Johannes Bitzer1
(1)
Department of Obstetrics and Gynecology, University Hospital Basel, Basel, Switzerland
Johannes Bitzer
Email: Johannes.Bitzer@usb.ch
7.1 Introduction
Contraceptive counseling and care are one of the mainstays of ambulatory women’s health care. Prescribing the pill seems to be a very easy and simple procedure and not worth of further mentioning. Looking closer, it becomes however evident that there are some indicators that point to the fact that there is still room for amelioration:
· A persistent rather high prevalence of unwanted pregnancies [1–6]
· Some rare but severe complications in young healthy women [7–14]
· Frequent complaints about side effects with subsequent discontinuation and risk of unwanted pregnancy [15–18]
It seems therefore justified to look at the contraceptive decision-making process from a total quality management point of view, which includes a concept of quality-related outcome objectives [19–21]. Contraceptive counseling should thus [22, 23]:
· Maximize contraceptive efficiency
· Minimize health risk
· Optimize tolerability
· Realize additional health benefits
· Avoid unnecessary costs
7.2 The Contraceptive Triangle
To reach these objectives the medical professional needs to work with what we have called the contraceptive triangle. By this we mean the interaction of three entities: method variables, patient variables, and situation variables[22–25] (Fig. 7.1).
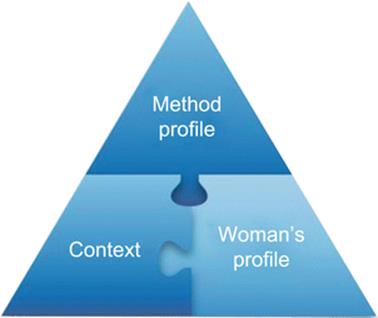
Fig. 7.1
The contraceptive triangle
The science and art in contraceptive counseling and care consists in providing a perfect fit between these three interacting groups of variables.
7.2.1 Method Variables
· Objective characteristics derived from experimental data: pharmacodynamic properties, pharmacokinetics, way of application, interactions with other drugs, and metabolizing enzymes
· Probability characteristics derived from statistical data: Pearl index (Life Table Analysis), health risks, health benefits, and side effects
7.2.2 Patient Variables
· Subjective characteristics: individual preferences, values, and family planning objectives
· Biopsychosocial profile:
· Biomedical characteristics: diseases and drugs (personal history), age, weight, blood pressure, and genetic outfit
· Psychosocial characteristics: education and professional situation
7.2.3 Situation Variables
· Phase of life
· Relationship characteristics
7.3 Contraceptive Counseling and Care as an Interactive, Structured Process
A structured process is best suited to provide the optimal interaction of these variables. This process follows basic bioethical principles [26]:
· Respect of autonomy. This means in contraceptive counseling that the objectives and values of the patient (client) are respected.
· Nonmaleficence. Do not harm means that she or he is effectively protected against unwanted pregnancies and that health risks and negative side effects are minimized.
· Beneficence. This principle means that the health and well-being of the patient is promoted and cared for.
· Justice. All patients (clients) should have the same access to contraceptive information and care.
7.3.1 Step 1: Assess the Woman’s Expectations and Preferences (Respect of Autonomy)
In the first step the professional should assess the individual woman’s expectations, objectives, and preferences. This corresponds to the ethical principle of respect of autonomy.
Women may have very different objectives and values. The following are some examples:
Objectives
· Absolute need to prevent pregnancy in the context of life situation
· Pregnancy not absolutely unwanted but not now
· Spacing pregnancies
· Family planning fulfilled, no more children
· Etc.
Values and needs
· Natural method
· Easy to use
· Independent of partner
· Involvement of partner
· Safety high priority
· Fear of health risks
· Etc.
The information obtained in this part of the interview is important for step 4a in the decision-making process.
7.3.2 Step 2: Assess the Woman’s Biopsychosocial Profile and Life Situation (Objective Patient and Situation Variables)
We differentiate between findings (objective facts), risks (probabilities), and conditions (elements of quality of life) [22–25].
