Mona Akbari1 and Jacqueline L. Wolf1
(1)
Beth Israel Deaconess Medical Center, Department of Gastroenterology, Boston, MA, USA
Jacqueline L. Wolf
Email: jwolf1@bidmc.harvard.edu
Keywords
Gastroesophageal reflux (GERD)ConstipationIrritable bowel syndrome (IBS)Functional bowel disordersInflammatory bowel disease (IBD)Crohn’s diseaseUlcerative colitisLaxativesBiologic medicationsImmunomodulator therapy
Abbreviations
5-ASA
5-aminosalicylic acid
6-MP
6-mercaptopurine
AAP
American Academy of Pediatrics
ADA
Adalimumab
AGA
American Gastroenterology Association
anti-TNF
Antitumor necrosis factors
AZA
Azathioprine
CD
Crohn’s disease
CZP
Certolizumab pegol
ECCO
European Crohn’s and Colitis Organization
EGD
Esophagogastroduodenoscopy
FDA
Food and Drug Administration
FODMAP
Fermentable oligo-, di-, and monosaccharrides and polyols
GERD
Gastroesophageal reflux disease
GLB
Golimumab
IBD
Inflammatory bowel disease
IBS
Irritable bowel syndrome
IFX
Infliximab
IPAA
Ileal pouch/anal anastomosis
LES
Lower esophageal sphincter
NAT
Natalizumab
PPI
Proton pump inhibitor
RPC
Restorative proctocolectomy
TCA
Tricyclic antidepressants
UC
Ulcerative colitis
VDZ
Vedolizumab
Gastroesophageal Reflux
Case 1
P.A. is a 27-year-old healthy primigravida who is currently 24 weeks pregnant with a single fetus. She has a history of gastroesophageal reflux disease (GERD) diagnosed several years ago. At that time she took daily omeprazole for symptoms that occurred approximately 3–5 times per week, 1–2 times per day. Her BMI was 26 (ideal BMI 18.5–24.9), and weight loss was recommended. Over the course of a year, she managed to lose weight with diet and exercise. Her symptoms improved, and, ultimately, she was able to discontinue omeprazole without difficulty. She never underwent endoscopic evaluation, as her symptoms were uncomplicated and resolved with proton pump inhibitor (PPI) therapy. Her mother is overweight with hypertension and her maternal grandfather has diabetes. She has one younger sibling whose only medical issue consists of seasonal allergies. She has no family history of gastrointestinal malignancies. She is a nonsmoker and denies current alcohol consumption.
For the past 2 weeks, she has experienced symptoms of increasing regurgitation and an acid taste in her mouth that is reminiscent of her gastroesophageal reflux disease (GERD) symptoms years ago. These symptoms occur two to three times per week, typically after meals and occasionally at night. She has not taken any medications for her symptoms, as she is reluctant to take medications during her pregnancy. Her only medication at this time is a prenatal vitamin. Her prepregnancy BMI was 24. She has gained a total of 12 pounds. Because she had problems with similar symptoms when she was heavy and not pregnant, she wonders if her weight gain during this pregnancy has precipitated these symptoms. Her pregnancy thus far has been uncomplicated.
Discussion
GERD is a disease characterized by abnormal reflux of gastric contents into the esophagus. GERD symptoms are common during pregnancy. Approximately 45-80 % of women will experience GERD symptoms at some point in their pregnancies [1, 2]. In women with a diagnosis of GERD prior to pregnancy, symptoms can worsen as their pregnancy progresses. The clinical features of GERD are similar in pregnancy as in the nonpregnant general population. Symptoms include burning, acid taste, regurgitation, and provocation after meals and when lying supine. Symptoms occur more commonly in the pregnant woman than in the nonpregnant woman, and the prevalence of GERD appears to increase throughout the course of pregnancy [1–3].
The pathogenesis of GERD during pregnancy is likely multifactorial and related to a series of functional and structural changes. Possible factors that contribute to GERD symptoms during pregnancy include reduced lower esophageal sphincter (LES) pressure and impaired LES contractility in response to pharmacologic stimuli. Alterations in esophageal motility are another potential mechanism, whereas increased abdominal pressure from the enlarged gravid uterus is less likely to explain symptoms [4]. There is evidence to suggest that the functioning and responsiveness of the LES are altered, even early in pregnancy. While in the first trimester the basal LES pressure remains within normal limits, the LES is less responsive to hormonal, pharmacological, and physiological stimuli such as pentagastrin, methacholine, and protein meals [5]. Increased circulating levels of estrogen and progesterone and potentially loss of the intra-abdominal LES segment result in a decrease in basal LES pressure as pregnancy progresses [4, 6]. Esophageal motility and 24-h pH monitoring demonstrate that in the second and third trimesters, the LES pressure falls [7] and nadirs at 36 weeks [8]. The LES pressure appears to return to prepregnancy values by 4 weeks postpartum [7, 8].
