José M. Palacios-Jaraquemada1 and Claudio Hernán Bruno1
(1)
Fundación Científica del Sur, Lomas de Zamora, Provincia de Buenos Aires Argentina and School of Medicine, University of Buenos Aires, Avenida Hipólito Irigoyen 8680, Lomas de Zamora, Buenos Aires, B1832BQS, Argentina
José M. Palacios-Jaraquemada
Email: jpalacios@fmed.uba.ar
14.1 Introduction
Abnormal invasive placenta describes a group of conditions that produce the abnormal infiltration of placental tissue into the uterus or also into the surrounding tissues. This condition usually produces severe complications for the mother, such as massive haemorrhage, organ damage, organ failure and even death [1]. Within the term “accreta”, different degrees of placental invasion are involved, named as placenta increta and percreta according to a histological classification, but they were also described as a group clinically termed abnormal invasive placenta [2, 3] (AIP). Increasing rates of caesarean are an emergent problem worldwide, and they are related to the exponential risk from AIP. Ultrasound and Doppler are regarded as excellent tools to perform a diagnosis in these cases [4, 5]. Although diagnostic accuracy in all methods is operator dependent, this problem is particularly important in ultrasound studies because image acquisition is personally guided by the operator. Recently, this question has been analysed in some papers and also the necessity to create an international consensus to establish the most accurate value to the diagnostic signs [6].
MRI for diagnosis of placental disorders was used for the first time in 1991 [7] (pMRI, placental MRI). At that moment, it was a tool to confirm diagnosis in cases of doubt after a US study. At first, the operation time to perform MRI slices was slow, and artefacts due to foetal movements were frequent. For this reason and with the purpose to get better images, a paramagnetic contrast named gadolinium was used [8]. The use of this product in AIP was approved by the FDA in cases when maternal risk was greater than a potential side effect in the foetus. But some years later, technical improvements in MRI equipment and software made it possible to reduce times to perform the slices, and a different acquisition mode turned unnecessary the routinary use of contrast [9]. MRI T2 fast techniques provide a natural white contrast for liquids, and although names of sequences could vary among equipment, techniques are equivalents (Table 14.1). Due to the fact that most cases of AIP are lower and anterior, when the bladder is filled, contrast with the placenta and the uterus (hypointensive) is excellent.
Table 14.1
Equivalences among MRI equipment
|
MRI equipment |
Picker 1.5 edgepicker 1.5 edge |
Siemens Espree 1.5 T |
GE 3 T signa HDxt |
Philips Achieva 1.5 T |
|
Sequences |
FSE |
Haste |
ss-FSE3 |
ssh-TSE4 |
|
TR |
3500 |
850 |
2399 |
712 |
|
TE |
168 |
101 |
141 |
110 |
|
FOV |
290–360 |
300–400 |
300–400 |
300–400 |
|
Thickness |
8 |
6 |
6 |
6 |
|
Matrix |
192 × 256 |
320 × 320 |
320 × 320 |
288 × 288 |
During the second stage, obstetricians requested for pMRI to know the degree of invasion [10], although this difference usually didn’t modify the tactic or surgical technique. In addition, it is known that different degrees of invasion usually coexist in the same patient [11]. This difference was recognised by a pathologist, who says that a simple sample is not a gold standard for diagnosis of abnormal invasive placentation [12].
During the third stage, pMRI was requested to know the anatomy of invasion, especially to examine the parametrium and the interface among placenta, invaded myometrium and the bladder [9–13]. But for a few authors, this information didn’t improve or modify the surgical plan. Part of this is a consequence of using long-standing techniques (1933) such as to leave the placenta in situ [14], but mainly because obstetricians are not formally trained to read these images. This situation also happens with radiologists, who perform the study without previous formal training in AIP images [15]. The lack of background after the surgery to re-evaluate the image findings is part of this problem. At a point, pMRI has a big difference compared with ultrasound, because US is performed mainly by obstetricians who have access of to perform the surgery, a situation that allows building a background of informed images immediately.
However, pMRI is an excellent tool for abnormal invasive placenta evaluation, because it is a multiplanar study with total acquisition of the affected area [16]. This fact makes it possible to study specific zones in detail not easily accessible with ultrasound, like the parametrium or posterior invasions. Besides, and as it was mentioned before, different degrees of invasion may coexist in the same patient, and although it is not common, the same patient may have an anterior placenta accreta and a massive parametrial invasion, information that completely modifies the surgical technique [17].
Due to the potential life threat of this condition, the use of different image techniques increases confidence before starting the surgery. In other words, ultrasound, Doppler and pMRI are not different methods to confirm the same diagnosis; they are complementary techniques to obtain different information to make active decisions in order to plan and anticipate further complications [18].
14.2 Definitions and Diagnostic Problems
For a long time, obstetricians and image studies used a histological classification of placental invasion to inform images of AIP cases, but these criteria may end in surgical problems or misinterpretations [11]. Histological classification names as placenta accreta are those placentas that invaded the myometrium without interposition of tunica basalis. Those placentas that invade the myometrium deeply are called placenta increta, and the term percreta is for those in which placental invasion reaches the myometrium or passes beyond it. But, due to the fact that most abnormal placentations are located in or near the previous scar, most of them should be called percretas, because the scar area is generally thin and the placenta usually reaches the serosa, a fact that from a surgical point of view is not right. So, the use of a histological classification can be confusing to perform a prenatal diagnosis of AIP. Likewise, when the placenta protrudes by a dehiscent scar, this would be termed percretas from histological point of view, a fact that is incorrect and it may be the cause of definitive over treatment like hysterectomy, even in young women [19]. For this reason, we need to know that diagnosis of AIP has some limitations which may confirm and also possibly modify the prenatal plan after surgical exploration.
From a prenatal and surgical point of view, it is possible to classify these placental invasions into 3 groups:
1.
2.
3.
AIP grade 0, 1 and 2 may also extend to the lateral part of the uterine segment (parametrium), although the most common is the grade 0 or 1. Grade 2 is particularly unusual but common in initial uterine segment lateral implantations (Fig. 14.4). The presence of vessels associated to this type of lateral invasions is evident in axial pMRI slices. This surgery is usually a nightmare because access to this deep and narrow area is quite difficult due to the presence of communication vessels from the ureter and pelvic wall branches from the iliac internal and even from the iliac external artery [11].
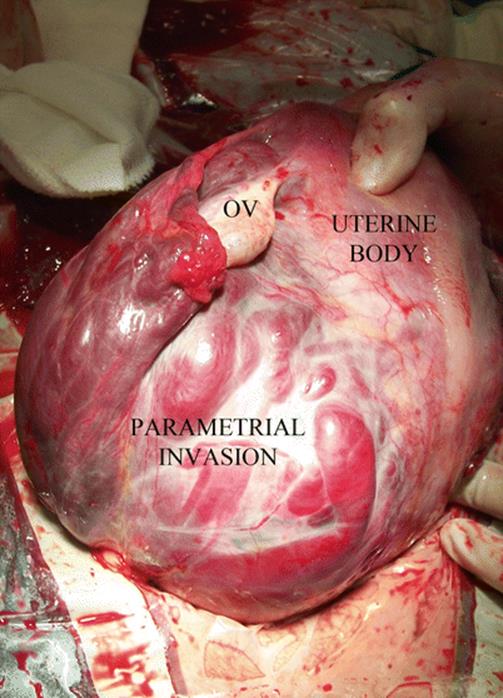
Fig. 14.4
Surgical view: complete and left lateral parametrial invasion. All lateral side of the uterus is infiltrated by the placenta. Patient background: 1 CS, curettage after caesarean by aberrant low cotyledon, second pregnancy after 4 months of previous CS
This disparity between histology, images and surgical features can be confusing to make decisions, especially for those obstetricians not specialised in this condition (Fig. 14.5). For this reason, agreement on diagnosis and terms is highly recommended to know exactly what type of invasion is described and then which was a better treatment for them. Many centres discuss all the prenatal and postnatal findings among all specialists involved, who comment on weaknesses and strengths in diagnosis in order to learn and modify surgical approaches and techniques. Although this learning activity is enriching in terms of improving diagnosis and treatments, for many reasons, it is not a wide-ranging practice.
