Robert J. Harrison, MD, MPH
ACIDS & ALKALIS
Acids and alkalis are of great importance as industrial chemicals. When ranked by volume of production, the inorganic acids and alkalis (including chlorine and ammonia) are 8 of the 50 major chemicals produced yearly in the United States.
1. Acids
ESSENTIALS OF DIAGNOSIS
![]() Acute Effects
Acute Effects
• Irritative dermatitis, skin burn.
• Respiratory irritation, pulmonary edema.
![]() Chronic Effects
Chronic Effects
• Hydrofluoric acid: osteosclerosis.
• Nitric acid (oxides of nitrogen): bronchiolitis fibrosaobliterans.
• Chromic acid: nasal ulceration, perforation, skin ulceration.
• Sulfuric acid: laryngeal cancer.
![]() General Considerations
General Considerations
An inorganic acid is a compound of hydrogen and one or more other elements (with the exception of carbon) that dissociates to produce hydrogen ions when dissolved in water or other solvents. The resulting solution has the ability to neutralize bases and turn litmus paper red. Inorganic acids of greatest industrial use are chromic, hydrochloric, hydrofluoric, nitric, phosphoric, and sulfuric acids. Inorganic acids share certain fire, explosive, and health hazards.
Organic acids and their derivatives include a broad range of substances used in nearly every type of chemical manufacture. All have primary irritant effects depending on the degree of acid dissociation and water solubility.
![]() Use, Production, & Occupational Exposure
Use, Production, & Occupational Exposure
A. Inorganic Acids
1. Sulfuric acid—Sulfuric acid is the leading chemical in production volume. It is less costly than any other acid, can be handled easily, reacts with many organic compounds to produce useful products, and forms a slightly soluble salt with calcium oxide or calcium hydroxide. The majority of sulfuric acid is used in the manufacture of phosphate and other fertilizers, petroleum refining, production of ammonium sulfate, iron and steel pickling, manufacture of explosives and other nitrates, synthetic fiber manufacture, and as a chemical intermediate. Workers with potential exposure to sulfuric acid include electroplaters, jewelers, metal cleaners, picklers, and storage-battery makers. Occupational exposure can occur both by skin contact and by inhalation of sulfuric acid mist.
2. Phosphoric acid—Phosphoric acid is used predominantly in the manufacture of fertilizers and agricultural feeds, in water treatment, and as a component of detergents and cleansers. Other uses include the acid treatment (pickling) of sheet metal, chemical polishing of metals, as a tart flavoring agent for carbonated beverages, as a refractory bonding agent, and for boiler cleaning, textile dying, lithographic engraving, and rubber latex coagulation. Occupational exposure occurs primarily to the liquid acid by skin contact.
3. Chromic acid—Chromic acid is produced by roasting chromite ore with soda ash and treatment with sulfuric acid to form chromic acid anhydride, chromic acid (chromium trioxide), and dichromic acid. Chromic acid is used in chromium plating, process engraving, cement manufacturing, anodizing, metal cleaning, tanning, and the manufacture of ceramic glazes, colored glass, inks, and paints. Without local exhaust ventilation, occupational exposure to chromic acid mist during metal-plating operations can range up to several milligrams per cubic meter, but with a local exhaust system, this can be reduced markedly to nearly undetectable limits.
4. Nitric acid—Nitric acid is produced from the oxidation of ammonia in the presence of a catalyst to yield nitric oxide, which is then further oxidized and absorbed in water to form an aqueous solution of nitric acid. Nitric acid is used to produce ammonium and potassium nitrate, explosives, adipic acid, isocyanates, fertilizers, nitroparaffins, and nitrobenzenes. Occupational exposure can occur by topical contact with the liquid acid, as well as by inhalation of nitrogen oxides evolved when nitric acid reacts with reducing agents (eg, metals or organic matter) or during the combustion of nitrogen-containing materials (eg, welding, glass blowing, underground blasting, and decomposition of agricultural silage). Reports of occupational exposure to nitric acid are limited to measurements of nitrogen oxides that evolved by these reactions.
5. Hydrochloric acid—Hydrochloric acid is an aqueous solution of hydrogen chloride and is used in steel pickling, chemical manufacturing, oil- and gas-well acidizing, and food processing. Hydrochloric acid gas also may evolve from thermal degradation of polyvinyl chloride, a hazard to firefighters.
6. Hydrofluoric acid—Hydrofluoric acid (hydrogen fluoride) is a colorless liquid manufactured by reaction of sulfuric acid with calcium fluoride in heated kilns. It evolves as a gas and then is condensed as liquid anhydrous hydrogen fluoride. Hydrofluoric acid is used as an intermediate in the production of fluorocarbons, aluminum fluoride, and cryolite; as a gasoline alkylation catalyst; and as an intermediate in the production of uranium hexafluoride. It is used in metal cleaning, glass etching, and polishing applications. Occupational exposure can occur both by direct skin contact and by inhalation of fumes.
7. Organic acids—Among the saturated monocarboxylic acids, formic acid is used mainly in the textile industry as a dye-exhausting agent, in the leather industry as a deliming agent and neutralizer, as a coagulant for rubber latex, and as a component of nickel plating baths. Propionic acid is used in organic synthesis, as a mold inhibitor, and as a food additive. The unsaturated monocarboxylic acid acrylic acid is used widely in the manufacture of resins, plasticizers, and drugs. The aliphatic dicarboxylics maleic, fumaric, and adipic acids find use in the manufacture of synthetic resins, dyes, surface coatings, inks, and plasticizers. The halogenated acetic acids are highly reactive chemical intermediates used in glycine, drug, dye, and herbicide manufacture. Glycolic acid and lactic acid are used widely in the leather, textile, adhesive, and plastics industries, and lactic acid is also used as a food acidulant.
![]() Metabolism & Mechanism of Action
Metabolism & Mechanism of Action
Both inorganic and organic acids, by virtue of their water solubility and acid dissociation, will cause direct destruction of body tissue, including mucous membranes and skin. The extent of direct skin damage depends on the concentration of acid and length of exposure, whereas the damage to the respiratory tract by inhalation of acid mists will depend in addition on particle size. Hydrofluoric acid, one of the most corrosive of the inorganic acids, readily penetrates the skin and travels to deep tissue layers, causing liquefaction necrosis of soft tissues and decalcification and corrosion of bone. The intense pain that may accompany hydrogen fluoride burns is attributed to the calcium-precipitating property of the fluoride ion, which produces immobilization of tissue calcium and an excess of potassium that stimulates nerve endings. The fluoride ion also may bind body calcium, causing life-threatening systemic hypocalcemia after acute skin exposure or osteosclerotic bone changes after chronic exposure to hydrogen fluoride mist.
![]() Clinical Findings
Clinical Findings
A. Symptoms and Signs
1. Acute exposure—All acids act as primary irritants of the skin and mucous membranes.
A. SKIN—All acids on contact with the skin cause dehydration and heat release to produce first-, second-, or third-degree burns with pain. Sensitization is rare. Hydrofluoric acid solutions of less than 50% may cause burns that may not become apparent for 1–24 hours; stronger solutions cause immediate pain and rapid tissue destruction, appearing reddened, pasty-white, blistered, macerated, or charred.
B. RESPIRATORY EFFECTS—Inhalation of vapors or mists causes immediate rhinorrhea, throat burning, cough, burning eyes, and conjunctival irritation. High concentrations may cause shortness of breath, chest tightness, pulmonary edema, and death from respiratory failure. Inhalation of acid vapors or mists generally causes immediate symptoms because of high water solubility in mucous membranes, but respiratory effects may be delayed for several hours. Noncardiogenic pulmonary edema has been reported following acute inhalation exposure to sulfuric acid fumes, with almost complete recovery except for slightly decreased diffusion capacity on pulmonary function testing. For nitric acid exposure with oxides of nitrogen, overexposure tends to produce delayed symptoms 1–24 hours after inhalation, beginning with dyspnea followed by pulmonary edema and cyanosis. Rapidly progressive pulmonary edema of delayed onset may follow the inhalation of fumes from accidental nitric acid exposure. In these cases, postmortem electron microscopy of lung tissue suggests increased permeability as a result of microvascular injury.
Chlorine species are highly reactive, resulting in a variety of dose-related lung effects ranging from respiratory mucous membrane irritation to pulmonary edema. Obstructive or restrictive pulmonary defects can result immediately following exposure, with complete resolution over a few days to weeks in most individuals. A few patients have long-term, persistent obstructive or restrictive pulmonary deficits or increased nonspecific airway reactivity after high-level exposure to chlorine gas.
Exposure to lower levels of acid vapors or mists over months may increase the risk of developing irritant-associated asthma. Aluminum potroom workers with exposure to fluorides have an increased risk of respiratory symptoms, with a greater prevalence of airway responsiveness as measured by nonspecific airway challenge. Occupational asthma also has been reported following exposure to chloramines in indoor swimming pool air.
C. SYSTEMIC EFFECTS—Several deaths have been reported as a result of persistent hypocalcemia and hypomagnesemia following exposure to concentrated hydrofluoric acid, with the exposures involving as little as 2.5% of total body surface area. Systemic toxicity involving gastrointestinal hemorrhage, acute renal failure, and hepatic injury has been reported following chromic acid ingestion.
2. Chronic exposure—
A. SKIN—Chromate compounds can be allergens and can cause pulmonary as well as skin sensitization, but chromic acid results only in direct irritant dermatitis. Ulceration of the skin and ulceration and perforation of the nasal septum have been reported following chronic exposure to chromic acid.
B. DENTAL EROSION—Exposure to inorganic and organic acid fumes is reported to cause tooth surface loss. An increase in periodontal pockets but not oral mucous membrane lesions was found among acid-exposed workers.
C. RESPIRATORY EFFECTS—Bronchiolitis fibrosaobliterans, a chronic interstitial lung disease, has been described after acute pneumonitis from nitric acid and oxides of nitrogen. No significant change in lung function has been found among workers exposed to phosphoric acid while refining phosphorus. Acids and a variety of other irritants have been recognized to cause vocal cord dysfunction, with chronic symptoms of hoarseness and loss of voice.
D. SYSTEMIC EFFECTS—Osteosclerosis has been found in workers exposed to hydrofluoric acid and fluoride-containing compounds. Farmers with formic acid exposure have increased renal ammoniagenesis and urinary calcium excretion, possibly as a result of interaction with the oxidative metabolism of renal tubular cells.
E. CANCER—Studies of workers exposed to sulfuric acid mists show an excess risk of laryngeal and nasopharyngeal cancer. The International Agency for Research on Cancer (IARC) concludes that there is sufficient evidence that occupational exposure to strong inorganic acid mists containing sulfuric acid is carcinogenic (group 1). Battery manufacturers and steel workers exposed to mineral acid mists have an increased risk of upper aerodigestive tract cancer. For chromic acid, IARC concludes that there is sufficient evidence of carcinogenicity in humans and animals (group 1). Airborne hexavalent chromium exposure results in an increased risk of lung cancer among chromium platers. National Institute for Occupational Safety and Health (NIOSH) recommends that chromic acid be regulated as a carcinogen. An increase in the number of sister-chromatid exchanges has been found in lymphocytes of workers exposed to acid aerosols at a phosphate fertilizer factory. IARC finds that hydrochloric acid is not classifiable in terms of carcinogenicity to humans (group 3). The cancer risk was not increased among cohorts of chemical manufacturing workers exposed to hydrogen chloride and nitric acid.
B. Laboratory Findings
In cases where inhalation exposure may cause more extensive mucosal irritation, the chest radiograph may show interstitial or alveolar edema, and hypoxemia may be evident by arterial blood gas analysis. Nonspecific abnormalities in liver and kidney function have been reported following massive inhalation exposures to sulfuric acid and hydrofluoric acid. Urine fluoride levels can be used as biologic indices of exposure in hydrofluoric acid intoxication, with a normal mean value in urine of 0.5 mg/L (recommended occupational post-shift urinary biologic standard of 7 mg/L).
![]() Differential Diagnosis
Differential Diagnosis
There are many respiratory irritants, including gases such as ammonia, phosgene, halogens (chlorine, bromine), sulfur dioxide, and ozone; solvents such as glycol ethers; and dusts such as fibrous glass. The symptoms and clinical course of lung disorders caused by these substances and by the acids discussed in this chapter do not differ; thus the history is essential. Likewise, hundreds of industrial chemicals may cause direct irritant dermatitis.
![]() Prevention
Prevention
A. Work Practices
When possible, highly corrosive acids should be replaced by acids that present fewer hazards, and if use of corrosives is essential, only the minimum concentration should be used. Proper storage practices should include fire-resistant buildings with acid-resistant floors, retaining sills, and adequate drainage; containers should be adequately protected against impact, kept off the floor, and labeled clearly. Wherever possible, handling should be done through sealed systems or the substances transported in safety-bottle carriers. Decanting should be done with special siphons or pumps. The potential for violent or dangerous reactions (eg, when water is poured into nitric acid) can be avoided by appropriate training.
Where processes produce acid mists (as in electroplating), local exhaust ventilation should be installed. Workers potentially exposed to splashes or spills must wear acid-resistant hand, arm, eye, and face protection, and respiratory protection should be available for emergency use. Emergency showers and eyewash stations should be strategically located.
B. Medical Surveillance
Preplacement and periodic examinations should include medical history of skin and respiratory disease and examination of the skin, teeth, and lungs. For potential hydrofluoric acid exposure near or above the permissible exposure limit, periodic postshift urinary fluoride in excess of 7 mg/L (adjusted for urine specific gravity of 1.024) may indicate poor work practices. Elemental analysis of hair for fluoride has been correlated with fluoride levels in serum and urine.
![]() Treatment
Treatment
Immediate on-site first aid treatment of acid burns to the eye or skin includes copious flushing with running water with removal of all contaminated clothing. First- or second-degree burns involving a small area generally can be treated at the on-site medical facility with debridement and application of suitable burn dressings. All other acid burns should be treated at a hospital emergency facility.
For hydrofluoric acid burns, the definitive treatment is aimed at deactivation of the fluoride ion in tissue with calcium, magnesium, or quaternary ammonium solution. If the hydrogen fluoride concentration is 20% or more, if the patient has been exposed to a long delay of a lower concentration, or if a large tissue area has been affected by a lower concentration, then calcium gluconate solution should be used. Calcium gluconate solution is prepared by mixing 10% calcium gluconate with an equal amount of saline to form a 5% solution and is infiltrated with a small needle in multiple injections (0.5 mL/cm2 of tissue) into and 5 mm beyond the affected area. Dramatic pain relief should occur. Vesicles and bullae should be debrided carefully, with removal of necrotic tissue; if periungual or ungual tissues are involved, the nail should be split to the base. A burn dressing then is applied along with calcium gluconate 2.5% gel or magnesium sulfate paste. Hydrofluoric acid burns of the hand have been treated successfully with repeated application of an occlusive glove over topical calcium carbonate gel. Repeated intra-arterial infusion over 4 hours with 10 mL of 10% calcium chloride diluted with 40 mL of normal saline also has been recommended for the treatment of hydrofluoric acid extremity burns. Careful monitoring of serum magnesium and calcium levels is required. If the hydrogen fluoride concentration is 20% or less and only a small surface area is involved, the burn can be flushed with water and then treated with 10% magnesium sulfate solution under a soft dressing. The eye burned with hydrogen fluoride should be irrigated copiously and then evaluated by an ophthalmologist. Calcium gluconate 1% in normal saline can be used as an irrigant.
Systemic effects from absorption should be anticipated from skin burns from hydrogen fluoride of greater than 50% concentration or from extensive burns at any concentration. Hypocalcemia can be life-threatening and should be monitored by repeated measurement of serum calcium and electrocardiography for QT-interval prolongation. Calcium gluconate 10% intravenously with adequate hydration should be used for calcium depletion.
For inhalation of acid vapors or mists, the victim should be removed immediately from the source of exposure and treated on-site with 100% oxygen. If there are symptoms of shortness of breath, chest tightness, or persistent cough, the patient should be evaluated at the hospital. Patients who are minimally symptomatic with normal peak expiratory flow rate and oxygen saturation values can be discharged from the emergency department after several hours of observation and instructed to return if dyspnea occurs. Upper body or facial burns are a clue that inhalation may have occurred with possible serious lower airway damage. Evaluation should include a chest radiograph and arterial blood gas analysis for oxygen. Hypoxemia should be treated with 100% oxygen by mask or by intubation in the event of severe hypoxemia, acidosis, or respiratory distress. Fluid balance should be monitored carefully and intracardiac pressure measured directly if necessary. Bronchospasm may be treated with inhaled bronchodilators or intravenous aminophylline and steroids if necessary. The benefits of steroids in the management of noncardiogenic pulmonary edema caused by acid inhalation are unknown, but the drugs may be used empirically to speed recovery and prevent the subsequent development of interstitial lung disease. Nebulized calcium gluconate 5% solution has been used successfully for treatment of inhalational exposure to hydrofluoric acid.
2. Alkalis
ESSENTIALS OF DIAGNOSIS
![]() Acute effects
Acute effects
• Skin and eye burns.
• Respiratory irritation.
![]() Chronic effects
Chronic effects
• Corneal opacities of the eye (untreated).
• Obstructive lung disease.
![]() General Considerations
General Considerations
Alkalis are caustic substances that dissolve in water to form a solution with a pH higher than 7. These include ammonia, ammonium hydroxide, calcium hydroxide, calcium oxide, potassium hydroxide, potassium carbonate, sodium hydroxide, sodium carbonate, and trisodium phosphate. The alkalis, whether in solid form or concentrated liquid form, are more destructive to tissue than most acids. They tend to liquefy tissues and allow for deeper penetration, depending on concentration, duration of contact, and area of the body involved.
![]() Use, Production, & Occupational Exposure
Use, Production, & Occupational Exposure
In the United States, all sodium hydroxide (caustic soda) is produced by the electrolysis of sodium or potassium chloride in mercury cells. In this process, pure saturated brine is decomposed by electric current to liberate chlorine gas at the anode and sodium metal at the cathode. The latter reacts with water to form sodium hydroxide. Most caustic soda is produced as a 50% aqueous solution. Sodium hydroxide is used in pulp and paper production, water treatment, and manufacture of a wide variety of organic and inorganic chemicals, soaps and detergents, textiles, and alumina. Annual US production is more than 22 billion pounds.
Sodium carbonate (soda ash) is produced by the ammonium chloride process, by the reaction of sodium chloride and sulfuric acid, or by leaching out of rock deposits. Sodium carbonate is used in glass manufacturing, as a component of cleaning-product formulations, in pulp and paper processing and water treatment, and as a chemical intermediate.
Potassium carbonate (potash) is produced by carbonating potassium hydroxide solutions obtained by electrolysis. Potassium carbonate is used in the manufacture of soap, glass, pottery, and shampoo; in tanning and finishing leather; in photographic chemicals, fire-extinguishing compounds, and rubber antioxidant preparations; and as an alkalizer and drainpipe cleaner.
Potassium hydroxide (caustic potash) is produced by electrolysis of potassium chloride solution and is used as a chemical intermediate in the manufacture of potassium carbonate, potassium phosphate, soaps, tetrapotassium pyrophosphate, liquid fertilizers, dyestuffs, and herbicides.
Calcium oxide (quicklime) is made by calcining limestone. Calcium oxide is used in metallurgy as a flux in steel production, for ammonia recovery in the Solvay process for sodium carbonate, in construction applications and water purification and softening, in beet and sugar cane refining, in kraft paper pulp production, and in sewage treatment.
![]() Metabolism & Mechanism of Action
Metabolism & Mechanism of Action
Occupational exposure to the alkalis is primarily by direct contact with the eyes, skin, and mucous membranes.
Inhalation of caustic mists generally is limited by the irritant properties of the compound. Contact of the eyes with alkalis causes disintegration and sloughing of corneal epithelium, corneal opacification, marked edema, and ulceration. Alkaline compounds will combine with skin tissue to form albuminates and with natural fats to form soaps. They gelatinize tissue and result in deep and painful destruction. Accidental or intentional ingestion of alkalis may cause severe esophageal necrosis with subsequent stenosis.
![]() Clinical Findings
Clinical Findings
A. Symptoms and Signs
1. Acute exposure—In contrast to acids, skin contact with the alkalis may not elicit immediate pain but may start to cause immediate damage with erythema and tissue necrosis within minutes to hours. Splashes of alkali to the eyes, if not treated within minutes, may result in corneal necrosis, edema, and opacification.
