Mediastinum: Overview
![]() The mediastinum is the space in the thorax between the pleural sacs of the lungs. It is divided into two parts: superior and inferior. The inferior mediastinum is further divided into anterior, middle, and posterior portions.
The mediastinum is the space in the thorax between the pleural sacs of the lungs. It is divided into two parts: superior and inferior. The inferior mediastinum is further divided into anterior, middle, and posterior portions.
Fig. 7.1 Divisions of the mediastinum
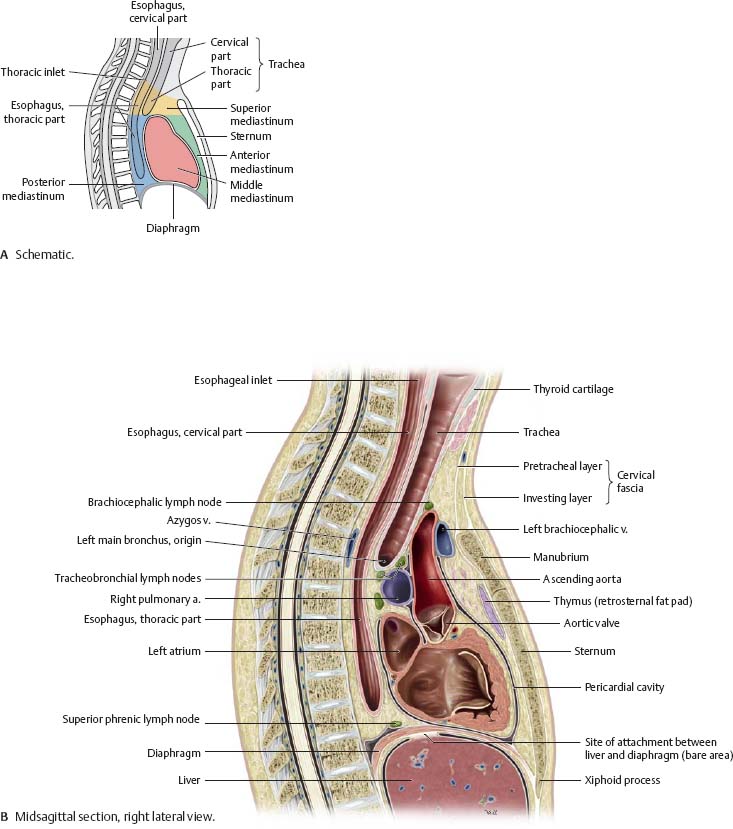
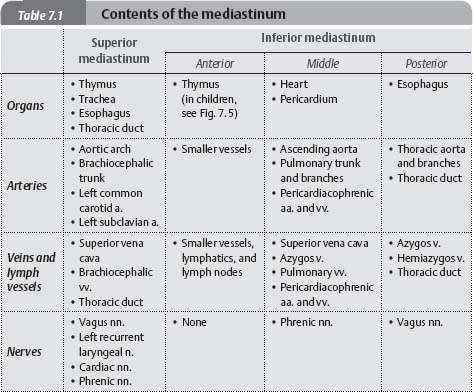
Fig. 7.2 Contents of the mediastinum
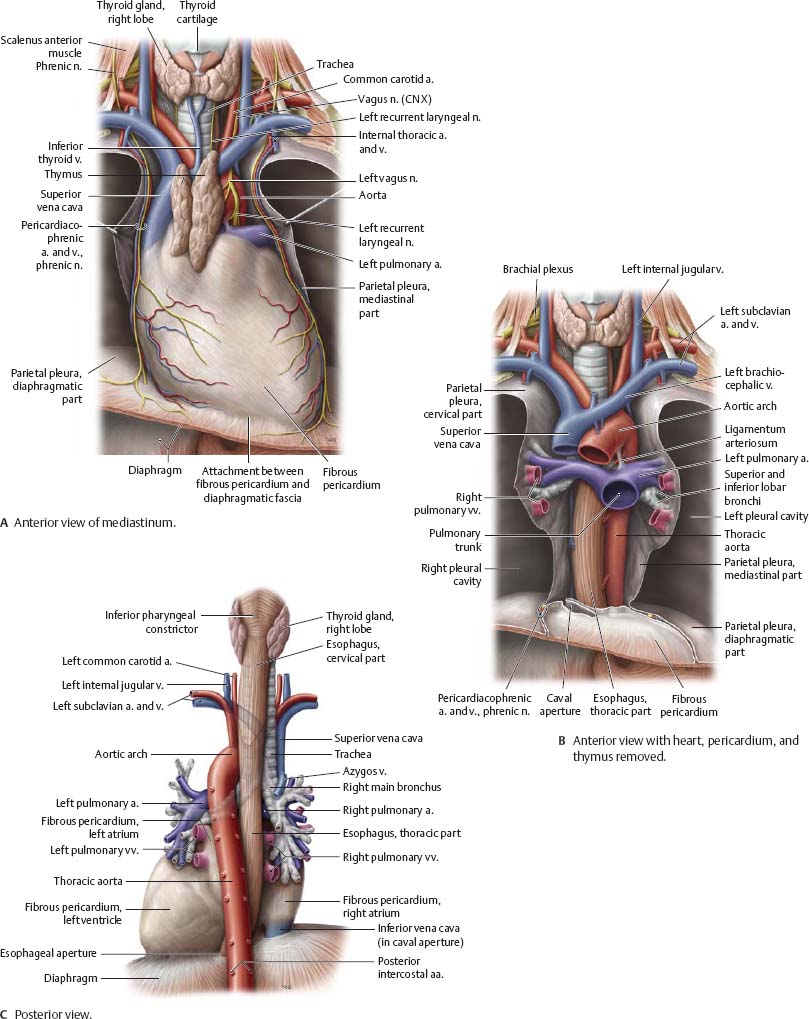
Mediastinum: Structures
Fig. 7.3 Mediastinum
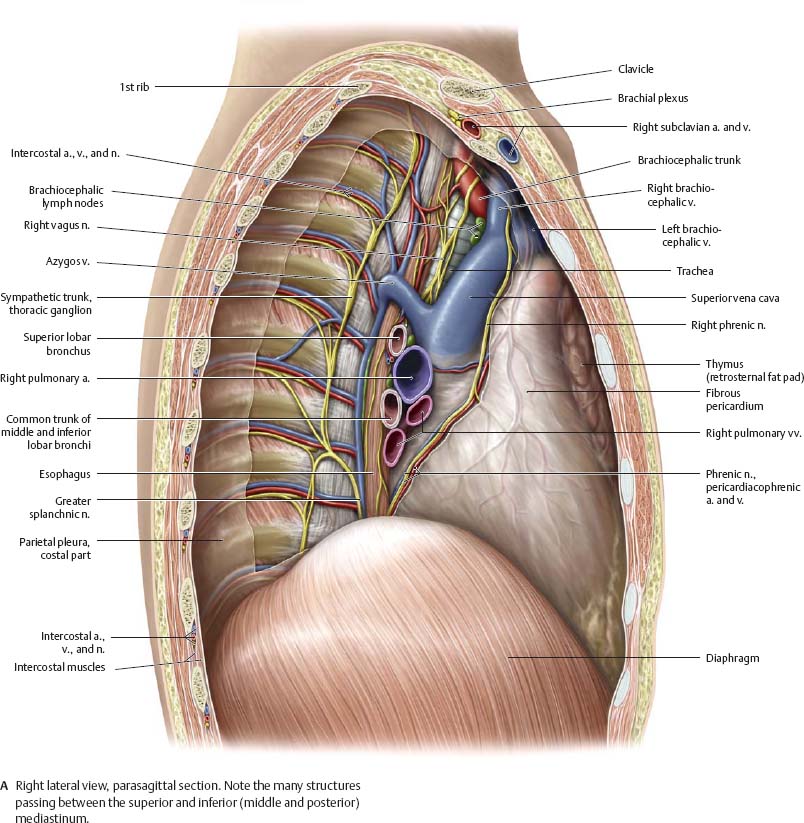
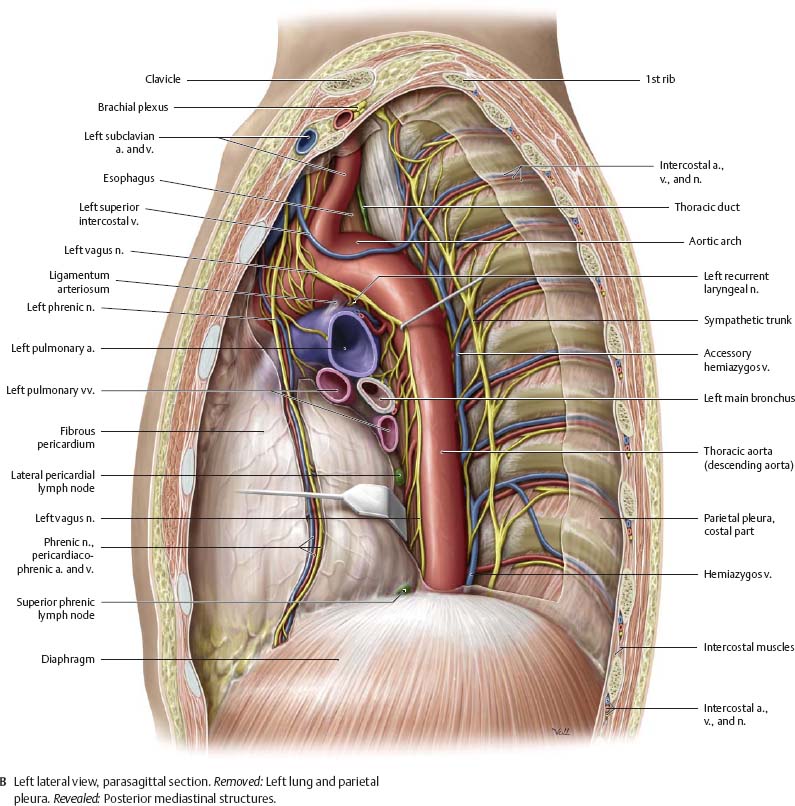
Thymus & Pericardium
Fig. 7.4 Thymus and pericardium in situ
Anterior view of coronal section. The thymus lies in the superior mediastinum.