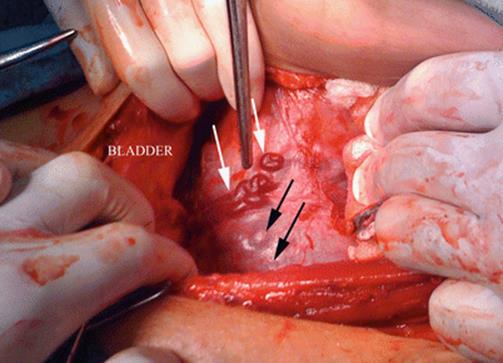
Fig. 14.5
Surgical view: parallel vessels over uterine segment surface (white arrows). Peripheral placental circulation (black arrows) plus parallel vessels could mimic placenta percreta. Surgical exploration in patients with AIP is necessary to confirm prenatal diagnosis and avoid under or over treatment
14.3 Obstetric MRI Versus pMRI
Although at first both studies, obstetric MRI and pMRI, may be considered the same, analysis of the placenta needs a special attention in order to get significant and useful images for diagnosis and surgical planning. Obstetrical MRI is indicated to study a wide range of foetal anatomy, such as spine, neck, nervous system, thorax, urogenital tract and extremities, among others. Placental MRI places a special focus on the placental anatomy in relation to the surrounding tissues, especially with the bladder and pelvic organs. The information provided by pMRI is particularly important as a prenatal guide to perform a surgical approach and also the vascular control. The image must include all the pelvic anatomy very clearly and not in the peripheral area as an obstetric MRI.
14.4 Placental MRI: Diagnosis
During the last decade, there were a great number of studies that analysed and compared the sensitivity and specificity of pMRI with ultrasound and Doppler [20]. None of them showed significant differences, although availability and cost favour ultrasound. It is common that after initial work by Dr. D. Levine [21], most authors accepted some pMRI advantage for the posterior invasions in comparison to US diagnosis [22]. However, none of these studies showed any image of posterior invasions. Analysis of ultrasound diagnostic failure in AIP cases is not demonstrative of specific causes. It is usual to read in papers that ultrasound was not able to perform a diagnosis, but there is no analysis regarding why. It is probable that this lack of prenatal diagnosis is associated to expertise and interpretation of US signs [6]. Recently, some authors have cast some doubt on the values of classic ultrasound signs, to indicate a lower value than what was published before. But there are no formal experiences to evaluate the same US study among obstetricians, although recent international experiences have shown a wide range of opinions and diagnosis among AIP experts and nonspecialists. These differences were minor or inexistent among AIP specialists, a fact that suggests two possible levels of competence: one that can be made by nonspecialists, who can determine cases which qualify as highly suspicious, and other levels that can be made by specialists in AIP diagnosis, who are able to provide accurate information to make definitive decisions [13].
Initially it was believed that pMRI improved or corrected these diagnostic differences, but as it happens with ultrasound or with Doppler, pMRI is a study that needs a deep experience and continuous feedback [11]. Some details about AIP are relevant to know why study preparation is important. Among positive or significant signs of MRI signs, like presence of lagoons, thinning of myometrium (Fig. 14.6) the existence of newly-formed vessels is one of the most important to diagnose AIP. These vessels are a consequence of the action of growth factors over the microanastomotic vessels between the uterus and the surrounding tissues, usually the bladder. These vessels are not strictly newly formed, because they are connected organs although are unnoticed to the naked eye. Due to the fact that they are not under normal arterial pressure, the tunica media (muscular) is poorly developed or practically inexistent [11]. For this reason, these newly formed vessels collapse very easily. To be evident in the pMRI, they mustn’t compressed by the bladder. For this reason, the bladder could be semi-filled at the time of performing a pMRI, because it is the best way to see the abnormal circulation. If the bladder is empty, the placenta crushes vessels and the bladder walls against the symphysis, and they are not seen in the study. On the other hand, if the bladder is overdistended, the newly formed vessels are collapsed against the placenta, and also, they are not visible by pMRI [9]. A better visualisation of the uterine-vesical interface could be done if the patient intake 600 ml of liquid 45 min before the study.
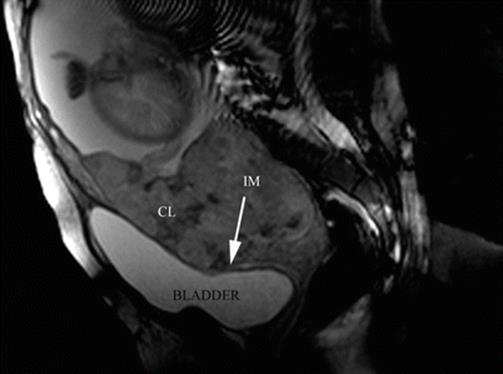
Fig. 14.6
Sagittal T2 image: confluent lagoons (CL) are evident near to uterine-bladder interphase. The uterine segment myometrium is interrupted (IM) or extremely thinning. In this area, the placenta reaches to the uterine serosa
Anterior AIP is the most common location worldwide, because it is also the most common site of the previous scar (caesarean). However, the lateral part of the uterus (parametrium) could not be seen properly by US due to the lack of natural contrast or liquid. But in recent years, some studies have investigated the possibility to explore the parametrial area in cervical cancer [23, 24]. Although parametrial invasions are not common in all countries, it is a condition relatively frequent associated after unsafe abortions. This variety must also be suspected in cases of lateral retained placenta, curettage by aberrant cotyledon or by curettage after short-time caesarean interval [11]. Coronal and axial slices of pMRI are really accurate (Figs. 14.7 and 14.8) to demonstrate this kind of invasion [13]. As it was commented before, lateral invasions may have two subtypes: (1) There is a lack of support of myometrium, because it appears thin. The placenta clearly protrudes until the lateral pelvic wall. (2) Besides placenta protruding (lateral bulging) a vascular signal of engorged vessels is evident around the invasion. The last type of invasion is very rare, though highly problematic to solve. Although other invasions are suitable to leave the placenta in situ, this is particularly dangerous in parametrial invasion type 2. Postpartum uterine contractions may produce an unexpected laceration of the weak lateral myometrium and start a massive haemorrhage. An emergency scenario includes a patient with a severe hypovolaemic shock, intra-abdominal or retroperitoneal bleeding and invaded placenta in a deep space plenty of enlarged vessels, which is a true surgical nightmare [25]. Except for a few special cases, this episode is the cause of death, because it is almost impossible to solve all problems very quickly. Although these cases are not reported or published, the specialist knows about them through informal talking with other specialists in congresses.

Fig. 14.7
Coronal T2 image: MRI was requested by US doubt of anterior invasion (black asterisk). Parametrial invasion (PI) was evident in the right side of the uterus. After study, the patient admitted having an abortion
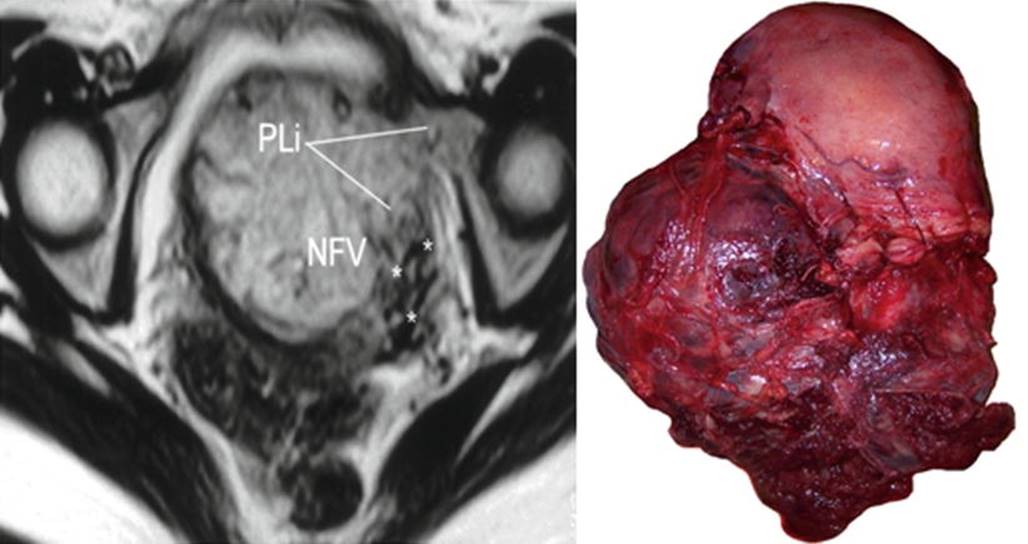
Fig. 14.8
Left: axial T2 image, PLi parametrial invasion, NFV newly formed vessels (asterisks). Right: massive low parametrial invasion. Uterine segment was not invaded by the placenta. MRI was requested by US doubt of anterior invasion
Although the number of cases with parametrial invasion is not known, diagnosis of this variation could imply several changes in the surgical plan [13]. Parametrial types 1 and 2 are formal indications of ureteral catheterisation in order to prevent unexpected damage during resective procedures (hysterectomy). When catheterisation is not possible, surgical identification is extremely useful to prevent ureteral damage. Although this manoeuvre is usually performed by urologist, it is sometimes very difficult because of the lack of anatomical landmarks, which are modified by placental protruding against a lateral pelvic wall [26]. Diagnosis of parametrial invasion type 2 suggests a possibility of heavy bleeding from vessels that have no relation with the uterine artery. In these cases, an accurate and efficient method of proximal vascular control must be performed before starting a hysterectomy. In the low parametrial area, the vessels come from different sources, and embolisation is usually not useful at all (Fig. 14.9). Attempt to control in these cases implies a high risk of failure and also a nontarget embolisation.
