I. DEFINITIONS
A. An epileptic seizure is a transient and reversible alteration of behavior caused by a paroxysmal, abnormal, and excessive neuronal discharge.
B. Epilepsy usually is defined as two or more recurring seizures not directly provoked by intracranial infection, drug withdrawal, acute metabolic changes, or fever.
II. CLASSIFICATIONS
There are two classifications—one of seizure types and one of epilepsy or epileptic syndromes. Accurate diagnosis of the type of epileptic seizure and the categorization of epilepsy (or epileptic syndrome) for each patient are essential for proper selection of antiepileptic drug (AED) therapy and for prognosis.
A. Classification of epileptic seizures is based on the patient’s behavior during seizures and on the associated EEG characteristics. Epileptic seizures are classified into two main types—partial and generalized.
1. Partial (focal) seizures arise at specific loci in the cerebral cortex and are associated with focal interictal and ictal EEG changes. Clinically, a partial seizure can range in intensity from a disorder of sensation without loss of consciousness to a generalized convulsion.
a. Simple partial seizures. Consciousness remains intact. Such seizures can be motor seizures (focal motor twitching or Jacksonian seizures), sensory seizures (numbness or tingling involving parts of the body), autonomic seizures, or seizures with psychic symptoms.
b. Complex partial seizures. Consciousness is impaired during complex partial seizures. Previously, these seizures were called psychomotor seizures. They constitute the most common type of seizure in adults. Approximately 85% have an epileptogenic focus in the temporal lobe, whereas the remaining 15% are of extratemporal origin, usually frontal.
c. Secondarily generalized (tonic–clonic) seizures. During any focal seizure (simple or complex partial), the epileptic excitation can spread widely to the entire brain, resulting in a generalized tonic–clonic convulsion.
2. Generalized seizures are characterized by generalized involvement of the brain from the outset and have no consistent focal areas of ictal onset. There are many subtypes.
a. Absence seizures were formerly known as petit mal seizures. The dominant feature is a brief loss of consciousness with no or minimal motor manifestations (e.g., twitching of the eyelids). During the seizure, EEG shows 3-Hz generalized spike–wave discharges.
b. Myoclonic seizures are brief jerks involving part of the body or the entire body.
c. Clonic seizures are rhythmic twitching of the body.
d. Tonic seizures are brief attacks of stiffness in part of the body or the entire body.
e. Atonic seizures are losses of posture with resultant drop attacks.
f. Tonic–clonic seizures are generalized convulsions or grand mal seizures. It is important to emphasize that some tonic–clonic (grand mal) seizures are generalized from the outset and some are secondarily generalized (they start as focal seizures and then become generalized). The second type is the most common among adults. The presence of an aura, focal manifestations during the seizure, and postictal focal deficits favor a secondarily generalized tonic–clonic seizure.
Confusion can arise in differentiating absence seizures and complex partial seizures. Both can present with a brief loss of awareness or altered responsiveness, and in both there may be automatic activities of various kinds. Diagnosis is aided by EEG findings (generalized spike–wave discharges in absence seizures and focal epileptiform abnormalities in complex partial seizures). Correct diagnosis is critical for instituting proper AED therapy.
B. Classification of epilepsy or epileptic syndromes. Classifying the seizure type, although useful, is of limited value because seizures usually appear as part of a cluster of other symptoms and signs that include etiologic factor, site of seizure onset, age, precipitating factors, response to medication, and prognosis. Hence, the ultimate goal is to diagnose epilepsy or an epileptic syndrome. This is very important. It helps to choose the appropriate AEDs to control seizures and to avoid using an AED, which may not only be ineffective but may even exacerbate seizures.
1. Localization-related (focal or partial) epilepsy or epileptic syndromes are disorders in which a localized origin of the seizures can be established. The patient has focal or secondarily generalized tonic–clonic seizures. EEG shows focal epileptiform discharges overlying the epileptogenic focus.
a. Most localization-related forms of epilepsy are acquired or symptomatic. Temporal lobe epilepsy is the common localization-related epilepsy among adults, and it is often associated with mesial temporal sclerosis on the MRI scan.
b. There are age-related idiopathic or primary localization-related epileptic syndromes. The best known is benign rolandic epilepsy of childhood.
2. Generalized epilepsy or epileptic syndromes are disorders that involve one or more types of generalized seizures. EEG shows generalized epileptiform abnormalities.
a. Primary (idiopathic) generalized epilepsy (PGE) is characterized by generalized seizures without any identifiable etiologic factor. EEG shows generalized spike–wave or polyspike–wave discharges with a normal background activity. Genetic factors predominate in these forms of epilepsy. Common syndromes include childhood absence epilepsy, juvenile myoclonic epilepsy, juvenile absence epilepsy, and tonic–clonic seizures occurring often in the early morning (“awakening” grand mal).
b. Secondarily (symptomatic) generalized epilepsy is characterized by various types of generalized seizures resulting from acquired cerebral diseases (e.g., seizures secondary to ischemic–hypoxic encephalopathy or following severe cerebral trauma or intracranial infection) or from inborn errors of metabolism (e.g., lipidosis and progressive myoclonus epilepsy). EEG shows generalized, irregular spike–wave, 2.5 Hz in frequency with an abnormally slow background activity. Patients usually have varying degrees of cognitive and neurologic deficits, and the seizures are often drug resistant. Within this category are two commonly recognized age-related syndromes—West’s syndrome (infancy) and Lennox–Gastaut’s syndrome (childhood).
III. EVALUATION
It is essential to establish that the spells or episodes are indeed epileptic seizures. Nonepileptic physiologic disorders that result in transient, reversible alterations of behavior or function, such as syncope, migraine, breath-holding spells, anxiety episodes, transient ischemic attacks, hypoglycemic episodes, and narcoleptic–cataplectic attacks, must be differentiated from epileptic seizures. Moreover, there are nonepileptic psychogenic seizures or pseudoseizures that are conversion reactions characterized by episodes of motor activity and loss of consciousness but not associated with ictal EEG patterns and without an underlying physiologic basis.
A. A history of the episodes, obtained not only from the patient but also from one or more observers, is perhaps the most essential element in making the diagnosis of epileptic seizures and differentiating them from nonepileptic disorders. The history can aid in ascertaining the type of epileptic seizures.
B. Physical and neurologic examinations can help detect the underlying cause of the brain disorder responsible for the epilepsy by uncovering evidence of a focal cerebral lesion or another organic disorder, such as tuberous sclerosis or neurofibromatosis.
C. Neuroimaging. Although CT of the head with and without contrast enhancement is performed on most patients believed to have epilepsy, MRI of the head is the imaging procedure of choice. MRI is particularly sensitive in detecting hamartoma, cavernous malformation, and low-grade glioma and in providing evidence of mesial temporal sclerosis in patients with temporal lobe epilepsy.
D. EEG is the most informative test for confirmation of the diagnosis of epilepsy and proper classification of the seizure type and even the epileptic syndrome. It also aids in initiation, selection, and discontinuation of antiepileptic therapy (see Chapter 33).
Not all patients with epilepsy have interictal epileptiform abnormalities; approximately 50% have such abnormalities in a routine awake-and-asleep EEG study that includes hyperventilation and intermittent photic stimulation. The yield increases with repeated EEG studies with sleep deprivation and extra recording electrodes. On the other hand, 1% to 2% of healthy persons without clinical seizures have epileptiform abnormalities in EEG studies. Hence, an interictal EEG alone can neither prove nor exclude a diagnosis of epilepsy. Similarly, the presence of interictal epileptiform EEG abnormalities does not automatically warrant AED therapy, and the absence of such abnormalities is not sufficient grounds for discontinuing AED treatment.
E. Intensive video EEG monitoring. Patients with drug-resistant epilepsy or poorly characterized episodes may need intensive monitoring that consists of simultaneous monitoring of the patient’s behavior and EEG to provide detailed clinical and EEG correlation of episodic events. This is an expensive and time-consuming technique and thus is left to the discretion of a consulting neurologist specialized in epilepsy. Only 5% to 10% of patients believed to have epilepsy need this technique to characterize and classify the epileptic episodes. Video EEG is mandatory in the presurgical evaluation of patients to document epileptic seizures prior to surgical resection to treat epilepsy, and even before placing a patient on vagal nerve stimulation (VNS). It is most helpful in patients who have frequent episodes that are suspected to be of the nonepileptic type. These episodes are not accompanied by the characteristic ictal pattern in the simultaneously recorded EEG.
IV. GENERAL PRINCIPLES IN TREATING PATIENTS WITH EPILEPSY
A. AED therapy should be initiated only when the diagnosis of epileptic seizures is well-established. If the patient’s episodes are yet to be clearly defined and there is reasonable doubt of their being epileptic in nature, it is prudent to wait until the diagnosis of epilepsy can be confirmed.
B. First seizure. AED therapy usually is not initiated after the first tonic–clonic seizure and postponed until a second seizure occurs when the diagnosis of recurrent seizures or epilepsy is made. The incidence of recurrence is 25% to 65% after the first and over 75% after the second seizure. The first seizure can be an isolated episode and not necessarily the onset of epilepsy. This is particularly true if a single tonic–clonic seizure was related to sleep deprivation, physical or mental stress, drug or alcohol withdrawal, or use of prescribed (e.g., Welbutrin) or recreational drugs (e.g., cocaine). In general, 50% of patients have recurrence over a 3-year follow-up period after the first tonic–clonic seizure. The incidence is <25% among subjects with low-risk factors to 65% or more for those with two or more of the following risk factors: strong family history of seizures in siblings, history of febrile convulsions, focal-onset seizure, postictal paralysis, abnormal findings at cognitive and neurologic examinations, evidence of a structural cerebral lesion at neuroimaging, and the presence of epileptiform abnormalities at EEG. Patients with two or more of these risk factors therefore may need prompt initiation of therapy even after the first tonic–clonic seizure.
C. Monotherapy is preferable to the use of several drugs because it has fewer toxic side effects, less likelihood of drug interactions, and better compliance. The chosen AED (Table 39.1) should be slowly increased until seizures are controlled or until clinical signs of toxicity develop. If seizures are not adequately controlled at the maximum tolerable dosage, a second AED is slowly introduced. After the second drug attains therapeutic levels, the first drug is gradually withdrawn. Monotherapy adequately controls new-onset epilepsy in about two-thirds of the patients.
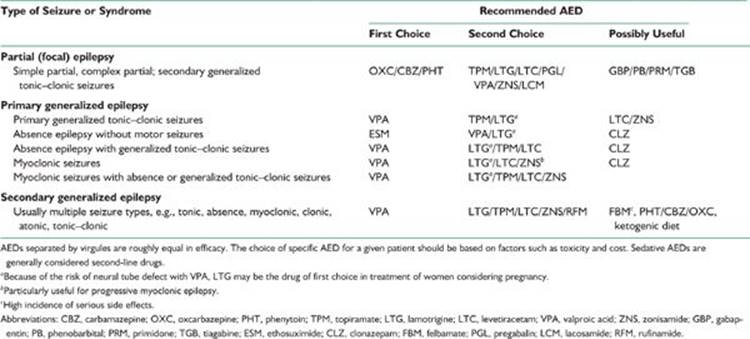
TABLE 39.1 AEDs of Choice for Specific Type of Seizures and Epilepsy Syndromes
D. Polytherapy with a combination of two AEDs (usually one traditional and one newer AED) becomes necessary only if monotherapy with two or more first-line AEDs has been unsuccessful. When using two AEDs, select those with different mechanism of action. Avoid using more than two AEDs simultaneously. If a combination of two AEDs in the treatment of a compliant patient with blood levels in the therapeutic range fails to provide adequate control of epileptic seizures, referral to an epileptologist or epilepsy center is indicated for further evaluation and management. After two appropriate AEDs fail to control seizures, monotherapy with a third AED or polytherapy is successful in only 4% of the patients.
