PROSTATE CANCER
GENERAL PRINCIPLES
Prostate cancer is the most common non-cutaneous cancer among men in the United States and is the second leading cause of cancer death in men after lung cancer. The American Cancer Society estimated that there would be 217,730 new cases and 32,050 deaths from prostate cancer in 2010. One man in 6 would get prostate cancer during his lifetime and one in 36 die of this disease. The course is often indolent and the average age at presentation is late in life. As a result of widespread screening, most patients today are diagnosed with asymptomatic, prostate-confined disease.
Risk Factors
Age is the most significant risk factor, and 2 out of every 3 prostate cancers are found in men older than 65 years, as per the American Cancer Society. Increased risk is also conferred to patients with a positive family history and those of African American descent. High-fat and red meat diets appear to correlate positively with prostate cancer development. Benign prostatic hypertrophy is not a risk factor.
Prevention
No method of prevention has been found to be effective in clinical trials. 5-Alpha-reductase enzyme inhibitors, finasteride1 and dutasteride2, have been found to temporarily shrink tumors that probably would not have been fatal, without being able to prevent cancer from development. Furthermore, they suppress levels of prostate-specific antigen (PSA) and may provide a false sense of security that may have contributed to finding of higher grade tumors found in patients treated with finasteride compared with placebo.
DIAGNOSIS
![]() Screening for prostate cancer in asymptomatic patients remains controversial. US death rates from prostate cancer have been falling since introduction of PSA testing. However, it’s unclear whether the harms of testing outweigh the benefits for the general asymptomatic population. Preliminary results from the US Prostate, Lung, Colorectal, and Ovarian (PLCO) Cancer Screening Trial showed no mortality benefit from combined screening with PSA testing and digital rectal exam during a medical follow-up of 11 years.3 Meanwhile, preliminary results from the European Randomized Study of Screening for Prostate Cancer (ERSPC) Trial noted that 1410 men needed to be screened and 48 needed to be treated to prevent one prostate cancer– related death in a 10-year period.4 These trials as well as several others, such as Prostate Cancer Intervention Versus Observation Trial (PIVOT) in the United States, are ongoing and the final results are pending.
Screening for prostate cancer in asymptomatic patients remains controversial. US death rates from prostate cancer have been falling since introduction of PSA testing. However, it’s unclear whether the harms of testing outweigh the benefits for the general asymptomatic population. Preliminary results from the US Prostate, Lung, Colorectal, and Ovarian (PLCO) Cancer Screening Trial showed no mortality benefit from combined screening with PSA testing and digital rectal exam during a medical follow-up of 11 years.3 Meanwhile, preliminary results from the European Randomized Study of Screening for Prostate Cancer (ERSPC) Trial noted that 1410 men needed to be screened and 48 needed to be treated to prevent one prostate cancer– related death in a 10-year period.4 These trials as well as several others, such as Prostate Cancer Intervention Versus Observation Trial (PIVOT) in the United States, are ongoing and the final results are pending.
![]() The American Cancer Society does not currently recommend routine PSA testing and recommends individualized discussion between the patient and his physician. US Preventive Services Task Force most recent recommendations from 2008 recommend against screening for men older than 75 years and conclude there is insufficient data to recommend for or against routine testing for younger men. As of November 2009, the American Urological Association continues to recommend PSA testing starting at age 40 years.
The American Cancer Society does not currently recommend routine PSA testing and recommends individualized discussion between the patient and his physician. US Preventive Services Task Force most recent recommendations from 2008 recommend against screening for men older than 75 years and conclude there is insufficient data to recommend for or against routine testing for younger men. As of November 2009, the American Urological Association continues to recommend PSA testing starting at age 40 years.
Clinical Presentation
Often patients with prostate cancer are asymptomatic. However, obstructive symptoms as well as dysuria, back pain, and hematuria can be initial presenting symptoms. In some cases, disease may become evident only after investigation of metastatic symptoms such as spinal cord compression or bone pain, which is the favored site of metastasis.
Physical Exam
Carcinoma of the prostate can develop within the posterior surfaces of the lateral lobes, which are palpable during the digital rectal exam (DRE). Sensation of hard irregular nodules on the DRE is characteristic. Deeper or more anterior lesions, however, are not detectable on routine DRE. One should also recognize that detection sensitivity also varies significantly between examiners owing to differences in experience and technique. Trials for detecting early disease suggest that the physical exam, or even an ultrasonography (U/S) exam, is less sensitive than measurement of PSA.5 On occasion, disease disseminates to the lymph nodes, causing evidence of scrotal or lower-extremity lymphedema on physical exam.
