LEUKOPENIA
General Principles
![]() The normal white blood cell (WBC) count varies with gender and ethnicity but, in general, ranges from 4 × 109 to 11 × 109 cells/L (Table 2-1) and is composed of those cells committed to the leukocyte lineage: granulocytes (neutrophils, eosinophils, and basophils), monocytes, and lymphocytes. Neutrophils make up about 60% of the peripheral blood nucleated cells. A person’s gender and ethnic background should be taken into consideration when determining normal ranges.
The normal white blood cell (WBC) count varies with gender and ethnicity but, in general, ranges from 4 × 109 to 11 × 109 cells/L (Table 2-1) and is composed of those cells committed to the leukocyte lineage: granulocytes (neutrophils, eosinophils, and basophils), monocytes, and lymphocytes. Neutrophils make up about 60% of the peripheral blood nucleated cells. A person’s gender and ethnic background should be taken into consideration when determining normal ranges.
![]() Leukopenia is defined as a WBC count <3.8 × 109 cells/L. This lower limit of normal varies with age (infants have lower absolute neutrophil counts [ANCs] than adults) and race (lower ANCs in persons of African ancestry, West Indians, Arab Jordanian, and Yemenite Jews),1 and 5% of thenormal population may fall outside of the normal reference range. Leukopenias can be divided according to clinically relevant cell lineages: neutrophils and lymphocytes.
Leukopenia is defined as a WBC count <3.8 × 109 cells/L. This lower limit of normal varies with age (infants have lower absolute neutrophil counts [ANCs] than adults) and race (lower ANCs in persons of African ancestry, West Indians, Arab Jordanian, and Yemenite Jews),1 and 5% of thenormal population may fall outside of the normal reference range. Leukopenias can be divided according to clinically relevant cell lineages: neutrophils and lymphocytes.
NEUTROPENIA
General Principles
Definition
The ANC is obtained by taking the percentage of neutrophils identified on a 100-cell differential or by the Coulter counter and multiplying by the total WBC count. Neutropenia is classified as mild (ANC, <1.5 × 109 to 1 × 109/L), moderate (ANC, 1 × 109 to 0.5 × 109/L), or severe (ANC, <0.5 × 109 /L). Agranulocytosis is the total absence of granulocytes.
Epidemiology
Neutropenia is five times more prevalent in African Americans than Caucasians.
Etiology
Causes of neutropenia in adults are reported in Table 2-2. Severe neutropenia can be congenital or acquired. Congenital causes are usually suggested by family history. Most cases of neutropenia are acquired and related to decreased granulocyte production and, less often, increased destruction. Pseudo-neutropenia may be obtained by analyzing blood several hours old and in the presence of paraproteinemia and certain anticoagulants that can cause clumping. Lower ANCs occur in African Americans as a result of defective release of neutrophils from the marrow, a poor marrow reserve, or an increased marginated pool of neutrophils.2
Pathophysiology
Neutropenia results from decreased production, ineffective granulopoiesis, increased margination to peripheral pools, or increased peripheral destruction. Acquired neutropenias are usually a result of infection, toxins/drugs, or immune disorders. Viral, parasitic, or bacterial infections may cause neutropenia, and this is usually short lived. The underlying mechanism involves increased margination, sequestration, and increased destruction by circulating antibodies. Drug and toxin exposure usually follows a temporal course, with neutropenia developing after continued drug exposure of days to months. The mechanism of drug-induced neutropenia is either antibody-mediated or direct toxic effects on the marrow. Certain drugs at higher risk of causing neutropenia are highlighted in Table 2-3. Primary immune disorders mediate neutropenia through antibody-mediated neutrophil destruction.
