Lower extremity pain and paresthesia are common symptoms of peripheral nervous system (PNS) disorders. Diagnosing a mononeuropathy requires that the motor, reflex, and sensory changes be confined to a single nerve and can, if necessary, be supported by electrodiagnostic studies (EDS).
Diagnosis and management of PNS disorders was once the sole domain of specialists. However, “reemergence” of generalists in health care has resulted in at least two major trends. The first, as expected, is that persons with such disorders are no longer under the sole care of a specialist. The second, somewhat “unintentional” effect, is that the role of specialists has become more demanding. Specialists need to develop a greater proficiency in differentiating neuropathy from radiculopathy, plexopathy, and other nonneurologic syndromes of pain, disturbed sensation, or weakness. This increasing competency occurs in the setting of fewer and more carefully selected laboratory investigations.
Fortunately, recognition of a neuropathy has always necessitated careful attention to the history and examination, skills that are expected of specialists, but “within reach” of a generalist. After localization, a ranking of potential etiologies is formulated, and further diagnostic evaluation is planned.
This chapter provides an outline of common as well as some infrequent forms of lower extremity neuropathy. Symptoms and findings are emphasized, and the most frequent etiologic considerations are mentioned. The importance of bedside examination is assumed throughout, but the application of diagnostic tests is also reviewed.
I. EVALUATION
A. History. Various aspects of the history help in narrowing the etiologies of a mononeuropathy. The nature of onset (abrupt or insidious), preceding events (injury, surgery, or illness), associated symptoms (fever, weight loss, or joint swelling), and aggravating or alleviating features (joint position or specific activities) are all important. Because the observed deficit can be similar regardless of etiology, historical information is instrumental in limiting the differential diagnosis.
B. Physical examination. Although motor and sensory symptoms and signs correspond to the distribution of a single peripheral nerve or branch, the degree of deficit and constellation of findings can vary. Motor signs may be clinically absent, or varying degrees of weakness, atrophy, or fasciculation may be found. Likewise, sensory symptoms can be positive (e.g., tingling, pricking, and burning), negative (hypesthesia), or, while corresponding to a sensory distribution of a nerve, may be most pronounced in its distal distribution. Therefore, the sensory examination should begin with the patient’s description of the area of involvement. The course of the nerve should be evaluated and local areas of discomfort or the presence of a Tinel’s sign (pain or paresthesia in the cutaneous distribution of a nerve elicited by light percussion over that nerve) sought. The relationship of sites of discomfort to adjacent anatomic structures helps in identifying sites of nerve entrapment or compression.
C. Diagnostic studies. Further evaluation may be necessary in confirming the presence and severity of a mononeuropathy and excluding more proximal sites of involvement (plexus or root) that can clinically mimic a mononeuropathy.
1. EDS. An EMG and nerve stimulation studies (NSS) are quite useful in the evaluation of mononeuropathies. They can aid in localization, detect bilateral but asymmetric nerve involvement (or detect an underlying polyneuropathy), define severity, and provide prognostic information.
2. Laboratory testing is directed at identification of a systemic or generalized disease that may be a predisposing factor and with newer imaging techniques, at times, targeted fascicular nerve biopsy. Owing to the practical nature of this section, an exhaustive listing of the medical, systemic diseases or structural disorders that can cause a mononeuropathy is not provided. The Recommended Readings provide extensive tabulations of frequent as well as unusual etiologies.
3. Imaging studies. Radiographic imaging was previously undertaken to identify intrathoracic, abdominal, retroperitoneal, or pelvic masses that may lead to nerve root, plexus, or nerve injury, but now it is also applied to imaging the PNS directly. This role of imaging studies has considerably advanced now that MRI is the method of choice for delineating a focal site of involvement, characterizing and at times assisting in the diagnosis of a nerve lesion. However, its effectiveness not only depends on the necessary MRI hardware and software, but clinical expertise through an interdisciplinary and collaborative effort among physicians and clinical departments. Routine X-ray studies play a less significant role.
