Michael A. O’Malley, MD, MPH
Pesticides include chemicals as well as biological and physical agents used to control a diverse array of pests. In the United States, currently registered pesticides include 1023 active ingredients formulated into 13,400 separate products including adjuvants and antimicrobial compounds. The long list of agents can best be understood by grouping them to target pest or use categories and further subdividing them by chemical structure (Table 34–1). In the United States, pesticide use is about 1.1 billion pounds per year, and estimates run as high as 5.2 billion pounds used worldwide. These estimates do not include antimicrobial agents, which can account for as much as half of all pesticides used, bringing US consumption alone to more than 2 billion pounds annually.
Table 34–1. Pesticide use categories, ingredients, and chemical structure.

REGULATION
In the United States, the Environmental Protection Agency (EPA) regulates the registration, sale, and conditions of use of all pesticides and is responsible for the protection of agricultural workers exposed to pesticides. Prior to enactment of the Federal Insecticide, Fungicide, and Rodenticide Act (FIFRA) in 1970, there was little regulation and testing of pesticides in the United States. Since then, the EPA has applied an increasingly strict registration for sale and use of pesticides. The data required by the EPA for registration of a pesticide include product chemistry, environmental fate, acute and chronic toxicology, and studies of hazards to nontarget organisms (Table 34–2).
Table 34–2. US EPA data requirements for pesticide registration.

Labeling Requirements
When a specific pesticide is approved for use, its use is specified as either general or restricted (to be applied only through permit to a licensed pest control operator), and it is registered and assigned an EPA registration number. The EPA also specifies use instructions, hazard information, and first aid information that must be listed on the product label. The hazard information depends on the assigned acute toxicity category (Table 34–3). Additional safety measures, such as post-application field reentry, also may appear on the product labels.
Table 34–3. Environmental protection agency toxicity labeling categories.

Worker Protection Standard
Although labeling provides the chief means of regulating the use of pesticides by the EPA, some additional safety measures are imposed by regulations. For example, the Worker Protection Standard (WPS) contains several detailed requirements: (1) prohibition against applications that may expose workers at the application site or allow off-site exposure to unprotected individuals, (2) restricted post-application field reentry intervals, (3) personal protective equipment, (4) notification of workers regarding treated areas, (5) requirement for adequate supplies of water, soap, and towels for routine washing and emergency decontamination, (6) provisions for emergency assistance, (7) pesticide safety training for applicators and other handlers, and (8) access to pesticide labeling and application records. Responsibility for enforcement of the WPS requirements lies with the pesticide regulatory agency within each state, most often the state department of agriculture.
Other Federal & State Regulatory Bodies
The Occupational Safety and Health Administration (OSHA) is responsible for the protection of manufacturing and formulation workers. It also periodically investigates unusual hazardous conditions in industry. OSHA also has responsibility for enforcing regulations regarding field sanitation, heat exposure illness, and hazard communication.
The National Institute for Occupational Safety and Health (NIOSH) develops criteria documents for engineering measures to control exposures in manufacturing and formulating operations and occupational exposure to pesticides. Other activities at NIOSH have included a hazard alert regarding safety issues and efforts to promote state surveillance programs through the sentinel event reporting program (SENSOR).
State agriculture and health departments, along with county agriculture and health departments and other state and local agencies, as well as OSHA, may have a variety of regulatory or advisory functions with regard to the use of pesticides. Structural pest control—the application of pesticides to commercial and residential buildings—may fall under one or another of these jurisdictions or is regulated by separate state agencies.
OCCUPATIONAL & ENVIRONMENTAL PESTICIDE EXPOSURES
Table 34–4 lists typical occupational and nonoccupational pesticide exposure situations. The nature, extent, and route of exposure may vary among these different circumstances and the physical properties—particularly the vapor pressure—of individual pesticides.
Table 34–4. Occupational and environmental pesticide exposure situations.

The nature of exposure depends on whether exposure is to the commercial formulation of a pesticide, as applied in a field or structure, or to the active ingredient, as occurs in a manufacturing facility. A pesticide, as applied, consists of the technical-grade chemical (“active” ingredient), diluents (often organic solvents), additives (“adjuvants”), and other “inert” ingredients. The pesticide then is applied mixed or unmixed as sprays, dusts, aerosols, granular, impregnated preparations, fumigants, baits, or systemics. Inert ingredients are not necessarily nontoxic; many are organic solvents. Most typical solvents used are petroleum distillates, but other organic solvents such as methylene chloride and propylene glycol have been used.
Systemic pesticides are water-soluble chemicals that are taken up by a plant and translocated to a part of the plant where a pest, usually an insect, feeds on plant juices and ingests the pesticide. This term is also used for animal systemics, or feed-through pesticides, which are fed to an animal so that pests that feed on feces also ingest the pesticide. The use of systemic, granular, bait, and impregnated pesticide formulations can result in significantly reduced exposure during application.
Pesticides used by consumers for home and garden often are nearly identical in formulation to those used by commercial applicators or differ only in reduced concentration of active ingredient. The most serious exposures occur from accidental or deliberate ingestions. Although pesticides account for a relatively small percentage of the total childhood ingestions, childhood ingestions of organophosphates (OPs), carbamates, and dipyridyl herbicides (diquat and paraquat) may result in serious illness or death. Children also frequently attempt to ingest pesticides used at floor or ground level, such as anticoagulant rodenticides, snail baits, and ant traps, but these less often cause serious poisonings.
HIGH-RISK GROUPS
The highest exposures and incidences of poisoning occur in individuals involved in agricultural pest-control operations: mixing, loading, applying, and flagging. Mixers and loaders are exposed to concentrated pesticides and large volumes. The use of closed systems for mixing and loading reduces these exposures and poisonings considerably. The exposure to applicators varies with the type of application, from backpack sprayers to enclosed-cab vehicles with filtered cooled air. Leaking or poorly maintained equipment may fail and produce large overexposures with any type of application device, including closed mixing/loading systems. Exposures in most manufacturing facilities are low owing to the use of automated closed systems, but exposures that require unscheduled maintenance occur during development of new processes and process breakdowns or leaks. Exposures in formulating facilities may be much higher, particularly if dusty formulations (ie, dusts, powders, and granules) are produced in open systems.
Communities with minimal zoning and mixed agricultural and urban or suburban land use may be at risk for environmental exposure to pesticides. In recent years, most problems were the result of high-volume applications of volatile, soil fumigation products with low environmental effect levels.
ROUTES OF EXPOSURE
Most pesticide inhalation exposures derive from aerosols generated at the time of application or from pesticide adsorbed to household or environmental dust. With fumigants and a few insecticidal compounds, exposures to vapors are also a significant issue.
The most important route for most occupational exposures is dermal. A high percentage of pesticides are absorbed across intact human skin because of a combination of relatively low-molecular-weight and high-lipid solubility. This correlates with the requirement of many compounds to be absorbed through the protective coverings of insects or plants.
The ratio of dermal median lethal dose (LD50) to oral LD50 values that are available for most pesticide provides a rough indication of degree of dermal absorption. A low ratio of dermal LD50 to oral LD50indicates a probable high degree of dermal absorption (eg, the organophosphate insecticide mevinphos has reported oral LD50s ranging from 3.7 to 6.8 mg/kg and reported dermal LD50s ranging from 4.2 to 7.0 mg/kg). For the carbamate insecticides, however, this general rule can be misleading. Because of rapid metabolic deactivation, dermal toxicity is best reflected by the dermal dose that causes 50% cholinesterase inhibition rather than the dermal LD50.
Agricultural Industrial Hygiene—Evaluation of Workplace & Environmental Exposures
The agricultural work environment presents unique problems, many of them the result of the widely scattered seasonal work force. Hazards are transient and may be difficult to evaluate after-the-fact. For the workers, supervisors, and ranch operators living on farm properties, there is also a very limited division between work and living space. Unexpected hazards include intermittent presence of pesticide residues or pesticide application equipment in children’s play areas or accidental intrusion of soil applied nematicides and fumigants into well water.
A. Evaluation of Dermal Contact
Dermal exposure is monitored by using external patches (placed to represent regional anatomic exposure), or cotton shirts can be analyzed after a work shift. Clothing samples provides direct evidence of exposure. Clothing samples are usually collected by an enforcement officer or regulatory agencies.
Interpreting the results of dermal samples can be difficult in the absence of occupational exposure limits or standard reference levels. Even if a reference study evaluating dermal exposure on a comparable work task is available, one cannot be certain what percentage of actual exposure is represented by the measured sample. Most commonly the interpretation is therefore qualitative. After an event, the results are used to establish that some level of exposure occurred. If a previously conducted exposure study of a comparable activity is available, it may be possible to make at least a semiquantitative estimate of the exposure.
Evaluating field worker exposure to pesticides with low vapor pressure also depends upon evaluating residues on the leaves of vegetation. The half-life of residues depends upon humidity, temperature, and other environmental variables. Currently legal organochlorines (endosulfan and dicofol) have the slowest deposition times. Relatively volatile organophosphates (DDVP and mevinphos) have half-lives of 1 or 2 days, but low vapor pressure compounds (phosalone) may have half-lives longer than 1 month in low-humidity environments. Evaluation of residue levels that are safe for reentry is complicated by variations in exposure by crop and by work activity.
B. Biomonitoring
Biomonitoring of pesticide exposures is complex, but can provide both qualitative and quantitative evidence of exposure when suitable markers of exposure are available. It can complement dermal exposure monitoring by providing an estimate of dose when the percentage of absorbed material excreted as a given metabolite is known. Where no direct measure of absorbed dose is possible, it can be estimated from measurements of dermal exposure and studies of the percentage of dermal contamination absorbed over a specified time interval. It is currently possible to monitor cholinesterase enzyme levels and urinary metabolites of some compounds (organophosphates and some carbamates, selected pyrethroid insecticides, halogenated fumigants containing bromine and fluorine, bipyridyl and phenoxy herbicides, and thiocarbamate fungicides metabolized to ethylenethiourea).
C. Air-Monitoring and Air-Contaminant Modeling
Air monitoring is principally useful for gauging exposure to fumigants. Direct reading instruments, available for some compounds such as phosphine and methyl bromide, include colorimetric tubes and flame-ionization gas detectors. For other compounds, the only available methods employ air pumps and sample collection tubes, usually measuring average exposure over a work shift.
For evaluation of community environmental episodes, it is seldom possible to directly measure contaminant levels at the time of exposure. Approximate levels of exposures can be constructed after an event if there is sufficient information available. These usually include
Hourly weather data in the vicinity of the incident, including wind speed, temperature, and wind direction
Source information. Typically the number of treated acres, application method, and application rate (pounds of active ingredient/treated acre), modeled as an area source rather than a point source
Previous air-monitoring study with the same contaminant and a comparable method of application, used to estimate the expected rate of off-gassing
Models, such as the industrial source complex (ISC) or Gaussian model predict vapor concentrations downwind from the point of application. Nonvolatile contaminants that do not generate appreciable vapor can be modeled for off-site movement at the time of application using the fugitive dust model (FDM). In addition to weather data and application details, it is necessary to have knowledge of the particle size distribution. Numerous studies have determined that particle sizes are related most directly to the type of spray nozzle used in the application. Under ideal conditions, with accurate information on weather, application information, and particle size distribution, the FDM model can account for about 70% of variation in drift deposition. It does not directly evaluate exposures to individuals.
D. Estimation of Occupational Exposure and Environmental Exposure From Pesticide Use Data
Numerous studies have employed pesticide use data as a means of estimating either occupational or environmental exposure. All of the studies have in common the assumption is that the low-level exposures typical of off-site exposure are capable of causing chronic health effects such as cancer or Parkinson disease.
Even for field workers, there is at best indirect correlation between exposures and reported quantities of material applied. Use reports have several limitations because they do not account for several key factors related to exposure. Field work can be timed so that it takes place prior to pesticide applications or long afterwards. Significant drift exposures may affect field workers working in areas adjacent to treated fields.
EFFECTS, PREVENTION, & TREATMENT OF PESTICIDE TOXICITY
![]() Clinical Findings
Clinical Findings
A. Symptoms and Signs
1. Acute exposure— The manifestations of acute toxicity vary among pesticide families, but diagnosis generally relies on the following features: (1) signs and symptoms consistent with exposure to one or more chemical families of pesticides (in which a relatively specific clinical constellation [toxidrome] is present), (2) a temporal relationship to known exposure to pesticides or field work, even in the absence of known recent pesticide application (temporal relationships vary depending on the type of pesticide, the route and duration of exposure, and the nature of the toxic effect), and (3) evidence of poisoning in other workers or family members.
Severe acute poisoning usually does not present a diagnostic challenge because a history of significant acute exposure usually is available, and a full spectrum of clinical manifestations is normally present. Mild acute or subacute poisoning may not be readily apparent because the signs and symptoms are likely to be nonspecific and similar to influenza or other common illness. The history of exposure may not be particularly remarkable or even known to the patient.
B. Laboratory Findings
For acute pesticide poisoning, clinical laboratories are able to assess cholinesterase inhibition by organophosphate and possibly carbamate pesticides. Measurement of the pesticide or its metabolites in body fluids done at the time of initial presentation may not be helpful until the follow-up evaluation. The use of biologic levels is not helpful in the diagnosis of chronic toxicity because adequate dose-response data are unavailable for most pesticides, and biologic levels at the time of diagnosis, if present at all, may not reflect those present during exposure.
![]() Prevention
Prevention
A. Work Practices
Manufacturing and formulation workers, mixers, loaders, applicators, and flaggers all are exposed directly to the concentrated or dilute product and so can only be protected by engineering controls and personal protective clothing and devices. Field workers are exposed primarily to residues on plants and in soil. They are protected primarily by reentry intervals—the minimum time allowed between application of a pesticide on a field and entry into that field. The rate of degradation and the toxicity of the degradation products are important determinants of the extent and effect of exposure in this group. Pesticide degradation rates often vary among geographic regions, so reentry intervals may need to be specific to an area or climate. A common cause of acute pesticide intoxication in agriculture is the early entry of a group of field workers into a field where an acutely toxic pesticide was applied recently.
Because skin contamination is the most important route of most occupational exposures, the focus of prevention is on reducing dermal exposure through the use of respirators by manufacturing or formulation workers or pesticide applicators. Contamination of clothing, irritated skin, heat, and sweat are all factors common in agricultural work that promote absorption through the skin. The use of protective clothing in agriculture usually is impeded by the fact that most agricultural work takes place in hot and frequently humid environments. Therefore, the need for skin protection, which is difficult to quantify, must be balanced against the risk of heat-related disorders. The use of personal protective equipment for structural pest control sometimes is hampered by the need to work in tight areas, such as crawl spaces, but the confined nature of these spaces often makes their use necessary.
B. Medical Surveillance
Specific medical and biologic monitoring is available for cholinesterase-inhibiting organophosphate pesticides. For most other pesticides, surveillance is limited to general and occupational histories and physical examinations, with available tests discussed under laboratory findings for each family.
![]() Treatment
Treatment
Treatment of pesticide poisoning in general proceeds in three steps.
A. Decontamination
Decontamination is the first priority, unless lifesaving measures are required. In the case of acute dermal overexposure, the skin and clothing are reservoirs for continued exposure, as is the gastrointestinal tract in the case of ingestion. All clothing should be removed and placed in double plastic bags for later analysis, decontamination, or disposal. The skin and, if necessary, the hair should be washed with soap. Contamination should be checked for under the fingernails. If the eyes have been contaminated, they should be irrigated. The need for gastrointestinal lavage or activated charcoal instillation should be determined on a case-by-case basis (ie, depending on the pesticide, whether vomiting or diarrhea has occurred, and the level of consciousness). All procedures should be done in such a way as to minimize the contamination of medical personnel and equipment without compromising patient care.
B. Specific Antidotes
Specific antidotes are available only in the form of atropine and pralidoxime for cholinesterase-inhibiting pesticides, as discussed in detail below, and chelating agents for heavy metal pesticides such as arsenic and mercury, which rarely result in the need for treatment, except in cases of ingestion.
C. Supportive Care
Supportive care may be the only treatment indicated and may be lifesaving. Assessment of respiratory status and provision of appropriate ventilatory support are critical because most fatal or serious acute pesticide poisonings are indicated, at least in part, through respiratory embarrassment or arrest. Certain medications that otherwise might be given based on clinical diagnosis may be contraindicated once the diagnosis of a specific pesticide intoxication is known. An example is the use of morphine, which can precipitate cardiac arrhythmia, for pulmonary edema in the presence of organophosphate poisoning.
1. Dermatologic effects— Approximately one-third of all reported pesticide-related diseases are dermatologic, about the same percentage as estimated for other chemicals. Most skin reactions are secondary to allergic or irritant contact dermatitis. A few pesticides have been reported to cause other reactions such as contact urticaria, erythema multiforme, chloracne, vitiligo, and porphyria cutanea tarda. Key structural factors related to irritation and sensitivity include protein reactivity and physicochemical properties related to increased absorption, binding, and transport.
Diagnosis of contact reactions depends on careful evaluation of the pattern of exposure and its relation to the distribution and character of subsequent skin lesions. This task may be especially difficult in cases of dermatitis in field workers who may not know what pesticide residues are present on the plants they are in contact with. They also may be exposed to plants known to cause primary irritant or allergic contact dermatitis. Definitive diagnosis of irritant dermatitis depends on noting the above-described correspondence between pattern of exposure and pattern of skin reaction in addition to recognizing the irritant properties of the suspected materials.
Allergic dermatitis can be confirmed only by diagnostic patch testing (type IV allergy), open patch applications, or prick testing (type I allergy). Patch tests are available for a number of pesticides and plants known to be sensitizers and may be made for others, provided preliminary testing of control subjects is conducted to identify the maximum nonirritating concentration of the new test material. The distinction between pesticide and plant allergy and allergic dermatitis is important from an exposure/management standpoint because irritant dermatitis often can be prevented by reducing exposure through use of personal protective equipment or administrative measures such as reentry intervals.
Prevention of allergic contact dermatitis requires complete removal from exposure. Individual pesticides or weeds generally are simple to avoid, given a cooperative employer, but allergy to crop plants presents a greater problem. This is an infrequent problem with most food crops but may be relatively frequent among nursery workers handling Alstroemeria (Peruvian lily), carnations, primrose, chrysanthemums, and other allergenic ornamental crops. The distinction is particularly important for field workers because a pesticide-related cause may mean transfer from the field for several days at one time during a season, whereas a plant-related cause may mean permanent avoidance of a particular crop for at least part of its growing cycle.
Medical treatment consists of alleviation of symptoms with corticosteroids and moisturizers. Prevention of further exposure sufficient to cause recurrence usually is possible with protective clothing. Additional details regarding the skin effects of pesticides are given in the sections on individual classes of pesticides below.
ANIMAL TERATOGENS, REPRODUCTIVE TOXINS, ENDOCRINE DISRUPTORS, & RELATED EFIDEMIOLOGIC STUDIES
Animal studies of teratogenicity frequently employ albino rabbits but may use a variety of other species. Evaluation of study results depends on comparison of developmental abnormalities with controls and comparing doses producing fetal effects with doses producing maternal toxicity. Compounds with teratogenic effects at doses that do not cause maternal poisoning provoke special concern.
Individual compounds identified as teratogens or reproductive toxins in multigenerational studies in animals are identified in separate tables for each major pesticide use category. No currently registered pesticides are recognized human teratogens or female reproductive toxins. However, case reports and a few epidemiologic studies have described either teratogenicity or fetotoxicity at doses that also cause maternal toxicity. For example, workers developed acute organophosphate poisoning after entering a cauliflower field contaminated with residues of oxydemeton-methyl, mevinphos, and methomyl. A crew member who was 4 weeks pregnant at the time of the poisoning subsequently gave birth to a child with multiple cardiac defects, bilateral optic nerve coloboma microphthalmia of the left eye, cerebral and cerebella atrophy, and facial anomalies.
Similarly, the massive amounts of methyl isocyanate (MIC) released from plant producing the insecticide carbaryl at Bhopal in India was associated with a fourfold increased incidence of spontaneous abortion in women who survived the acute pulmonary syndrome that it also provoked. In both examples, the adverse reproductive effects resulted from single short-term rather than cumulative exposures.
![]() Endocrine Disruption
Endocrine Disruption
Since passage of the Food Quality Protection Act (FQPA), the possible interference of pesticides with endocrine function has been an intensely studied topic. Environmental endocrine effects have been studied with epidemiologic studies, field observations of animal populations, and laboratory models. Epidemiologic studies of endocrine effects have included more or less direct measurement of hormonal function, and others have been studies of hormonally influenced endpoints, especially cancers of the male or female reproductive tract. Many studies have evaluated interactions between pesticides and reproductive hormones, especially estrogen, estrogen agonists, and estrogen receptors. However, potential effects may involve many other targets in the neuroendocrine axis. These include neurotransmitters and receptors in the hypothalamus and pituitary peptides that stimulate end-organ targets.
![]() Chronic Health Effects Without Recognized Acute Effects
Chronic Health Effects Without Recognized Acute Effects
A. Cancer
No pesticides currently in use are recognized human carcinogens, with the exception of inorganic arsenic. Arguable cases may be made for the wood preservatives creosote and chromic acid and for the fumigant ethylene oxide. Studies of most occupational carcinogens are often limited by small numbers.
For pesticides, these types of classic occupational cohort studies therefore are only capable of identifying very potent human carcinogens. Recently, 4-chloro-ortho-toluidine (4-COT), the principal metabolite of the insecticide chlordimeform, was found to be a carcinogen in a study of 120 manufacturing workers in Germany, provoking an incidence of bladder cancer 72 times higher than expected. Chlordimeform was taken off the market in 1986 when this information became known. Animal studies previously identified chlordimeform and 4-COT as carcinogens.
1. Studies of farmers and other occupations with pesticide exposure—Epidemiologic studies of cancer in farmers show a relatively consistent increase in certain cancers, notably leukemia, lymphoma, and multiple myeloma. Although these findings are suggestive of an increase in cancer caused by pesticides, specific pesticides could not be incriminated, and other causes related to farm work (eg, viral exposures associated with animal handling) could not be ruled out. Studies of meat packing workers potentially exposed to biologic agents but not to pesticides provide inconsistent support for the viral hypothesis. Some case-control studies of lymphomas in this population have identified significant associations with phenoxy herbicides, atrazine, and organophosphate insecticides.
A few studies have examined cancer incidence in professional pesticide applicators. Two of these studies indicate elevated risks for lung cancer and one indicates an elevated risk for bladder cancer without being able to associate the effect with any specific pesticide.
2. Pesticides recognized as animal carcinogens—Epidemiologic information on carcinogenicity is constrained by the small populations with unique exposures to specific pesticides. Consequently, animal test data represent the only means for evaluating the effect of most compounds.
The EPA’s scientific advisory panel devised a system for summarizing the large volume of cancer bioassay data in 1986. The system differentiates among the small number of compounds recognized to definitely cause cancer in humans, those likely to cause cancer in sufficiently exposed populations, possible carcinogens, and those unlikely to cause cancer. The panel revised the system again in 1996 and in 1999. The final result resembles both the 1986 EPA classification system and that used by the International Agency for Research on Cancer (IARC) (Table 34–5). The current list of carcinogens available from the EPA principally employs the 1986 system because the agency has reviewed relatively few individual compounds since the 1999 classification changes.
Table 34–5. EPA versus IARC schemes for classification of carcinogens.

