ANTHONY C. WANG, KHOI DUC THAN, AND OREN SAGHER
Neurosurgeons play an important role in the diagnosis and treatment of central nervous system (CNS) infections. Before the introduction of antibiotics, the treatment of CNS infections was the exclusive realm of the surgeon, as most treatable infections were abscesses. With the development of antibiotics and increasingly sophisticated noninvasive diagnostic methods, the spectrum of treatable CNS infections has expanded rapidly, and the role of the neurosurgeon has changed. Medical intervention has become the treatment mainstay for most CNS infections. Diagnosis of most infectious processes can now be made on the basis of radiologic appearance, serologic markers, and specific culturing techniques. Nevertheless, a number of CNS infections continue to mandate neurosurgical intervention for diagnosis and/or management. In this chapter, we discuss the neurosurgical aspects of the management of CNS infections. The pathophysiology, microbiology, and medical management of these infections are discussed in other chapters, and this effort is not duplicated in this chapter. Rather, specific attention is paid to issues relevant to neurosurgical intervention.
HISTORY
Long before the discovery of microbes, the introduction of antisera, or the development of antibiotics, infections were routinely managed by various surgical procedures. One of the first descriptions of the surgical treatment of abscesses is found in the Edwin Smith Papyrus (roughly 2500 BC) (1); surgical drainage has continued to be a mainstay of abscess treatment to this day.
It is unclear when the general practice of surgical abscess drainage was applied to cranial infections. Possibly trephination of the skull, a practice dating back to antiquity, was performed in some cases for drainage of cranial abscess (2). However, the first description of surgical treatment for a CNS infection does not appear until the seventeenth century. Prince Rupert of the Rhine Palatinate, a nephew of King Charles I of England, underwent trephination of the skull for drainage of an epidural abscess in 1667 (3). This procedure was apparently successful, and the Prince lived for 27 additional years. The first successful treatment of an intracerebral abscess was recorded by the French surgeon, Morand, in 1772; following the opening of a mastoid abscess, he opened the dura and resected the capsule of an intracerebral abscess with his finger (4). More than a century later, the Scottish surgeon, Sir William Macewen, reported on a series of patients with a variety of CNS infections, including meningitis, epidural abscess, and brain abscess (5). Macewen, considered one of the founders of modern neurosurgery, made many important observations on the pathogenesis and natural history of CNS infections. His operative technique for the treatment of brain abscesses consisted of drainage of the abscess cavity, gentle irrigation with a dilute antiseptic solution (boric acid or phenol), and placement of an external drain. Without the aid of antibiotics, computed tomography (CT), or modern anesthetic techniques, he reported an astounding 95% success rate for surgical drainage of brain abscesses and a 100% success rate for epidural abscesses so treated. His results have served as a benchmark for other treatments to this day.
BACTERIAL BRAIN ABSCESS
A brain abscess is a focal suppurative process affecting the brain parenchyma and is the most common CNS infection requiring neurosurgical intervention. Although the incidence of brain abscesses overall is generally quite low (0.4 to 1.1 per 100,000 population per year) (6,7), they comprise up to 8% of all intracranial lesions in some parts of the world (8), approximately 1,500 to 2,500 cases arising yearly in the United States (9).
Pathogenesis
Brain abscesses form as a result of either contiguous invasion of infection or hematogenous spread from a distant infection. Their most common etiology is local transmission from the ear, nose, or an adjacent air sinus, accounting for 40% to 50% of all cases (10). Vigilance has improved worldwide, and rates of abscess formation complicating otitis have begun to fall (11). Hematogenous spread of a distant infection typically arises from the lungs or heart valves and is slightly less common, accounting for approximately one third of cases (12). In children, meningitis is more likely to incite the development of brain abscess than in adults (13). Because contiguous spread frequently leads to the formation of a single, often large brain abscess (14), most patients present with symptoms of mass effect. Multiple small abscesses are more frequently the result of hematogenous spread of distant infection.
Predisposing Factors
The relationship between chronic ear or paranasal sinus infection and the development of brain abscess is well documented (15–19), as are cases of cardiovascular anomalies (20–23). Source control and treatment of predisposing factors are as important in the management of brain abscess as the treatment of the abscess itself. Consequently, surgical drainage and débridement of infected air sinuses are important steps in the prevention of the intracranial complications of such infections, which include brain abscess as well as epidural abscess, meningitis, subdural empyema, and septic venous sinus thrombosis.
Pathology
The development of brain abscess following an infectious event proceeds through several well-defined stages (24). Acute cerebritis at the site of organism inoculation is followed by the development of a collagenous capsule as the host attempts to contain and eradicate the infection. The capsule that forms around the localized region of cerebritis is not of uniform thickness. Evidence from animal studies (25) as well as clinical data (24,26) suggest that capsule thickness is maximal at the superficial aspect of the abscess and minimal or incomplete at its deep margin. This asymmetric finding likely correlates with the tendency of untreated abscesses to migrate toward the ventricle (25).
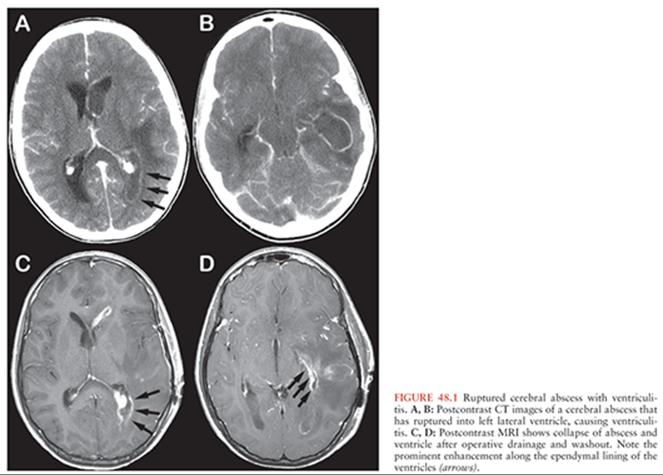
Thus, an untreated abscess is likely to eventually rupture its contents into the ventricular system (Fig. 48.1), a potentially catastrophic complication, having a mortality rate of up to 90% (15,27–29). Aggressive medical management demonstrates some efficacy in this setting, lowering mortality to approximately 40% (30). Nevertheless, prompt administration of antibiotics and consideration of surgical drainage are extremely important for those abscesses located in the periventricular region, as early intervention may prevent intraventricular rupture. In addition, care should be taken during surgical treatment of a periventricular abscess in order to avoid spillage of purulent material into the ventricular system.
The abscess capsule serves to contain the infectious process. In isolating the infectious process, however, capsule formation also creates within it an inflammatory fluid collection poorly permeable to host defenses and antibiotic penetration (31). A large abscess can also reduce regional blood flow by exerting mechanical pressure on the surrounding brain, thereby compromising host defenses. In addition, some organisms produce collagenase or hyaluronidase, which may lead to disruption of the capsule and further spread of the infection (32). Finally, the chemical environment within an abscess may inhibit the action of systemic antibiotics, even if antibiotic levels are sufficient within the abscess cavity (33). Abscess formation, therefore, restricts the infectious process in the brain but may impede its resolution.
Microbiology
Brain abscesses are most commonly formed by multiple aerobic and anaerobic bacteria and typically reflect the bacterial profiles of the source infections from which they are derived. Isolation of organisms from the blood may be indicative of the bacteria within a brain abscess propagated by hematogenous spread, but in the more common ear- or sinus-derived brain abscesses, blood cultures are often negative or misleading. Cerebrospinal fluid (CSF) cultures obtained by lumbar puncture may be positive in 10% to 20% of brain abscesses, but the potential for exacerbating cerebral herniation in the setting of intracranial hypertension precludes this from being a universal diagnostic option (34). Sinus cultures may give a more reliable indication of adjacent brain abscess bacteriology and can be used to guide antibiotic treatment should direct aspiration of the brain abscess be undesirable. With the advent of less invasive neurosurgical techniques and accompanying lower morbidity, direct aspiration of the abscess must be considered a first step in determining the microbiology of the lesion.
Clinical Features
The possibility of a brain abscess should be considered in any patient who presents with signs or symptoms of an intracranial mass lesion. Because the infectious process is usually localized and encapsulated at presentation, symptoms are usually a result of the expanding mass within the brain. Consequently, patients typically present with headaches (70% to 97%), focal neurologic deficits (50% to 60%), depressed sensorium (50% to 65%), and nausea and vomiting (50%) (17,18,28,35). Meningeal signs are found in 25% of cases and may indicate concurrent meningitis. Systemic signs such as fever are highly unreliable and may be absent in up to 55% of cases (15,17,18,27). The tempo of symptom progression is highly variable and may occur in a stepwise manner (36). Therefore, vigilance is still the most important tool in the diagnostic armamentarium of the physician.
Radiology of Brain Abscesses
The typical description of a bacterial brain abscess on CT or magnetic resonance imaging (MRI) is that of a circumscribed lesion that restricts diffusion, is encapsulated by a uniformly contrast-enhancing rim, and is accompanied by surrounding vasogenic edema (Fig. 48.2). The appearance of the enhancing rim, however, is variable in that it is often asymmetric (classically thinner on the deep surface nearest the ventricle) and may be altered by incomplete encapsulation. The use of corticosteroids may delay capsule formation and suppress contrast enhancement. Abscesses with a somewhat more heterogeneous or nodular rim can be mistaken for neoplasms. The distinction between cerebritis and encapsulated abscess may also be difficult to make, even with advanced imaging. During the cerebritis stage of abscess formation, ring enhancement may be absent or incomplete. The extent of surrounding edema is highly variable and is not a reliable radiologic feature. Uncommonly, brain abscesses may spontaneously bleed, presenting as an intracerebral hemorrhage (37).

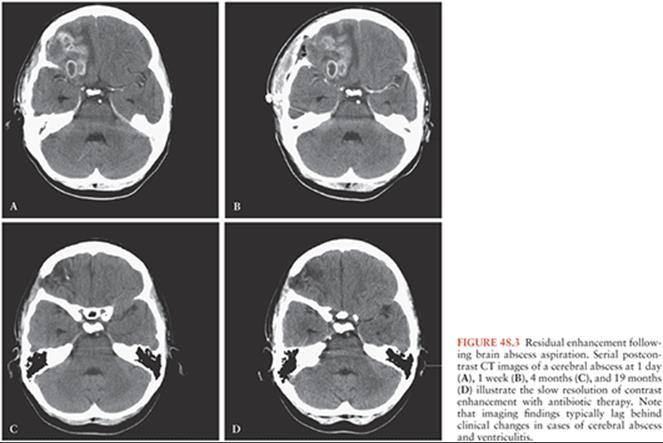
Surgery of brain abscess has been revolutionized by the development of cross-sectional imaging modalities. CT and MRI have allowed the surgeon not only to assess the size and location of an abscess but also to gain information on its stage of maturity, the existence of other abscesses, and the presence of loculation within the lesion. Although serial imaging provides detailed information on the response to treatment, imaging findings typically lag behind clinical response to treatment, so some patients will exhibit persistent brain edema and contrast enhancement following aspiration of a brain abscess. For example, Rosenblum et al. (38) reported several cases that underwent medical treatment only and found that the size of an abscess may not change for more than 2 weeks after the institution of eventually successful therapy. On average, it took 10 weeks before the abscesses resolved on CT. Abnormal enhancement may persist for months or years following the successful treatment of brain abscess (Fig. 48.3) (39,40), reflecting mild breakdown of the blood–brain barrier and not residual infection. Serial imaging, therefore, is a useful tool in the management of brain abscess, although it is prone to lag behind clinical improvement and cannot be relied on as the sole measure of treatment efficacy.
MRI has become the preferred modality in the assessment of brain abscess. The sensitivity of MRI is superior to that of CT in detecting small or multiple abscesses. MRI scanning renders superior images of the posterior fossa and may be better than CT at disclosing abscesses in this and other locations (Fig. 48.4). In addition, MRI is more sensitive in distinguishing cerebritis from necrosis (41). Diffusion-weighted imaging can aid in distinguishing abscesses from neoplasms (42,43), and MR spectroscopy has been used to differentiate likely bacterial etiologies of abscesses (44). Thus, MRI is the preferred imaging modality in the assessment of intracranial infections.

Surgical Therapy
Surgical treatment of brain abscess is guided by its clinical presentation. For example, multiple small abscesses—which are often metastatic—may not present with signs of focal brain compression. Surgical intervention in such cases is more a matter of diagnosis than treatment. In these cases, surgical drainage is aimed at the most accessible lesion in order to reduce operative morbidity. Large abscesses producing focal neurologic deficit are treated surgically regardless of their location (45). Surgical treatment is required from both therapeutic and diagnostic standpoints in the latter instance. In either situation, however, surgical intervention is a matter of some urgency and should ideally be performed before administration of antibiotics, although the diagnostic yield from samples obtained on antibiotics is adequate to justify cultures obtained within 3 days of initiation of antibiotics (46).
The location of a brain abscess also will direct the need for any secondary means of treatment. Lesions located in the cerebellum, for example, may compress CSF pathways and thereby lead to dangerously high intracranial pressure. These lesions may carry a mortality rate of 20% to 50% (47), even with the advent of intracranial imaging and appropriate surgical drainage. The current recommendations are to drain these abscesses through a posterior fossa craniotomy as well as perform CSF diversion through an external ventricular drain in patients with any radiologic signs of hydrocephalus. A study of 77 patients treated with or without CSF diversion demonstrated a reduction in mortality from 29% to 11% in patients who received a prophylactic CSF diversion procedure at the time of admission, even when these patients did not yet have clinical signs of hydrocephalus (47).
Mechanical drainage of an abscess improves the effectiveness of antibiotic therapy. Several studies have indicated that the contents of a brain abscess confer resistance to antibiotics (31,33,48). Smith and Wood (31) demonstrated that phagocytes within mature abscesses were ineffective despite adequate antibiotic levels. In 1973, Black et al. (33) aspirated brain abscesses in patients undergoing systemic antibiotic treatment and showed that several antibiotics were unable to inhibit organism growth despite adequate levels. A more recent study suggested that intracavitary antibiotic levels were higher in those abscesses that underwent prior drainage (49). Surgical drainage of brain abscess, therefore, plays a crucial role in both its diagnosis and treatment.
Over the last two centuries, a number of surgical techniques have been devised to treat brain abscess. These techniques range from the tube-drainage methods of the past to modern image-guided, minimally invasive approaches. For purposes of this discussion, the surgical techniques for abscess treatment are described in a historical context.
Tube Drainage
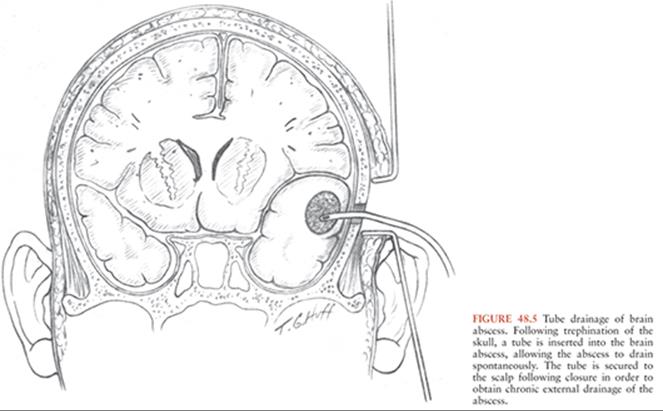
Tube drainage is the oldest known surgical treatment for brain abscess. The technique involves entry into the abscess cavity with a needle, followed by placement of a tube into the cavity for postoperative drainage (Fig. 48.5). Tube drainage of brain abscess offers the advantage of allowing spontaneous drainage of the abscess contents and decompression of the surrounding brain. This technique also minimizes trauma to the brain. However, tube drainage is often complicated by obstruction of the tube and occasional perforation of the deep wall of the abscess by the tube, and the reported mortality with this technique is high (30% to 45%) (50–52). Tube drainage is now employed much less frequently, except when used in conjunction with more recent aspiration methods.
Marsupialization
Another early method of brain abscess treatment was marsupialization of the abscess. Described in 1924 by King (53), this technique involved creating a broad opening of the cortex over the abscess and packing the cavity with gauze (Fig. 48.6). The gauze was then gradually removed as the infection resolved. The purported advantages of this method were that it prevented the accumulation of inspissated debris and the formation of secondary pockets. This method was used by a number of neurosurgeons and reported in various small series (a total of 123 patients in 13 series reviewed) with an average mortality rate of 23% (54). The technique has not gained wide use, as it involves the sacrifice of potentially functional brain regions overlying the abscess cavity and a persistent CSF leak until the abscess cavity is clean and closed.

