Gowri Dorairajan1
(1)
Obstetrics and Gynecology, JIPMER, Puducherry, India
A 24-year-old primigravida married for 1 year was referred from a village in the second stage of labour with an intrauterine foetal demise to the emergency room. There was mild tachycardia. She was not anaemic. The blood pressure was normal. A midline vertical sub-umbilical scar on the abdomen was claimed to be for an ovariotomy done during adolescence. The patient was exhausted. There was secondary inertia of the uterus. Vaginal examination confirmed full dilatation with fully rotated vertex at +2 station. Forceps delivery was uneventfully performed to deliver a 2.8 kg dead baby. Massive postpartum haemorrhage followed the delivery. There was no trauma to the lower genital tract. Since the brisk bleeding very quickly started exsanguinating the patient, she was taken up for urgent laparotomy, only to realize there was transverse rupture of a previous hysterotomy scar (Fig. 2.1).
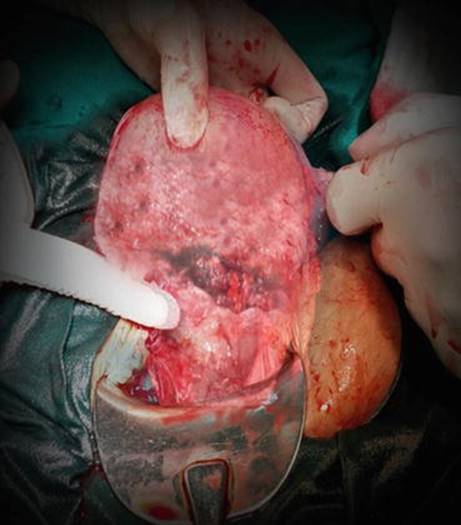
Fig. 2.1
Photograph showing the high rupture in the lower segment
The rent appeared to be rather high in the lower segment. The rent was repaired. This presentation was the first ever case I saw nearly 23 years back. In this presentation of a ruptured scarred uterus, the foetus was still intrauterine probably because the head and shoulders were deep in the pelvis. The trunk of the foetus must have acted as a tamponade and prevented exsanguination till the uterine contents were emptied vaginally. The women later confirmed hysterotomy for an unwanted pregnancy before wedlock which had been obviously hidden for social reasons.
Previous caesarean scar ruptures are the commonest cause of rupture of the uterus. It can present in various ways.
2.1 Asymptomatic Rupture
The most benign and harmless are those which are recognized as full-thickness rupture at the time of elective repeat caesarean section. Some of these caesarean sections are performed as before labour as emergency caesarean section due to suprapubic pain or unexplained tachycardia. Qureshi [35] had graded the intraoperative scars into four grades. Grades III and IV (incomplete or complete dehiscence) are occasionally observed in elective caesarean sections. Fortunately, the maternal as well as foetal outcome is unaffected in such situations.
With the rising caesarean section rate in the last two decades, one is likely to encounter pregnancies with previous caesarean sections. Scar rupture among women undergoing labour after previous caesarean sections has varied presentations.
Women with previous caesarean during labour should be carefully and diligently monitored. The decision for allowing them to labour either spontaneous or induced should be taken with prudence.
2.2 Scar Rupture During Labour
2.2.1 Scar Rupture in the First Stage of Labour
A woman was being supervised in her second pregnancy in our antenatal clinic. In her first pregnancy two and a half years back, a caesarean section had been performed for foetal distress during labour at term at a private nursing home. The baby had weighed 2.8 kg and was doing well. The caesarean operation and the postoperative recovery were uneventful. In the present pregnancy, there were no comorbidities or any complications. The foetus was in left occipito-anterior position, and the estimated birth weight was 2.7 kg. The pelvis was normal. She was willing for the trial of labour (TOLAC), and so we waited for spontaneous labour. The woman came back in spontaneous labour and was admitted in active labour. At admission, her pulse rate was 84/min with normal blood pressure. Abdomen revealed single-term foetus in left occipito-anterior position with regular good uterine contractions. The foetal heart trace was good. Vaginal examination confirmed 4 cm dilatation with full effacement and clear liquor draining. The pelvis was adequate, and the vertex was at -2 station. Two hours later she suddenly developed tachycardia with foetal bradycardia. The cervix was 6 cm and vertex was at -1 station, but the liquor was meconium stained. She was taken up for immediate caesarean section; there was full-thickness full-length scar rupture (Fig. 2.2). We delivered an asphyxiated 2.8 kg foetus from within the uterus. The rent was repaired.
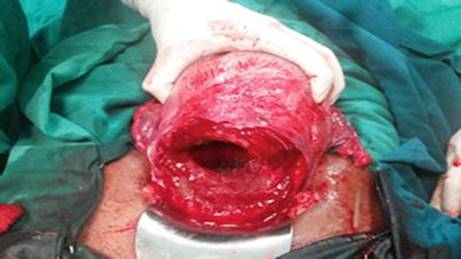
Fig. 2.2
Intraoperative photograph showing full-thickness scar rupture after delivering the foetus
The baby died in the NICU after 24 h. I felt miserable about the decision. Later I got to know that in that nursing home as a matter of routine, caesareans were performed by surgeons who would close uterus in single-layer locking sutures. Over a period, we have learnt lessons by facing the mishaps of rupture and scar dehiscence. We have identified the hospitals and the nursing homes surrounding our hospital whose caesarean scars have failed to withstand labour. We no longer post the women who underwent caesareans in these hospitals for TOLAC. It is a cause of great concern as there is an increase in the rate of repeat caesareans for fear of rupture. It is the moral and ethical duty of those who perform primary caesareans to follow the scientific techniques of repairing the incision and documenting the same properly in the discharge slips.
The commonest reason for concern suspecting the integrity of the scar is maternal tachycardia. However, there may be situations when one may take the decision to deliver by caesarean due to maternal tachycardia only to find a healthy intact previous scar. It is, therefore, important to give adequate pain relief and keep her well hydrated to prevent tachycardia due to other reasons confusing the picture.
Pain in the suprapubic region is another symptom of concern and might be one of the reasons for performing an emergency caesarean section. Cohen and colleagues [13] observed that abdominal pain alone is a poor predictor of scar rupture, but in the presence of an additional symptom or sign, it has a nearly 60 % positive predictive value for rupture.
It is also commonplace to find foetal heart rate abnormalities as the most important and only manifestation of scar dehiscence. Quite often during monitoring, one finds foetal tachycardia, variable deceleration, or bradycardia in a labouring woman with a previous caesarean scar.
In a population-based case-control study among women with previous caesarean section undergoing a trial of labour, Andersen and co-authors [5] observed that 77. 5 % of women with rupture had pathological (cardiotocography) CTG in early labour. Foetal tachycardia was significantly higher in women with scar rupture than in controls (OR = 2.5). Severe recurrent variable decelerations were also found to predict rupture.
