Myopathy is an abnormality of the skeletal muscle in which striated muscle cells or connective tissue elements are affected. Myopathy can result from abnormalities of skeletal muscle proteins (Duchenne muscular dystrophy), alterations of the sarcolemmal ion channels (hyperkalemic periodic paralysis), mitochondrial alterations (mitochondrial myopathy), or cell-mediated autoimmune mechanisms (polymyositis), to name a few examples. Because of the myriad abnormal mechanisms, treatments vary from one condition to the next. Progress in molecular biology, genetics, and immunology has considerably expanded our understanding of these complicated diseases. This chapter emphasizes current therapeutic approaches to the care of patients with relatively common forms of myopathy.
I. IDIOPATHIC INFLAMMATORY MYOPATHY
Idiopathic inflammatory myopathies are autoimmune diseases characterized by muscle weakness, pain, and fatigue. Inflammatory damage of muscle fibers is the underlying pathology. Polymyositis can occur in isolation or accompany other connective tissue disorders or associated systemic autoimmune disorders. Dermatomyositis, inclusion body myositis (IBM), and polymyalgia rheumatica (PMR) are the other major categories of idiopathic inflammatory myopathy. The incidence of these diseases is approximately 1 case among 100,000 persons.
A. Natural history and prognosis.
1. Polymyositis usually affects upper and lower girdle muscles in a symmetric pattern after the second decade of life. Patients with no family history of muscle weakness have subacute (weeks to months), progressive weakness of the deltoid, trapezius, neck flexor and extensor, biceps, triceps, iliopsoas, gluteus, quadriceps, and other muscles.
Patients characteristically have problems arising from a sitting position, washing their hair, or using stairs. Muscle pain may accompany the weakness. Pharyngeal muscle compromise can lead to dysphagia. The tongue, extraocular muscles, and facial muscles are usually spared. Sensation is not affected. Cardiac involvement can occur in as many as 40% of cases. Pulmonary involvement can result from primary weakness of respiratory muscles or from pulmonary interstitial fibrosis.
Polymyositis can occur in association with connective tissue and systemic autoimmune disorders. Polymyositis is not associated with an increased incidence of malignant disease. T-cell-mediated immunity plays a prominent role in the pathogenesis of polymyositis.
2. Dermatomyositis is characterized by a rash that accompanies or precedes muscle weakness. The characteristic skin abnormality is a heliotrope rash over the orbits and zygomatic arch with erythema on the rest of the face, upper trunk, and knuckles (Gottren’s papules). Subcutaneous nodular calcifications and dilated capillaries in the nail beds occur.
In children, extramuscular manifestations are more frequent than they are in adults. Dermatomyositis usually occurs alone but may be associated with systemic sclerosis, mixed connective tissue disease, or malignant lesions. Ten percent of patients with dermatomyositis will be found to have an underlying malignancy. It is a humorally mediated microangiopathic disorder with vascular deposition of immunoglobulin G (IgG), C3, and membrane attack complex. This suggests that the primary immunologic event is generation of antibodies against antigens within the walls of intramuscular blood vessels.
3. IBM involves distal and proximal muscles. Weakness and atrophy can be slightly asymmetric; quadriceps and finger flexors are commonly affected. It is a late-onset myopathy (sixth or seventh decade). It is treatment resistant. Occasionally, the diagnosis of IBM is made retrospectively when a patient fails to respond to treatment for polymyositis. Hereditary forms of IBM have been described: an autosomal dominant variant, which has a younger age of onset and an autosomal recessive variant, caused by a mutation in the GNE gene, on chromosome 9.
4. PMR affects elderly men and women with a peak incidence at 74 years of age. Patients describe diffuse muscle aching with neck and shoulder stiffness. Pain predominates over weakness or atrophy. Approximately 15% of the patients also have temporal arteritis. The erythrocyte sedimentation rate (ESR) is elevated to >40 mm per hour.
5. Noninfectious inflammatory myositis can also occur in the context of systemic lupus erythematosus, progressive systemic sclerosis, Sjögren’s syndrome, rheumatoid arthritis, mixed connective tissue disease, sarcoidosis, hypereosinophilic syndromes, and other disorders.
B. Diagnosis. In addition to the clinical features, the diagnosis of inflammatory myopathy is supported by results of measurement of muscle enzymes, electromyography (EMG), and muscle biopsy.
1. Muscle enzymes. Creatine kinase (CK) is released from the sarcoplasm into the serum after muscle destruction, and the level may be elevated as much as 50-fold in polymyositis/dermatomyositis. Other muscle enzymes such as lactate dehydrogenase, aldolase, and aminotransferases are commonly elevated. In IBM, CK level may be elevated as much as 10-fold or remain normal. In childhood dermatomyositis and in patients with myopathy associated with connective tissue diseases, CK levels may be normal. ESR should be determined, especially for suspected PMR.
2. The main value of EMG resides in its ability to show that peripheral neuromuscular weakness originates from the muscle itself and not from denervation or from a defect in neuromuscular transmission. It can also help ascertain the presence of disease activity. The classic EMG findings include short-duration, small-amplitude motor unit potentials, and increased insertional activity. These findings should not be considered specific for inflammatory myopathy, because they can also be found in acute toxic or metabolic myopathy and in dystrophy.