Irreversible obstructive lung injury has developed after acute inhalation of sodium hydroxide in a poorly ventilated space. Workers have suffered severe skin and inhalational injuries following exposure to “black liquor” used in the pulp and paper industry. Fatal injury has occurred after a relatively brief inhalation and dermal contact with a hot concentrated caustic solution. Acute tracheobronchitis and respiratory failure as a result of high-dose ammonia inhalation may result in permanent, severe, and fixed airways obstruction. Bronchiolitis obliterans caused by occupational exposure to incinerator fly ash has been reported.
2. Chronic exposure—Chronic exposure to caustic dusts does not increase the mortality rate significantly. Long-term sodium hydroxide inhalation has been reported to cause severe obstructive airway disease with significant air trapping. Chronic exposure to ammonia of over 7.5 ppm is associated with pulmonary function decrements among swine production facility workers. An increased prevalence of coughing, wheezing, and ocular and nasal irritation was reported among community residents exposed to alkali dust. Corneal opacities have resulted from untreated corneal alkali burns. An increased risk of nasopharyngeal carcinoma has been observed among Chinese textile workers exposed to acid and caustics.
B. Laboratory Findings
No specific laboratory tests are of value in the diagnosis and management of problems resulting from alkali exposure.
![]() Differential Diagnosis
Differential Diagnosis
Many other industrial chemicals, including acids, may cause eye and skin burns.
![]() Prevention
Prevention
A. Work Practices
Insofar as possible, solutions of caustics should be handled in closed systems that will prevent contact with or inhalation of the chemical. All persons with potential exposure to caustics should wear proper protective clothing and equipment, such as a full-face shield, safety goggles, apron or suit, rubber gloves, and boots. Emergency showers and eyewashes must be located where eye or skin contact may occur.
B. Medical Surveillance
Medical examination of the eyes, skin, and respiratory tract is recommended for all workers with caustic exposure.
![]() Treatment
Treatment
Sodium and potassium hydroxide may cause more extended and deeper damage as a result of rapid penetration through ocular tissues. Alkali burns of the eye and skin should be treated within minutes by copious irrigation with tap water and removal of all contaminated clothing. Irrigation with a weak acid such as 5% acetic acid also has been suggested. First aid treatment with prompt and continuous eye irrigation is essential to prevent permanent corneal damage and visual loss. Topical use of a synthetic metalloproteinase inhibitor has been shown to reverse or stop the progression of corneal ulceration following an experimental alkali burn. A relatively new hypertonic, polyvalent, amphoteric chelating compound (Diphoterine) also appears to be of benefit for emergent eye and skin decontamination. A physician or health practitioner should be consulted for eye burns and careful examination of the eye performed. If eye damage is suspected, follow-up with an ophthalmologist is recommended. Intensive topical steroids, antibiotics, and amniotic membrane transplantation may be required.
ACRYLAMIDE & ACRYLONITRILE
1. Acrylamide
ESSENTIALS OF DIAGNOSIS
![]() Acute effects
Acute effects
• Dermatitis.
![]() Chronic effects
Chronic effects
• Peripheral neuropathy.
![]() General Considerations
General Considerations
Pure acrylamide is a white crystalline solid at room temperature and is highly soluble in water. It is a vinyl monomer with high reactivity with thiols and with hydroxy and amino groups. Commercial acrylamide is shipped in 50% aqueous form in stainless steel drums, tank trucks, and cars. Acrylamide manufacture is from the catalytic hydration of acrylonitrile.
![]() Use
Use
The major use of acrylamide monomer is in the production of polymers, which are useful as flocculators. Polyacrylamides are used for waste and water treatment flocculants, in products for sewage dewatering, and in a variety of products for the water treatment industry. Other uses include strengtheners for papermaking and retention aids, drilling-mud additives, textile treatment, and surface coatings. One of the more important uses is as a grouting agent, particularly in mining and tunnel construction.
![]() Occupational & Environmental Exposure
Occupational & Environmental Exposure
Monomer manufacturing workers are potentially exposed to acrylamide, as are papermaking workers, soil-stabilization workers, textile workers, tunnel workers, and well drillers. Biomedical laboratory workers can be exposed to arcylamide used to make polyacrylamide gels. Intoxication has been reported in the manufacture of acrylamide monomer, in the handling of a 10% aqueous solution in a mine, in the production of flocculators, in the use of a resin mixture containing residual monomer, and in the production of polymers while manufacturing paper coating materials. One nonoccupational incident occurred in Japan, where a family ingested well water containing 400 ppm acrylamide.
Acrylamide may be formed at elevated temperatures in cooking, particularly of carbohydrate-rich foods such as potatoes (eg, crisps, chips, and fries). Residual levels of acrylamide also can be found in cosmetic products.
![]() Metabolism & Mechanism of Action
Metabolism & Mechanism of Action
Acrylamide is absorbed easily in animals following all routes of administration. The peripheral nerve terminal is a primary site of acrylamide action, with possible inhibition of membrane-fusion processes impairing neurotransmitter release. Quantitative data on absorption or excretion in humans are not available. Following intravenous administration in rats, acrylamide is distributed throughout total body water within minutes and then excreted largely in the urine with a half-life of less than 2 hours. Protein-bound acrylamide or acrylamide metabolites have a half-life in blood and possibly in the central nervous system of about 10 days. The primary metabolite of acrylamide is N-acetyl-S-(3-amino-3-oxypropyl) cysteine, and it is excreted predominantly in the urine.
![]() Clinical Findings
Clinical Findings
A. Symptoms and Signs
Acrylamide polymer may cause dermatitis but does not cause neurotoxicity. The monomer can produce numbness and tingling of hands and weakness of the hands and legs. Acrylamide is neurotoxic in many experimental animals, causing distal axonopathy and central neuronal degeneration.
More than 60 cases of acrylamide-associated neurotoxicity have been reported in humans. Subclinical peripheral neuropathy has been found in tunnel workers exposed to acrylamide during grouting work. Similar to the neuropathy associated with the hexacarbons n-hexane and methyl-n-butyl ketone, acrylamide neuropathy is considered a typical example of a dying-back disorder, where degeneration begins at the distal ends of the longest and largest fibers and spreads proximally. In most cases, toxicity results from skin contact and dermal absorption, although acrylamide may be absorbed by inhalation as well. The cellular and molecular site of acrylamide neurotoxicity may involve alterations in fast anterograde transport or sulfhydryl groups on presynaptic proteins. The neurologic features of acrylamide intoxication vary depending on the speed of intoxication. In the Japanese family that ingested contaminated well water, encephalopathy with confusion, disorientation, memory disturbances, hallucinations, ataxia, and peripheral neuropathy developed in approximately 1 month. Reported time to onset of symptoms in occupational cases has varied from 4 weeks to approximately 24 months. Clinically, acrylamide peripheral neuropathy affects both motor and sensory nerve fibers predominantly in the distal limbs. Difficulty in walking and clumsiness of the hands are usually the first symptoms, followed by numbness of the feet and fingers. Distal weakness is found on examination, with loss of tendon reflexes and vibration sensation. Evidence of excessive sweating affecting predominantly the extremities has been reported commonly, along with redness and exfoliation of the skin. In acute cases, central nervous system involvement may result in truncal ataxia, lethargy, and dysarthria. Major histologic findings are swelling of axons and/or a decrease in large-diameter axons. The axonopathy is reversible slowly over time, but complete recovery depends on the severity of intoxication.
Acrylamide has been found to increase the tumor yield in mice and is genotoxic in animal studies. Acrylamide reacts with hemoglobin to form DNA adducts and heritable translocations in animal studies. Human studies have shown increased DNA adducts and chromosomal aberrations among workers exposed to acrylamide. Cohort mortality studies have shown no significant excess of cancer among acrylamide-exposed workers, and initial studies suggest that there is not an excess of cancer owing to consumption of acrylamide in food. There is some evidence to suggest that acrylamide results in adverse developmental or reproductive effects in animal studies.
The State of California has listed acrylamide as a carcinogen since 1990. The IARC has concluded that there is sufficient evidence in experimental animals for acrylamide to be classified as a carcinogen (group 2A).
B. Laboratory Findings
Electrophysiologic studies of workers with signs and symptoms of neurotoxicity have shown only a slight effect on maximal conduction velocity of either motor or sensory fibers. Sensory nerve action potentials usually are reduced and are the most sensitive electrophysiologic test.
Sural nerve biopsies performed on two patients during recovery from acrylamide neuropathy showed axonal degeneration affecting mainly large-diameter fibers. Recent studies have suggested the use of urinary S-carboxyethyl cysteine and mercapturic acid metabolites of acrylamide for biomonitoring use in the workplace and general populations, respectively.
![]() Differential Diagnosis
Differential Diagnosis
The combination of truncal ataxia with peripheral neuropathy—predominantly motor—accompanied by excessive sweating and redness and peeling of the skin makes the diagnosis of acrylamide-associated neurotoxicity likely. Other occupational toxic agents associated with peripheral neuropathy must be considered (see Chapter 27), along with the presence of other underlying metabolic diseases, drug use, and endocrine disorders.
![]() Prevention
Prevention
A. Work Practices
Mechanized bag loading of polymerization reactors, closed-line transfer of liquid acrylamide, and other closed-system processes are important to minimize exposure. Where necessary, personal protective equipment designed to prevent dermal and inhalation exposure to acrylamide should be available. General population consumption of acrylamide in foods can be reduced by changes in food-manufacturing raw materials and preparation methods.
B. Medical Surveillance
Preplacement and periodic examinations should exclude symptomatic peripheral neuropathies. Hemoglobin adducts have been used to monitor occupational exposure to both acrylamide and acrylonitrile. A neurotoxicity index involving electrophysiologic measures was correlated with urinary 24-hour mercapturic acid levels, hemoglobin adducts of acrylamide, employment duration, and vibration sensitivity. Vibration threshold may be a sensitive indicator of early neurotoxicity caused by acrylamide exposure.
![]() Treatment
Treatment
Skin contaminated with acrylamide should be washed immediately with soap and water, and contaminated clothing should be removed. There is no known treatment for acrylamide intoxication. Removal from exposure is the only effective measure that can be taken. Full recovery has been observed in most cases after 2 weeks to 2 years, although in severe cases some residual neurologic abnormalities have been noted.
2. Acrylonitrile
ESSENTIALS OF DIAGNOSIS
![]() Acute effects
Acute effects
• Respiratory irritation, nausea, dizziness, and irritability, followed by convulsions, coma, and death.
![]() Chronic effects
Chronic effects
• Nausea, dizziness, headache, apprehension, fatigue
![]() General Considerations
General Considerations
Acrylonitrile is a volatile colorless liquid with a characteristic odor resembling that of peach seeds, discernible at 20 ppm or less. It is a highly reactive compound. Pure acrylonitrile polymerizes readily in light, and storage requires the addition of polymerization inhibitors. Its vapors are explosive and flammable and may release hydrogen cyanide on burning.
![]() Use
Use
Acrylonitrile was not an important product until World War II, when it was used in the production of oil-resistant rubbers. Nearly all world production of acrylonitrile is now based on a process where propylene, ammonia, and air react in the vapor phase in the presence of a catalyst. Hydrogen cyanide and acrylonitrile are the chief by-products formed; the latter undergoes a series of distillations to produce acrylonitrile.
Much of acrylonitrile monomer is used for the manufacture of acrylic fibers for the apparel, carpeting, and home furnishings industries. Acrylonitrile-containing plastics, particularly the resins acrylonitrile-butadiene-styrene (ABS) and styrene-acrylonitrile (SAN), are used in pipe and pipe fittings, automotive parts, appliances, and building components. Nitrile elastomers are used for their oil- and hydrocarbon-resistant properties in the petrochemical and automobile industries. Acrylonitrile is also used to make acrylamide.
![]() Occupational & Environmental Exposure
Occupational & Environmental Exposure
Potential exposure to acrylonitrile may occur in monomer-, fiber-, resin-, and rubber-producing plants. Potential exposure to acrylonitrile in acrylic fiber production is greatest when the solvent is removed from newly formed fibers and during decontamination of acrylonitrile processing equipment, loading, surveillance of the processing unit, and product sampling.
![]() Metabolism & Mechanism of Action
Metabolism & Mechanism of Action
Acrylonitrile is absorbed readily in animals following ingestion or inhalation. There is a biphasic half-life of 3.5 hours and 50–77 hours, with elimination predominantly in the urine. Acrylonitrile is metabolized to cyanide, and its metabolites are eliminated in the urine. In humans, absorption can occur through both inhalation and skin contact. The acute toxicity of acrylonitrile in humans is thought to be due to the action of cyanide, and thiocyanate is detected in blood and urine of workers. Acrylonitrile is an electrophilic compound and binds covalently to nucleophilic sites in macromolecules. Hemoglobin adducts have been used for exposure assessment in experimental animal studies and for follow-up of acute exposure to acrylonotrile in accidentally exposed workers. It has been postulated that the mutagenic effect of acrylonitrile is caused by glycidonitrile, a reactive intermediate able to alkylate macromolecules.
![]() Clinical Findings
Clinical Findings
A. Symptoms and Signs
A few deaths have been reported from acrylonitrile exposure, with respiratory distress, lethargy, convulsions, and coma at 7500 mg/m3. Acrylonitrile was implicated in four cases of toxic epidermal necrosis that developed 11–21 days after the victims returned to houses fumigated with a 2:1 mixture of carbon tetrachloride and acrylonitrile. One patient had measurable blood cyanide levels at autopsy. Symptoms of acute poisoning are described as irritability, respiratory irritation, limb weakness, respiratory distress, dizziness, nausea, cyanosis, collapse, convulsions, and cardiac arrest; these resemble cyanide poisoning.
Chronic human toxicity has been reported in rubber workers exposed to 16–100 ppm of acrylonitrile for periods of 20–45 minutes, with complaints of nasal irritation, headache, nausea, apprehension, and fatigue. Acrylonitrile is carcinogenic in rats after 2 years of feeding and inhalation, inducing brain tumors and stomach papillomas. An excess risk of colon and lung cancers occurred among acrylonitrile polymerization workers from a textile fibers plant. Epidemiologic studies suggest that acrylonitrile is associated with an increased lung cancer risk with a latency period of 20 years and that it should be regarded as probably carcinogenic in humans. However, meta-analyses of mortality studies among acrylonitrile-exposed cohorts do not reveal consistent evidence for carcinogenicity. The IARC has concluded that there is sufficient evidence in experimental animals for acrylonitrile to be classified as a carcinogen (group 2A).
B. Laboratory Findings
The use of biomarkers such as chromosomal aberrations and hemoglobin adducts has shown some promise as a tool to understand susceptibility for health effects and to monitor acutely exposed workers. Elevated serum cyanide or urine thiocyanate levels may be found in cases of acute intoxication.
![]() Differential Diagnosis
Differential Diagnosis
Acute poisoning with acrylonitrile may mimic cyanide intoxication.
![]() Prevention
Prevention
A. Work Practices
Controls have proved effective in reducing employee exposure to acrylonitrile. NIOSH has recommended that acrylonitrile be handled in the workplace as a potential human carcinogen and has published detailed recommendations for adequate work practices.
B. Medical Surveillance
Preplacement and annual medical examinations should include special attention to the skin, respiratory tract, and gastrointestinal tract, as well as to the nonspecific symptoms of headache, nausea, dizziness, and weakness that may be associated with chronic exposure. Treatment kits for acute cyanide intoxication (see Chapter 33) should be immediately available to trained medical personnel at each area where there is a potential for release of or contact with acrylonitrile.
Biologic monitoring may be useful to reflect exposure to acrylonitrile. The relationship between the degree of exposure to acrylonitrile and the urinary excretion of thiocyanate and acrylonitrile was determined in Japanese workers from acrylic fiber factories. A mean postshift urine thiocyanate concentration of 11.4 mg/L (specific gravity 1.024) was found to correlate with an 8-hour average acrylonitrile exposure of 4.2 ppm. Normal urinary thiocyanate levels in nonsmokers do not exceed 2.5 mg/g of creatinine. Mean urinary acrylonitrile levels of 30 μg/L in Dutch plastics workers were found to correlate with a mean 8-hour time-weighted average (TWA) exposure level of 0.13 ppm and were used to monitor adequate work practices.
![]() Treatment
Treatment
Treatment of acute intoxication with acrylonitrile is similar to that of cyanide poisoning. A combination of N-acetylcysteine with sodium thiosulfate has been suggested as an appropriate measure for acrylonitrile intoxication.
AROMATIC AMINES
ESSENTIALS OF DIAGNOSIS
![]() Acute effects
Acute effects
• Dermatitis.
• Asthma.
• Cholestatic jaundice.
• Methemoglobinemia.
![]() Chronic effects
Chronic effects
• Bladder cancer.
![]() General Considerations
General Considerations
The aromatic amines are a class of chemicals derived from aromatic hydrocarbons, such as benzene, toluene, naphthalene, anthracene, and diphenyl, by the replacement of at least one hydrogen atom by an amino group. Some examples are shown below.

![]() Use
Use
Aromatic amines are used mainly in the synthesis of other chemicals. The principal commercial use of benzidine was as a chemical intermediate in dye manufacture, especially for azo dyes in the leather, textile, and paper industries. Benzidine once was used in clinical laboratories for the detection of blood, but this has been discontinued because of safety concerns. Benzidine is no longer produced for commercial sale in the United States. Any benzidine production must be captive consumption and maintained in closed systems.
Aniline is used as a chemical intermediate in the production of methylene diisocyanate, rubber products, dyes, pesticides, pigments, and hydroquinones. p,p′-Methylene dianiline is used as a chemical intermediate in the production of polyurethanes, dyes, and polyamide and polyimide resins and fibers and as a laboratory analytic reagent. o-Toluidine is used as a component of printing textiles, in the preparation of ion-exchange resin, as an antioxidant in rubber manufacture, and in the synthesis of dyestuffs. 1,4-Phenylenediamine may be found in some hair dyes. 4,4-Methylenebis(2-chloroaniline) (MBOCA) has been used as a curing agent in urethane and epoxy resins. It is no longer manufactured commercially in the United States.
Because of the demonstrated carcinogenicity of b-naphthylamine, its manufacture and use have been banned in many countries. Production of b-naphthylamine ceased in the United States in 1972.
![]() Metabolism & Mechanism of Action
Metabolism & Mechanism of Action
The aromatic amines are nearly all lipid-soluble and are absorbed through the skin. Metabolism is largely via the formation of hydroxylamine intermediates. These metabolites are transported to the bladder as N-glucuronide conjugates and hydrolyzed by the acid pH of urine to form reactive electrophiles that bind to bladder transitional epithelial DNA. The polymorphic enzyme N-acetyltransferase-2 is involved in the metabolism of the aromatic amines; slow acetylator status is a genetic risk factor for bladder cancer. Increased susceptibility for bladder cancer also may be related to glutathione S-transferase M1 gene deficiency. Urine pH (influenced by diet) may have a strong effect on the presence of free urinary aromatic compounds and on urothelial cell DNA adduct levels.
![]() Clinical Findings
Clinical Findings
A. Symptoms and Signs
1. Acute exposure—
A. DERMATITIS—Because of their alkaline nature, certain amines constitute a direct risk of dermatitis. Many aromatic amines can cause allergic dermatitis, notably p-aminophenol and p-phenylenediamine. The latter was known as fur dermatitis and caused asthma among fur dyers and currently may cause contact dermatitis among hairdressers.
B. RESPIRATORY EFFECTS—Asthma caused by p-phenylenediamine has been reported.
C. HEMORRHAGIC CYSTITIS—Hemorrhagic cystitis can result from exposure to o- and p-toluidine and 5-chloro-o-toluidine. The hematuria is self-limited, and no increase in bladder tumors has been noted.
D. HEPATIC INJURY—Cholestatic jaundice has resulted from industrial exposure to diaminodiphenyl methane, which also caused toxic jaundice as a consequence of contaminated baking flour (Epping jaundice). The hepatitis is reversible after cessation of exposure. Acute liver dysfunction has been reported among workers exposed to 5-nitro-o-toluidine.
E. METHEMOGLOBINEMIA—Acute poisoning by aniline and its derivatives results in the formation of methemoglobin. A significant elevation of methemoglobin levels has been demonstrated in adult volunteers after ingestion of 25 mg aniline. The mean lethal dose is estimated to be between 15 and 30 g, although death has followed ingestion of as little as 1 g aniline. It has been postulated that a toxic metabolite, phenylhydroxylamine, is responsible for the methemoglobin. Peak levels of methemoglobin are observed within 1–2 hours of ingestion. Cyanosis becomes apparent at levels of methemoglobin of 10–15%, and headache, weakness, dyspnea, dizziness, and malaise occur at levels of 25–30%. Concentrations of methemoglobin greater than 60–70% may cause coma and death.
