewhat complicated as each pessary has its own measurement system, but familiarity with each pessary over time simplifies this task. The pessary should be lubricated and inserted with its widest dimension in the oblique diameter of the vagina to avoid painful distention at the introitus. With a finger of the opposite hand, depress the perineum to widen the introitus. Each pessary type has an optimal method for insertion.
Once a pessary is in place, the forefinger should pass easily between the sides of the frame and the vaginal wall at any point; if it cannot, the pessary is too large. After the pessary has been fitted, the patient should be asked to stand, walk, and squat to determine whether pain occurs or whether the pessary becomes displaced. The patient should be shown how to withdraw the pessary if it becomes displaced or is uncomfortable and cautioned that a contraceptive vaginal diaphragm cannot be used while a vaginal pessary is in place.
During the initial period of pessary wear, any discomfort, bleeding, or disturbance in defecation or urinary function should be reported immediately. The patient should be examined 1–2 weeks after insertion to inspect for the presence of pressure and inflammatory or allergic reactions. A repeat exam in 4 weeks can be done; then visits should be done at 3- to 6-month intervals to assess for continued proper fit and to evaluate for vaginal erosion and inflammation as a result of pessary use. For women who are unable to remove and clean the pessary themselves, the pessary should be changed approximately every 2–3 months.
The pessary should be maintained with an acidic pH gel such as Trimo-San (Milex Products, Chicago, IL). In post-menopausal patients, topical estrogen can vitalize the vaginal mucosa and reduce ulceration. An estrogen-containing ring can also be used in conjunction by “piggybacking” the ring with the pessary and then changing it every 3 months.
Vaginal pessaries are not curative of prolapse, but they may be used for months or years for palliation with proper supervision.
A neglected pessary may cause fistulas or promote genital infections, but there is no clear evidence that cancer occurs as a result of wearing a modern pessary.
2. Pelvic Floor Muscle Exercises—In some patients, improvement of pressure symptoms and of urinary control may be obtained by using pelvic floor muscle exercises, also referred to as Kegel exercises. These exercises are aimed to tighten and strengthen the pubococcygeus muscles. Evidence strongly supports use of Kegel exercises as first-line management in the treatment of urinary and fecal incontinence; however, they may also have some benefit in the relief of POP symptoms. Kegel exercises work best after specific instruction on how to perform them as most women do not perform them either correctly or in optimal fashion without supervised instruction and feedback.
3. Estrogens—In postmenopausal women, local estrogen therapy for a number of months may improve the tone, quality, and vascularity of the musculofascial supports. It is available in cream, parvule, and ring insert forms. With counseling, local estrogen can be offered to all post-menopausal women to reduce urogenital atrophy. For postmenopausal patients with exposed prolapse, who are awaiting surgery, or using a pessary, local therapy should be recommended to promote healthy epithelium particularly in preparation for surgery.
B. Surgical Measures
1. Anterior Vaginal Prolapse
A. ANTERIOR VAGINAL COLPORRHAPHY—Anterior vaginal colporrhaphy is the most common surgical treatment for anterior vaginal prolapse (Fig. 42–17). Traditional anterior colporrhaphy (anterior repair) is a vaginal approach that involves dissecting the vaginal epithelium from the underlying fibro-muscular connective tissue and bladder, and then plicating the vaginal muscularis across the midline. Excess vaginal epithelium may be excised and the wound closed. Recurrence of anterior prolapse as high as 52% has been reported and has always been a limitation of all reparative procedures. Modifications involving permanent suture material and graft materials have been introduced in the hope of increasing durability.
Figure 42–17. Repair of anterior vaginal prolapse.
B. PARAVAGINAL REPAIR—The etiology of the anterior vaginal prolapse has been much debated, beginning with White in 1912. The repair of defects in the anterior vaginal segment has traditionally been done by midline plication. An alternative method based on the anatomic observations by Richardson and colleagues advocates identification of the specific defect in the pubocervical fascia underlying the anterior vaginal epithelium and repairing the discrete breaks (Fig. 42–9). This relationship and a lack of correction of apical defects may help explain why no single operative repair should be universally applied to patients with anterior vaginal wall defects and why traditional repair has resulted in high recurrence rates.
Paravaginal repair is performed for anterior vaginal prolapse that is confirmed to be a result of detachment of the pubocervical fascia from its lateral attachment at the arcus tendineus fascia pelvis (white line). This defect can be unilateral or bilateral. It can be confirmed preoperatively by noting loss of the lateral sulci and lack of rugation over the epithelium along the base of the bladder and elongation to the anterior vaginal wall. Clinically, vaginal examination using a speculum reveals a preponderance of the prolapse lateralized to 1 side as the speculum is withdrawn. In addition, a ring forceps can be used by gently exerting anterior traction along the vaginal sulci. If the defect is reduced, then the defect is consistent with a paravaginal defect and can be approached with a paravaginal repair technique.