3. Muscle biopsy helps establish the diagnosis.
a. In polymyositis, light microscopic examination displays intrafascicular inflammatory infiltrates, necrosis, atrophy and regeneration of muscle fibers, and increased amounts of connective tissue.
b. In dermatomyositis, the inflammatory infiltrates are present around the vessels or in the interfascicular septa, and perifascicular atrophy is characteristic. Small blood vessels with hyperplastic endothelia may be occluded.
c. IBM is characterized by basophilic granular inclusions around the edges of vacuoles (rimmed vacuoles).
d. Muscle biopsy has limitations. Because of sampling error, a biopsy sometimes fails to disclose abnormalities expected from the clinical presentation.
C. Therapy.
1. Prednisone.
a. Administration. High-dose prednisone is the initial line of therapy for polymyositis and dermatomyositis. For prednisone, the recommended dosage is 1.0 mg per kg a day in a single daily dose for 30 to 60 days. The total dose should not exceed 100 mg. Daily administration should be used until there is unquestionable improvement muscle strength with recovery of ambulation. Then the dosage can be slowly reduced over 10 weeks to 1 mg per kg every other day. If no deterioration occurs, the dose is further reduced by 5 to 10 mg every 3 to 4 weeks until the lowest dose that controls the disease is reached. The dose should not be reduced if strength decreases. If treatment is effective, strength should improve within 3 months. If after 3 months of therapy no improvement has been achieved, prednisone should be tapered off and another immunosuppressant medication begun.
b. Side effects. Patients need to become acquainted with the numerous side effects of long-term prednisone treatment. Infections, fluid retention, potassium depletion, hypertension, diabetes, osteoporosis, premature cataracts, peptic ulcer disease, and skin bruising are some of the side effects that can occur.
Prevention of osteoporosis requires supplemental calcium gluconate or carbonate (500 to 1,000 mg per day) and calcitriol (0.2 to 0.5 mg per day) as well as axial exercise and adequate passive range of motion maneuvers. Bisphosphonates (alendronate, risedronate among others) can be used. A baseline dual energy X-ray absorption densitometry scan to measure bone density should be obtained for every patient before steroid treatment is started. The scan should be repeated every 6 months.
Proton pump inhibitors should be used to prevent peptic ulcers. Periodic eye examinations are needed for diagnosis of incipient cataracts or glaucoma. Periodic laboratory tests for serum glucose and electrolytes are recommended. Steroid myopathy, a side effect of long-term steroid use, will be addressed later in this chapter.
2. Sometimes patients with dermatomyositis need separate therapy for the rash.
3. Azathioprine is considered when complications preclude use of steroids, when the disease is not responding to adequate dosages of prednisone, or to add a steroid-sparing agent. A therapeutic response may take 3 to 6 months.
a. Administration. Azathioprine can be administered at 2 (up to 3) mg per kg a day. The initial dose should be approximately 50 mg per day, gradually increasing to BID dosing.
b. Side effects. The most common side effects of azathioprine are fever, nausea, rash. However, bone marrow suppression and liver toxicity can occur. CBC count with differential and platelets and liver function tests should be performed weekly for the first month and monthly thereafter. Patients with absent thiopurine S-methyltransferase (TPMT) enzyme activity are at high risk for azathioprine toxicity. TPMT testing is commercially available and may be considered before beginning treatment with azathioprine.
4. Mycophenylate mofetil inhibits proliferation of T and B lymphocytes and can be used as an alternative to azathioprine. Doses typically start at 500 mg per day and are increased to 1,000 mg twice a day. Side effects include immunosuppression, gastrointestinal side effects, hepatotoxicity and bone marrow inhibition, possible reactivation of chronic infection such as tuberculosis, and remote risk of malignant disease.
5. Methotrexate (15 to 25 mg per week by mouth) is used as another method to spare use of prednisone or if prednisone has not been effective. Hepatotoxicity, leukopenia, alopecia, stomatitis, and risk of neoplasia can occur. Methotrexate should not be used in patients with anti-Jo-1 antibodies and polymyositis because they are at increased risk for pulmonary fibrosis.
6. High-dose intravenous (IV) gamma globulin is effective in the management of polymyositis and in dermatomyositis. The recommended dosage is 0.5 mg per kg IV for 4 days, repeating each month as needed. Side effects include headaches, hypertension, acute renal failure, and hyperviscosity. Aseptic meningitis can occur and may respond to prednisone treatment. IgA-depleted preparations reduce the risk of reactions related to anti-IgA antibodies. Treatments are expensive. Despite these reservations, high-dosage IV gamma globulin might benefit patients who have been unresponsive to other medications.
7. In refractory cases, or if interstitial lung disease occurs, cyclophosphamide (1 to 2 g per m2 a month IV) may be considered. Side effects include nausea, vomiting, alopecia, hemorrhagic cystitis, teratogenicity, bone marrow suppression, carcinogenesis, and pulmonary fibrosis. Cyclophosphamide can also be used orally at doses of 1 to 2 mg per kg a day.