Diagnostic Testing
Laboratories
Because signs and symptoms are often nonspecific or nonexistent, diagnosis is often suggested by use of serum markers. The relatively high sensitivity (70% to 80%) and noninvasive nature of the total serum PSA assay have made it the most often used test. Although PSA levels fall on a continuum, a normal level is considered to be <4 ng/mL. Of note, levels typically elevate with age and following prostatic massage, and they can also be elevated in patients with benign prostatic hypertrophy and prostatitis. On the other hand, men with high Gleason score tumors may have tumors so undifferentiated that they do not synthesize large amounts of PSA. In the general population, sensitivity of PSA >4 has been estimated at 70% to 80%, while the specificity is estimated to be about 60% to 70%.6 Perhaps the most important feature of PSA testing is the ability to follow its change over time (PSA velocity).
Imaging
Although transrectal ultrasound has been used for screening and staging in some situations, its greatest use is to guide prostatic biopsies. Bone scan is useful in identifying bone metastasis and is recommended for men with PSA >20. CT can supplement clinical evaluation and should also be used with PSA >20 or T3/T4 stage tumors to evaluate for lymph node involvement. Role of PET and MRI is not yet clear.
Diagnostic Procedures
Biopsy is essential for diagnosis. The sensitivity can also be increased when more needle cores are obtained. A minimum of 6 cores is standard, although many patients routinely have 8 to 12 cores/biopsy sessions. Histologic grade is an important determinant of disease course and patient survival. Adenocarcinomas represent >95% of prostate cancers and are graded histologically using the Gleason scoring system. This system takes the two most predominant histologic patterns in the area of the tumor and assigns each a number from 1 to 5. These numbers are then added together to give the total score. Higher scores correlate with more poorly differentiated tumors and worse prognosis. Squamous and transitional cell tumors make up the majority of the remaining prostate tumors, with another important subset being the high-grade neuroendocrine or small-cell tumors.
TREATMENT
The key determinants in considering the optimal treatment of prostate cancer are estimates of life expectancy and risk of cancer progression. The risk of cancer progression can be estimated using the pathologic stage, which is determined by the clinical stage (based on DRE), preoperative PSA, and biopsy Gleason score. The National Comprehensive Cancer Network publishes a staging monogram based on these three features in their Clinical Practice Guidelines in Oncology. It is freely accessible at http://www.nccn.org/. Patients with prostate cancer can be divided for the purposes of treatment into three groups to guide treatment: localized prostate cancer, locally advanced prostate cancer, or advanced prostate cancer.
![]() Localized prostate cancer. By definition, cancer is confined to the prostate. These patients are further subdivided into three risk categories:
Localized prostate cancer. By definition, cancer is confined to the prostate. These patients are further subdivided into three risk categories:
![]() Low risk (T1-T2a, Gleason score ≤6, PSA <10): Expectant management, radical prostatectomy (RP), external beam radiation therapy (EBRT), or brachytherapy are all reasonable options for treatment. If expectant management is chosen, PSA should be checked at least every 6 months and DRE at least once a year. Curative therapy is initiated at onset of disease progression. No clinical trials have compared expectant management with immediate treatment. Choice of therapy otherwise is based on patient preferences, as no clinical trials have found any treatment modality to be superior.
Low risk (T1-T2a, Gleason score ≤6, PSA <10): Expectant management, radical prostatectomy (RP), external beam radiation therapy (EBRT), or brachytherapy are all reasonable options for treatment. If expectant management is chosen, PSA should be checked at least every 6 months and DRE at least once a year. Curative therapy is initiated at onset of disease progression. No clinical trials have compared expectant management with immediate treatment. Choice of therapy otherwise is based on patient preferences, as no clinical trials have found any treatment modality to be superior.
![]() Intermediate risk (T2b-T2c or GS 7 or PSA 10 to 20): Unless expected survival is less than 10 years, expectant management is not acceptable in this category. Otherwise, RP, EBRT, or brachytherapy are equivalent management options.
Intermediate risk (T2b-T2c or GS 7 or PSA 10 to 20): Unless expected survival is less than 10 years, expectant management is not acceptable in this category. Otherwise, RP, EBRT, or brachytherapy are equivalent management options.
![]() High risk (T3a or GS 8 to 10 or PSA >20): These patients are treated with either RP with pelvic lymph node dissection or EBRT combined with at least 2 to 3 years of androgen deprivation therapy7 (ADT; see below).
High risk (T3a or GS 8 to 10 or PSA >20): These patients are treated with either RP with pelvic lymph node dissection or EBRT combined with at least 2 to 3 years of androgen deprivation therapy7 (ADT; see below).
![]() Locally advanced prostate cancer (T3b-T4). Similar to patients with high-risk localized disease, these patients are treated with either RP with pelvic lymph node dissection or EBRT combined with 2 to 3 years of ADT. It is also acceptable to treat this group with only ADT.
Locally advanced prostate cancer (T3b-T4). Similar to patients with high-risk localized disease, these patients are treated with either RP with pelvic lymph node dissection or EBRT combined with 2 to 3 years of ADT. It is also acceptable to treat this group with only ADT.
