Sibylle Loibl1 and Bianca Lederer1
(1)
German Breast Group, c/o GBG-Forschungs GmbH, Martin-Behaim-Str. 12, Neu-Isenburg, 63263, Germany
Sibylle Loibl
Email: Sibylle.Loibl@gbg.de
Keywords
Breast cancerPregnancyYoung womenChemotherapy
Introduction
Breast cancer during pregnancy (BCP) accounts for approximately 2 % of primary breast cancer patients. The term pregnancy-associated breast cancer (PABC) refers to all cancers diagnosed during pregnancy and up to 1 year after delivery. This article will focus on breast cancer diagnosed and treated during pregnancy because treatment might need adaptation. An increase in breast cancer incidence and a rise in maternal age in the past decades have led to more cases of breast cancer diagnosed during pregnancy. This upward trend in the occurrence of BCP in recent years has prompted an increased awareness for management strategies of this rare and delicate disease. While BCP has shown to generally present in more advanced stages compared to breast cancer in non-pregnant women, all available evidence suggests that it has a similar prognosis provided that standard treatment is administered. In 2006, the first recommendations for diagnosis and treatment of BCP were published [1] with the consensus that treatment during pregnancy should adhere as closely as possible to the general recommendations for young non-pregnant women. With recent advances in breast cancer therapy, including the use of carboplatin, dose-dense chemotherapy, trastuzumab, neoadjuvant therapy and sentinel lymph node biopsy as sole treatment, options for breast cancer patients have increased. Therefore, a panel of experts has recently reviewed latest treatment strategies and adaptations for BCP and published a consensus paper on BCP management recommendations [2]. In general, all management strategies need to weigh maternal treatment efficacy against foetal safety. The main aspects will be highlighted in this chapter.
Management Strategies in Breast Cancer During Pregnancy
Diagnosis: Imaging - Pathology - BRCA Testing
Routine examination of the breast is not part of the general examinations for pregnant women in contrast to the Pap smear for early detection of cervical cancer. There are no typical signs or symptoms for BCP, and tumours will not be screen detected, because general screening is not indicated in this age group. Signs and symptoms should not be neglected, and a lump should be biopsied for histology if it is not disappearing within 4 weeks to avoid unnecessary delays in the right diagnosis and treatment, even if 80 % of breast biopsies performed in pregnant women will prove to be benign. A 1-month delay in diagnosis can increase the risk of nodal involvement by 0.9–1.8 %. Core biopsy is the standard procedure to obtain tissue, whereas fine needle biopsy or aspiration cytology is not recommended. For any woman with a confirmed or particularly suspicious malignant lesion in the breast, bilateral mammography is recommended. However, especially during pregnancy, many patients and physicians are concerned about radiation safety. A prospective study on exposure during a standard bilateral mammogram, however, has shown that radiation doses to all organs other than the breast are extremely low (less than 3 mGy) and that the dose to the uterus or foetus, especially in the early phase of pregnancy, is minimal [3]. Although many factors, like gestational age, anatomic site, modality and technique, can influence maternal and foetal radiation exposure and dose, breast ultrasound and mammography can generally be considered safe during pregnancy. In general, imaging and staging procedures should only be conducted in advanced stages, i.e. where they might alter the treatment, and, like in non-pregnant breast cancer patients, unnecessary or less accurate procedures should be avoided.
The diagnosis of BCP based on histopathologic evaluation of core biopsies from suspicious lesions is considered the gold standard. In this context, it is crucial that the pathologist is informed about the pregnancy of the patient when examining the tumour. Generally, the histological features of tumours from BCP patients are not different from those in young non-pregnant women with breast cancer. The vast majority are ductal invasive, mainly hormone-receptor negative and undifferentiated [4]. Moreover, tumour mutations do not differ between pregnant and non-pregnant young women. Small series on biological features at the genomic level showed significant differences in gene expression. In particular, tumours diagnosed during pregnancy showed higher expression of the IGF1 (insulin-like growth factor 1) gene and an activation of the GPCR (G protein-coupled receptor) and the serotonin receptor pathway. Moreover during pregnancy, the PD1 (programmed cell death 1)-PDL-1 (programmed cell death ligand 1) interaction is activated to allow foetomaternal tolerance. A high expression of PD1 during pregnancy is an appealing topic due to the recent efficacy data on anti-PD1 and anti-PDL-1 inhibitors [5]. However, no definite conclusions for general practice can be drawn from these analyses. The selection of appropriate control cohorts, matched by treatment and/or histology, as well as by age, will be required in future research in this challenging field.