7.3.2.1 Biomedical Dimension
· Findings (age, BMI, blood pressure)
· Risks (cardiovascular risks, neoplastic risks, metabolic risks, STI risks, bone risks)
· Conditions (hyperandrogenemic signs, dysmenorrhea, mastalgia, PMS, PMDD, etc.)
These data are assessed through a careful clinical history and examination. Findings and risks are important for step 4 (see below) in the structured process. Information about preexisting physical complaints and elements of quality of life are significant for step 5 (see below).
7.3.2.2 Psychological Dimension
· Findings (depression, anxiety, drug abuse)
· Risks (behavioral risks, STI risks)
· Conditions (somatoform disorder, sexual dysfunction)
The psychosocial history, the open interview, and the assessment of present complaints provide this information. Of special importance are the behavioral characteristics and preexisting emotional problems for step 4 and step 5 (see below).
7.3.2.3 Social Dimension
· Findings (socioeconomic status, education)
· Risks (cultural restrictions, dependence, migration)
· Conditions (stress, relationship issues)
Especially the sociocultural background and the present relationship characteristics are important elements of this part of the clinical interview, and they have an impact on step 4 (see below).
7.3.3 Step 3: Educate Women About Methods with Understandable Risk/Benefit Information Based on EBM Results (Method Variables) (Beneficence)
This step should empower women and help them to make informed choices. It is thus part of beneficence duty of the professional.
Women should be informed about the methods in a way that responds to two demands:
(a)
(b)
The first request is quite easily feasible; the second is much more difficult. Some general guidelines of information giving are as follows:
· Elicit preexisting knowledge and questions.
· Provide clear and simple information:
· Give information in small units.
· Encourage questions.
· Summarize and repeat.
· Elicit the understanding and the meaning given by the patient.
The issues and questions concerning the information exchange are as follows.
7.3.3.1 How Effective is Method x, y, and z to Prevent an Unwanted Pregnancy?
This question can be answered by a visual aid showing the efficiency of the methods if used by 100 women for 1 year.
The typical information to be given refers to the lowest expected percentage of women with a pregnancy during the first year of use [6, 15].
Highly effective methods:
· Combination pill 0.1 %, progestin only 0.5 %, IUD copper T380A 0.6 %, levonorgestrel IUD 0.1 %, implant 0.1 %, injectable 0.3 %, female sterilization 0.2 %, and male sterilization 0.1 %
Medium effective methods:
· Male condom 3 %, ovulation method 3 %, symptothermal 2 %, and post ovulation 1 %
Methods with low effectiveness:
· Withdrawal 4 %, spermicides 6 %, diaphragm and spermicides 6 %, calendar 9 %, and cervical cap (parous women 20 %, nulliparous 9 %)
7.3.3.2 How Much is the Effectiveness Dependent on Proper Use?
The discussion of this issue points to the discrepancy between theoretical and typical Pearl index and allows a classification with respect to the necessary compliance and discipline [6, 15].
Methods with high demand on compliance:
· Combination pill (lowest expected rate 0.1 % versus typical use rate 5–7.6 %)
· Condom (lowest expected 3 % versus typical use rate 10–14 %)
Methods with low demand on compliance:
· IUDs (lowest expected rate 0.1–0.6 % versus typical use rate 0.1–0.8 %)
· Implants (lowest expected rate 0.2 % versus typical use rate 0.2 %)
7.3.3.3 What are the Health Risks of the Method?
This is a crucial part of the information exchange process. It is important to give a risk background and to describe risks in absolute numbers or give some points of reference [15, 23, 27].
High risk: between >1 and 1 in 100
· For example: Gastrointestinal side effects of antibiotics 10 in 100; risk of pregnancy while using spermicides 6 in 100
Moderate risk: between 1 in 100 and 1 in 1,000
· For example: Death due to smoking 10 cigarettes/day: 5 in 1,000
· Having a baby with Down syndrome at age 35 years: 5 in 1,000
Low risk: between 1 in 1,000 and 1 in 10,000
· For example: Venous thromboembolism attributable to COC: 3 to 6 in 10,000
· Death due to car accident: 12 in 10,000
· Diagnosis of breast cancer under 45 years possibly attributable to current OC use: 8 in 10,000
Very low risk: between 1:10,000 and 1:100,000
· For example: Death due to soccer playing: 4 in 100,000
· Risk of death from all causes in OC users: 1 in 100,000
After the general understanding of risk, the patient should be informed about the typical risks or the methods [15, 24, 27–34].
