Edited by Peter Cameron
OUTLINE
9.1 Approach to undifferentiated fever in adults 428
9.2 Meningitis 434
9.3 Septic arthritis 440
9.4 Urinary tract infections 442
9.5 Skin and soft-tissue infections 450
9.6 Hepatitis 458
9.7 HIV/AIDS 463
9.8 Sexually transmitted infections 471
9.9 Antibiotics in the emergency department 478
9.10 Needlestick injuries and related blood and body fluid exposures 489
9.11 Tropical infectious diseases 494
9.1 Approach to undifferentiated fever in adults
Jonathan Knott
Essentials
1 Over one-third of patients who have fever for more than 2–3 days with no localizing symptoms and signs are likely to have a bacterial infection; half of these will be in the respiratory or urinary tracts.
2 An unexplained fever in a person over the age of 50 should be regarded as due to a bacterial infection until proved otherwise.
3 An undifferentiated fever in an alcoholic patient, an intravenous drug user or an insulin-dependent diabetic is generally an indication for admission to hospital.
4 Any fever in a traveller returned from a malaria-endemic area should be regarded as due to malaria until proved otherwise.
5 Severe muscle pain, even in the absence of overt fever, may be an early symptom of meningococcaemia, staphylococcal or streptococcal bacteraemia.
6 An unexplained rash in a febrile patient should be regarded as meningococcaemia until proved otherwise.
7 The diagnosis of meningococcaemia should be considered in every patient with an undifferentiated fever.
8 There will always be a small number of febrile patients whose sepsis is not initially recognized because they do not appear toxic and their symptoms are non-specific. It is essential that all patients are encouraged to seek review if they have any clinical deterioration.
Introduction
Fever is a common presenting symptom to the emergency department (ED); about 5% of patients give fever as the reason for their visit. Most patients with fever have symptoms and signs that indicate the site or region of infection. A prospective study of patients aged 16 years or older who presented to an ED with fever≥37.9°C found that 85% had localizing symptoms and signs that suggested or identified a source of fever and 15% had unexplained fever after the history and examination [1].
Fever with no localizing symptoms or signs at presentation is often seen in the first day or two of the illness. Many patients with such a problem will ultimately prove to have self-limiting viral infections, but others will have non-viral infections requiring treatment. Among this latter group are illnesses that may be serious and even rapidly fatal.
Over one-third of patients who have fever for more than a few days with no localizing symptoms and signs are likely to have a bacterial infection [1,2].
If no cause is found in an adult with fever present for over 3 days, there is a good chance the patient will have a bacterial infection that needs treatment. Over half of these infections are likely to be in the respiratory or urinary tracts [1].
The most important task in the ED for febrile patients without localizing features is not to miss early bacterial meningitis, bacteraemia, such as meningococcaemia and early staphylococcal and streptococcal toxic shock syndromes.
Approach
The management of febrile patients varies according to the severity, duration and tempo of the illness, the type of patient and the epidemiological setting. Although the steps in management of a febrile patient in the ED, listed below, may be set out in a sequential manner, in reality the mental processes involved occur simultaneously by the bedside.
![]() Step 1: identify the very ill.
Step 1: identify the very ill.
![]() Step 2: find localizing symptoms and signs.
Step 2: find localizing symptoms and signs.
![]() Step 3: look for ‘at-risk’ patients.
Step 3: look for ‘at-risk’ patients.
Step 1: identify the seriously ill patient who requires urgent intervention
The first step in managing febrile patients is to identify those in need of immediate resuscitation, urgent investigations and empirical therapy. The presence of any of the following features justifies immediate intervention: shock, coma/stupor, cyanosis, profound dyspnoea, continuous seizures and severe dehydration.
Step 2: identify those with localized infections or easily diagnosable diseases
Having excluded those who need urgent intervention, the doctor has more time to attempt a diagnosis. The history and physical examination are usually sufficient to localize the source of community-acquired fever in most cases, especially if the illness has been present for several days.
History
A precise history remains the key to diagnosis of a febrile illness. An inability to give a history and to think clearly is a sign of potential sepsis.
Illness
An abrupt onset of fever, particularly when accompanied by chills or rigors and generalized aches, is highly suggestive of an infective illness.
Localizing symptoms, their evolution and relative severity, help to identify the site of infection; localized pain is particularly valuable in this way.
The severity and the course of the illness can be assessed by the patient’s ability to work, to be up and about, to eat and sleep and the amount of analgesics taken.
Previous state of health
Underlying diseases predispose patients to infection at certain sites or caused by certain specific organisms. Knowledge of any defects in the immune system is similarly helpful. For example, asplenic patients are more prone to overwhelming pneumococcal septicaemia and renal transplant patients to Listeria meningitis.
A past history of infectious diseases, particularly if properly documented, may be useful in excluding infections such as measles and hepatitis.
Predisposing events
Recent operations, accidents and injuries and medications taken may be the direct cause of the illness (e.g. drug fever or rash from co-trimoxazole, ampicillin) or may affect the resistance of the patient, predisposing to certain infections. Concurrent menstruation raises the possibility of toxic shock syndrome.
Epidemiology
Information on occupation, exposure to animals, hobbies, risk factors for blood-borne viruses and travel overseas or to rural areas may suggest certain specific infections, e.g. leptospirosis, acute HIV infection, hepatitis C, malaria, etc.
Contact with similar diseases and known infectious diseases
This information is useful in the diagnosis of problems such as meningococcal infection, viral exanthema, respiratory infection, diarrhoea, and zoonoses.
Examination
Physical examination in the febrile patient serves two purposes: to assess the severity of the illness and to find a site of infection.
Bedside assessment of severity and ‘toxicity’ based on intuitive judgement is frequently wrong and many patients with severe bacterial infections do not appear obviously ill or toxic.
Physical examination may yield a diagnosis in a febrile patient who has not complained of any localizing symptoms. A checklist of special areas to be examined is useful.
![]() Eyes: conjunctival haemorrhages are seen in staphylococcal endocarditis and scleral jaundice may be present before cutaneous jaundice is obvious.
Eyes: conjunctival haemorrhages are seen in staphylococcal endocarditis and scleral jaundice may be present before cutaneous jaundice is obvious.
![]() Skin: rashes of any sort, especially petechial rash; cellulitis in the lower legs may present with fever and constitutional symptoms before pain in the leg develops. Evidence of intravenous drug use should be sought at the common injection sites.
Skin: rashes of any sort, especially petechial rash; cellulitis in the lower legs may present with fever and constitutional symptoms before pain in the leg develops. Evidence of intravenous drug use should be sought at the common injection sites.
![]() Heart: murmurs and pericardial rubs.
Heart: murmurs and pericardial rubs.
![]() Lungs: subtle crackles may be heard in pneumonic patients without respiratory symptoms.
Lungs: subtle crackles may be heard in pneumonic patients without respiratory symptoms.
![]() Abdominal organs: tenderness and enlargement without subjective pain may be the only clue to infections in these organs.
Abdominal organs: tenderness and enlargement without subjective pain may be the only clue to infections in these organs.
![]() Lymph nodes: especially the posterior cervical glands. Tenderness of the jugulodigastric glands is a good sign of bacterial tonsillitis.
Lymph nodes: especially the posterior cervical glands. Tenderness of the jugulodigastric glands is a good sign of bacterial tonsillitis.
![]() Sore throat may be absent in the first few hours of streptococcal tonsillitis. Examination of the throat may give the diagnosis. Oedema of the uvula is also a useful sign of bacterial infection in that region.
Sore throat may be absent in the first few hours of streptococcal tonsillitis. Examination of the throat may give the diagnosis. Oedema of the uvula is also a useful sign of bacterial infection in that region.
![]() Marked muscle tenderness is a frequent sign of sepsis.
Marked muscle tenderness is a frequent sign of sepsis.
![]() Neck stiffness may be a clue to meningitis in a confused patient who cannot give a history.
Neck stiffness may be a clue to meningitis in a confused patient who cannot give a history.
![]() Any area that is covered, e.g. under plasters or bandages, for evidence of sepsis.
Any area that is covered, e.g. under plasters or bandages, for evidence of sepsis.
There are two caveats when assessing local symptoms and signs:
![]() localizing features may not be present or obvious early in the course of a focal infection, e.g. the absence of cough in bacterial pneumonia, sore throat in tonsillitis or diarrhoea in gastrointestinal infections in the first 12–36 hours of the illness
localizing features may not be present or obvious early in the course of a focal infection, e.g. the absence of cough in bacterial pneumonia, sore throat in tonsillitis or diarrhoea in gastrointestinal infections in the first 12–36 hours of the illness
![]() localizing features may occasionally be misleading. For example, diarrhoea, which suggests infection of the gastrointestinal tract, may be a manifestation of more generalized infection, such as Gram-negative septicaemia, and crepitations at the lung base may indicate a subdiaphragmatic condition rather than a chest infection.
localizing features may occasionally be misleading. For example, diarrhoea, which suggests infection of the gastrointestinal tract, may be a manifestation of more generalized infection, such as Gram-negative septicaemia, and crepitations at the lung base may indicate a subdiaphragmatic condition rather than a chest infection.
Step 3: look for the ‘at-risk’ patient
If no diagnosis is forthcoming after the first two steps, the next task is to identify the ‘at-risk’ patient who may not appear overtly ill but who, nonetheless, requires medical intervention. This applies particularly to those with treatable diseases that can progress rapidly, such as bacterial meningitis, bacteraemia and toxic shock syndromes.
Four sets of pointers are helpful in identifying these ‘at-risk’ patients: the type of patient (host characteristics), exposure history, the nature of the non-specific symptoms and how rapidly the illness evolves.
Clinical pointers: type of patient
Clinical manifestations of infections are often subtle or non-specific in young children, the elderly and the immunocompromised. The threshold for intervention in these patients should be lowered. The issue of fever in children is not addressed in this chapter.
Elderly patients
Elderly patients with infections often do not mount much of a febrile response and fever may be absent in 20–30% of these patients [3].
Infectious diseases in the elderly, as in the very young, often present with non-specific or atypical symptoms and signs and may progress rapidly [4].
In adult patients with unexplained fever, up to one-third may have bacteraemia or focal bacterial infection. This proportion is even higher in those over the age of 50 [1]. In the elderly, a fever>38°C indicates a possible serious infection [5] and is associated with increasing risk of death [6].
The urinary tract is the most frequent site of infection and source of bacteraemia; symptoms of urinary tract infection are frequently absent in the elderly. The respiratory tract is the next most common site of infection; fever and malaise may be the only clues of pneumonia in the elderly. Urinalysis and chest X-ray will identify about half of occult infections [1].
An unexplained fever in a person over the age of 50 should be regarded as being caused by a bacterial infection until proved otherwise and is generally an indication for admission to hospital.
Alcoholic patients
Alcoholic patients present with multiple problems, many of which cause fever. Most are caused by infections, the commonest of which is pneumonia. Multiple infections may occur at the same time [7].
Non-infectious causes of fever frequently coexist with infections and conditions such as subarachnoid haemorrhage, alcoholic withdrawal and alcoholic hepatitis and require admission.
The initial history and physical examination in the alcoholic may be unreliable and diagnosis may be difficult.
Alcoholic patients with fever for which no obvious cause is found should be admitted to hospital for investigations and observation.
Injecting drug users
The risk of injecting drug users acquiring serious or unusual infections is high through repeated self-injection with non-sterile illicit substances, the use of contaminated needles and syringes and poor attention to skin cleansing prior to injections [8].
Many intravenous drug users presenting with fever have a serious infection. Some have obvious focal infections, such as cellulitis and pneumonia. Others present simply with fever and the presence of bacteraemia and endocarditis must be suspected.
Clinical assessment cannot differentiate trivial from potentially serious conditions in these patients [8]. A history of chills, rigors and sweats strongly suggest the presence of a transient or ongoing bacteraemia. Back pain may be a subtle symptom of endocarditis or vertebral osteomyelitis.
It is difficult to distinguish the patient with endocarditis from other drug users with fever due to another cause. Hospitalization of febrile injecting drug users would be prudent if 24-hour follow up is not possible. Intravenous drug use in the previous 5 days is a predictor of occult major infection and is an indication for admission to hospital [9].
Patients with diabetes mellitus
Diabetic patients are more prone to developing certain bacterial infections [1]. A diabetic patient with an unexplained fever is more likely to have an occult bacterial infection than a non-diabetic patient. In general, an insulin-dependent diabetic patient, especially if aged over 50, with fever and no obvious source of infection, should be investigated and preferably admitted.
Febrile neutropaenic patients
Febrile neutropaenic patients (absolute neutrophil count<500/μL or<1000/μL and falling rapidly) must be hospitalized regardless of their clinical appearance. Infections may become fulminant within hours in these patients and the clinical manifestations of their infective illnesses are frequently modified by the underlying disease, therapy received and coexisting problems.
Splenectomized patients
Splenectomized patients with fever must be very carefully assessed because of their increased risk of overwhelming bacterial infection. If the fever cannot be readily explained, admission for intravenous antibiotics is usually indicated.
Other immunocompromised patients
Fever in transplant patients (renal, hepatic or cardiac) and those with HIV infection is not an absolute indication for admission, but the threshold of intervention should be considerably lowered and they are best assessed by their usual treating doctors.
Patients recently discharged from hospital may have hospital-acquired infections or infections caused by multiresistant organisms. Recent operations or procedures may be a clue to the site of infection.
Clinical pointers: exposure history
Overseas travellers or visitors
Returned travellers or overseas visitors may have diseases such as malaria and typhoid fever that need early diagnosis and treatment. Any fever in a traveller returned from a malaria-endemic area should be regarded as due to malaria until proved otherwise.
Influenza in febrile returned travellers is a concern to EDs worldwide. Outbreaks of avian influenza occur periodically in bird populations throughout Asia. Although the virus does not typically infect humans, direct bird-to-human transmission of H5N1 influenza has been documented. The virus is highly pathogenic and the mortality of the disease is high. Travellers acquiring influenza overseas may also introduce this infection. Most cases occur within 2–4 days after exposure, but incubation is as long as 8 days. Suspected influenza infection requires isolation and respiratory precautions. The peak season is generally during the winter months, but can vary, especially in the tropics [10].
Although rare, viral haemorrhagic fever in returned travellers represents a true medical emergency and a serious public health threat. Viral haemorrhagic fevers are caused by several distinct families of virus, including Ebola and Marburg, Lassa fever, the New World arenaviruses (Guanarito, Machupo, Junin, and Sabia) and Rift Valley fever and Crimean Congo haemorrhagic fever viruses. Most exist in Africa, the Middle East or South America. Although some types cause relatively mild illnesses, many can cause severe, life-threatening disease. Viral haemorrhagic fever should be considered in any febrile patient who has returned from an area in which viral haemorrhagic fever was endemic, especially if they have come into contact with blood or other body fluids from a person or animal infected with viral haemorrhagic fever or worked in a laboratory or animal facility handling viral haemorrhagic fever specimens. All these infections have incubation periods of up to 2–3 weeks, so it may be possible to exclude viral haemorrhagic fever on epidemiological grounds alone. Isolation measures should be instituted immediately in these persons [11].
Staff working in emergency departments should be aware of regional outbreaks of unusual pathogens. These are reported by State and National Departments of Health. Returning travellers who are unwell will commonly go directly to an emergency department and this may be a critical point to limit further spread.
Contact with animals
A contact history with animals, either at work or at home, is frequently the clue to a zoonosis, particularly if the illness is a perplexing fever of several days’ duration. The occurrence of multiple cases at work or at home should also make one suspect these infections early.
Contact with meningococcal and Haemophilus meningitis
Close contacts of patients with these infections have a high risk of acquiring the same infections. Early symptoms may be subtle and a high index of suspicion must be maintained.
Clinical pointers: non-specific clinical features (Table 9.1.1)
There are several non-specific clinical features whose presence should suggest the possibility of sepsis. These warrant careful scrutiny even when the patient does not appear toxic. They are by no means specific indicators of serious problems and there will be many false positives. However, ignoring them is frequently the cause of missed or delayed diagnosis of sepsis.
Table 9.1.1
Clinical pointers: non-specific clinical features (‘alarm bells’)
Severe pain in muscles, neck or back
Impairment of conscious state
Vomiting, especially in association with headache or abdominal pain
Severe headache in the presence of a normal CSF
Unexplained rash
Jaundice
Severe sore throat or dysphagia with a normal looking throat
Repeated rigors
Severe pain in muscles, neck or back
Severe muscle pain, even in the absence of overt fever, may be an early symptom of meningococcaemia, staphylococcal or streptococcal bacteraemia. It is also a feature of myositis and necrotizing fasciitis.
Impairment of conscious state
A change in conscious state may be the sole presenting manifestation of sepsis, especially in the elderly.
Vomiting
Unexplained vomiting, especially in association with headache or abdominal pain, should raise concern. Vomiting without diarrhoea should not be attributed to a gastrointestinal infection. It is a common symptom of CNS infections and occult sepsis.
Severe headache in the presence of a normal CSF
This is especially important in a person who seldom gets headaches. Severe headache in a febrile patient with normal CSF should not be diagnosed as a viral infection; many focal infections, e.g. pneumonia and bacterial enteritis, may also present in this manner. CSF may be normal in cerebral abscess and in the prodromal phase of bacterial meningitis.
Unexplained rash
An unexplained rash in a febrile patient should be regarded as meningococcaemia until proved otherwise, even in the absence of headache or CSF pleocytosis.
Jaundice
Jaundice in the febrile patient is associated with a greatly increased risk of death, admission to ICU and prolonged hospital stay [6]. Jaundice in a febrile patient is unlikely to be due to viral hepatitis, but occurs in serious bacterial infections, such as bacteraemia, cholangitis, pyogenic liver abscess and malaria.
Sore throat or dysphagia
Severe sore throat or dysphagia with a normal-looking throat is frequently the presenting symptom of Haemophilus influenzae epiglottitis in adults.
Repeated rigors
Although repeated rigors may occur in some viral infections, they should generally be regarded as indicators of sepsis, in particular abscesses, bacteraemia, endocarditis, cholangitis and pyelonephritis.
Clinical pointers: evolution of illness (Table 9.1.2)
How rapidly the illness evolves is often an indication of its severity. Previously healthy individuals do not seek medical attention unless they are worried. Notice should be taken of any person seeking help within 24 hours of the onset of illness or a person whose illness appears to have progressed rapidly within 24–48 hours (e.g. from being up and about to being bedridden). Similarly, the patient who presents to the ED on more than one occasion over a 24–48-hour period warrants a careful work-up.
Table 9.1.2
Clinical pointers: evolution of illness
Those presenting early (<24 hours)
Those presenting with rapidly evolving symptoms
Patients presenting to ED on>1 occasion over a 24–48-hour period
Step 4: a final caveat
A major concern in the management of undifferentiated fever in adults is missing the diagnosis of meningococcal bacteraemia when the patient does not appear ill on presentation.
There are a number of infections that must be treated rapidly to minimize morbidity and mortality (Table 9.1.3). With the exception of meningococcal bacteraemia, there are usually some clues in the history or physical examination.
Table 9.1.3
Infections requiring urgent treatment
|
Disease |
Clues |
|
Meningococcaemia |
Myalgia, rash. May be none |
|
Falciparum malaria |
Travel history, blood film |
|
Bacterial meningitis |
Headache, change in conscious state, CSF findings |
|
Post-splenectomy sepsis |
Past history, abdominal scar |
|
Toxic shock syndromes |
Presence of shock and usually a rash |
|
Infections in the febrile neutropaenic |
Past history, blood film |
|
Infective endocarditis |
Past history, murmur, petechiae |
|
Necrotizing soft-tissue infections |
Pain, tenderness, erythema and swelling in skin/muscle, toxicity |
|
Space-occupying infection of head and neck |
Localizing symptoms and signs |
|
Focal intracranial infections |
Headache, change in conscious state, neurological signs, CT findings |
Meningococcal infection is peculiar in its wide spectrum of severity and variable rate of progression in different individuals. It may be fulminant and cause death within 12 hours or it may assume a chronic form that goes on for weeks.
When the patient presents with fever and a petechial rash, meningococcaemia can easily be suspected if one remembers the golden rule of medicine that ‘fever plus a petechial rash is meningococcaemia (or staphylococcal bacteraemia) until proved otherwise’. However, only 40% of meningococcal diseases present with a petechial rash.
It is less well known that the early meningococcaemic rash may be macular, i.e. one that blanches with pressure. This is the basis of another golden rule in infectious disease: early meningococcal rash may resemble a non-specific viral rash.
Rarely, meningococcal disease presents with symptoms and signs of a localized infection other than meningitis, e.g. pneumonia, pericarditis or urethritis. These presentations should not pose any management problems.
The risk of missing the diagnosis increases markedly when the patient with meningococcal disease presents with fever and non-specific symptoms without a rash. Abrupt onset of fever and generalized aches may be due to influenza, but it could be due to meningococcaemia.
It is prudent to single out meningococcal disease and ask oneself: could this patient have meningococcaemia? If in doubt, the safest course is to take cultures, give antibiotics and admit.
Clinical investigations
Most febrile patients seen in the ED justify a fever work-up.
Full blood examination is of limited use. White cell count (>15×109/L), marked left shift, neutropaenia or thrombocytopaenia are pointers to a possible bacteraemia or occult bacterial infections, but they may also be seen in viral infections [12]. Similarly, non-specific markers of inflammation, such as C-reactive protein and erythrocyte sedimentation rate, have not been shown to be useful in predicting outcomes for febrile patients in the ED [13].
Urinalysis and urine culture should be done in febrile adults over the age of 50 unless the pathology clearly lies in another body system. However, if the history does not suggest urinary sepsis and the dipstick urinalysis is normal, then urine cultures are usually negative [14].
A chest X -ray is usually indicated unless a definite diagnosis has been made, e.g. chickenpox, tonsillitis.
Blood cultures should be done in anyone suspected of having bacteraemia, endocarditis or meningitis, in compromised patients with a fever, all febrile patients over the age of 50 and, possibly, in anyone with an unexplained high fever. It should be noted that only 5% of blood cultures in this setting will be positive and less than 2% will alter clinical management [15]. In general, a patient considered ‘sick enough’ to warrant blood cultures should be admitted to hospital or followed up within 24 hours.
Disposition
Patients who have any of the following features are in need of resuscitation, followed by work-up and admission: shock, coma/stupor, cyanosis, profound dyspnoea, continuous seizures and severe dehydration.
With few exceptions, the following groups of febrile adults should be investigated and admitted:
![]() those over 50 years of age
those over 50 years of age
![]() patients with diabetes mellitus
patients with diabetes mellitus
![]() alcoholic patients
alcoholic patients
![]() injecting drug users
injecting drug users
![]() immunologically compromised patients
immunologically compromised patients
![]() overseas travellers or visitors
overseas travellers or visitors
![]() those with ‘alarm bells’ as described in Step 3.
those with ‘alarm bells’ as described in Step 3.
In general, there should be close liaison with the admitting unit and the issue of empirical therapy for septic patients should be discussed. For the dangerously ill, e.g. those with septic shock or bacterial meningitis, antibiotics should be commenced almost immediately.
There is an increasing tendency to start antibiotics in the ED as soon as possible to reduce the length of hospital stay. Time to antibiotic therapy is used as a key performance indicator for the ED, e.g. for febrile immunocompromised patients.
Patients who do not require intervention after the basic work-up in the ED are discharged home after a period of observation. Because of the time taken to interview the patient, perform investigations and wait for the results, the patient will usually have been observed for 1–2 hours and progression or lack of progression may be a help in deciding what to do. During observation one must be aware that the apparent improvement of the patient may be the result of pain relief or a fall in temperature due to antipyretics.
Arrangement must be made for the patient to be reviewed by their general practitioner or at the hospital. This is an essential component of the care of a febrile patient seen in an ED.
There is no easy way of detecting occult bacterial sepsis. The infectious process is a dynamic one and the doctor must maintain contact with the patient or family during the 24–72 hours following the initial visit.
Patients with fever>39°C must be seen within 24 hours. Review by a doctor within 6–12 hours may be necessary in those who have had a lumbar puncture and is advisable in those who have had blood cultures taken. A verified phone number should be clearly recorded in the medical history.
All febrile patients discharged from the ED should be encouraged to seek review if there is any adverse change to their condition. A patient re-presenting to the ED has provided an opportunity to ensure that they are being managed appropriately and to rectify any errors.
Fever due to most common viral infections will resolve by about 4 days. Many other infections will be diagnosed when new symptoms or signs appear.
If fever persists beyond 4–5 days without any localizing symptoms or signs, a less common infection or non-infective cause should be suspected and the patient should be thoroughly investigated. In this situation, the threshold of admission to hospital should be low.
The establishment of ED short-stay units allows fast-track treatment and observation, usually for 24–48 hours, for carefully selected febrile patients who are not suitable for immediate discharge home.
Future research directions
![]() The subject of undifferentiated fever of short duration in the adult has not been well studied. There are few data on the spectrum of diseases producing this clinical problem.
The subject of undifferentiated fever of short duration in the adult has not been well studied. There are few data on the spectrum of diseases producing this clinical problem.
Controversies
![]() Whether empirical antibiotics should be given to adult patients with undifferentiated fever of short duration in order to minimize the risk of death from unrecognized sepsis or meningitis is a perennial question and there are no algorithms capable of directing management of this problem.
Whether empirical antibiotics should be given to adult patients with undifferentiated fever of short duration in order to minimize the risk of death from unrecognized sepsis or meningitis is a perennial question and there are no algorithms capable of directing management of this problem.
![]() The safe and ideal course of action is to admit for observation all those patients who are ill enough to warrant a blood culture or a lumbar puncture. The limitation of hospital beds precludes this policy and there will be unnecessary admissions. The introduction of ED short-stay units provides an alternative for selected patients.
The safe and ideal course of action is to admit for observation all those patients who are ill enough to warrant a blood culture or a lumbar puncture. The limitation of hospital beds precludes this policy and there will be unnecessary admissions. The introduction of ED short-stay units provides an alternative for selected patients.
References
1. Mellors JW, Horowitz RI, Harvey MR, et al. A simple index to identify occult bacterial infection in adults with acute unexplained fever. Arch Intern Med. 1987;147:666–671.
2. Gallagher EJ, Brooks F, Gennis P. Identification of serious illness in febrile adults. Am J Emerg Med. 1994;12:129–133.
3. Norman DC, Yoshikawa TT. Fever in the elderly. Infect Dis Clin N Am. 1996;10:93–99.
4. Fontanarosa PB, Kaeberlein FJ, Gerson FW, et al. Difficulty in predicting bacteraemia in elderly emergency patients. Ann Emerg Med. 1992;21:842–848.
5. Marco CA, Schoenfeld CN, Hansen KN, et al. Fever in geriatric emergency patients: clinical features associated with serious illness. Ann Emerg Med. 1995;26:18–24.
6. Tan SL, Knott JC, Street AC, et al. Outcomes of febrile adults presenting to the emergency department. Emerg Med. 2002;14:A22.
7. Wrenn KD, Larson S. The febrile alcoholic in the emergency department. Am J Emerg Med. 1991;9:57–60.
8. Marantz PR, Linzer M, Feiner CJ. Inability to predict diagnosis in febrile intravenous drug abusers. Ann Intern Med. 1987;106:823–826.
9. Samet JH, Shevitz A, Fowle J, et al. Hospitalisation decisions in febrile intravenous drug users. Am J Med. 1990;89:53–57.
10. Beigel JH, Farrar J, Han AM, et al. Avian influenza A (H5N1) infection in humans. N Engl J Med. 2005;353:1374–1385.
11. Ufberg JW, Karras DJ. Commentary (viral haemorrhagic fever). Ann Emerg Med. 2005;45:324–326.
12. Wasserman MR, Keller EL. Fever white blood cell count, and culture and sensitivity: their value in the evaluation of the emergency patient. Top Emerg Med. 1989;10:81–88.
13. Van Laar PJ, Cohen J. A prospective study of fever in the accident and emergency department. Clin Microbiol Infect. 2003;9:878–880.
14. Sultana RV, Zalstein S, Cameron PA, et al. Dipstick urinalysis and the accuracy of the clinical diagnosis of urinary tract infection. J Emerg Med. 2001;20:13–19.
15. Kelly A. Clinical impact of blood cultures in the emergency department. J Acad Emerg Med. 1998;15:254–256.
Further reading
1. Talan DA. Infectious disease issues in the emergency department. Clin Infect Dis. 1996;23:1–14.
9.2 Meningitis
Andrew Singer
Essentials
1 Bacterial meningitis can be a rapidly progressive and fatal illness. A high level of suspicion is necessary, as well as rapid diagnosis and treatment.
2 Eighty-five per cent of cases have headache, fever, meningism and mental obtundation, but these are often absent or diminished in very young or old patients, those partially treated with oral antibiotics and those with some form of immunocompromise.
3 Treatment should not be delayed if lumbar puncture cannot be performed within 20 minutes of arrival in the emergency department. Blood cultures should be taken prior to the first dose of antibiotics, if at all possible.
4 The combination of a third-generation cephalosporin and benzylpenicillin will treat most cases of suspected bacterial meningitis and should be given as soon as the diagnosis is suspected (benzyl penicillin is sufficient in the pre-hospital setting).
5 Steroids are potentially of benefit to both adults and children with bacterial meningitis, reducing the incidence of deafness and other neurological complications in Haemophilus influenzae and Streptococcus pneumoniae infections. They should be given either before or with the first dose of antibiotic.
Introduction
Definition
Meningitis is an inflammation of the leptomeninges, the membranes that line the central nervous system, as well as the cerebrospinal fluid (CSF) in the subarachnoid space. It is usually the result of an infection, but can be due to an inflammatory response to a localized or systemic insult.
Classification
Meningitis is usually classified according to the aetiology or location as bacterial, aseptic (viral, tuberculous, fungal or chemical) or spinal (where the infection specifically affects the spinal meninges).
Aetiology
Bacterial
Bacterial meningitis is a serious cause of morbidity and mortality in all age groups. The causes vary according to age, as shown in Table 9.2.1. Neisseria meningitidis serogroups A and C tend to cause endemic cases of meningitis, especially in Aboriginal populations, whereas serogroup B is more commonly associated with epidemics [1]. There has been an increase in the incidence of penicillin-resistant Streptococcus pneumoniae, especially in children [2].
Table 9.2.1
Causes of meningitis
|
Viral |
Bacterial |
Other |
|
Echovirus 6, 9,11, 30 |
Neonates (<3 months old): |
Mycobacterium tuberculosis |
|
Coxsackie viruses A9, A16, B1, B5, B6 |
Group B streptococcus (Streptococcus agalactiae) |
Cryptococcus neoformans (especially in immunocompromised) Aseptic |
|
Cytomegalovirus Varicella zoster Epstein-Barr virus |
Coagulase-negative Staphylococcus aureus |
|
|
Children (<6 years old): |
||
|
Haemophilus influenzae type b |
||
|
Neisseria meningitidis |
||
|
Streptococcus pneumoniae |
||
|
Adults |
||
|
Neisseria meningitidis (especially in young adults) |
||
|
Streptococcus pneumoniae |
||
|
Listeria monocytogenes (especially in adults over 50) |
||
|
Klebsiella pneumoniae |
||
|
Staphylococcus aureus |
||
|
Escherichia coli (in the immunocompromised) |
Aseptic
Aseptic meningitis may be either due to an immune response to a systemic infection (usually viral) or to a chemical insult.
Viral
Enteroviruses are the most common cause of meningitis, often in clusters of cases. Herpes viruses often cause meningitis as part of a more generalized infection of the brain (meningoencephalitis) or as part of an immune response to a systemic infection. A generalized viraemia may also cause aseptic meningitis, owing to an immune reaction without direct infection.
Fungal
Fungal causes of meningitis, especially that due to Cryptococcus neoformans, tend to occur in immunocompromised patients, such as those with HIV/AIDS or those on immunosuppressant medication or cancer chemotherapy. It can occur in immunocompetent individuals as well, particularly the elderly.
Tuberculous
Tuberculous meningitis is rare in industrialized countries, but can occur in all age groups. It tends to follow an insidious course, with a lack of classic signs and symptoms. Diagnosis is often difficult, owing to the low yield from CSF staining and the 4-week time frame required to culture the organism. Suspicion should be high in patients with immunocompromise or chronic illness. It tends to have a high mortality.
Spinal
Spinal meningitis is usually bacterial and due to direct spread from a localized infection in the spine.
Epidemiology
The epidemiology of meningitis is different for groups according to age, as well as immunocompetence:
![]() Neonates: Table 9.2.1 shows the main causes of bacterial meningitis in neonates. There is an overall incidence of 0.17–0.32 cases per 1000 live births. There is 26% mortality, which is even higher in premature infants [3].
Neonates: Table 9.2.1 shows the main causes of bacterial meningitis in neonates. There is an overall incidence of 0.17–0.32 cases per 1000 live births. There is 26% mortality, which is even higher in premature infants [3].
![]() Children: until the introduction of Haemophilus influenzae type b (Hib) immunization in the early 1990 s, this organism was the major cause of bacterial meningitis in children under 5 years (until 1990, the incidence of childhood Hib meningitis was 26.3 per 100 000 [152 per 100 000 in Aboriginal children]) [4]. Between 1990 and 1996 there was a 94% reduction in the incidence of Hib disease. N. meningitidis and S. pneumoniae remain common causes of both meningitis and generalized sepsis [5]. The introduction of immunization programmes for some strains of both of these bacteria will reduce the incidence of meningitis caused by these organisms in the future, although it is important to understand that not all strains are covered by vaccines.
Children: until the introduction of Haemophilus influenzae type b (Hib) immunization in the early 1990 s, this organism was the major cause of bacterial meningitis in children under 5 years (until 1990, the incidence of childhood Hib meningitis was 26.3 per 100 000 [152 per 100 000 in Aboriginal children]) [4]. Between 1990 and 1996 there was a 94% reduction in the incidence of Hib disease. N. meningitidis and S. pneumoniae remain common causes of both meningitis and generalized sepsis [5]. The introduction of immunization programmes for some strains of both of these bacteria will reduce the incidence of meningitis caused by these organisms in the future, although it is important to understand that not all strains are covered by vaccines.
![]() Adults: N. meningitidis and S. pneumoniae are common causes in all age groups, with N. meningitides predominating in adults under 24 years. Listeria monocytogenes is more common in adults over 50 years. The overall incidence in adults is 3.8 per 100 000 population [6]. More unusual organisms occur in patients following neurosurgery or chronic illness, such as alcoholism, hepatic cirrhosis, chronic renal failure and connective tissue disease [7] (GNRs, coagulase-negative Staphylococcus aureus, Mycobacterium tuberculosis, Klebsiella pneumoniae).
Adults: N. meningitidis and S. pneumoniae are common causes in all age groups, with N. meningitides predominating in adults under 24 years. Listeria monocytogenes is more common in adults over 50 years. The overall incidence in adults is 3.8 per 100 000 population [6]. More unusual organisms occur in patients following neurosurgery or chronic illness, such as alcoholism, hepatic cirrhosis, chronic renal failure and connective tissue disease [7] (GNRs, coagulase-negative Staphylococcus aureus, Mycobacterium tuberculosis, Klebsiella pneumoniae).
![]() Patients with HIV/AIDS: Cryptococcus neoformans is relatively common, with an incidence of 5 per million of population or 10% of HIV-infected patients. Tuberculosis, Listeria, Klebsiella and syphilis are also causes of meningitis in this group, as well as viral causes of meningoencephalitis [8].
Patients with HIV/AIDS: Cryptococcus neoformans is relatively common, with an incidence of 5 per million of population or 10% of HIV-infected patients. Tuberculosis, Listeria, Klebsiella and syphilis are also causes of meningitis in this group, as well as viral causes of meningoencephalitis [8].
![]() Tuberculous meningitis occurs in around 2% of patients with TB and around 10% of HIV-infected patients with TB. It has a poor prognosis, with 20% mortality.
Tuberculous meningitis occurs in around 2% of patients with TB and around 10% of HIV-infected patients with TB. It has a poor prognosis, with 20% mortality.
Pathogenesis
Initially, there is colonization of the infectious agent, commonly in the nasopharynx in the case of the enteroviruses and bacteria, such as meningococcus and Hib. Other infections may spread from already established foci, such as otitis media or sinusitis (e.g. pneumococcus). There is either haematogenous or local spread to the meninges and subarachnoid space, with inflammation of this area and the production of a purulent exudate approximately 2 hours after invasion of the area. The inflammatory response is initiated by bacterial subcapsular components, such as lipoteichoic acid in S. pneumoniae, a lipo-oligosaccharide in H. influenzae and other Gram-negative endotoxins. These substances stimulate the release of cytokines, such as interleukin-1 and -6, tumour necrosis factor (TNF) and arachidonic acid metabolites, as well as the complement cascade. There is a subsequent increase in neutrophil and platelet activity, with increased permeability of the blood–brain barrier. This response is often worse after the initial destruction of bacteria by antibiotics. If left untreated, fibrosis of the meninges may occur. In viral and aseptic meningitis, there is a more limited inflammatory response, with mild-to-moderate infiltration of lymphocytes. In the more chronic causes, such as fungi or tuberculosis, the exudate is fibrinous, the main cells being a mixture of lymphocytes, monocytes/macrophages and plasma cells. The base of the brain is most commonly affected.
Presentation
History
There are some differences in the history with different causes of meningitis, which may allow an early differential diagnosis to be made. There are no pathognomonic single symptoms or signs for meningitis, so a high index of suspicion is necessary.
The combination of fever, headache, meningism and mental obtundation is found in approximately 85% of cases of bacterial meningitis [9]. It is also a common pattern in viral or aseptic meningitis, where obtundation is less of a feature. In fungal or tuberculous meningitis, these symptoms are much less common (less than 40% of cases of cryptococcal meningitis). Elderly patients or those who have had recent neurosurgery may present with subtle or mild symptoms and lack a fever [10].
The headache is usually severe and unrelenting. It may be either global or located in a specific area. The main symptoms of meningism are nuchal rigidity (neck stiffness) and photophobia. The nuchal rigidity is something more than merely pain on movement of the neck. It is clinically important when the patient complains of a painful restriction of movement in the sagittal plane (i.e. forwards and backwards only). Up to 35% of cases have associated nausea and vomiting.
As a general rule, the height of the fever is a poor indication of the possible cause, although the fever may often only be mild in tuberculous or fungal meningitis or in bacterial meningitis that has been partially treated by antibiotics. The spectrum of mental obtundation can range from mild confusion, to bizarre behaviour, delirium or coma. The severity of obtundation is a good indication of the severity of the illness.
Focal neurological signs occur in around 10–20% of cases of bacterial meningitis, but are also associated with cerebral mass lesions, such as toxoplasmosis or brain abscess. They are also a feature of tuberculous meningitis. Seizures are relatively uncommon (13–30%), but may occasionally be the only sign of meningitis if the patient has been partially treated with oral antibiotics.
There may also be associated systemic symptoms. Myalgias and arthralgias are often associated with viral causes, but may also be the sole presenting symptom in meningococcal meningitis. HIV/AIDS patients may show stigmata associated with that disease.
The course of the illness may also indicate the cause. Meningococcal or pneumococcal meningitis is often characterized by a rapid, fulminating course, often going from initial symptoms to death over an interval of hours. Viral causes tend to be a slower course over days. Fungal or tuberculous meningitis shows a more chronic course over days to weeks, with milder symptoms.
Risk factors for meningitis include the extremes of age, pre-existing sinusitis or otitis media, recent neurosurgery, CSF shunts, splenectomy, immunological compromise and chronic diseases such as alcoholism, cancer, connective tissue disorders, chronic renal failure and hepatic cirrhosis.
Examination
The physical examination will often reflect symptoms elicited in the history, with fever, physical evidence of meningism, stigmata of AIDS, etc.
As stated above, neck stiffness is only clinically significant when it occurs in the sagittal plane. There will be a restriction of both passive and active movement. Other tests to elicit meningism include Kernig’s sign and Brudzinski’s sign, although these are only present in 50% of adult cases of bacterial meningitis. Kernig’s sign is elicited by attempting to extend the knee of a leg that has been flexed at the hip with the patient lying supine and the other leg flat on the bed. The sign is positive if the knee cannot be fully extended due to spasm in the hamstrings. The test can be falsely positive in patients with shortening of the hamstrings or other problems involving the legs or lumbar spine. In Brudzinski’s sign, flexing the head causes the thighs and knees to also flex. It can also be tested in children by the inability to touch the nose with the flexed hips and knees in the sitting position. These are both late signs.
Focal neurological signs should be a cause for concern, as they can indicate a poor prognosis.
Papilloedema is rare and late, as is a bulging fontanelle in infants and should alert one to alternative diagnoses.
A rash, often starting as a macular or petechial rash on the limbs, is seen in sepsis due to N. meningitidis and S. pneumoniae. A petechial rash is a particularly serious sign and is an indication to start antibiotics immediately. A maculopapular rash is also a feature of viral causes.
Investigations
Lumbar puncture
A CSF sample via a lumbar puncture (LP) is an important source of information for making the diagnosis and determining the likely aetiology and treatment. As the procedure may be time-consuming, treatment should not be delayed if there will be more than a 20-minute delay before the lumbar puncture and there is a reasonable clinical suspicion that a bacterial cause is present. Blood cultures should be taken prior to the administration of antibiotics.
Indications
![]() Symptoms suggestive of meningitis, especially the combination of fever, headache, neck stiffness and photophobia.
Symptoms suggestive of meningitis, especially the combination of fever, headache, neck stiffness and photophobia.
![]() Any patient with fever and an altered level of consciousness.
Any patient with fever and an altered level of consciousness.
![]() Fever associated with seizures, especially in a neonate, older child or adult.
Fever associated with seizures, especially in a neonate, older child or adult.
![]() Seizures in any patient who has been on oral antibiotics.
Seizures in any patient who has been on oral antibiotics.
Precautions
![]() Deep coma: a patient with a Glasgow coma score (GCS) of 8 or less should have the lumbar puncture delayed until they are more conscious. A normal brain computed tomography (CT) scan does not exclude the risk of herniation in this group.
Deep coma: a patient with a Glasgow coma score (GCS) of 8 or less should have the lumbar puncture delayed until they are more conscious. A normal brain computed tomography (CT) scan does not exclude the risk of herniation in this group.
![]() Focal neurological signs: the patient should have CT first, to exclude a space-occupying lesion, which may increase the risk of cerebral herniation following the lumbar puncture.
Focal neurological signs: the patient should have CT first, to exclude a space-occupying lesion, which may increase the risk of cerebral herniation following the lumbar puncture.
![]() Surgery to the lumbar spine.
Surgery to the lumbar spine.
![]() Local skin infection around the lumbar spine.
Local skin infection around the lumbar spine.
The main features to note during lumbar puncture are the opening pressure and the physical appearance of the CSF. The sample should be sent for Gram staining, culture, sensitivities, polymerase chain reaction (PCR) analysis for bacteria and Herpes simplex virus, a cell count and protein and glucose levels. If fungal meningitis is suspected, an India-ink stain and cryptococcal antigen screen should be requested. If tuberculous meningitis is suspected, multiple 5 mL samples of CSF will be required to increase the likelihood of a positive result. If there has been prior administration of antibiotics, a bacterial antigen screen should also be requested.
Turbid CSF is indicative of a significant number of pus cells and is an indication for immediate administration of antibiotics. The patient should usually rest supine for a few hours after the procedure to prevent a worsening of the headache. This has been known to occur up to 24 hours following the procedure. The evidence for the benefits of enforced rest after lumbar puncture is equivocal.
The pattern of cell counts and glucose and protein levels is shown in Table 9.2.2. This can act as a guide only and the clinician needs to be guided by the complete clinical picture.
Table 9.2.2
Expected CSF values in meningitis

A leucocyte count (WCC) of more than 1000/μL with a predominantly neutrophilic pleocytosis is considered positive for bacterial meningitis. Ten per cent of cases, especially early in the course of the illness, may have a predominance of lymphocytes. As a general rule, bacterial meningitis is characterized by a raised CSF protein and a low CSF glucose level. The ratio of CSF to serum glucose levels is also lowered. The combination of CSF glucose<1.9 mmol/L, CSF to serum glucose ratio<0.23, CSF protein>2.2 g/L and either a total WCC>2000/μL or a neutrophil count of>1180/μL has been shown to have a 99% certainty of diagnosing bacterial meningitis [11]. Aseptic meningitis will often have cell counts near the normal range. This does not exclude infection with less common agents, such as herpes viruses or L. monocytogenes.
CT scan
CT scanning of the brain is indicated as a prelude to lumbar puncture in the presence of focal neurological signs, mental obtundation or abnormal posturing. It must be noted though, that a normal CT does not exclude the risk of cerebral herniation in bacterial meningitis [12] and, therefore, those with the above signs should have lumbar puncture delayed until they are conscious and stable.
Microbiology
Apart from microscopy and culture of CSF, there are a number of other methods that may allow the causative organism to be identified.
Skin lesion aspirate
In cases where a petechial rash is present, Gram staining or culture from some of the skin lesions may yield the causative organism. This has a reported sensitivity of 30–70%.
Throat swab
Throat swabs are useful in identifying a bacterial cause spread by nasopharyngeal carriage and should be performed in a case of suspected bacterial meningitis.
Polymerase chain reaction
This potentially allows identification of the causative organism and even the serotype for organisms, such as meningococcus. The test can be performed on CSF or EDTA blood samples and may remain positive for up to 72 hours after the commencement of antibiotics. In CSF, the reported sensitivity is 89% with a specificity of 100% and in blood a sensitivity of 81% with a specificity of 97% [13].
Serology
Tests to detect IgM to specific organisms are available for meningococcus and some viruses. For meningococcus, the test has a sensitivity and specificity of 97% and 95%, but is only reliable in adults and children over 4 years old and takes 5–7 days after onset of the illness to reach diagnostic levels.
Antigenic studies
Latex agglutination, immunoelectrophoresis or radioimmunoassay techniques can be used to screen for antigens from S. pneumoniae, Hib, group B streptococcus (S. agalactiae), Escherichia coli K1, N. meningitidisand C. neoformans. The tests can be performed on serum, CSF or urine. Serum or urine samples tend to allow greater sensitivities (around 96–99%) than CSF (82–99%). The test is no more sensitive in untreated cases than either a positive Gram-stain or the presence of CSF pleocytosis [14]. The main purpose of antigenic studies is in allowing rapid identification of the causative organism in cases confirmed by the CSF findings or in cases where partial treatment with antibiotics renders the CSF sterile on culture. In many laboratories, these tests have been superseded by PCR methods.
General investigations
Full blood count (FBC), urea and electrolyte counts (UEC), blood cultures, erythrocyte sedimentation rate (ESR) and a throat swab can assist in building an overall picture.
Blood cultures should be taken prior to parenteral antibiotics, especially in patients where lumbar puncture has been delayed. One study found that blood cultures grew the causative organism in 86% of proven cases of bacterial meningitis and that the combination of blood culture, CSF Gram staining and antigen testing identified the cause in 92% of cases [15].
Differential diagnosis
![]() Generalized viral infections, with meningism as a component.
Generalized viral infections, with meningism as a component.
![]() Encephalitis: this is a more generalized viral infection of the brain. Clinically, there may be no difference.
Encephalitis: this is a more generalized viral infection of the brain. Clinically, there may be no difference.
![]() Brain abscess: this tends to produce focal signs due to local pressure at the site of the abscess.
Brain abscess: this tends to produce focal signs due to local pressure at the site of the abscess.
![]() Focal cerebral infections, such as those due to Toxoplasma gondii in HIV/AIDS patients.
Focal cerebral infections, such as those due to Toxoplasma gondii in HIV/AIDS patients.
![]() Subarachnoid haemorrhage: this will often produce identical symptoms of meningism, but generally without any other evidence of infection, such as fever.
Subarachnoid haemorrhage: this will often produce identical symptoms of meningism, but generally without any other evidence of infection, such as fever.
![]() Migraine and other vascular headaches: again, meningism is a similar feature. The patient will often have a known history of the illness.
Migraine and other vascular headaches: again, meningism is a similar feature. The patient will often have a known history of the illness.
![]() Severe pharyngitis with cervical lymphadenopathy causing neck stiffness.
Severe pharyngitis with cervical lymphadenopathy causing neck stiffness.
Management
Management depends on the likely causative agents, as well as the severity of the illness.
General
Patients should rest in bed, particularly following a lumbar puncture. A quiet, darkened room will be beneficial to those with headache or photophobia. Simple analgesics may be used to treat the headache, with or without codeine. Opiates may be required in severe headache.
Sedation may be necessary if the patient is very agitated or delirious. Suitable drugs are diazepam 5–10 mg IV or midazolam 2–10 mg IV or IM, with or without the addition of an antipsychotic, such as haloperidol 5–20 mg IV or IM, or chlorpromazine 12.5–50 mg IV or IM.
Seizures should be treated appropriately, initially with a benzodiazepine, then maintenance with phenytoin or phenobarbitone. Meningitis can occasionally be associated with status epilepticus, which should be treated in the standard way.
Patients with raised intracranial pressure may need pressure monitoring and measures to reduce the pressure, such as nursing the patient 30° head up and the administration of hyperosmotic agents, such as mannitol. Hyperventilation is controversial as it may reduce intracerebral pressure at the expense of reduced cerebral perfusion. Obstructive hydrocephalus requires appropriate neurosurgical treatment with CSF shunting.
If septic shock has intervened, it should be treated in the usual way, with IV fluids and inotropes.
Antimicrobials
The choice of antimicrobial agent will be determined by the likely causative organism and is therefore determined primarily by age and immune status. It is important that antibiotic therapy is not delayed by investigations such as lumbar puncture or CT and should be administered as soon as the diagnosis is made. Table 9.2.3 shows the recommended choice of antimicrobial for different situations and organisms. Table 9.2.4 shows the recommended dosage of each. As a general rule, the combination of a third-generation cephalosporin and benzylpenicillin will cover most organisms in all age groups. It is important to note that there is emerging resistance to penicillins in S. pneumoniae (currently 7.6% of isolates in Australia). If Gram-positive diplococci are found or S. pneumoniae is identified on antigen or PCR testing, vancomycin should be added to the therapy.
Table 9.2.3
Choice of antimicrobial in meningitis [16]