At diagnosis, assessment of the family history of the patient is important, and genetic counselling should be provided according to national guidelines. Based on personal and family history, germline BRCAtesting can be offered to patients who are likely to have an inherited mutation. The results of the germline BRCA determination will be of treatment relevance. The majority of patients with BCP have triple-negative breast cancer (TNBC), and it has been shown that in young TNBC patients under the age of 40, the probability of detecting a germline BRCA mutation is around 40 % [6].
Local Treatment: Surgery - SLNB - Radiotherapy
In terms of surgery, patients with BCP should be treated with the same approach as non-pregnant patients. Mastectomy used to be the standard treatment for patients developing BCP. However, nowadays, the pregnancy alone is no indication for mastectomy because of the resulting delay of the radiotherapy. Breast conservation has been shown to be an option, and, especially when performed in the second or third trimester, it is possible to delay subsequent radiotherapy until after delivery. In this case, chemotherapy would generally be given during pregnancy followed by radiotherapy after delivery. For patients requiring or opting for a mastectomy, immediate breast reconstruction is an essential component in the patient management and particularly important for patients diagnosed at a young age. Tissue expander insertion has been discussed for women diagnosed with BCP as it ensures a short operation time and does not seem to be associated with considerable morbidity to the patient or the foetus [7]. However, it needs to be well considered if this is the ideal time point to start breast reconstruction.
According to ASCO guidelines 2014, patients with BCP should not undergo sentinel lymph node biopsy (SLNB) based on cohort studies and/or informal consensus. However, it has been shown that this procedure can be safely performed during pregnancy [8]. Radioactivity doses injected loco-regionally during SLNB are relatively low and show a rapid clearance as well as substantial and stable uptake at the injection site, which is shortly thereafter removed by surgery. In optimised protocols for radiopharmaceuticals and the amounts of activity typically used for SLNB, the doses absorbed by the foetus are mostly below 20 μGy for 10–20 MBq (about 1 μGy/MBq) [9]. Also, for the BCP patient herself SLNB appears to be safe having shown a low recurrence rate [10]. Therefore, pregnant breast cancer patients should be offered SLNB rather than axillary clearance whenever it is indicated according to general practice in non-pregnant patients. In terms of recommended protocols, an advised option to minimise radiation exposure is to inject colloid in the morning (1-day protocol). Using blue dye as a sole procedure is not recommended outside pregnancy. Due to a low (1 %) but potentially harmful underlying risk of an anaphylactic maternal reaction, this method should not be used in BCP, although a small series of 25 women receiving SLNB during pregnancy did not show any SLNB-associated complications for the seven women who received blue dye for mapping [11].
Radiation therapy (RT) during pregnancy is not generally recommended since the available information on long-term consequences of in utero exposure is limited. Generally, it is therefore recommended to delay RT until after delivery whenever possible.
Gestational age plays an important role in the consequences that RT may have during pregnancy. The radiation dose received by the foetus is dependent on the distance between the RT field and the position of the foetus, which is dependent on gestational age but also on the amount of leakage of irradiation outside the radiation field. The use of effective shielding can reduce the dose to the foetus by up to 75 %. RT administered during BCP with low foetal doses has been reported for several cases to result in the delivery of healthy babies [12]. Therefore, in case it is absolutely indicated, RT can be considered in the first or early second trimester, if the risk of postponing or omitting RT for the mother might outweigh the harm to the foetus.
Systemic Therapy: Chemotherapy - Endocrine Therapy - Targeted Therapy - Special Considerations
Systemic treatments such as chemotherapy, hormonal therapy, and targeted therapies are key in modern breast cancer treatment regimen, but due to their harmful side effects, administration in pregnancy needs to be considered carefully.