In a large multicenter case-control study [16], the authors observed that grade 3 foetal heart abnormalities (as defined by the FIGO guidelines [17]), in a woman with a previous caesarean scar in labour, are significantly likely to be associated with scar dehiscence, with an odds ratio of 4.1. It is important to view the foetal heart rate abnormality seriously and consider termination by caesarean section instead of an overenthusiastic attempt at resuscitation and continuing the trial because though the foetal heart rate may transiently recover, it may present a little later as a florid rupture where the risk of losing the baby increases many times. In this regard, the American [1] Royal College of Obstetrics and Gynaecology [36] and the Canadian Society [44] have recommended the use of continuous electronic foetal heart rate monitoring in women undergoing a trial of labour after previous caesarean section.
The other manifestation of scar dehiscence during labour is bleeding from the vagina. Typically it may not be excessive. Sometimes it may be perceived as the excessive show. Patients with abruption may further confuse the picture. That brings me to this case. A woman presented at 34 weeks of pregnancy with acute onset bleeding. It was the first episode associated with diffuse abdominal pain. There was a loss of foetal movements. She was in her second pregnancy and had one live issue. Previous was a caesarean section done in a private hospital for foetal distress. On examination she was pale. The pulse rate was 110/min. The blood pressure was 100/60 mm of Hg. The abdomen revealed a 36-week size uterus that was tense and tender. The foetal heart sound was absent. The uterine contour was made out, and the uterus was contracting. There was haematuria. Scar rupture was a close differential diagnosis, but because of the uterus acting and relaxing, it was less likely. On pelvic examination the cervix was 2 cm; 50 % effaced, membranes were present with the vertex at -3 station. Artificial rupture of membranes confirmed blood-stained liquor. Thus, the working diagnosis was grade IIIb abruption. The labour was augmented with low-dose oxytocin, and parallel resuscitation and correction of coagulation failure were carried out. Thus, it is very important to understand that even though there may be maternal tachycardia, bleeding, foetal demise, and haematuria in a woman with previous caesarean section, other causes like abruption could be an underlying feature, and the fact that uterus is contracting and relaxing may be the only feature against scar dehiscence.
She eventually delivered, but there was a continuous trickle of bleed with the uterus having a tendency to relax. The scar appeared thinned at digital exploration. Given continued postpartum haemorrhage and suspected scar integrity, she was taken up for laparotomy. Strangely the whole lower segment was bruised (Fig. 2.3).
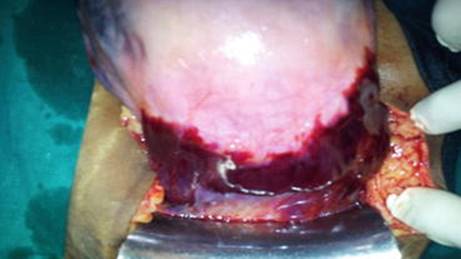
Fig. 2.3
Photograph showing bruising of the entire lower segment
There was couvelaire uterus. The utero-vesical fold was opened. The bruised lower segment was freshened and sutured. She recovered well. Whether the bruising was a manifestation of coagulation failure or impending rupture is a debatable issue.
A woman presented in labour at term. Her previous delivery was by lower segment caesarean section 3 years back for non-progress of labour in a government hospital. It was a 3 kg baby. The postoperative period was uneventful. At the time of admission, the pulse rate was 84/min. There was a single foetus in right occipito-anterior position palpable three-fifths above the brim. The expected foetal weight was 2.7 kg. The contractions were regular and moderate. Vaginal examination revealed 4 cm dilated fully effaced well-applied cervix. The clear amniotic fluid was draining, and membranes were absent. The position was confirmed as ROA and the station was -2. The pelvis was normal. She was monitored closely for the progress of labour. The foetal heart rate was well preserved with good variability and no decelerations. Two hours later the contractions seemed to become less intense and regular. The frequency reduced to once in 4 min and would last only 25 s. There was no maternal tachycardia. Foetal heart trace was good. Vaginal examination revealed a protracted dilation and descent. Labour was augmented with low-dose oxytocin drip. The contractions transiently improved in intensity but became erratic with few contractions lacking the intensity and remaining ill-sustained. Labour augmentation was continued while the woman and the foetus were carefully monitored. The foetal heart trace was well preserved; there was no maternal tachycardia or bleeding from the vagina. After 2 h vaginal examination revealed arrested dilation and descent, and so the decision for termination by caesarean section was taken. There was scar rupture of 1 cm length at the right end with the loss of all the layers and the foetal head seen through (Fig. 2.4). The foetus was delivered in good condition, and the rent was repaired. The recovery was uneventful.
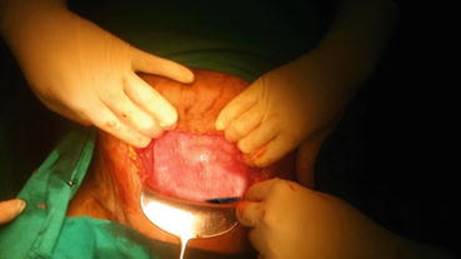
Fig. 2.4
Intraoperative photograph showing the full-thickness scar dehiscence
I wonder if there was any further prolongation, there might have been a full-blown rupture affecting the foetal outcome. It is extremely important to be vigilant about the changing patterns of uterine contractions in a woman with previous caesarean scars. In the above case, there was no maternal tachycardia or bleeding or foetal heart rate abnormities. Any incoordinate uterine contractions or tendency for secondary inertia might be the only subtle sign of early dehiscence as in this case.
Thus, diligent monitoring of various possible manifestations (Box 2.1) of a scar giving way is very important during the first stage of labour.
Box 2.1 Features of Scar Dehiscence in the First Stage of Labour
· Maternal tachycardia
· Suprapubic pain
· Vaginal bleeding
· Foetal heart rate abnormalities
· Incoordinate uterine contractions
· Secondary uterine inertia
· Haematuria
2.2.2 Scar Rupture in the Second Stage of Labour
The second stage is a particularly testing period for the strength of the scar. For this reason, some authors [22] have advocated forceps delivery to cut short the second stage of labour.
I have come across many cases where caesarean was decided for the non-descent of the head with deceleration even in the second stage to realize that there was scar dehiscence at the time of caesarean delivery.