The prevalence of GERD increases throughout pregnancy. Approximately 12.5–22 % of women in their first trimester report heartburn, and by the third trimester, up to 35.3–72 % of women may report heartburn [3, 9, 10]. Symptoms of acid taste in the mouth and regurgitation become more prevalent as pregnancy progresses. By the third trimester, approximately 50–80 % of women will report GERD symptoms [2, 3, 9, 11]. The severity of symptoms also progresses throughout pregnancy. GERD symptoms prior to pregnancy are a risk factor for pregnancy-related GERD [2, 10]. Other potential predictors of heartburn in pregnancy include multigravidity, high prepregnancy body mass index, and pregnancy weight gain [10]. Symptoms often abate after delivery; however, there is some limited evidence that pregnancy-related GERD may be a risk factor for developing frequent heartburn 1 year after delivery [2].
Case 1 Continued
She returns for a clinic visit and is now 30 weeks pregnant. She has tried avoiding fatty foods and eating late at night, factors that aggravated her GERD symptoms before her pregnancy. These modifications helped initially for her symptoms, but now she feels her symptoms are occurring more frequently. She has had appropriate weight gain during her pregnancy and continues to take only a prenatal vitamin daily. She denies tobacco and alcohol consumption. Her pregnancy remains uncomplicated. She has not tried any over-the-counter medications, but wonders what medications she can safely take during pregnancy.
Discussion
The risks and benefits of medical management should be weighed in pregnant women. Typically a step-up approach (Fig. 1) is adopted for the management of pregnancy-related GERD, and the first-line recommendation for all patients should include lifestyle modifications. Lifestyle modifications include eating smaller meals and avoiding late night eating. Though the data to support the avoidance of caffeine and/or spicy foods is limited, patients should avoid any foods that trigger symptoms. In a nonpregnant individual, mint can relax the LES and therefore should be avoided, even though there are no data during pregnancy if mint also triggers reflux. For patients with nighttime symptoms, elevating the head of the bed with a wedge or blocks can reduce symptoms.
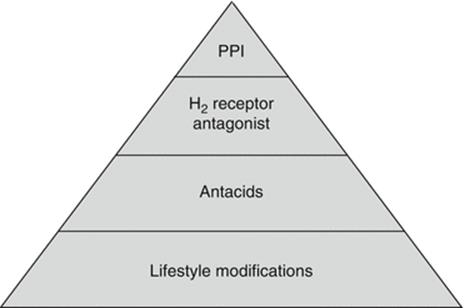
Fig. 1
Management of GERD during pregnancy, step-up approach. PPI proton pump inhibitor, H 2 -receptor antagonist histamine2-receptor antagonist
For symptoms not responding to lifestyle changes, the risks and benefits of drug therapy should be discussed (Tables 1 and 2). For the pregnant patient with mild to moderate GERD symptoms, initial therapy can begin with either an antacid or a histamine2 (H2)-receptor antagonist. Some expert opinion suggests that the initial medical therapy for pregnant women with mild GERD symptoms be calcium- and magnesium-based antacids [12].