Migration Technique
One particularly innovative approach to the treatment of brain abscess is the “migration technique” described by Kahn in 1937 (55). He noted that when a craniectomy was performed for the relief of increased intracranial pressure, brain abscesses tended to migrate toward the cranial opening. Based on this observation, he devised a two-stage technique composed of a decompressive craniectomy followed by a second procedure 3 to 4 days later for either excision or drainage of the abscess (Fig. 48.7). Kahn (56) also injected radiopaque dye into abscess cavities in order to follow their progress by skull x-rays. The migration technique is of historic interest and has not been reported by other neurosurgeons since its initial description. Of note, a similar technique was also commonly used for spinal cord ependymomas.


Excision
The two surgical techniques that continue to be used routinely in the treatment of brain abscess are excision and aspiration. Excision, the older of the two techniques, was first described by Krönlein in 1910 in a case where an abscess was mistaken for a brain tumor (57). The total extirpation of abscesses subsequently gained considerable popularity, with over 1,500 patients reported in 48 series (54). The excision of brain abscess relies on the existence of a collagenous capsule around the mature cavity and takes advantage of the clear plane of dissection that this capsule provides (Fig. 48.8). The main advantage of this method is that it achieves complete removal of the infectious process and immediate relief from the mass effect the abscess exerts.

In the 80 years since its initial description, several prerequisites for excision of an abscess have emerged. First, the abscess should reside in a surgically accessible region. Second, the lesion should be excised in the chronic stage, so that a well-developed capsule separates the abscess from the surrounding brain. Third, the integrity of the capsule should be maintained in order to avoid spillage of its contents and possible spread of the infection. Last, intraoperative entry into the ventricular system should be avoided. Many proponents of total excision have reported zero mortality, but a comprehensive literature review suggests that the mortality of this technique is near 20% (54). Fungal abscesses, which tend to be more invasive and less encapsulated than their bacterial counterparts, usually do not lend themselves readily to surgical excision. These abscesses also carry a much worse prognosis, with high mortality rates (58).
Aspiration
Aspiration of a brain abscess was described by Mayfield and Spurling in 1937 (59), but the treatment has its roots in the older practice of simple tapping, which was advocated by Walter Dandy in 1926 (60). The guiding principle of abscess aspiration is that simple evacuation and decompression of abscess contents is sufficient, with antimicrobial therapy, to allow natural resolution of the infectious process. Aspiration can usually be performed via a burr-hole opening, reducing trauma to the surrounding brain (Fig. 48.9). The main advantages of this technique are that it can be performed with relatively low morbidity and that lesions in eloquent brain or deep lesions can be drained. Its principal disadvantage is that it does not remove the infection but simply decompresses the cavity. Therefore, abscess reaccumulation is a regular occurrence following aspiration, and repeat aspiration or excision must be performed in 50% to 70% of cases (28,61).

Loculations within abscesses also present a potential difficulty, as adequate decompression via needle aspiration may prove impossible in these cases. Brain abscesses caused by Nocardia asteroides are particularly likely to be multiloculated and may not be successfully treated by aspiration; one recent series suggests that, in the case of Nocardia, craniotomy with abscess excision must be performed to effect a cure (62). Another potential complication of abscess aspiration relates to disruption of the capsule by the aspiration needle, which may result in parenchymal hemorrhage or leakage of abscess contents (63–65). Despite these potential problems, aspiration of abscesses remains the treatment of choice in cases of multiple abscesses, in cases of deep-seated lesions, or in abscesses located within eloquent brain.
The aspiration of brain abscesses gained considerable popularity before the development of CT scanning or stereotactic techniques, but the technique was traditionally performed on patients who were more seriously ill than those offered excision. Consequently, the mortality of abscess aspiration before the introduction of CT was approximately 45% (302 deaths in 670 patients from 31 series), compared to a mortality of 19% with primary excision in the same time period (259 deaths in 1,333 patients from 43 series) (54). Since 1990, the mortality of abscess aspiration is 6.6% compared to 12.7% for primary excision (66). A more recent study found no difference between patients who underwent burr-hole drainage versus open craniotomy in terms of reoperation rate, hospital length of stay, duration of antibiotic treatment, and discharge and long-term functional outcome (67). This suggests that burr-hole aspiration of abscesses is an effective first-line treatment.
Image Guidance
Since the introduction of cross-sectional imaging and the refinement of stereotactic techniques, however, aspiration has become much more commonly and safely performed. Stereotaxis consists of assigning three-dimensional coordinates to any point within the brain and uses these coordinates to guide the surgical approach (68–70). Stereotactic techniques have traditionally required affixing a coordinate frame to the skull before radiologic imaging, but frameless stereotactic techniques are now prevalent (71–75). The precision of this technique, which is better than 1 to 2 mm in frame-based and frameless stereotactic systems, has led stereotactic aspiration to be used in cases of small abscesses or in abscesses within deep or eloquent brain (76–79). Mortality rates for patients treated in this fashion fell to 0% to 21% with the introduction of stereotaxis (63,64,74,77,78,80–93). One risk of stereotactic needle aspiration is the development of a hematoma that cannot be primarily controlled. This risk of stereotactic needle placement for all pathologies ranges from 1% to 4%, with an approximate mortality rate of 0.5% to 2% (94,95). The incidence of postoperative hematoma may be increased in immunocompromised patients (95–98), perhaps because more of the lesions are due to toxoplasma or lymphoma.
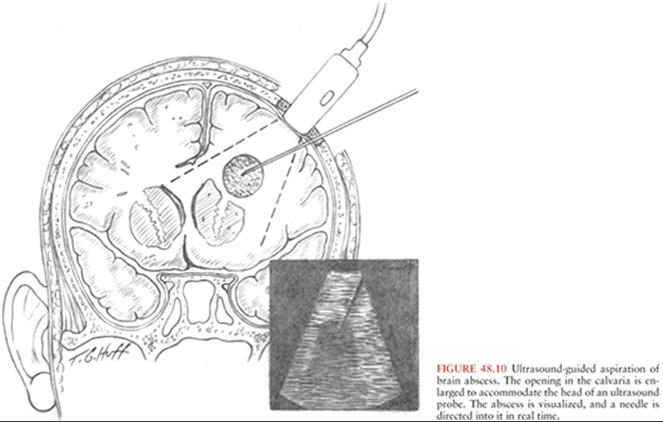
Intraoperative ultrasonography is an image-guided operative technique that does not require the use of a stereotactic instrument. Sonography allows the surgeon to view a brain abscess with excellent detail through intact dura (Fig. 48.10). The additional benefit of ultrasonography is its ability to yield images in real time, guiding the surgical approach and allowing the surgeon to adjust the planned approach as needed (99).
Antibiotic Irrigation
The use of intracavitary antibiotic irrigation has been reported in a number of series (100–102). Proponents suggest that irrigation of the abscess with antibiotic solution raises antibiotic levels and reduces the bacterial load, allowing the infectious process to resolve more quickly (102). This notion, however, remains unproven. Local administration of antibiotics is not routinely used because intravenous antibiotics used for brain abscess treatment penetrate the brain and abscess capsule, achieving therapeutic intracavitary levels (49). The placement of an indwelling intracavitary catheter is not without risk and can result in bleeding within the abscess cavity (64). One scenario in which antibiotic irrigation may prove helpful is the case of rupture of a brain abscess into the ventricle (29,103,104).
Nonsurgical Therapy
Surgery is the initial treatment of choice for most brain abscesses. Surgery confirms the diagnosis of brain abscess, allows direct identification of the causative organisms, and decompresses the abscess cavity. Nevertheless, selected patients may be treated by empirical medical therapy alone. Nonoperative management of brain abscess was initially reported by Heineman et al. in 1971 (105). In that series, six patients with abscesses were successfully treated with antibiotics alone; however, the lesions were in the cerebritis stage. The first report of an encapsulated abscess cured by medical therapy alone was published by Chow et al. in 1975 (106). In a 1986 review of nonsurgical series, Rosenblum et al. (10) found a total of 50 patients with mature abscesses so treated, with a success rate of 74% and a mortality rate of 4%. Based on their experience and review of the literature, Rosenblum et al. (38) recommended that nonoperative treatment of brain abscess be reserved for patients with known systemic or adjacent infection whose abscesses are smaller than 1.5 cm or for patients with uncontrollable bleeding diatheses. However, with the increased availability of accurate stereotactic techniques, it is now possible to approach abscesses smaller than 1.5 cm with accuracy, resulting in minimal morbidity. In a review of 16 patients with multiple brain abscesses, all patients with small abscesses underwent stereotactic aspiration for diagnostic purposes; the only death in the series occurred in a patient who suffered transtentorial herniation preoperatively (88). These encouraging results suggest that even the most conservative treatment for abscess should involve surgery to identify the organism(s) and drain the mass rather than empirical medical therapy without an accurate bacteriologic diagnosis.
PITUITARY ABSCESS
Infection of the pituitary gland is a rare but potentially fatal disease. Roughly 200 cases of pituitary abscess have been reported in the literature, with an estimated incidence of 0.38% to 0.69% (107–111). The pathogenesis of this lesion is not entirely clear. Traditionally, pituitary abscess was thought to arise by direct extension from an infected sphenoid sinus or as a complication of a sellar CSF leak (111–115). Other cases have been reported following resection of pituitary adenomas and occasionally within tumors that had not undergone surgical resection (116,117). It has also been suggested that many pituitary abscesses are not infectious at all but merely the result of necrosis within pituitary neoplasms (108).
The diagnosis of pituitary abscess is elusive and is most often made at operation for a presumed pituitary adenoma (112–114,116). Often pituitary abscess manifests itself in a manner identical to other intrasellar mass lesions, with headache, visual disturbances, and signs and symptoms of hypopituitarism. Preoperative diagnosis in such cases is particularly difficult. When pituitary abscess develops in the setting of sphenoid sinusitis, the diagnosis may be more apparent. Signs of CNS infection, such as fever and meningeal signs (114–117), are often absent in patients with pituitary abscess (107,111). Radiologic examination is often nonspecific. Plain x-rays may demonstrate sphenoid sinusitis or an eroded sella turcica, whereas cranial CT scans often show only a mass in the sellar region. In some instances, however, CT or MRI scanning may demonstrate ring-enhancing lesions consistent with abscess within the pituitary fossa or sella turcica (Fig. 48.11) and can show restricted diffusion (118). However, imaging characteristics attributed to pituitary abscess are inconsistent, and the presence of prior pituitary surgery or known pituitary tumor may complicate the diagnosis of abscess even in the face of such radiologic changes.

The treatment of pituitary abscess is a matter of some urgency. The tempo of symptom progression is unpredictable, and patients with long-standing symptoms of pituitary dysfunction may suddenly deteriorate (114,116,119). Rapid neurologic decline may be caused by endocrine failure, rupture of the abscess into the subarachnoid space, or spasm of the internal carotid artery within the cavernous sinus (120). Sudden visual loss may ensue if the abscess expands rapidly and compresses the optic pathways (112–114).
Treatment of pituitary abscess consists of antibiotics, endocrine support, and surgery. As is the case in other types of brain abscess, surgical decompression and broad-spectrum antibiotics are of primary importance in the initial management of pituitary abscess. In a series of 24 pituitary abscess cases seen at one institution, 58% had tissue Gram stains or cultures positive for a causative organism (111), whereas only 5 of 30 (17%) were positive in another series (110). Most of the offending organisms were gram-positive cocci, although Neisseria, Clostridium, Micrococcus, and Citrobacter species were also represented. In addition, cultures from some patients grew more than one organism. Because pituitary abscess commonly causes endocrine failure, hormone replacement therapy is paramount. Hormone replacement should be tailored to the needs of each patient and usually includes stress doses of hydrocortisone (e.g., 100 mg every 6 hours intravenously) (116). Close monitoring of electrolytes and fluid balance is also necessary, as some degree of diabetes insipidus is common (117,121). Hormone function is unlikely to return to normal following treatment (27%), and abscess can recur as well (110).
Surgery for pituitary abscess is similar to that for other pituitary lesions and usually consists of a transsphenoidal approach with drainage and débridement of purulent debris (107,111,116). The transsphenoidal approach allows drainage of the abscess and decompression of the optic structures with minimal morbidity. If the diagnosis of pituitary abscess is entertained preoperatively, or if the contents of the sella appear purulent intraoperatively, tissue is sent both for pathologic examination and for microbiologic analysis. Every attempt is made to avoid entry into the subarachnoid space, and some authors caution against an overly vigorous removal of the abscess capsule for fear of creating a CSF fistula (115). Drainage of the abscess may also be performed through a craniotomy, although this is less desirable, as it necessarily involves exposure of CSF to the contents of the abscess.
BACTERIAL SUBDURAL EMPYEMA
Subdural empyemas are relatively uncommon infections characterized by a purulent collection in the subdural space. The name “subdural empyema” was coined by Kubik and Adams (122) in 1943, although various other names have been associated with this type of infection, including pachymeningitis interna, circumscript meningitis, phlegmonous meningitis, and purulent pachymeningitis (123). Subdural empyemas account for approximately 10% to 20% of suppurative intracranial processes (124) and carry a mortality rate as high as 55% (125), although with advances in imaging and surgical techniques, most series now report mortality rates less than 20% (126,127). Subdural empyema may occur as a complication of meningitis or of skull fractures and neurosurgical procedures such as craniotomies and burr-hole drainage of chronic subdural hematomas (128,129). However, in most series, infections originating in the paranasal sinuses account for the majority of subdural empyemas (126,130–132).
Subdural empyema may be acute or subacute. Rapid clinical progression is characteristic of acute subdural empyema. Typically, patients present with a febrile illness followed by rapid, often catastrophic neurologic decline. Symptoms are primarily related to cortical inflammation, edema, vasculitis, and venous thrombosis (133). Headache is almost universal in conscious patients and is followed by signs of increased intracranial pressure, such as disturbances in consciousness and vomiting. Herniation can occur. Nathoo et al. (134) noted seizures in only 14.7% of patients. Subacute subdural empyemas, in contrast, often present with a history of headache and low-grade fever in the absence of profound neurologic compromise, and in fact, these patients may exhibit only minimal changes on neurologic examination (135).
Subdural empyemas spread along the subdural spaces and are not well encapsulated, as are parenchymal abscesses. Pus tends to track along the falx cerebri as well as over the cerebral convexity (Fig. 48.12); this is in contrast to sterile subdural fluid collections (i.e., hematomas, hygromas), which have a predilection for the convexity rather than the interhemispheric region. Purulent fluid collections in the subdural space can also be fairly unimpressive in size when compared to clinically significant sterile collections. CT scans may disclose abnormal enhancement of the meninges around the subdural space as well as evidence of brain edema or hemorrhage caused by cortical inflammation and venous thrombosis. Often, however, radiologic imaging belies the clinical picture. Because the mass effect of these collections tends to be more diffuse than that exerted by encapsulated parenchymal abscesses, it is not as readily apparent on CT scanning. Similarly, brain inflammation and swelling may be diffuse, resulting in an unimpressive radiologic picture. MRI with gadolinium contrast is more sensitive than CT scanning in demonstrating small subdural empyemas or enhancement around the purulent collection (136–139) (Fig. 48.13) and can also be helpful in differentiating between empyema and effusion (135). Lumbar puncture for CSF analysis and culture rarely provides useful information in patients with subdural empyemas and carries with it a risk of exacerbating herniation. In one reported series, 3 of 280 patients referred for treatment of subdural empyema died as a result of lumbar puncture, whereas another 33 sustained neurologic deterioration (127). When CSF is collected, it often shows only nonspecific, low-grade pleocytosis with a normal glucose and protein. CSF cultures are expected to be negative in cases of nonmeningitic empyema (126,135). Because of the risk of lumbar puncture in the setting of subdural empyema, patients presenting with fever, neurologic alteration, and purulent nasal or aural discharge should always undergo cranial imaging prior to lumbar puncture, even in the absence of focal neurologic abnormalities (127).
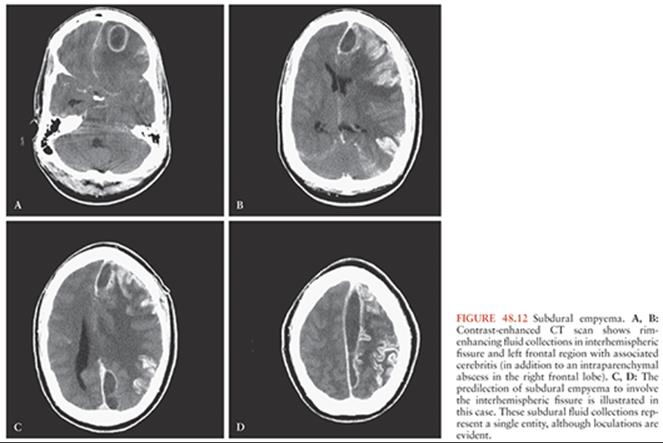