Diagnosis
Clinical Presentation
![]() Neutropenia is often incidentally discovered on a complete blood count (CBC) but may present with fever or infection. Signs of infection, such as purulence, may be less evident, given the low neutrophil count. The risk of infection is directly related to the degree and duration of neutropenia. The risk of infection increases at an ANC <1 × 109/L, but clinical symptoms usually do not become manifest until the ANC falls below 0.5 × 109/L.3
Neutropenia is often incidentally discovered on a complete blood count (CBC) but may present with fever or infection. Signs of infection, such as purulence, may be less evident, given the low neutrophil count. The risk of infection is directly related to the degree and duration of neutropenia. The risk of infection increases at an ANC <1 × 109/L, but clinical symptoms usually do not become manifest until the ANC falls below 0.5 × 109/L.3
![]() The initial evaluation should include a complete history and physical exam. The history should focus on systemic symptoms of infection, recent exposures or new medications, history of neutropenia, and family history of neutropenia. The physical exam may suggest the cause of neutropenia, and attention should be paid to vital signs that would suggest sepsis or infection, oral cavity exam for gingivitis or tooth abscess, macroglossia to suggest vitamin deficiency, lymphadenopathy to suggest malignancy or infection, skin and joint changes suggesting a rheumatologic disorder, and splenomegaly (sequestration and Felty’s syndrome).
The initial evaluation should include a complete history and physical exam. The history should focus on systemic symptoms of infection, recent exposures or new medications, history of neutropenia, and family history of neutropenia. The physical exam may suggest the cause of neutropenia, and attention should be paid to vital signs that would suggest sepsis or infection, oral cavity exam for gingivitis or tooth abscess, macroglossia to suggest vitamin deficiency, lymphadenopathy to suggest malignancy or infection, skin and joint changes suggesting a rheumatologic disorder, and splenomegaly (sequestration and Felty’s syndrome).
Diagnostic Testing
Initial laboratory evaluation starts with the CBC with complete differential and review of the peripheral blood smear. Additional testing to consider includes nutritional studies of vitamin B12, folate, and possibly copper. If a clonal process is suspected, lymphocyte immunophenotyping by flow cytometry and T-cell receptor gene rearrangement studies may be useful. Antinuclear antibody and antineutrophil antibody testing can be sent to evaluate for autoimmune neutropenia. HIV and EBV serologies start the initial infectious workup. If anemia or thrombocytopenia occurs in combination with neutropenia, direct examination of the bone marrow via bone marrow biopsy is usually warranted unless a cause is obvious. In cases of asymptomatic mild neutropenia, serial CBC examination to rule out cyclic neutropenia may be considered. In mild cases of neutropenia that do not improve in a couple of months with observation, a bone marrow biopsy should be considered.
Treatment
![]() Treatment is guided by the underlying etiology and severity of neutropenia. This can range from close observation in patients with benign neutropenia to growth factor support and antibiotics in patients with neutropenic fevers.
Treatment is guided by the underlying etiology and severity of neutropenia. This can range from close observation in patients with benign neutropenia to growth factor support and antibiotics in patients with neutropenic fevers.
![]() Growth factors can be used to speed count recovery in drug-induced neutropenia. The major complication associated with neutropenia is infection.
Growth factors can be used to speed count recovery in drug-induced neutropenia. The major complication associated with neutropenia is infection.
![]() Supportive care with broad-spectrum antibiotics in the ill or febrile patient is an essential part of initial care while the workup for a cause of neutropenia is under way. Common sites of infection include mucous membranes, skin, perirectal and genital areas, bloodstream, and lungs. Most commonly, endogenous bacterial flora is the pathogen (Staphylococcus from skin or gram-negative organisms from the gut). Antibiotics should be continued until the ANC is >500/L for 2 days and the fever subsides. If fever and neutropenia persist, empiric antifungal coverage should be considered.
Supportive care with broad-spectrum antibiotics in the ill or febrile patient is an essential part of initial care while the workup for a cause of neutropenia is under way. Common sites of infection include mucous membranes, skin, perirectal and genital areas, bloodstream, and lungs. Most commonly, endogenous bacterial flora is the pathogen (Staphylococcus from skin or gram-negative organisms from the gut). Antibiotics should be continued until the ANC is >500/L for 2 days and the fever subsides. If fever and neutropenia persist, empiric antifungal coverage should be considered.
![]() Cases caused by drug toxicity should improve, with removal of the drug within 1 to 3 weeks. Drug-related neutropenia can be confirmed by testing antineutrophil-associated drug antibodies. Infectious etiologies resolve with treatment of the infection or shortly after a viral infection has subsided. Autoimmune diseases can be treated by immunosuppression with corticosteroids and can be confirmed by testing antineutrophil antibodies. Congenital etiologies are often supported with growth factors such as granulocyte colony-stimulating factor (G-CSF). The involvement of other blood cell lineages (RBCs and platelets) suggests aplastic anemia, leukemia, myelodysplastic syndromes, or megaloblastic anemia.