Table 1
US Food and Drug Administration (FDA) categoriesa
|
FDA category |
Definition |
|
Category A |
Controlled studies show no risk to the fetus |
|
Category B |
Animal studies show no risk to the fetus; however, there are no adequate and well-controlled studies in pregnant women, or animal studies show risk but adequate and well-controlled human studies have failed to demonstrate risk |
|
Category C |
Animal studies show risk but there are no adequate and well-controlled studies in pregnant women or no adequate studies in animal or pregnant women |
|
Category D |
Fetal risk based on data from investigational, marketing experience, studies in human, but potential benefits may warrant use despite potential risks |
|
Category X |
Animal or human studies show fetal abnormalities; risks outweigh benefits |
aFDA ratings of drugs in pregnancy and lactation will be changed as of June 30, 2015
Table 2
Safety of medication use during pregnancy
|
Drug |
FDA classification |
Comments |
|
Antacids |
||
|
Calcium based |
None |
Low risk, preferred first line after lifestyle modification |
|
Magnesium based |
None |
Low risk, preferred first line after lifestyle modification. Has tocolytic properties; avoid in the last several weeks of pregnancy |
|
Aluminum based |
None |
Likely low risk in low doses, calcium- and magnesium-based antacids preferred |
|
Histamine2-receptor antagonist |
||
|
Cimetidine |
B |
Low risk |
|
Ranitidine |
B |
Low risk |
|
Famotidine |
B |
Limited safety data |
|
Nizatidine |
B |
Limited safety data |
|
Proton pump inhibitors |
||
|
Omeprazole |
C |
Animal study suggests fetal toxicity; epidemiologic studies in human suggest low risk |
|
Lansoprazole |
B |
Likely low risk based on epidemiologic studies. Avoid during 1–4 weeks prior to conception given possible risk of birth defects |
|
Pantoprazole |
B |
Likely low risk based on epidemiologic studies |
|
Esomeprazole |
B |
Likely low risk based on epidemiologic studies |
|
Sucralfate |
B |
Poorly absorbed, likely low risk |
|
Metoclopramide |
B |
Likely low risk, avoid long-term use given neurologic side effects |
Calcium-, magnesium-, and aluminum-based antacids are thought to be low risk in pregnancy [4, 13]. Low-dose (<1g/day) and high-dose (≥1 g/day) calcium supplementation during pregnancy appear to be associated with a reduced risk of hypertension and preeclampsia [14]. Oral magnesium supplementation has been shown to reduce the risk of high blood pressure compared to placebo, with conflicting results [15, 16]. Magnesium has tocolytic properties and its use is not advised during the last several weeks of pregnancy. Aluminum-containing antacids in large and chronic doses carry several potential concerns, such as constipation, malabsorption, skeletal impairment, and potential fetal neurotoxicity. For these reasons, high-dose aluminum antacids should be avoided and calcium-/magnesium-based antacids are preferred [4, 12]. Antacids containing sodium bicarbonate, which have the potential of causing metabolic alkalosis and fluid overload, should be avoided during pregnancy [4, 17].
H2 receptor antagonists inhibit gastric secretion stimulated by histamine and can be used in management of GERD symptoms concomitant with antacids. In prospective and retrospective studies, H2 receptor antagonist use during pregnancy was not associated with increased congenital malformations [18–20] or preterm delivery and low birth weight [19, 20]. For more severe symptoms or patients nonresponsive to therapy outlined above, a proton pump inhibitor (PPI) can be initiated. The US Food and Drug Administration (FDA) classifies all PPIs, except for omeprazole, as category B medications. Omeprazole, the first PPI approved on the market, at doses 5–56 times the human dose in animal studies resulted in embryo toxicity [21] and is therefore classified as category C.
In an earlier study, PPI use in the first trimester was associated with hypospadia [22]. However, this study was based on a small number of exposures (12 women exposed to lansoprazole, omeprazole, or esomeprazole). Moreover, there was no increased risk of hypospadia in a follow-up population-based study that included nearly 3,000 women exposed to PPI (including omeprazole) at conception or during pregnancy [23]. In a subgroup analysis restricted only to omeprazole use, the risk of hypospadia in the PPI exposed was similar to that in the unexposed group. In a meta-analysis of seven studies including data from 134,940 patients (1,530 exposed to PPIs), there was no difference in the risk of congenital malformations with first-trimester use of a PPI [24]. In many of these studies, the most common [25, 26], or often only [18, 27, 28], PPI exposure was omeprazole. In secondary analysis there was no difference in spontaneous abortions and preterm delivery. Analysis limited to omeprazole, alone, showed similar results. In two large epidemiologic studies published since this meta-analysis, exposure to PPIs in the first, second, and third trimester was not associated with congenital malformations [29, 30]. Similarly, a subgroup analysis of omeprazole use during pregnancy failed to show an increased risk of congenital malformations [29].