![]() Metastatic prostate cancer. Surgery is not usually used for these patients. Radiation or TURP can be used to palliate obstructive symptoms. Otherwise, initial choice is ADT followed by chemotherapy in castrate-resistant prostate cancer.
Metastatic prostate cancer. Surgery is not usually used for these patients. Radiation or TURP can be used to palliate obstructive symptoms. Otherwise, initial choice is ADT followed by chemotherapy in castrate-resistant prostate cancer.
![]() PSA only recurrence. These patients have a “biochemical recurrence” that occurs after either radiation therapy or surgical resection, and no source of recurrence other than elevated PSA can be found clinically or through imaging. Treatment options include watchful waiting, radiation therapy if they had previously had RP, or in selected patients salvage RP if they were originally treated with radiation therapy.
PSA only recurrence. These patients have a “biochemical recurrence” that occurs after either radiation therapy or surgical resection, and no source of recurrence other than elevated PSA can be found clinically or through imaging. Treatment options include watchful waiting, radiation therapy if they had previously had RP, or in selected patients salvage RP if they were originally treated with radiation therapy.
![]() Watchful waiting. This refers to complete deferment of therapy and instead proceeding with palliative therapy. Prostate cancer is often indolent in nature, which allows for watchful waiting as a reasonable approach in selected patients. In general, this option is reserved for patients whose life expectancy is <10 years and/or who have other comorbidities limiting treatment options.
Watchful waiting. This refers to complete deferment of therapy and instead proceeding with palliative therapy. Prostate cancer is often indolent in nature, which allows for watchful waiting as a reasonable approach in selected patients. In general, this option is reserved for patients whose life expectancy is <10 years and/or who have other comorbidities limiting treatment options.
![]() Androgen deprivation therapy. Prostate cancer is testosterone dependent, and androgen deprivation often aids in controlling the disease. ADT can be achieved either surgically through orchiectomy or through hormonal suppression. Most men opt for hormonal therapy instead of orchiectomy for psychological reasons. GnRH agonists are first-line therapy for ADT and are as efficacious as bilateral orchiectomy. Continuous treatment with GnRH agonists induces suppression of LH synthesis and, therefore, testosterone production. Due to being agonists, however, they cause a temporary flare in symptoms on initiation. Therefore, GnRH agonists should be coupled with androgen receptor antagonists when first initiated and continued for at least 7 days. There are several GnRH agonists available, such as leuprolide and goserelin. If patients progress through GnRH agonists, an androgen receptor antagonist (e.g., flutamide, bicalutamide, nilutamide) can be added to the regimen to achieve complete androgen blockade.
Androgen deprivation therapy. Prostate cancer is testosterone dependent, and androgen deprivation often aids in controlling the disease. ADT can be achieved either surgically through orchiectomy or through hormonal suppression. Most men opt for hormonal therapy instead of orchiectomy for psychological reasons. GnRH agonists are first-line therapy for ADT and are as efficacious as bilateral orchiectomy. Continuous treatment with GnRH agonists induces suppression of LH synthesis and, therefore, testosterone production. Due to being agonists, however, they cause a temporary flare in symptoms on initiation. Therefore, GnRH agonists should be coupled with androgen receptor antagonists when first initiated and continued for at least 7 days. There are several GnRH agonists available, such as leuprolide and goserelin. If patients progress through GnRH agonists, an androgen receptor antagonist (e.g., flutamide, bicalutamide, nilutamide) can be added to the regimen to achieve complete androgen blockade.
![]() Another option for androgen deprivation is suppression of adrenal androgen synthesis with high dose Ketoconazole. Corticosteroids are also effective for suppressing the adrenal androgen synthesis by reducing the production of pituitary ACTH.
Another option for androgen deprivation is suppression of adrenal androgen synthesis with high dose Ketoconazole. Corticosteroids are also effective for suppressing the adrenal androgen synthesis by reducing the production of pituitary ACTH.
![]() Themain adverse effects of ADT include hot flashes, decreases in libido, and loss of muscle mass. Osteoporosis also occurs at a higher rate with long-term androgen deprivation. A baseline bone density screen is therefore recommended prior to initiation of ADT. Patients should be supplemented with calcium and vitamin D and bisphosphonate therapy should also be offered to men who are found to be osteopenic or osteoporotic.
Themain adverse effects of ADT include hot flashes, decreases in libido, and loss of muscle mass. Osteoporosis also occurs at a higher rate with long-term androgen deprivation. A baseline bone density screen is therefore recommended prior to initiation of ADT. Patients should be supplemented with calcium and vitamin D and bisphosphonate therapy should also be offered to men who are found to be osteopenic or osteoporotic.