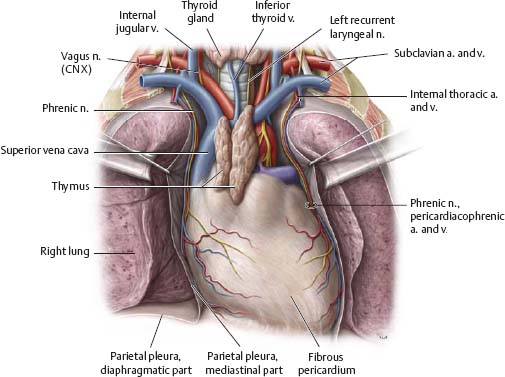
Fig. 7.5 Thymus
Anterior view of opened thorax of a 2-year-old child. The thymus is well developed at this age, extending inferiorly into the anterior mediastinum (compare with Fig. 7.4). The thymus grows throughout childhood; at puberty, high levels of circulating sex hormones cause the thymus to atrophy.
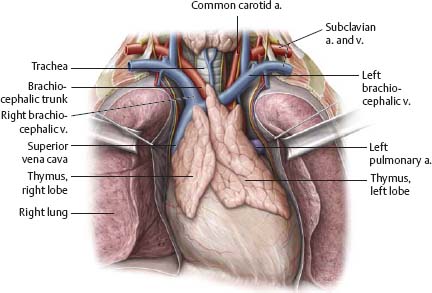
Fig. 7.6 Pericardium
Anterior view of opened thorax with flaps of fibrous pericardium reflected.
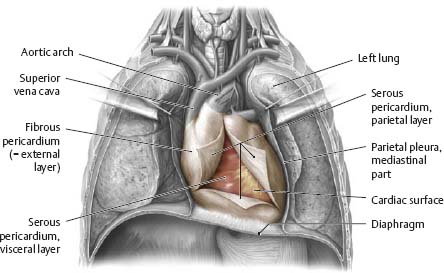
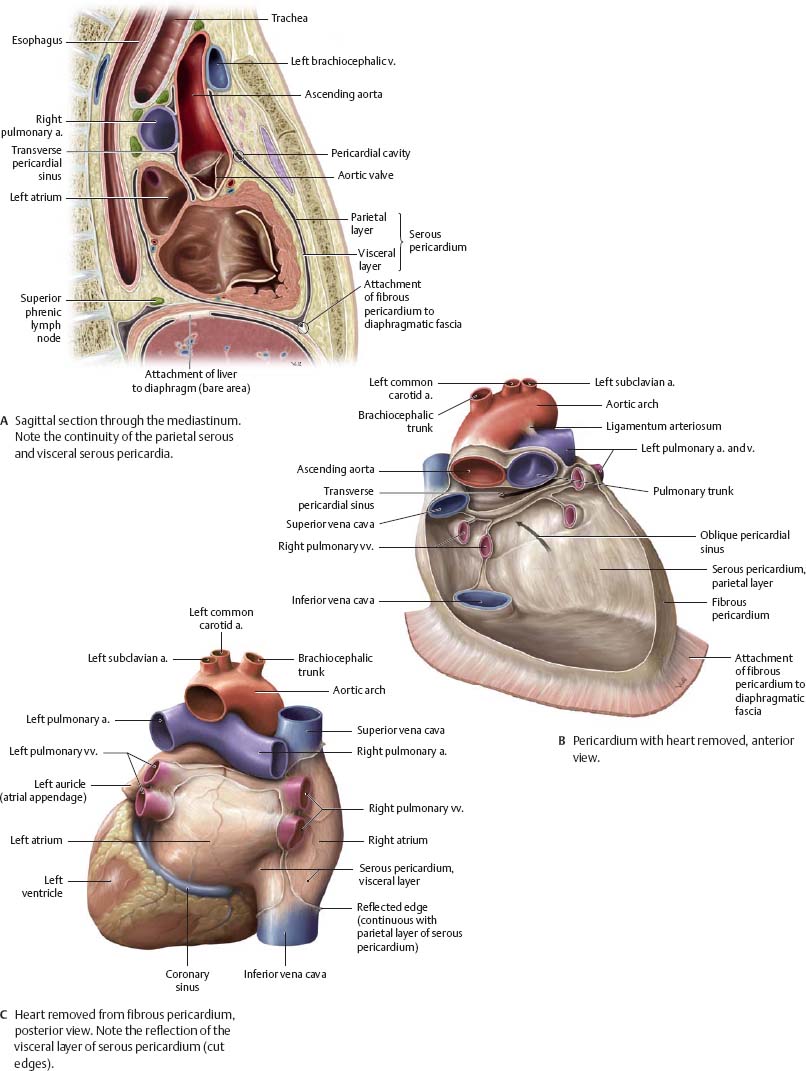
Fig. 7.7 Serous pericardial reflections
Anterior view. The parietal and visceral serous pericardium are continuous with one another around the great vessels of the heart. The passage between the arterial- and venous-associated reflections is the transverse pericardial sinus (see B).
Heart in Situ
![]() The heart is located posterior to the sternum in the middle portion of the inferior mediastinum. The heart projects into the left side of the thoracic cavity.
The heart is located posterior to the sternum in the middle portion of the inferior mediastinum. The heart projects into the left side of the thoracic cavity.
Fig. 7.8 Topographical relations of the heart
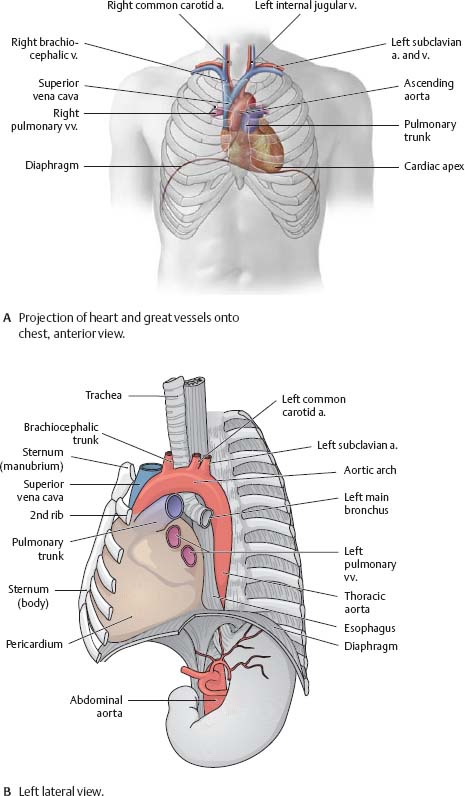
Fig. 7.9 Circulation
Red: Oxygenated blood. Blue: Deoxygenated blood. See p. 94 for prenatal circulation.