This particular case I now narrate is just to discourage heroic decisions. A 28-year-old second gravida was supervised from the early pregnancy. She had undergone a caesarean 3 years back in our hospital for foetal distress to deliver a 3 kg baby. The records confirmed an uneventful lower segment caesarean section, and the incision had been closed in two layers. The postpartum recovery was uneventful. In the ongoing pregnancy, there were no comorbidities. She crossed her dates and would not go into labour. It was a 3.5 kg expected birth weight. The pelvis was normal and adequate. The foetus was in occipito-transverse position at -3 station. In an attempt to reduce the caesarean section rate, it was decided to induce labour as everything was conducive and the woman was willing for TOLAC. The cervix was ripened with Foleys catheter bulb. Labour was induced with a low dose of oxytocin. Active labour got established though it became slightly protracted. Labour augmentation was continued with oxytocin in spite of the protracted labour. The foetal heart was well preserved, and the liquor was clear. She achieved the second stage. There was an arrest of descent of the head beyond +1 station even after 1 h of being in the second stage. Foetal bradycardia was noticed, and she was taken up for caesarean section. There was a full-thickness scar rupture. A 3.75 kg stillborn foetus was extracted, and the uterus was repaired. It is prudent to consider elective caesarean section if the baby weight is on the higher side. As I already brought out, the other important though subtle signals we need to be watchful about are protracted labour, after she has achieved active labour especially so with a baby weight on the higher side. In a nested case-control study by Harper et al. [23], the authors observed that women who had scar rupture or failed TOLAC had protracted progress after 7 cm dilation.
2.2.3 Scar Rupture Diagnosed Postpartum
Scar rupture at the second stage may have varied presentations. I came across an extremely interesting case. A woman with previous caesarean section presented to the hospital in labour. She was found to have severe preeclampsia. The previous caesarean was done 2 years back for a nonrecurring indication. At the time of admission, she was not pale. The blood pressure was 160/ 100 mm of Hg. She was 4 cm dilated with good foetal heart sounds and a 2.5 kg expected baby weight. Artificial rupture of membranes revealed clear liquor. The labour progressed well, and she delivered a 2.6 kg baby uneventfully spontaneously after 4 h of admission. There was no postpartum haemorrhage. The blood pressure settled.
After 12–14 h of delivery, she developed mild abdominal distension and pain. The pulse rate was 100 per minute. She did not appear pale. The blood pressure was 130/80 mm of Hg. The abdomen was mildly distended and tender but not guarded. Urine was mildly high coloured, and output was around 40 ml per hour. We observed the patient and infused fluids. Sonography revealed slight free fluid in the abdomen, but all other organs were normal. There was no bowel dilatation observed. The uterus was puerperal with an empty cavity. The abdominal girth slowly increased, and she developed tachypnoea. Abdominal paracentesis revealed haemoperitoneum. At laparotomy, there was a complete full-length scar rupture. The edges had retracted, and there was slow ooze from the edges. The presentation could be as indolent as this with a good perinatal outcome.
In yet another case, I was called by the registrar from the family planning operation theatre. She was doing puerperal sterilization for a woman who had a successful, uneventful vaginal birth after caesarean section (VBAC) 2 days ago with us in the hospital. The registrar suspected something wrong because haemoperitoneum showed up even on opening the abdomen by mini-laparotomy incision. On extending the abdominal incision, a full-length complete rupture became evident; the margins had nearly stopped oozing on their own. The same was easily repaired. How lucky are a few patients and how varied is the presentation of scar rupture that follows delivery of the foetus!
As illustrated by the above two cases, the scar can rupture late in the second stage and have an indolent presentation where fortunately both mother and foetus escape serious harm. However, continued monitoring in the postoperative period would reveal slowly developing symptoms and signs. A high index of suspicion is necessary especially because the foetus is born alive and the mother does not exsanguinate herself. Sometimes I wonder how many patients like this must have escaped notice and intervention and gone home and healed the small rents on their own. It is also possible that such cases that go home with the dehiscence unnoticed after successful vaginal delivery come back after a few days with puerperal sepsis and peritonitis if infection supervened and interfered with the healing as illustrated by a case later.
That brings me to the controversy of whether to explore the scar routinely after delivery. The answer is difficult, and it should be reserved only for cases with excessive bleed or other signs suggestive of scar rupture. Perrotin and colleagues [34] recommended that the exploration of the scar should be done in symptomatic patients only. The Canadian [44] as well as RCOG [36] guidelines also recommend the same.
A defect may feel like a full-thickness defect or a breach of the mucosa and inner layers only, under an intact serosa.
I would like to narrate a few more situations I came across. A woman had a successful VBAC and delivered a healthy luscious baby spontaneously, but had postpartum haemorrhage (PPH). The brisk bleeding compelled an immediate laparotomy. There was full-thickness scar rupture. The right edge of the rupture had involved the uterine artery and hence the brisk bleed. Of course, this patient was managed by suturing the rent, and she consented for sterilization also.
In yet another case, the woman delivered uneventfully. The bleeding appeared slightly excessive. Scar was explored and a rent suspected. Full bladder abdominal and a transvaginal scan confirmed the diagnosis. There was a discontinuity of the lower segment (Fig. 2.5).
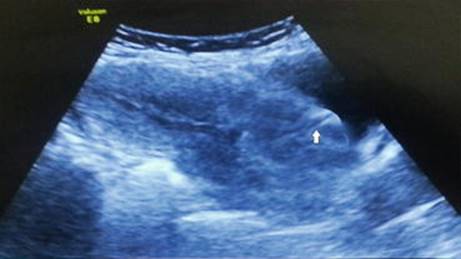
Fig. 2.5
Postpartum sonography confirms discontinuity of scar (block arrow)
Since the patient was stable and not anaemic, she was managed conservatively with antibiotics and monitoring for any deterioration. She recovered uneventfully and was discharged on the 14th day with the advice for interval sterilization and the need to avoid future pregnancies.
A woman was referred to our hospital on the 3rd postpartum day with a fever. She had a successful vaginal delivery after caesarean section at a nursing home. There was no postpartum haemorrhage. She was discharged home 48 h after delivery. At admission the pulse rate was 100/min. There were fever spikes of 101 °F. There were no other localizing symptoms or signs of fever. The abdominal examination revealed mild resistance to palpation, but there were no frank signs of peritonitis. Speculum exam revealed blood-stained minimal discharge and healthy vagina. The uterus was 18-week size. The os was closed, and there was minimal uterine tenderness. She was administered broad-spectrum antibiotics with a working diagnosis of puerperal sepsis. The ultrasound revealed a puerperal uterus and an empty uterine cavity. The uterus was surrounded by minimal free fluid. Blood culture grew Candida species, and so parenteral antifungal was administered.
The patient’s condition started deteriorating in spite of antibiotics after 48 h with fever spikes persisting and the abdominal fluid collection increasing. She developed tachypnoea and dyspnea, and the oxygen saturation started dropping and so exploratory laparotomy was carried out. There was pus in the peritoneal cavity and full-thickness scar rupture with friable and oedematous margins of the lower segment. The same was repaired. Peritoneal lavage was done, and a drain was inserted. It is possible that the scar must have given way at the second stage without affecting the perinatal outcome. The scar must have got infected due to ascending infection eventually causing peritonitis and deterioration. The woman was not immune-compromised or diabetic. The vagina would have been infected with Candidawhich was not recognized or treated before delivery resulting in such a florid infection of the ruptured scar. A similar case was reported way back in 2005 by Sun and colleagues [46]. However, it was an unscarred uterine dehiscence. She had presented 20 days after the successful vaginal delivery. The authors managed her with laparoscopic repair.