Data on the fungicide captan illustrate the principles of classification for probable carcinogens. Male and female rats fed ad libitum on a diet containing 8000 ppm captan (presumably a maximum tolerated dose [MTD]) for 21 weeks and containing 4000 ppm for an additional 59 weeks showed increased levels of benign and malignant endocrine tumors. Female mice also had increased numbers of neoplasms in the mammary glands, ovaries, and livers. Male rats also demonstrated testicular atrophy. Similar findings occurred in animals treated at 50% MTD dose. Mice fed on diets containing either 8000 or 16,000 ppm captan showed increased levels of duodenal neoplasms. In a separate study, male rats (70 per treatment group) fed captan at doses of 0, 25, 100, or 250 mg/kg per day for 2 years showed a slight but apparently dose-related increase in duodenal neoplasms with a tumor incidence of 1/70, 1/70, 3/70, and 4/70 in increasing dosage groups. Based on the demonstration of cancer effect in two rodent species at both the MTD and 50% of the MTD, captan is classified as a probable human carcinogen.
Compounds producing tumors only at the MTD in a single species are classified as class C or class D depending on the quality of the study demonstrating the effect. For example, the fungicide fosetyl-Al (Aliette or aluminum tris-[O-ethyl-phosphonate] received a group C classification because of benign and malignant urinary bladder tumors in a study involving male Charles River rats feeding ad libitum on food containing between 30,000 and 40,000 ppm. No tumors were seen in female rats in this study, nor in mice tested in another study (2500, 10,000, or 20,000/30,000 ppm). Because fosetyl-Al is a relatively simple compound, structurally unrelated to known classes of bladder carcinogens (aniline dyes or polynuclear aromatic hydrocarbons), the observed tumors probably represent no significance for human health.
3. Structural elements related to the occurrence of cancer— Structural elements associated with positive animal bioassays have been studied for some time. Many animal carcinogens are potent electrophiles or have electrophilic metabolic activation products.
Data on the carcinogenicity of aromatic amines illustrate the current approach to modeling structure-activity relationships. Important parameters include fat solubility (as characterized by the octanol/water partition coefficient [log p]), the energy of the highest occupied and lowest unoccupied molecular orbitals, and the polariz-ability of substituent amino (NH2) groups. These factors to some degree correspond with the elements in the scheme described earlier for predicting dermal irritation and allergenicity. As illustrated by data on fungicides such as chlorothalonil, many animal carcinogens cause skin irritation, skin allergy, or both.
B. Male Reproductive Effects
Examples of chronic effects with no recognized preceding acute illness include male reproductive toxicity, as well as occupational cancer. Two examples involve a cluster of azoospermia and sterility in workers employed in the formulation of the soil fumigant dibromochloropropane (DBCP) and chronic neurologic disorders in workers manufacturing chlordecone. In 1977, a number of male workers were discovered to have reduced or absent sperm, infertility or sterility, and testicular atrophy as a result of exposure to DBCP. A short time later, workers exposed to chlordecone (Kepone) in a manufacturing facility were found to have similar testicular changes; subsequent animal tests were confirmatory.
Identification of sterility related to the soil nematocide DBCP remains a landmark in the study of male reproductive toxicology. DBCP has moderate toxicity by oral administration (oral LD50 100 mg/kg) and low dermal toxicity (dermal LD50 1400 mg/kg). Although not producing other acute or subacute effects, rabbits inhaling 1.0 ppm DBCP for 14 weeks (estimated 490 mg/kg cumulative dose) showed reversible testicular effects. Rats also showed infertility after repeated exposures by gavage or injection (cumulative doses of 60 mg/kg or higher). Biochemical effects of the DBCP treatments include increases in luteinizing hormone (LH) and follicle-stimulating hormone (FSH).
The clinical findings in the DBCP animal studies exactly mirror the effect of DBCP on humans. The dermal exposures most commonly occurring in the formulation of DBCP did not produce acute effects, but the effect on sperm production was proportional to the duration of exposure. Specific metabolic changes associated with DBCP include decreased metabolism of glucose by sperm cells and unscheduled DNA synthesis. Nevertheless, the mechanism of the toxic effect of DBCP on germinal epithelium remains incompletely understood. Crucial endpoints in the animal studies on DBCP include sperm count, sperm morphology, and histology of testicular seminiferous tubules and epididymis.
Studies of the cumulative effect of environmental as opposed to occupational exposure to pesticides have been reported recently. A study of men evaluated at a Massachusetts fertility clinic looked at sperm quality in relationship to presence of urinary metabolites of two cholinesterase-inhibiting insecticides: carbaryl and chlorpyrifos. The authors reported increased odds of below-normal sperm count associated with increased levels of the carbaryl urinary metabolite 1-naphthol. (For low, medium, and high levels of 1-naphthol, the odds ratios [Ors] for decreased sperm count in the low, medium, and high groups were equal to 1.0, 4.2, and 4.2, respectively; p value for trend = .01.) These results were considered consistent with an earlier study of workers formulating carbaryl, a group that presumably had markedly higher exposure. The study showed a significantly higher level of abnormal sperm morphology in carbaryl workers compared with control subjects employed at the same plant.
A case-control study nested within a larger population fertility study evaluated urinary pesticide metabolites in relationship to subjects with high (control) and low (case) mean sperm concentrations. No association with urinary 1-naphthol was found. However, subjects had high levels of metabolites for three herbicides (alachlor, metolachlor, and atrazine), as well as for the insecticide diazinon. The authors were not able to identify the source of exposure to the pesticide in question or rule out a multicomparison effect as the explanation for the study findings. They also acknowledged that the short biologic half-life of the pesticides evaluated relative to the approximately 2-month process of spermatogenesis strongly suggests that the metabolites measured were surrogate measures of some other exposure.
ORGANOPHOSPHATE & CARBAMATE CHOLINESTERASE-INHIBITING INSECTICIDES
ESSENTIALS OF DIAGNOSIS
![]() Acute effects
Acute effects
• Abdominal cramps.
• Diarrhea.
• Vomiting.
• Excessive salivation.
• Sweating.
• Seizures.
• Lacrimation.
• Constricted pupils.
• Wheezing.
• Bronchorrhea.
• Skeletal muscle fasciculations.
• Weakness.
• Respiratory arrest.
• Initial tachycardia may be followed by bradycardia.
• Symptoms may persist or recur for days, especially with highly lipid-soluble agents such as fenthion or dimethoate.
![]() Chronic effects
Chronic effects
• Persistent central nervous system (CNS) dysfunction (organophosphates)—irritability, anxiety, mood lability, fatigue, short-term memory, and impaired concentration for weeks or months after acute exposures described in case reports; subclinical neurobehavioral effects in multiple studies of poisoned workers.
• Organophosphate delayed neuropathy—rapid onset of distal symmetrical sensorimotor neuropathy.
![]() General Considerations
General Considerations
Organophosphates are esters of phosphoric acid that exist in two forms: -thion and -oxon. Potency depends on the three-dimensional shape of individual compounds and their ability to bind with the cholinesterase molecule. Irreversible binding occurs with a serine molecule in the heart of the enzyme’s active site and the nonphosphate portion of the molecule (leaving group) cleaved by hydrolysis. Under most circumstances, the inhibition becomes irreversible after 24–48 hours.
Carbamates are esters of carbamic acid. The organophosphates and N-methyl carbamates are considered here a single class because they share a common mechanism of acute toxicity cholinesterase inhibition, with similar signs and symptoms of acute poisoning. Carbamates differ in causing reversible rather than irreversible cholinesterase inhibition and typically have a short clinical course. The thiocarbamates and dithiocarbamates do not inhibit cholinesterase, but many have activity against plants and fungi.
Together the carbamates and organophosphates represent one of the largest and most important classes of insecticides. Table 34–6 lists commonly used compounds according to acute toxicity. They vary widely in their cholinesterase-inhibiting potency, as reflected in their LD50 values.
Table 34–6. Organophosphate and carbamate insecticides.

As a result of their widespread use and acute toxicity, the organophosphates and carbamates are common causes of acute insecticide intoxication. Cholinesterase inhibitors produce a relatively stereotypical clinical presentation that in conjunction with determination of cholinesterase levels makes diagnosis more accurate than with other pesticides. Specific and nonspecific antidotes are available for treatment. Case reports indicate that some patients acutely poisoned by organophosphates may display persistent CNS dysfunction for weeks or months after acute poisoning. A small number of organophosphate pesticides also cause a delayed neuropathy that is correlated with inhibition of the enzyme neurotoxic esterase.
![]() Use
Use
Organophosphate pesticides were developed following World War II as a consequence of the synthesis of the organophosphate nerve gases sarin, soman, and tabun. During the 1950s and 1960s, they began replacing inorganic pesticides and organochlorines as the principal insecticides used in agriculture. Mevinphos and ethyl parathion, two of the most toxic organophosphates, were prohibited from use in the United States during the 1990s.
Subsequent to passage of the FQPA, organophosphates have been replaced by pyrethroids for termiticide applications and for crops destined to be used in processed foods. Some of the recent decrease in pesticide illnesses is probably attributable to the changes brought about by regulation of the more toxic compounds. The number of hospitalized poisoning cases has declined markedly. This decline is almost entirely due to fewer cases related to agricultural cholinesterase inhibitors.
SYSTEMIC VERSUS CONTACT INSECTICIDES
A number of organophosphates are systemic insecticides, a property that correlates to some extent with the water solubility (sol) of individual compounds. Examples include demeton (sol 3.3 g/L at 20°C [68°F]), dimethoate (sol 25 g/L at 21°C [69.8°F]), disulfoton (sol 25 mg/L at 23°C [73.4°F]), phosphamidon, and trichlorphon (sol 120,000 mg/L at 20°C [68°F]).
Many highly toxic organophosphates are no longer used, but some, such as methyl parathion, are still used in agriculture. However, even the low toxicity compounds such as diazinon and malathion are no longer sold for household use. Chlorpyrifos was perhaps the insecticide used most frequently by structural pest control operators against cockroaches and other structural pests, but its household use has also been curtailed under FQPA regulations.
A. Carbamates
The cholinesterase-inhibiting N-methyl carbamates have insecticidal properties. Carbaryl is used extensively because of its slow mammalian toxicity and relatively wide spectrum of activity. Aldicarb (sol 6000 mg/L), carbofuran (sol 320 mg/L), and methomyl (sol 57.9 g/L) are highly water soluble, systemic insecticides with use limited to agriculture. Illegal applications of these compounds do occur occasionally in urban settings. Propoxur is used by structural pest control operators and in ready-to-use home formulations.
![]() Occupational & Environmental Exposure
Occupational & Environmental Exposure
Organophosphates and carbamates are applied by a variety of techniques from aerial spraying to hand application. Granular and bait formulations significantly reduce exposure so that even highly toxic compounds such as aldicarb (0.5 mg/kg) can be used safely given proper precautions.
Organophosphate compounds show variable dissipation times. Compounds with high vapor pressures, including dichlorvos, naled, and mevinphos, have environmental half-lives measured in hours and may dissipate completely in less than 24 hours. Residues of dimethoate (LD50 180–330 mg/kg) have an environmental half-life ranging from 24 to 48 hours. Phosalone (LD50 82–205 mg/kg) residues, by contrast, have half-lives of 30 days or longer. Many organophosphates degrade rapidly in wet coastal environments but may persistent for prolonged periods in hot, dry climates. Consequently, long reentry intervals (eg, 90 days or more for ethyl parathion on citrus crops) have proved necessary to prevent acute poisoning of field workers.
The risk posed by a given level of residue depends on the crop and work activity. Residues of 7 μg/cm2 of phosalone, for example, cause no cholinesterase inhibition in workers picking citrus and peaches. Levels less than 1 μg/cm2 are associated with poisoning of workers harvesting wine and raisin grapes. A dermal residue transfer coefficient (in units of cm2/h) is used to summarize the relative levels of exposure associated with various agricultural tasks. Among various hand-harvested crops, transfer factors ranged from 5000 to 9000 cm2/h for row crops to 10,000 cm2/h for orchard crops and up to 130,000 cm2/h for hand-labor tasks (cane turning) in production of table grapes. The concept of a transfer coefficient is a useful generalization, but in practice the rate of transfer may vary considerably between fields planted with the same crop.
The available literature contains comparatively few studies on dissipation of carbamate compounds. Environmental fate data required by EPA include basic physical and chemical properties such as the Henry law constant, vapor pressure, water solubility, ultraviolet spectra, and residue data at time of harvest but not residue dissipation studies. However, summary values exist for carbaryl, aldicarb, propoxur, and carbofuran. For propoxur, residual systemic activity has been reported for up to 1 month. Data on carbaryl do not give a half-life but indicate that residues generally dissipate in less than 2 weeks. The half-life of carbofuran leaf residue is reported as longer than 4 days. Aldicarb presents a complicated picture because of its tendency to leach into groundwater. Plants convert aldicarb to systemic sulfoxide and sulfone transformation products, previously associated with episodes of consumer poisoning from watermelons and cucumbers. Variability in dissipation observed in extensive studies on methomyl suggests the need for caution in generalizing from limited data. A study in California established a 0.1 μg/cm2 safe-level for hand labor in methomyl-treated vineyards after an illness episode. Residue monitoring later revealed much longer dissipation times. It was therefore necessary to adjust the hand labor reentry interval from 7 days to 21 days.
![]() Mechanism of action & Clinical Findings
Mechanism of action & Clinical Findings
Organophosphates and carbamates are absorbed easily by inhalation, skin contact, and ingestion; the primary route of occupational exposure is dermal. They differ from one another in lipid solubility and therefore distribution in the body, particularly to the CNS.
Many commercial organophosphates are applied in thethion (sulfur-containing) form but readily undergo conversion to the -oxon (oxygen-containing) form (Figure 34–1B). Most of the -oxon forms have much greater toxicity than their corresponding -thion analogues. The conversion occurs in the environment, so the residues that crop field workers are exposed to may be more toxic than the pesticide that was applied. Some of the sulfur is released in the form of mercaptans, which produce the typical odor of the -thion form of organophosphates. The mercaptans have very low-odor thresholds, and the reactions to their noxious odor, including headache, nausea, and vomiting, often are mistaken for acute organophosphate poisoning.
The conversion from-thion to -oxon also occurs in vivo as a result of hepatic microsomal metabolism, so the -oxon becomes the active form of the pesticide in both animal pests and humans. Hepatic esterases rapidly hydrolyze organophosphate esters, yielding alkyl phosphates and phenols, which have little, if any, toxicologic activity and are excreted rapidly. Carbamates also are metabolized by the liver and excreted as metabolites in urine without evidence of significant accumulation.
Organophosphates and carbamates exert effects on insects and mammals, including humans, by inhibiting acetylcholinesterase at nerve endings. The normal function of acetylcholinesterase is the hydrolysis and inactivation of acetylcholine (Figure 34–1A). Figure 34–1B shows the reactions of organophosphates and acetylcholinesterase. The enzyme then can be dephosphorylated spontaneously and reactivated (step 3a) or aged through the hydrolysis of an alkyl (–R) group, resulting in irreversible inactivation.

![]() Figure 34–1. The irreversible inactivation of organophosphate pesticides.
Figure 34–1. The irreversible inactivation of organophosphate pesticides.
Carbamates initially react with acetylcholinesterase in the same fashion as organophosphates, resulting in accumulation of acetylcholine in the same distribution as organophosphates. The carbamyl enzyme product does not progress to an aging reaction but instead dissociates relatively rapidly. As a family, the carbamates have no known health effects other than those resulting from this acute, reversible inhibition of cholinesterase and resulting overactivity of acetylcholine.
The clinical manifestations of acute organophosphate or carbamate poisoning reflect the organs where acetylcholine is the transmitter of nerve impulses (Table 34–7). Rapid rates of cholinesterase inhibition are associated with clinical illness at levels of inhibition that may not be associated with symptoms following slower rates of inhibition. Asymptomatic subacute inhibition of acetylcholinesterase results in a state in which exposure to a dose of an organophosphate that previously would have had no effect now may lower acetylcholinesterase levels below a critical threshold and result in clinical illness.
Table 34–7. Signs and symptoms of acute organophosphate poisoning by site of acetylcholine neurotransmitter activity.

