GENERAL PRINCIPLES
Transfusion of donated blood products is used to treat many disorders, including anemia and impaired hemostasis. This treatment may be life saving, but all blood products carry the risk of transfusion complications, some of which may be life threatening. The functions of blood centers and hospital blood banks include consultation in appropriate blood product use and the delivery of blood products that have undergone adequate precautions for safety.
PRETRANSFUSION SCREENING AND TESTING
Donor Screening
Donors undergo extensive screening and testing to ensure the safety of both the donors and the eventual recipients of their donated products. At the time of donation, donors must meet requirements for age, weight, vital signs, and hematologic indices, and they are asked a battery of questions to assess their risk of transfusion-transmitted diseases and other conditions that may affect donation and transfusion safety. Their blood is screened for infectious diseases (Table 10-1). Transfusion-transmitted infections are extremely rare due to effective screening, but they cannot be completely eliminated because screening cannot identify new infections during the initial, undetectable “window period.”
Compatibility Testing
Blood collection centers and blood banks provide routine laboratory testing of both donors and recipients to prevent hemolytic transfusion reactions due to ABO antibodies and other clinically relevant antibodies to RBC antigens, as well as to prevent the formation of alloantibodies against the Rh D antigen in Rh-negative recipients. This testing is performed by serology. RBCs from a patient or donor are incubated with plasma from a patient or donor or a reagent known to contain specific antibodies. If IgM antibodies (such as ABO antibodies) are present against an antigen on the RBCs, antibody binding will cause the formation of a visible cell clump due to agglutination after the sample is centrifuged. This technique is called immediate spin, and is primarily used to quickly identify ABO type, ABO antibodies, and Rh D type. IgG antibodies do not routinely cause visible