In yet another interesting presentation [32], the woman was found to have omentum protruding from the vagina in the fourth stage of labour after she had a successful VBAC.
Thus, the scar integrity is put to test throughout labour and needs careful monitoring for the various signs alerting a possible dehiscence right from the onset of labour till 48 h or so after delivery.
Box 2.2 summarizes the features that are likely to raise suspicion of scar rupture in the second stage.
Box 2.2 Features of Scar Dehiscence in the Second Stage of Labour
· Arrest of descent requiring caesarean section
· Arrest of descent requiring instrumental delivery
· Foetal heart rates abnormalities
· Foetal demise and stillbirth
· Cessation of uterine contractions
· Bleeding from the vagina in the second stage
· Significant postpartum haemorrhage noticed vaginally
· Postpartum haemoperitoneum
· Postpartum grade III or IV sepsis after VBAC
· Haematuria
· Suspicion on scar exploration
Though the American and UK College recommend a trial of labour in a woman with previous two caesarean sections, one has to be very cautious. As a policy in our country, we prefer elective caesarean delivery for a woman with previous two caesarean sections. There have been instances where a woman with previous two caesareans came late in labour and delivered uneventfully.
2.3 Rupture Early in Pregnancy in a Scarred Uterus
I would like to narrate the following case. A woman presented in her third pregnancy at 24 weeks of pregnancy in shock. The first delivery was by caesarean section for obstructed labour, and the baby died of birth asphyxia. The second was an elective caesarean section with a good perinatal outcome. In the present pregnancy, there were no other comorbidities. At admission, she was in haemorrhagic shock. The uterine contour was absent, there was haemoperitoneum, and the foetus was dead. There was haematuria. She was resuscitated, and laparotomy revealed a full-thickness scar dehiscence and bladder rent at the dome of the bladder (Fig. 2.6).

Fig. 2.6
(a) Photograph showing ragged rupture of the lower segment in the woman with previous two caesarean sections. (b) Urinary bladder rent
The same was repaired, and sterilization was carried out after obtaining informed consent. In this case, the uterus ruptured in the second trimester itself even though the scar had never ruptured in the previous pregnancies. The previous caesarean section was 3 years back. The previous documents showed that the uterus had been closed with a single locking suture due to an adherent urinary bladder that had been drawn up to the lower segment. Inability to achieve double-layer closure and possible high up scar during the previous caesarean could be a possible reason for the extremely weak scar that gave way in the second trimester.
2.4 Scar Ruptures Involving Urinary Bladder
Scar ruptures can sometimes be very complex and difficult involving the urinary bladder. Bladder involvement would substantially increase the morbidity to the mother. It is easy to recognize bladder injury before laparotomy due to the associated haematuria. The primary repair can be very demanding due to the associated friability and the tissue oedema of the bladder wall and surrounding structures.
I have encountered many such referred cases. One such case is of particular interest. She came to the emergency room. Her previous delivery was a caesarean section done in a private nursing home. She complained of vaginal bleed, loss of foetal movements, and diffused pain in the abdomen. She was at 36 weeks of pregnancy and had been in labour for 3 h before presenting to the emergency room. She was pale. There was tachycardia. The systolic blood pressure was 90 mm of Hg. Abdomen showed signs of superficial foetal parts with the loss of contour of the uterus. The foetus was dead. Bladder catheter revealed frank haematuria. The woman was resuscitated. At laparotomy, the foetus and placenta were in the abdominal cavity. After extracting out the baby and placenta, we found that the previous scar had ruptured completely, and the tear had extended downwards towards the vagina on the right side. There was a large linear rent on the posterior wall of the urinary bladder stopping 1 cm short of the ureteric orifice. We had already alerted the urologist. We sutured the lower segment incision. The edges of the downward extension of the lower segment were very oedematous and friable. It was extremely difficult to suture. The extension was closed separate of the lower segment after completely mobilizing the bladder. We used fine catgut sutures to repair as the friable edges were cutting through. The ureteric orifices were visualized. The bladder edges appeared bluish, oedematous, and friable. Layered closure of bladder rent was done with fine sutures. An omental flap was inserted in between the vagina and the bladder. A suprapubic cystotomy was done. The woman was administered broad-spectrum antibiotics. I had thought the sutures will give way, or she will develop a fistula. We continued the bladder drainage for 14 days. Fortunately, she had an uneventful recovery and healed well. Usually, one would encounter bladder involvement transversely on the posterior bladder wall or dome. In the above case, the associated colporrhexis and vertical tear on the posterior bladder wall made it particularly a demanding surgery. Yang [49] observed three cases of the ruptured uterus with bladder injury in a 6-month period. All of them had involved the posterior bladder wall and the anterior wall of the uterus.
Ho and co-authors [24] reported an interesting case that had an uneventful vaginal delivery after caesarean section. They noticed haematuria after the delivery, and it was confirmed to be rupture of the scar involving the posterior bladder wall. If bladder injury is associated with scar dehiscence or rupture, it is very important to follow the principles of tension-free layered closure of the bladder wall. Bladder involvement requires a proper mobilization of the bladder before suturing. The proximity of the tear to the ureteric orifices needs to be looked at in situations with posterior wall or base or bladder involvement. Ureteric catheterization may be necessary if the edge of the tear is very close to the ureteric orifice as there may be kinking of the ureters at the time of double-layer closure. Interposing an omental or peritoneal flap could reduce the risk of fistula formation if the suture lines of the bladder and the lower segment or vagina are very close. Bladder rest in the form of an indwelling catheter for 7 days to sometimes 14 days is necessary depending on the tissue oedema and friability. The indwelling catheter could be urethral or suprapubic depending on the site of repair and its proximity to the bladder neck.
Some important steps during the primary caesarean could help prevent bladder adhesions and the risk of injury to it if the lower segment ruptures. In this regard following a secondary analysis of cases undergoing repeat caesarean sections, the authors recommend that closure of uterus in two layers is less likely to result in bladder adhesions compared to single-layer closure [7].
2.5 Interesting Cases from Literature and Discussion
The literature has many cases of rupture reported after a caesarean section.
I enumerate a few of the cases listed in the literature below for their extremely rarity and challenging individualized management.
Zhang and colleagues [50] reported an interesting case of asymptomatic large full-thickness defect in the lower uterine segment at repeat caesarean section in a woman who had undergone uterine packing to control haemorrhage at previous caesarean section.