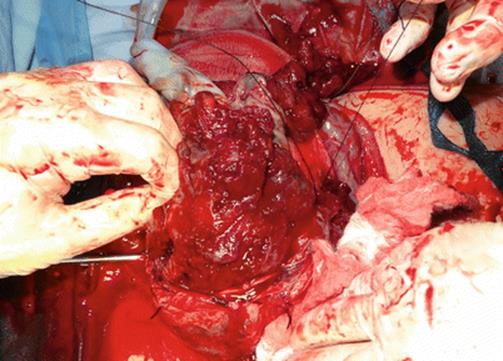
Fig. 14.9
Surgical view: unexpected parametrial invasion discovered during hysterectomy. Iliac internal occlusion was not effective to stop the bleeding and dissection was unmanageable. Emergency aortic balloon was inserted and an expertise group was called to complete the surgery
14.5 Placental MRI: Topography of the Invasion
Historically, 2 pedicles have been described for uterine irrigation, the uterine and the ovary arteries. However, after the use of embolisation to manage different types of obstetric haemorrhage, some complications and failures couldn’t be explained according the classic anatomy. A preliminary study made to use a uterine compression device (myomas treatment) demonstrated that lateral compression of uterine tissues above uterine arteries produces uterine necrosis in 6 h [27]. This was the first published study that demonstrated that the upper pedicle (ovary and round ligament artery) is not able to replace the uterine blood flow after uterine artery ligature or embolisation. A few years later, an anatomical study showed a thick and wide communicated anastomosis between vaginal and uterine arteries [28]. Irrigation areas from upper and lower uterine pedicles are different and also their origin. Uterine arteries arise from the anterior division of iliac internal artery, while vaginal ones arise from the posterior division. A practical division of uterine irrigation areas is the peritoneal reflection, which determines area S1 (above peritoneal reflection) and area S2 below this. S1 sector is mainly irrigated by the uterine arteries, while S2 sector by collaterals of the pudendal internal arteries. The topography of placental invasion indicated which is the most effective method for proximal vascular control [29]. For S1 invasions (less frequent in AIP), anterior iliac internal control is effective, but for S2 placental invasions, it is necessary to control the blood flow of the pudendal internal branches and their anastomotic connection, so the most accurate vascular control is the iliac common or aortic vascular control [30].
Identification of S1 and S2 areas can be established by pMRI, sagittal slice (Fig. 14.10). A line that perpendicularly crosses the middle of the posterior bladder wall determines an upper area named S1, which mainly corresponds to the uterine body, and an area below this line, named S2, which involves the lower segment, cervix and upper vagina [9]. Most part of AIP is located in S2 area, which also explained the high rate of failures with the use of uterine or internal iliac vascular control.
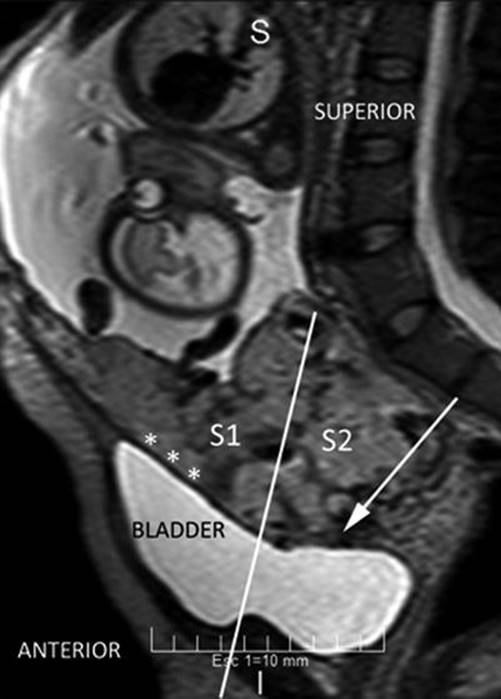
Fig. 14.10
Sagittal MRI T2 image: perpendicular plane which divide the posterior bladder wall determine an areas S1 and S2. White arrow shows an interrupted myometrium. Lagoons and vessels are clearly visible in the myometrial-bladder interphase
14.6 Placental MRI: Depth of Invasion
During a second stage, pMRI was requested to know the depth of placental invasion [10], especially to know whether the bladder is invaded. But bladder invasion is a vascular phenomenon, completely different from tissue invasion than neoplasia. In these cases, the placenta takes irrigation from the external layer of the bladder, but the state of uterine-bladder interface may be different according to thinning of myometrium, bladder wall, fibrous tissue between them or a combination of all these variables. For some authors, when the bladder is compromised, this is a formal indication to leave the placenta in situ, but this is not a really mandatory option. Placental MRI allows seeing this interface in great detail in axial T2 slices. When this series (axial T2) is performed perpendicularly to the posterior bladder wall, the anatomy of the placental-bladder interface is not distorted and shows a clear anatomy [13]. This interface can be distorted by peripheral circulation in a placental surface (engorged superficial vessels and lagoons) or by the presence of newly formed vessels between the placenta and the bladder. The presence of circular hypointensive signals (newly formed vessels) in the lower and posterior part of the bladder (cervicotrigonal area) is a reasonable sign of a very difficult dissection [13].
Placental MRI is a total acquisition study; for this reason, it allows analysing the whole invaded area, which can show different invasion degrees in the same patient. Sagittal slices are more suitable to evaluate the integrity and thickness of the anterior uterine wall in perspective. This is especially helpful when conservative-resective technique is the plan, because it allows knowing if there is healthy myometrium below the invaded area to perform a uterine anterior wall repair (prior to the surgery). As it was mentioned before, parametrial invasion is not a common event, but when it’s present, the surgery could change radically. For this reason, it is important to highlight that pMRI is not an initial method to perform diagnosis of AIP, but some additional information, especially topographic, like parametrium [9–22], may not be available with other methods. Although some specialists consider that the cost of MRI does not justify its use in AIP, when serious complications appear [31–35], it is obvious that the cost is completely validated to reduce severe complications.
14.7 Placental MRI: Diagnostic Signs of AIP
Diagnostic signs of AIP on pMRI are similar to those described in US [13, 23–38]. Interrupted or myometrial thinning is a common sign but not specific of AIP. Previous caesarean scar is prone to produce some degree of dehiscence when the placenta is located in the lower segment. For this reason, this sign itself is not indicative of AIP, although histologically it could agree with a placenta accreta, increta or percreta. The presence of lagoons, especially multiple, confluent and intercommunicated, is probably the most reliable sign of AIP (US and pMRI). Tenting of bladder was described as a sign of AIP [20], but this sign is not specific and surgically represents the bladder adhesion to the previous scar. Although uterine bulging was described as one of the useful signs for diagnosis, this is not specific at all, because a simple uterine scar dehiscence in a placenta previa can produce a same sign. According to some authors, absence of dark placental bands excludes a diagnosis of AIP, but this asseveration is made based on a small series. Presence of lobulations is frequent in AIP [38], although their absence is not an exclusion sign. For this reason, and as it happens in US analysis, prenatal diagnosis of AIP is based on a group of signs, although some of them are more predictable of AIP than others. Full area acquisition of MRI allows re-evaluation anytime by different observers (Figs. 14.11 and 14.12), a fact that is not usually possible for US, because image acquisition is dependant of the operator.