2. Chronic exposure—An excess of bladder tumors was recognized in 1895 among German workers who used aromatic amines in the production of synthetic dyes. British dyestuffs workers had a high risk for the development of bladder cancer. In the United States, bladder cancer has occurred in workers exposed to β-naphthylamine or benzidine in the manufacture of dyes and in chemical workers exposed to o-toludine.
Workers involved in the production of auramine and magenta from aniline and those working with 4-aminobiphenyl have an increased risk of bladder tumors. Workers exposed to 4-chloro-o-toluidine have a 73-fold excess of bladder cancer. Animal studies show an increased risk of bladder tumors after exposure to benzidine, o-toluidine, o-dianisidine-based dyes, MBOCA, and other aromatic amines. European studies of individual susceptibility to the development of aromatic amine–associated bladder cancer suggest some modulation by genetic polymorphisms.
The IARC considers benzidine carcinogenic to humans (group 1A) and MBOCA probably carcinogenic to humans (group 2A). The IARC has concluded that there is sufficient evidence in experimental animals for the carcinogenicity of o-toluidine and p,p′-methylene dianiline (group 2B) and finds limited evidence for the carcinogenicity of aniline in animals (group 3).
Results from cohort and case-control studies strongly support the association between occupational aromatic amine exposure (ie, benzidine, naphthylamines, MBOCA, and o-toluidine) and bladder cancer. Since these compounds have been declining in use, the population-attributable risk for bladder cancer (approximately 25%) also may decline.
B. Laboratory Findings
Methemoglobin levels can help in the detection of excess absorption of the single-ring aromatic compounds. Normal individuals have methemoglobin concentrations of 1–2%. A biologic threshold limit value (TLV) of 5% has been proposed.
Determination of the metabolites p-aminophenol and p-nitrophenol can be useful to monitor exposure to aniline and nitrobenzene. After 6 hours of exposure to 1 ppm nitrobenzene, the urinary concentration of p-aminophenol should not exceed 50 mg/L, and the recommended biologic threshold value is 10 mg/L. Levels of free MBOCA in the urine can be used to monitor exposure to this compound. Levels of free MBOCA in urine should be minimized to the limit of detection and used as an index of the adequacy of existing work practices and engineering controls. For workers exposed to the known or suspected carcinogenic aromatic amines, periodic screening of urine for red blood cells and evidence of dysplastic epithelium may detect early bladder cancer.
![]() Differential Diagnosis
Differential Diagnosis
Aliphatic nitrates (eg, ethylene glycol dinitrate), aliphatic nitrites, inorganic nitrites, and chlorates also may cause methemoglobinemia. Occupation-associated bladder cancer may account for 10–15% of all cases of bladder cancer. Exposure to arsenic in drinking water also causes an increased risk of bladder cancer. Cigarette smoking, with inhalation of carcinogenic arylamines (eg, 2-aminonaphthalene), is also a significant risk factor.
![]() Prevention
Prevention
A. Work Practices
Every effort should be made to eliminate use of the carcinogenic aromatic amines by substitution of safer alternatives. Appropriate engineering controls for manufacturers of polyurethane products who use MBOCA—particularly the use of automated systems and local exhaust ventilation—can reduce the potential for exposures successfully. Because most cases of aniline exposure occur through skin and clothing contamination, emphasis should be placed on providing appropriate gloves and protective clothing.
For the benzidine-based dyes, worker exposure should be reduced to the lowest feasible levels through appropriate engineering controls, including the use of closed-process and liquid metering systems, walk-in hoods, and specific local exhaust ventilation. Dust levels can be minimized by the use of dyes in pellet, paste, or liquid form. Restricted access to areas with potential exposure and provision of suitable protective clothing and respirators should be instituted.
B. Medical Surveillance
Preemployment and periodic measurement of postshift urinary p-aminophenol is useful for biologic monitoring of aniline exposure. Similarly, periodic postshift urine samples for free MBOCA can be an important adjunct to industrial hygiene measures of exposure.
The ACGIH (American Conference of Governmental Industrial Hygienists)-recommended biologic exposure limit (BEL) for o-toluidine, MBOCA, and aniline is methemoglobin in blood in excess of 1.5% during or at the end of the work shift. Biologic monitoring by high-pressure liquid chromatographic (HPLC) methods for analysis of urinary o-toluidine, aniline, and MBOCA may be useful. Measurement of methylene dianaline (MDA) using the sensitive gas chromatography–mass spectrometry (GC-MS) assay in urine correlates with hemoglobin adducts of MDA in polyurethane production workers and may serve as a sensitive index of exposure (particularly for dermal exposure) at levels below air-monitoring-detection limits. Hemoglobin adducts also have been used for biologic monitoring of workers exposed to 3-chloro-4-fluoroaniline.
High-risk populations with past or current exposure to carcinogenic aromatic amines should be screened on a periodic basis with exfoliative bladder cytology. Positive findings are followed up with direct urologic examination. Biomolecular screening using voided urine samples for DNA ploidy, bladder tumor–associated antigen p300, and a cytoskeletal protein has been used in one cohort of workers exposed to benzidine.
![]() Treatment
Treatment
The definitive treatment of methemoglobinemia caused by aniline poisoning is administration of the reducing agent methylene blue. However, an excessive amount of methylene blue may itself provoke the formation of methemoglobin. Additionally, the ability of methylene blue to reduce methemoglobin can be impaired by hereditary glucose-6-phosphate dehydrogenase (G6PD) deficiency and can precipitate frank hemolysis. The recommended dose of methylene blue for the initial management of methemoglobinemia is 1–2 mg/kg of body weight intravenously, equivalent to 0.1–0.2 mL of a 1% solution. Maximal response to methylene blue usually occurs within 30–60 minutes. Repeated doses should be spaced about 1 hour apart and based on methemoglobin levels; most patients, unless they are anemic, can tolerate a level of 30% or less. Methylene blue administration should be discontinued if either a negligible response or an increase in methemoglobin levels results after two consecutive doses or if the total dose exceeds 7 mg/kg. It is advisable to continue to monitor methemoglobin levels even after an initial response to methylene blue because there is a potential for continued production of methemoglobin by aniline.
Treatment of bladder cancer associated with aromatic amine exposure is identical to that of nonoccupationally associated bladder tumors. Early detection through screening programs may improve prognosis.
CARBON DISULFIDE
ESSENTIALS OF DIAGNOSIS
![]() Acute effects
Acute effects
• Irritability, manic delirium, hallucinations, paranoia.
• Respiratory irritation.
![]() Chronic effects
Chronic effects
• Coronary artery disease.
• Neurobehavioral abnormalities.
• Retinal microaneurysms.
• Peripheral neuropathy with ascending symmetric paresthesias and weakness.
![]() General Considerations
General Considerations
Carbon disulfide is a colorless volatile solvent with a strong, sweetish aroma. The average odor threshold of 1 ppm is below the permissible exposure limit; therefore, carbon disulfide is a material with good warning properties. It evaporates at room temperature, and its vapor is 2.6 times heavier than air; it may form explosive mixtures in a range of 1–50% by volume in air.
![]() Use
Use
Carbon disulfide is used in the manufacture of rayon, cellophane, carbon tetrachloride, and rubber chemicals and as a grain fumigant.
![]() Occupational & Environmental Exposure
Occupational & Environmental Exposure
In the production of viscose rayon, carbon disulfide is added to alkali cellulose to yield sodium cellulose xanthate. The latter is dissolved in caustic soda to yield viscose syrup, which can be spun to form textile yarn, tire yarn, or staple fiber or cast to form cellophane. Exposure to high concentrations of carbon disulfide can occur during the opening of sealed spinning machines and during cutting and drying.
![]() Metabolism & Mechanism of Action
Metabolism & Mechanism of Action
Inhalation is the major route of absorption in occupational exposure, and 40–50% of carbon disulfide in inhaled air is retained in the body. Excretion of carbon disulfide by the lung accounts for 10–30% of absorbed dose, and less than 1% is excreted unchanged by the kidney. The remainder is excreted in the form of various metabolites in the urine.
Carbon disulfide is metabolized by formation of dithiocarbamates and reduced glutathione conjugates, as well as by oxidative transformation. Thiourea, mercapturic acids, and the glutathione conjugate 2-thiothiazolidine-4-carboxylic acid (TTCA) can be detected in urine of exposed workers. Formation of dithiocarbamate may account in part for the nervous system toxicity of carbon disulfide, whereas oxidation yields carbonyl sulfide, a hepatotoxic metabolite. Carbon disulfide reacts with protein amino functions to form adducts of dithiocarba-mate, which then undergo oxidation or decomposition to an electrophile, which reacts with protein nucleophiles to result in protein cross-linking. Cross-linked neurofilaments then may accumulate within axonal swellings.
![]() Clinical Findings
Clinical Findings
A. Symptoms and Signs
1. Acute exposure—Acute carbon disulfide intoxication was described in the 1920s among workers in the viscose rayon industry, involving exposure to concentrations of hundreds or thousands of parts per million. Signs and symptoms included extreme irritability, uncontrolled anger, rapid mood changes (including manic delirium and hallucinations), paranoid ideas, and suicidal tendencies.
Exposure to 4800 ppm of carbon disulfide for 30 minutes may cause rapid coma and death. High concentrations of vapor may cause irritation of the eyes, nose, and throat; liquid carbon disulfide may cause second- or third-degree burns.
2. Chronic exposure—Chronic effects of lower-level exposure to carbon disulfide include the following:
A. EYE—Viscose rayon workers have been reported to have a high incidence of eye irritation. A high incidence of retinal microaneurysms and delayed fundal peripapillary filling by fluorescein angiography has been reported in Japanese and Yugoslavian workers exposed to carbon disulfide. Color vision has been reported to be disturbed in Chinese workers below the current TLV.
B. EAR—Carbon disulfide exposure enhances noise-induced high-frequency hearing loss. Vestibular symptoms of vertigo and nystagmus also may occur.
C. HEART—Epidemiologic studies indicate that workers exposed to carbon disulfide are at increased risk for cardiovascular disease mortality. There is a correlation between blood pressure, elevated triglyceride, and decreased lipoprotein levels and exposure to carbon disulfide. The pathophysiologic mechanism is unclear but may include an effect on oxidative stress in plasma or alteration of arterial elastic properties. Carbon disulfide may cause increased heart rate variability with persistent effects after exposure has ended. A greater risk of ischemic electrocardiographic changes has been seen in a longitudinal study of viscose rayon workers.
D. NERVOUS SYSTEM—Studies show persistent neurobehavioral changes in psychomotor speed, motor coordination, and personality in workers exposed to low concentrations (5–30 ppm) of carbon disulfide. There is a reduction in peripheral nerve conduction on exposure to less than 10 ppm, although clinical symptoms of polyneuropathy are not present. Distal latency, motor nerve-conduction velocity, and sensory amplitude were found to be sensitive indicators of polyneuropathy in viscose rayon workers exposed to carbon disulfide. Lower levels of exposure have been correlated with decreased slow-fiber-conduction velocity with prolongation of the refractory period of the peroneal nerve. Impaired motor and sensory nerve conduction has been demonstrated in prospective studies of workers exposed to carbon disulfide near the TLV. Cerebellar atrophy with extrapyramidal symptoms with atypical parkinsonism and cerebellar signs has been reported. Small-vessel disease with cerebral lesions in the basal ganglia, subcortical white matter, and brainstem has been reported. Peripheral nerve signs and symptoms may persist for as long as 3 years after exposure has ceased.
E. REPRODUCTIVE EFFECTS—Carbon disulfide exposure was associated with a significant effect on libido and potency but not on fertility or semen quality. Women exposed to concentrations of less than 10 ppm may have an increased rate of menstrual abnormalities, spontaneous abortions, and premature births. No other effects on general endocrine function have been observed.
B. Laboratory Findings
Nonspecific elevations of liver enzymes and creatinine have been reported in acute intoxication. With chronic exposure, peripheral nerve-conduction velocity can be decreased, and neurobehavioral testing may show abnormalities in psycho-motor skills and measures of personality function.
Urinary metabolites that catalyze the reaction of iodine with sodium azide can be used to detect exposure above 16 ppm (iodine-azide reaction). The concentration of end-of-shift urinary TTCA is related to exposure and can detect uptake as low as 10 ppm over the whole working shift. The ACGIH BEI is 5 mg TTCA per gram of creatinine in urine at the end of a shift. Heavy physical work and greater skin contact are correlated with higher TTCA levels. Biopsy of the sural nerve in cases of suspected peripheral nerve damage may be indicated and may show degeneration of both axon and myelin with a predominant loss of large myelinated fibers.
![]() Differential Diagnosis
Differential Diagnosis
Cardiac disease from carbon disulfide intoxication must be differentiated from atherosclerotic heart disease from other causes. Peripheral polyneuropathy should be distinguished from that caused by alcohol, drugs, diabetes, and other toxic agents. Neuropsychiatric symptoms may be a result of depression, post-traumatic stress syndrome, or other toxic exposures such as organic solvents.
![]() Prevention
Prevention
A. Work Practices
Control of exposure must rely largely on engineering controls, with enclosure of processes and machines and proper use of ventilation systems. Operator rotation and respiratory protection during peak exposures should be implemented. Potential sources of ignition are prohibited in areas where carbon disulfide is stored or handled, and the substance must not be allowed to accumulate to concentrations higher than 0.1%. Impervious clothing, gloves, and face shields should be worn to prevent skin contact.
B. Medical Surveillance
Initial medical examination should include the central and peripheral nervous systems, eyes, and cardiovascular system. Visual acuity and color vision should be measured and a baseline electrocardiogram obtained. Periodic medical surveillance to detect early signs or symptoms of toxicity should include questions regarding cardiac, nervous system, and reproductive function, with evaluation of blood pressure, peripheral nerve function, and mental status. Neurobehavioral testing, exercise electrocardiography, and nerve-conduction velocity testing may be indicated. Reduced color discrimination may be a sensitive marker for carbon disulfide neurotoxicity. Measurement of finger tremor frequencies may provide an early indication of chronic carbon disulfide intoxication. Magnetic resonance imaging (MRI) may show periventricular hyperintensity and lacunar infarct, which may be of diagnostic use in selected patients with neurobehavioral effects from carbon disulfide exposure.
Measurement of TTCA in urine collected at the end of the work shift following the first workday is the test of choice for biologic monitoring. Skin disease and increased absorption of carbon disulfide may be important in exposure assessment. Five milligrams per gram of creatinine corresponds to an 8-hour exposure (TWA) to the current TLV. The widely used iodine-azide test is insensitive at carbon disulfide levels of less than 16.7 ppm. The presence of preexisting neurologic, psychiatric, or cardiac disease should be considered relative contraindications for individual exposure.
![]() Treatment
Treatment
Skin and eye contact with carbon disulfide should be treated immediately by washing with large amounts of water, and all contaminated clothing should be removed. No specific treatment is available for chronic carbon disulfide toxicity.
CHLOROMETHYL ETHERS
ESSENTIALS OF DIAGNOSIS
![]() Acute effects
Acute effects
• Respiratory irritation
• Skin rash
![]() Chronic effects
Chronic effects
• Lung cancer.
![]() General Considerations
General Considerations
The haloethers bis(chloromethyl) ether (BCME) and chloromethylmethyl ether (CMME) are highly volatile, colorless liquids at room temperature, miscible with many organic solvents. The haloethers are alkylating agents that are highly reactive in vivo. Technical-grade CMME contains 1–8% BCME as an impurity.
![]() Use
Use
BCME is formed when formaldehyde reacts with chloride ions in an acidic medium. It has been used in the past primarily for chloromethylations (eg, in the preparation of ion-exchange resins), where a polystyrene resin is chloromethylated and then treated with an amine.
![]() Occupational & Environmental Exposure
Occupational & Environmental Exposure
Occupational exposure to the chloromethyl ethers occurs in anion-exchange resin production. Since 1948, approximately 2000 workers have been exposed to BCME in ion-exchange resin manufacture, where exposure levels ranged from 10–100 ppb. Small quantities are produced in the United States and only in closed systems to make other chemicals.
BCME also may be a potential hazard in the textile industry, where formaldehyde-containing reactants and resins are used in fabric finishing and as adhesives in laminating and flocking fabrics. Thermosetting emulsion polymers containing methylacrylamide as binders may liberate formaldehyde on drying and curing and then form BCME in the presence of available chloride. A NIOSH study of textile finishing plants found from 0.4–8 ppb BCME in the workroom air. This led to the use of low-formaldehyde resins and chloride-free catalysts.
![]() Clinical Findings
Clinical Findings
A. Symptoms and Signs
1. Acute exposure—The chloromethyl ethers are potent skin and respiratory irritants. There are no reported cases of acute overexposure to either BCME or CMME.
2. Chronic exposure—Both BCME and CMME are carcinogenic and mutagenic in animal and cellular test systems. When rats are exposed to 0.1 ppm BCME by inhalation for 6 hours a day, 5 days a week, a high incidence of esthesioneuroblastomas and squamous cell carcinoma of the respiratory tract is observed. Both BCME and CMME produce skin papillomas and squamous tumors on direct application or subcutaneous injection. In humans, an excess of lung cancer has been suspected. An industry-wide survey of plants using chloromethyl ethers has documented a strikingly increased risk of lung cancer in exposed workers. More than 60 cases of BCME-associated lung cancer have been identified, with oat cell the principal histologic type. The historical average time-weighted exposure in these cases is estimated to be between 10 and 100 ppm, and the latency period between exposure and lung cancer ranges from 5 to 25 years. An increasing incidence is observed with intensity and length of exposure. In addition, the risk of lung cancer is increased in smokers versus nonsmokers. The mortality rate from respiratory tract cancer is significantly (almost three times) higher among chloromethyl ether–exposed workers, with a latency of 10–19 years. The risk of cancer among exposed workers declines after 20 years from first exposure. NIOSH recommends that BCME be regulated as a potential human carcinogen. The IARC considers BCME carcinogenic to humans (group 1A).
B. Laboratory Findings
The lung carcinoma associated with BCME and CMME presents in similar fashion to nonoccupationally associated carcinoma. Chest radiography may show a mass that should lead to appropriate diagnostic testing. Alternatively, sputum cytology may be abnormal in the presence of a normal chest radiograph and thus may be useful as a screening technique in individual cases. Sputum cytology may be of limited value in the follow-up of workers exposed to known carcinogens who remain at risk for many years following exposure.
![]() Differential Diagnosis
Differential Diagnosis
Known occupational lung carcinogens include asbestos, arsenic, chromium, and uranium; consequently, a careful occupational history should be obtained from an individual who presents with lung carcinoma.
![]() Prevention
Prevention
A. Work Practices
Enclosed chemical processes are essential to reduce exposure below 1 ppb, and continuous monitoring has been used successfully to warn of excessive exposures to BCME and CMME. Since the number of potentially exposed workers has markedly declined since the 1970s, medical follow-up of past exposed workers has assumed a greater role.
B. Medical Surveillance
Preplacement and annual lung examination should be included in medical surveillance of exposed workers. Periodic sputum cytology may be of limited value in detecting early lung cancer.
![]() Treatment
Treatment
The treatment of lung carcinoma associated with BCME/ CMME exposure does not differ from that of nonoccupational cases.
DIBROMOCHLOROPROPANE
ESSENTIALS OF DIAGNOSIS
![]() Acute effects
Acute effects
• Oligospermia, azoospermia.
![]() General Considerations
General Considerations
Dibromochloropropane (DBCP) is a brominated organochlorinenematocide that was used extensively since the 1950s on citrus fruits, grapes, peaches, pineapples, soybeans, and tomatoes. Millions of pounds were produced in the United States. In 1977, employees at a California pesticide formulation plant were found to be infertile, and further investigation documented azoospermia and oligospermia among workers exposed to DBCP. In the United States, its use has been restricted since 1980 to a soil fumigant against plant-parasitic nematodes in pineapples. However, two American companies continued to export DBCP to less developed countries for use on bananas. This practice has largely stopped in recent years, but DBCP is one of many pesticides still in use in developing countries that lack regulation and enforcement. DBCP may remain persistent in soil and continues to be detected as a groundwater contaminant in areas of high past use.
In DBCP-exposed men with both azoospermia and elevation of follicle-stimulating hormone (FSH) levels, follow-up evaluation generally has shown permanent destruction of germinal epithelium. A 17-year follow-up of DBCP-exposed workers found sperm count recovery at 36–45 months in three of nine azoospermic and three of six oligozoospermic men, with no improvement thereafter. A significant increase in plasma levels of FSH and luteinizing hormone was found in the most severely affected workers, with incomplete recovery of sperm count and motility.
