Carolyn Chooljian
Deep venous thrombosis (DVT) of the lower extremity is a relatively common diagnosis made in the emergency department (ED), with an annual incidence of 67 per 100 000 in the United States. Two million patients are diagnosed with DVT and 600 000 have pulmonary embolism (PE) each year. Thromboembolic disease is responsible for an estimated 200 000 deaths annually and ranks third only to myocardial infarction and stroke as a life-threatening cardiovascular disease in the United States (1). Millions more experience sequelae such as stasis dermatitis and venous ulcers. The risk for development of DVT as described by Virchow is the triad of stasis, hypercoagulability, and endothelial injury. Additional conditions that either cross categories or separately place patients at risk are listed in Table 96.1. These risk factors are additive in nature and have been used to develop a prediction tool to establish likelihood of the disease in patients.
TABLE 96.1
Risk Factors for Deep Venous Thrombosis

The initiating stimulus for thrombus formation is often an intimal defect. The dynamic interplay of coagulation and fibrinolytic forces then results in clot propagation or dissolution. Conditions affecting the balance of these forces are what place patients at increased risk for symptomatic thrombus formation. If the process is of sufficient severity or duration, the associated vein can become inflamed; the condition is then referred to as thrombophlebitis.
It is useful to divide DVT of the lower extremities into two categories: distal and proximal. Distal DVT is confined to the calf veins and proximal DVT involves the popliteal, femoral, and/or iliac veins. Most DVTs start in the calf. Isolated calf DVT is usually silent and rarely causes clinically significant PE. About 25% of untreated symptomatic calf DVT progresses to the proximal veins within 1 week (2). Up to 40% of patients with DVT have clinically silent PE (3). PE occurs in 50% of proximal vein DVTs, which are the source of most fatal emboli, and over 90% of acute PE are from proximal lower extremity DVT (4). Although isolated superficial thrombophlebitis generally does not pose a significant risk for embolization, 23% of patients with symptoms of superficial thrombophlebitis have been shown to have involvement of the deep system as well (5).
Superficial thrombophlebitis involving the proximal saphenous vein or saphenous–femoral junction has a much higher risk of progression to DVT (6). In addition, patients with a first episode of DVT complicated by superficial thrombophlebitis have a 2-fold increased risk of recurrent DVT compared to patients without superficial thrombophlebitis (7). Although embolization to the lung is the most serious complication of DVT, it can also result in other complicating conditions. Venous hypertension and persistent clot can destroy valves, thereby leading to a postthrombotic syndrome (chronic leg pain and venous stasis) that occurs in 28% of patients after symptomatic DVT. Other patients develop only recurrent edema. Occasionally, acute cases can develop a compartment syndrome of the thigh or lower leg.
CLINICAL PRESENTATION
Patients with DVT can present with a symptom complex that ranges from completely occult to obvious. Most commonly, they present with lower extremity pain and swelling that may worsen with standing or walking. Manifestations are almost always unilateral, except in the rare life-threatening case of inferior vena cava (IVC) involvement. Generally, DVT symptoms develop over a period of several days, whereas sudden severe pain is more likely related to an injury. Associated symptoms, such as chest pain or dyspnea, may also be present, particularly as they relate to the presence of PE. Risk factors can be identified in the majority of patients.
DIFFERENTIAL DIAGNOSIS
Several medical conditions can present with symptoms suggestive of DVT (Table 96.2).
TABLE 96.2
Differential Diagnosis of Deep Venous Thrombosis

Many of these conditions generally cause bilateral leg swelling resulting from either increased production of edema fluid or an IVC obstructive process. However, if prior DVT has resulted in damage to the venous valves, these conditions can exacerbate asymmetric swelling. In addition, abdominal masses (e.g., a gravid uterus) can occasionally compress a single iliac vein, resulting in unilateral venous stasis. One of the most difficult conditions to distinguish from DVT is the postthrombotic syndrome. Recurrent DVT in the same leg predisposes to postthrombotic syndrome. Patients with prior DVT can develop recurrent unilateral pain and swelling that is clinically indistinguishable from acute DVT. Other conditions that are most often confused with DVT are cellulitis, superficial thrombophlebitis, and septic thrombophlebitis. There is much clinical overlap among these conditions, and a low threshold for objective diagnostic testing is required.