8. Rituximab, a CD20 monoclonal antibody that depletes B cells, is being investigated for use in resistant disease. It is administered at a dose of 375 mg per m2 every week for a month, with possible repeat courses in 6 months or 1 year. Opportunistic infections have occurred with this medication.
9. In PMR, prednisone rapidly provides benefits. Duration of treatment and dosage have to be individualized. In general, a starting dosage of 1 mg per kg a day should be appropriate. In patients suffering from temporal arteritis, corticosteroid treatment needs to be initiated immediately.
D. Prognosis. Dermatomyositis and, to a lesser degree, polymyositis are responsive to treatment, whereas IBM is usually resistant. Patients with interstitial lung disease have a higher mortality rate. When management of polymyositis is unsuccessful, the patient should be reevaluated and the muscle biopsy specimen reexamined to exclude IBM or muscular dystrophy of the limb-girdle type. Finally, it is important to emphasize the need to evaluate the patient’s strength and activities of daily living as measures of improvement, rather than simply adjusting treatment on the basis of CK levels alone.
II. VIRAL INFLAMMATORY MYOPATHY
Viruses and retroviruses can cause acute or subacute inflammatory myopathy.
A. Reye’s syndrome is acute encephalopathy with fatty degeneration of the liver that develops after varicella or influenza infections. This rare condition that affects children and adolescents begins with repeated vomiting and continues with confusion, lethargy, and coma. The mortality is high. There is acute liver dysfunction. The level of CK MM isoenzyme derived from skeletal muscle may be increased 300-fold. The level of CK correlates with prognosis. Salicylates may precipitate the syndrome. Treatment is supportive. The incidence of this syndrome has decreased precipitously over the years because aspirin is no longer being used to treat children with flu-like symptoms.
B. HIV may cause subacute or chronic myopathy early or late in relation to the infection. Proximal, symmetric involvement of lower or upper limbs manifests as weakness with or without atrophy. Serum CK levels may be elevated 10 to 15 times. The syndrome is almost identical to polymyositis. Thus, in the evaluation of patients with polymyositis, evaluation for HIV is recommended.
III. PARASITIC INFLAMMATORY MYOPATHY
In North America, trichinosis, cysticercosis, and toxoplasmosis are rarely the cause of a myopathy. These causes may need to be considered in an acute–subacute onset myopathy in an immunocompromised patient or one who has been in an endemic area or has had possible exposure to the parasite.
IV. PERIODIC PARALYSES
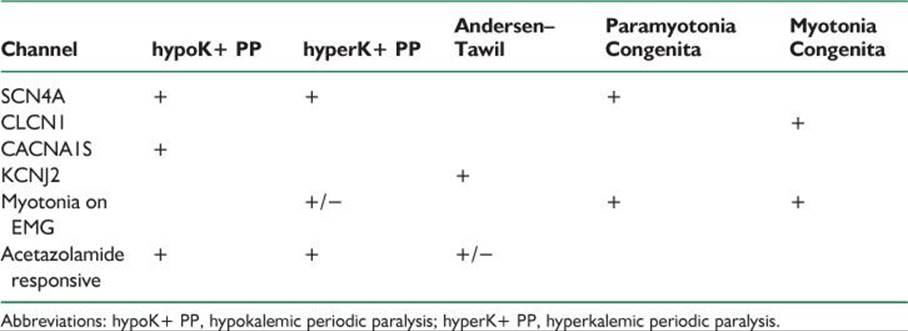
Periodic paralyses are disorders characterized by episodes of flaccid muscle weakness that can evolve into paralysis. Attacks usually last hours. Periodic paralysis is either a primary autosomal dominant disorder or a secondary disorder. The inherited forms of these diseases are caused by channelopathies, or defects in genes coding for muscle membrane ion channels (Table 47.1).
A. Natural history and prognosis.
1. Primary hypokalemic periodic paralysis affects young and middle-aged persons. Attacks usually occur at night or after strenuous exercise. On awakening, patients may be paralyzed and unable to get out of bed. The flaccid paralysis usually spares the respiratory and cranial muscles. During attacks, serum potassium level decreases. An ECG may reveal hypokalemic changes, including progressive flattening of T waves, depression of the ST segment, and appearance of U waves. Some patients eventually develop a progressive myopathy.
Seventy percent of patients with this disorder have a defect in the calcium channel gene: CACNA1S, which is located on chromosome 1q31. A small percentage of patients, however, have a sodium channel defect (SCN 4A). For approximately 20% of patients the gene defect is yet to be identified.
2. Secondary hypokalemic periodic paralysis.
a. Thyrotoxic periodic paralysis occurs 70 times more often in men than in women, despite the increased prevalence of hyperthyroidism among women. In nearly all cases, the condition is sporadic and the attacks cease when thyroid function is normalized. Every patient with hypokalemic periodic paralysis needs screening for thyrotoxicosis. This condition is more common in patients of Asian, Hispanic American, and Amerind origin.
TABLE 47.1 Channelopathies
b. Periodic paralysis secondary to urinary or gastrointestinal potassium loss can result from primary hyperaldosteronism, excessive thiazide therapy, excessive mineralocorticoid therapy for Addison’s disease, renal tubular acidosis, the recovery phase of diabetic coma, sprue, laxative abuse, villous adenoma of the rectum, or prolonged gastrointestinal intubation or vomiting.