This type of cumulative inhibition of acetylcholinesterase is unlikely to occur from carbamates owing to the rapidly reversible nature of the enzyme inhibition. For a large proportion of patients with acute intoxication, the clinician will not know the identity of the particular pesticide or pesticides at the time of initial presentation, and decisions’ regarding diagnosis and management will need to be made on the basis of clinical signs, symptoms, and laboratory data.
A health effect of organophosphate pesticides that is entirely unrelated to cholinesterase inhibition is organophosphate-induced delayed neuropathy. Inhibition of an enzyme known as neurotoxic esterase(NTE), found in the central and peripheral nervous systems of various species, is an indicator of neurotoxic potential and a potential tool for biologic monitoring. Animal studies indicate that irreversible inhibition of NTE to 75% of initial activity will be followed 10–14 days later by a rapidly progressive ascending peripheral neuropathy. Currently used organophosphate pesticides with evidence of neurotoxicity include such weak cholinesterase inhibitors as merphos (S,S,S-tri-n-butyl phosphorotrithioite) and DEF (S,S,S-tri-n-butyl phosphorotrithioate), which are used as cotton defoliants rather than as insecticides.
A. Symptoms and Signs
Despite the popularity of mnemonics such as MUDDDLES (miosis, urination, diarrhea, defecation, diaphoresis, lacrimation, excitation, and salivation), the signs and symptoms of acute intoxication with organophosphates and carbamates are best learned on a neurophysiologic basis by grouping them according to the affected class of cholinergic receptor (see Table 34–7). There is some variability in para-sympathetic nervous system manifestations because they are opposed by the sympathetic nervous system, which has preganglionic cholinergic innervation. Thus the heart rate may be slow, normal, or fast and the pupils may be small, normal, or large depending on which system predominates. In one large series of organophosphate-poisoned patients, 90% had at least muscarinic manifestations, 40% both muscarinic and nicotinic manifestations, 30% had muscarinic and CNS manifestations, and 10% had all three. The number of systems involved increases with the severity of intoxication. Mild poisoning usually is manifested by mild muscarinic signs and symptoms only.
The cause of death in acute organophosphate poisoning usually is respiratory failure. Bronchorrhea or pulmonary edema, bronchoconstriction, and respiratory muscular paralysis all contribute to respiratory failure. Seizures are not uncommon in cases of severe poisoning.
1. Cardiac rhythm disturbances—Cardiac arrhythmias, such as bradycardia and heart block and cardiac arrest, are less common causes of death. Ventricular arrhythmias have been observed in some of these cases, including torsade de pointes arrhythmias, associated with prolongation of the QT interval. Medications affecting the QT interval (eg, use of odansetron for treatment of nausea) should probably therefore be avoided.
Atrial fibrillation has been reported in cases of both carbamate and organophosphate poisoning.
During the 1995 terrorist attack on Tokyo using the OP nerve agent sarin, a case of coronary spasm was observed in the precordial ECG leads, attributed to the direct effect of acetylcholine on coronary nicotinic receptors. Atherosclerotic compromise of the coronary circulation was excluded by a thallium exercise study after successful treatment of the acute poisoning.
Severe poisoning from occupational exposure to carbamates is uncommon. Owing to the rapid spontaneous reactivation of acetyl cholinesterase, workers who become ill on the job are often better by the time they are seen at a medical facility. Recorded instances of serious poisoning have involved accidental reentry poisoning or accidental direct exposures to handlers involving high-toxicity (category I) carbamates.
B. Laboratory Findings
1. Cholinesterase—A number of nonspecific laboratory findings may be present in an individual with acute poisoning, including leukocytosis, proteinuria, glucosuria, and hemo-concentration. However, changes in cholinesterase activity, along with the typical signs and symptoms, provide sufficient information for the diagnosis and management of most cases. Red cell cholinesterase is called “true” cholinesterase because it is the same enzyme present in nerve endings and because its activity more closely parallels that in the nervous system than does plasma cholinesterase, particularly in the time course of recovery, after inhibition. However, red cell cholinesterase is more difficult to measure and therefore more susceptible to analytic error than plasma cholinesterase. Organophosphates and carbamates may differentially inhibit one enzyme relative to the other, so if one and not the other appears depressed, it is conservative to assume that neuronal cholinesterase more closely corresponds to the lower of the two. For example, the commonly used organophosphate chlorpyrifos (Dursban, Lorsban) preferentially depresses plasma cholinesterase, causing illness without significant depression of red cell cholinesterase.
A number of analytic methods are used to measure both red cell and plasma cholinesterase. Results obtained by one method usually cannot be compared with results from another, even if the units expressed by each are the same. There is considerable variability in cholinesterase activity in unexposed persons, so reports of results relative to “normal” may not reflect the true level of inhibition present.
Individuals with a genetic trait for atypical plasma cholinesterase have lowered plasma but not red cell cholinesterase. They have prolonged muscular paralysis after administration of succinyl choline and other neuromuscular blocking agents that are normally metabolized by plasma cholinesterase, but they are not more susceptible to cholinesterase-inhibiting pesticides. Unlike red cell cholinesterase, plasma cholinesterase is not a reliable indicator of exposure or poisoning in these individuals.
Plasma cholinesterase production may be lowered as a result of liver disease extensive enough to impair the production of proteins such as albumin. Albumin-losing conditions, such as nephrotic syndrome, may be accompanied by elevated levels of plasma cholinesterase as a result of increased hepatic protein synthesis. The only medical conditions known to influence red cell cholinesterase activity are those associated with reticulocytosis, such as recovery from hemorrhage, pernicious anemia, and some other anemias.
Two circumstances in which cholinesterase determinations may be useful are (1) routine biologic monitoring of exposure to organophosphates and (2) diagnosis of acute poisoning. In assessing exposure to carbamates, cholinesterase depression may prove difficult to document unless treatment facilities can run cholinesterase assays on-site shortly after phlebotomy.
Severe poisoning usually is accompanied by cholinesterase levels well below normal for the laboratory. However, patients with mild to moderate poisoning often have cholinesterase levels reported as equivocal, normal, and even above normal. The diagnosis can be confirmed retrospectively by periodic (ie, weekly or biweekly) determinations of cholinesterase until levels fluctuate by no more than 30%. If the average level at this time—the retrospective baseline—is more than 30% higher than the level at the time of illness, exposure to cholinesterase-inhibiting pesticides almost certainly was present, and the illness may have been due to that exposure. The rate of recovery of red cell cholinesterase, in the absence of treatment with pralidoxime and of further exposure, depends on the rate of formation of new red cells, which is approximately 1% per day. Red blood cell cholinesterase levels will reach a plateau in about 60–70 days and plasma cholinesterase in 30–50 days.
2. Intact pesticides and metabolites—Measurement of the parent organophosphate or carbamate, or their metabolites, in blood or urine has been investigated to a limited extent. No such measurements are currently likely to be helpful in the diagnosis of acute intoxication. Measurement of alkyl phosphate metabolites in urine has not been of use in biologic monitoring of exposure because of its lack of specificity and instability. Measurement of p-nitrophenol in urine can be useful for monitoring exposure to parathion; 0.5 mg/L in a sample collected at the end of an exposure interval corresponds to exposure to parathion at the current threshold limit value (TLV). Measurement of 1-naphthol in urine is used to monitor exposure to carbaryl.
![]() Differential Diagnosis
Differential Diagnosis
Mild acute poisoning from organophosphates or carbamates most closely resembles acute viral influenza, respiratory infections, gastroenteritis, asthma, or psychological dysfunction. The most significant differential diagnosis is between severe organophosphate poisoning and acute cerebrovascular accident; unequal pupils caused by the local effect of a direct-inhibiting (oxon) organophosphate or n-methyl carbamate in one eye of a comatose patient is a potential source of misdiagnosis. Other conditions to be distinguished from acute organophosphate poisoning include heat stroke, heat exhaustion, and infections.
The major disorder to be distinguished from organophosphate-induced delayed neuropathy is idiopathic acute symmetric polyneuropathy. Other toxic and disease-related neuropathies generally are insidious in onset and slowly progressive in course.
![]() Treatment
Treatment
Treatment that is otherwise indicated should never be delayed pending determination of cholinesterase levels. The initial diagnosis can be made on clinical grounds alone, samples sent to the laboratory, and a test dose of atropine delivered. Atropine blocks the effects of acetylcholine at muscarinic receptors. A dose of atropine sulfate (0.5 mg intravenously) produces signs of mild atropinization (ie, dry mouth, dry eyes, increased heart rate, and large pupils) in a normal adult; it has no effect in an individual with organophosphate poisoning. A dose of 1–2 mg intravenously will produce marked signs of atropinization in a nonpoisoned adult and may reverse the signs of cholinergic excess in a case of poisoning.
Samples must be sent for cholinesterase measurement before administration of pralidoxime, which will regenerate cholinesterase in red cells and plasma as well as nerves. Atropine has no effect on cholinesterase levels.
Treatment of acute intoxication must be predicated on assessment of the severity of poisoning, which largely depends on clinical judgment and experience. For some occupational poisonings, removal from further exposure to cholinesterase-inhibiting insecticides may prove to be the only treatment necessary. Treatment with specific antidotes should be reserved for patients observed in the hospital setting.
Assessment of severity should focus primarily on the respiratory system because it is affected by all three types of cholinergic sites and is the critical one for survival and serious morbidity. The most commonly used severity rating defines mild toxicity as involving only muscarinic signs and symptoms, moderate toxicity as involving more than one system but not requiring assisted breathing, and severe toxicity as requiring ventilatory assistance.
Treatment modalities include the following:
1. Decontamination, including bathing of skin, shampooing of hair, or emptying of stomach, as dictated by the route of exposure.
2. Atropine sulfate in a dosage of 1–2 mg intravenously for mild to moderate poisoning, 2–4 mg intravenously for severe poisoning, as often as every 15 minutes, as needed. There is no maximum dosage. Atropine blocks muscarinic activity but not the nicotinic (muscle paralysis) or CNS effects. Patients without evidence of muscle weakness or respiratory depression may be treated with atropine alone until one or more signs of mild atropinizadon appear (ie, tachycardia, flushing, dry mucous membranes, or dilated pupils). Multiple doses may need to be administered over a prolonged time.
3. For organophosphate poisoning only, give pralidoxime chloride (2-PAM, Protopam) slowly, 1 g intravenously (no more than 0.5 g/min), repeated once in 1–2 hours and then at 10- to 12-hour intervals, if needed. Pralidoxime acts by breaking the bond between acetylcholinesterase and organophosphate, reactivating the enzyme and restoring acetylcholine activity to normal (Figure 34–1C). Its advantages over atropine include acting at the neuromuscular junction to reverse muscular paralysis and possibly crossing the blood-brain barrier to reverse CNS depression. Overdosage is not a problem if the drug is administered slowly to avoid inducing hypotension. The decision to use pralidoxime must be made reasonably soon after diagnosis because it is ineffective once aging has occurred. A high incidence of atropine toxicity may result from the often-recommended regimen of first using atropine until primary signs of atropine toxicity appear and then using pralidoxime if necessary. This may be avoided by making the decision to use pralidoxime early.
The use of pralidoxime for carbamate poisoning is controversial. Fortunately, it is rarely indicated. There is experimental evidence that pralidoxime may be helpful in the management of poisoning by some rarely used carbamates, but for most of the commonly used carbamates, this drug has not been studied. One animal study indicated that pralidoxime may be harmful in the treatment of carbaryl poisoning.
Morphine, aminophylline, and phenothiazines are contraindicated because of the increased risk of cardiac arrhythmias. Diuretics for pulmonary edema and fluids for hypotension are also contraindicated. It is recommended that atropine be withheld until adequate ventilation has reversed hypoxia because atropine may generate arrhythmias in the presence of hypoxia.
4. Emergency supportive measures: Artificial ventilation, ventilatory assistance, oxygen, and clearance of secretions.
5. Evaluation of delayed symptoms: By the time the diagnosis of organophosphate-induced delayed peripheral neuropathy is made, the initial manifestations of cholinesterase inhibition, if once present, are likely to have resolved. Administration of atropine or pralidoxime, initially or later, does not influence the course of neuropathy. Treatment of delayed neuropathy is supportive; in a few cases, mechanical ventilation has been required because of respiratory failure caused by muscular paralysis.
![]() Prognosis
Prognosis
If treatment for organophosphate or carbamate poisoning is initiated before hypoxia results in tissue damage, antidotal therapy and respiratory support should ensure complete recovery, even in the most severe cases. Persistence of manifestations beyond 24 hours indicates the possibility of continued absorption of pesticide and the need to carefully consider and examine the skin, fingernails, eyes, and gastrointestinal tract as possible reservoirs.
Sudden death can occur in a small percentage of organophosphate-poisoned patients (2% in one series) 24–48 hours after apparent complete recovery from the acute phase of poisoning and is caused by, in at least some cases, ventricular arrhythmia. Sudden relapse of acute signs and symptoms within a few days of apparent recovery has been reported occasionally, perhaps as a result of release of pesticide from fat following mobilization of the patient from bed.
Deaths have been reported as a result of accidental or deliberate ingestion of carbamates, as a result of large doses and prolonged gastrointestinal absorption, and perhaps as a complication of delayed or inadequate treatment. Intoxication from occupational exposure may be serious but is rarely fatal and usually is of brief duration. Poisoning from contaminated fruits and vegetables with high water content also may be serious but not persistent.
A number of reports describe persistent CNS symptoms in a small percentage of patients following well-documented incidents of acute poisoning from organophosphates but not carbamates. Typical symptoms include irritability, depression, mood lability, anxiety, fatigue, lethargy, difficulty in concentrating, and short-term memory loss. Limited studies suggest that neurobehavioral test results and electroencephalograms may be different for such patients when compared with controls. Symptoms may persist for weeks or months after the initial intoxication and are difficult to distinguish from psychological reactions likely to occur after such an event. Sympathetic counseling and judicious use of antianxiety agents, when appropriate, generally will be more effective than intensive psychotherapy and antipsychotic medicine.
![]() Skin Effects
Skin Effects
Organophosphates generally have high octanol/water partition coefficients and high dermal absorption rates, but most cause minimal skin irritation. Skin effects derive from the reactivity of the nonphosphate portion (termed the leaving group) of individual compounds. For example, the irritant compounds dichlorvos and naled both have reactive halogen atoms in their leaving groups. Dichlorvos also has an unconjugated carbon-carbon bond. Some organophosphate formulations produce transient irritation in the Draize assay, including acephate, diazinon, dimethoate, malathion, methamidophos, methidathion, oxydemetonmethyl, phosmet, and sulfotep; many cause mild primary irritation in the challenge (epicutareous) phase of the guinea pig maximization test. Clinically, acute irritation with these compounds occurs most frequently with accidental direct exposure to pesticide handlers (mixer/loader/applicators). These types of exposures also may provoke systemic effects; in cases of organophosphate-associated dermatitis reported from Japan, approximately 25% had at least mild coincident symptoms of systemic poisoning. Systemic poisoning also was reported in a US case of irritant dermatitis caused by dichlorvos.
Buehler (epicutaneous) sensitization assays show negative findings for acephate, chlorpyrifos, dimethoate, malathion, methamidophos, methidathion, and phosmet. Nevertheless, several are sensitizers in the guinea pig maximization test (induction of allergy by subcutaneous injection), including diazinon, fenitrothion, and methidathion. Cases of possible contact sensitivity to organophosphates have been reported for omethoate and dimethoate. A case-control study of dermatitis in farmers identified allergic reactions to malathion and oxydemeton-methyl, as well as the carbamate compounds carbofuran and carbaryl. Further studies identified allergic contact dermatitis caused by malathion and naled, but the patch testing conducted did not meet current standards, especially with regard to identifying nonirritant concentrations to conduct of the patch procedure.
A case report from Australia identified an isomer and contaminant of diazinon called isodiazinon (2-isopropyl-6-methyl-4-S-pyrimidinyl diethylthiophosphate) as a possible cause of porphyria cutanea tarda in a sheep rancher. Investigation in a rat study showed that isodiazinon affected porphyrin synthesis by inhibiting the liver enzyme ferrochelatase. Other noncontact reactions include a case of erythema multiforme associated with indoor use of methyl parathion, an unusual contact reaction to ethyl parathion resembling erysipeloid, and a case of systemic organophosphate poisoning.
![]() Chronic Health Effects
Chronic Health Effects
A. Neurobehavioral Effects
Conjectured persistent sequelae of organophosphate poisoning remains a subject of controversy.
Numerous studies document subclinical neurobehavioral deficits relative to control subjects in previously poisoned workers and to a lesser extent in workers with applicators with long-term exposures who never experienced acute poisoning. The recorded deficits include vibrotactile sensitivity, decreased sustained attention, and decreased speed of information processing, memory and abstraction, and cognitive tests.
Poisoning by the organophosphate nerve agent sarin produced persistent neurobehavioral deficits, including significant amnesia in some victims of the 1995 terrorist attack on the Tokyo subway. The most severe deficits were seen in patients who experienced prolonged hypoxia. Cases of posttraumatic stress also occurred. Findings in less severely poisoned cases more closely resembled those seen in studies of applicators poisoned by organophosphate insecticides.
Studies of workers who handled organosphosphates without a history of overt poisoning show less consistent findings of subclinical neurobehavioral impairment. A study of sheep dippers handling organophosphates showed findings similar to the studies of overtly poisoned workers. Other studies of nonpoisoned organophosphate handlers demonstrated equivocal or negative findings.
Although none of the studies of poisoned workers shows significant clinical impairment, all involve cross-sectional measurement of neurobehavioral function, most less than 10 years after poisoning. From currently available information, it cannot be ascertained if the subclinical deficits observed eventually might progress to clinically significant impairments. A study of aging did show an association between pesticide exposure recorded at the outset of the study and mild cognitive defects recorded 3 years subsequently but did not identify exposures to particular pesticides or classes of pesticides.
B. Reactive Airways
Possible reactive airways cases, an asthma-like respiratory condition that occurs in some individuals exposed to environmental irritants, are commonly diagnosed, by means of a specialized pulmonary function test called a methacholine challenge. Occasionally, cases of reactive airways disease or new-onset asthma are associated with organophosphate exposure or organophosphate contaminants. Experimental studies in guinea pigs demonstrate that reactive airways following organophosphate exposure is more severe in animals previously sensitized to ovalbumin. Sensitized animals also demonstrated an increased pharmacologic sensitivity to airway constriction induced by inhibition of cholinesterase. The case described below provides a possible example:
A severe case of asthma in a nursery worker was reported to the California illness registry in 2006. The worker entered a greenhouse shortly after a Bt application and an application of diazinon in a neighboring area of the nursery. She had a prior history of sometimes poorly controlled asthma. After work she suffered a near respiratory arrest and was hospitalized. An extensive investigation followed, documenting prior workplace history. She had positive RAST/immunocap in vitro reactions to multiple pollens, plants and molds. Initial testing at an academic allergy center appeared to show an in vitro reaction to Bacillus thuringiensis (Bt) antigens, similar to those reported from farm workers in Ohio and from Denmark, but direct provocation testing (prick testing) was negative. Her near-respiratory arrest may have been solely related to her poorly controlled allergically mediated asthma. However, as noted above, studies in guinea pigs demonstrate that reactive airways following organophosphate exposure is more severe in previously sensitized animals (ovalbumin antigen). Sensitized animals also demonstrated an increased pharmacologic sensitivity to airway constriction induced by inhibition of cholinesterase.
C. Carcinogenicity, Teratogenicity, Effects on Childhood Development, Male Reproductive toxicity
1. Carcinogenicity—Most of the carbamates and organophosphates show no evidence of carcinogenicity in animal tests. Exceptions include probable (cancer classification B2) animal carcinogens propoxur (bladder cancer and liver cancer) and dichlorvos (gastric tumors in female mice, leukemia in male rats). Possible (cancer classification C) carcinogens include acetamide, a metabolite of methomyl and thidiocarb (liver cancer in male and female rats), acephate, dimethoate, parathion, methidathion, phosphamidon, tetrachlorvinphos, and tribufos.
Some cancer case-control studies conducted in the 1990s showed associations between handling organophosphates and occurrence of non-Hodgkin lymphoma and leukemia. Effects of the reported expoures on the immune system were hypothesized as a possible mechanism. An effect of a specific compound, for example, dichlorvos, which is recognized as an animal carcinogen, also could explain the findings. A case-control study investigating causes of aplastic anemia in Thailand also revealed a strong association with dichlorvos and with the carbamate insecticide propoxur. In common with the studies of lymphoma, the study employed questionnaire information to assess exposure, and the findings could have been attributed to recall bias.
2. Teratogenicity and effects on childhood neural development— The organophosphate compounds generally are not teratogenic below maternally toxic doses. As discussed earlier, the carbamate compound carbaryl is a spermatotoxin in rodents; a study in manufacturing and formulating workers demonstrated, as discussed earlier, an effect on sperm morphology. Sperm effects related to environmental exposures to carbaryl also have been reported (see above). Carbaryl is also teratogenic to beagle dogs but not to rodent species.
The FQPA has inspired considerable work on animal models for developmental neurotoxicity, including prenatal and postnatal exposures. Cohort studies in New York and California evaluating possible long-term effects of prenatal organophosphate exposures have studied multiple outcomes including cognitive development, attention disorders and other neurobehavioral outcomes. Parallel studies on animals have shown similar effects at somewhat different dose levels.
Studies in both cohorts measured exposures prospectively. Although total exposures during pregnancy were not quantifiable, they were assumed to be related to short-term measurements done by air sampling, blood measurements, and measuring of urinary metabolites. Exposures to both groups were probably higher than the reference population in the NPB cohort but well below the no effect levels in animal studies of impaired neurodevelopment related to chlorpyrifos and other organophosphates.
Some differences in outcomes were noted between the two cohorts, but both observed an increased number of abnormal neonatal reflexes, attention deficit problems in early childhood, and effects on cognition apparent as the children reached school age. Both outcomes and the excretion of OP metabolites were associated with PON1 activity. PON1, or serum paraoxonase, is an enzyme encoded by the PON1 gene. PON1 is responsible for hydrolysing organophosphate. It may be a confounding factor in these studies that has not been completely explored. Most studies of both cohorts involved batteries of tests and psychological instruments, raising the possibility that some identified significant outcomes were related to multiple comparisons.
3. Male reproductive effects—Animal feeding studies show effects on some measures of sperm quality and effects on reproductive hormones and after prolonged exposures to organophosphates or carbamates. Possible mechanisms of action include alkylating or phosphorylating DNA or nuclear proteins, enzyme inhibition in reproductive cells and endocrine effects. Most studies were conducted in rodents, but rabbits and fish were also employed.
Some cross-sectional studies demonstrated associations between pesticide OP exposure and levels of reproductive hormones. However, the results were not entirely consistent between studies. There were more consistent associations between high dose OP exposures and measures of damage to sperm, including aneuploidy and DNA fragmentation.
ORGANOCHLORINE INSECTICIDES
ESSENTIALS OF DIAGNOSIS
![]() Acute effects
Acute effects
• CNS excitation—irritability, excitability, dizziness, dis-orientation, paresthesias, tremors, and convulsions.
![]() Chronic effects
Chronic effects
• Cancer in animals.
• Case reports of aplastic anemia.
![]() General Considerations
General Considerations
The organochlorine insecticides are chlorinated hydrocarbon compounds of cyclic structure and high molecular weight. In contrast to chlorinated hydrocarbon solvents and fumigants, they are of low volatility and are CNS stimulants rather than general anesthetics. The prototype organochlorine, dichlorodiphenyltrichloroethane (DDT), was discovered in 1939 and first registered in the United States in 1948. Until it was banned from most uses in the United States in 1972, more than 4 billion pounds were applied in agriculture and in control programs aimed at mosquitoes and other insects that transmit human disease, such as yellow fever and malaria. The cyclodiene family compounds aldrin, dieldrin, chlordane, and heptachlor were first registered in the United States between 1948 and 1952. They were phased out after the prohibition of DDT and then banned completely in 1987 (aldrin and dieldrin) and 1988 (chlordane and heptachlor). Similar to DDT, the cylcodienes were environmentally persistent and accumulated in the wildlife food chain. The agricultural registrations of lindane have been canceled, although it is still available in most of the United States as a treatment of resistant scabies (Table 34–8).
Table 34–8. Organochlorine pesticides still in use in the United States.