Ahmadi and co-authors [2] reported an extremely interesting case of scar dehiscence noticed in a patient with previous caesarean section at 24 weeks of pregnancy which was managed conservatively till 34 weeks when repeat caesarean section was performed to deliver a live baby. The woman had complained of pain in the abdomen, and the dehiscence with amniotic sac bulging was diagnosed on the scan. The foetus was doing well. As she was stable and was very keen to continue the pregnancy, the decision for conservative management was taken after discussion and counselling with the woman and her husband. The decision was collectively taken by the couple and the medical team after understanding the possible risks. One-to-one intense monitoring was carried out after admission in the unit.
A similar case has been reported by other authors [21, 33]. In the latter case, the woman was conservatively managed till 33 weeks on an outpatient basis, though the defect had been diagnosed at 17 weeks of pregnancy. It is interesting to note that in spite of an obvious full-thickness defect, the pregnancy was continued till the foetus was mature enough to be salvaged by elective caesarean resulting in a favourable outcome.
Stitely and co-authors [45] reported a woman with previous caesarean section. She was planned for surgical termination of pregnancy at 13 weeks of gestation. She developed scar dehiscence when preoperative priming of the cervix was carried out with misoprostol.
Recently Bolla and colleagues [8] reported a pregnant woman with previous caesarean section who was found to have a scar dehiscence at 8 weeks of gestation. The authors sutured the same by laparoscopy, and elective caesarean section was carried out later at 38 weeks. These above situations are rare when the scar dehiscence is not associated with maternal compromise and foetal jeopardy. Suturing and continuation till term could be a possible alternative.
There is a rise in the number of previous caesarean pregnancies. It is therefore very essential to identify the factors which might result in a weak scar that might give way during labour. Extensive research has been done to predict scar dehiscence in a woman undergoing a trial of labour.
Guise et al. [20] undertook a review of various studies that included pregnant women after one caesarean section. They shortlisted 203 studies. They found that the risk of scar rupture after a caesarean was 3 per 1000. The risk was 4.7/1000 in women planned for the trial of labour, and it was 0.26 per 1000 in elective repeat caesarean section.
Al-Zirqi et al. [4] observed a rate of rupture of a total of 2/1000 for the pre-labour caesarean section. It was the lowest (0.7 %) for women who had elective repeat caesarean section, and the rupture was found to be significantly higher at 6.7/1000 among women who had a trial of labour. They further observed that among the 80 women who had rupture after the trial of labour, five were after spontaneous vaginal delivery and five had vacuum delivery. Age above 40 years, failed trial of labour, unplanned emergency pre-labour caesarean section, induction of labour, and gestational age at or above 41 weeks were found to be factors that significantly increased the risk of rupture.
In a 1-year case-control study from the UK [18], the risk of uterine rupture was found to be 0.2 per 1000 maternities overall and 2.1 and 0.3 per 1000 maternities in women with a previous caesarean delivery planning vaginal or elective caesarean delivery, respectively. The risk increased three times in women with previous two caesarean sections and those with interpregnancy interval shorter than12 months since their last caesarean delivery. Labour induction and oxytocin use increased the risk by four times. They observed a case fatality rate of 1.3 % and 124 per 1000 perinatal mortality.
Kok et al. [26] conducted a prospective cohort analysis from Dutch perinatal registry. They studied the risk of rupture among women delivering after a previously planned caesarean section and compared it with women delivering after a previous emergency caesarean section. The risk of scar rupture during a trial of labour in a subsequent pregnancy after previously planned elective caesarean was significantly higher (OR1.6) at 0.3 % compared to 0.2 % in those who had a previous emergency caesarean. In the group who underwent elective repeat caesarean also, the incidental finding of scar dehiscence was higher (0.07 %) in those who had a previously planned caesarean compared to 0.04 % among those who had a previous emergency caesarean section.
Thus, the presence of labour before a caesarean section gives a stronger scar subsequently probably because labour helps to identify the lower segment better and gives a scope of double-layer closure with a better formed lower segment.
In a 7-year review of the cases in a large tertiary institute in India by Sinha et al. [42], the authors found the incidence of rupture to be 0.318 % in women with a history of prior uterine surgery and to be 0.02 % in women without a history of any prior uterine surgery. Among the 47 cases of rupture, 14 did not have any scar, and all these cases were in labour. 24 out of the 33 cases of rupture of scarred uterus had a low transverse caesarean scar. Among these 24 cases, labour was induced in 9, 4 were not in labour, and 11 had spontaneous labour.
One has to be very prudent in selecting the cases with previous caesarean section for allowing for vaginal delivery. Various factors affect the healing of the uterine incision like the previous indication, the interval from the last childbirth, the intraoperative details including the extension of the uterine incision, the type of uterine incision, the type of closure of the uterine incision, and the postoperative infection of the uterine incision. Certain factors in the ongoing pregnancy like the estimated weight of the foetus and the adequacy of the pelvis for the foetus also influence the decision for TOLAC.
One must follow the guidelines (RCOG revised in 2015) before planning TOLAC in a woman with previous caesarean section. The risk of dehiscence of a previous low transverse caesarean scar is 0.2–0.7 % which increases to 2–9 % if the previous scar involved the upper segment (classical/J-shaped extension). The risk increases by two- to threefold when the inter-delivery interval is 12–24 months (RCOG 2015) [36].
Veena et al. [48] carried out a retrospective study in a referral Institute of South India over a 2-year period. They observed 93 cases in the period studied. The incidence of ruptured uterus was 0.28 %. 95 % were multiparous, and more than three-fourths of the cases with rupture were in women with a previous caesarean scar. Sixty-four percent of women with a previous caesarean had interpregnancy interval of <18 months’ duration from the last caesarean section. Fifty percent of ruptures in previous caesarean cases were following the trial of labour. Seven (all without scars) out of the 93 women with rupture had undergone induction of labour.
In a recent study, Brahmalakshmy and Kushtagi [9] analysed the obstetric and non-obstetric variables among women undergoing repeat elective caesarean section and correlated the intraoperative scar grade with the various factors. They observed that primary caesarean performed before term, single-layer closure, and a shorter than 54 months interpregnancy interval were significantly associated with thinner scars. Further age more than 35 years, prolonged rupture of membranes more than 18 h and baby weight more than 3 kg at the primary caesarean, and postpartum fever were also associated with thin scar though it was not statistically significant.
Sevket et al. [40] conducted a randomized trial of women undergoing elective caesarean. They randomly assigned them to the closure of caesarean incision with either single layer or double layer. The authors demonstrated that the healing ratio and the myometrium covering the scar (the measures of uterine scar healing) at 6 months as measured by hydrosonography were significantly higher in those who underwent closure by double layer versus those by a single layer.
In a study by Micek et al. [30], previous vaginal delivery (either VBAC or otherwise) and spontaneous onset of labour were found to be the most important factor predicting successful VBAC. Similar findings were observed by other authors [28].