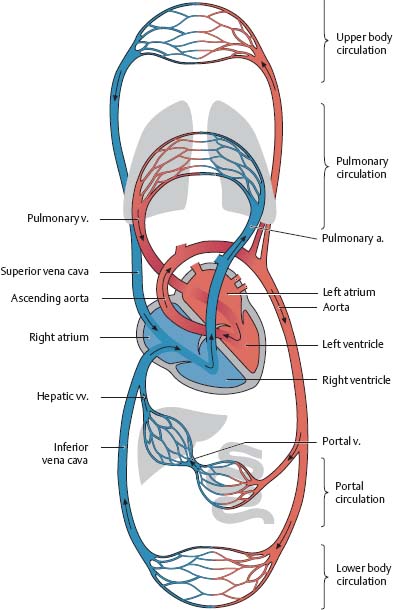
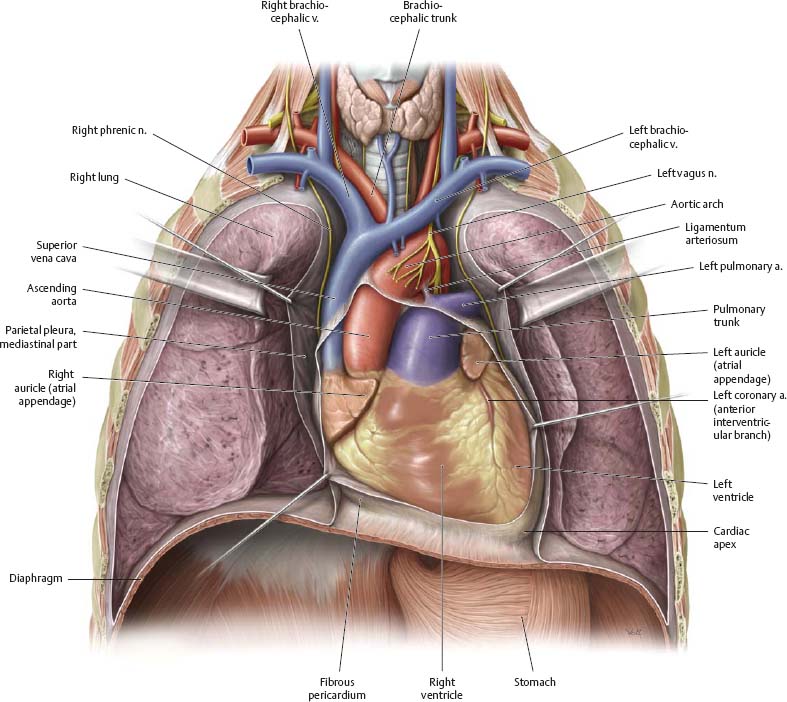
Fig. 7.10 Heart in situ
Anterior view.
Heart: Surfaces & Chambers
![]() Note the reflection of visceral serous pericardium to become parietal serous pericardium.
Note the reflection of visceral serous pericardium to become parietal serous pericardium.
Fig. 7.11 Surfaces of the heart
The heart has three surfaces: anterior (sternocostal), posterior (base), and inferior (diaphragmatic).
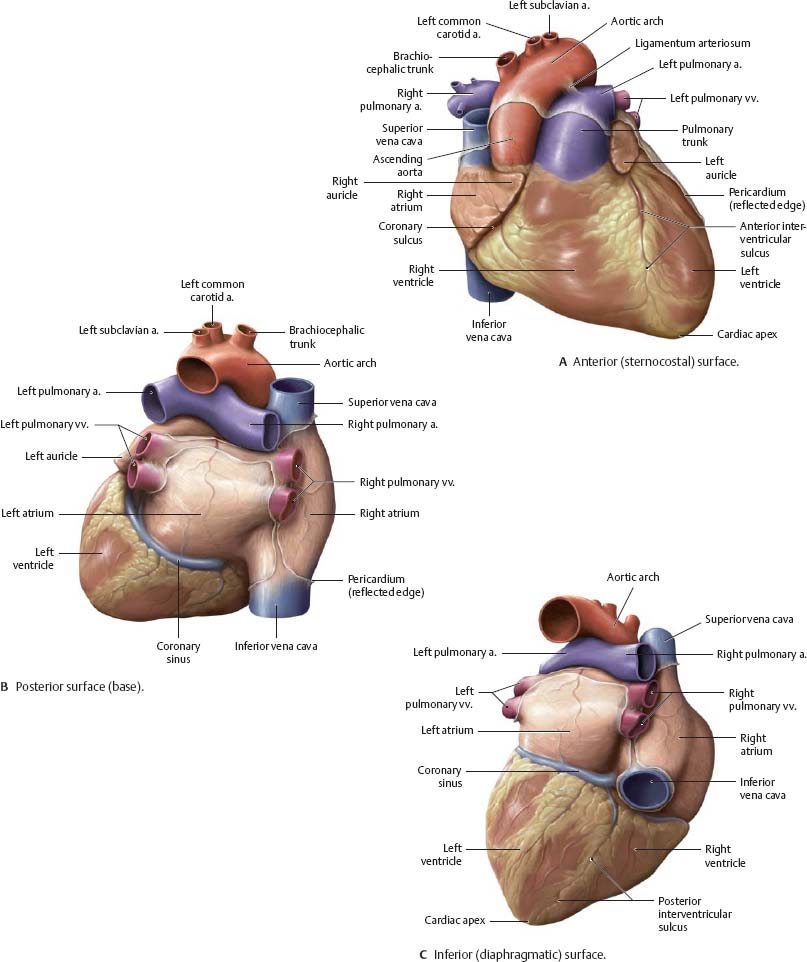
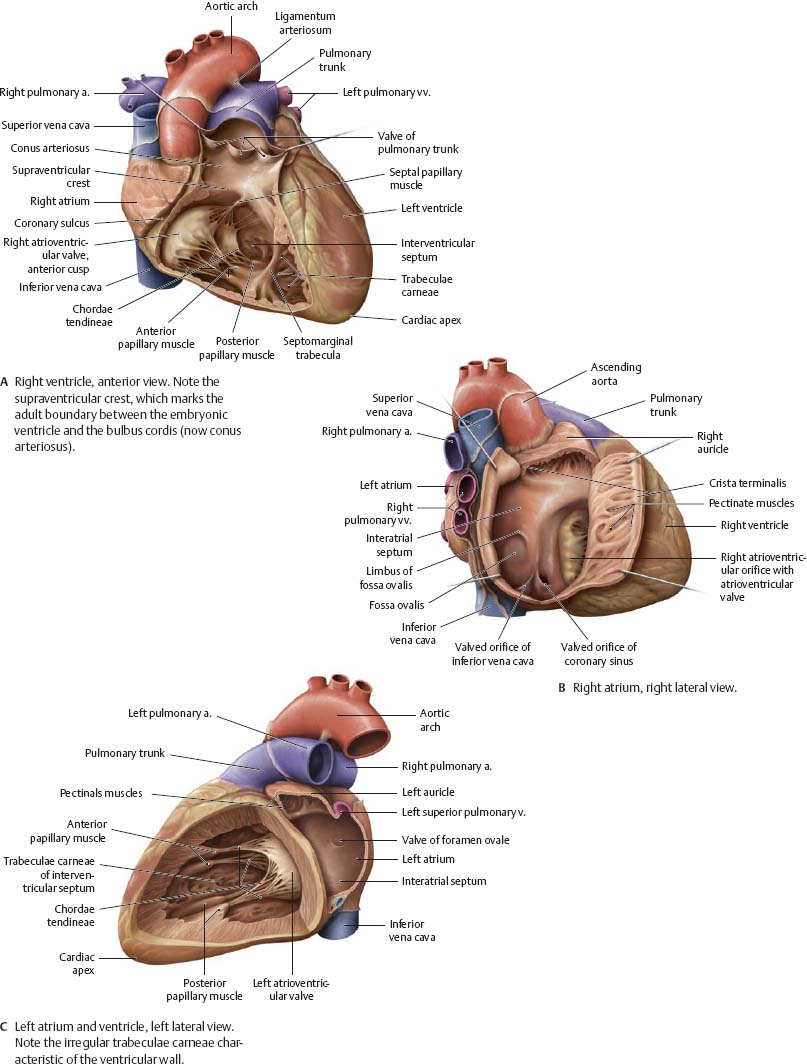
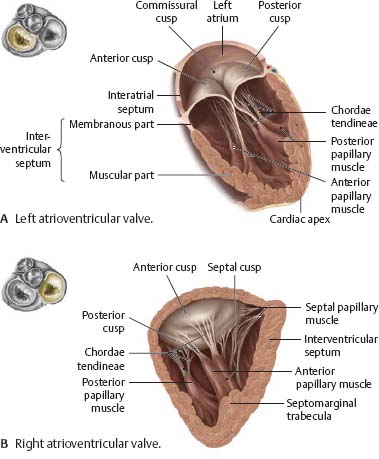
Fig. 7.12 Chambers of the heart
Heart: Valves
![]() The cardiac valves are divided into two groups: semilunar and atrioventricular. The two semilunar valves (aortic and pulmonary) located at the base of the two great arteries of the heart regulate passage of blood from the ventricles to the aorta and pulmonary trunk. The two atrioventricular valves (left and right) lie at the interface between the atria and ventricles.
The cardiac valves are divided into two groups: semilunar and atrioventricular. The two semilunar valves (aortic and pulmonary) located at the base of the two great arteries of the heart regulate passage of blood from the ventricles to the aorta and pulmonary trunk. The two atrioventricular valves (left and right) lie at the interface between the atria and ventricles.
Fig. 7.13 Cardiac valves
Plane of cardiac valves, superior view. Removed: Atria and great arteries.