Chemotherapy is contraindicated during the first trimester of pregnancy due to a higher risk of inducing foetal malformations as well as abortions. A recent report shows that the prevalence of malformations following chemotherapy in the first trimester is 14 %, but decreases to 3 % if given later in pregnancy [13], which is comparable to rates reported for the general population in the USA (3 %) and data from a German registry (6.7 %). Completely postponing chemotherapy treatment in pregnant patients until after delivery might seem to be an option. However, in non-pregnant young women, postponing of chemotherapy has been shown to be associated with an increased risk of relapse. Therefore, it is recommended to treat women with BCP during the second and third trimester following guidelines for non-pregnant young patients as closely as possible [1]. Other anticancer agents used as systemic treatment, such as trastuzumab, tamoxifen and endocrine agents, should in general be avoided during pregnancy, given their potential foetal toxicity [1]. The standard adjuvant or neoadjuvant combination of anthracyclines, cyclophosphamide and taxanes for non-pregnant patients is also recommended for the treatment of BCP after the first trimester [14]. Epirubicin/cyclophosphamide (EC) followed by weekly paclitaxel (EC-Pw) or the reverse sequence, starting with a taxane, can be used during pregnancy. Regimen that are not standard or no longer indicated for breast cancer therapy in non-pregnant women, such as anthracycline- or taxane-free regimen or 5-fluorouracil, should be avoided in pregnant as well as in non-pregnant patients. Platinum derivatives may have a role in the treatment of triple-negative tumours of breast cancer patients. The addition of carboplatin in neoadjuvant trials has led to significantly higher pathological complete response rates in two phase II prospectively randomised trials, but data are still immature for survival analyses [15, 16]. Carboplatin can thus also be considered during the second and third trimesters of pregnancy. Whether or not carboplatin is more effective than cisplatinum remains unclear; however, it may have less overall toxicity and seems therefore the preferred platinum agent during pregnancy.
Dose-dense (same dose administered over a shorter interval) or intensified dose-dense (IDD, higher dose over a shorter interval) chemotherapy regimen have been shown to lead to better survival compared with conventionally dosed chemotherapy regimen, especially in high-risk patients [17]. While dose-dense chemotherapy (e.g. EC every 2 instead of every 3 weeks) seems to be an acceptable option during pregnancy, there are no systematic studies and only a small number of reports on IDD chemotherapy [18]. Due to a high rate of grade 2–4 anaemia (59 %), with a need for transfusion in 20 % of patients, and a high risk of febrile neutropenia (7 % despite primary G-CSF prophylaxis) [17], the administration of IDD chemotherapy in BCP patients demands for a very strict risk/benefit analysis. Therefore, to date, IDD cannot generally be recommended in BCP.
In addition to the choice of therapy for BCP patients, there are some special considerations based on the physiological aspects during pregnancy. The variations in drug pharmacokinetics during pregnancy raise important concerns regarding optimal drug dosing in pregnant patients. Thus, physiologic alterations associated with pregnancy may result in lower maximal concentrations of chemotherapy and a lower area under the concentration-time curve. Since most anticancer agents are empirically prescribed according to body surface area (BSA), a large inter-patient variability for dosage exists, even outside the pregnancy setting. Another aspect of changed pharmacokinetics in pregnancy is the increased activity of major enzymes involved in the metabolism of taxanes and anthracyclines (including cytochrome p450 isoforms such as CYP3A4 or CYP2C8) during the late trimesters of pregnancy, which may result in decreased drug exposure. Additionally, albumin concentrations vary significantly during pregnancy, and, since taxanes are highly protein bound, this may lead to significant changes in taxane pharmacokinetics [19].
A comparison of pregnant versus non-pregnant patients in terms of pharmacokinetics of anthracyclines and taxanes showed that taxane serum levels significantly decreased during pregnancy, especially for paclitaxel, whereas the exposure to anthracyclines was not significantly modified by pregnancy in a very limited number of investigated patients [19]. Whether or not doses should be increased in pregnancy remains uncertain, given that such increases could result in severe toxicities, with potentially serious consequences for both mother and child. In overweight women, who also have altered pharmacokinetics, the dose is not increased. Furthermore, it was shown that the chemotherapy is as active in pregnant as in non-pregnant women [20]. Women who were treated with neoadjuvant chemotherapy for breast cancer during pregnancy achieved the same pathological complete response rate as non-pregnant women treated with neoadjuvant chemotherapy and women with BCP receiving neoadjuvant therapy after delivery. Thus, dosing based on BSA, using the current patient weight (prior to every course), should be the standard, as well as using the same dose for pregnant as non-pregnant women.