After eTG complete [internet]. Melbourne: Therapeutic Guidelines Limited; 2013 July with permission.
Table 9.2.4
Antibiotic doses in treating meningitis [17]

After eTG complete [internet]. Melbourne: Therapeutic Guidelines Limited; 2013 July with permission.
Steroids
Steroids have been shown to improve the prognosis of bacterial meningitis in both adults and children. There is a reduction in complications, such as sensorineural deafness and short-term neurological deficits (in high-income countries). The most benefit appears to be derived with infections from H. influenzae and S. pneumoniae. No clear mortality benefit has been established. Steroids are usually administered as dexamethasone 0.15 mg/kg IV q 6 h (up to 10 mg), started before or with the first dose of antibiotics and continued for 4 days. The main adverse effect is gastrointestinal bleeding, which may be reduced by limiting treatment to 2 days [17].
Disposition
All cases of bacterial meningitis require admission for IV antibiotics, as well as supportive therapy. They often require intensive therapy, especially if septic shock has supervened. Viral meningitis will usually require supportive therapy only, but this may require admission. Mild cases of viral or aseptic meningitis, with a clear diagnosis, can be safely sent home.
Prognosis
Over the last 20 years, the mortality of bacterial meningitis has ranged from 6 to 20% and is higher in the very young or the very old. Meningitis in immunocompromised individuals carries a high mortality of up to 50%. Bacterial meningitis in children can lead to a number of long-term sequelae, such as sensorineural hearing loss, learning difficulties, motor problems, speech delay, hyperactivity, blindness, obstructive hydrocephalus and recurrent seizures. These sequelae are less common in adults.
Prevention
Prophylaxis should be offered in cases of H. influenzae type b, or Meningococcus infection to:
![]() the index case
the index case
![]() all household or childcare contacts who have either stayed overnight in the same house or have been in the same room as the index case for any period of 4 hours or more in the preceding 7 days (in Hib, if less than 24 months old or less than 4 years and incompletely immunized against Hib)
all household or childcare contacts who have either stayed overnight in the same house or have been in the same room as the index case for any period of 4 hours or more in the preceding 7 days (in Hib, if less than 24 months old or less than 4 years and incompletely immunized against Hib)
![]() passengers adjacent to the index case on a trip of 8 hours’ or longer duration
passengers adjacent to the index case on a trip of 8 hours’ or longer duration
![]() any person who has potentially shared saliva (such as eating utensils or drink bottles) with the index case
any person who has potentially shared saliva (such as eating utensils or drink bottles) with the index case
![]() healthcare workers who have given mouth-to-mouth resuscitation to an index case
healthcare workers who have given mouth-to-mouth resuscitation to an index case
![]() Appropriate regimens are:
Appropriate regimens are:
![]() for meningococcus:
for meningococcus:
■ ciprofloxacin 500 mg orally as a single dose – preferred for females on oral contraceptives
■ ceftriaxone 250 mg (125 mg in children<12 years IM in 1% lignocaine – preferred in pregnant women
■ rifampicin 600 mg orally 12-hourly for 2 days (5 mg/kg in neonates<1 month, 10 mg/kg in children).
![]() for Hib:
for Hib:
■ rifampicin 600 mg orally daily for 4 days (10 mg/kg in neonates<1 month, 20 mg/kg in children)
■ ceftriaxone 1 g IM daily for 2 days (50 mg/kg in children)
■ if the index case is<24 months old, Hib vaccination should be given as a full course as soon as possible after recovery. Unvaccinated contacts under 5 years of age should be immunized as soon as possible.
Casual, neighbourhood or hospital contacts are not required to receive prophylaxis.
Meningococcal vaccine should be considered in populations where cases are clustered. The vaccine is currently only available for serogroup C.
Controversies
![]() Whether all patients should have a CT scan before lumbar puncture. In general, it is safe without CT in those with a clear history consistent with meningitis and normal sensorium. Comatose patients should have lumbar puncture delayed until they are conscious.
Whether all patients should have a CT scan before lumbar puncture. In general, it is safe without CT in those with a clear history consistent with meningitis and normal sensorium. Comatose patients should have lumbar puncture delayed until they are conscious.
![]() The use of steroids. Steroids have been shown to improve hearing and short-term neurological outcome in adults and children in high-income countries, but are known to cause adverse outcomes in patients with generalized sepsis.
The use of steroids. Steroids have been shown to improve hearing and short-term neurological outcome in adults and children in high-income countries, but are known to cause adverse outcomes in patients with generalized sepsis.
References
1. Munro R, Kociuba K, Jelfs J, et al. Meningococcal disease in urban south western Sydney, 1990–1994. Aust NZ J Med. 1996;26:526–532.
2. Collignon PJ, Bell JM. Drug-resistant Streptococcus pneumoniae: the beginning of the end for many antibiotics? Australian Group on Antimicrobial Resistance. Med J Aust. 1996;164:64–67.
3. Francis BM, Gilbert GL. Survey of neonatal meningitis in Australia: 1987–1989. Med J Aust. 1992;156:240–243.
4. Bower C, Payne J, Condon R, et al. Sequelae of Haemophilus influenzae type b meningitis in Aboriginal and non-Aboriginal children under 5 years of age. J Paediatr Child Hlth. 1994;30:393–397.
5. Herceg A. The decline of Haemophilus influenzae type b disease in Australia. Commun Dis Intell. 1997;21:173–176.
6. Sigurdardottir B, Bjornsson OM, Jonsdottir KE. Acute bacterial meningitis in adults A 20-year overview. Arch Intern Med. 1997;157:425–430.
7. Segreti J, Harris AA. Acute bacterial meningitis. Infect Dis Clin N Am. 1996;10:797–809.
8. Jones PD, Beaman MH, Brew BJ. Managing HIV Part 5: Treating secondary outcomes 5.5 HIV and opportunistic neurological infections. Med J Aust. 1996;164:418–421.
9. Tunkel AR, Scheld WM. Acute bacterial meningitis. Lancet. 1995;346:1675–1680.
10. Miller LG, Choi C. Meningitis in older patients: how to diagnose and treat a deadly infection. Geriatrics. 1997;52:43–44.
11. Spanos A, Harrell Jr FE, Durack DT. Differential diagnosis of acute meningitis: an analysis of the predictive value of initial observation. J Am Med Assoc. 1989;262:2700–2707.
12. Rennick G, Shann F, de Campo J. Cerebral herniation during bacterial meningitis in children. Br Med Jo. 1993;306:953–955.
13. Communicable Diseases Network Australia, Australian Government Department of Health and Ageing. Guidelines for the early clinicaI and public health management of Meningococcal Disease in Australia. The 2007 revision of the document is available at:<http://www.health.gov.au/internet/main/publishing.nsf/Content/cda-pubs-other-mening-2007.htm>.
14. Feuerborn SA, Capps WI, Jones JC. Use of latex agglutination testing in diagnosing pediatric meningitis. J Fam Pract. 1992;34:176–179.
15. Coant PN, Kornberg AE, Duffy LC, et al. Blood culture results as determinants in the organism identification of bacterial meningitis. Pediatr Emerg Care. 1992;8:200–205.
16. Therapeutic Guidelines Limited. Therapeutic Guidelines, Antibiotic, version 14, 2010.
17. Brouwer MC, McIntyre P, de Gans J. Corticosteroids for acute bacterial meningitis. Cochrane Database Syst Rev. 2010;9:CD004405.
9.3 Septic arthritis
Trevor Jackson and Varadarajulu Suresh
Essentials
1 Delayed or inadequate treatment can lead to irreversible joint damage.
2 Diagnosis is based on clinical features and synovial fluid examination; imaging techniques have a role in difficult cases.
3 Staphylococcus aureus and Neisseria gonorrhoeae are the most frequent pathogens.
4 Successful treatment hinges on rapid and complete joint drainage and high-dose parenteral antibiotics guided by culture results.
5 Outcomes are good in paediatric and gonococcal subgroups, but the presence of chronic arthritis or polyarticular involvement is associated with up to 15% mortality and 50% chronic joint morbidity.
Introduction
Septic arthritis is defined as bacterial infection of the synovial space. The knee is the most commonly affected joint in adults and the hip joint in the paediatric age group [1].
Aetiology, pathogenesis and pathology
Septic arthritis can be caused by haematological spread or direct invasion. Bacteria are the usual pathogens by haematogenous seeding of the joint. Direct spread from adjacent infection or via trauma are less common. Once inside the joint, bacterial growth and invasion can occur uninhibited. Phagocytic and neutrophil responses to the bacteria lead to proteolytic enzyme release and cytokine production, resulting in synovial abscess formation and cartilage necrosis [2]. Co-morbidity or deficient host defences are risk factors for infection [3] and can be associated with more rapid and severe disease (Table 9.3.1).
Table 9.3.1
Risk factors for septic arthritis

The majority of cases are community acquired and occur in children and young adults [4]. Prosthetic joint surgery and invasive management of chronic arthritis are factors in the increased prevalence observed in older age groups.
Epidemiology
The incidence of proven and probable septic arthritis in Western Europe is 4–10 per 1 00 000 patients per year. This is more in lower socioeconomic groups in both Northern Europe and Australia.
The prevalence is 29 cases per 1 00 000 of the Aborigine population with a relative risk of 6.6 compared with the white Northern Territory Australian population.
The incidence of septic arthritis is increasing and is linked to an increase in orthopaedic-related infection, an ageing population, more invasive procedures being under taken and an enhanced use of immunosuppressive treatment [5].
Clinical features
History
This will usually reveal the recent onset of a painful, hot and swollen joint, most commonly the hip or knee, although any joint may be affected. Systemic features of fever or rigors should be sought, plus the presence of any risk factors.
Examination
Typical findings include a hot, tender joint with marked limitation of passive or active movement owing to pain. An effusion will be evident in most cases. A polyarticular presentation is more common in gonococcal infection or in the setting of chronic arthritis. In general, fever is low grade and few patients will appear ‘toxic’ and unwell. The elderly and immunosuppressed may present non-specifically with anorexia, vomiting, lethargy or fever.
Differential diagnosis
Non-septic arthritis or synovitis may be differentiated on clinical features and joint fluid analysis. Fractures will generally be evident on joint radiographs, but detection of osteomyelitis may require more advanced imaging techniques, such as nuclear or computed tomography (CT) scanning. Rheumatic fever and brucellosis are rare causes.
Clinical investigations
Synovial fluid examination and culture
Aspiration should be performed promptly with local anaesthetic and a large-bore needle for cell count, crystals, Gram stain and culture to confirm the diagnosis. Typical findings in septic arthritis and its differential diagnoses are shown in Table 9.3.2[6].
Table 9.3.2
Synovial fluid characteristics

Most infections are acute and bacterial (Table 9.3.3) [6], although fungal and mycobacterial pathogens have been recognized in chronic infections.
Table 9.3.3
Bacterial causes of septic arthritis
|
Age group |
Typical bacteria |
|
Children |
Staphylococcus aureus |
|
Group A streptococci (B in neonates) |
|
|
Haemophilus influenza |
|
|
Young adults |
Neisseria gonorrhoeae |
|
Older adults |
Staphylococcus aureus |
|
Gram-negative species* |
|
|
Group A streptococci |
*Pseudomonas spp. and Enterobacteriaceae
Other laboratory investigations
Blood cultures should always be taken and may be positive in up to 50%. Inflammatory markers (erythrocyte sedimentation rate and C-reactive protein) are elevated, with typically a neutrophil-predominant leucocytosis. These are non-diagnostic, but aid in monitoring response to therapy.
Imaging studies
Plain radiographs should be performed in all cases: they may reveal effusions or local oedema and help to exclude alternative conditions. Ultrasound is very sensitive in detecting effusions and excellent for facilitating needle aspiration.
Fluoroscopy may also be used. Nuclear medical studies are very sensitive early, but not specific for sepsis. CT and magnetic resonance imaging (MRI) have a small role in difficult joints (e.g. hip and sacroiliac).
Criteria for diagnosis [7]
This depends on positive culture of synovial fluid from an affected joint, a positive Gram stain or blood culture in the context of an inflamed joint suspicious of sepsis, macroscopic pus aspirate and appropriate response to antibiotics.
Management
Joint drainage and empiric parenteral antibiotic therapy must take place without delay. Surgical drainage is usually employed in children, with needle drainage more commonly first line in adults. Newer arthroscopic techniques are increasingly being used [2,8–10]. Repeated drainage procedures will often be necessary to ensure complete resolution of the infection.
Antibiotic therapy is initiated after culture specimens have been obtained, with clinical presentation and Gram stain guiding the choice of agents. All regimens must include an antistaphylococcal agent with Gram-negative cover as indicated by the clinical setting.
Suggested initial empiric regimen [11]
Di(flu)cloxacillin: 2 g (25–50 mg/kg up to 2 g) intravenously, 6-hourly. If Gram-negative bacteria are suspected, add ceftriaxone 2 g (25–50 mg/kg up to 2 g) intravenously daily. If methicillin resistance is suspected, add vancomycin 1 g (25 mg/kg) intravenously 12-hourly.
Definitive therapy will be tailored to later laboratory identification of the organism and its sensitivities.
The duration and route of therapy remain controversial but, in uncomplicated acute cases, parenteral antibiotics will be required for at least 3 days in children and 2 weeks in adults, with a total treatment duration of 3–6 weeks [10,12]. Specific organisms, such as Neisseria spp., will respond more rapidly, whereas chronic infections and co-morbidity will necessitate aggressive and more prolonged therapy.
General care, with initial joint rest, appropriate analgesia and physical therapy, is important. All patients require admission until their joint sepsis is controlled. Thereafter, ongoing therapy may be monitored as an outpatient or via domiciliary hospital services.
Prognosis
This depends upon the organism, patient co-morbidity and the adequacy and rapidity of treatment. Gonococcal and paediatric infections have a generally good response, with low rates of ensuing joint morbidity. Polyarticular sepsis in rheumatoid arthritis has been associated with mortality rates of up to 15% and major morbidity in up to 50% of survivors [2,6,12].
Prevention
Safe sexual practice can reduce gonorrhoeal infections. Strict aseptic technique, good patient selection and prophylactic antibiotics help prevent cases associated with invasive joint procedures. The overall incidence of infection after arthroplasty ranges from 0.5 to 2% [2].
Table 9.3.4
Likely developments in future [14]

After Carpenter CR, Schuur JD, Everett WW, Pines JM. Evidence-based diagnostics: adult septic arthritis. Acad Emerg Med 2011;18:781–96.
Controversies
![]() The total duration of therapy has gradually been reduced, but optimum duration is unclear, as is the balance between parenteral and oral routes [13].
The total duration of therapy has gradually been reduced, but optimum duration is unclear, as is the balance between parenteral and oral routes [13].
![]() Consensus has not been reached on the best method of joint drainage. Surgical arthrotomy is usually employed for the hip and in children, but arthroscopic techniques are also available. Most centres still use repeated needle aspiration as first line for most joints.
Consensus has not been reached on the best method of joint drainage. Surgical arthrotomy is usually employed for the hip and in children, but arthroscopic techniques are also available. Most centres still use repeated needle aspiration as first line for most joints.
![]() Difficulties still exist with the differentiation of septic arthritis from new-onset non-septic arthritis, especially when polyarticular joint fluid analysis and medical imaging are used, but nuclear and CT scanning techniques may have difficulty in distinguishing infective from non-infective inflammation.
Difficulties still exist with the differentiation of septic arthritis from new-onset non-septic arthritis, especially when polyarticular joint fluid analysis and medical imaging are used, but nuclear and CT scanning techniques may have difficulty in distinguishing infective from non-infective inflammation.
References
1. Visser S, Tupper J. Septic until proven otherwise. Can Fam Phys 55.
2. Goldenberg DL. Bacterial arthritis. In: Kelley WN, Harris ED, Ruddy S, Sledge CB, eds. Textbook of rheumatology. 4th ed. Philadelphia: WB Saunders; 1993;1449–1466.
3. Goldenberg DL. Septic arthritis. Lancet. 1998;351:197–202.
4. Sonnen GM, Henry N. Paediatric bone and joint infections. Paediatr Clin N Am. 1996;4:933–947.
5. Matthews CJ, Weston VC, Jones A. Bacterial septic arthritis in adults. Lancet. 2010;375:846–855.
6. Brooks GF, Pons VG. Septic arthritis. In: Hoeprich PD, Jordan MC, Ronald AR, eds. Infectious diseases. 5th ed. Philadelphia: JB Lippincott; 1994;1382–1389.
7. Ma L, Cranney A, Holroyd-Leduc JM. Acute monoarthritis: what is the cause of my patient’s painful swollen joint? Can Med Assoc J. 2009;180:1.
8. Stanitski CL, Harwell JC, Fu FH. Arthroscopy in acute septic knees. Clin Orthop. 1989;241:209.
9. Broy SB, Schmid FR. A comparison of medical drainage (needle aspiration) and surgical drainage (arthrotomy or arthroscopy) in the initial treatment of infected joints. Clin Rheumatol Dis. 1986;12:501–522.
10. Manadan AM, Block JA. Daily needle aspiration versus surgical lavage for the treatment of bacterial septic arthritis in adults. Am J Ther. 2004;11:412–415.
11. Skin, muscle and bone infections. In: Therapeutic guidelines. Antibiotic, 13th ed.<http://www.tg.com.au.qelibresources.health.wa.gov.au/index.php>.
12. Youssef PP, York JR. Septic arthritis: a second decade of experience. Aust NZ J Med. 1994;24:307–311.
13. Syrogiannopoulos GA, Nelson JD. Duration of antimicrobial therapy for acute suppurative osteoarticular infections. Lancet. 1988;1:37–40.
14. Carpenter CR, Schuur JD, Everett WW, Pines JM. Evidence-based diagnostics: adult septic arthritis. Acad Emerg Med. 2011;18:781–796.
9.4 Urinary tract infections
Salomon Zalstein
Essentials
1 Urinary tract infection (UTI) is the most common bacterial infection.
2 By age 32, 50% of women will report at least one UTI.
3 Sexual activity is the most important risk factor in young women.
4 Most UTIs are caused by Escherichia coli, but Staphylococcus saprophyticus is responsible for up to 15% of infections in young, sexually active women.
5 There is a genetic predisposition in some women to recurrent UTI.
6 There are specific bacterial virulence factors determining uropathogenic strains of bacteria.
7 Bacteriological diagnosis is based on isolation of 105 cfu/mL of urine. Up to half of women presenting with dysuria and frequency will have fewer than this number of bacteria and about half of these do have bacterial UTI.
8 For the majority of outpatients with typical symptoms urine culture is not necessary.
9 In hospitalized patients, urinary catheterization produces infection in 10% of patients per day.
10 In institutionalized elderly patients, non-specific symptoms or decline in function correlate poorly with UTI despite the presence of pyuria and bacteriuria. Non-UTI causes must be sought.
11 Asymptomatic bacteriuria should not be sought or treated except in pregnant women and in patients about to undergo significant urological procedures.
Introduction
Urinary tract infections are the most common bacterial infections and the major cause of Gram-negative sepsis in hospitalized patients [1,2].
Definitions
Urinary tract infection
The term urinary tract infection (UTI) is non-specific and may refer to a variety of clinical conditions, including asymptomatic bacteriuria, urethritis, cystitis, female urethral syndrome and acute and chronic pyelonephritis. The most common clinical presentations are cystitis and acute pyelonephritis, although the clinical distinction between these diagnoses may not be as straightforward as the terms imply, with up to 50% of patients having unrecognized pyelonephritis [3].
UTI is considered in two main groups: simple (or uncomplicated) and complicated. Simple UTIs occur in an otherwise healthy person with a normal urinary tract, most commonly a young non-pregnant female. A complicated UTI is one associated with anatomical abnormality, urinary obstruction or incomplete bladder emptying due to any cause: instrumentation or catheterization, pregnancy or significant underlying disease, such as immunosuppression or diabetes mellitus.
Significant bacteriuria
Significant bacteriuria most commonly refers to more than 105 bacteria/mL of urine, reported as colony forming units per mL (cfu/mL). This usually represents infection as opposed to contamination (see Quantitative culture), although there are significant exceptions to this generalization (see Urethral syndrome).
Asymptomatic bacteriuria
Asymptomatic bacteriuria (ASB) refers to significant bacteriuria in the absence of symptoms of infection.
Epidemiology
UTIs are very common, particularly in women in whom age, degree of sexual activity and the form of contraception used are all factors that affect the incidence and prevalence of infection. While the overall rate of infection is difficult to estimate since UTI is not a reportable disease, in a USA health survey, the self-reported incidence of UTI is 12.1% among women and 3% among men. By age 32, 50% of women will report at least one UTI [4]. In non-pregnant women aged 18–40 years, the rate of infection has been stated to be between 0.5 and 0.7 per person per year, with much higher rates in pregnancy [5].
In males, the prevalence of bacteriuria beyond infancy is 0.1% or less. Between the ages of 21 and 50, infection rates may be as low as 0.6–0.8/1000 [6]. With increasing prostatic disease, the frequency of bacteriuria may rise to 3.5% in healthy men and to more than 15% in hospitalized men by age 70 [7]. Homosexual men are at increased risk of UTI.
In the presence of chronic disease and institutionalization in the elderly, the incidence of bacteriuria may be as high as 50%, although this is most commonly asymptomatic [8].
Aetiology
The aetiology of uncomplicated UTI has remained unchanged for decades, although increased antibiotic resistance in the bacteria responsible has been well documented. In community-acquired UTI, Escherichia coli accounts for 75–90% of cases, Staphylococcus saprophyticus accounts for 5–15% (especially in young, sexually active women), with enterococci and Gram-negative organisms, such as Klebsiella spp. and Proteus mirabilis, responsible for 5–10% [9,10]. Which bacteria are isolated is influenced by factors such as whether the infection is initial or recurrent; the presence of obstruction, instrumentation or anatomical abnormalities; and whether the patient is an inpatient or outpatient. In simple acute cystitis, the most common presentation of UTI, a single organism is usually isolated. On the other hand, in complicated UTI, E. coli is isolated in 20–50% of cases and non-E. coli organisms, such as Proteus and Klebsiella species, are more commonly seen. In the presence of structural abnormalities, it is more common to isolate multiple organisms and antibiotic resistance is frequently found [10].
Pathogenesis
In healthy individuals, the perineum, vagina, vaginal introitus and urethra and periurethral areas each have their respective flora and are normally colonized by bacteria different from those commonly associated with UTI, that is by non-pathogens. The periurethral area may become colonized by such UTI-causing (uropathogenic) bacteria, which then ascend via the urethra into the bladder and thence may ascend further to the kidney, causing pyelonephritis. The reservoir for these bacteria is the gastrointestinal tract [4]. There are host and bacterial mechanisms involved in determining whether a UTI will occur.
Host mechanisms
Anatomic considerations (men) and prostatic secretions
In males, the length of the urethra, its separation from the anus and the presence of prostatic secretions all contribute to the prevention of colonization and subsequent UTI.
Sexual activity, contraceptive practices, use of diaphragm/spermicides
Sexual activity is the most important risk factor for acute cystitis, with recent or frequent sexual activity increasing that risk. The use of a diaphragm with a spermicide (an inhibitor of normal vaginal flora) promotes vaginal colonization with uropathogenic bacteria and has also been shown to increase the risk of UTI [4].
Secretor/non-secretor status
Blood group antigens are secreted in the body fluids by some women. The urethral and periurethral mucosae in women who do not secrete these antigens (non-secretors) in their body fluids, have a higher affinity for bacterial adhesins (see below) than the mucosae of women who do. These non-secretors are more susceptible to recurrent infections [11].
Entry of bacteria into the bladder
Instrumentation of the bladder (see below) is a well-recognized mechanism by which bacteria are introduced into the bladder. Other factors have been considered but have not been conclusively demonstrated. These include frequency and timing of voiding, hormonal changes and personal hygiene habits [12].
Bladder defence mechanisms
The healthy bladder can normally clear itself of bacteria. There are three factors involved: voiding; urinary bacteriostatic substances, such as organic acids, high urea concentrations and immunoglobulins; and active resistance by the bladder mucosa to bacterial adherence.
Obstruction
This may be extrarenal (congenital anomalies, such as urethral valves, calculi, benign prostatic hypertrophy) or intrarenal (nephrocalcinosis, polycystic kidney disease, analgesic nephropathy). Complete obstruction of the urinary tract predisposes to infection by haematogenous spread. In the absence of such obstruction, haematogenous seeding of bacteria to the kidneys accounts for about 3% of infections. Partial obstruction does not have this effect.
Vesicoureteric reflux
Incompetence of the vesicoureteric valve is a congenital problem that is five times more common in boys than in girls, but tends not to be a significant factor in adults. It allows infected urine to ascend to the kidney and is the most common factor predisposing to chronic pyelonephritic scarring.
Instrumentation
Although any instrumentation of the urinary tract predisposes to infection, catheterization is the most common of these. A single catheterization will result in UTI in 1% of ambulatory patients but, in hospitalized patients, 10% of women and 5% of men will develop a UTI after one catheterization. Once in place, catheters produce infection in up to 10% of patients per day and nearly all catheterized patients will be bacteriuric by 1 month [13]. All chronically catheterized patients are bacteriuric.
Pregnancy
Changes to the urinary tract occur normally during pregnancy as a result of both anatomical alterations and hormonal effects: dilatation of the ureters and renal pelves, decreased peristalsis in the ureters and decreased bladder tone. These changes begin before the end of the second month. The prevalence of bacteriuria rises with age and parity. A large proportion of asymptomatic bacteriuric women develop symptomatic pyelonephritis later in that pregnancy, with significant increases in toxaemia and prematurity (see Asymptomatic bacteriuria).
Diabetes mellitus
The relationship between diabetes mellitus on the one hand and asymptomatic bacteriuria and UTI on the other has been debated in the past. Current evidence indicates that asymptomatic bacteriuria is more common in diabetic women than non-diabetic women. The evidence in men is less clear cut. Good evidence from prospective studies for increased incidence of symptomatic urinary tract infection in diabetics is lacking. What appears clear is that diabetes is a significant and independent risk factor for pyelonephritis, complicated UTI, urosepsis, hospitalization and other, often rare, complications (such as emphasematous pyelonephritis, papillary necrosis and candidal infections). The precise pathogenetic mechanism is unclear but involves many factors not necessarily related to glycaemic control [14–16].
Ageing
UTI is the most frequent bacterial infection in residents of long-term care facilities. Asymptomatic bacteriuria is highly prevalent in residents of long-term care facilities with up to 30% of men and 50% of women showing such bacteriuria. The likelihood of bacteriuria correlates with the degree of functional impairment. Several factors may be involved: chronic degenerative neurological diseases may impair bladder function as well as bladder and bowel continence, prostatic enlargement in men and oestrogen deficiency in women can both lead to incomplete bladder emptying, the use of devices, such as indwelling catheters or condom drainage, predisposes to bacteriuria [8].
Bacterial factors
A number of studies [17–19] have shown that the strains of E. coli (and a number of other Gram-negative bacteria) that cause UTI are not just the most prevalent in the bowel of the patient at the time of the infection, but have specific characteristics, termed virulence factors, that give them certain capabilities: increased intestinal carriage, persistence in the vagina and the ability to ascend and invade the normal urinary tract. Thus, there are clearly uropathogenic strains of these bacteria. In cases of complicated UTI (e.g. those associated with reflux, obstruction or foreign body), these virulence factors are not significantly involved.
Presentation
History
A careful history should be taken in any patient presenting with symptoms of apparent UTI, looking for risk factors for complicated or recurrent infection (such as previous UTIs and their treatment, the presence of known anatomical abnormalities and investigations or instrumentation, the possibility of pregnancy and history of diabetes mellitus), as well as seeking to identify those patients with urethritis and vaginitis. In men, the most common cause of recurrent lower tract UTI is prostatitis, so evidence of prostatitis, such as chills, dysuria and prostatic tenderness, should be sought.
Lower tract infections (cystitis) typically present with irritative micturition symptoms, such as dysuria and frequency, suprapubic discomfort and, sometimes, macroscopic haematuria. There is usually no fever. Women presenting with dysuria and frequency without vaginal discharge or irritation have a 90% probability of cystitis [20]. The classic symptom complex of loin pain, fever (>38°C), chills and urinary symptoms is usually associated with pyelonephritis. Severe pain should raise the suspicion of a ureteric calculus that, combined with infection, poses a greater risk of sepsis and of permanent injury to the kidney.
Patients with chronic indwelling catheters usually have no lower tract symptoms at all, but may develop loin pain and fever.
In elderly patients, particularly in long-term care facilities, the long-held view that symptoms of increased confusion and reduced mobility in the absence of fever, are due to urinary tract infection has been cast into doubt (see Treatment of specific groups: elderly patients) [8].
Examination
The clinical signs of lower UTI are few and non-specific, however, patients should be examined to exclude other causes for their symptoms, particularly vaginitis in women and prostatitis in men. The presence of renal angle tenderness, associated with fever, chills and dysuria suggests pyelonephritis.
Investigations
The key step in the diagnosis of UTI is examination of the urine, most commonly a midstream specimen (MSU). Catheterization is appropriate in patients with altered mental state or who cannot void because of neurological or urological reasons. Suprapubic aspiration is commonly used in paediatric practice but can be used in adults if other techniques have failed or are unable to be used.
The next step is to look for the presence of pyuria and, subsequently, the specimen may be sent for quantitative culture and antibiotic sensitivity testing. Testing for haematuria, proteinuria and nitrites may be of supportive value but is not diagnostic.
Reagent test strips
In considering the use of reagent strips in the diagnosis of UTI, it should be noted that variations in published sensitivity and specificity exist and are due to: (1) the use of different brands of reagent strips; (2) the use of different ‘gold standards’ against which comparison is made (e.g. counting chamber or cells/HPF counts, ‘cut-off’ criterion of the test used); (3) the nature of the study (blinded, unblinded); (4) the reader of the test (lab worker, doctor, nurse); and most importantly, (5) the clinical setting or target population (e.g. symptomatic emergency department patients rather than an asymptomatic population in a clinic or office environment) – in other words, the pre-test probability.
A reagent strip test for leucocyte esterase is now the most common screening test for pyuria (see below). Taken alone, this has a sensitivity of 48–86% and a specificity of 17–93% for detecting pyuria (as defined below). A positive predictive value (in symptomatic individuals) of 50% and a negative predictive value of 92% makes it a valuable test for screening the emergency department population. Most studies indicate that when the combination of leucocyte esterase and nitrite is considered, the sensitivity of the test is 68–88% and a negative test excludes the presence of infection [21]. Recent work by Sultana and others has shown that reagent strips significantly improve the clinician’s accuracy in diagnosing UTI in symptomatic emergency department patients [22]. The clinical probability of UTI must be considered when using such screening tests. In the patient with typical urinary tract symptoms, it may provide an adequate screen. It should, however, be used with great caution in the presence of fever of unknown cause in the elderly, the patient with an indwelling catheter or the patient with an impaired mental state, as pyuria and the implied bacteriuria may not be the cause of the problem.
Pyuria
Pyuria, indicates inflammation in the urinary tract and, as an indicator of infection, is second only to bacteriuria determined by quantitative culture (see below). The ‘gold standard’ definition of pyuria is based on early work involving the measurement of the rate of excretion of polymorphs in the urine. This work showed that excretion of 400 000 polymorphs per hour was always associated with infection and was also found to be represented by 10 polymorphs per mm3 in a single (unspun) midstream specimen of urine [23]. Thus, ‘significant pyuria’ was defined as 10 000 polymorphs per mL of urine. It was subsequently shown that more than 96% of symptomatic patients, defined as having significant bacteriuria, had significant pyuria and conversely less than 1% of asymptomatic people without bacteriuria have this degree of pyuria. Other definitions of pyuria, such as>5 leucocytes/high power field are based on examination of either the urinary sediment or of centrifuged urine and are inherently inaccurate because they cannot be standardized, but are nevertheless often used [24]. ‘Sterile’ pyuria indicates the presence of significant pyuria without the presence of bacterial growth in standard culture (Table 9.4.1).
Table 9.4.1
Common causes of sterile pyuria
Non-specific urethritis in males
Prostatitis
Renal tract neoplasm
Renal calculi
Catheterization
Renal TB
Previous antibiotic treatment
Nitrites
This reagent strip-based test is dependent on the bacterial reduction of urinary nitrate to nitrite, a function of coliform bacteria but not of Enterococcus spp. nor S. saprophyticus. The test has a low sensitivity (45–60%), better specificity (85–98%) but a high false-negative rate (about 45% in many studies). False-negative results are likely if the infecting organism is Gram positive or Pseudomonas, if the diet lacks nitrate or if there is diuresis or extreme frequency, as a period of bladder incubation is necessary to form nitrites.
Haematuria
Although a frequent accompaniment of UTI, this finding is non-specific as there are many other causes of haematuria.
Proteinuria
Most commonly with UTI protein excretion is less than 2 g/24 h. It is another common but non-specific finding.
Quantitative culture
Urine culture is not essential in the management of the pre-menopausal sexually active female with an uncomplicated UTI as the probability of UTI in these patients is 90% [20]. Culture should always be performed in patients with recurrent infection, possible pyelonephritis, potentially complicated UTI, males, the elderly or in cases where the cause of infection is not clinically evident. In symptomatic patients, a single specimen with a bacterial count in urine of>105 colony forming units (cfu)/mL has a 95% probability of representing infection [25]. However, it has been shown that 30–50% of women with symptoms of dysuria will have bacterial counts less than 105 cfu/mL [26]. Of these, about one-half have bacterial UTI with low numbers of bacteria. The rest may be considered in two groups; one group has urethritis due to Chlamydia trachomatis or Neisseria gonorrhoeae and the other has negative cultures and may have Ureaplasma urealyticum urethritis. In men, counts as low as 103 cfu/mL suggest infection [27].
In patients with indwelling urethral or suprapubic catheters, or those who intermittently self-catheterize and have symptoms or signs of UTI, a colony count of≥103 cfu/mL of more than one bacterial species in a single catheter or MSU specimen if the catheter has been removed within the previous 48 hours does indicate UTI [28].
Blood cultures
Blood cultures are normally not taken in afebrile patients with symptoms of cystitis. Current evidence indicates that blood cultures do not alter management and are therefore unnecessary in the majority of cases of uncomplicated pyelonephritis since the infecting organism is able to be isolated from a urine specimen [29,30]. Blood cultures should be taken in the following circumstances:
![]() recent instrumentation
recent instrumentation
![]() known anatomic abnormality
known anatomic abnormality
![]() failure of empiric treatment
failure of empiric treatment
![]() immunosuppression
immunosuppression
![]() significant co-morbidity, such as diabetes mellitus
significant co-morbidity, such as diabetes mellitus
![]() major sepsis
major sepsis
![]() fever of unclear cause.
fever of unclear cause.
Imaging
Imaging is not required in cases of uncomplicated cystitis. In pyelonephritis, imaging should be performed if there is:
![]() pain suggestive of renal colic or obstruction
pain suggestive of renal colic or obstruction
![]() failure to defervesce within 72 hours
failure to defervesce within 72 hours
![]() rapid relapse on cessation of antibiotic treatment or within 2 weeks
rapid relapse on cessation of antibiotic treatment or within 2 weeks
![]() infection with an unusual organism.
infection with an unusual organism.
These circumstances have been shown to be associated with stones or renal scarring. CT scanning is the preferred modality as this has greater sensitivity for demonstrating not only stones and obstruction, but rare gas-forming infections, haemorrhage and inflammatory masses.
Management
Ideally, treatment of UTI should rapidly relieve symptoms, prevent short-term complications, such as progression from cystitis to pyelonephritis and subsequent sepsis or long-term sequelae, such as renal scarring, and prevent recurrences by eliminating uropathogenic bacteria from vaginal and perineal reservoirs. Treatment should be cost-effective and have few or no side effects.
There is no evidence that non-specific treatments, such as pushing fluids or attempting to alter urinary pH, improve the outcome of normal antibiotic treatment.
Antibiotic treatment
Serum levels of antibiotics are largely irrelevant in the elimination of bacteriuria. Reduction in urinary bacterial numbers correlates with the sensitivity of the organism to the urinary concentration of the antibiotic. Inhibitory concentrations are usually achieved in the urine after oral doses of the commonly used antibiotics. On the other hand, blood levels are vitally important in the treatment of bacteraemic or septic patients or those with renal parenchymal infections. In considering antibiotic treatment, it is important to recognize the rapidly and constantly evolving antibiotic resistance patterns of common urinary pathogens. In order to ensure effective treatment, while participating in worldwide efforts to mitigate the rapid development of highly resistant forms, clinicians should always refer to the latest available guidelines. Empirically, the choice of antibiotic is based on the clinical presentation and the bacteria likely to be involved (Table 9.4.2).
Table 9.4.2
Choice of treatment depending on bacteria involved (see text)