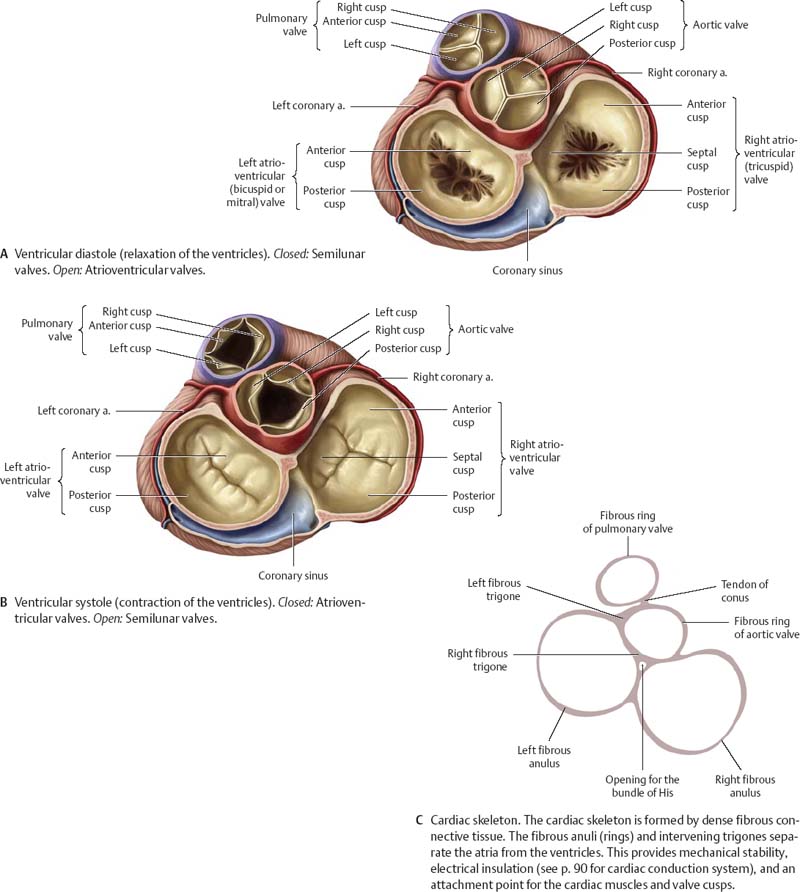
Fig. 7.14 Semilunar valves
Valves have been longitudinally sectioned and opened.
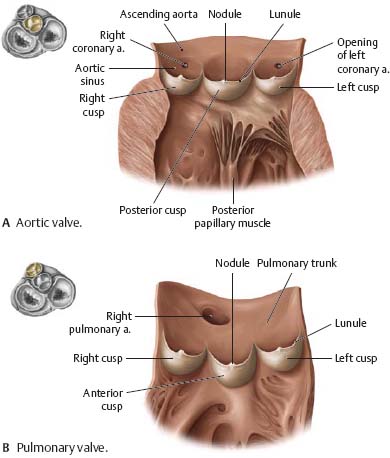
Fig. 7.15 Atrioventricular valves
Anterior view during ventricular systole.
![]() Clinical
Clinical
Auscultation of the cardiac valves
Heart sounds, produced by closure of the semilunar and atrioventricular valves, are carried by the blood flowing through the valve. The resulting sounds are therefore best heard “downstream,” at defined auscultation sites (dark circles). Valvular heart disease causes turbulent blood flow through the valve; this produces a murmur that may be detected in the colored regions.
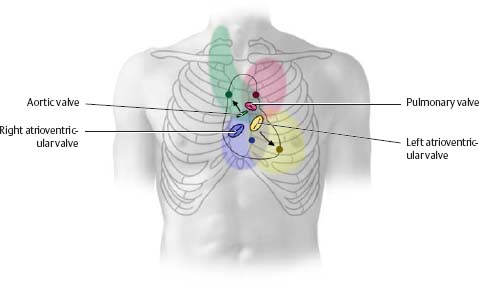
|
Table 7.2 Position and auscultation sites of cardiac valves |
||
|
Valve |
Anatomical projection |
Auscultation site |
|
Aortic valve |
Left sternal border (at level of 3rd rib) |
Right 2nd intercostal space (at sternal margin) |
|
Pulmonary valve |
Left sternal border (at level of 3rd costal cartilage) |
Left 2nd intercostal space (at sternal margin) |
|
Left atrioventricular valve |
Left 4th/5th costal cartilage |
Left 5th intercostal space (at midclavicular line) or cardiac apex |
|
Right atrioventricular valve |
Sternum (at level of 3rd costal cartilage) |
Left 5th intercostal space (at sternal margin) |
Arteries & Veins of the Heart
Fig. 7.16 Coronary arteries and cardiac veins
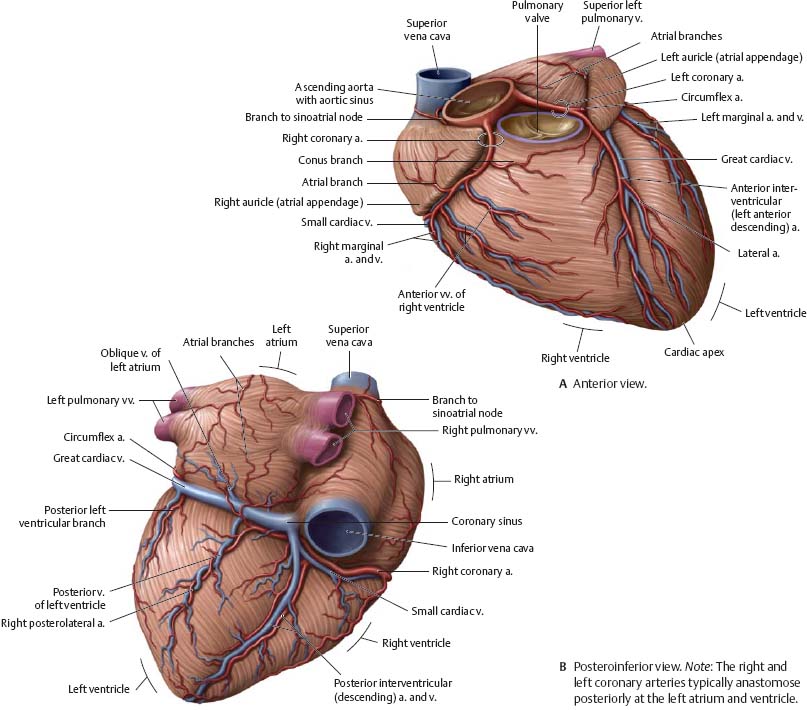

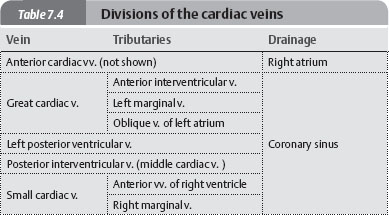
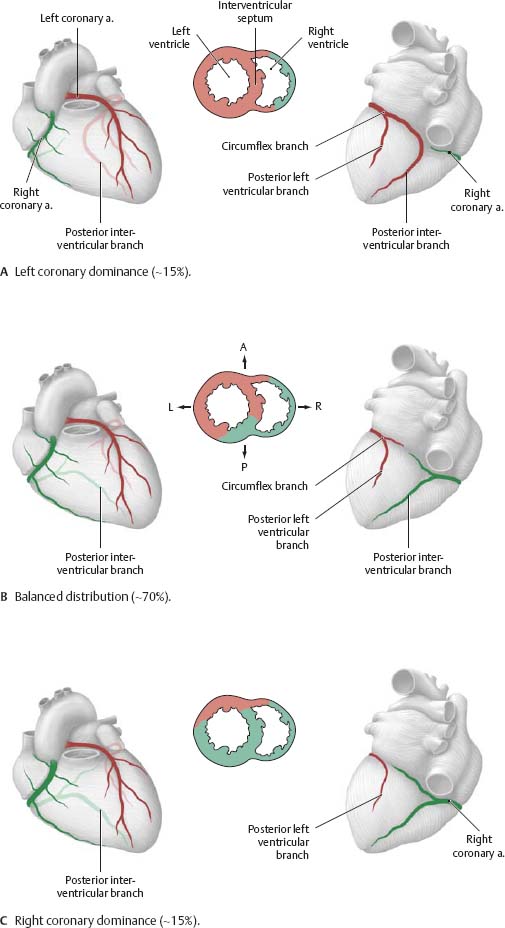
Fig. 7.17 Distribution of the coronary arteries
Anterior and posterior views of the heart, with superior views of transverse sections through the ventricles. The distribution of the coronary arteries differs from person to person. Right coronery artery and branches (green); left coronary artery and branches (red).