II. SPECIFIC FORMS OF MONONEUROPATHY
A. Femoral and saphenous neuropathy. Formed within the psoas muscle by fusion of the posterior divisions of the ventral rami of the L2–4 spinal nerves, the femoral nerve exits from the lateral border of the psoas and descends between it and the iliacus muscles (which it may also innervate), but under the fascia of the iliacus. Emerging under the inguinal ligament, lateral to the femoral artery, the nerve divides into motor branches, which supply the quadriceps muscles, and sensory branches to the anterior portion of the thigh (Figure 25.1). One major division, the saphenous nerve, descends medially within Hunter’s (adductor) canal, accompanying the femoral artery. At the medial superior aspect of the knee, it emerges from the canal and, accompanying the saphenous vein, descends medially down the leg and ends at the medial aspect of the foot. The saphenous nerve supplies the sensory innervation to the medial aspect of the leg and foot.
1. Etiology. Femoral neuropathy is usually caused by trauma from surgery (intrapelvic, inguinal, or hip operations), stretch or traction injuries (prolonged lithotomy position in childbirth), or direct compression (hematoma within the iliacus compartment). Although diabetes mellitus is described as a frequent etiologic factor, such cases are misnomers and represent a restricted plexopathy or more diffuse lesions, but predominantly affecting femoral nerve function. Saphenous neuropathy is most often attributable to injury following surgery (peripheral vascular, saphenous vein removal, or knee operations).
2. Clinical manifestations.
a. History. The patient reports leg weakness (as if the leg will “fold under”) on attempting to stand or walk. Pain in the anterior part of the thigh accompanied by the abrupt onset of leg weakness is a frequent presentation of an iliacus (retroperitoneal) hematoma. A similar pattern of pain, but usually subacute in onset, can be observed in cases of “femoral neuropathy” occurring in diabetes mellitus. With the exception of pain, sensory involvement tends to be infrequent and a minimal symptom of femoral neuropathy.
Because of its association with surgery, sensory loss in saphenous neuropathy may initially go unnoticed or be of little concern to the patient. However, pain may be prominent, and in such cases, it usually appears sometime after the assumed injury to the nerve.
b. Physical examination.
(1) Neurologic. Examination reveals weakness of the quadriceps muscles, absent or diminished patellar reflex, and sensory loss over the anterior thigh and, with saphenous nerve involvement, the medial aspect of the leg and foot.
(2) General. Examination or palpation within the inguinal region and, in cases of saphenous nerve involvement, the medial aspect of the knee may identify focal areas of pain and perhaps the site of involvement. The proximity of a surgical scar or point of injury can provide additional etiologic clues. In cases in which retroperitoneal hemorrhage is suspected, peripheral pulses may be normal, but there is characteristic posturing of the leg (held flexed at the hip), and attempts to extend or perform a reverse straight-leg test exacerbate the pain.
3. Differential diagnosis. Discovery of hip adduction weakness suggests a more proximal process, plexus or root as the site of involvement, although a superimposed obturator neuropathy cannot be excluded.
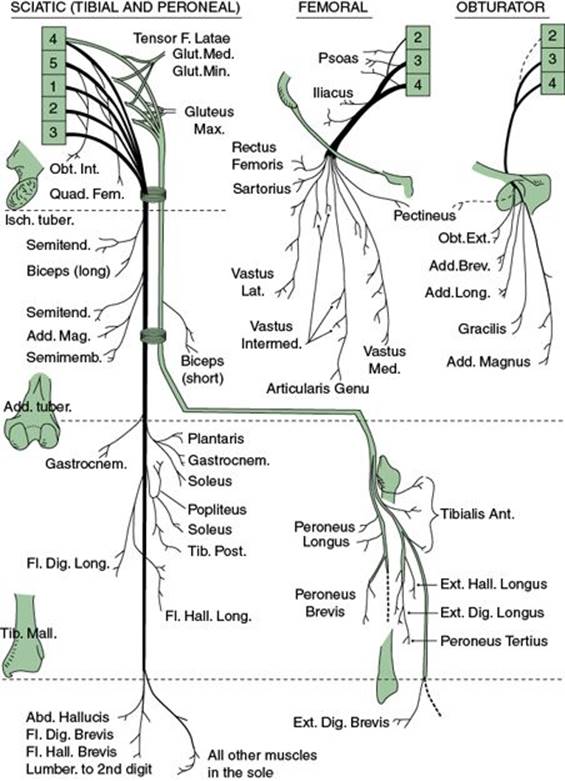
FIGURE 25.1 Motor distribution of the nerves of the lower limb. (Modified from Basmajian JV. Grant’s Method of Anatomy. 9th ed. Baltimore, MD: Williams and Wilkins; 1975.)
