Dag Orstavik
1.1 Introduction
Endodontology includes pulp and periapical biology and pathology. As a clinical discipline, however, endodontics mainly deals with treatment of the root canal and the placement of a root filling, or treatment by surgical endodontics. The technical procedures associated with treatment focus on the particular problems of asepsis and disinfection of the pulp canal system. Treatment measures to preserve pulp vitality are a shared responsibility with conservative dentistry, and include specific techniques in dental traumatology. Recent research has shown the importance of asepsis and disinfection procedures also for treatment of pulps exposed by caries or trauma, extending classical endodontic treatment principles to the management of deep caries (see Chapter 9).
For vital teeth requiring partial or full pulp removal, the initial diagnoses and the difficulties associated with treatment may be related to the state of the pulp, but the purpose of treatment is no longer the preservation of the pulp but the prevention and/or elimination of infection in the root canal system. The ultimate biological aim of this treatment is to prevent apical periodontitis. For teeth with infected/necrotic pulpal with an established apical disease process, the biological aim is to cure apical periodontitis. Of the endodontic diseases, apical periodontitis is therefore prominent as it is a primary indication for root canal treatment and because it is by far the most common sequel when treatment is inadequate or fails (Figure 1.1). Even the measures taken to preserve pulp vitality may be viewed as ultimately preventing root canal infection and the development of apical periodontitis.
The importance of microbes in the initiation, development and persistence of apical periodontitis has been thoroughly documented (see Chapter 4). The emphasis in this book is on the infectious etiology of apical periodontitis and on the aseptic and antiseptic principles applied during treatment. Furthermore, new research findings have impact on aspects of diagnosis, treatment, prognosis and evaluation of outcome in endodontics. It is therefore important to use the acquired knowledge to build treatment principles logically, and to show how all these fundamental aspects can be applied in clinical practice.
1.2 Terminology
Both pulp and pulp-periodontal diseases have been subject to many classification systems with variable terminology. Periodontitis caused by infection of the pulp canal system has been termed apical periodontitis, apical granuloma/cyst, periapical osteitis and periradicular periodontitis, among other terms. Sub-classifications have been acute/ chronic, exacerbating/Phoenix abscess and symptomatic/asymptomatic, among others [18]. The two most accepted classification schemes are presented in Table 1.1. These are quite similar, but symptomatic teeth according to the AAE classification may include more cases than teeth with acute apical periodontitis according to the ICD. The latter term is for cases presenting with subjective needs for immediate treatment, while symptomatic teeth may include teeth that only slightly affect the patients and that are diagnosed by chairside testing (see Chapter 7). The term “chronic” is useful for prognostication and follow-up studies: symptomatic or not, it implies the presence of a radiolucent lesion, which is a major predictor for treatment success [24]. The term “symptomatic” confirms that there are objective signs verifying the diagnosis.
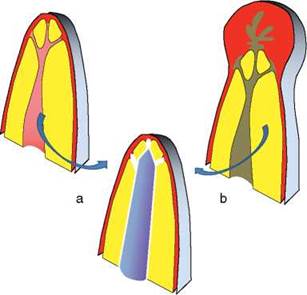
Figure 1.1 Pulp extirpation (a) prevents and root canal disinfection (b) cures apical periodontitis. Both need a root filling of the entire pulpal space.
Table 1.1 Classification of apical periodontitis [18].
|
AAE |
ICD-10 |
|
Symptomatic apical periodontitis SAP1 |
K04.4 Acute apical periodontitis of pulpal origin2 |
|
Asymptomatic apical periodontitis AAP |
K04.5 Chronic apical periodontitis |
|
Chronic apical abscess |
K04.6 Periapical abscess with sinus3 |
|
Acute apical abscess |
K04.7 Periapical abscess without sinus |
|
Condensing osteitis4 Radicular cyst |
K04.8 Radicular cyst |
1 presents with a broad range of symptoms
2 presents with strong pain
3 further subdivided in relation to sinus tract location on surfaces
4 may be seen as a variant of AAP or Chronic apical periodontitis
Apical periodontitis includes dental abscess, granuloma and radicular cyst as manifestations of the same basic disease. The balance between the virulence and extent of infection on the one hand and the body's response on the other, determines whether the condition is symptomatic/acute versus asymptomatic/ chronic. The historical emphasis on the differential diagnosis of a cyst versus a granuloma has been abandoned. This is due to the fact that radiographs, even from CBCT, are not very sensitive in discriminating between cysts and granulomas [6]; they share the same etiology and basic disease processes (Chapters 3 and 4); and their treatment and prognosis are also similar (Chapters 11 and 12). However, so-called true cysts separated from the root canal infection that initiated them may show impaired healing [27] and require surgical removal, but there are no means for diagnosing such cases without scrupulous histological investigation of surgical biopsies [32].