Cases caused by drug toxicity should improve, with removal of the drug within 1 to 3 weeks. Drug-related neutropenia can be confirmed by testing antineutrophil-associated drug antibodies. Infectious etiologies resolve with treatment of the infection or shortly after a viral infection has subsided. Autoimmune diseases can be treated by immunosuppression with corticosteroids and can be confirmed by testing antineutrophil antibodies. Congenital etiologies are often supported with growth factors such as granulocyte colony-stimulating factor (G-CSF). The involvement of other blood cell lineages (RBCs and platelets) suggests aplastic anemia, leukemia, myelodysplastic syndromes, or megaloblastic anemia.
LYMPHOPENIA
General Principles
Definition
Lymphopenia is defined as an absolute lymphocyte count <1.2 109/L. The absolute lymphocyte count is 80% T cells and 20% B cells. Sixty-six percent of the T-cell population is CD4+ cells and the remaining is mainly CD8+cells.
Etiology
Lymphopenia is most often acquired, but congenital causes should also be considered. Etiologies of lymphopenia are listed in Table 2-4 and are mainly acquired. Patients who appear to have low or absent CD4 cells should be evaluated for OKT4 epitope deficiency. This condition is found in 8% of individuals of African descent. Individuals with OKT4 epitope deficiency usually have normal CD4+ number and do not develop infections.4
Treatment
Most causes of lymphocytopenia are acquired, and the management focuses on treating the underlying illness. The most common infectious cause is acquired immunodeficiency syndrome (AIDS). Other viral and bacterial diseases also cause lymphocytopenia, which usually resolves a couple of weeks after antimicrobial therapy. Zinc deficiency responds to repletion of zinc and should be part of the initial screen, along with examination of the peripheral blood smear. Inherited causes predispose to recurrent and opportunistic infections, and detailed discussion of management is beyond the scope of this text. In general, prophylactic antibiotics can be used, as well as best supportive care.
LEUKOCYTOSIS
General Principles
An elevated WBC most commonly reflects a normal bone marrow response to inflammation or infection. Occasionally leukemia or myeloproliferative disorders are to blame. The maturation of WBCs is influenced by G-CSFs, interleukins (ILs), tumor necrosis factor, and complement components.
Definition
Leukocytosis is defined as a WBC count >10 × 109 cells/L.
Classification
Leukocytosis should be divided into granulocytosis, monocytosis, and lymphocytosis to guide the workup and differential diagnosis.
Etiology
Most cases of leukocytosis are a result of the bone marrow reacting to inflammation or infection. A leukemoid reaction is an excessive WBC response (usually >50,000) associated with a cause outside of the bone marrow (growth factors, infection, or differentiating agents such as all-trans retinoic acid [ATRA]). Leukocytosis may also be caused by physical and emotional stress and usually resolves in hours once the stress is eliminated. In postsplenectomy patients, a transient leukocytosis can be seen, lasting for weeks to months secondary to the demargination of leukocytes typically stored in the spleen. Other etiologies include medications, but leukocytes should not rise above 20,000 to 30,000 in this case. The leukocytosis seen in hemolytic anemias (sickle cell and autoimmune types) is related to the nonspecific effects of increased erythropoiesis and inflammation. Nonhematopoietic malignancy can also cause a leukocytosis that is multifactorial in etiology. Finally, acute and chronic leukemias and myeloproliferative disorders usually present with a leukocytosis.

Pathophysiology
The pathophysiology of leukocytosis stems from the production, maturation, and survival of leukocytes. Stem cells give rise to erythroblasts, myeloblasts, and megakaryoblasts. Seventy-five percent of nucleated cells in the bone marrow are committed to production of leukocytes. At any given time, 90% of WBCs remain in storage in the bone marrow, with 7% to 8% in the tissue compartment and the remainder in circulation. This large storage pool allows for a rapid increase in WBCs (mostly neutrophils). In addition, a percentage of circulating WBCs is marginated along blood vessel walls and is mobilized by inflammatory stimuli. The two basic causes of leukocytosis are a normal bone marrow response to external stimuli or the effect of a primary bone marrow disorder.