![]() Chemotherapy. Many men receiving ADT will eventually develop increasing PSA values despite continued therapy. This progression of disease is termed androgen-independent or castrate-resistant prostate cancer. This resistance is due to adaptations at the cellular level that allows the prostate cancer cell to grow despite a low-androgen environment. A docetaxel-based regimen with prednisone is the current standard-of-care therapy for patients with androgen-independent prostate cancer, based on a demonstrated survival benefit of 2 to 3 months over mitoxantrone- and corticosteroid-based regimens in two phase III trials (Southwest Oncology Group [SWOG] 99168 and TAX 3279).
Chemotherapy. Many men receiving ADT will eventually develop increasing PSA values despite continued therapy. This progression of disease is termed androgen-independent or castrate-resistant prostate cancer. This resistance is due to adaptations at the cellular level that allows the prostate cancer cell to grow despite a low-androgen environment. A docetaxel-based regimen with prednisone is the current standard-of-care therapy for patients with androgen-independent prostate cancer, based on a demonstrated survival benefit of 2 to 3 months over mitoxantrone- and corticosteroid-based regimens in two phase III trials (Southwest Oncology Group [SWOG] 99168 and TAX 3279).
RENAL CELL CARCINOMA
GENERAL PRINCIPLES
Primary cancers of the kidney can be divided into cancers of the renal parenchyma and cancers of the renal pelvis that are generally transitional cell tumors. This section focuses on cancers of the renal parenchyma, which are generally adenocarcinomas (renal cell carcinoma [RCC]). Histologically RCC comprises of four main subtypes. Clear cell is the most common and accounts for ~80% of the cases. Papillary or chromophilic RCC accounts for 10%, followed by chromophobic RCC and collecting duct RCC.
Epidemiology
The American Cancer Society estimates 58,240 new cases of RCC and 13,040 cancer deaths in 2010. The overall incidence of RCC is rising for unclear reasons. It is more common in men currently and slightly more common in blacks. The higher incidence in males may change as smoking rates equalize between men and women.
Risk Factors
As with prostate cancer, age is the major risk factor for RCC. Accordingly, disease predominantly presents in the sixth to eighth decades of life. Other risk factors include cigarette smoking, obesity, hypertension, acquired cystic kidney disease associated with dialysis, polycystic kidney disease, and occupational exposure to heavy metal, asbestos, or petroleum products. Although not common, there are several hereditary forms of renal cancer as well. A well-known example is von Hipple–Lindau syndrome in which approximately two-thirds of patients develop RCC.
DIAGNOSIS
Clinical Presentation
![]() Unfortunately many patients are asymptomatic until advanced stages of disease. Hematuria, abdominal pain, and a palpable flank or abdominal mass are the classic triad of RCC diagnosis, but occur in combination only 10% of the time.10
Unfortunately many patients are asymptomatic until advanced stages of disease. Hematuria, abdominal pain, and a palpable flank or abdominal mass are the classic triad of RCC diagnosis, but occur in combination only 10% of the time.10
![]() Hematuria is the most common finding in RCC. Other symptoms include fever, night sweats, malaise, and weight loss. One interesting potential presenting symptom in men is a left-sided varicocele, secondary to obstruction of the left testicular vein. Paraneoplastic syndromes are rare, but include erythrocytosis and hypercalcemia from overproduction of erythropoietin or parathyroid hormone-related protein, respectively. Stauffer’s syndrome is another RCC paraneoplastic syndrome to keep in mind, which consists of hepatic dysfunction in absence of liver metastasis. This dysfunction may be due to production oftumorcytokines.
Hematuria is the most common finding in RCC. Other symptoms include fever, night sweats, malaise, and weight loss. One interesting potential presenting symptom in men is a left-sided varicocele, secondary to obstruction of the left testicular vein. Paraneoplastic syndromes are rare, but include erythrocytosis and hypercalcemia from overproduction of erythropoietin or parathyroid hormone-related protein, respectively. Stauffer’s syndrome is another RCC paraneoplastic syndrome to keep in mind, which consists of hepatic dysfunction in absence of liver metastasis. This dysfunction may be due to production oftumorcytokines.
Diagnostic Testing
Imaging
The best initial imaging modality for a suspected RCC is an ultrasound. It is inexpensive, is without radiation, and has a high sensitivity. It is also very useful for distinguishing simple cysts from complex and malignant masses. If a suspicious mass is found, ultrasound is typically followed by abdominal CT or MR to further characterize lymph node and regional involvement. Role of PET in initial diagnosis is unclear. Bone scan is indicated if patients have bone pain or unexplained elevated alkaline phosphatase that could be due to Stauffer’s syndrome (see “Clinical presentation”).
Diagnostic Procedures
Despite the sensitivity of imaging studies, biopsy of suspected metastatic lesions or nephrectomy is needed to definitely establish RCC diagnosis, as well as to delineate histologic subtype. Needle biopsy of the renal mass is not recommended due to concern for seeding of the peritoneum, as well as sampling errors that result in a negative biopsy.