In vitro, in vivo, and human genotoxicity studies indicate that DBCP can act as a mutagen and clastogen. No correlation has been found between DBCP contamination in drinking water and mortality rates from leukemia or gastric cancer. Birth outcomes (low birth weight and birth defects) did not differ among DBCP-exposed workers or community residents exposed to DBCP-contaminated drinking water.
NIOSH recommends that DBCP be regulated as a potential human carcinogen. The IARC finds that there is sufficient evidence of carcinogenicity in animals (group 2B).
DIMETHYLAMINOPROPIONITRILE
Dimethylaminopropionitrile was a component of catalysts used in manufacture of flexible polyurethane foams. In 1978, NIOSH reported urinary dysfunction and neurologic symptoms among workers at facilities that used dimethylaminopropionitrile. Workers at polyurethane-manufacturing plants developed neurogenic bladder dysfunction after the introduction of a catalyst containing dimethylaminopropionitrile. Workers had urinary retention, hesitancy, and dribbling. Examination showed a pattern of decreased sensation confined to the lower sacral dermatomes, abnormal retention of contrast material on intravenous pyelogram, or abnormal cystometrograms. Nerve-conduction velocity studies were normal. Symptoms of persistent sexual dysfunction were found 2 years after the original epidemic, and one worker had residual sensorimotor neuropathy. Following these findings, production of catalysts containing dimethylaminopropionitrile was discontinued voluntarily.
Dimethylaminopropionitrile appears to be a unique example of a neurotoxin that produces localized autonomic dysfunction without peripheral nervous system damage. Urotoxic effects may be related to metabolism via a cytochrome P450–dependent mixed-function oxidase system, with formation of reactive intermediate metabolites that interfere with axoplasmic transport. The discovery of this toxicity by an alert clinician underscores the role of the community practitioner in the discovery of new occupational diseases.
ETHYLENE OXIDE
ESSENTIALS OF DIAGNOSIS
![]() Acute effects
Acute effects
• Respiratory tract irritation.
• Skin rash.
• Headache, drowsiness, weakness.
![]() Chronic effects
Chronic effects
• Increased sister chromatid exchanges in lymphocytes.
• Possible increased risk of cancer.
![]() General Considerations
General Considerations
Ethylene oxide is a colorless flammable gas with a characteristic ether-like odor. At elevated pressures, it may be a volatile liquid. It is completely miscible with water and many organic solvents. The threshold of detection in humans is about 700 ppm but is quite variable, and smell cannot be relied on to warn of overexposure. To reduce the explosive hazard of ethylene oxide used as a fumigant or sterilant, it is often mixed with carbon dioxide or halocarbons (15% ethylene oxide and 85% dichlorofluoromethane).
![]() Use
Use
Ethylene oxide is used in the manufacture of ethylene glycol (used for antifreeze and as an intermediate for polyester fibers, films, and bottles), nonionic surface-active agents (used for home laundry detergents and dishwashing formulations), glycol ethers (used for surface coatings), and ethanolamines (for soaps, detergents, and textile chemicals). It is used as a pesticide fumigant and as a sterilant in hospitals, medical products manufacture, libraries, museums, beekeeping, spice and seasoning fumigation, animal and plant quarantine, transportation vehicle fumigation, and dairy packaging.
![]() Occupational & Environmental Exposure
Occupational & Environmental Exposure
Most ethylene oxide is used as a chemical intermediate in plants where closed and automated processes generally maintain exposure levels below 1 ppm. The greatest potential for worker exposure occurs during loading or unloading of transport tanks, product sampling, and equipment maintenance and repair.
Although only approximately 0.02% of production is used for sterilization in hospitals, NIOSH estimates that 75,000 health care workers have potential exposure to ethylene oxide. Approximately 10,000 ethylene oxide sterilization units are in use in 8100 hospitals in the United States. Field surveys of hospital gas sterilizers generally have found that 8-hour TWA exposures to ethylene oxide are below 1 ppm. However, occupational exposure may be several hundred parts per million for brief periods during the opening of the sterilizer door, in the transfer of freshly sterilized items to the aeration cabinet or central supply area, during tank changes, and at the gas-discharge point.
![]() Metabolism & Mechanism of Action
Metabolism & Mechanism of Action
Ethylene oxide is absorbed through the skin and respiratory tract. It is an alkylating agent that binds to DNA and may cause cellular mutation.
![]() Clinical Findings
Clinical Findings
A. Symptoms and Signs
1. Acute exposure—Ethylene oxide is irritating to the eyes, respiratory tract, and skin, and at high concentrations it can cause respiratory depression. Symptoms of upper respiratory tract irritation occur at between 200 and 400 ppm, and above 1000 ppm ethylene oxide may cause headache, nausea, dyspnea, vomiting, drowsiness, weakness, and incoordination. Direct contact of the skin or eyes with liquid ethylene oxide can result in severe irritation, burns, or contact dermatitis.
2. Chronic exposure—
A. REPRODUCTIVE EFFECTS—Ethylene oxide is toxic to reproductive function in both male and female experimental animals. Retrospective studies of reproductive function show a higher rate of spontaneous abortions and preterm birth in women exposed to ethylene oxide.
B. CARCINOGENIC EFFECTS—Ethylene oxide is genotoxic in a variety of animal test systems. Chronic inhalation bioassays in rats have shown that ethylene oxide results in a dose-related increase in mononuclear cell leukemia, peritoneal mesothelioma, and cerebral glioma. Intragastric administration of ethylene oxide in rats produces a dose-dependent increase of squamous cell carcinomas of the forestomach. Studies show a dose-related increase in chromosomal aberrations, sister chromatid exchange in lymphocytes and micronuclei in bone marrow cells of exposed workers; and a dose-related increase in the level of hemoglobin adducts. The GSTT1-null genotype is associated with increased formation of hemoglobin adducts in relation to ethylene oxide exposure, suggesting that individuals with homozygous deletion of the GSTT1 gene may be more susceptible to the genotoxic effects of ethylene oxide.
Retrospective cohort mortality studies have suggested an excess of lymphatic and hematopoietic cancers in ethylene oxide–exposed workers. The IARC considers ethylene oxide to be carcinogenic to humans (group 1). NIOSH recommends that ethylene oxide be treated as a potential human carcinogen.
C. NEUROLOGIC TOXICITY—Impairment of sensory and motor function has been observed in animals exposed to 357 ppm ethylene oxide over 48–85 days, and four cases of peripheral neuropathy were described among workers exposed to a leaking sterilizing chamber for 2–8 weeks. Central neurotoxicity has been reported following chronic ethylene oxide exposure, including neuropsychological abnormalities, lower P300 amplitude, and peripheral neuropathy.
D. OTHER—Occupational asthma also has been reported following acute exposure.
B. Laboratory Findings
No specific finding is characteristic of ethylene oxide exposure. Lymphocytosis has been noted after acute exposure. Where inhalation results in respiratory symptoms, the chest radiograph may show interstitial or frank alveolar edema. Where suspect, a complete blood count may be helpful in the diagnosis of leukemia. Cytogenetic analysis (ie, sister chromatid exchange) of peripheral lymphocytes cannot be used in individual cases to quantitate exposure or estimate cancer risk.
![]() Differential Diagnosis
Differential Diagnosis
The mixture of chlorofluorocarbons found in sterilant cylinders also may produce upper respiratory symptoms on inhalation exposure. Many other genotoxicants, including cigarette smoke and other alkylating agents, can cause an increase in sister chromatid exchanges and chromosomal aberrations.
![]() Prevention
Prevention
A. Work Practices
Proper engineering controls are essential for reducing short-term exposures to hospital sterilizer staff during procedures where ethylene oxide levels have been found to be greatest. A NIOSH survey found that engineering controls are extremely effective in hospitals in reducing ethylene oxide exposure during sterilization. These controls include effective sterilization chamber ethylene oxide purging, local exhaust ventilation at the sterilizer door, adequate ventilation of floor drains, efficient handling of product carts from sterilizer to aerator, and installation of ethylene oxide tanks in ventilated cabinets. Self-contained breathing apparatus or airline respirators are the only respirators acceptable for ethylene oxide and must be worn when concentrations of ethylene oxide are unknown, such as when entering walk-in chambers or for emergency response. With the implementation of effective engineering controls, work shift exposures to ethylene oxide may decrease, but intermittent peak excursions and accidental exposures still may occur.
B. Medical Surveillance
Preplacement and periodic examinations should include attention to the pulmonary, hematologic, neurologic, and reproductive systems. Consistent changes in hematologic parameters have not been demonstrated among workers monitored for ethylene oxide exposure. The mean absolute numbers of eosinophils and red blood cells and percentage of hematocrit were significantly elevated among a group of workers with higher cumulative doses of ethylene oxide. Other studies have not demonstrated the utility of the complete blood count as a screening test for medical surveillance of ethylene oxide–exposed hospital workers. Biologic monitoring studies of ethylene oxide–exposed workers show an increase in chromosomal aberrations, sister chromatid exchanges, micronuclei, and hemoglobin adducts. Personnel trained in emergency response for use of self-contained breathing apparatus should be evaluated for cardiorespiratory fitness with pulmonary function or exercise testing.
![]() Treatment
Treatment
Removal from the work environment after inhalation of the gas should be immediate. If respiratory symptoms are evident, oxygen should be administered and the victim brought to the emergency room. Any contaminated clothing should be removed immediately and, where appropriate, the skin thoroughly washed with soap and water. A chest radiograph should be obtained if warranted by respiratory symptoms, and the patient should be observed for several hours for the onset of pulmonary edema. No other specific treatment is indicated.
FORMALDEHYDE
ESSENTIALS OF DIAGNOSIS
![]() Acute effects
Acute effects
• Eye irritation causing lacrimation, redness, and pain.
• Cough, chest tightness, shortness of breath.
• skin irritation, contact dermatitis.
![]() Chronic effects
Chronic effects
• Bronchitis, exacerbation of asthma.
![]() General Considerations
General Considerations
Formaldehyde is a colorless flammable gas with a pungent, irritating odor. Known to physicians as a tissue preservative and disinfectant, formaldehyde is a basic feedstock of the modern chemical industry. It also may be encountered as formalin (37–50% formaldehyde), methyl aldehyde, methanal (methanol-formaldehyde mixture), methylene glycol, paraform, or paraformaldehyde (a linear copolymer of formaldehyde).
![]() Use
Use
The largest use for formaldehyde is the manufacture of ureaformaldehyde and polyacetal and phenolic resins and as an intermediate in the manufacture of ethylenediaminetetraacetic acid, methylene dianiline, hexamethyl-enetetramine, and nitriloacetic acid. Other important uses include wood industry products, molding compounds, foundry resins, adhesives for insulation, slow-release fertilizers, manufacture of permanent-press finishes of cellulose fabrics, and formal-dehyde-based textile finishes. Formaldehyde is used in relatively small quantities for preservation and disinfection. It is a by-product of the incomplete combustion of hydrocarbons and is found in small amounts in automobile exhaust and cigarette smoke.
![]() Occupational & Environmental Exposure
Occupational & Environmental Exposure
Occupational exposure to formaldehyde above 1 ppm occurs in the production of formaldehyde resin and plastics and in the manufacture of apparel, plywood particle board and wood furniture, paper, and paperboard; workers at risk include urea-formaldehyde foam insulation dealers and installers, mushroom farmers, embalmers, and laboratory workers. NIOSH industrial hygiene surveys have found formaldehyde levels of up to 8 ppm in hospital autopsy rooms and up to 2.7 ppm in gross anatomy laboratories. Wildland firefighters may be exposed to formaldehyde as a result of vegetation combustion.
Residential exposure to formaldehyde up to several parts per million occurs from urea-formaldehyde foam insulation (UFFI) and particle board in mobile homes. Levels of formaldehyde are highest in new residences and decline with a half-life of 4–5 years for mobile homes and of less than 1 year for UFFI homes. Mean levels for mobile homes are about 0.5 ppm and for UFFI homes about 0.1 ppm. Diurnal and seasonal variations in exposure levels may occur.
![]() Metabolism & Mechanism of Action
Metabolism & Mechanism of Action
Formaldehyde is formed intracellularly as N5,N20-methylenetetrahydrofolic acid, an important metabolic intermediate. Exogenous formaldehyde can be absorbed by inhalation, ingestion, or dermal absorption. More than 95% of an inhaled dose is absorbed and metabolized rapidly to formic acid by formaldehyde dehydrogenase. Formaldehyde disappears from plasma with a half-life of 1–1.5 minutes, so an increase cannot be detected immediately following inhalation exposure to high concentrations. Most formaldehyde is converted to CO2 via formate, and a small fraction is excreted in the urine as formate and other metabolites. Formaldehyde interacts with macromolecules such as DNA, RNA, and protein. This probably accounts for its carcinogenic effect.
![]() Clinical Findings
Clinical Findings
A. Symptoms and Signs
1. Acute exposure—Formaldehyde vapor exposure causes direct irritation of the skin and respiratory tract. Both direct irritation (eczematous reaction) and allergic contact dermatitis (type IV delayed hypersensitivity) occur. After a few days of exposure to formaldehyde solutions or formaldehyde-containing resins, the individual may develop a sudden urticarial eczematous reaction of the skin of the eyelids, face, neck, and flexor surfaces of the arms. Allergic contact dermatitis may occur from exposure to phenol-formaldehyde resins, water-based paints, or photographic products. There appears to be no relationship between cutaneous disease from formaldehyde and personal or family history of atopy. Direct irritation of the eyes, nose, and throat occurs among most people exposed to 0.1–3 ppm of formaldehyde vapor.
The odor threshold is 0.05–1 ppm; some individuals may note irritation of the upper respiratory tract at or just above the odor threshold. Shortness of breath, cough, and chest tightness occur at 10–20 ppm. Exposure to 50–100 ppm and above can cause pulmonary edema, pneumonitis, or death. Irritant symptoms caused by formaldehyde exposure do not elicit a consistent immunologic response with elevated levels of immunoglobulin (Ig) E or IgG antibody to formaldehyde–human serum albumin.
Several studies show respiratory irritation from exposure to formaldehyde and wood dust. Embalmers report more frequent symptoms of respiratory irritation with exposures during embalming exceeding permissible limits. Formaldehyde exposures in gross anatomy dissection may exceed exposure limits, causing significantly increased upper respiratory symptoms and decrements in airflow during exposure. Respiratory irritant effects are significantly associated with formaldehyde exposure in mobile homes. Residents of homes insulated with urea-formaldehyde foam had a higher prevalence of respiratory symptoms than did residents of control homes but had no demonstrated changes in various hematologic or immunologic parameters.
2. Chronic exposure—
A. CANCER—Squamous cell carcinomas of the nasal epithelium were induced in rats and mice exposed for prolonged periods (up to 2 years). Biochemical and physiologic studies in rats have shown that inhaled formaldehyde can depress respiration, inhibit mucociliary clearance, stimulate cell proliferation, and cross-link DNA and protein in the nasal mucosa.
Epidemiologic studies have suggested that occupational exposure to formaldehyde increases the risk for lung and thyroid cancer and myeloid leukemia, whereas other studies have found no association between formaldehyde exposure and deaths from malignant respiratory disease. Generally consistent results have been found in studies of nasopharyngeal and hypopharyngeal cancer and exposure to formaldehyde, with several studies showing an increased risk of sinonasal cancer (particularly adenocarcinoma) with exposure to formaldehyde. Three cases of malignant melanoma of the nasal mucosa have been reported in persons occupationally exposed to formaldehyde. An increased risk of pancreatic cancer has been observed among embalmers exposed to formaldehyde. The IARC has found sufficient evidence to conclude that formaldehyde is carcinogenic in humans (nasopharyngeal cancer). NIOSH recommends that formaldehyde be regulated as a potential human carcinogen.
B. RESPIRATORY—Occupational asthma has been reported as a result of exposure to formaldehyde resin dust, with studies reporting workers with asthma and positive specific bronchial challenge to formaldehyde. However, exposure-chamber studies have not demonstrated increased airway responsiveness among asthmatics following formaldehyde challenge. Tests of formaldehyde-specific IgE antibodies and cutaneous reactivity also generally have been negative, and formaldehyde sensitization does not correlate with symptoms. A study of students exposed to formaldehyde showed short-term decrements in peak expiratory flow rates. Workers exposed to formaldehyde have significantly greater cross-shift reduction in forced expiratory volume in 1 second (FEV1) and significantly lower respiratory symptoms than do unexposed controls. However, the rate of decline of lung function in formaldehyde-exposed workers is not greater than expected.
C. OTHER EFFECTS—Chronic formaldehyde exposure has been linked in case reports to a variety of neuropsychologic problems, but cohort studies have not been performed to confirm these findings. Spontaneous abortions in cosmetologists and laboratory workers have been associated with the use of formaldehyde-based disinfectants and formalin, respectively. Wood workers exposed to formaldehyde had significantly delayed conception. However, a meta-analysis does not confirm these findings.
B. Laboratory Findings
1. Liver and kidney—Routine tests of hepatic and renal function generally are unremarkable. Measurement of formic acid in the urine generally is not helpful because of the short half-life of formaldehyde.
2. Skin—If contact dermatitis is suspected, patch testing should be performed with appropriate concentrations of formaldehyde.
3. Respiratory system—Cough, shortness of breath, or wheezing may be associated with decreased FEV1 by pulmonary function testing. Peak-flow recordings while at work may show a decrease in maximal airflow during or after exposure to formaldehyde. After exposure to over 20–30 ppm of formaldehyde, chest radiographs may show interstitial or alveolar edema with a resulting reduction in arterial oxygen content on blood gas analysis.
![]() Differential Diagnosis
Differential Diagnosis
Numerous workplace gases and vapors may produce symptoms of upper respiratory tract irritation. Symptoms of eye and throat irritation among office workers may be a result of inadequate ventilation, cigarette smoke, or glues and solvents emitted from newly installed synthetic materials.
Asthmatics may be particularly sensitive to the effects of formaldehyde exposure to indoor environments.
![]() Prevention
Prevention
A. Work Practices
Ventilation engineering controls are effective at significantly reducing exposure to formaldehyde in anatomy laboratories and during embalming procedures. Safety goggles or a full-length plastic face mask should be worn where splashing is possible. At air concentrations above the permissible exposure limit, a full-facepiece respirator with organic vapor cartridge is required. Protective neoprene clothing and boots and gloves impervious to formaldehyde should be worn to prevent skin contact.
B. Medical Surveillance
A preplacement history of asthma or allergy should be obtained, along with a baseline FEV1 and forced vital capacity (FVC). Biologic monitoring using urinary formate concentration is not useful with the possible exception of populations where ambient formaldehyde concentrations are greater than 1 ppm.
Low-level exposure to formaldehyde during embalming is associated with cytogenetic changes in epithelial cells of the mouth and in blood lymphocytes. These cytogenetic effects may be useful markers in biologic monitoring of formaldehyde-exposed workers. Various pathologic changes have been observed in the nasal mucosa of formaldehyde-exposed workers, including ciliary loss, goblet cell hyperplasia, squamous metaplasia, and mild dysplasia.
![]() Treatment
Treatment
In case of eye and skin contact, immediately flush the contaminated area with water for 15 minutes and remove any contaminated clothing. Immediate removal to fresh air is required for inhalation exposure, with administration of oxygen for shortness of breath or hypoxemia. For formaldehyde exposure exceeding 20–30 ppm, emergency department observation with periodic evaluation of respiratory status is necessary for 6–8 hours.
NITRATES: NITROGLYCERIN & ETHYLENE GLYCOL DINITRATE
ESSENTIALS OF DIAGNOSIS
![]() Acute effects
Acute effects
• Headache.
• Angina.
• Fall in blood pressure.
![]() Chronic Effects
Chronic Effects
• Sudden death.
• Increased incidence of ischemic heart disease.
![]() General Considerations
General Considerations
Nitroglycerin (glyceryltrinitrate, trinitropropanetriol) and ethylene glycol dinitrate (dinitroethanediol) are liquid nitric acid esters of monohydric and polyhydric aliphatic alcohols. Those of the tetrahydric alcohols (erythritoltetranitrate, pentaerythritoltetranitrate) and the hexahydric alcohol (mannitolhexanitrate) are solids. They are less stable than aromatic nitro compounds.
Nitroglycerin is readily soluble in many organic solvents and acts as a solvent for many explosive ingredients, including ethylene glycol dinitrate. It is an oily liquid at room temperature with a slightly sweet odor. The sensitivity of nitroglycerin decreases with decreasing temperature, so ethylene glycol dinitrate may be added to nitroglycerin-bearing dynamites to depress the freezing point. Explosions of nitroglycerin may occur when the liquid is heated or when frozen nitroglycerin is thawed. Ethylene glycol dinitrate is an oily colorless liquid that is more stable and less likely than nitroglycerin to explode when it burns.