E. AEDs with sedative or hypnotic side effects need to be avoided unless first-choice AED does not work. Drugs with these effects include phenobarbital, primidone, and clonazepam. Often a patient undergoing polytherapy that includes one of the aforementioned sedative AEDs is best served by very gradual withdrawal of the sedative AED while the dosage of the other AED is maximized. Discontinuation of sedative–hypnotic AEDs is followed not only by a reduction in side effects but also by better control of seizures in many instances.
F. Simplify drug schedules. Most AEDs have long elimination half-lives (see Tables 39.3 and 39.5), and thus can be prescribed in a single daily dose or two divided daily doses. Exceptions include the traditional AEDs, such as valproic acid and carbamazepine (extended release valproate and sustained release carbamazepine are now available), and the new AEDs such as gabapentin and tiagabine, which have to be given in two or three divided daily doses. With multiple AEDs, half-lives of some AEDs are shorter than when the same drug is given in monotherapy. Thus, larger doses and multiple dosing are required.
G. Compliance must be emphasized. Medication is best taken at the time of meals for easy remembrance. For most AEDs, an occasional missed dose can be made up by taking an additional dose within the same 24-hour period. It is also convenient for the patient to put the medication in a plastic pillbox with divided compartments and to ensure at bedtime that the entire day’s medication has been taken.
H. Advise the patient to maintain a seizure diary. Such a diary provides an accurate record of the frequency of seizures and assists in evaluating the effectiveness of the therapy.
I. Emphasize to the patient the need for constant medical follow-up care. Once AED therapy is well-established and the seizures have been brought under satisfactory control, the patient should be examined every 6 to 12 months. During the follow-up visits, evaluate the patient for evidence of drug toxicity or development of a progressive neurologic disorder. CBC, liver function tests, and serum electrolytes may need to be performed every 6 to 12 months to detect untoward effects of AEDs on the bone marrow and liver. However, routine blood testing at periodic intervals is a controversial issue because serious side effects are rare and, when they occur, they do so over a short period to be detected by periodic monitoring.
J. A patient who achieves good control with drug therapy may have a “breakthrough” seizure during periods of physical or mental stress, sleep deprivation, or infection. Appropriate management of such precipitants rather than increases in dose or changes in the AED is indicated.
K. Generic substitution for brand-name AEDs can reduce the cost of medication, but the bioavailabilities of generic and proprietary AEDs are not the same. Generic preparations are required by the U.S. Food and Drug Administration (FDA) to provide bioavailabilities within ±20% of those of the corresponding proprietary formulations, but some patients may be sufficiently sensitive to these fluctuations that replacing one with the other leads to either loss of seizure control or signs of neurotoxicity. This problem applies primarily to phenytoin and carbamazepine. The proprietary phenytoin (Dilantin; Pfizer, New York, NY, USA) is more slowly absorbed than is generic phenytoin, so blood levels are maintained with less fluctuation, and no more than one or two daily doses are required. Similarly, brand-name carbamazepine (Tegretol; Novartis, East Hanover, NJ, USA) is absorbed more slowly than is the generic formulation. It is probably prudent to continue name brand AED if the patient is seizure free on that formulation. If changed to generic AED the patient needs to be warned of the possibility of break-through seizures or drug toxicity. The patient should avoid switching between formulations of AEDs. When a generic AED is used, the formulation by the same manufacturer should be refilled.
L. Therapeutic drug levels are rough guides to the ranges that in most patients provide best seizure control while avoiding dose-related side effects. The blood should be drawn preferably before the morning dose of AEDs so as to obtain the lowest (trough) levels. The blood levels are not to be followed rigidly for a given patient. Some patients may attain complete seizure control at low “therapeutic” levels, and increasing the dose to attain idealized levels is not indicated. On the other hand, there are patients who need higher than “therapeutic” levels for control of their seizures and tolerate such levels without significant untoward side effects. In such patients, it is fully justified to maintain phenytoin level as high as 20 to 30 µg per ml, valproic acid level as high as 100 to 150 µg per ml, and carbamazepine level as high as 12 to 15 µg per ml. Anticonvulsant blood levels are indicated under the following circumstances:
1. To determine the baseline plasma dose level.
2. When the patient is believed to be noncompliant.
3. When the patient does not respond adequately to the usual dosage of an AED.
4. When symptoms and signs of clinical toxicity are suspected.
5. When there is a question of drug interaction.
6. To establish the correct dosage for a pregnant patient or a patient with diseases affecting the pharmacokinetics of the AEDs (hepatic, renal, or gastrointestinal disorders).
Total serum levels of AEDs usually are obtained. When metabolism of AEDs may be altered or serum protein levels are likely to be low (e.g., hepatic or renal disorders and pregnancy), free levels of highly protein-bound AEDs may become necessary.
M. Emphasize the need to regularize the time and duration of sleep, because sleep deprivation tends to potentiate seizures.
N. Concomitant use of other drugs. Alcohol in any form is best avoided or used in small amounts (e.g., one drink) because of possible interactions with most AEDs. Be aware of drugs that lower the seizure threshold (e.g., tricyclic antidepressants, Welbutrin, and phenothiazines) or those that can cause drug interactions (increasing or decreasing the levels of AEDs); they should be used with caution. Some AEDs affect the elimination kinetics of many drugs metabolized in the liver (e.g., birth control pills, corticosteroids, anticoagulants, and cyclosporine), necessitating proper dose adjustment of these comedications.
O. Encourage the patient to make the adjustments necessary for leading a normal life as much as possible. Moderate exercise does not affect seizure frequency. Encourage a regular exercise program. Participation in highly competitive sports increases the risk of physical injury. Individualize instructions to the patient by considering the risk of a particular sport against the patient’s needs. Swimming may be permitted under supervision for a patient with good control of seizures. Bathing in a bathtub is to be avoided; taking a shower is recommended instead.
P. Most adults who have epilepsy are able to maintain competitive employment and should be encouraged to do so. This improves their self-esteem and their acceptance in the mainstream of society. There are, however, some realistic limitations on the types of work a patient with epilepsy can be permitted to do. Certain occupations, such as working with heavy machines, working above ground level, working close to water or fire, driving trucks or buses, and flying planes, may be off limits for reasons of personal and public safety. There are still scores of jobs such as secretary, lawyer, physician, accountant, and stockbroker that are acceptable.
Q. Family members or caregivers should be educated regarding proper care of the patient when a seizure occurs. During a grand mal seizure, the patient should be helped to lie on the ground, a bed, or a couch and should be turned on one side to avoid aspiration. An object such as a spoon or a finger should never be thrust into the patient’s mouth. It is a myth that the tongue can be swallowed during a seizure. Pushing a hard object into the mouth often results in broken teeth. The patient must be closely watched and the sequence of events observed during the seizure, which can help determine the type of seizure.
R. For a patient with a known history of seizures, an isolated self-limiting seizure does not constitute a need to call for an ambulance and have the patient rushed to an emergency department. However, if the seizure lasts longer than 5 minutes or if the patient has repeated seizures without regaining consciousness between them, prompt transfer to a nearby hospital becomes essential. Prompt medical attention must also be sought if patient had a fall and sustained bodily injury.
S. Driving. Most states have laws denying driving privileges to patients with uncontrolled epilepsy but permit driving once the seizures have been brought under control with AEDs. In a few states, doctors are required to report cases of epilepsy. The period of time that the patient must remain seizure-free before being permitted to drive varies from 3 months to 2 years, depending on the state. Rare patients who have only nocturnal seizures or who have only simple partial seizures (no loss of consciousness) may be exempted from driving restrictions. Reinstitution of driving privileges may require reapplication, a letter from the treating physician, or a determination made by a state-appointed board. Some states require the treating physician to certify at regular intervals that the patient has continued to remain seizure-free before reissuing the driving permit. Because the laws regarding driving vary widely among different states and are frequently changing, physicians are best advised to obtain their current state registration.
In general, patients with frequent seizures with altered consciousness must be advised to refrain from driving until seizures can be satisfactorily controlled. That the patient has been properly advised must be documented in the patient’s record.
There is no consensus as to how long the patient should be advised not to drive after having a breakthrough seizure after a long seizure-free period. If such a seizure follows a known precipitant such as infection, mental or physical stress, prolonged sleep deprivation, or poor compliance, observation for at least 3 to 6 months is required before driving is again permitted.
V. SELECTION OF AED
Table 39.1 lists AEDs effective for managing various forms of epilepsy and epileptic syndromes.
A. Symptomatic partial (localization-related) epilepsy. Several well-designed studies have shown that for simple partial, complex partial, and secondarily generalized tonic–clonic seizures, several AEDs, including phenytoin, carbamazepine, valproate, phenobarbital, and primidone, have similar antiepileptic efficacy but differ greatly in toxicity. Primidone and phenobarbital are more often associated with neurotoxicity, probably less effective against simple and complex partial seizures, hence, usually avoided. Valproate is somewhat less effective against complex partial seizures than carbamazepine. There are very small differences in overall effectiveness and basic mechanism of action between carbamazepine and phenytoin. Hence, carbamazepine and phenytoin can be considered as the first-line “traditional” AEDs.
Phenytoin is relatively inexpensive, which can be titrated rapidly in 2 to 3 days, is better tolerated in the initial period of therapy, and can be given in one to two divided daily doses. However, it has a high incidence of chronic dysmorphic side effects, such as hirsutism, coarsening of facial features, and acneiform eruptions, which makes its use rather unacceptable in women. Its nonlinear kinetics makes the dose adjustment difficult during maintenance therapy. Carbamazepine has no dysmorphic effects and hence is better accepted by adolescent and young adult female patients. Its short half-life usually necessitates using it in three or four divided doses, but sustained-release preparations are now available and given in two daily doses. It needs to be slowly titrated over 3- to 4-week period due to autoinduction. Cognitive side effects with long-term use of phenytoin or carbamazepine have been found to be equally frequent in recent studies and both require periodic blood monitoring for bone marrow and liver functions. Oxcarbazepine, which has lesser side effects, rapid titration, and no requirement for blood monitoring, is emerging as a more favored newer AED alternative to carbamazepine since its approval by the FDA for monotherapy in adults with focal epilepsy.
Most physicians now recommend using oxcarbazepine or carbamazepine be used initially as monotherapy. If one of the two is ineffective, it can be replaced with the other. If monotherapy with carbamazepine or oxcarbazepine fails to achieve satisfactory control, a combination therapy with valproic acid is tried—carbamazepine plus valproic acid, or oxcarbazepine with valproic acid. Alternatively, adjunctive therapy with newer AEDs should be strongly considered by adding levetiracetam, lamotrigine, topiramate, zonisamide or lacosamide to carbamazepine, or oxcarbazepine, based on the lesser risk of toxicity and drug interaction of these newer AEDs. Combination therapy is more likely to produce cognitive and other side effects.
Even with adequate AED therapy, only 40% to 60% of patients with symptomatic partial epilepsy, particularly those with complex partial seizures (the most common type of seizures among adults) attain full control of seizures. In one study of new onset epileptic seizures, initial monotherapy was effective in 47% of patients. Changing to a second drug controlled another 13% of patients. Use of a third AED or combined therapy with two or more AEDs controlled only 4% of additional patients. Most experts believe that, if an adequate trial with two or three AEDs (including at least one newer AED) either as monotherapy or combined therapy fails, the patient has medically refractory epilepsy. Such a patient will need a referral to a comprehensive epilepsy center for further management.