TREATMENT
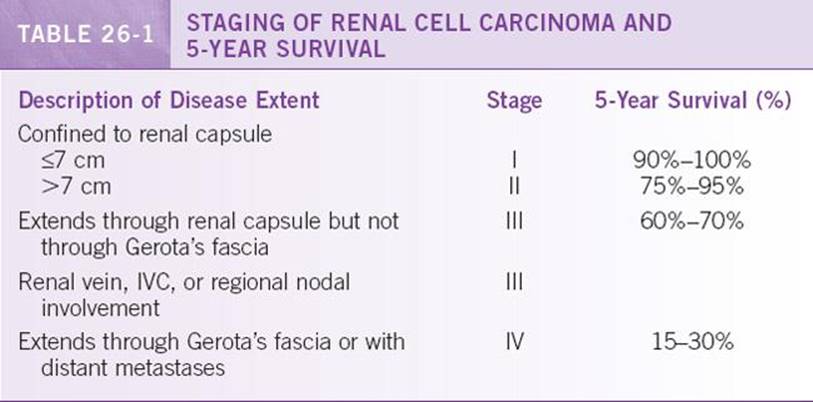
Table 26-1 lists a simplified staging of RCCs and their associated prognosis based on contemporary studies.11 RCC is one of the most resistant tumors to therapy, so surgery remains the gold standard. Treatment decision and prognosis is based mostly on feasibility of surgical resection, and it is controversial whether histological subtype affects management.
![]() Resectable disease. Surgical resection can be curative and is the treatment of choice in patients with stage I, II, and III disease. This includes even patients with tumor thrombus involving the inferior vena cava (IVC). Patients with a solitary metastasis should also be considered for resection of primary renal mass and site of metastasis. Either radical nephrectomy or partial nephrectomy are performed depending on the size of mass and the patient’s reliance on the affected kidney for total renal function.
Resectable disease. Surgical resection can be curative and is the treatment of choice in patients with stage I, II, and III disease. This includes even patients with tumor thrombus involving the inferior vena cava (IVC). Patients with a solitary metastasis should also be considered for resection of primary renal mass and site of metastasis. Either radical nephrectomy or partial nephrectomy are performed depending on the size of mass and the patient’s reliance on the affected kidney for total renal function.
![]() Adjuvant therapy. There is no role for either chemotherapy or radiation in the adjuvant setting currently, even though a portion of these patients will have recurrence of disease.
Adjuvant therapy. There is no role for either chemotherapy or radiation in the adjuvant setting currently, even though a portion of these patients will have recurrence of disease.
![]() After surgical resection, patients should be followed with repeat imaging for evidence of recurrence. A repeat surgical resection can be considered in suitable patients with recurrence.
After surgical resection, patients should be followed with repeat imaging for evidence of recurrence. A repeat surgical resection can be considered in suitable patients with recurrence.
![]() Unresectable disease. Until 2005, only high dose interleukin-2 had been approved by the FDA for treatment of advanced RCC. Although toxic, this treatment was effective in a very small minority of patients. Studies on patho-physiology of RCC have now revealed reliance of a majority of the tumors on von Hipple–Lindau protein inactivation and subsequent activation of hypoxia-induced vascular endothelial growth factor (VEGF) overexpression for tumor angiogenesis. Two oral, small-molecule kinase inhibitors, sunitinib and sorafenib, which inhibit VEGF, are now approved on two phase III trials.12,13 Both drugs were found to improve progression-free survival in patients with metastatic clear-cell renal carcinoma. Other treatments based on the same rationale include bevacizumab (anti-VEGF antibody)14 and temsirolimus (an inhibitor of the mammalian target of rapamycin [mTOR])15 can be used. Both drugs improved survival in patients with metastatic RCC and are currently approved by the FDA for metastatic RCC.
Unresectable disease. Until 2005, only high dose interleukin-2 had been approved by the FDA for treatment of advanced RCC. Although toxic, this treatment was effective in a very small minority of patients. Studies on patho-physiology of RCC have now revealed reliance of a majority of the tumors on von Hipple–Lindau protein inactivation and subsequent activation of hypoxia-induced vascular endothelial growth factor (VEGF) overexpression for tumor angiogenesis. Two oral, small-molecule kinase inhibitors, sunitinib and sorafenib, which inhibit VEGF, are now approved on two phase III trials.12,13 Both drugs were found to improve progression-free survival in patients with metastatic clear-cell renal carcinoma. Other treatments based on the same rationale include bevacizumab (anti-VEGF antibody)14 and temsirolimus (an inhibitor of the mammalian target of rapamycin [mTOR])15 can be used. Both drugs improved survival in patients with metastatic RCC and are currently approved by the FDA for metastatic RCC.