Management of specific groups (Fig. 9.4.1)
Frequency dysuria syndrome: presumed simple cystitis
A non-pregnant, non-diabetic woman first presenting from the community with typical lower urinary symptoms should have vulvovaginitis excluded and an MSU taken and examined or tested by dipstick for pyuria. If pyuria is confirmed, culture of the urine specimen is not necessary and treatment should be commenced empirically.
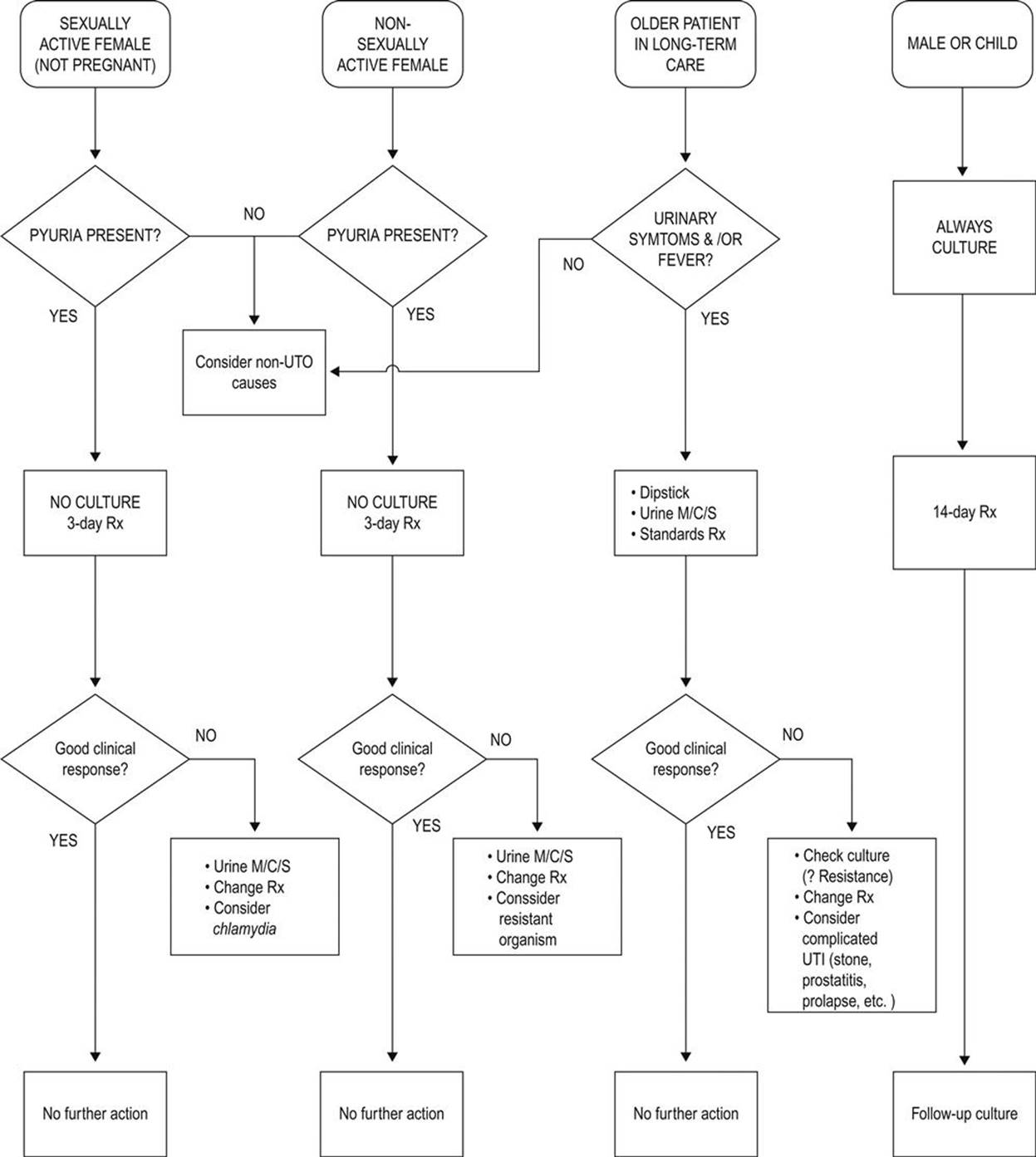
FIG. 9.4.1 Suggested management flow chart for simple cystitis.
There is now good evidence that in this group of patients short-course treatment is effective in both treating the infection and eradicating uropathogenic strains of bacteria from reservoirs. Three-day treatment is superior to a single dose in eradicating the reservoirs of uropathogenic organisms, thereby reducing the incidence of recurrence. Longer courses have an increased incidence of side effects but not higher cure rates. The antibiotic of choice for 3-day treatment is trimethoprim [31,32]. The emergence of trimethoprim resistant uropathogens has been well documented in some communities. The subsequent overuse of fluoroquinolones as first-line agents has resulted in a rapid rate of development of resistance to these agents in parts of Europe and in North America [33,34]. In general, fluoroquinolones should not be used as first-line agents for simple cystitis [31]. Awareness of local antibiotic resistance patterns is thus an important factor in choice of the most appropriate antibiotic.
Amoxycillin/clavulanic acid, nitrofurantoin and cephalexin are suitable for 5-day therapy, but amoxycillin alone should not be used as there is a high incidence of resistant E. coli in community-acquired UTI. If there is no clinical response, MSU should be sent for culture and, in sexually active women, treatment for C. trachomatis commenced (doxycycline 100 mg bd). In non-sexually active women, further treatment is guided by the results of sensitivity testing. Short-course treatment is inappropriate in women who are at risk of upper urinary tract infection (despite lower tract symptoms), which includes those with a history of previous infections due to resistant organisms, with symptoms for more than 1 week or those with diabetes mellitus.
Males with UTI should be investigated for urinary tract abnormality, associated prostatic or epididymal infection, must have urine culture initially and should have at least 14 days of treatment with any of the agents used for treatment of young women with simple cystitis (see Table 9.4.2) [31]. In men over 50 years, there is a high probability of invasion of prostatic tissue and treatment may need to be continued for 4–6 weeks.
Recurrent UTI
Recurrent UTI is defined as a symptomatic UTI which follows resolution of a previous UTI. These may be re-infections (with the same organism or another) or relapses (regrowth of the same organism within 2 weeks of treatment). Re-infections are more common than relapses, but the two may be indistinguishable [35]. It is important to consider the risk factors specific to the age and gender of the patient, e.g. sexual activity and use of spermicides in the young pre-menopausal woman or the higher rate of asymptomatic bacteriuria in the older patient. A careful search for causes and reversible factors (e.g. of complicated UTI due to stone or obstruction, previously undiagnosed diabetes mellitus, prostatitis in males) should be made together with urine culture and sensitivity testing. Treatment is generally as for pyelonephritis, with an appropriate antibiotic guided by the results of sensitivity tests for at least 10–14 days. Female patients may benefit from post-intercourse or maintenance prophylaxis with, e.g. cephalexin 250 mg or trimethoprim 150 mg at night for several months (see Host factors). There has recently been burgeoning interest in the use of cranberry, either as juice or in tablet form, for UTI prophylaxis. Evidence is variable, but a recent Cochrane review of 24 studies concluded that cranberry cannot be recommended for UTI prevention [36].
Acute pyelonephritis
Patients presenting with the typical symptoms of pyelonephritis are at risk of bacteraemia or sepsis syndrome and therefore must rapidly have adequate concentrations of appropriate antibiotics delivered to both the blood and the urine. In order to meet this requirement, particularly in patients who are vomiting, parenteral (intravenous) treatment is usually required initially, but seldom for longer than 24–48 hours, by which time the patient is usually afebrile and not vomiting.
The choice of antibiotics is of necessity empirical at this stage. In cases of mild infection in patients who are not vomiting, 10-day treatment with one of the antimicrobials used for simple cystitis is appropriate, with ciprofloxacin or norfloxacin reserved for resistant organisms or proven Pseudomonas aeruginosa. For severe infections, parenteral ampicillin or amoxycillin (2 g 6-hourly) together with gentamicin (4–6 mg/kg and up to 7 mg/kg as initial dose for severe sepsis) are appropriate, with a third-generation cephalosporin as an alternative to gentamicin when use of aminoglycosides is contraindicated. In patients with hospital- acquired infections and suspected Gram-negative sepsis or infections with Pseudomonas aeruginosa, broader-spectrum agents, such as ceftazidime, piperacillin/tazobactam, ticarcillin/clavulanic acid and imipenem, perhaps in combination with aminoglycosides, may be required. Parenteral treatment is followed by oral therapy for 2 weeks.
The use of short-stay observation units is now a standard part of the practice of emergency medicine. The safety and efficacy of treatment of pyelonephritis in such units with intravenous antibiotics and fluid administration, followed by oral therapy is widely accepted [37]. ‘Hospital in the home (HITH)’ programmes are also now commonplace, allowing close supervision of these patients by hospital-based staff and once- or twice-daily intravenous antibiotic administration at home. The efficacy and safety of HITH is well established, but requires careful patient selection to exclude those at risk of complicated infections. Appropriate follow up is essential, although repeat urine cultures are not recommended in asymptomatic patients following simple pyelonephritis [38,39].
Pregnancy
UTI in pregnancy are associated with an increased incidence of premature delivery and low birthweight infants. This has also been demonstrated to occur with asymptomatic bacteriuria, although up to 40% of asymptomatic women develop acute pyelonephritis later in pregnancy. Therefore, screening for bacteriuria and treatment of pregnant women is essential and urine must be sent for culture and antibiotic sensitivity testing. Three-day courses of treatment are not widely recommended, although it may be reasonable to use them with close follow up in an effort to reduce antibiotic usage; however, 10-day treatment courses are the norm. Cephalexin, nitrofurantoin or amoxycillin/clavulanate are appropriate for use in pregnancy, sulphas and trimethoprim being contraindicated [31].
Complicated UTI
As there is a greater range of organisms causing infection in these circumstances and a higher probability of antibiotic resistance, urine culture is essential and initial empiric treatment must cover the broader spectrum of organisms potentially involved. If possible, antibiotic treatment should be delayed until results of urine culture and antibiotic sensitivities are known and, if empirical therapy is instituted, management should be reviewed as soon as such results are available [40]. Trimethoprim or a quinolone is appropriate for mild infections. More serious infections may need combinations of agents, such as aminoglycosides with amoxycillin or imipenem/cilastatin.
Catheter-associated UTI
Catheter-associated UTI (CA-UTI) are the most common nosocomial infections. In patients with short-term catheters who develop infection, the catheter must be removed or changed and treatment instituted as for complicated UTI. For those with chronic indwelling catheters (such as patients with spinal injuries), bacteriuria is universal and treatment is only indicated in the presence of symptoms such as fever, chills or loin pain. Patients with chronic spinal injuries may present with autonomic dysreflexia syndrome, the symptoms of which include sudden hypertension, muscle spasm and sweating, with or without fever. Antibiotic selection should again be based on culture or empirically as for complicated UTI. The most important strategy for prevention of CA-UTI is minimizing catheter use and duration whenever possible. Preventative strategies based on use of methanemine, cranberry or prophylactic antibiotics at time of catheterization or catheter change are not recommended [28,31].
Elderly patients
As previously stated, asymptomatic bacteriuria and UTI are very common in older patients and more so with increasing functional impairment. Symptomatic infection is a significant cause of morbidity and mortality as this age group also has a higher incidence of bacteraemia associated with pyelonephritis and septic shock commonly follows. Given the high rate of asymptomatic bacteriuria, the diagnosis of UTI in such individuals is difficult. The traditional view that non-specific symptoms, such as increased confusion (without fever), falling or deteriorating mobility are due to UTI has been called into question. Evidence indicates that UTI should only be considered in patients with fever or specific genitourinary symptoms or both. In patients with non-specific symptoms, non-infective causes should be sought and, in the case of fever alone, other potential sources of infection must be considered [8].
Antibiotic treatment of symptomatic UTI in the elderly patient is no different initially to that of younger patients; however, it should be borne in mind that a greater variety of organisms may be cultured in this age group and urine for culture should be obtained at the outset whenever possible.
Asymptomatic bacteriuria
Asymptomatic bacteriuria (ASB) is defined as the presence of significant bacteriuria (as previously defined) in a person without signs or symptoms of UTI. The presence of pyuria per se should not be taken to indicate bacteriuria – a quantitative culture is essential for the diagnosis. Current evidence indicates that many patient groups may be harmed, or at least not benefit from antibiotic treatment for asymptomatic bacteriuria. Antibiotics should therefore not be given to:
![]() pre-menopausal, non-pregnant women
pre-menopausal, non-pregnant women
![]() diabetic women
diabetic women
![]() older people either living in the community or in institutions
older people either living in the community or in institutions
![]() patients with long-term indwelling catheters.
patients with long-term indwelling catheters.
Conversely, two significant groups receive clear benefit from antibiotic treatment for ASB: pregnant women and patients about to undergo urological procedures in which mucosal bleeding is anticipated (e.g. TURP).
Pregnant women with ASB have a 20–30-fold increased risk of developing pyelonephritis during pregnancy with consequent premature delivery and low birthweight infants. Therefore, pregnant women should be actively screened for ASB in early pregnancy and treated as for uncomplicated cystitis (without nitrofurantoin), with repeat cultures to confirm bacterial clearance. Periodic re-testing is recommended.
Patients who undergo urological procedures with mucosal bleeding (e.g. TURP) have a 60% rate of bacteraemia, with sepsis in 6–10% of these. These patients should be screened for ASB prior to the procedure and antibiotic treatment commenced shortly prior to the procedure and continued until after the procedure or removal of the post-procedure catheter [41].
Disposition
Patients with simple UTI should have follow up to confirm clinical cure. Failure of symptomatic improvement in 48 hours may indicate antibiotic resistance, which requires urine culture to elucidate. Recurrence of symptoms within 1–2 weeks may indicate occult renal infection and necessitates urine culture and at least 7 days’ treatment.
Prognosis
In adults with normal urinary tracts, UTI does not cause long-term sequelae. In the presence of urinary-tract abnormalities, infection may be a factor in producing renal damage or altering its rate of onset. Imaging of adults as part of their follow up should detect this group of patients.
Controversies
![]() The level of bacteriuria representing infection – traditional 105 cfu/mL or lower counts, such as 102 cfu/mL.
The level of bacteriuria representing infection – traditional 105 cfu/mL or lower counts, such as 102 cfu/mL.
![]() Best first-line treatment of uncomplicated cystitis in the face of emerging resistance of uropathogens to common antibiotics such as trimethoprim and fluoroquinolones.
Best first-line treatment of uncomplicated cystitis in the face of emerging resistance of uropathogens to common antibiotics such as trimethoprim and fluoroquinolones.
![]() The role of blood cultures as part of the investigation of pyelonephritis. Although traditionally used, they add little to the diagnosis and management.
The role of blood cultures as part of the investigation of pyelonephritis. Although traditionally used, they add little to the diagnosis and management.
![]() Asymptomatic bacteriuria in older, institutionalized patients. Differentiation from symptomatic infection and appropriate management.
Asymptomatic bacteriuria in older, institutionalized patients. Differentiation from symptomatic infection and appropriate management.
References
1. Bergeron M. Treatment of pyelonephritis in adults. Med Clin North Am. 1995;79:619–649.
2. Kreger BE, Craven DE, Carling PC, McCabe WR. Gram-negative bacteremia III Reassessment of etiology, epidemiology and ecology in 612 patients. Am J Med. 1980;68:332–343.
3. Fairley KF, Carson NE, Gutch RC, et al. Site of infection in acute urinary-tract infection in general practice. Lancet. 1971;298:615–618.
4. Foxman B. Epidemiology of urinary tract infections Transmission and risk factors, incidence, and costs. Infect Dis Clin N Am. 2003;17:227–241.
5. Hooton T, Scholes D, Hughes JP. A prospective study of risk factors for symptomatic urinary tract infection in young women. N Engl J Med. 1996;335:468.
6. Vorland L, Carlson K, Aalen O. An epidemiological survey of urinary tract infections among outpatients in Northern Norway. Scand J Infect Dis. 1985;17:277.
7. Nicolle L, Bjornson J, Harding G, MacDonell J. Bacteriuria in elderly institutionlaized men. N Engl J Med. 1983;309:1421–1425.
8. Nicolle LE. Urinary tract infection in geriatric and institutionalized patients. Curr Opin Urol. 2002;12:51–55.
9. Gupta K, Hooton T, Stamm W. Increasing antimicrobial resistance and the managment of uncomplicated community-acquired urinary tract infections. Ann Intern Med. 2001;135:41–50.
10. Ronald A. The etiology of urinary tract infection: traditional and emerging pathogens. Am J Med. 2002;113:14S–19S.
11. Kinane D, Blackwell C, Brettle R, et al. ABO blood group, secretor state and susceptibility to recurrent urinary tract infection in women. Br Med J. 1982;285:7–9.
12. Scholes D, Hooton T, Roberts P, et al. Risk factors for recurrent urinary tract infection in young women. J Infect Dis. 2000;182:1177–1182.
13. Turck M, Goffe B, Petersdorf R. The urethral catheter and urinary tract infection. J Urol. 1962;88:834–837.
14. O'Sullivan D, Fitzgerald M, Meyness M, Malins J. Urinary tract infection, a comparative study in the diabetic and general population. Br J Med. 1961;1:786.
15. Ronald A, Ludwig E. Urinary tract infection in adults with diabetes. Int J Antimicrob Agents. 2001;17:287–292.
16. Stapleton A. Urinary tract infections in patients with diabetes. Excerpta Med. 2002;113:80S–84S.
17. Mabeck C, Orskov R, Orskov I. Escherichia coli serotypes and renal involvement in urinary tract infection. Lancet. 1971;1:1312–1314.
18. Hagberg L, Hull R, Hull S, et al. Contribution of adhesion to bacterial persistence in the mouse urinary tract. Infect Immun. 1983;40:265–272.
19. Svanborg-Eden C, Hausson S, Jodal Y, et al. Host-parasite interactions in the urinary tract. J Infect Dis. 1988;157:421–426.
20. Bent S, Nallamothu BK, Simel DL, et al. Does this woman have an acute uncomplicated urinary tract infection? J Am Med Assoc. 2002;287:2701–2710.
21. Deville WL, Yzermans JC, van Duijn NP, et al. The urine dipstick test useful to rule out infections A meta-analysis of the accuracy. BMC Urol 2004;4.
22. Sultana R, Zalstein S, Cameron P, Campbell D. Dipstick urinalysis and the accuracy of the clinical diagnosis of urinary tract infection. J Emerg Med. 2001;20:13–19.
23. Brumfitt W. Urinary cell counts and their value. J Clin Pathol. 1965;18:550.
24. Stamm W. Measurement of pyuria and its relation to bacteriuria. Am J Med. 1983;75:53–58.
25. Kass E. Bacteriuria and the diagnosis of infection of the urinary tract. Arch Intern Med. 1957;100:709.
26. Stamm WE, Counts G, Running K, et al. Diagnosis of coliform infection in acutely dysuric women. N Engl J Med. 1982;307:463.
27. Lipsky B. UTI in men: epidemiology, pathophysiology, diagnosis and treatment. Ann Intern Med. 1989;110:138.
28. Hooton T, Bradley S, Cardenas D, et al. Diagnosis, prevention, and treatment of catheter-associated urinary tract infection in adults: 2009 international clinical practice guidelines from the infectious diseases society of America. Clin Infect Dis. 2010;50:625–663.
29. McMurray B, Wrenn K, Wright S. Usefulness of blood cultures in pyelonephritis. Am J Emerg Med. 1997;15:137–140.
30. Velasco M, Martinez J, Moreno-Martinez A, et al. Blood cultures for women with uncomplicated pyelonephritis: are they necessary? Clin Infect Dis. 2003;37:1127–1130.
31. Antibiotic Expert Group. Urinary tract infections. eTG Complete [CD-ROM] Melbourne: Therpeutic Guidlelines Limited; 2012.
32. Gupta K, Hooton T, Naber KG, et al. Executive summary: international clinical practice guidelines for the treatment of acute uncomplicated cystitis and pyelonephritis in women: a 2010 update by the Infectious Diseases Society of America and the European Society for Microbiology and Infectious Diseases. Clin Infect Dis. 2011;52(5):561–564.
33. Zhanel G, Hisanaga T, Laing N, et al. Antibiotic resistance in Escherichia coli outpatient urinary isolates: final results from the North American Urinary Tract Infection Collaborative Alliance (NAUTICA). Int J Antimicrob Agents. 2006;27:468–475.
34. Naber K, Schito G, Botto H, et al. Surveillance study in Europe and Brazil on clinical aspects and antimicrobial resistance epidemiology in females with cystitis (ARESC): implications for empiric therapy. Eur Urol. 2008;54:1164–1178.
35. Franco AV. Recurrent urinary tract infections. Best Pract Res Clin Obstet Gynaecol. 2005;19:861–873.
36. Jepson R, Williams G, Craig J. Cranberries for preventing urinary tract infections. Cochrane Database Syst Rev 2012;10.
37. Ward G, Jorden R, Severance H. Treatment of pyelonephritis in an observation unit. Ann Emerg Med. 1991;20:258–261.
38. Montalto M, Dunt D. Home and hospital intravenous therapy for two acute infections: an early study. Aust NZ J Med. 1997;27:19–23.
39. Caplan G, Sulaiman N, Mangin D, et al. A meta-analysis of “hospital in the home”. Med J Aust. 2012;197:512–519.
40. Nicolle L. Complicated urinary tract infection in adults. Can J Infect Dis Med Microbiol. 2005;16:349–360.
41. Nicolle L, Bradley S, Colgan R, et al. Infectious Diseases Society of America guidelines for the diagnosis and treatment of asymptomatic bacteriuria in adults. Clin Infect Dis. 2004;40:643–654.
9.5 Skin and soft-tissue infections
Rabind Charles
Essentials
1 The time-honoured principles of wound management, together with the judicious evidence-based use of antibiotics remain the basis for preventing and treating skin and soft-tissue infections.
2 All wounds, no matter how trivial, should be assumed to be tetanus prone and treated accordingly.
3 Skin and soft-tissue infections are common and range from mild to life threatening; they occasionally require surgical intervention and usually respond to narrow-spectrum antibiotics.
4 Deep soft-tissue infections have high morbidity and mortality and, unless treated aggressively, can rapidly result in loss of limb or death of the patient.
5 Infections due to unusual organisms, including organisms not usually considered to be pathogenic, frequently cause serious infections in the immunocompromised, patients with diabetes and patients with hepatic disease.
Introduction
Infectious disease is one of the most common reasons for patients to present to the emergency department (ED) and skin and soft- tissue infections (SSTIs) make up an important subset of these. SSTIs are a diverse group of aetiologically and anatomically distinct infections. Bacteria cause the majority of SSTIs encountered in the ED. The pathogenesis of these infections usually involves direct inoculation of bacteria as a result of violation of the skin or its defences, although there may also be spread of infection from a distant source via the haematogenous or lymphatic systems. The severity of infections encountered may range from mild to life threatening. Most recommendations for the diagnosis and treatment of SSTIs are based on tradition or consensus, as there are few randomized clinical trials on the subject. Some of the challenges to the emergency physician include:
![]() early and accurate diagnosis of the type of infection, based on clinical grounds and limited use of laboratory and radiological investigations
early and accurate diagnosis of the type of infection, based on clinical grounds and limited use of laboratory and radiological investigations
![]() early identification of potentially high-risk situations when the initial presentation is seemingly innocuous by looking at patient factors (e.g. diabetes, immunosuppression) and local factors (bite wounds, site of infection, e.g. orbital cellulitis)
early identification of potentially high-risk situations when the initial presentation is seemingly innocuous by looking at patient factors (e.g. diabetes, immunosuppression) and local factors (bite wounds, site of infection, e.g. orbital cellulitis)
![]() role of antibiotics: (1) appropriate choice of pharmacotherapeutic agent where indicated, taking into account the emergence of new infections and changing bacterial resistance patterns; (2) optimal route of delivery, i.e. topical versus oral versus initial intravenous or intramuscular bolus, followed by oral antibiotics versus intravenous therapy; (3) duration of the antibiotic treatment
role of antibiotics: (1) appropriate choice of pharmacotherapeutic agent where indicated, taking into account the emergence of new infections and changing bacterial resistance patterns; (2) optimal route of delivery, i.e. topical versus oral versus initial intravenous or intramuscular bolus, followed by oral antibiotics versus intravenous therapy; (3) duration of the antibiotic treatment
![]() need for surgical intervention, e.g. drainage of abscess, early debridement in necrotizing fasciitis
need for surgical intervention, e.g. drainage of abscess, early debridement in necrotizing fasciitis
![]() disposition issues: whether the patient can be discharged with outpatient follow up or will need hospitalization for management.
disposition issues: whether the patient can be discharged with outpatient follow up or will need hospitalization for management.
Aetiology
The majority of SSTIs are caused by aerobic Gram-positive bacteria, commonly Staphylococcus aureus and group A streptococcus. Deeper complicated infections, commonly seen in the immunocompromised host, are usually caused by Gram-negative, anaerobic or mixed organisms (Table 9.5.1).
Table 9.5.1
Causes of skin and soft-tissue infections
|
Risk factor/setting |
Expected pathogen |
|
Simple cutaneous infection |
Staphylococcus aureus. Also Staph. epidermidis, Staph. hominis, Streptococcus viridans |
|
Perianal, genital, buttocks, ungual and cervical areas |
Bacteroides fragilis, Escherichia coli, Klebsiella and Proteus |
|
Immunocompromised host |
Cryptococccus neoformans, Coccidioides, Aspergillus, Mycobacterium kansasii, M. tuberculosis and Yersinia enterocolitica |
|
Human bite |
Eikenella corrodens, Fusobacterium, Prevotella, streptococci |
|
Dog bite |
Pasteuralla multocida, Capnocytophaga canimorsus |
|
Cat bite |
P. multocida |
|
Injection drug abuse |
S. aureus, Clostridium spp., E. corrodens, S. pyogenes |
|
Body piercing |
S. aureus, S. pyogenes, P. aeruginosa, C. tetani |
|
Hot tub/wading pool |
Ps. aeruginosa |
|
Fresh water injury |
A. hydrophila |
|
Salt water injury |
V. vulnificus |
|
Fish tank exposure |
Mycobacterium marinum |
Examination
History
When taking a history, it is important to elicit the following:
![]() Any event leading to a breach in skin integrity may precipitate an infection, e.g. human, insect or animal bite, ‘clenched fist’ injury, excoriation, fungal infection or puncture wound. This is important because it will help in determining the likely pathogen and choice of antibiotics, as well as the need to rule out any potential foreign body that may be embedded in the wound.
Any event leading to a breach in skin integrity may precipitate an infection, e.g. human, insect or animal bite, ‘clenched fist’ injury, excoriation, fungal infection or puncture wound. This is important because it will help in determining the likely pathogen and choice of antibiotics, as well as the need to rule out any potential foreign body that may be embedded in the wound.
![]() The speed with which the infection has progressed serves as a guide as to how aggressive the infection is and the urgency of treatment needed.
The speed with which the infection has progressed serves as a guide as to how aggressive the infection is and the urgency of treatment needed.
![]() Patient factors that may complicate the treatment of the infection:
Patient factors that may complicate the treatment of the infection:
![]() history of immunosuppression, e.g. diabetes, steroid use, chronic liver disease, alcoholism, malnourishment, HIV, oncology patients on chemotherapy, nephrotic syndrome
history of immunosuppression, e.g. diabetes, steroid use, chronic liver disease, alcoholism, malnourishment, HIV, oncology patients on chemotherapy, nephrotic syndrome
![]() recent use of antibiotics, i.e. failed treatment
recent use of antibiotics, i.e. failed treatment
![]() history of prosthetic heart valves, mitral valve prolapse with regurgitation, previous history of endocarditis
history of prosthetic heart valves, mitral valve prolapse with regurgitation, previous history of endocarditis
![]() chronic venous stasis or lymphoedema in limbs; surgery that includes lymph node dissection or saphenous vein resection
chronic venous stasis or lymphoedema in limbs; surgery that includes lymph node dissection or saphenous vein resection
![]() intravenous drug use (IVDU)
intravenous drug use (IVDU)
![]() Tetanus status
Tetanus status
![]() Contamination with soil or water, which would suggest unusual pathogens as the cause of the infection.
Contamination with soil or water, which would suggest unusual pathogens as the cause of the infection.
Physical examination
![]() Identification of severe sepsis: unstable vital signs, hyperpyrexia, ‘toxic’-looking patient.
Identification of severe sepsis: unstable vital signs, hyperpyrexia, ‘toxic’-looking patient.
![]() Specific features of the infection to help narrow down the diagnosis, e.g. raised erythematous margins in erysipelas; presence of bullae and crepitus or tenderness out of proportion to physical signs, suggestive of necrotizing fasciitis; fetid odour suggesting anaerobic infection or green exudates typical of Pseudomonas spp.
Specific features of the infection to help narrow down the diagnosis, e.g. raised erythematous margins in erysipelas; presence of bullae and crepitus or tenderness out of proportion to physical signs, suggestive of necrotizing fasciitis; fetid odour suggesting anaerobic infection or green exudates typical of Pseudomonas spp.
![]() The extent of the infection, e.g. mapping areas of erythema to track progress, fluctuance that indicates fluid and a likely abscess that may need incision and drainage or lymphangitic spread.
The extent of the infection, e.g. mapping areas of erythema to track progress, fluctuance that indicates fluid and a likely abscess that may need incision and drainage or lymphangitic spread.
![]() Location of the infection, as involvement of certain critical areas (e.g. head, face, perineum) may require more intensive inpatient management and specialist consultation.
Location of the infection, as involvement of certain critical areas (e.g. head, face, perineum) may require more intensive inpatient management and specialist consultation.
![]() Complicating factors that might impair successful treatment, e.g. needle tracks in IV drug users, the presence of prosthetic heart valves.
Complicating factors that might impair successful treatment, e.g. needle tracks in IV drug users, the presence of prosthetic heart valves.
Investigations
SSTIs are usually diagnosed from their clinical presentation. Laboratory and radiological investigations play a secondary and limited role in routine evaluation but may be useful in the ED management of immunocompromised patients or those with signs and symptoms of severe sepsis. In such situations, the following parameters should be considered [1]:
![]() Full blood examination with differential: presence of marked leucocytosis, leucopaenia or an extreme left shift in the white cell differential; new-onset anaemia or thrombocytopaenia may suggest sepsis syndrome.
Full blood examination with differential: presence of marked leucocytosis, leucopaenia or an extreme left shift in the white cell differential; new-onset anaemia or thrombocytopaenia may suggest sepsis syndrome.
![]() Urea/creatinine: elevated levels suggest intravascular volume depletion or renal failure.
Urea/creatinine: elevated levels suggest intravascular volume depletion or renal failure.
![]() Creatine kinase: elevated levels may indicate myonecrosis caused by necrotizing fasciitis.
Creatine kinase: elevated levels may indicate myonecrosis caused by necrotizing fasciitis.
![]() Blood culture and drug susceptibility tests: the yield from these may be less than 10% and may be compounded by false-positive results [2]. In addition, emergency physicians do not have the luxury of time to await blood culture results before initiating the appropriate antibiotic treatment.
Blood culture and drug susceptibility tests: the yield from these may be less than 10% and may be compounded by false-positive results [2]. In addition, emergency physicians do not have the luxury of time to await blood culture results before initiating the appropriate antibiotic treatment.
![]() Other investigations: it is prudent to test for diabetes mellitus in patients presenting with an abscess because of the strong association of the two.
Other investigations: it is prudent to test for diabetes mellitus in patients presenting with an abscess because of the strong association of the two.
Patients with a chronic, recurrent or unusual infection should have their immune status checked, including serology for HIV. Soft-tissue radiographs may demonstrate a foreign body or gas in deep tissues. Computed tomography (CT) or magnetic resonance imaging (MRI) may be needed to define the depth and extent of the infective process when entertaining the diagnoses of fasciitis or myonecrosis. Ultrasonography in the ED may be a useful adjunct in evaluating soft-tissue infections for the presence of subcutaneous abscesses.
Management
Key points in the management of SSTIs include:
![]() analgesia
analgesia
![]() appropriate use of antibiotics
appropriate use of antibiotics
![]() appropriate surgical intervention
appropriate surgical intervention
![]() tetanus and other prophylaxis
tetanus and other prophylaxis
![]() disposition plans and options.
disposition plans and options.
Analgesia
Oral or parenteral analgesia should be prescribed, as most patients with SSTIs will present with pain. Simple measures, such as immobilization, elevation, heat or moist warm packs, should not be overlooked as they may help to alleviate pain in cellulitis. Abscess pain is best resolved by timely incision and drainage.
It is important to have a high index of suspicion for necrotizing fasciitis in any patient who has cellulitis with an inordinate amount of pain, or exquisite muscle tenderness where there is no history of musculoskeletal trauma.
Antibiotic therapy
Antibiotics are recommended for patients with signs of systemic toxicity, high fever, tachycardia, who are flushed and who look unwell, who are immunocompromised, who have abscesses in high-risk areas (hands, perineal region or face) and where deep necrotizing infection is suspected [2].
It is important for the emergency physician to recognize patients with serious skin and soft-tissue infections and to initiate appropriate care. The choice of antibiotic is often empiric and thus must be guided by the patient’s history, where they have been recently institutionalized and knowledge of the typical range of pathogens associated with each type of infection and their resistance patterns. The antibiotic of choice is the one that has proven efficacy against the range of expected pathogens, is associated with minimal toxicity and is cost-effective. Where possible, narrow- spectrum antibiotics should be used in preference to broad-spectrum ones [3]. Published guidance on antibiotic therapy is often deliberately non-prescriptive, reflecting the wide variety between differing patient populations, resistance patterns, methicillin-resistant Staph. aureus (MRSA) risk and local governance policies. It is also prudent to remember that SSTI clinical trials often exclude the most severely ill patients and may be powered to demonstrate non-inferiority only. The Infectious Diseases Society of America recently released guidelines for treating MRSA in SSTIs [4]. Unlike inpatient or chronic care settings, emergency physicians more frequently have to initiate empiric antibiotics based on clinical judgement and prevailing antibiograms, due to absence of culture and susceptibility results.
In the last decade, new agents active against Gram-positive bacteria have emerged and those licensed for treating complicated SSTIs include linezolid, daptomycin and tigecycline.
Surgical intervention
Certain SSTIs are best treated surgically. Effective treatment of abscesses and carbuncles and large furuncles entails incision, drainage of pus and breaking up of loculations, followed by regular dressings. Necrotizing fasciitis requires early aggressive surgical debridement together with broad-spectrum antibiotics in order to achieve best morbidity and mortality outcomes [5].
Tetanus and other prophylaxis
All wounds should be considered to be tetanus prone and treated accordingly. The patient’s immunization status should be checked and, where appropriate, tetanus toxoid plus tetanus immunoglobulin should be administered. Deep and penetrating wounds and wounds that have significant tissue devitalization or where there is heavy contamination (e.g. soil, dust, manure and wood splinters) are best treated with prophylactic antibiotic cover. The antibiotic of choice is penicillin; patients who are allergic to penicillin should receive cephalexin. If there is a history of severe penicillin allergy, use erythromycin or vancomycin.
Rabies prophylaxis should be considered for all feral and wild animal bites and in geographical areas where there is a high prevalence of rabies.
In cases involving human bites, consideration should also be given to screening for blood-borne pathogens such as hepatitis B virus, hepatitis C virus, HIV and syphilis.
Disposition
SSTIs are among the most frequently encountered conditions in the emergency observation setting. Good candidates for the observation/short-stay unit include patients likely to respond to empirical therapy, with a low likelihood of infection with unusual and/or resistant organisms.
Patients who have systemic toxicity (fever, tachycardia, rigors, altered mentation, severe pain), involvement of vital structures (fingers, hand, face and neck, genitourinary, scrotal and anal regions), those unable to take oral medication, who have failed outpatient therapy or who are immunocompromised (HIV positive, cancer, diabetes mellitus, hepatic or renal failure) are highly likely to require admission. Other prognostic factors include low serum bicarbonate, elevated creatinine, elevated creatine kinase and marked left shift polymorphonuclear neutrophils. The emergency physician must also be alert to scenarios requiring not just inpatient care but also urgent subspecialty consultation, e.g. necrotizing fasciitis.
Superficial skin infections
Clinical presentation
Patients usually present with a complaint of localized pain, redness and swelling. They may have been self-treating or have had previous treatment with oral antibiotics without success. Frequently, an abscess is fluctuant and indurated with surrounding erythema. The patient may also have associated lymphadenitis, regional lymphadenopathy and cellulitis. If the patient is febrile or there is systemic involvement, their immune status needs to be examined.
The possibility of a foreign body associated with an abscess needs to be considered. A careful history needs to be taken to determine whether this is possible and radiography may be necessary. Ultrasound can be useful in identifying the presence of a foreign body. The patient should also be questioned in relation to use of immunosuppressive agents.
Impetigo
This is a localized purulent skin infection, usually caused by group A streptococcus (Strep. pyogenes) and is seen more in warm humid climates. Topical therapy with mucipirocin often suffices, but oral antibiotics (first-generation cephalosporin or erythromycin) may be needed in cases with extensive lesions or perioral lesions.
Folliculitis
A superficial infection characterized by reddened papules or pustules of the hair follicles. Most cases are caused by Staph. aureus. Pseudomonas aeruginosa may be the cause following swimming pool or hot tub (spa) exposure. Treatment may only require the use of an antibacterial soap or solution. Removal of the hair in limited infections usually results in rapid resolution.
Furuncle and carbuncle
A furuncle arises secondarily to an infected hair follicle, where an abscess forms in the subcutaneous tissue. Furuncles most commonly occur on the back, axilla or lower extremities. Staphylococcus species are the most common associated organism. When the infection extends to involve several adjacent follicles, resulting in a coalescent inflammatory mass, the lesion is termed a carbuncle. Small furuncles are best treated with moist heat. Larger furuncles and all carbuncles require incision and drainage. The most common site is the back of the neck and diabetics are particularly prone to this. Systemic antibiotics are usually unnecessary unless there is extensive surrounding cellulitis or fever or if the patient has diabetes or is immunocompromised, in which case di(flu)cloxacillin (500 mg q 6 h oral), cephalexin (500 mg q 6 h) or clindamycin (450 mg q 8 h oral) can be used.
Erysipelas
Erysipelas is a rapidly progressive, erythematous, indurated, painful, sharply demarcated superficial skin infection caused by Strep. pyogenes (other causes are non-group A streptococci, Haemophilus influenzae, Staph. aureus and Strep. pneumoniae). The classic description is of a butterfly facial distribution, but recent evidence suggests that erysipelas is commonly found on the lower limbs. There is a clear line of demarcation between involved and uninvolved skin. It is common in young children and the elderly. Systemic symptoms (fever, chills, rigors and diaphoresis) are common and 5% will have bacteraemia. Erysipelas may rapidly progress to cellulitis (i.e. involvement beyond the upper dermis), abscess formation and, occasionally, fasciitis. Treatment consists of the use of antibacterial soap and oral penicillin (di(flu)cloxacillin 500 mg q 6 h oral).
Herpetic whitlow
Herpetic whitlow is a superficial infection with herpes simplex virus and is an occupational hazard of jobs having contact with oral mucosa, e.g. dentistry and anaesthesiology. Incision and drainage is contraindicated and may in fact spread the viral infection.
Cellulitis
Cellulitis is an acute spreading infection of the skin involving the deeper dermis and subcutaneous fat. In patients with a normal immune system who are otherwise healthy, the infection is caused by bacteria that normally colonize skin, principally Staph. aureus and group A β-haemolytic streptococci. Predisposing factors include conditions leading to a disrupted cutaneous barrier and/or impaired local host defences, such as trauma and inflammatory dermatoses, e.g. eczema, oedema from venous insufficiency or lymphatic obstruction.
Despite the common occurrence of cellulitis, there is a paucity of published research on issues such as criteria for antibiotics and admission and severity assessment. The presence of an underlying abscess should be considered in patients who ‘fail’ initial antibiotic therapy: treatment failure may be caused by an undrained abscess rather than inadequate antimicrobial therapy and bedside soft-tissue ultrasonography is a useful tool that is increasingly available in EDs.
Treatment consists of elevation of the affected part and administration of an antistaphylococcal penicillin such as di(flu)cloxacillin (2 g q 6 h IV) or a first-generation cephalosporin, such as cephazolin (2 g q 12 h IV).
Therapy may need to be escalated in special settings, such as diabetes, or particular anatomical areas. Anaerobes or Gram-negative organisms have been identified in 95% of affected diabetic foot ulcers, with Staph. aureus found in approximately 33%. Broad-spectrum antibiotic treatment, e.g. metronidazole (400 mg q 12 h orally) plus cephazolin (2 g q 12 h IV), is recommended.
Infections that originate from wounds involving the feet may be due to Pseudomonas aeruginosa; this organism is also associated with osteomyelitis of the foot. Antibiotic treatment should consist of an antipseudomonal β-lactam, such as carbenacillin, or a third- generation cephalosporin, such as ceftriaxone, and an aminoglycoside.
Cellulitis is a well-known complication in women who have undergone axillary lymph node dissection and surgery for breast cancer. The major mechanism is thought to be an altered lymphatic and/or venous circulation related to the surgical procedure and to radiation therapy. Empiric antibiotic therapy is targeted at Staph. aureus and β-haemolytic streptococci and choices include cephalexin or cefazolin. If the patient has received recent chemotherapy and is neutropaenic, then the antibiotic regimen must be broadened to include coverage for aerobic Gram-negative bacilli, including Pseudomonas aeruginosa.
Facial cellulitis, including periorbital and orbital cellulitis is a serious infection occurring in adults and children [6]. The causal organisms include Staph. aureus, H. influenzae type b and Staph. pneumoniae. This type of cellulitis may arise from an infected sinus. Broad-spectrum antibiotic therapy is required, the agent of choice being dicloxacillin 1 g IV 6-hourly. Radiological evaluation, including CT scanning, may be necessary to identify underlying sinusitis.
Abscesses
Pilonidal abscess
Pilonidal abscesses occur in the superior gluteal fold and arise from the disruption of the epithelium, causing the formation of a pit lined with epithelial cells that may become plugged with hair and keratin, leading to an abscess. Treatment involves incision and drainage, usually in the operating theatre, although smaller abscesses can be drained in the ED under local anaesthetic. They are usually associated with mixed organisms, both aerobic and anaerobic.
Hidradenitis suppurativa
This is a chronic suppurative abscess of the upper apocrine sweat glands in the groin and axilla. It is much more common in females, in obesity and in patients who have poor hygiene or who shave the region. Organisms include Staph. aureus, Strep. viridans and Proteus spp. Treatment is incision and drainage, usually in the operating theatre. Definitive treatment may require removal of the apocrine sweat glands from the region.
Bartholin’s abscess
This occurs as a result of the obstruction of a Bartholin’s duct and is usually composed of mixed vaginal flora. Neisseria gonorrhoeae and Chlamydia trachomatis may also be involved. Treatment is incision, drainage and marsupialization of the cyst in the operating theatre.
Paronychia
This is a superficial abscess of the lateral aspect of the nail, commonly associated with patients whose hands are frequently wet. Common organisms involved are Staph. aureus, Candida and anaerobes. Some cases may require incision and drainage, with advice to keep the hands dry.
Perianal abscess
These are thought to originate in the anal crypts and extend into the ischiorectal space. Patients frequently complain of pain on defaecation and sitting. Perianal abscesses may be associated with inflammatory bowel disease and fistula formation. Treatment should be incision and drainage in the operating theatre under general anaesthesia. When the abscess is superficial and ‘pointing’, drainage in the ED is possible.
Infected sebaceous cyst
Sebaceous cysts become infected when the duct is obstructed. They can occur anywhere on the body, but tend to favour the head and neck region. Treatment is incision and drainage, recurrence is not uncommon.
Treatment
Incision and drainage of cutaneous abscesses is the key to treatment. Some patients require oral antibiotic therapy. Patients who are immunosuppressed or who have diabetes mellitus should be treated with appropriate antibiotic therapy based on a knowledge of the probable pathogen. Patients at risk of developing bacterial endocarditis require prophylactic antibiotics prior to incision and drainage. The treatment of superficial skin abscesses has in recent years been complicated by the emergence of MRSA. Proponents of the practice of ‘routine culture’ of abscess fluid say that surveillance of antimicrobial susceptibility allows therapeutic adjustment. Detractors point out that for simple abscesses, incision and drainage without antibiotics is usually sufficient and thus if antibiotics are not considered clinically useful it is unlikely that culture results will alter the management.
Deep soft-tissue infections
Necrotizing fasciitis
Necrotizing fasciitis is a rare, rapidly progressing, life-threatening infectious process involving primarily the superficial fascia (i.e. all the tissue between the skin and underlying muscles – the subcutaneous tissue). Patients usually present with the triad of exquisite pain – often out of proportion to initial physical findings – swelling and fever. Early diagnosis is sometimes thwarted by the paucity of cutaneous findings early in the course of the disease. The clinician should have a high index of suspicion based on the clinical presentation as well as the patient’s underlying co-morbidities (diabetes, chronic alcoholism, and immunosuppression). Laboratory values may be used in risk scoring, e.g. the Laboratory Risk Indicator for Necrotizing Fasciitis (LRINEC), which has been validated prospectively and has a high sensitivity and positive predictive value of 92% in patients with scores of six points and above. Patients with scores of five points and below are considered at low risk of necrotizing fasciitis [7,8].
Numbness of the involved area is characteristic of advanced necrotizing fasciitis – this is a result of infarction of the cutaneous nerves. Eighty per cent of cases show clear origins for an accompanying skin lesion (insect bite, minor abrasion, furuncle, and IVDU injection site) but, in the remaining 20%, no skin lesion can be found [9,10].
Patients appear extremely toxic with a high fever, tachycardia and malaise. Pathognomonic features include extensive undermining of the skin and subcutaneous tissues, with separation of the tissue planes. The subcutaneous tissues may have a hard, wooden feel. Bullous lesions and skin ecchymoses may also be evident. Crepitation may be clinically evident and gas may be visualized on X-ray in some 80% of patients. The gas is typically layered along fascial planes. CT or MRI may aid in confirming the clinical suspicion.
Bacteria involved in this infection are usually mixed: Staph. aureus, haemolytic streptococci, Gram-negative rods and anaerobes. Sometimes only group A streptococci, either alone or in combination with Staph. aureus, are found. Aggressive therapy is essential, as mortality approaches 50%. Immediate surgical intervention extensively to open and debride the wound is required, as myonecrosis may be present [11]. Appropriate antimicrobial therapy should be commenced immediately: meropenam (1 g q 8 h IV) plus clindamycin (600 mg q 8 h IV) or lincomycin (600 mg q 8 h IV). Hyperbaric oxygen therapy should be considered.
Fournier’s gangrene is a form of necrotizing fasciitis which involves the scrotum, penis or vulva and is usually seen in diabetics. It usually originates from perianal or urinary tract infections (which extend into the periurethral glands) and can progress explosively. The management is early recognition and surgical debridement and intravenous antibiotics.
Gas gangrene
Gas gangrene is an acute life- and limb- threatening deep-tissue infection, also known as clostridial myonecrosis. Aetiological agents include Clostridium perfringens, Cl. histolyticum, Cl. septicum and Cl. novyi. Cl. perfringens is the most common cause in traumatic gas gangrene, whereas spontaneous gangrene is principally associated with Cl. septicum. This infection is characterized by the rapid development (often within hours) of intense pain in the region of a wound, followed by local swelling and a haemoserous exudate. A characteristic foul smell is also a good indication of the diagnosis. The area becomes tense and may develop a bluish and bronze or dusky discoloration. The presence of gas is typical, although it may be a late finding. It is frequently found on X-ray, where it has a feathered pattern as gas develops within the muscle itself. Aggressive treatment is required as the patient may present in an advanced stage with tachycardia, altered mental status, shock and haemolytic anaemia.
Classically, the gas gangrene occurs in extensive and or deep wounds with predisposing factors, including vascular ischaemia, diabetes and presence of foreign bodies. Gram stain frequently reveals relatively few white blood cells and large numbers of club-shaped Gram-positive rods.
Early surgical intervention is essential, including wide debridement of necrotic muscle and other tissues, administration of high-dose penicillin C (benzylpenicillin 2.4 g q 4 h IV), an aminoglycoside and hyperbaric oxygen therapy. Early hyperbaric oxygen therapy has been demonstrated to result in improved outcome [3,10–13].
One should note that the presence of gas certainly raises the suspicion of a deep-tissue infection, including gas gangrene, but that it may also be present because of previous wound manipulation, self-injection of air, localized gas abscess or other gas-producing organisms, including anaerobes, E. coli, streptococci and staphylococci.
Pyomyositis
Pyomyositis is the presence of pus within individual muscle groups and the usual culprit is Staph. aureus. A positive blood culture yield is found in only 5–30% of cases. Typical presenting symptoms included localized pain in a single muscle group, usually in an extremity, and fever. Ultrasonography or CT may be warranted to differentiate the condition from a suspected deep vein thrombosis.
Toxic complications of wound infections
A number of bacteria produce toxins that result in systemic symptoms.
Tetanus
Tetanus, albeit rare in developed countries, still occurs despite the fact that immunization is completely effective in preventing it. All wounds should be treated as tetanus prone. Tetanus may occur with trivial wounds that may not even be apparent. The incubation period is variable, ranging from 3 days to several weeks after inoculation; the disease is more severe at the extremes of age. Difficulty in swallowing and a fever with progression to stiffness and trismus is pathognomonic. Tetanus is also associated with autonomic nervous system dysfunction. Occasionally, localized tetanus may occur with muscle spasm in the area adjacent to the wound. This is sometimes associated with cranial nerve dysfunction. Treatment is largely supportive, often requiring deep sedation, paralysis and ventilation for prolonged periods. Antibiotic therapy with high-dose penicillin should also be given in addition to tetanus immunization and tetanus immunoglobulin (Table 9.5.2).
Table 9.5.2
Tetanus prophylaxis