Combined OCs:
· Venous thromboembolic disease, myocardial infarction, and stroke – very low absolute risk (1 case in 10,000 users down to 3 in 100,000 users)
· Breast cancer and cervical neoplasia – very low absolute risk
Progestogen only:
· No or very low health risk
IUDs copper:
· Perforation, PID, and ectopic pregnancy – low to very low risk
Levonorgestrel IUS:
· Perforation, PID, and ectopic pregnancy – low to very low risk
7.3.3.4 What are the Factors that Influence the Health Risks (Increase or Decrease)?
This information helps the patient to understand her personal nonmodifiable and modifiable risks. She can learn about the impact of a healthy lifestyle on her individual risk during the use of different contraceptives [15, 27, 35, 36].
· Reduce cardiovascular risks by controlling weight, exercising, stopping smoking, and lowering cholesterol.
· Reduce risk of STI, PID, and CIN by using condom and avoiding sex with “risky” partners or under risk conditions (alcohol, drugs, etc.).
7.3.3.5 What are the Possible Noncontraceptive Health Benefits of the Method?
It is important to give not only information about risks but also about possible noncontraceptive health benefits of the methods.
For example, condoms diminish considerably the risk to acquire a sexually transmitted disease (for HIV with consistent use at about 97 %, against HSV highly effective, against HPV not very effective) [37].
For example, OC benefits are less endometrial cancer, less ovarian cancer, fewer ectopic pregnancies, more regular menses (less flow, less dysmenorrheal, less anemia), less salpingitis, increased bone density, probably less endometriosis, possibly less benign breast disease, possibly less rheumatoid arthritis, possibly protection against atherosclerosis, possibly fewer fibroids, and possibly fewer ovarian cysts [15, 38].
For example, levonorgestrel IUD reduces blood loss in hypermenorrhea, possibly diminishes fibroma size, and may protect against PID [34].
7.3.3.6 What are the Most Frequent Possible Negative and Positive Side Effects Having an Impact on the Quality of Life?
It is important to comment on possible negative and positive side effects at the same time. Many of the effects observed by the patient are very individual and cannot be predicted. Mentioning possible negative effects is important to avoid unnecessary anxiety, disappointment, and insecurity.
Hormonal Contraception
For example: combined OCs
· The most important possible negative side effects which should be discussed including information about their frequency are changes in bleeding pattern (frequent), breast tension (sometimes), headache (rare), mood swings (rare), and skin changes (rare).
· The most important possible positive side effects (health benefits) are diminution of dysmenorrhea (frequent), diminution of blood loss (frequent), and amelioration of acne (frequent) [15, 24, 27, 38].
For example: progestogen implant
· The most important negative side effect is the occurrence of bleeding disorders. Positive side effects are in some women the establishment of a stable amenorrhea (if desired) and the decreased necessity of regular intake or compliance (see above).
For example: levonorgestrel intrauterine system
· The most important possible side effect is irregular bleeding during the first months; much rarer are systemic side effects like depressed mood or acne. Even more seldom are functional ovarian cysts.
· Positive possible side effects are induced hypomenorrhea and amenorrhea if desired by the woman [34].
Nonhormonal Contraception
For example: copper IUD
· The most frequent side effects are hypermenorrhea and pain with a 15 % rate of removal due to bleeding and pain [15]. There are special positive side effects.
· For example: condom and diaphragm
· The most important side effect is the possible negative impact on sexual spontaneity and sexual interaction (condom) and an increased incidence of UTI (diaphragm).
7.3.3.7 When Do They Occur, for How Long Do They Typically Last, and What Medical Significance Do They Have?
Patients should be informed about the probable duration of side effects, whether these subside after the first months and whether or not patients must expect a recurrence. This time frame gives patients more control about the treatment and increases compliance.