agglutination, and therefore a reagent anti-human globulin (Coomb’s reagent) is added, and the sample is incubated for 30 to 60 minutes at 37°C to strengthen the reaction due to antibody crosslinking. This technique is called Coomb’s test, and is primarily used to identify common clinically relevant antibodies responsible for non-ABO hemolytic transfusion reactions, such as Rh (D, C, and E), Kell, Duffy, and Kidd. The common compatibility tests performed in the blood bank are summarized in Table 10-2.

Emergency Release
In certain critical situations, the immediate need for transfusion supersedes the need for standard and potentially time-consuming compatibility testing. In these situations, it is appropriate for clinicians to request an emergency release of blood products before compatibility testing is completed. In these cases, type O RBCs (which lack ABO antigens) and type AB plasma (which lacks ABO antibodies) are transfused until testing is complete, in order to avoid an acute hemolytic reaction due to ABO antibodies.
BLOOD PRODUCTS
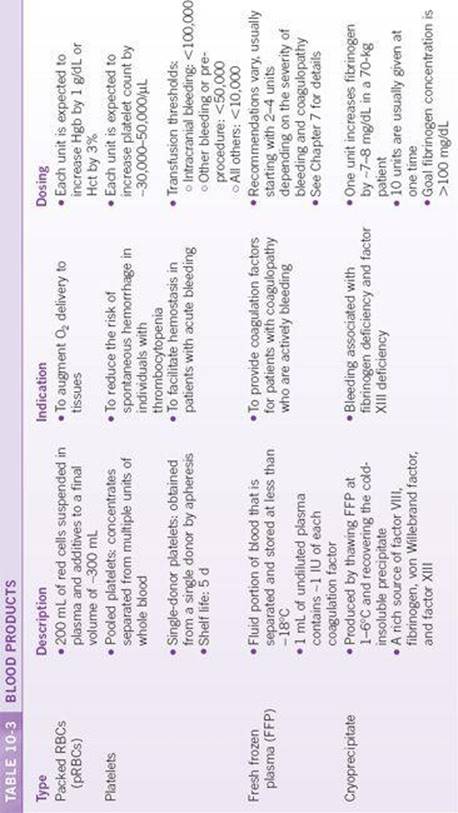
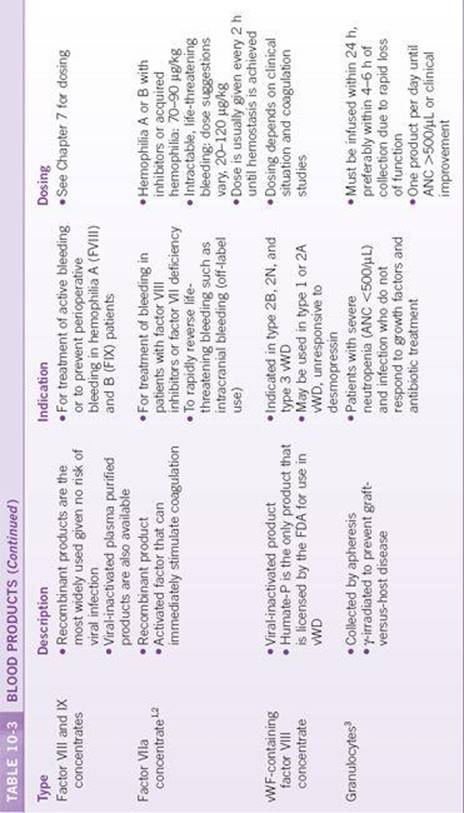
Most blood products are produced from whole blood, which is separated into components by centrifugation and manual separation. Packed RBCs, platelets, and plasma may also be collected separately by apheresis. Coagulation factor concentrates are produced by fractionation of multiple units of donated plasma or by synthesis of recombinant proteins. The most common blood products available are described in Table 10-3.
PRODUCT MODIFICATIONS
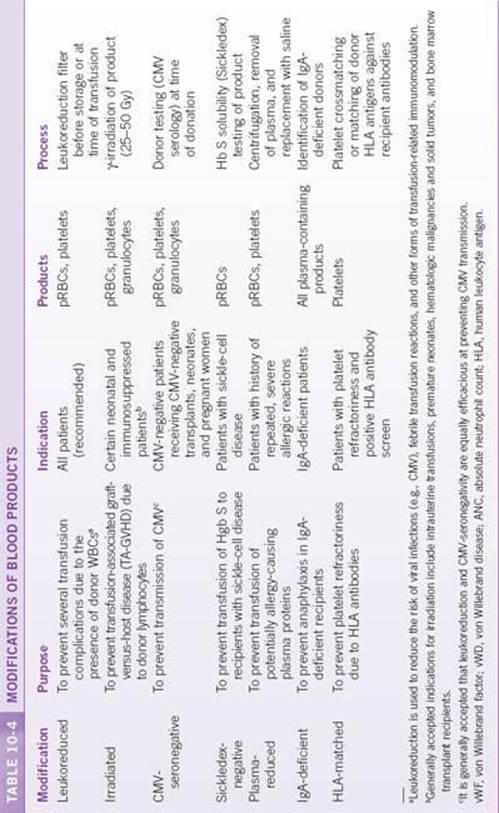
Depending on the clinical situation, modifications and specifications may be requested for blood products. The most common are described in Table 10-4.

PLATELET REFRACTORINESS
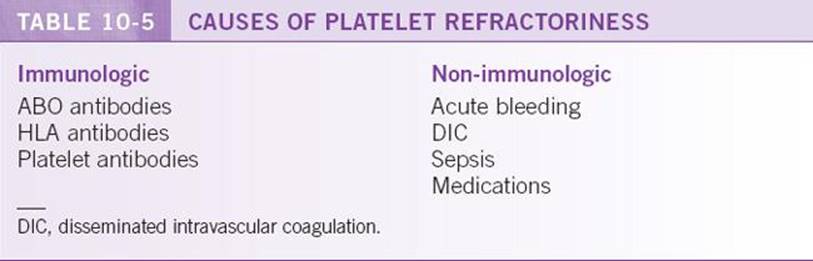
In some patients, response to platelet transfusions may be lower than expected. This condition is known as platelet refractoriness and may have multiple causes (Table 10-5). Platelet refractoriness is typically suspected if a patient’s platelet count rises by <30,000/μL 10 to 60 minutes following transfusion.