Reproduced with permission from Antibiotic Expert Group. Melbourne: Therapeutic guidelines Limited, 2013 eTG [14].
Toxic shock syndrome
Toxic shock syndrome (TSS) is a life-threatening multisystem disease caused by inflammatory immune responses to toxogenic strains of Staph. aureus. TSS has been classically associated with the use of tampons, although 10–40% of cases are non-menstrual related. Onset of menstrual TSS symptoms occurs within 3 days of the menstrual cycle and usually has no preceding clinically apparent infection. Non-menstrual cases occur after childbirth, abortions, in bone and skin infections including postoperative wound infections, burns, mastitis and varicella-related cellulitis. The wound itself may look insignificant. There is a rapid onset of fever, usually>38.9°C, hypotension and an initial diffuse and later desquamating erythematous rash. Multiorgan involvement may include muscular (myalgia), neurological (headache, and altered sensorium) and gastrointestinal (nausea, and diarrhoea) symptoms. Occasionally, Staph. aureus can be cultured locally, although blood cultures are rarely positive. Antibiotics do not affect the course of TSS but may lower the recurrence rate by 59–73% [15]. An antistaphylococcal agent should be given with an aminoglycoside. Patients are frequently haemodynamically compromised, requiring aggressive fluid resuscitation and inotropic support. Debridement of necrotic wounds, if present, and elimination of the source of infections – e.g. removal of the tampon – should be carried out urgently. A similar syndrome can develop due to infection with group A β-haemolytic streptococci. This is known as ‘wound’ or ‘surgical’ scarlet fever. Treatment is the same as for TSS.
Special infections
Human bites
Human bite wounds may occur as a result of an accidental injury, deliberate biting or closed fist injuries. The bacteriology reflects the normal oral flora of the biter: streptococci in 50–80% of wounds, staphylococci, Eikenella corrodens and anaerobic organisms. Therapy consists of irrigation and topical wound cleansing and prophylactic antibiotics should be initiated as early as possible in all patients, regardless of the appearance of the wound.
Clenched fist injuries over the metacarpophalangeal joint warrant hospitalization for formal washout and intravenous antibiotics. Appropriate antibiotic choices include amoxicillin-clavulanate (875+125 mg q 12 h oral), metronidazole (400 mg q 12 h oral) plus either ceftriaxone (1 g daily IV) or cefotaxime (1 g daily IV). In cases of β-lactam allergy, metronidazole plus doxycycline, ciprofloxacin or trimethoprim-sulfamethoxazole may be used.
Animal bites
Most bites are from dogs (80%) or cats, but bites from exotic pets and feral animals also occur. Pasteurella and bacteriodes species are the most common bacterial isolates and Capnocytophaga canimorsus can cause bacteraemia and fatal sepsis, especially in patients with underlying liver disease or asplenia. Infected bites presenting<12 hours after injury are more likely to be infected with Pasteurella spp., whereas those presenting>24 hours post-bite are more likely to be infected with staphylococci or anaerobes. Wounds should be cleansed with sterile normal saline and infected wounds should not be closed. Cat bite wounds have less crush injury and wound trauma than dog bites, but have a higher proportion of osteomyelitis and septic arthritis. The oral agent of choice for both dog and cat bites is amoxicillin–clavulanate, with doxycycline as an alternative. Intravenous options include second-generation cephalosporins, piperacillin–tazobactam and carbapenams. Cellulitis and abscesses usually respond to 5–10 days of therapy. Rabies prophylaxis should be considered for all feral and wild animal bites and in geographical areas where there is a high prevalence of rabies.
Water-related infections
Water-related infections may be caused by unusual organisms. Vibrio vulnificus, V. alginolyticus and other non-cholera vibrios are found in salt and brackish water and can result in serious and life-threatening infections, especially in patients with hepatic disease. Aggressive infection can progress rapidly over 2–4 hours. It is associated with saltwater exposure or the ingestion of raw shellfish. Infections can mimic gas gangrene, with rapid progression and tissue destruction; septicaemia may occur and can be fatal. If parenteral therapy is required, a third- generation cephalosporin can be combined with an aminoglycoside and/or doxycycline.
Exposure to fresh or brackish water (rivers, mud and caving) can result in infection with the Gram-negative bacillus Aeromonas hydrophila[16]. Aeromonas infections can result in superficial skin infections, myositis and septicaemia. Treatment consists of administration of cefotaxime 1 g IV 8-hourly or ceftriaxone 1 g IV daily. If oral therapy is possible, consider ciprofloxacin 500 mg orally 12-hourly.
Mycobacterium marinum, M. ulcerans, M. chelonei, M. gordanae. and M. fortuitum are found in fish tanks and can result in ‘fish fancier’s finger’. After 2–6 weeks of incubation, an ulcerating granuloma develops. Treatment options include clarithromycin, trimethoprim–sulfamethoxazole or a combination of ethambutol and rifampicin. Systemic infection is uncommon.
Saltwater fish handlers may develop infections due to Erysipelothrix rhusiopathiae; this causes erysipeloid, a type of cellulitis. It also causes infections in people handling fish, poultry, meat and hides. Coral cuts are often infected with Streptococcus pyogenes; other marine pathogens may be involved (including Vibrio species). Treatment should consist of phenoxymethylpenicillin 500 mg 6-hourly.
Mastitis
Infections of the breast can occur in both sexes and in all ages; however, breast infections are most common in nursing mothers and the prevalence of lactational mastitis in Australia is estimated at 20% [17]. Staph. aureus is the most common pathogen in infective mastitis.
Treatment consists of regular emptying of the breast. If breastfeeding needs to be stopped because of the severity of the infection or the risk to the neonate, a pump or manual expression methods should be employed (at least temporarily). If symptoms are not resolving within 12–24 hours of effective milk removal and analgesia, antibiotic treatment should be commenced to prevent abscess formation. Eleven per cent of patients who are not treated appropriately with antibiotics will develop an abscess. Options include di(flu)cloxacillin (500 mg q 6 h oral) or a first-generation cephalosporin, such as cephalexin (500 mg q 6 h oral) or erythromycin (250–500 mg q 6 h oral). Severe infections may require parenteral or more prolonged therapy. Local care to the region is also important, including warm compresses, breast support, analgesia and the application of a moisturizing cream to the nipple and areolar region. Patients who develop an abscess will require percutaneous aspiration or open drainage [18].
Decubitus ulcers
Decubitus ulcers are cutaneous ulcerations caused by prolonged pressure that results in ischaemic necrosis of the skin and underlying soft tissue. They are most commonly found in patients who are bedbound, particularly elderly nursing home patients and patients with sensory deficits, such as paraplegia and quadriplegia. Immobility, compounded by vascular insufficiency and neuropathy, results in ulcer formation and, unless treated aggressively, serious complications can follow [19]. Complications include cellulitis and deep soft-tissue necrosis, osteomyelitis, septic thrombophlebitis, bacteraemia and sepsis. Culture of the ulcer invariably reveals a mixed bacterial flora of both aerobes and anaerobes, which do not distinguish between colonization and tissue infection. The most common organisms found are staphylococci, streptococci, coliforms and a variety of anaerobes. Systemic antibiotics are required for patients with clinical signs of sepsis or osteomyelitis.
Varicose ulcers
Varicose ulcers are cutaneous ulcers caused by oedema and poor tissue drainage as a result of dysfunction of the venous system, including varicose veins. These are more common in the elderly and obese. They may be chronic, and healing is often difficult. Complications include cellulitis and, occasionally, bacteraemia. Culture of the ulcer variably reveals a mixed bacterial flora of both aerobes and anaerobes that cannot distinguish between colonization and tissue infections. The most common organisms found are staphylococci, streptococci, coliforms and a variety of anaerobes.
Treatment consists of debridement of necrotic tissue, pressure area and general nursing care, as well as treatment of infection, if present. Antibiotic treatment is only indicated where there is systemic evidence of infection or where there is a complicating infection, such as osteomyelitis or bacteraemia. Surgical debridement is frequently as important, if not more important, than antibiotic therapy, particularly where the bacterial infection is localized.
Diabetic foot infections
Foot infections are a common complication of diabetes and require both local (foot) treatment and systemic (metabolic) optimization, which is best undertaken by a multidisciplinary team including surgeons, podiatry services and the endocrinologist or physician.
The peripheral neuropathy associated with diabetes results in the loss of protective pain sensation and results in repetitive injuries, followed by the development of ulcers that become infected. Vascular insufficiency and impaired immune function contribute to the increased risk of acute and chronic infection. Infections in foot ulcers are often polymicrobial and both the number of bacterial groups and bacterial density are thought to affect healing [16]. Aerobes include Staph. aureus, coagulase-negative staphylococci and streptococci. Enterobacteriaceae and Corynebacterium are not uncommon. Anaerobes which have been isolated from up to 48% of patients include Bacteroides and Clostridium spp. The presence of anaerobes is associated with a high frequency of fever, foul-smelling lesions and the presence of an ulcer. Cultures obtained using curettage following debridement should be used in preference to wound swabs to identify causative organisms and sensitivities.
Local signs and symptoms predominate and include those secondary to infection, vasculopathy and neuropathy. Pain and tenderness are often minimal due to the neuropathy and pulses are frequently reduced or absent. Wound infections must be diagnosed clinically on the basis of local (and occasionally systemic) signs and symptoms of inflammation. Laboratory (including microbiological) investigations are of limited use for diagnosing infection, except in cases of osteomyelitis radiography and/or a bone scan may be warranted to exclude osteomyelitis.
A recent systemic review [20] reported that there is no strong evidence for any particular antimicrobial agent in the prevention of amputation, resolution of infection or ulcer healing. For mild to moderate infections with no evidence of osteomyelitis or septic arthritis, consider amoxicillin–clavulanate (875+125 mg q 12 h oral) for at least 5 days. Alternatives include ciprofloxacin 500 mg q 12 h with clindamycin 600 mg q 8 h. For severe limb- or life-threatening infections, intravenous piperacillin–tazobactam 4+0.5 g q 8 h or ticarcillin–clavulanate 3+0.1 g q 6 h or meropenem 500 mg q 8 h are all acceptable empiric therapy. Prolonged use of appropriate bactericidal antibiotics may be required, especially in the setting of osteomyelitis or septic arthritis.
Surgical-site/postoperative wound infection
Surgical-site infections are the most commonly occurring adverse events in patients who have undergone surgery, accounting for as much as 38% of nosocomial infections in postoperative patients. Surgical-site infections are usually diagnosed by the usual features of inflammation: wound pain, redness, swelling and purulent discharge. These external signs of inflammation may manifest late in morbidly obese patients or those with deep, multilayer wounds. Most bacterial wound infections present with fever only after 48 hours. Earlier symptoms may be seen in Strep. pyogenes and clostridial infections.
The mainstay of treatment for surgical site infections is early opening of the incision, coupled with evacuation of any infected material and sending off of wound cultures. This should be done after consultation with the surgeon involved, where possible. There has been a paucity of evidence regarding the use of antibiotics combined with drainage [21], but expert consensus generally advocates the use of empirical antibiotics for patients with temperature>38.5°C and/or pulse rate>100 in the presence of obvious wound infection [5].
Post-traumatic wound infection
The goals of wound care are to avoid infection and to achieve a functional and cosmetically acceptable scar. Adequate wound management requires a thorough history, with particular attention directed at factors adversely affecting healing. Factors, such as the extremes of age, diabetes, chronic renal failure, malnutrition, alcoholism, obesity and patients on immunosuppressive agents, cause an increased risk of infections and impaired wound healing. Wounds located in highly vascular areas, such as the scalp or face, are less likely to become infected than wounds in less vascular areas.
In order to reduce the incidence and severity of infections, wounds need to be thoroughly cleansed and irrigated. Devitalized tissue should be removed, injuries to associated structures need to be excluded and the wound closed appropriately. The method of closure depends on the location of the wound, the level of contamination and whether it is an ‘old’ wound (over 6 hours old). Wounds that should not be closed because of a high risk of infection, such as heavily contaminated wounds, should be treated by delayed primary closure 3–5 days after initial management. Where primary closure is possible the wound should be closed and a protective non-adherent dressing applied for a minimum of 24–48 hours, with both the wound and the dressing kept dry [22].
The use of prophylactic antibiotics is not recommended except where there is significant bacterial contamination, foreign bodies, the patient is immunosuppressed or the wound is the result of a bite (human or animal) or associated with an open fracture. Most wounds can be treated with amoxicillin–clavulanate (875+125 mg q 12 h oral) or metronidazole (400 mg q 12 h oral) plus di(flu)cloxacillin (500 mg q 6 h oral). Broad-spectrum antibiotics should be limited to heavily contaminated and bite wounds and immunosuppressed patients (see Table 9.5.2).
Intravenous drug users
Intravenous drug users frequently develop unusual infections because the needles and the drug paraphernalia used are contaminated. They also have alterations to their skin and flora and frequently have poor nutrition and immune function [23]. Many are hepatitis B, C and HIV positive. Intravenous drug users frequently have mixed organisms, particularly anaerobes, including Klebsiella, Enterobacter, Serratia and Proteus. They have mixed Gram-positive and Gram-negative infections. Some develop fungal infections, including candidaemia. Subacute bacterial endocarditis and endocarditis need to be considered in IV drug users. If endocarditis is not suspected, treatment should consist of flucloxacillin 2 g IV 6-hourly and gentamicin 5–7 mg/kg/day as a single daily dose.
Controversies
![]() The timing and method of closure of contaminated or ‘old’ (more than 6 hours since injury) wounds.
The timing and method of closure of contaminated or ‘old’ (more than 6 hours since injury) wounds.
![]() The prophylactic use of antibiotics in patients with ‘clean’ wounds.
The prophylactic use of antibiotics in patients with ‘clean’ wounds.
![]() Which antibiotics to use in treating skin and soft-tissue infections: narrow-spectrum, first-generation cephalosporin or broad-spectrum third-generation cephalosporin? Do you use antibiotics to cover Gram-negative, Gram-positive organisms, anaerobes and aerobes?
Which antibiotics to use in treating skin and soft-tissue infections: narrow-spectrum, first-generation cephalosporin or broad-spectrum third-generation cephalosporin? Do you use antibiotics to cover Gram-negative, Gram-positive organisms, anaerobes and aerobes?
![]() Can more patients be treated wholly as outpatients using parenteral therapy or after early discharge once the acute toxic phase is over?
Can more patients be treated wholly as outpatients using parenteral therapy or after early discharge once the acute toxic phase is over?
![]() Management of cutaneous abscesses: are antibiotics necessary after incision and drainage? Are cultures of the abscess fluid needed?
Management of cutaneous abscesses: are antibiotics necessary after incision and drainage? Are cultures of the abscess fluid needed?
References
1. Simonart T, Simonart JM, Derdelinckx I, et al. Value of standard laboratory tests for the early recognition of group A beta-hemolytic streptococcal necrotizing fasciitis. Clin Infect Dis. 2001;32:E9–12.
2. Perl B, Gottehrer NP, Raveh D, et al. Cost-effectiveness of blood cultures for adult patients with cellulitis. Clin Infect Dis. 1999;29:1483–1488.
3. Antibiotic Expert Group. Therapeutic guidelines: antibiotics eTG complete [Internet] Melbourne: Therapeutic Guidelines Limited; 2013; <http://online.tg.org.au.proxy1.athensams.net/ip/>[Accessed Feb. 2013].
4. Liu C, Bayer A, Cosgrove SE, et al. Clinical Practice Guidelines by the Infectious Diseases Society of America for the Treatment of Methicillin-resistant Staphylococcus aureus infection in adults and children. Clin Infect Dis. 2011;52:e18–e55.
5. Stevens DL, Bisno AL, Chambers HF, et al. Infectious diseases Society of America Practice guidelines for the diagnosis and management of skin and soft-tissue infections. Clin Infect Dis. 2005;41:1373–1406.
6. Leong WC, Lipman J, Hon H. Severe soft-tissue infections – a diagnostic challenge The need for early recognition and aggressive therapy. S Afr Med J. 1997;87 648–52, 654.
7. Wong CH, Khin LW, Heng KS, et al. The LRINEC (Laboratory Risk Indicator for Necrotizing Fasciitis) score: a tool for distinguishing necrotizing fasciitis from other soft tissue infections. Crit Care Med. 2004;32:1535–1541.
8. Hasham S, Matteuci P, Stanley PR, Hart NB. Necrositing fasciitis. Br Med J. 2005;330:830–833.
9. Gabillot-Carre M, Roujeau JC. Acute bacterial skin infections and cellulitis. Curr Opin Infect Dis. 2007;20:118–123.
10. Wong CH, Chang HC, Pasupathy S. Necrotizing fasciitis: clinical presentation, microbiology, and determinants of mortality. J Bone Joint Surg. 2003;85A:1454–1460.
11. Bosshardt TL, Henderson VJ, Organ Jr CH. Necrotizing soft-tissue infections. Arch Surg. 1996;131:846–852.
12. Lille ST, Sato TT, Engrav LH, et al. Necrotizing soft tissue infections: obstacles in diagnosis. J Am Coll Surg. 1996;182:7–11.
13. Ben-Aharon U, Borenstein A, Eisenkraft S, et al. Extensive necrotizing soft tissue infection of the perineum. Isr J Med Sci. 1996;32:745–749.
14. Antibiotic Expert Group. Tetanus prophylaxis. In: eTG complete [Internet]. Melbourne: Therapeutic Guidelines Limited<http://online.tg.org.au.proxy1.athensams.net/ip/>; 2013 [Accessed Feb. 2013].
15. Nakase JY. Update on emerging infections from the centers for disease control and prevention. Ann Emerg Med. 2000;36:268–269.
16. Weber CA, Wertheimer SJ, Ognjan A. Aeromonas hydrophila – its implications in freshwater injuries. J Foot Ankle Surg. 1995;34:442–446.
17. Amir LH, Forster DA, Lumley J, et al. A descriptive study of mastitis in Australian breastfeeding women: incidence and determinants. BMC Publ Hlth. 2007;25:62.
18. File Jr TM, Tan JS. Treatment of skin and soft-tissue infections. Am J Surg. 1995;169:27S–33S.
19. Lertzman BH, Gaspari AA. Drug treatment of skin and soft tissue infections in elderly long-term care residents. Drugs Aging. 1996;9:109–121.
20. Nelson EA, O’Meara S, Golder S, DASIDU Steering Group, et al. Systematic review of antimicrobial treatments for diabetic foot ulcers. Diabet Med. 2006;23:348–359.
21. Huizinga WK, Kritzinger NA, Bhamjee A. The value of adjuvant systemic antibiotic therapy in localised wound infections among hospital patients: a comparative study. J Infect Dis. 1986;13:11–16.
22. Singer AJ, Hollander JE, Quinn JV. Evaluation and management of traumatic lacerations. N Engl J Med. 1997;337:1142–1148.
23. Henriksen BM, Albrektsen SB, Simper LB, et al. Soft tissue infections from drug abuse A clinical and microbiological review of 145 cases. Acta Orthop Scand. 1994;65:625–628.
9.6 Hepatitis
Helen E Stergiou and Biswadev Mitra
Essentials
1 Acute and chronic viral hepatitis are of global public health importance.
2 Owing to the non-specific symptomatology in the early phases of acute viral hepatitis, definitive diagnosis may be delayed in the emergency setting.
3 Supportive care is fundamental in the management of hepatitis.
4 Prevention of viral hepatitis is possible via the introduction of public health programmes which include appropriate education regarding high-risk practices.
Introduction
Hepatitis is a non-specific clinicopathological term that encompasses all disorders characterized by hepatocellular injury and by histological evidence of a necroinflammatory response [1]. Prolific research has resulted in the identification of specific hepatotrophic viruses. An important distinction is that between acute and chronic viral hepatitis. Acute viral hepatitis refers to a process of self-limited liver injury of less than 6 months’ duration [1]. Chronic viral hepatitis is diagnosed on pathological criteria and is characterized by a duration of more than 6 months [1].
Clinical presentations of viral hepatitis
An appropriate clinical pattern of illness and specific laboratory confirmation are necessary for the diagnosis of acute viral hepatitis. Patients with acute viral hepatitis may present quite variably: they may be asymptomatic with only mildly deranged liver function tests (LFTs), they may be symptomatic with or without jaundice or they may present with fulminant disease (severe liver failure which develops within 8 weeks of symptom onset) [2].
Various clinical phases characterize acute viral hepatitis [1–4]. The incubation phase is the time between the original infection and the initial symptoms and is the time of viral replication and laboratory evidence of hepatitis. During the pre-icteric phase non-specific symptoms evolve, such as malaise, fatigue, anorexia, nausea, vomiting, myalgias, arthralgias, abdominal discomfort. If fever is present, it is generally low grade. Cough, coryza, pharyngitis and a distaste for alcohol and tobacco smoke may be evident. Rarely meningoencephalitis may occur.
The icteric phase features a variable degree of jaundice, dark urine (bilirubinuria), pale stools (absence of bile pigment in the stool), pruritus, hepatomegaly and splenomegaly. During the convalescent phase symptoms resolve, as do liver enzyme abnormalities. In patients presenting to the emergency department (ED) during the pre-icteric phase, the diagnosis may be challenging, given their non-specific symptomatology. If the patients present during the icteric phase, focused history taking, examination and the appropriate investigations should result in a definitive diagnosis.
Laboratory investigations
Blood test abnormalities are a prominent aspect of acute viral hepatitis. Serum transaminases are typically elevated at>500 μ/L and often>1000 μ/L [5]. Alanine aminotransferase (ALT) may be characteristically higher than aspartate aminotransferase (AST) [5]. Alkaline phosphatase may be normal or mildly elevated. Serum bilirubin is variably elevated and is usually divided between conjugated and unconjugated fractions. Albumin and the prothrombin time should be normal unless hepatic synthetic function is significantly impaired. Neutropaenia and lymphopaenia may be evident transiently. Severe acute hepatitis may cause hypoglycaemia. Further specific laboratory tests for viral hepatitis will be presented subsequently.
Management
In cases of acute viral hepatitis, the fundamental management is supportive care. Many of these patients can be managed on an outpatient basis. Patients require hospitalization when they have intractable vomiting with inadequate oral intake and when they demonstrate clinical features of liver failure. Bed rest is recommended during the symptomatic phase. A well-balanced diet is beneficial. It is recommended that alcohol be avoided during the acute phase, but there is no definitive evidence that alcohol consumption post-recovery causes either relapses or progression to chronic disease [2]. Given that the liver is involved in the metabolism of a plethora of drugs, all medications must be carefully prescribed to patients with acute hepatitis.
In managing fulminant hepatic failure, it is imperative that potential patients be identified as early as possible. In the emergency setting, intubation and the concomitant critical care are necessary for patients with progressive encephalopathy. Early referral to an appropriate intensive care unit (ICU) is mandatory.
Prevention and immunization
Prevention of viral hepatitis is possible via the introduction of public health programmes, improved sanitation and vaccination programmes. Post-exposure prophylaxis regimens are particularly relevant to healthcare workers.
Hepatitis A virus (HAV)
As the most common cause of viral hepatitis, HAV contributes significantly to the global burden of disease. Multiple genotypes exist and infection with one genotype confers immunity against others [6]. (See Table 9.6.1 for virology.)
Table 9.6.1
Characteristics of the main hepatitis viruses

Epidemiology
HAV is highly endemic in developing countries and can often be traced to contaminated water or food.
Natural history
Virus is excreted in the stool of the infected person for 1–2 weeks prior to and for 1 week after the onset of symptoms. A non-specific prodrome may be followed by jaundice and tender hepatomegaly. The clinical severity of the illness increases with age, with more than 80% of children being asymptomatic [1]. HAV has been associated with extrahepatic features, such as cutaneous vasculitis, renal failure, pancreatitis, bradycardia and, rarely, convulsions, transverse myelitis and aplastic anaemia [7]. Relapsing hepatitis has been described in 20% of those with HAV infection [2]. Relapses are generally benign and may occur 4–15 weeks after the original illness. Complete recovery is the typical outcome. Fulminant hepatic failure occurs in less than 1% of cases. Chronic infection never ensues.
Laboratory investigations
Serum antibody is present from the onset of HAV disease in both IgM and IgG forms. After approximately 3–12 months, anti-HAV IgM disappears and anti-HAV IgG persists, thereby conferring lifelong immunity against re-infection.
Management
Supportive management is of primary importance. Bed rest is indicated until any jaundice settles. Potentially hepatotoxic medications must be ceased. Alcohol must not be consumed during acute episodes because of the direct nephrotoxic effects.
Prevention and immunization
General measures are imperative – safe water supplies, proper sewage disposal and careful hand washing. HAV vaccines can prevent HAV infection and, importantly, they have excellent safety profiles. Persons who have been exposed to HAV and who have not been previously vaccinated should receive the vaccine within 2 weeks of exposure. Travellers to endemic areas require inactivated hepatitis vaccine, which confers long-term immunity to more than 90% of persons.
Hepatitis B virus (HBV)
Of the viral causes of hepatitis few are of greater global importance than HBV. HBV infection is endemic in certain parts of the world – Southeast Asia, China and sub-Saharan Africa. It is estimated that there are 350 million carriers worldwide [8]. By 2017, it is estimated there will be a two- to threefold increase in the number of hepatitis B-induced liver cancer cases and a marked increase in the number of deaths attributable to hepatitis B under current treatment patterns in Australia [9]. (See Table 9.6.1 for virology.)
Epidemiology
Transmission occurs by percutaneous and mucosal exposure to infected blood products and bodily fluids, hence unprotected sexual contact with infected individuals, the use of contaminated paraphernalia during intravenous drug use and vertical transmission from mother to infant are commonly implicated.
Natural history
Many acute HBV infections are asymptomatic, particularly in younger patients. The non- specific symptoms of the acute episode may be preceded by a serum-sickness syndrome with fevers, urticaria and arthralgias [5].
Approximately 90% of patients completely recover from an acute episode of HBV infection. Fulminant hepatic failure may develop in 1% of patients and has a mortality rate of up to 80%.
Progression to chronic HBV infection occurs in 5–10% of cases, with 90% of these experiencing an asymptomatic carrier state and the remaining 10% proceeding to cirrhosis and hepatocelluar carcinoma. The risk of developing chronic disease is related to the age at which HBV is first contracted – there is a greater than 90% risk of developing chronic HBV in neonates and a less than 5% risk in immunocompetent adults [1,2]. Although chronic HBV infection is generally a lifelong condition, a small percentage of infected individuals will experience complete viral eradication. In chronic HBV infection, the incidence of cirrhosis is about 2–3% per year [10]. Variables associated with progression to cirrhosis are persistence of viral replication, older age, elevation of ALT levels and HBeAg positivity [11].
Laboratory investigations
HBsAg indicates acute hepatitis or a carrier state if it persists beyond 6 months. Anti-HBc IgM indicates acute HBV and high infectivity. Anti-HBc IgG indicates previous infection. HBeAg indicates ongoing viral replication, high infectivity or chronic hepatitis.
Management
Supportive care is the primary aim of management. Household contacts require adequate education. In cases of chronic HBV infection, the aims are to suppress HBV replication and to reduce liver injury. Interferon-alpha (IFN-α) has antiviral, antiproliferative and immunomodulatory effects and is an effective treatment option against HBV. Patients with normal serum ALT levels have a poor response to interferon- alpha because the lack of hepatic dysfunction is suggestive of low immune-mediated hepatic inflammation. The limiting factor in the use of IFN-α is the side-effect profile, which includes an influenza-like illness, gastrointestinal symptoms, psychological sequelae (particularly depression), bone marrow suppression, thyroid dysfunction and possible birth defects [5]. Lamivudine is an oral nucleoside analogue that potently inhibits HBV DNA synthesis. Human monoclonal antibodies may be directed against different epitopes of HBsAg, bind HBV particles and reduce serum viral titres and HBsAg levels. Further research is ongoing.
Prevention and immunization
The pre-exposure administration of HBV vaccine is fundamental to immunoprophylaxis. The vaccine is protective in over 90% of individuals [1]. Current recommendations include all infants at birth and individuals with high exposure risk, such as healthcare personnel, injecting drug users and high-risk sexual workers. Antibody titres may decrease with time, but the protective effects persist. The risk of HBV infection in the occupational setting is related primarily to the degree of contact with blood and to the HBeAg status of the donor. In needle-stick injuries, the risk of developing clinical hepatitis if the blood is both HBsAg- and HBeAg-positive has been estimated to be up to 30%. Post-exposure prophylaxis involves the administration of hepatitis B immunoglobulin in addition to the recombinant vaccine series.
Hepatitis C virus (HCV)
International studies estimate that up to 3% of the world’s population is infected with HCV [2,12]. (See Table 9.6.1 for virology.) The identification of six major genotypes of the HCV has important clinical implications in that such genomic sequence variation makes vaccine development extremely difficult.
Epidemiology
Parenteral exposure leads to HCV infection, the use of contaminated needles and syringes being a predominant factor. Sexual and perinatal transmission of HCV is negligible. Transfusion-related HCV transmission has essentially been eradicated via donor screening. Up to 10% of HCV cases do not have an identifiable source of infection.
Natural history
A pre-icteric phase featuring non-specific symptoms develops in 15–20% of patients. When the icteric phase develops it typically lasts for 1–2 weeks. Fulminant hepatic failure rarely results from acute HCV infection.
Following an acute episode, 75–85% of adults and 55% of children will enter a chronic phase [12]. There is a high proportion of subclinical chronic HCV infection, hence patients may not manifest any pathology until incidental blood tests or end-stage liver disease many years after the initial infection. Approximately 20–30% of chronic HCV patients develop cirrhosis, with subsequent hepatocellular carcinoma occurring in up to 20% of the latter group [1,5].
Laboratory investigations
A fluctuating titre of HCV RNA is detectable within days to weeks of the initial HCV infection. The rate at which HCV antibodies develop is variable. Notably, HCV antibodies are neither neutralizing nor protective. It may not be possible to distinguish between acute and chronic HCV infection, given that the same laboratory markers can be present in both conditions. Further specific laboratory tests for viral hepatitis are presented in Table 9.6.2.
Table 9.6.2
Laboratory tests in viral hepatitis
|
Test |
Interpretation of positive test |
Clinical significance |
|
Tests for HAV |
||
|
Anti-HAV IgM |
Recently acquired HAV |
Acute hepatic illness |
|
Anti-HAV IgG |
Previous infection/vaccination |
Immunity |
|
Tests for HBV |
||
|
HBsAg (surface Ag) |
Current/chronic infection |
Structural viral component |
|
Anti-HBsAg (surface Ab) |
Previous infection/vaccination |
Immunity |
|
Anti-HBclgM (core Ab) |
Recently acquired HBV |
Test for acute HBV |
|
HBeAg |
Marker of viral replication |
High infectivity |
|
Anti-HBeAg |
No viral replication |
Low infectivity |
|
HBV DNA |
Complete virus present |
High infectivity |
|
Tests for HCV |
||
|
Anti-HCV |
HCV exposure |
Variable infectivity |
|
HCV RNA |
Virus present |
|
|
Tests for HDV |
||
|
Anti-HDV IgG/IgM |
HDV exposure |
Acute or chronic HDV |
|
Delta Ag |
HDV present |
Acute or chronic HDV |
|
Tests for HEV |
||
|
Anti-HEV IgM |
Recently acquired HEV |
Acute hepatic illness |
|
Anti-HEV IgG |
Previous exposure |
Modified from Talley N, Martin C. Clinical Gastroenterology: A Practical Problem-based Approach, 2nd edn. Edinburgh: Churchill Livingstone, 2006 with permission.
Management
Supportive management is fundamental in addressing HCV infection. Relevant education and counselling regarding high-risk behaviours and referrals to appropriate support networks are necessary. Avoidance of alcohol is advisable as some studies indicate that alcohol may promote the progression of HCV infection [13,14]. Standard therapy for HCV infection has consisted of a combination of pegylated interferon-α and ribavirin. However, the combination therapy leads to cure in only abut 50% of cases [15]. Recent advances include the use of HCV protease inhibitors, polymerase inhibitors, NS5A inhibitors and host factor inhibitors, such as cyclophilin antagonists [16,17].
Prevention and immunization
Currently, there is no effective vaccination available against HCV infection, nor is there any specific post-exposure prophylaxis regimen. Vaccination against HAV and HBV is advisable. HCV is not transmitted efficiently through occupational exposures to blood. The average incidence of anti-HCV seroconversion after accidental exposure from an HCV-positive source is<2% [18].
Hepatitis D virus (HDV)
As a defective virus, HDV requires the presence of HBV for virion assembly and for viral replication [1,2]. (See Table 9.6.1 for virology.)
Epidemiology
Only patients with acute or chronic HBV infection are susceptible to infection with HDV. An estimated 5% of HBV carriers are infected with HDV worldwide [1,2,5]. Parenteral exposure is the primary transmission mode. HDV can occur as a co-infection with acute HBV (acquired at the same time) or as a superinfection in chronic HBV carriers.
Natural history
In cases of HDV and HBV co-infection, acute HDV infection generally presents as a benign acute hepatitis with subsequent resolution in up to 80–95% of patients. Chronic HDV/HBV infection may occur in 5–10% of patients [2]. HDV superinfection results in progression to chronic HDV/HBV in 70–80% of cases [5]. Chronic HDV/HBV infection manifests as a chronic healthy carrier state or severe liver disease. HDV superinfection may result in fulminant hepatitis in 2–20% of cases [5]. Chronic HDV infection leads to more severe liver disease than HBV monoinfection and is associated with accelerated fibrosis progression, earlier hepatic decompensation and an increased risk for the development of hepatocellular carcinoma.
Laboratory investigations
HBsAg must be detected to diagnose acute HDV/HBV co-infection. Anti-HDV IgM is transiently present in acute infections. Anti-HDV IgG appears late in acute infections.
Management
There is no specific cure for HDV infection other than suppressing HBV replication. IFN-α treatment has proven antiviral activity against HDV in humans and has been linked to improved long-term outcomes. Studies conducted in the past 2 years on the use of PEG-IFN-α show that a sustained virologic response to therapy, measured in terms of undetectable serum HDV RNA levels, can be achieved in about one-quarter of patients with hepatitis D [19].
Prevention and immunization
Currently, there is no vaccine for preventing HDV infection. HBV immunization has been shown to provide protection against the development of HDV.
Hepatitis E virus (HEV)
See Table 9.6.1 for virology.
Epidemiology
HEV is endemic in developing countries, such as Southeast and Central Asia and the Indian subcontinent. The primary transmission mode is the faecal–oral route, with contaminated drinking water and food supplies being primary sources of infection. Young adults are often predominantly affected.
Natural history
The clinical course is similar to that of acute HAV infection. Full recovery from the acute HEV infection is the norm. There have not been any recorded cases of chronic HEV infection.
Overall mortality from acute HEV infection is about 5%. For reasons which remain unclear, fulminant hepatic failure with a subsequent high mortality rate occurs in 25% of women with HEV infection during the third trimester of pregnancy. Liver transplant recipients may be at a greater risk for HEV infection, which can lead to chronic hepatitis.
Laboratory investigations
Anti-HEV IgM occurs between 1 week and 6 months after the illness onset. Anti-HEV IgG is evident during the convalescent phase or post-exposure.
Management
Supportive management is the key.
Prevention and immunization
Disease control depends on good personal hygiene and improved environmental sanitation. There is no effective vaccine.
Hepatitis G virus (HGV)
Exposure to blood products is a recognized route of acquisition of HGV infection in humans. Chronic viraemia results and reported prevalences of HGV infection range from 1% to 3% in most populations, incidences that are higher than those of either HBV or HCV in these populations [20]. A causal relationship between the prevalence of HGV and hepatitis, however, has not been proven. Although HGV RNA may persist in serum of patients acutely infected with HGV for as long as 16 years, in about 90% of these patients persistence is not accompanied by evidence of hepatocellular injury [21]. Currently, there should be no need to test for HGV in the emergency setting.
Non-hepatotrophic viruses
Several non-ABCDE viruses cause viral hepatitis. The cytomegalovirus (CMV) and Epstein–Barr virus (EBV) commonly contribute to abnormal LFTs and icteric hepatitis may also occasionally be noted. In immunocompromised patients, herpes simplex may lead to a hepatitic picture. Progression to chronic hepatitis has not been demonstrated with any of these viruses.
Non-viral hepatitis
Of the causes of non-viral hepatitis, the following are important in the emergency setting: alcoholic hepatitis, non-alcoholic steatohepatitis (NASH), drug-induced hepatitis and autoimmune hepatitis.
Alcoholic hepatitis
Alcoholic hepatitis is an important clinical syndrome which is variably characterized by anorexia, nausea, jaundice, hepatomegaly and features of portal hypertension, such as ascites and encephalopathy. Cirrhosis and death are possible sequelae if the patients do not cease their alcohol consumption.
Non-alcoholic steatohepatitis
Defects in the processing of fatty acids through the liver may cause steatosis-induced inflammation (steatohepatitis). Ten to 50% of patients with NASH are at risk of developing cirrhosis [14].
Drug-induced hepatitis
Toxic exposure to certain medications, vitamins, herbal remedies and food supplements may result in a drug-induced hepatitis. Drug-induced hepatitis may occur as an expected consequence of a drug’s toxicity profile or as an idiosyncratic reaction to a standard dose. Hepatotoxic agents result in variable clinicopathological patterns of liver injury via toxic and immune mechanisms [22]. Commonly, the formation of reactive hepatotoxic metabolites is the primary underlying mechanism [23]. Extensive lists of hepatotoxic drugs can be found in the literature. Acute liver injury may be necroinflammatory (e.g. paracetamol), cholestatic (e.g. chlorpromazine) or of a mixed type. Table 9.6.3 lists drugs which may induce hepatitis and which are encountered in the emergency setting [22,23].
Table 9.6.3
Hepatitis-inducing drugs [13,14]
|
Drug |
Pathology |
|
Allopurinol |
Hepatic granulomas |
|
Cloxacillin |
Lobular hepatitis |
|
Chlorpromazine |
Cholestatic hepatitis |
|
Dantrolene |
Cytolytic hepatitis |
|
Erythromycin |
Cholestasis with hepatitis |
|
Flucloxacillin |
Cholestatic hepatitis |
|
Halothane |
Hepatocellular injury |
|
Isoniazid |
Cytolytic hepatitis |
|
Non-steroidals |
Primarily cholestasis |
|
Paracetamol |
Cytolytic hepatitis |
|
Phenothiazines |
Cholestatic hepatitis |
|
Phenytoin |
Non-caseating granulomas |
|
Sulphonamides |
Cytolytic hepatitis |
After Thomas D, Astemborski J, Rai R. The natural history of hepatitis C virus infection. Journal of the American Medical Association 2000;284:45; Friedman L, Keeffe E, Schiff E. Handbook of Liver Disease, 2nd edn. Edinburgh: Churchill Livingstone, 2004.
Autoimmune hepatitis
Autoimmune hepatitis is a self-perpetuating hepatocellular inflammation of unknown cause which is associated with hypergammaglobulinaemia and serum antibodies [14]. Fatigue, anorexia and jaundice may progress to liver failure. Corticosteroids are the basis of treatment.
Future directions
![]() Global emphasis on adequate public health schemes, including vaccination programmes, to control the transmission of viral hepatitis.
Global emphasis on adequate public health schemes, including vaccination programmes, to control the transmission of viral hepatitis.
![]() Emphasis on public education regarding high-risk practices.
Emphasis on public education regarding high-risk practices.
![]() Surveillance of the long-term immunity conferred by the hepatitis A and B vaccinations.
Surveillance of the long-term immunity conferred by the hepatitis A and B vaccinations.
![]() Development of a vaccine for hepatitis C.
Development of a vaccine for hepatitis C.
![]() Optimization of the management algorithms for chronic viral hepatitis.
Optimization of the management algorithms for chronic viral hepatitis.
References
1. Yamada T, Hasler W, Inadomi J, et al. Handbook of gastroenterology 2nd ed. Baltimore: Lippincott Williams & Wilkins; 2005.
2. Mandell G, Bennett J, Dolin R. Principles and practice of infectious diseases 6th ed. Edinburgh: Churchill Livingstone; 2005.
3. Talley N, Martin C. Clinical gastroenterology: a practical problem-based approach 2nd ed. Edinburgh: Churchill Livingstone; 2006.
4. Boon N, Colledge N, Walker B, et al. Davidson’s principles and practice of medicine 20th ed. Edinburgh: Churchill Livingstone; 2006.
5. Friedman S, McQuaid K, Grendell J. Current diagnosis and treatment in gastroenterology 2nd ed. McGraw-Hill 2003.
6. Lemon S, Jansen RW, Brown EA, et al. Genetic, antigenic and biological differences between strains of hepatitis A virus. Vaccine. 1992;10:S40–S44.
7. Schiff E. Atypical clinical manifestations of hepatitis A Vaccine. 1992;10:S18–S20.
8. Kane M, Clements J, Hu D. Disease control priorities in developing countries. In: Jamison D, Mosley W, Measham A, Bobadilla J, eds. Disease control priorities in developing countries. New York: Oxford University Press; 1993;330.
9. Homewood J, Coory M, Dinh B. Cancer among people living in rural and remote indigenous communities in Queensland: an update 1997–2002. Queensland Health 2005.
10. Liaw YF, Tai DI, Chu CM, et al. The development of cirrhosis in patients with chronic type B hepatitis: a prospective study. Hepatology. 1988;8:493–496.
11. McMahon BJ, Holck P, Bulkow L, et al. Serologic and clinical outcomes of 1536 Alaska natives chronically infected with hepatitis B virus. Ann Intern Med. 2001;135:759–768.
12. Lavanchy D. Public health measures in the control of viral hepatitis: a World Health Organization perspective for the next millennium. J Gastroenterol Hepatol. 2002;17:S452–S459.
13. Thomas D, Astemborski J, Rai R. The natural history of hepatitis C virus infection. J Am Med Assoc. 2000;284:45.
14. Friedman L Keeffe E, Schiff E. Handbook of liver disease 2nd ed. Edinburgh: Churchill Livingstone; 2004.
15. Fried MW, Shiffman ML, Reddy KR, et al. Peginterferon alfa-2a plus ribavirin for chronic hepatitis C virus infection. N Engl J Med. 2002;347:975–982.
16. Gane EJ, Roberts SK, Stedman CA, et al. Oral combination therapy with a nucleoside polymerase inhibitor (RG7128) and danoprevir for chronic hepatitis C genotype 1 infection (INFORM-1): a randomised, double-blind, placebo-controlled, dose-escalation trial. Lancet. 2010;376:1467–1475.
17. Lok AS, Gardiner DF, Lawitz E, et al. Preliminary study of two antiviral agents for hepatitis C genotype 1. N Engl J Med. 2012;366:216–224.
18. Mitsui T, Iwano K, Masuko K. Hepatitis C virus infection in medical personnel after needlestick accident. Hepatology. 1992;16:1109–1114.
19. Wedemeyer H, Manns MP. Epidemiology, pathogenesis and management of hepatitis D: update and challenges ahead. Nat Rev Gastroenterol Hepatol. 2013;7:31–40.
20. Yoshibo M, Okamoto H, Mishiro S, et al. Detection of GBV-C virus genome in serum of patients with fulminant hepatitis of unknown origin. Lancet. 1995;346:1131–1134.
21. Alter MJ. The cloning and clinical implications of HGV and HGBV-C. N Engl J Med. 1996;334:1536–1537.
22. Farrell G. Drug-induced liver injury New York: Churchill Livingstone; 1994.
23. Bircher J, Benhamou J-P, Mclntyre N, et al. Oxford textbook of clinical hepatology 2nd ed. Oxford: Oxford University Press; 1999.
9.7 HIV/AIDS
Alan C Street
Essentials
1 Patients with previously undiagnosed human immunodeficiency virus (HIV) infection may present to the emergency department at any time during the course of infection, from early (acute seroconversion illness) to late (AIDS-defining illness) stages.
2 Patients with previously diagnosed HIV infection may present with complications of antiretroviral therapy or, if therapy has failed or is not taken, with a range of HIV-related clinical syndromes.
3 Globally, heterosexual transmission accounts for most HIV infections but, in Australia, HIV infection remains predominantly a disease of homosexual and bisexual men.
4 Most AIDS-defining illnesses occur when the CD4 T-lymphocyte count is<0.2×109/L (bacterial pneumonia and tuberculosis are exceptions).
5 Serious non-AIDS events which are not classically associated with HIV infection, such as cardiovascular disease, bone disease, renal disease and cognitive impairment, cause significant morbidity, may occur at higher CD4 cell counts and are possibly related to chronic inflammation.
6 Combination antiretroviral therapy with well-tolerated and potent once-daily regimens dramatically reduces HIV mortality and morbidity, reduces the risk of HIV transmission from the infected individual to his or her partner and may decrease HIV transmission at a population level (treatment as prevention).
7 Close liaison between emergency department staff and the patient’s hospital or local doctor is vital for optimal management of HIV-infected patients.
Introduction
HIV medicine is a complex and specialized field and emergency physicians are not the usual primary care providers for people with HIV infection. However, the emergency department (ED) is often the first point of contact for patients presenting with acute HIV-related complications, whether or not they have already been diagnosed with HIV.
Emergency medicine physicians do not need to be HIV experts, but they should develop knowledge and skills in the following areas:
![]() the natural history and clinical manifestations of HIV infection
the natural history and clinical manifestations of HIV infection
![]() the principles of HIV diagnosis, including the ability to engage patients in discussions about HIV testing and test results
the principles of HIV diagnosis, including the ability to engage patients in discussions about HIV testing and test results
![]() the principles of early management of patients with common HIV-related disease syndromes
the principles of early management of patients with common HIV-related disease syndromes
![]() familiarity with antiretroviral agents in current use, including toxicity and drug interactions.
familiarity with antiretroviral agents in current use, including toxicity and drug interactions.
The first cases of AIDS were recognized in the USA in 1981 and in Australia in 1982. The causative agent, human immunodeficiency virus (HIV), was discovered in 1984 and a diagnostic blood test developed soon thereafter. In 1986, the first effective antiviral drug (AZT, later renamed zidovudine) became available. Since the late 1990s, the use of combination antiretroviral therapy has led to dramatic reductions in HIV-associated morbidity and mortality in resource-rich countries. Antiretroviral use is rapidly increasing in poor countries, but the global HIV situation remains serious; in 2011, the World Health Organization (WHO) estimated that there were 2.5 million new HIV infections, 1.7 million deaths and 34 million people living with HIV, 23.5 million in sub-Saharan Africa and 4.8 million in Asia [1]. A major challenge in the coming years will be to develop an effective HIV vaccine.
Epidemiology
Globally, the great majority of HIV infections arise as a result of heterosexual transmission. In developed countries, injecting drug use and sex between men account for a greater proportion of HIV infections, although the contribution of specific behaviours to overall transmission varies greatly within and between countries and over time.
In Australia up to December 2011, more than 31 000 people had been diagnosed with HIV infection, of whom an estimated 24 730 are living with HIV. Seventy-nine per cent of people infected with HIV report male-to-male sex, 15% have become infected through heterosexual transmission, 4% of cases have occurred in injecting drug users and 2% in recipients of contaminated blood or blood products. Women account for 9% of HIV-infected people and children for less than 1% [2]. Approximately 1000 new HIV diagnoses have been notified for each of the past few years, mostly among gay men. Compared to some other countries, the prevalence of HIV infection in injecting drug users has remained low, of the order of 1–2%.
Pathogenesis
Once HIV infection becomes established, one billion or more HIV virus particles are produced per day, chiefly in lymph nodes and other lymphoid tissue, accompanied by the daily turnover of up to 1 billion CD4 T lymphocytes. The number of CD4 cells falls secondary to mechanisms such as immune activation and direct infection of CD4 cells, resulting in reduced helper function for cell-mediated and humoral immunity [3].
HIV replication occurs at a relatively constant rate, producing a stable level of HIV in the blood and this can be measured with quantitative HIV RNA detection tests. The HIV viral load is used as a prognostic marker (because it is associated with the rate at which CD4 T lymphocytes are lost) and to monitor the efficacy of antiretroviral therapy.
The peripheral blood CD4 T-lymphocyte count is an accurate indicator of the degree of immunosuppression. The normal count is 0.5–1.5×109/L; susceptibility to opportunistic infection and to most other serious HIV-related complications is greatest when the CD4 cell count is less than 0.2×109/L. In untreated patients, the average rate of CD4 cell decline is 0.05–0.1×109/year.
A wide variety of chronic medical conditions not previously associated with HIV infection, such as cardiovascular, bone and kidney disease, mild cognitive impairment and non-HIV related cancers, are more common in HIV-infected patients [4]. Predisposition to these serious non-AIDS events results from a complex interplay between an ageing HIV-infected population, traditional risk factors, side effects of some antiretroviral agents and HIV infection itself. Chronic inflammation induced by HIV infection, which may persist despite effective antiretroviral therapy, is thought to mediate some of the direct HIV effects.
Classification and natural history (Fig. 9.7.1)
HIV infection can be conveniently divided into four stages on the basis of time after infection, CD4 T-lymphocyte count and the presence of complications [5]:
![]() primary infection: a febrile illness that occurs soon after acquisition of HIV infection (discussed in more detail below)
primary infection: a febrile illness that occurs soon after acquisition of HIV infection (discussed in more detail below)
![]() early infection: CD4 cell count>0.5×109/L – generally asymptomatic period
early infection: CD4 cell count>0.5×109/L – generally asymptomatic period
![]() intermediate infection: CD4 cell count 0.2–0.5×109/L – asymptomatic or less serious complications
intermediate infection: CD4 cell count 0.2–0.5×109/L – asymptomatic or less serious complications
![]() late infection: CD4 cell count<0.2 × 109/L – susceptibility to AIDS-defining opportunistic infections and malignancies.
late infection: CD4 cell count<0.2 × 109/L – susceptibility to AIDS-defining opportunistic infections and malignancies.

FIG. 9.7.1 Natural history of untreated HIV infection. ‘Time’ represents time after infection. (Modified with permission from Stewart G (ed.). Managing HIV. Sydney: Australasian Medical Publishing Co, 1997.)
Patients are categorized as having AIDS when they develop a defined opportunistic infection, an HIV-related malignancy, a wasting syndrome or AIDS dementia complex.
Presentation
Patients with underlying HIV infection who present to the ED fall into three distinct groups. First, they may present with a manifestation of previously unrecognized HIV infection. To identify these patients, the physician must know who is potentially at risk of HIV infection (see Epidemiology above) and be aware of the many different ways in which previously undiagnosed HIV infection may present. Prompt consideration of the possibility of HIV infection is important because the differential diagnosis of the presenting problem will broaden to encompass a variety of other conditions, some of which may be life threatening and require a different approach to initial investigation and treatment.
The second group includes those who are already known to be HIV infected. Many of these patients will have been started on antiretroviral therapy; serious HIV-related infections or malignancies are uncommon in this group, but ED presentations may be related to complications of therapy or to the chronic medical conditions associated with HIV infection discussed in the section above. A smaller group of patients have developed resistance to or are intolerant of antiretroviral agents or decline to start or remain on treatment; these patients usually present with one of a limited number of classic, HIV-related clinical syndromes, such as fever and cough or shortness of breath, diarrhoea, unexplained fever or neurological symptoms. The initial diagnostic and treatment approach is based on knowledge of the differential diagnosis for each of these syndromes.
Finally, there will be patients whose ED presentation is not related to an HIV complication at all but who have clinically silent, ‘incidental’ HIV infection. Readily identifiable groups who may be in this category are patients with a sexually transmitted infection and patients with hepatitis B or hepatitis C infection. Otherwise, a brief history, including a sexual history, is required to elicit HIV risk factors. Presentation of these patients to the ED offers an important opportunity to explore HIV risk factors and to discuss the benefits of earlier HIV diagnosis and the desirability of HIV testing.
Previously undiagnosed HIV infection [6]
Primary HIV infection (acute seroconversion illness) (Table 9.7.1)
Up to 50% of patients will develop a glandular fever-like illness of varying severity 2–3 weeks after acquiring HIV. The most common features are fever, myalgia, headache, erythematous maculopapular rash, diarrhoea, lymphadenopathy and mouth ulcers. Complications include aseptic meningitis, encephalitis and Guillain–Barré syndrome. The diagnosis is often missed at this stage; patients may be thought to have a ‘viral illness’, such as infectious mononucleosis or, if a patient develops a complication, more common causes (for example, herpes simplex in a patient with encephalitis) and not HIV are considered.
Table 9.7.1
Manifestations of primary HIV infection
|
Common (present in>30% of patients) |
Less common |
Complications |
|
Fever |
Diarrhoea |
Aseptic meningitis |
|
Rash |
Generalized lymphadenopathy |
Guillain–Barré syndrome |
|
Myalgia/arthralgia |
Painful swallowing |
Encephalitis |
|
Headache |
Abdominal pain |
Interstitial pneumonitis |
|
Pharyngitis |
Cough |
Rhabdomyolysis |
|
Cervical lymphadenopathy |
Photophobia |
Haemophagocytic syndrome |
|
Mouth ulcers |
Tonsillitis |
Early infection (CD4 cell count>0.5×109/L)
People are generally healthy during this phase. Thrombocytopaenia may occur and so HIV infection should be considered in appropriate patients with idiopathic thrombocytopenia.
Intermediate HIV infection (CD4 cell count 0.2–0.5 × 109/L)
This is a phase when previously undiagnosed HIV-infected patients often present with HIV-related conditions, but the clues may not be recognized as such and the underlying diagnosis can be missed. Manifestations that will alert the astute clinician include:
![]() minor infections: shingles, severe or very frequent orolabial or genital herpes, oral thrush
minor infections: shingles, severe or very frequent orolabial or genital herpes, oral thrush
![]() skin conditions: extensive seborrhoeic dermatitis, worsening psoriasis
skin conditions: extensive seborrhoeic dermatitis, worsening psoriasis
![]() constitutional symptoms: fever, weight loss, diarrhoea
constitutional symptoms: fever, weight loss, diarrhoea
![]() generalized lymphadenopathy
generalized lymphadenopathy
![]() more serious complications: bacterial pneumonia (especially recurrent), tuberculosis and, rarely, Kaposi’s sarcoma or non-Hodgkin’s lymphoma.
more serious complications: bacterial pneumonia (especially recurrent), tuberculosis and, rarely, Kaposi’s sarcoma or non-Hodgkin’s lymphoma.
Late HIV infection (CD4 cell count<0.2×109/L)
It is often not appreciated that patients may remain completely well during the early and intermediate stages of HIV infection and only present when they develop a serious HIV-related complication, such as an opportunistic infection; the ED is a common point of initial care for such patients. If the history reveals risk factors for HIV infection, HIV testing can be performed and initial investigations directed at specific HIV-related complications. However, if the patient does not volunteer this information, is not specifically asked about HIV risk factors or does not belong to a ‘conventional’ HIV risk group, diagnosis of the presenting illness and the underlying HIV infection are often delayed.
The following clinical situations (discussed in more detail in the following section) should prompt consideration of the possibility of underlying HIV infection:
![]() Diffuse bilateral pulmonary infiltrates (as a manifestation of Pneumocystis jiroveci pneumonia, PCP) – this is the commonest serious opportunistic infection in patients with previously undiagnosed HIV infection; it is often misdiagnosed as atypical pneumonia, leading to incorrect initial treatment with a macrolide agent or doxycycline
Diffuse bilateral pulmonary infiltrates (as a manifestation of Pneumocystis jiroveci pneumonia, PCP) – this is the commonest serious opportunistic infection in patients with previously undiagnosed HIV infection; it is often misdiagnosed as atypical pneumonia, leading to incorrect initial treatment with a macrolide agent or doxycycline
![]() Ring-enhancing space-occupying cerebral lesion – in an HIV-uninfected patient, the usual causes are tumour or bacterial brain abscess and brain biopsy is required whereas, in the setting of HIV infection, cerebral toxoplasmosis is the most likely diagnosis and brain biopsy can usually be avoided
Ring-enhancing space-occupying cerebral lesion – in an HIV-uninfected patient, the usual causes are tumour or bacterial brain abscess and brain biopsy is required whereas, in the setting of HIV infection, cerebral toxoplasmosis is the most likely diagnosis and brain biopsy can usually be avoided
![]() Tuberculosis – although the overlap between those at risk for HIV and tuberculosis is not as great in Australia as in resource-poor countries with a high HIV burden, all patients with tuberculosis should be encouraged to undergo HIV testing after appropriate counselling
Tuberculosis – although the overlap between those at risk for HIV and tuberculosis is not as great in Australia as in resource-poor countries with a high HIV burden, all patients with tuberculosis should be encouraged to undergo HIV testing after appropriate counselling
![]() Kaposi’s sarcoma – well-developed lesions (purple, oval and nodular) are easy to recognize, but early lesions are often non-descript (brown or pink and flat) and biopsy may be required for diagnosis
Kaposi’s sarcoma – well-developed lesions (purple, oval and nodular) are easy to recognize, but early lesions are often non-descript (brown or pink and flat) and biopsy may be required for diagnosis
![]() Other presentations – unexplained cytopaenias (anaemia or pancytopenia) and other AIDS-defining conditions, such as non-Hodgkin’s lymphoma, cryptococcal meningitis, chronic cryptosporidial diarrhoea or AIDS dementia complex (manifesting as impaired cognition and motor performance) are occasionally the first manifestation of previously unsuspected HIV infection.
Other presentations – unexplained cytopaenias (anaemia or pancytopenia) and other AIDS-defining conditions, such as non-Hodgkin’s lymphoma, cryptococcal meningitis, chronic cryptosporidial diarrhoea or AIDS dementia complex (manifesting as impaired cognition and motor performance) are occasionally the first manifestation of previously unsuspected HIV infection.
Previously diagnosed HIV infection [7]
Patients with known HIV infection are much less likely now than in the past to present with the classic AIDS-related clinical syndromes indicative of advanced immunodeficiency because of the effectiveness of modern antiretroviral treatment. However, some previously diagnosed patients have failed antiretroviral therapy or have elected not to start or continue treatment and are still susceptible to AIDS-defining conditions. (Otherwise, these presentations involve patients with previously unrecognized HIV infection, as discussed in the above section.) Patients with known HIV infection may also present with complications of antiretroviral therapy, with chronic medical conditions now associated with HIV and of course with a problem not related to HIV at all.
Cough, shortness of breath, fever
Respiratory pathogens are listed in Table 9.7.2. The most important issue to decide is whether the patient has Pneumocystis jiroveci pneumonia or not, because this complication is common and potentially serious. Tuberculosis must also be considered because of the need to place the patient in respiratory isolation.
![]() PCP (occurs in patients with<0.2×109 CD4 cells/L): the presentation is subacute or chronic, with a non-productive cough, dyspnoea, fever and chest tightness. Physical examination reveals fever, tachypnoea and reduced chest expansion, but chest auscultation is often normal. PCP is very unlikely in patients taking regular co-trimoxazole because this drug is virtually 100% effective as PCP prophylaxis.
PCP (occurs in patients with<0.2×109 CD4 cells/L): the presentation is subacute or chronic, with a non-productive cough, dyspnoea, fever and chest tightness. Physical examination reveals fever, tachypnoea and reduced chest expansion, but chest auscultation is often normal. PCP is very unlikely in patients taking regular co-trimoxazole because this drug is virtually 100% effective as PCP prophylaxis.
![]() Bacterial pneumonia (may occur when the CD4 cell count is>0.2×109/L): patients usually present with a short history, a productive cough and sometimes pleuritic chest pain. Physical examination may be normal or reveal signs of consolidation, a pleural rub or pleural effusion.
Bacterial pneumonia (may occur when the CD4 cell count is>0.2×109/L): patients usually present with a short history, a productive cough and sometimes pleuritic chest pain. Physical examination may be normal or reveal signs of consolidation, a pleural rub or pleural effusion.
![]() Tuberculosis: the clinical features vary according to the degree of immunosuppression. If the CD4 cell count is>0.2×109/L, patients usually present with typical symptoms and signs of tuberculosis (chronic cough, haemoptysis, fever and weight loss) but, in late-stage infection, atypical manifestations, such as disseminated disease, are common and diagnosis is more difficult.
Tuberculosis: the clinical features vary according to the degree of immunosuppression. If the CD4 cell count is>0.2×109/L, patients usually present with typical symptoms and signs of tuberculosis (chronic cough, haemoptysis, fever and weight loss) but, in late-stage infection, atypical manifestations, such as disseminated disease, are common and diagnosis is more difficult.
Table 9.7.2
Respiratory complications in HIV-infected patients