![]() Use, Production, & Occupational Exposure
Use, Production, & Occupational Exposure
Alfred Nobel first used a mixture of nitroglycerin with diatomaceous earth and later a more stable mixture of nitroglycerin, sodium nitrate, and wood pulp to form dynamite. The major application of nitroglycerin is in explosives and blasting gels, as in low-freezing dynamite in mixture with ethylene glycol dinitrate. Other explosive uses are in cordite in mixture with nitrocellulose and petroleum and in blasting gelatin with 7% nitrocellulose. Nitroglycerin also has medical therapeutic applications for the treatment of angina.
Nitroglycerin may be manufactured by a process in which glycerin is added to a mixture of nitric and sulfuric acids. Dynamite is formed by adding “dope,” or mixtures of sodium nitrate, sulfur, antacids, and nitrocellulose. Ethylene glycol dinitrate is made by nitration of ethylene glycol with mixed acid.
Occupational exposures to nitroglycerin and ethylene glycol dinitrate can occur during their manufacture and during the manufacture and handling of explosives, munitions, and pharmaceuticals. Skin absorption for both nitroglycerin and ethylene glycol dinitrate has not been quantified but is generally greater than respiratory absorption. Air sampling in dynamite plants where both nitroglycerin and ethylene glycol dinitrate are manufactured and used to produce explosives has shown that short-term higher exposures (in the range of 2 mg/m3 of ethylene glycol dinitrate) occur among mixers, cartridge fillers, and cleanup or maintenance workers.
![]() Metabolism & Mechanism of Action
Metabolism & Mechanism of Action
Both nitroglycerin and ethylene glycol dinitrate pass readily through the skin. Although there is an excellent correlation between blood nitrate ester levels and airborne exposures, skin absorption is more significant. Both nitroglycerin and ethylene glycol dinitrate are hydrolyzed to inorganic nitrates. The biologic half-life of both nitroglycerin and ethylene glycol dinitrate is about 30 minutes. Both act directly on arteriolar and venous smooth muscle, causing vasodilation within minutes with a consequent drop in blood pressure and an increase in regional myocardial blood flow. The headache associated with nitrate esters is secondary to cerebral vessel distension.
The tolerance that develops after 2–4 days of continuous exposure appears to be the result of an increased sympathetic compensatory mechanism. The pathogenesis of sudden death caused by nitroglycerin and ethylene glycol dinitrate is postulated to be a rebound vasoconstriction resulting in acute hypertension or myocardial ischemia. NIOSH recommends that workplace exposure to nitroglycerin and ethylene glycol dinitrate be controlled so that workers are not exposed at concentrations that will cause vasodilation, as indicated by the development of throbbing headaches or decreases in blood pressure. At this exposure level, workers should be protected against work-related angina pectoris, other signs or symptoms of ischemia or cardiac damage, and sudden death.
![]() Clinical Findings
Clinical Findings
A. Symptoms and Signs
1. Acute exposure—Symptoms of acute illness include loss of consciousness, severe headache, difficulty breathing, weak pulse, and pallor. Tolerance to these effects develops in dynamite production workers after 1 week of exposure, but symptoms recur on return to work after an absence of 2 days or more. The headache associated with nitroglycerin (powder headache) frequently begins in the forehead and moves to the occipital region, where it can remain for hours or days. Associated symptoms include depression, restlessness, and sleeplessness. Alcohol ingestion may worsen the headache.
An acute drop in mean blood pressure of 10 mm Hg systolic and 6 mm Hg diastolic occurs on return to work after 2–3 days off. Mean blood pressure measurements increase over the week as compensatory mechanisms develop.
Blood pressure reduction has been noted after exposure to 0.5 mg/m3 for 25 minutes, and some workers develop headaches after inhalation exposure of more than 0.1 mg/ m3. Both irritant and allergic contact dermatitis as a consequence of nitroglycerin exposure have been reported.
2. Chronic exposure—Angina pectoris and sudden death have been described among dynamite workers handling nitroglycerin and ethylene glycol dinitrate. In affected workers, the angina usually occurs on the weekend or early in the work shift following periods away from work. The angina is relieved by reexposure to nitroglycerin or ethylene glycol dinitrate in contaminated clothes or by taking nitroglycerin sublingually. Sudden deaths without premonitory angina also have been recorded in dynamite workers. There is an excess risk of cardiac disease among nitroglycerin and ethylene glycol dinitrate workers.
Other reported chronic effects include symptoms of Raynaud phenomenon and peripheral neuropathy. At high concentrations, the aliphatic nitrates may give rise to methemoglobinemia. A retrospective cohort mortality study of munitions workers exposed to nitroglycerin and dinitrotoluene showed an increase in ischemic heart disease mortality for those younger than age 35 years.
B. Laboratory Findings
Coronary angiography has shown normal coronary arteries in workers with angina, and atheromatous coronary vessels generally have not been found on autopsy of workers who died suddenly. The incidence of ectopy is not increased in dynamite workers, and electrocardiograms may be normal. Abnormalities in digital plethysmography show changes in the digital wave pulse with inhalation exposures of 0.12–0.41 mg/m3.
![]() Differential Diagnosis
Differential Diagnosis
An increased incidence of cardiovascular disease has been found in carbon disulfide–exposed workers. Sudden cardiac death may occur after exposure to carbon monoxide or to hydrocarbon solvents.
![]() Prevention
Prevention
A. Work Practices
Avoidance of headaches, blood pressure reduction, angina, or sudden death is achieved by reduction of exposure through proper work practices. Control of exposure is best accomplished by closed systems, local ventilation, and the use of proper seals, joints, and access ports. The danger of detonation can be minimized by the use of nonsparking equipment, prevention of smoking and open flames, and other safety measures. Natural and synthetic rubber gloves accelerate absorption of nitrate esters, so only cotton or cotton-lined gloves should be worn. Dermal contact with nitrates should be minimized because this may be an important route of absorption.
B. Medical Surveillance
Preplacement and periodic examination should stress a history of cardiovascular disease and physical examination of cardiac abnormalities. Urinary glycerol dinitrates may have potential as a biologic monitoring tool. A small experimental study in humans has shown that urinary N-methylnicotinamide may have potential as a biomarker for nitrate exposure, but further studies are necessary to determine its importance in the occupational setting. Methemoglobin is not sensitive for routine monitoring of exposure.
![]() Treatment
Treatment
Treatment of cardiac symptoms caused by nitrate ester exposure does not differ from that of symptoms of coronary insufficiency caused by underlying coronary artery disease. Sublingual nitroglycerin should be used immediately for anginal symptoms. New-onset angina or a change in anginal patterns should be evaluated by noninvasive cardiac imaging or angiography if indicated.
NITROSAMINES
ESSENTIALS OF DIAGNOSIS
![]() Acute effects
Acute effects
• Liver damage.
![]() Chronic effects
Chronic effects
• Probable human carcinogen (selected).
![]() General Considerations
General Considerations
N-Nitrosamines have the general structure shown below:

where R and R can be alkyl or aryl or aryl, for example, N-nitrosodimethylamine (NDMA), N-nitrosodiethylamine (NDEA), N-nitrosodiethanolamine (NDELA), and N-nitrosodiphenylamine (NDPhA). Derivatives of cyclic amines also occur, for example, N-nitrosomorpholine (NMOR) and N-nitrosopyrrolidine (NPyR). N-Nitrosamines are volatile solids or oils and are yellow because of their absorption of visible light by the NNO group.
Reactions of nitrosamines involve mainly the nitroso group and the CH bonds adjacent to the amine nitrogen. Enzymatic reactions leading to the formation of carcinogenic metabolites are thought to occur at the alpha carbon.

![]() Use, Production, & Exposure
Use, Production, & Exposure
Nitrosamines are formed by the reaction of a secondary or tertiary amine with nitrite ion in an acidic medium, according to the general equation shown below:
![]()
Appreciation of the carcinogenicity of the nitrosamines has led to their characterization in many occupational and environmental circumstances. Humans may be exposed to nitrosamines in several ways: formation in the environment and subsequent absorption from food, water, air, or industrial and consumer products; formation in the body from precursors ingested separately in food, water, or air; from the consumption or smoking of tobacco; and from naturally occurring compounds. There is no commercial production in the United States of nitrosamines. Prior to 1976, NDMA was used in the production of dimethylhydrazine, a rocket propellant. NDMA now is used primarily as a research chemical. Other uses of NDMA include the control of nematodes, inhibition of nitrification in soil, as a plasticizer for rubber and acrylonitrile polymers, in the preparation of thiocarbonyl fluoride polymers, as a solvent in the plastics and fiber industry, and as an antioxidant. NDELA is a known contaminant of cosmetics, lotions, shampoos, certain pesticides, antifreeze, and tobacco. NDEA is used primarily as a research chemical, a gasoline and lubricant additive, an antioxidant, a stabilizer in plastics, a fiber industry solvent, a copolymer softener, and a starting material for synthesis of 1,1-diethylhydrazine. The major uses of NDPhA have been in the rubber industry as an antiscorching agent or vulcanization retarder. NDPhA reacts with other amines in the rubber to form N-nitrosamines.
The largest nonoccupational exposure to preformed nitrosamines is derived from tobacco products and tobacco smoke, which may contain NDMA, NDEA, NPyR, and others. Nitrosamine content is greater in sidestream smoke and from cigars. Low levels of nitrosamines occur in several types of food, including cheese, processed meats, beer, and cooked bacon. Many cosmetics, soaps, and shampoos are contaminated with NDELA as a result of the nitrosation of triethanolamine by bactericides.
Nitrate can be reduced to nitrite in vitro and in human saliva in vivo. The reaction of ingested nitrites with amines will yield in vivo nitrosamines in the acidic medium of the stomach. Main contributors to gastric nitrite load are vegetables, cured meats, baked goods, cereals, fruits, and fruit juices.
![]() Occupational Exposure
Occupational Exposure
NDMA has been detected in the workroom air of a rubber sealing factory, fish meal producer, manufacturer of surface-active agents, rubber footwear plant, and chrome and leather tanneries. Approximately 750,000 workers are employed by about 1000 cutting-fluid manufacturing firms, and an undetermined number of machine shop workers have the potential to be exposed to nitrosamines in cutting oils. Direct contact with cutting fluids and the presence of airborne mists provide the opportunity for ingestion or skin absorption.
The greatest exposure to the population as a whole occurs from cigarette smoking and the ingestion of nitrite-preserved meats. Certain classes of pesticides have been found to contain identifiable N-nitroso contaminants formed during synthesis or as a result of interaction with nitrate fertilizers applied simultaneously to crops. The Environmental Protection Agency (EPA) requires testing for nitrosamines of suspect formulation. NDMA has been found in drinking water, probably associated with the chloramine drinking water disinfection process when nitrogen species are added for chloramination.
![]() Metabolism & Mechanism of Action
Metabolism & Mechanism of Action
The nitrosamines are metabolized rapidly after skin or gastrointestinal absorption with a biologic half-life for NDMA of several hours. NDMA is enzymatically demethylated to form monomethylnitrosamine, which then yields an unstable diazohydroxide. The carcinogenic action of the nitrosamines is attributed to this electrophilic species, which can react covalently with DNA.
![]() Clinical Findings
Clinical Findings
A. Symptoms and Signs
1. Acute exposure—Two cases of industrial poisoning caused by NDMA were reported in 1937 in chemists producing an anticorrosion agent. They developed headaches, backache, abdominal cramps, nausea, anorexia, weakness, drowsiness, and dizziness; both workers developed ascites and jaundice, and one died with diffuse hepatic necrosis. Five family members who ingested lemonade accidentally contaminated with NDMA developed nausea, vomiting, and abdominal pain within a few hours, and two died 4 and 5 days later with generalized bleeding. Postmortem examination showed hepatic necrosis.
2. Chronic exposure—Approximately 85% of more than 200 nitrosamines tested in animals are carcinogenic, inducing tumors of the respiratory tract, esophagus, kidney, stomach, liver, and brain. N-Nitrosodimethylamine, NDMA, NDEA, NDPhA, NDELA, NPyR, and NMOR are carcinogenic in many animal species and are transplacental carcinogens.
Analyses of lung tissue have found higher levels of 7-methyl-dGMP (a metabolic product of N-nitrosamines) in association with specific genotypes. Genetic polymorphisms may be predictive of carcinogen adduct levels and therefore may predict the risk of cancer following carcinogen exposure. DNA adducts derived from exposure to aromatic amines have been detected in pancreatic tissues in relation to cancer risk. Exposure to nitrosamines among rubber workers is associated with a significantly increased mortality from cancers of the esophagus, oral cavity, and pharynx. Studies of workers exposed to metalworking fluids indicate an association between metalworking fluid and stomach, pancreatic, laryngeal, liver, and rectal cancer. Although it remains to be determined which specific constituents of metalworking fluids are responsible for the increased risk of various cancers, N-nitrosamines are one of the suspect chemicals. The IARC considers that NDEA and NDMA are probably carcinogenic to humans (group 2A) and that NDELA, NMOR, and NPyR are possibly carcinogenic to humans (group 2B). NIOSH recommends that NDMA be regulated as a potential human carcinogen.
Nitrates may be found in drinking water and have been associated in epidemiologic studies with a greater risk of gastric cancer. Case-control studies of gastric cancer and occupational exposures have suggested a slight increase in risk associated with exposure to nitrosamine. Maternal dietary exposure to N-nitroso compounds (NOC) or to their precursors during pregnancy has been associated with risk of childhood brain tumors.
Liver cirrhosis has been reported following chronic exposure to NDMA.
B. Laboratory Findings
In the few fatalities reported, elevated liver enzymes consistent with hepatic necrosis were noted.
![]() Prevention
Prevention
A. Work Practices
Nitrosamines should be handled in well-ventilated fume hoods. To minimize the potential for formation of nitrosamines, nitrate-containing materials should not be added to metalworking fluids containing ethanolamines. Reduction of nitrosamine exposure in the rubber industry includes the avoidance of compounds that give rise to nitrosamines. Adequate engineering controls should be instituted for working with raw polymers, elastomers, and rubber parts containing dialkylamine compounds that may emit nitrosa-mine when heated.
B. Medical Surveillance
Increased single-strand DNA breaks in peripheral mono-nuclear cells have been found in metalworkers exposed to NDELA in cutting fluids. Screening for mutagenicity of cutting fluids containing nitrite and NDELA has been suggested as a means to assess risk of hazardous exposure. Use of biologic samples for exposure to NDELA has been employed to monitor exposure of workers to metalworking fluids. No specific medical surveillance for nitrosamines is recommended.
![]() Treatment
Treatment
There is no treatment for nitrosamine exposure.
PENTACHLOROPHENOL
ESSENTIALS OF DIAGNOSIS
![]() Acute effects
Acute effects
• Skin and respiratory tract irritation.
• Systemic collapse.
![]() Chronic effects
Chronic effects
• Skin rash (chloracne secondary to chlorodibenzodioxin).
![]() General Considerations
General Considerations
Pentachlorophenol (PCP) is a crystalline solid with low water solubility and a characteristic pungent phenolic odor. Its commercial production proceeds by the direct chlorination of phenol in the presence of chlorine and a catalyst or by the alkaline hydrolysis of hexachlorobenzene; both processes result in 4–12% tetrachlorophenol and less than 0.1% trichlorophenol in the final product. In addition, the required elevated temperatures to produce PCP result in the formation of condensation products, including the toxic dimers dibenzo-p-dioxin and dibenzofuran. Analyses of commercial PCP have reported ranges of chlorinated dioxins and furans from 0.03 to 2510 ppm. Tetrachlorodibenzodioxin has been found in a commercial sample of PCP, but it was not the most toxic 2,3,7,8-isomer. High serum dioxin levels have been reported among chlorophenol workers after occupational exposures. Thus evaluation of the health effects of PCP must be considered separately from those of its impurities.
![]() Use
Use
PCP is used as a wood preservative, herbicide, defoliant, fungicide, and chemical intermediate in the production of pentachlorophenate. A 0.1% solution in mineral spirits, fuel oil, or kerosene is commonly applied as a wood preservative. PCP is used in pressure treatment of lumber at a 5% concentration. About 80% of PCP is used by the wood-preserving industry to treat products such as railway ties, poles, pilings, and fence posts. Treated wood products have a useful product life five times that of untreated wood, resulting in significant economic savings and conservation of timber resources. PCP is usually applied to wood products as a 5% solution in mineral spirits, fuel oil, or kerosene. In the United States, commercial and industrial use of PCP as a preservative is concentrated in the South, Southeast, and Northwest. The remaining 20% is used in production of sodium PCP, in plywood and fiberboard waterproofing, in termite control, and as an herbicide for use in rights of way and industrial sites. PCP is registered by the EPA as a termiticide, fungicide, herbicide, algicide, and disinfectant and as an ingredient in anti-fouling paint. It can be applied as a microbial deterrent in the preservation of wood pulp, leather, seeds, rope, glue, starch, and cooling-tower water. It may not be used for domestic purposes because it is a restricted-use pesticide by the EPA.
Because of the risk of teratogenicity and fetotoxicity, the EPA, since 1984, has required that PCP products in concentrations of 5% or less be used only by certified applicators and has restricted the use of PCP on products that may come in contact with bare skin, food, water, or animals.
![]() Occupational & Environmental Exposure
Occupational & Environmental Exposure
Occupational exposure to PCP occurs primarily in the gas, electric service, and wood preservative industries. Air sampling at 25 wood treatment plants using PCP showed an average exposure of 0.013 mg/m3, and newer automated processes and closed systems at larger facilities are further reducing exposure. Acute exposure may occur with the opening of pressure-vessel doors or in tank cleaning, solution preparation, and the handling of wood after treatment. Hand application of PCP also may pose a risk of overexposure. Dermal exposure is the principal route, either through direct contact with PCP or through contact with treated wood.
Nonoccupational exposure to PCP can occur after the wood has been treated and shipped, where handling may result in dermal exposure. Six months after treatment, PCP will be present on the wood surface at a concentration of about 0.5 mg/ft2. Elevated levels of PCP have been found in the blood and urine of residents of log homes where the logs have been dipped in PCP prior to construction; air samples showed an indoor air concentration of up to 0.38 μg/m3 five years after construction.
![]() Metabolism & Mechanism of Action
Metabolism & Mechanism of Action
Absorption of PCP in the occupational setting is largely through inhalation and skin absorption. The latter is increased when PCP is dissolved in organic solvents. Metabolic studies in rodents and human liver homogenates indicate that PCP undergoes oxidative dechlorination to form tetrachlorohydroquinone, which results in lipid peroxidation and cell death. PCP is excreted mainly in urine as free PCP and as a conjugate with glucuronic acid. Pharmacokinetics are characterized in a single-dose oral administration study by first-order absorption, enterohepatic circulation, and first-order elimination, with 74% of the oral dose of PCP excreted unchanged within 8 days. The half-life for elimination was approximately 30 hours. However, in chronically exposed workers during 2- to 4-week vacations, the terminal half-life of elimination ranges from 30 to 60 days.
Acute intoxication with PCP is caused by interference with cellular electron transport and the uncoupling of oxidative phosphorylation in mitochondria and endoplasmic reticulum. Interaction with energy-rich phosphate compounds results in hydrolysis and free-energy release, leading to a hypermetabolic state with peripheral tissue hyperthermia.
![]() Clinical Findings
Clinical Findings
A. Symptoms and Signs
1. Acute exposure—
A. SKIN—Commercial PCP can cause skin irritation after single exposures to more than a 10% concentration of the material or after prolonged or repeated contact with a 1% solution. Skin sensitization has not been demonstrated. Chloracne may occur after exposure to PCP contaminated with dioxins and dibenzofurans, particularly associated with direct skin contact.
B. EYE, NOSE, AND THROAT—Irritation can occur at levels above 0.3 mg/m3.
C. SYSTEMIC INTOXICATION—Systemic intoxication caused by PCP became evident in the 1950s after two workers died following cutaneous exposure in a wood-dipping operation. Since that time, fatalities from PCP have occurred among chemical production workers, herbicide sprayers, and wood manufacturers. A unique poisoning tragedy occurred in 20 babies wearing diapers inappropriately laundered in 23% sodium pentachlorophenate; two babies died.
Acute intoxication is characterized by the rapid onset of profuse diaphoresis, hyperpyrexia, tachycardia, tachypnea, weakness, nausea, vomiting, abdominal pain, intense thirst, and pain in the extremities. An intense form of muscle contraction is observed before death. Postmortem examination of one acutely intoxicated worker showed cerebral edema with fatty degeneration of the viscera. The minimum lethal dose of PCP in humans is estimated to be 29 mg/kg.