Because platelets do express some ABO and human leukocyte antigen (HLA) activity, platelet-refractory patients may respond better to ABO-matched and/or HLA-matched platelet transfusions. ABO matching is performed by serologic compatibility testing as with RBCs. It is advisable to begin with ABO matching, as the serologic techniques are easy and inexpensive to perform. If ABO matching is not successful, HLA matching is indicated; this is particularly common in patients who have received bone marrow transplants and/or multiple transfusions. HLA matching may be done by crossmatching selected units, or by obtaining an HLA antibody screen of the recipient, and selecting donors whose HLA type is compatible with the recipient’s screen.
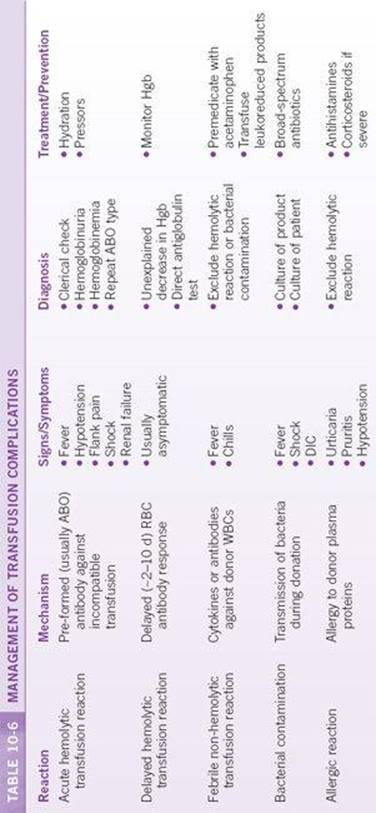
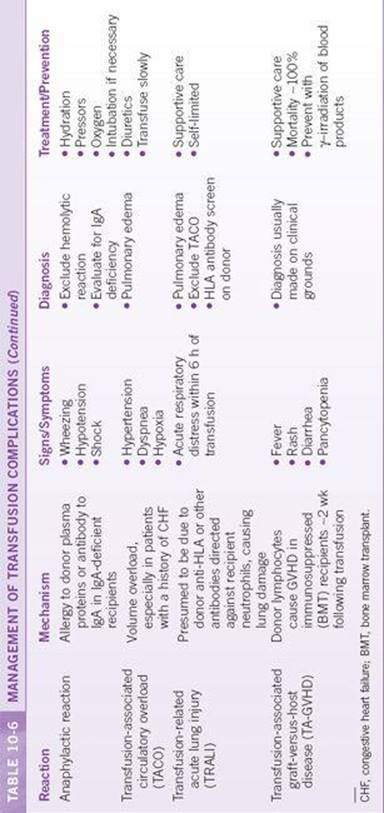
TRANSFUSION COMPLICATIONS
Every blood product carries with it the risk of transfusion complications, some of which may be severe and life-threatening. It is important to recognize the signs and symptoms of a transfusion complication and treat it appropriately. Recommendations for diagnosis and treatment are shown in Table 10-6. In all cases, if a suspected transfusion reaction occurs during transfusion, it should be stopped immediately, and appropriate measures taken.
REFERENCES
1. Rodriguez-Merchan EC, Rocino A, Ewenstein B, et al. Consensus perspectives on surgery in haemophilia patients with inhibitors: summary statement. Haemophilia. 2004;10(Suppl 2):50.
2. Freeman WD, Brott TG, Barrett KM, et al. Recombinant factor VIIa for rapid reversal of warfarin anticoagulation in acute intracranial hemorrhage. Mayo Clin Proc. 2004;79(12): 1495–1500.
3. Vamvakas EC, Pineda AA. Meta-analysis of clinical studies of the efficacy of granulocyte transfusions in the treatment of bacterial sepsis. J Clin Apher. 1996;11(1):1–9.