TESTICULAR CANCER
GENERAL PRINCIPLES
Due to remarkable advances in treatment, testicular cancer is now one of more curable solid organ cancers. This is especially important since it affects a much younger population than most neoplasms. Five-year survival rate is now over 95%.
Classification
Testicular tumors are divided into seminoma and nonseminoma tumors. Thenonseminoma tumors include embryonal carcinomas, teratomas, choriocarcinomas, and mixed germ cell types. Leydig, granulosa, and Sertoli cell tumors occur rarely. Nonseminomas are clinically more aggressive tumors. If elements of both seminoma and nonseminoma are found on biopsy, management follows that of nonseminoma, which is the more aggressive tumor type.
Epidemiology
As per the American Cancer Society, ~8500 new cases of testicular cancer were diagnosed in 2010 with 350 dying of the disease. It remains the most common cancer among men aged 15 to 35 years.
Risk Factors
Although it can affect men of any age, 9 out of 10 cases are in men between 20 and 54 years. White men have five times the risk of black men and three times the risk of Asian Americans for unclear reasons. Other risk factors include cryptorchidism, Klinefelter syndrome, and family history.
DIAGNOSIS
Clinical Presentation
A painless testicular mass is the classic presenting symptom. Testicular pain or swelling can be present, but typically suggests epididymitis or orchitis, and an initial course of antibiotics may be appropriate in this situation. Gynecomastia as the first sign of testicular cancer is seen is ~10% of patients. It is due to tumor production of human chorionic gonadotropin (hCG).
Physical Exam
A thorough physical exam is essential in patients with suspected testicular cancer. Exam should focus on the testicles, lymphadenopathy (particularly supraclavicular), scrotal edema, and evaluation for gynecomastia. Early metastases to bone are rare but possible, and back pain can also result from bulky retroperitoneal lymphadenopathy.
Laboratories
Three serum markers have roles in testicular cancer: alpha fetoprotein (AFP), β-hCG, and lactate dehydrogenase (LDH). These tumor markers are useful for diagnosis, prognosis, and assessment of treatment. Although nondiagnostic by themselves, it is important to know that pure seminomas do not produce AFP, and β-hCG is elevated in only 15% to 20% of these cancers. By contrast, AFP and/or β-hCG are elevated in more than 80% of non-seminomas.
Imaging
Testicular ultrasound is the initial test of choice in suspected testicular cancer. Subsequent tests include a chest x-ray to rule out pulmonary metastasis and abdominal and pelvic CT exam with oral and intravenous contrast to assess nodal enlargement and staging. Chest CT should be included if any retroperitoneal lymph-adenopathy is present or if there is high suspicion of pulmonary metastasis. Other imaging options, such as bone scan or brain imaging, are guided by the patient’s symptoms.
Diagnostic Procedures
Biopsy of a testicular mass should be avoided due to a concern for seeding and dissemination of an otherwise curable cancer. A radical inguinal orchiectomy is therefore recommended when workup, including imaging and labs, indicates testicular cancer. Trans-scrotal orchiectomy has an increased risk of seeding. In addition, retroperitoneal lymph node dissection is frequently used in patients with low-stage nonseminomas for staging and curative purposes, and to remove remaining viable tumor tissue in the lymph nodes.
TREATMENT
Treatment is based on histology type and stage. In general, stage I disease is localized to the testis, stage II disease has spread to retroperitoneal lymph nodes, and stage III disease is metastatic or has spread to non-regional lymph nodes. Prognosis is generally very favorable with >90% of patients being cured with therapy, including 70% to 80% of patients with advanced tumors. Sperm banking should be discussed before initiation of any therapy.
![]() Seminomas are very radiation and chemo sensitive. Stage I tumors are treated with either adjuvant radiation therapy or single-agent carboplatin for one to two cycles. Active surveillance is also an acceptable option in motivated patients due to low risk of spread. Stage II seminomas are treated with radiation therapy in non-bulky disease and cisplatin-based combination chemotherapy, such as BEP (bleomycin, etoposide, cisplatin) in bulky disease. Stage III seminomas are treated similarly to bulky stage II with chemotherapy.
Seminomas are very radiation and chemo sensitive. Stage I tumors are treated with either adjuvant radiation therapy or single-agent carboplatin for one to two cycles. Active surveillance is also an acceptable option in motivated patients due to low risk of spread. Stage II seminomas are treated with radiation therapy in non-bulky disease and cisplatin-based combination chemotherapy, such as BEP (bleomycin, etoposide, cisplatin) in bulky disease. Stage III seminomas are treated similarly to bulky stage II with chemotherapy.
![]() Nonseminomas are less radiosensitive, and patients often require additional surgical therapies following orchiectomy. Most of these patients will undergo retroperitoneal lymph node dissection for either diagnostic or therapeutic purposes. The major morbidity of this surgery is retrograde ejaculation with resulting infertility, which occurs is ~10% of cases using an open nerve dissection technique. Adjuvant chemotherapy is often recommended if the surgical resection reveals lymph node involvement. Etoposide and cisplatin ± bleomycin is the chemotherapeutic regimen of choice.