3. Hyperkalemic periodic paralysis produces episodic attacks of weakness accompanied by elevations in serum potassium level (up to 5 to 6 mmol per L). It can be associated with myotonia (inability to relax the muscle) or paramyotonia (muscle stiffness worsened by exercise or cold). It is inherited in an autosomal dominant manner. Attacks start in the first decade of life. Patients usually have brief periods of generalized weakness. Static weakness is rare. Sustained mild exercise may prevent attacks. Cardiac arrhythmias can occur.
a. The genetic abnormality is a mutation in the SCN 4A (sodium channel) gene.
b. Needle EMG may detect myotonia, which supports the diagnosis.
4. Andersen’s syndrome (or Andersen–Tawil’s syndrome) is a triad of facial dysmorphism, long QT syndrome, and periodic paralysis. Short stature is often also present. Andersen syndrome is an autosomal dominantly inherited disease with a young age of onset and phenotypic variability. Fatal cardiac dysrhythmias may occur, making early recognition of this condition important. Mutations in the KCNJ2 gene, which codes for an inward-rectifying potassium channel, have been found in some patients.
5. Chloride channel mutations produce myotonia congenita with dominant and recessive forms (Thomsen and Becker, respectively), and with more myotonia than weakness. The dominant form manifests as painless muscle stiffness. Muscle stiffness is relieved after repeated exercise (warm-up), but it returns after rest. Cooling does not produce a significant change. These disorders result from missense mutations in the chloride channel gene CLCN1.
B. Prevention and therapeutic approach.
1. Primary hypokalemic periodic paralysis. Mild attacks may not require treatment. For attacks of general paralysis, oral potassium chloride can be used (0.25 mEq per kg), repeated every 30 minutes until the weakness is relieved. Muscle strength usually recovers within approximately 1 hour. IV potassium is not recommended because of the danger of cardiac arrhythmias and should be avoided.
For prevention of attacks, acetazolamide is the drug of choice, starting at 125 mg every other day, which can be increased to 250 mg three times a day. Side effects include increased incidence of nephrolithiasis, paresthesia, anorexia, and metallic taste. In severe cases, patients should eat a low-salt diet and be given the aldosterone antagonist spironolactone (100 mg twice a day) or triamterene (150 mg per day). Both drugs promote renal potassium retention.
2. Thyrotoxic periodic paralysis. Return to euthyroid status is curative. Propranolol (40 mg four times a day) and other β-adrenergic blocking agents may prevent attacks, possibly by suppressing the adrenergic overactivity induced by hyperthyroidism.
3. Hyperkalemic periodic paralysis.
a. Preventive measures consist of low-potassium diet, avoidance of fasting, and avoiding strenuous exercise. Slight exercise or ingestion of carbohydrates at the onset of weakness may prevent or abort attacks.
b. A thiazide diuretic, acetazolamide, or inhalation of a β-adrenergic agent (metaproterenol or salbutamol) may abort an attack. Dilantin (300 mg per day) can also be useful. For long-term preventive therapy, a thiazide diuretic or acetazolamide is recommended at the lowest possible dosage (hydrochlorothiazide, 25 mg every other day).
4. In myotonia congenita, mexiletine has been used. The starting dose is 150 mg by mouth twice a day, up to 1,200 mg per day. Mexiletine is contraindicated in the case of patients with second- and third-degree heart block; other cardiac arrhthymias can occur.
V. MUSCULAR DYSTROPHY
Muscular dystrophy is the term for inherited defects of cellular muscle structure, producing intrinsic muscle weakness. Some forms present at childbirth, others as late as the seventh decade. Family history, clinical examination, and temporal profile are necessary when considering a muscular dystrophy. The number of inherited dystrophies and the enormous variety of phenotypes prevent complete coverage in this forum. The following dystrophies will be discussed: X-linked dystrophinopathy (Duchenne and Becker muscular dystrophy), facioscapulohumeral dystrophy, myotonic, limb-girdle, and oculopharyngeal.
A. Natural history and prognosis.
1. Dystrophinopathy is an X-linked disorder caused by a mutation in the short arm, locus 21, of the X chromosome in the enormous gene that codes for the protein dystrophin. Dystrophin is a filamentous protein present in striated and cardiac muscle and other tissues. Although the role of dystrophin is not precisely known, anchoring and structural functions have been proposed for this protein.
In the most severe form of dystrophinopathy—Duchenne muscular dystrophy— almost no dystrophin is detected in skeletal muscle. In milder allelic forms—phenotypically denominated as Becker muscular dystrophy—some muscle fibers express dystrophin, which may be structurally abnormal. Almost all patients with dystrophinopathy are male. The disease can be caused by spontaneous mutations, which are more common than in other genetic disorders, probably because of the large size of the gene. Approximately 70% of patients with Duchenne and Becker muscular dystrophy have detectable mutations on routine DNA testing of peripheral blood. Deletions of varying sizes can be found in approximately 65% of cases; 5% of patients have gene duplications. A negative result of a DNA test does not exclude the diagnosis because approximately 30% of patients have an undetected mutation. The diagnosis in these patients depends on dystrophin analysis at muscle biopsy.
a. Duchenne muscular dystrophy affects children early in life. Motor developmental delay is noticeable after the first year, but muscle necrosis and serum enzyme elevation can be found in neonates. Onset of walking may be delayed past 15 months of age. Signs are present before the age of 5 years. They include difficulties in running and climbing stairs.