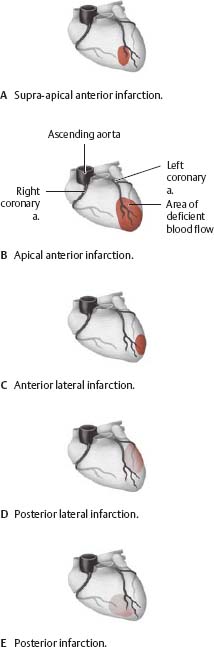
![]() Clinical
Clinical
Disturbed coronary blood flow
Although the coronary arteries are connected by structural anastomoses, they are end arteries from a functional standpoint. The most frequent cause of deficient blood flow is athero sclerosis, a narrowing of the coronary lumen due to plaque-like deposits on the vessel wall. When the decrease in luminal size (stenosis) reaches a critical point, coronary blood flow is restricted, causing chest pain (angina pectoris). Initially, this pain is induced by physical effort, but eventually it persists at rest, often radiating to characteristic sites (e.g., left arm, left side of head and neck). A myocardial infarction occurs when deficient blood supply causes myocardial tissue to die (necrosis). The location and extent of the infarction depends on the stenosed vessel (see A-E, after Heinecker).
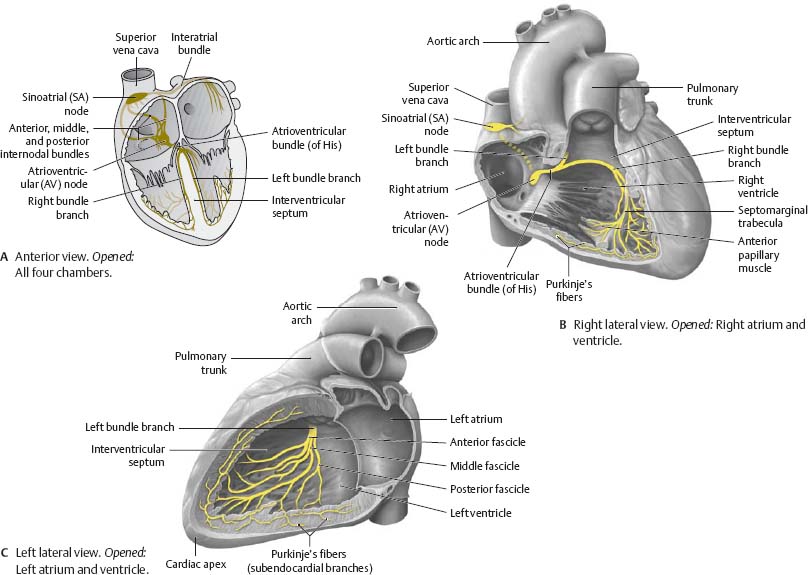
Conduction & Innervation of the Heart
![]() Contraction of cardiac muscle is modulated by the cardiac conduction system. This system of specialized myocardial cells generates and conducts excitatory impulses in the heart. The conduction system contains two nodes, both located in the atria: the sinoatrial (SA) node, known as the pacemaker, and the atrioventricular (AV) node.
Contraction of cardiac muscle is modulated by the cardiac conduction system. This system of specialized myocardial cells generates and conducts excitatory impulses in the heart. The conduction system contains two nodes, both located in the atria: the sinoatrial (SA) node, known as the pacemaker, and the atrioventricular (AV) node.
Fig. 7.18 Cardiac conduction system
![]() Clinical
Clinical
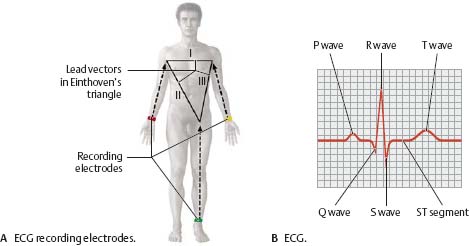
Electrocardiogram (ECG)
The cardiac impulse (a physical dipole) travels across the heart and may be detected with electrodes. The use of three electrodes that separately record electrical activity of the heart along three axes or vectors (Einthoven limb leads) generates an electrocardiogram (ECG). The ECG graphs the cardiac cycle (“heartbeat”), reducing it to a series of waves, segments, and intervals. These ECG components can be used to determine whether cardiac impulses are normal or abnormal (e.g., myocardial infarction, chamber enlargement). Note: Although only three leads are required, a standard ECG examination includes at least two others (Goldberger, Wilson leads).
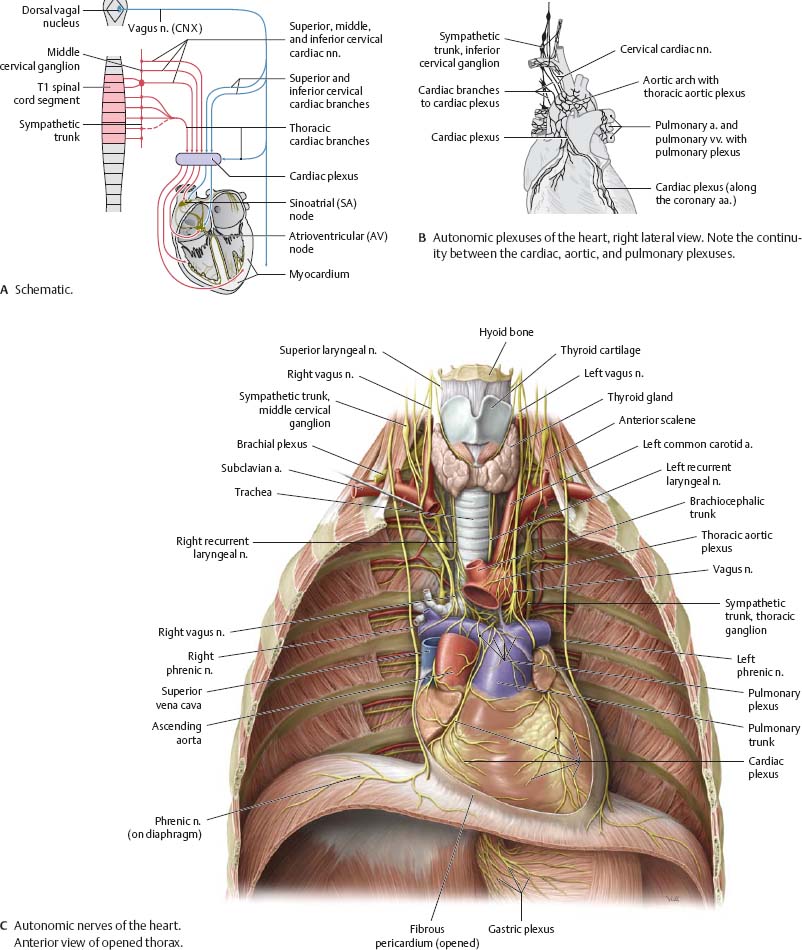
![]() Sympathetic innervation: Presynaptic neurons from T1 to T6 spinal cord segments send fibers to synapse on postsynaptic neurons in the cervical and upper thoracic sympathetic ganglia. The three cervical cardiac nerves and thoracic cardiac branches contribute to the cardiac plexus. Parasympathetic innervation: Presynaptic neurons and fibers reach the heart via cardiac branches, some of which also arise in the cervical region. They synapse on postsynaptic neurons near the SA node and along the coronary arteries.
Sympathetic innervation: Presynaptic neurons from T1 to T6 spinal cord segments send fibers to synapse on postsynaptic neurons in the cervical and upper thoracic sympathetic ganglia. The three cervical cardiac nerves and thoracic cardiac branches contribute to the cardiac plexus. Parasympathetic innervation: Presynaptic neurons and fibers reach the heart via cardiac branches, some of which also arise in the cervical region. They synapse on postsynaptic neurons near the SA node and along the coronary arteries.