Terminology should not be considered a straitjacket for authors or clinicians. Therefore, variants of the terms and references to other diagnostic schemes, in this book and other texts, are inevitable, and can even be desirable. However, given that insurance companies and other third parties require codes or terms for reimbursements, and legal issues dictate clear basic diagnoses as basis for treatment, selection and proper usage of a recognized classification scheme is mandatory.
1.3 Pulp Infection and Periapical Inflammation
The oral cavity is an extension of the skin/ mucosal barrier to the external environment. In the digestive tract, it may be viewed as the first battleground for the body's efforts to maintain homeostasis and keep infection away from the vulnerable interior parts of the body. Infection occurs when pathogenic or opportunistic microorganisms infiltrate or penetrate the body surface. In the oral/dental sphere, the body surface is either the mucosa or the enamel/dentine coverage of underlying soft tissue. Endodontic treatment aims to re-establish the muco-cutaneo-odontobarrier with a complete seal from the coronal to the apical end of the treated root, whereas voids or leaks in the restoration may present an opportunity for bacteria to establish themselves close and eventually ingress into the body's interior. The emphasis on coronal as much as apical leakage of bacteria and bacterial products reflects this line of reasoning.
The evolution of permanent teeth in a dentition with multiple functions is integral to the evolution of animals [40], not least primates and man. However, the structure of these teeth is such that if fracture occurs, microorganisms may enter the body and establish a foothold in the exposed dentinal and pulpal tissues. Unless protective mechanisms were developed, such infections would be life-threatening and presented a strong survival disadvantage in the young [40]. Employing and modifying general mechanisms of inflammation, apical periodontitis evolved to combat and contain the infections in the compromised dental pulp spreading through its ramifications and the tubules of dentin (Figure 1.2). While defining the disease, apical periodontitis works therefore to our advantage; it is the underlying infection that is the cause for concern.
The protection by tissue responses comes at a cost, however. Clinical symptoms that accompany the inflammation may be distressing to the patient, and the granuloma or cyst are not always effective in containing the invading microbes. The pain sometimes following the inflammation of the pulp and periapical tissues can be excruciating and is a testimony to the potential danger of the infection. This pain is also the starting point for human attempts to combat dental disease. Thus, acute pulpal and periapical inflammation were the among the first targets of the dental profession.
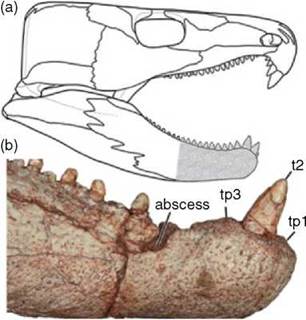
Figure 1.2 Evidence of dental and mandibular pathology in Labidosaurus hamatus, a basal reptile from the Lower Permian of Oklahoma. (a) Skull reconstruction in right lateral view. (b) right hemi-mandible in lateral view. Reproduced with permission from [40].
Teeth, cheek cells, tongue crypts, tonsillar irregularities, gingival sulci and other anatomical structures are safe havens for microbial populations of the mouth. From these areas, microbes of varying virulence may emigrate and cause infections such as tonsillitis, gingivitis, pericoronitis, marginal periodontitis, dental caries, pulpitis and apical periodontitis. Whereas physiological and mechanical clean- sing activities tend to reduce the level of microorganisms in the mouth, environmental factors sometimes favor infection rather than its prevention. Current research on oral microbial communities emphasizes the concept of biofilm formation and development, with particular physiological, genetic and pathogenic properties of the organisms expressed as consequences of the conditions within the biofilm (see Chapter 4).