Children have hyperlordosis with prominent abdomen and calf pseudohypertrophy. Tiptoe walking is common. To stand up from the floor, patients use their hands (Gower’s sign). Joint contractures of the iliotibial bands, hip flexors, and heel cords develop in most patients by 6 to 9 years of age. By the age of 10 years, many of these patients lose the ability to walk or stand and must use a wheelchair. By the midteens they lose upper-extremity function. Cognitive dysfunction occurs in 10% of cases. The disease is usually fatal by the end of the second decade. Death is usually related to pulmonary infection, respiratory failure, or cardiomyopathy. Approximately 8% of female carriers have myopathy of the limb-girdle type. Female carriers may also have isolated cardiomyopathy.
Muscle biopsy specimens from patients with Duchenne muscular dystrophy have abnormal variations in fiber size, fiber splitting, central nuclei, and replacement by fat and fibrous tissues. The diagnosis of Duchenne’s muscular dystrophy can be confirmed by an absence of dystrophin immunostaining.
An EMG obtained early in the course of the disease shows findings compatible with those of myopathy. In end stage, there are decreased numbers of muscle fibers and the tissue can even become inexcitable.
b. Becker’s muscular dystrophy is a milder variety of dystrophinopathy in terms of severity and molecular abnormality. The diagnosis has been variably defined as either a patient who remains ambulatory or who has symptom onset after age 12. Patients may live many decades with mild to moderate symptoms, which can be indistinguishable from those of limb-girdle dystrophy. Patients are at risk for cardiomyopathy.
2. Facioscapulohumeral muscular dystrophy is an autosomal dominant disease that has high penetrance. It affects both men and women and starts before 30 years of age. Ninety-five percent of patients have a deletion in a sequence of a 3.3-kilobase repetitive unit (known at D4Z4) in chromosome 4q35.
Clinically, facial muscles are affected early. Bell’s phenomenon (failure of eyelids to close completely when the patient is sleeping or blinking) and drooping of the lower lip are noticeable. Patients may be unable to whistle. Facioscapulohumeral muscular dystrophy also involves the trapezius, rhomboid, and serratus anterior scapular muscles. Scapular winging is noticed with forward arm movement because of serratus anterior weakness. Deltoid function and rotator cuff muscles are better preserved. Patients typically seek medical attention because of the involvement of the shoulder rather than of the facial muscles. Lower-extremity weakness is found later in the disease.
This disorder has wide phenotypic variability, even within the same family. Some patients remain ambulatory all their lives, whereas others progress to using a wheelchair. The heart is usually spared. Trunk weakness may also occur.
3. Oculopharyngeal muscular dystrophy is an autosomal dominant disease of late onset. This syndrome manifests as ptosis and progressive dysphagia. Muscle biopsy shows rimmed vacuoles in muscle biopsy specimens, and tubulofilamentous inclusions within the striated muscle cell nucleus. The differential diagnosis includes myasthenia gravis and mitochondrial myopathies (Kearns–Sayre’s syndrome).
4. Limb-girdle muscular dystrophy is a heterogeneous collection of both autosomal recessive and autosomal dominant disorders that affect pelvic and upper girdle muscles and spare the face. Some disorders present in childhood, others into late adulthood.
5. Myotonic dystrophy is the most common muscular dystrophy among adults. Rather than being restricted to the skeletal muscle, it is a multisystemic, autosomal dominant disorder. It also involves the pancreas, gonads, thyroid, myocardium, and brain. Myotonic dystrophy is produced by a trinucleotide repeat expansion in chromosome 19 (19q13.2–13.3) that codes for myotonin protein kinase, a ubiquitous enzyme related to protein phosphorylation. The molecular gene defect consists of a CTG repeat expansion (>80) within the gene and the length of the repeat is correlated with the severity of the disease and inversely correlated with symptom onset. Clinical diagnosis is supported by the presence of myotonic discharges on EMG.
a. Muscle features. Weakness of facial muscles is typical. The face is hatched and thin with early frontal balding. Ptosis is present but is not as severe as in myasthenia gravis or Kearns–Sayre’s syndrome. Temporalis and masseter atrophy is a characteristic feature. Limb involvement is predominantly distal. Proximal limb muscles are usually preserved until the late stages. Myotonia, which is the delay of muscle relaxation after contraction, is present. Myotonia can be elicited by percussion of the thenar eminence or tongue. Patients may be unable to release their grip after a handshake.
b. Generalized features. Many patients have prominent systemic symptoms. Common abnormalities include cataracts, testicular atrophy, adult-onset diabetes mellitus, thyroid dysfunction, heart block, and arrhythmias. Hypersomnia and excessive daytime somnolence are reported and patients are often found to have mixed obstructive and central apneas. Because of the cardiorespiratory compromise, patients are susceptible to complications during surgery and anesthesia. Cognitive dysfunction, apathy, and lethargy are seen in more severely affected patients.