For example: OCs and progestogen-only contraception
· Intermenstrual bleeding and spotting occurs typically during the first months of treatment and disappears afterwards. These bleeding abnormalities do not have a pathologic significance.
7.3.4 Step 4: Shared Decision Making About Contraceptive Method
The medical professional can now enter into what is called shared decision making about the contraceptive method to be used by the individual patient. With respect to informed consent and shared decision making, we can distinguish four typical clinical situations:
1.
2.
3.
4.
Contraceptive counseling corresponds mainly to situation 2 and sometimes to situation 4. The shared decision-making process in contraception comprises two steps of exclusion of methods, one step of search for additional benefit and a final step of personal choice.
7.3.4.1 Step 4a: Exclusion of Methods Not Matching Patient’s Expectations and Preferences (Respect of Autonomy)
In this step the patient has the lead. The values, past experiences, and health beliefs expressed during step 1 of the interview exclude some methods (Fig. 7.2).
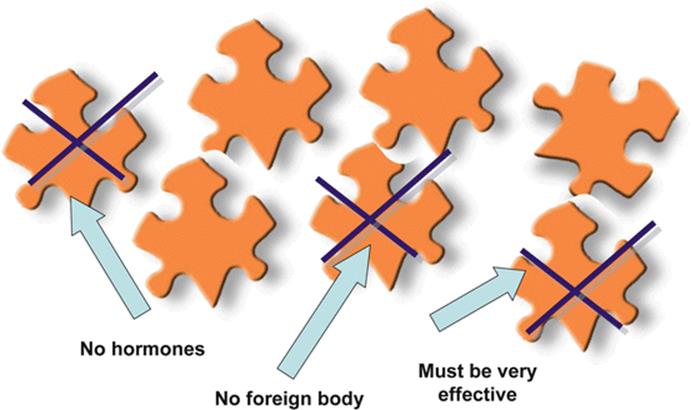
Fig. 7.2
Shared decision making about contraceptive method – women’s expectations and preferences
For example, some patients do not want to take any hormonal contraceptive. It is of course important to discuss the background of this attitude or judgment, but it is important to show an a priori respect for this position and after information exchange in case of persisting wishes of the patient to accept her exclusions.
7.3.4.2 Step 4b: Exclusion of Methods with Respect to Major Somatic and Psychosocial Health Risks (Nonmaleficence)
By combining the results of the biopsychosocial profile of the patient (step 2) with the EBM-based characteristics of the methods (step 3), the medical professional can contribute his or her knowledge and experience [28, 29, 39–44] to exclude methods which should not be used because:
A.
B.
Example A: Identified clotting abnormality of any kind; any past proven arterial or venous thrombosis excludes combined oral contraceptives.
Example B: Family history of parent or sibling <45 with thromboembolic disease with recognized precipitating factor would under usual circumstances exclude combined OCs, and personal history of high-risk sexual and addictive behavior would under usual circumstances exclude oral hormonal contraception.
Conditions which correspond to the description A are called absolute contraindications in older terminology and now are categorized by WHO as category 4 [28], meaning that the method should never be used in a patient with this diagnosis.
Condition B is much more directing towards the importance of the clinical judgment of the family planning professional. If, for example, the individual preference or value system of the patient would exclude medically safer methods (e.g., the IUD mentioned in the above example), then it is important for the clinician to negotiate with the patient and make the decision-making process transparent by visualizing the pros and cons and their respective weight [28].
7.3.4.3 Step 4c: Evaluation of Methods with Additional Health Benefits or in Case of Side Effects with the Actually Used Method Search for Alternatives (Beneficence)
After the two steps of exclusion, the information about relevant health conditions of the patient (step 2) is used to positively select methods that can contribute to an amelioration of clinical conditions and quality of life.
This is either in the case of, first, the active use of knowledge about additional health benefits (a) or, in the case of side effects with the present method, the use of knowledge about side effect specific characteristics of methods (b) [15, 34, 38, 45–49] (Fig. 7.4).