Caries has been the dominant dental infection for decades, and infection and inflammation of the pulp and periapical tissues are often an extension of the dental caries process. Researchers studied the occurrence and epidemiology of apical periodontitis as part of caries investigations. The infectious nature and the possibilities for spreading and complications of apical periodontitis should form the basis for independent surveys of public health consequences of endodontic diseases.
1.4 Biological and Clinical Significance of Apical Periodontitis
1.4.1 Apical Periodontitis as an Infection
Root canal infections and apical periodontitis is common today and is a frequent finding in skulls from archaeological investigations (Figure 1.3). In the pre-antibiotic era, infections of the pulp and periapical tissues were potentially serious and needed close monitoring. In the early days of antibiotics, it was found that most of these infections were readily susceptible to penicillin, and there- fore the spread of infection to regional spaces was often controllable by antibiotics. Today, it is recognized that pulp infection may be caused by organisms of different virulence (see Chapter 4), and that control of the infection is not always easily accomplished.
The flora of the mouth fortunately has relatively few pathogenic organisms, which usually have low virulence. Most are opportunistic, causing disease only in mixed infections or in hosts compromised by other diseases. However, organisms that are not normally pathogenic in the oral cavity may exhibit features of virulence if allowed access to the pulp or periapical tissues. Studies of the infected pulp have shown the presence of oral bacteria that normally inhabit the mouth, which do not normally cause disease. The apical periodontitis response to pulp infection may be viewed as a way of taming and coping with expressions of virulence by the infecting organisms. Thus, the pain frequently encountered in the early stages of disease development usually subsides in response to the tissue reactions. Furthermore, the initial expansion of the lesion of apical periodontitis is soon followed by periods of quiescence, possibly even regression or at least consolidation of the lesion. This dynamic process is accompanied in time by changes in the composition of the flora recoverable from the root canal.
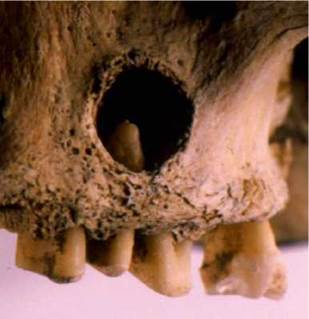
Figure 1.3 Apical periodontitis in an upper premolar of a woman's skull found in Iceland and dating to the 12th century. Trauma or wear caused exposure of the pulp with infection and lesion development.
Some forms of apical periodontitis have been associated with particular species dominating in the pulp canal flora. However, evidence from molecular analysis implies that endodontic infections may be more opportunistic than specific, and include many more species than previously thought (Chapter 4). Research into microbiological causes and interactions in apical periodontitis are imperative for improvements in diagnosis and treatment. Particularly, this would apply to the so-called “therapy-resistant” cases of apical periodontitis, in which infection per- sists despite apparently adequate root canal treatment, and to retreatment cases. Modern microbiological techniques have demonstrated an almost endless complexity and variability of the endodontic infections [19, 53], opening new avenues for research and expanding our understanding of the disease.
1.4.2 Infection Control
The outcome of endodontic treatment is dependent on use of an aseptic technique and antiseptic measures to prevent and/or eliminate infection. However, the critical role of infection control may not always be given the prominence it deserves. The transmission of hepatitis viruses has been an issue for a long time, and there is concern about prion transmission via contaminated instruments. The sterilization procedures for contaminated endodontic instruments have limitations, so there is a strong tendency towards applying single-use instruments. Most contemporary, machine-operated instruments are designed for single use, a practice that benefits the local treatment and prevents cross-infections.
1.4.3 Microbial Specificity and Host Defense
The host responses to root canal infection have been the subject of much research. There is great similarity between the pathogenic processes in marginal and apical periodontitis, many of the findings in periodontal research have direct relevance to apical periodontitis. Our understanding of the immunological processes involved in the development of apical periodontitis is expanding (Chapter 3). Microbiological variability and virulence factors in infected root canals have been demonstrated, and the bacterial flora may vary with the clinical condition of the tooth involved (persistent infection, therapy-resistant infection) (Chapters 4 and 11). Thus, different strategies of antimicrobial measures may be possible and even desirable depending on the microbiological diagnosis in a given case.