Dicofol and endosulfan are the only organochlorine compounds still in use in the United States. These compounds have shorter environmental and biologic half-lives than the canceled organochlorines, although longer than typical organophosphates or typical pyrethroids. For example, dicofol has a half-life of 60 days in soil, and a variable half-life in water, depending upon the pH. The soil half-life of endosulfan varies from 19 to 33 days. By comparison, the half-life of DDT in the soil ranges from 2 to 15 years.
![]() Occupational & Environmental Exposure
Occupational & Environmental Exposure
There is little information on current occupational exposure to organochlorines. Owing to their persistence and bioaccumulation, environmental exposure to organochlorines will continue for years, even to compounds no longer in use, although there is evidence that levels are decreasing. Most of the world’s population has measurable levels of DDT and its metabolites in fat and blood. Elimination times for dicofol are approximately 192 hours, for endosulfan days to weeks. Metabolism of lindane (γ-isomer of hexachlorocyclohexane) is rapid.
![]() Mechanism of Action & Clinical Findings
Mechanism of Action & Clinical Findings
The organochlorines are well absorbed by inhalation or ingestion but are absorbed more slowly through the skin. Most are highly fat soluble and are distributed to adipose tissue, the liver, and the nervous system. Most are metabolized by the liver and excreted in urine as metabolites. For some, this is a slow process, so accumulation in adipose tissue occurs during chronic exposure. DDT is metabolized and excreted slowly and is found in the fat of most persons; average DDT levels in the fat of Americans have been decreasing since cessation of its use in the United States.
Although the clinical picture of acute intoxication is similar for members of this family of compounds, their precise mechanism of action is unknown, and whether they share a common mechanism is, for that reason, uncertain. These chemicals cause CNS excitation and dysfunction with little pathologic change presumably as a result of changes in the transmission of nerve impulses. They cause hepatocellular necrosis in high doses, hepatocellular hypertrophy, and carcinomas—particularly in mice—at lower doses, and are inducers of hepatic microsomal enzymes.
A. Symptoms and Signs
Acute or subacute intoxication from organochlorines produces a picture of generalized CNS excitability and dys-function: apprehension, excitability, dizziness, headache, disorientation, confusion, disequilibrium, weakness, paresthesias, muscle twitching, tremor, convulsions, and coma. Nausea and vomiting are common after ingestion but not after dermal exposure, which is the primary route in the workplace. Most organochlorines are formulated with organic solvents, which may account for CNS depression, particularly after ingestion. Fever commonly occurs after seizures but may be a result of seizure activity rather than an effect of the pesticide. Chlordecone, no longer in use, caused a unique chronic intoxication, as discussed below.
There are a number of case reports and case series suggesting an association between aplastic anemia and exposure to organochlorine insecticides. These reports cannot exclude the possibility of coincidental occurrence of this rare condition with relatively common exposures.
B. Laboratory Findings
With the exception of measurement of parent compounds or metabolites in biologic samples, laboratory findings are nonspecific. Electroencephalography may show generalized seizure activity. For some compounds, a correlation between biologic levels and degree of poisoning is known, but such levels are rarely available in time to assist in management of acute poisonings.
![]() Differential Diagnosis
Differential Diagnosis
Severe organochlorine poisoning usually occurs following obvious overexposure and, as a result, does not present a problem in diagnosis. Other causes of CNS overactivity or seizures must be considered, particularly drug intoxications. Infections of the nervous system must be considered in the presence of seizures. Pneumonitis may be present as a result of aspiration of organic solvent.
![]() Treatment
Treatment
There are no antidotes, so treatment is supportive, directed primarily at maintenance of respiratory function and prompt management of seizures with anticonvulsant medication. Decontamination of the skin, hair, and gut (as appropriate) is important, as in all cases of acute intoxication. Cholestyramine has been shown to accelerate the elimination of chlordecone (from an average half-life of 165 days to a half-life of 80 days) but has not been studied for use in management of poisoning with any other organochlorine.
![]() Prognosis
Prognosis
Uncontrolled seizures may result in anoxic brain or other organ damage. If hypoxia is prevented, recovery should be complete.
![]() Chronic Effects
Chronic Effects
Chlordecone caused a constellation of nervousness, tremor, weight loss, opsoclonus, pleuritic and joint pain, and a syndrome resembling pseudotumor cerebri in overexposed production workers. These chronic effects were mitigated by treatment with cholestyramine to enhance elimination of chlordecone.
Another concern regarding organochlorines is the risk of cancer. Most of the organochlorines cause liver cancer in one or more rodent species. Evidence for excess liver and biliary tract cancer in humans is equivocal. A study of manufacturing workers showed excess biliary tract cancer in a facility producing aldrin, dieldrin, endrin, and DBCP and a nonsignificant increase in a plant producing DDT. Facilities producing other organochlorines (principally heptachlor and chlordane) did not show excess cancer. A principal limitation of the study was incomplete exposure information.
Dieldrin is a recognized animal carcinogen, producing liver tumors in mice, but no dieldrin-related elevation in the occurrence of cancer was noted in Dutch workers involved in manufacturing dieldrin and aldrin. A NIOSH mortality study of US workers producing DDT, chlordane, dieldrin, and aldrin showed an elevated risk of biliary tract cancer (SMR [standardized mortality ratios] = 4.87), but no increase of lung cancer.(SMR = 0.87). This finding was coherent with the increase in liver cancers in animal feeding studies
A 2007 publication based on the Agricultural Health Study (AHS) cohort evaluated the risks of cancer following exposure to organochlorines. A self-administered questionnaire completed at study enrollment assessed a history of ever using the insecticides aldrin, chlordane, DDT, dieldrin, heptachlor, lindane, or toxaphene. No clear evidence of an association was found between use of organochlorine insecticides and all incident cancers.
There was significant association (relative risk = 2.9) between high cumulative exposure to toxaphene (>25 lifetime days) and melanoma (5 exposed cases, with 53 total cases). It was unclear from review of prior literature whether this was a chance finding. Toxaphene causes liver and thyroid tumors in animal studies and appears genotoxic based on the sister chromatid exchanges assay and an epidemiologic study showing an increased frequency of chromosomal aberrations was reported among workers exposed to toxaphene.
A significant association (relative risk = 2.7) was also found for high cumulative exposure (>9 lifetime days) to chlordane and rectal cancer. There has been no consistent association between rectal cancer and either farming or pesticide exposure. A significantly elevated rate of rectal cancer was found in a cohort of Dutch workers producing dieldrin and aldrin (cyclodiene compounds related to chlordane), but the risk appeared to be highest in the workers with the lowest exposure.
A significant association was noted for high cumulative exposure to dieldrin (>9 lifetime days) and lung cancer (relative risk = 2.8), based upon 5 exposed cases, with 104 total cases. An elevated risk of lung cancer for applicators was also reported in the Agricultural Health Study. The AHS cohort study showed another significant association (relative risk = 2.6), between high cumulative exposure to the cyclodiene compounds chlordane/heptachlor (>9 lifetime days) and leukemia, based upon 13 exposed cases. Prior reports described cases of leukemia following exposures to chlordane/heptachlor exposures used in structural pest control.
A. Breast Cancer and DDT
Studies evaluating the relation between DDT/DDE and breast cancer strongly suggested by the 1993 study have been inconsistent. For example, a review of 26 studies published between 2000 and 2006, employing a variety of comparison points, most odds ratios overlapped one. Studies showing elevated risk included a 2003 study describing elevated risk in European whites with detectable DDT(>0.5 ng/g lipid vs <0.5) (OR ¼ 5.64; 95% CI, 1.81–17.65) or DDE (OR ¼ 2.21; 95% CI, 1.41–3.48). A 2000 study from Mexico City (highest quintile (>3490 ng/g lipid) compared with lowest quintile (<1170) (OR ¼ 3.81; 95% CI, 1.14–12.8). The remaining studies did not show a link between DDE and breast cancer and stratification by menopausal status, tumor hormone receptor status, parity, breastfeeding, or body mass index did not reveal consistent associations.
Apart from DDT/DDE, positive associations between other organochlorines and breast cancer have been reported. In a Danish case-control study serum levels of dieldrin were associated with increasing risk from the lowest exposure group (<6.9 ng/g lipid) to the highest (>36.0 ng/g lipid): odds ratio = 4.6 (1.8–11.5). The observed increased breast cancer risk associated with exposure to dieldrin derived from women who developed an estrogen receptor negative (ERN) tumor (odds ratio [OR] I vs IV quartile, 7.6, 95% confidence interval [95% CI] 1.4–46.1, p-value for linear trend 0.01). Because the compounds evaluated differed from study to study, it was not possible to check for consistency of results across multiple studies.
B. Prostate Cancer
A 2013 study evaluated the risk of lifetime exposure to 48 pesticides in 1,962 incident prostate cancer cases, and subset of 919 aggressive cancers: OP compounds fonofos (rate ratio [RR] for the highest quartile of exposure [Q4] versus nonexposed = 1.63, 95% CI: 1.22, 2.17; p [trend] <.001); malathion (RR for Q4 vs. non-exposed = 1.43, 95% CI: 1.08, 1.88;
P(trend) = 0.04); and terbufos (RR for Q4 vs nonexposed = 1.29, 95% CI: 1.02, 1.64; p [trend] = .03). The risk of aggressive cancer was elevated for the organochlorine compound aldrin (RR for Q4 vs nonexposed = 1.49, 95% CI: 1.03, 2.18; p [trend] = .02).
C. Male Reproductive Effects
The organochlorine compound chlordecone (Kepone) produced demonstrable effects on spermatogenesis in both animal studies and workers. Adult male rats fed 15 or 30 ppm chlordecone for 90 days showed reversible changes in mobility and viability of spermatozoa.
Workers affected by neurologic symptoms also experienced oligospermia and decreased sperm motility and had abnormal testicular biopsies. Because organochlorines have recognized estrogenic properties, it appears possible that the decreased sperm production associated with chlordecone toxicity derives from an endocrine mechanism rather than a direct effect on germinal epithelium. However, this supposition has not been supported by the findings in animal studies investigating the compound’s estrogenic properties.
PYRETHRUM & SYNTHETIC PYRETHROID INSECTICIDES
ESSENTIALS OF DIAGNOSIS
![]() Acute effects
Acute effects
• Topical paresthesias and irritation of the skin, ocular and respiratory irritation, systemic poisoning on ingestion.
• Allergic contact dermatitis: erythema, vesicles, papules, and itching.
• Allergic rhinitis: nasal congestion and sore throat.
• Asthma: wheezing, cough, chest lightness, and dyspnea.
![]() General Considerations
General Considerations
Pyrethrum is a partially refined extract of the chrysanthemum flower and has been used as an insecticide for more than 60 years. There are six known active compounds in pyrethrums, including two esters known as pyrethrins(pyrethrin I and pyrethrin II). Chrysanthemum and pyrethrum have long been recognized to cause allergies. In rodent acute toxicity tests, pyrethrin mixtures demonstrate remarkable variability in effect (Table 34–9), but most mixtures have oral LD50s greater than 1000 mg/kg. Pyrethrum and pyrethrin mixtures do not cause systemic toxicity on dermal application.
Table 34–9. Pyrethrin and pyrethroid acute toxicity.

Synthetic pyrethroids structurally resemble pyrethrins, with modifications that increase both toxicity and stability. Between two subtypes of synthetic pyrethroids, designated as type I and type II (cyanohalogen) compounds, the latter generally demonstrate greater toxicity. Animal studies also demonstrate some differential age-related toxicity of type II pyrethroids. Deltamethrin, for example, has an LD50 of 5.1 for 11-day-old male rats versus 81 mg/kg for 72-day-old male rats. Because of this possible increased susceptibility, illegally imported pyrethroid chalk products (containing deltamethrin), resembling a form of hard candy, pose a risk of systemic poisoning to children. A published case report describes poisoning of a 4-year-old child with a deltamethrin chalk at an estimated dose of 2 mg/kg.
![]() Use
Use
Between 1990 and 2010 the use of pyrethroids has gradually declined in the United States. There are several hundred commercial products containing pyrethrum (active ingredients pyrethrin I and pyrethrin II) and pyrethroids. Most contain piperonyl butoxide as a synergist and often with an additional insecticide, such as a carbamate or organophosphate to delay pyrethroid metabolism in the target insects. Many are available for home use against flies, mosquitoes, and fleas. The usual household formulation contains about 0.5% active ingredient. The greater stability of the synthetic pyrethrins has made them useful in agricultural applications. The petroleum solvents contained in most ready-to-use mixtures and “bug bombs” as “inert ingredients” have their own toxicity. A number of spectacular fires and explosions have occurred with overapplication of the indoor “bug bomb” products presumably because the petroleum distillates and propellants achieve such high concentrations.
![]() Occupational & Environmental Exposures
Occupational & Environmental Exposures
The low toxicity of pyrethrins has resulted in little interest in quantifying exposure levels apart from concern about hazards to small children from residues on interior surfaces from mixtures that contain organophosphates and carbamates. Occupational pyrethroid exposures in the United States principally result in topical symptoms, but systemic poisoning caused by pyrethroid compounds has been reported in applicators from China.
Indoor exposures to residue of pyrethroids also may produce irritant upper respiratory and ocular symptoms. Similar symptoms also have been reproduced in limited experimental studies in humans and in California orange harvesters working in orchards recently treated with cyfluthrin. These symptoms resemble acute allergy but probably represent an effect of the pyrethroids on nerve endings in the eye and the respiratory tract.
Residue dissipation for cyfluthrin has been characterized to only a limited extent, with available data showing marked variability. In some orchards in California’s central valley, rapid dissipation occurred. In others, there was biphasic decay with an initial half-life of 11 days, followed by a half-life of 32 days. At 65 days postapplication, 10–20% of the initial residue still remained.
Pyrethroids generally have not been thought to accumulate in the environment. Nevertheless, a recent study demonstrated permethrin and bifenthrin accumulating in runoff sediment in southern California. Under both aerobic and anaerobic conditions, the half-life for bifenthrin ranged from 8 to 17 months.
Concentrations in sediment may be high enough to affect growth of some crustaceans in soil sediment, such as the amphipod Hyalella azteca.
Indoor dissipation of pyrethroids also may be unpredictable. A German study, for example, demonstrated multiphasic dissipation of permethrin following indoor application, with a final slow-phase half-life of up to 2 years. The levels have been reported to be high enough to cause irritant symptoms in sensitive individuals.
![]() Mechanism of Action & Clinical Findings
Mechanism of Action & Clinical Findings
The pyrethrins and pyrethroids are absorbed from the gastrointestinal tract and hydrolyzed in the gut and tissues, and excreted rapidly. They are very slowly absorbed from the skin. They function principally by excitation of the sodium channels in the nervous system. Data on acute toxicity in animals shows moderate oral toxicity and very low systemic toxicity following skin contact (see Table 34–9).
Possible interactions between pyrethroids and other insecticides have received little notice. There may be a possible metabolic interaction with organophosphates and with the insect repellant diethyltoluamide (DEET). Workers exposed to the type II pyrethroid deltamethrin excreted principally metabolic transformation products. Workers also exposed to the organophosphate compound methamidophos excreted unchanged active pyrethroids (fenvalerate or deltamethrin) in the urine, implying an inhibition of pyrethroid metabolism by methamidophos. The effect has been documented clearly in fish exposed simultaneously to esfenvalerate and diazinon. In that experimental system, the esfenvalerate toxicity was enhanced by diazinon inhibition of the enzyme carboxyl esterase, usually responsible for pyrethroid metabolism.
A. Signs and Symptoms
The most common effect of pyrethrum exposure is allergic contact dermatitis, which is manifested by itching and an erythematous vesicular rash. Bullae, edema, and photosensitivity also may occur. Allergic rhinitis is not uncommon, with nasal congestion, sneezing, and sore throat. Asthma and hypersensitivity pneumonitis have been reported but are uncommon. Dyspnea, cough, and wheezing indicate asthma, although these manifestations plus fever, malaise, and pulmonary infiltrates are indicative of hypersensitivity pneumonitis. Anaphylaxis with bronchospasm, laryngeal edema, and shock has been reported occasionally after inhalation of pyrethrum. A case report published in 2000 reported an apparent fatal allergic reaction in an 11-year-old using a pet shampoo containing 0.2% pyrethrins.
These allergic manifestations have been reported only infrequently from exposure to synthetic pyrethroids, but cases of related allergic contact dermatitis have been documented. The most common effect of the pyrethroids appears to be topical paresthesias and ocular or respiratory irritation.
B. Laboratory Findings
Skin testing can aid in the diagnosis of sensitivity to pyrethrum. There are no biologic monitoring methods for exposure to pyrethrum or pyrethroids available routinely from commercial metabolites.
![]() Differential Diagnosis
Differential Diagnosis
Allergy to other pesticides, plants or flowers, insect stings, and household products must be considered in the evaluation of one of the allergic manifestations of pyrethrum.
![]() Treatment
Treatment
The key to treatment of any allergy is removal from exposure to the allergen. Allergic contact dermatitis may be treated with application of topical steroid preparations. Allergic rhinitis may be treated with antihistamines, decongestants, and a steroid nasal spray, if needed. Asthma is treated with bronchodilators and steroids as appropriate. Anaphylaxis may require epinephrine, aminophylline, or a parenteral corticosteroid. Topical application of creams containing vitamin E may alleviate pyrethroid-related paresthesias if applied shortly after skin contact. Acute respiratory irritation is short lived and does not require medical treatment unless lower respiratory symptoms are also present.
![]() Prognosis
Prognosis
If the diagnosis is correct, treatment is prompt, and removal from exposure is effective, recovery should be rapid and complete. In a pediatric ingestion case involving deltamethrin, transient CNS symptoms occurred, but recovery was rapid following gastric decontamination. The overall time course is comparable with that of an acute carbamate intoxication.
SYNERGISTS (PIPERONYL BUTOXIDE)
Although there are a few other examples, by far the most common synergistic insecticide combination, is that of piperonyl butoxide with pyrethrins.
![]() Use
Use
Piperonyl butoxide is used as an insecticide synergist with pyrethrins in ratios of 5:1 or 20:1 in a variety of formulations, many of which are available for home use. They are used primarily for flies, mosquitoes, and fleas, often in combination with a carbamate or organophosphate.
![]() Occupational & Environmental Exposure
Occupational & Environmental Exposure
Most exposure occurs with use of household ready-to-use formulations.
![]() Mechanism of Action & Clinical Findings
Mechanism of Action & Clinical Findings
Piperonyl butoxide is poorly absorbed from the gastrointestinal tract and probably poorly absorbed dermally. It is metabolized but also retained unchanged to an uncertain degree in rodents. Its mechanism of action is inhibition of enzymes that metabolize pyrethroids and pyrethrins in insects (hepatic mixed-function oxidases).
A. Symptoms and Signs
There are no reports of clinical illness occurring as a result of isolated exposure to piperonyl butoxide.
B. Laboratory Findings
There is no evidence of enzyme inhibition from piperonyl butoxide in humans. A single oral dose of 50 mg did not change the metabolism of antipyrine in eight volunteers.
![]() Differential Diagnosis
Differential Diagnosis
Any illness occurring in an individual exposed to a formulation containing piperonyl butoxide probably is caused by another ingredient, such as allergy to pyrethrum, an effect of a carbamate or organophosphate, or something other than the pesticide.
![]() Treatment
Treatment
Treatment, if required, is symptomatic. Vitamin E cream can diminish paresthesias after skin contact with pyrethroids if applied within a few hours of exposure, but has limited availability.
Treatment of ingestions may require a period of hospital observation depending upon the quantity ingested.
![]() Prognosis
Prognosis
The outcome depends on the actual diagnosis.
![]() Safety Issues
Safety Issues
Foggers containing pyrethroid and pyrethrin mixtures are commonly used to control household insect pests. Sold as ready-to-use (RTU) devices, the products are often used by renters or homeowners. Propellants in the devices are typically propane or another form of natural gas. When foggers are used without extinguishing pilot lights or other ignition sources, fires or explosions can result. Most do not result in pyrethroid illnesses in either householders or first responders, presumably because the active ingredients decompose in fires caused by the flammable propellants.
![]() Chronic Effects
Chronic Effects
Several of the synthetic pyrethroids cause cancer at maximum tolerated doses in animals, including bifenthrin, cypermethrin, permethrin, and tetramethrin. However, none are classified as probable human carcinogens. The pyrethroid compounds do not cause teratogenic effects in animal studies. For example, in a study of cyfluthrin, offspring of animals dosed at 0, 3, 10, or 40 mg/kg per day on gestation days 6–15 showed no signs of malformations. Treatment-related maternal toxicity occurred at doses of 10 and 40 mg/kg per day, including decreased weight gain, hypoactivity, locomotor incoordination, increased salivation, and a 15% mortality rate. Piperonly butoxide has complex, often biphasic effects on mixed function oxidases (cytochrome p450 system) in laboratory animals. Following oral administration, initial enzyme inhibition occurs, followed by induction. This may increase the metabolism of other xenobiotic compounds. In experimental studies this has been associated with relative tolerance to the organophosphate compounds methyl parathion, dimethoate, azinphos methyl and the neurotoxic esterase inhibitor triorthocresyl phosphate.
![]() Reproductive Effects
Reproductive Effects
Rats treated with 5 mg/kg deltamethrin daily for 4 weeks demonstrated increased levels of reactive oxygen species. Bone marrow DNA damage showed increases including chromatid breaks, deletions, fragments, and gaps. Hormonal effects were noted including decreased testosterone, LH, and FSH.
The effect of high-dose fenvalerate in utero exposure (40 mg/kg) on reproduction of offspring was evaluated. At post natal day (PND) 75, parameters female offspring had decreases in ovarian weight, preantral follicles, and corpora lutea. There was also an increase in the resorption number, when fertility test was performed at PND 80. Maternal fenvalerate exposure at 35 mg/kg produced a parallel decrease in fertility, marked by decreased the number of mature seminiferous tubules and reduced the number of epididymal spermatozoa in adult male offspring.
![]() Ecological Toxicity
Ecological Toxicity
Pyrethroids can be highly toxic to fish and aquatic invertebrates in the laboratory and are highly toxic to bees and other beneficial insects. The 96-hour LC50 of deltamethrin for fish, for example, ranges between 0.048 and 5.13 μg/L. Although pyrethroids dissipate rapidly through photolysis or hydrolysis, they may persist in aquatic sediments. Pyrethroids are also highly toxic to bees and other nontarget arthropods in the laboratory. Ecologic effects may be mitigated by dissipation of residues, allowing recovery of affected populations.
Neonicotinoid Insecticides
The nicotine analogues (including acetamiprid, clothianidin, imidacloprid and thiacloprid) selectively stimulate the nicotinic receptors in insect and mammalian nervous systems. Their differential toxicity depends upon their selectivity for stimulation of insect nicotinic receptors, and consequently they have lower toxicity profiles than nicotine and many of the cholinesterase-inhibiting insecticides. Many of the oral LD50s are comparable to low or moderate toxicity cholinesterase inhibitors, but have very low dermal toxicity and inhalation toxicity (Table 34–10).
Table 34–10 Neonicotinoid compounds, acute toxicity, pharmacokinetic, and environmental fate data