Based on the observations of a case (successful VBAC)-control (failed VBAC) study [6], the authors concluded that a previous history of vaginal delivery after the caesarean, spontaneous rupture of membranes with labour, and a cervix more than 3 cm at admission are associated with higher chances of successful vaginal delivery. They further observed that meconium staining of liquor, malposition, and history of stillbirth are more likely to be associated with failed VBAC. These authors did not find factors like inter-delivery interval, maternal age, the indication of the previous caesarean, and the birth weight to be significant determinants of successful VBAC.
The risk of rupture increases many times when labour is induced or augmented compared to the spontaneous onset of labour. Dekker et al.[15] conducted a population-based retrospective cohort study and observed a three- to fivefold increased risk of rupture for any induction and sixfold increased risk for prostaglandin combined with oxytocin, and the risk of rupture was highest at 14-fold for augmentation with oxytocin. The risk of rupture goes up many times when prostaglandins are used for induction compared to mechanical methods of induction. The serious perinatal outcomes increased by 41-fold in ruptures after induction of labour and by 15-fold in ruptures after spontaneous labour among women with previous caesarean scars [3]. The reason being the need for augmentation is likely when the labour has become protracted after achieving active labour which is an indirect warning about likely dehiscence or underlying disproportion.
Bujold et al. [10] conducted a case-control study with 1:3 ratio of the case (previous caesarean in labour who had rupture) with the control (previous caesarean in labour who did not rupture). They observed that the risk of rupture increases significantly with single-layer closure (OR = 2.69) and birth weight more than 3.5 kg (OR = 2.03).
In a meta-analysis of various studies, Roberge et al.[37] reviewed nine studies that included a total of 5810 women undergoing trial after previous caesarean section and concluded that previous closure with locked single-layer had five times higher risk of rupture compared to double-layer closure. The risk was not increased with non-locked single-layer closure of the previous caesarean when compared to previous double-layer closure.
However, in a retrospective comparative study, Hudic and colleagues [25] did not find any significant difference in the rate of ruptures among women with previous locked versus unlocked single-layer closure.
Scientists have looked at the scar thickness a week before delivery and have used various cutoffs to predict dehiscence.
Sharma et al. [41] studied a hundred women with or without previous caesarean and observed that the sonographic thickness of the scar was significantly less in women with previous caesarean sections.
Bujold and co-authors [11] conducted a prospective cohort study and recommended a cutoff value of 2.3 mm for scar thickness. The risk of rupture is very high if the scar is thinner than this and should preclude trial of labour.
Mohammed et al. [31] conducted a prospective controlled study on pregnant women. They had three groups: one group of patients with previous one caesarean, another group who had previous VBAC, and the third who had unscarred controls. They studied the scar thickness by the transabdominal and vaginal scan. The scar dehiscence rate was found to be 28 %, and it occurred only in the group without previous VBAC. The dehiscence was significantly more in women in whom the first caesarean section was performed for cephalopelvic disproportion and when the interpregnancy interval was short. The authors observed that a scar thinner than 2.5 mm on scan had a higher risk of rupture.
Vaginal and abdominal scans are recommended for measuring the lower uterine segment thickness. However, Laflammo et al. [27] reported two cases who had the previous caesarean performed at 29 weeks. In the subsequent pregnancy, the vaginal scan missed a scar dehiscence which was high up on the lower segment that was picked up on abdominal scan only. So it may be prudent to use both routes of the scan in patients to double check for the entire lower segment.
Uharček and colleagues [47] recommend a cutoff of 2.5 mm as the scar thickness for a sensitivity of 90.9 % and negative predictive value of 95.5 %.
Recently Mansour and colleagues [29] have recommended multiplanar view 3D scan for assessment of scar dehiscence before labour, and they found it to have nearly 100 % specificity and positive predictive value.
A few authors [39, 43] have tried to work out a prediction model for the possibility of rupture in the subsequent pregnancies based on the adjusted log likelihood ratio. An admission scoring system was designed by Flamm and Geiger [19]. Evidence-based patient decision aid in the form of a booklet has been developed recently [38].
High-Risk Factors for Intrapartum Scar Dehiscence
· Age >40 years
· Birth interval <18 months
· Previous classical caesarean
· Previous PT caesarean
· Previous caesarean for placenta previa
· Previous extension of uterine incision
· Previous uterine wound infection
· Expected birth weight more than 3.5 kg in the index pregnancy
· Scar thickness less than 2.5 mm
· Previous uterine closure with single layer locked
· Induction/augmentation with oxytocin
· Induction with prostaglandins
Randomized control trials would be the ideal way to study the risk of rupture with the type of closure, the number of layers of closure, and the sutures used at the closure.
The CAESER and CORONIS trials [12, 14] have shown that for various parameters like blunt versus sharp dissection, single-/double-layer closure, catgut versus polyglactin, and closure versus non-closure of the peritoneum, the short-term outcomes are comparable. The results for the long-term outcomes including the risk of rupture in subsequent pregnancies during labour are yet awaited.
Thus to summarize the scar rupture can occur silently without labour. It can occur anytime during labour including the second stage of labour. It may sometimes get recognized after delivery. The following table summarizes the rare or atypical presentations of scar rupture.
Rare Presentation
· Scar dehiscence on routine scan in antenatal period
· Abdominal pain anytime during pregnancy
· Shock in early pregnancy in a previous scarred uterus
· Antepartum haemorrhage
· Postpartum haemorrhage
· Postpartum peritonism and haemoperitoneum
· Puerperal sepsis grade 3 or 4
References
1.
ACOG Practice bulletin no. 115. Vaginal birth after previous cesarean delivery. Obstet Gynecol. 2010;116(2 Pt 1):450–63.
2.
Ahmadi F, Shiva S, Akhbari F. Incomplete cesarean scar rupture. J Reprod Infertil. 2013;14(1):43–5.PubMedPubMedCentral
3.
Al-Zirqi I, Stray-Pedersen B, Forsen L, et al. Uterine rupture after previous caesarean section. BJOG. 2010;117(7):809–20. doi:10.1111/j.1471-0528.2010.02533.x.Epub2010M.CrossRefPubMed
4.
Al-Zirqi I, Stray-Pedersen B, Forsén L, Daltveit AK, Vangen S. Uterine rupture: trends over 40 years. BJOG. 2015;123(5):780–7. doi:10.1111/1471-0528.13394 [Epub ahead of print].CrossRefPubMed
5.
Andersen MM, Thisted DL, Amer-Wåhlin I, Krebs L, Danish CTG, Monitoring during VBAC study group. Can intrapartum cardiotocography predict uterine rupture among women with prior caesarean delivery?: a population-based case-control study. PLoS One. 2016;11(2):e0146347. doi:10.1371/journal.pone.0146347. eCollection 2016.CrossRefPubMedPubMedCentral
6.
Birara M, Gebrehiwot Y. Factors associated with the success of vaginal birth after one caesarean section (VBAC) at three teaching hospitals in Addis Ababa, Ethiopia: a case control study. BMC Pregnancy Childb. 2013;13:31. doi:10.1186/1471-2393-13-31.CrossRef
7.