4. Evaluation.
a. Electrodiagnostic. NSS are not as helpful as EMG in the evaluation of a suspected femoral neuropathy. EMG includes study of other L2–4 innervated muscles and paraspinal muscles because they should not be involved in isolated femoral neuropathy.
b. Imaging. CT or MRI of the retroperitoneum helps to identify cases resulting from retroperitoneal hemorrhage or suspected mass lesion.
B. Obturator neuropathy. Arising within the psoas muscle from ventral divisions of the L2–4 spinal nerves, the obturator nerve exits from the psoas muscle at its lateral margin, descends into the pelvis, and exits through the obturator foramen. It innervates the gracilis, adductor magnus, longus, and brevis muscles and supplies sensation to the upper medial aspect of the thigh.
1. Etiology. Isolated neuropathy of the obturator nerve is unusual. In cases resulting from pelvic or hip fracture, involvement of other nerves to the lower extremity or lumbosacral plexus also occur. Benign and malignant pelvic masses can result in obturator neuropathy, as can surgical procedures performed on those masses or within the pelvis.
2. Clinical manifestations.
a. History. Leg weakness and difficulty walking are the most common first symptoms and usually overshadow sensory loss, if present.
b. Physical examination.
(1) Neurologic. Motor evaluation shows weakness of hip adduction, and sensory loss may be found along the upper medial thigh. The patellar reflex should be intact.
(2) General. Careful pelvic and rectal examinations can identify an intrapelvic tumor and are necessary when obturator paralysis occurs without trauma.
3. Differential diagnosis. The presence of hip flexor or knee extensor weakness or an impaired patellar reflex suggests a lumbosacral plexopathy or L3–4 radiculopathy. In addition, sensory loss, which extends below the knee, is inconsistent with the expected sensory deficit.
4. Evaluation.
a. Electrodiagnostic. NSS are not as helpful as EMG where involvement of other L2–4 muscles or paraspinal muscles identifies a more proximal lesion.
b. Imaging. When obvious trauma is not a consideration, radiological imaging of the pelvic cavity is helpful when a mass or infiltrative lesion is suspected.
C. Lateral femoral cutaneous neuropathy. Dorsal divisions of the ventral primary rami of the L2–3 spinal nerves contribute to the lateral femoral cutaneous nerve, which emerges from the lateral border of the psoas major muscle. It then crosses laterally, within the fascia of the iliacus muscle, and crosses over the sartorius muscle before passing under the lateral border of the inguinal ligament. Piercing the fascia lata, it divides into anterior and posterior branches that provide sensory innervation to the anterolateral aspects of the thigh. Anatomic variation is frequent in regard to origin (it can arise as a branch of the femoral or genitofemoral nerve), course of its branches and extent of its sensory innervation.
1. Etiology. In most cases, entrapment or compression at or near the inguinal ligament is the assumed etiologic factor. However, entrapment or compression at other sites (i.e., retroperitoneal mass), surgical procedures (especially those involving retroperitoneal structures, pelvis, or inguinal sites), and trauma to the thigh can also injure the lateral femoral cutaneous nerve.
2. Clinical manifestations.
a. History. Pain (burning or a “crawling” sensation) with variable loss of sensation on the anterolateral aspects of the thigh and exacerbated by walking or arising from a chair (meralgia paresthetica). Frequently, the patient rubs the thigh for relief.
b. Physical examination.
(1) Neurologic. The area of sensory change usually is small and over the lateral aspect of the thigh.
(2) General. Careful palpation along the inguinal ligament and anterior pelvic brim will infrequently detect an area of tenderness or precipitate symptoms.
3. Differential diagnosis. The primary differential diagnosis is a femoral neuropathy. Lumbar plexopathy and L2 radiculopathy are considerations, but limited sensory impairment, lack of motor involvement, and intact reflexes help in excluding them.
4. Evaluation. Although clinical features usually provide enough support for a diagnosis, when uncertainty or a preexisting illness complicates the issue (retroperitoneal mass), further testing may be needed.
