INTRODUCTION
Radiation oncology unifies the study of cancer with the therapeutic use of radiation. The radiation oncologist is the medical specialist who decides when and how to best use radiation. Under the supervision of the radiation oncologist, an array of nonmedical specialists, including physicists, dosimetrists, and technicians, assist in the planning and delivery of radiation to patients. This chapter introduces some of the basic principles of radiation oncology, some common treatment strategies, and an overview of the common toxicities that may be encountered in the inpatient setting.
PHYSICAL AND BIOLOGIC PRINCIPLES
![]() Ionizing radiation is energy that causes the ejection of an orbital electron. It may be either electromagnetic (photons or gamma rays) or particulate (electrons, protons, or other atomic particles). The energy of photons that can be generated in the clinic has increased over the years, which allows more doses to be delivered to internal malignancies while respecting the tolerance of the skin to radiation. Radiation dose is measured as energy per unit mass, where 1 joule (J)/kg is 1 gray (Gy). The previously used term, rad, is equal to 1 centigray (cGy).
Ionizing radiation is energy that causes the ejection of an orbital electron. It may be either electromagnetic (photons or gamma rays) or particulate (electrons, protons, or other atomic particles). The energy of photons that can be generated in the clinic has increased over the years, which allows more doses to be delivered to internal malignancies while respecting the tolerance of the skin to radiation. Radiation dose is measured as energy per unit mass, where 1 joule (J)/kg is 1 gray (Gy). The previously used term, rad, is equal to 1 centigray (cGy).
![]() Radiation causes DNA damage in both normal tissues and tumor cells. In general, cells are most susceptible in the G1, G2, and M phases of the cell cycle. Susceptible cells may enter apoptotic cell death by a variety of mechanisms or undergo necrosis. Hypoxic cells are thought to be less susceptible to radiation than well-oxygenated cells, owing to preferential free radical formation. Fractionated radiotherapy(radiation given in multiple small doses over a given period instead of a single large dose) allows normal tissue to repair sublethal damage and repopulate while the tumor cells re-sort themselves in the cell cycle and become better oxygenated. A great deal of current research involves the cell cycle–signaling pathways involved in each aspect of the damage, repair, and reoxygenation pathways.1
Radiation causes DNA damage in both normal tissues and tumor cells. In general, cells are most susceptible in the G1, G2, and M phases of the cell cycle. Susceptible cells may enter apoptotic cell death by a variety of mechanisms or undergo necrosis. Hypoxic cells are thought to be less susceptible to radiation than well-oxygenated cells, owing to preferential free radical formation. Fractionated radiotherapy(radiation given in multiple small doses over a given period instead of a single large dose) allows normal tissue to repair sublethal damage and repopulate while the tumor cells re-sort themselves in the cell cycle and become better oxygenated. A great deal of current research involves the cell cycle–signaling pathways involved in each aspect of the damage, repair, and reoxygenation pathways.1
RADIATION TREATMENT GOALS AND METHODS
![]() Radiation can be given by directing x-rays from a treatment machine to the patient (external beam radiotherapy) or by placing a radioactive source in close proximity to the patient (brachytherapy). Brachytherapy is most often used in gynecologic cancers, prostate cancer, and head and neck cancer. Sometimes both techniques are used to provide the optimal dose distribution.2
Radiation can be given by directing x-rays from a treatment machine to the patient (external beam radiotherapy) or by placing a radioactive source in close proximity to the patient (brachytherapy). Brachytherapy is most often used in gynecologic cancers, prostate cancer, and head and neck cancer. Sometimes both techniques are used to provide the optimal dose distribution.2
![]() Advances in imaging and external beam delivery now allow for ablative radiation techniques (stereotactic radiotherapy). These techniques require very precise immobilization and deliver large doses of highly focused radiation in a few (i.e., 1 to 5) fractions. Stereotactic radiotherapy works best for smaller, well-defined lesions such as limited brain/liver metastases or early-stage lung cancer.
Advances in imaging and external beam delivery now allow for ablative radiation techniques (stereotactic radiotherapy). These techniques require very precise immobilization and deliver large doses of highly focused radiation in a few (i.e., 1 to 5) fractions. Stereotactic radiotherapy works best for smaller, well-defined lesions such as limited brain/liver metastases or early-stage lung cancer.
![]() Radiation can be given for curative or palliative intent and commonly is the definitive treatment. Often, it is combined with chemotherapy and surgery in the complete cancer care of the patient. It can be given before (neoadjuvant) or after (adjuvant) the definitive therapy. Many solid tumors are treated with radiation and chemotherapy at the same time (concurrent chemoradiation).
Radiation can be given for curative or palliative intent and commonly is the definitive treatment. Often, it is combined with chemotherapy and surgery in the complete cancer care of the patient. It can be given before (neoadjuvant) or after (adjuvant) the definitive therapy. Many solid tumors are treated with radiation and chemotherapy at the same time (concurrent chemoradiation).
