Ramaprasad Srinivasan, Inger Lerra Rosner, and W. Marston Linehan
Renal cell cancer (RCC), a term that includes a variety of cancers arising in the kidney, comprises several histologically, biologically, and clinically distinct entities. Surgical resection for localized disease and immunotherapy for metastatic disease have been the mainstays of therapy for RCC. However, recent advances in our understanding of the molecular mechanisms underlying individual subtypes of the disease have led to newer, more effective, targeted approaches to managing metastatic RCC.
EPIDEMIOLOGY
■An estimated 64,000 new cases of cancer arising in the kidney and renal pelvis were diagnosed in the United States in 2012, leading to more than 13,000 deaths.
■Incidence is higher in men, with a male:female ratio of 1.6:1.
■Incidence from 2004 to 2008 increased by 4.1% per year in men and 3.3% per year in women, largely due to an increase in diagnosis of early-stage disease. Mortality has decreased during the same period by 0.4% per year in men and 0.6% in women.
■Largely a disease of adulthood, with a peak incidence after the fifth decade of life, RCC may also occur in children and infants.
ETIOLOGY AND RISK FACTORS
Nonhereditary Risk Factors
■Tobacco use. Up to one-third of cases in men and one-fourth of cases in women may be linked to smoking.
■Hypertension.
■Occupational exposure to trichloroethylene, cadmium, asbestos, and petroleum products.
■Obesity.
■Chronic kidney disease and acquired cystic disease of the kidney associated with long-term dialysis.
Genetic Predisposition/Familial Syndromes
Several familial kidney cancer syndromes have been identified. Although they represent a minority of RCC patients, individuals affected by these heritable disorders have a predisposition for developing kidney cancer, which is often bilateral and multifocal. Systematic evaluation of at-risk families has helped elucidate the molecular mechanisms underlying the origins of several types of kidney cancer. Several forms of sporadic kidney cancer have histologically similar familial counterparts with which they share aberrant oncogenic pathways. The following familial kidney cancer syndromes have been described:
von Hippel-Lindau Syndrome
■von Hippel-Lindau (VHL) syndrome is inherited in an autosomal-dominant pattern.
■Affected individuals have a predilection for developing a variety of tumors, including bilateral, multifocal renal tumors (clear cell RCC), pancreatic neuroendocrine tumors, renal and pancreatic cysts, CNS hemangioblastomas, retinal angiomas, pheochromocytomas, endolymphatic sac tumors, and epididymal/broad ligament cystadenomas.
■Genetic linkage analysis led to the identification of the VHL tumor suppressor gene located on chromosome 3p25. Affected individuals have a mutated/deleted allele of the VHL gene in their germ line. Acquisition of a somatic “second hit” that inactivates the normal copy of VHL leads to tumor formation in the affected organ(s).
Hereditary Papillary RCC
■Affected individuals have bilateral, multifocal type 1 papillary RCC. There are no known extrarenal manifestations of this disease.
■The underlying genetic alteration is an activating germ-line mutation in the MET proto-oncogene, located on the long arm of chromosome 7, accompanied by a nonrandom duplication of the aberrant chromosome 7 (resulting in trisomy 7).
■Patients usually present with renal tumors in or beyond the fifth decade of life, although an early-onset form that presents in the second or third decades has also been described.
Birt-Hogg-Dube Syndrome
■Affected individuals are at increased risk of developing cutaneous fibrofolliculomas, pulmonary cysts predisposing to the development of spontaneous pneumothoraces, and renal tumors.
■Several histologic types of renal tumors have been described in Birt-Hogg-Dube (BHD) syndrome, including chromophobe (34%), hybrid chromophobe-oncocytomas (50%), clear cell, and oncocytomas.
■The BHD gene, localized to chromosome 17p11, encodes a protein known as folliculin. Identification of somatic “second hit” mutations in BHD/folliculin indicates that this gene may function as a tumor suppressor.
Hereditary Leiomyomatosis and RCC
■Hereditary leiomyomatosis and RCC (HLRCC)–affected individuals have a predisposition to developing multiple cutaneous and uterine leiomyomas, as well as papillary RCC.
■Renal tumors are often solitary.
■Sometimes described histologically as a type of papillary type 2 RCC; may be mistaken for collecting duct RCC. The distinctive histopathologic hallmark of these tumors is the presence of a large nucleus with a prominent orangiophilic nucleolus surrounded by a halo.
■Tumors tend to metastasize early and have a characteristically aggressive clinical course.
■The underlying defect is a germ-line mutation in the gene for the Krebs cycle enzyme fumarate hydratase (FH), located on chromosome 1. Loss of FH and the accompanying alteration in Krebs cycle function result in a metabolic switch characterized by a reliance on aerobic glycolysis for cellular energy needs (Warburg effect). Other critical cellular events associated with loss of FH include dysregulated HIF1-α expression and downregulation of AMPK, a key cellular energy sensor.
Succinate Dehydrogenase and RCC
■Succinate dehydrogenase is a multiunit mitochondrial enzymatic complex that catalyzes the conversion of succinate to fumarate in the Krebs cycle.