The surgery can be performed either abdominally or vaginally. Both require identification of the white line and placement of serial sutures from the medial portion of the pubocervical fascia to the lateral side-wall at the level of the white line as it runs from the ischial spine over the obturator internus muscle to the posterior and inferior aspect of the pubic bone on the ipsilateral side. Reapproximation of the detached pubocervical fascia should reduce the anterior vaginal prolapse. This procedure can be done with other reconstructive procedures in the vagina as well as surgery to alleviate incontinence. Short-term surgical studies have shown good results, but no long-term or comparative data exist for this repair.
A transabdominal approach to the paravaginal repair may be elected to correct the anterior vaginal prolapse when an abdominal approach is necessary for other pelvic conditions such as abdominal hysterectomy, adnexal surgery, or, most commonly, with sacral colpopexy for apical prolapse repair.
2. Posterior Vaginal Prolapse—The traditional repair (Fig. 42–18) involves posterior midline incision, often high, to the level of the posterior fornix. The vaginal epithelium is separated off the underlying fibromuscular layer and endopelvic fascia. This fibromuscular layer is then serially plicated across the midline. Some describe adding levator muscle plication as well. No attempt at identifying specific fascial defects is made.

Figure 42–18. Repair of rectocele.
An alternate method of posterior vaginal defect (rectocele) repair relies on the identification of discrete defects in the rectovaginal fascia (Fig. 42–19). The surgeon inserts a finger of the nondominant hand into the rectum to inspect the rectovaginal fascia for defects. The rectal wall is brought forward to distinguish the uncovered muscularis (fascial defect) from the muscularis that was covered by the smooth semitransparent rectal vaginal septum. The defects are then repaired with interrupted sutures to plicate over the rectal wall. In this manner, the isolated defects are repaired, and the functional anatomy is optimally restored. Notably absent is any effort to plicate the levator ani musculature, as this often results in a bandlike stricture over the posterior wall—a likely cause of dyspareunia. Randomized trials do not support improved outcomes using this technique.

Figure 42–19. Site-specific repair of posterior vaginal prolapse. A: Dissection below vaginal epithelium exposes defect in rectovaginal (RV) fascia. B: Reflection of detached RV fascia. C: Restoration of the continuity of RV fascia by reapproximation with delayed absorbable suture.
Perineorrhaphy is generally combined with posterior vaginal repairs. This procedure is principally aimed at restoring the perineal body and reducing the vaginal outlet (genital hiatus) to more normal caliber. Reapproximation of the superficial transverse perinei muscle and the bulbocavernosus muscle rebuilds the perineum and lengthens the distance between vaginal opening and anal verge.
A. POSTOPERATIVE FACTORS—The prognosis after vaginal repair is excellent in the absence of a subsequent pregnancy or comparable factors (eg, constipation, obesity, large pelvic tumors, bronchitis, bronchiectasis, heavy manual labor) that increase intraabdominal pressure. The recurrence of the POP is probable when a specific defect of pelvic supports has been overlooked or ignored; in such cases, subsequent progression of the overlooked site may itself lead to new symptoms or even to disruption of the previously repaired segment.
Postoperative avoidance of straining, coughing, and strenuous activity is advisable. Careful instruction about diet to avoid constipation, about intake of fluids, and about the use of stool-softening laxatives and lubricating suppositories is necessary to ensure durable integrity of the rectocele repair.
B. MESH AUGMENTATION IN VAGINAL SURGERY—Efforts to reduce prolapse recurrence rates have ushered in a dramatic increase in the use of mesh for vaginal repairs. Current evidence would only support the use of synthetic mesh to augment anterior vaginal repairs but at the expense of increased rates of complications. Clinically significant rates of vaginal erosions, painful intercourse, and pelvic pain have all been reported with the use of permanent mesh materials.
3. Apical vaginal repair—Prolapse of the vaginal apex includes:
• Uterine prolapse
• Posthysterectomy vaginal cuff prolapse
• Enterocele
All of the preceding clinical conditions indicate a failure of apical support. The procedures used to address surgical repair require knowledge of the specific support structures available to reestablish normal anatomy.
Uterine prolapse is almost always accompanied by some degree of enterocele, and, as the degree of uterine descent progresses, the size of the hernial sac increases. Similarly, posthysterectomy prolapse of the vaginal vault may be the result of poor repair and identification of cuff support structure at the time of hysterectomy or may develop as a result of an enterocele that was overlooked (not repaired). Consequently, it is critical to always address apical cuff support at the time of surgery if a hysterectomy is being performed. Less common, after hysterectomy, the enterocele is located anterior to the vaginal vault, where it may be easily confused with typical anterior vaginal prolapse.