While maternal drug exposure is necessary to achieve optimal treatment efficacy, the transplacental transfer of active agents is a concern in terms of foetal safety. In addition to its functions in protecting the foetus and preparing the women for pregnancy and lactation, the placenta is also the central organ for foetal-maternal exchange. Transplacental transfer of drugs can be studied using the perfused human ex vivo placenta; however, toxic effects of cancer therapy on the human placenta are poorly understood. Data are limited, mainly because most animal models are devoid of central common features with the human placenta and even closely related species such as rhesus monkeys show diverging invasion patterns.
A preclinical study on transplacental transfer rates indicates similar and reassuring data on different anthracyclines and taxanes, although with marked inter-patient variability, particularly with docetaxel [21]. Consequently, paclitaxel might be preferred to docetaxel in the pregnancy setting regarding foetal safety. Carboplatin was also analysed and demonstrated significant transplacental transfer but long-term data remain limited [13].
Giving chemotherapy during pregnancy has been associated with a significantly higher incidence of small-for-gestational-age babies [4]. A potentially toxic influence on placental development leading to placental malfunction, e.g. via incomplete trophoblast invasion into the uterus, has been suggested, which could result in a decreased supply of nutrients for the foetus [4]. The start of chemotherapy is often considered after week 10 of gestation, since organogenesis is completed around that time. However, trophoblast invasion of the placenta is not completed until around week 20. Therefore, even starting chemotherapy at week 14 might interfere with late stages of placental development (Fig. 10.1), which is highly hypothetical and remains to be proven. Chemotherapy should not be started before the end of the first trimester and should be stopped around the 36th week of gestation to allow for a 2-week chemo-free interval prior to delivery and to allow for a term delivery (>37 weeks). Children exposed to chemotherapy in utero seem to have no adverse outcome when compared to age-matched children, on the short and the long run. However, close monitoring of the pregnancy, as well as an exact determination of the gestational age prior to start of the therapy, is indicated.
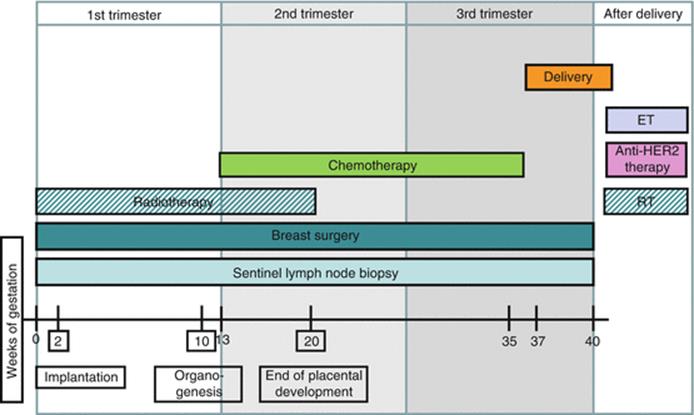
Fig. 10.1
Therapeutic options during pregnancy. Crucial phases: implantation (0–2w), organogenesis (2–10w) and foetal phase (>10w). Starting chemotherapy from week 13–14 instead of week 10 allows a ‘safety period’. If radiotherapy is indicated and decided (against preferred option) to not postpone until after delivery, it can be applied during the first and until early second trimester. Endocrine therapy and anti-HER2 treatment should be delayed until after delivery. RT radiotherapy, ET endocrine therapy
Anti-HER2 Treatment
Treatment with targeted agents such as trastuzumab has become an essential part of primary treatment in breast cancer patients with HER2+ tumours. An early introduction of trastuzumab and combined rather than sequential administration of cytotoxic agents has been associated with a better survival in non-pregnant patients. During pregnancy, it is generally recommended to avoid trastuzumab, since several case reports have demonstrated development of oligo-anhydramnios and reported foetal deaths [22]. However, inadvertent foetal exposure of 1–2 cycles of trastuzumab is no reason for termination of pregnancy [23]. In special high-risk situations, trastuzumab during pregnancy might even be considered, though not without carefully weighing foetal and maternal risks and benefits during an informed decision-making process. The double HER2 blockade with pertuzumab in addition to trastuzumab and chemotherapy has shown to increase pathological complete response rate in patients with HER2+ breast cancer, but data on the use of pertuzumab during pregnancy are currently lacking. Therefore, although the dual blockade with trastuzumab and pertuzumab is recommended as neoadjuvant treatment in breast cancer, this does not seem to be an option in women with BCP.