■Germ-line mutations in the genes encoding SDHB, SDHC, and SDHD have been identified in patients with hereditary forms of kidney cancer. Patients with germ-line SDHB mutations are also at risk for developing pheochromocytomas and paragangliomas.
■Loss of SDH activity leads to impaired Krebs cycle function and leads to metabolic and biochemical alterations similar to that seen with FH inactivation.
Other Genes Associated with Hereditary Kidney Cancer
■Mutations in multiple genes involving the LKB1/TSC/mTOR are associated with familial forms of RCC.
■Mutations in the genes responsible for tuberous sclerosis complex (TSC1/2) have been associated with kidney cancer. While the majority of renal tumors resulting from TSC mutations are benign (angiomyolipomas), clear cell, papillary, and other subtypes of RCC have also been described.
PATHOLOGIC CLASSIFICATION
Based on histopathologic features, RCC is divided into the following subtypes:
■Clear cell RCC. The most common variety, comprising 70% to 80% of all kidney cancers. Composed predominantly of cells with a clear cytoplasm.
■Papillary RCC. Further divided into type 1 and type 2 based on morphologic appearance. Represents approximately 10% to 15% of all kidney cancers.
■Chromophobe RCC. Represents approximately 5% of all malignant renal neoplasms. Characterized histologically by the presence of sheets of cells with pale or eosinophilic granular cytoplasm.
■Collecting duct RCC. Rare (<1%) variant believed to originate in the collecting system. Medullary RCC, which has some features suggestive of collecting duct RCC, is seen almost exclusively in patients with sickle cell trait and is characterized by an aggressive clinical course.
■Unclassified. Represents approximately 3% to 5% of renal tumors. Lack distinct features of a particular subtype or variant.
■Renal tumors with sarcomatoid features do not comprise a separate entity. Instead, they represent localized or diffuse sarcomatoid differentiation of one of the subtypes of RCC. Generally associated with poor prognosis.
MOLECULAR MECHANISMS
Identifying familial kidney cancer syndromes was an important step in unraveling the complex aberrant pathways leading to the development of several types of both hereditary and sporadic RCCs. This has enabled the development of therapeutic agents that target pathways critical to the development and growth of these tumors.
Clear Cell RCC
■Germ-line mutations in the VHL gene are the hallmark of VHL syndrome.
■The vast majority of patients with sporadic clear cell RCC show evidence of VHL inactivation in tumor tissue resulting from either mutation or promoter hypermethylation. The absence of functionally active VHL protein has several consequences, the best understood of which is the accumulation of a group of transcription factors called hypoxia-inducible factors (HIF).
■Increased intracellular HIF leads to transcriptional upregulation of several proangiogenic, growth and survival factors, such as vascular endothelial growth factor (VEGF), platelet-derived growth factor (PDGF), transforming growth factor-alpha (TGF-α), and the glucose transporter glut-1. This sequence of events appears to be important in the genesis and propagation of clear cell RCC.
■Several components of this pathway are targets for novel therapeutic agents.
Type 1 Papillary RCC
■MET is a cell surface receptor normally activated on binding its ligand, hepatocyte growth factor (HGF). The HGF/MET axis mediates a variety of biologic functions including cell growth, proliferation, and motility. Activating mutations in the MET proto-oncogene (which render the receptor constitutionally active) are responsible for the bilateral, multifocal, type 1 papillary renal tumors seen in patients with hereditary papillary RCC (HPRC).
■Activating somatic mutations in the tyrosine kinase domain of Met have also been identified in 10% to 15% of patients with sporadic papillary RCC. Duplication of chromosome 7, where genes for both MET and HGF are located, is seen more frequently in sporadic papillary tumors (approximately 70% in one series) and may represent an alternative mechanism for activation of the HGF/Met pathway.
■Agents targeting the MET pathway are currently being evaluated in patients with papillary RCC.
Type 2 Papillary RCC
■Includes tumors with papillary architecture but with features inconsistent with type 1 papillary tumors. Patients with HLRCC are at risk for developing renal tumors, which are sometimes described as type 2 papillary RCC.
■The underlying molecular defect in HLRCC-related tumors is inactivation of the Krebs cycle enzyme FH, leading to accumulation of its substrate fumarate. Fumarate interferes with HIF degradation and leads to its accumulation and consequent transcriptional activation of its target genes (VEGF, PDGF, TGF-α, etc.). While no sporadic counterpart for this tumor has been described, it is speculated that some sporadic type 2 tumors may be associated with impaired Krebs cycle activity.
Chromophobe RCC
■The precise biochemical aberrations underlying chromophobe RCC are being investigated; however, patients with BHD often present with chromophobe renal tumors.
■The gene for BHD (folliculin) appears to interact with the mTOR and AMPK pathways, which may be important in chromophobe tumors and, potentially, other histologic RCC subtypes seen in BHD.
Other Subtypes
■Other histologic subtypes of RCC include (1) medullary RCC, seen almost exclusively in association with sickle cell trait, and (2) collecting duct RCC, which shares similarities with upper urinary tract tumors.