Diagnosis
The differential diagnosis of leukocytosis is extensive, and common causes are listed in Table 2-5. Increases in the absolute numbers of lymphocytes, eosinophils, monocytes, or basophils are less common in leukocytosis than neutrophilia and help to direct the differential diagnosis.
NEUTROPHILIA
General Principles
Definition
Neutrophilia is defined as an ANC >6.6 × 109/L. The neutrophil count is influenced by shifts in neutrophils among four major compartments: the bone marrow, the circulation, the marginated pool, and the tissues. Only about 5% of neutrophils are in circulation at any given time, with a half-life of 6 to 10 hours. Most neutrophils and their precursors are contained in storage pools in the bone marrow at 10 to 20 times their circulating numbers. About 50% of peripheral blood neutrophils are circulating, and the other 50% marginated along vessel walls and in the spleen. This pool can be rapidly increased, within hours from the bone marrow stores or within minutes from demarginating neutrophils along blood vessel walls. Neutrophils move to sites of inflammation and infection and act as phagocytes. Their trafficking depends on chemotaxins and surface molecules such as selectins to mediate rolling and integrins to mediate adhesion and transmigration of blood vessels.
Pathophysiology
The pathophysiology of primary neutrophilia may be related to inherited deficiencies in adhesion molecules or, in the case of myeloproliferative disorders, constitutive expression and activation of a growth-promoting receptor tyrosine kinase such as bcr/abl or Jak2. Secondary neutrophilia, seen in infection and inflammation, is related to demargination from storage pools in the bone marrow and peripheral blood signaled by endotoxin and proinflammatory cytokines such as tumor necrosis factor-alpha, IL-6, IL-1B, IL-8, G-CSF, and granulocyte/macrophage colony-stimulating factor (GM-CSF).
Diagnosis
Differential Diagnosis
![]() Neutrophilia can be spurious, of primary hematologic origin, or related to secondary causes. Etiologies of neutrophilia are listed in Table 2-6. Spurious leukocytosis can be a result of the automated cell counter (Coulter counter) counting clumps of platelets as leukocytes and is usually associated with pseudo-thrombocytopenia. In addition, cryoglobulins can agglutinate and be counted as leukocytes at temperatures lower than body temperature.
Neutrophilia can be spurious, of primary hematologic origin, or related to secondary causes. Etiologies of neutrophilia are listed in Table 2-6. Spurious leukocytosis can be a result of the automated cell counter (Coulter counter) counting clumps of platelets as leukocytes and is usually associated with pseudo-thrombocytopenia. In addition, cryoglobulins can agglutinate and be counted as leukocytes at temperatures lower than body temperature.

![]() Primary causes of neutrophilia may be hereditary (usually resulting in splenomegaly and leukocyte counts of 20 × 109 to 100 × 109/L) or associated with familial syndromes. Other primary causes include myeloproliferative disorders (e.g., chronic myeloid leukemia [CML]) and leukocyte adhesion deficiency.
Primary causes of neutrophilia may be hereditary (usually resulting in splenomegaly and leukocyte counts of 20 × 109 to 100 × 109/L) or associated with familial syndromes. Other primary causes include myeloproliferative disorders (e.g., chronic myeloid leukemia [CML]) and leukocyte adhesion deficiency.
![]() Secondary causes are by far the most common cause of neutrophilia. Common secondary causes include infection, smoking (25% increase), chronic inflammation (e.g., rheumatoid arthritis and inflammatory bowel disease), stress, medications, chronic marrow stimulation (hemolytic anemia and idiopathic thrombocytopenic purpura), asplenia, marrow invasion, and nonhematologic malignancy.
Secondary causes are by far the most common cause of neutrophilia. Common secondary causes include infection, smoking (25% increase), chronic inflammation (e.g., rheumatoid arthritis and inflammatory bowel disease), stress, medications, chronic marrow stimulation (hemolytic anemia and idiopathic thrombocytopenic purpura), asplenia, marrow invasion, and nonhematologic malignancy.