Obstetrical/Perinatal Care
A further major concern in prenatal care of pregnant women treated with cancer therapy is preterm delivery and subsequent development of the children.
Antenatal chemotherapy exposure has been associated with an increased risk of preterm rupture of membranes (3 % vs. 0 %) and preterm labour (6 % vs. 2 %) [4]. Recent studies have reported a mean gestational age at delivery of 36–37 weeks, indicating that a significant proportion of patients deliver (iatrogenically) preterm. A long-term study of children exposed to chemotherapy in utero showed no impairment of cognitive, cardiac or general development of the children [24]. However, prematurity was correlated with worse cognitive outcome, independent of cancer treatment. Therefore, prematurity should be avoided whenever possible, and treatment during pregnancy may help to achieve a full-term pregnancy. The complex medical situation of BCP renders a primordial multidisciplinary discussion. A close collaboration of the obstetrician and perinatologist and frequent (at least once in 3 weeks) in addition to standard prenatal care are warranted. Moreover, a 2–3-week interval between last chemotherapy and delivery is recommended in order to allow the bone marrow to recover and prevent haematologic toxicity to the mother and child.
Conclusion
The management strategies in breast cancer during pregnancy should follow the general guidelines for young non-pregnant patients as closely as possible. Breast cancer during pregnancy is not per se a reason for abortion, but it is important to discuss and treat this complex medical situation within a multidisciplinary team. A careful risk/benefit analysis is crucial to avoid both over- and undertreatment in order to minimise foetal toxicity and not compromise on maternal treatment efficacy. Delivery should be as close as possible to term to reduce the risk for developmental shortfalls.
To improve treatment strategies for breast cancer during pregnancy, large prospective cohort studies are needed. Long-standing international collaborations have already provided the basis of current knowledge on management strategies of BCP and will continue to do so. Patients can be registered online through the German Breast Group (www.gbg.de).
References
1.
Loibl S, von Minckwitz G, Gwyn K, et al. Breast carcinoma during pregnancy. International recommendations from an expert meeting. Cancer. 2006;106(2):237–46.CrossRefPubMed
2.
Loibl S, Schmidt A, Gentilini O, Kaufman B, Kuhl C, Denkert C, von Minckwitz G, Parokonnaya A, Stensheim H, Thomssen C, van Calsteren K, Poortmans P, Berveiller P, Markert UR, Amant F. Breast Cancer Diagnosed During Pregnancy: Adapting Recent Advances in Breast Cancer Care for Pregnant Patients. JAMA Oncol. 2015;1(8):1145–53.
3.
Sechopoulos I, Suryanarayanan S, Vedantham S, D’Orsi CJ, Karellas A. Radiation dose to organs and tissues from mammography: Monte Carlo and phantom study. Radiology. 2008;246(2):434–43.CrossRefPubMedPubMedCentral
4.
Loibl S, Han SN, von Minckwitz G, et al. Treatment of breast cancer during pregnancy: an observational study. Lancet Oncol. 2012;13(9):887–96.CrossRefPubMed
5.
Azim Jr HA, Brohée S, Peccatori FA, et al. Biology of breast cancer during pregnancy using genomic profiling. Endocr Relat Cancer. 2014;21(4):545–54.CrossRefPubMed
6.
Couch FJ, Hart SN, Sharma P, et al. Inherited mutations in 17 breast cancer susceptibility genes among a large triple-negative breast cancer cohort unselected for family history of breast cancer. J Clin Oncol. 2015;33(4):304–11.CrossRefPubMedPubMedCentral
7.
Lohsiriwat V, Peccatori FA, Martella S, et al. Immediate breast reconstruction with expander in pregnant breast cancer patients. Breast. 2013;22(5):657–60.CrossRefPubMed
8.
Gentilini O, Cremonesi M, Toesca A, et al. Sentinel lymph node biopsy in pregnant patients with breast cancer. Eur J Nucl Med Mol Imaging. 2010;37(1):78–83.CrossRefPubMed
9.
Pandit-Taskar N, Dauer LT, Montgomery L, St Germain J, Zanzonico PB, Divgi CR. Organ and fetal absorbed dose estimates from 99mTc-sulfur colloid lymphoscintigraphy and sentinel node localization in breast cancer patients. J Nucl Med. 2006;47(7):1202–8.PubMed
10.