Unlike the situation with other entrapment syndromes, responsiveness to treatment may help to “confirm” the diagnosis of lateral femoral cutaneous neuropathy. Subcutaneous injection of an anesthetic agent or steroid at the assumed exit point of the lateral femoral cutaneous nerve (medial to the anterior superior iliac spine and under the inguinal ligament) or at a site of local tenderness may relieve symptoms and support but does not confirm the diagnosis.
a. Electrodiagnostic. Difficulty in eliciting a response from healthy or control subjects during NSS has limited the use of such testing. However, EMG studies play a more important role by clarifying unusual or unclear symptoms, because detection of clinically silent motor involvement implies involvement of more than the lateral femoral cutaneous nerve.
b. Imaging. Radiographic imaging is not indicated. However, unexplained or concomitant gastrointestinal or urogenital symptoms should raise suspicion of a retroperitoneal or pelvic mass and then further evaluation is appropriate.
D. Sciatic neuropathy. The sciatic nerve arises from the ventral rami of the L4–5 spinal nerves, which, by way of the lumbosacral trunk, fuse with those from S1 to S3. Passing along the inner wall of the pelvis, it exits through the sciatic notch and passes under the piriformis muscle, where it lies between the ischial tuberosity and greater trochanter. Remaining in this deep location, the sciatic nerve descends into the extremity and proximal to the knee, divides into the peroneal and tibial nerves. The sciatic nerve itself is clearly divisible into two trunks—the medial, which receives contributions from the L4 to S3 rami and gives rise to the tibial nerve, and the lateral, the contributions of which are from L4 to S2 and from which the common peroneal nerve is derived. The sciatic nerve itself has no sensory branches. The lateral trunk provides innervation to the short head of the biceps femoris muscle and, by way of the medial trunk, the semitendinosus, semimembranosus, and long head of the biceps femoris muscles. With the obturator nerve, the adductor magnus muscle is also innervated.
1. Etiology. Most cases of sciatic neuropathy, whether involved at the gluteal level or the thigh, are secondary to trauma. (The sciatic nerve is possibly second to the peroneal nerve in this regard.) This includes injury to adjoining or neighboring structures in fracture of the pelvis, hip, or femur as well as from direct injury. Injection injuries are no longer as frequent, but compression injuries are increasing and often occur in the setting of prolonged immobility such as in various operative procedures (i.e., cardiac bypass graft surgery). Miscellaneous causes include entrapment by fibrous constricting bands, local hematoma, or tumor.
Mention must be made of the piriformis syndrome. At this time, few cases rigorously support the assumed pathogenesis of this syndrome, compression of the sciatic nerve by the overlying piriformis muscle, although it remains a frequent clinical diagnosis. Point tenderness of the sciatic nerve at the level of the piriformis muscle can be found among patients with plexopathy or lumbosacral radiculopathy and does not necessarily confirm pathologic compression of the sciatic nerve by the piriformis muscle.
2. Clinical manifestations.
a. History. Complete lesions are associated with paralysis of the hamstring muscles and all muscles below the knee. Sensory loss occurs in the tibial and peroneal distributions. Partial lesions, especially those of the lateral trunk, make up most cases of sciatic neuropathy and often manifest as foot-drop.
b. Physical examination.
(1) Neurologic. Although variable paralysis of muscles innervated by both the medial and lateral trunk can be present, involvement of muscles innervated by the lateral trunk is the most frequent presentation. Sensory loss is variable, but restricted to the distribution of the sensory branches of the peroneal and tibial nerves. The muscle stretch reflexes of the hamstring and Achilles’ tendons can be depressed.
(2) General. Palpation along the course of the nerve may help identify masses or locate points of pain and tenderness, but cannot entirely exclude more proximal nerve lesions.
3. Differential diagnosis. Care must be taken to ensure that a radiculopathy (especially L5–S1) is not masquerading as a sciatic neuropathy. The straight-leg-raise test, frequently positive in radiculopathy, can also be present in cases of lumbosacral plexopathy and sciatic neuropathy. A careful rectal and pelvic examination is indicated when sciatic neuropathy is suspected because involvement of the sacral plexus by a pelvic mass may not otherwise be identified. Finally, an isolated common peroneal or tibial neuropathy must be considered.