Fig. 14.11
Initial radiologic evaluation was informed as anterior placenta accreta, which was agree with US inform. After that, a second MR expert described a left parametrial invasion (PI) In asterisk, interrupted lateral myometrium is showed
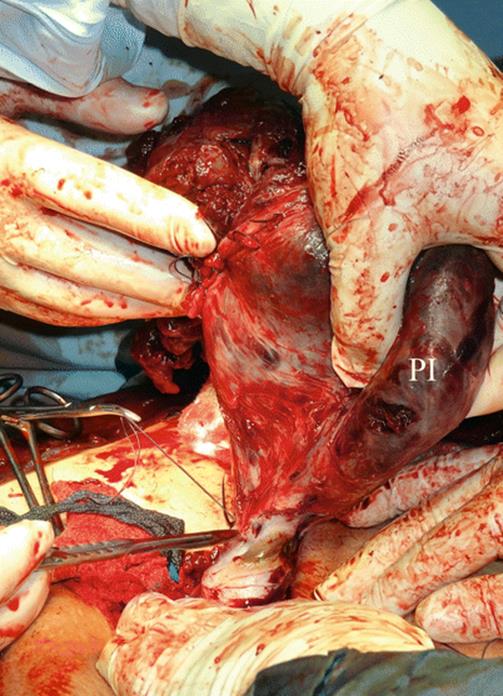
Fig. 14.12
Surgical view of Fig. 14.11: after dissection, left parametrial invasion (PI) is clearly seen
14.8 Placental MRI: Information Accuracy
Only a few obstetricians know that initially, the use of ultrasound for AIP diagnosis was retrospective [39]. This knowledge started in 1983, and it was continuously studied by a group of excellent researchers, who found objective signs to perform an accurate diagnosis of AIP later. Even today, the value of these signs is under investigation, because there is not always a right correlation between US signs and surgical features [40]. This is not a minor detail, because it is possible that mild cases end in hysterectomy (when uterine preservation is possible) or that invasion has been underestimated and the result is worse than it was expected. For this reason, it is essential that radiologists and obstetricians work together to provide and receive feedback, a fact that will improve future diagnosis. There are some logistic problems that hinder this process: (1) Obstetricians have a formal training in US and Doppler but not in the interpretation of pelvic MRI images; (2) obstetricians perform the US, Doppler and frequently also the surgery, which provides immediate feedback; and (3) in only a few units are radiologists invited to review the images after the surgery. As a result of these problems, potential help with pMRI is usually limited for the obstetricians. Most part of pMRI reports are evaluated by radiologists without extensive experience in placental disorders, and since it is common that reports only confirm the US findings; consequently, it is logical that some obstetricians do not feel comfortable with this information. From a surgical point of view, the obstetrician who plans to operate on a patient with MAP needs to know (1) if the diagnosis is reliable and agrees with the medical background; (2) the topography of the placental invasion, with special attention to the bladder, parametrium and trigonal area (Figs. 14.13, 14.14, 14.15, 14.16, 14.17 and 14.18); (3) the depth of invasion by areas; and (4) if placental invasion is associated to a presence of newly vascularisation or with tissue dehiscence [30].
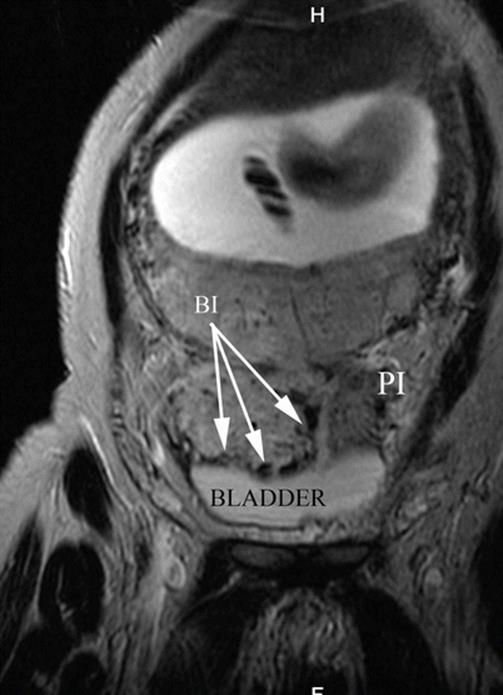
Fig. 14.13
Coronal MRI T2 image: white arrows, massive bladder invasion by AIP type 2 (percreta). Notice that placenta is lobulated. Naturally liquid contrast of bladder allows the proper identification of newly formed vessels, given their hypointense signal. PI parametrial invasion
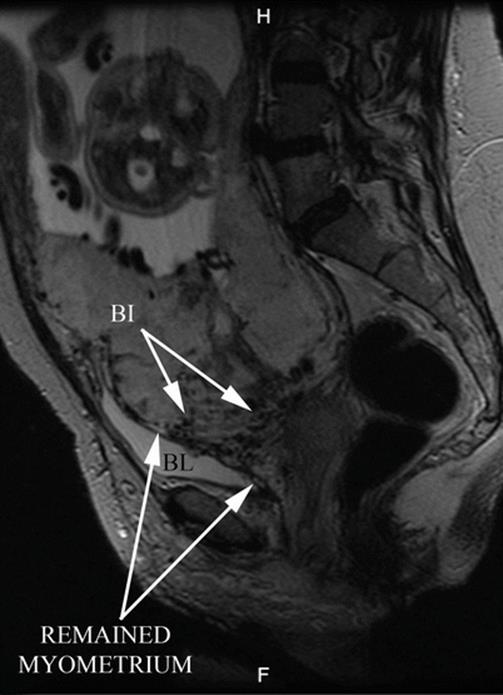
Fig. 14.14
Sagittal MRI T2 image: white arrows, bladder vascular invasion. Placenta is lobulated and the anterior myometrium is interrupted. Confluent and irregular lagoons are evident
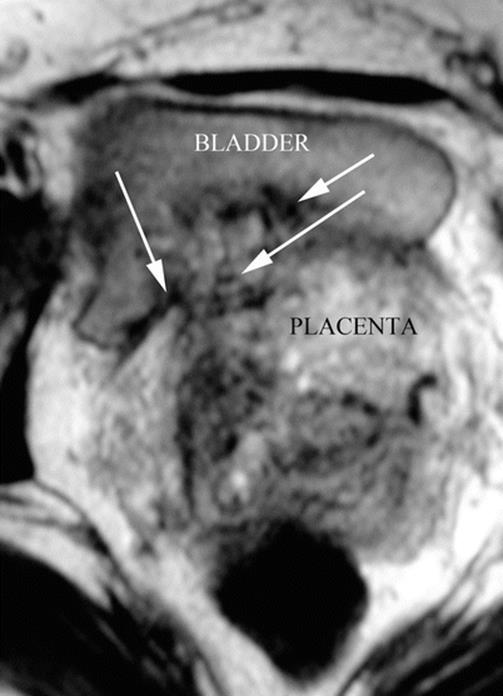
Fig. 14.15
Axial MRI T1 image: axial slices made perpendicular to the posterior bladder wall are the most useful to see a vesical invasion. In this case, fibrous adherence between bladder and anterior myometrium is seen. White arrows: newly formed vessels. AIP type 2 (percreta)
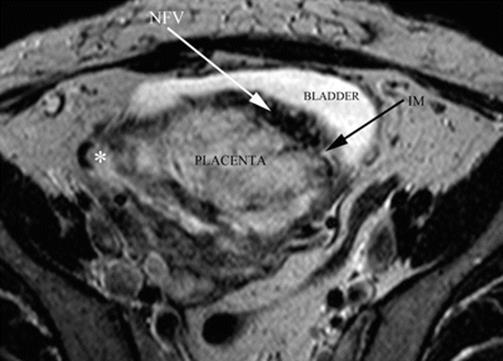
Fig. 14.16
Axial MRI T2 image: white arrow, newly formed vessels between uterus and the bladder. Black arrow: interrupted myometrium. AIP type 2 (percreta) myometrial-bladder interphase is completely distorted
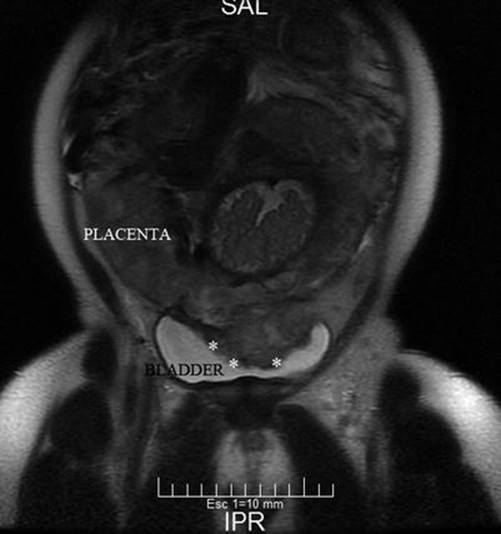
Fig. 14.17
Coronal MRI T2 image: placenta is seen inside of the bladder (asterisk). Part of them was a uterine dehiscence and another a AIP grade 2 (percreta)
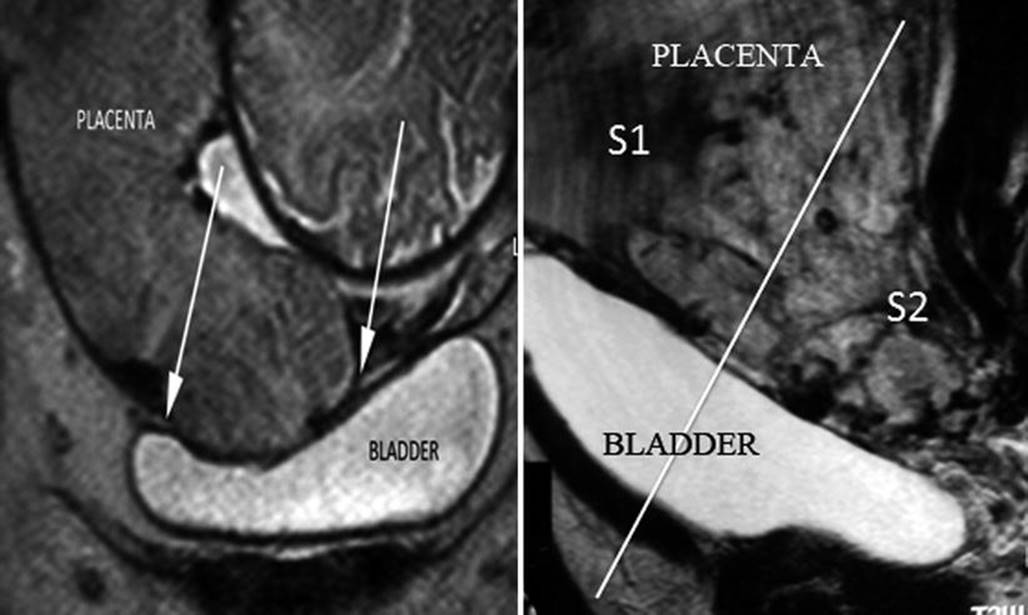
Fig. 14.18
Left: coronal MRI T2 image, notice that bladder is seen inside the bladder but without any newly formed vessels. The myometrium is clearly interrupted. Lack of other features of AIP is in favour of AIP grade 0 (uterine scar dehiscence) which was proved in surgical exploration. Inadequate evaluation of this study can precipitate to perform an unnecessary hysterectomy. Right: sagittal MRI T2 image, detail of bladder vascular invasion (lagoons and newly formed vessels are evident). S1 and S2 uterine sectors are divided by line that crosses the middle of posterior bladder wall
Ultrasound diagnosis is highly reliable when it is performed by a trained operator. In this respect, the qualified specialist knows small differences among signs perfectly to make an excellent report. In some countries, when suspicion is noticed during ultrasound scan, a full study is then performed by experts to distinguish between slight, hazardous or false-positive cases. However, specialists may recommend pMRI to confirm diagnosis in special locations (Fig. 14.19) or when signs are small or inconclusive, like posterior uterine wall [41, 42]. Except for a small indication, comparative diagnoses between qualified ultrasound and pMRI are minor [43].