2. Chronic exposure—Long-term exposure to PCP is associated with conjunctivitis, sinusitis, and bronchitis. Chloracne may occur among PCP-exposed workers and may persist for years after exposure has ceased. Occupational exposure to PCP does not cause adverse effects on the peripheral nervous system, and consistent immunologic effects have not been demonstrated following prolonged exposure to PCP.
Paternal exposure to chlorophenate wood preservatives is associated with congenital anomalies in offspring of sawmill workers. Bone marrow aplasia has been reported after exposure to PCP. Cytogenetic studies of PCP-exposed workers have not demonstrated increased sister chromatid exchanges or chromosomal breakage.
An increased risk for non-Hodgkin lymphoma has been observed following exposure to PCP and phenoxyacetic acids. The IARC finds that pentachlorophenol is possibly carcinogenic to humans (group 2B). The EPA concluded that the use of PCP poses a risk of oncogenicity because of the contaminants hexachlorodibenzodioxin and hexachlorobenzene. PCP and its contaminants cause teratogenic and fetotoxic effects in test animals, but little is known concerning adverse reproductive outcomes in humans.
B. Laboratory Findings
Acute intoxication with PCP can result in elevation of blood urea and creatinine, with metabolic acidosis and increased anion gap. Increased serum lactic acid dehydrogenase activity and reduced creatinine clearance have been measured in chronically PCP-exposed workers.
Blood levels of PCP in fatal cases have ranged from 40 to 170 mg/L. Urine levels have ranged from 29 to 500 mg/L in fatal cases and from 3 to 20 mg/L in nonfatal cases of intoxication. In PCP-exposed workers, mean urine PCP levels were 0.95–1.31 mg/L. In nonoccupationally exposed individuals in the United States, urine values of PCP average 6.3 μg/L, with a range from 1 to 193 μg/L and an average of 15 μg/L in hemodialysis patients.
![]() Differential Diagnosis
Differential Diagnosis
Acute intoxication can be confused with hyperthermia from other causes, including heat stroke or sepsis. Symptoms of respiratory irritation may be due to the solvent carrier or other occupational irritants. Chloracne is associated with polychlorinated biphenyls, polychlorinated dibenzodioxins, or polychlorinated dibenzofurans.
![]() Prevention
Prevention
A. Work Practices
Appropriate respiratory protection must be worn where exposure to PCP may exceed permissible limits, particularly in higher-risk operations such as formulating plants and pressure-vessel and tank maintenance. Gloves of nitrile and polyvinyl chloride provide the best protection against both aqueous sodium pentachlorophenate and PCP in diesel oil. Clothing contaminated with PCP must be removed, left at the workplace, and laundered before reuse. Washing and showering facilities should be available to prevent contamination of food, drink, and family. Coating PCP-treated logs of home interiors with a sealant will reduce PCP exposure to the residents.
B. Medical Surveillance
Preemployment urine analysis for PCP should be performed and repeated at intervals. Samples should be collected prior to the last shift of the work week and PCP measured by methods that incorporate hydrolysis. The recommended ACGIH BEI is 2 mg of total PCP per milligram of creatinine in urine or 5 mg of free PCP per milligram of creatinine in plasma before the last shift of work. Discontinuation of PCP exposure will not result in persistent excretion of total PCPs in urine.
Routine medical surveillance should include attention to skin rash and mucous membrane irritation. Hot weather appears to be a predisposing factor for PCP intoxication, so exposure to PCP should be minimized during those times. Significant skin absorption of PCPs may occur and can be documented by urinary PCP monitoring.
![]() Treatment
Treatment
Solutions of PCP spilled on the skin are treated with prompt and thorough washing with soap and water. Eyes contaminated with PCP should be flushed for 15 minutes with water. All contaminated shoes and clothing should be removed immediately.
In the event of acute PCP intoxication, adequate intravenous hydration and efforts to maintain normal body temperature are essential to prevent cardiovascular collapse. Rapid onset of muscular spasms may prevent intubation and resuscitation, so careful monitoring of respiratory status is critical. Metabolic acidosis should be treated with sodium bicarbonate. Atropine sulfate is contraindicated.
POLYCHLORINATED BIPHENYLS
ESSENTIALS OF DIAGNOSIS
![]() Acute effects
Acute effects
• Skin rash (chloracne).
• Eye irritation.
• Nausea, vomiting.
![]() Chronic Effects
Chronic Effects
• Weakness, weight loss, anorexia.
• Skin rash (chloracne).
• Numbness and tingling of extremities.
• Elevated serum triglycerides.
• Elevated liver enzymes.
![]() General Considerations
General Considerations
Polychlorinated biphenyls (PCBs) are a large family of chlorinated aromatic hydrocarbons prepared by the chlorination of biphenyl. Commercial products are a mixture of PCBs with variable chlorine content and are named according to the percentage of chlorine. In addition, all PCBs are contaminated with small but highly toxic concentrations of polychlorinated dibenzofurans.
![]() Use
Use
Between 1930 and 1975, approximately 1.4 billion pounds of PCBs were produced in the United States. The fire-resistant nature of PCBs, combined with their outstanding thermal stability, made them excellent choices as hydraulic and heat-transfer fluids. They also were used to improve the waterproofing characteristics of surface coatings and were used in the manufacture of carbonless copy paper, printing inks, plasticizers, special adhesives, lubricating additives, and vacuum-pump fluids. In the United States, commercial PCBs were marketed under the name Aroclor. In 1977, Congress banned the manufacture, processing, distribution, and use of PCBs.
![]() Occupational & Environmental Exposure
Occupational & Environmental Exposure
Leakage of PCBs from capacitors and transformers while in storage, shipment, or maintenance results in transient exposure risks for utility repair crews, railroad maintenance workers, building engineers, and custodians. Improper storage of used PCB electrical equipment may result in environmental contamination and community exposure. Electrical fires occurring in transformers containing PCBs may release polychlorinated dibenzofurans and polychlorinated dibenzodioxins formed through incomplete combustion of PCBs and chlorinated benzenes. Incidents of widespread building contamination caused by PCB transformer fires have occurred in many cities. The EPA maintains a database of PCB transformers that were in use or in storage for reuse that may pose a significant risk to the general public if leakage or fire should occur.
![]() Metabolism & Mechanism of Action
Metabolism & Mechanism of Action
Chlorinated biphenyl compounds are readily absorbed through the respiratory tract, gastrointestinal tract, and skin.
Distribution is primarily into fat. Biphenyls are metabolized in the liver as the primary site of biotransformation. PCB mixtures cause induction of the hepatic microsomal mono-oxygenase systems. Induction is related to chlorination, and PCB mixtures containing higher percentages of chlorine are more potent than mixtures with lower levels of chlorination. More highly chlorinated isomers are also more resistant to metabolism and therefore are more persistent. Hydroxy metabolites can be detected in bile, feces, and breast milk, but urinary excretion is quite low. This leads to bioaccumulation in fat at low exposure levels and the persistence of PCBs in fatty tissue years after exposure. The formation of electrophilic arene oxide metabolites may cause DNA damage and the initiation of tumor growth.
![]() Clinical Findings
Clinical Findings
A. Symptoms and Signs
1. Acute—Acute exposure to PCBs results in mucous membrane irritation and nausea and vomiting. Transient skin irritation may result from direct handling of PCBs containing mixtures of solvents.
In the mass food poisoning incident, which was a result of rice oil contamination, in western Japan in 1968 (yusho, or rice oil disease), ingestion of PCBs resulted in chloracne. Chloracne probably results from interference with vitamin A metabolism in the skin, with disturbances of the epithelial tissues of the pilosebaceous duct. Typical chloracne presents with cystic or comedonal lesions over the face, ear lobes, retroauricular region, axillae, trunk, and external genitalia and may occur at any age. Yusho patients also showed dark pigmentation of the gingivae, oral mucosa, and nails, with conjunctival swelling. It is not clear whether all or some of these findings were a result of trace contamination of the PCBs with dibenzofurans; the latter compound may have increased during cooking.
2. Chronic—In addition to the acute symptoms of upper respiratory tract irritation, chronic workplace exposure to PCBs also has resulted in chloracne. The relationship between dose of exposure and the appearance of chloracne is inconsistent, although chloracne persists for years after exposure has ceased.
PCBs have an efficient transplacental transfer, and adverse reproductive effects of PCBs have been reported in many animal species; these include failure of implantation, increased number of spontaneous abortions, and low birth weight of litters. In yu-cheng(oil disease), mothers were exposed to PCBs and their heat-degradation products from the ingestion of contaminated rice oil in 1979. Children of these mothers were born growth retarded, with dysmorphic physical findings, delayed cognitive development, and increased activity levels. Rare cases of chloracne and, more commonly, nail abnormalities have been found in yu-cheng children. Higher prenatal exposure to PCBs predicts poorer cognitive abilities, impaired development, and endocrine abnormalities in the offspring of women with exposure to PCBs in the environment or from eating PCB-contaminated fish; these effects appear long-lasting in follow-up studies.
Cytogenetic analysis of peripheral blood lymphocytes has shown increased chromosome aberrations and sister chromatid exchanges among PCB-exposed workers. PCBs fed to test animals produce hepatocellular carcinomas. Cohort studies and case reports of workers exposed to PCBs show an increased risk of malignant melanoma and brain, liver, biliary, stomach, thyroid, hematopoietic, and colorectal cancer. Case control studies show that PCBs significantly increase the risk of non-Hodgkins lymphoma.
PCBs are known as environmental endocrine-disrupting chemicals, with a variety of end-organ hormonal effects. For example, low doses of PCBs potentially can interfere with thyroid hormone receptor–mediated transactivation and alter prenatal steroid hormones. Some PCBs exert dioxin-like activity mediated through receptors that can interfere with sexual hormone–mediated processes. To determine whether these exert an important clinical effect, several studies of environmental PCB exposure and breast cancer incidence have been performed over the past several years. A significant association between PCB levels and breast cancer risk has been demonstrated in some but not all studies. One recent study indicated that PCB-exposed women had an excess of amyotrophic lateral sclerosis and, among those most highly exposed, an excess of Parkinson disease and dementia.
B. Laboratory Findings
Mild elevations of serum triglyceride concentrations have been found in yusho patients and occupationally exposed individuals. PCB-exposed workers have been reported to have significant correlations between the serum PCB level and the γ-glutamyl transpeptidase level.
If exposure to PCB is suspected, serum or fat levels of PCBs may be measured to document absorption. In a steady state, serum is as good a reflection of body burden as is fat. Results must be interpreted in light of established normal values for geographic area and laboratory technique. PCBs can be measured in human tissue by a variety of analytic methods and have been variously reported as total PCB content related to a commercial mixture, as quantification of chromatographic peaks, or by characterization of specific congeners. Analysis of coplanar mono-ortho-substituted and di-ortho-substituted PCB levels in human blood may be useful following acute or chronic exposure. These more toxic congeners contribute significantly to dioxin toxic equivalents in blood from US adults. Normative PCB values among US adults have been published by the Centers for Disease Control and Prevention (CDC).
![]() Differential Diagnosis
Differential Diagnosis
Occupational exposure to PCBs may be accompanied by exposure to chlorinated dibenzodioxin and dibenzofuran contaminants and may be responsible for chronic toxicity. Concurrent exposure to solvents is important because these substances may cause chronic fatigue and elevated liver enzymes. Mild chloracne should not be confused with other papular rashes. A biopsy may be necessary to establish the diagnosis.
![]() Prevention
Prevention
A. Work Practices
Work practices to avoid exposure to PCBs include the use of special PCB-resistant gloves and protective clothing. Adequate ventilation should be maintained during spill cleanup or maintenance of vessels containing PCBs; if this is not possible, approved respirators should be provided. Provision should be made for proper decontamination or disposal of contaminated clothing or equipment. Locations where PCBs are stored should be clearly posted as required by law. Environmental sampling may be necessary to ensure adequate worker protection or safety for public reentry to contaminated areas. Reentry or cleanup levels have been established for dioxins and PCBs to protect workers who reoccupy buildings following a PCB fire.
B. Medical Surveillance
Workers intermittently exposed to PCBs should have a baseline skin examination and liver function tests. Follow-up examination can be limited to symptomatic individuals and those exposed as a consequence of accidental contamination. Routine serum measurements are not recommended.
![]() Treatment
Treatment
Acute exposure should be treated by immediate decontamination of the skin with soap and water to prevent skin absorption. No specific measures are available for respiratory tract or skin absorption. No treatment is available for chronic PCB toxicity. Chloracne is treated with topical therapy for symptomatic relief.
POLYCYCLIC AROMATIC HYDROCARBONS
ESSENTIALS OF DIAGNOSIS
![]() Acute effects
Acute effects
• Dermatitis, conjunctivitis (coal tar pitch volatiles).
![]() Chronic effects
Chronic effects
• Excess cancer rates in selected occupations.
![]() General Considerations
General Considerations
Polycyclic aromatic hydrocarbons (PAHs) are organic compounds consisting of three or more aromatic rings that contain only carbon and hydrogen and share a pair of carbon atoms. They are formed by pyrolysis or incomplete combustion of such organic matter as coke, coal tar and pitch, asphalt, and oil. The composition of the products of pyrolysis depends on the fuel, the temperature, and the time in the hot area. PAHs are emitted as vapors from the zone of burning and condense immediately on soot particles or form very small particles themselves. Such processes always lead to a mixture of hundreds of PAHs. Compounds with three or four aromatic rings predominate. Carcinogenic PAHs are found among those with five or six rings. The simplest fused ring is naphthalene. Some important PAHs in the occupational environment are shown below:
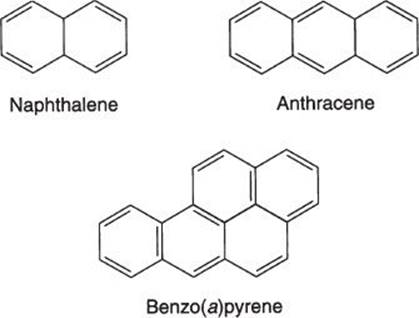
![]() Use, Production, & Exposure
Use, Production, & Exposure
Pure PAHs have no direct use except for naphthalene and anthracene. Anthracene is used in the manufacture of dyes, synthetic fibers, plastics, and monocrystals; as a component of smoke screens; in scintillation counter crystals; and in semiconductor research. Benzo(a)pyrene (BaP) is used as a research chemical and is not produced commercially in the United States. Bitumens are contained in road-paving, roofing, and asphalt products. The majority of carbon black is used as a pigment for rubber tires, with the remainder used in a variety of products such as paint, plastics, printing inks, pigment in eye cosmetics, carbon paper, and typewriter ribbons.
Creosote is used extensively as a wood preservative, usually by high-pressure impregnation of lumber, and as a constituent of fuel oil, lubricant for die molds, and pitch for roofing. Creosote contains over 300 different compounds, the major components of which are PAHs, phenols, cresols, xylenols, and pyridines.
Coal tar pitch is used as a raw material for plastics, solvents, dyes, and drugs. Crude or refined coal tar products are used for waterproofing, paints, pipe coatings, roads, roofing, and insulation; as a sealant, binder, and filler in surface coatings; and as a modifier in epoxy resin coatings.
Naphthalene is used as a chemical intermediate in the production of phthalic anhydride, carbamate insecticides, -naphthol, sulfonic acids, and surfactants and as a moth repellent and tanning agent. PAHs as contaminants can be found in air, water, food, and cigarette smoke, as well as in the industrial environment.
![]() Occupational Exposure
Occupational Exposure
A. Coal Tars & Products
Exposures to PAHs may occur among carbon black production workers, wildland firefighters, petroleum tanker deck crews, meat smokehouse workers, and printing press room operators. The most important source of PAHs in the air of the workplace is coal tar. Tars and pitches are black or brown liquid or semisolid products derived from coal, petroleum, wood, shale oil, or other organic materials. Coal tars are byproducts of the carbonization of coal to produce coke or natural gas. The coke-oven plant is the principal source of coal tar. Coal tar pitch and creosote are derived from the distillation of coal tar. Numerous PAHs have been identified in coal tar, coal tar pitch, and creosote. Coal tar pitch volatiles are the volatile matter emitted into the air when coal tar, coal tar pitch, or their products are heated, and they may contain several PAHs.
The major use for coal tar pitch is as the binder for aluminum smelting electrodes; other uses include roofing material, surface coatings, pipe-coating enamels, and as a binder for briquettes and foundry cores. Creosote is used almost exclusively as a wood preservative.
Occupational exposure to PAHs in coal tar and pitches may occur in gas and coke works, aluminum reduction plants, iron and steel foundries, and coal gasification facilities and during roof and pavement tarring and the application of coal tar paints.
B. Carbon Black
Carbon black is derived from the partial combustion (pyrolysis) of natural gas or petroleum. It is used primarily in pigmenting and reinforcing rubber products and in inks, paints, and paper.
C. Bitumens
Bitumens are viscous solids or liquids derived from refining processes of petroleum. They are used principally for road construction when mixed with asphalt, in roofing felt manufacture, in pipe coatings, and as binders in briquettes. Occupational exposure may occur in these operations.
D. Soots
Soots are mixtures of particulate carbon, organic tars, resins, and inorganic material produced during incomplete combustion of carbon-containing material. Occupational exposure is primarily to chimney soot; potential exposure occurs to chimney sweeps, brick masons, and heating-unit service personnel.
E. Diesel Exhaust
Exposure to PAHs (methylated naphthalenes and phenanthrenes) has been documented among several occupational groups exposed to diesel exhaust, including truck drivers, underground miners, and railroad workers.
![]() Environmental Exposure
Environmental Exposure
PAHs occur in the air primarily as a result of coal burning and settle on soil, where they may leach into water. They are found in smoked fish and meats and form during the broiling and grilling of foods. They are inhaled in cigarette smoke from the burning of tobacco.
![]() Metabolism & Mechanism of Action
Metabolism & Mechanism of Action
PAHs are absorbed readily by the skin, lungs, and gastrointestinal tract of experimental animals and are metabolized rapidly and excreted in the feces. In humans, they are largely absorbed from carrier particles via the respiratory route. They are activated by aryl hydrocarbon hydroxylase to a reactive epoxide intermediate and then conjugated for excretion in urine or bile. The reactive epoxide may bind covalently with DNA and probably accounts for the carcinogenic activity.
![]() Clinical Findings
Clinical Findings
A. Symptoms and Signs
1. Acute exposure—Acute inhalation exposure to naphthalene may cause headache, nausea, diaphoresis, and vomiting. Accidental ingestion has caused hemolytic anemia. Naphthalene also may cause erythema and dermatitis on repeated skin contact. Exposure to coal tar products may cause phototoxicity, with skin erythema, burning, and itching, and eye burning and lacrimation.
2. Chronic exposure—The PAHs are genotoxic, as demonstrated by increased DNA adducts, micronuclei, and chromosomal aberrations among exposed workers. Many PAHs are carcinogenic in animals. Often benzo(a)pyrene is measured to indicate the presence of PAHs where exposure to carcinogens is suspected.
Evidence for human carcinogenicity was described initially by Percivall Pott in 1775, when he associated scrotal cancer in chimney sweeps with prolonged exposure to tar and soot. Subsequently, scrotal cancer has been reported among mule spinners exposed to shale oil and among workers exposed to pitch.
Excess cancer mortality has been found among coke oven workers (lung and prostate), foundry workers (lung), aluminum smelter workers (lung and bladder), and roofers (lung and stomach). Workers exposed to diesel exhaust have an increased risk of lung and, possibly, prostate cancer. In one study, exposure to carbon black experienced by dockyard workers was associated with a twofold increased risk of bladder cancer. Road-paving workers may have a slightly higher rate of lung cancer and a moderately higher rate of stomach cancer than their nonexposed counterparts.
The IARC considers coal tar pitch volatiles to be carcinogenic to humans (group 1), BaP and creosote possibly carcinogenic to humans (group 2A), and carbon black possibly carcinogenic to humans (group 2B). NIOSH considers that coal tar products, carbon black, and anthracene are carcinogenic and recommends that exposures be limited to the lowest feasible level. There is evidence that extracts of refined bitumens are carcinogenic in animals. There are insufficient data to assess cancer risk among workers exposed to bitumens (such as highway maintenance workers and road pavers).