Han SN, Amant F, Sangalli C, et al. Sentinel lymph node biopsy for breast cancer treatment during pregnancy – on behalf of the International Network of Cancer. Infertility and pregnancy (INCIP) and the German Breast Group (GBG). Ann Oncol. 2014;25(Suppl 4):266PD.
11.
Gropper AB, Calvillo KZ, Dominici L, et al. Sentinel lymph node biopsy in pregnant women with breast cancer. Ann Surg Oncol. 2014;21(8):2506–11.CrossRefPubMed
12.
van der Giessen PH. Measurement of the peripheral dose for the tangential breast treatment technique with Co-60 gamma radiation and high energy X-rays. Radiother Oncol. 1997;42(3):257–64.CrossRefPubMed
13.
National Toxicology Program. NTP monograph: developmental effects and pregnancy outcomes associated with cancer chemotherapy use during pregnancy. NTP Monogr. 2013;(2):i–214. http://www.ncbi.nlm.nih.gov/pubmed/24736875
14.
Cardonick E, Bhat A, Gilmandyar D, Somer R. Maternal and fetal outcomes of taxane chemotherapy in breast and ovarian cancer during pregnancy: case series and review of the literature. Ann Oncol. 2012;23(12):3016–23.CrossRefPubMed
15.
von Minckwitz G, Schneeweiss A, Loibl S, et al. Neoadjuvant carboplatin in patients with triple-negative and HER2-positive early breast cancer (GeparSixto; GBG 66): a randomised phase 2 trial. Lancet Oncol. 2014;15(7):747–56.CrossRef
16.
Sikov WM, Berry DA, Perou CM, et al. Impact of the addition of carboplatin and/or bevacizumab to neoadjuvant once-per-week paclitaxel followed by dose-dense doxorubicin and cyclophosphamide on pathologic complete response rates in stage II to III triple-negative breast cancer: CALGB 40603 (Alliance). J Clin Oncol. 2015;33(1):13–21.CrossRefPubMedPubMedCentral
17.
Moebus V, Jackisch C, Lueck HJ, et al. Intense dose-dense sequential chemotherapy with epirubicin, paclitaxel, and cyclophosphamide compared with conventionally scheduled chemotherapy in high-risk primary breast cancer: mature results of an AGO phase III study. J Clin Oncol. 2010;28(17):2874–80.CrossRefPubMed
18.
Cardonick E, Gilmandyar D, Somer RA. Maternal and neonatal outcomes of dose-dense chemotherapy for breast cancer in pregnancy. Obstet Gynecol. 2012;120(6):1267–72.PubMed
19.
van Hasselt JG, van Calsteren K, Heyns L, et al. Optimizing anticancer drug treatment in pregnant cancer patients: pharmacokinetic analysis of gestation-induced changes for doxorubicin, epirubicin, docetaxel and paclitaxel. Ann Oncol. 2014;25(10):2059–65.CrossRefPubMed
20.
Loibl S, Han S, Mayer K, et al. Neoadjuvant chemotherapy for patients with breast cancer during pregnancy (BCP). J Clin Oncol. 2014;32:5s(Suppl; abstr 1071).
21.
Amant F, Loibl S, Neven P, Van Calsteren K. Breast cancer in pregnancy. Lancet. 2012;379(9815):570–9.CrossRefPubMed
22.
Zagouri F, Sergentanis TN, Chrysikos D, Papadimitriou CA, Dimopoulos MA, Bartsch R. Trastuzumab administration during pregnancy: a systematic review and meta-analysis. Breast Cancer Res Treat. 2013;137(2):349–57.CrossRefPubMed
23.
Zagouri F, Psaltopoulou T, Dimitrakakis C, Bartsch R, Dimopoulos MA. Challenges in managing breast cancer during pregnancy. J Thorac Dis. 2013;5 Suppl 1:S62–7.PubMedPubMedCentral
24.
Amant F, Vandenbroucke T, Verheecke M, Fumagalli M, Halaska MJ, Boere I, Han S, Gziri MM, Peccatori F, Rob L, Lok C, Witteveen P, Voigt JU, Naulaers G, Vallaeys L, Van den Heuvel F, Lagae L, Mertens L, Claes L, Van Calsteren K; International Network on Cancer, Infertility, and Pregnancy (INCIP). Pediatric Outcome after Maternal Cancer Diagnosed during Pregnancy. N Engl J Med. 2015;373(19):1824–34.