Neonicotinoids have low vapor pressures and do not dissipate by evaporation, potentially resulting in long environmental dissipation times. Imidacloprid, for example, has a soil half-life of 26.5–229 days, depending upon temperature, pH, environmental moisture, and incorporation into layers of soil where no photolysis occurs. For acetamiprid the reported soil half life is 1– 8 days in soil and 34 days in aqueous solution when degradation takes place solely by photolysis. The soil half-life of chlothianidin is extraordinarily long, more than 1000 days, but residues close to surface have a photolysis half-life of 34 days. The aqueous anaerobic half-life is 27 days.
Neonicotinoid Illnesses
Illnesses associated with neonicotinoids usually involved mixed exposure to multiple categories of insecticides (typically cholinesterase inhibitors and/or pyrethroids). These were principally topical effects on eye or skin or mild systemic illnesses. Most were agricultural or associated with the use of flea control. Data on 68 poisoning cases were recently reported from Sri Lanka. Typical illnesses were mild, associated with nausea, vomiting, headache, and diarrhea.
Unlike the cholinesterase inhibitors, whose actions may partially be imitated by ingestion of imidacloprid and related compounds, no specific antidote exists for treatment of poisoning by neonicotinoids. As illustrated by the cases above, supportive ventilation may prove necessary in some cases, but most cases treated prior to frank respiratory arrest are likely to survive. Treatment of agitation and delirium may also be necessary for a number of days.
Specific laboratory testing: cholinesterase levels are not useful except to exclude simultaneous exposure to organophosphates and carbamates; measurement of imidacloprid in gastric aspirate or in the blood levels is not likely to be clinically useful but may provide documentation of the cause of poisoning.
Ecological Effects
Neonicotinoids are absorbed into plant tissues after application, with measurable levels in leaves, pollen, and plant vascular fluids. This creates the possibility of secondary poisoning of non-target insects, including honeybees, parasitic wasps, and predaceous ground beetles. The effect on nontarget insects depends upon species-sensitivity, method of application and route of intake. Honey bees appear to be uniquely sensitive to the effects of these compounds compared to birds, fish, and mammals, as well as even other insects. Neonicotinoids metabolites may be even more toxic to the bees than the active ingredients.
Although these data suggest the neonicotinoids may contribute to hive collapse or other problems with bee colonies, in practice the data are less clear. A 2013 review of the hive collapse disorder problem by a committee of USDA and EPA scientists, on the other hand indicated a consensus regarding multiple factors underlying the disorder: viral infections, the parasitic mite Varroa destructor, the bacterial disease European foulbrood, nutrition, changes in gut microbial flora, and acute and cumulative effects of pesticides.
BIOLOGIC INSECTICIDES
Although the pyrethrins and pyrethrum extract could be termed biologic insecticides the term usually applies to compounds that interfere with mating called pheromones, to plant extracts other than pyrethrum, insect toxins produced by microorganisms such as Bacillus thuringiensis (Bt), and compounds that interfere with regulation of insect growth. In recent years the number of biologic insecticides has increased, perhaps because of the simplified data requirements promulgated by the EPA. The extensive chronic toxicity testing required for most pesticides is not required for the biologic materials.
Microbial Insecticides
Currently registered microbial insecticides include 12 Bt variants, Bacillus sphaericus, 2 strains of the fungus Beaveria bassiana, Chromobacterium substugae, spores of Nosema, locustate, Paecilomyces fumosoroseus, Metarhizium anisopliae strain F52, Apopka strain 97, codling moth granulosis virus, and polyhedral occlusion bodies (OBs) of the nuclear polyhedrosis virus of Helicoverpa zea (corn earworm).
CYD-X Biological Insecticide
CYD-X contains a naturally occurring host-specific granulovirus that infects codling moth larvae (Cydia pomonella) after the ingestion of occlusion bodies or granules, before or during initial entry into fruit. The virus degrades quickly by the action of ultraviolet light, a property that limits its residual effect after application. It does not infect beneficial insects, fish, wildlife, livestock, or humans.
Chromobacterium subtsugae strain PRAA4-1(T), a motile, gram-negative, violet-pigmented bacterium, is toxic to Colorado potato beetle larvae and other insects. It has specific action to control the black pecan aphid Melanocallis caryaefoliae and coddling moth on walnuts. The product label states it is toxic to bees present at the time of treatment or exposed to residues on weeds and blooming crops.
The fungal insect pathogen (entomopathogen) Paecilomyces fumosoroseus spores germinate on the body of the target pest (Diptera such as the Mediterranean fruit fly). It penetrates the cuticle and grows within the hemolymph and other tissues of the infected insects. Sporulation from dead pests leads to infections of other insects.
Bt Mechanism of Action
Bt insecticides produce delta-endotoxin, also called Crystal (Cry) and Cytolitic (Cyt) proteins with activity against several insect orders—Lepidoptera (moths and butterflies), Coleoptera (beetles), Diptera (flies)—and also against nematodes These proteins binds to epithelial cells in the insect midgut, creating cell membranes pores, leading to cell lysis. After cell lysis, bacterial spores germinate, leading to a lethal blood poisoning. B. sphaericus produces insecticidal toxins with a mode of action very similar to Bt.
Beauveria bassiana is a fungal pathogen of insects (entomopathogen). The fungal active ingredient kills the adult house fly pest by growing on the insect’s exoskeleton and secreting enzymes into the pest’s soft body parts to kill it.
Bt Infection
Because Bt is closely related to Bacillus cereus, the possibility of human infections, especially with immunocompromised host, has been raised. Case reports have documented prior episodes of infection following direct ocular contact with Bt. Although recommended antibiotic treatment does not necessarily change after bacterial speciation, it would be reasonable to request a delta-endotoxin assay when there is a known exposure to Bt and a possibly related illness. An alternative approach would be to consider B. cereus infections in agricultural workers handling Bt as potentially representing infections with Bt could be missed by standard lab evaluations.
Bt formulations have low capacity to cause skin irritation. Bt formulations can irritate the eyes or skin without causing infection.
Microbial By-Products
A. Spinosad
The gram-positive soil bacterium Saccharopolyspora spinosa (an Actinomycetes species) produces a macrolide compound called spinosad, a mixture of spinosyn A, spinosyn D, and smaller amounts of other compounds. It has a complex mode of action: similar to abamectin, it interacts with the insect nicotinic receptors, but also interacts with gamma-aminobutyric acid (GABA) receptors. The effect is relatively specific to insects and its mammalian toxicity is low by both the oral and dermal routes.
B. Avermectins
Abamectin is a mixture of two complex compounds, byproducts of a bacterium found in soil called Streptomyces avermitilis. The two avermectin compounds B1a and B1b differ very slightly from each other in chemical structure, an ethyl group in B1a versus a methyl group in B1b at the substituent labeled R in the complex molecule of a class described as a macrocyclic lactone. Its molecular weight is 873, 50% larger than the aminoglycoside antibiotic streptomycin produced by the species Streptomyces griseus. It consequently has a very low vapor pressure and very low solubility in both water and organic solvents. Avermectins act by stimulation of GABA receptors in the insect nervous system.
The oral LD50 demonstrates its potentially high mammalian toxicity once absorbed. It has a low dermal toxicity, probably related to slow dermal absorption. Cases of accidental direct exposure to abamectin typically involved eye or skin irritation. Ingestion of abamectin can produce more severe poisoning than typical occupational exposure cases.
Pheromone Products
Currently registered pheromone products are shown in Table 34–11. Pheromones control insects by a selective mechanism, interfering with mating, but not killing target pests. They have an excellent track record in agriculture, but have generated controversy when used in aerial spraying programs. A community interest group proposed to use pheromones as alternative to Bt for gypsy moth pest control in Oregon/Washington. However, in a California program to control the light brown apple moth, use of a pheromone product generated 473 complaints, including 321 (70%) describing respiratory symptoms. It was not possible to determine whether any of the complaints were related to the spraying, at a rate of approximately 1 ounce per acre. The crude rate of symptom reporting was 1.2 individuals/1000 residents in the treated area.
Table 34–11 Some currently registered pheromone products.

Insect Growth Regulators
Humans do not make or use the hormones insects in molting, egg production, or chitin synthesis. Therefore, insect growth regulators are considered to have little human toxicity.
A. Azadirachtin and Neem Oil
Azadirachtin is the principal insecticidal ingredient in oil from the neem tree, Azadirachta indica, native to India. The oil has reported medical uses including treatment of parasitic and helminthic infections. Azadirachtin interferes with synthesis of the insect developmental hormone prothoracicotropic hormone (PTTH), which stimulates the release of the insect molting hormone. Perhaps because of its selective mode of action, azadirachtin has low mammalian toxicity.
B. Inhibitors or Analogues of Juvenile Growth Hormone
Insect juvenile growth hormone refers to a complex of hormones that regulate development, reproduction, dormancy, and expression of genetic traits in between insect molts. It has become a common biochemical target for control of insects. Other compounds affect development affect by interrupting egg production.
Fenoxycarb and pyriproxyfen both act as juvenile hormonal agonists that produce an overload of hormonal activity. Both have low mammalian toxicity.
C. Inhibitors of Egg Production
Methoprene, hydroprene, and kinoprene interfere with the development of insect eggs by inhibiting the production of the egg yolk glycolipoprotein vitellogenin.
The acute mammalian toxicity of all the compounds is low. None cause prolonged reactions in the Draize eye and skin irritation assays.
D. Chitin Synthesis Inhibitors
The chitin synthesis inhibitors include the thiadiazine compound buprofezin, the benzoylphenyl ureas noviflumuron and novaluron, and the diacylhydrazine compound tebufenozide. After molting, chitin inhibitors cause gradual death by preventing growth of a new exoskeleton. The acute systemic toxicity of the chitin inhibitors is very low; none caused persistent effects in the Draize irritation test for either the eye or the skin.
Nonspecific Growth Regulators
Cyromazine and hexythiazox are both insect growth regulators but do not have completely defined mechanisms of action. Cyromazine belongs to the s-triazine class of chemicals, but lacks herbicidal activity. It is effective against fly larvae and leaf miners. The exact mode of action of the mite growth regulator hexythiazox is not well understood. Hexythiazox kills the eggs before the mites hatch and also some immature mites. Adult mites are not killed, although adults exposed to residues may lay eggs that are not viable.
A. Rotenone
Rotenone derives from the roots of Asian Derris plants, South American Lonchocarpus species, and other plants. It is acutely toxic after oral administration. Rotenone has low aqueous solubility, but high solubility in organic solvents (log P of 4.26). Pesticide registration data for rotenone do not include a dermal absorption study, but estimated dermal absorption is 9%, based upon a surrogate study using fluazifop-P-butyl (with a comparable molecular weight (383.4) and log P value [4.5]). This is consistent with its low recorded dermal toxicity. It is markedly toxic to fish, as demonstrated by its 48 hour LC50 in the rainbow trout of 28 ppb.
This property accounts for the reported use of Derris and other rotenone-containing plants as piscicides in traditional cultures. No illnesses were reported to the California illness registry associated with rotenone between 1982 and 2008. Although rotenone’s limited use does not make it a plausible explanation for many cases of Parkinson disease, it has definite effects on dopaminergic neurons in the brain in experimental studies in rodents, accompanied by decreased motor activity.
Miscellaneous Insecticides
A. Insecticidal Soaps
Insecticidal soaps can help control soft-bodied insects including aphids, leafhoppers, spider mites, and whiteflies. The soaps act by disrupting cell membranes, requiring direct contact at the time of application to be effective. Residuals of the soaps on plant foliage do not have insecticidal activity.
Acute toxicity data for insecticidal soaps demonstrate very low mammalian toxicity, but Draize testing shows a clear tendency to cause eye and skin irritation. There are a few cases of systemic illness reported, along with eye irritation following direct accidental contact with soap, with skin irritation, and infrequently with respiratory irritation.
B. Propargite
The acute systemic toxicity of propargite is low. Propargite causes severe irritation of the eyes and the skin. Cases of eye and skin irritation remained common in propargite applicators.
Animal testing data identify propargite as a potential carcinogen. In a rat oncogenicity study conducted for pesticide registration, technical propargite (87.2% purity) was fed to albino rats for 104 weeks at concentrations of 0, 80, 400, and 800 ppm. The no-effect-level for general chronic toxicity was 80 ppm (reduced bodyweights in males at 400 [slight] and for females at 800 ppm). Based upon the known irritant effects of propargite, the sarcomas may have been related to hyperplasia caused by acute epithelial irritation (a threshold effect) rather than genotoxic effects of propargite (a non-threshold or stochastic effect). There are no human epidemiologic studies implicating propargite as a carcinogen.
C. Insect Repellents
Frequently used insect repellents include biological oils and products containing diethyl-toluamide (DEET). Concentrated formulations of DEET (34.3–98.1%) provide 10–12 hours of protection but lower concentrations (7.15%.) may provide only 2 hours of protection. Adverse effects of DEET have been described extensively. Repeated use of high-dose formulations have been associated with irritant dermatitis. Allergic reactions have also been reported including contact urticaria and anaphylaxis.
Because DEET is easily absorbed through skin, concerns have been raised about possible systemic effects, especially neurologic effects on children. Encephalopathy in adults has also been reported. A 27-year-old previously healthy man applied 25% DEET to his arms, neck, and legs repeatedly during a fishing trip on a hot and humid afternoon. He initially reported paresthesias of the limbs and face, then auditory hallucinations, progressive confusion, disorientation, and agitation He did not return to normal mental function until the third hospital day.
D. Alternative Insect Repellents
Natural products that have some efficacy as insect repellents include pyrethrum and oils extracted from neem, garlic, aniseed, thyme, geranium, bergamot, eucalyptus, lavender, bergamot, pyrethrum, lavender oil, coconut oil, birchwood tar, soybean, nutmeg, pine, orange blossom, clove, cinnamon, pennyroyal, and peppermint.
Linalool, is a terpene alcohol found in mints, laurels, citrus, and birch. The 17 products currently registered include carpet sprays and two products described as repellent “lanterns”, but none that are directly applied to human skin. It has low mammalian toxicity, but has been reported as a human eye irritant in concentrations greater than 320 ppm.
In a series of dermatitis patients, contact allergy to oxidized linalool was present in 6.9%. A 2011 report described a prevalence of 2.3%. A cluster of six illnesses (designated as priority episode 61-LA-93) was reported in a California airlines office treated during nonwork hours with a product containing linalool.
Another alternative product, Eucalyptus oil, has low systemic toxicity, but eye, skin, and respiratory irritation are listed as adverse effects.
STRUCTURAL PEST CONTROL
Borates
Active ingredients containing boron include boric acid, also known as orthoboric acid, sodium tetraborate (pentahydrate), barium metaborate, perboric acid sodium salt, and other compounds. The discussion below is limited to boric acid. There are currently 59 household insect control formulations registered in addition to a 100% boric acid dust intended for manufacturing use. There are 10 additional registered formulations containing sodium tetraborate (pentahydrate) and 3 fungicidal formulations containing barium metaborate.
![]() Mode of Action & Acute Toxicity
Mode of Action & Acute Toxicity
Boric acid functions by disrupting water balance in insects, but the detailed mechanism has not been identified. It has low acute toxicity in mammals and minimal irritant effects. Acute accidental ingestion of boric acid causes no symptoms in more than 75% of cases. However, fatal adult and pediatric poisonings have been reported. Borate urine or blood concentrations are useful primarily for documentation of exposure. Unusual conditions associated with chronic exposure include alopecia associated with both ingestion and topical exposure. Chronic pediatric boron poisoning has also been reported following repeated use of a boric acid formulation for diaper rash.
In most cases only supportive treatment is required. Advanced measures in chronic ingestion or high dose acute ingestions have included dialysis, benzodiazepines for seizures and forced diuresis.
Sulfluramid & Fipronil
Sulfluramid is classified as a halogenated alkyl sulphonamide; it has potent metabolites that inhibits energy production by uncoupling oxidative phosphorylation. It does not produce the same effect in mammals because of differences in metabolism. It has moderate oral toxicity but low dermal toxicity in mammals.
Fipronil is a member of the phenylpyrazole class of insecticides. Fipronil is an antagonist that binds to the GABA channel, producing overstimulation of the nervous system because of the absence the GABA associated inhibitory activity. It has moderate mammalian oral toxicity, but its dermal absorption is very slow.
There are 75 currently registered fipronil products, including multiple products for control of ectoparasites on pets. Fipronil has been used extensively in Europe, Asia and elsewhere as an agricultural insecticide, with applications as a seed treatment for corn, cotton, wheat, and other crops.
![]() Ecological Effects of Fipronil
Ecological Effects of Fipronil
Fipronil has high toxicity for termites, bees, ants, and other colony insects, typified by the very low LD50 for bees (LD50 = 0.004 μg/bee). This has led to concerns about fipronil’s use as an agricultural insecticide. Regulatory action to date has included a temporary ban in France after hive losses in the southwest region of the country in 2003, and a limited ban in China in 2009. In Madagascar, two species of lizard and an endemic mammal declined because termites form part of their food chain.
Recent acute illness cases reported in the United States mostly occurred in residential settings (ant or roach baits or treatments for pets). One occupational case suffered a brief seizure, blurred vision, and dizziness after doing an application with minimal protective equipment. Another applicator required 7 days of hospitalization for dyspnea, diaphoresis, tremor, paresthesia, and slurred speech while applying the 80% termite control formulation of fipronil.
Novel Agents in Agricultural Pest Control
A. Insect Ryanodine Receptor Compounds
Ryanodine receptor insecticides stimulate a calcium channel in the sarcoplasmic reticulum of muscle cells and a similar channel in the endoplasmic reticulum of other cells. The model compound is ryanodine, an insecticidal compound found in the South American plant Ryania speciosa. At low ryanodine concentrations the calcium channel is partially open, stimulating muscle action, but the channel is closed at high concentrations.
The ryanodine receptor is present in mammals, but the insecticidal ryanodine compounds are highly selective for insect ryanodine receptors. This is reflected in the low systemic toxicity of the two synthetic ryanodine compounds.
B. Tetronic Acid Derivatives
New tetronic acid derivatives function by inhibiting insect lipid synthesis and the development of the respiratory system. They are effective against mites and a variety of insect pests. The mammalian toxicity of all four compounds is low and they do not cause prolonged eye or skin irritation in Draize test.
C. Physical Agents
Because of concerns about chemical agents, some structural pest control companies have promoted the use of physical agents.
A system using liquid nitrogen to freeze termites in wall spaces raosed safety concerns about traditional termite control methods. In terms of efficacy, the method appeared to be comparable to a chemical spot treatment, but was not the equivalent of tenting a structure and applying a traditional fumigant. An applicator died applying liquid nitrogen in a narrow crawl space adjacent to a bath tub. Post-mortem evaluation was negative for drugs of abuse, structural heart disease, and the final diagnosis was simple asphyxia.
FUMIGANTS & NEMATOCIDES
ESSENTIALS OF DIAGNOSIS
![]() Acute effects
Acute effects
• Respiratory tract irritation—burning eyes, nose, throat, cough, shortness of breath, and pulmonary edema.
• CNS depression—headache, nausea, vomiting, dizziness, drowsiness, fatigue, slurred speech, loss of balance, disorientation, loss of consciousness, and respiratory depression.
• Encephalopathy (methyl bromide) —tremors, seizures, elevated serum bromide level, late personality changes, and cognitive dysfunction.
![]() Chronic effects
Chronic effects
• Liver damage (halogenated hydrocarbons)—anorexia, abdominal pain, jaundice, and abnormal liver function tests.
• Peripheral neuropathy (methyl bromide)—progressive distal symmetric sensorimotor neuropathy, ascending paresthesias, numbness, and weakness.
• Asthma and reactive airways secondary to metamsodium by-products and to chloropicrin.
• Immune effects of metam sodium and methyl isothiocyanate (MITC) in animal studies.
![]() General Considerations
General Considerations
The fumigants have in common innately high vapor pressures or by-products with high vapor pressure. They also have a high degree of chemical and biological reactivity, potentially leading to many secondary effects (Table 34–12). Recorded effects include respiratory and eye irritation, CNS injury, hepatic and renal injury, as well as carcinogenicity and reproductive effects in animal studies. Use of insecticides has gradually shifted to a diverse group of low toxicity compounds. The reactivity and toxicity of prospective fumigants, by contrast, tends to present barriers to registration and widespread use.
Table 34–12. Selected fumigants and nematicides.