Focal neurological signs, convulsions or altered conscious state
These features generally indicate the presence of an intracerebral space-occupying lesion, the most common causes of which are:
![]() Cerebral toxoplasmosis: this infection occurs when the CD4 cell count is<0.2×109/L. The specific focal features depend on the site of the usually multiple lesions and may include hemiparesis, visual field defects, personality change or cerebellar signs.
Cerebral toxoplasmosis: this infection occurs when the CD4 cell count is<0.2×109/L. The specific focal features depend on the site of the usually multiple lesions and may include hemiparesis, visual field defects, personality change or cerebellar signs.
![]() Primary intracerebral lymphoma: this complication occurs with advanced HIV infection (CD4 cell count usually<0.05×109/L), developed in 2–3% of AIDS patients prior to the development of effective antiretroviral therapy and is closely associated with Epstein–Barr virus (EBV) infection. Clinical presentation is indistinguishable from that of cerebral toxoplasmosis.
Primary intracerebral lymphoma: this complication occurs with advanced HIV infection (CD4 cell count usually<0.05×109/L), developed in 2–3% of AIDS patients prior to the development of effective antiretroviral therapy and is closely associated with Epstein–Barr virus (EBV) infection. Clinical presentation is indistinguishable from that of cerebral toxoplasmosis.
![]() Progressive multifocal leucoencephalopathy: caused by JC virus (a polyoma virus); patients present with cognitive decline or focal signs and seizures are relatively uncommon. Differentiation from cerebral toxoplasmosis and primary cerebral lymphoma requires computed tomography (CT) or magnetic resonance imaging (MRI) (see below).
Progressive multifocal leucoencephalopathy: caused by JC virus (a polyoma virus); patients present with cognitive decline or focal signs and seizures are relatively uncommon. Differentiation from cerebral toxoplasmosis and primary cerebral lymphoma requires computed tomography (CT) or magnetic resonance imaging (MRI) (see below).
Diarrhoea, with or without abdominal pain or fever
A wide range of gastrointestinal pathogens cause diarrhoea in HIV-infected patients (Table 9.7.3). Patients should be asked about recent travel or antibiotic use. Bloody, small-volume diarrhoea with cramping lower abdominal pain is suggestive of a large bowel pathogen, such as cytomegalovirus, Entamoeba histolytica or Clostridium difficile, whereas profuse watery diarrhoea suggests an infection of the small bowel, such as cryptosporidiosis. However, clinical features are often of limited diagnostic value and the specific diagnosis rests on identification of the pathogen in a faecal or biopsy specimen. Prominent anal pain or tenesmus suggests the possibility of proctitis due to a sexually acquired infection, such as gonorrhoea, Chlamydia (including lymphogranuloma venereum) or herpes.
Table 9.7.3
Gastrointestinal pathogens in HIV-infected patients

Fever without localizing features
This is chiefly a problem in those with a CD4 cell count<0.2×109/L. The differential diagnosis is extensive, the major causes being:
![]() disseminated opportunistic infections: disseminated Mycobacterium avium complex (MAC), disseminated tuberculosis, disseminated histoplasmosis (USA and South America), Salmonella bacteraemia, CMV
disseminated opportunistic infections: disseminated Mycobacterium avium complex (MAC), disseminated tuberculosis, disseminated histoplasmosis (USA and South America), Salmonella bacteraemia, CMV
![]() focal opportunistic infections with non-focal presentation: PCP, cryptococcal meningitis, tuberculosis
focal opportunistic infections with non-focal presentation: PCP, cryptococcal meningitis, tuberculosis
![]() bacterial infections: sinusitis, bacterial pneumonia, primary bacteraemia (especially in patients with an indwelling long-term intravenous device, or neutropenia)
bacterial infections: sinusitis, bacterial pneumonia, primary bacteraemia (especially in patients with an indwelling long-term intravenous device, or neutropenia)
![]() non-HIV specific infections: right-sided endocarditis, secondary syphilis
non-HIV specific infections: right-sided endocarditis, secondary syphilis
![]() non-infectious causes: non-Hodgkin’s lymphoma, drug fever.
non-infectious causes: non-Hodgkin’s lymphoma, drug fever.
Difficult or painful swallowing
This is usually due to Candida oesophagitis, in which case coexisting oral candidiasis is often present. Other causes include CMV oesophagitis, herpes simplex oesophagitis and idiopathic aphthous ulceration.
Headache, fever, neck stiffness
Cryptococcal meningitis is the most common cause of this syndrome, although headache may be mild and signs of meningism subtle or absent. Less common causes include tuberculous meningitis, syphilitic meningitis, HIV itself and lymphomatous meningitis.
Complications of antiretroviral therapy
Antiretroviral drugs are discussed in more detail below and drug side effects are outlined in Table 9.7.4. Examples of more serious side effects and treatment complications that may prompt presentation to the ED include:
![]() pancreatitis – didanosine*
pancreatitis – didanosine*
![]() hepatitis – nevirapine
hepatitis – nevirapine
![]() drug rash – nevirapine, abacavir, fosamprenavir*, efavirenz
drug rash – nevirapine, abacavir, fosamprenavir*, efavirenz
![]() renal calculi – indinavir*, atazanavir
renal calculi – indinavir*, atazanavir
![]() lactic acidosis – zidovudine
lactic acidosis – zidovudine
![]() renal impairment – tenofovir
renal impairment – tenofovir
![]() anaemia – zidovudine
anaemia – zidovudine
![]() jaundice – atazanavir (unconjugated hyperbilirubinaemia)
jaundice – atazanavir (unconjugated hyperbilirubinaemia)
![]() immune reconstitution inflammatory syndrome – patients who commence antiretroviral therapy with a very low CD4 cell count may develop an exacerbation of symptoms and signs of a recently diagnosed opportunistic infection or a previously unrecognized infection may be ‘unmasked’; this occurs with mycobacterial infections (notably tuberculosis) and a range of other infections.
immune reconstitution inflammatory syndrome – patients who commence antiretroviral therapy with a very low CD4 cell count may develop an exacerbation of symptoms and signs of a recently diagnosed opportunistic infection or a previously unrecognized infection may be ‘unmasked’; this occurs with mycobacterial infections (notably tuberculosis) and a range of other infections.
Table 9.7.4
Side effects of antiretroviral agents*

*Drugs that are licensed in Australia but are no longer commonly used have been omitted.
Other presentations
![]() Cutaneous manifestations: Kaposi’s sarcoma, infections (e.g. secondary syphilis, zoster, warts, molluscum contagiosum, and crusted scabies), eosinophilic folliculitis, drug rashes
Cutaneous manifestations: Kaposi’s sarcoma, infections (e.g. secondary syphilis, zoster, warts, molluscum contagiosum, and crusted scabies), eosinophilic folliculitis, drug rashes
![]() Abdominal pain: pancreatitis due to antiretroviral therapy, HIV cholangiopathy, intra-abdominal lymphadenopathy secondary to MAC or lymphoma, lactic acidosis and hepatic steatosis associated with antiretroviral therapy
Abdominal pain: pancreatitis due to antiretroviral therapy, HIV cholangiopathy, intra-abdominal lymphadenopathy secondary to MAC or lymphoma, lactic acidosis and hepatic steatosis associated with antiretroviral therapy
![]() Neuropsychiatric manifestations: depression, mania, cognitive decline
Neuropsychiatric manifestations: depression, mania, cognitive decline
![]() Visual complaints: CMV retinitis (when CD4 cell count<0.05×109/L), syphilitic uveitis or chorioretinitis, rarely toxoplasma or cryptococcal chorioretinitis.
Visual complaints: CMV retinitis (when CD4 cell count<0.05×109/L), syphilitic uveitis or chorioretinitis, rarely toxoplasma or cryptococcal chorioretinitis.
Clinically silent HIV infection with risk factors
Sexually transmitted infections
Perianal or rectal sexually transmitted infections (STIs) in men are obvious markers of HIV infection risk and should prompt testing for infection. However, STIs often ‘hunt in packs’, so any patient diagnosed with gonorrhoea, Chlamydia, syphilis, genital warts, genital herpes or another STI should also be investigated for HIV.
Other risk groups
Other patients who present with a problem unrelated to HIV but with whom the desirability of HIV testing should be discussed include those with the following risk factors:
![]() unprotected male-to-male intercourse
unprotected male-to-male intercourse
![]() sharing of injecting equipment
sharing of injecting equipment
![]() being from a country with a high HIV prevalence
being from a country with a high HIV prevalence
![]() being the sexual partner of either an HIV-positive person or a person at risk of HIV.
being the sexual partner of either an HIV-positive person or a person at risk of HIV.
Investigations
Requesting an HIV test
In Australia, doctors who request an HIV test are obliged to provide patients with information about the medical, psychological and social consequences of a positive or negative HIV test and to provide the result to the patient in person. More detailed information about HIV testing can be found in guidelines issued by Australian authorities [8].
Unfortunately, practical issues mean that the emergency department is usually not an ideal setting for HIV testing. First, discussion about sensitive personal information, such as sexual history (especially if it has to be obtained via an interpreter) is difficult in an open-design, overcrowded and noisy ED. Second, many EDs do not have a mechanism for follow up of patients for provision of test results. For these reasons, a more appropriate arrangement may be to refer patients who are discharged from the ED to their local doctor or local sexual health clinic for testing, while testing of patients admitted to hospital can be the responsibility of the admitting unit.
Widespread ‘opt out’ HIV testing of hospital patients is recommended in the USA but has not been adopted in Australia. However, if the ‘treatment as prevention’ approach is to be effective, increasing rates of testing among groups at risk of HIV will be necessary in order to reduce the number of undiagnosed HIV infections and increase HIV treatment uptake. Rapid point-of-care HIV tests, performed by appropriately trained people (but not patients), are now licensed in Australia and do not require a follow-up visit but whether these tests will have a role in settings such as emergency departments is currently unclear.
Primary HIV infection
![]() Full blood examination, heterophile antibody test
Full blood examination, heterophile antibody test
![]() HIV antibody/p24 antigen enzyme immunoassay (EIA) test: may be negative initially, in which case it is vital to repeat the test in 2, 4 and 6 weeks. A positive EIA is confirmed with a positive Western blot test
HIV antibody/p24 antigen enzyme immunoassay (EIA) test: may be negative initially, in which case it is vital to repeat the test in 2, 4 and 6 weeks. A positive EIA is confirmed with a positive Western blot test
![]() HIV RNA (viral load) test: not generally recommended for diagnosis of primary HIV infection because false positives may occur (although with a low viral load, whereas true positives usually have a very high viral load).
HIV RNA (viral load) test: not generally recommended for diagnosis of primary HIV infection because false positives may occur (although with a low viral load, whereas true positives usually have a very high viral load).
Previously unrecognized HIV infection (not including primary HIV infection)
The HIV antibody/antigen EIA test will be positive in all patients and other tests are not needed for diagnosis.
Previously diagnosed HIV infection – disease syndromes
Cough, shortness of breath, fever
If the CD4 cell count is>0.2×109/L, most patients can be managed as if they did not have HIV infection. Investigations required for patients with suspected bacterial pneumonia or tuberculosis include a chest X-ray, full blood examination, sputum examination and blood cultures.
If the CD4 cell count is<0.2×109/L, investigation is almost always indicated, the extent of which will be guided by the patient’s condition and the likely diagnostic possibilities and may include some or all of the following:
![]() oxygen saturation
oxygen saturation
![]() chest X-ray
chest X-ray
![]() blood cultures
blood cultures
![]() sputum Gram stain, culture and acid-fast bacillus (AFB) smear and culture
sputum Gram stain, culture and acid-fast bacillus (AFB) smear and culture
![]() induced sputum for detection (by microscopy or polymerase chain reaction [PCR]) of PCP
induced sputum for detection (by microscopy or polymerase chain reaction [PCR]) of PCP
![]() bronchoscopy – usually during inpatient admission.
bronchoscopy – usually during inpatient admission.
A high index of suspicion for tuberculosis must be maintained; the diagnosis is generally suggested by one or more suggestive epidemiological, clinical or radiological features.
Focal neurological signs, convulsions or altered conscious state
A brain CT scan (with contrast) should be done in all patients, often as a matter of some urgency, and should always precede a lumbar puncture. MRI will often provide additional important information. The commonest causes of focal lesions are cerebral toxoplasmosis and primary intracerebral lymphoma. A Toxoplasma gondii IgG test will have usually been performed in those with previously diagnosed HIV infection: if positive, this indicates prior infection and a predisposition to the development of cerebral toxoplasmosis; if negative, toxoplasmosis is much less likely. Diagnosis of cerebral lymphoma is primarily based on non-response to empiric treatment for cerebral toxoplasmosis; CSF cytology, detection of EBV DNA in CSF by PCR or, occasionally, brain biopsy are required for a specific diagnosis. Progressive multifocal leucoencephalopathy manifests as focal white matter lesions visible on T2-weighted MRI scans.
Diarrhoea, with or without abdominal pain and fever
Faecal examination (preferably two to three fresh specimens collected on different days) for:
![]() microscopy for ova, cysts and parasites
microscopy for ova, cysts and parasites
![]() Cryptosporidium antigen test or stain, microsporidium stain
Cryptosporidium antigen test or stain, microsporidium stain
![]() culture for Salmonella, Campylobacter and Shigella
culture for Salmonella, Campylobacter and Shigella
![]() Clostridium difficile culture and toxin, especially if recent antibiotic therapy.
Clostridium difficile culture and toxin, especially if recent antibiotic therapy.
Selected patients with undiagnosed diarrhoea may require colonoscopy or upper GI endoscopy if infections such as CMV, MAC or microsporidiosis are suspected. Swabs for gonorrhoea, Chlamydia and herpes should be taken from patients with symptoms of proctitis.
Fever without localizing features
If the CD4 cell count is>0.2×109/L, serious HIV-related causes are uncommon and so investigation will be guided by clinical features, severity of illness and so on. If the CD4 cell count is<0.2×109/L, most patients will need investigation, beginning with the following basic work-up:
![]() blood cultures, including mycobacterial blood cultures if CD4 cell count<0.05×109/L
blood cultures, including mycobacterial blood cultures if CD4 cell count<0.05×109/L
![]() chest X-ray
chest X-ray
![]() serum cryptococcal antigen.
serum cryptococcal antigen.
Additional tests for selected patients include faecal examination, sputum examination, abdominal ultrasonography or CT scanning and, occasionally, bone marrow or liver biopsy.
Difficult or painful swallowing
Oesophagoscopy and biopsy are reserved for those who fail an empirical course of antifungal therapy (see below).
Headache, fever, neck stiffness
The serum cryptococcal antigen test is a useful screening test for cryptococcal meningitis because a negative result effectively excludes the diagnosis. A lumbar puncture should only be performed after a CT brain scan and if the CT does not show a space-occupying lesion or evidence of increased intracranial pressure. CSF should be routinely sent for the following:
![]() protein and glucose
protein and glucose
![]() Gram stain and culture (and AFB smear and culture if tuberculosis is suspected)
Gram stain and culture (and AFB smear and culture if tuberculosis is suspected)
![]() India ink stain and cryptococcal antigen
India ink stain and cryptococcal antigen
![]() cytology
cytology
![]() VDRL or RPR test – only indicated if serum syphilis serology is positive.
VDRL or RPR test – only indicated if serum syphilis serology is positive.
Management
Primary HIV infection
![]() Symptomatic treatment.
Symptomatic treatment.
![]() Specific antiretroviral therapy – role not determined.
Specific antiretroviral therapy – role not determined.
Specific HIV syndromes
ED physicians should consult doctors experienced in treating HIV-infected patients for advice about the management of specific syndromes and opportunistic infections. The following guidelines focus on initial and empiric therapy and provide examples of treatment options, but detailed information about indications for specific agents, toxicity and so on is omitted. For more comprehensive treatment recommendations, a specialized text should be consulted [9,10].
Cough, fever, shortness of breath
Any person with suspected pulmonary tuberculosis must be placed in respiratory isolation until the diagnosis is excluded. Otherwise, on the basis of the initial diagnostic evaluation, patients can be categorized and management proceed as follows:
![]() significant infection unlikely: no treatment
significant infection unlikely: no treatment
![]() possible PCP: empirical PCP therapy with co-trimoxazole, and corticosteroids if PaO2 on room air<60 mmHg
possible PCP: empirical PCP therapy with co-trimoxazole, and corticosteroids if PaO2 on room air<60 mmHg
![]() possible bacterial pneumonia:
possible bacterial pneumonia:
![]() non-severe, outpatient – oral amoxicillin with or without either macrolide (e.g. clarithromycin) or doxycycline
non-severe, outpatient – oral amoxicillin with or without either macrolide (e.g. clarithromycin) or doxycycline
![]() non-severe, inpatient – IV penicillin plus either oral macrolide (e.g. roxithromycin) or doxycycline
non-severe, inpatient – IV penicillin plus either oral macrolide (e.g. roxithromycin) or doxycycline
![]() severe – IV ceftriaxone plus IV azithromycin
severe – IV ceftriaxone plus IV azithromycin
![]() possible tuberculosis: admission, and respiratory isolation; treatment with isoniazid, rifampicin, pyrazinamide and ethambutol if diagnosis confirmed; empirical therapy is sometimes necessary depending on clinical circumstances (e.g. suspected coexisting tuberculous meningitis).
possible tuberculosis: admission, and respiratory isolation; treatment with isoniazid, rifampicin, pyrazinamide and ethambutol if diagnosis confirmed; empirical therapy is sometimes necessary depending on clinical circumstances (e.g. suspected coexisting tuberculous meningitis).
Focal neurological signs, convulsions, altered conscious state
Treatment is guided by the results of the brain CT scan. If a space-occupying lesion is found patients are treated empirically for cerebral toxoplasmosis with sulphadiazine and pyrimethamine. The CT scan is repeated after 2–3 weeks and, if no response is evident, a brain biopsy might be considered in selected patients to diagnose cerebral lymphoma. If the CT scan is normal or non-diagnostic, MRI scanning is usually indicated, and supplemented by lumbar puncture.
Diarrhoea, with or without abdominal pain and fever
Any infection identified on initial faecal examinations is treated on its merits. Symptomatic treatment with an antimotility agent, such as loperamide, is contraindicated if bloody diarrhoea and fever are present, but otherwise can be given safely to most patients. Endoscopy is generally reserved for those in whom no specific cause is identified on initial evaluation and whose diarrhoea persists despite antimotility therapy.
Fever without localizing features
Empirical antibacterial therapy (with an antipseudomonal agent, such as piperacillin/tazobactam, with or without an aminoglycoside or vancomycin) is indicated for patients with an absolute neutrophil count<0.5×109/L; otherwise the need for specific treatment is guided by the condition of the patient and the results of the diagnostic work-up. Any long-term IV access device should be removed if infection of the device is confirmed on clinical or microbiological grounds or if diagnostic evaluation reveals no other focus of infection. Treatment for disseminated MAC (with clarithromycin and ethambutol, with or without rifabutin) is generally given only after the organism has been isolated, although occasional patients with debilitating fevers, weight loss and no other diagnosis may be treated empirically.
Difficult or painful swallowing
Empirical antifungal therapy is started with an azole agent, usually oral fluconazole. Patients with resistant Candida infections need treatment with an alternative azole agent, such as posaconazole, or a short course of IV amphotericin B. Patients undergo endoscopy if they do not respond to antifungal treatment and the results of histology and cultures determine subsequent treatment.
Headache, fever, neck stiffness
Patients with confirmed cryptococcal meningitis are treated with a combination of IV amphotericin B and oral 5-fluorocytosine for 2 weeks, then remain on suppressive therapy with oral fluconazole. If tuberculous meningitis is suspected empirical therapy should be started immediately, pending the results of CSF cultures.
Specific treatment of other infections
![]() CMV infections: IV ganciclovir or IV foscarnet
CMV infections: IV ganciclovir or IV foscarnet
![]() Salmonella infections (non-enteric fever): ciprofloxacin
Salmonella infections (non-enteric fever): ciprofloxacin
Antiretrovirals in the management of HIV infection [11]
Combination antiretroviral therapy has transformed the lives of people living with HIV infection, by improving their quality of life and by reducing the incidence of HIV-related complications and deaths by 80% or more. More than 90% of patients starting treatment with one of the current recommended antiretroviral regimens will achieve a non-detectable plasma HIV viral load and a substantial CD4 cell count increase and, in the great majority of patients, these benefits are sustained in the long term. Modern antiretroviral regimens are much more convenient, less toxic and more potent than earlier combination antiretroviral therapy, but antiretroviral therapy is not without its costs: difficulty in maintaining life-long adherence, short- and long-term toxicities of antiretroviral agents and the potential development of antiretroviral resistance.
Effective treatment is also available for patients failing therapy because of drug resistance or intolerance, using antiretroviral agents that belong to the same classes of drugs used for initial therapy or that have novel mechanisms of action. Examples include ‘new-generation’ protease inhibitors (tipranavir and darunavir) and non-nucloeside reverse transcriptase inhibitors (etravirine), inhibitors of CCR5 (maraviroc), a host chemokine receptor involved in HIV cell entry and the fusion inhibitor enfuvirtide [12].
The emergency physician does not require a detailed knowledge of antiretroviral therapy, but should be aware of the agents in current use, their side effects and the potential for clinically significant drug–drug interactions. More detailed information can be referenced in regularly updated antiretroviral guidelines; examples are those produced by a panel of the US Department of Health and Human Services with an added Australian commentary, accessible at http://www.ashm.org.au/aust-guidelines/ and British HIV treatment guidelines, accessible at http://www.bhiva.org/.
Indications
![]() Symptomatic HIV infection
Symptomatic HIV infection
![]() Asymptomatic HIV infection – CD4 cell count<0.500×109/L
Asymptomatic HIV infection – CD4 cell count<0.500×109/L
![]() Asymptomatic HIV infection – CD4 cell count>0.500×109/L
Asymptomatic HIV infection – CD4 cell count>0.500×109/L
![]() pregnant women with HIV infection [13]
pregnant women with HIV infection [13]
![]() patients with chronic hepatitis B that requires treatment
patients with chronic hepatitis B that requires treatment
![]() patients with HIV-associated nephropathy
patients with HIV-associated nephropathy
![]() Treatment of the infected individual in a serodiscordant relationship, to prevent transmission to the uninfected partner [14]
Treatment of the infected individual in a serodiscordant relationship, to prevent transmission to the uninfected partner [14]
![]() After significant HIV exposure sustained by a healthcare worker (see Chapter 9.10) or following likely or definite exposure to HIV sexually.
After significant HIV exposure sustained by a healthcare worker (see Chapter 9.10) or following likely or definite exposure to HIV sexually.
Classes of drug†
![]() Nucleoside/nucleotide reverse transcriptase inhibitors (NRTIs): tenofovir, emtricitabine (FTC), abacavir, lamivudine (3TC), zidovudine (ZDV or AZT)
Nucleoside/nucleotide reverse transcriptase inhibitors (NRTIs): tenofovir, emtricitabine (FTC), abacavir, lamivudine (3TC), zidovudine (ZDV or AZT)
![]() Non-nucleoside reverse transcriptase inhibitors (NNRTIs): nevirapine, efavirenz, etravirine, rilpivirine
Non-nucleoside reverse transcriptase inhibitors (NNRTIs): nevirapine, efavirenz, etravirine, rilpivirine
![]() Protease inhibitors (Pls): atazanavir, lopinavir, fosamprenavir, tipranavir, darunavir (all co-administered with low-dose ritonavir)
Protease inhibitors (Pls): atazanavir, lopinavir, fosamprenavir, tipranavir, darunavir (all co-administered with low-dose ritonavir)
![]() Fusion inhibitors: enfuvirtide
Fusion inhibitors: enfuvirtide
![]() Entry (chemokine receptor 5 [CCR5]) inhibitors: maraviroc
Entry (chemokine receptor 5 [CCR5]) inhibitors: maraviroc
![]() Integrase inhibitors: raltegravir
Integrase inhibitors: raltegravir
Initial regimens – at least three drugs
![]() Two NRTIs plus one NNRTI: examples are tenofovir+FTC+efavirenz (Atripla) OR abacavir+3TC (Kivexa) plus efavirenz OR tenofovir+FTC+rilpivirine (Eviplera)
Two NRTIs plus one NNRTI: examples are tenofovir+FTC+efavirenz (Atripla) OR abacavir+3TC (Kivexa) plus efavirenz OR tenofovir+FTC+rilpivirine (Eviplera)
![]() Two NRTIs (as listed above) plus one PI (atazanavir) boosted with low-dose ritonavir
Two NRTIs (as listed above) plus one PI (atazanavir) boosted with low-dose ritonavir
![]() Two NRTIs (as listed above) plus raltegravir.
Two NRTIs (as listed above) plus raltegravir.
Side effects [15] (see Table 9.7.4)
If an antiretroviral drug is suspected or known to be the cause of a serious side effect, the patient’s treating HIV doctor or a hospital HIV doctor should be consulted. In the interim, or unless advised otherwise by the treating or hospital doctor, all antiretroviral medications, and not just the incriminating drug, should be withheld to reduce the risk of development of resistance on a less than fully suppressive therapy.
Drug–drug interactions
Some commonly used drugs metabolized by or that induce hepatic cytochrome P450 oxidases are contraindicated with certain Pls or NNRTIs; in addition, many other drugs will require dose modification or closer monitoring. Always check before prescribing any new drug to a patient on antiretroviral therapy; a very useful website (from the University of Liverpool, UK) is www.hiv-druginteractions.org.
Disposition
Patients with newly diagnosed HIV infection should be referred to a specialized HIV clinic or to a doctor with expertise in HIV medicine.
HIV medicine is a complex and rapidly changing field. For this reason, the management of patients with known HIV infection presenting to the ED should always involve consultation with a hospital doctor knowledgeable about HIV infection, such as an infectious diseases physician or immunologist. The patient’s usual HIV doctor (a hospital specialist, sexual health physician or general practitioner with a high HIV caseload) can be contacted to obtain important details, such as recent CD4 cell count and current antiretroviral agents, in the event that such information is not otherwise immediately available. In general, patients with a suspected or confirmed serious opportunistic infection will need to be admitted for investigation and management. Patients in the final stages of AIDS (fortunately an uncommon group nowadays in wealthy countries like Australia) or those with less serious complications, can often be managed in the community, in which case liaison with the local doctor, home-care nurses or community-care agencies is vital.
Prognosis
Prior to the widespread use of opportunistic infection prophylaxis and effective antiretroviral therapy, 50% of patients developed AIDS 10 years after becoming HIV infected and 75% of patients after 13 years. Following an AIDS-defining illness, the median survival was 12–24 months. Long-term non-progressors, who have a normal CD4 cell count and no HIV-related complications without antiretroviral therapy after 10 or more years of HIV infection, comprise less than 5% of patient cohorts.
Most AIDS-defining infections, such as PCP, now have low mortality and high 1-year survival rates if the infection is treated appropriately and patients are started promptly on combination antiretroviral therapy, but survival rates following diagnosis of disseminated MAC and CMV end-organ disease are lower because these two opportunistic infections usually occur at a very advanced stage of HIV infection. Combination antiretroviral therapy has reduced the mortality and incidence of opportunistic infections by over 80%.
Data from several large cohort studies indicate that average life expectancy in developed countries for patients on long-term antiretroviral therapy is close to but still lower than that of the HIV-uninfected population [16]. This difference is partly accounted for by an excess of deaths due to chronic conditions not typically associated with HIV infection, such as cardiovascular disease, non-HIV associated malignancy and renal disease (discussed earlier in the chapter); classic HIV complications, such as opportunistic infections or HIV-related malignancies, still occur but do not contribute substantially to this difference.
Prevention
Prevention of HIV transmission
![]() Public health and educational efforts to encourage the adoption of safer sex practices
Public health and educational efforts to encourage the adoption of safer sex practices
![]() HIV screening of blood, blood products and tissue donors
HIV screening of blood, blood products and tissue donors
![]() Non-sharing and use of clean needles and syringes by injecting drug users
Non-sharing and use of clean needles and syringes by injecting drug users
![]() Observance of standard precautions by workers in healthcare settings
Observance of standard precautions by workers in healthcare settings
![]() Use of antiretroviral therapy and avoidance of breastfeeding to prevent transmission from an HIV-infected mother to her baby
Use of antiretroviral therapy and avoidance of breastfeeding to prevent transmission from an HIV-infected mother to her baby
![]() Use of antiretroviral therapy to prevent transmission from an infected individual to an uninfected partner [14]
Use of antiretroviral therapy to prevent transmission from an infected individual to an uninfected partner [14]
![]() Use of antiretroviral prophylaxis after significant occupational exposure (see Chapter 9.10) or sexual exposure to HIV (post-exposure prophylaxis)
Use of antiretroviral prophylaxis after significant occupational exposure (see Chapter 9.10) or sexual exposure to HIV (post-exposure prophylaxis)
![]() Use of antiretroviral prophylaxis by an uninfected individual before HIV exposure (pre-exposure prophylaxis) [17]
Use of antiretroviral prophylaxis by an uninfected individual before HIV exposure (pre-exposure prophylaxis) [17]
![]() Male circumcision – shown to reduce the acquisition of HIV infection by 60% in studies in sub-Saharan Africa [18].
Male circumcision – shown to reduce the acquisition of HIV infection by 60% in studies in sub-Saharan Africa [18].
Prevention of HIV-related complications [19]
|
Infection |
Preventive measure |
|
Pneumococcal pneumonia |
Pneumococcal vaccination |
|
Latent tuberculous infection |
Isoniazid |
|
PCP |
Co-trimoxazole |
|
Toxoplasmosis |
Co-trimoxazole |
|
MAC |
Azithromycin or rifabutin |
Controversies
![]() With the availability of more potent, more convenient and better-tolerated antiretroviral drugs, should all patients and not just those below a certain CD4 cell count threshold be treated?
With the availability of more potent, more convenient and better-tolerated antiretroviral drugs, should all patients and not just those below a certain CD4 cell count threshold be treated?
![]() Will the ‘treatment as prevention’ approach (broadening treatment indications for people already known to be HIV-infected and increasing testing rates in at-risk groups in order to diagnose and then treat people with unrecognized HIV) lead to a reduction in HIV transmission at the population level?
Will the ‘treatment as prevention’ approach (broadening treatment indications for people already known to be HIV-infected and increasing testing rates in at-risk groups in order to diagnose and then treat people with unrecognized HIV) lead to a reduction in HIV transmission at the population level?
![]() What is the role of other prevention measures that are known to be effective, such as pre-exposure prophylaxis and male circumcision?
What is the role of other prevention measures that are known to be effective, such as pre-exposure prophylaxis and male circumcision?
![]() What is the relative contribution of HIV infection, antiretroviral therapy and standard risk factors to the risk of developing chronic medical conditions that are responsible for most of the morbidity and mortality in patients on effective antiretroviral therapy, and what is the nature of the association?
What is the relative contribution of HIV infection, antiretroviral therapy and standard risk factors to the risk of developing chronic medical conditions that are responsible for most of the morbidity and mortality in patients on effective antiretroviral therapy, and what is the nature of the association?
![]() Can an effective HIV vaccine be developed?
Can an effective HIV vaccine be developed?
![]() Can HIV ever be eradicated?
Can HIV ever be eradicated?
References
1. UNAIDS. Global report: UNAIDS report on the global AIDS epidemic 2012. Geneva: WHO, 2012.
2. The Kirby Institute. HIV, viral hepatitis and sexually transmissible infections in Australia Annual Surveillance Report 2012. The University of New South Wales, Sydney: The Kirby Institute, 2012.
3. Cameron PU, Kelly M. HIV immunopathology. In: Hoy J, Lewin S, Post JJ, Street A, eds. HIV management in Australasia: a guide for clinical care. Darlinghurst: Australasian Society for HIV Medicine; 2009;19–37.
4. Phillips AN, Neaton J, Lundgren J. The role of HIV in serious diseases other than AIDS. AIDS. 2008;22:2409–2418.
5. Stewart G, ed. Could it be HIV?. Sydney: Australasian Medical Publishing Company Ltd; 1994.
6. Giles M, Workman C. Clinical manifestations and the natural history of HIV. In: Hoy J, Lewin S, Post JJ, Street A, eds. HIV management in Australasia: a guide for clinical care. Darlinghurst: Australasian Society for HIV Medicine; 2009;125–133.
7. Post JJ, Kelly C, Clezy M, et al. Key opportunistic infections. In: Hoy J, Lewin S, Post JJ, Street A, eds. HIV management in Australasia: a guide for clinical care. Darlinghurst: Australasian Society for HIV Medicine; 2009;133–166.
8. National HIV Testing Policy Expert Reference Committee 2011. National HIV testing policy 2011. Commonwealth of Australia, Canberra.<http://testingportal.ashm.org.au/hiv>[Accessed Feb. 2013].
9. Hoy J, Lewin S, Post JJ, Street A, eds. HIV management in Australia: a guide for clinical care. Darlinghurst: Australasian Society for HIV Medicine; 2009.
10. Crowe S, Hoy J, Mills J, eds. Medical management of the HIV-infected patient. 2nd ed. Cambridge: Martin Dunitz; 2001.
11. Pett S, Pierce A. Antiretroviral therapy. In: Hoy J, Lewin S, Post JJ, Street A, eds. HIV management in Australasia: a guide for clinical care. Darlinghurst: Australasian Society for HIV Medicine; 2009;59–73.
12. Hirschel B, Perneger T. No patient left behind–better treatments for resistant HIV infection. Lancet. 2007;370:3–5.
13. Panel on Treatment of HIV-Infected Pregnant Women and Prevention of Perinatal Transmission. Recommendations for use of antiretroviral drugs in pregnant HIV-1-infected women for maternal health and interventions to reduce perinatal HIV transmission in the United States (last updated 31 July 2012).<http://aidsinfo.nih.gov/contentfiles/lvguidelines/PerinatalGL.pdf>[Accessed Feb. 2013].
14. Cohen MS, Chen YQ, McCauley M, et al. Prevention of HIV-1 infection with early antiretroviral therapy. N Engl J Med. 2011;365:493–505.
15. Carr A, Vujovic O. Approaches to the management of antiretroviral therapy toxicity. In: Hoy J, Lewin S, Post JJ, Street A, eds. HIV management in Australasia: a guide for clinical care. Darlinghurst: Australasian Society for HIV Medicine; 2009;103–113.
16. The ART Cohort Collaboration. Life expectancy of individuals on combination antiretroviral therapy in high-income countries: a collaborative analysis of 14 cohort studies. Lancet. 2008;372:293–299.
17. Grant TM, Lama JR, Anderson PL, et al. Preexposure chemoprophylaxis for HIV prevention in men who have sex with men. N Engl J Med. 2010;363:2587–2599.
18. Gray RH, Kigozi G, Serwadda D, et al. Male circumcision for HIV prevention in men in Rakai, Uganda: a randomised trial. Lancet. 2007;369:657–666.
19. Panel on Opportunistic Infections in HIV-Infected Adults and Adolescents. Guidelines for the prevention and treatment of opportunistic infections in HIV-infected adults and adolescents: recommendations from the Centers for Disease Control and Prevention, the National Institutes of Health, and the HIV Medicine Association of the Infectious Diseases Society of America. Available at http://aidsinfo.nih.gov/contentfiles/lvguidelines/adult_oi.pdf. Accessed (25/8/13).
9.8 Sexually transmitted infections
Jane Terris
Essentials
1 Sexually transmitted infections (STIs) are among the commonest infections worldwide and affect around 340 million new patients per year.
2 STIs account for a significant number of emergency department (ED) visits per year and, despite public health efforts, are increasing each year in Australia.
3 ED staff should be competent to screen, diagnose, treat, notify and improve future sexual health through advice and referral.
4 Emergency physicians should aim to provide effective, confidential, non-judgemental care and this may involve staff challenging their own prejudices around sexual behaviour in order to remain objective.
5 Patients may present at any stage of the STI and with multiple co-existent STIs. Presentation with genital ulcers or with signs of discharge, urethritis or cervicitis are the usual clinical manifestations.
6 STIs may be asymptomatic, missed or undertreated leading to adverse outcomes including chronic infection and infertility.
7 A detailed and specific sexual history should be normalized in the context of the general medical history and should be confidential.
8 The essentials of the sexual history can be summed up by the five Ps: Partners, Practices, Pregnancy, Protection, Past STIs.
9 Empirical treatment may need to be commenced in the ED. Screening test results are rarely available at presentation currently, although point of care testing is becoming more widespread.
10 Syndromic treatment, according to symptoms, is likely to be more successful in males, especially in high prevalence areas. Most cases of vaginal discharge in females are not STI related and many STIs in females are asymptomatic.
Introduction
Sexually transmitted infections (STIs) are among the commonest infections worldwide and continue to be epidemic in all societies. STIs are a major public health problem and are contracted by around 340 million patients per year according to World Health Organization figures [1]. STIs account for a significant number of emergency department (ED) visits per year and notification rates in Australia [2], in common with the UK [3] and the USA [4], currently show a yearly increase. The true incidence of STIs is difficult to ascertain in view of variations in regional reporting and likely under reporting.
EDs should aim to provide effective and confidential care in a sensitive and non-judgemental environment. This may be a challenge for a busy, noisy department with multiple simultaneous care priorities.
The ED management of STIs should include screening, diagnosis, treatment of patients and contacts, notifying confirmed cases to public health authorities and optimizing future sexual health through advice and referral.
Patients will continue to use ED to access general and sexual healthcare needs, therefore, emergency physicians (EPs) must have a sound working knowledge of STI management. This may be the only opportunity to intervene.
Patients may present at any stage of the STI and with multiple coexistent STIs. A detailed and specific sexual history should be normalized within the context of the general medical history.
STIs may be asymptomatic, missed or undertreated, resulting in adverse outcomes including ectopic pregnancy, chronic infection and infertility.
EPs should maintain a high index of suspicion, avoid stereotyping patients and be prepared to treat empirically, especially in males in areas of high prevalence.
Patients should be referred for follow up including HIV and hepatitis screening.
ED patients have high rates of asymptomatic STIs [5] and over half of females presenting with chlamydia or gonorrhoea are discharged without adequate treatment [6]. Conversely, most females presenting with vaginal discharge do not have STIs and are more likely to have candidiasis or bacterial vaginosis. Debates around whether to screen for STIs and HIV in EDs versus specialist clinics continue. The cost of screening, especially if adequate and accessible specialized facilities exist nearby and the potential for increasing antibiotic resistance with empirical treatment must be weighed against the consequences of missed infections and the significant public health, social and economic burden of undiagnosed and untreated STIs. Alternative facilities may not be accessible at the time of presentation. There is, therefore, an important opportunity for the EP to intervene and capture these patients at presentation.
Epidemiology
Sexually transmitted infections are increasing overall in Australia, the UK and the USA, although likely under reporting means that true figures are unknown. There are several well-documented high-risk groups for STIs and some newly emerging patient groups. Those known to be high risk include young people in the 15–24 age group, patients who exchange sex for money or drugs, also known as transactional sex, pregnant women and men who have sex with men (MSM). Recently reported at-risk groups include users of erectile dysfunction medication [7] and widowers. A high index of suspicion should be maintained in view of the fact that STIs can affect any sexually active person and stereotyping should be avoided.
The commonest STI in Australia, the UK and the USA is chlamydia, with an increase in Australia from 103 to 357 cases per 100 000 population between 2001 and 2011 [2]. The next commonest is gonorrhoea with an increase of 53 cases from 31 between 2001 and 2011 [2].
Syphilis is currently increasing overall in Australia, as in the UK [3] and the USA [4] with 659 reported cases in Australia in 2005 to 1285 in 2011 [2]. No cases of chancroid or donovanosis have been reported in Australia for over 10 years [2], although there are still some endemic regions including Papua New Guinea and parts of India and Africa, with reported cases mainly in tropical and subtropical regions.
Prevention
Public health campaigns have been active around STI prevention, although rates of infection continue to increase. It is not clear whether this is linked to increased awareness and reporting. Advice should include discussion around high-risk behaviour and safe sex practices. This should include effective barrier contraception in the form of latex condoms, rather than the less protective non-latex alternatives, the need to abstain from sex until STI treatment is complete and the need for regular STI screening, especially in high-risk groups.
History
Effective communication is especially important within the context of a sexual history and patients are often embarrassed and anxious. Wherever possible a private, clean, comfortable cubicle with a door should be used for taking the sexual history. Attention should be paid to the initial greeting, appropriate body language, eye contact, which varies culturally, and non-verbal cues from the patient. Be prepared that this may take a little longer than the focused history often used in EM. It may be necessary for the EP to reflect on their own personal attitudes to sexual behaviour in order to normalize the sexual history within the overall medical history and make an objective assessment. Communication needs of specific patient groups in ED with regard to language, hearing difficulty and cultural context should be addressed. Resources may include interpreter services, websites and local support organizations.
Efforts should be made to reinforce the confidential nature of the interview in order to encourage candour. It may help to display local STI clinic posters and literature within the ED. Students and observers will need the consent of the patient to be present and this is not always given due to the sensitive nature of the interview and the need for confidentiality. A detailed and specific history is important to identify those patients at risk for sexually transmitted infections, including HIV, and to ascertain which anatomical sites to focus on for screening. The history may start with open statements and questions, such as telling the patient it is important to ask questions around their sexual behaviour, and progress to closed questions around specifics of the five Ps of the history, below. It may be necessary to explain clearly the need to ask certain questions in order to avoid offence. For example, explaining that asking about the gender of a partner is needed to identify MSM in order to offer rectal, pharyngeal and hepatitis screening tests and that questions around partners are necessary to allow contact tracing and follow up. The history often opens with the presenting local symptoms, including discharge or genital ulcers. Further questions should ask about the characteristics of any discharge, abdominal and pelvic pain, dyspareunia, dysuria, joint and eye symptoms and skin rashes. The history should look for risk factors for STI in general, for specific features of STIs and should also screen for complicated, disseminated or recurrent infection. A previous history of STI, or partner infection and treatment, and the possibility of pregnancy should be explored. It is important to adapt the questioning style within the cultural context as many patients will be unfamiliar and uncomfortable with discussing details of their sexual practices.
The sexual history can be summarized in terms of the five Ps: Partners, Practices, Pregnancy, Protection and Past STIs (Table 9.8.1). The skill of taking a focused sexual history including the points below, in the time available in ED, takes practice.
Table 9.8.1
Essentials of the sexual history
|
The five Ps of the sexual history |
Essential points to cover |
|
Partners |
Last 3 months who, how many, where from, risk factors in partners? |
|
Practices |
Is sexual contact vaginal, oral, anal and with whom? Are condoms used sometimes, always, never? |
|
Protection |
How is risk reduced, e.g. monogamy, condoms? |
|
Pregnancy |
Plans around becoming or preventing pregnancy and details of contraception used |
|
Past STIs |
In patient and partners – what infections, when and how were they treated, how were they followed up? Screening since? |
Partners
Ask how many in the past year and how many in the past 3 months, what gender, current length of relationship, risk factors of partners, for example IV drug use, and other partners outside the relationship.
Practices
Number and genders of recent partners within the past 3 months, whether condoms are used always, sometimes, never and, if not, in which situations condoms are not used, whether sexual acts are vaginal, anal insertive or anal receptive, oral and with whom. A history of recent travel may identify infections in areas where specific pathogens or antibiotic resistance are known.
Pregnancy
Assess for current risk of pregnancy, which may affect treatment options and follow up, whether contraception is used or not and what type, in addition to any pregnancy-related symptoms, including last menstrual period. All females of childbearing age should have a pregnancy test. The need for emergency contraception should be assessed and a cervical cytology history included.
Protection
It is useful to ask what the patient does to protect themselves from STIs and HIV, including monogamy, condoms, safer sexual practices and also to gauge the patient’s perception of their own and partners’ risks.
Past STIs
Previous STIs may indicate higher risk behaviour and also repeat infection. Ask specifically whether the patient has ever had, and been treated for, gonorrhoea, chlamydia or any other STI, including HIV testing and results, and about hepatitis testing and results.
General principles of examination and screening
Following the full sexual history, a comprehensive STI check should be offered.
A chaperone should be available for all intimate examinations. Examination should be performed in a comfortable private cubicle with a door that closes and, preferably, with screens available for additional privacy. A good light source is essential and swabs and specimen pots should be readily available.
General physical examination including the mouth, pharynx, lymph nodes and skin should then be followed by genital examination, with palpation for inguinal lymph nodes, careful inspection of the genital and perianal areas for discharge, papules, ulcers, warts, lice or nits and signs of local trauma. Examination beneath the foreskin in the male is important and inspection of the urethral meatus for lesions and discharge. The scrotum, testes and epididymis should be examined for lesions and tenderness and the anorectal area examined, including a digital exam and proctoscopy in patients at risk of rectal disease or presenting with anorectal symptoms.
Examination of the vulva, Bartholin’s glands, vagina, cervix and perianal area is important in the female and should include bimanual pelvic examination to assess for tenderness and masses. A pregnancy test should be performed on all women of childbearing age.
It is important to confirm with the laboratory which specimen tubes and transport media are needed for which tests and how specimens should be stored, for example gonorrhoea swabs should be kept at room temperature. A ready made testing pack supplied by the laboratory is useful and will generally include swabs with charcoal transport medium for urethral and high vaginal smear and culture, glass slides for high vaginal or urethral smear, wire cottontip swabs with plastic shaft tube for chlamydia, gonorrhoea and herpes NAAT and clotted blood tubes for serological tests. Specimen collection is a specialist skill upon which the diagnosis rests and advice from laboratory staff prior to collection is invaluable. Swabs should be taken from the appropriate areas as detailed below, for microscopy, culture and sensitivity (M, C and S) and nucleic acid amplification testing NAAT for chlamydia, gonorrhoea, trichomonas and other organisms as indicated. This may include the genital area, anorectal area and pharynx.
Urine should be sent as first void specimen for M, C and S and NAAT testing for specific organisms and a midstream specimen for general M, C and S. Swab or lesion scraping should be sent if ulcers are present. Blood should be taken for syphilis, HIV and hepatitis serology.
Clinical features of specific infections
STIs may be asymptomatic or may present with constitutional or focal symptoms (Table 9.8.2). Focal symptoms are commonly those of urethritis or cervicitis or genital ulcers. Disseminated infection may present with skin rash, joint or eye symptoms. One or more STDs may coexist and, as the clinical features may be indistinguishable, empirical treatment, especially in males in areas of high prevalence, is indicated. Syndromic treatment, according to symptoms, has been shown to be neither sensitive nor specific in females presenting with vaginal discharge because most cases of vaginal discharge are not caused by STIs and many STIs in females are asymptomatic. In areas of high prevalence, a judgement should be taken on each individual case as to whether symptoms are likely to be STI related and whether treatment is indicated prior to results. Around one-third of cases of vaginal discharge presenting to ED remain undiagnosed pathologically [7], which may reflect inadequate specimen sampling or possibly other non-infective physiological causes.
Table 9.8.2
Clinical presentation of STIs and differential diagnosis