Fig. 7.19 Autonomic innervation of the heart
Heart: Radiology
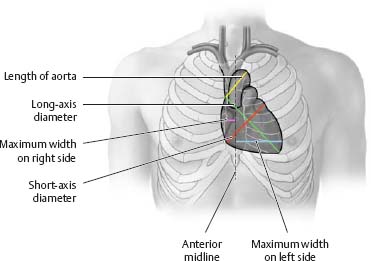
Fig. 7.20 Cardiac borders and configurations
|
Table 7.5 Borders of the heart |
|
|
Border |
Defining structures |
|
Right cardiac border |
Right atrium |
|
Superior vena cava |
|
|
Apex |
Left ventricle |
|
Left cardiac border |
Aortic arch (“aortic knob”) |
|
Pulmonary trunk |
|
|
Left atrium |
|
|
Left ventricle |
|
|
Inferior cardiac border |
Left ventricle |
|
Right ventricle |
|
Fig. 7.21 Radiographic appearance of the heart
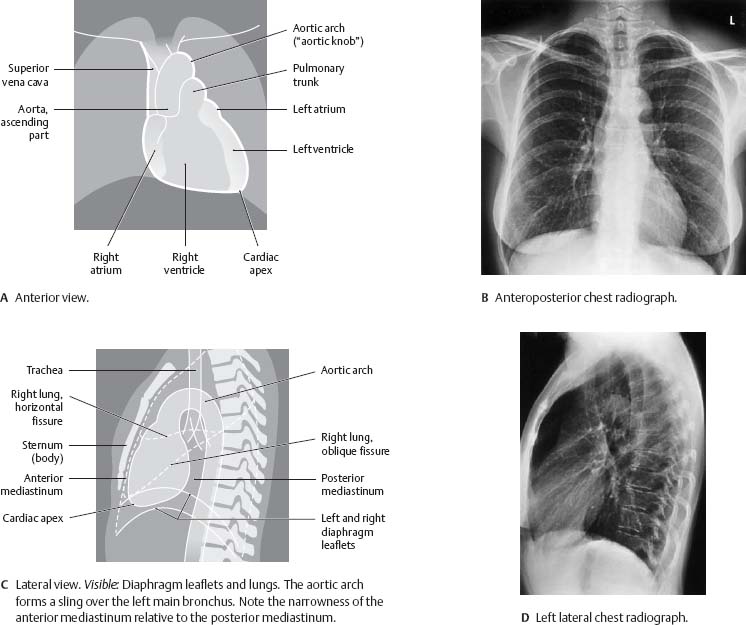
Fig. 7.22 Heart in transverse section
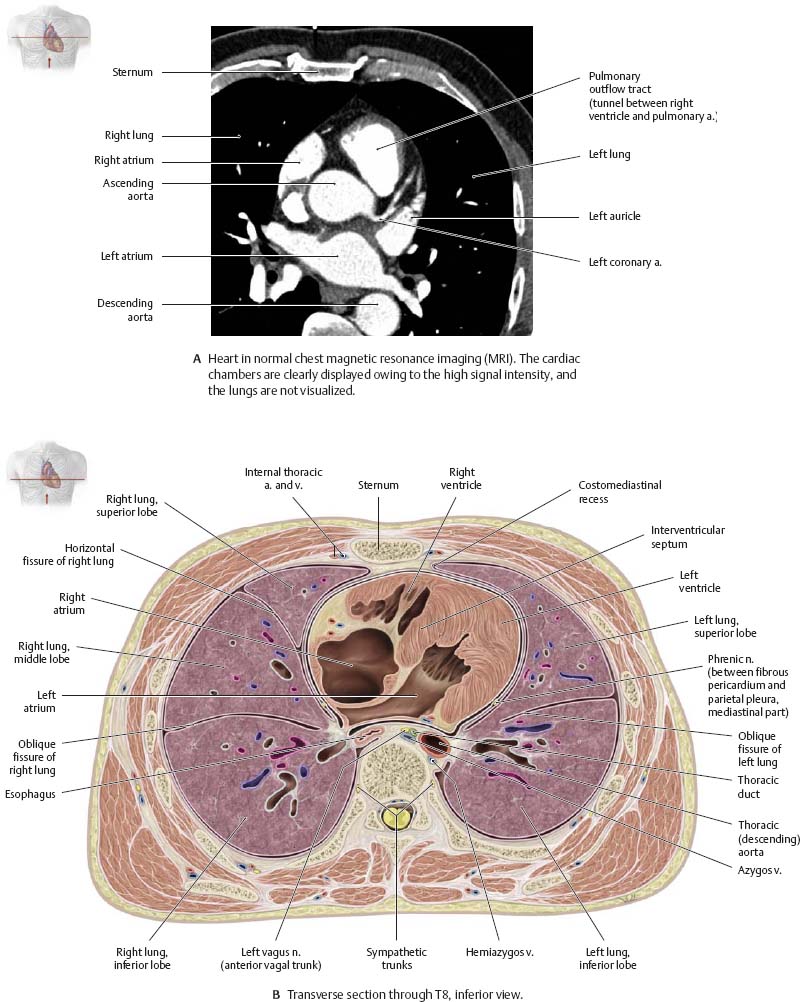
Pre- & Postnatal Circulation
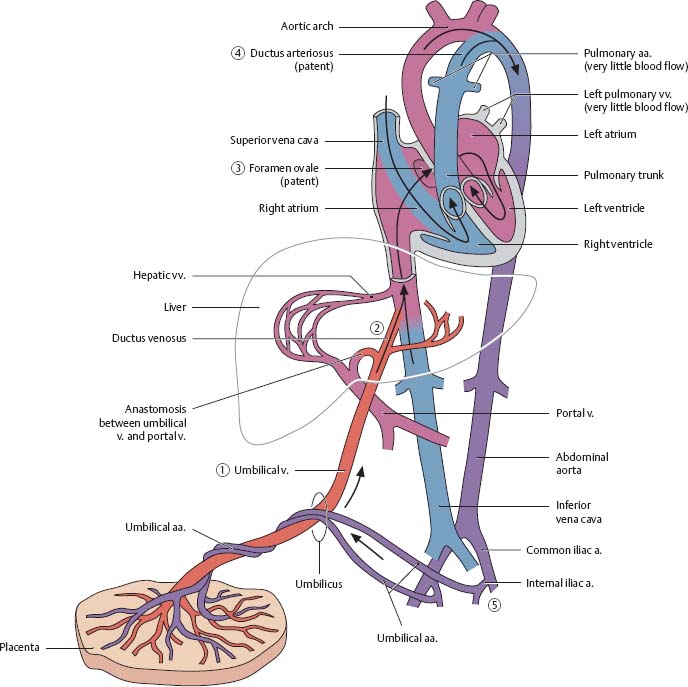
Fig. 7.23 Prenatal circulation
After Fritsch and Kühnel.
1. Oxygenated and nutrient-rich fetal blood from the placenta passes to the fetus via the umbilical vein.
2. Approximately half of this blood bypasses the liver (via the ductus venosus) and enters the inferior vena cava. The remainder enters the portal vein to supply the liver with nutrients and oxygen.
3. Blood entering the right atrium from the inferior vena cava bypasses the right ventricle (as the lungs are not yet functioning) to enter the left atrium via the foramen ovale, a right-to-left shunt.
4. Blood from the superior vena cava enters the right atrium, passes to the right ventricle, and moves into the pulmonary trunk. Most of this blood enters the aorta via the ductus arteriosus, a right-to-left shunt.
5. The partially oxygenated blood in the aorta returns to the placenta via the paired umbilical arteries that arise from the internal iliac arteries.
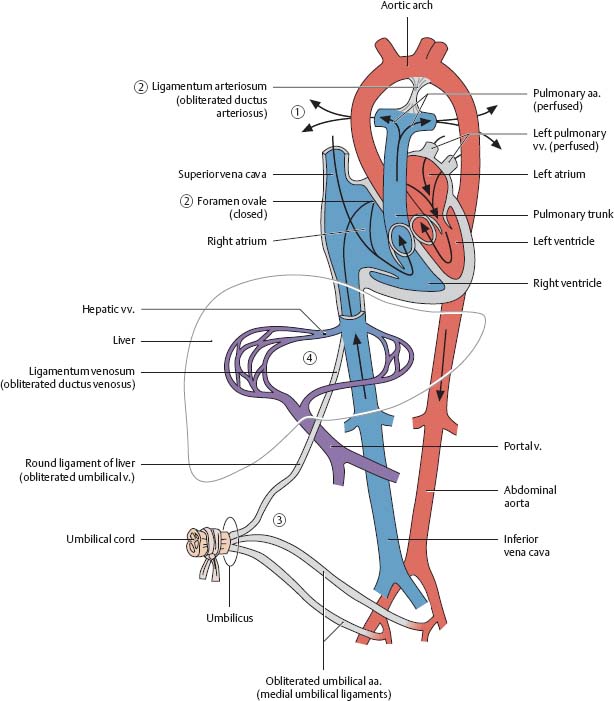
1. As pulmonary respiration begins at birth, pulmonary blood pressure falls, causing blood from the right pulmonary trunk to enter the pulmonary veins.
2. The foramen ovale and ductus arteriosus close, eliminating the fetal right-to-left shunts. The pulmonary and systemic circulations in the heart are now separate.
3. As the infant is separated from the placenta, the umbilical arteries occlude (except for the proximal portions), along with the umbilical vein and ductus venosus.
4. Blood to be metabolized now passes through the liver.
Fig. 7.24 Postnatal circulation
After Fritsch and Kühnel.
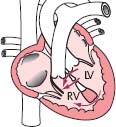
![]() Clinical
Clinical
Septal defects
Septal defects, the most common type of congenital heart defect, allow blood from the left chambers of the heart to improperly pass into the right chambers during systole. Ventrical septal defect (VSD, shown below) is the most common form. Patent foramen ovale, the most prevalent form of atrial septal defect (ASD), results from improper closure of the fetal shunt.
|
Table 7.6 Derivatives of fetal circulatory structures |
|
|
Fetal structure |
Adult remnant |
|
Ductus arteriosus |
Ligamentum arteriosum |
|
Foramen ovale |
Fossa ovalis |
|
Ductus venosus |
Ligamentum venosum |
|
Umbilical v. |
Round ligament of the liver (ligamentum teres) |
|
Umbilical a. |
Medial umbilical ligament |
Esophagus
![]() The esophagus is divided into three parts: cervical (C6-T1), thoracic (T1 to the esophageal hiatus of the diaphragm), and abdominal (the diaphragm to the cardiac orifice of the stomach). It descends slightly to the right of the thoracic aorta and pierces the diaphragm slightly to the left, just below the xiphoid process of the sternum.