ED EVALUATION
The clinical evaluation of the patient with possible DVT focuses both on identifying findings consistent with DVT and on diagnosing alternative conditions that may explain the patient’s presentation. It is important to question the patient about risk factors for DVT (Table 96.1) including a family history. As many as one-fourth of patients with acute DVT have a history of prior DVT. The physical examination can provide useful findings but must be interpreted with caution. Although patients with cellulitis and other infections usually have a fever, DVT can also result in an elevated temperature, although often only low grade. Unilateral warmth and erythema are common with both DVT and cellulitis. The presence of an abscess, enlarged lymph nodes, and ulcerations can be important clues for distinguishing an infectious etiology. A difference of 3 cm in the circumference of the calves, measured 10 cm below the tibial tuberosity, or 3 cm in the circumference of the thighs, measured equally from the patella, is clinically significant and one of the most specific findings for DVT. The presence of palpable “cords” in the thigh is considered fairly specific but quite insensitive for thrombosis. In the popliteal fossa, they can easily be confused with a Baker cyst. Homan sign was classically taught as being important in the evaluation of DVT, but it is now well known that its sensitivity and specificity are only about 50%.
The assessment for DVT begins with an estimation of the probability of disease based on the presence of patient-based clinical variables. This can be combined with the measurement of a derivative of cross-linked fibrin, the D-dimer. D-Dimer levels are nonspecific, being elevated in DVT as well as infection, cancer, trauma, and other inflammatory states. Different D-dimer assays exist and can be divided into high-sensitivity (ELISA, second-generation latex agglutination-immunoturbidometric) and moderate-sensitivity (non-ELISA) forms (8,9).
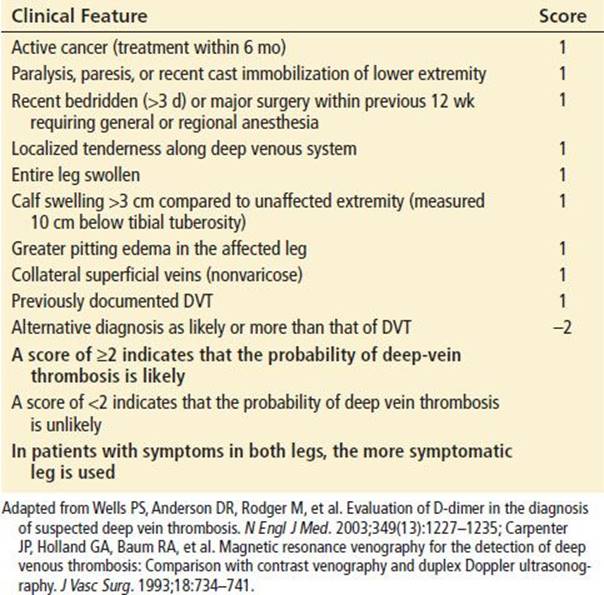
Wells et al. described a clinical predictive model with a relatively simple scoring system that accurately identified patients at low, intermediate, or high risk of DVT (10). This model was subsequently combined with D-dimer testing to further stratify patients’ risk of DVT (11,12). A simplification of the Wells model divided the probability estimate into two categories: DVT likely or DVT unlikely (see Table 96.3) (13). A meta-analysis showed that the combination of a low-probability clinical estimate and a negative high- or moderate-sensitivity D-dimer was sufficient to exclude DVT without further testing. In cases where the D-dimer assay used had high sensitivity, a negative result also reduced the probability of DVT to ≤1% in moderate-risk patients. No D-dimer assay was adequate to eliminate DVT in high-risk patients to this level of probability and therefore it is not recommended to use D-dimer alone in assessing these patients (10,14).