Infections presenting with discharge, urethritis and cervicitis
These symptoms may be caused by chlamydia, gonococcus, Trichomonas vaginalis, Ureaplasma urealyticum and Mycoplasma genitalium. Occasionally, genital herpes may cause discharge, although this is seldom the only symptom. Candida and bacterial vaginosis also present with discharge, although these are not classically sexually transmitted infections.
Chlamydia
Chlamydia is the most common STI in Australia and is increasing yearly in Australia, the UK and the USA. It is caused by the organism Chalamydia trachomatis and often coexists with gonorrhoea and other STIs. Chlamydia is often asymptomatic, especially in women [9]. This leads to underdiagnosis and treatment, which may result in long-term complications, including pelvic inflammatory disease and infertility. Screening in sexually active females under 25 and older females with new or multiple sexual partners is advised, although routine screening is not advised in sexually active males [10]. Symptoms, if present, are most commonly a mucopurulent cervical or vaginal discharge, intermenstrual bleeding and dysuria in females, or symptoms of urethritis, proctitis and epididymitis in males including urethral discharge, itching and sterile pyuria. Males may present with Reiter’s syndrome.
Diagnosis is made by Gram stain of urethral, vaginal or cervical discharge or first voided urine for NAAT.
Treatment for uncomplicated infection is with azithromycin 1 g oral as a single dose. If pelvic inflammatory disease is likely, ceftriaxone 500 mg IM once and metronidazole 400 mg oral bd for 14 days should be added.
Gonorrhoea
Gonococcal infections are increasing and are the second most common STI in Australia [2], the UK [3] and the USA [4]. They are caused by the Gram-negative intracellular diplococcus Neisseria gonorrhoeae which has an incubation period of 10–14 days. Infection in females is often asymptomatic and may coexist with chlamydia. Untreated gonococcal infection may lead to pelvic inflammatory disease and ectopic pregnancy in females and epididymitis and prostatitis in males. Symptomatic presentation is usually with purulent penile discharge in males and pelvic discomfort and mucopurulent cervicitis in females. Rectal infection is seen in up to 50% of females and in MSM. The latter may also have pharyngeal gonorrhoea which is often asymptomatic. Gonococcus in urban Australia is isolated more commonly in males in pharyngeal and rectal specimens and in genital isolates in both genders in rural areas [11]. Gonococcal infections may disseminate to cause constitutional symptoms of fever and malaise and focal signs of septic arthritis, tenosynovitis and a distinctive skin rash of pustular lesions on an erythematous base on the palms and fingers.
Diagnosis is made by Gram stain of swabs and NAAT testing of swabs and urine. Treatment of uncomplicated infection is with ceftriaxone 500 mg IM with 2 mL 1% lignocaine. Gonococcal antibiotic sensitivities change rapidly, with fluoroquinolone resistance widely documented and some regions, for example the Northern Territory of Australia and Southeast Asia, have penicillin-sensitive strains. Current local advice should always be sought.
Trichomoniasis
This infection occurs mainly in females and is caused by the protozoan Trichomonas vaginalis. The incubation period is up to 1 month. Infection may be asymptomatic or may present with genital irritation and vaginal discharge or urethritis in males. The discharge is rarely frothy and green coloured but, more commonly, clear and offensive. Other symptoms may include dysuria, dyspareunia and pelvic pain. Untreated trichomonas may cause pelvic inflammatory disease. Diagnosis is by visualization of the protozoa if a wet mount is readily available within 20 minutes, but more accurate diagnostic methods include high vaginal or urethral swabs for NAAT. Treatment is with metronidazole 2 g oral single dose or tinidazole 2 g oral if not pregnant. Pelvic inflammatory disease should be treated with combination antibiotics as described previously.
Urethritis or urethral discharge in males
This is usually caused by gonorrhoea or chlamydia, but may also be caused by Ureaplasma urealyticum, Mycoplasma genitalium, Trichomonas vaginalis or the herpesvirus. Diagnosis is made by the clinical history (although this will not indicate the causative organism) plus urethral swab and first void urine specimen for NAAT, culture and cell count. Midstream urine should be sent for general M, C and S as UTI, especially in older males, may be the cause. Treat empirically if STI is likely for gonorrhoea and chlamydia with ceftriaxone 500 mg IM once and azithromycin 1 g oral once.
Candida
Vaginal candidiasis may feature in the differential diagnosis of STIs causing irritation and discharge, but is generally not a sexually transmitted infection. It is caused by the fungus Candida albicans in most cases and characterized by an itchy white, curd-like discharge. Swabs for Gram stain or wet mount specimens may confirm the diagnosis by visible yeast and pseudohyphae. Treat with clotrimazole 500 mg single dose vaginal pessary or fluconazole 150 mg oral single dose.
Bacterial vaginosis
Bacterial vaginosis is not caused by any one specific organism and may occur in women who are not sexually active. It is associated with having multiple sexual partners and is a differential diagnosis of STI-related vaginal discharge. It is characterized by an overgrowth of normal vaginal flora by anaerobic bacteria and may be asymptomatic or present with discharge. Diagnosis is by Gram stain, combined with the characteristic thin, offensive discharge and clue cells on microscopy. Treat with metronidazole 400 mg bd oral for 7 days.
Infections presenting with genital ulcers
Genital ulceration may be caused by the herpes virus, syphilis and, rarely, chancroid, lymphogranuloma venereum (LGV) and lymphogranuloma inguinale (Donovanosis). These infections have higher rates of HIV co-transmission. Genital ulceration may rarely be caused by malignancy and referral should be made for any lesion that does not respond to treatment. There have been recent reports of an increase in vulval cancer in young indigenous Australian females [12].
Herpes simplex
Herpes simplex virus (HSV) infections are common worldwide, although most are asymptomatic. Genital ulceration is more commonly caused by HSV type 2 and occurs in up to one-quarter of patients who are seropositive for the virus. HSV type 1 generally causes oro-labial blisters, but may also cause genital ulceration identical to HSV-2. Subclinical infections may spread by viral shedding during sex. The primary outbreak of genital ulceration is usually accompanied by constitutional symptoms of fever, malaise, headache and painful bilateral regional lymphadenopathy which may precede the ulceration by 1 or 2 days. Prodromal tingling or paresthesia in the affected dermatomes may occur. The lesions are initially vesicular with an itchy, erythematous base and then become ulcerated before forming a scab. They may occur around the vulva, anus, thighs or buttocks in women and on or around the penis, perianal area, thighs and buttocks in males. They are usually very painful and adequate analgesia is important. Females may experience urinary retention due to the pain of voiding and sacral radiculopathy and may require admission for catheterization and intravenous antiviral treatment. The virus is shed for up to 2 weeks after the rash appears and lesions usually heal within 3 weeks. Recurrent episodes are generally less severe. Genital herpes is diagnosed by the characteristic clinical features and confirmation may be possible from viral swabs of lesion fluid, although treatment should be commenced empirically. Severe or disseminated infection should be treated by high-dose intravenous antivirals with a duration adequate to ensure lesion healing. The patient should be informed that treatment does not cure and there may be recurrent episodes. Future episodes can be attenuated if treatment is commenced at the onset of symptoms. Frequent recurrences (more than six per year) may indicate the need for prophylactic antiviral therapy. Diagnosis is clinical and confirmation is by dry swab of the ulcer base or blister fluid for NAAT. Treatment is with valacyclovir 500 mg bd for 7–10 days for the primary episode or 3 days for recurrent episodes.
Chancroid
Chancroid is a disease mainly seen in Asia, Africa and the Caribbean, with few cases reported in the developed world. There have been no reports in Australia for 10 years to 2012. It is caused by the Gram-negative bacillus Haemophilus ducreyi and presents with painful genital ulcers of up to 2 cm diameter. Painful inguinal lymph nodes may go on to suppurate if the infection is untreated. Chancroid may coexist with other genital ulcerating infections including syphilis and herpes simplex. Unlike herpes, chancroid is rarely accompanied by constitutional symptoms and is rarely recurrent. Diagnosis is generally clinical, although lesion swabs for culture and NAAT may confirm the diagnosis. Treatment is with azithromycin 1 g oral once or ceftriaxone 500 mg IM once.
Lymphogranuloma venereum
LGV is caused by Chlamydia trachomatis and is endemic in parts of Africa, South America, India, Southeast Asia and the Caribbean, although rarely seen in the developed world. Occasional cases occur in Australia and North America. There are three clinical stages. The initial presentation with a painless ulcer or papule may be missed. The second stage involves painful inguinal lymphadenopathy which is commonly unilateral. The third stage involves strictures, fistulae and scarring around the perianal area. Diagnosis is clinical with exclusion of other causes. Serology is not available everywhere.
Treatment is with doxycycline 100 mg orally twice daily for 21 days or erythromycin 500 mg four times daily for 21 days or azithromycin 1 g oral weekly for 3 weeks.
Donovanosis
This is now a rare infection overall, although there are pockets of increased prevalence in desert areas of central Australia and in rural tropical and subtropical areas including South East India, South Africa, Papua New Guinea and the Caribbean. Despite this, there have been no reported cases for over 10 years in Australia to 2012. It is caused by Klebsiella granulomatis and is seen more commonly in males. The incubation period is up to 12 weeks. Red papules in the genital and perianal area evolve into nodules of friable granulation tissue that bleed easily. The initial lesions may resemble chancroid and progress to spread and necrose with loss of genital tissue and depigmentation if untreated. Diagnosis is generally clinical and may be confirmed by lesion swab or scraping for NAAT, although specialized laboratory services may be required for testing. Treat with azithromycin 1 g oral weekly for at least 4 weeks or until fully healed.
Syphilis
Syphilis is currently increasing especially among MSM, with increased reporting worldwide including Australia, the UK and the USA. The causative organism is the spirochaete Treponema pallidum. Patients may present with symptoms in any of the three stages of primary, secondary or tertiary infection or may present in the latent phase. Primary syphilis has an incubation period of up to 3 weeks and classically presents with a painless genital ulcer, known as the primary chancre (E-Fig. 9.8.2 ). This may last for up to 6 weeks and is not accompanied by constitutional symptoms. If untreated, the primary stage may evolve into the secondary stage within 6 weeks, characterized by the distinctive macular pink rash which may resemble pityriasis rosea. It is often present on the flexor surfaces, trunk, palms and soles. The secondary stage is often accompanied by constitutional symptoms of fever, malaise and headache. Tertiary syphilis occurs up to 20 years after the primary infection in around one-third of patients with untreated secondary syphilis. The presentation includes widespread granulomas, known as gummas, or may present with meningitis, dementia, thoracic aneurysm or neuropathy, known as tabes dorsalis. There may be extensive involvement of the cardiovascular and nervous systems.
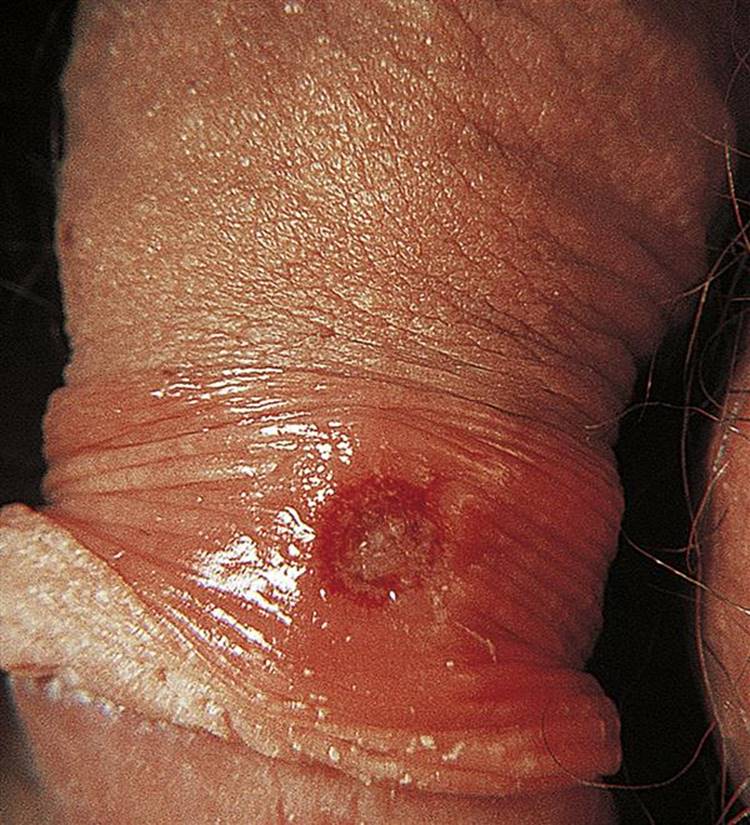
E-FIG. 9.8.2 Syphilis primary chancre. (Reproduced with permission from Campbell JL, Chapman MS, Dinulos JGH, Zug KA. Skin Disease: Diagnosis and Treatment, 3rd edn, Chapter 7, Sexually transmitted infections. Elsevier Inc., 2011, 184–209.)
Diagnosis depends on serological confirmation of treponemal or non-treponemal tests. Treponemal tests remain positive for life. Non-treponemal tests respond to treatment. An NAAT for syphilis may be requested from swabs or scrapings from rash or ulcers. Treatment is with benzathine penicillin 2.4 million units IM single dose if under 2 years’ duration or three doses at weekly intervals if over 2 years’ duration. There is no documented treponemal resistance as yet to penicillin, although treatment failures have occurred occasionally and are thought to be either re-infection or individual variation in decline of the non-treponemal test titres in response to treatment.
Patients should be notified to the regional syphilis register.
Genital warts
Genital warts are caused by the human papillomavirus. Up to three-quarters of sexually active adults are infected, although most infections are subclinical. Multiple warts, which may cluster, are seen over the vulva and penis. Internal warts may be seen in the rectum and around the cervix. Diagnosis is clinical and the differential diagnosis of molluscum contagiosum, secondary syphilis (condylomata) and carcinoma must be considered. Treatment is with podophllin 0.5% lotion applied twice daily to lesions for 3 days, then no treatment for 4 days, and repeat until lesions resolve. This may take four or more cycles of treatment.
Principles of clinical investigations
Diagnosis is initially clinical and treatment may need to be empirical. Attempts should be made to confirm the diagnosis by laboratory analysis of swabs and urine and it is important to communicate with laboratory staff to discuss collection, transport and testing of specimens and the specific type of swab, transport medium and temperature for each organism. All specimens should be correctly labelled with patient details in leakproof containers. Common tests include dark field microscopy for syphilis, Gram staining for gonorrhoea, chancroid and bacterial vaginosis, Tzanck smear for herpes and Donovanosis, wet mount for trichomoniasis and potassium hydroxide wet mount for candidiasis. NAAT are now seen as the gold standard for confirmation of chlamydia, gonorrhoea and some other STIs. Swabs from the affected areas for M, C and S and swabs and urinalysis for NAAT should be taken. Male swabs should include urethral smear for Gram stain, urethral, throat and rectal swab for culture, urinalysis including NAAT, blood for syphilis, HIV and hepatitis serology.
Female swabs should include lateral vaginal wall smear for Gram stain, vaginal swab for candida and trichomonas, endocervical, urethral, throat and rectal swabs for gonorrhea and urinalysis including pregnancy test, NAAT and culture and sensitivity. Referral for cervical cytology and serology for syphilis, HIV and hepatitis should be made.
If lesions are suggestive, scraping the lesion for syphilis microscopy and specialist swabs for HSV and Haemophilus ducreyi may be helpful.
The criteria used for diagnosing urethritis include one or more of: purulent or mucoid urethral discharge showing more than 5 white blood cells (WBC) per high power field, Gram stain of urine sediment showing more than 10 WBC per high power field and positive leucocyte esterase test on early morning first urine. NAAT may identify specific organisms.
Criteria for a diagnosis of cervicitis include purulent endocervical discharge and friable endocervical mucosa which bleeds easily when gently scraped. Direct visualization of organisms in discharge by wet mount, if available, or microscopy, Gram stain and culture of swab material or, if possible NAAT testing of discharge or urine specimens may confirm the diagnosis and add additional diagnoses.
Treatment
See Table 9.8.3 for an example summary guideline.
Table 9.8.3
Treatment guideline summary

Treatment is subject to current local guidelines and sensitivities and may vary according to regional strains and sensitivities. Where the infection likely originated is an important part of the history. Always check with local laboratories for current antimicrobial guidelines.
Follow up
Referral to a local STI clinic for follow up and contact tracing and treatment of partners is essential to stop the spread of STIs.
The patient should be advised of the need for partner treatment and of the need to abstain from sex until the infection has been treated adequately. The opportunity for health education around safer sexual practices and STI prevention should be taken. Notifiable diseases include chlamydia, gonorrhoea, syphilis, chancroid, LGV and Donovanosis.
Controversies and future directions
![]() There are conflicting views over whether the ED is the right place to screen for STIs including HIV, although it should be considered, especially in high prevalence areas. Screening while the patient is in the ED may be the only potential for diagnosis and treatment.
There are conflicting views over whether the ED is the right place to screen for STIs including HIV, although it should be considered, especially in high prevalence areas. Screening while the patient is in the ED may be the only potential for diagnosis and treatment.
![]() Debate exists in some regions over whether to treat infections empirically and the potential for increasing antibiotic resistance versus the burden of untreated disease. Syndromic treatment in areas of low prevalence is likely to be more effective in males as most cases of vaginal discharge in females are not STI related.
Debate exists in some regions over whether to treat infections empirically and the potential for increasing antibiotic resistance versus the burden of untreated disease. Syndromic treatment in areas of low prevalence is likely to be more effective in males as most cases of vaginal discharge in females are not STI related.
![]() More accurate and easier diagnostic testing, preferably at point of care is needed. Near patient testing is likely to become more accurate and more available in the future.
More accurate and easier diagnostic testing, preferably at point of care is needed. Near patient testing is likely to become more accurate and more available in the future.
![]() Changing antibiotic sensitivities of STIs mandates current local treatment guidelines and close liaison with colleagues in sexual health, infectious disease, microbiology and virology.
Changing antibiotic sensitivities of STIs mandates current local treatment guidelines and close liaison with colleagues in sexual health, infectious disease, microbiology and virology.
References
1. World Health Organization database. [Accessed Dec. 2012].
2. Australian Government Department of Health and Ageing. National Notifiable Diseases Surveillance System, Australia.<http://www9.health.gov.au/cda>[Accessed Dec. 2012].
3. British Association for Sexual Health and HIV (BASHH) Guidelines 2011.<www.bashh.org./guidelines>.
4. Trends in sexually transmitted diseases in the United States. Centers for Disease Control and Prevention Dec. 2012.<www.cdc.gov/std/stats>.
5. Mehta S, Hall J, Lyss S, et al. Adult and pediatric emergency department sexually transmitted disease and HIV screening programmatic overview and outcomes. Acad Emerg Med. 2007;14:250.
6. Bachmann L, Pigott D, Desmond R, et al. Prevalence and factors associated with gonorrhoea and chlamydia infection in at risk females presenting to an urban emergency department. Sex Transm Dis. 2003;30:335–339.
7. Smith KP, Christakis NA. Association between widowhood and risk of diagnosis with a sexually transmitted infection in older adults. Am J Publ Hlth. 2009;99:2055–2062.
8. Anderson M, Klink K, Cohrssen A. Evaluation of vaginal complaints. J Am Med Assoc. 2004;291:1368–1379.
9. Borhart J, Birnbaumer D. Emergency department management of sexually transmitted infections. Emerg Med Clin N Am. 2011;29:587–603.
10. Evans C, Das C, Kinghorn GA. Retrospective study of recurrent Chlamydia infection in men and women: is there a role for targeted screening for those at risk? Int J STD AIDS. 2009;3:188–192.
11. Annual report of the Australian Gonococcal Surveillance Programme 2011. Commun Dis Intell 2012: 36:E166-73.
12. Condon J, Rumbold A, Thorn J, et al. A cluster of vulval cancer and vulvar intraepithelial neoplasia in young Australian indigenous women. Cancer Causes Control. 2009;20:67–74.
Further reading
1. California STD/HIV Prevention.<www.stdhivtraining.org>.
2. Hall J, Lyss SB, et al. Adult and paediatric emergency department sexually transmitted disease and HIV screening; programmatic overview and outcomes. Acad Emerg Med. 2007;14:250–258.
3. Northern Territory Government, Department of Health, Australia. Guideline for the management of sexually transmitted infections in the primary care setting, February 2012.
4. Tapsall JW, Ndowa F, Lewis DA, Unemo M. Meeting the public health challenge of multidrug and extensively drug-resistant Neisseria gonorrhoeae. Expert Rev Anti Infect Ther. 2009;7:821–834.
9.9 Antibiotics in the emergency department
John Vinen
Essentials
1 Patients with infections and infectious diseases commonly present to emergency departments.
2 There are also changing patterns of infectious disease, largely due to immunosuppression from chemotherapy, continuing development of bacterial resistance, HIV-associated infections and new and emerging infections.
3 Many bacteria are becoming increasingly resistant to available antimicrobials, with some resistant to multiple agents including many community-acquired infections.
4 The growing world trade in wildlife, factory farming, increasing air travel and increased population density increases the risk of infectious disease transmission.
5 There are relatively few new antimicrobials to counter these changing patterns of resistance.
6 Antimicrobial prescribing should follow evidence-based guidelines.
7 Some patients with infection can be treated wholly as outpatients using parenteral therapy or after early discharge once the acute toxic phase is over.
8 Early administration of guideline-based antibiotics combined with supportive therapy is the key to a good outcome in patients with serious infections.
9 The increasing incidence of terrorism may result in patients presenting with novel, unusual or clusters of infections caused by biological agents.
Principles of antimicrobial therapy
The first decision to be made regarding antimicrobial therapy is whether the administration of these agents is truly indicated. In many cases, antibiotics are administered without clear indications. This practice is potentially dangerous, as some agents can cause serious toxicity, diagnoses may be masked if appropriate cultures are not taken prior to therapy, serious adverse events can result and microorganism resistance may emerge.
Ideally, antibiotic therapy is determined by isolation of the organism(s) involved and determination of antibiotic susceptibility pattern. As this information is rarely available, it is necessary to make treatment decisions without precise knowledge of infectious source or microbial species in which case empiric treatment is commenced based on the type of infection (if known) and the likely organisms involved utilizing recognized guidelines.
In specific situations (e.g. suspected meningitis, meningococcal infection, necrotizing fasciitis, sepsis, peritonitis, febrile neutropaenia, and pneumonia), early empiric therapy can be life saving.
The choice of an appropriate antimicrobial agent requires consideration of the following factors.
The microorganism
The identity of the infecting organism(s) needs to be identified or suspected. In the emergency department (ED) setting, almost all antimicrobial decisions will be made without the benefit of cultures, with treatment commencing based on the most likely to cause infection in a given clinical setting [1]. However, certain ‘rapid methods’ of microbial identification may be employed. These include Gram-stain preparations (bacterial, some fungal and leucocyte identification) and immunological methods for antigen detection (enzyme-linked immunoabsorbent assay, latex agglutination, polymerase chain reactions).
Microorganism susceptibility
The emergency physician is unlikely to have this information and therapeutic decisions will generally be based on a knowledge of likely susceptibilities [1]. For example, group A streptococci remain susceptible to the penicillins and cephalosporins and virtually all anaerobes (except Bacteroides spp.) are susceptible to penicillin G. However, when the identity or susceptibility of the infecting organism is sufficiently in doubt, the patient’s clinical condition is atypical, serious or potentially serious or where antimicrobial resistance is suspected, it is good practice to obtain appropriate specimens for culture and susceptibility testing prior to empirical broad-spectrum antimicrobial therapy (Table 9.9.1).
Table 9.9.1
Antimicrobial agents of choice in selected infections