6. Myotonic dystrophy type 2 or proximal myotonic myopathy is an autosomal dominant disorder characterized by progressive weakness, myotonia, and cataracts. This is caused by a tetranucleotide repeat (CCTG) in the zinc finger protein 9 gene (ZNF9). These patients are a phenotypically milder form of myotonic dystrophy, with a later onset of symptoms.
7. Mitochondrial myopathies are a category of inherited diseases in which the genetic defect is either in the mitochondrial DNA or in a nuclear DNA gene that encodes for a protein involved in the mitochondrial respiratory chain. Because these defects all produce mitochondrial dysfunction, leading to decreased cellular energy production, patients manifest with symptoms related to oxidative stress. Seizures, encephalopathy, strokes, cardiomyopathy, muscle weakness (especially extraocular muscles), short stature, and hearing loss are common symptoms. The genetics underlying mitochondrial myopathies are complex. Mitochondrial DNA defects are maternally inherited and heteroplasmy (unequal distribution of affected mitochondria) may occur. Nuclear DNA genetic abnormalities follow Mendelian genetics.
Measurement of function of the components of the respiratory chain, muscle biopsy, MRI of affected organs, pedigree analysis, and genetic analysis of the mitochondrial genome can assist in making the diagnosis. Some of the more common phenotypes are mitochondrial encephalopathy with ragged red fibers, mitochondrial encephalopathy with lactic acidosis and stroke-like syndrome, chronic progressive external ophthalmoplegia, Kearns–Sayre’s syndrome, and Leigh’s disease.
B. Therapeutic approach.
1. Duchenne muscular dystrophy.
a. Family and patient education is important. A multidisciplinary clinic which specializes in muscular dystrophy can provide specialists and support to the patient and families.
Genetic counseling is recommended. Mothers and female siblings can be assessed for carrier status by assessment of serum CK or, if the patient has a documented genetic defect, through peripheral blood genetic analysis. However, negative results of mutation analysis in the mother do not rule out the risk of Duchenne muscular dystrophy affecting future pregnancies. Even with normal results of peripheral blood gene analysis, a mutation can be present in a percentage of oocytes (germline mosaicism).
b. Physical therapy is used to preserve mobility and to prevent early contractures. Passive range of motion exercises and adequate orthotics may prolong ambulation but do not stop disease progression.
Orthotics and splints can assist with managing contractures. All patients progress to wheelchair dependency: proper wheelchair assessments and fittings can lessen the development of scoliosis.
Patients with Duchenne muscular dystrophy are at high risk of side effects of general anesthesia. Succinylcholine or halothane should not be used because of the risk of episodes that resemble malignant hyperthermia. Adverse effects can be reduced with the use of nondepolarizing muscle relaxants.
c. Respiratory therapy. In later stages, noninvasive intermittent positive-pressure ventilation is useful, especially when patients retain carbon dioxide. Pulmonary exercises, use of a cough assist device and use of a respiratory vest may prevent pulmonary infections.
d. Medications. Prednisone (0.75 mg per kg a day) is recommended when children are still ambulatory to prolong this phase of life. Prednisone can improve neuromuscular strength after 1 month of treatment. The maximum effect is reached by 3 months. Side effects need to be addressed and close monitoring is needed.
e. Therapy for Becker muscular dystrophy follows principles similar to those of therapy for Duchenne disease, tailored to each patient’s level of strength.
2. In facioscapulohumeral muscular dystrophy and limb-girdle muscular dystrophy, treatment varies with individual patients. In patients with minimal symptoms, screening for cardiomyopathy and genetic counseling may be all that is needed. For patients with a foot-drop, ankle-foot orthoses may be prescribed. Physical therapy will be useful for range of motion, stretching, and gait assessment. When ambulation is impaired, a mobility evaluation can assess for wheelchair or motorized scooter needs.
3. In oculopharyngeal muscular dystrophy, blepharoplasty with resection of the levator palpebrae muscles may be needed. Dysphagia may be relieved with cricopharyngeal myotomy.
4. In myotonic dystrophy, only when myotonia is disabling, phenytoin (100 mg by mouth three times a day) can alleviate myotonia. In general, patients with myotonic dystrophy are not greatly concerned about the myotonia. The main goals are prevention and management of the systemic disease, especially cardiac arrhythmias.
5. Treatment of mitochondrial myopathies depends upon which organ systems are involved. Avoiding oxidative stress (hypoxia, ischemia, hypoglycemia and infection) may help prevent exacerbations or worsening. A “mitochondrial cocktail” of antioxidants and vitamins has been developed to promote respiratory chain function and includes co-enzyme Q10, riboflavin, creatine, carnitine, B complex vitamins, and vitamins E and C.
VI. METABOLIC MYOPATHY
Metabolic myopathies comprise a group of inherited disorders in which the defect is an alteration in the processing of carbohydrates or fats. Acid maltase deficiency, McArdle’s disease, and carnitine-O-palmitoyltransferase (CPT II) deficiency are reviewed (Table 47.2).