■Translocation RCCs are so named because of the presence in these tumors of characteristic translocations involving members of the Microphthalmia Transcription Factor/Transcription Factor E (MITF/TFE). In its most common form, tumors exhibit translocations involving TFE3. These tumors are more common in children and young adults and can exhibit aggressive clinical behavior with a propensity for early metastasis.
CLINICAL PRESENTATION
■Many renal masses are found incidentally during evaluation for unrelated medical issues or metastatic foci.
■Only 10% of patients present with the classic triad of hematuria, pain, and flank mass.
■Initial presentation may be a paraneoplastic syndrome or laboratory abnormality, including elevated erythrocyte sedimentation rate, weight loss/cachexia, hypertension from increased renin, anemia, hypercalcemia (release of PTH-like substance), elevated alkaline phosphatase, polycythemia (increased erythropoietin), and Stauffer syndrome (reversible, nonmetastatic hepatic dysfunction that usually resolves once the primary tumor is removed).
■Approximately 50% of RCC patients present with localized disease, 25% with locally advanced disease, and 25% to 30% with metastatic disease.
■Common sites of metastatic spread include lung (70% to 75%), lymph nodes (30% to 40%), bone (20% to 25%), liver (20% to 25%), and CNS.
DIAGNOSIS AND EVALUATION
■Initial workup for a patient with a renal mass includes a history and physical examination, complete blood count with differential, full chemistry panel, and PT/PTT.
■CT scan of the abdomen and pelvis, with and without contrast, is standard for evaluating the renal mass and regional lymph nodes. If the CT scan suggests renal vein and/or inferior vena cava involvement, an MRI of the abdomen and chest imaging is warranted.
■Chest x-ray is also recommended. Chest CT is indicated in the presence of an abnormal x-ray, a large primary tumor, or symptoms suggestive of pulmonary or mediastinal involvement such as cough, hemoptysis, or chest pain.
■Bone scan is indicated in patients with elevated alkaline phosphatase, hypercalcemia, pathologic fracture, or bone pain.
■MRI of the brain is usually reserved for patients with clinical features suggesting brain metastases, but is increasingly performed in some centers as part of initial staging in asymptomatic patients with known metastatic disease.
STAGING
The most commonly used system for staging RCC is the Tumor–Lymph Node–Metastasis (TNM) staging system outlined by the American Joint Committee for Cancer (AJCC). Stage I disease encompasses any tumor not greater than 7 cm in greatest dimension and is limited to the kidney. Stage II includes any tumor greater than 7 cm in greatest dimension but is limited to the kidney. Stage III disease is present if there are metastases to regional lymph nodes, or the tumor extends into major veins or perinephric tissues but not the ipsilateral adrenal gland nor Gerota fascia. Stage IV disease includes any distant metastasis or tumor invading beyond Gerota fascia or contiguous extension into the ipsilateral adrenal gland.
PROGNOSTIC FACTORS
■Several tumor and patient characteristics appear to influence outcome for patients with localized kidney cancer. Nomograms based on factors such as tumor stage and nuclear grade, tumor histology, mode of presentation, and performance status are used to predict the risk of disease recurrence following nephrectomy. Several such nomograms are currently available and are gaining acceptance in both clinical practice and clinical trial design as an effective means of risk stratification.
■In patients with metastatic disease, clinical characteristics (performance status, prior nephrectomy, number of metastatic sites, etc.) as well as laboratory parameters (serum lactate dehydrogenase, serum calcium, hemoglobin, etc.) are predictive of survival. A widely used prognostic model based on outcomes in patients treated with either cytokines or chemotherapeutic agents (Memorial Sloan-Kettering Cancer Center prognostic criteria) implicates the following features in poor outcome:
– Poor performance status (Karnofsky PS <80)
– Elevated LDH (>1.5 × upper limit of normal)
– Elevated corrected calcium (>10 mg/dL)
– Low hemoglobin (< lower limit of normal)
– Absence of prior nephrectomy
■The presence or absence of one or more of these prognostic features allows stratification of patients into the following prognostic categories:
– Favorable: No risk factors, median survival 19.9 months
– Intermediate: One or two risk factors, median survival 10.3 months
– Poor: Three to five risk factors, median survival 3.9 months
■A similar prognostic scheme has been proposed for stratification of patients receiving agents targeting the VEGF and mTOR pathways, but remains to be independently validated.
TREATMENT OF LOCALIZED RCC
Surgery
■For patients with early-stage localized RCC, surgical resection is often curative; for small renal masses (<4 cm) a partial nephrectomy/nephron-sparing surgery is typically performed using an open, laparoscopic, or robotic assisted approach.
■For tumors >4 cm, radical nephrectomy (open or laparoscopic procedure) is the treatment of choice. However, recent literature supports nephron-sparing procedures for tumors 4 to 7 cm in selected patients. Patients with primary tumors larger than 7 cm and disease localized to the kidney generally undergo a radical nephrectomy with curative intent.
■Active surveillance of small renal masses is also an alternative option in selected patients including the elderly and those with significant competing health risks and comorbidities.