Apical vaginal repair may be accomplished transabdominally or transvaginally. A review of vaginal operations includes, among others, sacrospinous ligament suspension, iliococcygeal fixation, and high uterosacral ligament suspension (high McCall culdoplasty).
Because the normal vaginal axis is directed some distance posteriorly (almost horizontally when the patient is in an erect position) over the levator plate, operative correction by any means, whether by the vaginal or the abdominal route, should restore a normal vaginal axis. This is accomplished by suspension of the vaginal apex far back on the uterosacral ligaments, the presacral fascia, or the sacrospinous ligaments.
A. SACROSPINOUS LIGAMENT FIXATION—A popular method of vaginal vault suspension is that of unilateral or bilateral fixation to the sacrospinous ligament. In this technique, the vaginal epithelium is separated from the rectovaginal tissues. Perforation through the rectal pillar is accomplished by directing blunt dissection toward the ischial spine through the loose areolar tissue. After an appropriate location on the sacrospinous ligament is identified (usually 2–3 cm medial to the ischial spine), one of several techniques may be used to safely pass 2 or more permanent (or delayed absorbable) ligatures through the ligament to the submucosal apex of the vagina. Tying the sutures brings the vaginal apex to that sacrospinous ligament, and a posterior colporrhaphy is then performed (as noted previously). Closing the dead space by intermittently suturing the vaginal mucosa to the underlying reconstituted rectovaginal septum may be useful.
Vaginal vault suspension to 1 or both sacrospinous ligaments has the potential of injury to the pudendal nerve or pudendal vessels and is often technically difficult. Because gluteal and posterior leg pain is a potential complication of this procedure, particularly if the branches of the sacral plexus are disturbed by suturing deep to the ligament, the procedure requires a skilled vaginal surgeon and should be undertaken only by those familiar with the technique.
B. ILIOCOCCYGEAL VAGINAL SUSPENSION—First described in 1962, this procedure uses the fascia overlying the iliococcygeal muscle. Although not nearly as commonly used as other procedures, this point of attachment allows reliable apical fixation without the need to gain peritoneal access. It is generally a safe procedure requiring a posterior vaginal incision in the midline with wide dissection of the overlying epithelium. Bilateral placement of permanent or delayed absorbable suture can be used.
C. BILATERAL UTEROSACRAL LIGAMENT SUSPENSION—The use of the uterosacral ligaments to attach the vaginal cuff has become a re-appreciated technique in apical repairs. Several modifications of the procedure have been described since its introduction in 1938. This technique, as with the other vaginal procedures, can be done at the time of vaginal hysterectomy or to correct posthysterectomy apical cuff prolapse. After entrance into the peritoneum is complete, traction on the ipsilateral posterior vaginal wall with rectal digital examination will facilitate transperitoneal identification of the uterosacral ligament. Placement of a pair of permanent sutures in a lateral-to-medial fashion, 1 at the level of the ischial spine and another placed more cephalad, can be performed bilaterally. These sutures are then brought to the ipsilateral vaginal apices. Fixation of the cuff at this level reproduces cuff placement to the normal position of the cervicovaginal junction. Anterior vaginal repair should be performed before tying down the vaginal cuff.
A risk of this procedure is medial displacement and kinking of the ureters, which has been reported to occur in up to 11% of patients undergoing this procedure. Cystoscopic assessment of ureteral function without and with tension on the fixation sutures, before tying down the vaginal apices, is critical to identify any potential compromise intraoperatively. If ureteral flow reduction is identified, then removal of the sutures on the affected side will often restore normal function.
D. ABDOMINAL SACROCOLPOPEXY—Vaginal vault suspension can also be performed abdominally by attaching the vaginal cuff to the sacral promontory. Abdominal sacrocolpopexy is an excellent primary procedure for apical vaginal prolapse and enterocele and is the procedure of choice for those who are already having an abdominal approach for hysterectomy or for another indication. In this procedure, a laparotomy is performed, and the cul-de-sac and peritoneum overlying the sacrum are visualized. A window in the peritoneum over the sacral promontory is created, and 2 permanent sutures are placed through the anterior longitudinal ligament, approximately at the level of S1. The vaginal cuff is then exposed by dissecting off the overlying peritoneum. Fixation of a graft over the anterior and posterior vagina is then performed fashioning a Y-shape or 2 individual strips of graft. This Y graft is then brought posteriorly along the hollow of the sacrum and affixed to the anterior longitudinal ligament sutures overriding the sacral promontory. Avoidance of undue tension is critical to prevent postoperative dyspareunia.