Reports of apical periodontitis with particularly aggressive microbes are fortunately very rare. Root canal infections with bacteria causing necrotizing fasciitis have been reported with very serious, even life-threatening consequences [48], and bacteria with resistance to common antibiotics may pose a problem, particularly in patients with impaired immune system [5]. However, it is important to remember that incomplete and inadequate root canal treatment can lead to infections requiring hospitalization and extensive medical treatment [17].
1.4.4 Endodontic Infection and General Health
The focal infection theory has been a source of both frustration and inspiration in dental practice and research. Both irrelevant and sometimes incorrect arguments and concepts were used to dictate an unnecessary wave of tooth extractions in healthy individuals for decades. Unsubstantiated opinions on the subject restricted clinical developments in the field of endodontics for a very long time. The controversy, however, has also sparked important new discoveries, and it is, even today, an important part of the frame of reference in studies of endodontic microbiology and host defense mechanisms.
1.4.4.1 Influence of General Health on Apical Periodontitis
Apical periodontitis and other disease processes may mutually affect each other. The root canal infection meets a response that is defined by the host's condition and dependent on genetic and constitutional factors, including systemic diseases. This variable tissue response may limit or allow expansion of the apical lesion, and it may promote or impair healing responses during and after treatment of the infection.
Diabetes is the classical example: it causes a general, reduced defense against infections and diabetic patients may have more and larger lesions [41, 45].
Smoking has a general, adverse effect on infection defenses and affects marginal periodontitis and wound healing negatively; it may also affect the incidence and healing rate of apical periodontitis [25, 36], but the effect may be weak or questionable [42], and confounding factors (age, marginal periodontitis) make conclusions about its effect difficult [25].
There is speculation that infection by the varicella zoster virus may be causally associated with root resorption and development of apical periodontitis [35, 47], but the evidence is very limited and inconclusive [22]. Similarly, other viral infections have been implicated in the pathogenesis of otherwise bacterially initiated apical periodontitis [20, 21, 26, 29]
Sickle cell anemia may cause pulp necrosis, preferentially in the mandible, apparently in the absence of microbial infection [12]. Sub- sequent infection causes classic apical periodontitis. The mechanisms for this increased susceptibility are poorly understood, but patients express high levels of genes for inflammatory cytokines [13]
Systemic medicaments influencing the immune and host response status in patients will influence the biological processes associated with apical periodontitis as well [9]. Moreover, as study designs and research methodologies become more sophisticated, dental diseases are found to be linked with diseases in other locations. Patients with inflammatory bowel disease have a higher prevalence of apical periodontitis and their lesions are larger [37], as they are in diabetic patients with poor glycemic control. Immuno- suppressive medicaments generally, or the diseases for which they are used, may not influence healing after endodontic treatment significantly [2, 33]. Other medicaments may favor healing; one study found that statin intake improved the incidence of healing after endodontic therapy [1].
There is a well-establish relationship of marginal periodontitis and preeclampsia and preterm births [34], which is traditionally linked with a raised level of inflammatory blood markers [10, 50]. Similarly, preeclampsia occurs more frequently in the presence of apical periodontitis [23].
Antimicrobial and pain-relieving medicaments have their place in treatment of apical periodontitis. However, when applied for other indications, they may mask symptoms [38], possibly impair body defenses [52] and the microbial population may develop resistance to the antibiotics.
Patients treated with immunosuppressants or who otherwise have compromised immune systems need special consideration. A number of the blood dyscrasias, notably leukemias, are associated with potentially serious sequels to apical periodontitis: infection spreads easily and may require extensive antimicrobial therapy. The irradiated patient is a special case: the incidence of osteoradionecrosis [8] after oral surgical procedures places high demands on effective, conservative treatment of endodontic conditions. Similarly, patients on bisphosphonate therapy by intra- venous injections may be at risk for tissue necrosis after surgical endodontics [31]. Case reports of complications from endodontic therapy in patients with reduced resistance [5, 46] point to the importance of meticulous and complete endodontic treatment in such patients.
1.4.4.2 Apical Periodontitis Affecting Other Tissues and Organs
Distant and systemic consequences of apical periodontitis is the other side of the coin.
Sinusitis may be induced by root canal infections of maxillary molars or, in very few instances, second premolars [49]. In the lower jaw, inflammation may cause paresthesia of the mandibular or mental nerve. These com- plications normally subside after successful treatment of the affected tooth [51].