TABLE 96.3
Clinical Model for Predicting Pretest Probability of Deep Venous Thrombosis
Diagnostic studies for DVT include ultrasonography, contrast venography, impedance plethysmography, magnetic resonance venography (MRV), and computerized tomography venography (CTV). The primary imaging study to identify patients with DVT is ultrasonography. Lack of vein compressibility is highly sensitive (>95%) and specific (>95%) for proximal DVT. Ultrasound does not detect isolated thrombi in the iliac or femoral veins in the adductor canal. Ultrasound can also occasionally identify other conditions such as tumors, cysts, or aneurysms. In addition, the test is less accurate in the second half of pregnancy and for pelvic vein thrombus, for which the Doppler mode of the duplex ultrasound scan can be helpful. In these situations, or in patients felt to be at very high risk for DVT, it is recommended to use additional tests such as venography or close outpatient follow-up with serial ultrasound examinations within 5 to 7 days (14).
Patients with high clinical probability for DVT who have been evaluated and found to have a negative D-dimer and negative duplex ultrasound have a <1% probability of DVT and do not require serial ultrasound studies (10,15). However, if the initial ultrasound study is negative in a high-probability patient and the D-dimer is elevated, a repeat ultrasound should be obtained on day 5 to 7. MRV has a sensitivity of 100% and specificity 98% to 99% for pelvic and proximal DVTs, as well as 68% for calf DVTs. Advantages of MRV include the ability to distinguish between acute and chronic DVT, evaluate the pelvis and is the test of choice in pregnancy (16,17). CTV evaluates pelvis, thighs, and calves and has similar sensitivity and specificity to US for proximal DVT. It can be done with CT angiography for PE. CTV can diagnose pelvic and calf DVTs. Disadvantages of CT include radiation exposure and cost (18). DVT can also develop in the upper extremities, usually in the subclavian or axillary veins (19). Ultrasound may not be as sensitive as in the lower extremity, but it is still recommended as the first-line diagnostic test. Inadequate or negative examinations in high-risk patients may be followed with contrast venography.
KEY TESTING
• D-dimer
• Ultrasonography
ED MANAGEMENT
Patients with isolated DVT rarely require immediate lifesaving interventions. Treatment is important to prevent clot extension, acute PE, thrombosis recurrence, postthrombotic syndrome and chronic thromboembolic pulmonary hypertension. Many of these patients have occult PEs and all are at risk for new emboli. When a PE is strongly suspected, starting heparin prior to imaging studies is appropriate. In addition, occasional patients can present with complete venous obstruction, a condition referred to as “phlegmasia dolens.” These patients should also undergo immediate heparinization and fluid resuscitation prior to imaging studies when the diagnosis is suspected. They may also require thrombolytics and occasionally, urgent thrombectomy.
For most patients, the initial treatment for DVT is anticoagulation with a non-vitamin-K-dependent anticoagulant, usually a form of heparin for a short time, in addition to a vitamin K antagonist, for example, coumadin. Combination therapy was shown to be superior by a randomized controlled trial in which recurrent DVT had three times greater incidence if only a vitamin K antagonist was used (20). Heparin blocks extension of thrombus and reduces the risk of further emboli. It does not actively lyse clot but allows the body’s own fibrinolytic mechanism to operate over a period of several days to weeks. Heparin can be given in the unfractionated form, which is usually delivered by intravenous infusion or subcutaneously with one of the low-molecular-weight forms. Unfractionated heparin (UFH) is best dosed on a weight-based regimen. One of the most commonly used regimens is an 80-U/kg bolus followed by an 18-U/kg per hour infusion (21). Complex nomograms are reliable at attaining therapeutic anticoagulation adjustment while monitoring the aPTT. The efficacy of heparin therapy depends on achieving a critical therapeutic level of 1.5 to 2.5 above the normal aPTT (22).