Treatment relies on a twofold approach using surgical intervention and intravenous antibiotics. Evacuation of the purulent collection within the subdural space allows culture and identification of the causative organisms, reduces the cortical irritation, and relieves the associated mass effect. In a study of 90 patients with subdural empyema, more than 65% of cultures taken intraoperatively identified causative organisms (126), including streptococci, staphylococci, and gram-negative bacilli. The prevalence of streptococci is greatest in subdural empyema arising from sinus spread (140,141). Broad-spectrum antibiotics should be started as early as possible in subdural empyema patients, with tailoring of the antibiotic regimen once organism sensitivity has been identified. The duration of antibiotic administration is open to debate, although most authors recommend a 3-week course of intravenous antibiotics followed by at least a 3-week course of oral antibiotics if suitable options exist (126,127,141).
Prompt surgical intervention plays an integral role in the successful treatment of subdural empyema. Delay in diagnosis and treatment has been shown to affect the outcome of subdural empyema adversely (132). In addition to identification of the causative organisms, evacuation of the purulent collection within the subdural space reduces cortical irritation and relieves the associated mass effect. Although little disagreement exists about the need to perform surgery on these lesions, considerable controversy exists regarding the choice of surgical approach. Both burr-hole drainage and craniotomy have been advocated for the treatment of subdural empyemas (126,131,140–143). Advocates of burr-hole drainage point out that CT scanning allows the precise placement of burr holes and adequate removal of the purulent collection without the complications of a craniotomy in a swollen, irritated brain (Fig. 48.14). In addition, the potential for infection of the cranial bone flap may necessitate not replacing it after craniotomy, resulting in a cranial defect until the infection resolves. On the other hand, proponents of craniotomy suggest that burr-hole evacuation, with or without catheter lavage, may not adequately drain collections that may be loculated, thick, and tenacious. Moreover, with the burr-hole approach, there may be a greater need for multiple operations. In 1993, Bok and Peter (126) analyzed a series of 90 patients, most of whom underwent burr-hole evacuation of subdural empyemas. The mortality rate was 4% following burr-hole evacuation and 5% after craniotomy or burr holes with concurrent craniectomy. They concluded that given the propensity for these cases to be performed by junior staff under acute conditions, CT-guided burr-hole drainage was the easiest and safest choice for management. In a series of 65 patients, Banerjee et al. (144) found a 23% rate of reoperation required after burr-hole evacuation compared with 10% after craniotomy. Nathoo et al. (127) retrospectively analyzed a series of 699 patients treated over the period 1983 to 1997. The mortality rate was significantly higher for patients who received no surgery (28%) or burr holes only (23.3%) compared to those who received burr holes with craniectomy (11.5%) or craniotomy alone (8.4%). They concluded that craniotomy was the treatment of choice in most patients, with burr-hole drainage reserved only for infants with meningitis-related subdural empyema. It should be noted that the retrospective nature of this study placed it at risk of selection bias, and detailed preoperative comparison between the groups was not undertaken. It is also unclear how the choice of surgery was undertaken on a case-by-case basis.
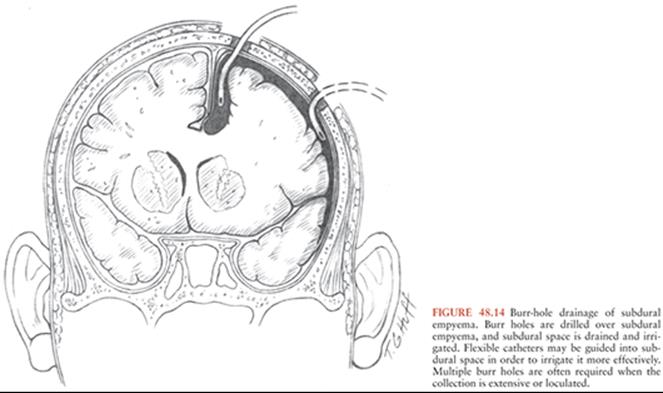
Direct comparison of the two techniques is fraught with difficulties, as the less aggressive burr-hole surgery is often performed on sicker patients and therefore may appear to carry a higher morbidity. Overall, mortality associated with burr holes is generally comparable to the mortality with craniectomy (Table 48.1). The choice of procedure often reflects the preferences of individual centers rather than a prospective analysis of outcomes.

The role of intraoperative placement of drainage catheters is likewise unknown; when used, however, the operative surgeon must be aware that the swollen brain is friable and can be easily damaged by such drainage catheters (127). Finally, in patients who develop subdural empyema as a consequence of infection of the paranasal sinuses or mastoid air cells, definitive surgical therapy should include débridement of such sources of infection.
CRANIAL BACTERIAL EPIDURAL ABSCESS
Cranial epidural abscesses are infections localized to the potential space between the calvaria and the dura. They are most often related to frontal sinusitis or to a previous surgical procedure, although they have been associated with many of the risk factors for brain abscess (141,145). These lesions are less common than subdural empyemas, representing only 1% to 2% of intracranial infections. By and large, epidural abscess is more indolent in its presentation than infection in the subdural space, as the degree of cerebral irritation is far less severe. However, epidural infections may extend into the subdural or subarachnoid spaces or into the brain itself (146). Patients typically present with complaints of fever, nuchal rigidity, and periorbital or perinasal swelling. The initial diagnostic test should be neuroimaging with either CT or MRI, ideally with contrast and fine cuts through the paranasal and mastoid sinuses (141). MRI may allow for greater determination of the extent of infection, especially in tissue adjacent to bone. As with subdural empyemas, lumbar puncture should be considered with reservation, given the risk of neurologic deterioration and low diagnostic yield.
Definitive management of cranial epidural abscess is surgical evacuation and intravenous, often followed by oral, antibiotic therapy; recommended treatments vary widely based on the source of infection, organism, and response to therapy. In cases of postneurosurgical epidural abscess, concurrent osteomyelitis of the overlying bone flap must be assumed. In such cases, surgery should include removal of the infected bone flap as well as evacuation of pus within the epidural space; the presumptive organism is methicillin-resistant Staphylococcus aureus until culture and sensitivities can be obtained. Staphylococcus epidermidis, Escherichia coli, and anaerobic bacilli are also common. Salvage of bone flaps in postcraniotomy epidural abscess is feasible (147).
The treatment of epidural abscess associated with sinusitis is somewhat more controversial than the treatment of postneurosurgical infection. Both burr hole and craniotomy have been advocated for evacuation of epidural collections. Advocates of burr-hole evacuation point out that this method adequately removes the purulent collection without creating a devitalized bone flap over the infected space. On the other hand, proponents of craniotomy suggest that this method allows for complete removal of the infected fluid collection and the ability to treat any underlying subdural spread of infection. There is little evidence to support the superiority of either approach. The decision regarding which technique is used often falls to the preference of the individual surgeon. In all cases, however, the potential need for repeat surgery for source control should be considered. The outcome following surgical evacuation and antibiotic therapy is generally favorable, with most patients (>95%) experiencing good outcomes (135,141). As is the case in patients with subdural empyema, definitive surgical therapy also includes débridement for source control.
The propensity of cranial epidural abscess to be associated with subdural empyema has prompted some to advocate routine subdural exploration during evacuation (148). However, the likelihood that such a maneuver could seed a previously sterile subdural space is worrisome. Subdural exploration is advocated by other authors only in cases in which there is clinical or radiographic evidence of subdural spread (141,145).
Some authors have suggested that surgery to drain extradural pus can be avoided in neurologically intact patients with small fluid collections (141). These patients were treated with intravenous antibiotics and followed closely for any signs of worsening neurologic status or infection such as increased white blood cell count or erythrocyte sedimentation rate. The authors stress that such treatment should only be used in patients who have had a surgical eradication of the primary source of infection. This treatment is not fully accepted (135) and should only be attempted in patients who are available for close observation.
CALVARIAL OSTEOMYELITIS
Osteomyelitis of the calvaria most commonly occurs as a result of trauma, either incidental or operative. Surgical management must be tailored as each case demands, and a detailed discussion regarding these options is beyond the scope of this chapter. Calvarial osteomyelitis also occurs spontaneously from hematogenous dissemination, which is treated adequately with antibiotic administration in the vast majority of cases. Finally, calvarial osteomyelitis may occur as a result of invasion from adjacent structures. Osteomyelitis of the skull base frequently occurs as a direct extension from otitis, sinusitis, mastoiditis, or dental sources. Although many of these cases are also adequately managed with antibiotic administration, advanced stage of presentation and intracranial involvement in these cases can necessitate operative intervention.
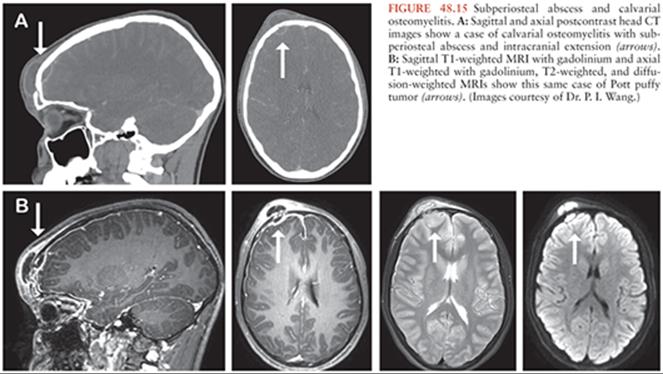
In particular, calvarial osteomyelitis resulting in subgaleal abscess and intracranial extension (Fig. 48.15) may require urgent surgical management. Pott puffy tumor, first described by Sir Percivall Pott in 1760 (149), is most commonly found in adolescents and is defined by frontal osteomyelitis with adjacent subgaleal abscess (150). It is most commonly suspected to arise from adjacent frontal sinusitis, although similar infectious processes have been reported in areas of the skull not contiguous with any sinus (151–153). The subgaleal abscess causes a soft raised mass—hence the moniker, “puffy tumor.” Intracranial extension can lead to meningitis, epidural abscess, subdural empyema, cerebral abscess, and thrombophlebitis of the superficial cortical veins. Venous sinus thrombosis is theorized to occur by septic emboli in the diploic veins that drain from the frontal sinus to the dural venous sinuses (149). Devastating venous infarcts are a major source of morbidity in cases of Pott puffy tumor, and thus, all cases suspected of progressing to intracranial extension warrant urgent evaluation for operative intervention. Although the frequency of cases has seemingly plummeted with the introduction of antibiotics and cross-sectional imaging (154), surgery remains the primary treatment modality in addition to antibiotic therapy (155) in this relatively rare presentation.
NEUROCYSTICERCOSIS
Cysticercosis is a common parasitic disease caused by infection with the larval form of pork tapeworm, Taenia solium. It is endemic in many parts of the world, including Africa, Eastern Europe, Central and South America, and Mexico (156). Because of the migration of people from endemic areas to the United States, cysticercosis is being reported with increasing frequency, such that it is listed as one of the neglected parasitic infections targeted by the Centers for Disease Control and Prevention (157–160). Depending on the extent of CNS involvement with cysticerci, neurocysticercosis may cause high mortality. This mortality rate has declined as better treatments have been defined, from 20% to 100% in the 1960s (161,162) to 0.9% to 18.5% in more recent reviews (163,164).
The larvae of T. solium (cysticerci) have a marked predilection for neural tissue and readily invade the brain, subarachnoid space, and ventricles. Invasion of the CNS in cysticercosis is extremely common, occurring in 60% to 90% of all cases (165,166). The propensity for brain involvement in cysticercosis makes neurocysticercosis the most common parasitic disease of the CNS (167). In rare instances, the larvae can invade the spinal cord, causing intramedullary cysticercosis, which manifests with paraparesis or tetraparesis (168).
Cysticerci typically develop in muscles, subcutaneously, and adjacent to the CNS. Cysticerci can involve the spinal cord, eye, subarachnoid space, ventricular CSF space, or the brain parenchyma. Typical cysticerci are small (<2 cm), round, thin-walled cysts found in the brain parenchyma or CSF spaces and initially provoke only minor inflammation. Intraventricular cysticerci can obstruct the CSF outflow tract, and hydrocephalus is a fairly common presentation of the disease. Racemose neurocysticercosis refers to a large, actively growing form of the disease in which grapelike clusters of cysts form in the basal subarachnoid cisterns or ventricles. The racemose form is poorly tolerated and causes intense inflammation with resultant seizures, intracranial hypertension, and hydrocephalus due to the size of the cysts and the accompanying inflammatory response.
Parenchymal cysts are usually multiple and may present with symptoms ranging from seizures to hemiparesis, movement disorders, visual loss, or brainstem dysfunction. Focal or generalized seizures are the most common symptom of parenchymal neurocysticercosis and may be present in up to 92% of cases (158,159,166,169–172). In children, parenchymal involvement may be diffuse and cause a clinical picture of acute encephalitis with cerebral edema (173,174).
Invasion of the ventricular system with cysticerci occurs relatively rarely. Cysts within the ventricles tend to be free floating and do not usually adhere to the ependymal lining. Both free-floating cysts and pedunculated cysts, which project into the ventricles, cause symptoms by obstructing midline CSF pathways. The episodic nature of the CSF obstruction may give rise to intermittent, severe headaches, drop attacks, or sudden death related to the sharp rise in intracranial pressure caused by obstruction. Hydrocephalus may or may not accompany such intermittent obstruction.
Cysts are found in the subarachnoid space in approximately 27% to 56% of cases (165). Typical cysts have a predilection for the dorsolateral subarachnoid space, usually do not present with significant symptoms, and rarely require surgical intervention. However, racemose neurocysticercosis, which has a predilection for the basal subarachnoid cisterns, causes symptoms of a chronic meningitis or, if CSF pathways are sufficiently blocked, hydrocephalus. In some cases, cranial nerves and cerebral arteries may be entrapped by the inflammatory process, giving rise to a clinical picture similar to meningeal carcinomatosis or vasculitis (159,166,171,172,175–177). This vasculitis may lead to the development of aneurysms within the cerebrovascular system through local inflammation of the arterial wall (175). Racemose involvement of the basal cisterns carries the worst prognosis (161,162).
The symptoms of neurocysticercosis are related to the size and location of the cysticercus cysts. Cysts within the brain parenchyma present in a manner similar to brain abscesses, with symptoms of increased intracranial pressure and seizures. Subarachnoid spread of the disease causes meningitis and communicating hydrocephalus. Those cysts that are located in the ventricular system frequently lodge at the foramen of Monro or aqueduct of Sylvius, causing obstructive hydrocephalus. These cysts may degenerate during the final stage of development, leading to a hyperintense T1-weighted lesion that can mimic the appearance of colloid cysts within the ventricular system (178).
The diagnosis of neurocysticercosis is generally made by clinical, serologic, and radiographic criteria. Because of the variety of possible presenting symptoms and physical examination findings, a diagnostic algorithm has been proposed that divides patients into “definitive” or “probable” cases on the basis of imaging, histopathology, serologic assays, response to antihelminthic therapy, and epidemiologic criteria (179). Subcutaneous nodules or visualization of subretinal parasites from systemic cysticercosis may be present on physical examination. An ophthalmologic examination is imperative in all suspected cases, as cysts in the vitreous humor may result in blindness if ruptured. Both serum and CSF markers for cysticercosis may reveal antibodies to T. solium antigens. A detailed discussion of the diagnostic tests available for neurocysticercosis is found elsewhere in this book. However, several caveats bear mention in this discussion. First, physical findings are inconstant in neurocysticercosis (179). Second, the existence of antibodies, especially serum antibodies, is not entirely specific (179). Third, CSF antibodies may be absent in up to 15% to 30% of cases of neurocysticercosis, particularly when lesions are restricted to parenchyma (159,166,168,180). Taken together, the presence of serum antibodies and compatible radiographic lesions in a patient from an endemic area is sufficient for the diagnosis of neurocysticercosis. The absence of such correlative findings, however, mandates surgical intervention in order to establish the diagnosis, because the only definitive test for neurocysticercosis is histopathologic proof.
Effective antihelminthic medications for the treatment of neurocysticercosis have quickly become the primary mode of management. Both albendazole and praziquantel have significantly reduced the need for surgery in the treatment of the CNS manifestations of neurocysticercosis (165,167,181–183). The use of steroids in addition to an antihelminthic agent has been shown to improve prognosis (184–187). Nevertheless, the need for surgery in this disease persists. Medical treatment often elicits a massive inflammatory response, one that can require operative intervention. Surgery remains the treatment of choice for patients with hydrocephalus secondary to subarachnoid or intraventricular cysts or for treatment of symptomatic mass effect (188). In endemic areas, neurocysticercosis accounts for as much as 11% of all neurosurgical procedures (189).
Single parenchymal cysts can be a diagnostic dilemma, as serologic markers are frequently negative in this setting. Single cysts that are located in surgically accessible regions can be safely excised with excellent results (Fig. 48.16A). When a single cyst is asymptomatic or seizures are well controlled, surgery may be avoided in favor of medical treatment. Most cysticerci will die within 5 to 7 years of their arrival in the brain (159); treatment with albendazole or praziquantel accelerates this process.