Applications for fumigants include structural pest control, treatment of stored commodities, and control of soil pests. Structural and commodity fumigations usually target insects, but soil fumigation also assists in control of nematodes, weed and some soil fungi.
Equipment used for soil fumigation includes shank injection equipment, broadcast sprinklers on risers, and drip irrigation lines. Structures such as houses, warehouses, grain elevators, and greenhouses may be sealed, fumigated, and then aerated before being reoccupied. Tarps made of impermeable material are also used to cover fields treated halogenated hydrocarbon fumigants (dichloropropene and methyl bromide). To limit off-gassing of highly water soluble by-products of metam sodium and other fumigants that release MITC, postapplication water treatments are employed. Phosphide compounds are applied in solid formulations, as pellets of aluminum, magnesium or zinc phosphide, which liberate phosphine gas when in contact with water in the environment or after ingestion by pests such as rodents. The mixture of phosphine gas and carbon dioxide is used for commodity fumigation and pumped under pressure from tanks into commodity containers or warehouse storage spaces.
![]() Industrial Hygiene Measures
Industrial Hygiene Measures
Unlike most other categories of pesticides, fumigants can usually be measured by air sampling.
Colorimetric tubes are available for measuring air concentrations of methyl bromide and phosphine. Phosphine can also be detected near the 0.3 ppm TLV with flame ionization equipment. Other fumigants can be measured with air sampling pumps and charcoal or other appropriate media. This does not allow the immediate feedback often necessary for short-term control of excess exposure.
![]() Occupational & Environmental Exposure
Occupational & Environmental Exposure
Workers applying fumigants may be exposed when leaks occur in equipment, when buildings are not sealed adequately, and when checking for leaks and entering chambers or buildings before complete aeration without appropriate protective equipment. Exposure of applicators, field workers, and bystanders to soil fumigant can occur downwind from application sites. Off-gassing of fumigated commodities has the potential to affect warehouse workers not directly involved in fumigation operations.
Newly Registered Compounds & Methyl Bromide Alternatives
Since 2000 newly registered fumigants have included a phosphine/carbon dioxide (CO2) gas mixture, intended as a safer variation of the phosphide salts that release phosphine gas. Safety issues with the phosphide fumigants are discussed below, but do not apply to the gas formulation because of the CO2 content. Although the phosphine gas/CO2 formulation is gradually increasing in use, its application is relatively complex compared to the use of aluminum phosphide tablets.
Methyl bromide alternatives that have not been registered include included propargyl bromide. Its use is limited by both toxicity and safety concerns because of its tendency to decompose with mild shock. Dimethyl disulfide has also been evaluated, as well as a mixture of allyl isothiocyanate and furfural. Allyl isothiocyanate is currently only registered as a animal repellant. Dimethadione is structurally similar to furfural, a natural compound that targets nematode cuticles. The relatively selective mechanism of action thus distinguishes from the broad biocidal activity of most other fumigants.
![]() Mechanism of Action & Clinical Findings
Mechanism of Action & Clinical Findings
A. Halogenated Hydrocarbons
1. Methyl bromide, methyl iodide, 1,3-dichloropropene, and chloropicrin—Halogenated fumigants, including chloropicrin, are electrophilic chemicals, reacting with nucleophilic amino acids (glutamine, asparagines, cysteine, and serine) at or near the active sites of enzymes in target pathogens or target pests.
Most halogenated fumigants and nematicides are well absorbed by all routes of exposure and are excreted rapidly without significant bioaccumulation. Both methyl bromide and 1,3-dichloropropene fit this pattern. Inhalation of vapors is the most common route of exposure, although dermal absorption of vapors or liquid also can occur. The vapors and liquids usually are primary irritants and in some cases are quite potent. At sufficient concentrations, acute CNS depression may occur. The halogenated hydrocarbon fumigants share many of the effects of the halogenated hydrocarbon solvents, including cardiac sensitization, direct cellular toxicity to the liver and kidneys, and carcinogenicity in laboratory animals.
2. Methyl bromide—Halogenated hydrocarbon fumigant poisoning may occur gradually over hours of inhalation exposure, even at levels inside of tarped buildings. For individuals impaired by alcohol, drugs, or mental illness, the eye and respiratory irritation provoked by chloropicrin (present as a warning agent) may not be sufficient to make them leave a treated building. Symptoms may be nonspecific and difficult for victims to recognize as poisoning, including headache, nausea, vomiting and dizziness, drowsiness, fatigue, slurred speech, loss of balance, and disorientation. Tremors, myoclonus, and generalized seizures may occur. If such cases survive until hospitalization, death may occur after several days due to simultaneous liver and renal failure, possibly complicated by adult respiratory distress syndrome.
Acute and chronic poisoning from methyl bromide may be followed by prolonged and, in some cases, permanent organic brain damage marked by personality changes and cognitive dysfunction. Workers have been diagnosed as suffering from severe psychological disorders until a source of methyl bromide exposure was recognized.
Direct contact with liquid halogenated hydrocarbons may result in erythema and blisters. Damage to the skin can be severe if liquid is spilled on clothing and shoes, which retard evaporation. Many current agricultural formulations contain 20–50% chloropicrin, adding to the potential problems with irritation.
Topical exposures frequently lead to skin burns, eye irritation, or respiratory irritation, but may also show systemic effects. Since serious systemic poisoning sometimes occurs following dermal exposure, these cases deserve careful evaluation.
Serious systemic poisonings and fatalities commonly occurred after exposure in confined spaces, usually associated with structural pest control applications. Many occurred after deliberate violations of tented structures, but failure to properly clear a treated apartment building caused two fatalities in a 1991 case in California. Partial impairment by alcohol may have contributed to the death, because other tenants left the building after experiencing symptoms of acute irritation.
Two noteworthy occupational poisonings occurred in the cold storage industry in 2010 over a period of several weeks. Two produce inspectors unknowingly inhaled methyl bromide off-gassing from produce treated on entry to the United States after shipment from Chile. Both had transient ataxia, cognitive impairment, and elevated levels of serum bromide. A subsequent industrial hygiene survey of the cold storage industry showed elevated methyl bromide air concentrations in enclosed locations in other facilities. These ranged as high as 20 ppm, 20 times the California 1-ppm 8-hour exposure standard. Numerous mitigation measures implemented since the poisonings were identified have markedly lowered exposures and no additional cases have been reported.
A. CHRONIC EXPOSURES AND SEQUELAE OF ACUTE POISONING—Chronic exposure to methyl bromide can result in progressive peripheral neuropathy with ascending paresthesias, numbness, and weakness and with or without depressed deep tendon reflexes.
Acute and chronic poisoning from methyl bromide may be followed by prolonged and, in some cases, permanent neurologic damage, including personality changes and cognitive dysfunction. Workers have been diagnosed as suffering from severe psychological disorders until the methyl bromide exposure was recognized In some cases of acute or chronic poisoning, neurologic deficits associated with exposure prove reversible, in others not. In the 2010 cases affecting the cold storage produce inspectors, neither had any overt permanent impairment. Acute massive exposures associated with prolonged entry into a treated space have historically had poor outcomes.
Because methyl bromide is a strong alkylating agent it seems likely to be a carcinogen. However, the only positive animal bioassay is a gavage feeding study where the initially reported tumors were reinterpreted as hyperplasia caused by mucosal irritation.
A 2012 report described an epidemiologic study of cancer risk in agricultural workers previously handling methyl bromide. This showed a dose-related increase in stomach cancer, based upon a total of 15 exposed cases. This could have been a chance finding since methyl bromide exposure was correlated with some demographic and behavioral risk factors, such as smoking. Data were not available on other significant risk factors for stomach cancer, including infection with Helicobacter pylori.
A rabbit teratogenicity study evaluated the effect of methyl bromide (0, 20, 40, and 80 ppm) administered by inhalation 6 hours daily between day 7 and day 19 of gestation. The 80-ppm group showed some evidence of maternal neurotoxicity and decreased body weight gain, as well as abdominal wall defects (omphalocele), agenesis of the gallbladder, abnormal location of the subclavian artery behind the esophagus, and fused sternal bone segments in the offspring. Because of the large safety factor customarily required to minimize potential exposure to teratogenic agents, this study proved to be a critical endpoint in the California risk assessment process. Although the current California occupational 8-hour exposure limit for methyl bromide is 1 ppm, the current California reference level for environmental exposures and setting permit conditions is 210 ppb over 24 hours.
3. Dichloropropene—The level of dichloropropene use has decreased drastically in recent years because of regulatory concerns related to its carcinogenicity. A cancer bioassay evaluated the response of mice to lifetime inhalation exposure of 1,3-dichloropropene. There was a positive trend of increase with dose in pulmonary adenomas in male mice. Adenomas but also malignant alveolar/bronchiolar carcinomas were observed in male mice exposed to Telone II by oral gavage. The treatments resulted in statistically significant increase in forestomach tumors and liver tumors at multiple doses.
4. Methyl iodide—The spectrum of pests methyl iodide controls comes close to that of methyl bromide, including nematodes, fungi, and weeds.
The acute effects of methyl iodide are similar to those produced by methyl bromide, but occur at somewhat lower doses (see Table 34–12). In addition to neurotoxicity, skin burns can be significant problems. Experience with methyl iodide is limited to case reports, each involving neurologic symptoms very similar to those produced by methyl bromide.
A recent report described poisoning in an American facility after breakdown in his protective equipment barrier caused unsuspected dermal exposure and contact burns. He subsequently required extensive full thickness grafting during a 17 day hospitalization. He had a urinary infection with Staphylococcus aureus, complicated by an episode of acute delirium. Neurobehavioral symptoms developed after discharge from the hospital and healing of his skin grafts. The patient reported difficulty with memory and concentration and his spouse reported altered behavior and personality. Neurobehavioral testing showed below median scores in overall intellectual functioning and verbal skills, but no baseline reference was available. Abnormal results were noted for measures of attention, memory, information processing, and performance of simultaneous tasks.
An English chemical worker suffered a burn on the extensor surface of his right wrist while handling methyl iodide a week before presenting to the hospital with symptoms that resembled a right sided stroke. His brain imaging did not show evidence of either a hemorrhagic or ischemic stroke and his laboratory evaluation was normal. While in the hospital he had visual and auditory hallucinations. Abnormal neurologic examination findings included bilateral nystagmus, slurred speech, an ataxic gait, and past pointing on the right side. His neurologic function had almost returned to normal at follow-up 6 months after his hospitalization.
A. POSSIBLE LONG-TERM EFFECTS IDENTIFIED IN ANIMAL OR EPIDEMIOLOGIC STUDY—Prominent effects observed in chronic animal studies of methyl iodide include thyroid hyperplasia, increased TSH levels, and degeneration of the nasal olfactory epithelium. Injection of methyl iodide subcutaneously produces sarcomas near the injection sites and intraperitoneal injection produces lung tumors. Thyroid tumors were reported in multiple animal studies.
5. Chloropicrin—Chloropicrin best serves to control fungi and has less activity against nematodes and weeds than other soil fumigants. Much of it was applied as mixtures with halogenated hydrocarbons (dichlorpropene, chloropicrin, methyl bromide, and methyl iodide), but some applications were made using formulations containing only chloropicrin. It cannot be mixed with nucleophilic compounds such as metam sodium.
A. TOXICITY—Chloropicrin has high acute toxicity relative to other fumigants on inhalation, and both oral and topical (dermal) administration. Nevertheless, its most noteworthy effect is eye irritation, noticeable at levels below the threshold of respiratory irritation and systemic toxicity. In a subchronic inhalation study with rats, rhinitis was the most sensitive endpoint. Lung tumors and mammary fibroadenomas were identified as possible treatment-related effects in rodent cancer bioassays/chronic effects studies.
6. Sulfuryl fluoride—Sulfuryl fluoride has been the main replacement for methyl bromide in structural pest control. It’s acute toxicity is lower (Table 34–12). In animal studies with sulfuryl fluoride, the interval between initial exposure and death depends on the concentration of sulfuryl fluoride present, as indicated by the series of acute inhalation toxicity (LC50) studies and time-to-incapacitation studies. For sulfuryl fluoride concentrations similar to those present during typical fumigations (800–4000 ppm), animal studies demonstrate a poisoning syndrome with an exposure-fatality interval of several hours. At concentrations of 20–40,000 ppm, incapacity and death occurs in a matter of time.
A. SIGNS AND SYMPTOMS—Ocular, dermal, and respiratory irritant symptoms produced by sulfuryl fluoride overlap with those produced by chloropicrin and may accompany accidental exposures. Systemic symptoms may include acute delirium and seizures. Most human poisoning occurs after violation of tarped structures in a fashion similar to methyl bromide.
B. LABORATORY MEASUREMENTS—Exposure to sulfuryl fluoride can be evaluated by measuring fluoride levels in serum. Reference values range from 0.01 to 0.2 μg fluoride per milliliter of serum.
C. CHRONIC EFFECTS—Cases of residual neurotoxicity similar to those caused by methyl bromide usually do not occur following exposure to sulfuryl fluoride. In a NIOSH study of sulfuryl fluoride applicators, repeated exposure in the year preceding examination was associated with subclinical effects on memory and also on the sense of smell.
Chronic inhalation studies in multiple animal species did not show oncogenic effects. However, dental fluorosis, respiratory tract injury, and brain vacuoles were seen in multiple species.
MITC-RELEASING FUMIGANTS
The soil fumigants metam sodium, metam potassium, and dazomet, all function as profumigants, releasing the active fumigant MITC after reacting with air or water. Dazomet is used in small quantities for landscape maintenance and for fumigating soil mixtures used to grow ornamental plants
The principal degradation product (or by-product) of metam (either the sodium or potassium salt) is MITC, but can include a complex mixture of irritant compounds (methylamine, carbon disulfide, hydrogen sulfide, and low levels of methyl isocyanate [MIC]), depending upon the soil pH and other environmental conditions.
Metam Sodium
The degradation products of metam sodium include a complex mixture of irritant compounds: the primary pesticidal agent MITC, MIC (approximately 4% of the level of MITC), carbon disulfide, hydrogen sulfide, and methyl amine. The toxicologic effects of the mixture have not been characterized.
![]() Signs & Symptoms
Signs & Symptoms
Exposure to airborne MITC for 1 hour produces burning eyes and other irritant symptoms at concentrations of 800 ppb. Exposure for 4 minutes may produce similar symptoms at concentrations of 1900 ppb. Those with asthma or smoking-related pulmonary disease may experience respiratory problems at concentrations that only produce eye symptoms in others. In community exposures, nonspecific systemic symptoms such as nausea, headache, and diarrhea accompany the irritant symptoms in a portion of those exposed. The symptoms produced by hydrogen sulfide, carbon disulfide, and methyl amine have a high degree of overlap with those produced by MIC and MITC, but quantitative experimental data on dose-response for these compounds do not exist.
Irritant dermatitis is associated initially with direct skin contact in metam sodium applicators. However, dermatitis has not been a frequent problem in community exposure to airborne metam-sodium by-products
![]() Laboratory Findings
Laboratory Findings
No specific clinical tool exists for monitoring exposure to MITC metam-sodium by-products. Measurement of environmental levels of MITC in excess of the 0.8-ppm irritation threshold clearly would suggest the cause of concurrent ocular or upper respiratory symptoms. However, permit conditions and the metam-sodium applicators trade association encourage the use of odor monitoring in the place of industrial hygiene techniques. Odor monitoring is a poor means of detecting MITC because the average odor threshold (1700 ppb) is approximately twice the 800-ppb 1-hour ocular irritation threshold.
Pulmonary function studies and methacholine challenge tests may prove of value in cases of residual airway reactivity.
![]() Chronic Effects
Chronic Effects
Metam sodium is a carcinogen in animal bioassays, producing mouse liver tumors and angiosarcomas in rats. Metam exposure has moderate systemic toxicity on ingestion, but significant irritant properties. Occupational handler cases continue to occur sporadically.
Numerous environmental illness episodes have occurred following applications near rural communities, correlated with concentrations of MITC at or above its 800 ppb lowest-observed effect level (LOEL) for eye irritation in human volunteers. In illness episodes associated with metam applications both ocular and respiratory effects typically occur at the same time.
Dazomet
Use of dazomet is much lower than use of metam sodium and it causes far fewer episodes of illness. However, the comparatively small volumes of dazomet incorporated in soil can pose a risk to gardeners and landscapers.
Phosphide Fumigants
Phosphide fumigants generate phosphine (PH3) gas on contact with either moisture in the environment or acid in the intestinal tract. It is relatively more toxic than other fumigants, but is generally applied in far smaller quantities. Aluminum phosphide is the model compound, but there is gradually increasing use of a new product containing phosphine and carbon dioxide that does not present a risk of uncontrolled oxidation.
Most fatal cases of poisoning occur in cases of aluminum phosphide ingestion. These occur most frequently in countries that do not restrict the sale to professional applicators. Fumigation of rodent burrows near residential structures can present serious potential hazards, illustrated by the tragic death of two children in Utah in 2010. The adults exposed in the same residence had short term illnesses only.
![]() Signs & Symptoms
Signs & Symptoms
Cases of acute aluminum phosphide ingestion result in death from pulmonary edema, seizures, and respiratory depression. Nonfatal cases have been marked by liver injury with abdominal pain, nausea, vomiting, jaundice, elevated hepatic enzymes, and coagulopathy with bleeding. Occupational cases infrequently cause fatal outcomes but may result in symptoms severe enough to require hospitalization. Severe poisoning and fatal outcomes have occurred during fumigation in transit by either ship or rail.
![]() Laboratory Findings
Laboratory Findings
Following inhalation exposure to phosphine, no specific tests are available in most laboratories to aid in diagnosis. However, in cases of phosphide ingestion, aluminum may be elevated in serum and other tissues. Phosphine can be measured in expired air in either inhalation or ingestion. It may prove possible to use colorimetric tubes or direct-reading industrial hygiene instruments.
![]() Chronic Effects
Chronic Effects
An investigation of excess lymphoid cancers in members of a midwestern grain millers union demonstrated G-banded chromosomal translocations in workers handling phosphine and corresponding lesions in lymphocyte cultures exposed to the gas. No specific measures of exposure were reported. A follow-up study of applicators did not show any indication of genotoxicity. Passive-diffusion badges and colorimetric tubes were used to measure exposures; none exceeded 2.4 ppm.
![]() Differential Diagnosis
Differential Diagnosis
CNS effects from halogenated hydrocarbons must be distinguished from exposure to other CNS depressants, including drugs and alcohol, which are sometimes simultaneous exposures.
The acute irritation produced by chloropicrin, formaldehyde, acrolein, and sulfur dioxide will be marked by the presence of their distinctive odors. Phosphine has a garlic-like odor that often can be detected on a victim’s breath, especially in cases of ingestion. The encephalopathy and peripheral neuropathy from methyl bromide are similar to those from other organic causes of central or peripheral disease, such as alcohol, drugs, and other neurotoxins. Toxicity can occur from exposure to levels without a detectable odor, making diagnosis difficult without a history of exposure.
![]() Treatment
Treatment
Treatment of all fumigant poisonings except cyanide is symptomatic: Respiratory support and anticonvulsants should be provided as indicated. Dimercaprol (British anti-Lewisite [BAL]) has been used in early methyl bromide poisoning but without evidence of benefit; given its toxicity, it cannot be recommended. Treatment to increase the excretion of inorganic bromide has no rational basis.
Monitoring calcium levels and replacing depleted calcium may be of benefit in acute care of sulfuryl fluoride inhalation.
![]() Prognosis
Prognosis
Toxicity from the irritants chloropicrin, formaldehyde, acrolein, and sulfur dioxide is limited to their acute reversible effects. On the other hand, deaths have been reported from the use of most of the other fumigant and nematocides. Recovery from nonfatal poisoning usually is complete, except for methyl bromide, which has caused permanent organic brain damage and a prolonged, if not permanent, peripheral neuropathy. Acute liver necrosis followed by cirrhosis and liver cancer has been reported from industrial solvent use of carbon tetrachloride, but not from use as an agriculture fumigant.
RODENTICIDES
Rats are the most prevalent pest in many developing countries and consume up to 20% of stored grain. They represent a significant threat to the food supply in developed countries as well. Other rodents and small mammals such as squirrels, gophers, and rabbits compete for food and act as reservoirs for diseases that affect humans and are considered pests for that reason.
Poisoning with rodenticides is the most widely used method of control of small mammals. To be effective, rodenticides must be attractive to a rat as food, which is difficult because they are fastidious eaters. Rodenticides also must be delayed in action if used as bait because rats will avoid returning to feed where another rat has died after eating. Unfortunately, what is attractive, edible, and ultimately lethal to a rat is also appealing to pets and other animals and small children. Because application of baits results in negligible exposure to applicators, the primary human health hazard from most rodenticides is childhood poisoning from ingestion, although serious poisoning from single ingestions of warfarin is rare.
Table 34–13 lists the rodenticides in current use, along with compounds that were used historically. Data on pounds sold indicate that the most heavily used compound currently is zinc phosphide. The anticoagulants include short- and intermediate-acting compounds, represented by warfarin and diphacinone, and the long-acting compounds (“superwarfarins”), represented by 4-hydroxycoumarin, brodifacoum, bromodialone, difenacoum; and the inadione derivative chlorophacinone.
Table 34–13. Rodenticides currently used in the United States.