The esophagus is divided into three parts: cervical (C6-T1), thoracic (T1 to the esophageal hiatus of the diaphragm), and abdominal (the diaphragm to the cardiac orifice of the stomach). It descends slightly to the right of the thoracic aorta and pierces the diaphragm slightly to the left, just below the xiphoid process of the sternum.
Fig. 7.25 Esophagus: Location and constrictions
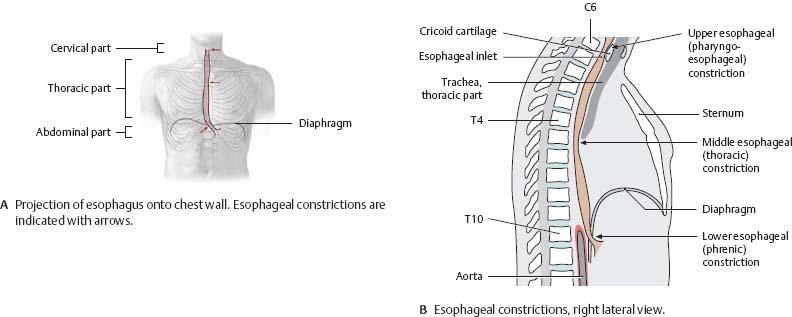
Fig. 7.26 Esophagus in situ
Anterior view.
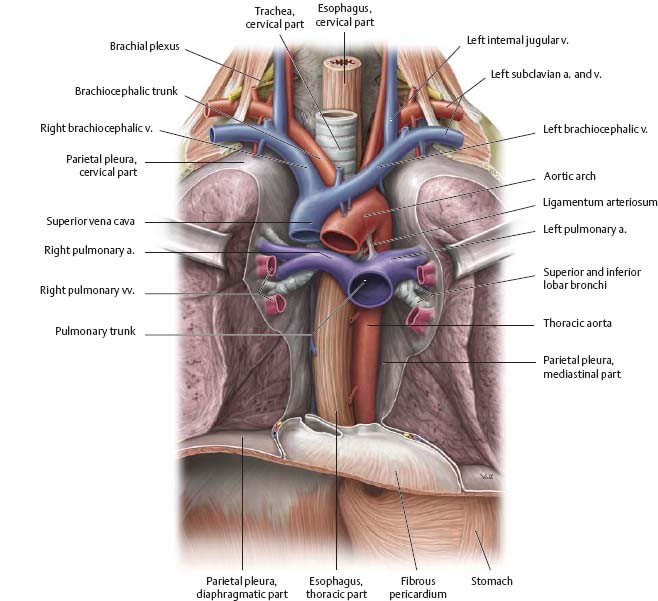
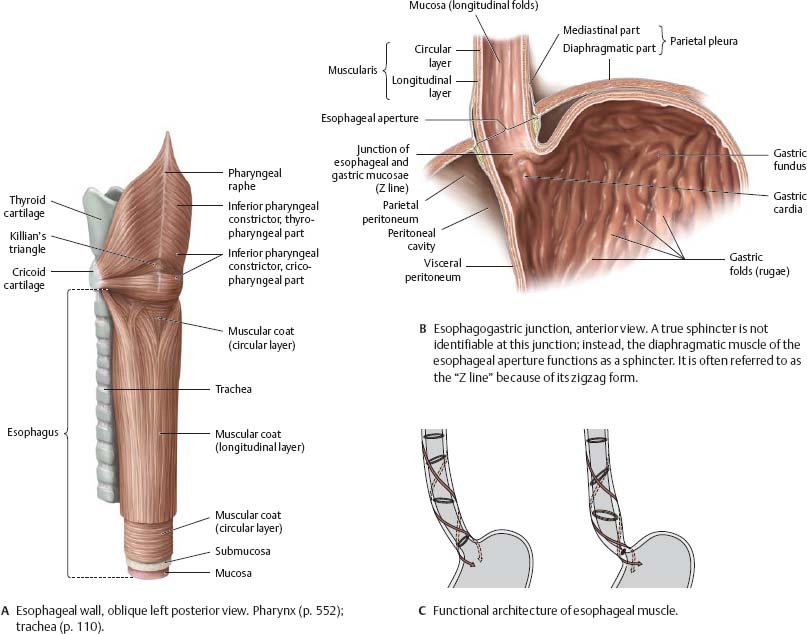
Fig. 7.27 Structure of the esophagus
![]() Clinical
Clinical
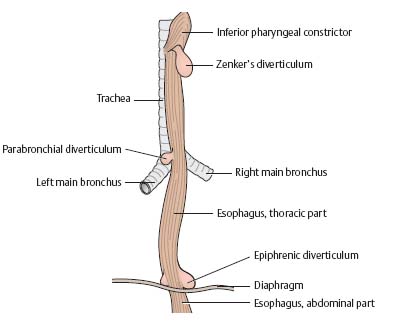
Esophageal diverticula
Diverticula (abnormal outpouchings or sacs) generally develop at weak spots in the esophageal wall. There are three main types of esophageal diverticula:
· Hypopharyngeal (pharyngo-esophageal) diverticula: Outpouchings occurring at the junction of the pharynx and the esophagus. These include Zenker's diverticula (70% of cases).
· “True” traction diverticula: Protrusion of all wall layers, not typicallyoccurring at characteristic weak spots. However, they generally result from an inflammatory process (e.g., lymphangitis) and are thus common at sites where the esophagus closely approaches the bronchi and bronchial lymph nodes (thoracic or parabronchial diverticula).
· “False” pulsion diverticula: Herniations of the mucosa and submucosa through weak spots in the muscular coat due to a rise in esophageal pressure (e.g., during normal swallowing). These include parahiatal and epiphrenic diverticula occurring above the esophageal aperture of the diaphragm (10% of cases).
Neurovasculature of the Esophagus
![]() Sympathetic innervation: Presynaptic fibers arise from the T2-T6 spinal cord segments. Postsynaptic fibers arise from the sympathetic chain to join the esophageal plexus. Parasympathetic innervation: Presynaptic fibers arise from the dorsal vagal nucleus and travel in the vagus nerves to form the extensive esophageal plexus. Note: The postsynaptic neurons are in the wall of the esophagus. Fibers to the cervical portion of the esophagus travel in the recurrent laryngeal nerves.
Sympathetic innervation: Presynaptic fibers arise from the T2-T6 spinal cord segments. Postsynaptic fibers arise from the sympathetic chain to join the esophageal plexus. Parasympathetic innervation: Presynaptic fibers arise from the dorsal vagal nucleus and travel in the vagus nerves to form the extensive esophageal plexus. Note: The postsynaptic neurons are in the wall of the esophagus. Fibers to the cervical portion of the esophagus travel in the recurrent laryngeal nerves.
Fig. 7.28 Autonomic innervation of the esophagus
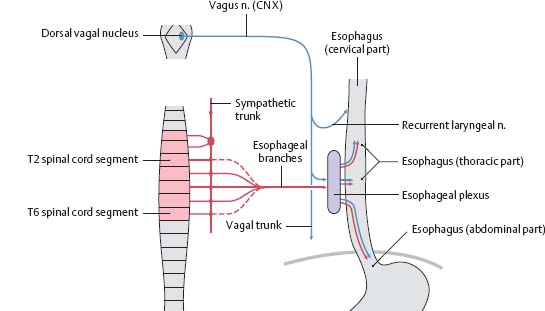
Fig. 7.29 Esophageal plexus
The left and right vagus nerves initially descend on the left and right sides of the esophagus. As they begin to contribute to the esophageal plexus, they shift to anterior and posterior positions, respectively. As the vagus nerves continue into the abdomen, they are named the anterior and posterior vagal trunks.
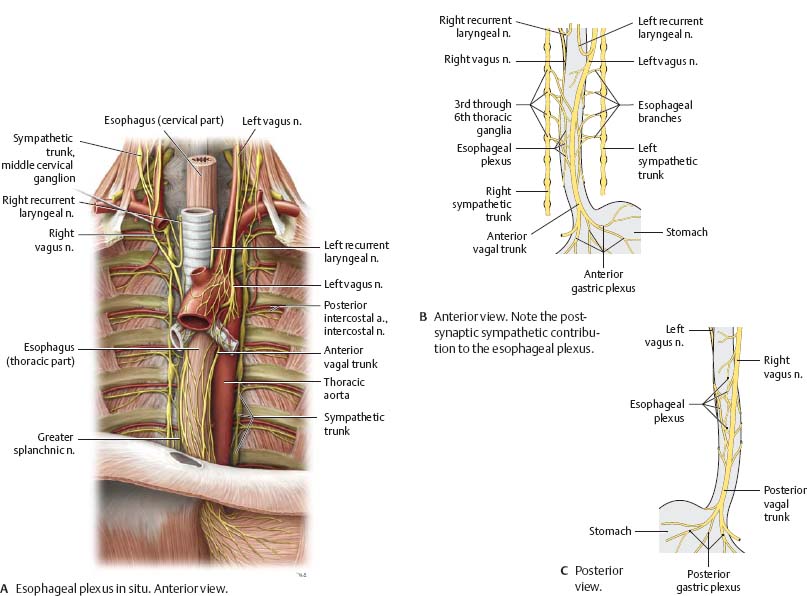
Fig. 7.30 Esophageal arteries
Anterior view.