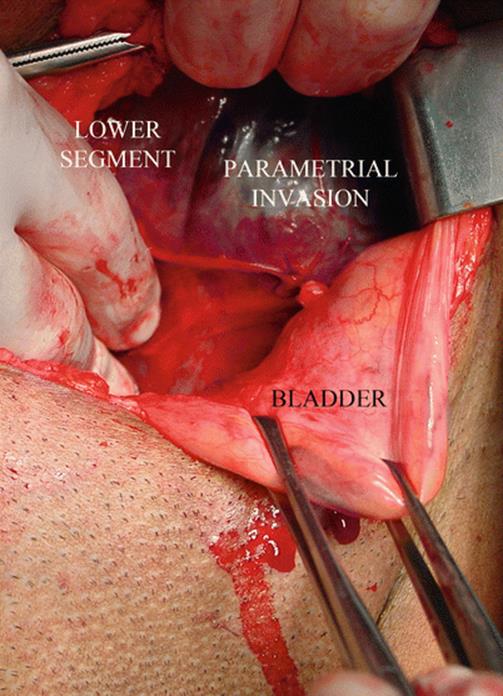
Fig 14.19
Surgical view: US examination suspects some compromise of lateral wall, but pMRI was refused by patient. During surgical exploration, lateral parametrial involvement was confirmed. Complete posterior wall bladder dissection is mandatory to see this kind of invasion
A map or topography of invasion is essential to plan the surgical approach, especially in resective procedures like hysterectomy or one-step conservative surgery [44]. Both need an accurate dissection to disconnect invaded and normal tissues; in this respect, three areas deserve a special care: the bladder, the trigon area and the parametrium. Bladder invasion is not a usually tissue invasion, it is a vascular invasion (Fig. 14.20); for this reason, it is not necessary or mandatory to resect part of the bladder in surgical treatments of placenta accreta or percreta. Initially, the placenta grows into the uterine scar (poorly vascularised area) and stimulates the development of newly formed vessels among surrounding tissues to take additional blood for the placenta. By proximity, the most affected organ is the bladder, in special, the detrusor (vesical muscle), because it has plenty of anastomotic vessels. Diagnosis of vascular connection between bladder and uterus is known as vesical invasion, and for many authors, this is a contraindication to resective surgery. However, topographic identification of these vessels can modify the surgical technique. Upper and posterior wall of bladder is the most common location of newly formed vessels. In this respect, multiplanar pMRI allows to identify placement and extension of vascular bladder invasion [13]. Due to the shape of the pelvis, lower vascular invasions are more difficult to operate on especially those located between the trigon and the cervix. This vascular invasion is produced by enlargement of colpo-uterine anastomosis in a narrow and fibrous space. The location of this vascular hyperplasia is particularly suitable to identify by pMRI, as a hypointense signal like a ball, in the posterior and lower wall of the bladder. It is very important to know that it is a secondary vascular effect of placental invasion, but this is not a tissue invasion in itself. Image identification of this vascular invasion may change the resection from total to subtotal, because unwanted vessel cutting could produce massive and uncontrollable bleeding. Growing of vessels in the detrusor thickness frequently reduces resistance to traction forces, a fact that makes this area particularly breakable. An unpublished case of an attempt to cervical resection in known cervicotrigonal hyperplasia ended in a complete trigon detachment and a massive haemorrhage, which was only controlled after an aortic compression. To date, this type of vascular hyperplasia has only been described in pMRI studies, and it is clinically associated to cases of macroscopic haematuria [13]. This relationship between macroscopic haematuria (Fig. 14.21) and cervicotrigonal vascular hyperplasia (in AIP cases) also has other therapeutic implications. Due to the fact that newly formed vessels do not have a tunica media developed, the use of electrocautery by cystoscopy to stop the haematuria is particularly dangerous, because it worsens the vascular damage and it can cause life-threatening bleeding within minutes [1].
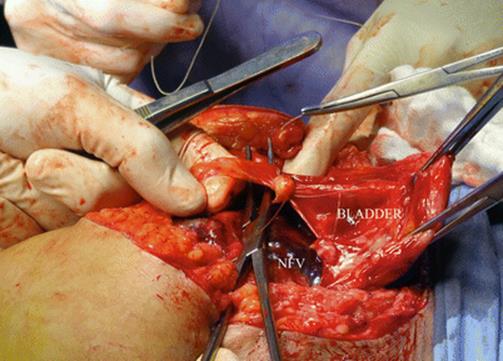
Fig. 14.20
Surgical view: vascular invasion from uterus to the bladder is gently separated between ligatures. This manoeuvre allows avoiding resecting any bladder tissue during the surgery. AIP grade 2 (percreta)

Fig. 14.21
Macroscopic haematuria is not frequent in AIP cases, but it is strongly associated to trigon invasion. These cases need a special attention and full resources to operate on. Bleeding from thick vessels between trigon and cervix is very difficult to solve, and it could be cause of life-threatening haemorrhage
Placental invasion depth is not homogeneous (Fig. 14.22); this fact is well-known by pathologists, and histological reports may agree or not with the surgical findings depending on which area the sample was taken. This assessment is useful on resective surgery and especially during dissection manoeuvres, because extension of the invaded area may promote additional measures, such as ureteral catheterisation or a specific type of vascular control. Although diagnosis of invasion depth is particularly useful in surgical resective-reconstructive techniques [44], there has not been a randomised trial that demonstrated its benefits.