Low-molecular-weight heparins (LMWHs) are fractionated versions of standard heparin that have a more predictable effect and have fewer side effects or complications. Compared to controls treated with UFH in the inpatient setting, these outpatient protocols have demonstrated very favorable outcomes with respect to short-term (2 weeks) events such as PEs, propagation of thrombus, and minor bleeding (23). Advantages over UFH include greater bioavailability, once or twice a day dosing, lack of need for laboratory monitoring except in certain circumstances (severe renal failure, pregnancy—which may require monitoring Factor anti-Xa levels), and lower risk of heparin-induced thrombocytopenia (HIT). Enoxaparin and dalteparin are two LMWHs used in the treatment of venous thromboembolism. The recommended dosing for these medications is as follows: enoxaparin, 1 mg/kg/dose every 12 hours or 1.5 mg/kg once daily subQ for inpatient treatment and 1 mg/kg/dose subQ every 12 hours for outpatient treatment. Dalteparin dosing is 200 U/kg once daily subQ or 100 units/kg twice daily subQ. These medications are predominantly eliminated by the kidneys. In patients with renal impairment and creatinine clearance >30 mL/min no specific adjustment is recommended, but in patients with severe renal failure lower doses should be used and anti-Xa levels monitored. Enoxaparin is not approved by the FDA for use in dialysis patients.
Fondaparinux is a synthetic heparin pentasaccharide analogue that binds to antithrombin and is given subcutaneously. It has been found to be effective in the treatment of venous thromboembolism (24). Dosing is weight based: <50 kg, 5 mg once daily; 50 to 100 kg, 7.5 mg once daily; >100 kg, 10 mg once daily. It should be adjusted in renal insufficiency and is contraindicated in severe renal failure. In patients with HIT, direct thrombin inhibitors (DTIs) should be used. Two direct thrombin inhibitors approved in the United States for use in patients with HIT are lepirudin and argatroban (25). Thrombolytic therapy has a role in treating limb-threatening thrombosis of the iliofemoral system, and can be considered in this setting, but is otherwise discouraged in the treatment of DVT (26).
Once a non-vitamin K anticoagulant has been initiated, warfarin (5 to 10 mg orally) may be begun, although this is generally not administered in the ED.
Those patients with absolute contraindications to anticoagulation require inferior vena caval interruption with an IVC filter (27). These devices are approximately 95% effective in preventing further PEs but have a 10% complication rate on insertion. However, this rate is not appreciably higher than that of full anticoagulation.
DVT below the knee should be managed in the same way as proximal DVTs. Similarly, there has been controversy regarding the ideal treatment of superficial thrombophlebitis of the leg. Though the danger of acute embolism may be less than that of calf DVT, propagation to the deep system must be ruled out at the time of the ED visit and close follow-up must be arranged. Compression bandages, anti-inflammatory drugs, and elevation are standard recommendations but anticoagulation should be considered in patients with a prior history of DVT. If the patient is an intravenous drug user or appears toxic, septic thrombophlebitis must be ruled out. Septic thrombophlebitis most commonly occurs at a site of intravenous catheter placement, and because the most common causative organism is Staphylococcus aureus, appropriate antibiotic therapy should be administered.
Postthrombotic syndrome occurs in 20% to 50% of patients with lower extremity DVT, usually within the first 2 years but has been seen as late as 8 years out (28). Endovascular interventions to reduce the thrombus burden have been shown reduce the incidence and severity of postthrombotic syndrome (29,30). Endovascular interventions may include catheter-directed thrombolysis and thrombosuction to prevent postthrombotic syndrome. The American College of Chest Physicians recommend consideration of catheter-directed thrombolysis or pharmacomechanical thrombolysis if appropriate expertise and resources are available for patients who have iliofemoral DVT, symptoms less than 14 days, good functional status, life expectancy > 1 year, and low risk of bleeding (31).
Patients referred for endovascular intervention consultation should be started on anticoagulation therapy in the ED.
CRITICAL INTERVENTIONS
• Anticoagulation with UFH, LMWH, or fondaparinux should be initiated in the ED.
• Traditional loading doses of heparin result in inconsistent anticoagulation of many patients. Weight-based dosing is more effective.
• LMWHs or fondaparinux are effective and allow outpatient therapy in appropriate cases.
• Isolated DVT distal to the knee is not as benign as previously thought. It should be treated more like proximal DVT in terms of therapeutic aggressiveness.
• Early consultation to consider catheter-directed thrombolysis or pharmacomechanical thrombolysis in appropriate patients with extensive acute proximal DVT in addition to anticoagulant therapy.