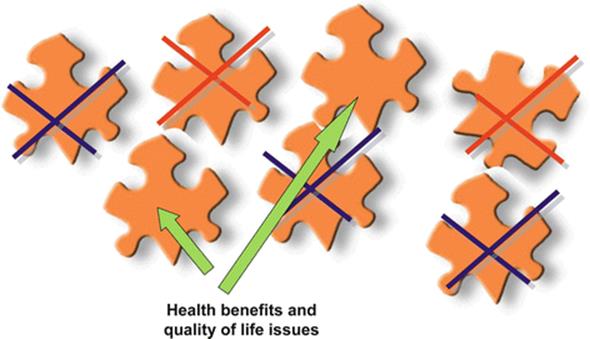
Fig. 7.4
Shared decision making – positive selection of methods with additional health benefits and QoL impact
Example A: Skin problems respond positively to OCs, but especially to combined OCs with a progestogen having antiandrogenic properties.
PMS-like symptoms seem to correspond well to a drospirenone-containing preparation.
Preexisting cycle irregularities seem to respond well to vaginal hormonal contraception.
Example B: Nausea, headache, and breast tension under combined OC would request the change to a less estrogen dominant OC or progestogen-only contraception.
Bleeding irregularities under progestogen-only or low-dose combined OCs can be resolved by changing to higher dosage of EE or the vaginal contraceptive ring.
Lack of sexual desire while using OCs could be due to the SHBG increase and androgen decrease. Changing to an OC with less SHBG increase and possibly a progestogen with androgenic properties would then be helpful.
7.3.4.4 Step 4d: Personal Choice of the Contraceptive Method
Having gone together through these steps mentioned before, the medical professional and the patient will come to a shared decision about the contraceptive method to be used.
This structured process seems in our opinion to be the best precondition to reach the optimal outcomes described above.
7.3.5 Step 5: Evaluation of the Outcome and Ongoing Adaptation of Choices
Each follow-up consultation serves the purpose to evaluate whether the abovementioned objectives have been reached or whether there is room or necessity for amelioration and change.
7.4 Summary
The objectives of contraceptive counseling are to maximize contraceptive efficiency, minimize health risk, optimize tolerability, realize additional health benefits, and avoid unnecessary costs.
To reach these outcomes, contraceptive counseling and care has to be provided in an interactive, structured way following five steps which integrate patient-centered communication to assess patient’s aims and values, biopsychosocial competence to determine the patient profile, professional patient education about methods, principles of shared decision making to choose the optimal method for the individual patient, and continuous evaluation to assess whether the objectives of contraceptive counseling and care for the individual patient have been reached.
References
1.
Westhof CF (1988) Unintended pregnancy in America and abroad. Fam Plann Perspect 20:254CrossRef
2.
Henshaw SK, Singh S, Haas T (1999) The incidence of abortion worldwide. Int Fam Plan Perspect 25(Suppl):30–38CrossRef
3.
Finer LB, Henshaw SK (2006) Disparities in rates of unintended pregnancy in the United States, 1994 and 2001. Perspect Sex Reprod Health 38:90PubMedCrossRef
4.
Alan Guttmacher Institute (2004) Contraceptive use. Facts in brief. Alan Guttmacher Institute, New York/Washington, DC
5.
Fu H, Darroch JE, Haas T, Ranjit N (1999) Contraceptive failure rates: new estimates from the 1995 National Survey of Family Growth. Fam Plann Perspect 31:56PubMedCrossRef
6.
Trussell J (2004) Contraceptive failure in the United States. Contraception 70:89PubMedCrossRef
7.
Schwartz SM, Petitti DB, Siscovick DS et al (1998) Stroke and use of low-dose oral contraceptives in young women: a pooled analysis of two US studies. Stroke 29:2277PubMedCrossRef
8.
Mant J, Painter R, Vessey M (1998) Risk of myocardial infarction, angina, and stroke in users of oral contraceptives: an updated analysis of a cohort study. Br J Obstet Gynaecol 105:890PubMedCrossRef
9.