![]() Accurate tumor localization is essential for optimum delivery of radiation. This can be done clinically (e.g., in palliative cases and gynecologic cancers) or using radiographic studies. Clinical localizations are quick and allow the patient to start treatment immediately but do not allow for conformal delivery of radiation. To plan most radiotherapy, the patient needs to have a simulation (“sim”) in which he or she is brought to the radiation oncology department to make a treatment plan. This plan will aid in delivering the maximal dose of radiation to tumor tissue while attempting to avoid healthy tissue. Often, specialized immobilization devices are constructed for the patient during simulation to reduce intertreatment variation in patient position. After the simulation, the radiation oncologist develops a treatment plan. Because the time between the initial consultation and the first treatment is often 2 to 3 weeks, patients are best served by early radiation oncology consultation.
Accurate tumor localization is essential for optimum delivery of radiation. This can be done clinically (e.g., in palliative cases and gynecologic cancers) or using radiographic studies. Clinical localizations are quick and allow the patient to start treatment immediately but do not allow for conformal delivery of radiation. To plan most radiotherapy, the patient needs to have a simulation (“sim”) in which he or she is brought to the radiation oncology department to make a treatment plan. This plan will aid in delivering the maximal dose of radiation to tumor tissue while attempting to avoid healthy tissue. Often, specialized immobilization devices are constructed for the patient during simulation to reduce intertreatment variation in patient position. After the simulation, the radiation oncologist develops a treatment plan. Because the time between the initial consultation and the first treatment is often 2 to 3 weeks, patients are best served by early radiation oncology consultation.
![]() In general, for external-beam radiotherapy, a patient will be placed on a flat, mobile treatment table each day during the course of radiation therapy. Marks on the patient’s skin and any immobilization devices are used to obtain accurate and precise patient positioning. Each treatment may last 10 to 30 minutes. The total course of radiation therapy can vary from 1 day (prostate brachytherapy, some stereotactic treatments) to several weeks (fractionated external-beam radiotherapy). Most fractionated treatment is given once a day, 5 days per week, although some treatments are given more frequently.
In general, for external-beam radiotherapy, a patient will be placed on a flat, mobile treatment table each day during the course of radiation therapy. Marks on the patient’s skin and any immobilization devices are used to obtain accurate and precise patient positioning. Each treatment may last 10 to 30 minutes. The total course of radiation therapy can vary from 1 day (prostate brachytherapy, some stereotactic treatments) to several weeks (fractionated external-beam radiotherapy). Most fractionated treatment is given once a day, 5 days per week, although some treatments are given more frequently.
INDICATIONS FOR URGENT RADIATION THERAPY
Urgent radiation therapy is useful in certain oncologic emergencies. Spinal cord compression is the only true radiation oncology emergency and is described further in Chapter 35. It is imperative that radiation oncology and surgical services are consulted early and that an MRI of the entire spine is obtained as soon as possible. Brain metastases can be treated with radiation therapy, with timing based on symptoms and performance status. Uncontrolled bleeding of tumors (commonly breast, gynecologic, lung, colon, or bladder) often responds well to radiation therapy. Superior vena cava (SVC) syndrome,in which the SVC is compressed by tumor (typically small-cell lung cancer), can be palliated by radiation, with resolution after weeks of therapy. Although SVC syndrome by itself is rarely fatal, these tumors frequently encase other critical structures of the mediastinum.
LATE EFFECTS AND TISSUE TOLERANCE
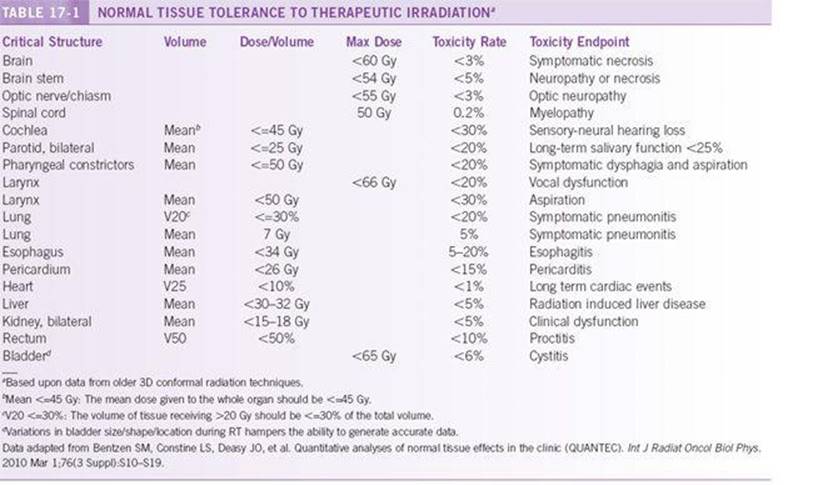
Radiation therapy balances side effects to normal tissue with the need to deliver adequate doses to the tumor. Side effects are considered to be either late (months to years after completion of radiation therapy) or acute (during radiation therapy). The radiation tolerance of normal tissue varies from patient to patient and depends on dose, fractionation scheme, and exposed tissue volume. The most common fractionation scheme is 1.8 to 2 Gy/d. Table 17-1briefly summarizes the best available human data. These data represent only general parameters, and a radiation oncologist may elect to exceed these dose levels or be more conservative based on individual patient considerations.