Nonseminomas are less radiosensitive, and patients often require additional surgical therapies following orchiectomy. Most of these patients will undergo retroperitoneal lymph node dissection for either diagnostic or therapeutic purposes. The major morbidity of this surgery is retrograde ejaculation with resulting infertility, which occurs is ~10% of cases using an open nerve dissection technique. Adjuvant chemotherapy is often recommended if the surgical resection reveals lymph node involvement. Etoposide and cisplatin ± bleomycin is the chemotherapeutic regimen of choice.
MONITORING/FOLLOW-UP
Patients with testicular cancer should have close follow-up after diagnosis and treatment. This includes serial chest x-rays, CT scanning of the abdomen and pelvis, and blood work for relevant tumor markers, in addition to a detailed clinical exam. The National Comprehensive Cancer Network recommends tumor markers and chest x-ray monthly for the first year and every 2 months for the second year, and specific guidelines for follow-up is found on its Web site (www.nccn.org). It also recommends abdominal and pelvic CT every 3 months in the first 2 years. PET scan may have a role in surveillance, but this has yet to be fully determined. Screening for late effects of platinum-based chemotherapy, most commonly dyslipidemias, cardiovascular disease, and cerebrovascular diseases, should be included in follow-up as well.
BLADDER CANCER
GENERAL PRINCIPLES
Bladder cancer is primarily a malignancy of the epithelium. Urothelial (transitional cell) carcinomas are the most common histological subtype and account for >97% of bladder tumors. Other histologic subtypes include squamous cell carcinoma, adenocarcinoma, and small-cell tumor.
Epidemiology
The American Cancer Society estimates that over 70,000 new cases of bladder cancer were diagnosed in 2010 with 14,680 deaths. Similar to prostate cancer, the median age at presentation is late in life (65 years). Medical comorbidities and life expectancy play key roles in management decisions.
Risk Factors
Bladder cancer affects more men than women almost threefold. The most important risk factor is cigarette smoking, with smokers twice as likely to get bladder cancer as nonsmokers. Other known risk factors include occupational exposure to aromatic amines in the dye industry, and work in certain other industries such as rubber, leather, textile, paint, printing, and hairdressing industries. Although uncommon in the United States, squamous cell carcinoma is a prevalent subtype in areas endemic for Schistosoma haematobium.
DIAGNOSIS
Clinical Presentation
Symptoms are not always appreciated by patients, but hematuria is present is ~90% of individuals with bladder cancer. This may be intermittent or constant, frank or microscopic, and is occasionally associated with symptoms of urinary frequency or urgency. Otherwise unexplained hematuria in an individual older than 40 years denotes bladder or renal cancer unless proven otherwise.
Diagnostic Testing
Laboratories
Urinalysis, including microscopic and gross examination, should be performed to detect presence and degree of hematuria. Urine cytology has low sensitivity, but it is usually sent as noninvasive adjunctive test that may yield the diagnosis.
Imaging
Imaging studies are performed to determine extent of disease involvement usually after bladder cancer is already confirmed by cystoscopy. Abdominal and pelvic CT, with and without IV contrast, is the test of choice. MRI is also efficacious but is more expensive. Ultrasound has no role in evaluation of bladder cancer. The role of PET is being studied.
Diagnostic Procedures
Cystoscopy is the diagnostic test of choice for evaluation and initial staging of suspected bladder cancer. If a suspicious lesion is found, cystoscopy is usually repeated underanesthesiatoobtainbiopsy.
TREATMENT
For purposes of treatment, bladder cancer is divided into three categories: superficial, muscle invasive, or metastatic.
![]() Superficial bladder cancer. Up to 75% of bladder cancers are superficial at presentation. Most can be completely resected by transurethral resection of the bladder tumor (TURBT). Most patients with superficial bladder cancer will have recurrence within 5 years, even with seemingly complete initial resection. Therefore, it is recommended that patients undergo cystoscopy every 3 months after initial resection and, depending on level of progression, be considered for intravesical therapy. Bacillus Calmette-Guérin (BCG) is the most common and effective intravesicular agent used. Other agents, including mitomycin, inter-feron, and anthracyclins, are also used. None has been shown to be superior to BCG.
Superficial bladder cancer. Up to 75% of bladder cancers are superficial at presentation. Most can be completely resected by transurethral resection of the bladder tumor (TURBT). Most patients with superficial bladder cancer will have recurrence within 5 years, even with seemingly complete initial resection. Therefore, it is recommended that patients undergo cystoscopy every 3 months after initial resection and, depending on level of progression, be considered for intravesical therapy. Bacillus Calmette-Guérin (BCG) is the most common and effective intravesicular agent used. Other agents, including mitomycin, inter-feron, and anthracyclins, are also used. None has been shown to be superior to BCG.