Chasan-Taber L, Stampfer MJ (1998) Epidemiology of oral contraceptives and cardiovascular disease. Ann Intern Med 128:467PubMedCrossRef
10.
Acute myocardial infarction and combined oral contraceptives: results of an international multicentre case–control study. WHO Collaborative Study of Cardiovascular Disease and Steroid Hormone Contraception (1997) Lancet 349:1202
11.
Siritho S, Thrift AG, McNeil JJ et al (2003) Risk of ischemic stroke among users of the oral contraceptive pill: the Melbourne Risk Factor Study (MERFS) Group. Stroke 34:1575PubMedCrossRef
12.
Schwingl PJ, Ory HW, Visness CM (1999) Estimates of the risk of cardiovascular death attributable to low-dose oral contraceptives in the United States. Am J Obstet Gynecol 180:241PubMedCrossRef
13.
Baillargeon JP, McClish DK, Essah PA, Nestler JE (2005) Association between the current use of low-dose oral contraceptives and cardiovascular arterial disease: a meta-analysis. J Clin Endocrinol Metab 90:3863PubMedCrossRef
14.
Haemorrhagic stroke, overall stroke risk, and combined oral contraceptives: results of an international, multicentre, case–control study. WHO Collaborative Study of Cardiovascular Disease and Steroid Hormone Contraception (1996) Lancet 348:505
15.
Speroff L, Darney P (2005) A clinical guide for contraception, 4th edn. Williams and Wilkins, Baltimore
16.
Rosenberg MJ, Waugh MS, Meehan TE (1995) Use and misuse of oral contraceptives: risk indicators for poor pill taking and discontinuation. Contraception 51:283PubMedCrossRef
17.
Trussell J, Vaughan B (1999) Contraceptive failure, method-related discontinuation and resumption of use: results from the 1995 National Survey of Family Growth. Fam Plann Perspect 31:64PubMedCrossRef
18.
Foster DG, Parvataneni R, de Bocanegra HT et al (2006) Number of oral contraceptive pill packages dispensed, method continuation, and costs. Obstet Gynecol 108:1107PubMedCrossRef
19.
Trussell J, Leveque JA, Koenig JD et al (1995) The economic value of contraception: a comparison of 15 methods. Am J Public Health 85:494PubMedCentralPubMedCrossRef
20.
Trussell J (2007) The cost of unintended pregnancy in the United States. Contraception 75:168PubMedCrossRef
21.
Halpern V, Grimes D, Lopez L, Gallo M (2006) Strategies to improve adherence and acceptability of hormonal methods for contraception. Cochrane Database Syst Rev 1, CD004317PubMed
22.
Bitzer J (2002) Kontrazeptionsberatung bei Jugendlichen. In: Wolf AS, Esser Mittag J (eds) Kinder und Jugendgynäkologie. Schattauer Verlag, Ulm/Düsseldorf
23.
Bitzer J, Tschudin S, Frey B (2006) Tirri Kontrazeption bei Jugendlichen. Schweiz Med Forum 6:1004–1010
24.
Bitzer J (2005) Familienplanung. In: Riecher-Rössler A, Bitzer J (eds) Frauengesundheit. Ein Leitfaden für die ärztliche und psychotherapeutische Praxis. Elsevier GmbH/Urban & Fischer Verlag, München, pp 535–549
25.
Bitzer J (2005) Improving compliance with contraception old concepts and new ideas. In: Genazzani AR, Schenker J, Artini PG, Simoncini T (eds) Human reproduction. CIC Edizioni Internazionali, Rome, pp 594–598. ISBN 88-7141-694-5
26.
Beauchamp TL, Childress JF (2001) Principles of biomedical ethics, 5th edn. Oxford University Press, New York
27.
Guillebaud J (2004) Contraception today. Martin Dunitz, London
28.
World Health Organization (WHO) (2003) Improving access to quality care in family planning. In: Medical eligibility criteria for contraceptive use, 3rd edn. World Health Organization, Geneva
29.