B. Primary (idiopathic) generalized epilepsy. Most patients with PGE have either absence, myoclonic, or tonic–clonic seizures, and most have more than one type of seizure, although one type may dominate. Depending on seizure type, several epileptic syndromes are identified under the heading PGE. The best example is juvenile myoclonic epilepsy, which is characterized by myoclonic seizures in the early hours after waking, but most patients also have occasional tonic–clonic seizures. Less often, even absence seizures may occur. Other syndromes include primary tonic–clonic seizures (contrasted to secondarily generalized tonic–clonic seizures, which are part of focal epilepsy), which commonly occur in the morning hours and hence are called “awakening” grand mal seizures. Absence seizures as the dominant manifestation of PGE commonly occur in childhood (childhood absence epilepsy), but in rare cases start in adolescence or early adulthood (juvenile absence epilepsy).
1. For absences, both ethosuximide and valproic acid are equal and very effective in controlling these seizures in over 80% of patients. Patients with childhood or juvenile absence epilepsy who have absence seizures only can, therefore, be treated with either ethosuximide or valproic acid. Ethosuximide is preferred because of its lesser toxicity and longer half-life allowing it to be taken in only one or two divided daily doses. But those patients who have additional myoclonic and/or tonic–clonic seizures do need valproic acid for treatment of both absence and motor seizures because ethosuximide is ineffective against motor events.
2. Valproic acid has clearly been the drug of choice for PGE where multiple seizure types are likely to be present, including the syndrome of juvenile myoclonic epilepsy, primary grand mal seizures, or combined absence–grand mal epilepsy. The advantage of valproic acid is that it is effective against all seizure types comprising PGE.
3. Appropriate AED therapy is very effective, and good seizure control is possible for as many as 80% to 90% of patients with PGE.
4. Clonazepam is an effective AED for myoclonic and absence seizures but has certain disadvantages. It has a high incidence of sedative and cognitive side effects, and patients develop a tolerance to its antiepileptic potency after several months of therapy. Furthermore, its use may start or exacerbate tonic–clonic seizures in juvenile myoclonic epilepsy. Use of clonazepam has, therefore, declined since the introduction of newer AEDs. If used for myoclonic or absence seizures, clonazepam can be added to valproic acid if the latter by itself fails to be fully effective.
5. Because of the high incidence of fetal malformations, polycystic ovarian syndrome (PCOS), and weight gain, the use of valproic acid in women with PGE needs to be restricted because recent well-designed studies have demonstrated effectiveness of the newer AEDs, several of which are broad spectrum in their efficacy.
a. Lamotrigine is effective against absences, has low teratogenicity, and is a weight neutral AED. It is a good alternative for treating absences associated with childhood and juvenile absence epilepsy. Very recent studies, however, have shown it to be somewhat less effective compared with ethosuximide or valproic acid.
b. Lamotrigine is also found to be effective in juvenile myoclonic epilepsy, although it can occasionally exacerbate myoclonic seizures.
c. Topiramate is effective in PGE manifesting with generalized tonic–clonic seizures and approved by the FDA for this indication.
d. Levetiracetam is effective in all seizure types accompanying PGE and particularly against myoclonic seizures.
e. Zonisamide is probably effective, also, against all seizure types of PGE, but has more sedative side-effects than lamotrigine and levetiracetam.
6. Although most patients with PGE can be well-controlled with appropriate AEDs, some 15% are medically refractory. Some are pseudo-refractory because they are on wrong AEDs. In recent years, sodium-channel blocking and gamma-aminobutric acid (GABA)-enhancing AEDs have been recognized to exacerbate certain seizure types in patients with PGE. Carbamazepine, oxcarbazepine, phenytoin, vigabatrin, tiagabine, gabapentin, and pregabalin commonly exacerbate absence seizures, myoclonic jerks, or both, and should not be used in the treatment of PGE. It is, therefore, critical that the patient needs to have the syndromic diagnosis of PGE established even though the precise subtype of PGE may not be identified. This will prevent iatrogenic exacerbation of seizures.
In truly refractory cases, combination therapy using Depakote with either levetiracetam, lamotrigine (but be aware of markedly decreased elimination of lamotrigine by valproic acid), topiramate, or zonisamide may be more effective. Combination of Depakote and ethosuximide may control absences more effectively than either drug alone.
C. Secondarily (symptomatic) generalized epilepsy, which is secondary to multifocal or diffuse cerebral disorders (static or progressive), occurs mostly among children and less often among adults. Patients have multiple seizure types, including atypical absence seizures, myoclonic seizures, tonic seizures, tonic–clonic seizures, and drop attacks.
In general, response to any AED is poor, only 20% to 40% of patients attaining acceptable seizure control. Such patients commonly end up being treated with polypharmacy, which not only fails to provide better seizure control than do one or two AEDs but may even exacerbate certain types of seizures (absence seizures, myoclonic seizures, and drop attacks).
Valproic acid is the AED of first choice for secondarily generalized epilepsy and may be started as monotherapy. However, most patients need an addition of newer AEDs. Of these, lamotrigine, topiramate,zonisamide,and levetirecetam have been found useful in clinical trials. Felbamate and rufinamide have been approved by FDA for the control of seizures associated with secondary generalized epilepsy of the Lennox–Gastaut’s type but felbamate has potentially serious hepatic and bone marrow toxicity. When drug combinations are prescribed, appropriate dosages should be used to avoid sedation, which tends to exacerbate minor seizures as well as precipitate statuses in such patients.
VI. TRADITIONAL AEDS
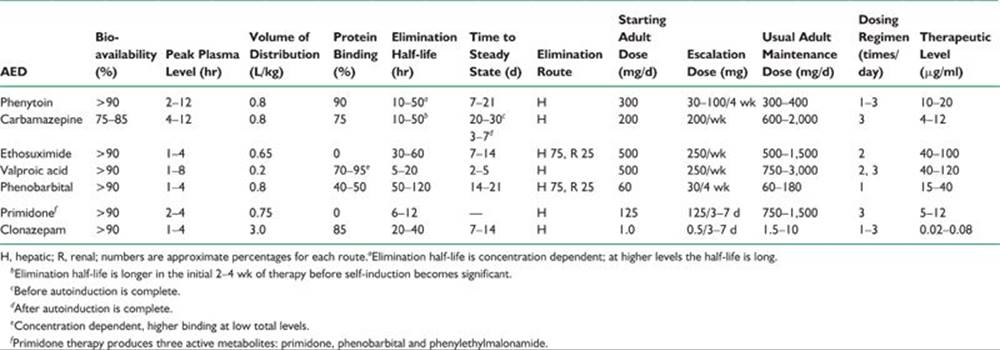
Mechanism of action, indications, and common side effects of traditional AEDs are listed in Table 39.2, and their pharmacokinetic characteristics and adult dosages in Table 39.3.
A. Efficacy. In general, the traditional AEDs have limited efficacy against epileptic seizures especially those associated with focal or partial epilepsy.
1. The first landmark VA study in 1985 compared carbamazepine, phenytoin, primidone, and phenobarbital in the treatment of partial epilepsy. All four of those AEDs were shown to be equally effective as monotherapy for secondarily generalized tonic–clonic seizures, whereas carbamazepine and phenytoin were more effective and better tolerated than primidone and phenobarbital in the treatment of complex partial seizures.
2. The second VA study in 1992 demonstrated that carbamazepine was superior to valproic acid in the treatment of complex partial seizures but both were equivalent for secondarily generalized tonic–clonic seizures.
TABLE 39.2 Mechanism of Action, Indications and Undesirable Side Effects of Traditional AEDs
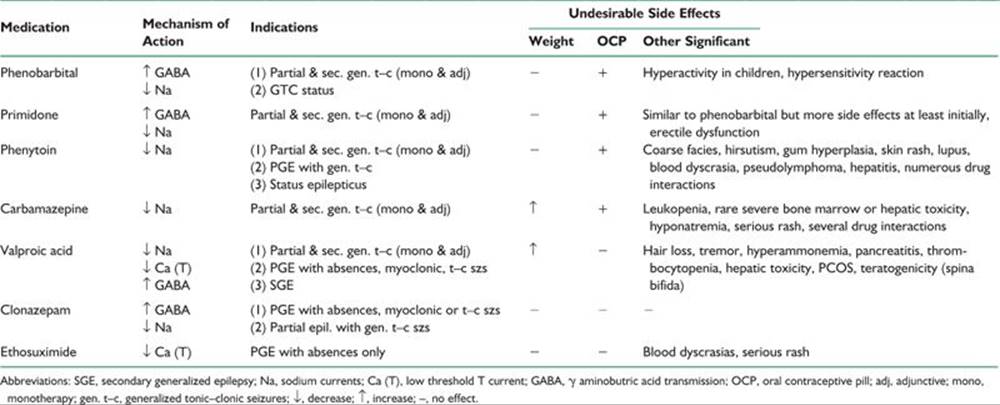
TABLE 39.3 Pharmacokinetics and Adult Dosages of Traditional AEDs
3. In both VA cooperative studies, only 25% to 48% patients were seizure-free after 12 months of therapy.
4. Ethosuximide has a narrow spectrum of efficacy, controlling only absence seizures.
5. Valproic acid is the only broad spectrum AED among the traditional drugs, being effective both in partial and generalized epilepsy.
B. Side effects.
1. All traditional AEDs are associated with neurotoxic side effects such as tiredness, somnolence, dizziness, ataxia, blurred or double vision, nystagmus, difficulty with concentration, behavioral disturbances, and cognitive dysfunction.
2. Phenobarbital, primidone, and clonazepam have higher incidence of sedative/hypnotic side effects.
3. Paradoxical hyperactivity occurs in children treated with phenobarbital or primidone.
4. Skin rashes have been reported with phenytoin, carbamazepine, ethosuximide, and other AEDs.
5. Phenytoin has several side effects such as coarsening of the facies, facial/body hirsutism in women, and gum hyperplasia. These cosmetic effects are concerning to women.
6. Valproic acid, when used in larger doses, is associated with tremor.
7. Valproic acid and carbamazepine are commonly associated with weight gain.
8. Serious reactions to traditional AEDs are reported but are uncommon. These include hepatotoxicity, bone marrow suppression (aplastic anemia and agranulocytosis), exfoliative dermatitis, and even Stevens–Johnson’s syndrome. Valproic acid is also reported to produce pancreatitis. If not detected in time, these reactions may be fatal. Hence, periodic monitoring of hepatic and hematopoietic parameters is recommended, although there is no evidence that routine testing detects those serious conditions much prior to a time when the effects can still be prevented.
Precise frequency of serious side effects from the traditional AEDs is unknown. Probably the incidence is somewhat higher than their spontaneous occurrence except in the case of hepatotoxicity associated with valproic acid. The highest risk group includes children below the age of 2 years receiving polytherapy, which includes valproic acid, the incidence being as high as 1 in 500.
Usually benign leukopenia occurs in 10% to 20% patients, especially on carbamazepine. A total white cell count of 2,000 per cmm and an absolute granulocyte count of 1,000 per cmm are well-tolerated.
Valproic acid is reported to produce thrombocytopenia, interference in the platelet aggregation, and an increased tendency toward bleeding. However, platelet counts above 60,000 per cmm are acceptable.
9. Because of its inhibition of cytochrome P-450 system, valproic acid in women is associated with abnormal metabolism of gonadal and adrenal sex hormones, often resulting in PCOS. It is characterized by infertility, hirsutism, obesity, increased serum androgen levels, frequent anovulatory cycles, and other menstrual abnormalities. The incidence of PCOS is 10% to 20% in women with epilepsy compared with 5% to 6% of women in the general population. The incidence of this syndrome is highest in women with PGE treated with valproic acid. How much each of the two factors, type of epilepsy and valproic acid, play a role in causing PCOS is still being debated.