There is limited evidence from one epidemiologic study that PPI exposure at 1–4 weeks before conception was associated with birth heart and urinary tract defects [29]. Approximately 28 % of births exposed to PPI 1–4 weeks before conception were also exposed to other medications, such as corticosteroids, beta-blockers, and ACE inhibitors, 58 % were from mothers above the age of 30, and 23 % were from mothers reporting smoking during pregnancy. Subgroup analysis demonstrated that the risk of birth defects was significant only for lansoprazole, and not omeprazole, pantoprazole, or esomeprazole. Twenty-nine defects were reported in 541 births exposed to lansoprazole. In additional analysis looking at groups of birth defects, the odds of heart and urinary tract defects seemed to be associated with PPI exposure at 1–4 weeks before conception. The overall number of exposed cases was small with 30 cases of heart defects and 12 cases of urinary tract defects reported in 1,969 live births exposed to PPIs 1–4 weeks before conception. Further studies are needed to further explore this possible association. There are no guidelines on the use of PPI for patients contemplating pregnancy, although patients trying to become pregnant should be counseled on this possible risk [31]. If tolerated and not medically necessary, cessation of PPI therapy should be attempted in all women.
Other agents that can be considered for management of GERD symptoms include sucralfate and metoclopramide. Sucralfate, a surface agent and mucosal protectant, is poorly absorbed systemically with few side effects other than constipation and generally is regarded to be safe in pregnancy [32]. Metoclopramide, a prokinetic agent primarily used for treatment of delayed gastric emptying and nausea, may decrease GERD and has not been shown to be associated with increased adverse pregnancy outcomes [33, 34]. Metoclopramide could be considered for patients not responding to PPI therapy [31]. Its long-term use should be avoided given serious risks of neurologic complications, such as dystonia and akathisia.
Case 1 Continued
She is now 35 weeks pregnant. Her symptoms continued despite lifestyle modifications and use of calcium-based antacids and ranitidine 150 mg twice a day. She stopped these medications and it was suggested that she start a trial of pantoprazole 40 mg daily. Her symptoms have improved, although occasionally she will experience breakthrough reflux. She does admit to not taking pantoprazole daily, because she remains concerned about taking medications during her pregnancy. She denies signs or symptoms of burning or pain during swallowing, difficulty swallowing foods or food getting stuck, nausea, vomiting, and black or bloody stools. She has mild, infrequent generalized abdominal and back discomfort that developed during the course of her pregnancy. She continues to have appropriate weight gain during her pregnancy. Her labs have all been within normal limits. She wonders if she will require any further work-up now or after her pregnancy.
Discussion
The diagnosis of GERD can be made based on symptoms and more invasive testing during pregnancy is typically not necessary.
Barium radiographs, which expose the fetus to radiation, are not necessary for the diagnosis of GERD and therefore may be avoided during pregnancy. Esophageal manometry and pH studies are safe but are rarely necessary during pregnancy. For the general population, progressive symptoms or symptoms not responding to therapy can be evaluated with esophagogastroduodenoscopy (EGD). Signs and symptoms of dysphagia, odynophagia, and significant or continued gastrointestinal bleeding are considered to be indications for endoscopy during pregnancy by the American Society for Gastrointestinal Endoscopy (ASGE) [35]. Other experts have suggested that a moderate indication for EGD is recurrent nausea and emesis in patients past 16–18 weeks with a concern for peptic ulcer disease that have had inadequate response to PPI therapy [36]. Weak indications for EGD include self-limited nausea, emesis, or abdominal pain and GERD symptoms (aside from dysphagia) not responsive to empiric PPI therapy [36]. In this patient, who has no alarm features and symptoms and has been partially responsive to therapy, an EGD is not necessary. Symptoms should be monitored, daily PPI use recommended, and therapy increased with the addition of an H2 blocker at bedtime if needed.
The data on the safety and efficacy of EGD during pregnancy is sparse and limited to case series. The potential risks include maternal hypoxia, hypotension, and inferior vena cava compression during maternal positioning, as well as medication exposure to the fetus. The risks and benefits should be discussed in a multidisciplinary fashion. Maternal blood pressure and oxygen should be carefully monitored. As recommended by the American Society of Anesthesiologists and American College of Obstetrics and Gynecologists, when the fetus is previable, the fetal heart rate should be assessed before and after non-obstetric surgery, and when viable, at a minimum, fetal heart rate and contraction monitoring should be performed before and after procedure [37].
Judicious sedation is recommended. Propofol is category B by the FDA for use during pregnancy, while fentanyl and meperidine are category C and benzodiazepines are category D. The safety of propofol early in pregnancy is not known, but is considered relatively safe in pregnancy when given by a trained anesthesia provider. Meperidine and its active metabolites have been demonstrated to cross the placenta [38, 39]. Studies of teratogenicity from meperidine in animal models and humans are lacking. However, it is generally felt to be relatively safe for use during pregnancy. Fentanyl crosses the placenta as well [40], but is short acting and has faster procedural recovery time. Benzodiazepines are category D and diazepam has been associated with congenital abnormalities, including cleft lip and palate, with conflicting results [41–44]. Reports of infant floppy syndrome and neonatal withdrawal symptoms have been described in late third-trimester use of benzodiazepines [45]. If used, midazolam is the preferred benzodiazepines and should be given at its lowest effective dose.