Exposure-related respiratory effects in carbon black–exposed workers have included reduction in airflow, symptoms of chronic bronchitis, and small opacities on chest radiograph. Elevated liver enzymes have been found in a group of coke oven workers heavily exposed to PAHs, and excess mortality from cirrhosis of the liver has been observed in a cohort of workers heavily exposed to chlorinated naphthalenes. Some studies have indicated that occupational PAH exposure causes fatal ischemic heart disease with a consistent exposure-response relationship. Occupational creosote exposure is a risk for squamous papilloma and carcinoma of the skin.
B. Laboratory Findings
Photopatch testing may demonstrate photodermatitis in workers with occupational exposure to coal tar pitch and fumes.
![]() Differential Diagnosis
Differential Diagnosis
Exposure to other known or potential carcinogens in the work environment should be investigated.
![]() Prevention
Prevention
A. Work Practices
Reduction of emissions from coke ovens, aluminum works, foundries, and steel works is essential. Where gaseous emissions occur during loading or transferring of heated coal tar products, fume and vapor control systems will reduce personal exposure. Skin exposure to tars, pitches, and oils containing PAHs is avoided by wearing gloves and changing contaminated work clothes.
B. Medical Surveillance
Periodic examination of workers exposed to coal tar pitch volatiles should include a history of skin or eye irritation and physical examination with attention to the skin, upper respiratory tract, and lungs. Urinary 1-hydroxypyrene (1-OHP) has been used for biologic monitoring of many worker populations, including coal liquefaction workers, coke oven workers, foundry workers, aluminum smelter potroom workers, underground miners, electrode paste plant workers, fireproof stone manufacturing workers, graphite electrode production workers, artificial shooting target factory workers, automotive repair workers, carbon black production workers, roofers, road pavers, asphalt workers, firefighters, and policemen. Good correlation has been found between airborne PAH exposure and urinary 1-OHP, with significant contribution from dermal exposure. Urinary 1-naphthol has been used as a biomarker of PAH exposure among naphthalene oil distillation workers, foundry workers, and creosote-impregnated wood assemblers. Urinary PAHs also may be useful biomarkers of occupational exposure. Enzyme radioimmunoassay techniques to measure PAH-DNA adducts in white blood cells also have been used as a biomarker of PAH exposure among several types of PAH-exposed workers, including foundry workers, coke oven workers, fireproof material workers, aluminum smelter potroom workers, roofers, and wildland firefighters. Dietary sources of PAHs (eg, charbroiled food) and cigarette smoking contribute to PAH-DNA adduct or urinary 1-OHP levels and should be evaluated as confounding factors. Tetrahydrotetrol metabolites of BaP in urine also may prove to be useful for biomonitoring of PAH exposures.
![]() Treatment
Treatment
Photodermatitis should be treated with cortisone-containing preparations, barrier creams, or removal from exposure.
STYRENE
ESSENTIALS OF DIAGNOSIS
![]() Acute effects
Acute effects
• Eye, respiratory tract, and skin irritation.
![]() Chronic effects
Chronic effects
• Weakness, headache, fatigue, dizziness.
• Neuropsychological deficits, color vision loss, sensory nerve conduction slowing.
![]() General Considerations
General Considerations
Styrene, also known as vinyl benzene and phenylethylene, has the chemical formula C5H5CH:CH2. It is a colorless volatile liquid at room temperature with a sweet odor at low concentrations. The odor threshold of 1 ppm is below the permissible exposure limit, and the material has adequate warning properties. Styrene monomer must be stabilized by an inhibitor to prevent exothermic polymerization, a process that may cause explosion of its container.
![]() Use
Use
Commercial styrene was first produced in the 1920s and 1930s. During World War II, styrene was important in the manufacture of synthetic rubber. More than 90% of styrene is produced by the dehydrogenation of ethylbenzene. Styrene is used as a monomer or copolymer for polystyrenes, acrylonitrile-butadiene-styrene (ABS) resins, styrene-butadiene rubber (SBR), styrene-butadiene copolymer latexes, and styrene-acrylonitrile (SAN) resins. Styrene is also used in glass-reinforced unsaturated polyester resins employed in construction materials and boats and in the manufacture of protective coatings.
![]() Occupational & Environmental Exposure
Occupational & Environmental Exposure
In closed polymerization processes, worker exposure to styrene generally is low, but exposure peaks may occur during cleaning, filling, or maintenance of reaction vessels or during transport of liquid styrene. Styrene exposure during manual application of resins (hand lamination) or spraying in open molds may exceed exposure limits. The most significant exposure to styrene occurs when it is used as a solvent-reactant for unsaturated polyester products that are reinforced with fibrous glass. Reinforced plastics/composites are used in the manufacture of boats, storage tanks, wall panels, tub and shower units, and truck camper tops. In this process, alternating layers of chopped fibers or woven mats of fibrous glass are hand applied with catalyzed resin; up to 10% of the styrene may evaporate into the workplace air as the resin cures. Average styrene exposures in plants where the reinforced products are manufactured can range from 40–100 ppm, with short-term individual exposures of up to 150–300 ppm. In a NIOSH study of the reinforced-plastics industry, directly exposed workers engaged in the manufacture of truck parts and boats had the highest exposure to styrene, with a mean 8-hour TWA of 61 and 82 ppm, respectively.
![]() Metabolism & Mechanism of Action
Metabolism & Mechanism of Action
Occupational exposure occurs mainly via inhalation, with approximately 60% of inhaled styrene retained by the lungs. The odor threshold is 0.02–0.47 ppm. Percutaneous absorption is not significant. Styrene is metabolized by the microsomal enzyme system to styrene oxide, which is hydrated to phenylethylene glycol (styrene glycol). Styrene glycol then is metabolized to mandelic acid or to benzoic acid and then hippuric acid. Mandelic acid is further metabolized to phenylglyoxylic acid. Styrene oxide is also metabolized directly to hydroxyphenylethylmercapturic acid. The styrene oxide intermediate is genotoxic and is probably the key factor in the carcinogenic effect of styrene. Genetic polymorphisms of xenobiotic-metabolizing enzymes (EPHX1, GSTT1, GSTM1, GSTP1) appear to play an important role in styrene biotransformation.
After short-term exposure, the venous half-life of styrene is approximately 40 minutes. The half-lives of mandelic acid and phenylglyoxylic acid are about 4 and 8 hours, respectively. In the chronically exposed worker, the half-life for mandelic acid excretion may range from 6 to 9 hours.
![]() Clinical Findings
Clinical Findings
A. Symptoms and Signs
1. Acute exposure—Concentrations of styrene from 100 to 200 ppm may cause eye and upper respiratory tract irritation. Styrene is a defatting agent and a primary skin irritant, resulting in dermatitis. Experimental human exposure to several hundred parts per million causes typical organic solvent anesthetic symptoms, with listlessness, drowsiness, impaired balance, difficulty in concentrating, and decrease in reaction time. Styrene exposure acutely enhances serum pituitary hormone secretion. There are no reports of fatalities as a consequence of styrene exposure.
2. Chronic exposure—Weakness, headache, fatigue, poor memory, and dizziness can occur in workers chronically exposed to styrene in concentrations of less than 100 ppm. Mean reaction time and visuomotor performance may be decreased in exposed workers. The incidence of abnormal electroencephalographs (EEGs) is significantly greater as well.
Studies of styrene-exposed workers have shown detectable blood levels of styrene-7,8-oxide, with dose-related increases in lymphocyte DNA adduct levels, styrene-7,8-oxide hemoglobin adduct levels, single-strand DNA breaks, chromosomal aberrations, lymphocyte micronuclei, and sister chromatid exchanges. Higher hypoxanthine–guanine phosphoribosyltransferase (HRPT) gene mutant frequencies have been detected in styrene-exposed individuals, associated with years of employment and styrene in blood. Several studies of styrene-exposed workers have demonstrated an association between styrene exposure and degenerative disorders of the nervous system, pancreatic cancer, and lymphohematopoietic cancer. Significant associations have been observed in large European studies between the risk of leukemia and exposure to styrene. Other authors suggest that these findings may be confounded by concomitant exposures to other solvents (1,3-butadiene, benzene). The IARC considers styrene possibly carcinogenic to humans (group 2B).
A number of neurotoxic effects have been observed after styrene exposure, including electroencephalographic abnormalities, sensory nerve-conduction slowing, prolonged somatosensory-evoked potentials, and neuropsychological deficits. Neuropsychological symptoms generally are reversible, but some deficits such as visuomotor performance and perceptual speed persist. Neuropsychological effects may correlate with microsomal epoxide hydrolase activity. Styrene exposure among glass-reinforced-plastic workers and plastic-boat manufacturing workers has been associated with early color and contrast vision dysfunction. The effects on contrast sensitivity increase with long-term cumulative exposure, probably reflecting chronic damage to the neurooptic pathways. An effect on hearing acuity has been observed, possibly owing to disorganization of the cochlear membranous structures.
Moderate exposure to styrene has been associated with an altered distribution of lymphocyte subsets in worker populations and may alter leukocyte adherence in experimental test systems. Results of these studies suggest that styrene may alter the cell-mediated immune response of T lymphocytes and result in leukocyte alterations in exposed workers. Styrene also has been found to increase the risk of acute ischemic heart disease mortality among the most highly exposed workers at a synthetic rubber plant.
Styrene may be embryotoxic or fetotoxic in animals. Human reproductive studies (spontaneous abortions, congenital malformations, low birth weight, or reduced fertility) have been inconsistent or limited by methodologic shortcomings.
B. Laboratory Findings
A dose-response relationship exists between styrene exposure and hepatic transaminase, direct bilirubin, and alkaline phosphatase concentrations. However, these tests are nonspecific and should be interpreted in light of other confounders.
The most reliable indicator of styrene exposure is mandelic acid in the urine. Postshift mandelic acid levels in urine show a good correlation with average TWA styrene exposure over the range of 5–150 ppm. Levels of 500 mg mandelic acid per liter of urine may indicate recent exposure to at least 10 ppm styrene. A concentration of 1000 mg mandelic acid per liter of urine corresponds to an average 8-hour TWA styrene exposure of 50 ppm.
![]() Differential Diagnosis
Differential Diagnosis
Exposure to other solvents during the production of styrene and in the manufacture of reinforced-plastic products may cause similar symptoms of central nervous system toxicity such as headache, fatigue, and memory loss.
![]() Prevention
Prevention
A. Work Practices
Styrene poses a significant fire hazard, and proper handling and storage are essential to prevent ignition of the liquid and vapor and a potential explosive reaction. Exposures should be reduced through general and local ventilation systems or through the use of automated processes and closed molds. Intensive local exhaust ventilation is the best way to reduce styrene vapor concentrations during construction of large reinforced-plastic objects, although dilution ventilation is used widely to reduce styrene vapor exposure in the boat industry.
When worker exposure cannot be controlled adequately by engineering controls, protective clothing and respirators may be needed. Where workers may come into contact with liquid styrene, appropriate gloves, boots, overshoes, aprons, and face shields with goggles are recommended. Polyvinyl alcohol and polyethylene gloves and protective clothing give good protection against styrene. To prevent eye irritation at moderately low concentrations, full-facepiece respirators are recommended.
B. Medical Surveillance
Initial medical evaluation should include a history of nervous system disorders and an examination with particular attention to the nervous system, respiratory tract, and skin. Annual medical examinations should be performed on all workers with significant air exposure above the action level or with potential for significant skin exposure. The ACGIH recommended BEI is 240 mg phenylglyoxylic acid per gram of creatinine, 300 mg mandelic acid per gram of creatinine in urine, or 0.55 mg/L in venous blood at the end of the work shift. Styrene in exhaled air also has been used as an indicator of low-level styrene exposure. Measurement of monoamine oxidase type B activity in platelets and the glycophorin A assay also have been suggested as biomarkers of styrene exposure.
![]() Treatment
Treatment
Hands should be washed after skin exposure, and clothing saturated with styrene should be removed immediately. In the case of eye contact, flush the eye immediately with copious amounts of water for 15 minutes. No specific treatment is recommended for acute or chronic styrene exposure.
2,3,7,8-TETRACHLORODIBENZO-P-DIOXIN
ESSENTIALS OF DIAGNOSIS
![]() Acute effects
Acute effects
• Eye and respiratory tract irritation.
• Skin rash, chloracne.
• Fatigue, nervousness, irritability.
![]() Chronic effects
Chronic effects
• Chloracne.
• Soft-tissue sarcoma, non-Hodgkin lymphoma,
• Hodgkin disease.
![]() General Considerations
General Considerations
Polychlorinated dibenzo-p-dioxins (PCDDs) and polychlorinated dibenzofurans (PCDFs) are two large series of tricyclic aromatic compounds that exhibit similar physical, chemical, and biologic properties.

However, there is a pronounced difference in potency among the different PCDD and PCDF isomers. The most extensively studied is the 2,3,7,8-tetrachlorodibenzo-p-dioxin isomer (2,3,7,8-TCDD). Dioxin is the name used for at least 75 chlorinated aromatic isomers, including 22 isomers of the tetrachlorinated dioxin. 2,3,7,8-TCDD is the specific dioxin identified as a contaminant in the production of 2,4,5-trichlorophenol (TCP), 2-(2,4,5-trichlorophenoxy)propionic acid (Silvex), and 2,4,5-trichlorophenoxyacetic acid (2,4,5-T). In its pure form, 2,3,7,8-TCDD is a colorless crystalline solid at room temperature, sparingly soluble in organic solvents, and insoluble in water. The degree of toxicity of the dioxin compounds is highly dependent on the number and position of the chlorine atoms; isomers with chlorination in the four lateral positions (2,3,7,8) have the highest acute toxicity in animals. Under laboratory conditions, 2,3,7,8-TCDD is one of the most toxic synthetic chemicals known. The chlorinated dibenzofurans are contaminants found in some PCBs used in transformers and capacitors, including the most toxic 2,3,7,8-tetrachlorinated dibenzofuran.
![]() Use
Use
2,3,7,8-TCDD is formed as a stable by-product during the production of TCP. Normally, 2,3,7,8-TCDD persists as a contaminant in TCP in amounts ranging from 0.07 to 6.2 mg/kg. Production of 2,4,5-T and Silvex ceased in the United States in 1979, although stockpiles are still being distributed and used. Agent Orange, used in Vietnam as a defoliant during the 1960s, was a 50:50 mixture of esters of the herbicides 2,4-D and 2,4,5-T. Between 10 and 12 million gallons was sprayed over 3–4 million acres in Vietnam; in Agent Orange, the 2,3,7,8-TCDD concentration was about 2 ppm.
The combustion of 2,4,5-T can result in its conversion to small amounts of 2,3,7,8-TCDD. Polychlorinated biphenyls can be converted to PCDFs. Soot from PCB transformer fires may be contaminated with more than 2000 μg/g PCDFs, including the most toxic 2,3,7,8 isomers. A complex mixture of PCDDs and PCDFs may occur in fly ash from municipal incinerators. 2,3,7,8-TCDD is not used commercially in the United States.
![]() Occupational & Environmental Exposure
Occupational & Environmental Exposure
Occupational exposure to 2,3,7,8-TCDD can occur during the production and use of 2,4,5-T and its derivatives. Since 1949, there have been 24 accidents in chemical plants manufacturing chlorinated phenols in which workers were exposed to PCDDs. The explosion of a TCP chemical plant in 1976 in Seveso, Italy, exposed some 37,000 residents of surrounding communities to 2,3,7,8-TCDD.
Workers may be exposed to PCDDs during the production of TCP, 2,4,5-T, and pentachlorophenol. Herbicide sprayers using 2,4,5-T or Silvex have been exposed to 2,3,7,8-TCDD during application. Environmental contamination occurred from spraying waste oil that contained 2,3,7,8-TCDD for dust control on the ground in Missouri. Workers exposed to slag and fly ash from municipal waste incinerators may have increased blood concentrations of PCDDs and PCDFs. The EPA banned most uses of 2,4,5-T and Silvex in 1979, although their use was allowed on sugar cane and in orchards, and miscellaneous noncrop uses were permitted. In October 1983, the EPA published its intent to cancel the registration of all pesticide products containing 2,4,5-T or Silvex. It is not possible to accurately estimate the number of US workers currently exposed to 2,3,7,8-TCDD during decontamination of worksites, from waste materials contaminated with 2,3,7,8-TCDD (such as metal recycling), or from cleanup after fires in transformers containing PCBs.
![]() Metabolism & Mechanism of Action
Metabolism & Mechanism of Action
2,3,7,8-TCDD is an extremely lipophilic substance that is absorbed readily following an oral dose in rats. It accumulates mainly in the liver and after a single dose is largely eliminated unmetabolized in the feces with a whole-body half-life of about 3 weeks. After repeated dosing in small laboratory animals, it is stored in adipose tissue. The half-life of 2,3,7,8-TCDD in humans is 9 years. Dermal absorption may be important in workers exposed to phenoxy acids and chlorophenols. Exposure to 2,3,7,8-TCDD as a vapor normally is negligible because of its low vapor pressure.
Dioxin-like compounds are characterized by high-affinity binding to the Ah receptor, and most biologic effects are thought to be mediated by the ligand-Ah receptor complex. A second protein is required for DNA-binding capability and transcriptional activation of target genes. Growth factors, free radicals, the interaction of 2,3,7,8-TCDD with the estrogen-transduction pathway or protein kinases also may play a role in signal-transduction mechanisms. Relative potency factors have been assigned to the dioxin-like compounds on the basis of a comparison of potency with that of 2,3,7,8-TCDD. Each chemical is assigned a toxic equivalency factor (TEF), some fraction of 2,3,7,8-TCDD, and the total toxic equivalency of the mixture (TEQ) is the sum of the weighted potencies. TEF values have been calculated for PCDDs, PCDFs, and dioxin-like PCBs.
![]() Clinical Findings
Clinical Findings
A. Signs and Symptoms
1. Acute exposure—In some animals, 2,3,7,8-TCDD is lethal in doses of less than 1 μg/kg. Acute toxicity results in profound wasting, thymic atrophy, bone marrow suppression, hepatotoxicity, and microsomal enzyme induction.
In humans, the acute toxicity of 2,3,7,8-TCDD is known from accidental release caused by runaway reactions or explosions. A process accident in Nitro, West Virginia, in 1949, was followed by acute skin, eye, and respiratory tract irritation, headache, dizziness, and nausea. These symptoms subsided within 1–2 weeks and were followed by an acneiform eruption; severe muscle pain in the extremities, thorax, and shoulders; fatigue, nervousness, and irritability; dyspnea; and complaints of decreased libido and intolerance to cold. Workers exhibited severe chloracne, hepatic enlargement, peripheral neuritis, delayed prothrombin time, and increased total serum lipid levels. Long-term follow-up studies of dioxin-exposed workers have found persistence of chloracne and some evidence of liver disease.
2. Chronic exposure—In animals, 2,3,7,8-TCDD is a teratogen and is toxic to the fetus. Two-year feeding studies in rats and mice have demonstrated an excess of liver tumors; the feeding level at which no observable effects in rats occurred was 0.001 μg/kg per day.
Chloracne can result within several weeks after exposure to 2,3,7,8-TCDD and can persist for decades. Among production workers, the severity of chloracne is related to the degree of exposure. In some workplaces, exposed persons had chloracne but no systemic illnesses; in others, workers experienced fatigue, weight loss, myalgias, insomnia, irritability, and decreased libido. The liver becomes tender and enlarged, and sensory changes, particularly in the lower extremities, have been reported. In exposed production workers, systemic symptoms—except for chloracne—have not persisted after exposures ceased.
Immunotoxic, reproductive, and endocrine effects appear to be among the most sensitive indicators of dioxin toxicity. Research indicates that 2,3,7,8-TCDD inhibits multiple estrogen-induced responses in rodent uterus and mammary tissue and in human breast cancer cells. Antiestrogenic effects are thought to be mediated via the aryl hydrocarbon receptor. Laboratory studies in animals suggest that dioxin-like compounds cause altered development (low birth weight, spontaneous abortions, congenital malformations) and adverse changes in reproductive health (fertility, sex organ development, reproductive behavior). 2,3,7,8-TCDD may be transferred transplacentally and via breast milk, and elevated levels of 2,3,7,8-TCDD have been detected in adult children of female chemical production workers exposed to dioxins. A correlation has been found between serum dioxin levels and menstrual cycle characteristics, particularly among premenarcheal women. Minimal effects were observed on the incidence of endometrioses in this cohort. Epidemiologic studies suggest an association between paternal herbicide exposure and an increased risk of spina bifida in offspring. No effect on the risk of spontaneous abortion or sex ratio of the offspring has been observed.
A number of immunologic effects also have been seen in animal studies. Human studies show alteration in delayed-type hypersensitivity after exposure to dioxins. A relation between serum 2,3,7,8-TCDD concentration and a decrease in circulating CD26 cells and decreased spontaneous background proliferation has been observed. Evidence for an effect of dioxin on the humoral immune system is sparse, and no consistent cytogenetic effects have been seen from 2,3,7,8-TCDD exposure.