10. Teratogenic effects occur in 5% to 10% pregnancies with the use of many traditional AEDs, highest incidence with valproic acid (see below).
11. Almost all of the traditional AEDs are associated with bone loss, even in young adults, and affect both genders following their use of 1 to 2 years. The bone loss is more severe in elderly patients, with polytherapy and with prolonged use. Possible mechanisms include hepatic induction of cytochrome P-450 enzymes (phenobarbital, primidone, phenytoin, and carbamazepine) leading to increased catabolism of vitamin D, secondary hyperparathyroidism, and increased bone turnover. Valproic acid, a noninducer, does produce bone loss by increased ostoclastic activity.
Reduced bone density increases the risk, two to six times, for fractures, particularly of the hip and spine. Bone loss is best detected by dual-energy X-ray absorptometry and expressed as a T-score. Osteopenia is defined by a T-score of 21 to 22.5 and osteoporosis by a score 22.5.
Optimal management for bone loss is exercise, nutrition, and calcium and vitamin D supplementation. If osteoporosis is detected, treat with biphosphonate and other measures.
C. Pharmacokinetics. There are several undesirable pharmacokinetic issues related to the traditional AEDs.
1. Most of them are eliminated primarily through hepatic metabolism, through microsomal cytochrome P-450 enzymes; hence, there are several drug interactions. These enzyme-inducing AEDs lower hormonal levels including oral contraceptives, Warfarin, steroids, tricyclics, cyclosporine, digitalis, and antipsychotics. Carbamazepine is notorious in causing autoinduction, so that a smaller dose at the start is associated with blood levels which are achievable only by two to three times that dose after a month or so.
On the other hand, competitive enzymatic inhibition increases the levels of these AEDs causing clinical toxicity. Erythromycin markedly inhibits metabolism of carbamazepine. Cimetidine and propxyphene have similar but lesser effects. These drugs, as well as grapefruit juice, may result in rise of carbamazepine to toxic levels.
2. Valproic acid is a strong competitive inhibitor of certain hepatic enzymes, increasing levels of many drugs including phenobarbital, lorazepam, lamotrigine, gonadal, and adrenal androgens.
3. Phenytoin and valproic acid have extensive plasma protein binding (80% to 95%), another undesirable pharmacokinetic property. Valproic acid, in addition, has nonlinear binding; at higher levels, the proportion of protein-bound drug is less, and there is a disproportionately high level of free or unbound drug. It is the free fraction that relates to drug efficacy and clinical toxicity.
4. Another troubling pharmacokinetic characteristic of phenytoin is its nonlinear elimination resulting from saturation of hepatic microsomal P-450 enzymes. Small increments (by 30 mg capsules) need to be made within the therapeutic window of 10 to 20 µg per ml. Giving a 100 mg capsule to a patient with a blood level of 15 µg per ml on a dose of 300 mg per day may lead to levels close to 30 µg per ml with the risk of clinical toxicity. Similarly, higher serum levels require a relatively small decrease in the dose to optimize the level.
5. Shorter elimination half-lives of some of the traditional AEDs (e.g., carbamazepine and valproic acid) necessitate giving them in three or four divided doses. However, the introduction of sustained- or extended-release preparations of these AEDs allows two divided doses ensuring better compliance.
VII. NEWER AEDS
Approximately 60% to 70% of patients with newly diagnosed epilepsy become seizure-free when treated with traditional AEDs as monotherapy. The remaining 30% to 40% are either unresponsive to traditional AEDs or obtain satisfactory seizure control only with polytherapy, but at a price of side effects and drug interactions. Furthermore, many of these AEDs are associated with neurotoxicity, teratogenic effects, drug–drug interaction, and many undesirable pharmacokinetic properties. Hence, newer AEDs are needed that may have a better side-effect profile, work through novel mechanisms to control epileptogenesis, and have more desirable pharmacokinetics. An ideal AED should be effective against multiple seizure types, effective against seizures resistant to traditional AEDs, have low neurologic and systemic toxicity, and should have favorable pharmacokinetics: complete oral absorption, minimal binding to plasma protein, primarily renal elimination, long elimination half-life, linear kinetics, and no enzyme induction or inhibition.
After a lapse of more than a decade, a number of new AEDs have been introduced in the United States, starting in 1993. Although none satisfy all the criteria of being an ideal AED, many show marked improvement when compared with the traditional AEDs. They are briefly discussed below. Their mechanism of action, indications, and side-effect profile are listed in Table 39.4, and the pharmacokinetics and dosages for adults are summarized in Table 39.5. Because a combined AED therapy is often used, knowledge of pharmacokinetic interactions between the AEDs is essential. These drug interactions are summarized in Table 39.6.
TABLE 39.4 Mechanism of Action, Indications and Undesirable Side Effects of Newer AEDs
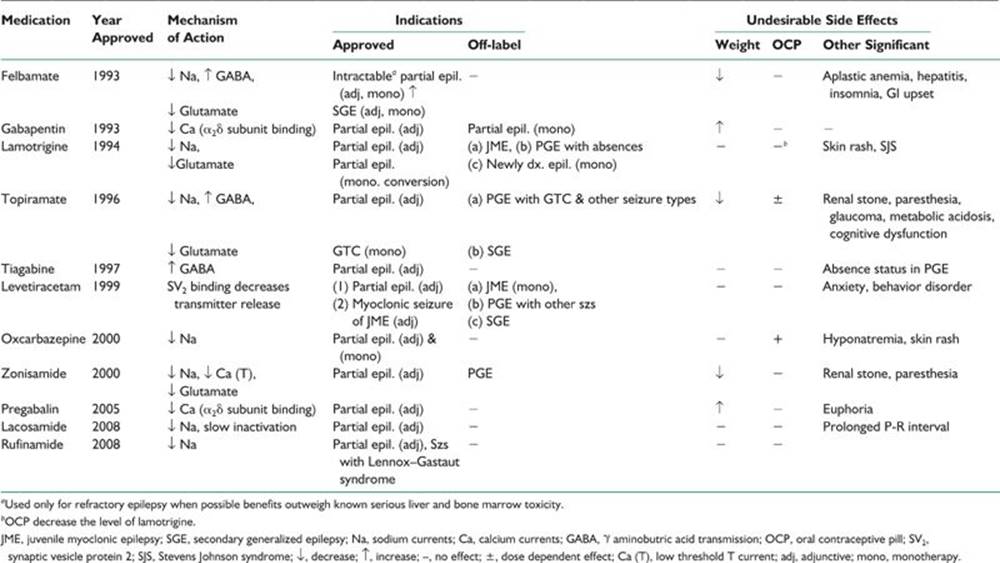
TABLE 39.5 Pharmacokinetics and Adult Dosages of Newer AEDs
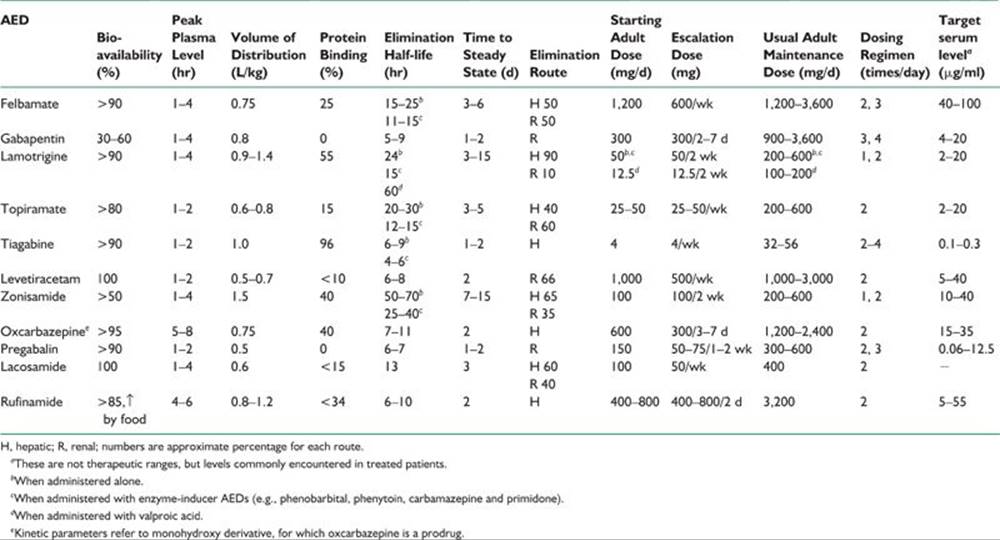
TABLE 39.6 Pharmacokinetic Interactions among AEDs
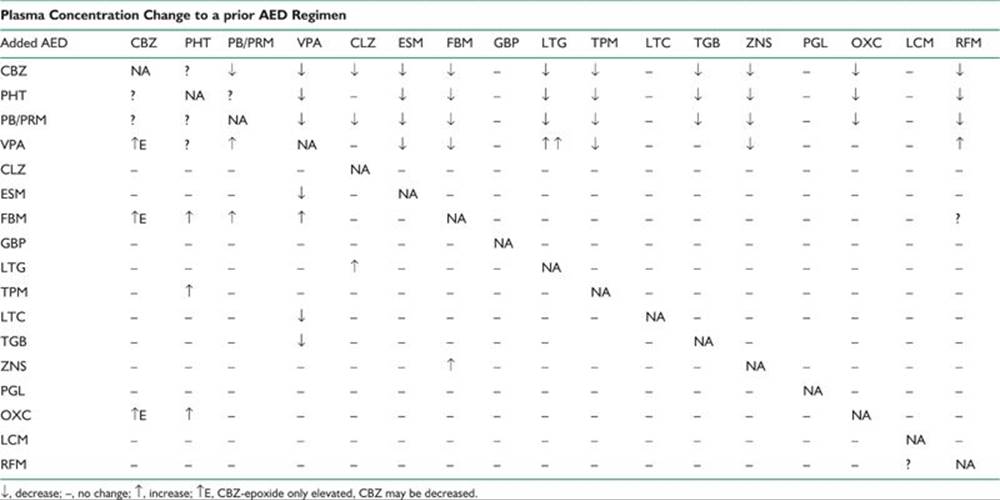
The utility of many newer AEDs is still evolving but there is a clear trend for increasing their use in partial and generalized epilepsy, refractory to traditional AEDs.
A. Efficacy.
1. Most of the newer AEDs are approved by the FDA for adjunctive therapy in the treatment of partial epilepsy refractory to traditional AEDs. The exception are felbamate and rufinamide, which are approved, also, for seizures associated with secondary generalized epilepsy of the Lennox–Gastaut’s type.
2. Oxcarbazepine is now approved as monotherapy because of a better side-effects profile and more desirable pharmacokinetics; it is preferred over carbamazepine as the initial drug of choice for partial epilepsy.
3. Levetiracetam, lamotrigine, topiramate, and zonisamide have broad-spectrum activity against seizures associated with both focal and generalized epilepsies.
4. Lamotrigine has shown efficacy against absences and tonic–clonic seizures generalized from the onset. It is increasingly used to treat patients with PGE to avoid the side effects associated with valproic acid, especially in women.
5. Similarly, topiramate is approved for generalized tonic–clonic seizures (primary or secondarily generalized) and levetirecetam is found to be effective against myoclonic seizures associated with PGE.
6. Gabapentin and tiagabine are not used as often as others because of less efficacy and sedative side effects.
B. Side effects.
1. Many of the newer AEDs (e.g., oxcarbazepine, gabapentin, zonisamide, and tiagabine) are associated with neurotoxic side effects similar to those with traditional AEDs. Lamotrigine, levetiracetam, and topiramate have relatively less neurotoxicity in the commonly used daily doses.