A. Natural history, prognosis, and treatment.
1. Acid maltase deficiency is an autosomal recessive glycogen storage disease caused by a deficiency in lysosomal α-glucosidase, which normally participates in the metabolism of glycogen into glucose. The infantile form is called Pompe disease; there is also a juvenile and adult onset form of this deficiency. Infants with Pompe disease have hypotonia, macroglossia, cardiomegaly, and hepatomegaly. Usually the infantile form is fatal. Adults suffer from slowly progressive myopathy with respiratory failure. Diaphragm, biceps, shoulder, and thigh adductor muscles are preferentially affected.
The cause of this disorder is a mutation in the acid alpha-glucosidase gene (GAA), located on chromosome 17q25. GAA enzyme activity evaluation is diagnostic, but in borderline cases, molecular genetic testing is available.
Treatment is now available for acid maltase deficiency, in the form of recombinant human α-glucosidase (Myozyme; Genzyme, Cambridge, MA, USA), given as repeated infusions. Enzyme replacement therapy has been clinically shown to lengthen the time before ventilator dependence in the infantile form. Adults have been documented to show increased strength and increased respiratory function.
2. McArdle’s disease, or myophosphorylase deficiency, affects children and adults and manifests with myalgia, fatigue, and muscle stiffness. Myoglobinuria and renal failure can develop. CK level is increased. EMG shows changes supportive of myopathy. The forearm ischemic exercise in affected patients shows no increase in venous lactate (as expected in normal controls). Muscle biopsy discloses subsarcolemmal deposits of glycogen. Immunohistochemistry staining of muscle biopsy tissue will show absence of myophosphorylase.
TABLE 47-2 Metabolic Myopathies
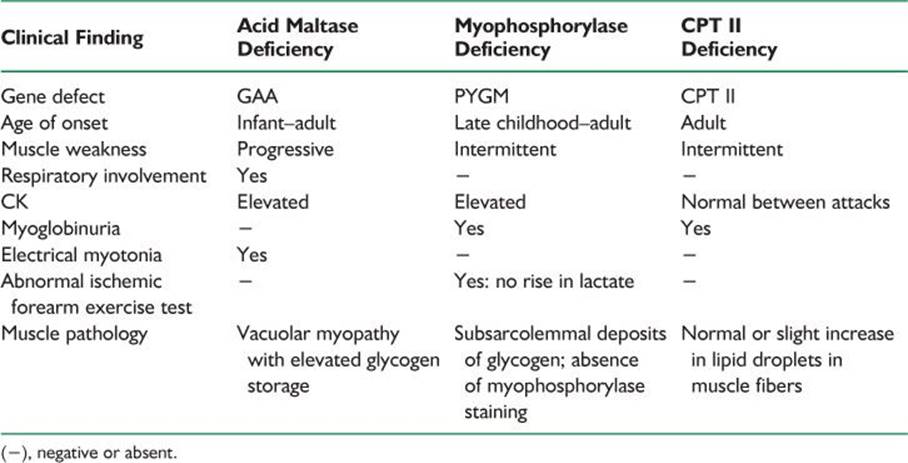
The disease is autosomal recessive, caused by homozygous or compound heterozygous mutations in the glycogen phosphorylase (PYGM) gene.
Prognosis is rather benign. Some patients can tolerate the deficits and learn to avoid brief, strong exercises that precipitate attacks. No definite treatment is available.
3. Carnitine-O-palmitoyltransferase II (CPT II) deficiency manifests in the adult patient with intermittent cramps, myalgia, and myoglobinuria. Renal failure, resulting from myoglobinuria, or respiratory failure may ensue. Between attacks, muscle strength is normal, CK and EMG are normal between attacks. The symptoms are precipitated by intense exertion. The capacity to perform short exercise is not impaired. Fasting, exposure to cold, high fat intake, viral infections, and general anesthesia can precipitate rhabdomyolysis. Increased lipid content may be seen on histochemical staining on muscle biopsy, but is not always present. The disease is autosomal recessive. The diagnosis is made by sequencing and mutation analysis of the carnitine palmitoyltransferase II (CPT2) gene. Therapy includes avoidance of triggers (general anesthesia, prolonged exercise, cold exposure and prolonged fasting) and a diet high in carbohydrates and low in fats.
VII. TOXIC MYOPATHY
Toxic myopathy is myopathy associated with either systemic disease or medication effect. Some medications produce direct muscle fiber necrosis, while others produce electrolyte imbalances with rhabdomyolysis. The most important types of toxic myopathy are necrotizing, autophagic, antimicrotubular, and steroid.
A. Natural history, prognosis, and treatment.
1. Endocrine myopathy.
a. Thyrotoxic myopathy manifests as weakness and little muscle wasting. Fatigue and heat intolerance are also present. Hypokalemic periodic paralysis (see IV.2.a.) and myasthenia gravis are associated with hyperthyroidism and should be included in the differential diagnosis. Treatment relies on correcting the hyperthyroid state; ß-adrenergic blocking agents may be of help. Glucocorticoids should be used in thyroid storm to block the peripheral conversion of thyroxine in triiodothyronine.
b. Hypothyroid myopathy manifests as enlargement of muscles, weakness, painful cramps, myoedema, and slow-recovery reflexes. This disease is more common among women. Rhabdomyolysis or respiratory muscle involvement may be present. Serum level of CK may be elevated. The diagnosis is supported by abnormal results of thyroid function tests. Treatment is to restore the euthyroid state.