COMMON TREATMENT GUIDELINES AND
ASSOCIATED ACUTE EFFECTS
The following is a brief description of current “off-protocol” treatment regimens. It must be emphasized that many patients are treated according to research protocols, which vary considerably. Acute toxicities of radiation therapy typically result from direct tissue damage within the radiation path. Most radiation-alone acute effects can be managed on an outpatient basis. However, with concurrent chemoradiation, acute effects increase substantially and may require inpatient management. This is especially seen with head and neck, lung, and gastrointestinal cancers.
Palliative Therapy
Palliative treatment of brain and bone metastases is given at doses of 20 to 40 Gy over 1 to 3 weeks. This is a larger fraction size than used in most curative treatments, because there is less concern about late effects and a greater interest in minimizing treatment time for patient convenience.
Bone Metastases
Bone metastases can be treated with 6- to 8-Gy single doses, or with 30 Gy in fractions of 3 Gy/d, depending on the number and location of lesions and the patient’s life expectancy. For patients with multiple bone metastases, an infusion of radioactive strontium, samarium, or yttrium can be used to decrease pain.
Lung Cancer
Stage III non-small-cell lung cancer is often treated with definitive radiotherapy, typically combined with chemotherapy and to a dose of 60 to 70 Gy. Recently, stereotactic radiation therapy has been shown to be promising for medically inoperable stage I and II lung cancer patients. In many patients, the mediastinum is irradiated, which can result in the acute toxicity of esophagitis. The resultant odynophagia can lead to dehydration or significant weight loss, which may require inpatient management. Shortly after the completion of radiotherapy, radiation pneumonitis or, very rarely, radiation pericarditis may occur. Anti-inflammatory steroid therapy is the mainstay of treatment for both of these conditions. Three-dimensional conformal radiation therapy, involving treatment planning based on CT scans, is being investigated as a method to increase dose to tumor while avoiding normal lung. Radiation therapy is used as adjuvant treatment in select cases of locally advanced non-small-cell lung cancer treated with definitive surgery. Both chemotherapy and radiation therapy play a central role in the definitive management of limited-stage small-cell lung cancer.
Esophageal Cancer
Radiation therapy with concurrent chemotherapy is used either as definitive treatment for esophageal/gastroesophageal junction tumors or as neoadjuvant treatment. Esophagitis is the major acute toxicity.
Central Nervous System Cancer
After maximal safe surgical resection, primary brain tumors may be treated to 50 to 60 Gy, depending on the area of the brain involved. Brain metastases are normally treated to 20 to 30 Gy. Stereotactic radiosurgery, either by linear accelerator or by gamma knife, can be used for patients with few metastatic lesions that are <4 cm in size. Radiosurgery involves a single day of treatment. Mild mental deterioration is seen in children and the elderly. Neurologic changes requiring hospitalization are generally due to tumor progression, not toxicity from radiosurgery.
Head and Neck Cancer
In general, early-stage head and neck cancers are treated equally well with surgery or radiation. Advanced head and neck cancers require surgery and postoperative radiation, radiotherapy alone, or chemoradiation. Doses are typically 60 to 70 Gy. Multiple treatments per day (hyperfractionation) are often used. Side effects include xerostomia, odynophagia, dysphagia, and hoarseness. Acute toxicity often results in dehydration, which may require administration of IV fluids or placement of a gastric tube for nutritional support. Chemotherapy has been shown to improve outcome in the definitive radiotherapy of advanced head and neck cancers at the cost of 1% to 3% treatment-associated mortality and 20% to 30% risk of hospitalization for esophagitis. Treatment-associated deaths are rare with radiotherapy alone, and the frequency of hospitalization for acute toxicity is generally ≤10%.
Breast Cancer
Radiation is used in breast conservation therapy as well as postmastectomy patients with large initial tumors, positive lymph nodes, or positive resection margins. The dose to the breast is normally 54 to 60 Gy. For early-stage breast conservation therapy, partial breast radiation therapy is being investigated as a shorter-course, lower-volume alternative to whole-breast radiation. Acute side effects are usually limited to skin reactions, but pneumonitis, lymphoedema, and carditis can occur as late reactions. Hospitalization is a distinctly rare event for any acute radiotherapy effect in the breast.