2. Serious reactions are rare with newer AEDs with the exception of a high incidence of felbamate-associated aplastic anemia and toxic hepatitis detected after its release. This has led to a marked drop in its use.
3. Lamotrigine causes a skin rash in 5% to 10% of patients, and even Stevens–Johnson’s syndrome may occur, although rarely. These complications are more common with rapid titration of the dose and comedication with valproic acid.
4. Topiramate produces cognitive dysfunction, for example, slower mentation, word-finding difficulty, problems in concentration, thinking abnormalities, impaired memory, and encephalopathy. These are less common with daily doses below 400 mg. Rarely, acute myopia associated with the secondary angle closure glaucoma has been reported.
5. Levetiracetam is associated with anxiety and behavioral side effects.
6. Zonisamide and topiramate may produce renal calculi in approximately 1% patients as well as paresthesiae in the limbs.
7. Like carbamazepine, oxcarbazepine is associated with marked hyponatremia in 3% of patients, more often in elderly patients or those who are on other medications. It becomes evident in the first few months of therapy but rarely necessitates discontinuation of the drug.
8. Lacosamide may prolong the P-R interval; hence, it should be used with caution in patients with known cardiac problems.
C. Newer AED versus traditional AEDs. Newer AEDs have substantial advantages over traditional AEDs including absence of side-effects, none or fewer drug interactions, and favorable pharmacokinetics. However, there is no evidence that any new AED has better efficacy compared with carbamezapine, phenytoin, or valproic acid in well-control trials of recent onset epilepsy.
D. Pharmacokinetics.
1. Except zonisamide and gabapentine (dose-related absorption), all the newer AEDs have 90% or more bioavailability with oral intake.
2. Hepatic metabolism is significant only with zonisamide and tiagabine, but they do not cause significant enzymatic induction.
3. Lamotrigine and oxcarbazepine, although metabolized in the liver, do not involve cytochrome P-450 enzyme system (phase I reaction). They undergo extensive metabolism by glucuronidation (phase II reaction).
4. Topiramate, levetirecetam, gabapentine, and pregabalin are eliminated either completely or largely by renal route without undergoing significant hepatic metabolism.
5. Oxcarbazepine and topiramate do induce the metabolism of oral contraceptives, but the effect of topiramate is significant only with doses over 400 mg per day.
6. Most of the newer AEDs, with the exception of tiagabine, has either no or clinically insignificant binding to the plasma proteins.
7. All of the newer AEDs have linear kinetics and no autoinduction.
8. Significant drug-to-drug interactions are absent with the use of gabapentine, pregabalin, levetiracetam, and zonisamide.
9. Most relevant interaction is between valproic acid and lamotrigine; valproic acid decreases the metabolic elimination of lamotrigine, increasing its half-life three to four times. In patients on valproic acid, lamotrigine needs to be started at a very low dose, titrated up very slowly, and the maintenance dose needs to be much smaller (100 to 200 mg) than when lamotrigine is used either alone or with enzyme-inducing AEDs, such as carbamazepine.
10. There has been little available data regarding the effect of newer AEDs on bone density or teratogenicity. Pregnancy registries have provided the most extensive data only on lamotrigine, which has a low adverse effect on fetal development.
VIII. STATUS EPILEPTICUS
A. Definition. The definition of status epilepticus has evolved over last two decades. The World Health Organization defines status epilepticus as follows: “A condition characterized by an epileptic seizure that is sufficiently prolonged or repeated at sufficiently brief intervals so as to produce an unvarying and enduring epileptic condition.” Most critical is the duration of seizure activity, and 30 minutes is the most commonly used duration. Hence, status epilepticus is considered to be present if continuous seizure activity persists for at least 30 minutes or two or more sequential seizures repeat within 30 minutes without full recovery of consciousness between seizures. Because the neuronal injury increases with duration, some experts now suggest that if seizure activity persists beyond 5 minutes, or, if at least two seizures have occurred over this period without full recovery, one may consider status epilepticus and treat it as such.
B. Types. Overall incidence is 1 in 5,000, and an estimated number of 60,000 new cases of status occur in the United States per year. Any type of seizure can manifest as status epilepticus, but the common forms include the following:
1. Generalized convulsive status epilepticus (GCSE) manifests as repeated major motor convulsions without full recovery of consciousness between seizures. In the past, the term status epilepticus implied essentially this form of status.
2. Nonconvulsive status epilepticus produces a continuous or fluctuating “epileptic twilight” state. This includes absence status and complex partial status. Only an EEG can establish the diagnosis.
3. Simple partial status epilepticus is characterized by repeated focal motor seizures, epilepsia partialis continua, and focal impairment of function (e.g., aphasia) without accompanying alteration of consciousness.
C. Cause of GCSE. GCSE is the most common and most serious type of status epilepticus. Unconsciousness associated with convulsive major motor seizures is the cardinal feature. The motor activity varies in symmetry and form depending on the history of seizures, duration of status, duration of treatment, and associated pathologic processes in the brain. GCSE occurs mainly in the following settings:
1. Acute cerebral insult or acute encephalopathy accounts for one half of cases of GCSE. These disorders include meningitis, encephalitis, head trauma, hypoxia, hypoglycemia, drug intoxication (e.g., cocaine), drug withdrawal, and strokes or metabolic encephalopathy.
2. GCSE can occur in patients with a history of epilepsy due to remote neurologic insults. Common precipitants of GCSE include changes in AEDs, sudden discontinuation or reduction in AEDs, systemic infection, physical and emotional stress, and sleep deprivation.
3. It can occur as an initial unprovoked epileptic event in an otherwise healthy person. Such “idiopathic” cases may account for one-third of all cases of GCSE.
D. Prognosis. GCSE is an emergency associated with substantial morbidity and mortality. The overall mortality may be as high as 30% among adults. GCSE associated with acute neurologic insults has the poorest prognosis, which essentially depends on the underlying cerebral etiologic factor.
When GCSE is the first epileptic event for an otherwise neurologically intact patient, or when it occurs in a patient with a previously known history of epilepsy but has a benign or reversible cause (e.g., hypoglycemia or drug or alcohol withdrawal), the prognosis is good if therapy is instituted promptly.
Without adequate and prompt treatment, GCSE can progress to a state of electromechanical dissociation in which the patient becomes increasingly unconscious or encephalopathic from the ongoing status, but the convulsive activity becomes increasingly subtle although EEG continues to show an ictal pattern. Patients with this condition, which is often called subtle status epilepticus, are considered to be candidates for an aggressive therapy, as are those with overt GCSE.
E. Management of GCSE. Immediate treatment needs to be instituted after establishing that the patient has an ongoing seizure lasting over 5 minutes, or has had two separate tonic–clonic seizures over a short period without recovery in between. The treatment protocol outlined in Table 39.7 is useful in the management of GCSE.
1. Diagnose status epilepticus by observing either continued seizure activity beyond 5 minutes or two generalized convulsions without full recovery of consciousness.
2. Assess vital functions and systemic abnormalities and stabilize the vital functions as much as possible.
a. Maintain an adequate airway and oxygenation. This usually can be accomplished with an oral airway. The airway should be suctioned periodically to maintain patency. Oxygen should be administered through a nasal cannula or with a mask and a bag-valve-mask ventilator. If, after bagging, respiratory assistance is still needed, endotracheal intubation should be considered.
TABLE 39.7 Treatment Protocol for Status Epilepticus (Generalized Tonic–Clonic) in Adults
|
Time (min) |
Treatment |
|
0–5 |
Diagnose status by observing either continued seizure activity or one additional seizure |
|
Assess vital functions, insert oral airway, and give oxygen |
|
|
6–10 |
Establish IV infusion line with normal saline solution. Monitor temperature and BP |
|
Draw blood for electrolytes, glucose, Ca, Mg, AED levels, CBC, BUN, and AST |
|
|
Administer 100 mg of thiamine followed by 50 ml of 50% glucose IV push |
|
|
11–20 |
Give lorazepam 0.1 mg/kg IV push at a rate of <2 mg/min |
|
21–60 |
Whether or not lorazepam stops the seizures, administer phenytoin 20 mg/kg IV no faster than 50 mg/min. Preferably, use fosphenytoin IV in PE doses |
|
Monitor BP and ECG during phenytoin (or fosphenytoin) infusion, and if hypotension or ECG changes occur, slow the infusion or temporarily withhold the drug. If seizures continue, give an additional 5–10 mg/kg of phenytoin (or fosphenytoin) |
|
|
>60 |
If seizures continue, intubate the patient, transfer to ICU, and start continuous EEG |
|
Start pentobarbital (or propofol or midazolam) coma. Use an IV loading dose to produce at least a suppression-burst pattern on EEG. Continue a maintenance dose by IV drip to maintain seizure free state or at least suppression-burst pattern on EEG |
|
|
Slow the rate of infusion after 24–96 hr periodically to determine whether seizures have stopped clinically and on EEG |
|
|
Monitor continuous EEG, BP, ECG, and respiratory function |
|
|
Abbreviations: Ca, calcium; Mg, magnesium; AST, aspartate aminotransferase. |
|
b. Assess blood pressure (BP) and maintain it at a normal or high-normal level during prolonged GCSE. Use vasopressors if necessary.
c. Establish an IV infusion line using normal saline solution. Blood should be drawn initially for CBC, blood sugar, blood urea nitrogen (BUN), serum electrolytes (including calcium and magnesium), and AED levels, and both urine and blood should be obtained for toxicology screening.
d. Assess oxygenation by means of oximetry or periodic arterial blood gas determination.
e. Monitor rectal temperature. Body temperature can increase to a high level during prolonged status epilepticus as a result of increased motor activity.
f. If hypoglycemia is documented or if it is impossible to obtain prompt blood sugar determination, administer 50 ml of 50% glucose by means of IV push. In adults, thiamine (100 mg) is always given before glucose to protect a thiamine-deficient patient from exacerbation of Wernicke’s encephalopathy.
g. Administer bicarbonate therapy only if serum pH is so low as to be immediately life-threatening. Acidosis commonly develops during GCSE, but acidosis usually responds promptly once the seizure activity is controlled.
h. For rare patients with GCSE resulting from hyponatremia (serum sodium concentration <120 mEq per L), hypocalcemia, or hypomagnesemia, administer appropriate electrolytes by means of IV drip.
3. Drug therapy for the control of GCSE. The goals of therapy are rapid termination of the clinical and EEG evidence of seizure activity and subsequent maintenance of a seizure-free state. Present recommendation is to treat GCSE by a combination of a short-acting benzodiazepine (such as lorazepam or diazepam) for rapid termination of seizure activity and a long-acting anticonvulsant (such as phenytoin or fosphenytoin) to subsequently maintain a seizure-free state. The use of IV lorazepam (2 mg) or diazepam (5 mg) by the emergency medical personnel prior to arrival in the emergency room is increasingly recommended for out of the hospital status.
In the emergency room, most physicians prefer IV lorazepam first, followed by fosphenytoin.
a. Lorazepam is administered at 0.1 mg per kg IV at a rate of <2 mg per minute. Diazepam has been largely replaced by lorazepam in the acute treatment of GCSE.
Although diazepam can stop GCSE slightly faster than can lorazepam, it has a much shorter duration of effectiveness allowing seizures to recur within 30 minutes of IV administration of diazepam bolus. Lorazepam has relatively rapid effectiveness and yet has a prolonged duration of action against status epilepticus. Both diazepam and lorazepam can produce serious respiratory depression or hypotension, particularly when given in combination with barbiturates.
b. Phenytoin or fosphenytoin. If seizures are still continuing after the administration of lorazepam, or even if they have been successfully terminated, IV administration of phenytoin (or preferably fosphenytoin) is usually needed to prevent recurrence of convulsions.