DISPOSITION
Historically, all patients with proximal lower extremity or upper extremity DVT required admission to the hospital. However, many institutions now have protocols by which a select subset of patients is discharged directly home from the ED on subcutaneous LMWH with close outpatient follow-up. This practice is supported by the American College of Physicians and American Academy of Family Physician guidelines if the required support systems are in place (17). Referral for possible endovascular intervention to prevent postthrombotic syndrome should be made early in the course. Outpatient treatment is not appropriate in cases of massive DVT (e.g., iliofemoral DVT), symptomatic PE, patients at high risk of bleeding, and those with other conditions who would require hospitalization. Conversely, the treatment of patients with DVT distal to the knee was historically similar to that of superficial thrombophlebitis. Now, with increasing concerns that these patients have risks for embolization that are only modestly lower than those patients with proximal DVT, it is recommended they be treated as those with proximal DVT or entered into a close outpatient follow-up protocol. Patients with phlegmasia dolens require more aggressive therapy. Emergent vascular surgery consultation and intensive care admission is indicated. Transport to a higher level of care should be considered if vascular surgery is unavailable.
Common Pitfalls
• Patients with a painful or swollen leg should be considered to have DVT until proven otherwise. Clinical evaluation alone is not sufficient to rule out the diagnosis.
• The traditional physical examination findings of DVT are notoriously inaccurate. The Homan test should be abandoned, and the presence of a palpable cord is not reliable.
• A negative highly sensitive D-dimer (ELISA, second generation latex agglutination-immunoturbidometric) test is useful only in patients with a low and intermediate clinical risk of DVT. It should not be used to exclude the diagnosis in high-risk patients. Moderate-sensitivity (non-ELISA) D-dimers are only adequate in assessing low clinical risk patients.
• A negative ultrasound combined with a negative moderate or high-sensitivity D-dimer leaves a <1% probability of DVT even in a high clinical probability patient. In these patients further ultrasound testing may not be needed (18).
REFERENCES
1. Cushman M, Tsai AW, White RH, et al. Deep venous thrombosis and pulmonary embolism in two cohorts: The longitudinal investigation of thromboembolism etiology. Am J Med. 2004;117:19–25.
2. Kearon C. Natural history of venous thromboembolism. Circulation. 2003;107:I22–I30.
3. Moser KM, Fedullo PF, LitteJohn JK, et al. Frequent asymptomatic pulmonary embolism in patients with deep venous thrombosis. JAMA. 1994;271:223–225.
4. Galanaud JP, Sevestre-Pietri MA, Bosson JL, et al. Comparative study on risk factors and early outcome of symptomatic distal versus proximal deep vein thrombosis: Results from the OPTIMEV study. Thromb Haemost.2009;102:493–500.
5. Jorgensen JO, Hanel KC, Morgan AM, et al. The incidence of deep venous thrombosis in patients with superficial thrombophlebitis of the lower limbs. J Vasc Surg. 1993;18:70–73.
6. Verlato F, Zucchetta P, Prandoni P, et al. An unexpectedly high rate of pulmonary embolism in patients with superficial thrombophlebitis of the thigh. J Vasc Surg. 1999;30:1113–1115.
7. Schonauer V, Kryle PA, Weltermann A, et al. Superficial thrombophlebitis and risk for recurrent venous thromboembolism. J Vasc Surg. 2003;37:834–838.
8. Stein PD, Hull RD, Patel KC, et al. D-Dimer for the exclusion of acute venous thrombosis and pulmonary embolism. A systematic review. Ann Intern Med. 2004;140:589–602.
9. Bates SM, Kearon C, Crowther M, et al. A diagnostic strategy involving a quantitative latex D-dimer assay reliably excludes deep venous thrombosis. Ann Intern Med. 2003;138(10):787–794.
10. Wells PS, Owen C, Doucette S, et al. Does this patient have deep vein thrombosis? JAMA. 2006;295:199–207.
11. Shields GP, Turnipseed S, Panacek EA, et al. Validation of the Canadian probability model for acute venous thrombosis. Acad Emerg Med. 2002;9:561–566.