■Less invasive techniques such as radiofrequency ablation and cryotherapy are being evaluated and may be effective in eradicating smaller renal tumors; however, studies demonstrate an increased risk of local recurrence when compared to surgery.
Adjuvant Therapy
Although a variety of agents such as cytokines and vaccines have been evaluated in the adjuvant setting, none has proved effective in reducing the risk of recurrence or improving long-term outcome. Ongoing adjuvant trials are evaluating the role of targeted therapeutic agents such as sunitinib, pazopanib, everolimus, and sorafenib in patients with high-risk disease following resection of the primary tumor.
TREATMENT OF METASTATIC RCC
Surgery
■In selected patients with isolated metastases, surgical resection may provide extended disease-free periods. Five-year survival rates of 30% to 50% have been reported in retrospective analyses using this approach.
■Cytoreductive nephrectomy preceding systemic cytokine therapy has been the subject of several studies. At least two randomized phase 3 trials have demonstrated a survival advantage in patients receiving interferon-alpha (IFN-α) following nephrectomy versus patients receiving IFN-α alone. Careful patient selection is key to the success of this approach, and patients with limited metastatic burden, favorable tumor kinetics, and good performance status are most likely to benefit. Cytoreductive nephrectomy as a prelude to antiangiogenic targeted therapies is currently under evaluation in randomized phase 3 trials.
■Cytoreductive nephrectomy can be performed for palliation of intractable hematuria and pain associated with RCC.
Systemic Therapy
■Conventional cytotoxic chemotherapy is ineffective in the vast majority of patients with metastatic RCC (approximately 5% to 6% overall response rate with single agent) and is not part of the standard approach to this disease. However, some patients with sarcomatoid variants of RCC are responsive to gemcitabine-based regimens.
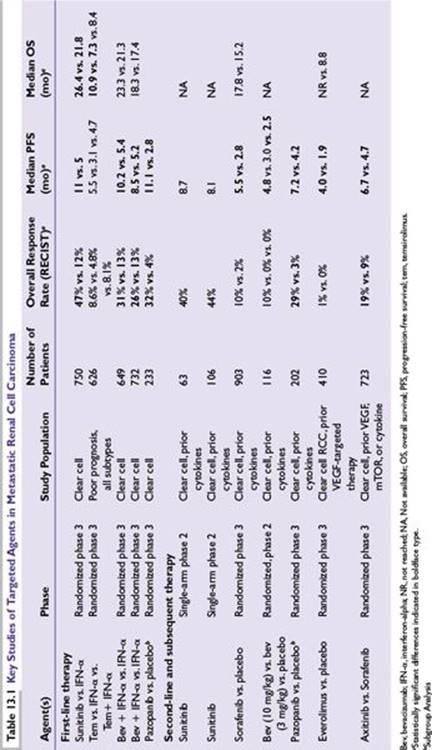
■Targeted agents directed against the VEGF/PDGF and mammalian target of rapamycin (mTOR) pathways have been evaluated in patients with metastatic RCC and have largely supplanted cytokines as standard first-line agents in the management of clear cell RCC (Table 13.1). The standard initial approach for most patients with metastatic clear cell RCC is treatment with small molecule inhibitors of angiogenesis, although cytokine-based therapy with interleukin (IL)-2 should be considered in selected patients.
■There are currently no standard treatments for non–clear cell variants, although several interesting mechanism-based approaches are under investigation.
VEGF Pathway Inhibitors
Upregulation of proangiogenic factors such as VEGF and PDGF is an important consequence of VHL inactivation and provides the basis for the efficacy of anti-VEGF agents in RCC. Several agents targeting the VEGF pathway are approved by the US FDA for the treatment of metastatic RCC.
Sunitinib
■An oral tyrosine kinase inhibitor with potent activity against VEGF receptor 2 (VEGFR-2) and PDGF receptor (PDGFR).
■Initial single-arm phase 2 studies demonstrated a remarkably high overall response rate of 30% to 40% in patients with cytokine-refractory disease. A randomized phase 3 study comparing sunitinib with IFN-α in previously untreated clear cell RCC patients has demonstrated a significantly higher response rate (47% vs. 12%), improved progression-free survival (PFS) (median 11 vs. 5 months), and superior overall survival (OS) (26.4 vs. 21.8 months) with sunitinib.
■Dosage is 50 mg per day over 4 weeks, followed by a 2-week rest period.
■Fairly well tolerated by the majority of patients. Common side effects include hypertension, fatigue, cutaneous side effects (rash, hand–foot syndrome), gastrointestinal symptoms (nausea, vomiting, diarrhea, anorexia, constipation), and cytopenia.
■Sunitinib is one of the most widely used first-line agents in metastatic clear cell RCC.
Pazopanib
■Oral angiogenesis inhibitor that targets VEGFR-1, -2, and -3. Approved in 2009 by the FDA for the treatment of advanced RCC.
■In a phase 3 trial of patients with advanced RCC with 0 or 1 prior cytokine treatment comparing pazopanib versus placebo, PFS was increased in the pazopanib group (9.2 months) compared to the placebo cohort (4.2 months). In subgroup analyses, the improvement in PFS was seen in both treatment naïve patients and those who had received prior cytokine therapy.