Multiple parenchymal lesions are not generally amenable to surgery because the disease is too diffuse. In children, a frequent form of the disease is characterized by multiple cysts and severe intracranial hypertension. In these cases, decompressive craniectomy or internal decompression may be performed as a life-saving measure (188). Despite the use of decompressive procedures, this form of neurocysticercosis has a poor prognosis (190).
Intraventricular cysts that cause obstruction of midline CSF pathways require surgical intervention (Fig. 48.16B). In these cases, the preferred surgical procedure is extirpation of the often free-floating cysts. Most often, these cysts lodge at the foramen of Monro, in the aqueduct of Sylvius, or in the fourth ventricle and can be safely extirpated either by open neurosurgical procedures or through endoscopic techniques. Of note, cyst rupture is a frequent result of removal but does not appear to predispose to ventriculitis, as long as the ventricular system is well flushed prior to closure (191). CSF diversion procedures are less effective in relieving the hydrocephalus caused by intraventricular cysts than that caused by subarachnoid seeding, as intraventricular cysts can lead to poor communication between the various ventricular chambers. In cases that do require CSF shunting, treatment with antihelminthic postprocedure leads to a significant reduction in shunt failure (192).
Involvement of the basal subarachnoid cisterns with neurocysticercosis often causes hydrocephalus. In these cases, the extensive meningeal involvement and arachnoiditis caused by the cysts make surgical exposure difficult. Nevertheless, large subarachnoid cysts causing brain or brainstem compression should be surgically removed (158,193). Treating the hydrocephalus with a CSF shunt in this setting is also difficult, as both inflammatory debris and encysted parasites may obstruct the shunt tubing (191). Morbidity from such shunt-related complications is common, and some neurosurgeons have advocated third ventriculostomy for the treatment of obstructive hydrocephalus at the time of endoscopic cyst removal (191). In cases of basal subarachnoid infection, treatment with antihelminthic agents is mandatory, as the disease is often too diffuse for surgery to offer any more than a palliative option.
MYCOTIC ANEURYSMS
The term “mycotic aneurysm” was coined by Sir William Osler in 1885 in reference to the mushroom-like morphology of an aneurysmal dilation in the aorta of a patient with infective endocarditis (194). The term is now commonly used to describe infectious arterial aneurysms in general, although these infectious aneurysms rarely occur as a result of fungal infection (195). Cerebral mycotic aneurysms occur in 2% to 10% of cases of bacterial endocarditis (196). A septic embolus lodges in the arterial wall of a small vessel, disrupting its integrity and resulting in aneurysmal enlargement of the vessel. Emboli demonstrate a tendency toward lodging in preexisting atherosclerotic plaques and areas of turbulent flow (197). The morphology of mycotic aneurysms is characterized by friable fusiform dilations that are prone to spontaneous rupture leading to intracranial hemorrhage (Fig. 48.17). Approximately 5% of patients with infective endocarditis develop an intracranial hemorrhage from a ruptured mycotic aneurysm; in some cases, a ruptured aneurysm is the first clinical manifestation of infective endocarditis (198,199). The rupture of a mycotic aneurysm is frequently a catastrophic event and is associated with a high rate of mortality (195).

Cerebral angiography is a very sensitive diagnostic tool for detecting mycotic aneurysms. The evolution of mycotic aneurysms is a dynamic process, and aneurysms may enlarge, shrink, or disappear within a short period of time (195,200,201). Serial angiography often shows this variable natural history (Fig. 48.18). Less invasive vascular studies such as CT angiography and magnetic resonance (MR) angiography have been used as well (202), but adequate sensitivity of these studies for the identification of such small, distal aneurysms has yet to be proven. In addition to providing important information on the location, size, and morphology of a ruptured aneurysm, angiography often discloses other unruptured mycotic aneurysms (203). Cerebral angiography should be performed if mycotic aneurysm is suspected. It has also been suggested that cerebral embolic phenomena in the setting of known infection be investigated by cerebral angiography, as these symptoms may represent a prodrome to aneurysmal rupture (204).

The risk factors for aneurysmal rupture remain unclear, and no randomized, prospective trials to inform treatment yet exist (196,203,205,206). In a recent review of 20 cases from one institution, Chun et al. (200) suggest a treatment strategy guide that incorporates medical antibiotic management, cerebral angiography, and endovascular or surgical intervention. Medical management alone consisted of a minimum 6-week course of appropriate antibiotics and was reserved only for patients with unruptured mycotic aneurysms. These patients were followed with serial angiography to assess for aneurysm progression and to determine the need for invasive intervention (200). Endovascular treatment was used in patients without an acute need for operative decompression and in aneurysms that did not involve eloquent cortex, whereas surgical treatment was mandated for all patients who had aneurysms in eloquent cortex or in those patients who had acute signs of increased intracranial pressure or expanding hematoma.
Because mycotic aneurysms are often necrotic or fusiform in shape, the standard neurosurgical approach using clip ligation of the aneurysm neck with preservation of the parent artery is often not possible (206,207). Rather, surgery of mycotic aneurysms usually involves resection of the aneurysmal segment of the affected artery or parent vessel sacrifice. The use of bypass grafting of the distal arterial segment has been advocated by some authors in order to preserve blood flow to the affected cortex (208). However, bypass grafts are technically difficult and, in the setting of disseminated intravascular infection, are prone to disruption and embolism. The surgical approach to mycotic aneurysms is also complicated by their predominant location in the distal cerebral vasculature. This difficulty can sometimes be circumvented by using stereotactic guidance techniques for aneurysm localization (200,206,209).
Endovascular techniques have proven useful in the management of mycotic aneurysms. The mainstay of endovascular treatment of mycotic aneurysms remains parent artery sacrifice. Endovascular obliteration of mycotic aneurysms with stents, balloons, glue, or thrombogenic coils has been attempted, although the potential risks of this approach, such as perforation of the necrotic aneurysm wall, are considerable (210,211). The natural history of mycotic aneurysm formation involves disruption of the internal elastic lamina and inflammatory infiltration of the media and adventitia. Thus, these mycotic aneurysms resemble pseudoaneurysms and aneurysms that occur as a result of transmural angiitis, more than they do true aneurysms (212). As such, the risk of intraprocedural rupture is extraordinarily high.
Other authors have recommended a more aggressive approach, including surgical excision of mycotic aneurysms whenever possible (213), whereas still others have recommended surgery in the case of previously ruptured or enlarging mycotic aneurysms. These latter recommendations are extrapolated from the natural history of congenital cerebral aneurysms, which suggest that these aneurysms are at higher risk of hemorrhage (206,207). Because mycotic aneurysms are rare and their natural history is variable, a uniform set of indications for surgical intervention has not been established. A clinical decision must therefore be made in each case, based on the condition of the patient, the location of the aneurysm, and the apparent response to appropriate medical therapy.
SPINE INFECTIONS
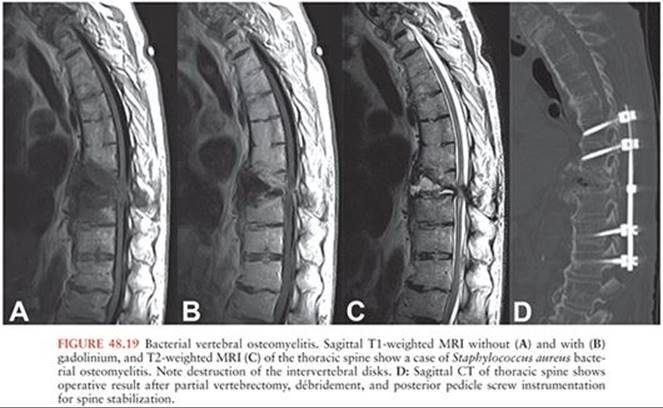
Infections of the spine are often classified anatomically, using terms such as osteomyelitis, diskitis, or epidural abscess. However, most pyogenic spine infections affect multiple structures (214). Spine infections most typically involve bacteria, particularly gram positives, and the most common organism is S. aureus (215). Inoculation occurs via arterial blood flow, retrograde flow through Batson vertebral venous plexus, or by direct invasion, whether from an adjacent infectious source or from a contaminated external source (such as a surgeon). Diagnosis is typically made by MRI, when the finding of back pain or neurologic deficit indicates this study. Enhancement of the vertebrae and paravertebral tissues is typical of spine infection, and destruction of the vertebral endplates and disk spaces suggests osteomyelitis rather than malignancy (Fig. 48.19). Spinal deformity frequently occurs with osteomyelitis as well.
Several atypical pathogens cause osteomyelitis, including fungi and mycobacteria. Many of these organisms first involve the lung, including Mycobacterium tuberculosis, Aspergillus, Cryptococcus, and Coccidioides. Many of these are liable to occur in the setting of immunosuppression, and correction of this immunocompromised state as well as initiation of antifungal medications are the mainstay of treatment. Surgical decompression and débridement is an option in cases demonstrating neurologic decline and is frequently required in cases of candidiasis (216), aspergillosis (217), and coccidioidomycosis (218).
The pathophysiology of spinal osteomyelitis caused by M. tuberculosis, known as Pott disease (Fig. 48.20), is characterized by slow growth, propensity for an oxygen-rich environment, and the absence of proteolytic enzymes. Tuberculous osteomyelitis is quite different from pyogenic spine infections in that it follows an indolent course over months to years and demonstrates a less painful evolution than the pyogenic infections. Serum markers for inflammation may remain normal, even when an abscess already exists. Imaging features typical of spinal tuberculosis infection include relative preservation of the intervertebral disks due to the lack of proteolytic enzymes, spread along the anterior longitudinal ligament, involvement of the psoas muscles, clumping of the lumbosacral nerve roots, and granulomatous accumulation in the basal cisterns (219). Calcification within a psoas abscess is commonly considered to be diagnostic for tuberculosis. Surgical intervention is reserved primarily for deformity correction and spinal cord decompression, and thus the timing of surgery is highly variable. In the absence of spinal cord compression, surgery for deformity correction is typically performed after completion of antibiotic therapy.

Culture and speciation is a critical part of the diagnostic process, and although blood cultures are easily and quickly obtained, sampling of infected tissue is often required via image-guided needle aspiration or, in rare cases, open biopsy. Biopsy cultures are typically obtained for suspected cases of polymicrobial osteomyelitis or in cases of negative blood cultures, where they can improve the diagnostic yield from an estimated 58% to 77% (220). Little data exist to suggest an advantage of open biopsy over image-guided needle biopsy. The impact of antibiotic administration prior to obtaining biopsy specimens on culture results remains a controversial topic. Small studies have shown both damaging effects and little effect on the diagnostic yield of needle biopsy performed after antibiotics have been initiated (221,222).
The role for surgery in cases of spine infection largely revolves around the question of neurologic deficit. A progressive neurologic deficit should trigger consideration for urgent surgical intervention, regardless of whether an epidural abscess or acute kyphotic deformity is readily visible on imaging. However, no definitive study yet exists to prove the benefit of urgent operative decompression in spine infections, and there is at least a small amount of debate about the issue, particularly in the case of spinal epidural abscess, as discussed in the following section.
SPINAL EPIDURAL ABSCESS
Suppuration in the spinal epidural space is an infrequent but potentially devastating condition, one that has traditionally been considered a neurosurgical emergency. Spinal epidural abscesses were first described by Morgagni in 1769 (223). They were most frequently identified on postmortem examination until the 1930s (224). A number of series have been published since then, outlining the pathogenesis and treatment of these rather uncommon infections (225–233).
Spinal epidural abscess typically arises either by hematogenous spread or by direct extension. Known risk factors for spinal epidural abscess include intravenous drug abuse, diabetes mellitus, dialysis-dependent renal failure, immunosuppression, alcoholism, and trauma. Hematogenous spread accounts for up to 63% of cases and may be associated with skin abscesses (223,226,230), infected vascular catheters, pneumonia, or otitis media (232). Direct extension from adjacent osteomyelitis accounts for up to 38% of cases (226). Contiguous spread of infection from retropharyngeal, psoas, and pulmonary abscesses have also been reported to cause infection in the spinal epidural space. Finally, spinal epidural abscesses have been reported as rare complications of surgical procedures and epidural anesthesia (234–239). Most spinal epidural abscesses are caused by S. aureus (Fig. 48.21), although the number of infections caused by gram-negative aerobes and M. tuberculosis is significant (223,226,230). Atypical organisms that cause meningitis, osteomyelitis, and diskitis are also prone to cause epidural abscess, most famously, the fungus Exserohilum rostratum involved in the 2012 outbreak of infections in patients who received contaminated methylprednisolone (Fig. 48.22).