(1) Administration. The usual loading dose of phenytoin is 20 mg per kg, given through a syringe into the IV port close to the patient at a rate <50 mg per minute. Injection is preferably performed by a physician. BP and ECG are continuously monitored throughout the infusion. The rate of infusion should be slowed or temporarily stopped if hypotension, widening of the QT interval, or arrhythmias develops.
(2) There is a significant risk of skin complications when IV phenytoin is given. These include phlebitis, tissue sloughing after extravasation, and most serious, purple glove syndrome after infusion into a dorsal hand vein. The latter is a delayed soft tissue injury that can result in severe edema, arterial occlusion, and tissue necrosis that can necessitate amputation. These complications occur because parenteral phenytoin has 40% propylene glycol (antifreeze). Its pH is adjusted to 12.2 with sodium hydroxide (drain cleaner). Use of IV phenytoin is now virtually replaced by fosphenytoin, a much safer preparation for IV administration.
(3) Fosphenytoin, a phosphate ester of phenytoin, is enzymatically converted to phenytoin by serum phosphatases. It is available only for parenteral use (IV or IM). Because it is dissolved in TRIS buffer at a pH of 8 to 9, fosphenytoin does not cause tissue injury as phenytoin does. Three parts of phenytoin are bioequivalent to two parts of phenytoin, but the fosphenytoin dose is labeled in phenytoin equivalents (PE), so that 150 mg of fosphenytoin is labeled 100 mg PE. Although somewhat confusing, it apparently allows easy conversion of phenytoin to fosphenytoin dosing. Fosphenytoin can be administered more rapidly (up to 150 mg PE per minute compared with up to 50 mg per minute for phenytoin). The shorter infusion time compensates for the time needed for its conversion to active phenytoin. The result is that peak levels of phenytoin are attainable with IV fosphenytoin as rapidly as with phenytoin infusion itself. BP and ECG monitoring are recommended during fosphenytoin infusion, as they are with IV phenytoin.
(4) If the standard loading dose of 20 mg per kg of phenytoin (or 20 mg PE per kg of fosphenytoin) fails to stop GCSE, an additional 5 to 10 mg per kg may be given. Some patients may need high blood levels (30 to 40 µg per ml) before the seizure terminates. Phenytoin or fosphenytoin is effective in the treatment of 40% to 90% of patients with GCSE.
(5) Phenytoin or fosphenytoin should be used cautiously in the care of elderly patients, patients with known cardiac abnormalities, and patients with low baseline BP.
4. Management of refractory GCSE. If status fails to respond to combined lorazepam and fosphenytoin administration, the status is termed as refractory GCSE. There is a high probability that the patient has an acute cerebral insult responsible for the status and prognosis is more guarded, mortality being around 50%.
a. GCSE is successfully terminated in approximately 75% of patients by lorazepam, phenytoin (or fosphenytoin) combination. Remaining 25% constitute refractory GCSE.
b. The patient needs to be intubated now and placed in ICU for managing refractory GCSE.
c. The patient needs general anesthesia to eliminate not only clinical seizures but also electrical discharges indicative of continuing seizure activity. There is no randomized comparative study of the management of refractory GCSE on which to base the choice of the drug to induce general anesthesia. Continuous IV infusion of pentobarbital, propofol, and midazolam has been reported to be effective. The choice of a drug is largely based on the clinical experience of the physician. If one agent in optimal dosages is unsuccessful in controlling status, another should be tried. Continuous EEG monitoring is mandatory. The rate of administration of the drug is adjusted to ensure at least suppression burst or, as some experts recommend, cessation of all epileptiform activity, which may require making the EEG pattern almost isoelectric. However, the maximal EEG suppression is associated with an increased frequency of hypotension requiring the use of pressor agents. The anesthesia is continued for 1 to 4 days before an attempt is made to lighten it. If clinical seizures or ictal EEG patterns return, the infusion is appropriately increased.
Pentobarbital-induced anesthesia has been used for a long period for refractory GCSE. It is started intravenously with a loading dose of 5 to 15 mg per kg, followed by a maintenance dosage of 0.5 to 1.0 mg/kg/hour to maintain an EEG suppression-burst pattern or bring about cessation of all epileptiform discharges.
Propofol, a short-acting agent, is becoming the drug of choice. It is started IV with a loading dose of 1 to 2 mg/kg, given over 5 minutes, followed by a maintenance dosage of 1 to 5 mg/kg/hour to maintain suppression-burst pattern or complete control of all epileptiform discharges on the EEG.
Midazolam is administered at a loading dose of 0.2 mg/kg by slow IV bolus followed by a maintenance dose of 0.1 to 0.5 mg/kg/hour.
There are no prospective randomized trials comparing these three infusion agents for the treatment of refractory status. One systematic review demonstrated no difference between the three, at least for mortality rate. It appears that pentobarbital anesthesia, although, very effective, is associated with serious hypotension. Similarly, propofol if used in large doses (>5 mg/kg/hour) is associated with serious hypotension. On the contrary, midazolam appears to be safer and may be an excellent agent for refractory status epilepticus.
5. Additional diagnostic studies. When the seizures have been stopped or sufficiently controlled and the patient’s vital signs have been stabilized, additional diagnostic studies, such as chest radiographs, lumbar puncture, brain imaging, and EEG, are performed to evaluate the cause of the GCSE and the effectiveness of the antiepileptic therapy.
6. Long-term antiepileptic therapy. After the episode of GCSE has been brought under control, most patients need continuation of some form of AED therapy. Long-term AED therapy is indicated when GCSE is caused by a structural brain lesion or when the patient has a history of epileptic seizures. When GCSE constitutes the patient’s first seizure and no cause is found, the decision to initiate long-term AED therapy should be individualized, but most physicians initiate long-term treatment under such circumstances. If the GCSE was caused by acute CNS involvement, such as metabolic encephalopathy, meningoencephalitis, or cerebrovascular compromise, antiepileptic therapy is continued for a short period of 3 to 6 months.
F. Management of other types of status epilepticus. Other forms of status epilepticus do not pose the same emergency situation that GCSE poses. Complex partial status epilepticus has been reported to result in long-term neurologic deficits (e.g., permanent memory impairment) and should be controlled promptly with a benzodiazepine (e.g., lorazepam) followed by IV phenytoin (or fosphenytoin) or IV levitracetam. IV anesthesia is rarely indicated.
Management of absence status or simple partial status (focal motor seizure or epilepsia partialis continua without loss of consciousness) is not standardized. Absence status is best managed with an IV benzodiazepine (diazepam or lorazepam), which is effective in most cases. If a benzodiazepine is not effective, valproic acid can be given intravenously, in a dose of 25 mg per kg to promptly achieve therapeutic blood levels. After the absence status is controlled, valproic acid therapy is continued orally. Simple partial status responds to phenytoin (or fosphenytoin) usually in large doses to maintain blood levels as high as 30 g per ml. IV levitracetam or valproic acid are other alternatives. Benzodiazepines are not desirable because of their sedative side effects, except using one or two doses of IV lorazepam or diazepam initially.
IX. MANAGEMENT OF ACUTE SEIZURE CLUSTER
Some patients have repetitive series or clusters of epileptic seizures that occur within a short period, not meeting the definition criteria of status epilepticus. Such seizure clusters can be intermittently managed with either of the following approaches:
A. Most physicians use benzodiazepine (lorazepam or diazepam) to manage a cluster. Lorazepam is given in doses of 2 to 4 mg orally (sublingually) or parenterally (IM or IV), usually becoming effective in approximately 30 minutes. Administration may be repeated in doses of 1 to 2 mg with a maximum dose of 6 to 8 mg in 24 hours.
B. Diazepam gel (Diastat) is approved for rectal administration in the management of repetitive seizures. It is as effective, if not better, than lorazepam. Diazepam gel is administered in doses of 0.2 to 0.5 mg per kg rectally (usual adult dose is 10 to 20 mg); it is effective in 15 minutes and the effect lasts as long as 8 hours. If social or logistic reasons make the use of rectal administration difficult, sublingual or oral lorazepam is a better alternative.
C. Home treatment with rectal diazepam reduces the risk for subsequent status and the need for frequent visits to the emergency room in patients at risk for seizure-clusters or who have history of status epilepticus.
X. EPILEPSY, PREGNANCY, AND OTHER WOMEN’S ISSUES
A. Major problems. Pregnancy in a woman with epilepsy (WWE) is considered to constitute a high risk and needs to be followed by a high-risk obstetrician. Recently, American Academy of Neurology (AAN) has issued guidelines regarding the management of WWE, which are incorporated in the following discussion:
1. Obstetrical complications. In short, there is no conclusive evidence of increased obstetrical complications in WWE on AEDs.
a. There is a weak evidence that WWE on AEDs has a slightly higher risk for cesarean section but a good evidence that this risk is not >2 times expected.
b. There is a good evidence that there is no greater risk for late pregnancy bleeding, early contractions or early labor and delivery in WWE on AEDs. However, these complications are substantially increased in WWE who smoke.
c. There is insufficient evidence to support or refute an increased risk for preeclamsia, hypertension, or miscarriage.
2. Effect of pregnancy on epilepsy. It is usually believed that seizures become more frequent during pregnancy in one-third WWE especially in second and third trimesters. According to the AAN guidelines, there is insufficient evidence to support or refute an increased risk in seizure frequency or occurrence of status epilepticus. Furthermore, if a WWE has been seizure free for at least 9 months prior to becoming pregnant, there is a high likelihood of her remaining seizure free during pregnancy.
3. Alteration of the pharmacokinetics of AEDs (increased clearance) results in decreased serum concentrations of almost all AEDs. Carbamazepine is least and lamotrigine is most affected. Changes in AED levels and in the hormonal status, poor compliance, psychological stresses, and sleep deprivation during pregnancy are some of the factors for exacerbation of seizures in some WWE.
4. The incidence of fetal malformation is two to three times higher among mothers with epilepsy than among women without epilepsy. Most of this effect is a result of fetal exposure to AEDs. Malformations include minor malformations, which are deviations from normal morphology not requiring treatment, and major malformations, requiring medical and/or surgical treatment. The overall incidence of major birth defects (oropalatal clefts, urogenital and congenital heart anomalies, and neural tube defects) among infants of epileptic mothers is approximately 7%, compared with 2% to 3% in women not receiving AEDs. All major AEDs have potential teratogenic effects, but certain facts have emerged from a number of pregnancy registries: Avoid the use of polytherapy during pregnancy and strongly consider monotherapy.
a. Incidence of malformation increases with polytherapy compared with monotherapy. There is also an evidence for reduced cognitive outcome in the offspring of WWE receiving polytherapy during pregnancy.
b. Valproic acid is associated with the highest incidence (6% to 10%) of major malformations, which include neural tube defects, facial clefts, and hypospadias. Hence, avoid the use of valproic acid in women during child bearing years.
c. Valproic acid shows dose-related adverse effects on the fetus, the incidence of major malformation reaches three times with a dose above 1 g per day.
d. Children born to women who received AEDs, particularly valproic acid, show higher incidence of developmental delay and lower mean verbal IQ, often requiring special education. This is yet another reason to avoid the use of valproic acid in women who want or may become pregnant.
e. Phenytoin, carbamezapine, and phenobarb also, increase the risk of fetal malformation, cleft palate with phenytoin use, posterior cleft palate with carbamezapine use, and cardiac malformation with phenobarb use. Hence, avoiding the use of these AEDs during pregnancy would reduce major fetal malformations.
f. Lamotrigine, which is the most extensively studied new AED, appears to have the least teratogenicity (3%), probably no different from the general population. However, there seems to be an increased risk for nonsyndromic orofacial clefts (cleft lip/palate). The UK registry has also shown dose-response effect of lamotrigine-associated malformations, 2% incidence with doses up to 200 mg per day compared with 5.4% for above 200 mg per day. Polytherapy use of lamotrigine with valproic acid markedly increases the incidence of malformation (12%).