4. Evaluation.
a. Electrodiagnostic. Both NSS and EMG are useful in differentiating sciatic mononeuropathy from L5 to S2 radiculopathies or plexopathy and require careful study of the paraspinal and gluteal muscles. Yet at times there can still be diagnostic confusion. When the lateral division of the sciatic nerve is “selectively” involved, EMG and NSS may show a pattern that suggests peroneal nerve involvement, whereas tibial nerve studies may appear to be normal.
b. Imaging. In cases in which radiculopathy and plexopathy cannot be excluded, further radiological studies can provide useful information. In addition, in cases in which only sciatic nerve involvement is found, MRI with gadolinium infusion may effectively depict the course of the nerve and help in identifying focal abnormalities.
E. Peroneal neuropathy. Arising from posterior divisions of the L4–S2 ventral rami of spinal nerves, the common peroneal nerve descends into the leg as the lateral division of the sciatic nerve. At the level of the popliteal fossa, it branches from the sciatic nerve and moves toward the lower lateral portion of the popliteal fossa. Two cutaneous sensory branches arise at this point, one to the sural nerve and the other, the lateral (sural) cutaneous nerve of the calf, providing sensation to the upper lateral calf. Exiting laterally from the popliteal space, the peroneal nerve is in close juxtaposition to the fibula, winds below its head, and passes through a tendinous arch formed by the peroneus longus muscle. At its exit from the arch, the nerve divides into the superficial and deep peroneal nerves. The superficial peroneal nerve descends adjacent to the peroneus longus and brevis muscles, which it innervates, and in the distal third of the leg, it pierces the fascia. The terminal branches (medial and lateral) of the superficial peroneal nerve provide sensation to the lateral dorsal surface of the foot. The deep peroneal nerve enters the extensor compartment of the leg and with the tibial artery descends on the interosseous membrane, innervating the tibialis anterior, extensor hallucis longus, and extensor digitorum longus muscles. The terminal portion of this nerve then passes under the extensor retinaculum at the ankle, where a lateral branch innervates the extensor digitorum brevis muscle and a medial branch provides sensory innervation to the first and second toes.
1. Etiology. Most cases of peroneal neuropathy are caused by external compression (anesthesia and casts), trauma (blunt injury, arthroscopic knee surgery, and fractures) and less frequently by other etiologies (e.g., tumor, constriction by adjacent structures, involvement in systemic disease, and traction injuries from severe ankle strain).
2. Clinical manifestations.
a. History. Most patients present with foot-drop, and sensory disorders are usually minimal or of no concern. Less prominent degrees of weakness or only affecting intrinsic foot muscles may not elicit alarm in the patient. On review of the history, careful attention needs to be paid to possible episodes of trauma, compression, or unusual sustained postures that may have preceded the problem (e.g., squatting and kneeling).
b. Physical examination.
(1) Neurologic. The characteristic presentation is foot-drop, and with complete involvement of the common peroneal nerve, paralysis of ankle dorsiflexion, ankle eversion, and toe extension (dorsiflexion). Sensory loss occurs over the anterolateral lower leg and the dorsum of the foot and toes.
(2) General. Palpation in the popliteal fossa and along the fibular head may elicit signs of tenderness or discovery of a mass and define the site of involvement and possible etiology. Examination of the dorsum of the ankle and distal lateral leg, where the terminal branch of the deep peroneal nerve emerges, may reveal similar signs and suggest a distal injury. The most common sites at which focal pathologic processes can affect the nerve or its branches include the fibular head and its proximal neck, the outer compartment of the leg, and the superior and inferior extensor retinaculum at the ankle, beneath which branches of the peroneal nerve pass. However, the peroneal nerve serves as a reminder that, in cases of focal compression, there can be variable fascicular involvement. Motor impairment of only the deep or superficial component, sensory dysfunction only, or various combinations may be the result of nerve compression at the fibular head.
3. Differential diagnosis. The primary differential diagnoses are other causes of foot-drop: involvement of the L5 root, the lumbosacral trunk, and the lateral division of the sciatic nerve.
4. Evaluation. The extent of evaluation depends on the history. In cases in which an identifiable episode of compression is present, supportive care, after elimination of continuing compression, is often all that is needed. When disruption (laceration) of the nerve is suggested, the onset of the problem is insidious, or physical findings are inconclusive (incomplete common peroneal neuropathy), further evaluation is indicated.
a. Electrodiagnostic. NSS assist in identification of the site of involvement, extent of axonal injury and prognosis by comparison to the asymptomatic leg, but could identify bilateral, although asymmetric, nerve involvement suggesting an underlying generalized nerve disorder (polyneuropathy). EMG helps to further define the extent of axonal injury or evidence of etiology of the patient’s symptoms, if abnormalities are found in other L4–5 innervated muscles or paraspinal muscles.
b. Imaging. Although X-ray studies can be useful when joint trauma or a mass is detectable at examination, CT and MRI are more useful in defining lesions of the nerve and delineating the relationship of adjacent structures.