![]() Occupational & Environmental Exposure
Occupational & Environmental Exposure
There are no reports of harmful exposure from the manufacture, formulation, or application of dry anticoagulant rodenticides. There is one report of bleeding in a farmer following extensive and prolonged skin contact with a liquid warfarin solution. Childhood ingestion of these compounds is common, although bleeding as a result is uncommon unless the compounds are ingested repeatedly. The long-acting anticoagulants require fewer doses to cause bleeding.
![]() Mechanism of Action & Clinical Findings
Mechanism of Action & Clinical Findings
Warfarin is well absorbed from the gastrointestinal tract. Diphacinone is absorbed following ingestion, but there is no information regarding its dermal absorption. All the anticoagulants act through inhibition of hepatic synthesis of prothrombin (factor II) and factors VII, IX, and X. In humans and rats, the half-lives of these factors are longer than the half-life of the anticoagulants, so repeated doses are necessary before significant depression and bleeding occur. Resistance to warfarin in humans and rats appears to be genetic and may be a result of rapid metabolism.
The anticoagulants also produce capillary damage through an uncertain mechanism, although this, too, is reversed by administration of vitamin K. Skin necrosis and dermatitis have been reported as rare. Complications of therapeutic use of warfarin, but have not been reported as a result of exposure to rodenticides. The indanediones cause neurologic and cardiovascular toxicity in some animal species, but these effects have not been reported in humans.
A. Symptoms and Signs
Most cases of accidental ingestion do not result in evidence of toxicity even without treatment because doses usually are single and relatively small. Repeated doses could, be followed by bleeding, primarily from the mucous membranes such as the gums and nasal passages and into the skin, joints, and gastrointestinal tract. Abdominal, flank, back, and joint pain reflect bleeding into those areas.
B. Laboratory Findings
Prolonged prothrombin time may appear 24–48 hours after ingestion of an anticoagulant and is often the only evidence of toxicity following a single exposure. Coagulation time will be increased in cases of significant poisoning, but bleeding time may be normal. Specific factors other than prothrombin may be depressed. Warfarin can be measured in plasma and its metabolites in urine, but these measurements have little utility.
![]() Differential Diagnosis
Differential Diagnosis
Most cases of rodenticide ingestion are observed or reported episodes and do not result in significant toxicity. Failed suicidal or homicidal use may result in otherwise unexplained bleeding and depressed prothrombin time.
![]() Treatment
Treatment
Treatment of single acute ingestions is usually unnecessary, but patients should be observed (at home) for 4–5 days following ingestion. Vitamin K may be administered orally in a dosage of 15–25 mg for adults and 5–10 mg for children with a history of ingestion, or intramuscularly in a dosage of 5–10 mg for adults and 1–5 mg (up to 0.6 mg/kg) for children with prolonged prothrombin time or bleeding. Following treatment, prothrombin times should be determined every 6–12 hours and used as the basis for further treatment. If bleeding is severe, slow intravenous infusion may be considered, but carries a risk of adverse reactions including flushing, dizziness, hypotension, dyspnea, cyanosis, and death. Transfusion and iron to replace blood loss should be considered.
![]() Prognosis
Prognosis
Treatment is usually effective within 3–6 hours. The prognosis is determined by the extent and location of bleeding and is usually good.
FUNGICIDES (PHTHALIMIDES, DITHIOCARBAMATES, SUBSTITUTED AROMATICS, TRIAZOLES, COPPER FUNGICIDES, STROBILURIN & MISCELLANEOUS COMPOUNDS)
Pesticides classified as fungicides overlap with compounds classed as algaecides and antibacterial agents. Most fungicides are distinct from other pesticides owing to the unique plant characteristics of fungi and the fact that the chemical must kill or inhibit the fungi without adversely affecting the host plant. Quantitative structure-activity relationship (QSAR) models of fungicide activity highlight the importance of hydrophobicity (measured by log P) and three-dimensional steric configuration, important in inhibiting fungal enzymes such as 14α-demethylase, involved in sterol synthesis.
Approximately 150 fungicides are available, mostly synthetic organic chemicals of relatively recent development. This discussion is confined to the most commonly used classes of compounds, the phthalimido compounds, dithiocarbamates, substituted aromatics, and miscellaneous compounds with important adverse effects (Table 34–14).
Table 34–14. Fungicides currently used in the United States.