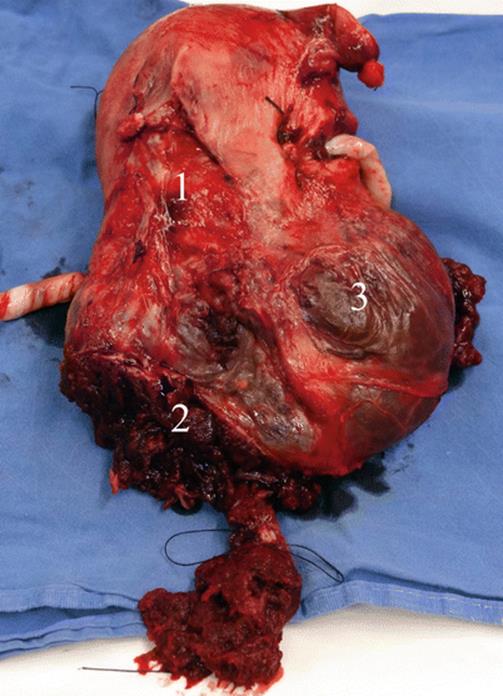
Fig. 14.22
Hysterectomy specimen: different degrees of invasion could be seen in the same specimen: (1) AIP grade 1 (accreta), AIP grade (2) (percreta) and also parametrial invasion (3). Depending in which place the pathology sample is taken, histological diagnosis could be completely different to a surgical features (not gold standard diagnosis)
The presence of new vascularisation associated to AIP indicates a different approach or surgical treatment. Although these differences may be seen by US, it can be difficult to differentiate peripheral placental circulation (lacunae blood flow) from newly formed vessels, especially when the bladder wall is very thin. In this respect, diagnosis may be difficult by US and also by pMRI, although analysis of multiplanar images may bring some benefits for MRI, because it is possible to examine the same area from different views. In any case, complementary information from any source is especially useful to make active decisions [45]. This is not a minor issue, because in recent years, many cases of AIP have happened after the first caesarean, so a decision whether or not to perform a hysterectomy must be carefully taken.
14.9 Placental MRI: Feedback of Informed Images
Retrospective analysis of images after surgery is essential to improve studies, knowledge and information to make future decisions. Radiologists are interested in this, but this activity also depends on interactive teaching between obstetricians and radiologists. Feedback may include fix MRI and surgical images, especially those that show all area below the peritoneal reflection (S2 area). However, sometimes it is not easy to obtain them. In some cases, only the presence of a bulging area and the presence of newly formed vessels is an indication to leave the placenta in situ or to perform a hysterectomy; consequently, a retrospective analysis is never done.
Another point is to know whether prenatal differences in diagnosis could modify the surgical plan. Current options include hysterectomy, to leave the placenta in situ, and other resective-reconstructive techniques such as one-step conservative surgery (Figs. 14.23, 14.24, 14.25, and 14.26) or the triple P procedure. Besides prenatal diagnosis, additional information to make decisions in the two first options is not necessary. Maybe for this reason, some obstetricians cannot use pMRI anymore. However, an unexpected invasion may be discovered in the surgical room and makes it more difficult or quite impossible to perform the hysterectomy. Unfortunately, these cases are not published for many reasons and the problem is unknown to most obstetricians. Although cases like this are not common, maybe the cost-benefit relation deserves that each case to be fully studied by images before making a surgical plan. One-step conservative surgery needs topographic information, to know the extension and placement of specific details to plan surgical actions. In the triple P procedure, the dissection reaches to the peritoneal reflection, above the bladder, so topographic information below this is not necessary.
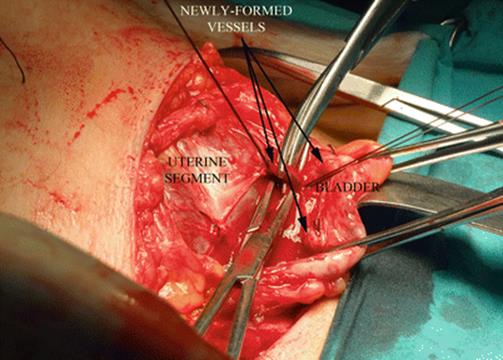
Fig. 14.23
Surgical view (one-step conservative surgery): all connection between placenta-myometrium and the bladder are separated between ligatures to expose invaded tissues
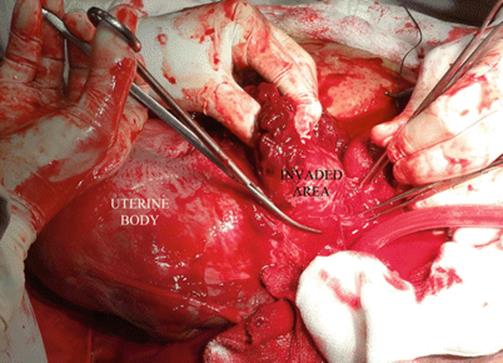
Fig. 14.24
Surgical view (one-step conservative surgery): after delivery, all invaded tissues and the entire placenta are removed
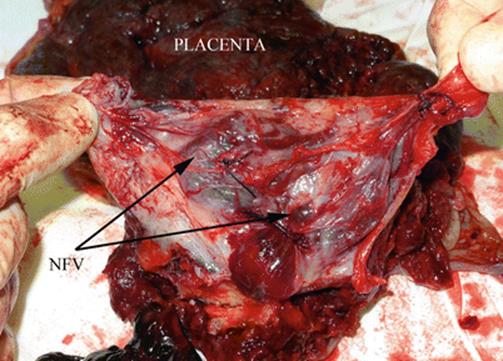
Fig. 14.25
Surgical view (one-step conservative surgery): the picture shows the resection of invaded myometrium (pathological sample). It is possible to identify the ligated newly formed vessels between uterus and the bladder
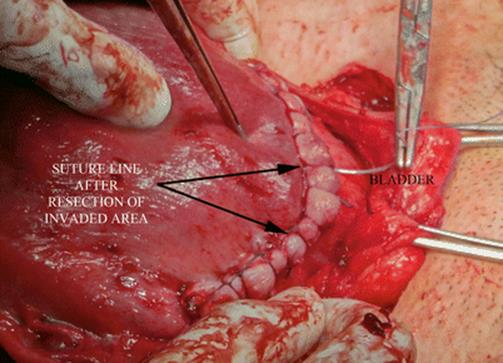
Fig. 14.26
Surgical view (one-step conservative surgery): uterine aspect after two planes of suture. Identification of healthy tissue below of invasion is mandatory to perform this conservative-resective technique
14.10 Placental MRI: AIP and Abortions
In some countries in which abortion is illegal, it is very important to examine all uterine walls when abortion is confirmed or suspected. As it is showed in Fig. 14.7, myometrial damage after abortion could result in unsuspected AIP. Posterior location of AIP is not frequent at all, but it could be diagnosed easily by pMRI (Figs. 14.27, 14.28). Low-posterior AIPs are usually irrigated by a posterior anastomotic pedicle (Fig. 14.29) which arises from the rectal superior artery (RSA). This is not a minor issue, because RSA is a terminal branch of the inferior mesenteric artery, which arises above the aortic division. Consequently, placental detachment in these cases could result in a massive hemorrhage even if abdominal aorta or both common iliac arteries are occluded. Vascular control in these cases could be done by aortic balloon placed 4–5 below of the renal arteries or by rectus elastic clamping.