In regard to the use of sedation during lactation, many sedatives are excreted in breast milk. Midazolam is excreted in breast milk and levels of midazolam and its metabolite appear to be undetectable after 4 h. Therefore, nursing should be withheld for at least 4 h after its exposure [35]. Fentanyl and propofol are excreted in breast milk, though at very low doses. Within 24 h of administration, 0.027 % of the propofol dose and 0.033 % of the fentanyl dose were detected in breast milk [46]. Fentanyl and propofol are considered to be compatible with breastfeeding [35]. Meperidine can be detected in breast milk up to 24 h after administration, and because of potential neurobehavioral effects on the infant, fentanyl is preferred when possible [35].
Functional Bowel Disorders
Case 2
S.T. is a 33-year-old gravida 2 para 1 woman who is currently 15 weeks pregnant. She has a history of asthma that is well controlled with rare use of albuterol inhaler as needed. During her first pregnancy, she reported constipation during her second and third trimesters. This was managed with increased fiber intake and occasional Metamucil. After her first pregnancy, her symptoms of hard stools generally resolved, though occasionally she found herself straining depending on her diet. Currently, she reports straining and hard stools, almost daily. She finds these symptoms to be more bothersome and frequent than what she experienced in her first pregnancy. She denies any vomiting, abdominal pain, or blood in her stools. She has been gaining weight appropriately. She has never had a colonoscopy. She has a paternal grandfather who had colon cancer in his seventies, but no other family members with colorectal cancer. She takes prenatal vitamins and occasionally Benadryl at night as a sleep aid. She feels that her symptoms interfere with her daily activity and would like to discuss this further.
Discussion
Functional bowel disorders such as irritable bowel syndrome (IBS) and functional constipation consist of gastrointestinal symptoms for which investigation does not reveal an organic cause. Alterations in the brain-gut axis as well as dysbiosis of the gut microbiome and its metabolic byproducts are thought to contribute to the underlying physiology of functional bowel disorders. Functional bowel symptoms can be common during pregnancy, with up to two thirds of women reporting one or more functional bowel symptoms during their first trimester of pregnancy. In a survey-based study of women in their first trimester, 46 % reported constipation, 49 % reported bloating, 44 % had irritable bowel syndrome, and 5 % reported diarrhea [47].
Constipation, often defined as hard stools, straining, incomplete evacuation, or infrequency of defecation, is a common gastrointestinal complaint during pregnancy. The prevalence of constipation during pregnancy has been reported with variable results. Approximately 45 % of women in their first trimester self-report symptoms of constipation [47, 48], and 51 % of patients self-report constipation at some point during their pregnancy [49]. Using a more narrow definition of constipation (fewer than three bowel movements per week and straining in more than 25 % of defecations), one study reported that only 5–9 % of women are affected by constipation throughout their pregnancy. The prevalence of constipation defined by the Rome criteria (Table 1), which includes symptoms of incomplete evacuation, hard stools, and use of manual maneuvers for defecation, during the first, second, and third trimester ranges from 24 to 30 %, 19 to 26 %, and 16 to 22 %, respectively [48, 49]. Similarly, in the postpartum period, the prevalence of constipation as defined by the Rome criteria is 24 % [48, 49]. Some studies suggest that the prevalence of constipation declines by the third trimester [49, 50] and in the postpartum period [50]. Sex hormones and decreased colonic transit time from elevated progesterone and reduced motilin levels may contribute to constipation. Other risk factors include decreased activity and vitamin supplementation (iron and calcium). Women with a history of treatment for constipation prior to pregnancy are more likely to report constipation during their pregnancy [49].
Despite its potentially high prevalence, bowel dysfunction and its recommended treatment are not always addressed during clinic visits, possibly reflective of underreporting or patient/provider perceptions that constipation symptoms are part of pregnancy. However, quality of life studies suggest that pregnant women reporting one or more functional bowel complaints have lower mean overall quality of life scores. Similarly, body image, health worry, activity interference, and food avoidance scores were lower for women who complained of functional bowel disorders during their pregnancy [47].