All strains should be examined in vitro for sensitivity to various antimicrobial agents.
*G1, first-generation cephalosporin; G2, second-generation cephalosporin; G3, third-generation cephalosporin.
Host factors
An adequate history of drug allergies must be obtained in order to prevent the administration of an antimicrobial that may have serious or fatal consequences. Where this is not possible, avoid administration of penicillin and associated antimicrobials. The age of the patient may have clinically significant effects on drug absorption (e.g. penicillin absorption is increased in the young and the elderly) [2], metabolism (e.g. reduced chloramphenicol metabolism in the neonate) [2] and excretion (e.g. declining renal function with age [3] may reduce the excretion of penicillins, cephalosporins and aminoglycosides). Furthermore, tetracyclines bind and discolour the developing bone and tooth structures in children aged 8 years or less [2]. Pregnant women and nursing mothers may pose certain problems in the selection of appropriate antimicrobial agents, as all of these agents cross the placenta to varying degrees. The administration of antibiotics to pregnant patients must be based on guidelines [4]. Whether or not antibiotic use has an effect on the efficacy of combined oral contraceptive pills (OCPs) has been a matter of controversy. A significant pharmacokinetic interaction between combined OCPs and antibiotics, apart from rifampicin and griseofulvin, has not been proven. It has been suggested that if an interaction does exist, it is likely that it occurs in a small number of predisposed individuals. It is not possible at this time to predict who is at risk for potential interaction [5]. Other host factors that may require consideration include the patient’s renal and hepatic function, their genetic (e.g. liver acetylation rate) or metabolic abnormalities (e.g. diabetes mellitus) and the site of the infection [6].
Route of administration
In general, the oral route is chosen for infections that are mild and can be managed on an outpatient basis. In this situation, consideration needs to be given to compliance with treatment, the variability of absorption with food in the stomach and interaction of the agent with concomitant medications [7]. The parenteral route is used for agents that are inefficiently absorbed from the gastrointestinal tract and for the treatment of patients with serious infections in whom high concentrations of antimicrobial agents are required [7]. Intramuscular administration (not in patients on anticoagulants or who are coagulopathic) will provide adequate serum concentrations for most infections and may be appropriate where antimicrobial depots are desirable, e.g. procaine penicillin injections where patient compliance with oral medication is doubtful. Intravenous administration allows large doses of drugs to be given with a minimal amount of discomfort to the patient, e.g. infection prophylaxis in compound fractures, life-threatening infections and shock. For intravenous administration, large veins should be used followed by saline flushing of the veins to help to minimize the incidence of venous irritation and phlebitis.
Supportive care
Supportive care in association with antimicrobial therapy is essential in many infections, fluid resuscitation and vasopressors being essential for a good outcome in sepsis [8].
Adverse drug events involving antibiotics
Antibiotics are one of the top medication classes resulting in ED visits for adverse drug events.
There is a 1:1000 risk that an individual prescribed an antibiotic will require a visit to the ED because of an antibiotic side effect.
Antibiotics are responsible for 19% of ED visits for adverse drug events:
![]() in children (<18 years), antibiotics are the most common cause of ED visits for adverse drug events
in children (<18 years), antibiotics are the most common cause of ED visits for adverse drug events
![]() 79% of ED visits for antibiotic-associated adverse drug events are due to allergic reactions [9].
79% of ED visits for antibiotic-associated adverse drug events are due to allergic reactions [9].
Antibiotic resistance
Bacteria can be resistant to an antimicrobial agent because the drug fails to reach the target or is inactivated or because the target is altered [10–12]. Bacteria may produce enzymes that inactivate the drug or have cell membranes impermeable to the drug. Having gained entry into the microorganism, the drug must exert a deleterious effect. Natural variation or acquired changes at the target site that prevent drug binding or action can lead to resistance.
Resistance is most commonly acquired by horizontal transfer of resistance determinants from a donor cell, often of another bacterial species, by transformation, transduction or conjugation. Resistance may also be acquired by mutation and passed vertically by selection to daughter cells. Antimicrobial agents can affect the emergence of resistance by exerting strong selective pressures on bacterial populations favouring those organisms capable of resisting them [13].
The increasing emergence of antibiotic resistance is a very serious development that threatens the end of the antibiotic era. Penicillin-resistant strains of pneumococci account for>50% of isolates in some European countries. The worldwide emergence of Haemophilus and gonococci that produce β-lactamase is a major therapeutic problem [14]. Methicillin-resistant strains of Staphylococcus aureus are widely distributed among hospitals and are increasingly being isolated from community-acquired infections [15]. There are now strains of enterococci (VRE), Pseudomonas and enterobacters that are resistant to all known drugs [16]. Epidemics of multiply drug-resistant strains of Mycobacterium tuberculosis have been reported [16].
A more responsible approach to the use of antimicrobial agents is essential to slow the development of multidrug-resistant organisms. Their use should be avoided in viral infections and rational policies for their use in prophylaxis and in established bacterial infections must be developed and followed [1]. The use of narrow-spectrum antimicrobial agents to which the organism is susceptible is encouraged and, in certain circumstances, the use of combinations of agents may prevent the emergence of resistant mutants during therapy.
Prophylactic use of antibiotics
Antimicrobial prophylaxis is the use of antimicrobial agents in order to prevent infection developing. It is indicated in many circumstances, including the prevention of recurrent rheumatic fever, endocarditis, meningitis, tuberculosis and urinary tract and surgical infections [1]. Antimicrobial prophylaxis in the ED is usually indicated to prevent trauma-related infection following contamination of soft tissue, crush injuries, bites, clenched fist injuries and compound fractures. Other risk factors for wound infection include ‘old’ wounds (>6 hours), penetrating injuries, contaminated wounds, co-morbid illness, shock, colon injury and massive haemorrhage [17].
Antimicrobial prophylaxis should be considered where there is a significant risk of infection, but cannot be relied upon to overcome excessive soiling, damage to tissues, inadequate debridement or poor surgical technique. Adequate wound care, with splinting and elevation of the affected area as indicated, will continue to be important factors in trauma-related infection prophylaxis.
Antimicrobial prophylaxis should be directed against the likely causative organism(s). However, an effective regimen need not necessarily include antimicrobials that are active against every potential pathogen. Regimens that only reduce the total number of organisms may assist host defences and prevent infection [1]. The type, dose, duration and route of administration of antimicrobial therapy will vary according to the nature, site and aetiology of the injury, as well as host factors and should be based on established guidelines. In all cases of open traumatic injury, no matter how trivial, tetanus prophylaxis must be considered.
Penicillins
Chemistry and mechanism of action
The penicillins constitute one of the most important groups of antimicrobial agents and remain the drugs of choice for a large number of infectious diseases. The basic structure of the penicillins consists of a thiazolidine ring connected to a β-lactam ring, and a side chain. The penicillin nucleus is the chief structural requirement for biological activity, whereas the side chain determines many of the antibacterial and pharmacological characteristics of the particular type of penicillin.
Peptidoglycan is an essential component of the bacterial cell wall and provides mechanical stability by virtue of its highly cross-linked latticework structure. Penicillin is thought to acetylate and inhibit a transpeptidase enzyme responsible for the final cross-linking of peptidoglycan layers. Penicillin also binds to penicillin-binding proteins (PBPs), causing further interference with cell wall synthesis and cell morphology. The lysis of bacteria is ultimately dependent on the activity of cell wall autolytic enzymes – autolyses and murein hydrolases. Although the relationship between the inhibition of PBP activity and the activation of autolysins is unclear, the interference with peptidoglycan assembly in the face of ongoing autolysis activity might well lead to cell lysis and death.
Bacterial resistance to penicillins
Microorganisms may be intrinsically resistant to the penicillins because of structural differences in PBPs. Resistance may be acquired by the development of high molecular weight PBPs that have reduced affinity for the antibiotic [12]. Bacterial resistance can also be caused by the inability of the agent to penetrate to its site of action. Unlike Gram-positive bacteria, Gram-negative bacteria have an outer membrane of lipopolysaccharide which functions as an impenetrable barrier to some antibiotics. However, some broader-spectrum penicillins, such as ampicillin and amoxicillin, can diffuse through aqueous channels (porins) of this outer membrane to reach their sites of action.
Bacteria can destroy penicillins enzymatically. Different bacteria elaborate a number of different β-lactamases and individual penicillins vary in their susceptibility to these enzymes. In general, Gram-positive bacteria produce a large amount of β-lactamase, which is secreted extracellularly. Most of these enzymes are penicillinases which disrupt the β-lactam ring and inactivate the drug. In Gram-negative bacteria, β-lactamases are found in relatively small amounts strategically located between the inner and outer bacterial membranes for maximal protection.
Classification of penicillins
Benzylpenicillin (penicillin G) and phenoxymethyl penicillin (penicillin V)
These drugs are the so-called ‘natural penicillins’. The antimicrobial spectra of benzyl penicillin (penicillin G) and phenoxymethyl penicillin (penicillin V) are very similar for aerobic Gram-positive microorganisms. Benzyl penicillin is the drug of choice against many Gram-positive cocci (streptococci, penicillin-sensitive staphylococci), Gram-negative cocci (Neisseria meningitidis and N. gonorrhoeae), Gram-postive bacilli (Bacillus anthracis, Corynebacterium. diphtheriae), anaerobes (peptostreptococcus, Actinomyces israelii, Clostridium and some Bacteroides), Pasteurella multocida and Treponema pallidum. Phenoxymethyl penicillin is an acceptable alternative for Streptococcus pneumoniae, Strep. pyogenes (A) and Actinomyces israelii.
The sole virtue of benzylpenicillin compared to phenoxymethyl penicillin is that it is more stable in an acid medium and therefore much better absorbed from the gastrointestinal tract. Benzylpenicillin is administered parenterally but has a half-life of only 30 minutes. Accordingly, repository preparations (penicillin G procaine, penicillin G benzathine) are often used and probenecid may be administered concurrently to block the renal tubular secretion of the drug. Once absorbed, both penicillins are distributed widely throughout the body. Significant amounts appear in the liver, bile, kidney, semen, joint fluid, lymph and intestine. Importantly, penicillin does not readily enter the CSF when the meninges are normal. However, when the meninges are acutely inflamed penicillin penetrates into the CSF more easily. Under normal circumstances, penicillin is eliminated unchanged by the kidney, mainly by tubular secretion.
The penicillinase-resistant penicillins
These drugs remain the agents of choice for most staphylococcal disease. Methicillin is a penicillin resistant to staphylococcal β-lactamase, although the increasing incidence of isolates of methicillin-resistant microorganisms is cause for concern. Methicillin-resistant Staph. aureus (MRSA) contain a high molecular weight PBP with a very low affinity for β-lactam antibiotics [12]. From 40 to 60% of strains of Staph. epidermidis are also resistant to penicillinase-resistant penicillins by the same mechanism. As bacterial sensitivities are usually not known in the ED, methicillin is rarely administered in this setting.
The isoxazolyl penicillins (oxacillin, cloxacillin, dicloxacillin and flucloxacillin) are congeneric semisynthetic penicillins which are pharmacologically similar. All are relatively stable in an acid medium and are adequately absorbed after oral administration. These penicillins undergo some metabolism but are excreted primarily by the kidney with some biliary excretion. All are remarkably resistant to cleavage by penicillinase and inhibit both penicillin-sensitive and some penicillin-resistant staphylococci. Methicillin-resistant staphylococci are resistant to these penicillins. Isoxazolyl penicillins inhibit streptococci and pneumococci but are virtually inactive against Gram-negative bacilli.
The aminopenicillins
Ampicillin is the prototypical agent in this group. It is stable in acid medium and, although well absorbed orally, is often administered parenterally. Amoxicillin is a close chemical and pharmacological relative of ampicillin. The drug is stable in acid and was designed for oral use. It is more rapidly and completely absorbed from the gastrointestinal tract than is ampicillin. The antimicrobial spectra of these agents are essentially identical, with the important exception that amoxicillin appears to be less effective for shigellosis. Ampicillin is the penicillin of choice for many Gram-negative bacilli (H. influenzae, Escherichia coli, Proteus mirabilis, Salmonella typhi and Salmonella spp.), some Gram-positive bacilli (Listeria monocytogenes) and some Gram-positive cocci (Enterococcus faecalis). It also has activity against Pneumococcus spp., Neisseria spp., Peptostreptococcus, Fusobacterium, Clostridium and Erysipelothrix.
Bacterial resistance to these drugs is becoming an increasing problem. Many pneumococcal isolates have varying levels of resistance to ampicillin. H. influenzae and the viridans group of streptococci are usually inhibited by very low concentrations of ampicillin. However, strains of H. influenzae (type b) that are highly resistant to ampicillin have been recovered from children with meningitis. It is estimated that 30% or more cases of H. influenzae meningitis are now caused by ampicillin-resistant strains. Similarly, ampicillin-resistant strains of H. influenzae have been increasingly isolated from cases of acute otitis media. An increasing percentage of N. gonorrhoeae, E. coli, P. mirabilis, Salmonella and Shigella are now resistant to ampicillin and practically all species of Enterobacter are now insensitive.
β-Lactamase inhibitors have been introduced to combat many penicillin-resistant microorganisms. These molecules bind to β-lactamases and inactivate them, thereby preventing the destruction of β-lactamase antibiotics. Clavulanic acid binds to the β-lactamases produced by a wide range of Gram-positive and Gram-negative microorganisms. It is well absorbed orally and can also be given parenterally. It has been combined with amoxicillin as an oral preparation (Augmentin) and with ticarcillin (a carboxypenicillin) as a parenteral preparation (Timentin). Augmentin is effective for β-lactamase-producing strains of staphylococci, H. influenzae, gonococci and E. coli. Sulbactam is another β-lactamase inhibitor which also can be administered orally or parenterally. In combination with ampicillin (Unasyn), good coverage is provided for Gram-positive cocci (including β-lactamase-producing strains of Staph. aureus), Gram-negative anaerobes (but not Pseudomonas) and anaerobes.
Adverse reactions to penicillin
Hypersensitivity reactions are the major adverse effects of penicillins. Penicillins are capable of acting as haptens to combine with proteins contaminating the solution or with human protein after the penicillin has been administered. Penicilloyl and penicillanic derivatives are the major determinants of penicillin allergy. All acute hypersensitivity reactions to penicillin are mediated by the IgE antibody and range in severity from rash to anaphylaxis. Anaphylactic reactions are uncommon, occurring in only 0.2% of 1000 courses of treatment, with 0.001% out of 100 000 courses resulting in death [18]. Morbilliform eruptions that develop after penicillin therapy are likely to be mediated by IgM antibodies and the uncommon serum sickness is likely to be mediated by IgG antibodies. All forms of penicillin are best avoided in patients with a history of penicillin allergy.
Otherwise, the penicillins are generally well tolerated. CNS toxicity, in the form of myoclonic seizures, can follow the administration of massive doses of benzylpenicillin (penicillin G), ampicillin or methicillin. Massive doses have also been associated with hypokalaemia. Haematological toxicity – usually neutropaenia – and nephrotoxicity have also been reported. Gastrointestinal disturbances have followed the use of all oral penicillins, but have been most pronounced with ampicillin. Enterocolitis due to the overgrowth of Cl. difficile is well documented and abnormalities in liver function have been reported, especially with flucloxacillin [19].
Cephalosporins
The antimicrobial activity of cephalosporins, like that of other β-lactam antibiotics, results at least in part from their ability to interfere with the synthesis of the peptidoglycan component of the bacterial cell wall. However, the exact bactericidal and lytic effects of cephalosporins are not completely understood.
Classification and uses
The first-generation compounds (cephalothin, cefazolin, cefalexin) have a relatively narrow spectrum of activity focused primarily on the Gram-positive cocci, especially penicillin-sensitive streptococci and methicillin-sensitive Staph. aureus. These compounds have modest activity against Gram-negative organisms, including E. coli and Klebsiella spp. Cefaclor has extended Gram-negative activity and is active against H. influenzae and M. catarrhalis.
The second generation of cephalosporins (cefuroxime, cefamandole) are more stable against Gram-negative β-lactamases. They have variable activity against Gram-positive cocci, but have increased activity against Gram-negative bacteria (E. coli, Proteus, Klebsiella). In spite of relatively increased potency against Gram-negative aerobic and anaerobic bacilli (Bacteroides fragilis), the cephamycins (cefoxitin, cefotetan) are included in this generation.
The third-generation cephalosporins (cefotaxime, ceftriaxone, ceftazidime, cefpirome) have very marked activity against Gram-negative bacteria. Most are useful against Ps. aeruginosa, Serratia and Neisseriaspecies and some Enterobacteriaceae. Some of these compounds have limited activity against Gram-positive cocci, particularly methicillin-sensitive Staph. aureus. This generation of cephalosporins is particularly effective in meningitis because of their better penetration into the CSF and higher intrinsic activity. However, as these third-generation drugs are more expensive and have a wide antimicrobial spectrum, their use should be based on established guidelines.
Recently, several compounds have been considered as possibly meriting classification as a fourth generation. Cefepime has activity against Gram-positive cocci and a broad array of Gram-negative bacteria, including Ps. aeruginosa and many of the Enterobacteriaceae with inducible chromosomal β-lactamases.
Adverse reactions
Hypersensitivity reactions are the most common side effects of the cephalosporins and all compounds have been implicated. The reactions appear to be identical to those caused by the penicillins. Immediate reactions, such as anaphylaxis, bronchospasm, angio-oedema and urticaria, have been reported. More commonly, a maculopapular rash develops, usually after several days of therapy. Because of the similarity in structure between the penicillins and the cephalosporins, patients allergic to one class of agents may manifest cross-reactivity when a member of the other class is administered. Studies indicate that about 0.5% of patients allergic to penicillin will demonstrate a clinically apparent reaction when a first-generation cephalosporin is administered (0% for second- and third-generation cephalosporins) [20]. Patients with a mild or temporarily distant reaction to penicillin appear to be at low risk of rash or other allergic reactions following the administration of a cephalosporin. However, subjects with a recent history of an immediate reaction to penicillin should not be given a cephalosporin. Other reactions to cephalosporins are uncommon and include diarrhoea, nephrotoxicity, intolerance of alcohol and bleeding disorders.
Penicillin allergy cross-reactivity with cephalosporins is significantly overstated. Cross-reactivity between penicillins and cephalosporins is much less than the 10% commonly cited. Cephalothin, cephalexin, cefadroxil and cefazolin confer an increased risk of allergic reaction among patients with penicillin allergy.
Cefuroxime, cefpodoxime, ceftazidime and ceftriaxone do not increase risk of an allergic reaction.
No cross-reactivity exists between penicillins and third-generation cephalosporins. However, if a patient has known anaphylaxis to penicillin, caution with cephalosporin use is still warranted.
Bacterial resistance
The most prevalent mechanism for resistance to cephalosporins is their destruction by β-lactamase hydrolysis. The cephalosporins have variable susceptibility to β-lactamase, with the later-generation compounds being more resistant to the β-lactamases produced by Gram-negative bacteria. However, third-generation cephalosporins are susceptible to hydrolysis by inducible, chromosomally encoded (type 1) β-lactamases. The induction of type 1 β-lactamases by treatment of infections due to many aerobic Gram-negative bacilli with second- or third-generation cephalosporins may result in resistance to all third-generation cephalosporins.
Macrolides
Erythromycin was originally isolated from soil bacteria and contains a many-membered lactone ring to which are attached one or more deoxy sugars. Clarithromycin, azithromycin and roxithromycin are new semisynthetic derivatives of erythromycin. Clarithromycin differs only by methylation of a hydroxyl group and azithromycin contains a methyl-substituted nitrogen atom in the lactone ring. Roxithromycin is a good alternative to oral erythromycin and has good oral bioavailability, but is more expensive. The macrolides are usually bacteriostatic and inhibit protein synthesis by binding reversibly to 50S ribosomal subunits of sensitive microorganisms. They are thought to inhibit the translocation step wherein a newly synthesized peptidyl tRNA molecule moves from the acceptor site on the ribosome to the peptidyl (donor) site.
Clinical uses
Erythromycin is most effective against aerobic Gram-positive cocci and bacilli. It is active against Strep. pyogenes, Strep. pneumoniae, Cl. perfringens, Cl. diphtheriae, L. monocytogenes and some staphylococci. Useful activity has also been seen with P. multocida, Borrelia spp., B. pertussis, Campylobacter jejuni, Legionella pneumophila, M. pneumoniae, C. trachomatis and some atypical mycobacteria. It has modest activity in vitroagainst some Gram-negative organisms, including H. influenzae and N. meningitidis and excellent activity against most strains of N. gonorrhoeae.
Clarithromycin is more potent against erythromycin-sensitive strains of streptococci and staphylococci, but has only modest activity against H. influenzae and N. gonorrhoeae. However, it has good activity against M. catarrhalis, Chlamydia spp., L. pneumophila and Mycoplasma pneumoniae. Azithromycin is generally less active than erythromycin against the Gram-positive organisms and is more active than the other two macrolides against H. influenzae and Campylobacter spp. Azithromycin is very active against M. catarrhalis, P. multocida, Chlamydia spp., M. pneumoniae, L. pneumophila and N. gonorrhoeae.
Adverse reactions
Erythromycin is one of the safest antibiotics and causes serious adverse effects only rarely. Dose-related abdominal cramps, nausea, vomiting, diarrhoea and flatulence occur, but are uncommon in children and young adults. Allergic reactions observed include fever, eosinophilia and skin eruptions. Cholestatic hepatitis, transient hearing loss, polymorphic ventricular tachycardia, superinfection of the gastrointestinal tract and pseudomembranous colitis have been reported. Intravenous use of erythromycin is often associated with thrombophlebitis, but the incidence of this complication can be reduced with appropriate dilution of the dose. Adverse reactions to the other macrolides, at the usual dose, are rare and usually confined to the gastrointestinal tract. For this reason, roxithromycin is often prescribed instead of erythromycin.
Erythromycin and, to a lesser extent, the other macrolides, has been reported to cause clinically significant drug interactions [21]. Erythromycin has been reported to potentiate astemizole, terfenadine, carbamazepine, corticosteroids, digoxin, theophylline, valproate and warfarin, probably by interfering with cytochrome P450-mediated drug metabolism. Care should be used in the concurrent administration of the macrolides with these drugs.
Bacterial resistance
Resistance to erythromycin may be the result of reduced permeability through the cell envelope. This form of resistance is exhibited by the Enterobacteriaceae and Pseudomonas spp. Alteration of ribosomal proteins, especially the 50S protein, often affects binding of the drug and has led to the emergence of resistant strains of B. subtilis, Strep. pyogenes and Strep. pneumoniae, Campylobacter spp., E. coli, Staph. aureus, Cl. perfringens, Listeria spp. and Legionella spp. Finally, enzymatic degradation of the drug has conferred high-level resistance among strains of Enterobacteriaceae.
Tetracycline
Tetracyclines are generally bacteriostatic and are thought to inhibit bacterial protein synthesis by binding to the 30S bacterial ribosome and preventing access of aminoacyl tRNA to its acceptor site.
Clinical uses
The antimicrobial spectra of all the tetracyclines are almost identical. They possess a wide range of antimicrobial activity against aerobic and anaerobic Gram-positive and Gram-negative bacteria. Clinically, the tetracyclines are useful against Strep. pneumoniae, H. influenzae, Neisseria spp., E. coli, Brucella spp., H. ducreyi, Vibrio cholerae, Campylobacter spp. and some Shigella and Mycobacterium spp. Many pathogenic spirochaetes are susceptible, including Borrelia burgdorferi. They are also effective against some microorganisms that are resistant to cell-wall active antimicrobial agents, such as Rickettsia, Coxiella burnetti, Mycoplasma pneumoniae,Chlamydia spp., Legionella spp. and Plasmodium spp.
Adverse reactions
The tetracyclines all produce gastrointestinal irritation in some individuals, although doxycycline is usually well tolerated. Epigastric discomfort, nausea, vomiting and diarrhoea are commonly reported. Renal and liver toxicity and photosensitivity may occur. Tetracyclines are deposited in the skeleton and teeth during gestation and childhood and can cause abnormalities of bone growth and discoloration of the teeth. It is therefore essential not to administer these agents to pregnant women or children under 8 years of age. Hypersensitivity reactions, including skin reactions, burning of the eyes, pruritus ani, vaginitis, angio-oedema and anaphylaxis, are rarely seen.
Bacterial resistance
Bacteria develop resistance to the tetracyclines mainly by preventing the accumulation of the drug within the cell. This is accomplished by reducing the influx or increasing the ability of the cell to export the antibiotic. Rarely, the tetracyclines are inactivated biologically or inhibited in their ribosomal attachment [22]. Resistance to one tetracycline usually means resistance to all. Clinically, most strains of enterococci are now resistant to tetracycline; group B streptococci are 50% susceptible and only 65% of Staph. aureus remain susceptible. Resistant pneumococci are now found in many geographical areas and many strains of Neisseriaspp. are now resistant.
Aminoglycosides
Each aminoglycoside demonstrates concentration-dependent bactericidal activity against susceptible microorganisms. Gentamicin is the most commonly administered aminoglycoside in the ED and is a mixture of three closely related constituents. It binds to a specific area on the interface between the smaller (30S) and the larger (50S) bacterial ribosomal subunits, causing an increase in misreading of messenger RNA and a measurable decrease in protein synthesis. However, these effects do not provide a complete explanation for the rapidly lethal effect of gentamicin on bacteria.
Clinical uses
The antibacterial activity of gentamicin is directed primarily against aerobic and facultative Gram-negative bacilli. It has little activity against anaerobic microorganisms and facultative bacteria under anaerobic conditions and its activity against most Gram-positive bacteria is very limited. Gentamicin is clinically effective against Pseudomonas aeruginosa, Proteus mirabilis, Klebsiella pneumoniae, E. coli, Enterobacter spp. and Serratia spp. It is particularly effective when used in combination with cell-wall active antimicrobial agents, e.g. penicillin, cephalosporin. Interactions between these agents result in synergistic effects on bacterial death and may be useful against enterococci, Strep. pyogenes, some staphylococci, Enterobacteriaceae and Pseudomonas aeruginosa.
Adverse reactions
Like most other aminoglycosides, gentamicin has the potential to cause injury to the renal proximal convoluted tubules, damage to the cochlear and/or vestibular apparatus and neuromuscular blockade. As the drug is eliminated almost entirely by glomerular filtration, gentamicin dosing in renal failure must be undertaken with care and drug-level monitoring is recommended. Gentamicin has little allergenic potential. Anaphylaxis, rash and other hypersensitivity reactions are unusual.
Bacterial resistance
Bacteria defend themselves against the aminoglycosides by a combination of alteration of uptake, synthesis of modifying enzymes and a change of ribosomal binding sites.
In several centers, a significant percentage of clinical isolates are highly resistant to all aminoglycosides. At present, other widespread bacterial resistance to the aminoglycosides remains limited. However, there are reports of resistance emerging among some strains of Ps. aeruginosa, Enterobacteriaceae, E. coli, Serratia spp. and Staph. aureus.
Metronidazole
The toxicity of metronidazole is due to short-lived intermediate compounds or free radicals that produce damage by interaction with DNA and possibly other macromolecules.
Clinical uses
Metronidazole is active against a wide variety of anaerobic protozoal parasites. It is directly trichomonicidal. Sensitive strains of Trichomonas vaginalis are killed by very low concentrations of the drug under anaerobic conditions. The drug also has potent amoebicidal activity against E. histolytica, even in mixed culture, and substantial activity against the trophozoites of Giardia lamblia. Metronidazole manifests antibacterial activity against all anaerobic cocci and both anaerobic Gram-negative bacilli and anaerobic spore-forming Gram-positive bacilli. Bacteroides, Clostridium, Helicobacter, Fusobacterium, Peptococcus and Peptostreptococcusspp. are all susceptible.
Adverse reactions
In general, metronidazole is well tolerated. The most common side effects are headache, nausea, dry mouth and a metallic taste. Vomiting, diarrhoea and abdominal distress are occasionally experienced [23]. Furry tongue, glossitis and stomatitis may occur during therapy and are associated with a sudden intensification of moniliasis. Of clinical importance is metronidazole’s well-documented disulfiram-like effect (Antabuse). Some patients experience abdominal distress, vomiting, flushing or headache if they drink alcohol during therapy with this drug.
Bacterial resistance
Fortunately, very few strains of Bacteroides spp. have demonstrated resistance. Some resistant strains of T. vaginalis have been isolated from patients with refractory cases of trichomoniasis, but these patients have usually responded to higher doses of metronidazole and prolonged courses of therapy [24].
Co-trimoxazole
Co-trimoxazole is a combination of sulphamethoxazole, a sulphonamide antibiotic, and trimethoprim, a diaminopyrimidine. The antimicrobial activity of this combination results from actions on two steps of the enzymatic pathway for the synthesis of tetrahydrofolic acid. Sulphamethoxazole inhibits the incorporation of PABA into folic acid and trimethoprim prevents the reduction of dihydrofolate to tetrahydrofolate. The latter is the form of folate essential to bacteria for one-carbon transfer reactions. Mammalian cells utilize preformed folate from the diet and do not synthesize this compound. This combination has been associated with serious sulphonamide-induced side effects. It has been recommended that the combination product be restricted to the few situations where combined use is the treatment of choice [1].
Clinical uses
Trimethoprim is effective in the treatment of most urinary tract infections and should be used alone for this indication. However, co-trimoxazole is active against a wide range of Gram-positive and Gram-negative microorganisms. C. diphtheriae and N. meningitidis are susceptible, as are most strains of Strep. pneumoniae. From 50 to 95% of strains of H. influenzae, Staph. aureus and epidermidis, Strep. pyogenes and viridans, E. coli, Proteus mirabilis, Enterobacter spp., Salmonella, Shigella and Serratia are inhibited. Also sensitive are Klebsiella spp., Brucella abortis, Pasteurella haemolytica and Yersinia spp. Co-trimoxazole has an important place in the treatment and prophylaxis of P. carinii infection and the treatment of L. monocytogenes and Nocardia infection.
Adverse reactions
In routine use, the combination appears to produce little toxicity. About 75% of adverse reactions involve the skin. These reactions are typical of those produced by sulphonamides and include a wide variety of rashes, erythema nodosum, erythema multiforme and Stevens–Johnson syndrome, exfoliative dermatitis and photosensitivity. Severe reactions tend to be more common among the elderly and HIV-infected patients. Gastrointestinal reactions include nausea and vomiting, but rarely diarrhoea. Glossitis and stomatitis are relatively common. Central nervous system reactions (headache, depression and hallucinations) and haematological disorders (anaemias, coagulation disorders and granulocytopaenia) have been reported.
Bacterial resistance
The frequency of development of bacterial resistance to co-trimoxazole is lower than it is to either of the constituent compounds alone. Resistance to sulphamethoxazole is presumed to originate by random mutation and selection or by transfer of resistance by plasmids. Such resistance is usually persistent and irreversible. Resistance to all sulphonamides is now becoming widespread in both community and nosocomial strains of bacteria, including streptococci, staphylococci, Enterobacteriaceae, Neisseria spp. and Pseudomonas spp. Trimethoprim-resistant microorganisms may arise by mutation, but resistance in Gram-negative bacteria is often associated with the acquisition of a plasmid that codes for an altered dihydrofolate reductase. Increasing incidences of resistance have been found in Enterobacteriaceae, Ps. aeruginosa, Staph. aureus, E. coli, Salmonella and Shigella.
Quinolones
The 4-quinolones, including nalidixic acid, are a family of compounds that contain a carboxylic acid moiety attached to a basic ring structure. The newer fluoroquinolones also contain a fluorine substituent, e.g. ciprofloxacin, and ofloxacin. Some may also contain a piperazine moiety. Bacterial DNA gyrase is an essential enzyme involved in DNA function. The quinolones inhibit the enzymatic activities of DNA gyrase and promote the cleavage of DNA within the enzyme–DNA complex.
Clinical uses
The early quinolones are most active against aerobic Gram-negative bacilli, particularly Enterobacteriaceae and Haemophilus spp. and against Gram-negative cocci, such as Neisseria spp. and M. catarrhalis. The fluoroquinolones are significantly more potent and have a much broader spectrum of antimicrobial activity. Relative to nalidixic acid, the fluoroquinolones also have additional activity against Ps. aeruginosa and some staphylococci. Ciprofloxacin remains the most potent fluoroquinolone against Gram-negative bacteria. Several intracellular bacteria are inhibited by the fluoroquinolones, including Chlamydia, Mycoplasma, Legionella,Brucella and some mycobacteria. Recently, a new drug, moxifloxacin, has been released that is useful for sinusitis, community-acquired pneumonia and acute bronchitis.
Adverse reactions
Generally, these drugs are well tolerated. Gastrointestinal symptoms of anorexia, nausea, vomiting, diarrhoea and abdominal discomfort are commonly seen, particularly with the older quinolones. Headache, dizziness, insomnia and alteration in mood are the next most commonly reported symptoms. Allergic and skin reactions, including phototoxicity, may occur. Rarely, arthralgias and joint swelling, leucopaenia, eosinophilia, thrombocytopaenia and haemolysis are reported.
Bacterial resistance
Resistance patterns over time have indicated that resistance increased following the introduction of fluoroquinolones and occurred most often with Pseudomonas spp. and staphylococci and in soft-tissue infections and in infections associated with foreign bodies. Possibly reflecting the pressures of extensive use, increasing fluoroquinolone resistance has been reported among strains of Cl. jejuni and E. coli. Focused quinolone use should be considered to avoid compromising the utility of the fluoroquinolones.
Nitrofurantoin
The mechanism of action is poorly understood, but activity in many cases appears to require enzymatic reduction within the bacterial cell [25]. The reduced derivatives are thought to bind to and damage intracellular proteins, including DNA, and inhibit bacterial respiration, pyruvate metabolism and the synthesis of inducible enzymes.
Clinical uses
Nitrofurantoin is active against over 90% of clinical strains of E. coli, Citrobacter spp., Staph. saprophyticus and E. faecalis. However, most species of Proteus, Pseudomonas, Serratia, Providencia, Morganella and many Enterobacter and Klebsiella spp. are resistant. Given its spectrum of activity and concentration in the urine, nitrofurantoin is usually administered for the treatment of urinary tract infections or for urinary antisepsis. However, it may have activity against bacteria not usually associated with urinary tract infections, including Salmonella, Shigella, Staph. aureus, Strep. pneumoniae and pyogenes and Bacteroides. Fortunately, bacteria that are susceptible to nitrofurantoin rarely become resistant during therapy.
Adverse reactions
Gastrointestinal upsets, particularly nausea, vomiting and diarrhoea, are the commonest side effects of nitrofurantoin. The frequency of these symptoms may be reduced if the macrocrystalline formulation is administered. Rashes, presumably allergic in nature, have been seen quite commonly. Cholestatic jaundice, acute and chronic hepatitis, pulmonary and haematological reactions and peripheral neuropathies have all been reported.
Colistin link parenteral
Colistin Link Parenteral has activity against Gram-negative bacilli: Enterobacter aerogenes, E. coli, Klebsiella pneumoniae and Pseudomonas aeruginosa.
Serious infection due to strains of Pseudomonas aeruginosa that exhibit resistance to all common antipseudomonal antimicrobials is an increasingly serious problem.
Pseudomonas aeruginosa is the Gram-negative pathogen that most commonly causes nosocomial pneumonia and is associated with the highest rates of crude and attributable mortality, even among patients who receive appropriate antimicrobial therapy.
Colistin Link Parenteral is eliminated mainly by renal excretion, it should be used with
caution when the possibility of impaired renal function exists. The decline in renal function with advanced age should be taken into consideration, it can also cause nephrotoxicity. Maximum daily dose should not exceed 5 mg/kg/day with normal renal function [26].
Antiviral drugs
Several antiviral drugs are available, although famciclovir, acyclovir and valacyclovir (prodrug of acyclovir that requires a lower dosage frequency) are the most frequently prescribed. Their mechanism of action is similar. Each drug targets virus-infected cells and inhibits viral DNA polymerase. Consequently, viral DNA synthesis and therefore viral replication are inhibited.
Clinical uses
These drugs are primarily used for the management of herpes zoster (within 72 hours of rash onset), treatment and suppression of genital herpes and the management of patients with advanced symptomatic HIV disease. Famciclovir is well absorbed in the gut and has the advantage of three times daily dosage compared to five times daily for acyclovir.
Acyclovir is also used to treat herpex simplex encephalitis (HSE).
HSE needs to be distinguished from herpes simplex meningitis, which is more commonly caused by HSV-2 than by HSV-1 and which often occurs in association with a concurrent herpetic genital infection.
Empiric treatment with acyclovir is essential in patients with suspected HSE pending confirmation of the diagnosis because acyclovir is the drug of choice and is relatively non-toxic and, if commencement of treatment is delayed, the prognosis for untreated HSE is poor.
Adverse reactions
These drugs are generally well tolerated. However, headache, gastrointestinal disturbance, dizziness and fatigue have been reported. Adverse effects are generally mild.
Antiviral agents for influenza
Zanamivir and oseltamivir are related antiviral medications known as neuraminidase inhibitors. These two medications are active against both influenza A and B viruses. They differ in pharmacokinetics, safety profiles, route of administration, approved age groups and recommended dosages.
The two other drugs used to treat influenza, amantadine and rimantadine are related antiviral drugs classified as adamantanes. These medications are active against influenza A viruses but not influenza B viruses. Widespread adamantane resistance among influenza A (H3N2) virus strains has made this class of medications less useful clinically.
Early antiviral treatment can shorten the duration of fever and symptoms and may reduce the risk of complications from influenza (e.g. otitis media in young children, pneumonia, respiratory failure) and death and shorten the duration of hospitalization. Clinical benefit is greatest when antiviral treatment is administered early, especially within 48 hours of influenza illness onset.
Antiretroviral drugs
Emergency physicians are unlikely to initiate these drugs as they form the basis of HIV treatment.
The only exception is prophylaxis following needle stick injury or body fluid exposure where close adherence to the hospital’s policy is essential.
However, an appreciation of their uses and side effects is useful. Furthermore, the management of patients with HIV disease can be difficult and advice from an appropriate specialist source is essential.
Standard antiretroviral therapy (ART) consists of the combination of at least three antiretroviral (ARV) drugs to suppress maximally the HIV virus and stop the progression of HIV disease.
Clinical uses
The antiretrovirals are used in the treatment of established HIV infection. This includes patients with HIV-associated illnesses (e.g. CNS disease, malignancies, opportunistic diseases) and asymptomatic patients with low CD4 cell counts and/or high HIV viral loads. The drugs are also of use in the prevention of maternofetal transmission and as post-exposure prophylaxis for significant exposure from a known HIV-infected source.
Three major classes of antiretroviral drugs are available. For initial therapy, two to three drugs are generally used in combination (see Chapter 9.7).
Antifungal agents
Systemic fungal infections are becoming more and more common. Candidiasis and aspergillosis are the most common infections; other systemic fungal infections include histoplasmosis, blastomycosis, coccidioidomycosis.
Severe systemic fungal infection in hospitals are commonly seen in:
![]() neutropaenic patients following chemotherapy and other oncology patients with immune suppression
neutropaenic patients following chemotherapy and other oncology patients with immune suppression
![]() persons that are immune compromised due to acquired immune deficiency syndrome caused by HIV infection
persons that are immune compromised due to acquired immune deficiency syndrome caused by HIV infection
![]() patients in intensive care (ICU), who are not necessarily neutropaenic, but are compromised due to the presence of long-term intravascular lines or other breaches in their integument, severe systemic illness or burns and prolonged broad-spectrum antibiotic therapy.
patients in intensive care (ICU), who are not necessarily neutropaenic, but are compromised due to the presence of long-term intravascular lines or other breaches in their integument, severe systemic illness or burns and prolonged broad-spectrum antibiotic therapy.
Other predisposing factors include:
![]() APACHE score>10
APACHE score>10
![]() renal dysfunction
renal dysfunction
![]() haemodialysis
haemodialysis
![]() surgery for acute pancreatitis, splenectomy
surgery for acute pancreatitis, splenectomy
![]() recurrent GIT perforation
recurrent GIT perforation
![]() Hickmann catheters.
Hickmann catheters.
Systemic fungal infections cause ≈25% of infection-related deaths in leukaemics. Infections due to Candida species are the fourth most important cause of nosocomial bloodstream infection.
The mainstay of antifungal therapy for severe systemic mycoses is amphotericin B.
Cryptococcal meningitis
Cryptococcus neoformans is an encapsulated yeast. The most serious infections usually develop in patients with defective cell-mediated immunity including, patients with:
![]() AIDS
AIDS
![]() organ transplantation
organ transplantation
![]() reticuloendothelial malignancy
reticuloendothelial malignancy
![]() corticosteroid treatment
corticosteroid treatment
![]() sarcoidosis.
sarcoidosis.
The incidence of cryptococcosis is increasing and now represents a major life-threatening fungal infection in AIDS patients.
Occupational risk factors for the infection include arborists and those exposed to bird droppings.
The initial site or sites of infection (pulmonary, CNS, and disseminated disease) determine the medical history of patients with symptomatic cryptococcal disease.
Patients with CNS infections, which are usually subacute or chronic in nature, present with headaches, neck pain, confusion, lethargy, malaise, and then, as the untreated infection progresses, focal neurological defects and decreased LOC. Fever, nausea and vomiting are not uncommon.
Treatment
Amphotericin B at 0.7–1 mg/kg/day for 2 weeks, with or without 2 weeks of flucytosine at 100 mg/kg/day in 4 divided doses, followed by fluconazole at 400 mg/day for a minimum of 8–10 weeks.
Initial therapy should be considered successful only after CSF culture is negative for cryptococcal organisms and the patient has had significant clinical improvement.
Initial therapy should be followed with maintenance therapy using fluconazole at 200 mg/day for life.
Amphotericin B
Amphotericin B is useful in treatment of infection with Blastomyces, Coccidioides, Histoplasma, Paracoccidioides, Candida and Cryptococcus, but does have substantial risk of toxicity. It is a ‘polyene’ and works on fungi by binding to ergosterol in the fungal cell membrane, disrupting the membrane and killing the fungus.
Other antifungal agents include fluconazole, which is mainly used for C. albicans infection (and some other susceptible Candida spp. but not C. krusei, and has variable activity against C. glabrata). C. albicansmay acquire resistance, especially with chronic or recurrent treatment in AIDS patients. Fluconazole may be effective against Cryptococcus neoformans meningitis and coccidioidomycosis.
Outpatient parenteral antibiotic therapy
Outpatient parenteral antibiotic therapy (OPAT) has been widely used for the treatment of moderate to serious infections, either as an alternative to hospitalization or following initial hospitalization and early discharge once the patient is over the toxic phase of the infection. A wide range of infections are suitable for OPAT therapy (Table 9.9.2).
Table 9.9.2
Conditions that can be treated on an outpatient basis with parenteral antibiotic therapy
|
AIDS |
Soft-tissue infections |
|
Associated infections |
Cellulitis |
|
Wound infections/abscesses |
|
|
Cardiac |
Bone and joint infections |
|
Endocarditis |
Osteomyelitis |
|
Prosthetic-valve infections |
Septic arthritis Prosthetic infections |
|
Neurological infections |
|
|
Meningitis |
|
|
Genitourinary |
Other infections |
|
Pyelonephritis |
Bacteraemia |
|
Complicated urinary tract infections |
Mastoiditis |
|
Prostatitis |
|
|
Pelvic inflammatory disease |
|
|
Respiratory |
|
|
Pneumonia |
|
|
Lung abscess |
Significant savings, both in terms of direct and indirect costs, are possible utilizing OPAT. Appropriate patient selection is essential for safe and effective outpatient parenteral therapy (Tables 9.9.3 and 9.9.4). Patients should be clinically stable, willing to participate and physically and mentally capable of being treated at home (Table 9.9.4).
Table 9.9.3
Patient selection process
Condition suitable for outpatient therapy
Patient does not fulfil need to admit criteria (see Table 9.9.5)
OR
Patient meets discharge criteria (see Table 9.9.6)
Home environment suitable
Patient/family consent
Table 9.9.4
Patient selection criteria
Able to give consent
Adequate social support at home
The antibiotic(s) chosen is/are appropriate for OPAT use
Patient’s condition is stable
Concurrent illness does not require hospital care
Adequate venous access can be maintained
Patient is mobile
The infection is amenable to outpatient parenteral therapy
Adequate monitoring by the treating medical team is possible
Some patients require initial hospitalization (Table 9.9.5), following which they may be suitable for early discharge to continue treatment at home.
Table 9.9.5
Criteria for admission to hospital
Confused
Persistent high fever
Systolic blood pressure<100 mmHg
Respiratory rate>30/min
Pulse rate>100/min
Requires specialized nursing care assistance with activities of daily living
Hypoxic on room air (PaO2<80 mmHg)
Concurrent illness requiring inpatient care
Personal or social reasons
Pneumonic consolidation in more than one lobe
Once patients comply with predefined discharge criteria (Table 9.9.6), they may be able to be discharged into an outpatient parenteral therapy programme.
Table 9.9.6
Discharge criteria
Medical
Afebrile
Clinical improvement
No specialized nursing care required
Stable
Bacterial pathogens identified
Response to inpatient therapy
Complications unlikely
Social
Parents interested and motivated
Parents capable
Home environment acceptable
Telephone and transport access
Close patient monitoring is essential, with daily reviews by a nurse either by telephone or face to face while patients are in the programme. Patients should be reviewed at least weekly by a physician.
The benefits of OPAT include a reduction in overall costs of patient care through avoidance or reduction in hospitalization, reduction of the costs associated with the hazards of hospitalization and increased patient satisfaction [27,28].
Other issues
The risks associated with bioterrorism need to be taken into account with each and every patient presenting with a febrile illness or signs and symptoms of infection.
Numerous bacterial agents and bacterial toxins have been identified as potential biological agents. Patients presenting in clusters or with unusual or uncommon infections, particularly those that can be used as biological agents, should be quarantined, with staff utilizing post-exposure prophylaxis and strict infection control procedures. It may be necessary to activate the hospital’s Mass Casualty Incident Plan when biological agents are suspected [29].
Recent updates from the medical literature
In August 2012, the CDC announced changes to the 2010 sexually transmitted disease guidelines for gonorrhoea treatment. The Gonococcal Isolate Surveillance Project (GISP) described a decline in cefixime susceptibility among urethral N. gonorrhoeae isolates in the USA during 2006–2011. Because of cefixime’s lower susceptibility, new guidelines were issued that no longer recommend oral cephalosporins for first-line gonococcal infection treatment [30].
Likely developments over the next 5–10 years
The most important challenge regarding infectious disease in the future will be:
![]() The containment of and management of antimicrobial resistance patterns. In part, these patterns have emerged as a result of poor prescribing habits [31].
The containment of and management of antimicrobial resistance patterns. In part, these patterns have emerged as a result of poor prescribing habits [31].
![]() Fewer new antimicrobial drugs are being developed, with the result that with developing resistance patterns there will be very few effective antibiotics available for use against infection [9].
Fewer new antimicrobial drugs are being developed, with the result that with developing resistance patterns there will be very few effective antibiotics available for use against infection [9].
![]() The implementation of prescribing guidelines based on scientific evidence will form the basis of all antibiotic prescribing.
The implementation of prescribing guidelines based on scientific evidence will form the basis of all antibiotic prescribing.
![]() Human behaviour, wildlife trade, factory farming, poor hygiene, global warming and increasing travel will increase the risk of pandemics, evolution and spread of new and old infections [32,33].
Human behaviour, wildlife trade, factory farming, poor hygiene, global warming and increasing travel will increase the risk of pandemics, evolution and spread of new and old infections [32,33].
Detailed descriptions of the drugs described above are available on the Internet by accessing MIMS Online and Antibiotic Guidelines [1].
References
1. Therapeutic Guidelines Limited. Therapeutic Guidelines: Antibiotic. Version 2010;14 Therapeutic Guidelines Limited, Melbourne.
2. Weinstein L, Dalton AC. Host determinants of response to antimicrobial agents. N Engl J Med. 1968;279:467.
3. Moellering Jr RC. Factors influencing the clinical use of antimicrobial agents in elderly patients. Geriatrics. 1978;33:83.
4. Philipson A. The use of antibiotics in pregnancy. J Antimicrobiol Chemother. 1983;12:101.
5. Black A, Francoeur D, Rowe T. SOCG Clinical Practice Guidelines No. 143, Part 2 2004. Canadian Contraception Consensus.
6. Moellering Jr RC. Principles of anti-infective therapy. In: Mandell GL, Bennett JE, Dolin R, eds. Principles and practice of infectious diseases. 4th ed. New York: Churchill Livingstone; 1995;199–212.
7. Welling PG. Effects of food on drug absorption. Annu Rev Nutrit. 1996;16:383–415.
8. Schlichting D, McCollam JS. Recognising and managing severe sepsis: a common and deadly threat. South Med J. 2007;100:594–600.
9. Cephalosporin-resistant CDC. Neisseria gonorhoeae Public Health Response Plan. MMWR. 2012;61:5.
10. Davies J. Inactivation of antibiotics and the dissemination of resistance genes. Science. 1994;264:375–382.
11. Nikaido H. Prevention of drug access to bacterial targets: permeability barriers and active efflux. Science. 1994;264:382–388.
12. Spratt BG. Resistance to antibiotics mediated by target alterations. Science. 1994;264:388–393.
13. Kopecko D. Specialized genetic recombination systems in bacteria: their involvement in gene expression and evolution. Proc Mol Subcell Biol. 1980;7:135–243.
14. Elwell LP, Roberts M, Mayer LW, et al. Plasmid-mediated beta-lactamase production in Neisseria gonorrhoeae. Antimicrobiol Agents Chemother. 1977;11:528–533.
15. Lyon BR, Skurray R. Antimicrobial resistance of Staphylococcus aureus: genetic basis. Microbiol Rev. 1987;5:88–134.
16. Chambers HF, Sande MA. Antimicrobial agents. Goodman and Gilman’s The pharmacological basis of therapeutics 9th ed. New York: McGraw-Hill; 1995; 1029–1032.
17. Stillwell M, Caplan ES. The septic multiple-trauma patient. Infect Dis Clin N Am. 1989;3:155.
18. Kobayashi Y, Takahashi T, Nakae T. Diffusion of beta-lactam antibiotics through liposome membranes containing purified porins. Antimicrobiol Agents Chemother. 1982;2:775–780.
19. Idsoe O, Gothe T, Wilcox RR, et al. Nature and extent of penicillin side reactions with particular reference to fatalities from anaphylactic shock. Bull WHO. 1968;38:159.
20. Pichichero ME. A review of evidence supporting the American Academy of Pediatrics recommendation for prescribing cephalosporin antibiotics for penicillin-allergic patients. Pediatrics. 2005;115:1048–1057.
21. Periti P, Mazzei T, Mini E, Novelli A. Pharmacokinetic drug interactions of macrolides. Clin Pharmacokinet. 1992;23:106–131.
22. Spera RV Jr, Farber BF. Multiply-resistant Enterococcus faecium. The nosocomial pathogen of the 1990s. J Am Med Assoc 1992; 268:2563–64.
23. Lau AH, Lam NP, Piscitelli SC, et al. Clinical pharmacokinetics of metronidazole and other nitroimidazole anti-infectives. Clin Pharmacokinet. 1992;2:328–364.
24. Johnson PJ. Metronidazole and drug resistance. Parasitol Today. 1993;9:183–186.
25. McCalla DR, Reuvers A, Kaiser C. Mode of action of nitrofurantoin. J Bacteriol. 1970;104:1126–1134.
26. Linden PK, Kusne S, Coley K, et al. Use of parenteral colistin for the treatment of serious infection due to antimicrobial-resistant Pseudomonas aeruginosa. Clin Infect Dis. 2003;37:154–160.
27. Tice AD. Outpatient parenteral antibiotic therapy (OPAT) in the United States: delivery models and indications for use. Can J Infect Dis. 2000;11A:45A–48A.
28. Vinen JD. Intravenous antibiotic treatment outside the hospital: safety and health economic aspects. Rev Contemp Pharmacother 1995; 6:435–44, 525.
29. <www.bt.cdc.gov/Agent/agentlist.asp>[Accessed Nov. 2007].
30. Gross EA, Stephens D. Multi-drug resistant bacteria: implications for the emergency physician. Emerg Med Rep 2007;28.
31. WHO World Health Report. A Safer Future.<www.who.com>; 2007.
32. Singer JL, Williams M. Imported infections in pediatric travelers. Emerg Med Rep 2007;28.
33. CDC. 2012 Travellers’ Health: Yellow Book<www.n.cdc.gov/travel/contentYellowBook.aspx>[Accessed Nov. 2012].
9.10 Needlestick injuries and related blood and body fluid exposures
Sean Arendse and Muhammad Shuaib Afzal
Essentials
1 Avoiding blood and other body fluid exposure remains the primary means of preventing occupationally-acquired blood-borne virus infections.
2 The risks of acquiring infection after occupational exposure to blood-borne viruses are: HIV 0.3%, hepatitis B (HBV) 12–30%, hepatitis C (HCV) 1.8%.
3 HBV immunization is an integral part of workplace safety.
4 Effective post-exposure prophylaxis (PEP) is available for both HBV and HIV, but not HCV.
5 Significant emotional distress often complicates needlestick and related occupational injuries.
Introduction
Management of the healthcare worker who sustains an occupational exposure to blood or other potentially infectious body fluids (e.g. semen, vaginal secretions, CSF and fluids containing visible blood) is an important issue for the emergency department (ED) doctor. It is estimated that around 600 000–800 000 needlestick or other percutaneous exposures happen annually in the USA [1]. This figure is a conservative estimate as many needlestick injuries go unreported. HBV, HCV and HIV are the most important occupationally acquired blood-borne pathogens; however, many other organisms, including malaria, syphilis, cytomegalovirus and possibly the prion diseases, such as Creutzfeld–Jakob disease, may also be transmissible via this route.
When evaluating healthcare providers (HCP) at risk for occupational infection with HIV, ‘exposure’ is defined as contact with potentially infectious blood, tissue or body fluids in a manner that allows for possible transmission of HIV and therefore requires consideration of post-exposure prophylaxis (PEP).
Such potentially infectious contacts are:
![]() a percutaneous injury (e.g. a needlestick or cut with a sharp object)
a percutaneous injury (e.g. a needlestick or cut with a sharp object)
![]() contact of mucous membrane or non-intact skin (e.g. exposed skin that is chapped, abraded, or afflicted with dermatitis).
contact of mucous membrane or non-intact skin (e.g. exposed skin that is chapped, abraded, or afflicted with dermatitis).
Body fluids of concern include:
![]() body fluids implicated in the transmission of HIV: blood, semen, vaginal secretions, other body fluids contaminated with visible blood
body fluids implicated in the transmission of HIV: blood, semen, vaginal secretions, other body fluids contaminated with visible blood
![]() potentially infectious body fluids (undetermined risk for transmitting HIV): cerebrospinal, synovial, pleural, peritoneal, pericardial and amniotic fluids.
potentially infectious body fluids (undetermined risk for transmitting HIV): cerebrospinal, synovial, pleural, peritoneal, pericardial and amniotic fluids.
Fluids that are not considered infectious unless they contain blood include faeces, nasal secretions, saliva, gastric secretions, sputum, sweat, tears, urine and vomitus.
In addition, any direct contact (i.e. without barrier protection) to concentrated HIV in a research laboratory or production facility is considered an ‘exposure’ that requires clinical evaluation and consideration of PEP.
Intact skin is an effective barrier against HIV infection and contamination of intact skin with blood or other potentially contaminated fluids is not considered an exposure and does not require PEP.
Most exposures do not result in infection and the risk of infection following significant exposure varies with factors such as:
![]() the pathogen involved (hepatitis B, hepatitis C or HIV)
the pathogen involved (hepatitis B, hepatitis C or HIV)
![]() the fluid involved – blood is generally the most infectious body fluid
the fluid involved – blood is generally the most infectious body fluid
![]() the type of exposure – percutaneous or mucous membrane/non-intact skin
the type of exposure – percutaneous or mucous membrane/non-intact skin
![]() the amount of blood or other infectious body fluid involved in the exposure
the amount of blood or other infectious body fluid involved in the exposure
![]() the amount of virus in the patient’s blood at the time of exposure.
the amount of virus in the patient’s blood at the time of exposure.
General issues
Prevention of needlestick injuries
The old adage ‘prevention is better than cure’ certainly rings true when considering needlestick injuries, as the cost of managing one needlestick injury can range from 376 USD to 2456 USD [2].
The potentially infectious nature of all blood and bodily fluids necessitates the implementation of infection control practices. The universal application of standard precautions should be the minimum level of infection control when treating patients to prevent blood-borne virus transmission. The important elements of standard precautions are:
![]() the use of gloves when contact with blood, body fluids or secretions is anticipated
the use of gloves when contact with blood, body fluids or secretions is anticipated
![]() the use of masks and protective eyewear during procedures that have the potential to generate splashes or sprays of blood or bodily fluids
the use of masks and protective eyewear during procedures that have the potential to generate splashes or sprays of blood or bodily fluids
![]() the use of gowns to protect skin and clothing from soiling by blood and other bodily fluids
the use of gowns to protect skin and clothing from soiling by blood and other bodily fluids
![]() correct handling and disposal of needles and other sharp instruments:
correct handling and disposal of needles and other sharp instruments:
![]() disposal of sharps directly from patient immediately into sharps bins
disposal of sharps directly from patient immediately into sharps bins
![]() locating sharps bins conveniently to reduce the unnecessary transportation of uncapped devices
locating sharps bins conveniently to reduce the unnecessary transportation of uncapped devices
![]() avoiding overfilling sharps containers
avoiding overfilling sharps containers
![]() never re-sheathing or re-capping needles
never re-sheathing or re-capping needles
![]() 100% attention when handling sharps.
100% attention when handling sharps.
More than 50 products with features designed to prevent needlestick injuries are currently available and fall broadly into two categories: those providing ‘passive’ or automatic protection and those with a safety mechanism that the user must activate.
It has been demonstrated that most needlestick injuries are preventable [3] and that use of safety engineered devices reduces needlestick injuries [3,4]. The passive devices are most effective in preventing needlestick injuries [5].
Hospital systems
Hospitals need to have appropriate policies and procedures to deal with occupational exposures to blood and body fluids; these are best implemented through a comprehensive and coordinated occupational exposure programme. Depending on the individual institution, such a programme is usually managed by infection control personnel and also involves staff health, occupational health, laboratory services, the ED and the infectious diseases service.
Staff need to be aware of the appropriate steps to take in the event that they sustain an exposure, such as who to notify, incident reporting requirements and where and how to seek medical evaluation. The programme should develop processes for consent and testing of the source individual (including situations where the individual refuses or is unable to give consent), prompt blood-borne virus testing and communication of results to the exposed person. Clear written guidelines and clinical pathways should be accessible to medical staff involved in managing these exposures (including specific recommendations for exposures involving a blood-borne virus positive source and antiretroviral post-exposure prophylaxis).
Management
The initial management of all needlestick injuries is the same – first aid measures, documentation of the event, determining the status of the source and counselling of exposed worker.
Initial management
Occupational exposure to blood or other potentially infectious body fluids should be considered a medical emergency to ensure timely management. Following exposure, the exposed person should be removed from the area and general first aid measures applied:
![]() for skin exposures – wash the exposed area well with soap and water; if no water is available, use an alcohol-based antiseptic. Other antiseptics, such as iodophors, chloroxylenol (PCMX) and chlorhexidine (CHG) also inactivate HIV
for skin exposures – wash the exposed area well with soap and water; if no water is available, use an alcohol-based antiseptic. Other antiseptics, such as iodophors, chloroxylenol (PCMX) and chlorhexidine (CHG) also inactivate HIV
![]() for eye exposures – remove contact lenses if present and irrigate eyes with copious amounts of water or saline
for eye exposures – remove contact lenses if present and irrigate eyes with copious amounts of water or saline
![]() for oral mucous membrane exposures – spit out contaminating material and rinse the mouth with water several times.
for oral mucous membrane exposures – spit out contaminating material and rinse the mouth with water several times.
Documentation
Clinical information on the source patient for the exposure and the recipient HCP should be documented. This includes risk factors and serological tests for HIV and hepatitis B and C. The nature and time of the exposure should also be described. The exposure should be evaluated and documented on the basis of the definition of exposure given above. All potential exposures to blood or contaminated body fluids as defined above should be promptly evaluated. The following information should be obtained by trained medical personnel:
![]() name and identification of the source
name and identification of the source
![]() time and date of the exposure
time and date of the exposure
![]() nature of the exposure (i.e. non-intact skin, mucosal, or percutaneous exposure, human bite); type of fluid (i.e. blood, blood-contaminated fluid or other contaminated fluid)
nature of the exposure (i.e. non-intact skin, mucosal, or percutaneous exposure, human bite); type of fluid (i.e. blood, blood-contaminated fluid or other contaminated fluid)
![]() body location of the exposure and contact time with the contaminated fluid
body location of the exposure and contact time with the contaminated fluid
![]() infective status of the source (i.e. HIV, HCV, HBsAg), if known, including date of test
infective status of the source (i.e. HIV, HCV, HBsAg), if known, including date of test
![]() when the source is HIV positive, selection of the post-exposure prophylaxis regimen should consider the comparative risk represented by the exposure and information about the exposure source, including history of and response to antiretroviral therapy based on clinical response, CD4 cell counts, viral load measurements and current disease stage
when the source is HIV positive, selection of the post-exposure prophylaxis regimen should consider the comparative risk represented by the exposure and information about the exposure source, including history of and response to antiretroviral therapy based on clinical response, CD4 cell counts, viral load measurements and current disease stage
![]() for percutaneous injuries, a description of the injury (depth of wound, solid versus hollow needle, sharp use in source patient).
for percutaneous injuries, a description of the injury (depth of wound, solid versus hollow needle, sharp use in source patient).
The injured HCP should be questioned about the circumstances of the exposure (activity, time, device type, availability of PPE). The following information should be obtained from the injured person and verified from their medical/occupational health record:
![]() dates of hepatitis B immunizations
dates of hepatitis B immunizations
![]() post-immunization titre, if known
post-immunization titre, if known
![]() previous testing (if available) for HIV, HBV and HCV
previous testing (if available) for HIV, HBV and HCV
![]() tetanus immunization status
tetanus immunization status
![]() current medications
current medications
![]() current or underlying medical conditions that might influence drug selection (e.g. pregnancy, breastfeeding, renal or hepatic disease).
current or underlying medical conditions that might influence drug selection (e.g. pregnancy, breastfeeding, renal or hepatic disease).
Determining status of the source
All source cases should be tested for HBsAg, HCV and HIV, unless the source is known to be infectious. If feasible, a system should be devised to allow HIV test results to be obtained as soon as possible (i.e. within 24 hours). The rapid HIV test should be used to make an initial determination of the source patient’s HIV status and has the advantage that results are available in less than 60 minutes [6]. All positive tests should be confirmed by Western blot. Negative tests do not require confirmation. Determination of HBsAg status should be obtained as soon as possible, but not later than 7 days. Local and state laws regarding consent and counselling prior to HIV testing should be followed.
Clinicians should also be aware of rare case reports where the source patient tested HIV seronegative and was later found to have primary HIV infection [7]; these rare events do not alter guidelines for routine antibody testing but do highlight the importance of testing for HIV RNA if clinically indicated.
Counselling of exposed worker
Risk assessment is particularly important for the HCP to make educated decisions about PEP since the consequences are huge and the stress is extraordinary. They should also be well informed of the benefits and risks of PEP and of the importance of close follow up. Specifically, the following issues should be discussed with the exposed HCP:
![]() The HCP should be informed of the risk associated with the specific exposure experienced.
The HCP should be informed of the risk associated with the specific exposure experienced.
![]() The efficacy and side effects of PEP should be discussed.
The efficacy and side effects of PEP should be discussed.
![]() Risk reduction strategies should be employed to prevent transmission of HIV should the HCP acquire infection. In the event of HIV infection post-exposure, the greatest risk of transmission to other individuals is in the first 6–12 weeks. The exposed HCP should be instructed on condom use or abstinence from sex and refraining from blood, plasma, organ, tissue and semen donation until the 6-month serological test is negative. There is no need to modify a healthcare provider’s patient-care responsibilities after an exposure.
Risk reduction strategies should be employed to prevent transmission of HIV should the HCP acquire infection. In the event of HIV infection post-exposure, the greatest risk of transmission to other individuals is in the first 6–12 weeks. The exposed HCP should be instructed on condom use or abstinence from sex and refraining from blood, plasma, organ, tissue and semen donation until the 6-month serological test is negative. There is no need to modify a healthcare provider’s patient-care responsibilities after an exposure.
![]() Follow up is important to identify HIV infection or adverse effects of the PEP regimen, if administered.
Follow up is important to identify HIV infection or adverse effects of the PEP regimen, if administered.
![]() Specific counselling is warranted for women of childbearing age. The data from an HIV pregnancy registry suggests overall safety of antiretroviral drugs [8]. Temporary discontinuation of breastfeeding following exposure until the 6-month serological test is negative should be considered.
Specific counselling is warranted for women of childbearing age. The data from an HIV pregnancy registry suggests overall safety of antiretroviral drugs [8]. Temporary discontinuation of breastfeeding following exposure until the 6-month serological test is negative should be considered.
Hepatitis B
Hepatitis B vaccination is recommended for all healthcare workers who are involved in direct patient care or who handle human blood or tissues [9], and is an important infection control and occupational health strategy. Healthcare workers should be aware of their HBV immunization status and should undergo antibody testing 4–8 weeks after the last dose of the HBV vaccine to ascertain their immune status.
The risk of acquiring HBV from occupational blood/body fluid exposure from a patient positive for hepatitis B surface antigen (HBsAg) is well recognized and related primarily to the degree of contact with blood and the hepatitis B e-anitgen (HBeAg) status of the source. Following contact with a source positive for HBeAg, the risk of clinical hepatitis is 22–31% and serological evidence of HBV infection develops in 37–62% of exposed, non-immune individuals. In contrast, after exposure to HBeAg-negative blood, there is a 1–6% risk of clinical hepatitis and a 23–37% risk of serological evidence of HBV infection [10]. The average time from exposure to the development of symptoms is 10 weeks (range 4–26 weeks). Routine vaccination against HBV has been recommended for healthcare workers since the early 1980s [11], with a consequent marked reduction in the incidence of infection in this population.
Post-exposure management following an occupational blood/body fluid exposure to HBV requires evaluation of the source’s HBsAg status and the HBV vaccination and vaccine response status of the exposed person [12,13]. HB immunoglobulin (HBIG) is indicated for people who are non-immune (either because of no prior vaccination or because of vaccine non-responsiveness) and are exposed to blood or other infectious body fluids from an HBsAg-positive source. HBIG is prepared from human plasma (screened for blood-borne viruses) known to contain a high titre of antibody to HBsAg (antiHBs). The dose of HBIG is 400 IU, given IM. Concomitantly, HBV vaccination should be injected at a separate site and a full course completed. Table 9.10.1 provides more detailed information about specific indications for HBIG and hepatitis B vaccination following occupational exposures. The exposed person does not need to take any special precautions to prevent secondary transmission [14].
Table 9.10.1
HBV post exposure prophylaxis following occupational exposure

HBsAg: hepatitis B surface antigen; HBIG: hepatitis B immumoglobulin; anti-HBs: antibody to hepatitis B surface antigen.
From Updated US Public Health Service Guidelines for the Management of Occupational Exposures to HBV, HCV, and HIV and Recommendations for Postexposure Prophylaxis. 29 June 2001; 50:RR-11.
There are no apparent risks for adverse effects to developing fetuses when hepatitis B vaccine is administered to pregnant women (CDC, unpublished data, 1990). The vaccine contains non-infectious HBsAg particles which should pose no risk to the fetus. HBV infection during pregnancy might result in severe disease for the mother and chronic infection for the newborn.
Therefore, neither pregnancy nor lactation should be considered a contraindication to vaccination of women. HBIG is also not contraindicated for pregnant or lactating women [15].
Hepatitis C
The risk associated with occupational exposure to hepatitis C following a parenteral injury is estimated to range between 1.8% and 10% [16].
Transmission to healthcare workers has never been documented from skin contamination and rarely from mucous membrane exposure. In contrast to HBV, environmental contamination is not significant [17].
Recommendations for the management of occupational exposures to HCV are aimed at achieving early identification of infection. Recent data suggest that antiviral treatment of acute HCV infection increases rates of HCV clearance [18,19]. If the source HCV antibody test is positive, then polymerase chain reaction (PCR) testing for HCV RNA should be performed. Transmission is much less likely to occur from a source who is PCR negative and the exposed individual can be reassured that the transmission of HCV in this case is negligible. If the source is positive for HCV RNA, a baseline serum from the exposed person is tested for HCV RNA by PCR, anti-HCV antibody testing by enzyme-linked immunosorbent assay (ELISA) and alanine aminotransferase (ALT) with follow-up testing as shown in Table 9.10.2. HCV viraemia can be detected by PCR between 10 days and 6 weeks after infection [20,21].
Table 9.10.2
Serology testing for needle stick injury exposed person