5. Adverse perinatal outcomes: There is a good evidence that WWE taking AEDs have an increased risk of small-for-gestation offspring and a weak evidence for low apgar scores at 1 minute.
6. Hemorrhagic disease is reported among 7% of newborns delivered by women who have received hepatic enzyme-inducing AEDs (phenobarbital, primidone, phenytoin, and carbamazepine) because of their effect in decreasing the vitamin K-dependent clotting factors. According to the AAN guidelines, there is insufficient evidence to support or refute an increased risk of hemorrhagic complications in the newborn of WWE taking AEDs.
B. Management guidelines. The therapeutic challenge is to keep the patient free of seizures while minimizing the adverse effects of seizures and AEDs on the course of pregnancy and the fetus. The major guidelines, which preferably are initiated before the patient becomes pregnant, are as follows:
1. Counsel the family about the higher incidence of fetal malformation, but assure the patient that most pregnant women (>90%) exposed to AEDs still bear healthy offspring.
2. May withdraw AEDs before pregnancy if the patient has remained free of seizures for >2 years. Do not wean AED if epilepsy syndrome (e.g., JME and MTS) suggests a high probability of recurrence.
3. Before conception, determine the best AED for seizure control. If the patient is on polytherapy, reduce AEDs to appropriate monotherapy. Because of the relatively high incidence of neural tube defects with valproic acid and carbamazepine, avoid these AEDs (both are schedule D drugs) and replace them with other AEDs. For women with PGE, lamotrigine alone or with levetirecetam or topiramate are alternative AEDs.
4. Prescribe folic acid in any woman during childbearing age, but certainly start it prior to the pregnancy (see below) to reduce the overall incidence of neural defects in the offspring.
5. Address and eliminate other risk factors, for example, drugs, alcohol, and smoking.
6. Do not stop or change AED therapy after the pregnancy has been diagnosed. The risk of fetal malformation is highest during the first 4 to 8 weeks of pregnancy. It usually is too late to protect the fetus by the time pregnancy is confirmed. Stopping or changing the drug can induce more frequent and more violent seizures with adverse consequences on both the mother and the fetus.
7. Supplemental multivitamins and folic acid are prescribed during the entire pregnancy. The precise dose for women with epilepsy has not been defined, but 1.0 mg per day is commonly used. Women at risk of having a child with neural tube defect (previous child or family history) are advised to supplement with 4.0 mg per day. Folic acid supplementation is recommended throughout childbearing age for sexually active women with epilepsy.
8. Monitor the AED serum level (preferably free level) before conception, at each trimester, in the last month of pregnancy, and through 8 weeks postpartum, especially if the patient is on lamotrigine, carbamezapine, or phenytoin. Adjust the dosage accordingly. Maintain lowest effective blood levels.
9. Measure serum α-fetoprotein and acetylcholinesterase levels at 15 to 18 weeks of gestation to be followed by high-definition ultrasound imaging to detect or neural tube defects and other major malfunctions. Together, these detect over 99% of the major fetal abnormalities. Amniocentesis is indicated only if these tests do not provide positive exclusion of a neural tube defect.
10. Give oral vitamin K1 (phytonadione) to the mother at 10 to 20 mg per day during the last month of pregnancy. Administer vitamin K1 (1.0 mg IM) to the neonate immediately after birth. These measures help reduce the incidence of AED-related hemorrhagic disease of the newborn.
11. After delivery, check AED levels and adjust the doses of AED, usually at a lower level. Some AEDs, such as lamotrigine and oxcarbazepine, start reversing their elimination rate to the pre-pregnancy level even prior to the delivery. Their dose needs to be brought down within a week or so of the delivery.
12. Breast-feeding is allowed. Nearly all AEDs appear in breast milk at a concentration closely related to the AED’s free plasma level. Hence, highly protein-bound AEDs attain only a very small concentration in the milk to produce significant clinical effect on the breast-fed baby. Even many of the newer AEDs, which are not protein-bound, deliver only a small daily dose to the baby. Most AEDs, with the possible exception of ethosuximide, phenobarbital, or primidone, do not reach sufficient concentrations in the breast milk to pose a serious concern for the infant. Problems such as poor suck, lethargy, irritability, or decreased weight gain have been reported in some infants nursed by women on these AEDs.
C. Prevention of pregnancy with oral contraceptives.
1. Women with epilepsy may use oral hormonal contraceptives if they wish. They do not exacerbate epilepsy despite the warnings on package inserts.
2. The major concern in using oral contraceptives is the higher failure rate (>6% per year) among women taking hepatic enzyme-inducing AEDs (e.g., phenytoin, carbamazepine, phenobarbital, primidone, felbamate, oxcarbazepine, rufinamide, and topiramate [doses over 200 mg per day]). This is due to increased elimination of both estrogens and progestogens by these AEDs. The effect is on many contraceptive preparations including combined contraceptive pill, combined patch, progestogen-only pill, and progestogen implants.
The most commonly used is a combined “mini-pill” containing 35 µg or less of estrogen combined with progestogen. It may be less effective. Breakthrough bleeding can be a warning of decreased contraceptive efficiency. Patients taking these enzyme-inducing AEDs usually need a medium-dose combined contraceptive pill containing at least 50 µg of ethinyl estradiol. If breakthrough bleeding occurs, increase the dose to 75 or 100 µg per day. Theprogestogen-only pill is likely to be ineffective in women on enzyme-inducing AEDs. Progestogen implants are contraindicated. Intramuscular medroxyprogesterone injections (Depo-Provera) appear to be effective but patients are advised to take injections every 10 weeks rather than 12 weeks if they are on enzyme-inducing AEDs. Contraception using Mirena coils is highly effective and is not affected by enzyme-inducing AEDs because progestogen acts by being released locally in the uterus. It is a highly effective contraceptive option in women with poor compliance history.
Because the usual 7-day pill-free interval decreases the contraceptive effectiveness, some recommend not only a higher dose estrogen-containing pill (e.g., 5 mcg estrogen) but using it in a “tricycling regimen.” The pills are given consecutively for three cycles without a break, followed by a short pill-free period of 4 days. This tricycling method has the best contraceptive efficacy.
Because both estrogens and progestogens are eliminated faster by enzyme-inducing AEDs, the higher dose of estrogen needs to accompany a similarly higher dose of progestogen in the combined contraceptive pill.
3. Non-enzyme inducers are unlikely to cause failure of oral contraception. These include valproic acid, lamotrigine, gabapentin, levetiracetam, zonisamide, pregabalin, and lacosamide.
4. Hormonal contraceptives have little effect on AEDs except on lamotrigine blood levels, which drop to half the precontraceptive levels because of enzyme induction. During the 7-day-off period, the levels could double back to pre-hormone levels. This effect may result in a breakthrough seizure during the 3 weeks of hormonal ingestion and toxicity during the off-week.
D. Catamenial epilepsy. Some women have increased frequency of seizures related to their menstrual cycle probably due to changing hormonal concentration during menstrual cycle. Catamenial seizures happen in about 10% of women and occur at three different times. Commonly, they occur either just before or during the first few days of menstruation (perimenstrual). Some have increased seizures at ovulation (midcycle), whereas others who have anovulation cycles have increased seizure activity in the second half of the menstrual cycle.
Perimenstrual increase may be treated by the intermittent use of acetazolamide at 250 to 500 mg per day starting 2 to 4 days before the anticipated onset and then continuing during menstruation.
An alternative is to cover the period of increased seizure frequency by additional doses of the AED or the use of a benzodiazepine such as lorazepam for a few days.
Hormone therapy is another alternative approach. Progestogen supplements may be given perimenstrually or during the latter half (10 to 26 days) of the menstrual cycle. A combined oral contraceptive pill or depot progestogen may also be used.
XI. AED TREATMENT OF THE ELDERLY
Prevalence of epilepsy increases sharply in the elderly. Common causes of seizures are cerebrovascular disease, degenerative dementia, and neoplasms in that order of frequency. Seizures are nearly exclusively partial onset, being simple partial, complex partial, or secondarily generalized type. The postictal period is often longer in the elderly and may last several days after a generalized tonic–clonic seizure. EEG shows epileptiform abnormalities in 40% patients.
A. Major problems. Elderly patients with epilepsy constitute a special patient population because of age-related changes in the pharmacokinetics of AEDs, comorbidity, and multiple drug therapy. There is a decrease in plasma albumin with resultant overall decrease in protein binding of AEDs; hence, total concentration of highly bound AEDs (valproic acid, phenytoin, etc.) becomes misleading. Hepatic drug metabolism is decreased, and there is a steady decline in glomerular filtration rate, decreased tubular secretory function, and decreased renal blood flow with normal aging. These changes lead to longer elimination half-life and reduced clearance of many AEDs, with either primarily hepatic or renal elimination. In addition, concomitant medical problems and, consequently, multiple co-medications further affect the absorption, disposition, and metabolism of AEDs. Elderly patients also tend to experience more side effects of AEDs, especially somnolence, confusion, gait disturbances, and postural and other tremors.
B. Management guidelines for using AEDs to treat the elderly.
1. AEDs should be prescribed only when the diagnosis of epileptic seizures is firmly established and there is a high probability of recurrence.
2. AED therapy should be initiated at lower doses and titrated more slowly to a daily dose that is on average lower by 30% to 40% than used to treat younger adults.
3. For high protein-bound AEDs, measurement of free rather than total concentration may be more useful in evaluating seizure control or drug-related toxicity.
4. “Therapeutic ranges” of the traditional AEDs do not apply to the elderly mainly because of an overall decrease tolerability for drugs in this age group. Aim for a “low therapeutic” serum level, increasing the dose only if clinically necessary.
5. Carbamazepine, phenytoin, and valproic acid have been commonly used in the past because the seizures are focal-onset, but their pharmacokinetics makes them less ideal for the elderly. Oxcabazepine is well-tolerated and approved as monotherapy.
6. Generally, many of the newer AEDs are better for the elderly patient because of their fewer drug interactions, minimal or no protein binding, and predominant renal elimination (e.g., gabapentin, levetiracetam, pregabalin, or lacosamide). However, the dose has to be adjusted downward in the presence of renal impairment. Lamotrigine has only minor interactions and good tolerability; hence, it is another alternative among newer AEDs. These five newer AEDs are presently approved only for adjunctive therapy. Ongoing monotherapy trials may make them the initial AED of choice for seizures of the elderly. Many experts use off-label gabapentine or levetiracetam in the elderly.
XII. DISCONTINUATION OF AEDS
The decision to discontinue AEDs in the care of a patient who has been seizure-free for several years depends on many prognostic factors. The relapse rate after AED withdrawal is approximately 30% to 40% among adults with chronic epilepsy.
A. Prognostic factors.
1. Types of epilepsy.
a. Some childhood forms of epilepsy (e.g., benign Rolandic epilepsy and childhood absence epilepsy) usually remit during adolescence.
b. Patients with PGE with onset in adolescence or adulthood have a good prognosis. Many remit, but some (especially JME) need lifelong therapy.
c. Patients with focal epilepsy (simple partial seizures, complex partial seizures, and secondarily generalized tonic–clonic seizures) have a high recurrence rate (40% to 50%) after discontinuing AEDs. This is especially true of adults with complex partial seizures, for which the relapse rate can approach or exceed 50%.
d. Patients in whom seizures occur in the setting of an acute cerebral insult (e.g., trauma, infection, or stroke) may not have chronic epilepsy. Such patients should be considered for withdrawal of AED after a seizure-free period of 3 to 6 months.