12. Wells PS, Anderson DR, Bormanis J, et al. Value of assessment of pretest probability of deep-vein thrombosis in clinical management. Lancet. 1997;350:1795–1798.
13. Wells PS, Anderson DR, Rodger M, et al. Evaluation of D-dimer in the diagnosis of suspected deep-vein thrombosis. N Engl J Med. 2003;349:1227–1235.
14. Snow V, Qaseem A, Barry P, et al. Management of venous thromboembolism: A clinical practice guideline from the American College of Physicians and the American Academy of Family Physicians. Ann Intern Med.2007;146:204–210.
15. Moody J, Hafner J. Evidence-based emergency medicine/rational clinical examination abstract: The evidence-based diagnosis of deep venous thrombosis. Ann Emerg Med. 2009;54:461–464.
16. Carpenter JP, Holland GA, Baum RA, et al. Magnetic resonance venography for the detection of deep venous thrombosis: Comparison with contrast venography and duplex Doppler ultrasonography. J Vasc Surg. 1993;18:734–741.
17. Spritzer CE, Evans AC, Kay HH. Magnetic resonance imaging of deep venous thrombosis in pregnant women with lower extremity edema. Obstet Gynecol. 1995;85:603–607.
18. Garg K, Kemp JL, Wojcik D, et al. Thromboembolic disease: Comparison of combined CT pulmonary angiography and venography with bilateral leg sonography in 70 patients. Am J Roentgenol.2000;175:997–1001.
19. Ellis MH, Manor Y, Witz M. Risk factors and management of patients with upper limb deep vein thrombosis. Chest. 2000;117:43–46.
20. Brandjes DP, Heijboer H, Buller HR, et al. Acenocoumarol and heparin compared with acenocoumarol alone in the initial treatment of proximal-vein thrombosis. N Engl J Med. 1992;327:1485–1489.
21. Raschke RA, Reilly BM, Guidry JR. Weight-based heparin dosing nomogram compared with a standard care nomogram: A randomized controlled trial. Ann Intern Med. 1993;119:874–881.
22. Hull RD, Raskob GE, Brant RF, et al. The importance of initial heparin treatment on long-term clinical outcomes of antithrombotic therapy. Arch Intern Med. 1997;157:2317–2321.
23. Vinson DR, Berman DA. Outpatient treatment of deep venous thrombosis: A clinical care pathway managed by the emergency department. Ann Emerg Med. 2001;37:251–258.
24. Tran H, McRae S, Ginsberg J. Anticoagulant treatment of deep vein thrombosis and pulmonary embolism. Cardiol Clin. 2008;26:235–250.
25. Di Nisio M, Middeldorp S, Buller HR. Direct thrombin inhibitors. N Engl J Med. 2005;353:1028–1040.
26. American College of Emergency Physicians (ACEP) Clinical Policies Committee. Clinical policy: Critical issues in the evaluation and management of adult patients presenting with suspected lower-extremity deep venous thrombosis. Ann Emerg Med. 2003;42:124–135.
27. Ferris EJ, McCowan TC, Carver DK, et al. Percutaneous inferior vena caval filters: Follow-up of seven designs in 320 patients. Radiology. 1993;188:851–856.
28. Kahn SR. The post-thrombotic syndrome: The forgotten morbidity of deep venous thrombosis. J Thromb Thrombolysis. 2006;21:41–48.
29. Pollack CV Jr. Advanced management of acute iliofemoral deep venousthrombosis: Emergency department and beyond. Ann Emerg Med. 2011;57:590–599.
30. Imberti D, Maraldi C, Gallerani M. Interventional Approaches in VTE Treatment (Vena Cava Filters, Catheter-Guided Thrombolysis, Thrombosuction). Semin Respir Crit Care Med. 2012;33:176–185.
31. Kearon C et al. Antithrombotic therapy for venous thromboembolic disease: American College of Chest Physicians Evidence-Based Clinical Practice Guidelines (8th Edition). Chest. 2008;133:454S–545S.