F. Tibial neuropathy. Ventral rami from the L5 to S2 spinal nerves contribute to the tibial nerve, which descends into the extremity as part of the medial trunk of the sciatic nerve. At the distal portion of the extremity, the sciatic nerve bifurcates into both the tibial and peroneal nerves. Entering the calf, it descends to the depth of the gastrocnemius, which it innervates, and provides innervation to the soleus, tibialis posterior, flexor digitorum, and hallucis longus muscles. At the level of the ankle, it divides into its terminal branches (plantar nerves), which innervate all intrinsic foot flexor muscles as well as providing sensation to the sole.
1. Etiology. Tibial neuropathy is infrequent partly because of the deep anatomic location of the nerve but, severe ankle injuries can cause proximal or distal tibial nerve injuries through traction on the nerve. Major knee trauma is surprisingly an infrequent cause of severe tibial nerve injury.
2. Clinical manifestations.
a. History. Sensory loss is evident along the side of the foot and extends proximally if the contribution of the tibial nerve to the sural nerve is also involved. Weakness may not be noticed unless ankle plantar flexion is involved.
b. Physical examination.
(1) Neurologic. Sensory loss is present along the sole of the foot. Weakness may be limited to intrinsic toe flexor muscles, or, with more proximal muscle involvement, weakness of ankle dorsiflexion and inversion.
(2) General. Careful palpation over the course of the nerve, especially within the popliteal space, should be performed. The finding of a mass or precipitation of paresthesia or pain helps in localization and may suggest the cause as tumors involving the tibial nerve (or any nerve) may increase their sensitivity to such maneuvers.
3. Differential diagnosis. Because of its infrequent occurrence, any suspicion of tibial neuropathy should prompt a search for a more proximal lesion. Radiculopathy, plexopathy, or sciatic neuropathy can manifest clinically as an “isolated” tibial neuropathy. Careful examination of more proximal muscles, reflexes, as well as the sensory examination can help identify or suggest those conditions as more appropriate diagnoses.
4. Evaluation.
a. EDS play a crucial role in identifying as well as excluding a tibial neuropathy by identifying involvement of other nerves on NSS or EMG evidence of muscle involvement other than those innervated by the tibial nerve. At times, plantar nerve involvement rather than a more proximal tibial lesion is the cause of the observed sensory or motor deficits.
b. Imaging. Identification of a mass or point of tenderness in cases of unclear causation may necessitate MRI to identify the anatomic structure of the nerve and its relations to adjacent structures.
G. Medial and lateral plantar neuropathy. At the level of the ankle, the terminal portion of the tibial nerve is medial to the Achilles tendon. As it descends, it passes under the flexor retinaculum, which composes the roof of the tarsal tunnel. Within the tunnel, the tibial nerve divides into medial and lateral plantar nerves, which descend toward the foot, and a calcaneal or sensory branch, which provides sensation over the heel. Both plantar nerves then cross under the abductor hallucis muscle (which the medial plantar nerve innervates) and go on to innervate all the muscles of the foot as well as providing sensation to the sole and the toes (the medial nerve supplies the medial and the lateral supplies the lateral portion) through their distal divisions, which give rise to the digital nerves. Muscles innervated by the medial plantar nerve include the flexor hallucis brevis and digitorum brevis. The lateral plantar nerve innervates the interossei, the flexor and abductor digiti minimi, and the adductor hallucis.
1. Etiology. The proximity of the plantar nerves to osseous and fibrous structures predisposes them to injury or compression. At the level of the tarsal tunnel, external compression and ankle injury are the most frequent etiologic factors. A multitude of other less frequent structural abnormalities (synovial or joint changes and mass lesions) can also lead to nerve injury. Within the foot itself, the medial and lateral plantar nerves are susceptible to the effects of trauma or fracture of the foot bones.