2. EEG findings obtained just before discontinuation of AED therapy are useful predictors of the outcome. The relapse rate is four to five times higher if EEG shows persistent epileptiform abnormalities. Hence, most physicians consider continuing AEDs if EEG continues to show paroxysmal abnormalities.
3. Other predictors of outcome. A high frequency of seizures, a long duration before seizures are controlled with AEDs, and multiple seizure types in a patient carry a less favorable prognosis. Similarly, patients who have structural abnormalities responsible for seizures or who have mental retardation or neurologic deficits are more likely to have recurrence of seizures after discontinuation of AEDs.
4. Duration of seizure-free period. There is no consensus on how long a patient should remain seizure-free before drug withdrawal is considered. Most physicians recommend a seizure-free period of 2 to 5 years, but such recommendations must be individualized because of the serious socioeconomic effect of recurrence of seizures.
B. General guide lines for withdrawing AEDs.
1. After assessing the risks and benefits, discontinuation of AEDs may be considered by the physician if the patient has been seizure-free for 2 to 5 years while taking AEDs, has had a single type of seizure, has normal neurologic examination, and normalized EEG with treatment.
2. Adult patients with PGE who have remained seizure-free for 2 to 5 years and whose EEGs show no paroxysmal abnormalities or photoparoxysmal response are good candidates for withdrawal of therapy unless they have JME.
3. Women who want to bear children and who have been seizure-free for several years should be considered for withdrawal of medication before conception to avoid possible ill-effects on the fetus.
4. On the other hand, most adults with focal epilepsy, secondary generalized epilepsy, or PGE manifesting as multiple seizure types probably need long-term, if not lifelong, AED therapy unless continuation of the medication and the seizure-free state are possible only at the cost of unpleasant side effects.
C. AED withdrawal mode and precautions.
1. AEDs must be withdrawn slowly, typically over 3 months or longer, especially in the case of barbiturates and benzodiazepines.
2. Patients taking more than one AED should have the less or least effective drug withdrawn before the first-line drug. Only after the patient has remained seizure-free, taking only one drug for several months, is the final drug withdrawn.
3. During the withdrawal period, the patient is advised to follow a restricted lifestyle (no driving or hazardous occupational or recreational activities) to minimize the consequences should the seizures recur.
4. If the seizures recur, the patient is promptly placed on an adequate dosage of an appropriate AED, which then probably has to be continued on a lifelong basis.
5. After the withdrawal period, attention has to be paid to such lifestyle issues as getting adequate sleep, avoiding alcohol, and avoiding anxiety that can cause recurrence.
XIII. REFERRALS
A. Psycosocial problems. Patients with epilepsy face many personal and psychosocial difficulties that require counseling. There is a high incidence of mood disorder; depression occurs in 20% to 50% patients. Driving restrictions, concerns regarding pregnancy, and possible transmission of epilepsy to the offspring are concerns that have to be addressed. It is important to detect these problems, respond to them promptly, and refer patients to appropriate specialists for further therapy and counseling. Patients should also be put in touch with local support groups, state epilepsy associations, and the Epilepsy Foundation of America. Patients who need comprehensive care can be referred to a local comprehensive epilepsy center equipped with multidisciplinary teams capable of providing psychosocial and vocational counseling in addition to the appropriate medical therapy.
B. Experimental AEDs. Approximately 20% to 30% of patients with epilepsy who have medically intractable seizures may be helped by referral to regional comprehensive epilepsy centers, which can conduct ongoing clinical trials of new AEDs. Such patients often need combination therapy with two or more AEDs, which is best handled in a comprehensive epilepsy center.
C. Surgical treatment.
1. Resective surgery. Patients who have medically refractory partial epilepsy with complex partial or secondarily generalized tonic–clonic seizures should be referred to a comprehensive epilepsy center for presurgical evaluation to determine whether they are suitable candidates for surgical treatment. Epilepsy is considered medically refractory when seizure control is unsatisfactory despite trials of three AEDs, in monotherapy or in combination. After multimodality presurgical evaluation, if found to have a single epileptogenic focus that does not involve eloquent cortex, these patients may be candidates for resective surgery.
a. Anterior temporal lobectomy is the most frequently performed resective surgery when the seizures arise from antero-medial area or the lateral cortical area of the temporal lobe on one side. Epileptic patients with mesial temporal sclerosis on the MRI are excellent candidates because they are usually medically refractory but respond very well to surgery, two-thirds rendered seizure-free.
b. Extratemporal resection for epileptogenic foci outside the temporal lobe is increasingly performed, although the success rate is 50% or less. Localizing an extratemporal seizure focus, for example, in the frontal lobe, is more challenging (may require invasive monitoring with subdural electrodes) unless a structural lesion is present on the MRI scan.
c. Corpus callosotomy is performed in a few epilepsy centers for patients with secondarily generalized epilepsy who suffer from frequent drop attacks (atonic and/or tonic seizures). The procedure is followed by a number of complications such as disconnection syndrome, difficulty with motor and language function, and maintenance. Drop attacks and generalized seizures respond favorably but rarely do seizures totally remit.
d. Hemispherectomy is reserved for a selected few patients who have unilateral hemispheric insult (Rasmussen’s encephalitis, Sturge–Weber’s syndrome, extensive cerebral dysgenesis, hemimegalencephaly, etc.), resulting in medically refractory focal epilepsy and hemiparesis.
e. Subpial transaction is a surgical technique to treat focal epilepsy when the seizures arise from eloquent cortex such as the language or motor area. The procedure cuts horizontally connecting fibers so as to decrease propagation of seizure activity without resulting neurologic defects. The procedure is often combined with corticectomy.
2. Vagal nerve stimulation. The FDA has approved VNS as adjunctive therapy for medically refractory seizures in adults and children. VNS is considered for patients with focal or generalized epilepsy who have undergone unsuccessful polytherapy trials of several AEDs, including newer AEDs, who are not candidates for resective surgery or have undergone unsuccessful resective surgery.
VNS requires implantation of a programmable signal generator subcutaneously in the upper chest on the left side. This device is capable of delivering intermittent stimulation to the left vagus nerve in the neck (by means of bipolar electrodes) at the desired settings. In addition, the patient or a companion can activate the generator by placing the accompanying magnet over the generator for several seconds to interrupt a seizure or reduce its severity if administered at seizure onset.
The mechanism of action of VNS is unknown, but several large trials showed that approximately 30% to 40% of patients with refractory partial-onset seizures had a 50% or greater reduction in seizure frequency. Only a few had complete control of seizures. Overall, VNS has the same efficacy as many of the newer AEDs but without drug-related side effects. Patients with aurae are particularly helped because they can activate the device to abort an impending seizure. The common side effects include hoarseness, throat pain, coughing, dyspnea, and muscle pain. Possible surgical complications of hematoma, wound infection, left vocal cord paralysis, and injury to the vagus nerve or carotid sheath are rare when the procedure is performed by an experienced surgeon.
3. Future trends. Gamma knife surgery, deep brain stimulation of the anterior thalamic nuclei, and responsive neurostimulation (Neuropace) are some of the promising surgical techniques, which are being evaluated at present and may prove to be useful in providing better seizure control in the future.
![]()
Recommended Readings
Bourgeois BFD. Pharmacokinetic properties of current antiepileptic drugs. What improvements are needed? Neurology. 2000;55(suppl 3):S11–S16.
Browne TR. The pharmacokinetics of antiepileptic drugs. Neurology. 1998;51(suppl 4):2–7.
Browne TR, Holmes GL. Handbook of Epilepsy. 3rd ed. Philadelphia, PA: Lippincott Williams & Wilkins; 2004.
Cloassen J, Hirsch LJ, Emerson RG, et al. Treatment of refractory status epilepticus with pentobarbital, propofol or midazolam: a systematic study. Epilepsia. 2002;43:146–153.
Commission on Classification and Terminology of the International League against Epilepsy. Proposal for revised clinical and electroencephalographic classification of epileptic seizures. Epilepsia. 1981;22:489–501.
Commission on Classification and Terminology of the International League against Epilepsy. Proposal for revised classification of epilepsies and epileptic syndromes. Epilepsia. 1989;30:389–399.
Engle J, Pedley TA, eds. Epilepsy: A Comprehensive Textbook. Vol 2. Philadelphia, PA: Lippincott–Raven Publishers; 1997.
Engle J Jr, Wiebe S, French J, et al. Special report, practice parameter: temporal lobe and localized neocortical resection for epilepsy. Epilepsia. 2003;44:741–751.
French JA, Kanner AM, Bautista J, et al. Efficacy and tolerability of the new antiepileptic drugs I: treatment of new onset epilepsy. Neurology. 2004;62:1252–1260.
French JA, Kanner AM, Bautista J, et al. Efficacy and tolerability of the new antiepileptic drugs II: treatment of refractory epilepsy. Neurology. 2004;62:1261–1273.
Levy RH, Mattson RH, Meldrum BS, eds. Antiepileptic Drugs. 4th ed. New York, NY: Raven Press; 1995.
Lowenstein DH, Alldredge BK. Status epilepticus. N Engl J Med. 1998;338:970–976.
Mattson RH. Efficacy and adverse effects of established and new antiepileptic drugs. Epilepsia. 1995;36(suppl 2):S12–S26.
Mattson RH, Cramer JA, Collins JF. A comparison of valproate with carbamazepine for the treatment of complex partial seizures and secondarily generalized tonic–clonic seizures in adults. N Engl J Med. 1992;27:765–771.
Mattson RH, Cramer JA, Collins JF, et al. Comparison of carbamazepine, phenobarbital, phenytoin, and primidone in partial and secondarily generalized tonic–clonic seizures. N Engl J Med. 1985;313:145–151.
O’Brien MD, Guillebaud J. Critical review, contraception for women with epilepsy. Epilepsia. 2006;47:1419–1422.
Quality Standards Subcommittee of the American Academy of Neurology. Practice parameter: a guideline for discontinuing antiepileptic drugs in seizure free patients [summary statement]. Neurology. 1996;47:600–602.
Quality Standards Subcommittee of the American Academy of Neurology. Practice parameter update: management issues for women with epilepsy—focus on pregnancy (an evidence-based review): teratogenesis and perinatal outcomes. Neurology. 2009;73(2):126–132.
Report of the Quality Standards Subcommittee of the American Academy of Neurology. Practice parameter: management issues for women with epilepsy [summary statement]. Neurology. 1998;51:944–948.
Rowan AJ. Reflections on the treatment of seizures in the elderly population. Neurology. 1998;51(suppl 4):28–33.
Stowe ZN, Herzog AG, eds. The Use of Antiepileptic Drugs, Long-Term Management Issues. Stanford, CA: Stanford School of Medicine and Imprint Science; 2007.
Therapeutics and Technology Assessment Subcommittee of the American Academy of Neurology. Assessment: generic substitution for antiepileptic medication. Neurology. 1990;40:1641–1643.
Therapeutic and Technology Assessment Subcommittee of the American Academy of Neurology. Reassessment: vagus nerve stimulation for epilepsy. Neurology. 1999;53:666–669.
Treiman DM, Meyers PD, Walton NY, et al. A comparison of four treatments for generalized convulsive status epilepticus. N Engl J Med. 1998;339:792–798.
Wilder BJ. Management of epilepsy: consensus conference on current clinical practice. Neurology. 1998;51(suppl 4):1–43.
Working Group on Status Epilepticus. Treatment of status epilepticus. JAMA. 1993;270:854–859.
Zahn CA, Morrell MJ, Collins SD, et al. Management issues for women with epilepsy: a review of the literature. Neurology. 1998;51:949–956.