■Adverse reactions include diarrhea, hypertension, nausea, fatigue, and abdominal pain. Hepatotoxicity with elevated transaminases was seen, and a small number of deaths from hepatic failure were noted on study. Liver function testing should be performed while on therapy.
•Although the adverse events seen with sunitinib and pazopanib are similar, head-to-head comparison in randomized studies suggested that pazopanib was better tolerated, with both patients and physicians indicating a preference for this agent over sunitinib based on better tolerability. Furthermore, in a phase 3 randomized study, the efficacy of pazopanib in clear cell RCC patients was shown to be noninferior to that seen with sunitinib.
Axitinib
■A highly selective oral tyrosine kinase inhibitor that targets VEGFR 1, 2, and 3. Approved by the FDA in 2012 for the treatment of advanced RCC in patients who had previously failed one prior systemic therapy.
■A phase 3 trial compared the efficacy of dose-escalated axitinib versus standard dose sorafenib following first-line treatment with either sunitinib, bevacizumab plus IFN-α, temsirolimus, or cytokine therapy. The PFS was 6.7 months in patients on axitinib versus 4.7 months in patients on sorafenib. In subgroup analysis the median PFS was consistently improved over the sorafenib group regardless of prior treatment, although the difference was more pronounced in patients who had received prior cytokine therapy.
■The agent appears to be fairly well tolerated. Adverse effects include hypertension, diarrhea, dysphonia, nausea, fatigue, and hand–foot syndrome.
•Treatment-related hypertension has been proposed as a clinically evaluable pharmacodynamic marker in patients receiving axitinib as well as other agents targeting the VEGF pathway. Retrospective studies demonstrate a correlation between the occurrence of hypertension (thought to indicate adequate plasma levels of the agent and consequently optimal inhibition of the VEGF pathway) and outcome.
•The effect of dose titration of axitinib on plasma levels of the agent and on treatment outcome is being currently evaluated in a phase 2 randomized study.
Bevacizumab
■A monoclonal antibody against VEGF-A, approved by the FDA in 2009 for the treatment of advanced RCC in combination with IFN-α.
■A randomized, three-arm phase 2 study comparing two different doses of bevacizumab (10 mg/kg and 3 mg/kg IV every 2 weeks) and placebo in cytokine-refractory patients showed a PFS advantage favoring the 10 mg/kg arm (4.8 vs. 2.5 months).
■Two multicenter randomized phase 3 studies with similar trial designs comparing IFN-α alone (9 million international units SC 3 times per week) versus the same dose of IFN-α plus bevacizumab (10 mg/kg IV every 2 weeks) showed superior PFS in the combination arm (5.4 vs. 10.2 months). These improvements in PFS did not appear to translate into significant OS benefits in either of these trials.
■Side effects include hypertension, headache, nosebleeds, headaches, and proteinuria and in some cases gastrointestinal perforation and difficulty with wound healing.
Sorafenib
■An oral tyrosine kinase inhibitor with activity against c-Raf, VEGFR-2, and PDGFR.
■A randomized phase 2 study showed significant improvement in PFS versus placebo (median 24 vs. 6 weeks) in patients with cytokine-refractory metastatic RCC. This finding was confirmed in a randomized phase 3 trial of sorafenib versus placebo (median PFS 5.5 vs. 2.8 months). OS was similar in the two groups (17.8 vs. 15.2 months) and may have been influenced by the trial’s crossover design (patients progressing on placebo could cross over to the sorafenib arm).
■A randomized phase 2 study in metastatic, untreated clear cell RCC failed to demonstrate the drug’s superiority over IFN-α.
■Typically administered at a dose of 400 mg twice a day. Adverse events are similar to those of sunitinib.
■Provides a reasonable option for patients who have failed sunitinib and/or other first-line agents.
mTOR Pathway Inhibitors
The mTOR inhibitors temsirolimus and everolimus are rapamycin analogs believed to act at least in part by downregulating mTOR-dependent translation of HIF.
Temsirolimus
■A prodrug of rapamycin-administered IV.
■The most convincing evidence for the activity of this drug in RCC comes from a randomized phase 3 trial of 626 patients with previously untreated high-risk metastatic RCC (defined as the presence of three or more poor prognostic criteria). All histologic subtypes of RCC were included in this trial. Patients were randomized to receive temsirolimus 25 mg IV per week or temsirolimus 15 mg IV per week plus IFN-α (6 million international units 3 times per week) or IFN-α alone (18 million international units three times per week as tolerated). Single-agent temsirolimus was associated with significantly prolonged disease-free survival and OS compared to IFN-α alone (median OS 10.9 vs. 7.3 months). An exploratory subgroup analysis suggested that both patients with clear cell and those with non–clear cell RCC benefited from temsirolimus. The combined temsirolimus/IFN-α arm had superior disease-free survival compared to IFN-α alone, but there was no difference in OS between the two groups.
■Common adverse events include rash, fatigue, mucositis, hyperglycemia, hypercholesterolemia, and interstitial pneumonitis. Rapamycin analogs are also associated with a risk of immunosuppression.