2. Toxic necrotizing myopathy. The cholesterol-lowering drugs include 3-HMG-CoA reductase inhibitors (or statins) can cause a myopathy. Onset can be acute or insidious, often with myalgia, occasionally with myoglobinuria, and usually involving the proximal lower-extremity muscles. Patients with renal failure are especially predisposed. Elevated serum CK levels are common, and the EMG findings are abnormal. Muscle fiber necrosis with phagocytosis and small regenerating fibers are found on biopsy. When the medication is stopped, symptoms resolve in a few weeks to months. Asymptomatic elevations of CK level occur in about 1% of patients taking statins. Cyclosporine and tacrolimus have also been associated with toxic myopathy.
3. Autophagic myopathy occurs with chloroquine (and its derivatives), systemic lupus erythematosus, scleroderma, and rheumatoid arthritis, and with amiodarone. The myopathy of chloroquine affects the proximal lower-extremity muscles and is usually not painful. The course is subacute or chronic. The heart can be affected. Elevation in CK level and myotonic potentials on the EMG can be found. Muscle biopsy shows vacuoles (lysosomes), which stain for acid phosphatase and contain debris and curvilinear structures (autophagic vacuoles). With amiodarone, severe proximal and distal weakness may occur in combination with distal sensory loss, tremor, and ataxia. The treatment is to discontinue the medication.
4. Antimicrotubular myopathy is produced by colchicine and vincristine. These drugs bind to nerve and muscle tubulin. The toxic etiology of this myopathy is often not recognized because of the insidious onset in patients who may have been taking colchicine for years. Concomitant axonal sensorimotor neuropathy occurs. Weakness resolves slowly and may take 6 months after discontinuing the medication. Because of the neuromyopathy, a mixed pattern of denervation and myopathy is seen on electrophysiologic studies. Muscle biopsy shows acid-phosphatase-positive autophagic vacuoles.
5. The effects of zidovudine can be indistinguishable from the myopathy of HIV infection. It is due to mitochondrial toxicity associated with this agent. CK levels are normal or mildly elevated. Differentiation from AIDS myopathy can be difficult solely on a clinical basis. The muscle biopsy may show “ragged red fibers,” a sign of mitochondrial disease, and its presence supports the diagnosis of zidovudine myopathy. Treatment consists of stopping the medication. Whereas myalgia (muscle pain) is usually relieved within weeks of discontinuing zidovudine, muscle weakness can persist for months.
6. Steroid myopathy is a type-2 fiber atrophy of muscles associated with long-term corticosteroid exposure. Doses of prednisone >30 mg per day carry the risk of myopathy. Fluorinated compounds (triamcinolone, betamethasone, and dexamethasone) have a greater risk. Patients have predominantly proximal muscle weakness and atrophy. Serum level of CK is usually normal. EMG findings are normal. Muscle biopsy shows type 2 fiber atrophy, especially type 2B (fast twitch glycolytic). Tapering to an alternate-day regimen, use of “steroid-sparing” drugs (e.g., azathioprine), use of nonfluorinated steroids, and exercise may reduce the incidence of this myopathy.
In poly- or dermatomyositis, clinical worsening in a patient being treated with steroids may represent either a progression of the primary disease or the onset of steroid myopathy. The decision to raise or lower the prednisone dose has to be made after careful evaluation of the patient’s muscle strength, mobility, CK levels, and medication changes in the preceding months.
![]()
Recommended Readings
Amato AA, Darras BT, Greenberg SA, et al. Muscle diseases. AAN Continuum. 2006;12(3): 140–168.
Brooke MH. A Clinician’s View of Neuromuscular Diseases. Baltimore, MD: Williams & Wilkins; 1986.
Dalakas MC, Hohlfeld R. Polymyositis and dermatomyositis. Lancet. 2003;362(9388):971–982.
Griggs RC. Periodic paralysis. In: Wilson JD, et al., Wilson JD, Braunwald E, Isselbacher KJ et al., eds. Harrison’s Principles of Internal Medicine. 12th ed. New York, NY: McGraw-Hill; 1991:2121.
Griggs RC, Mendell JR, Miller RG. Evaluation and Treatment of Myopathies. Philadelphia, PA: FA Davis Co; 1995.
Jurkat-Rott K, Lehmann-Horn F. Paroxysmal muscle weakness: the familial periodic paralyses. J Neurol. 2006;253(11):1391–1398.
Kirschner J, Bonnemann CG. The congenital and limb-girdle muscular dystrophies: sharpening the focus, blurring the boundaries. Arch Neurol. 2004;61(2):189–199.
Machuca-Tzill L, Brook D, Hilton-Jones D. Clinical and molecular aspects of the myotonic dystrophies: a review. Muscle Nerve. 2005;32:1–18.
Mastaglia FL, Phillips BA, Zilko P. Treatment of inflammatory myopathies. Muscle Nerve. 1997;20:651–664.
Walsh RJ, Amato AA. Toxic myopathies. Neurol Clin. 2005;23:397–428.