2,3,7,8-TCDD may inhibit uroporphyrinogen decarboxylation, and cases of porphyria cutanea tarda among exposed workers have been reported. However, recent studies have failed to find an association between 2,3,7,8-TCDD and porphyrin levels. No association has been observed among former chlorophenol production workers between 2,3,7,8-TCDD exposure and serum transaminase levels, induction of cytochrome P450 activity, peripheral neuropathy, chronic bronchitis or chronic obstructive pulmonary disease, and porphyria cutanea tarda. Serum dioxin levels have been positively associated with levels of luteinizing and follicle-stimulating hormones and inversely related to total testosterone levels. This finding is consistent with dioxin-related effects on the hypothalamic-pituitary-Leydig cell axis in animals.
An increased risk of peripheral neuropathy, heart disease, and liver disorders has been seen in studies of Vietnam veterans exposed to dioxin (Operation Ranch Hand). No significant clinical effect on acne, hematologic parameters, immunologic function, or cognitive functioning has been observed in this population. Combined analyses of the Ranch Hand subjects and a NIOSH cohort of industrial workers show modest evidence that exposed workers are at higher risk than nonexposed workers of diabetes or abnormal fasting glucose levels. There is a slight influence of serum 2,3,7,8-TCDD on lipid concentrations in the NIOSH cohort.
Excess risk of soft-tissue sarcoma has been associated with exposure to 2,3,7,8-TCDD and phenoxy herbicides. In a recent reanalysis of US chemical workers with 2,3,7,8-TCDD exposure, a positive trend was found between estimated log cumulative 2,3,7,8-TCDD serum level and overall cancer mortality. Long-term follow-up studies of the Seveso population and a large international cohort show an increase in all-cancer mortality, with increases in soft-tissue sarcoma and lymphohemopoietic neoplasms. Studies of the Operation Ranch Hand cohort suggest a modest increase in the risk of prostate cancer. Serum 2,3,7,8-TCDD levels were significantly related to breast cancer incidence in a long-term follow-up of women in the Seveso Womens’ Health Study. However, another study indicates that breast cancer risk does not appear to be associated with adipose levels of PCDDs. The IARC finds 2,3,7,8-TCDD to be carcinogenic to humans (group 1). NIOSH recommends that 2,3,7,8-TCDD be treated as a potential human carcinogen and that exposure be reduced to the lowest feasible concentration.
B. Laboratory Findings
Abnormalities reported most consistently are elevated liver enzymes, prolonged prothrombin time, and elevated cholesterol and triglyceride levels. Urinary porphyrins may be elevated. Following the Seveso accident, the incidence of abnormal nerve-conduction tests was significantly elevated in subjects with chloracne.
Very low levels of 2,3,7,8-TCDD (4–130 ppt) can be detected in adipose tissue of nonexposed populations. Concentration of polychlorinated compounds in plasma may be 1000-fold less than in adipose tissue. There is a high correlation between adipose and serum 2,3,7,8-TCDD levels; serum levels are a valid measure of body burden. The correlation between plasma and adipose tissue concentrations of 2,3,7,8-TCDD with signs and symptoms is uncertain. Normative PCDD and PCDF serum values among US adults have been published recently by the CDC.
![]() Differential Diagnosis
Differential Diagnosis
Known causes of an acneiform eruption in the workplace include petroleum cutting oils, coal tar, and the chlorinated aromatic compounds. With systemic complaints, such as weight loss, headache, myalgias, and irritability, other underlying medical illnesses should be ruled out before attributing the disorder to 2,3,7,8-TCDD.
![]() Prevention
Prevention
A. Work Practices
NIOSH recommends that 2,3,7,8-TCDD be considered a potential occupational carcinogen and that exposure in all occupational settings be controlled to the fullest extent possible. Specific guidelines for safe work practices must begin with environmental sampling to determine the presence of 2,3,7,8-TCDD contamination, including sampling of air, soil, and settled dust and wipe sampling of surfaces. For site cleanup, specific decontamination procedures should be adhered to for adequate worker protection. Protective clothing and equipment should consist of both outer and inner garments, with outer coveralls, gloves, and boots made of nonwoven polyethylene fabric. Appropriate respiratory protection must be worn, ranging from an air-purifying respirator to a self-contained breathing apparatus. Follow-up sampling should be conducted after decontamination of a site to ensure adequate cleanup.
B. Medical Surveillance
Production workers exposed to compounds contaminated with 2,3,7,8-TCDD, as well as site-decontamination personnel, should undergo baseline and periodic medical examinations with special attention to the skin and nervous system. Baseline laboratory testing should include liver enzymes, cholesterol, and triglycerides, with follow-up as required. Effective safety measures for dioxin cleanup workers will prevent clinical or biochemical disease (chloracne, liver disease, peripheral neuropathy, porphyria cutaneatarda). There has been considerable progress in the use of serum 2,3,7,8-TCDD levels, with the characterization of 2,3,7,8-TCDD body burdens in the Ranch Hand cohort, Seveso residents, herbicide production employees, and Vietnamese civilians. Serum dioxin levels may be useful for research purposes or to assess health-outcome risks for exposure reconstruction, but they are not recommended for routine medical monitoring.
![]() Treatment
Treatment
Skin contaminated with 2,3,7,8-TCDD should be washed immediately and any contaminated clothing removed and placed in marked containers and disposed of appropriately. Except for symptomatic treatment of chloracne, there is no treatment for acute or chronic health effects resulting from 2,3,7,8-TCDD exposure.
VINYL CHLORIDE MONOMER
ESSENTIALS OF DIAGNOSIS
![]() Acute effects
Acute effects
• Respiratory tract irritation.
• Lethargy, headache.
![]() Chronic effects
Chronic effects
• Acroosteolysis, Raynaud phenomenon, skin thickening.
• Hepatosplenomegaly.
• Hepatic angiosarcoma.
![]() General Considerations
General Considerations
Vinyl chloride monomer (chloroethene) is a colorless, highly flammable gas at room temperature. It usually is handled as a liquid under pressure containing a polymerization inhibitor (phenol). It is soluble in ethanol and ether. The odor threshold is variable, so odor cannot be used to prevent excess exposure.
![]() Use
Use
The vast majority of vinyl chloride monomer is used for the production of polyvinyl chloride resins. Polyvinyl chloride is used primarily in the production of plastic piping and conduit, floor coverings, home furnishings, electrical applications, recreational products (records, toys), packaging (film, sheet, and bottles), and transportation materials (automobile tops, upholstery, and mats).
![]() Occupational & Environmental Exposure
Occupational & Environmental Exposure
A 1977 NIOSH survey of three vinyl chloride monomer plants found that the 8-hour TWA ranged from 0.07 to 27 ppm. Following promulgation of the OSHA standard in 1974, exposures were reduced to less than 5 ppm. The highest exposures occur in polymerization plants, particularly during reactor-vessel cleaning.
![]() Metabolism & Mechanism of Action
Metabolism & Mechanism of Action
The chief route of exposure to vinyl chloride monomer (VCM) is through inhalation of the gas, although dermal absorption may be significant during manual reactor-vessel cleaning. Vinyl chloride is absorbed readily through the respiratory tract. Its primary metabolite is chloroethylene oxide, which forms the reactive intermediate epoxide that can bind to RNA and DNA in vivo and may be responsible for the carcinogenicity observed in animal and human studies. There may be increased risk of hepatic angiosarcoma in association with p53 gene mutations. Studies have suggested that polymorphisms of CYP 2E1, GSTT1, and ADH2 may be a major reason for genetic susceptibility in VCM-induced hepatic damage.
The half-life of VCM in expired air is 20–30 minutes. Thiodiglycolic acid (TdGA) is the major urinary metabolite, but it is of limited value in biomonitoring because of metabolic saturation of vinyl chloride, variable metabolism rates, and nonspecificity. One study has suggested that TdGA can be used as an exposure marker for polyvinyl chloride workers when the air VCM level to which they are exposed is greater than 5 ppm.
![]() Clinical Findings
Clinical Findings
A. Symptoms and Signs
1. Acute exposure—VCM has relatively low acute toxicity, causing respiratory irritation and central nervous system depression at high concentrations (10,000–20,000 ppm).
2. Chronic exposure—Chronic toxicity from VCM exposure can result in liver disease, osteolysis, Raynaud phenomenon, vasculiticpurpura, mixed connective-tissue disease, and scleroderma-like skin lesions.
A. ACROOSTEOLYSIS—Symptoms of Raynaud phenomenon, osteolysis in the terminal phalanges of some of the fingers, and thickening or raised nodules on the hands and forearms occurred in workers employed in production and polymerization, especially in workers assigned to clean the reactors. Vinyl chloride disease is a syndrome consisting of Raynaud phenomenon, acroosteolysis, joint and muscle pain, enhanced collagen deposition, stiffness of the hands, and scleroderma-like skin changes. An increase in circulating immune complex levels, cryoglobulinemia, B-cell proliferation, hyperimmunoglobulinemia, and complement activation has been found in these patients. Susceptibility to this disease has been associated with the HLA-DR5 allele. Vascular changes in the digital arteries of the hand associated with acroosteolysis have been demonstrated by arteriography, and circulating immune complexes have been identified.
B. LIVER DISEASE—Hepatic fibrosis, splenomegaly, and thrombocytopenia with portal hypertension have occurred. The characteristic pattern of changes consists of hypertrophy and hyperplasia of hepatocytes and sinusoidal cells, sinusoidal dilation associated with damage to the cells lining the sinusoids, focal areas of hepatocellular degeneration, and fibrosis of portal tracts, septa, and intralobular perisinusoidal regions.
In 1974, three cases of hepatic angiosarcoma among polyvinyl chloride polymerization workers were reported at a plant in Louisville, Kentucky. Since then, many cohort mortality studies have documented an increased risk of hepatic angiosarcoma, hepatocellular carcinoma, and liver cirrhosis. There have now been almost 200 cases of hepatic angiosarcoma reported worldwide, with an average latency of 22 years. Vinyl chloride is genotoxic, causing increased chromosomal aberrations, sister chromatid exchanges, and lymphocyte micronuclei among exposed workers. Specific gene mutations at the p53 locus and mutant p21 proteins have been linked to vinyl chloride angiosarcoma. These findings suggest an effect of chloroethylene oxide, a carcinogenic metabolite of vinyl chloride. The risk of hepatic angiosarcoma is related to the time since the first exposure, duration of employment, and the extent of exposure. The IARC finds that vinyl chloride is carcinogenic to humans (group 1), and NIOSH recommends that vinyl chloride be regulated as a potential human carcinogen.
Only two cases of hepatic angiosarcoma have been documented in the polyvinyl chloride processing industry, suggesting a significantly lower vinyl chloride–related neoplastic risk among fabrication workers. Hemangioendothelioma also has been reported after both vinyl chloride and polyvinyl chloride exposure.
C. PULMONARY EFFECTS—Cases of pneumoconiosis have been reported in workers exposed to polyvinyl chloride dust. Some polyvinyl chloride production and fabrication workers with high (>10 mg/m3) exposure to polyvinyl chloride dust have reduced pulmonary function and an increased incidence of chest radiograph abnormalities. Cumulative polyvinyl chloride dust exposure is associated with mild obstructive airway disease and a higher prevalence of small opacities on chest radiograph. One case of pneumoconiosis and systemic sclerosis following a 10-year exposure to polyvinyl chloride dust has been reported.
D. REPRODUCTIVE EFFECTS—Decreased androgen levels and complaints of impotence and decreased libido and sexual function have been found among male vinyl chloride–exposed workers. Few studies have evaluated the effects of vinyl chloride exposure on the reproductive function of female workers. A significant increase in congenital abnormalities has been found in communities located near a vinyl chloride processing plant, although other studies have failed to report significant development toxicity in association with parental exposure to vinyl chloride or proximity to vinyl chloride facilities.
B. Laboratory Findings
There may be elevated levels of liver enzymes and alkaline phosphatase in workers with vinyl chloride exposure, although in some workers with hepatic angiosarcoma the liver enzymes remain normal until the final stages of disease. Fasting levels of serum bile acids and urinary coproporphyrins have been suggested as clinically useful indicators of early chemical injury in VCM-exposed worker populations with asymptomatic liver dysfunction.
![]() Differential Diagnosis
Differential Diagnosis
Hepatic angiosarcoma has been associated with a history of arsenic exposure and thorium dioxide (Thorotrast) ingestion. The VCM-associated sclerotic changes in skin, with skin nodules, Raynaud phenomenon, and osteolysis, are clinically very similar to idiopathic scleroderma; however, sclerodactyly, calcinosis, and digital pitting scars are unusual in VCM disease.
![]() Prevention
Prevention
The risk of hepatic angiosarcoma should be greatly reduced if the 8-hour TWA is less than 1 ppm.
A. Work Practices
Worker isolation is achieved in most polyvinyl chloride plants through the use of isolated process control rooms. For operators, cleaners, and utility employees, extensive engineering controls in polyvinyl chloride polymerization plants are required to reduce 8-hour TWA worker exposures to less than 1 ppm. Preventing worker exposure during routine maintenance and cleanup operations by adequate degassing of autoclaves and reaction vessels is essential. Online gas chromatographic VCM-specific detectors can identify leaks before large emissions develop.
Employees should be required to wear half-face supplied-air respirators when the concentration of VCM exceeds 1 ppm. A full-face supplied-air respirator is required for reactor cleaning or other maintenance. Where skin contact is possible, protective uniforms, gloves, head coverings, and impervious boots are necessary.
B. Medical Surveillance
Preplacement medical examination should evaluate the presence of liver disease. Concurrent viral hepatitis and alcohol consumption should be evaluated because these factors increase the risk of liver disease in vinyl chloride-exposed workers. Preplacement and periodic measurements of liver enzymes are recommended by NIOSH, although the specificity and sensitivity of these tests are poor. An increased γ-glutamyl transpeptidase level is associated with vinyl chloride exposure and may offer greater specificity for medical surveillance. However, a recent study concluded that liver function assessment only including liver function tests is not able to detect VCM-induced liver damage and only revealed alterations owing to nonoccupational factors, such as dietary and/or metabolic dysfunction. Fasting levels of serum bile acids or plasma clearance of technetium-labeled iminodiacetate have been suggested as a sensitive measure of liver dysfunction among vinyl chloride–exposed workers. Liver ultrasonography is a useful diagnostic test for medical surveillance of vinyl chloride workers, with an increased incidence of periportal fibrosis among more highly exposed workers. Surveillance using biomarkers such as p53gene mutations and DNA adducts are under investigation but have not yet been proven as useful screening tools.
![]() Treatment
Treatment
The mean survival after diagnosis of hepatic angiosarcoma is several months. Computed tomography with intravenous contrast dynamic scanning shows a characteristic isodense appearance on delayed postcontrast scans. Chemotherapy may slightly improve the duration and quality of survival. Acroosteolysis appears to be irreversible after cessation of exposure.
REFERENCES
Burton C: Medium-density fibreboard and occupational asthma. Occup Med (Lond) 2011;61:357 [PMID: 218318].
Chen YC: Retrospective exposure assessment in a chemical research and development facility. Environ Int 2012;39:111 [PMID: 22208749].
Costa C: DNA damage and susceptibility assessment in industrial workers exposed to styrene. J Toxicol Environ Health A 2012;75:735 [PMID: 22788361].
Costa S: Cytogenetic and immunological effects associated with occupational formaldehyde exposure. J Toxicol Environ Health 2013;76:217 [PMID: 23514064].
Goldstein BD: Hematological and toxicological evaluation of formaldehyde as a potential cause of human leukemia. Hum ExpToxicol 2011;30:725 [PMID: 20729258].
Helmfrid I: Health effects and exposure to polychlorinated biphenyls (PCBs) and metals in a contaminated community. Environ Int 2012;44:53 [PMID: 22336529].
Kiran S: Occupationalexposure to ethylene oxide and risk of lymphoma. Epidemiology 2010;21:905 [PMID: 20811284].
Kramer S: Current status of the epidemiologic evidence linking polychlorinated biphenyls and non-hodgkin lymphoma, and the role of immune dysregulation. Environ Health Perspect 2012;120:1067 [PMID: 22552995].
Li MC: Mortality after exposure to polychlorinated biphenyls and dibenzofurans: 30 years after the “Yucheng accident.” Environ Res 2013;120:71 [PMID: 23026800].
Lin YS: Environmental exposure to dioxin-like compounds and the mortality risk in the U.S. population. Int J Hyg Environ Health 2012;215:541 [PMID:22429684].
Luo JC: Blood oxidative stress in Taiwan workers exposed to carbon disulfide. Am J Ind Med 2011;54:637 [PMID: 21630299].
Mahboubi A: Assessment of the effect of occupational exposure to formaldehyde on the risk of lung cancer. Scand J Work Environ Health 2013;39:401 [PMID: 23329145].
Manuwald U: Mortality study of chemical workers exposed to dioxins: follow-up 23 years after chemical plant closure. Occup Environ Med 2012;69:636 [PMID: 22767868].
Mikoczy Z: Cancer incidence and mortality in Swedish sterilant workers exposed to ethylene oxide. Int J Environ Res Public Health 2011;8:2009 [PMID: 21776215].
Pelucchi C: Exposure to acrylamide and human cancer—a review and meta-analysis of epidemiologic studies. Ann Oncol 2011;22:1487 [PMID: 21239401].
Persky V: Polychlorinated biphenyl exposure, diabetes and endogenous hormones. Environ Health 2012;11:57 [PMID: 22931295].
Sharma N: Course and outcome of accidental sodium hydroxide ocular injury. Am J Ophthalmol 2012;154:740 [PMID: 22840487].
Valdez-Flores C: Quantitative cancer risk assessment for ethylene oxide inhalation in occupational settings. Arch Toxicol 2011;85:1189 [PMID: 21347664].
vanTongeren M: Assessing Occupational Exposure to Chemicals in an International Epidemiological Study of Brain Tumours. Ann Occup Hyg 2013;57:610 [PMID: 23467593].
Wang L: Polychlorinated dibenzo-p-dioxins and dibenzofurans and their association with cancer mortality among workers in one automobile foundry factory. Sci Total Environ 2013;443:104 [PMID: 23178894].
![]() SELF-ASSESSMENT QUESTIONS
SELF-ASSESSMENT QUESTIONS
Select the one correct answer to each question.
Question 1: Hydrofluoric acid (hydrogen fluoride)
a. occupational exposure can occur both by direct skin contact and by inhalation of fumes
b. treatment is aimed at deactivation of the fluorideion in blood and tissue
c. burns may cause vesicles and bullae, but they should not be debrided
d. systemic effects from absorption occur only from skin burns
Question 2: Formaldehyde
a. is a colorless, nonflammable gas with an irritating odor
b. is no longer found in wood industry products
c. is primarily a by-product of the incomplete combustion of heavy metals
d. is found in small amounts in automobile exhaust and cigarette smoke
Question 3: Nitroglycerine
a. acute illness symptoms include loss of consciousness, severe headache, difficulty breathing, weak pulse, and pallor
b. symptoms increase in dynamite production with continued exposure
c. headache (powder headache) frequently begins in the occipital region
d. headache is relieved by alcohol ingestion
Question 4: Pentachlorophenol
a. is used as a wood preservative, herbicide, defoliant, and fungicide
b. may explode if used in pressure treatment of lumber
c. is usually applied to wood products as a 50% solution in mineral spirits, fuel oil, or kerosene
d. is registered by the FDA as a disinfectant and as an ingredient in antifouling paint
Question 5: Polychlorinated biphenyls
a. causes acute symptoms of nasal and pharyngeal irritation
b. chronic workplace exposure predictably always results in chloracne
c. have an efficient transplacental transfer
d. prenatal exposure predicts accelerated cognitive abilities
Question 6: Styrene
a. exposure acutely diminishes serum pituitary hormone secretion
b. chronic exposure may cause weakness, headache, fatigue, poor memory, and dizziness
c. may increase mean reaction time and visuomotor performance in exposed workers
d. exposure produces no abnormal electroencephalographs (EEGs) effects
Question 7: Vinyl chloride disease
a. is a syndrome consisting of Raynaud phenomenon, acroosteolysis, joint and muscle pain, enhanced collagen deposition, stiffness of the hands, and scleroderma-like skin changes
b. has a decrease in circulating immune complex levels, cryoglobulinemia, B-cell proliferation, hyperimmunoglobulinemia, and complement activation
c. resistance has been associated with the HLA-DR5 allele
d. is ruled out by a finding of circulating immune complexes