2. Clinical manifestations.
a. History. The first recognition of disorder occurs when sensory impairment develops, because foot pain or discomfort more frequently has an orthopedic origin. Sensory loss can be present in the sole or heel and at times can be precipitated by specific foot positions. Weakness of foot muscles usually produces no significant symptoms.
b. Physical examination.
(1) Neurologic. Sensory loss in the distribution of the plantar nerves or their distal divisions (digital nerves) should be sought. If foot involvement is asymmetric, changes in foot muscle bulk can be appreciated, as can weakness, although usually only toe flexion can be reliably evaluated clinically.
(2) General. Careful examination of the course of the nerve at the ankle and attempts to elicit a Tinel’s sign by means of light percussion over its course help confirm the presence and location of plantar nerve involvement. Joint changes, deformity, or swelling can also help to suggest a site of nerve involvement.
3. Differential diagnosis. One needs to consider a more proximal nerve (tibial) or root (S1) lesions that can cause foot pain or paresthesia. Motor and reflex changes should aid in this distinction. Polyneuropathy can be considered when bilaterality, depressed reflexes, and sensory involvement outside of the plantar sensory nerve distribution are found.
4. Evaluation.
a. EDS are helpful in demonstrating findings consistent with nerve entrapment (tarsal tunnel) of the medial or lateral plantar as well as calcaneal nerves. Because of the technical difficulty of such recordings, further study of asymptomatic or contralateral nerves clarifies the findings. An EMG is needed to exclude more proximal disorders (tibial neuropathy, sciatic neuropathy, or radiculopathy).
b. Imaging. Studies of possible sites of involvement (ankle) are not usually indicated. However, in cases of marked discomfort or disability, such studies may identify underlying orthopedic abnormalities and guide treatment.
H. Iliohypogastric, ilioinguinal, and genitofemoral neuropathy. The iliohypogastric, ilioinguinal, and genitofemoral nerves are described as a group because of the similarity of their origins, sensory innervation, and causes of dysfunction. These nerves arise from the L1 spinal roots (the genitofemoral nerve also has an L2 root contribution) and first pass through and then close to the psoas muscle in their intraabdominal course. The iliohypogastric nerve emerges above the iliac crest and supplies sensation to an area of skin over the lateral upper buttock and another area over the pubis or symphysis. The ilioinguinal nerve enters the inguinal canal at its lateral border and supplies the area above the inguinal ligament and the base of the genitalia. Both the iliohypogastric and ilioinguinal nerves also supply the muscles of the lower abdominal area. Clinically evident weakness of the lower abdominal musculature can occur, but perhaps more common with loss of function of both of those nerves. After it emerges from the psoas muscle, the genitofemoral nerve is retroperitoneal and descends to the inguinal ligament while resting on the surface of the psoas muscle. It supplies sensation to a small area over the proximal genitalia and anterior proximal thigh.
1. Etiology. Because of the location and course of these nerves, neuropathy usually results from surgical procedures, especially inguinal herniorrhaphy. The development of neuralgia is not infrequent after injuries to these nerves.
2. Clinical manifestations.
a. History. Patients have varying sensory problems, including numbness, paresthesia, or pain within the ipsilateral inguinal and perineal areas. If the cause is related to surgery, these difficulties may be evident immediately after the operation or may not become evident for several weeks.
b. Physical examination.
(1) Neurologic. Iliohypogastric neuropathies are infrequent, but result in sensory loss over the lateral upper buttock and suprapubic area. Ilioinguinal impairment results in sensory loss over the inguinal area and the base of the genitalia but typically resolves or results in minimal disability. In other cases, pain may appear both here and in the inferior abdomen and upper thigh and be worsened or precipitated by changes in leg position. Genitofemoral neuropathy usually accompanies inguinal nerve involvement because of the anatomic proximity of the genitofemoral and inguinal nerves. Symptoms and precipitating factors are similar as well, but sensory problems can extend into the medial and proximal areas of the genitalia.
(2) General. In ilioinguinal and genitofemoral neuropathy, areas of tenderness that often conform to the site of injury may be found in the inguinal region.