The most common symptoms of constipation during pregnancy are straining and hard stools. Occasionally women will report incomplete evacuation [49]. Anorectal obstruction and manual maneuvers to produce a stool appear to be the least commonly reported symptoms. Patients may report symptoms relating to constipation that do not conform to strict Rome criteria (Table 3). Often patients report constipation despite daily bowel movements, but further questioning will reveal symptoms such as straining, hard stools, unproductive urges, and incomplete evacuation. While the majority of pregnant women with constipation will have simple constipation without alarming underlying etiologies, history and physical examination are important in the evaluation of constipation. A number of medications, including prenatal multivitamins with iron, iron supplements, antihistamines, calcium channel blockers, and antidepressants, can be associated with constipation. A thorough review of medications, both prescription and over the counter, should be performed. Endoscopic evaluation is rarely necessary unless there are alarm signs such as gastrointestinal bleeding, uncontrolled diarrhea, new anemia not due to pregnancy, or possibly weight loss. In this patient, who has no alarm signs, endoscopic evaluation is not necessary. Limiting antihistamine use could be recommended as it can contribute to constipation.
Table 3
Rome diagnostic criteria
|
Rome II |
Rome IIIa |
|
|
IBS |
At least 12 weeks (need not be consecutive) in preceding 12 months of abdominal discomfort or pain with two or more of the following: 1. Improvement with defecation 2. Onset associated with change in stool frequency 3. Onset associated with change in stool form |
Recurrent abdominal pain or discomfort at least 3 days/month in the last 3 months associated with two or more of the following: 1. Improvement with defecation 2. Onset associated with change in stool frequency 2. Onset associated with change in stool form |
|
Functional constipation |
At least 12 weeks (need not be consecutive) in preceding 12 months of the following: 1. Straining > 1/4 of defecations 2. Lumpy or hard stools > 1/4 of defecations 3. Sensation of incomplete evacuation > 1/4 of defecations 4. Sensation of anorectal obstruction or blockage > 1/4 of defecations 5. Manual maneuvers to facilitate > 1/4 of defecations 6. <3 defecations per week Loose stools are not present and there are insufficient criteria for IBS |
I. Must include two or more of the following: 1. Straining in at least 25 % of defecations 2. Lumpy or hard stools in at least 25 % of defecations 3. Sensation of incomplete evacuation for at least 25 % of defecations 4. Sensation of anorectal obstruction or blockage for at least 25 % of defecations 5. Manual maneuvers to facilitate at least 25 % of defecations 6. Fewer than three defecations per week II. Loose stools are rarely present without the use of laxatives III. Insufficient criteria for IBS |
|
Functional diarrhea |
At least 12 weeks (need not be consecutive) in the preceding 12 months of: 1. Loose (mushy) or watery stools 2. Present > 3/4 of the time 3. No abdominal pain |
Loose (mushy) or watery stools without pain occurring in at least 75 % of stools |
IBS irritable bowel syndrome
aCriteria fulfilled for the last 3 months with symptom onset at least 6 months prior to diagnosis
Case 2 Continued
She returns for a follow-up visit 4 weeks later and states that she has tried increasing fiber intake daily, in the form of fiber-containing cereal and wheat bran, and has been taking Metamucil, which she took intermittently during her previous pregnancy. She has stopped taking Benadryl at night for a sleep aid. She experienced bloating, which she attributes to the increased fiber intake. She asks you if there are alternative management options.
Discussion
Initial management of constipation includes increase in dietary fiber, fluids, and exercise. Dietary fibers increase stool bulk and frequency and reduce transit time. The recommended daily dietary fiber dose is 20–35 g, though these doses are often not achieved. Soluble fibers, found in oat bran, barley, nuts, and seeds, attract water forming a gel and improve bowel symptoms in chronic constipation in the general population. Fiber supplement with corn-based biscuit and wheat bran has been shown to increase the number of bowel movements and soften stool consistency in pregnant women [51]. Light exercise should also be recommended, as it can promote regular bowel movements. If symptoms persist despite the above changes, a laxative can be considered (Tables 4 and 5).