Generally, any disease for which bacteremia poses an additional hazard is of concern in endodontics. Particularly, a history of infective endocarditis, congenital heart disease, rheumatic heart fever or the presence of an artificial heart valve or other susceptible vascular implants may necessitate the implementation of an antibiotic regimen in conjunction with the endodontic procedures. The magnitude of risk for cardiovascular complications due to bacteremia of dental origin is low and the need to controlling minor and chronic oral infections, including apical periodontitis [39], may be questioned [11]. The formalization of guidelines for antibiotic prophylactic needs by physicians and dentists to ensure safety for patients at risk has made decision making easier [28].
Atherosclerosis is central to the development of cardiovascular disease (CVD) [14]. The arterial plaques may be sites of colonization by microbes circulating during transient bacteremia, and oral infections may thus be a risk factor for CVD [44]. Specifically, apical periodontitis has been associated with an increased incidence of cardiovascular disease events [3, 4, 15, 16, 43]. Research into this association is complicated by a lack of strict criteria for assessing the nature of the periapical infection; radiographic observations give only the status at the time it is taken and cannot dis- criminate between ongoing infection and a healing lesion. Root-filled teeth, with or without a lesion, may represent a history of pulpitis or apical periodontitis. Pooling all root filled teeth with untreated teeth with apical periodontitis in an individual has been used as a measure of an “endodontic burden”; this is independently associated with CVD [16]. However, viewed isolated from other factors, root-filled teeth are associated with reduced incidence of cardiovascular disease [30]. The chain of events that may give apical periodontitis a role in CVD development is purely conjectural at this stage. However, the blood levels of sev- eral cytokines and other compounds associated with CVD are elevated in patients with apical periodontitis [3].
1.4.5 Tooth Loss and Replacement
Untreated apical periodontitis represents a chronic infection of the oral tissues at locations close to many important tissues. While these infections may remain quiescent for decades, they may also develop and spread with serious consequences for the individual. In the face of the risks of such chronic infection from involved teeth, their extraction and replacement by implants has been put forward and discussed as a viable alternative to endodontic treatment. The variable success rates (by strict criteria) of treatment procedures for the cure of apical periodontitis (Chapter 5) are sometimes used as an argument in favor of implants. However, what little evidence is available does not indicate a lower survival rate of endodontically treated teeth [7], and the superiority of tooth preservation compared to its replacement should be stated as a biological principle of preference. The challenge from other treatment options to endodontics as a discipline should act as a driving force to produce more scientifically solid evidence for the modalities of cure and prevention applied to our disease of interest, namely apical periodontitis.
1.5 Concluding Remarks
Pulp and periapical inflammation, the associated pain and the consequences of root canal infection remain significant aspects of dentistry today. New knowledge and insights provide better treatment opportunities and stimulate further research activities. The prevention and control of apical periodontitis has a solid scientific base, but the many variations in the clinical manifestations of the disease still leave technical and biological problems that need to be solved. Technological advances in treatment have made possible effective treatment of teeth that were previously considered untreatable, and further developments in microbiology, host biology and image technology are certain to improve the scientific foundation of endodontology in the near future.
References
1 Alghofaily, M. et al. (2018) Healing of apical periodontitis after nonsurgical root canal treatment: the role of statin intake. J Endod 44: 135-136.
2 Azim, A.A., Griggs, J.A., and Huang, G.T. (2016) The Tennessee study: factors affecting treatment outcome and healing time following nonsurgical root canal treatment. Int Endod J 49: 6-16.
3 Berlin-Broner, Y., Febbraio, M., and Levin, L. (2017) Apical periodontitis and atherosclerosis: Is there a link? Review of the literature and potential mechanism of linkage. Quintessence Int 48: 527-534.
4 Berlin-Broner, Y., Febbraio, M., and Levin, L. (2017) Association between apical periodontitis and cardiovascular diseases: a systematic review of the literature. Int Endod J 50: 847-859.
5 Blount, C.A. and Leser, C. (2012) Multisystem complications following endodontic therapy. J OralMaxillofac Surg 70: 527-530.