Blumenfeld YJ, Caughey AB, El-Sayed YY, Daniels K, Lyell DJ. Single- versus double-layer hysterotomy closure at primary caesarean delivery and bladder adhesions. BJOG. 2010;117(6):690–4. doi:10.1111/j.1471-0528.2010.02529.x. Epub 2010 Mar 12.CrossRefPubMed
8.
Bolla D, Raio L, Favre D, Papadia A, In-Albon S, Mueller MD. Laparoscopic ultrasound-guided repair of uterine scar isthmocele connected with the extra-amniotic space in early pregnancy. J Minim Invasive Gynecol. 2016;23(2):261–4. doi:10.1016/j.jmig.2015.09.010. Epub 2015 Sep 25.CrossRefPubMed
9.
Brahmalakshmy BL, Kushtagi P. Variables influencing the integrity of lower uterine segment in post-cesarean pregnancy. Arch Gynecol Obstet. 2015;291(4):755–62. doi:10.1007/s00404-014-3455-6. Epub 2014 Sep 11.CrossRefPubMed
10.
Bujold E, Goyet M, Marcoux S, Brassard N, Cormier B, Hamilton E, Abdous B, Sidi EA, Kinch R, Miner L, Masse A, Fortin C, Gagné GP, Fortier A, Bastien G, Sabbah R, Guimond P, Roberge S, Gauthier RJ. The role of uterine closure in the risk of uterine rupture. Obstet Gynecol. 2010;116(1):43–50. doi:10.1097/AOG.0b013e3181e41be3.CrossRefPubMed
11.
Bujold E, Jastrow N, Simoneau J, Brunet S, Gauthier RJ. Prediction of complete uterine rupture by sonographic evaluation of the lower uterine segment. Am J Obstet Gynecol. 2009;201(3):320. doi:10.1016/j.ajog.2009.06.014.e1-6.CrossRefPubMed
12.
CAESAR study collaborative group. Caesarean section surgical techniques: a randomised factorial trial (CAESAR). BJOG. 2010;117(11):1366–76. doi:10.1111/j.1471-0528.2010.02686.x.CrossRef
13.
Cohen A, Cohen Y, Laskov I, Maslovitz S, Lessing JB, Many A. Persistent abdominal pain over uterine scar during labor as a predictor of delivery complications. Int J Gynaecol Obstet. 2013;123(3):200–2. doi:10.1016/j.ijgo.2013.06.018. Epub 2013 Aug 31.CrossRefPubMed
14.
The CORONIS Collaborative Group. CORONIS – the International study of caesarean section surgical techniques: the follow-up study. BMC Pregnancy Childbirth. 2013;13:215. doi:10.1186/1471-2393-13-215. Published online 2013 Nov 21.CrossRefPubMedCentral
15.
Dekker GA, Chan A, Luke CG, Priest K, Riley M, Halliday J, King JF, Gee V, O’Neill M, Snell M, Cull V, Cornes S. Risk of uterine rupture in Australian women attempting vaginal birth after one prior caesarean section: a retrospective population-based cohort study. BJOG. 2010;117(11):1358–65. doi:10.1111/j.1471-0528.2010.02688.x. Epub 2010 Aug 17.CrossRefPubMed
16.
Desseauve D, Bonifazi-Grenouilleau M, Fritel X, Lathélize J, Sarreau M, Pierre F. Fetal heart rate abnormalities associated with uterine rupture: a case-control study: a new time-lapse approach using a standardized classification. Eur J Obstet Gynecol Reprod Biol. 2016;197:16–21. doi:10.1016/j.ejogrb.2015.10.019. Epub 2015 Dec 2.CrossRefPubMed
17.
FIGO consensus guidelines on intrapartum fetal monitoring: cardiotocography (October 2015) http://www.figo.org/news/available-view-figo-intrapartum-fetal-monitoring-guidelines-0015088. Accessed on 16 Apr 2016.
18.
Fitzpatrick KE, Kurinczuk JJ, Alfirevic Z, Spark P, Brocklehurst P, Knight M. Uterine rupture by intended mode of delivery in the UK: a national case-control study. PLoS Med. 2012;9(3):e1001184. doi:10.1371/journal.pmed.1001184. Epub 2012 Mar 13.CrossRefPubMedPubMedCentral
19.
Flamm BL, Geiger AM. Vaginal birth after cesarean delivery: an admission scoring system. Obstet Gynecol. 1997;90:907.CrossRefPubMed
20.
Guise J-M, Eden K, Emeis C, Denman MA, Marshall N, Fu R, Janik R, Nygren P, Walker M, McDonagh M. Vaginal Birth After Cesarean: New Insights. Evidence Report/Technology Assessment No.191. (Prepared by the Oregon Health & Science University Evidence-based Practice Center under Contract No. 290-2007-10057-I). AHRQ Publication No. 10-E003. Rockville, MD: Agency for Healthcare Research and Quality. March 2010.
21.
Hamar BD, Levine D, Katz NL, Lim KH. Expectant management of uterine dehiscence in the second trimester of pregnancy. Obstet Gynecol. 2003;102:1139–42.PubMed
22.
Haresh UD, Rohit KJ, Aarti AV. Prognostic factors for successful vaginal birth after cesarean section – analysis of 162 cases. J Obstet Gynaecol India. 2010;60(6):498–502.CrossRef
23.
Harper LM, Cahill AG, Roehl KA, Odibo AO, Stamilio DM, Macones GA. The pattern of labor preceding uterine rupture. Am J Obstet Gynecol. 2012;207(3):210. e1-6.CrossRefPubMedPubMedCentral
24.
Ho SY, Chang SD, Liang CC. Simultaneous uterine and urinary bladder rupture in an otherwise successful vaginal birth after cesarean delivery. J Chin Med Assoc. 2010;73(12):655–9. doi:10.1016/S1726-4901(10)70143-X.CrossRefPubMed
25.
Hudic I, Bujold E, Fatusic Z, Roberge S, Mandzic A, Fatusic J. Risk of uterine rupture following locked vs unlocked single-layer closure. Med Arch. 2012;66(6):412–4.CrossRefPubMed
26.
Kok N, Ruiter L, Hof M, Ravelli A, Mol BW, Pajkrt E, Kazemier B. Risk of maternal and neonatal complications in a subsequent pregnancy after planned caesarean section in a first birth, compared with emergency caesarean section: a nationwide comparative cohort study. BJOG. 2014;121(2):216–23. doi:10.1111/1471-0528.12483.CrossRefPubMed
27.
Laflamme SM, Jastrow N, Girard M, Paris G, Bérubé L, Bujold E. Pitfall in ultrasound evaluation of uterine scar from prior preterm cesarean section. AJP Rep. 2011;1(1):65–8. doi:10.1055/s-0031-1284222. Epub 2011 Jul 22.CrossRefPubMedPubMedCentral
28.