■Single-agent temsirolimus is a reasonable first-line option for patients with poor-prognosis RCC.
Everolimus
■An oral rapamycin analog.
■In a randomized phase 3 trial of metastatic RCC patients who had progressed on front-line VEGF-targeted therapy, everolimus improved disease-free survival compared to placebo (4 vs. 1.9 months).
■Side effects are similar to those of temsirolimus.
■Both temsirolimus and everolimus are reasonable treatment options for patients who have progressed on sunitinib or other VEGF antagonists.
Cytokines
Until the advent of VEGF-targeted therapy, cytokines were the mainstay of treatment for metastatic clear cell RCC. High-dose IL-2 and IFN-α are the most studied agents in this class.
IL-2
Since the early 1980s, numerous studies have demonstrated the efficacy of IL-2 in patients with metastatic RCC.
■High-dose IL-2 (600,000 to 720,000 international units/kg every 8 hours as tolerated up to a maximum 15 doses) has shown an overall response rate of 15% to 20%, with complete responses in 7% to 9% of patients. Since only a small subset of patients appears to benefit from this agent, no survival advantage has been demonstrated in randomized trials. However, most complete responses were durable, with very few recurrences noted during long-term follow-up. IL-2 is FDA approved for treatment of RCC.
■Responses to IL-2 are best characterized in patients with clear cell histology; its role in other subtypes of RCC is unclear.
■The major limitation of IL-2 is toxicity associated with the high-dose regimen. A high incidence of serious and life-threatening but often reversible complications (notably vascular-leak syndrome, hypotension, multiorgan failure, etc.) occurred in early trials, with resultant mortality rates of 1% to 5%. However, further experience with IL-2 has led to better management of side effects. A report of over 800 patients treated at the National Cancer Institute reported no treatment-related mortality.
■IL-2 has been evaluated in combination with a variety of other modalities, including cellular therapy with lymphokine-activated killer cells and tumor-infiltrating lymphocytes, chemotherapy, interferon, etc. However, combining any of these therapies with high-dose IL-2 appears to provide no additional benefit.
■Lower doses of either IV or SC IL-2 have been evaluated to determine if toxicity could be reduced without compromising efficacy. At least two randomized trials have demonstrated that lower-dose IL-2 leads to fewer responses and, more importantly, a decline in durable complete responses.
■Despite the availability of newer, better tolerated, VEGF-targeted agents, high-dose IL-2 remains a reasonable first-line option for selected patients with metastatic clear cell RCC.
IFN-α
■The overall response rate in treatment-naïve RCC patients treated with recombinant IFN-α is approximately 15%.
■Administered SC in a variety of dosages (5 to 18 million international units) and regimens (three to five times per week).
■Limited long-term follow-up data; durable complete responses relatively rare.
■Common side effects include constitutional symptoms, gastrointestinal toxicity, elevated hepatic transaminases, and bone marrow suppression.
■Several studies evaluating combined IL-2 and IFN-α have demonstrated no survival benefit over single-agent cytokine therapy.
■Single-agent IFN-α has fallen out of favor due to associated toxicity and the availability of more effective agents.
Allogeneic Stem Cell Transplantation
■Investigated in metastatic RCC to test the hypothesis that this malignancy may be susceptible to alloimmune donor-mediated graft-versus-solid tumor effects.
■Several groups have reported overall response rates of up to 30% to 40%, including some durable complete responses following nonmyeloablative or reduced-intensity conditioning peripheral blood stem cell transplants.
■Transplant-related morbidity and mortality and the availability of HLA-matched donors are limitations to this current investigational approach.
PD-1/PD-1 L Inhibitors
■Activation of inhibitory T cell receptors such as Programmed Death-1 (PD-1) is believed to play a major role in mediating resistance of some tumors to immune surveillance.
■Inhibitors of PD-1 as well as one of its activating ligands, PDL-1, are currently undergoing clinical evaluation. In an early-phase clinical trial, these agents have shown promising activity in patients with clear cell RCC who have progressed on conventional therapy (overall response rate of 27% with many durable responses in one study).
■Further studies are expected to define the role of these agents in the management of clear cell RCC.
Non–Clear Cell RCC
■There are currently no standard systemic options of proven benefit for the treatment of patients with advanced RCC of non–clear cell histology.
■Retrospective analyses and small phase 2 trials indicate that inhibitors of the VEGF and mTOR pathways are associated with modest activity in some subtypes. A subgroup analysis of patients from the phase 3 ARCC trial suggested that patients with poor-risk, non–clear cell RCC had better outcomes when treated with temsirolimus compared to IFN-α. The efficacy of the oral mTOR inhibitor, everolimus, is currently being evaluated in a phase 2 study in patients with papillary RCC.
■In a large phase 2 trial, foretinib, a novel inhibitor of MET and VEGFR2, was associated with activity in patients with papillary RCC, with an overall response rate of 13.5% and a median PFS of 9.3 months. Efficacy was most pronounced in patients with papillary type 1 RCC carrying a germ-line mutation in MET (overall response rate 50%), although patients without this alteration also appeared to benefit to some extent.