Symptoms and signs of spinal epidural abscess include back pain and fever, followed by radicular pain and paraparesis or tetraparesis. Patients may also have symptoms that mimic bacterial meningitis such as headache and neck stiffness. Symptoms may vary from indolent to rapidly progressive. Rapid progression is associated with poor outcome (227,230). Other poor prognostic factors at admission include paraplegia, location of the abscess in the cervicothoracic region, and elevated peripheral blood white blood cell count (232).
The management of a spinal epidural abscess has traditionally consisted of decompressive laminectomy and débridement of the inflammatory tissue, followed by long courses of intravenous and oral antibiotics (145,223,227). In the past, the spinal wound was commonly packed open and allowed to close by granulation (239). More recently, however, primary closure of wounds has been advocated, as this approach reduces the length of hospital stay and discomfort to the patient (145,223,227). Because many epidural abscesses involve long segments of the spinal column, decompressive laminectomies are performed at multiple levels, if necessary. For example, in a review of 188 patients from seven studies, laminectomies were performed at an average of four spinal levels (range, 1 to 26 levels) (227). This degree of spinal decompression, especially in children, raises the possibility of long-term spinal instability or deformity. Some authors have advocated a more limited exposure in children in order to minimize loss of structural stability (240,241); laminoplasty, as opposed to laminectomy, might contribute to this goal of preserving alignment and stability, although data to support this claim are limited.
Epidural abscesses arising from vertebral osteomyelitis (usually those in the cervical or thoracic region) are primarily ventral to the thecal sac and may be difficult to approach through a standard laminectomy. In such cases, an anterior or anterolateral approach has been advocated in order to remove the infected bone and adequately decompress the spinal canal. Anterior spinal procedures involve bone–autograft fusion of one or more vertebral bodies and are more technically complicated than the traditional laminectomy approach. The use of metal plates or screws, a standard adjunct in most anterior spinal fusions, is controversial in the setting of osteomyelitis and epidural abscess (242). Immediate stabilization in anterior fusions is therefore often achieved using rigid external bracing.
An alternative surgical approach to treatment of spinal epidural abscess is percutaneous drainage of the abscess, with or without image guidance (243–245). The difficulty with percutaneous drainage lies primarily in the consistency of the inflammatory tissue within the spinal canal; in more than 50% of cases, the spinal epidural abscess is composed of granulation tissue rather than liquid pus (223). Nonetheless, in cases that involve multiple spinal levels, some authors advocate the use of CT-guided percutaneous drainage as a means of avoiding extensive decompression and the risk of spinal instability.
Several studies have suggested that certain patients, primarily those with positive cultures and without neurologic deficits, may be treated without surgery (244,246–250). The uncertainty over which patients can be safely treated without surgery, however, has prevented this approach from gaining wide acceptance. In one report, several patients who were being treated medically with appropriate antibiotics deteriorated suddenly and irreversibly before surgical decompression (251). In addition, whether nonoperative treatment requires a longer course of antibiotics is unclear (223).
Potential for neurologic recovery with these different treatment modalities is also uncertain. The single most important prognostic factor for functional recovery is neurologic deficit at the time of decompression. Although there is no definitive comparison of surgical versus medical therapy for the acute presentation of spinal epidural abscesses, it is widely accepted that immediate operative decompression is preferable to conservative management as a means to preserve neurologic function. Although overall paralysis rates have traditionally been reported to be quite high (21% to 39%) (252–254), the potential for recovery of function after spinal epidural abscess appears to be better than after traumatic spinal cord injury (255).
CENTRAL NERVOUS SYSTEM INFECTIONS IN THE IMMUNOCOMPROMISED PATIENT
Immunocompromised patients are susceptible to infections not usually seen in the general population. Patients with impaired immunity present with CNS infections caused by organisms such as gram-negative bacteria, fungi, and parasites. The management of CNS infections in this group of patients presents the clinician with a unique set of difficulties. The list of opportunistic infections that occur in immunocompromised patients is lengthy, and a detailed discussion of rarely encountered infections is beyond the scope of this chapter. Because the immune system of these patients is compromised, infections are difficult to eradicate and tend to spread aggressively despite maximal therapy. Although medical therapy is the cornerstone of treatment for all such infections, surgery plays an important role in their management.
Fungal Infection
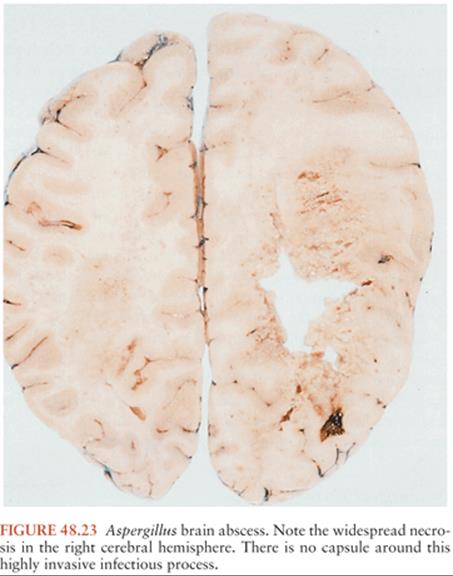
Aspergillus species are the most common cause of fungal brain abscess in immunosuppressed patients (58,256) but are rare in otherwise healthy individuals (257). Aspergillus fumigatus, the most common Aspergillus species isolated from abscesses, has a propensity for CNS invasion (20,258–260). Any CNS involvement is predominantly parenchymal, although meningitis or vasculitis may also occur (261). The angioinvasive nature of the fungus may result in multifocal hemorrhagic lesions or vascular thrombosis with infarction. Brain abscess caused by Aspergillus species is extremely difficult to treat. The fungal infection tends to be highly invasive and poorly encapsulated (Fig. 48.23) (98,101). Patients susceptible to this type of infection have a poor immune response and are likely to have serious systemic illnesses concurrent with the abscess.
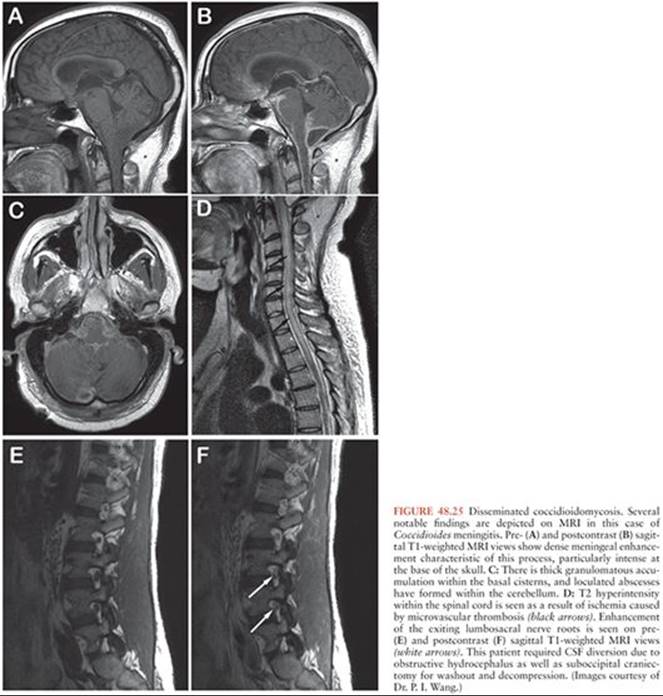
A number of other fungal agents, such as certain Candida species, Cryptococcus neoformans (Fig. 48.24), Cryptococcus gattii, Coccidioides immitis (Fig. 48.25), and certain members of the order Mucorales, may cause abscesses that require surgical intervention. The mainstay of treatment involves antibiotics, such as voriconazole, posaconazole, amphotericin B, anidulafungin, or caspofungin; surgical aspiration alone is ineffective and rarely successful in cases without a well-formed capsule. However, when the abscess is located in noneloquent regions of the brain, some authors favor an aggressive resection that includes a margin of uninvolved tissue (262,263); a few successful outcomes have been reported with this approach. Intracavitary or intraventricular administration of amphotericin B is also reported in treating fungal brain abscesses with some success (101,264–267). Although intrathecal injection of amphotericin B circumvents the penetration difficulties of intravenous administration of this medication, its efficacy remains unstudied. In addition, the neurotoxic effects of intracranially administered amphotericin B, such as seizures, psychosis, quadriparesis, and leukoencephalopathy, may limit the usefulness of this approach (268).

Rhinocerebral Mucormycosis
Cerebral involvement by fungus from the order Mucorales was described over 100 years ago and more commonly include the genera Rhizopus, Mucor, and Rhizomucor (269). Termed “rhinocerebral mucormycosis” (RCM) because of its propensity to involve the paranasal sinuses as well as the brain (Fig. 48.26), this infection was universally considered fatal until 1955. RCM begins when a susceptible patient inhales the airborne spores of the ubiquitous molds of Mucoraceae and establishes the infection in the nasal mucosa or palate. Direct invasion into the paranasal sinuses, skull base, or frontal lobes causes CNS symptoms such as headache, cranial nerve dysfunction, motor or sensory deficits, or altered mental status. With the introduction of amphotericin B and aggressive surgical treatment, survival rates have climbed steadily since then, reaching 40% to 80% (270–273). Mortality is highly dependent on the degree of intracranial extension, which conveys a dismal outcome (274,275).

RCM is most commonly associated with diabetic ketoacidosis, hematopoietic malignancy, and immunosuppression (276). In the United States, the incidence of RCM is declining in the overall population; a novel hypothesis attributes the decline to increased use of statins (277). Statins induce apoptosis in certain species of Mucorales (278). Diabetic patients are at risk of RCM because of the impaired neutrophilic activity that results from long-standing disease. A similar mechanism is present in patients treated with immunosuppressive medications. The susceptibility of AIDS patients to Mucor species is not a direct function of T-cell suppression but rather occurs in the setting of neutropenia (279,280).
Brain abscess caused by Mucor may occur by direct extension (e.g., through the cribriform plate) or by hematogenous spread. Mucor has an affinity for the internal elastic lamina of arteries, and vascular thrombosis is a well-known complication of RCM (281). Thrombosis of vessels in the paranasal sinuses may result in facial necrosis, a disfiguring and often fatal complication of RCM. Involvement of the orbital apex and cavernous sinus is common, resulting in ophthalmoplegia, proptosis, and visual loss (Fig. 48.27) (274,282). Thrombosis of the internal carotid artery in the cavernous sinus may also occur, with devastating consequences (283,284).