6 Chanani, A. and Adhikari, H.D. (2017) Reliability of cone beam computed tomography as a biopsy-independent tool in differential diagnosis of periapical cysts and granulomas: an in vivo study. J Conserv Dent 20: 326-331.
7 Chercoles-Ruiz, A., Sanchez-Torres, A., and Gay-Escoda, C. (2017) Endodontics, endodontic retreatment, and apical surgery versus tooth extraction and implant placement: a systematic review. J Endod 43: 679-686.
8 Chronopoulos, A. et al. (2018) Osteoradionecrosis of the jaws: definition, epidemiology, staging and clinical and radiological findings. A concise review. Int Dent J 68: 22-30.
9 Cotti, E. et al. (2014) An overview on biologic medications and their possible role in apical periodontitis. J Endod 40: 1902-1911.
10 da Silva, H.E.C. et al. (2017) Effect of intra-pregnancy nonsurgical periodontal therapy on inflammatory biomarkers and adverse pregnancy outcomes: a systematic review with meta-analysis. Syst Rev 6: 197.
11 Dayer, M. and Thornhill, M. (2018) Is antibiotic prophylaxis to prevent infective endocarditis worthwhile? J Infect Chemother 24: 18-24.
12 Demirbas Kaya, A., Aktener, B.O., and Unsal, C. (2004) Pulpal necrosis with sickle cell anaemia. Int Endod J 37: 602-606.
13 Ferreira, S.B. et al. (2015) Periapical cytokine expression in sickle cell disease. J Endod 41: 358-362.
14 Frostegard, J. (2013) Immunity, atherosclerosis and cardiovascular disease. BMC Med 11: 117.
15 Garg, P. and Chaman, C. (2016) Apical periodontitis - is it accountable for cardiovascular diseases? J Clin Diagn Res 10: Ze08-12.
16 Gomes, M.S. et al. (2016) Apical periodontitis and incident cardiovascular events in the Baltimore Longitudinal Study of Ageing. Int Endod J 49: 334-342.
17 Gronholm, L. et al. (2013) The role of unfinished root canal treatment in odontogenic maxillofacial infections requiring hospital care. Clin Oral Investig 17: 113-121.
18 Gutmann, J.L. et al. (2009) Identify and define all diagnostic terms for periapical/ periradicular health and disease states. J Endod 35: 1658-1674.
19 Iriboz, E. et al. (2018) Detection of the unknown components of the oral microflora of teeth with periapical radiolucencies in a Turkish population using next-generation sequencing techniques. Int Endod J.
20 Jakovljevic, A. and Andric, M. (2014) Human cytomegalovirus and Epstein-Barr virus in etiopathogenesis of apical periodontitis: a systematic review. J Endod 40: 6-15.
21 Jakovljevic, A. et al. (2016) Epstein-Barr virus infection induces bone resorption in apical periodontitis via increased production of reactive oxygen species. Med Hypotheses 94: 40-42.
22 Jakovljevic, A. et al. (2017) The role of varicella zoster virus in the development of periapical pathoses and root resorption: a systematic review. J Endod 43: 1230-1236.
23 Khalighinejad, N. et al. (2017) Apical periodontitis, a predictor variable for preeclampsia: a case-control study. J Endod 43: 1611-1614.
24 Kirkevang, L.-L. et al. (2014) Prognostic value of the full-scale Periapical Index. Int Endod J.
25 Kirkevang, L.-L. et al. (2007) Risk factors for developing apical periodontitis in a general population. Int Endod J 40: 290-299.
26 Lee, M.Y. et al. (2016) A case of bacteremia caused by Dialister pneumosintes and Slackia exigua in a patient with periapical abscess. Anaerobe 38: 36-38.
27 Lin, L.M. et al. (2009) Nonsurgical root canal therapy of large cyst-like inflammatory periapical lesions and inflammatory apical cysts. J Endod 35: 607-615.
28 Lockhart, P.B. et al. (2013) Acceptance among and impact on dental practitioners and patients of American Heart Association recommendations for antibiotic prophylaxis. J Am Dent Assoc 144: 1030-1035.
29 Makino, K. et al. (2015) Epstein-Barr virus infection in chronically inflamed periapical granulomas. PLoS One 10: e0121548.