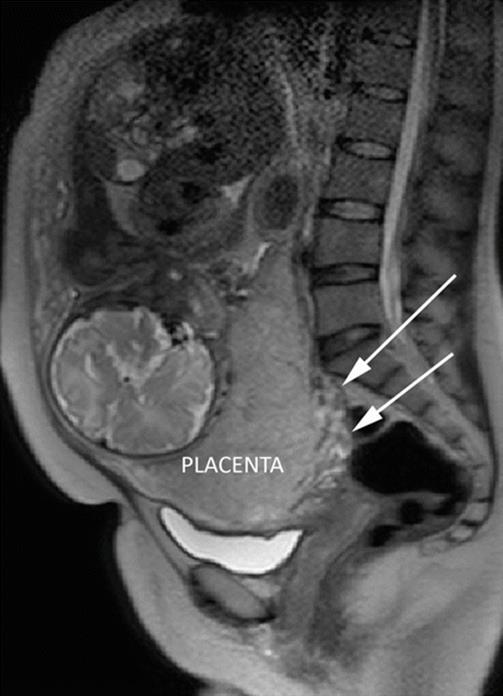
Fig. 14.27
Sagital T2 MRI. White arrows show a posterior AIP with prominent vessels. Placental MRI was requested due to doubt of anterior uterine wall (uterine segment); posterior invasion was a finding

Fig. 14.28
Sagital T2 MRI. Another slice of posterior invasion. The placenta is plenty of vessels in a supracervical area (white arrows). Patient with 2 CS, 2 miscarriages and 2 abortions
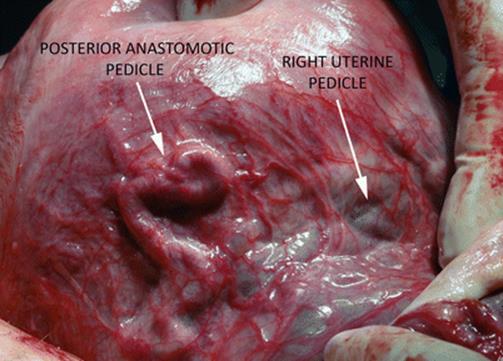
Fig. 14.29
Surgical view: Posterior view of the uterus, in the middle, a thick posterior anastomotic pedicle from rectal superior artery
14.11 Integrated Diagnosis
Due to the wide range of complications in AIP cases, the use of multiple methods of prenatal diagnosis could improve details and invasion features (Table 14.2). This information is closely related with the changes in the surgical approach [9] (Table 14.3). For this reason, in our opinion, the discussion must not focused in which study is better than others and, if not, in which information we need to perform a better prenatal evaluation (Table 14.4) to plan the surgery with a minimum risk, although the use of many of them increase a costs.
Table 14.2
Prenatal diagnosis features by method
|
Diagnosis |
Invasion deepness |
Invasion topography |
Bladder invasion |
Parametrial invasion |
CTVH |
|
|
pMRI |
High |
High |
High |
High |
High |
High |
|
Ultrasound |
High |
High/NC |
High/moderate |
High/moderate |
Poor |
ND |
|
Doppler |
High |
High |
Limited |
Moderate |
Poor |
ND |
|
3D Doppler |
High |
High/LE |
Limited |
High |
ND |
ND |
|
Cystoscopy |
Poor |
Poor |
Poor |
Moderate |
NP |
ND |
ND not described, CTVH cervicotrigonal vascular hyperplasia, NP not possible
Table 14.3
Induced changes by prenatal diagnosis
|
Diagnosis |
Bladder invasion |
Parametrial invasion |
CTVH |
S1 invasions |
S2 invasions |
|
Induced changes |
Ureteral catheterisation |
Ureteral catheterisation |
Ureteral catheterisation |
Possibility of resective-conservative technique |
Ureteral catheterisation |
|
Triple lumen foley catheter Methylene blue control |
Common iliac or aortic vascular control |
Common iliac or aortic vascular control |
Uterine or IIA vascular control |
Common iliac or aortic vascular control |
|
|
Eventual requirement of urologist or gynaecologist oncologist |
Deepness dissection. ureteral NFV haemostasis |
Subtotal hysterectomy |
High possibility for uterine resective conservation techniques |
Conservative technique if there is healthy myometrium below invasion |
NFV newly formed vessels, CTVH cervicotrigonal vascular hyperplasia
Table 14.4
Prenatal information before delivery for planning strategy
|
Diagnosis |
Invasion deepness |
Invasion topography |
Bladder invasion |
Parametrial invasion |
CTVH |
|
|
Hysterectomy |
Yes |
Yes |
Yes |
Yes |
Yes |
Yes |
|
One-step conservative |
Yes |
Yes |
Yes |
Yes |
Yes |
Yes |
|
Triple P |
Yes |
ND |
ND |
NN |
ND |
ND |
|
In situ placenta |
Yes |
No |
NN |
Yes |
ND |
ND |
ND not described, NN not necessary, CTVH cervicotrigonal vascular hyperplasia
14.12 Placental MRI: Future of Prenatal Study
It was proved by multiples studies that diagnostic differences between US and MRI are not significant [43] and they only position the pMRI indication only for non-conclusive cases [46] or when there is suspicion of posterior invasions. Maybe and it happened with US and Doppler, it is necessary to stimulate a close participation with obstetricians to improve knowledge of other AIP features. In other respects, pMRI may be necessary if other surgical conservative-repair techniques are incorporated globally. In this respect, there is a renewed interest to learn more about them, especially because a technique that leaves the placenta in situ has more complications than are usually reported. Although learning of resective-conservative techniques requires specific training and effort, skilled obstetricians can perform them with a guided assistance very quickly. Increasing rates of AIP worldwide may justify a comprehensive effort with all available possibilities.
References
1.
Abbas F, Talati J, Wasti S, Akram S, Ghaffar S, Qureshi R (2000) Placenta percreta with bladder invasion as a cause of life threatening hemorrhage. J Urol 164(4):1270–1274PubMed
2.
Guleria K, Gupta B, Agarwal S, Suneja A, Vaid N, Jain S (2013) Abnormally invasive placenta: changing trends in diagnosis and management. Acta Obstet Gynecol Scand 92(4):461–464PubMed
3.
Chantraine F, Langhoff-Roos J (2013) Abnormally invasive placenta–AIP. Awareness and pro-active management is necessary. Acta Obstet Gynecol Scand 92(4):369–371PubMed
4.
Comstock CH, Bronsteen RA (2014) The antenatal diagnosis of placenta accreta. BJOG 121(2):171–181PubMed
5.
D’Antonio F, Iacovella C, Bhide A (2013) Prenatal identification of invasive placentation using ultrasound: systematic review and meta-analysis. Ultrasound Obstet Gynecol 42(5):509–517PubMed
6.
Bowman ZS, Eller AG, Kennedy AM, Richards DS, Winter TC 3rd, Woodward PJ, Silver RM (2014) Interobserver variability of sonography for prediction of placenta accreta. J Ultrasound Med 33(12):2153–2158PubMed
7.
Kay HH, Spritzer CE (1991) Preliminary experience with magnetic resonance imaging in patients with third-trimester bleeding. Obstet Gynecol 78(3 Pt 1):424–429PubMed
8.
Palacios-Jaraquemada JM, Bruno C (2000) Gadolinium-enhanced MR imaging in the differential diagnosis of placenta accreta and placenta percreta. Radiology 216(2):610–611PubMed
9.
Palacios-Jaraquemada JM, Bruno CH (2005) Magnetic resonance imaging in 300 cases of placenta accreta: surgical correlation of new findings. Acta Obstet Gynecol Scand 84(8):716–724PubMed
10.
Algebally AM, Yousef RR, Badr SS, Al Obeidly A, Szmigielski W, Al Ibrahim AA (2014) The value of ultrasound and magnetic resonance imaging in diagnostics and prediction of morbidity in cases of placenta previa with abnormal placentation. Pol J Radiol 79:409–416PubMedCentralPubMed
11.
Palacios-Jaraquemada JM (2012) Placental adhesive disorders. DeGruyter Ed, Berlin
12.
Khong TY, Werger AC (2001) Myometrial fibers in the placental basal plate can confirm but do not necessarily indicate clinical placenta accreta. Am J Clin Pathol 116(5):703–708PubMed
13.
Palacios-Jaraquemada JM, Bruno CH, Martín E (2013) MRI in the diagnosis and surgical management of abnormal placentation. Acta Obstet Gynecol Scand 92(4):392–397PubMed
14.
Capechi E (1933) Placenta accreta abandonata in utero cesarizzato. Ritorno progressivo di questo allo stato normales enza alcuna complicanza (reasorbimiento autodigestione uterina della placenta?). Policlinic 40:347 [In Italian]
15.