The key to successful treatment of RCM begins with rapid diagnosis. Diagnosis can be definitively established by histopathology and culture of biopsy of the affected area, and biopsy is typically accessible through the nasal passage in the outpatient setting. Polymerase chain reaction (PCR) techniques have also been used for the diagnosis of Mucor infection (279,285).
Once the diagnosis of Mucor has been made, débridement of infected sinuses is mandatory. Surgical drainage and débridement of the Mucor abscess is the rule, because antibiotic therapy is rarely effective in the setting of the vascular thrombosis that often accompanies this disease process. Débridement has classically been achieved through an open procedure, although endoscopic sinus drainage may also be effective (286,287). Orbital exenteration has been advocated for cases in which the infection has spread to the orbital apex in an effort to prevent the extension of infection into the cavernous sinus (283,288). Because the base of the frontal lobe is most often involved, an aggressive surgical approach is usually well tolerated, allowing débridement of the anterior skull base, which is often necrotic (288). When the infection has destroyed the anterior skull base, a combined surgical approach is often necessary, with neurosurgical débridement of the infected intracranial contents as well as a débridement of the infected sinuses and skull base, followed by reconstruction of the cranial base in order to prevent CSF leak.
References
1. Breasted JH. The Edwin Smith Surgical Papyrus. Chicago: University of Chicago Press; 1930.
2. Clower WT, Finger S. Discovering trepanation: the contribution of Paul Broca. Neurosurgery. 2001;49:1417–1426.
3. Martin G. Successful drainage of an extradural abscess in 1667: Prince Rupert’s trephination. Br J Neurosurg. 1989;3:211–216.
4. Morand SF. Opuscules de Chirurgie. Paris: G Depsrez; 1772.
5. Macewen W. Pyogenic Infective Diseases of the Brain and Spinal Cord. Glasgow: James Maclehose and Sons; 1893.
6. Helweg-Larsen J, Astradsson A, Richhall H, et al. Pyogenic brain abscess, a 15 year survey. BMC Infect Dis. 2012;12:1471–2334.
7. Nicolosi A, Hauser WA, Beghi E, et al. Epidemiology of central nervous system infections in Olmsted County, Minnesota, 1950–1981. J Infect Dis. 1986;154:399–408.
8. Bhatia R, Tandon PN, Banerji AK. Brain abscess—an analysis of 55 cases. Int Surg. 1973;58:565–568.
9. Mathisen GE, Johnson JP. Brain abscess. Clin Infect Dis. 1997;25:763–781.
10. Rosenblum ML, Mampalam TJ, Pons VG. Controversies in the management of brain abscesses. Clin Neurosurg. 1986;33:603–632.
11. Brook I. Microbiology and antimicrobial treatment of orbital and intracranial complications of sinusitis in children and their management. Int J Pediatr Otorhinolaryngol. 2009;73:1183–1186.
12. Prasad KN, Mishra AM, Gupta D, et al. Analysis of microbial etiology and mortality in patients with brain abscess. J Infect. 2006;53:221–227.
13. Ersahin Y, Mutluer S, Guzelbag E. Brain abscess in infants and children. Childs Nerv Syst. 1994;10:185–189.
14. Carey ME, Chou SN, French LA. Experience with brain abscesses. J Neurosurg. 1972;36:1–9.
15. Beller AJ, Sahar A, Praiss I. Brain abscess. Review of 89 cases over a period of 30 years. J Neurol Neurosurg Psychiatry. 1973;36:757–768.
16. Garfield J. Management of supratentorial intracranial abscess: a review of 200 cases. Br Med J. 1969;2:7–11.
17. Morgan H, Wood MW, Murphey F. Experience with 88 consecutive cases of brain abscess. J Neurosurg. 1973;38:698–704.
18. Samson DS, Clark K. A current review of brain abscess. Am J Med. 1973;54:201–210.
19. Sennaroglu L, Sozeri B. Otogenic brain abscess: review of 41 cases. Otolaryngol Head Neck Surg. 2000;123:751–755.
20. Britt RH, Enzmann DR, Remington JS. Intracranial infection in cardiac transplant recipients. Ann Neurol. 1981;9:107–119.
21. Fischer EG, McLennan JE, Suzuki Y. Cerebral abscess in children. Am J Dis Child. 1981;135:746–749.
22. Kagawa M, Takeshita M, Yato S, et al. Brain abscess in congenital cyanotic heart disease. J Neurosurg. 1983;58:913–917.
23. Levy RM, Bredesen DE, Rosenblum ML. Neurological manifestations of the acquired immunodeficiency syndrome (AIDS): experience at UCSF and review of the literature. J Neurosurg. 1985;62:475–495.
24. Britt RH, Enzmann DR, Yeager AS. Neuropathological and computerized tomographic findings in experimental brain abscess. J Neurosurg. 1981;55:590–603.
25. Winn HR, Mendes M, Moore P, et al. Production of experimental brain abscess in the rat. J Neurosurg. 1979;51:685–690.
26. Waggener JD. The pathophysiology of bacterial meningitis and cerebral abscesses: an anatomical interpretation. Adv Neurol. 1974;6:1–17.
27. Nielsen H, Gyldensted C, Harmsen A. Cerebral abscess. Aetiology and pathogenesis, symptoms, diagnosis and treatment. A review of 200 cases from 1935–1976. Acta Neurol Scand. 1982;65:609–622.
28. Yang SY. Brain abscess: a review of 400 cases. J Neurosurg. 1981;55: 794–799.
29. Zeidman SM, Geisler FH, Olivi A. Intraventricular rupture of a purulent brain abscess: case report. Neurosurgery. 1995;36:189–193.
30. Takeshita M, Kawamata T, Izawa M, et al. Prodromal signs and clinical factors influencing outcome in patients with intraventricular rupture of purulent brain abscess. Neurosurgery. 2001;48:310–317.
31. Smith MR, Wood WB. An experimental analysis of the curative action of penicillin in acute bacterial infections: III. The effect of supparation upon the antibacterial action of the drug. J Exp Med. 1956;103:509–522.
32. Alderson D, Strong AJ, Ingham HR, et al. Fifteen-year review of the mortality of brain abscess. Neurosurgery. 1981;8:1–6.
33. Black P, Graybill JR, Charache P. Penetration of brain abscess by systemically administered antibiotics. J Neurosurg. 1973;38:705–709.
34. Wispelwey B, Scheld WM. Brain abscess. Clin Neuropharmacol. 1987;10: 483–510.
35. Mampalam TJ, Rosenblum ML. Trends in the management of bacterial brain abscesses: a review of 102 cases over 17 years. Neurosurgery. 1988;23: 451–458.
36. Diaz JM, Slagle DC. Sudden “stroke like” onset of hemiparesis due to bacterial brain abscess. Neurology. 1994;44:569–570.
37. Devi BI, Bhatia S, Kak VK, et al. Spontaneous haemorrhage associated with a brain abscess. Childs Nerv Syst. 1993;9:481–482.
38. Rosenblum ML, Hoff JT, Norman D, et al. Nonoperative treatment of brain abscesses in selected high-risk patients. J Neurosurg. 1980;52: 217–225.
39. Rotheram EB Jr, Kessler LA. Use of computerized tomography in nonsurgical management of brain abscess. Arch Neurol. 1979;36:25–26.
40. Whelan MA, Hilal SK. Computed tomography as a guide in the diagnosis and follow-up of brain abscesses. Radiology. 1980;135:663–671.
41. Brant-Zawadzki M, Enzmann DR, Placone RC Jr, et al. NMR imaging of experimental brain abscess: comparison with CT. AJNR Am J Neuroradiol. 1983;4:250–253.
42. Chang SC, Lai PH, Chen WL, et al. Diffusion-weighted MRI features of brain abscess and cystic or necrotic brain tumors: comparison with conventional MRI. Clin Imaging. 2002;26:227–236.
43. Guzman R, Barth A, Lovblad KO, et al. Use of diffusion-weighted magnetic resonance imaging in differentiating purulent brain processes from cystic brain tumors. J Neurosurg. 2002;97:1101–1107.
44. Garg M, Gupta RK, Husain M, et al. Brain abscesses: etiologic categorization with in vivo proton MR spectroscopy. Radiology. 2004;230:519–527.
45. Obana WG, Rosenblum ML. Nonoperative treatment of neurosurgical infections. Neurosurg Clin N Am. 1992;3:359–373.
46. Arlotti M, Grossi P, Pea F, et al. Consensus document on controversial issues for the treatment of infections of the central nervous system: bacterial brain abscesses. Int J Infect Dis. 2010;14:16.
47. Nadvi SS, Parboosing R, van Dellen JR. Cerebellar abscess: the significance of cerebrospinal fluid diversion. Neurosurgery. 1997;41:61–67.
48. de Louvois J, Hurley R. Inactivation of penicillin by purulent exudates. Br Med J. 1977;1:998–1000.
49. Yamamoto M, Jimbo M, Ide M, et al. Penetration of intravenous antibiotics into brain abscesses. Neurosurgery. 1993;33:44–49.
50. Bagley CJ. Brain abscess: clinical and operative data. JAMA. 1923;81: 2161–2166.
51. Coleman CC. The treatment of abscess of the brain. Arch Surg. 1929;18: 100–116.
52. McKenzie KG. The treatment of abscess of the brain. Arch Surg. 1929;18: 1594–1620.
53. King JE. The treatment of brain abscess by unroofing and temporary herniation of abscess cavity with the avoidance of usual drainage methods. Surg Gynecol Obstet. 1924;39:554–568.
54. Stephanov S. Surgical treatment of brain abscess. Neurosurgery. 1988;22: 724–730.
55. Kahn EA. The treatment of encapsulated brain abscess. JAMA. 1937;108: 87–90.
56. Kahn EA. Treatment of encapsulated brain abscess of the brain: visualization by colloidal thorium dioxide. Arch NeurPsych. 1939;41:158–165.
57. Stern WE. Surgery of the craniocerebral infections. In: Walker AE, ed. A History of Neurological Surgery. New York: Hafner; 1967:180–212.
58. Hagensee ME, Bauwens JE, Kjos B, et al. Brain abscess following marrow transplantation: experience at the Fred Hutchinson Cancer Research Center, 1984–1992. Clin Infect Dis. 1994;19:402–408.
59. Mayfield FH, Spurling RG. Brain abscess: a review of thirty-two surgical cases. South Med J. 1937;30:191–196.
60. Dandy WE. Treatment of chronic abscess of the brain by tapping: preliminary note. JAMA. 1926;87:1477–1478.
61. Tekkok IH, Erbengi A. Management of brain abscess in children: review of 130 cases over a period of 21 years. Childs Nerv Syst. 1992;8:411–416.
62. Mamelak AN, Obana WG, Flaherty JF, et al. Nocardial brain abscess: treatment strategies and factors influencing outcome. Neurosurgery. 1994;35: 622–631.
63. Kondziolka D, Duma CM, Lunsford LD. Factors that enhance the likelihood of successful stereotactic treatment of brain abscesses. Acta Neurochir (Wien). 1994;127:85–90.
64. Lunsford LD. Stereotactic drainage of brain abscesses. Neurol Res. 1987;9: 270–274.
65. Monobe T, Kawai S, Takemura K, et al. Aspiration and drainage for brain abscess causing massive bleeding from abscess capsule: case report [in Japanese]. No Shinkei Geka. 1994;22:363–365.
66. Ratnaike TE, Das S, Gregson BA, et al. A review of brain abscess surgical treatment—78 years: aspiration versus excision. World Neurosurg. 2011;76:431–436.
67. Smith SJ, Ughratdar I, MacArthur DC. Never go to sleep on undrained pus: a retrospective review of surgery for intraparenchymal cerebral abscess. Br J Neurosurg. 2009;23:412–417.
68. Horsely V, Clarke RH. The structure and function of the cerebellum examined by a new method. Brain. 1908;31:45–124.
69. Leksell L. The stereotaxic method and radiosurgery of the brain. Acta Chir Scand. 1951;102:316–319.
70. Spiegel EA, Wycis HT, Marks M, et al. Stereotaxic apparatus for operations on the human brain. Science. 1947;106:349–350.
71. Drake JM, Prudencio J, Holowaka S, et al. Frameless stereotaxy in children. Pediatr Neurosurg. 1994;20:152–159.
72. Guthrie BL, Adler JR Jr. Computer-assisted preoperative planning, interactive surgery, and frameless stereotaxy. Clin Neurosurg. 1992;38:112–131.
73. Kitchen ND, Lemieux L, Thomas DG. Accuracy in frame-based and frameless stereotaxy. Stereotact Funct Neurosurg. 1993;61:195–206.
74. Laborde G, Klimek L, Harders A, et al. Frameless stereotactic drainage of intracranial abscesses. Surg Neurol. 1993;40:16–21.
75. Paleologos TS, Dorward NL, Wadley JP, et al. Clinical validation of true frameless stereotactic biopsy: analysis of the first 125 consecutive cases. Neurosurgery. 2001;49:830–837.
76. Gildenberg PL. General concepts of stereotactic surgery. In: Lunsford LD, ed. Modern Stereotactic Surgery. Boston: Martinus Nijhoff; 1988:3–11.
77. Mohanty A, Venkatarama SK, Vasudev MK, et al. Role of stereotactic aspiration in the management of tuberculous brain abscess. Surg Neurol. 1999;51:443–447.
78. Nakajima H, Iwai Y, Yamanaka K, et al. Successful treatment of brainstem abscess with stereotactic aspiration. Surg Neurol. 1999;52:445–448.
79. Rajshekhar V, Chandy MJ. Successful stereotactic management of a large cardiogenic brain stem abscess. Neurosurgery. 1994;34:368–371.
80. Barlas O, Sencer A, Erkan K, et al. Stereotactic surgery in the management of brain abscess. Surg Neurol. 1999;52:404–411.
81. Bernays RL, Seifert B, Kollias SS, et al. Near-real-time guidance using intraoperative magnetic resonance imaging for radical evacuation of hypertensive hematomas in the basal ganglia. Neurosurgery. 2001;49:478.
82. Chacko AG, Chandy MJ. Diagnostic and staged stereotactic aspiration of multiple bihemispheric pyogenic brain abscesses. Surg Neurol. 1997;48:278–283.
83. Ciurea AV, Stoica F, Vasilescu G, et al. Neurosurgical management of brain abscesses in children. Childs Nerv Syst. 1999;15:309–317.
84. Dyste GN, Hitchon PW, Menezes AH, et al. Stereotaxic surgery in the treatment of multiple brain abscesses. J Neurosurg. 1988;69:188–194.
85. Hasdemir MG, Ebeling U. CT-guided stereotactic aspiration and treatment of brain abscesses. An experience with 24 cases. Acta Neurochir (Wien). 1993;125:58–63.
86. Itakura T, Yokote H, Ozaki F, et al. Stereotactic operation for brain abscess. Surg Neurol. 1987;28:196–200.
87. Lee GY, Daniel RT, Brophy BP, et al. Surgical treatment of nocardial brain abscesses. Neurosurgery. 2002;51:668–672.
88. Mamelak AN, Mampalam TJ, Obana WG, et al. Improved management of multiple brain abscesses: a combined surgical and medical approach. Neurosurgery. 1995;36:76–86.
89. Nauta HJ, Contreras FL, Weiner RL, et al. Brain stem abscess managed with computed tomography-guided stereotactic aspiration. Neurosurgery. 1987;20:476–480.
90. Skrap M, Melatini A, Vassallo A, et al. Stereotactic aspiration and drainage of brain abscesses. Experience with 9 cases. Minim Invasive Neurosurg. 1996;39:108–112.
91. Stapleton SR, Bell BA, Uttley D. Stereotactic aspiration of brain abscesses: is this the treatment of choice? Acta Neurochir (Wien). 1993;121:15–19.
92. Strowitzki M, Moringlane JR, Steudel W. Ultrasound-based navigation during intracranial burr hole procedures: experience in a series of 100 cases. Surg Neurol. 2000;54:134–144.
93. Yamamoto M, Fukushima T, Hirakawa K, et al. Treatment of bacterial brain abscess by repeated aspiration—follow up by serial computed tomography. Neurol Med Chir (Tokyo). 2000;40:98–105.
94. Chen T, Apuzzo M. Biopsy techniques and instruments. In: Gildenberg PL, Tasker RR, eds. Textbook of Stereotactic and Functional Neurosurgery. New York: McGraw-Hill; 1998:397–412.
95. Favre J, Taha JM, Burchiel KJ. An analysis of the respective risks of hematoma formation in 361 consecutive morphological and functional stereotactic procedures. Neurosurgery. 2002;50:48–57.
96. Gildenberg PL, Gathe JC Jr, Kim JH. Stereotactic biopsy of cerebral lesions in AIDS. Clin Infect Dis. 2000;30:491–499.