30 Meurman, J.H. et al. (2017) Lower risk for cardiovascular mortality for patients with root filled teeth in a Finnish population. Int Endod J 50: 1158-1168.
31 Moinzadeh, A.T. et al. (2013) Bisphosphonates and their clinical implications in endodontic therapy. Int Endod J 46: 391-398.
32 Nair, P.N. (1998) New perspectives on radicular cysts: do they heal? Int Endod J 31: 155-160.
33 Ng, Y.L., Mann, V., and Gulabivala, K. (2011) A prospective study of the factors affecting outcomes of nonsurgical root canal treatment: part 1: periapical health. International Endodontic Journal 44: 583-609.
34 Parihar, A.S. et al. (2015) Periodontal Disease: A Possible Risk-Factor for Adverse Pregnancy Outcome. J Int Oral Health 7: 137-142.
35 Patel, K. et al. (2016) Multiple apical radiolucencies and external cervical resorption associated with varicella zoster virus: a case report. J Endod 42: 978-983.
36 Persic Bukmir, R. et al. (2016) Influence of tobacco smoking on dental periapical condition in a sample of Croatian adults. Wien Klin Wochenschr 128: 260-265.
37 Piras, V. et al. (2017) Prevalence of apical periodontitis in patients with inflammatory bowel diseases: a retrospective clinical study. J Endod 43: 389-394.
38 Read, J.K. et al. (2014) Effect of ibuprofen on masking endodontic diagnosis. J Endod 40: 1058-1062.
39 Reis, L.C. et al. (2016) Bacteremia after endodontic procedures in patients with heart disease: culture and molecular analyses. J Endod 42: 1181-1185.
40 Reisz, R.R. et al. (2011) Osteomyelitis in a Paleozoic reptile: ancient evidence for bacterial infection and its evolutionary significance. Naturwissenschaften 98: 551-555.
41 Segura-Egea, J.J. et al. (2016) Association between diabetes and the prevalence of radiolucent periapical lesions in root-filled teeth: systematic review and meta-analysis. Clin Oral Investig 20: 1133-1141.
42 Segura-Egea, J.J., Martin-Gonzalez, J., and Castellanos-Cosano, L. (2015) Endodontic medicine: connections between apical periodontitis and systemic diseases. Int Endod J 48: 933-951.
43 Singhal, R.K. and Rai, B. (2017) sTNF-R Levels: apical periodontitis linked to coronary heart disease. Open Access Maced J Med Sci 5: 68-71.
44 Slocum, C., Kramer, C., and Genco, C.A. (2016) Immune dysregulation mediated by the oral microbiome: potential link to chronic inflammation and atherosclerosis. J Intern Med 280: 114-128.
45 Smadi, L. (2017) Apical periodontitis and endodontic treatment in patients with type II diabetes mellitus: comparative cross-sectional survey. J Contemp Dent Pract 18: 358-362.
46 Stalfors, J. et al. (2004) Deep neck space infections remain a surgical challenge. A study of 72 patients. Acta Otolaryngol 124: 1191-1196.
47 Talebzadeh, B. et al. (2015) Varicella zoster virus and internal root resorption: a case report. J Endod 41: 1375-1381.
48 Treasure, T., Hughes, W., and Bennett, J. (2010) Cervical necrotizing fasciitis originating with a periapical infection. J Am Dent Assoc 141: 861-866.
49 Vidal, F. et al. (2017) Odontogenic sinusitis: a comprehensive review. Acta Odontol Scand 75: 623-633.
50 Vivares-Builes, A.M. et al. (2018) Gaps in knowledge about the association between maternal periodontitis and adverse obstetric outcomes: an umbrella review. J Evid Based Dent Pract 18: 1-27.
51 von Ohle, C. and ElAyouti, A. (2010) Neurosensory impairment of the mental nerve as a sequel of periapical periodontitis: case report and review. Oral Surg Oral Med Oral Pathol Oral Radiol Endod 110: e84-99.
52 Yang, J.H. et al. (2017) Antibiotic-induced changes to the host metabolic environment inhibit drug efficacy and alter immune function. Cell Host Microbe 22: 757-765.
53 Zandi, H. et al. (2016) Antibacterial effectiveness of 2 root canal irrigants in root-filled teeth with infection: a randomized clinical trial. J Endod 42: 1307-1313.