Alamo L, Anaye A, Rey J, Denys A, Bongartz G, Terraz S, Artemisia S, Meuli R, Schmidt S (2013) Detection of suspected placental invasion by MRI: do the results depend on observer’ experience? Eur J Radiol 82(2):e51–e57PubMed
16.
Masselli G, Gualdi G (2013) MR imaging of the placenta: what a radiologist should know. Abdom Imaging 38(3):573–587PubMed
17.
Palacios-Jaraquemada JM, Pesaresi M, Nassif JC, Hermosid S (2004) Anterior placenta percreta: surgical approach, hemostasis and uterine repair. Acta Obstet Gynecol Scand 83(8):738–744PubMed
18.
Podrasky AE, Javitt MC, Glanc P, Dubinsky T, Harisinghani MG, Harris RD, Khati NJ, Mitchell DG, Pandharipande PV, Pannu HK, Shipp TD, Siegel CL, Simpson L, Wall DJ, Wong-You-Cheong JJ, Zelop CM (2013) ACR appropriateness Criteria® second and third trimester bleeding. Ultrasound Q 29(4):293–301PubMed
19.
Palacios-Jaraquemada JM, Bruno CH, Clavelli WA (2007) Morbid adherent placenta: prediction, diagnosis and management. Fetal Matern Med Rev 18(4):357–381
20.
D’Antonio F, Iacovella C, Palacios-Jaraquemada J, Bruno CH, Manzoli L, Bhide A (2014) Prenatal identification of invasive placentation using magnetic resonance imaging: systematic review and meta-analysis. Ultrasound Obstet Gynecol 44(1):8–16PubMed
21.
Levine D, Hulka CA, Ludmir J, Li W, Edelman RR (1997) Placenta accreta: evaluation with color Doppler US, power Doppler US, and MR imaging. Radiology 205(3):773–776PubMed
22.
Elhawary TM, Dabees NL, Youssef MA (2013) Diagnostic value of ultrasonography and magnetic resonance imaging in pregnant women at risk for placenta accreta. J Matern Fetal Neonatal Med 26(14):1443–1449PubMed
23.
Chiappa V, Di Legge A, Valentini AL, Gui B, Miccò M, Ludovisi M, Giansiracusa C, Testa AC, Valentin L (2014) Agreement of two-dimensional and three-dimensional transvaginal ultrasound with magnetic resonance imaging with regard to parametrial infiltration in cervical cancer. Ultrasound Obstet Gynecol. doi:10.1002/uog.14637
24.
Byun JM, Kim YN, Jeong DH, Kim KT, Sung MS, Lee KB (2013) Three-dimensional transvaginal ultrasonography for locally advanced cervical cancer. Int J Gynecol Cancer 23(8):1459–1464PubMed
25.
Seoud M, Cheaib S, Birjawi G, Tawil A, Jamali F (2010) Successful treatment of severe retro-peritoneal bleeding with recombinant factor VIIa in women with placenta percreta invading into the left broad ligament: unusual repeated ante-partum intra-abdominal bleeding. J Obstet Gynaecol Res 36(1):183–186PubMed
26.
Tam Tam KB, Dozier J, Martin JN Jr (2012) Approaches to reduce urinary tract injury during management of placenta accreta, increta, and percreta: a systematic review. J Matern Fetal Neonatal Med 25(4):329–334PubMed
27.
Wranning CA, Mölne J, El-Akouri RR, Kurlberg G, Brännström M (2005) Short-term ischaemic storage of human uterine myometrium–basic studies towards uterine transplantation. Hum Reprod 20(10):2736–2744PubMed
28.
Palacios-Jaraquemada JM, García Mónaco R, Barbosa NE, Ferle L, Iriarte H, Conesa HA (2007) Lower uterine blood supply: extrauterine anastomotic system and its application in surgical devascularization techniques. Acta Obstet Gynecol Scand 86(2):228–234PubMed
29.
Palacios-Jaraquemada JM, Karoshi M, Keith LG (2012) Uterovaginal blood supply: the S1 and S2 segmental concepts and their clinical relevance. In: A comprehensive textbook of postpartum hemorrhage: an essential clinical reference for effective management, 2nd edn. Sapiens Publishing Ltd, London, pp 19–23
30.
Palacios-Jaraquemada JM (2013) Caesarean section in cases of placenta praevia and accreta. Best Pract Res Clin Obstet Gynaecol 27(2):221–232, ReviewPubMed
31.
Al-Omari W, Elbiss HM, Hammad FT (2012) Placenta percreta invading the urinary bladder and parametrium. J Obstet Gynaecol 32(4):396–397PubMed
32.
Vahdat M, Mehdizadeh A, Sariri E, Chaichian S, Najmi Z, Kadivar M (2012) Placenta percreta invading broad ligament and parametrium in a woman with two previous cesarean sections: a case report. Case Rep Obstet Gynecol 2012:251381. doi:10.1155/2012/251381, Epub 2012 Oct 14PubMedCentralPubMed
33.
Borekci B, Ingec M, Kumtepe Y, Gundogdu C, Kadanali S (2008) Difficulty of the surgical management of a case with placenta percreta invading towards parametrium. J Obstet Gynaecol Res 34(3):402–404PubMed
34.
Lee LC, Lin HH, Wang CW, Cheng WF, Huang SC (1995) Successful conservative management of placenta percreta with rectal involvement in a primigravida. Acta Obstet Gynecol Scand 74(10):839–841PubMed
35.
Varghese B, Singh N, George RA, Gilvaz S (2013) Magnetic resonance imaging of placenta accreta. Indian J Radiol Imaging 23(4):379–385PubMedCentralPubMed
36.
Rezk MA, Shawky M (2014) Grey-scale and colour Doppler ultrasound versus magnetic resonance imaging for the prenatal diagnosis of placenta accreta. J Matern Fetal Neonatal Med 23:1–6
37.
Patenaude Y, Pugash D, Lim K, Morin L, Diagnostic Imaging Committee, Lim K, Bly S, Butt K, Cargill Y, Davies G, Denis N, Hazlitt G, Morin L, Naud K, Ouellet A, Salem S (2014) The use of magnetic resonance imaging in the obstetric patient. J Obstet Gynaecol Can 36(4):349–363PubMed
38.
Allen BC, Leyendecker JR (2013) Placental evaluation with magnetic resonance. Radiol Clin North Am 51(6):955–966PubMed
39.
Pasto ME, Kurtz AB, Rifkin MD, Cole-Beuglet C, Wapner RJ, Goldberg BB (1983) Ultrasonographic findings in placenta increta. J Ultrasound Med 2(4):155–159PubMed
40.
Koai E, Hadpawat A, Gebb J, Goffman D, Dar P, Rosner M (2014) Clinical outcomes and efficacy of antenatal diagnosis of placenta accreta using ultrasonography and magnetic resonance imaging. Obstet Gynecol 123(Suppl 1):61S. doi:10.1097/01.AOG.0000447363.70482.2
41.
Riteau AS, Tassin M, Chambon G, Le Vaillant C, de Laveaucoupet J, Quéré MP, Joubert M, Prevot S, Philippe HJ, Benachi A (2014) Accuracy of ultrasonography and magnetic resonance imaging in the diagnosis of placenta accreta. PLoS One 9(4):e94866. doi:10.1371/journal.pone.0094866, eCollection 2014PubMedCentralPubMed
42.
Abuhamad A (2013) Morbidly adherent placenta. Semin Perinatol 37(5):359–364PubMed
43.
Meng X, Xie L, Song W (2013) Comparing the diagnostic value of ultrasound and magnetic resonance imaging for placenta accreta: a systematic review and meta-analysis. Ultrasound Med Biol 39(11):1958–1965PubMed
44.
Palacios-Jaraquemada JM (2012) One-step conservative surgery for abnormal invasive placenta (placenta accreta–increta–percreta) section 5: placental abnormalities. In: A comprehensive textbook of postpartum hemorrhage: an essential clinical reference for effective management, 2nd edn. Sapiens Publishing Ltd, London, pp 263–274
45.
Primo LF, Arbogast K, Digiacomo T, Shepherd K, Gardner MO, Doyle NM (2014) Placenta accreta: can we forecast its arrival? Obstet Gynecol 123(Suppl 1):166S. doi:10.1097/01.AOG.0000447171.60522.8
46.
Maher MA, Abdelaziz A, Bazeed MF (2013) Diagnostic accuracy of ultrasound and MRI in the prenatal diagnosis of placenta accreta. Acta Obstet Gynecol Scand 92(9):1017–1022PubMed