97. Gildenberg PL, Langford L, Kim JH, et al. Stereotactic biopsy in cerebral lesions of AIDS. Acta Neurochir Suppl (Wien). 1993;58:68–70.
98. Levy RM, Berger JR. Neurosurgical aspects of human immunodeficiency virus infection. Neurosurg Clin N Am. 1992;3:443–470.
99. Lunardi P, Acqui M, Maleci A, et al. Ultrasound-guided brain biopsy: a personal experience with emphasis on its indication. Surg Neurol. 1993;39:148–151.
100. Broggi G, Franzini A, Peluchetti D, et al. Treatment of deep brain abscesses by stereotactic implantation of an intracavitary device for evacuation and local application of antibiotics. Acta Neurochir (Wien). 1985;76:94–98.
101. Camarata PJ, Dunn DL, Farney AC, et al. Continual intracavitary administration of amphotericin B as an adjunct in the treatment of Aspergillus brain abscess: case report and review of the literature. Neurosurgery. 1992;31:575–579.
102. Mahmood A, Abad RM, Updegrove JH. Brain abscess treated by continuous antibiotic perfusion: technical note. Neurol Res. 1993;15:63–67.
103. Sato M, Oikawa T, Sasaki T, et al. Brain abscess ruptured into the lateral ventricle: the usefulness of treatment by intraventricular irrigation with antibiotics. A case report [in Japanese]. No Shinkei Geka. 1994;22:689–693.
104. Takeshita M, Kagawa M, Yato S, et al. Current treatment of brain abscess in patients with congenital cyanotic heart disease. Neurosurgery. 1997;41:1270–1279.
105. Heineman HS, Braude AI, Osterholm JL. Intracranial suppurative disease. Early presumptive diagnosis and successful treatment without surgery. JAMA. 1971;218:1542–1547.
106. Chow AW, Alexander E, Montgomerie JZ, et al. Successful treatment of non-meningitic listerial brain abscess without operation. West J Med. 1975;122:167–171.
107. Berger SA, Edberg SC, David G. Infectious disease in the sella turcica. Rev Infect Dis. 1986;8:747–755.
108. Bjerre P, Riishede J, Lindholm J. Pituitary abscess. Acta Neurochir (Wien). 1983;68:187–193.
109. Dutta P, Bhansali A, Singh P, et al. Pituitary abscess: report of four cases and review of literature. Pituitary. 2006;9:267–273.
110. Liu F, Li G, Yao Y, et al. Diagnosis and management of pituitary abscess: experiences from 33 cases. Clin Endocrinol (Oxf). 2011;74:79–88.
111. Vates GE, Berger MS, Wilson CB. Diagnosis and management of pituitary abscess: a review of twenty-four cases. J Neurosurg. 2001;95:233–241.
112. Lindholm J, Rasmussen P, Korsgaard O. Intrasellar or pituitary abscess. J Neurosurg. 1973;38:616–619.
113. Selosse P, Mahler C, Klaes RL. Pituitary abscess. Case report. J Neurosurg. 1980;53:851–852.
114. Domingue JN, Wilson CB. Pituitary abscesses. Report of seven cases and review of the literature. J Neurosurg. 1977;46:601–608.
115. Robinson B. Intrasellar abscess after transsphenoidal pituitary adenomectomy. Neurosurgery. 1983;12:684–686.
116. Nelson PB, Haverkos H, Martinez AJ, et al. Abscess formation within pituitary tumors. Neurosurgery. 1983;12:331–333.
117. Obrador S, Blazquez MG. Pituitary abscess in a craniopharyngioma. Case report. J Neurosurg. 1972;36:785–789.
118. Shuster A, Gunnarsson T, Sommer D, et al. Pituitary abscess: an unexpected diagnosis. Pediatr Radiol. 2010;40:219–222.
119. Mohr PD. Hypothalamic-pituitary abscess. Postgrad Med J. 1975;51: 468–471.
120. Nelson DA, Holloway WJ, Kara-Eneff SC, et al. Neurological syndromes produced by sphenoid sinus abscess. Neurology. 1967;17:981–987.
121. Ahmed YS, Bradey N, Halaka AN, et al. Primary pituitary abscess: surgical management and endocrine assessment in three cases. Br J Neurosurg. 1989;3:409–414.
122. Kubik CS, Adams RD. Subdural empyema. Brain. 1943;66:18–42.
123. Khan M, Griebel R. Subdural empyema: a retrospective study of 15 patients. Can J Surg. 1984;27:283–285, 288.
124. Pathak A, Sharma BS, Mathuriya SN, et al. Controversies in the management of subdural empyema. A study of 41 cases with review of literature. Acta Neurochir (Wien). 1990;102:25–32.
125. Smith HP, Hendrick EB. Subdural empyema and epidural abscess in children. J Neurosurg. 1983;58:392–397.
126. Bok AP, Peter JC. Subdural empyema: burr holes or craniotomy? A retrospective computerized tomography-era analysis of treatment in 90 cases. J Neurosurg. 1993;78:574–578.
127. Nathoo N, Nadvi SS, Gouws E, et al. Craniotomy improves outcomes for cranial subdural empyemas: computed tomography-era experience with 699 patients. Neurosurgery. 2001;49:872–878.
128. Balch RE. Wound infections complicating neurosurgical procedures. J Neurosurg. 1967;26:41–45.
129. Post EM, Modesti LM. “Subacute” postoperative subdural empyema. J Neurosurg. 1981;55:761–765.
130. Dill SR, Cobbs CG, McDonald CK. Subdural empyema: analysis of 32 cases and review. Clin Infect Dis. 1995;20:372–386.
131. Feuerman T, Wackym PA, Gade GF, et al. Craniotomy improves outcome in subdural empyema. Surg Neurol. 1989;32:105–110.
132. Mauser HW, Van Houwelingen HC, Tulleken CA. Factors affecting the outcome in subdural empyema. J Neurol Neurosurg Psychiatry. 1987;50:1136–1141.
133. Wackym PA, Canalis RF, Feuerman T. Subdural empyema of otorhinological origin. J Laryngol Otol. 1990;104:118–122.
134. Nathoo N, Nadvi SS, van Dellen JR, et al. Intracranial subdural empyemas in the era of computed tomography: a review of 699 cases. Neurosurgery. 1999;44:529–536.
135. Bockova J, Rigamonti D. Intracranial empyema. Pediatr Infect Dis J. 2000;19:735–737.
136. Davidson HD, Steiner RE. Magnetic resonance imaging in infections of the central nervous system. AJNR Am J Neuroradiol. 1985;6:499–504.
137. Komori H, Takagishi T, Otaki E, et al. The efficacy of MR imaging in subdural empyema. Brain Dev. 1992;14:123–125.
138. Ogilvy CS, Chapman PH, McGrail K. Subdural empyema complicating bacterial meningitis in a child: enhancement of membranes with gadolinium on magnetic resonance imaging in a patient without enhancement on computed tomography. Surg Neurol. 1992;37:138–141.
139. Takamura Y, Uede T, Igarashi K, et al. Magnetic resonance imaging of supratentorial and parafalcial empyema. No Shinkei Geka. 1995;23:61–64.
140. Bannister G, Williams B, Smith S. Treatment of subdural empyema. J Neurosurg. 1981;55:82–88.
141. Nathoo N, Nadvi SS, van Dellen JR. Cranial extradural empyema in the era of computed tomography: a review of 82 cases. Neurosurgery. 1999;44:748–754.
142. Bhandari YS, Sarkari NB. Subdural empyema. A review of 37 cases. J Neurosurg. 1970;32:35–39.
143. Le Beau J, Creissard P, Harispe L, et al. Surgical treatment of brain abscess and subdural empyema. J Neurosurg. 1973;38:198–203.
144. Banerjee AD, Pandey P, Devi BI, et al. Pediatric supratentorial subdural empyemas: a retrospective analysis of 65 cases. Pediatr Neurosurg. 2009;45:11–18.
145. Krauss WE, McCormick PC. Infections of the dural spaces. Neurosurg Clin N Am. 1992;3:421–433.
146. Slager UT. Infection and parainfectious inflammations. In: Basic Neuropathology. Baltimore: Williams & Wilkins; 1970:89–135.
147. Erickson DL, Seljeskog EL, Chou SN. Suction-irrigation treatment of craniotomy infections. Technical note. J Neurosurg. 1974;41:265–267.
148. George B, Roux F, Pillon M, et al. Relevance of antibiotics in the treatment of brain abscesses. Report of a case with eight simultaneous brain abscesses treated and cured medically. Acta Neurochir (Wien). 1979;47:285–291.
149. Bordley JE, Bischofberger W. Osteomyelitis of the frontal bone. Laryngoscope. 1967;77:1234–1244.
150. Feder HM Jr, Cates KL, Cementina AM. Pott puffy tumor: a serious occult infection. Pediatrics. 1987;79:625–629.
151. Arnold PM, Govindan S, Anderson KK. Spontaneous cranial osteomyelitis in an otherwise healthy ten-year-old male. Pediatr Neurosurg. 2009;45:407–409.
152. Maheshwar AA, Harris DA, Al-Mokhthar N, et al. Pott’s puffy tumour: an unusual presentation and management. J Laryngol Otol. 2001;115: 497–499.
153. Martin-Hirsch DP, Habashi S, Page R, et al. Latent mastoiditis: no room for complacency. J Laryngol Otol. 1991;105:767–768.
154. Tattersall R. Pott’s puffy tumour. Lancet. 2002;359:1060–1063.
155. Bambakidis NC, Cohen AR. Intracranial complications of frontal sinusitis in children: Pott’s puffy tumor revisited. Pediatr Neurosurg. 2001;35: 82–89.
156. Scharf D. Neurocysticercosis. Two hundred thirty-eight cases from a California hospital. Arch Neurol. 1988;45:777–780.
157. Centers for Disease Control and Prevention. Parasites—cysticercosis. http://www.cdc.gov/parasites/cysticercosis/index.html. Accessed January 1, 2014.
158. Grisolia JS, Wiederholt WC. CNS cysticercosis. Arch Neurol. 1982;39: 540–544.
159. McCormick GF, Zee CS, Heiden J. Cysticercosis cerebri. Review of 127 cases. Arch Neurol. 1982;39:534–539.
160. Richards FO Jr, Schantz PM, Ruiz-Tiben E, et al. Cysticercosis in Los Angeles County. JAMA. 1985;254:3444–3448.
161. Obrador S. Cysticercosis cerebri. Acta Neurochir (Wien). 1962;10: 320–364.
162. Stepien L. Cerebral cysticercosis in Poland. Clinical symptoms and operative results in 132 cases. J Neurosurg. 1962;19:505–513.
163. Carabin H, Ndimubanzi PC, Budke CM, et al. Clinical manifestations associated with neurocysticercosis: a systematic review. PLoS Negl Trop Dis. 2011;5:24.
164. Sorvillo FJ, DeGiorgio C, Waterman SH. Deaths from cysticercosis, United States. Emerg Infect Dis. 2007;13:230–235.
165. Leblanc R, Knowles KF, Melanson D, et al. Neurocysticercosis: surgical and medical management with praziquantel. Neurosurgery. 1986;18: 419–427.
166. Loo L, Braude A. Cerebral cysticercosis in San Diego. A report of 23 cases and a review of the literature. Medicine (Baltimore). 1982;61:341–359.
167. Monteiro L, Almeida-Pinto J, Stocker A, et al. Active neurocysticercosis, parenchymal and extraparenchymal: a study of 38 patients. J Neurol. 1993;241:15–21.
168. Mohanty A, Venkatrama SK, Das S, et al. Spinal intramedullary cysticercosis. Neurosurgery. 1997;40:82–87.
169. Del Brutto OH. Single parenchymal brain cysticercus in the acute encephalitic phase: definition of a distinct form of neurocysticercosis with a benign prognosis. J Neurol Neurosurg Psychiatry. 1995;58:247–249.
170. Dixon HBF, Lipscomb FM. Cysticercosis: an analysis and follow-up of 450 cases. Medical Research Council Special Report Series. 1961;299: 1–58.
171. Latovitzki N, Abrams G, Clark C, et al. Cerebral cysticercosis. Neurology. 1978;28:838–842.
172. Shanley JD, Jordan MC. Clinical aspects of CNS cysticercosis. Arch Intern Med. 1980;140:1309–1313.
173. Lopez-Hernandez A, Garaizar C. Childhood cerebral cysticercosis: clinical features and computed tomographic findings in 89 Mexican children. Can J Neurol Sci. 1982;9:401–407.
174. Rodriguez-Carbajal J, Boleaga-Duran B. Neuroradiology of human cysticercosis. In: Flisser A, ed. Cysticercosis: Present State of Knowledge and Perspectives. New York: Academic Press; 1982:139–162.
175. Huang PP, Choudhri HF, Jallo G, et al. Inflammatory aneurysm and neurocysticercosis: further evidence for a causal relationship? Case report. Neurosurgery. 2000;47:466–468.
176. Levy AS, Lillehei KO, Rubinstein D, et al. Subarachnoid neurocysticercosis with occlusion of the major intracranial arteries: case report. Neurosurgery. 1995;36:183–188.
177. Rabiela-Cervantes MT, Rivas-Hernandez A, Rodriguez-Ibarra J, et al. Anatomopathological aspects of human brain cysticercosis. In: Flisser A, ed. Cysticercosis: Present State of Knowledge and Perspectives. New York: Academic Press; 1982:179–200.
178. Gupta A, Nadimpalli SP, Cavallino RP. Intraventricular neurocysticercosis mimicking colloid cyst. Case report. J Neurosurg. 2002;97:208–210.
179. Del Brutto OH, Rajshekhar V, White AC Jr, et al. Proposed diagnostic criteria for neurocysticercosis. Neurology. 2001;57:177–183.
180. Espinosa B, Flisser A, Plencarte A, et al. Immunodiagnosis of human cysticercosis: ELISA and immunoelectrophoresis. In: Flisser A, ed. Cysticercosis: Present State of Knowledge and Perspectives. New York: Academic Press; 1982:163–170.
181. Monteiro L, Coelho T, Stocker A. Neurocysticercosis—a review of 231 cases. Infection. 1992;20:61–65.
182. Sotelo J, Del Brutto OH. Therapy of neurocysticercosis. Childs Nerv Syst. 1987;3:208–211.
183. St Geme JW III, Maldonado YA, Enzmann D, et al. Consensus: diagnosis and management of neurocysticercosis in children. Pediatr Infect Dis J. 1993;12:455–461.
184. Baird RA, Wiebe S, Zunt JR, et al. Evidence-based guideline: treatment of parenchymal neurocysticercosis: report of the Guideline Development Subcommittee of the American Academy of Neurology. Neurology. 2013;80:1424–1429.
185. Garg RK, Potluri N, Kar AM, et al. Short course of prednisolone in patients with solitary cysticercus granuloma: a double blind placebo controlled study. J Infect. 2006;53:65–69.
186. Kishore D, Misra S. Short course of oral prednisolone on disappearance of lesion and seizure recurrence in patients of solitary cysticercal granuloma with single small enhancing CT lesion: an open label randomized prospective study. J Assoc Physicians India. 2007;55:419–424.
187. Mall RK, Agarwal A, Garg RK, et al. Short course of prednisolone in Indian patients with solitary cysticercus granuloma and new-onset seizures. Epilepsia. 2003;44:1397–1401.
188. Rueda-Franco F. Surgical considerations in neurocysticercosis. Childs Nerv Syst. 1987;3:212.
189. Madrazo I, Garcia-Renteria JA, Sandoval M, et al. Intraventricular cysticercosis. Neurosurgery. 1983;12:148–152.
190. Colli BO, Martelli N, Assirati Junior JA, et al. Cysticercosis of the central nervous system. I. Surgical treatment of cerebral cysticercosis: a 23 years experience in the Hospital das Clinicas of Ribeirao Preto Medical School. Arq Neuropsiquiatr. 1994;52:166–186.
191. Bergsneider M, Holly LT, Lee JH, et al. Endoscopic management of cysticercal cysts within the lateral and third ventricles. J Neurosurg. 2000;92:14–23.
192. Kelley R, Duong DH, Locke GE. Characteristics of ventricular shunt malfunctions among patients with neurocysticercosis. Neurosurgery. 2002;50:757–762.
193. Stern WE. Surgical considerations and management of central nervous system cysticercosis. In: Brown WJ, Voge M, eds. Neuropathology of Parasitic Infections. New York: Oxford University Press; 1982:240.
194. Osler W. Gulstonian lectures on malignant endocarditis. Lancet. 1885;1: 415–418.
195. Bohmfalk GL, Story JL, Wissinger JP, et al. Bacterial intracranial aneurysm. J Neurosurg. 1978;48:369–382.
196. Aspoas AR, de Villiers JC. Bacterial intracranial aneurysms. Br J Neurosurg. 1993;7:367–376.
197. Krings T, Geibprasert S, terBrugge KG. Pathomechanisms and treatment of pediatric aneurysms. Childs Nerv Syst. 2010;26:1309–1318.
198. Pruitt AA, Rubin RH, Karchmer AW, et al. Neurologic complications of bacterial endocarditis. Medicine (Baltimore). 1978;57:329–343.
199. Salgado AV, Furlan AJ, Keys TF. Mycotic aneurysm, subarachnoid hemorrhage, and indications for cerebral angiography in infective endocarditis. Stroke. 1987;18:1057–1060.