HIV: human immunodeficiency virus, HCV RNA PCR: hepatitis C virus RNA by polymerase chain reaction. ELISA: enzyme linked immunosorbant assay, ALT: alanine amino transferase, AST: aspartate amino transferase, PEP: post exposure prophylaxis.
Currently, there is no proven effective post-exposure prophylaxis for persons exposed to HCV blood or contaminated body fluids. Immunoglobulin (Ig) and antiviral agents are not recommended for post-exposure prophylaxis of HCV [22,23]. Post-exposure prophylactic use of interferon has not been demonstrated to reduce the rate of infection and interferon is associated with many side effects [24,25], but early recognition and antiviral treatment of acute HCV infection in healthcare workers vastly increases viral clearance [18].
Currently, no recommendations exist to restrict professional activities of healthcare workers with HCV infection. As recommended for all healthcare workers, those who are HCV-positive should follow strict aseptic technique and standard precautions, including appropriate use of hand washing, protective barriers and care in the use and disposal of needles and other sharp instruments [14].
Human immunodeficiency virus
The average risk of acquiring human immunodeficiency virus (HIV) infection from all types of reported percutaneous exposure to HIV-infected blood is 0.3% [26]. This is increased for exposures considered as high risk involving:
![]() a deep injury
a deep injury
![]() visible blood on the device causing the injury
visible blood on the device causing the injury
![]() a device previously placed in the artery or vein of the source patient
a device previously placed in the artery or vein of the source patient
![]() a source with terminal AIDS or who has died as a result of AIDS within 60 days of the exposure and thus is presumed to have a high titre of HIV [27].
a source with terminal AIDS or who has died as a result of AIDS within 60 days of the exposure and thus is presumed to have a high titre of HIV [27].
These factors are also probably significant for mucous membrane and skin exposures to HIV-infected blood, where the average risk of HIV transmission is approximately 0.09% and<0.09% respectively [28]. Prolonged or extensive skin contact or visibly compromised skin integrity would also suggest a higher risk.
Recommendations for post-exposure prophylaxis with antiretroviral agents have been guided by a better understanding of the pathogenesis of primary HIV infection, which indicates that HIV infection does not become established immediately; this leaves a brief window of opportunity during which post-exposure antiretroviral intervention might modify or prevent viral replication. An early case-control study demonstrated that use of zidovudine decreased the risk of occupational HIV seroconversion by 81% [29], and it is likely (but not proven) that combination antiretroviral therapy provides even greater protection. Animal data also support the use of antiretroviral prophylaxis after exposure to HIV, provided prophylaxis is administered promptly and for an adequate period. Failures of HIV PEP are well documented with both single drug and combination drug regimens [30].
HIV PEP should be initiated promptly, preferably within 2 hours of the exposure, although it may still be effective for up to 72 hours. Given the complexity of choosing and administering HIV PEP, whenever possible, consultation with an infectious diseases consultant or another physician who has experience with antiretroviral agents is recommended, but it should not delay timely initiation of PEP. There is a slight variability in the selection of drugs by different authorities. For the sake of simplicity and ease of understanding, a three-drug regimen is generally recommended for all high-risk injuries. The preferred PEP regimen is tenofovir+emtricitabine (lamivudine may be used in place of emtricitabine) plus raltegravir. Zidovudine is no longer recommended in the preferred PEP regimen. The recommended duration of PEP is 28 days. A 3–5-day supply of PEP antiretroviral agents (a ‘starter pack’) should be kept in the ED.
This regimen is now the preferred combination because of its excellent tolerability, proven efficacy, fewer side effects and drug–drug interactions and ease of administration. Studies have shown increased rates of adherence and regimen completion when tenofovir+either emtricitabine or lamivudine have been used as components of the PEP regimen [31–37]. Zidovudine is not a ‘must’ inclusion in the newer regimens, as it has no clear advantages in efficacy over tenofovir and significant treatment limiting side effects. Efavirenz should not be used in pregnant women or women of childbearing age. Niverapine, abacavir and didanosine should not be used as PEP because of significant side effects.
Most occupational exposures do not result in transmission of HIV and the potential benefits of PEP need to be carefully weighed against the toxicity of the drugs involved. Nearly 50% of healthcare workers taking HIV PEP experience adverse symptoms (e.g. nausea, malaise, headache, diarrhoea and anorexia) and approximately 33% cease taking drugs because of side effects [38,39]. In some other studies, adherence to PEP has been estimated to be around 40–60% [39–41]. The importance of completing the prescribed regimen needs to be stressed and measures taken to minimize side effects.
The emotional effect of an occupational HIV exposure is substantial [42] and often underestimated. The exposed person may need time off work, short-term use of a night-time sedative or even referral for formal psychological or psychiatric counselling. Patients should be advised of measures to prevent secondary transmission (e.g. safer sexual practices) during the follow-up period, especially the first 6–12 weeks.
Maintaining confidentiality for the staff member sustaining exposure is a priority, as it may have lasting implications both personally and professionally.
The circumstances surrounding the exposure should be reviewed as part of the hospital’s occupational exposure policy and appropriate preventive and educational measures taken if indicated.
Exposures that occur in the community
Blood or body fluid exposures may be sustained in the community, as well as in healthcare settings; examples include needlestick injuries from improperly discarded needles and syringes or blood splashes to the eye or mouth in the course of an altercation. The exposed person may be a member of the public or of an emergency service, such as a policemen or ambulance officer. These exposures are usually managed in the ED.
Although the principles of management are broadly similar to those for occupational exposures, there are some important differences. First, the source is almost never available for testing. (If the source syringe has been retrieved by the exposed person, this should not be tested for blood-borne viruses because such testing is only validated on serum.) Second, needlestick exposures almost always involve old dried blood; this is much less infectious than fresh blood because the viral titre falls with time and dried blood does not pass easily from the lumen of the needle into the exposed person’s subcutaneous tissue. Third, these exposures often provoke a considerable degree of distress in the affected person and there may be considerable pressure from the exposed person, a family member or a colleague to ‘do something’. Some of these incidents even attract media attention.
In Australia, only 1–2% of injecting drug users are HIV infected, so the risk of HIV transmission from a discarded needlestick injury is negligible: e.g. 1:100 (risk source is HIV positive) times 1:300 (risk of HIV transmission after needlestick) times undefined factor to account for old dried blood (say 1:5) – or approximately 1 in 150 000. Similar calculations show a potentially higher risk of hepatitis B and C transmission but, in reality, documented instances of blood-borne virus infection resulting from these community exposures are extremely rare and people should be reassured about this.
In Australia, antiretroviral prophylaxis is not recommended for these exposures unless there are particularly compelling epidemiological circumstances to indicate a high HIV risk in the source.
People not previously vaccinated against HBV should be given HBIG and the first dose of a hepatitis B vaccination course. Despite the low risk of blood-borne virus transmission, many patients feel more reassured if they are offered baseline and follow-up testing. As with exposures in the hospital setting, the attending doctor needs to provide the affected person with information, support and a sympathetic ear!
Provision of antiretroviral prophylaxis following sexual exposures in the community is a highly specialized field and is outside the scope of this chapter; advice should be sought from a doctor with HIV expertise. Interested readers are referred to guidelines produced by the Australian Department of Health and Ageing, available at http://www.ashm.org.au/pep-guidelines/
References
1. Report available at<http://www.healthsystem.virginia.edu/pub/epinet/EPINet2011-NeedlestickRpt.pdf>.
2. O’Malley EM, Scott II RD, Gayle J, et al. Costs of management of occupational exposures to blood and body fluids. Infect Control Hosp Epidemiol. 2007;28:774–782.
3. Cullen BL, Genasi F, Symington I, et al. Potential for reported needlestick injury prevention among healthcare workers through safety device usage and improvement of guideline adherence: expert panel assessment. J Hosp Infect. 2006;63:445.
4. Van der Molen HF, Zwinderman KA, Sluiter JK, et al. Interventions to prevent needle stick injuries among health care workers. Work 2012;41.
5. Tosini W, Ciotti C, Goyer F, et al. Needlestick injury rates according to different types of safety-engineered devices: results of a French multicenter study. Infect Control Hosp Epidemiol. 2010;31:402–407.
6. Landovitz RJ, Currier JS. Clinical practice Postexposure prophylaxis for HIV infection. N Engl J Med. 2009;361:1768.
7. Giulieri S, Schiffer V, Yerly S, et al. The trap: professional exposure to human immunodeficiency virus antibody negative blood with high viral load. Arch Intern Med. 2007;167:2524.
8. Committee, APRS, Antiretroviral Pregnancy Registry International Interim Report for 1 January 1989 through 31 July 2011; 2011.
9. Australian Immunization Handbook, 8th ed. National Health and Medical Research Council, 2003.<http://www.immunise.health.gov.au>.
10. Werner BG, Grady GF. Accidental hepatitis-B-surface-antigen-positive inoculations: use of e antigen to estimate infectivity. Ann Intern Med. 1982;97:367–369.
11. CDC. Recommendation of the Immunization Practices Advisory Commitee (ACIP) inactivated hepatitis B virus vaccine. MMWR. 1982;31:317–328.
12. Grady GF, Lee VA, Prince AM, et al. Hepatitis B immune globulin for accidental exposures among medical personnel; final report of a multicenter controlled trial. J Infect Dis. 1978;138:625–638.
13. Seeff LB, Zimmerman HJ, Wrught EC, et al. A randomized, double blind controlled trial of the efficacy of immune serum globulin for the prevention of post-transfusion hepatitis: a veterans administation cooperative study. Gastroenterology. 1977;72:111–121.
14. Updated US Public Health Service guidelines for the management of occupational exposures to HBV, HCV, and HIV and recommendations for postexposure prophylaxis. 29 June 2001;50:RR-11.
15. HIV/Viral hepatitis – a guide for primary care. Australian society for HIV medicine inc. 2001.
16. Polish LB, Tong MJ, Co RL. Risk factors for hepatitis C virus infection among health care personnel in a community hospital. Am J Infect Control. 1993;21:196–200.
17. Jaeckel E, Cornberg M, Wedemeyer H, et al. Treatment of acute hepatitis C with interferon alpha-2b. N Engl J Med. 2001;345:1452–1457.
18. Gerberding JL. Clinical practice Occupational exposure to HIV in health care settings. N Engl J Med. 2003;348:826.
19. Zaaijer HL, Cuypers HT, Reesink HW, et al. Reliability of polymerase chain reaction for detection of hepatitis C virus infection. Lancet. 1993;341:722–724.
20. Wang TY, Kuo HT, Chen LC, et al. Use of polymerase chain reaction for early detection and management of hepatitis C virus infection after needlestick injury. Ann Clin Lab Sci. 2002;32:137.
21. Cleveland JL, Cardo DM. Occupational exposures to human immunodeficiency virus, hepatitis B virus, and hepatitis C virus: risk, prevention, and management. Dent Clin N Am. 2003;47:681.
22. Beekmann SE, Henderson DK. Protection of healthcare workers from bloodborne pathogens. Curr Opin Infect Dis. 2005;18:331.
23. Chung H, Kudo M, Kumada T, et al. Risk of HCV transmission after needlestick injury, and the efficacy of short-duration interferon administration to prevent HCV transmission to medical personnel. J Gastroenterol. 2003;38:877.
24. Corey KE, Servoss JC, Casson DR, et al. Pilot study of postexposure prophylaxis for hepatitis C virus in healthcare workers. Infect Control Hosp Epidemiol. 2009;30:1000.
25. CDC. Case-control study of HIV seroconversion in health care workers after percutaneous exposure to HIV-infected blood – France, United Kingdom and United States, Jan. 1988–Aug. 1994. MMWR 1995;44:929–33.
26. Bell DM. Occupational risk of human immunodeficiency virus infection in health-care workers: an overview. Am J Med. 1997;102:9–15.
27. Gerberding JL. Management of occupational exposure to blood borne viruses. N Engl J Med. 1995;332:444–551.
28. Cardo DM, Culver DH, Ciesielski CA, et al. A case-control study of HIV seroconversion in health care workers after percutaneous exposure. N Engl J Med. 1997;337:1485–1490.
29. Jochimsen EM. Failures of zidovudine postexposure prophylaxis. Am J Med. 1997;102(suppl 5B):52–55.
30. Mayer KH, Mimiaga MJ, Cohen D, et al. Tenofovir DF plus lamivudine or emtricitabine for nonoccupational postexposure prophylaxis (nPEP) in a Boston Community Health Center. J Acquir Immune Defic Syndr. 2008;47:494–499.
31. Tosini W, Muller P, Prazuck T, et al. Tolerability of HIV postexposure prophylaxis with tenofovir/emtricitabine and lopinavir/ritonavir tablet formulation. AIDS. 2010;24:2375–2380.
32. Mayer K, Mimiaga M, Gelman M, et al. Tenofovir DF/emtricitabine/raltegravir appears safe and well-tolerated for non-occupational post-exposure prophylaxis. Presented at fifth IAS Conference on HIV Pathogenesis, Treatment, and Prevention. 19–22 July 2009. Cape Town, South Africa. Abstract WEAC104.
33. Mayer KH, Mimiaga MJ, Gelman M, et al. Raltegravir, tenofovir DF, and emtricitabine for postexposure prophylaxis to prevent the sexual transmission of HIV: safety, tolerability, and adherence. J Acquir Immune Defic Syndr. 2012;59:354–359.
34. Grant RM, Lama JR, Anderson PL, et al. Preexposure chemoprophylaxis for HIV prevention in men who have sex with men. N Engl J Med. 2010;363:2587–2599.
35. Baeten J. Antiretroviral pre-exposure prophylaxis for HIV-1 prevention among heterosexual African men and women: the partners PrEP study. HIV-1-infected adults. 6th IAS Conference on HIV Pathogenesis, Treatment and Prevention. 17–20 July 2011. Rome. Abstract MOAX0106.
36. Thigpen MC, Kebaabetswe PM, Smith DK, et al. Daily oral antiretroviral use for the prevention of HIV infection in heterosexually active young adults in Botswana: results from the TDF2 study. HIV-1-infected adults. Presented at 6th IAS Conference on HIV Pathogenesis, Treatment and Prevention. 17–20 July 2011. Rome. Abstract WELBC01.
37. Wang SA, Panlilio AL, Doi PA, et al. Experience of healthcare workers taking postexposure prophylaxis after occupational HIV exposure: findings of the HIV postexposure prophylaxis registry. Infect Control Hosp Epidemiol. 2000;21:780–785.
38. Parkin JM, Murphy M, Anderson J, et al. Tolerability and side effects of post-exposure prophylaxis for HIV infection. Lancet. 2000;335:722–723.
39. Day S, Mears A, Bond K, et al. Post-exposure HIV prophylaxis following sexual exposure: a retrospective audit against recent draft BASHH guidance. Sex Trasm Infect. 2006;82:236–237.
40. Lunding S, Katzenstein TL, Kronborg G, et al. The Danish PEP registry: experience with the use of postexposure prophylaxis (PEP) following sexual exposure to HIV from 1998–2006. Sex Transm Dis. 2010;37:49–52.
41. Armstrong K, Gordon R, Santorella G. Occupational exposures of health care workers (HCWs) to human immunodeficiency virus (HIV): stress reactions and counseling interventions. Social Work Health Care. 1995;21:61–80.
42. Henry K, Campbell S, Jackson B, et al. Long-term follow-up of health care workers with work-site exposure to human immunodeficiency virus. J Am Med Assoc. 1990;236:1765.
9.11 Tropical infectious diseases
Sander Manders
Essentials
1 Tropical diseases are a major cause of morbidity and mortality worldwide.
2 Due to climate change and increasing population mobility (migration and travel), health practitioners in non-tropical areas will increasingly need to diagnose and treat tropical diseases.
3 A significant proportion of northern Australia has a tropical climate and several tropical diseases occur in this area. Vigilant public health surveillance, case tracking and vector control are instrumental in controlling incursions of non-endemic tropical diseases into Australia.
4 Indigenous Australians are disproportionally affected by infections in tropical Australia.
5 Within tropical areas, the aetiological spectrum of common diseases is different from that in temperate areas. This is due to different local prevalences of common pathogens, as well as the existence of specific tropical agents. Knowledge of local protocols is important in choosing appropriate antibiotic cover for the treatment of common diseases in the tropics.
6 In travellers who have returned from the tropics and present to the emergency department, common infections not specific to the tropics should not be forgotten as likely causes. A good history and systematic approach may aid in the correct identification of tropical diseases. Expert consultation may be of great benefit and public health notification is essential.
Introduction
Tropical diseases cause an enormous burden of disease worldwide and many other diseases that are not specific to the tropics disproportionally affect people in developing countries.
Returned travellers or migrants may present to health practitioners with signs and symptoms of tropical diseases. Due to climate change and increasing population mobility (travel and migration), health practitioners in non-tropical areas will increasingly need to diagnose and treat tropical diseases. A high index of suspicion, a systematic approach and expert consultation contribute to the appropriate investigation and management of these cases.
Significant areas of northern Australia have a tropical climate, including the Top End (around Darwin), Far North Queensland (north of Cairns) and the Kimberley (in Western Australia). Several tropical diseases are endemic there, with others occurring only infrequently. Indigenous Australians are disproportionally affected by both tropical and non-tropical disease.
Diseases common to temperate climates also occur in the tropics and it is important to recognize that these may have different aetiologies there. Community-acquired pneumonia in tropical Australia, for instance, is most commonly caused by S. pneumoniae but, in severe cases, organisms such as Burkholderia pseudomallei and Acinetobacter baumannii should also be covered. Cryptococcus gattii should be considered in meningitis or subacute pneumonia. In undifferentiated sepsis, melioidosis is an important differential diagnosis. Knowledge of protocols, based on specific local circumstances, is important.
Vigilant public health systems are in place to help prevent spreading disease from endemic areas into non-endemic areas. Many of the diseases discussed in this chapter are notifiable in both Australia and New Zealand. An appropriate public health response may include case surveillance, contact tracing and vector control.
Parasitic tropical diseases
Malaria
Introduction and epidemiology
Malaria is often considered the most important tropical disease worldwide. Half of the world’s population is at risk, with over 200 million cases annually. An estimated 655 000 deaths occur each year, of which 90% are in subsaharan Africa [1]. A substantial number of malaria infections occur in South America, Southeast Asia and the Pacific.
In Australia, malaria was only officially considered eradicated in 1981 and there are ongoing concerns regarding the potential re-establishment of the disease due to the widespread presence of appropriate vectors and geographic proximity to endemic areas (particularly Indonesia and Papua New Guinea). An average of 600 cases are notified in Australia each year in travellers and migrants.
Malaria is caused by the protozoan parasite Plasmodium, of which six species are currently known to infect humans (Table 9.11.1). They have a complex life cycle and are transmitted by Anopheles mosquitoes, which bite from dusk to dawn. Less commonly, malaria can also be transmitted vertically. The parasites enter the blood and spread to the liver, where they replicate and are periodically released back into the bloodstream and then invade red blood cells.
Table 9.11.1
Plasmodium species that cause malaria in humans
|
Plasmodium species |
Area |
Notes |
|
P. falciparum (≈75%) |
Africa, South America, Southeast Asia |
Responsible for most severe cases and deaths |
|
P. malariae (≈20%) |
Africa, Southeast Asia, Pacific, South America |
Quartan malaria |
|
P. ovale curtisi |
West Africa, Southeast Asia |
Two subspecies of P. ovale have recently been described [3] |
|
P. ovale wallikeri |
West Africa, Southeast Asia |
|
|
P. vivax |
USA, South America, Asia, Africa |
Relatively benign |
|
P. knowlesi |
Southeast Asia (Malaysia) |
Can cause severe cases; macaques are reservoir |
The majority of malaria cases are caused by P. falciparum, which is the most severe and lethal form. Groups at particular risk include young children, pregnant women, immunocompromised patients (including those with HIV/AIDS) and travellers (due to a lack of immunity). Conversely, some genetic red blood cell variations, including sickle cell trait, thalassaemia trait, G6PD deficiency [2] and Melanesian ovalocytosis, provide some resistance against malaria.
Prevention
A large number of national and international organizations are involved in malaria prevention. Measures include vector control programmes, indoor residual spraying, insecticide-treated nets and intermittent preventative treatment for pregnant women. Travellers to endemic areas should use appropriate chemoprophylaxis, tailored to the locally occurring Plasmodium species and drug resistance patterns and avoid mosquito exposure. Efforts to develop a malaria vaccine are ongoing.
Clinical features
The incubation period is typically 10 days to 4 weeks, but can be longer. Mild cases of acute malaria are characterized by paroxysmal fevers, caused by periodic parasitaemia. Rigors herald 6–10 hours of high fever (>40°C), after which a relatively asymptomatic period follows. The rigors recur after approximately 40 hours (‘tertian’ fever) with P. vivax and P. ovale or approximately 64 hours (‘quartan fever’) with P. malariae. In P. falciparum malaria, fevers are less predictable and may be continuous. Additionally, there may be flu-like symptoms, diarrhoea, and mild jaundice.
Chronic malaria occurs when low-level parasitaemia persists, causing recurrent attacks and anaemia, hepatosplenomegaly and increased susceptibility to other infections. Secondary complications include massive splenomegaly, malarial nephropathy and Burkitt’s lymphoma.
Severe malaria is almost exclusively caused by P. falciparum. Important features have been summarized in Table 9.11.2. The WHO has published case definitions for severe malaria [4]. The prognosis is poor, especially in children. Non-falciparum malaria is usually more benign, but death due to splenic rupture can occur.
Table 9.11.2
Features of severe malaria [5]

ARDS: acute respiratory distress syndrome; RBC: red blood cell.
After Trampuz A, Jereb M, Muzlovic I, Prabhu RM. Clinical review: severe malaria. Critical Care 2003;7:315–23.
Diagnosis
Clinical findings are of limited utility in diagnosing malaria [6]; microscopic examination of thick and thin blood smears [7] remains essential. A thick smear (drop of blood on a slide) is used to detect the presence of parasites and a thin smear (drop of blood spread thin on a slide) may help to identify the Plasmodium species. One negative smear does not exclude malaria and usually three sets are obtained at 12–24 hour intervals. Rapid dipstick immunoassay tests exist, but can be false negative with low or very high levels of parasitaemia. Plasmodium-specific polymerase chain reaction (PCR) tests are sensitive and specific but not widely available in endemic areas. Many other laboratory abnormalities, such as thrombocytopaenia and hyperbilirubinaemia, can be seen in malaria, but these are not specific enough to make the diagnosis. Before the diagnosis of cerebral malaria can be made, bacterial meningitis should be ruled out by lumbar puncture.
Treatment
Early treatment reduces morbidity, mortality and malaria transmission. Emerging resistance to antimalarial drugs (chloroquine and sulfadoxine-pyrimethamine) is a recurring problem worldwide. Artemisinin, a compound derived from wormwood, combined with another agent (artemisinin combination therapy) is the best currently available treatment for falciparum malaria [8,9]. It is given orally for uncomplicated cases and intravenously for severe cases. Various regimens are available to treat other Plasmodium species. For travellers diagnosed with malaria, different drugs should be used for treatment than were taken for prophylaxis. Initial hospitalization with infectious disease specialist consultation is recommended for all cases.
Schistosomiasis (bilharzia)
This parasitic disease affects more than 200 million people worldwide, with more than 90% of infections occurring in Africa. Its global impact is second only to malaria, with an estimated 200 000 deaths per year and significant chronic morbidity in survivors.
Infected fresh water snails release free-swimming larvae (cercaria) into surface waters, which can penetrate the skin of humans who come into contact with the water. Schistosomula then circulate in the blood and replicate in the portal vessels. Subsequently, they migrate to blood vessels in other parts of the body and release their eggs, some of which are shed in human faeces and end up back in the surface waters. The eggs hatch in the water and produce miracidia, which enter suitable fresh water snails. After multiplying inside the snail, cercaria are released into the water, awaiting a new human host. The species of Schistosomaresponsible for human infections are listed in Table 9.11.3; mixed infections occur.
Table 9.11.3
Schistosoma species that infect humans
|
S. mansoni |
Latin America, Africa, Middle East |
|
S. haematobium |
Africa, Middle East, Turkey, India |
|
S. japonicum |
East Asia, Pacific |
|
S. intercalatum (≈1%) |
Sub-Saharan Africa |
|
S. mekongi (<1%) |
Cambodia, Laos (Mekong river basin) |
Acute infections are more likely to cause symptoms among non-residents of endemic areas. A pruritic rash in response to cercariae entering the skin (swimmers’ itch) can occur within a day, usually subsiding within 10 days. Acute toxaemic schistosomiasis (Katayama fever) is an uncommon but often severe seroconversion illness which may occur 1–3 months after the primary infection. Symptoms include fever, malaise, urticaria, cough, diarrhoea, hepatosplenomegaly and lymphadenopathy. It may last several weeks.
In chronic infection, the parasites migrate to species-specific areas in the host body, where their eggs induce a localized inflammatory response with fibrosis. This causes a high burden of disease [10–12]; common symptoms are listed in Table 9.11.4.
Table 9.11.4
Symptoms of chronic schistosomiasis

Prevention includes improving sewage management and personal protection, such as rubber boots. Fresh water exposure should be avoided where possible. A vaccine is under development.
The diagnosis is made by a history of fresh water exposure and the demonstration of eggs in the urine or faeces. During Katayama fever, no eggs may yet be seen. Serological tests are available. Abdominal X-rays may show bladder calcification in chronic genitourinary schistosomiasis.
Praziquantel (40–60 mg/kg in two divided doses) is an effective treatment but, due to high rate of re-infection, it may be difficult to achieve a cure in endemic areas. During Katayama fever, prednisone may be given to suppress the acute reaction and a repeat dose of praziquantel is recommended after 1–2 months. Community treatment programmes exist in endemic areas.
Leishmaniasis
Various species of Leishmania protozoa occur in South America, Africa, the Middle East and India, but also in southern Europe [13]. They are transmitted by sandflies from human and canine reservoirs. Preventative measures include DEET-containing insect repellents, covering exposed skin and insecticide spraying inside houses. Sandflies are so small they will pass through the mazes of bed nets that have not been treated with insecticide. Insecticide-impregnated dog collars have been shown to be effective.
Leishmania infections can have cutaneous or systemic manifestations, depending on parasite species and host factors; it can remain asymptomatic. HIV co-infection predisposes to severe or recurrent disease. The incubation period is usually 1–2 weeks to 6 months, but there can be a latent period of up to 3 years.
Cutaneous leishmaniasis manifests with skin ulcers, which are usually painless unless a secondary bacterial infection occurs. Most lesions heal spontaneously over a few months, leaving a scar. A mucocutaneous form of the disease causes destruction of the mucous membranes of the nose, mouth, throat and surrounding tissues and can occasionally be fatal.
Visceral leishmaniasis (also known as kala-azar) manifests as fever with rigors, malaise, anorexia, lymphadenopathy and non-tender hepatosplenomegaly. Malnutrition and anaemia occur as the disease becomes chronic. The mortality is very high within 2 years if the disease remains untreated, although milder chronic forms occur.
The diagnosis can be confirmed by microscopy, culture or PCR. Treatment of leishmaniasis varies by clinical manifestation and geographic region; options include amphotericin-B, pentavalent antimony-containing preparations and several other drugs.
Post-kala-azar dermal leishmaniasis (PKDL) can occur several months to years after recovery from visceral leishmaniasis and consists of maculopapular lesions which spread from around the mouth. It typically disappears within a year without treatment, but may require several months of treatment in some regions. PKDL patients can be long-term reservoirs of infection.
Trypanosomiasis
American trypanosomiasis (Chagas’ disease)
A major public health concern in Latin and South America, Chagas’ disease is caused by the flagellate protozoan Trypanosoma cruzi. It is spread to humans and other mammals by the faeces of insects of the Triatominae subfamily (‘kissing bugs’). Additionally, it can be spread vertically or by the administration of blood products. Prevention focuses on vector control, including the improvement of housing conditions and the use of insecticides and mosquito nets. Blood products and organ donors in the Americas are screened for T. cruzi.
The acute phase of the infection may have no or non-specific flu-like symptoms, but it can be fatal in children. Swelling around the site of inoculation in the face or around the eye (Romaña’s sign) is well described. The infection becomes asymptomatic within approximately 2 months. In the chronic phase, the parasites invade the myocardium and intestinal smooth muscle. The development of cardiomyopathy leads to congestive heart failure and arrhythmias and is fatal in 30%. Dilatation of the oesophagus and colon (10%) and neurological involvement may also occur.
The diagnosis can be made by direct visualization of the parasites in blood smears or serological testing. Benznidazole and nifurtimox are effective treatments if given soon after the infection occurs. Treatment for chronic infections is difficult and side effects are common. Supportive treatment for cardiac and gastrointestinal complications is important.
African trypanosomiasis (sleeping sickness)
This disease of sub-Saharan Africa is caused by Trypanosoma brucei, which is spread by bites of the tsetse fly. Several major epidemics have occurred in the last century and vector control programmes have been successful in reducing the number of cases reported.
Approximately 95% of cases are caused by Trypanosoma brucei gambiense (West and Central Africa). A chancre may develop at the site of inoculation, followed by an asymptomatic stage which can last months to years. Symptomatic infection then begins with the haemolymphatic stage, characterized by fever, arthralgias and pruritus. Posterior cervical lymphadenopathy (Winterbottom’s sign) is common. The neurological stage begins when the trypanosomes invade the central nervous system, causing headaches, personality changes, psychosis and focal motor, extrapyramidal and/or cerebellar signs. The final stages of the disease are characterized by daytime somnolence, seizures, coma and death.
A second type of the disease is caused by T. b. rhodesiense (5%) which occurs in southeastern Africa. Its course is more fulminant, with early multiple organ failure and death.
The diagnosis can be made by direct microscopic observation of the trypanosomes. Several serological screening tests (card agglutination trypanosoma test) exist for Gambian trypanosomiasis. The treatment is complex and depends on parasite subtype, regional drug resistance and the stage of the disease.
Filariasis
This variable disease is caused by a number of helminth species (worms) which occur throughout the (sub)tropics and are spread by mosquitoes and black flies. Lymphatic filariasis is the most common form; the worms develop in the lymphatic system and cause lymphoedema. Elephantiasis is the most extreme manifestation of this disease. Subcutaneous filariasis is caused by different species of helminths, producing a rash and arthritis. Onchocerca volvulus inhabits the eyes and is the world’s second cause of blindness (‘river blindness’). The diagnosis can be made with thick and thin blood smears, obtained on a species-specific time of the day or by PCR. Treatment is with diethyl-carbamazine or ivermectin and albendazole; sequelae often remain chronic.
Gastrointestinal parasites
A variety of gastrointestinal infections are prevalent throughout the tropics (Table 9.11.5); they represent a major cause of morbidity and childhood mortality. Most are transmitted by the faecal–oral route and prevention therefore includes improving sanitation and access to safe drinking water. The most important feature of management is appropriate oral or intravenous rehydration; specific antimicrobial therapy is secondary.
Table 9.11.5
Common gastrointestinal pathogens in tropical areas
|
Parasites |
Bacteria |
Viruses |
|
Protozoa |
Salmonella |
Rotavirus |
|
Giardia lamblia |
Shigella |
Norovirus |
|
Entamoeba histolytica |
Yersinia enterocolitica |
Adenovirus |
|
Cryptosporidium parvum, C. hominis |
Campylobacter jejuni |
Astrovirus |
|
Eschericia coli (enterotoxigenic) |
||
|
Helminths |
Staphylococcus aureus |
|
|
Ascaris lumbricoides (roundworm) |
Clostridium difficile |
|
|
Hookworms |
Clostridium botulinum |
|
|
Ancylostoma duodenale |
Vibrio cholerae |
|
|
Necator americanus |
||
|
Trichuris trichuria (whipworm) |
||
|
Enterobius vermicularis (threadworm) |
||
|
Strongyloides stercoralis (pinworm) |
In Australia, Strongyloides is sometimes called ‘roundworm’; In American English, ‘pinworm’ refers to Enterobius and ‘threadworm’ to Strongyloides.
Protozoa
Giardia lamblia is a protozoan parasite with worldwide distribution, including Australia and New Zealand. It can survive for a long time in freshwater lakes and streams contaminated with animal or human faeces. Mild infections can be asymptomatic but it often causes foul-smelling, loose stools which may become fatty and float on water. It is usually self-limiting (7–10 days) but can become more chronic and contribute to malnutrition. The diagnosis is made by microscopy and treatment is with oral metronidazole (30 mg/kg up to 2 g daily, for 3 days) or tinidazole (50 mg/kg up to 2 g, as a single dose).
Entamoeba histolytica infection is often asymptomatic or causes only mild diarrhoea, but it may lead to severe diarrhoea with mucus, pus and blood in the stools (dysentery). Complications include peritonitis from intestinal perforation and amoebal liver abscesses. Microscopy often needs to be repeated to make the diagnosis. Supportive treatment is important; the specific treatment is oral metronidazole (15 mg/kg up to 600 mg, 8-hourly, for 7–10 days) or tinidazole (50 mg/kg up to 2 g daily, for 3 days).
Several species of Cryptosporidium occur worldwide (including in Australia and New Zealand), causing self-limiting watery diarrhoea. Special microscopic techniques are required to make the diagnosis. Specific treatment is only required in immunocompromised patients (particularly those with AIDS) as it may become severe, even life threatening in this group.
Helminths (worms)
Soil-transmitted helminths infect humans when their eggs are ingested (Ascaris, Trichuris) or by active penetration of the skin by larvae (hookworms, Strongyloides). Most helminthic infections cause chronic abdominal discomfort without significant diarrhoea. Complications include intestinal obstruction (Ascaris), chronic diarrhoea (Trichuris) and iron deficiency anaemia (hookworms). The eggs and larvae of these species can be distinguished by microscopy. Benzimidazoles (albendazole, mebendazole) as a single dose or short course are effective treatment.
Strongyloides occurs worldwide, but it is hyperendemic in rural and remote indigenous communities in northern Australia with a reported prevalence of up to 60%. Due to a cycle of auto-infection, Strongyloidesinfection can be lifelong if untreated. Clinical features of strongyloidiasis are summarized in Table 9.11.6. Immunocompromised patients, including those given corticosteroids, may develop disseminated strongyloidiasis, which carries a high mortality. The diagnosis can be made by detection of larvae in stool or serology in specialized laboratories. Treatment is with ivermectin (200 μg/kg as a single dose) or albendazole (400 mg daily for 3 days); some authors advocate repeat treatments.
Table 9.11.6
Clinical features of strongyloidiasis [14,15]

After Adams M, Page W, Speare R. Strongyloidiasis: an issue in Aboriginal communities. Rural and Remote Health 2003; 3 (online); Johnston FH, Morris PS, Spears R, et al. Strongyloidiasis: a review of the evidence for Australian practitioners. Aust J Rural Health 2005;13: 247–54.
Viral tropical diseases
Many tropical viral infections are arthropod- borne (arboviruses), with flaviviruses being the most important subgroup (Table 9.11.7). Alphaviruses also cause some important infections in tropical and temperate areas.
Table 9.11.7
Major viral tropical diseases

*Commonly used abbreviation to indicate the causative virus. PNG: Papua New Guinea.
Yellow fever
In acute yellow fever, a flu-like syndrome develops after 3–6 days’ incubation and improves after another 3–4 days. In 15% of cases, a ‘toxic phase’ then develops, with high fever, liver dysfunction causing jaundice and haemorrhage, and renal impairment. Mortality in this group is approximately 50%; survivors recover without significant sequelae. There is no specific treatment, but an effective vaccine exists. Evidence of vaccination is required by health authorities in many countries for travellers returning from endemic areas.
Dengue
Four distinct serotypes of the dengue virus occur in most tropical areas of the world; most cases occur in Southeast Asia. In Australia, occasional outbreaks occur, mostly in Far North Queensland. Most cases remain asymptomatic. Dengue fever may develop after 3–14 days’ incubation; it lasts for 2–7 days. Arthralgias are often severe. The rash resembles that of measles. The most feared complication is dengue haemorrhagic fever (DHF), which may develop if a subsequent infection with another dengue serotype occurs. Features are summarized in Table 9.11.8. Laboratory diagnosis can be made with PCR testing. Supportive management with adequate hydration and analgesia is important; no specific therapy exists. A vaccine against all four serotypes is in an advanced stage of development [16].
Table 9.11.8
WHO case definitions for dengue

Arboviral encephalitis
Various arboviruses cause encephalitis and, while the vast majority of infections remain asymptomatic, potentially devastating sequelae occur in symptomatic patients. Clinical symptoms do not reliably differentiate between various arboviral causes of encephalitis; a definitive diagnosis can be obtained by serological testing.
In recent years, the West Nile virus (WNV) has been successful in extending its range into temperate areas including North America, Europe and Australia. Kunjin virus is a subtype of WNV endemic to Australia and Papua New Guinea [17]. The Murray Valley encephalitis virus is also endemic to northern Australia and Papua New Guinea and epidemics in the southern states of Australia have been well described [18]. Japanese encephalitis virus sporadically occurs on the Torres Strait Islands and in the northernmost tip of Queensland.
When symptomatic, these viruses may cause fever with a flu-like syndrome, rash, meningeal signs, convulsions and decreased level of consciousness. Encephalitis, meningitis or a poliomyelitis-like illness with flaccid paralysis have all been described. Disease progression is variable, ranging from full recovery to death. Long-term neuropsychiatric sequelae occur in a large proportion of survivors. Treatment is supportive. A vaccine exists for the Japanese encephalitis virus.
Alphaviruses
Several thousand cases of Ross River virus (RRV) and Barmah Forest virus (BFV) infections occur annually in Australia. They cause flu-like symptoms with arthralgia and a widespread maculopapular rash in 50% of cases. Arthritis, myalgia and fatigue can last for 6 months or longer. IgM serological tests for RRV or BFV may be false positive in patients with other infections such as malaria and dengue. The closely related chikungunya virus occurs in Africa and Southeast Asia and causes similar symptoms. There is no specific treatment.
Viral haemorrhagic fevers
Several families of viruses can cause fever with haemorrhagic diathesis (Table 9.11.9). They are carried by vectors, but person-to-person spread is mostly responsible for outbreaks. Dengue, yellow fever, and certain other flaviviruses occasionally cause haemorrhagic fever as well.
Table 9.11.9
Important viral haemorrhagic fevers

The clinical presentation is variable, but usually includes a flu-like prodrome with respiratory and sometimes central nervous system symptoms. The disease progresses to multiple organ failure with disseminated intravascular coagulopathy. Case fatality rates are high, up to 90%. Treatment is supportive, with no specific antiviral agents available. Vaccine research is ongoing.
Viral hepatitis
A high incidence of hepatitis A and B (HAV, HBV) occurs in developing countries. HAV is spread via the faecal–oral route, whereas HBV is spread via contaminated blood and other bodily fluids. Vaccinations are recommended for travellers to endemic areas.
The hepatitis C virus (HCV) is a non-arthropod-borne flavivirus. Transmission via non-sterilized medical equipment is a concern in certain countries and recommendations for travellers may include carrying their own needles; no vaccine is available.
Hepatitis E (HEV) causes a self-limiting disease similar to hepatitis A, except during pregnancy when fatal hepatitis is well documented.
Bacterial tropical diseases
Tuberculosis
Introduction
Approximately one-third of the world’s human population is infected by Mycobacterium tuberculosis, making it a major worldwide public health concern. Tuberculosis is by no means exclusively a tropical disease, but it disproportionally affects people in developing countries. Risk factors include HIV infection and other causes of immune suppression, diabetes, pulmonary disease and malnutrition. It is transmitted via inhalation of droplets produced by a coughing person with tuberculosis. Mortality if untreated is around 50%.
Screening and prevention
For screening purposes, the Mantoux (tuberculin) skin test and interferon-gamma release assays (IGRA) are commonly used. When positive, these tests indicate prior exposure to tuberculosis but do not prove active disease. False negatives and false positives are a concern. The Bacillus Calmette–Guérin (BCG) vaccination offers some protection to people at high risk of contracting tuberculosis. Some controversy remains over the validity of the Mantoux test after BCG vaccination.
Symptoms
Tuberculosis can be asymptomatic, but may produce non-specific symptoms including fever, night sweats, anorexia and cachexia.
The most common presentation is pulmonary tuberculosis, which causes a productive cough, haemoptysis, chest pain and dyspnoea. An exudative pleural effusion may occur. Chronic complications include bronchiectasis and pulmonary fibrosis.
Non-pulmonary tuberculosis can occur in virtually any part of the body, including lymph nodes, bones, meninges, pericardium, abdomen and genitourinary tract. Miliary tuberculosis is an aggressive form of haematogenously disseminated tuberculosis which occurs in infants and immunocompromised patients.
Latent tuberculosis occurs when mycobacteria persist intracellularly; patients are asymptomatic and not infectious. The disease can reactivate later, for example, when the patient becomes immunocompromised.
Diagnosis
Pulmonary tuberculosis may be suggested by chest X-ray appearance (Table 9.11.10). Mycobacteria can be demonstrated on acid-fast (Ziehl–Neelsen) staining of sputum or broncho-alveolar lavage fluid. Culture confirms the diagnosis by identifying the species of mycobacteria and also enables drug susceptibility testing. For non-pulmonary tuberculosis, samples appropriate to the site should be obtained and tested [19].
Table 9.11.10
Chest X-ray findings associated with pulmonary tuberculosis
Consolidation
Hilar lymphadenopathy
Ghon focus (calcified nodule that remains after resolution of initial consolidation)
Cavitating lesions
Fibrosis (dominant in upper lobes)
Calcifications
Miliary pattern (small nodules throughout lungs)
Tuberculoma (well-defined tuberculosis mass)
Management
Within an emergency department or other hospital setting, patients suspected to have infective tuberculosis should be in a negative-pressure room; staff and visitors should wear appropriate N95 face-masks (aerosol precautions).
Treatment of any form of tuberculosis requires expert consultation. Public health reporting with appropriate contact tracing is essential. A 6-month treatment regimen with four drugs initially (‘HRZE’, i.e. isoniazid, rifampicin, pyrazinamide, ethambutol) is often used to treat uncomplicated tuberculosis. Emerging multidrug resistance is of increasing concern worldwide [20,21].
Melioidosis
Introduction
The Gram-negative bacterium Burkholderia pseudomallei is the cause of melioidosis [22]. It is found throughout Southeast Asia and India and is highly endemic in northeast Thailand, Malaysia, Singapore and northern Australia (with sporadic cases seen further south). The bacteria live in the soil during the dry season but can be found in surface water and mud after heavy rainfall; they may also become airborne. Transmission occurs through the skin (cuts and sores), inhaled airborne dust or droplets and, rarely, through ingestion of contaminated water. Person-to-person transmission is extremely rare.
The most important risk factors are diabetes, renal disease, alcohol excess and chronic lung disease. Indigenous Australians are disproportionally affected. Healthy people can become infected when working in wet, muddy conditions without adequate hand and foot protection.
Symptoms
The most common presentation of melioidosis is pneumonia, which may be severe [23]. It can also cause multiple abscesses in the skin, prostate, spleen, kidney and liver. Septic arthritis, osteomyelitis and neurological disease also occur. In endemic areas, melioidosis is an important differential diagnostic consideration in community-acquired sepsis. Septic shock develops in approximately 20% and carries a high mortality. Unusual features of melioidosis include the development of sepsis after a long initial period of subclinical infection and its potential for recurrence after apparently appropriate antibiotic treatment.
Diagnosis and management
Melioidosis should be treated empirically when the diagnosis is suspected in endemic areas. Serological tests are of limited utility in populations with high background rates of infection. Cultures of blood, sputum, urine or swabs from an abscess or skin ulcer (in Ashdown’s selective medium) can confirm the diagnosis. A chest X-ray should be performed in all suspected cases and a CT-abdomen and pelvis is recommended to seek abscesses in any culture-positive case.
B. pseudomallei is resistant to penicillins, most cephalosporins and aminoglycosides. Although it has some susceptibility to ceftriaxone (2 g IV is recommended initially for adults), to treat melioidosis definitively, meropenem (25 mg/kg up to 1 g, 8-hourly), imipenem (25 mg/kg up to 1 g, 6-hourly) or ceftazidime (50 mg/kg up to 2 g, 6-hourly) needs to be given. Intravenous antibiotic therapy should be continued for at least 14 days and followed by oral therapy (usually with sulfamethoxazole/trimethoprim) for 3–6 months. Abscesses should be drained and septic joints washed out. Expert consultation is recommended.
Leptospirosis
The zoonotic, spirochaete bacteria of the Leptospira genus have a worldwide distribution (including Australia and New Zealand), with a higher prevalence in wet and humid tropical areas. Transmission occurs via the urine of infected animals and people with occupational or recreational exposure to animals or their urine are at particular risk.
Leptospira enter the body through damaged skin or mucous membranes, circulate in the blood and then invade kidneys, lungs and liver. The incubation period is 2–20 days. The initial (spiraemic) phase produces non-specific flu-like symptoms with conjunctivitis and occasional jaundice and hepatosplenomegaly. This is followed by a second (immune) phase which may include renal and hepatic failure, aseptic meningitis and pulmonary haemorrhage. Multiorgan failure can lead to death.
The diagnosis is usually made by leptospirosis serology (microagglutination test). However, initial serology is often negative, necessitating a convalescent serum sample for diagnosis. Polymerase chain reaction tests are also available. Cultures may only become positive after several weeks of incubation. Leptospira are sensitive to a wide variety of antibiotics, including doxycycline (100 mg PO, 12-hourly, for 5–7 days). For more severe disease, intravenous benzylpenicillin (30 mg/kg up to 1.2 g IV, 6-hourly, for 5–7 days) or ceftriaxone (25 mg/kg up to 1 g, daily for 5–7 days) are recommended.
Rickettsia
Rickettsia are a group of pleomorphic, Gram-negative, obligate intracellular bacteria, with various species occurring in different areas throughout the world. They are spread by ticks and several other vectors.
Spotted fever
This group of rickettsial diseases includes Rocky Mountain spotted fever (RMSF), African tick bite fever, Mediterranean spotted fever, Australian tick typhus and others. The disease typically begins with fever, nausea and vomiting, myalgia and headaches. After a few days, a maculopapular rash appears in the majority of cases. RMSF in particular can progress to severe disease with vasculitis involving the lungs, intra-abdominal organs, central nervous system and skin (petechial rash). Treatment with oral or intravenous doxycycline (100 mg, 12-hourly, for 7–10 days) should be commenced when there is sufficient clinical suspicion; confirmation from serological tests should not be awaited.
Typhus
Caused by Rickettsia that are spread by lice and fleas; epidemics occur after natural or man-made disasters. Symptoms include a high fever with rigors, cough, myalgias and delirium. After a few days, a centrifugal rash may be seen. Treatment with doxycycline (dosing as above) or azithromycin (500 mg orally on day 1, then 250 mg daily for a further 4 days) may be life saving.
Scrub typhus
This disease is caused by Orientia species, a bacterium similar to Rickettsia. It is endemic to East and Southeast Asia and northern Australia and is spread by larval stages of mites which occur in dense scrub vegetation. Symptoms include fever with chills, headache, cough, lymphadenopathy and sometimes a rash. An eschar (macule with black scab) often develops when the bite site ulcerates, usually in the groin, on the buttocks or in the axilla. Multiorgan failure can develop and fatal cases within Australia have been described. It is treated with doxycycline or azithromycin (dosing as above).
Enteric fever
Salmonella enterica, serovar Typhi causes a severe, acute febrile illness known as typhoid fever. Paratyphoid fever is a similar but usually less severe disease caused by serovar Paratyphi. Together, these entities are referred to as enteric fever. The causative bacteria occur throughout tropical areas of the world, with the majority of cases occurring in South Asia (India, Pakistan, Bangladesh). They are spread via the faecal–oral route, often via contaminated food or water. Oral and intramuscular vaccines are available, but are not completely effective.
Enteric fever is a systemic illness, different from the ‘simple’ gastroenteritis caused by non-typhoid Salmonella serovars. Initial symptoms include high fever for>2 days with malaise, headache, cough and constipation. After a week, prostration and high fevers with relative bradycardia become prominent. A rash (rose spots) and hepatosplenomegaly can occur. Delirium and profuse diarrhoea often develop in the third week. Most mortality is caused by complications, including intestinal haemorrhage or perforation, septicaemia, encephalitis and secondary abscess formation. Survivors slowly improve over another week or so.
The diagnosis can be made from blood, stool or bone marrow cultures. Management is complicated by increasing rates of antibiotic resistance, with multidrug resistance particularly problematic in Southeast Asia. Fluoroquinolones or third-generation cephalosporins are still largely effective treatments; azithromycin is recommended for Southeast Asia. For infections acquired in areas with less antibiotic resistance, other options may include chloramphenicol, amoxicillin and co-trimoxazole; expert consultation is recommended. Dexamethasone may reduce mortality in septic shock from enteric fever and good supportive management is essential.
Cholera
The flagellated, Gram-negative bacterium Vibrio cholerae secretes a toxin which causes profuse, watery diarrhoea [24]. It is transmitted via the faecal–oral route and occurs throughout sub-Saharan Africa, South Asia and South America. Oral vaccines are available.
Cholera causes high-volume watery diarrhoea (‘rice water’) leading to dehydration with electrolyte loss, metabolic acidosis and hypoglycaemia. Vomiting occurs in the majority of cases. Shock, renal failure and cardiac arrhythmias are the main causes of mortality.
In the event of an outbreak, the diagnosis is often made on clinical grounds alone. V. cholerae may be demonstrated on microscopy and cultures in selective media can confirm the diagnosis.
The cornerstone of management is rehydration (oral or intravenous fluids) with electrolyte replacement. In severe cases, antibiotics reduce diarrhoea duration and severity. Doxycycline and azithromycin (20 mg/kg up to 1 g orally as a single dose) are most commonly used; other antibiotics may also be effective, such as ciprofloxacin (25 mg/kg up to 1 g orally, as a single dose).
Approach to the returned traveller
People returning from tropical areas may present to healthcare providers with a variety of symptoms. A systematic approach is important to appropriately investigate and manage these patients. Ordered lists of common presenting symptoms, incubation times and geographical distributions can be found widely on the Internet [25]. Up-to-date information on endemic tropical diseases in the area of the patient’s travel should be sought, including any current outbreaks of tropical or non-tropical diseases (such as novel influenza viruses or SARS). It is essential that common non-tropical diseases, such as influenza or meningococcal meningitis, are included in the differential diagnosis.
History
Obtain a detailed previous medical history, including any predisposing factors, such as immune compromise, splenectomy and pregnancy, as well as a list of medications and known adverse drug reactions.
The travel history should include:
![]() exact locations, duration and dates of stay, including any stopovers
exact locations, duration and dates of stay, including any stopovers
![]() the nature of the accommodation (e.g. air-conditioned hotel or camping), use of bed nets and insect repellents
the nature of the accommodation (e.g. air-conditioned hotel or camping), use of bed nets and insect repellents
![]() vaccination history (routine and travel-related) and adherence to preventative medication (such as antimalarial drugs)
vaccination history (routine and travel-related) and adherence to preventative medication (such as antimalarial drugs)
![]() any behaviour that may have led to disease exposure: known insect or tick bites, contact with sick people, animals, fresh water (including leisure activities), potentially unsafe food or water and sexual or needle exposures.
any behaviour that may have led to disease exposure: known insect or tick bites, contact with sick people, animals, fresh water (including leisure activities), potentially unsafe food or water and sexual or needle exposures.
The history of the presenting complaint should include fever patterns (including rigors) and associated symptoms (e.g. respiratory, cutaneous, gastrointestinal and other symptoms). Ask about appearance and frequency of stools and whether they contained blood or mucus (dysentery).
Examination
The physical examination is often non-specific but can sometimes provide important clues to the diagnosis. Careful examination of the respiratory system, lymph nodes and skin is important (Table 9.11.11). Look for hepatosplenomegaly, jaundice and bleeding.
Table 9.11.11
Important tropical diseases by presenting symptom

Investigations
Potential non-tropical causes for the patient’s condition should be investigated as usual, e.g. chest X-ray for respiratory infections or a lumbar puncture for meningitis.
If the history and examination have raised suspicion for conditions for which specific tests (such as serology or cultures) are available, these should be sent off. Keep in mind that many of these will take days or longer to come back. Stool samples for culture, ova and parasites may be helpful. Specific tests for Giardia, Cryptosporidium and Entamoeba histolytica can be requested.
Routine blood tests may provide support for certain conditions, but they are rarely diagnostic. There should be a low threshold for obtaining three sets of thick and thin smears for malaria. Malaria rapid antigen detection tests are also very helpful, although relatively low sensitivity of these kits mandates that three blood films are still required to rule out malaria in any traveller with persisting fever who has returned from a malaria-endemic location. Eosinophilia is usually associated with parasitic (helminth) infection, but also with several other infections including HIV, HTLV and tuberculosis. Evidence of haemolysis or coagulopathy may correlate with worse outcome.
Management
Specific treatment may be commenced if available, depending on the likely diagnosis. This may need to be done without the benefit of laboratory confirmation. A low threshold for expert consultation is recommended to help guide investigations and management. Supportive treatment is very important and may include analgesia, fever control measures and the maintenance of adequate hydration and electrolyte replacement.
Patients who are unwell or suspected to have a high-risk condition should be admitted to the hospital. Consideration can be given to discharging low-risk patients, provided that adequate follow up can be arranged, including the results of any outstanding tests. Public health notification of any suspected or proven notifiable diseases is essential.
Acknowledgement
The author wishes to thank Professor Bart Currie, Infectious Diseases Physician at Royal Darwin Hospital and the Menzies School of Health Research, for his helpful suggestions.
The author strongly recommends checking all drug doses and regimens carefully. The latest version of the Australian Therapeutic Guidelines (Antibiotic) or other appropriate local guidelines should be consulted.
References
1. World Health Organization (WHO).<http://www.who.int/features/factfiles/malaria/en/index.html>and UNICEF<http://www.unicef.org/health/index_malaria.html>[Accessed Feb. 2013].
2. Hedrick PW. Population genetics of malaria resistance in humans. Heredity. 2011;107:283–304.
3. Sutherland CJ, Tanomsing N, Nolder D, et al. Two nonrecombining sympatric forms of the human malaria parasite Plasmodium ovale occur globally. J Infect Dis. 2010;201:1544–1550.
4. World Health Organization. Guidelines for the treatment of malaria 2nd ed. Geneva: WHO; 2010.
5. Trampuz A, Jereb M, Muzlovic I, Prabhu RM. Clinical review: severe malaria. Crit Care. 2003;7:315–323.
6. Taylor SM, Molyneux ME, Simel DL, et al. Does this patient have malaria? J Am Med Assoc. 2010;304:2048–2056.
7. Moody AH, Chiodini PL. Methods for the detection of blood parasites. Clin Lab Haematol. 2000;22:189–201.
8. South East Asian Quinine Artesunate Malaria Trial (SEAQUAMAT) group. Artesunate versus quinine for treatment of severe falciparum malaria: a randomised trial. Lancet. 2005;366:717–725.
9. Dondorp AM, Fanello CI, Hendriksen ICE, et al. Artesunate versus quinine in the treatment of severe falciparum malaria in African children (AQUAMAT): an open-label, randomised trial. Lancet. 2010;376:1647–1657.
10. Gryseels B, Polman K, Clerinx J, Kestens L. Human schistosomiasis. Lancet. 2006;368:1106–1118.
11. Ross AGP, Bartley PB, Sleigh AC, et al. Schistosomiasis. N Engl J Med. 2002;346:1212–1220.
12. MacConnachie. Schistosomiasis. J R Coll Physicians Edinb. 2012;42:47–50.
13. Pavli A, Maltezou HC. Leishmaniasis, an emerging infection in travelers. Internatl J Infect Dis. 2010;14:e1032–e1039.
14. Adams M, Page W, Speare R. Strongyloidiasis: an issue in Aboriginal communities, rural and remote health 2003;3(online).<http://rrh.deakin.edu.au>.
15. Johnston FH, Morris PS, Spears R, et al. Strongyloidiasis: a review of the evidence for Australian practitioners. Aust J Rural Hlth. 2005;13:247–254.
16. Dengue vaccine roll-out: getting ahead of the game. Bull World Health Org 2011;89:476–7.
17. Gray TJ, Burrow JN, Markey PG, et al. Case report: West Nile virus (Kunjin subtype) disease in the Northern Territory of Australia – a case of encephalitis and review of all reported cases. Am J Trop Med Hyg. 2011;85:952–956.
18. Knox J, Cowan RU, Doyle JS, et al. Murray Valley encephalitis: a review of clinical features, diagnosis and treatment. Med J Aust. 2012;196:5.
19. National Institute for Health and Clinical Excellence (NICE) clinical guideline 117. Tuberculosis: clinical diagnosis and management of tuberculosis, and measures for its prevention and control, 2011.<www.nice.org.uk/guidance/CG117>.
20. Ministry of Health. Guidelines for tuberculosis control in New Zealand, 2010; (online) HP 5148.<http://www.moh.govt.nz>.
21. Lawn SD, Zumla AI. Tuberculosis. Lancet. 2011;378:57–72.
22. Wiersinga WJ, Currie BJ, Peacock SJ. Melioidosis. N Engl J Med. 2012;367:1035–1044.
23. Currie BJ, Ward L, Cheng AC. The epidemiology and clinical spectrum of melioidosis: 540 cases from the 20 year Darwin prospective study. PLoS Neglected Trop Dis. 2010;11:e900 <www.plosntds.org>.
24. Harris JB, LaRocque RC, Qadri F, et al. Cholera. Lancet. 2012;379:2466–2476.
25. Centers for Disease Control.<http://wwwnc.cdc.gov/travel/yellowbook/2012/chapter-5-post-travel-evaluation/general-approach-to-the-returned-traveler.htm>[Accessed Feb. 2013].

E-FIG. 9.8.1 Genital herpes rash, primary attack. (Reproduced with permission from Campbell JL, Chapman MS, Dinulos JGH, Zug KA. Skin Disease: Diagnosis and Treatment, 3rd edn, Chapter 7, Sexually transmitted infections. Elsevier Inc., 2011, 184–209.)

E-FIG. 9.8.3 Secondary syphilis rash. (Reproduced with permission from Campbell JL, Chapman MS, Dinulos JGH, Zug KA. Skin Disease: Diagnosis and Treatment, 3rd edn, Chapter 7, Sexually transmitted infections. Elsevier Inc., 2011, 184–209.)

E-FIG. 9.8.4 Secondary syphilis rash. (Reproduced with permission from Campbell JL, Chapman MS, Dinulos JGH, Zug KA. Skin Disease: Diagnosis and Treatment, 3rd edn, Chapter 7, Sexually transmitted infections. Elsevier Inc., 2011, 184–209.)
*These agents are no longer in widespread use in Australia.
†Drugs that are licensed but are no longer in common use in Australia have been omitted.