■A better understanding of the molecular changes driving individual subtypes of non–clear cell tumors is likely to lead to the development of mechanism-based treatment strategies for each histologic/molecular variant.
ACKNOWLEDGMENT
This research was funded by the Intramural Research Program of the National Institutes of Health, National Cancer Institute, Center for Cancer Research, Bethesda, Maryland.
REVIEW QUESTIONS
1.In which of the following patients with metastatic RCC is cytoreductive nephrectomy most appropriate?
A.A 50-year-old male with an ECOG performance status of 0, a large 12 cm right renal mass, and four small pulmonary metastases
B.A 67-year-old female with an ECOG performance status of 0, a 7 cm left renal mass, retroperitoneal adenopathy, and hepatic metastases that have doubled in size over 4 weeks
C.An 81-year-old man with an asymptomatic 6 cm right renal mass, and multiple hepatic metastases who has declined sytemic therapy
D.A 72-year-old man with an ECOG performance status of 2, a 5 cm right renal mass, and mild dyspnea associated with numerous pulmonary metastases
2.Which of the following regarding IL-2 therapy for metastatic RCC is true?
A.IL-2 has demonstrable efficacy in clear cell as well as papillary RCC.
B.Randomized studies have demonstrated an OS benefit associated with high-dose IL-2.
C.Low-dose subcutaneous (SC) and high-dose intravenous (IV) IL-2 have comparable efficacy.
D.Durable complete responses are seen in a small proportion of patients receiving high-dose IL-2.
E.Newer formulations have led to better tolerability of high-dose IL-2.
3.A 68-year-old man undergoes a right radical nephrectomy for a 12 cm renal mass identified during evaluation of flank pain and unexplained weight loss. Histopathologic evaluation is consistent with clear cell RCC, Fuhrman grade III. Routine surveillance imaging 2 years later reveals multiple bilateral pulmonary nodules and three 2 to 3 cm liver masses. Biopsy of a liver lesion is consistent with clear cell RCC and he is referred to a medical oncologist for discussion of systemic therapy options. He has no symptoms attributable to metastatic disease, and his CBC and chemistry panel are normal. Reasonable treatment options, based on demonstration of clinical benefit in randomized phase 3 trials, include which of the following?
A.High-dose IL-2
B.Sunitinib
C.Axitinib
D.Temsirolimus
E.Everolimus
4.A 55-year-old woman is referred to you for further management of metastatic type 2 papillary RCC. She was initially diagnosed at the age of 54 when she underwent a left radical nephrectomy and retroperitoneal lymph node dissection for an 8 cm renal mass and associated regional lymphadenopathy. Complete staging evaluation at the time also revealed extensive mediastinal and hilar adenopathy and multiple bone lesions consistent with metastatic disease. She has received temsirolimus and sunitinib with disease progression following brief periods of stability with both agents. Which of the following statements best reflects available treatment options for this patient?
A.Foretinib and other antagonists of the MET pathway are associated with improved survival in this setting.
B.Treatment with axitinib should be considered as this agent has been shown to improve progression-free survival compared to sorafenib.
C.Everolimus has been shown to improve progression-free survival compared to sorafenib in patients with papillary RCC who have failed prior therapy with sunitinib.
D.There are no standard options of proven clinical benefit; appropriate clinical trials could be considered.
5.A 31-year-old man presents with a 4-week history of malaise and two to three episodes of gross hematuria. He has also noticed an unintentional weight loss of approximately 10 lb over the past 2 to 3 months. His family history is remarkable for kidney cancer in his mother. His physical examination is remarkable for several papular skin lesions, which are very sensitive to touch, temperature changes. A CT of the chest, abdomen, and pelvis reveals a 10 cm left renal mass but is otherwise normal. He is offered a radical left nephrectomy by his urologist, but asks if the procedure can be deferred for at least 4 weeks as he is the sole caregiver for his 24-year-old sister who is undergoing a hysterectomy next week for removal of several large uterine fibroids. Which of the following statements about this patient’s condition is most accurate?
A.This presentation is most consistent with hereditary papillary renal cell carcinoma and genetic evaluation will reveal a germ-line mutation in MET.
B.Imaging of the CNS in this patient and his sister will likely demonstrate the presence of hemangioblastomas.
C.He should undergo genetic counseling and should be evaluated for germ-line alterations in the fumarate hydratase gene.
D.His renal tumor will demonstrate loss of heterozygosity affecting the VHL gene.
Suggested Readings
1.Atkins MB, Dutcher J, Weiss G, et al. Kidney cancer. The Cytokine Working Group experience (1986–2001): part I. IL-2-based clinical trials. Med Oncol. 2001;18(3):197-207.
2.Childs R, Chernoff A, Contentin N, et al. Regression of metastatic renal-cell carcinoma after nonmyeloablative allogeneic peripheral-blood stem-cell transplantation. N Engl J Med. 2000;343(11):750-758.
3.Dutcher J, Atkins MB, Margolin K, et al. Kidney cancer: the Cytokine Working Group experience (1986–2001): part II. Management of IL-2 toxicity and studies with other cytokines. Med Oncol. 2001;18(3):209-219.