Table 4
Types of laxatives and their mechanism of action
|
Treatment |
Examples |
Mechanism of action |
Side effects |
|
Bulk-forming fiber agents |
Psyllium (Metamucil) Methylcellulose (Citrucel) Polycarbophil (FiberCon) Wheat dextrin (Benefiber) Inulin |
Increases luminal water binding Increases fecal mass and stool bulk |
Bloating and gas |
|
Stool softeners |
Docusate |
Lower surface tension of stool Facilitates passage of water into stool |
Few side effects, cramping |
|
Osmotic laxatives |
|||
|
Saline agents |
Magnesium citrate Magnesium hydroxide |
Poorly absorbed osmotic preparations Secretion of water into the intestine |
Dehydration and electrolyte disturbance |
|
Poorly absorbed sugars |
Polyethylene glycol (Miralax) Lactulose |
Increase osmolar tension Secretion of water into the intestine |
Bloating and gas Dehydration and electrolyte disturbance |
|
Stimulant laxatives |
Bisacodyl (Dulcolax) Senna (Senokot) |
Increase intestinal motor activity |
Cramping and abdominal pain Dehydration and electrolyte disturbance |
Table 5
Safety of medication use during pregnancy
|
Drug |
FDA classification |
Comments |
|
Bulk-forming agents |
||
|
Psyllium |
None |
Low risk, preferred after trial of increase in dietary fiber, fluids, and exercise |
|
Methylcellulose |
None |
Low risk, preferred after trial of increase in dietary fiber, fluids, and exercise |
|
Polycarbophil |
None |
Low risk, preferred after trial of increase in dietary fiber, fluids, and exercise |
|
Docusate |
None |
Low risk for short-term use, limited efficacy in treatment of constipation |
|
Osmotic laxatives |
Low risk |
|
|
Magnesium citrate |
C |
Avoid long-term use (risk of hypermagnesemia, hyperphosphatemia, and dehydration) |
|
Lactulose |
B |
Low systemic absorption, likely low risk for short-term use |
|
Polyethylene glycol |
C |
Low systemic absorption, likely low risk for short-term use |
|
Stimulant laxatives |
||
|
Bisacodyl |
B |
Low systemic absorption, likely low risk for short-term use |
|
Senna |
C |
Low systemic absorption, likely low risk for short-term use |
|
Cascara |
C |
Safety in pregnancy not well known |
|
Aloe |
None |
Not recommended |
|
Castor oil |
X |
Not recommended (possible induction of labor) |
|
Mineral oil |
None |
Not recommended (may interfere with absorption of maternal nutrients and vitamins, possible neonatal coagulopathy and hemorrhage) |
|
Peppermint oil |
None |
Likely low risk |
|
Dicyclomine |
B |
Likely low risk |
|
Hyoscyamine |
C |
Crosses placenta, little known on effects on fetus |
|
TCA |
C (desipramine, amitriptyline) D (nortriptyline) |
Crosses placenta, possible association with adverse outcomes in neonate |
|
Rifaximin |
C |
Associated teratogenicity in some but not all animal studies, no adequate studies in pregnant women |
Bulk-forming fiber agents, including psyllium, methylcellulose, polycarbophil, wheat dextrin, flax seed, and guar, are not systemically absorbed and are low risk for use during pregnancy. However, their effects may take days to work, and unwanted side effects include gas, bloating, and cramping. Psyllium (e.g., Metamucil), which has a soluble/insoluble fiber ratio 70/30, increases fecal water content, but patients may complain of unwanted side effects such as gas and bloating secondary to its fermentation in the colon. Methylcellulose (e.g., Citrucel), which is 100 % soluble, is a synthetic polymer fiber that increases fecal mass, stimulates motility, and reduces colonic time. It is resistant to bacterial fermentation. Polycarbophil (e.g., FiberCon) is a hydrophilic resin that is not metabolized by intestinal bacteria. Methylcellulose, polycarbophil, and wheat dextrin (e.g., Benefiber) are less likely to cause gas and bloating.
Docusate sodium, a surfactant and stool softener, is generally well tolerated. Its efficacy and safety in pregnancy has not been established, though there have been no reports demonstrating increased risk in congenital malformations [52, 53]. Docusate is considered likely low risk [13], although there has been one report of neonatal hypomagnesemia from a mother who reported daily maternal docusate sodium use (100–200 mg or more daily) throughout pregnancy [54].
Overall, there is limited data on the use of laxatives during pregnancy. Osmotic laxatives, such as lactulose, polyethylene glycol, and magnesium-containing salts, increase the amount of fluid retained in the gut. Lactulose is a poorly absorbed sugar, classified as category B, but can cause bloating, gas, and pain in patients. Polyethylene glycol may result in less bloating and gas and is considered the first-choice osmotic laxative by the American Gastroenterology Association (AGA) during pregnancy [13]. Its systemic absorption is also low, but its safety during pregnancy has not been well established and is classified as category C. Prolonged use could theoretically lead to electrolyte disturbances. Saline laxatives, like magnesium containing agents, are likely low risk. However, in certain patients (e.g., renal dysfunction), excessive absorption of magnesium may lead to electrolyte and volume overload. Their long-term use during pregnancy is not recommended. Moreover, given its tocolytic properties, magnesium-containing agents should be avoided during the last several weeks of pregnancy.