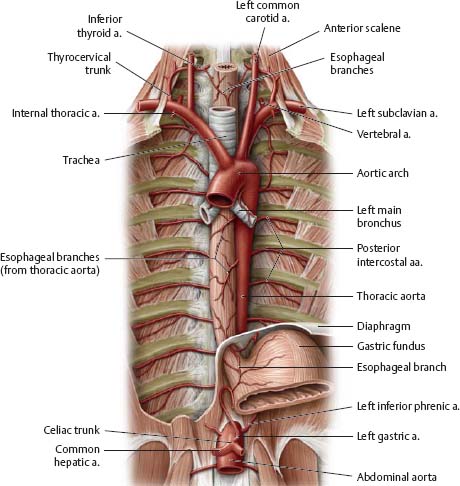
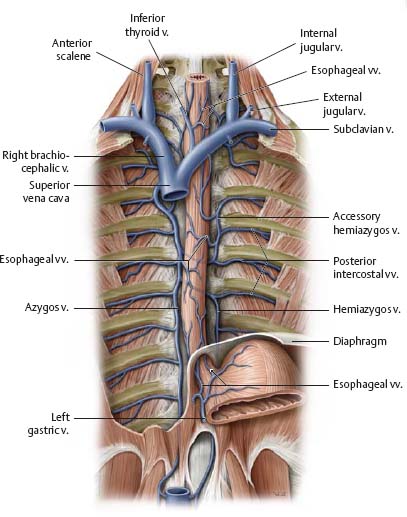
Fig. 7.31 Esophageal veins
Anterior view.

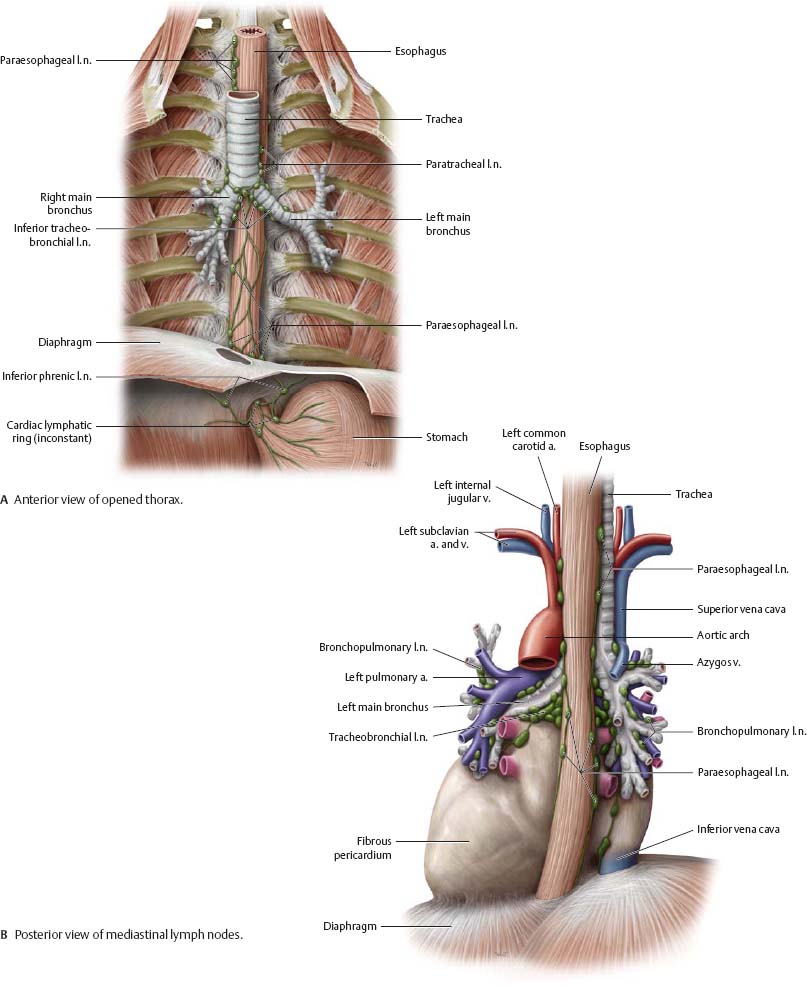
Lymphatics of the Mediastinum
![]() The superior phrenic lymph nodes drain lymph from the diaphragm, pericardium, lower esophagus, lung, and liver into the bronchomediastinal trunk. The inferior phrenic lymph nodes, found in the abdomen, collect lymph from the diaphragm and lower lobes of the lung and convey it to the lumbar trunk. Note: The pericardium may also drain superiorly to the brachiocephalic lymph nodes.
The superior phrenic lymph nodes drain lymph from the diaphragm, pericardium, lower esophagus, lung, and liver into the bronchomediastinal trunk. The inferior phrenic lymph nodes, found in the abdomen, collect lymph from the diaphragm and lower lobes of the lung and convey it to the lumbar trunk. Note: The pericardium may also drain superiorly to the brachiocephalic lymph nodes.
Fig. 7.32 Lymph nodes of the mediastinum and thoracic cavity
Left anterior oblique view.
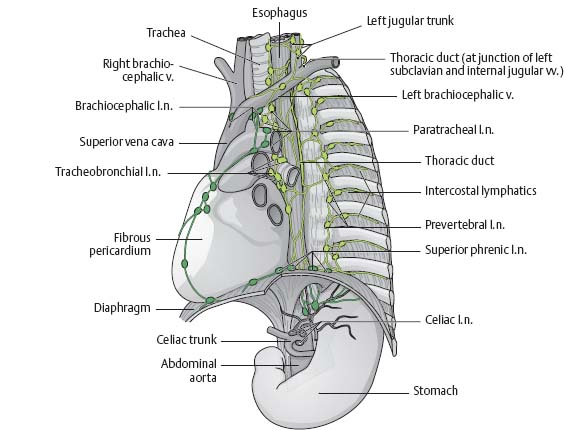
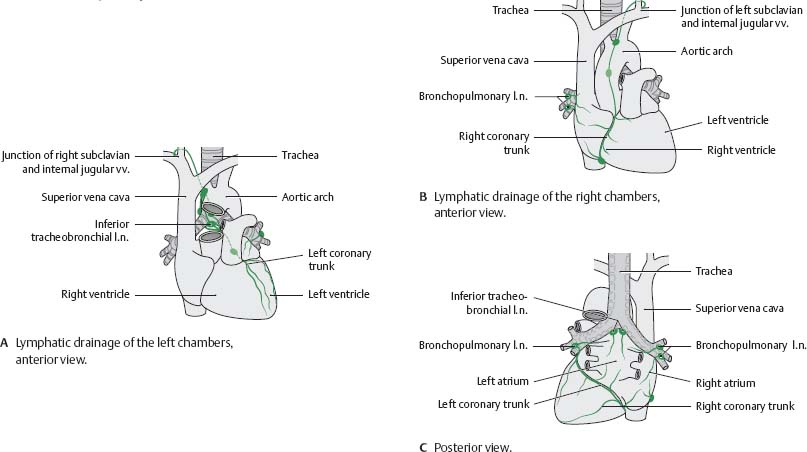
Fig. 7.33 Lymphatic drainage of the heart
A unique “crossed” drainage pattern exists in the heart: lymph from the left atrium and ventricle drains to the right venous junction, whereas lymph from the right atrium and ventricle drains to the left venous junction.
![]() The paraesophageal nodes drain the esophagus. Lymphatic drainage of the cervical part of the esophagus is primarily cranial, to the deep cervical lymph nodes and then to the jugular trunk. The thoracic part of the esophagus drains to the bronchomediastinal trunks in two parts: the upper half drains cranially, and the lower half drains inferiorly via the superior phrenic lymph nodes. The bronchopulmonary and paratracheal nodes drain lymph from the lungs, bronchi, and trachea into the bronchomediastinal trunk (see p. 118).
The paraesophageal nodes drain the esophagus. Lymphatic drainage of the cervical part of the esophagus is primarily cranial, to the deep cervical lymph nodes and then to the jugular trunk. The thoracic part of the esophagus drains to the bronchomediastinal trunks in two parts: the upper half drains cranially, and the lower half drains inferiorly via the superior phrenic lymph nodes. The bronchopulmonary and paratracheal nodes drain lymph from the lungs, bronchi, and trachea into the bronchomediastinal trunk (see p. 118).
Fig. 7.34 Mediastinal lymph nodes