Prostate Cancer
Prostate cancer can be treated with either external-beam radiation therapy or prostate brachytherapy (“seeds”). External-beam radiation consists of 70 to 80 Gy. The most common side effects are urethritis and cystitis, which are managed on an outpatient basis in the vast majority of patients. Rates of impotence and cure are similar among external-beam radiation therapy, brachytherapy, and radical prostatectomy.
Colon/Rectal Cancer
Radiation is not often used in colon cancers, except in those that are locally advanced (often fixed or perforated) and require preoperative radiation therapy. The confines of the pelvis make surgical resection of rectal cancer more challenging than that of colon cancer. Preoperative radiation therapy is used in most cases to facilitate surgical resection, for sphincter preservation, and to treat the poorly accessible presacral lymph nodes. Rectal cancer doses are 20 to 50 Gy, depending on the size of the lesion and whether radiation is given before or after surgery. Radiation therapy–induced proctitis is generally quite mild unless concurrent chemotherapy (generally 5-fluorouracil) is administered, in which case proctitis can be severe enough to cause dehydration leading to hospitalization. Patients may need nutritional support.
Anal Cancer
Most anal malignancies can be managed with definitive radiotherapy plus adjuvant chemotherapy, with surgery reserved for salvage. Although generally of squamous histology, anal cancers respond well to low doses of radiotherapy. Typical curative doses are 30 to 54 Gy, classically given with concurrent mitomycin-C and 5-fluorouracil. Acute toxicities are mainly myelosuppression caused by mitomycin-C, proctitis, cystitis, hemorrhoid exacerbation, and skin reaction in the perineum.
Pediatric Cancer
Radiation is used in many pediatric tumors. Total-body irradiation and irradiation of sanctuary sites are used in leukemia, and many lymphoma protocols involve local irradiation. Many sarcomas are treated with radiation after or instead of surgery. Most CNS tumors are treated in part with radiation. Treatment varies by site, and long-term effects on development limit dose.
Lymphoma
Lymphomas are very radioresponsive, and doses from 20 to 45 Gy are used, depending on the type of lymphoma and chemotherapy regimen. Treatment is normally very well tolerated, with some cytopenia and fatigue with larger fields and some nausea/vomiting when the abdomen is treated.
Total-Body Irradiation
Total-body irradiation is used as part of the preparative scheme for peripheral blood stem cell transplant protocols. Treatment with 550 cGy in a single fraction is very common, although fractionated total-body irradiation is also used in certain protocols. This is normally well tolerated, except for self-limiting parotitis and nausea and vomiting.
Gynecologic Cancer
Cervical and uterine corpus cancers are often treated with radiation therapy. Most advanced cervical cancers are treated with definitive radiotherapy, and numerous phase III trials have demonstrated equivalent survival outcome between radical hysterectomy and radiotherapy for early cervical cancer. Radiation is generally used as adjuvant therapy in uterine corpus tumors. Radiotherapy for most gynecologic malignancies uses brachytherapy, because it better delivers the dose to the at-risk tissues, while sparing the bladder and rectum. Doses are anywhere from 50 to 85 Gy. Patients can develop proctitis, enteritis, and urethritis/cystitis, depending on the dose and tumor location. As with other sites, the frequency of admission for acute toxicity increases with concurrent chemoradiation.
KEY POINTS TO REMEMBER
![]() Consult a radiation oncology specialist before starting therapeutic chemotherapy or surgery to allow for optimum multidisciplinary management of the malignancy. Evaluating patients in their presenting state is very valuable to the radiation oncologist.
Consult a radiation oncology specialist before starting therapeutic chemotherapy or surgery to allow for optimum multidisciplinary management of the malignancy. Evaluating patients in their presenting state is very valuable to the radiation oncologist.
![]() When radiation toxicity is in the differential diagnosis, consult a radiation oncolo-gist, who can help with diagnosis and treatment.
When radiation toxicity is in the differential diagnosis, consult a radiation oncolo-gist, who can help with diagnosis and treatment.
![]() The only true radiation oncology emergency is spinal cord compression, although there are other “urgent” indications for radiation therapy.
The only true radiation oncology emergency is spinal cord compression, although there are other “urgent” indications for radiation therapy.
![]() Consider urgent radiation oncology consults when faced with SVC syndrome, new brain metastases, or uncontrolled bleeding.
Consider urgent radiation oncology consults when faced with SVC syndrome, new brain metastases, or uncontrolled bleeding.
REFERENCES
1. Gunderson L, Tepper J, eds. Clinical Radiation Oncology. 2nd ed. Philadelphia: Churchill Livingstone; 2006.
2. Perez C, Brady L, eds. Principles and Practicesof Radiation Oncology. 5th ed. Philadelphia: Lippincott-Raven; 2007.