Diagnostic Testing
Initial laboratory evaluation starts with review of the peripheral blood smear to confirm automated counts and rule out spurious leukocytosis. The smear may suggest a secondary cause such as infection or inflammation with increased bands, vacuolization, Döhle bodies, and toxic granulations in neutrophils. A marrow-infiltrating process is suggested by a leukoerythroblastic reaction that shows a “left shift” (increased myelocytes and metamyelocytes in the marrow and bands in the peripheral blood) and nucleated RBCs. Acute leukemia is suggested by circulating blasts, which may be incorrectly counted as monocytes or neutrophils by the Coulter counter. If no secondary causes of neutrophilia can be identified, peripheral blood analysis for bcr/abl by fluorescence in situ hybridization (FISH) or cytogeneticsmay be helpful to exclude CML. A leukocyte alkaline phosphatase (LAP) score is of historical importance but is no longer commonly used because of intraoperator variability and the evolution of cytogenetic testing. A low LAP score can be seen in CML, and a high LAP score may suggest inflammation or infection.
Treatment
Treatment depends on the underlying etiology. Treatment of primary etiologies such as CML and myeloproliferative disorders are discussed elsewhere in this book. Treatment of neutrophilia related to a secondary cause revolves around treating the underlying cause.
EOSINOPHILIA
General Principles
Eosinophilia is defined as an absolute eosinophil count >0.5 × 109/L. Eosinophilia is most commonly due to secondary causes. Table 2-7 reviews causes of eosinophilia. Absolute eosinophil counts >4 × 109/L suggest primary eosinophilia as a result of either clonal expansion (chronic leukemia variant or acute leukemia variant) or hypereosinophilic syndrome.5
Diagnosis
Initial evaluation of eosinophilia should include review of the peripheral smear, stool examination for ova and parasites, and serum tryptase, cortisol, IgE, and IL-5 levels. If no secondary source can be identified, T-cell immunophenotyping and T-cell receptor gene rearrangement analysis and bone marrow biopsy with cytogenetic analysis and FISH for the platelet-derived growth factor receptor-alpha (FIP1L1-PDGFRA) rearrangement and bcr/ablshould be performed to evaluate for a clonal disorder.

Treatment
Hypereosinophilic syndrome can cause end organ damage, including cardiac involvement causing conduction defects and cardiomyopathy, as well as pulmonary involvement. In patients with evidence of end organ damage, treatment with corticosteroids and hydroxyurea may be needed to decrease the eosinophil count rapidly. Leukopheresis may be used as well to lower the eosinophil count rapidly. Recent evidence suggests that patients with idiopathic hypereosinophilic syndrome and chronic eosinophilic leukemia with the FIP1L1-PDGFRA rearrangement may be effectively treated with the tyrosine kinase inhibitor imatinib.6
BASOPHILIA
General Principles
Definition
Basophilia is defined as an absolute basophil count >0.2 × 109/L. Basophils are inflammatory mediators, and their granules contain histamine, glycosaminoglycans, major basic protein, proteases, and other inflammatory and vasoactive substances. They primarily function to activate the type 1 hypersensitivity reaction mediated through surface receptors for IgE.
Etiology
Basophilia can be associated with hypersensitivity reactions to drugs and food. It may also be seen in chronic inflammatory states such as tuberculosis and ulcerative colitis. However, these reactions are rare, and the most common setting of basophilia is in myeloproliferative disorders such as CML.
Diagnosis
Review of the peripheral smear confirms basophilia and management focuses on the underlying etiology. Peripheral blood can be sent for Jak2 and bcr/abl to evaluate for a myeloproliferative disorder. If suspicion of a myeloproliferative disorder is high, a bone marrow biopsy is necessary.
MONOCYTOSIS
General Principles
Definition
Monocytosis is defined as an absolute monocyte count >0.8 × 109/L. Monocytes are cells in transit to the tissues and are capable of transformation to macrophages in the tissues. They play a role in acute and chronic inflammatory reactions.
Etiology
Monocytosis usually represents a myeloproliferative disorder such as CML or acute monocytic leukemia. Secondary causes include infection (bacterial or tuberculosis) and relative monocytosis as seen with initial count recovery after chemotherapy and drug-induced neutropenia.