Most crops are susceptible to fungi and related diseases. Frequent application of fungicides often is necessary owing to the rapid replication of many fungi. With the exception of a few systemics, fungicides are only active where they have been left as a residue on a plant, making uniform application necessary. They are applied as sprays or dusts so that a film of residue is left on the plants. Many seeds are treated with fungicides. A limited number of general-purpose lawn and garden fungicides are available for home use.
![]() Occupational & Environmental Exposure
Occupational & Environmental Exposure
Field workers and employees in greenhouses and nurseries are more apt to be exposed to fungicides than to other pesticides on a routine basis because fungicides are only effective as long as a residue is present on plant surfaces, and application is often necessary at the same time plants must be handled by these workers. Seed-treatment facilities are an important site of exposure to fungicide.
Homeowners are exposed to lawn and garden treatments. Most fruits and vegetables have allowable residues (tolerances) for one or more fungicides. Water contamination has not been a significant problem to date.
Safety issues arise for few fungicides. Nevertheless, products are formulated with elemental sulfur because of its flammability. Hot-weather aerial applications present a particular risk, and several fires and crop-duster accidents associated with burning sulfur have occurred.
![]() Clinical Findings
Clinical Findings
A. Thiocarbamate Compounds
Disulfiram (Antabuse), used to treat alcoholism because of its ability to produce an adverse reaction in the presence of alcohol, is a dithiocarbamate and shares a number of properties with other compounds in the class. Several dithiocarbamates, including the ethylene-bis-dithiocarbamates (EBDCs), are used as accelerators in the vulcanization of rubber. These compounds are not acutely toxic; the primary recognized health effect is dermatitis, both irritant and allergic. An alcohol-dithiocarbamate reaction is marked by headache, nausea, vomiting, flushing, dizziness, confusion, and disorientation.
1. Chronic effects—Thiocarbamate compounds also contain varying levels of the contaminant ethylenethiourea (ETU), a material classified as a probable human carcinogen. ETU has affinity for the thyroid similar to the antithyroid drug propylthiouracil. Mancozeb and zineb both cause thyroid adenocarcinoma in animal tests. The IARC concluded that the mechanism of ETU-related thyroid cancer was not genetic and unlikely to cause cancer following low-dose exposure to humans. The EPA nevertheless classified the compound as a probable human carcinogen.
Sprayers in Mexico heavily exposed to maneb and mancozeb showed increased levels of thyroid-stimulating hormone (TSH) compared with controls but normal levels of thyroxine (T4). Sprayers also showed increased levels of sister chromatid exchange and chromosome translocations. In reproductive studies, ETU produces neural tube and brain malformations in rats. Other test species do not appear susceptible to this adverse effect.
B. Phthalimido Compounds
Captan and captafol have low systemic toxicity but can cause immediate and delayed allergic reactions. Skin irritation also may occur given sufficient exposure. In QSAR terms, the phthalimido compounds act as reactive electrophiles. This property also accounts for the carcinogenicity of the compounds in rodent bioassays. Both captan and folpet cause intestinal tumors in rodent species. Captafol causes lymphosarcoma in feeding studies with mice. The EPA ranking system designates all three compounds as probable human carcinogens.
Structural analogies with the drug thalidomide have raised concerns that the phthalimido compounds could cause teratogenicity in agricultural workers. Studies of teratogenic effects of captan show positive effects in chick embryos but negative in Syrian hamsters, rabbits, and non-human primates.
C. Substituted Aromatics
The substituted aromatics include several compounds with a marked tendency to cause immediate and delayed allergy, including chlorothalonil and pentachloronitrobenzene (PCNB).
Hexachlorobenzene (HCB) was used formerly as an antifungal seed treatment, but several episodes of accidental food contamination have curtailed its use. HCB caused a widespread outbreak of acquired porphyria and adverse reproductive outcomes. Serum levels of HCB showed strong correlation with risk for spontaneous abortion.
The EPA rankings designate both chlorothalonil and HCB as probable human carcinogens. Pentachlorophenol has complex dermal and systemic toxicity. Although no longer in use, it remains of concern because of residual contamination at some hazardous waste sites.
D. Triazole Compounds
This group structurally resembles ketoconazole and other antifungal compounds used in human medicine. Triadimefon, the prototypical compound in the group, has low to moderate toxicity but a maximum application rate of only 8 oz/acre. As with most of the compounds in the group, triadimefon is a possible human carcinogen. Feeding studies show that it causes liver adenomas in mice. Terrazole is the only compound in the group classified as a probable human carcinogen. It also causes testicular atrophy in the rat.
E. Miscellaneous Compounds
1. Benomyl—Benomyl and its breakdown product, carbendazim (MBC), also cause mouse liver adenomas. It produces developmental anomalies, including encephalocele, hydro-cephalus, microphthalmia, and anophthalmia, in various animal model systems over doses ranging from 15.6 to 125 mg/kg. Clusters of anophthalmia have been reported in Britain and Wales, speculatively associated with benomyl exposure (by means of residence in a high-use area). Benomyl also causes dose-dependent male reproductive effects.
2. Mercury—Mercury fungicides are no longer used as either seed treatments or paint preservatives, but exposure to mercury remains a significant environmental issue. Because of its persistence in the nervous system, developmental toxicity for these compounds is significant (effect on cerebellar function in behavioral tests). Pregnant mice administered single doses of methyl mercury have anatomically normal offspring that demonstrate functional and histologic cerebellar abnormalities. Currently, there are no mercury-containing seed-treatment fungicides with active US registrations. Organic mercuries used as preservatives in latex paint were phased out following reported cases of acrodynia associated with their use in 1990.
3. Iprodione—The EPA also classifies iprodione as a likely carcinogen based on male and female rat liver adenomas. Testicular adenomas occurred in animals that were fed 1600-ppm iprodione diets. In these doses, the compound also had a marked antiandrogenic effect.
4. Elemental sulfur—Between applications to grapes, tomatoes, and sugar beets, farmers use approximately 70 million pounds of inorganic sulfur annually. It has a low potency when compared with synthetic organic pesticides. Application rates for triadimefon, for example, range from 6 to 8 oz/acre compared with a range of 3–30 lb/acre for inorganic sulfur, often with multiple applications required to achieve control.
Animal testing indicate that inorganic sulfur is basically nonreactive. It produces no irritation in the Draize dermal irritation assay and is negative in predictive allergy tests submitted for pesticide registration. Nevertheless, sulfur is among the most frequently reported sources of illness in agricultural workers, a discrepancy perhaps explained by transformation of elemental sulfur to various sulfur oxide compounds. Some, such as sulfur dioxide and sulfuric acid, are known to cause irritation, and others, notably the sulfite compounds, are recognized causes of allergic reactions.
5. Copper naphthenate, copper sulfate, and other copper compounds—Copper sulfate has been used as a fungicide for many years. Uses include control of downy mildew, blights, leaf spots, apple scab, and peach leaf curl. In vineyards, it is mixed with lime and applied as “Bordeaux mixture.” It causes acute gastric irritation when ingested and also may cause irritation of the eyes and skin.
Systemic poisoning associated with ingestion of copper sulfate also may cause methemoglobinemia. This also has been reported in conjunction with ingestion of an organic copper fungicide, copper-8-hydroxyquinolate. A case reported to the California illness registry also demonstrated 16% methemoglobinemia and borderline elevated levels of serum copper in an adult with indoor exposure to copper naphthenate. Two children from the same residence had definite elevation of serum copper but did not demonstrate methemoglobinemia. Methemoglobinemia may be more likely in patients with partial or complete deficiency of glucose-6-phosphate dehydrogenase (G6PD). The majority of cases of indoor exposure to the compound have exhibited only upper respiratory irritation, eye irritation, and mild, nonspecific systemic symptoms. The tendency to cause irritant symptoms and the apparently long environmental half-life have led to prohibition of indoor applications of copper naphthenate. The compound is still legal to apply as an outdoor wood preservative.
6. Strobilurins—Strobilurins are a new class of fungicides that act by inhibition of the mitochondrial cytochrome oxidase complex at an outer location on the moleculue termed the “quinol” location because it binds hydroquinone. Azoxystrobin and pyraclostrobin are the most frequently used compounds. Trifloxystrobin has somewhat lower reported use but is applied to a similar broad spectrum of crops. Fluoxastrobin has had limited use, principally on nursery crops.
The compounds have very low systemic toxicity and low irritant capacity. It is a nonsensitizer in the Buehler (epicutaneous) testing system. Although they do not seem likely to cause a significant number of illnesses, they are important at this point because they are replacing older materials that have multiple sites of action and a higher degree of chemical reactivity.
![]() Laboratory Findings
Laboratory Findings
Confirmation of a hypersensitivity response to a fungicide requires patch testing for allergic contact dermatitis and for asthma inhalation challenge or pre- and postshift spirometry. Diagnosis of an alcohol dithiocarbamate reaction is based on the history of concurrent exposures. Serum copper levels and methemoglobin levels may be helpful in evaluating patients exposed to copper fungicides to ensure that only irritant symptoms are present. Normal levels of methemoglobin usually are less than 1%, and normal serum copper concentrations range between 65 and 145 μg/dL.
![]() Differential Diagnosis
Differential Diagnosis
Fungicide-induced allergic contact dermatitis must be distinguished from contact dermatitis caused by irritants and allergic contact dermatitis caused by other pesticides or plants. Asthma in a phthalimide manufacturing worker may be due to an intermediate in the manufacturing process.
![]() Treatment
Treatment
Deliberate ingestions of any fungicide may require hospitalization and prolonged supportive measures.
Allergic contact dermatitis is treated by withdrawal of the offending agent and treatment as needed with topical or systemic steroids. Asthma is treated by removal from exposure and symptomatic treatment as needed.
Treatment of antabuse reactions to thiocarbamate fungicides is supportive. In many cases it may be sufficient to give medication for nausea (promethazine or ondansetron) and abdominal discomfort.
![]() Prognosis
Prognosis
Ingestions with fungicides may produce fatal outcomes, but otherwise will likely have limited sequelae. Fungicide-related allergies should resolve following cessation of exposure, but probably will not resolve with continued expsoure. Antabuse reactions are also usually short-term.
HERBICIDES
Herbicides are pesticides that are intended to prevent or control the growth of unwanted plants or kill them once they have appeared. They have largely replaced mechanical methods of weed control and are currently the largest category of pesticides used in agriculture. Included here are plant growth regulators that alter plant development, defoliants that cause leaves to drop prematurely, and desiccants that accelerate the drying of plant parts. Nonselective herbicides affect all plants: selective herbicides affect specific target weeds, contact herbicides affect plant parts that touch the chemical, and translocated herbicides are absorbed by the plant and act at distant sites. Mode of actions for herbicides include hormonal disruptors of plant growth (auxins), inhibitions of plant root development enzymes and cell division, inhibition of fatty acid synthesis, Microtubule assembly inhibition, inhibition of amino acid synthesis, and inhibition of electron transport or inhibition of protoporphyrinogen oxidase (PPO) action in photosynthesis. Most herbicides selectively target plant-specific biochemistry and have low mammalian toxicity. As discussed below, the bipyridyl compounds are an exception in terms of toxicity and broad biological activity.
The herbicides that have serious recognized or suspected human health effects—the dipyridyls and the chlorophenoxyacetic acids—are considered separately in the following sections.
![]() Use
Use
In addition to agriculture, herbicides are used to clear rights-of-way along roadsides, railroads, power lines, fence lines, and property lines; to reduce competition for seedlings in forests; and for fire prevention by reducing the amounts of combustible grasses and brush available as fuel. They are usually sprayed in bands or strips, broadcast over an entire area, or focused on one area or group of weeds (spot or directed treatment). The timing of application may be preplanting (before planting crop), preemergence (after planting but before emergence of weeds or crop), and postemergence (after weeds or crops emerge).
![]() Occupational & Environmental Exposure
Occupational & Environmental Exposure
Occupational exposure to herbicides occurs as a result of dermal exposure to spray applicators and flaggers, whereas environmental exposure occurs in the form of residues on crops and food. Although a few studies suggest the possibility of health effects occurring in populations as a result of spray drift or other environmental contamination from herbicides, with the exception of cases of obvious spray drift with damage to nontarget plants, such exposure is difficult to document.
![]() Pharmacokinetics & Mechanism of Action
Pharmacokinetics & Mechanism of Action
Apart from the bipyridyl compounds paraquat and diquat, most herbicides do not cause significant mammalian toxicity (Table 34–15). For the low toxicity herbicides, there is often little information on human pharmacokinetics or medical experience with poisoning. Some otherwise low toxicity compounds may be formulated with irritating or sensitizing inert ingredients.
Table 34–15. Herbicide acute toxicity.
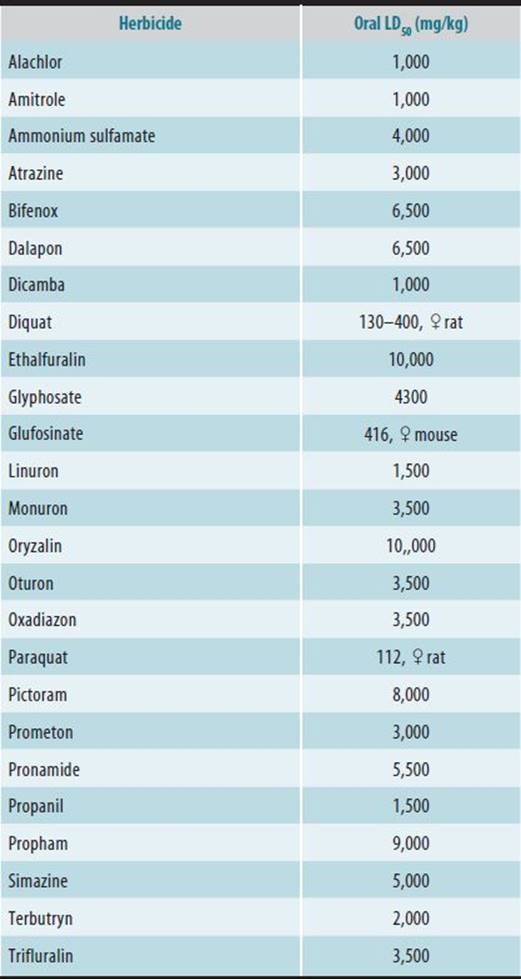
![]() Animal & Human Cancer Studies Implicating Herbicides
Animal & Human Cancer Studies Implicating Herbicides
Classes of herbicides demonstrating some evidence of carcinogenicity in animals include acetanilides (ie, acetochlor, alachlor, metolachlor, and butachlor), nitrobenzenes, phenol derivatives, and triazines. Human data for most of the compounds are lacking.
![]() Clinical Findings
Clinical Findings
A. Symptoms and Signs
Some formulations of herbicides contain organic solvents, surfactants, emulsifiers, or other vehicles and additives that may cause eye, nose, or throat irritation in applicators exposed to spray mists and dermatitis in mixers and loaders as a result of prolonged skin contact. Otherwise, these compounds have no known human health effects.
B. Laboratory Findings
There is little or no information regarding measurement of parent compounds or metabolites in biologic media.
![]() Differential Diagnosis
Differential Diagnosis
It is always possible that these compounds have effects not yet appreciated in humans, particularly from accidental or deliberate ingestion. The toxicity of “inert” ingredients should be considered in evaluating persons with symptoms following exposure.
![]() Treatment
Treatment
Because these compounds have little or no known human health effects, treatment of any symptoms resulting from their use should be symptomatic only. For evaluation of symptomatic patients, the manufacturer should be consulted, particularly for identification of the inert ingredients.
![]() Prognosis
Prognosis
Acute irritation and dermatitis from herbicide formulations should resolve shortly after cessation of exposure.
CHLOROPHENOXYACETIC ACIDS
ESSENTIALS OF DIAGNOSIS
![]() Acute effects
Acute effects
• Topical irritation—redness of skin, burning, soreness in throat and chest, and cough.
• Ingestion associated with CNS depression, myotonia, rhabdomyolysis, renal failure, nausea and vomiting, severe hypotension.
![]() Chronic effects
Chronic effects
• Scattered cases of peripheral neuropathy have been reported following either ingestion of 2,4-dichlorophenoxyacetic acid (2,4-D) or occupational exposure.
![]() General Considerations
General Considerations
The principal herbicidal derivatives of phenoxy acetic acid include 2,4-dichlorophenoxyacetic acid (2,4-D), 2,4,5-trichlorophenoxyacetic acid (2,4,5-T), 2-methyl-4-chlorophenoxyacetic acid (MCPA), and their salts and ester derivatives. Silvex, lcuron, and fenac are homologues of 2,4,5-T, whereas 2,4-DB and MCPB, MCPCA, and MCPP are homologues of 2,4-D and MCPA, respectively. They are translocated herbicides relatively selective for broad leaf plants. 2,4,5-T and its homologues are no longer manufactured or used in the United States because of their combination contamination with 2,3,7,8-tetrachlorodibenzo-p-dioxin (TCDD) and the controversy over health effects in environmentally exposed populations and Vietnam veterans. While certain batches of 2,4-D have been found to be contaminated with low levels of other lesser chlorinated dioxins such as dichlorodibenzo-p-dioxin, none of its contaminants has been found to be of toxicologic importance.
![]() Use
Use
The chlorophenoxy herbicides have had a wide variety of uses, including control of undesirable perennial hardwood trees and plants for “release” of desirable evergreen softwood trees.
![]() Occupational & Environmental Exposure
Occupational & Environmental Exposure
Occupational exposure occurs primarily as direct contact with liquid concentrate during mixing and loading and inhalation and contact with spray mist during application. Although concern has been expressed about environmental exposure of populations living near conifer forests where chlorophenoxy herbicides are applied, in the absence of obvious spray drift with nontarget crop damage, such exposure is difficult to document. 2,4-D is degraded rapidly in the environment, and water contamination has not been a major problem.
![]() Pharmacokinetics & Mechanism of Action
Pharmacokinetics & Mechanism of Action
The herbicidal mechanism of action of chlorophenoxyacetic acids is uncertain but appears to involve a mimicking of plant auxins (growth hormones) and effects on plant metabolism. They are absorbed by inhalation, dermal contact, and ingestion and are excreted rapidly unchanged in urine. The mechanisms of any health effects on humans other than irritation are uncertain. They are weak uncouplers of oxidative phosphorylation and may produce hyperthermia at extremely high doses as a result of increased heat production. A study reported to the EPA indicated that 2,4-D caused an increase in brain tumors in rats given 40 mg/kg per day by mouth.
![]() Clinical Findings
Clinical Findings
A. Symptoms and Signs
Some formulations produce skin irritation following contact with liquid; irritation of the eyes, nose, throat, and respiratory tract, with burning and cough from exposure to spray mist; and irritation of the gastrointestinal tract, with abdominal pain, nausea, and vomiting following ingestion. Ingestion of chlorophenoxyacetic acid herbicides has resulted in nausea, vomiting, abdominal pain, and diarrhea followed by muscle twitching, myotonia, metabolic acidosis, and a hypermetabolic state with fever, tachycardia, hypertension, sweating, convulsions, and coma.
Approximately six cases of peripheral neuropathy have been reported following relatively large dermal exposures to 2,4-D over the course of a few days. Clinically, these resembled idiopathic acute symmetric polyneuropathy (Guillain-Barre syndrome) and organophosphate-induced delayed neuropathy in their symptoms of an initial influenza-like illness associated with nausea, vomiting, diarrhea, and myalgias, followed by an asymptomatic interval and then, 7–10 days later, by rapidly ascending loss of both motor and sensory nerve function. Respiratory function was spared in most cases.
Chloracne has been reported as a result of exposure to TCDD in 2,4,5-T manufacturing plant workers. A number of epidemiologic studies have suggested an association between exposure to chlorophenoxyacetic acid herbicides and soft-tissue sarcomas and lymphomas. Follow-up of the US manufacturing workers cases exposed to TCDD in the manufacture of 2,4,5-T and pentachlorophenol demonstrated an increase of all cancers combined. The level of serum TCDD in this population far exceeded background levels in the US population and levels found in military personnel who handled 2,4,5-T containing herbicides during the Vietnam war.
B. Laboratory Findings
Exposure to a chlorophenoxyacetic compound can be confirmed through analysis of blood or urine by gas-liquid chromatography. Urine samples should be collected as soon as possible after exposure because the chemical may be excreted completely within 24–72 hours. There is insufficient information to relate a spot urine level precisely to a level of exposure. However, because these compounds are excreted almost entirely unchanged in urine, a dose can be measured by collecting and analyzing all urine, provided collection is begun promptly after exposure. Other laboratory findings in cases of acute intoxication are entirely nonspecific. In the few cases of peripheral neuropathy associated with exposure to 2,4-D where testing was done, nerve conduction velocities were normal or slightly depressed, and spinal fluid analyses were unremarkable.
![]() Differential Diagnosis
Differential Diagnosis
Acute irritation following direct exposure or acute intoxication following ingestion presents with obvious diagnoses. The differential diagnosis for a patient with peripheral neuropathy following exposure to 2,4-D includes idiopathic acute symmetric polyneuropathy and exposure to other neurotoxic compounds, including organophosphates.
![]() Treatment
Treatment
Treatment of acute irritation and peripheral neuropathy is entirely symptomatic. Because chlorophenoxyacetic compounds are weak organic acids, they are preferentially excreted in alkaline urine. In severe poisoning from ingestion of large doses, alkalinization of the urine can hasten elimination of the chemical and may improve the course of intoxication. Administration of large fluid volumes to achieve “forced” diuresis should be avoided owing to the risk of precipitating pulmonary edema.
![]() Prognosis
Prognosis
Although death from ingestion of a chlorophenoxyacetic acid has been reported, severe intoxications apparently have been infrequent, and most victims have survived. In cases of peripheral neuropathy following exposure to 2,4-D, maximum paralysis lasted approximately 1 week or less. Recovery of function usually was prolonged for up to 1 year following exposure, with some residual weakness in most cases.
![]() Chronic Health Effects
Chronic Health Effects
A number of studies suggest an association between phenoxy herbicides and soft-tissue sarcoma, but these studies have not been consistently replicated in other populations. Nevertheless, manufacturing populations have shown dose-response relation-ships between exposure (characterized by high levels of serum dioxin) and total cancer mortality.
DIPYRIDYLS (PARAQUAT & DIQUAT)
ESSENTIALS OF DIAGNOSIS
![]() Acute effects
Acute effects
• Contact with skin, eyes, and respiratory tract—irritation and fissuring of skin of hands, cracking and discoloration of fingernails, conjunctivitis, sore throat, and coughing.
• Ingestion of paraquat—early (1–4 days), oral and abdominal pain, nausea, vomiting, and diarrhea; later (24–72 hours), liver injury, jaundice, elevated hepatocellular enzymes, and renal injury (proteinuria, hematuria, pyuria, elevated serum urea nitrogen and creatinine); late (3–4 days), pulmonary fibrosis (cough, dyspnea, tachypnea, cyanosis, and respiratory failure).
• Ingestion of diquat—same as paraquat without late pulmonary fibrosis.
![]() General Considerations
General Considerations
Paraquat is used extensively in the United States and worldwide, diquat to a lesser extent. They are nonselective contact herbicides.
![]() Use
Use
The dipyridyls are used extensively as general-purpose herbicides owing to their ability to kill most plants on contact. They are also used as defoliants and desiccants because the foliage of plants becomes dry and frostbitten in appearance, resulting in the premature dropping of leaves.
![]() Occupational & Environmental Exposures
Occupational & Environmental Exposures
The most important occupational exposures occur by direct contact of the skin with liquid concentrate during mixing and loading and inhalation and skin contact with spray mist during application. A case of acute paraquat intoxication was reported in a flagger who endured extensive dermal exposure to spray mist. Environmental exposure through field residues, food residues, and water contamination has not been a concern. The program of the U.S. Drug Enforcement Agency to spray marijuana fields with paraquat generated controversy over the possibility of inhalation of paraquat by marijuana smokers. Most of the paraquat probably undergoes thermal decomposition before it is inhaled, but the possibility of adverse effects from paraquat or its decomposition products has not been ruled out.
![]() Pharmacokinetics & Mechanism of Action
Pharmacokinetics & Mechanism of Action
The dipyridyls affect both plants and mammals by damaging tissue through the generation of oxygen free radicals. Their effect on plants requires the presence of sunlight. They are absorbed by inhalation, dermal contact, or ingestion. They damage epithelial tissues such as skin, nails, cornea, gastrointestinal tract, and respiratory tract, as well as the liver and kidneys.
Paraquat is more toxic to humans than diquat. A small sip of the liquid concentrate can kill an adult, which accounts for the hundreds of deaths reported worldwide from accidental and deliberate ingestion of this herbicide. An experimental trial that consisted of adding an emetic to formulations of paraquat was instituted recently in an attempt to reduce the frequency of fatal ingestions.
A relatively small number of cases of serious poisoning from paraquat have been reported as a result of large dermal exposures, whereas none has been reported from inhalation exposure in the absence of significant skin contact. Pulmonary injury from chronic dermal or inhalation exposure has not been reliably reported or found in the few epidemiologic studies performed with applicators. Neither paraquat nor diquat has been adequately tested for carcinogenicity.
![]() Clinical Findings
Clinical Findings
A. Symptoms and Signs
Direct contact with concentrated liquid dipyridyls results in skin irritation and fissuring and in cracking, discoloration, and sometimes loss of the fingernails. Liquid splashed in the eye can cause conjunctivitis and opacification of the cornea. Inhalation of spray mist can irritate the nose and throat, causing nosebleeds and sore throat.
Ingestion of either paraquat or diquat can result in an early phase (1–4 days) of inflammation of the mouth and gastrointestinal tract, with soreness, ulceration, burning pain, nausea, vomiting, diarrhea, and sometimes hematemesis and melena. These symptoms can range from mild to severe, and their intensity may not predict the severity of the following phases. The second phase begins 24–72 hours after exposure and is marked by evidence of hepatic and renal injury. Hepatocellular injury is indicated by abdominal pain, nausea, and jaundice. Renal injury usually is asymptomatic unless oliguria or anuria develops. Renal and hepatic injury from ingestion of paraquat is common and frequently severe, whereas that from ingestion of diquat is less common and often milder.
A late phase (72–96 hours) of pulmonary fibrosis occurs from paraquat, but not diquat, presumably because paraquat, but not diquat, becomes concentrated in pulmonary epithelial tissue. Pulmonary edema has occurred occasionally following ingestion of either paraquat or diquat. In cases of paraquat poisoning, pulmonary fibrosis is marked by cough, shortness of breath, and tachypnea. Advanced fibrosis is indicated by progressive cyanosis.
B. Laboratory Findings
In the early phase of acute poisoning, the findings are non-specific and usually are related to dehydration from nausea and diarrhea. In the later phase, liver injury is indicated by elevated bilirubin and hepatocellular enzymes. Renal injury, primarily tubular, is indicated by proteinuria, hematuria, pyuria, and elevated serum urea nitrogen and creatinine. Oliguric renal failure typical of acute tubular necrosis may occur. Laboratory evidence of pulmonary fibrosis from paraquat in the form of a progressive decline in arterial oxygen tension and diffusion capacity for carbon monoxide commonly precedes the appearance of pulmonary symptoms. Later, pulmonary function findings are typical of restrictive lung disease. The diagnosis of acute intoxication from paraquat or diquat can be confirmed by analysis of either compound in blood and urine.
![]() Differential Diagnosis
Differential Diagnosis
The early phase of acute intoxication from a dipyridyl may be mild and, in the absence of a history of ingestion, may be mistaken for gastroenteritis or ingestion of another irritant chemical. The combination of renal and hepatic injury could occur following exposure to a chlorinated hydrocarbon solvent such as carbon tetrachloride. In the absence of a history of paraquat exposure, the differential diagnosis of the pulmonary injury is the same as for acute pulmonary fibrosis (see Chapter 23).
![]() Treatment
Treatment
The primary treatment during any phase of intoxication from paraquat or diquat is supportive, particularly during periods of organ failure. Bentonite and Fuller’s earth are more effective absorbents for dipyridyls in the gastrointestinal tract than activated charcoal. If available, they should be administered as a 7 g/dL suspension in normal saline in quantities of at least 2 L to any patient suspected of ingesting any quantity of a dipyridyl within the preceding several days. If neither bentonite nor Fuller’s earth is available, a similar quantity of the usual concentration of activated charcoal should be administered.
Saline catharsis then is recommended, using sodium sulfate rather than magnesium salts because of the risk of magnesium retention in the presence of impaired renal function. This cycle may be repeated for several days. Given the high fatality rate following paraquat ingestion, this extreme degree of gut cleansing is probably worth the risk of fluid and electrolyte imbalance, which must be monitored closely.
The issue of enhanced excretion of dipyridyl is controversial. There is no basis for the recommendation that glucose and electrolyte infusions be given in large quantities to minimize toxicant concentrations in tissues and force diuresis of the compounds. Memo-dialysis is clearly ineffective for removal of paraquat. Hemoperfusion with coated charcoal may be effective in removing paraquat from the blood if it is performed before the chemical has been distributed to tissues. However, few patients have a confirmed diagnosis and can be placed in a facility where the procedure can be performed early (24–48 hours after ingestion). The decision to perform hemoperfusion should be made by a physician with experience in the technique and familiarity with the issues and risks involved.
A number of therapies are available to attempt to retard pulmonary fibrosis from paraquat. Increased levels of alveolar oxygen increase the rate of production of oxygen free radicals and accelerate the process of pulmonary fibrosis. Animal studies show increased survival in low-oxygen atmospheres, but there are no comparable human studies. Early placement of a patient in an atmosphere of 15% oxygen has been recommended. Supplemental oxygen should be administered only as necessary to maintain minimally acceptable levels of oxygenation. Early experimental results with the free-radical scavenger superoxide dismutase have been disappointing. Corticosteroids and cytotoxic agents such as azathioprine have been tried with uncertain results.
![]() Prognosis
Prognosis
Once pulmonary fibrosis occurs as a result of paraquat ingestion, death from respiratory failure can be expected. Survival with disability from restrictive lung disease also may occur. Occasionally, recovery of lung function may take place over a course of weeks to months. Although death from liver and kidney necrosis may occur following diquat ingestion, recovery is more common than following paraquat ingestion.
Phosphonate Herbicides
Glyphosate and its numerous derivatives function by inhibiting the enzyme 5-enolpyruvylshikimate-3-phosphate synthase (EPSPS), blocking the synthesis of the aromatic amino acids phenylalanine, tyrosine, and tryptophan. It has low systemic toxicity. Glufosinate blocks the synthesis of gluta-mine. It has moderate systemic toxicity.
Glyphosate is typically formulated with surfactants that can cause eye or skin irritation, typically affecting applicators or other pesticide handlers. The formulation residue does not cause illness. Serious illnesses from glyphosate sometimes occur after ingestion, with symptoms also likely related to the surfactant content. Newer formulations that contain mixtures of glyphosate and diquat have toxicity in proportion to the concentration of diquat, especially in cases of deliberate or accidental ingestion.
![]() Chronic effects
Chronic effects
Treatment with glufosinate-ammonium interfered with gestation in rat feeding studies before and after implantation at daily doses of 27.8 mg/kg and greater in the absence of maternal toxicity. The no effect level was 9.1 mg/kg. Because the mechanism of action of glufosinate is relevant to human development, the observed effects are of potential public health concern. In a review sponsored by the European Union, the margin of safety for applicators handling glufosinate was high (995 with incomplete use of protective equipment and more than 4000 with complete protective equipment).
INFORMATION SOURCES
Information on the identity, exposure, toxicity, and clinical management of specific pesticides is often found through online sources. Information about pesticides used in local agriculture can be obtained through the county agricultural commissioner or extension agent in individual jurisdictions. Specific advice about medical treatment can be obtained through regional poison control centers.
For commonly used agricultural compounds, a consortium of university extension toxicologists also provides online information, often derived from data supplied by pesticide manufacturers, at Extoxnet. The EPA booklet, “Recognition and Management of Pesticide Poisonings,” is a concise guide to diagnosis and treatment.
REFERENCES
Barry KH: Methyl bromide exposure and cancer risk in the Agricultural Health Study. Cancer Causes Control 2012;23: 807 [PMID: 22527160].
Buckley DA: Allergy to oxidized linalool in the UK. Contact Dermatitis 2011;64:240 [PMID: 21392034].
Engel SM: Prenatal exposure to organophosphates, paraoxonase 1, and cognitive development in childhood. Environ Health Perspect 2011;119:1182 [PMID: 21507778].
Koutros S: Risk of total and aggressive prostate cancer and pesticide use in the Agricultural Health Study. Am J Epidemiol 2013;177:59 [PMID: 23171882].
Maund SJ: Ecotoxicology of synthetic pyrethroids. Top Curr Chem 2012;314:137 [PMID:22025065].
O’Malley M: Inhalation of phosphine gas following a fire associated with fumigation of processed pistachio nuts. J Agromedicine 2013;18:151 [PMID: 23540306].
Rauh VA: Brain anomalies in children exposed prenatally to a common organophosphate pesticide. Proc Natl Acad Sci 2012;109:7871 [PMID: 22547821].
Stromberg PE: Cases from NACCT acute and intensive care symposium: altered mental status, seizures, and rash in a fumigation company employee. Clin Toxicol 2013;51:182 [PMID: 23473464].
![]() SELF-ASSESSMENT QUESTIONS
SELF-ASSESSMENT QUESTIONS
Select the one correct answer to each question.
Question 1: In the United States, the Environmental Protection Agency (EPA)
a. regulates the registration, sale, and conditions of use of all pesticides
b. defers to OSHA the responsibility for the protection of agricultural workers exposed to pesticides
c. narrowly defines pesticides for registration for sale and use
d. ignores studies of hazards to nontarget organisms
Question 2: Organophosphates
a. are esters of phosphoric acid that exist in any number of forms
b. bind with the cholinesterase molecule
c. bind with the phosphate portion of the serine molecule
d. have no irreversible effects
Question 3: Carbamates
a. share a common mechanism of chronic toxicity with organophosphates
b. present unique signs and symptoms of acute poisoning
c. differ from organophosphates in causing reversible rather than irreversible cholinesterase inhibition
d. typically have a longer clinical course than organophosphate poisoning
Question 4: Treatment of organophosphate poisoning
a. should be instituted on clinical grounds alone
b. should be delayed pending determination of cholinesterase levels
c. may follow a test dose of atropine with marked signs of atropinization
d. may follow a test dose of atropine with no signs of atropinization
Question 5: Organochlorine insecticides
a. are chlorinated hydrocarbon compounds of cyclic structure and low molecular weight
b. are of low volatility and have CNS effects similar to general anesthetics
c. are poorly absorbed by inhalation or ingestion but are absorbed rapidly through the skin
d. are highly fat soluble and are distributed to adipose tissue, the liver, and the nervous system
Question 6: Fumigants
a. have innately high vapor pressures or by-products with high vapor pressure
b. have a low degree of chemical and biological reactivity
c. cause respiratory and eye irritation, CNS injury, and retinal injury
d. have no measurable carcinogenicity and reproductive effects in animal studies
Question 7: Halogenated hydrocarbon fumigants
a. are primary irritants with negligible potency
b. are excreted slowly leading to significant bioaccumulation
c. typically cause CNS stimulation
d. share many of the effects of the halogenated hydrocarbon solvents