Dissection and suture placement over the sacrum may introduce risk of operative hemorrhage. During placement of the sacral sutures, the nearby fragile sacral veins may be lacerated. Bleeding from these veins is difficult to control if the veins retract into the bone. Use of sterile thumbtacks to occlude these veins has been an operative technique used to stem potentially life-threatening hemorrhage.
Many different graft types have been described, as well as different methods of attaching these grafts to the vagina. Biologic grafts, however, have high failure rates when placed at the apex. Synthetic grafts are effective; however, they have erosion complication rates between 5% and 10%. As graft technologies continue to evolve, identification of the optimal graft material that maximizes durability and compatibility may materialize.
Numerous studies demonstrate this colpopexy to be highly curative of apical/uterine prolapse. Most surgeons consider the sacrocolpopexy to be the gold standard for apical repair. In the largest prospective evaluation of sacrocolpopexy outcome, the success rates are more than 95%. The procedure can also be performed laparoscopically. A prospective study evaluating outcomes of this approach in more than 100 women describe no apical recurrences and no mesh complication. Another retrospective study of 188 cases resulted in a 10% erosion rate; however, 13 of the 19 erosions occurred with concomitant hysterectomy.
The addition of robotic assistance has been introduced to facilitate the technical aspects of laparoscopic repair. To date no prospective trial exists on efficacy.
E. OBLITERATIVE VAGINAL OPERATIONS (COLPOCLEISIS AND LE FORT’S OPERATION)—These are used primarily for severe uterovaginal prolapse in elderly patients and chronically ill patients who no longer desire coital function. It has the advantage of being done with either regional or local anesthesia. These procedures are highly effective and generally well tolerated. Traction produced by the obliterating scar tissue under the bladder neck and the urethra that may actually cause or aggravate stress incontinence is associated with these operations. Closing the genital hiatus may reduce the chance of recurrence and can be achieved by performing an “extended” perineorrhaphy concomitantly (Table 42–11).
Table 42–11. Follow-up and cure rate after abdominal sacral colpopexy.

Burrows LJ, Meyn LA, Walters MD, Weber AM. Pelvic symptoms in women with pelvic organ prolapse. Obstet Gynecol 2004;104(5 Pt 1):982–988. PMID: 15516388.
Fitzgerald MP, Richter HE, Siddique S, Thompson P, Zyczynski H; Ann Weber for the Pelvic Floor Disorders Network. Colpocleisis: A review. Int Urogynecol J Pelvic Floor Dysfunct 2006;17:261–271. PMID: 15983731.
Handa VL, Garrett E, Hendrix S, Gold E, Robbins J. Progression and remission of pelvic organ prolapse: A longitudinal study of menopausal women. Am J Obstet Gynecol 2004;190:27–32. PMID: 14749630.
Haylen BT, de Ridder D, Freeman RM, et al. An International Urogynecological Association (IUGA)/International Continence Society (ICS) joint report on the terminology for female pelvic floor dysfunction. NeuroUrodyn 2010;29:4–20. PMID: 19941278.
Luber KM, Boero S, Choe JY. The demographics of pelvic floor disorders: Current observations and future projections. Am J Obstet Gynecol 2001;184:1496–1501. PMID: 11408873.
Lukacz ES, Lawrence JM, Contreras R, Nager CW, Luber KM. Parity, mode of delivery, and pelvic floor disorders. Obstet Gynecol 2006;107:1253–1260. PMID: 16738149.
Maher C, Feiner B, Baessler K, Adams EJ, Hagen S, Glazener CM. Surgical management of pelvic organ prolapse in women. Cochrane Database Syst Rev 2010;CD004014. PMID: 20393938.
Morgan DM, Larson K. Uterosacral and sacrospinous ligament suspension for restoration of apical vaginal support. Clin Obstet Gynecol 2010;53:72–85. PMID: 20142645.
NIH State-of-the-Science Conference Statement on cesarean delivery on maternal request. NIH Consens Sci Statements 2006; 23:1–29. PMID: 17308552.
Nygaard IE, McCreery R, Brubaker L, et al. Abdominal sacrocolpopexy: A comprehensive review. Obstet Gynecol 2004;104:805–823. PMID: 15458906.
Nygaard I, Barber MD, Burgio KL, et al. Prevalence of symptomatic pelvic floor disorder in U.S. women. JAMA 2008;300: 1311–1316. PMID: 18799443.
The Simon Foundation. www.simonfoundation.org. Accessed March 13, 2012.
Sung VW, Hampton BS. Epidemiology of pelvic floor dysfunction. Obstet Gynecol Clin North Am 2009;36:421–443. PMID: 19932408.