3. Differential diagnosis. In these cases, nerve involvement predominantly causes sensory impairment, and the differential diagnosis is directed at detecting other causes of sensory impairment outside the typical boundaries of these nerves, including abnormalities in the medial thigh (obturator nerve), anterior thigh (femoral nerve), and lateral thigh (lateral femoral cutaneous nerve), as well as dermatomal involvement caused by T12 or L1 radiculopathy. Because of these overlapping sensory innervations, the presence of motor deficits or reflex changes provides the strongest clue to the presence of one of these other disorders. Back pain, which can suggest a radiculopathy, or the absence of a previous operative procedure, which is the usual cause of these neuropathies, suggests another etiologic factor.
4. Evaluation.
a. EDS play little role in the identification of these neuropathies. However, they become indispensable in helping to identify more proximal lesions (plexus or root) or other forms of neuropathy (femoral) that may clinically resemble iliohypogastric, ilioinguinal, or genitofemoral neuropathy in regard to sensory deficits.
b. Imaging is performed if there is suspicion of radiculopathy or if a retroperitoneal, intraabdominal, or pelvic lesion is under consideration.
I. Miscellaneous neuropathy. This grouping and brief review is based on their infrequent occurrence or isolated involvement.
1. Superior gluteal neuropathy. Arising from, and receiving its contributions from the L4 to S1 components of the sacral plexus, the nerve passes through the sciatic notch above the piriformis muscle and innervates the gluteus medius and minimus muscles. Its isolated involvement is unusual and is most often the result of injury by a misplaced injection.
2. Inferior gluteal neuropathy. Arises from the L5 to S2 divisions of the sacral plexus and exits through the sciatic notch. Its proximity to the sciatic, pudendal, and posterior cutaneous nerves of the extremity results in concomitant injury to these nerves.
3. Posterior cutaneous nerve of the thigh. Arising from the S1 to S3 components of the sacral plexus, it descends through the sciatic notch close to the sciatic nerve and supplies sensation to the posterior portion of the buttock and thigh. At times it is susceptible to local compression, but its isolated involvement is unusual.
4. Pudendal neuropathy. Derived from the S2 to S4 components of the sacral plexus, it passes through the sciatic notch and descends toward the perineum. Supplying muscles of the perineum, including the anal sphincter and erectile tissue, it also provides sensory innervation to the perineum. Its deep location protects it, but prolonged compression can cause dysfunction or stretch injuries related to prolonged labor and manifested by fecal and urinary incontinence.
III. REFERRALS
A. Indications for and purposes of neurologic consultation.
1. Site of involvement unclear from examination or history, discrepancy between severity of the underlying disease and the neuropathy.
2. Progressive deterioration despite appropriate treatment.
3. Problem precipitated by trauma or injury.
4. Prior to more expensive or invasive evaluation (MRI or nerve biopsy) or more aggressive intervention (surgery).
5. Confirmation of diagnosis, etiologic factor, or treatment plan.
B. EMG and NSS evaluation.
1. Basic tenets of such testing.
a. Extension of the clinical examination and not a replacement.
b. Intended to clarify the clinical question to be answered (e.g., carpal tunnel syndrome or C6 radiculopathy).
c. Sensitivity and specificity vary according to the etiologic factor and process in question.
2. Role in the evaluation of neuropathy.
a. Confirmation of diagnosis or characterization, localization, and quantification of a disease process.
b. Defining prognosis.
c. Detection of subclinical disease.
d. Planning treatment or determining need for further evaluation or consultation.
![]()
Recommended Readings
Brazis PW, Masdeu JC, Biller J. Localization in Clinical Neurology. 6th ed. New York, NY: Lippincott Williams & Wilkins; 2011.
Donovan A, Rosenberg ZS, Cavalcanti CF. MR imaging of entrapment neuropathies of the lower extremity. Part 2. The knee, leg, ankle and foot. Radiographics. 2010;30:1001–1019.
O’Brien M. Aids to the Examination of the Peripheral Nervous System. 5th ed. New York, NY: W.B. Saunders; 2010.
Petchprapa CN, Rosenberg ZS, Sconfienza LM, et al. MR imaging of entrapment neuropathies of the lower extremity. Part 1. The pelvis and hip. Radiographics. 2010;30:983–1000.
Preston DC, Shapiro B. Electromyography and Neuromuscular Disorders: Clinical-Electrophysiologic Correlations. 2nd ed. Philadelphia, PA: Butterworth-Heineman; 2005.
Stewart JD. Focal Peripheral Neuropathies. 4th ed. West Vancouver: JBL Publishing; 2010.