Diagnosis
Review of the peripheral smear confirms monocytosis, and treatment is focused on the underlying etiology. Peripheral blood can be sent for Jak2 and bcr/abl to evaluate for a myeloproliferative disorder. If suspicion of a myeloproliferative disorder is high, a bone marrow biopsy is necessary.
LYMPHOCYTOSIS
General Principles
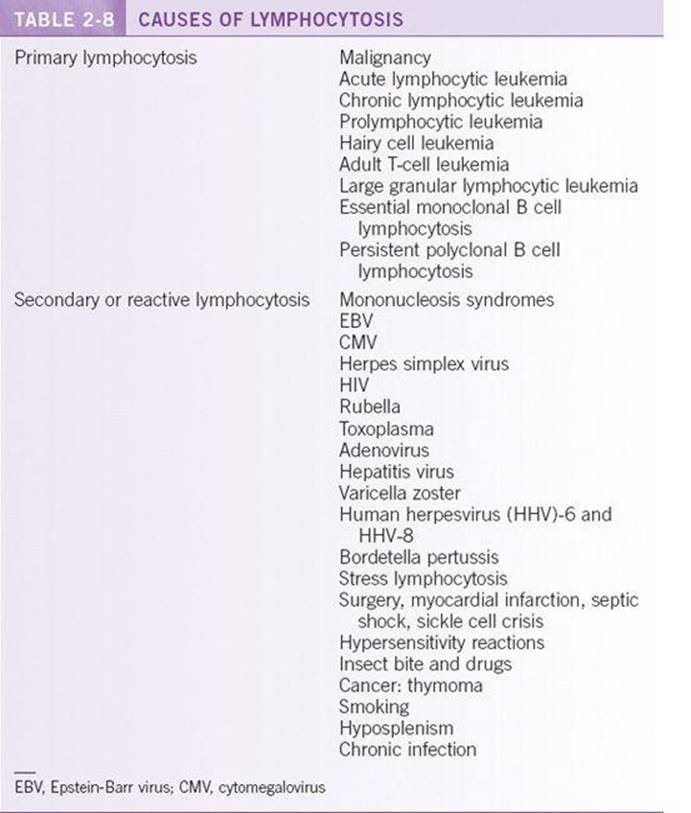
Lymphocytosis is defined as an absolute lymphocyte count >3.3 × 109/L. Lymphocytosis may be of primary or secondary origin. Table 2-8 reviews causes of lymphocytosis. Cell surface markers are important in determining primary from secondary lymphocytosis.
Diagnosis
The blood smear should be reviewed to look for evidence of reactive lymphocytes associated with infection, large granular lymphocytes associated with large granular lymphocytic leukemia, smudge cells associated with chronic lymphocytic leukemia (CLL), or blasts associated with acute leukemia. Peripheral blood flow cytometry immunophenotyping allows identification of clonal disorders. Immunoglobulin or T-cell receptor gene rearrangements support a clonal disorder.
Treatment
Management of hematological malignancies including CLL is discussed in Chapter 29. Resolution of infectious etiologies results in resolution of the lymphocytosis. Finally, removal of allergens such as drugs or venom results in resolution of the lymphocytosis associated with hypersensitivity reactions.
REFERENCES
1. Shoenfeld Y, Alkan ML, Asaly A, et al. Benign familial leukopenia and neutropenia in different ethnic groups. Eur J Haematol. 1988;41(3):273–277.
2. Andersohn F, Konzen C, Garbe E. Systematic review: agranulocytosis induced by nonchemotherapy drugs. Ann Intern Med. 2007;146 (9):657–665.
3. Brown AE. Neutropenia, fever, and infection. Am J Med. 1984;76(3):421–428.
4. Bach MA, Phan-Dinh-Tuy F, Bach JF, et al. Unusual phenotypes of human inducer T cells as measured by OKT4 and related monoclonal antibodies. J Immunol. 1981;127(3):980–982.
5. Tefferi A, Patnaik MM, Pardanani A. Eosinophilia:secondary, clonal and idiopathic. Br J Haematol. 2006;133(5):468–492.
6. Jovanovic JV, Score J, Waghorn K, et al. Low-dose imatinib mesylate leads to rapid induction of major molecular responses and achievement of complete molecular remission in FIP1L1-PDGFRA-positive chronic eosinophilic leukemia. Blood. 2007;109(11):4635–4640.