Madaan M, Agrawal S, Nigam A, Aggarwal R, Trivedi SS. Trial of labour after previous caesarean section: the predictive factors affecting the outcome. J Obstet Gynaecol. 2011;31(3):224–8. doi:10.3109/01443615.2010.544426.CrossRefPubMed
29.
Mansour GM, El-Mekkawi SF, Khairy HT, Mossad AE. Feasibility of prediction of cesarean section scar dehiscence in the third trimester by three-dimensional ultrasound. J Matern Fetal Neonatal Med. 2015;28(8):944–8.CrossRefPubMed
30.
Micek M, Kosinska-Kaczynska K, Godek B, Krowicka M, Szymusik I, Wielgos M. Birth after a previous cesarean section – what is most important in making a decision? Neuro Endocrinol Lett. 2014;35(8):718–23.PubMed
31.
Mohammed AB, Al-Moghazi DA, Hamdy MT, Mohammed EM. Ultrasonographic evaluation of lower uterine segment thickness in pregnant women with previous cesarean section. Middle East Fertil Soc J. 2010;15(3):188–93. doi:10.1016/j.mefs.2010.06.006.CrossRef
32.
Mwenda AS. 4th stage transvaginal omental herniation during VBAC complicated by shoulder dystocia: a unique presentation of uterine rupture. BMC Pregnancy Childbirth. 2013;13:76 (ISSN: 1471-2393).CrossRefPubMedPubMedCentral
33.
Oyelese Y, Tchabo JG, Chapin B, Nair A, Hanson P, McLaren R. Conservative management of uterine rupture diagnosed prenatally on the basis of sonography. J Ultrasound Med. 2003;22:977–80.PubMed
34.
Perrotin F, Marret H, Fignon A, Body G, Lansac J. Scarred uterus: is a routine exploration of the cesarean scar after vaginal birth always necessary? [Article in French]. J Gynecol Obstet Biol Reprod (Paris). 1999;28(3):253–62.
35.
Qureshi B, Inafuku K, Oshima K, Masamoto H, Kanazawa K. Ultrasonographic evaluation of lower uterine segment to predict the integrity and quality of cesarean scar during pregnancy: a prospective study. Tohoku J Exp Med. 1997;183(1):55–65.CrossRefPubMed
36.
RCOG guidelines Birth after Previous Caesarean Birth (Green-top Guideline No. 45) Published: 01/10/2015 https://www.rcog.org.uk/en/guidelines-research-services/guidelines/gtg45/. Accessed on 16 Apr 2016.
37.
Roberge S, Chaillet N, Boutin A, Moore L, Jastrow N, Brassard N, Gauthier RJ, Hudic I, Shipp TD, Weimar CH, Fatusic Z, Demers S, Bujold E. Single- versus double-layer closure of the hysterotomy incision during cesarean delivery and risk of uterine rupture. Int J Gynaecol Obstet. 2011;115(1):5–10. doi:10.1016/j.ijgo.2011.04.013. Epub 2011 Jul 26.CrossRefPubMed
38.
Schoorel EN, van Kuijk SM, Melman S, et al. Vaginal birth after a caesarean section: the development of a Western European population-based prediction model for deliveries at term. BJOG. 2014;121(2):194–201. doi:10.1111/1471-0528.12539; discussion 201.CrossRefPubMed
39.
Scifres CM, Rohn A, Odibo A, Stamilio D, Macones GA. Predicting significant maternal morbidity in women attempting vaginal birth after cesarean section. Am J Perinatol. 2011;28(3):181–6. doi:10.1055/s-0030-1266159. Epub 2010 Sep 14.CrossRefPubMed
40.
Sevket O, Ates S, Molla T, Ozkal F, Uysal O, Dansuk R. Hydrosonographic assessment of the effects of 2 different suturing techniques on the healing of the uterine scar after cesarean delivery. Int J Gynaecol Obstet. 2014;125(3):219–22. doi:10.1016/j.ijgo.2013.11.013. Epub 2014 Feb 28.CrossRefPubMed
41.
Sharma C, Surya M, Soni A, Soni P, Verma A, Verma S. Sonographic prediction of scar dehiscence in women with previous cesarean section. J Obstet Gynaecol India. 2015;65(2):97–103. doi:10.1007/s13224-014-0630-4. Epub 2014 Nov 4.CrossRefPubMed
42.
Sinha M, Gupta R, Gupta P, Rani R, Kaur R, Singh R. Uterine rupture: a seven year review at a tertiary care hospital in New Delhi, India. Indian J Community Med. 2016;41(1):45–9. doi:10.4103/0970-0218.170966.CrossRefPubMedPubMedCentral
43.
Smith GC, White IR, Pell JP, Dobbie R. Predicting cesarean section and uterine rupture among women attempting vaginal birth after prior cesarean section. PLoS Med. 2005;2(9):e252. Epub 2005 Sep 13.CrossRefPubMedPubMedCentral
44.
SOGC Clinical Practice Guidelines. Guidelines for vaginal birth after previous Caesarean birth. No 155 (Replaces guideline No 147), February 2005. http://sogc.org/wp-content/uploads/2013/01/155E-CPG-February2005.pdf 10.3109/14767058.2014.938626. Epub 2014 Jul 22. Accessed 16 Mar 2016.
45.
Stitely ML, Craw S, Africano E, Reid R. Uterine scar dehiscence associated with misoprostol cervical priming for surgical abortion: a case report. J Reprod Med. 2015;60(9–10):445–8.PubMed
46.
Sun CH, Liao CI, Kan YY. “Silent” rupture of the unscarred gravid uterus with subsequent pelvic abscess: successful laparoscopic management. J Minim Invasive Gynecol. 2005;12(6):519–21.CrossRefPubMed
47.
Uharček P, Brešťanský A, Ravinger J, Máňová A, Zajacová M. Sonographic assessment of lower uterine segment thickness at term in women with previous cesarean delivery. Arch Gynecol Obstet. 2015;292(3):609–12. doi:10.1007/s00404-015-3687-0. Epub 2015 Mar 27.CrossRefPubMed
48.
Veena P, Habeebullah S, Chaturvedula A. A review of 93 cases of ruptured uterus over a period of 2 years in a tertiary care hospital in South India. J Obstet Gynaecol. 2012;32(3):260–3. doi:10.3109/01443615.2011.638091.CrossRefPubMed
49.
Yang B. Bladder rupture associated with uterine rupture at delivery. Int Urogynecol J. 2011;22(5):625–7. doi:10.1007/s00192-010-1318-7. Epub 2010 Nov 25.CrossRefPubMed
50.
Zhang J, Chen SF, Luo YE. Asymptomatic spontaneous complete uterine rupture in a term pregnancy after uterine packing during previous caesarean section: a case report. Clin Exp Obstet Gynecol. 2014;41(5):597–8.PubMed