4.Escudier B, Eisen T, Stadler WM, et al. Sorafenib in advanced clear-cell renal-cell carcinoma. N Engl J Med. 2007;356(2):125-134.
5.Escudier B, Pluzanska A, Koralewski P, et al. Bevacizumab plus interferon alfa-2a for treatment of metastatic renal cell carcinoma: a randomised, double-blind phase III trial. Lancet. 2007;370(9605):2103-2111.
6.Flanigan RC, Salmon SE, Blumenstein BA, et al. Nephrectomy followed by interferon alfa-2b compared with interferon alfa-2b alone for metastatic renal-cell cancer. N Engl J Med. 2001;345(23):1655-1659.
7.Fyfe GA, Fisher RI, Rosenberg SA, Sznol M, Parkinson DR, Louie AC. Long-term response data for 255 patients with metastatic renal cell carcinoma treated with high-dose recombinant interleukin-2 therapy. J Clin Oncol. 1996;14(8): 2410-2411.
8.Grubb RL III, Franks ME, Toro J, et al. Hereditary leiomyomatosis and renal cell cancer: a syndrome associated with an aggressive form of inherited renal cancer. J Urol. 2007;177(6):2074-2079.
9.Hudes G, Carducci M, Tomczak P, et al. Temsirolimus, interferon alfa, or both for advanced renal-cell carcinoma. N Engl J Med. 2007;356(22):2271-2281.
10.Javidan J, Stricker HJ, Tamboli P, et al. Prognostic significance of the 1997 TNM classification of renal cell carcinoma. J Urol. 1999;162(4):1277-1281.
11.Latif F, Tory K, Gnarra J, et al. Identification of the von Hippel-Lindau disease tumor suppressor gene. Science. 1993;260(5112):1317-1320.
12.Linehan WM, Pinto PA, Srinivasan R, et al. Identification of the genes for kidney cancer: opportunity for disease-specific targeted therapeutics. Clin Cancer Res. 2007;13(2 Pt 2):671s-679s.
13.Linehan WM, Ricketts CJ. The metabolic basis of kidney cancer. Semin Cancer Biol. 2013;23(1):46-55.
14.Motzer RJ, Escudier B, Oudard S, et al. Efficacy of everolimus in advanced renal cell carcinoma: a double-blind, randomised, placebo-controlled phase III trial. Lancet. 2008;372(9637):449-456.
15.Motzer RJ, Hutson TE, Tomczak P, et al. Sunitinib versus interferon alfa in metastatic renal-cell carcinoma. N Engl J Med. 2007;356(2):115-124.
16.Motzer RJ, Mazumdar M, Bacik J, Berg W, Amsterdam A, Ferrara J. Survival and prognostic stratification of 670 patients with advanced renal cell carcinoma. J Clin Oncol. 1999;17(8):2530-2540.
17.Negrier S, Escudier B, Lasset C, et al. Recombinant human interleukin-2, recombinant human interferon alfa-2a, or both in metastatic renal-cell carcinoma. Groupe Francais d’Immunotherapie. N Engl J Med. 1998;338(18):1272-1278.
18.Ricketts CJ, Shuch B, Vocke CD, et al. Succinate dehydrogenase kidney cancer: an aggressive example of the Warburg effect in cancer. J Urol. 2012;188(6):2063-2071. doi:10.1016/j.juro.2012.08.030 [Epub October 18, 2012].
19.Rini BL, Escudier B, Tomczak P, et al. Comparative effectiveness of axitinib versus sorafenib in advanced renal cell carcinoma (AXIS): a randomized phase 3 trial. Lancet. 2011;378:1931-1939.
20.Rini BL, Halabi S, Rosenberg JE, et al. Phase III trial of bevacizumab plus interferon alfa versus interferon alpha monotherapy in patients with metastatic renal cell carcinoma: final results of CALGB 90206. J Clin Oncol. 2010;28: 2137-2143.
21.Schmidt LS, Nickerson ML, Angeloni D, et al. Early onset hereditary papillary renal carcinoma: germline missense mutations in the tyrosine kinase domain of the met proto-oncogene. J Urol. 2004;172(4 Pt 1):1256-1261.
22.Schmidt LS, Nickerson ML, Warren MB, et al. Germline BHD-mutation spectrum and phenotype analysis of a large cohort of families with Birt-Hogg-Dube syndrome. Am J Hum Genet. 2005;76(6):1023-1033.
23.Sternberg CN, Davis IS, Mardiak J, et al. Pazopanib in locally advanced or metastatic renal cell carcinoma: results of a randomized phase III trial. J Clin Oncol. 2010;28:1061-1068.
24.Yang JC, Haworth L, Sherry RM, et al. A randomized trial of bevacizumab, an anti-vascular endothelial growth factor antibody, for metastatic renal cancer. N Engl J Med. 2003;349(5):427-434.
25.Yang JC, Sherry RM, Steinberg SM, et al. Randomized study of high-dose and low-dose interleukin-2 in patients with metastatic renal cancer. J Clin Oncol. 2003;21(16):3127-3132.