![]() Muscle invasive bladder cancer. The standard treatment is radical cystectomy with pelvic node dissection. In males this also includes removal of prostate and seminal vesicles, and in females the uterus, ovaries, and fallopian tubes. Neoadjuvant chemotherapy has been found to improve outcome in patients with muscle invasive bladder cancer. MVAC (methotrexate, vinblastin, doxorubicin, cisplatin) is one of the commonly used regimens, as a trial demonstrated a doubling of median survival with this approach.16 A cisplatin-based adjuvant chemotherapy regimen can be used in patients who did not receive neoadjuvant therapy.
Muscle invasive bladder cancer. The standard treatment is radical cystectomy with pelvic node dissection. In males this also includes removal of prostate and seminal vesicles, and in females the uterus, ovaries, and fallopian tubes. Neoadjuvant chemotherapy has been found to improve outcome in patients with muscle invasive bladder cancer. MVAC (methotrexate, vinblastin, doxorubicin, cisplatin) is one of the commonly used regimens, as a trial demonstrated a doubling of median survival with this approach.16 A cisplatin-based adjuvant chemotherapy regimen can be used in patients who did not receive neoadjuvant therapy.
![]() Metastatic bladder cancer. Cisplatin-based combination therapy is the treatment of choice. MVAC is the most commonly used regimen. Sometimes TURBT or radiation is used palliatively for symptoms of obstruction. Unfortunately, median survival remains in the range of 12 months.
Metastatic bladder cancer. Cisplatin-based combination therapy is the treatment of choice. MVAC is the most commonly used regimen. Sometimes TURBT or radiation is used palliatively for symptoms of obstruction. Unfortunately, median survival remains in the range of 12 months.
REFERENCES
1. Thompson IM, Goodman PJ, Tangen CM, et al. The influence of finasteride on the development of prostate cancer. N Engl J Med. 2003;349:215–224.
2. Andriole GL, Bostwick DG, Brawley OW, et al. Effect of dutasteride on the risk of prostate cancer. N Engl J Med. 2010;362:1192–1202.
3. Andriole GL, Crawford ED, Grubb RL III, et al. Mortality results from a randomized prostate-cancer screening trial. N Engl J Med. 2009;360:1310–1319.
4. Schröder FH, Hugosson J, Roobol MJ, et al. Screening and prostate-cancer mortality in a randomized European study. N Engl J Med. 2009;360:1320–1328.
5. Catalona WJ, Richie JP, Ahmann FR, et al. Comparison of digital rectal examination and serum prostate specific antigen in the early detection of prostate cancer: results of a multi-center clinical trial of 6,630 men. J Urol.1994;151:1283–1290.
6. Brawer MK. Prostate-specific antigen: current status. Cancer J Clin. 1999;49:264–281.
7. Bolla M, deReijke TM, Tienhoven GV, et al. Duration of androgen suppression in the treatment of prostate cancer. N Engl J Med. 2009;360:2516–2527.
8. Petrylak DP, Tangen CM, Hussain MH, et al. Docetaxel and estramustine compared with mitoxantrone and prednisone for advanced refractory prostate cancer. N Engl J Med. 2004; 351:1513–1520.
9. Tannock IF, de Wit R, Berry WR, et al. Docetaxel plus prednisone or mitoxantrone plus prednisone for advanced prostate cancer. N Engl J Med. 2004;351:1502–1512.
10. Skinner DG, Colvin RB, Vermillion CD, et al. Diagnosis and management of renal cell carcinoma. A clinical and pathologic study of 309 cases. Cancer. 1971;28:1165–1177.
11. Pantuck AJ, Zisman A, Belldegrun AS. The changing natural history of renal cell carcinoma. J Urol. 2001;166:1611–1623.
12. Motzer RJ, Hutson TE, Tomczak P, et al. Sunitinib versus interferon alfa in metastatic renal-cell carcinoma. N Engl J Med. 2007;356:115–124.
13. Escudier B, Eisen T, Stadler WM, et al. Sorafenib in advanced clear-cell renal-cell carcinoma. N Engl J Med. 2007;356:125–134.
14. Escudier B, Bellmunt J, Négrier S, et al. Phase III trial of bevacizumab plus interferon alfa-2a in patients with metastatic renal cell carcinoma (AVOREN): final analysis of overall survival. J Clin Oncol. 2010;28:2144–2150.
15. Hudes G, Carducci M, Tomczak P, et al. Temsirolimus, interferon alfa, or both for advanced renal-cell carcinoma. N Engl J Med. 2007;356:2271–2281.
16. Grossman HB, Natale RB, Tangen CM, et al. Neoadjuvant chemo therapy plus cystectomy compared with cystectomy alone for locally advanced bladder cancer. N Engl J Med. 2003; 349:859–866.