ACOG Committee on Practice Bulletins-Gynecology (2006) ACOG practice bulletin. No. 73: use of hormonal contraception in women with coexisting medical conditions. Obstet Gynecol 107:1453CrossRef
30.
Vandenbroucke JP, Rosing J, Bloemenkamp KW et al (2001) Medical progress: oral contraceptives and the risk of venous thrombosis. N Engl J Med 344:1527PubMedCrossRef
31.
Gomes MP, Deitcher SR (2004) Risk of venous thromboembolic disease associated with hormonal contraceptives and hormone replacement therapy: a clinical review. Arch Intern Med 164:1965PubMedCrossRef
32.
van Vlijmen EF, Brouwer JL, Veeger NJ et al (2007) Oral contraceptives and the absolute risk of venous thromboembolism in women with single or multiple thrombophilic defects: results from a retrospective family cohort study. Arch Intern Med 167:282PubMedCrossRef
33.
Grimes DA (2000) Intrauterine device and upper-genital tract infection. Lancet 356:1013–1019PubMedCrossRef
34.
French R, Cowan F, Mansour D et al (2001) Hormonally impregnated intrauterine systems (IUSs), versus other forms of reversible contraceptives as effective methods of preventing pregnancy. Cochrane Database Syst Rev 2, CD001776PubMed
35.
Petitti DB (2003) Clinical practice. Combination estrogen-progestin oral contraceptives. N Engl J Med 349:1443PubMedCrossRef
36.
Cates W Jr, Stone KM (1992) Family planning, sexually transmitted diseases and contraceptive choice: a literature update–part I. Fam Plann Perspect 24:75PubMedCrossRef
37.
Davis KR, Weller SC (1999) The effectiveness of condoms in reducing heterosexual transmission of HIV. Fam Plann Perspect 31:272PubMedCrossRef
38.
Glasier A. Non-contraceptive benefits of contraception. Women Health Med 2(5):33–34
39.
Farmer RD, Lawrenson RA, Thompson CR et al (1997) Population-based study of risk of venous thromboembolism associated with various oral contraceptives. Lancet 349:83PubMedCrossRef
40.
Mohllajee AP, Curtis KM, Martins SL, Peterson HB (2006) Does use of hormonal contraceptives among women with thrombogenic mutations increase their risk of venous thromboembolism? A systematic review. Contraception 73:166PubMedCrossRef
41.
Oral-contraceptive use and the risk of breast cancer. The Cancer and Steroid Hormone Study of the Centers for Disease Control and the National Institute of Child Health and Human Development (1986) N Engl J Med 315:405
42.
Marchbanks PA, McDonald JA, Wilson HG et al (2002) Oral contraceptives and the risk of breast cancer. N Engl J Med 346:2025PubMedCrossRef
43.
Davidson NE, Helzlsouer KJ (2002) Good news about oral contraceptives. N Engl J Med 346:2078PubMedCrossRef
44.
Collaborative Group on Hormonal Factors in Breast Cancer (1996) Breast cancer and hormonal contraceptives: collaborative reanalysis of individual data on 53 297 women with breast cancer and 100 239 women without breast cancer from 54 epidemiological studies. Lancet 347:1713CrossRef
45.
Gallo MF, Grimes DA, Schulz KF (2003) Skin patch and vaginal ring versus combined oral contraceptives for contraception. Cochrane Database Syst Rev 4, CD003552
46.
Loder EW, Buse DC, Golub JR (2005) Headache as a side effect of combination estrogen-progestin oral contraceptives: a systematic review. Am J Obstet Gynecol 193:636PubMedCrossRef
47.
Rosenberg MJ, Waugh MS, Higgins JE (1996) The effect of desogestrel, gestodene and other factors on spotting and bleeding. Contraception 53:85PubMedCrossRef
48.
Forinash AB, Evans SL (2003) New hormonal contraceptives: a comprehensive review of the literature. Pharmacotherapy 23:1573PubMedCrossRef
49.
Edelman, A, Gallo, M, Jensen, J, et al. Continuous or extended cycle vs. cyclic use of combined oral contraceptives for contraception (2005) Cochrane Database Syst Rev CD004695