200. Chun JY, Smith W, Halbach VV, et al. Current multimodality management of infectious intracranial aneurysms. Neurosurgery. 2001;48: 1203–1214.
201. Moskowitz MA, Rosenbaum AE, Tyler HR. Angiographically monitored resolution of cerebral mycotic aneurysms. Neurology. 1974;24: 1103–1108.
202. Ahmadi J, Tung H, Giannotta SL, et al. Monitoring of infectious intracranial aneurysms by sequential computed tomographic/magnetic resonance imaging studies. Neurosurgery. 1993;32:45–50.
203. Bingham WF. Treatment of mycotic intracranial aneurysms. J Neurosurg. 1977;46:428–437.
204. Yamada M, Miyasaka Y, Takagi H, et al. Cerebral bacterial aneurysm and indications for cerebral angiography in infective endocarditis. Neurol Med Chir (Tokyo). 1994;34:697–699.
205. Ducruet AF, Hickman ZL, Zacharia BE, et al. Intracranial infectious aneurysms: a comprehensive review. Neurosurg Rev. 2010;33:37–46.
206. Malik JM, Kamiryo T, Goble J, et al. Stereotactic laser-guided approach to distal middle cerebral artery aneurysms. Acta Neurochir (Wien). 1995;132:138–144.
207. Morawetz RB, Karp RB. Evolution and resolution of intracranial bacterial (mycotic) aneurysms. Neurosurgery. 1984;15:43–49.
208. Day AL. Extracranial-intracranial bypass grafting in the surgical treatment of bacterial aneurysms: report of two cases. Neurosurgery. 1981;9: 583–588.
209. Steinberg GK, Guppy KH, Adler JR, et al. Stereotactic, angiography-guided clipping of a distal, mycotic intracranial aneurysm using the Cosman-Roberts-Wells system: technical note. Neurosurgery. 1992;30:408–411.
210. Frizzell RT, Vitek JJ, Hill DL, et al. Treatment of a bacterial (mycotic) intracranial aneurysm using an endovascular approach. Neurosurgery. 1993;32:852–854.
211. Khayata MH, Aymard A, Casasco A, et al. Selective endovascular techniques in the treatment of cerebral mycotic aneurysms. Report of three cases. J Neurosurg. 1993;78:661–665.
212. Kannoth S, Thomas SV. Intracranial microbial aneurysm (infectious aneurysm): current options for diagnosis and management. Neurocrit Care. 2009;11:120–129.
213. Frazee JG, Cahan LD, Winter J. Bacterial intracranial aneurysms. J Neurosurg. 1980;53:633–641.
214. Kuker W, Mull M, Mayfrank L, et al. Epidural spinal infection. Variability of clinical and magnetic resonance imaging findings. Spine (Phila Pa 1976). 1997;22:544–551.
215. Waldvogel FA, Papageorgiou PS. Osteomyelitis: the past decade. N Engl J Med. 1980;303:360–370.
216. Hendrickx L, Van Wijngaerden E, Samson I, et al. Candidal vertebral osteomyelitis: report of 6 patients, and a review. Clin Infect Dis. 2001;32:527–533.
217. Walsh TJ, Anaissie EJ, Denning DW, et al. Treatment of aspergillosis: clinical practice guidelines of the Infectious Diseases Society of America. Clin Infect Dis. 2008;46:327–360.
218. Galgiani JN, Ampel NM, Catanzaro A, et al. Practice guideline for the treatment of coccidioidomycosis. Infectious Diseases Society of America. Clin Infect Dis. 2000;30:658–661.
219. Moore SL, Rafii M. Imaging of musculoskeletal and spinal tuberculosis. Radiol Clin North Am. 2001;39:329–342.
220. Zimmerli W. Clinical practice. Vertebral osteomyelitis. N Engl J Med. 2010;362:1022–1029.
221. Marschall J, Bhavan KP, Olsen MA, et al. The impact of prebiopsy antibiotics on pathogen recovery in hematogenous vertebral osteomyelitis. Clin Infect Dis. 2011;52:867–872.
222. Rankine JJ, Barron DA, Robinson P, et al. Therapeutic impact of percutaneous spinal biopsy in spinal infection. Postgrad Med J. 2004;80: 607–609.
223. Del Curling O Jr, Gower DJ, McWhorter JM. Changing concepts in spinal epidural abscess: a report of 29 cases. Neurosurgery. 1990;27: 185–192.
224. Dandy WE. Abscesses and inflammatory tumors in the spinal epidural space (so-called pachymeningitis externa). Arch Surg. 1926;13:477–494.
225. Allen SS, Kahn EA. Acute pyogenic infection of the spinal epidural space. JAMA. 1932;98:875–878.
226. Baker AS, Ojemann RG, Swartz MN, et al. Spinal epidural abscess. N Engl J Med. 1975;293:463–468.
227. Danner RL, Hartman BJ. Update on spinal epidural abscess: 35 cases and review of the literature. Rev Infect Dis. 1987;9:265–274.
228. Huang CR, Lu CH, Chuang YC, et al. Clinical characteristics and therapeutic outcome of Gram-negative bacterial spinal epidural abscess in adults. J Clin Neurosci. 2011;18:213–217.
229. Huang PY, Chen SF, Chang WN, et al. Spinal epidural abscess in adults caused by Staphylococcus aureus: clinical characteristics and prognostic factors. Clin Neurol Neurosurg. 2012;114:572–576.
230. Kaufman DM, Kaplan JG, Litman N. Infectious agents in spinal epidural abscesses. Neurology. 1980;30:844–850.
231. Kempthorne JT, Pratt C, Smale EL, et al. Ten-year review of extradural spinal abscesses in a New Zealand tertiary referral centre. J Clin Neurosci. 2009;16:1038–1042.
232. Soehle M, Wallenfang T. Spinal epidural abscesses: clinical manifestations, prognostic factors, and outcomes. Neurosurgery. 2002;51:79–87.
233. Zimmerer SM, Conen A, Muller AA, et al. Spinal epidural abscess: aetiology, predisponent factors and clinical outcomes in a 4-year prospective study. Eur Spine J. 2011;20:2228–2234.
234. Elias M. Cervical epidural abscess following trigger point injection. J Pain Symptom Manage. 1994;9:71–72.
235. Hendlisz A, Rypens F, Gay F, et al. Spinal epidural abscess after endoscopic treatment for chronic pancreatitis. Pancreas. 1995;10:204–206.
236. Strafford MA, Wilder RT, Berde CB. The risk of infection from epidural analgesia in children: a review of 1620 cases. Anesth Analg. 1995;80: 234–238.
237. Wanscher M, Riishede L, Krogh B. Fistula formation following epidural catheter. A case report. Acta Anaesthesiol Scand. 1985;29:552–553.
238. Watters DA, Moussa SA, Buyukpamukcu N. Epidural abscess complicating Swenson procedure: a case report and a review of the literature. J Pediatr Surg. 1984;19:218–220.
239. Wright RL. Septic Complications of Neurosurgical Spinal Procedures. Springfield, IL: Thomas; 1970.
240. de Villiers JC, Cluver PF. Spinal epidural abscess in children. S Afr J Surg. 1978;16:149–155.
241. Fischer EG, Greene CS Jr, Winston KR. Spinal epidural abscess in children. Neurosurgery. 1981;9:257–260.
242. Redfern RM, Miles J, Banks AJ, et al. Stabilisation of the infected spine. J Neurol Neurosurg Psychiatry. 1988;51:803–807.
243. Cwikiel W. Percutaneous drainage of abscess in psoas compartment and epidural space. Case report and review of the literature. Acta Radiol. 1991;32:159–161.
244. Lyu RK, Chen CJ, Tang LM, et al. Spinal epidural abscess successfully treated with percutaneous, computed tomography-guided, needle aspiration and parenteral antibiotic therapy: case report and review of the literature. Neurosurgery. 2002;51:509–512.
245. Tabo E, Ohkuma Y, Kimura S, et al. Successful percutaneous drainage of epidural abscess with epidural needle and catheter. Anesthesiology. 1994;80:1393–1395.
246. Bouchez B, Arnott G, Delfosse JM. Acute spinal epidural abscess. J Neurol. 1985;231:343–344.
247. Latronico N, Tansini A, Gualandi GF, et al. Successful nonoperative treatment of tuberculous spinal epidural abscess with cord compression: the role of magnetic resonance imaging. Eur Neurol. 1993;33:177–180.
248. Leys D, Lesoin F, Viaud C, et al. Decreased morbidity from acute bacterial spinal epidural abscesses using computed tomography and nonsurgical treatment in selected patients. Ann Neurol. 1985;17:350–355.
249. Mampalam TJ, Rosegay H, Andrews BT, et al. Nonoperative treatment of spinal epidural infections. J Neurosurg. 1989;71:208–210.
250. Wheeler D, Keiser P, Rigamonti D, et al. Medical management of spinal epidural abscesses: case report and review. Clin Infect Dis. 1992;15: 22–27.
251. Hlavin ML, Kaminski HJ, Ross JS, et al. Spinal epidural abscess: a ten-year perspective. Neurosurgery. 1990;27:177–184.
252. Darouiche RO, Hamill RJ, Greenberg SB, et al. Bacterial spinal epidural abscess. Review of 43 cases and literature survey. Medicine (Baltimore). 1992;71:369–385.
253. Maslen DR, Jones SR, Crislip MA, et al. Spinal epidural abscess. Optimizing patient care. Arch Intern Med. 1993;153:1713–1721.
254. Sampath P, Rigamonti D. Spinal epidural abscess: a review of epidemiology, diagnosis, and treatment. J Spinal Disord. 1999;12:89–93.
255. Koo DW, Townson AF, Dvorak MF, et al. Spinal epidural abscess: a 5-year case-controlled review of neurologic outcomes after rehabilitation. Arch Phys Med Rehabil. 2009;90:512–516.
256. Chernik NL, Armstrong D, Posner JB. Central nervous system infections in patients with cancer. Medicine (Baltimore). 1973;52:563–581.
257. Kim DG, Hong SC, Kim HJ, et al. Cerebral aspergillosis in immunologically competent patients. Surg Neurol. 1993;40:326–331.
258. Casey AT, Wilkins P, Uttley D. Aspergillosis infection in neurosurgical practice. Br J Neurosurg. 1994;8:31–39.
259. Gridelli B, Lucianetti A, Rodriguez G, et al. Neurologic complications following pediatric orthotopic liver transplantation. Transplant Proc. 1994;26:193–194.
260. Hall WA. Neurosurgical infections in the compromised host. Neurosurg Clin N Am. 1992;3:435–442.
261. Salaki JS, Louria DB, Chmel H. Fungal and yeast infections of the central nervous system. A clinical review. Medicine (Baltimore). 1984;63: 108–132.
262. Green M, Wald ER, Tzakis A, et al. Aspergillosis of the CNS in a pediatric liver transplant recipient: case report and review. Rev Infect Dis. 1991;13:653–657.
263. Guppy KH, Thomas C, Thomas K, et al. Cerebral fungal infections in the immunocompromised host: a literature review and a new pathogen— Chaetomium atrobrunneum: case report. Neurosurgery. 1998;43: 1463–1469.
264. Adler DE, Milhorat TH, Miller JI. Treatment of rhinocerebral mucormycosis with intravenous interstitial, and cerebrospinal fluid administration of amphotericin B: case report. Neurosurgery. 1998;42:644–649.
265. Albayrak BS, Sirin S, Arpaci F, et al. Long-term follow-up of a previously reported case of cerebellar aspergillosis with the implication of the potential therapeutic effect of intracavitary amphotericin B application. Neurosurgery. 2010;67:E1469–E1470.
266. Elgamal EA, Murshid WR. Intracavitary administration of amphotericin B in the treatment of cerebral aspergillosis in a non immune-compromised patient: case report and review of the literature. Br J Neurosurg. 2000;14:137–141.
267. Itoh T, Yamada H, Yamaguchi A, et al. Percutaneous intracavitary antifungals for a patient with pulmonary aspergilloma; with a special reference to in vivo efficacies and in vitro susceptibility results. Intern Med. 1995;34:85–88.
268. Hamill RJ, Sobel JD, El-Sadr W, et al. Comparison of 2 doses of liposomal amphotericin B and conventional amphotericin B deoxycholate for treatment of AIDS-associated acute cryptococcal meningitis: a randomized, double-blind clinical trial of efficacy and safety. Clin Infect Dis. 2010;51:225–232.
269. Paltauf A. Mycosis mucorina. Virchows Arch Pathol Anat. 1885;102: 543–564.
270. Blitzer A, Lawson W, Meyers BR, et al. Patient survival factors in paranasal sinus mucormycosis. Laryngoscope. 1980;90:635–648.
271. Pillsbury HC, Fischer ND. Rhinocerebral mucormycosis. Arch Otolaryngol. 1977;103:600–604.
272. Price JC, Stevens DL. Hyperbaric oxygen in the treatment of rhinocerebral mucormycosis. Laryngoscope. 1980;90:737–747.
273. Spellberg B, Walsh TJ, Kontoyiannis DP, et al. Recent advances in the management of mucormycosis: from bench to bedside. Clin Infect Dis. 2009;48:1743–1751.
274. Nussbaum ES, Hall WA. Rhinocerebral mucormycosis: changing patterns of disease. Surg Neurol. 1994;41:152–156.
275. Ochi JW, Harris JP, Feldman JI, et al. Rhinocerebral mucormycosis: results of aggressive surgical debridement and amphotericin B. Laryngoscope. 1988;98:1339–1342.
276. Roden MM, Zaoutis TE, Buchanan WL, et al. Epidemiology and outcome of zygomycosis: a review of 929 reported cases. Clin Infect Dis. 2005;41:634–653.
277. Kontoyiannis DP. Decrease in the number of reported cases of zygomycosis among patients with diabetes mellitus: a hypothesis. Clin Infect Dis. 2007;44:1089–1090.
278. Roze LV, Linz JE. Lovastatin triggers an apoptosis-like cell death process in the fungus Mucor racemosus. Fungal Genet Biol. 1998;25:119–133.
279. Eucker J, Sezer O, Graf B, et al. Mucormycoses. Mycoses. 2001;44:253–260.
280. Hejny C, Kerrison JB, Newman NJ, et al. Rhino-orbital mucormycosis in a patient with acquired immunodeficiency syndrome (AIDS) and neutropenia. Am J Ophthalmol. 2001;132:111–112.
281. Schwartz JC. Rhinocerebral mucormycosis: three case reports and subject review. J Emerg Med. 1985;3:11–19.
282. Schwartz JN, Donnelly EH, Klintworth GK. Ocular and orbital phycomycosis. Surv Ophthalmol. 1977;22:3–28.
283. Anaissie EJ, Shikhani AH. Rhinocerebral mucormycosis with internal carotid occlusion: report of two cases and review of the literature. Laryngoscope. 1985;95:1107–1113.
284. Galetta SL, Wulc AE, Goldberg HI, et al. Rhinocerebral mucormycosis: management and survival after carotid occlusion. Ann Neurol. 1990;28:103–107.
285. Hammond SP, Bialek R, Milner DA, et al. Molecular methods to improve diagnosis and identification of mucormycosis. J Clin Microbiol. 2011;49:2151–2153.
286. Alobid I, Bernal M, Calvo C, et al. Treatment of rhinocerebral mucormycosis by combination of endoscopic sinus debridement and amphotericin B. Am J Rhinol. 2001;15:327–331.
287. Jiang RS, Hsu CY. Endoscopic sinus surgery for rhinocerebral mucormycosis. Am J Rhinol. 1999;13:105–109.
288. Anand VK, Alemar G, Griswold JA Jr. Intracranial complications of mucormycosis: an experimental model and clinical review. Laryngoscope. 1992;102:656–662.
289. Borzone M, Capuzzo T, Rivano C, et al. Subdural empyema: fourteen cases surgically treated. Surg Neurol. 1980;13:449–452.
290. Hockley AD, Williams B. Surgical management of subdural empyema. Childs Brain. 1983;10:294–300.
291. Morgan DW, Williams B. Posterior fossa subdural empyema. Brain. 1985;108(pt 4):983–992.
292. Hodges J, Anslow P, Gillett G. Subdural empyema—continuing diagnostic problems in the CT scan era. Q J Med. 1986;59:387–393.
293. Miller ES, Dias PS, Uttley D. Management of subdural empyema: a series of 24 cases. J Neurol Neurosurg Psychiatry. 1987;50:1415–1418.
294. Shokunbi MT, Malomo AO. Intracranial subdural empyema: burr hole exploration for diagnosis and treatment. Afr J Med Med Sci. 1993;22: 9–12.
295. de Falco R, Scarano E, Cigliano A, et al. Surgical treatment of subdural empyema: a critical review. J Neurosurg Sci. 1996;40:53–58.
296. Emery E, Redondo A, Berthelot JL, et al. Intracranial abscess and empyema: neurosurgical management [in French]. Ann Fr Anesth Reanim. 1999;18:567–573.
297. Yilmaz N, Kiymaz N, Yilmaz C, et al. Surgical treatment outcome of subdural empyema: a clinical study. Pediatr Neurosurg. 2006;42:293–298.
298. Mat Nayan SA, Mohd Haspani MS, Abd Latiff AZ, et al. Two surgical methods used in 90 patients with intracranial subdural empyema. J Clin Neurosci. 2009;16:1567–1571.