Stimulant laxatives should be reserved for patients who do not respond to dietary measures, exercise, fiber bulking agents, or osmotic laxatives. Stimulant laxatives increase intestinal fluid secretion and stimulate colonic motility. There is limited data on the use of senna and bisacodyl during pregnancy, and both are classified as categories C and B, respectively. Senna glycosides are minimally absorbed by the intestine and excreted in bile. In rat models senna given during organogenesis was not associated with teratogenicity [55], and in case-control studies, maternal use of senna was not associated with congenital malformation [56]. There have been no animal reproductive studies reported on bisacodyl, though this medication has very little systemic absorption and is likely low risk for short-term use. It can be associated with more abdominal cramping when compared to senna. In general, when possible, the lowest dose and shortest duration of stimulant laxative ingestion needed to control symptoms are recommended during pregnancy.
Cascara, an extract from the dried, aged bark of Rhamnus purshiana, is an anthraquinone purgative with laxative properties and is available as an herbal supplement. While its use has not been well studied for during pregnancy, there is no evidence that drugs in this class pose risk to the fetus [53]. Cascara is generally well tolerated and with few side effects, though in high and chronic doses cascara has been linked to several cases of liver injury [57, 58]. Aloe, which has a laxative effect, is not recommended in pregnancy given the possible association with congenital malformations [59]. Castor oil, category X, has a potential association with induction of labor and is therefore not recommended for use during pregnancy. Mineral oil is also not recommended during pregnancy as it may interfere with absorption of important nutrients and vitamins in mothers, as well as its possible association with neonatal coagulopathy and hemorrhage [13].
Linaclotide and lubiprostone are two therapies approved for the management of chronic constipation in the general population. Linaclotide (trade name Linzess), approved by the FDA in 2012, is a guanylin peptide that acts as a selective agonist at the guanylate cyclase-C receptor of intestinal enterocytes. Linaclotide promotes small intestinal secretion of chloride and bicarbonate ions and small intestinal fluid secretion and increases intestinal transit time. In animal studies fetal toxicity occurred at doses toxic to the mother [60]. The safety of its use in pregnant women is unknown. Lubiprostone (trade name Amitiza), approved in 2008, is a prostaglandin analogue and acts locally as a chloride channel activator. At high doses, fetal toxicity was noted in animal studies, but human studies are lacking [61]. Both linaclotide and lubiprostone are classified as category C, and given their relative newness to the market, there are no societal guidelines or recommendations for use during pregnancy.
Case 3
M.L. is a 25-year-old female who recently discovered she is pregnant. She has never been pregnant. She has a history of irritable bowel syndrome (IBS) since college. Because of persistent abdominal pain, she was prescribed amitriptyline over 2 years ago. While taking the amitriptyline, she noted improvement in her symptom, but because of unpleasant side effects, she tapered off amitriptyline. Her symptoms remained manageable and she now takes peppermint oil occasionally for symptoms. For the past year, she noticed increased bloating, as well as generalized mild to moderate abdominal pain associated with belching and passing gas. She does not have any weight loss, blood in stool, worsening of abdominal pain, or changes in bowel habit. She had an unremarkable colonoscopy in her early twenties, and recent labs including work-up for celiac disease were normal. She started a probiotic with some improvement in her bloating. Dietary modifications were recommended, but she has not made any changes in her diet thus far. She is concerned about medication use and control of her symptoms during pregnancy.
Discussion
Traditional management of IBS in the nonpregnant patient focuses on improving individual symptoms, improving global symptoms, preventing unnecessary procedures, and reducing the impact of IBS on the overall quality of life. Smooth muscle relaxants (e.g., antispasmodics) and tricyclic antidepressants (TCAs), selective serotonin reuptake inhibitors, and anticonvulsants are potential medical therapies used in the management of abdominal pain and discomfort.
Dicyclomine and hyoscyamine are antispasmodics used for management of IBS and are categories B and C, respectively. In clinical trials dicyclomine improves pain, tenderness, bowel habits, and overall condition [62], and in a meta-analysis of 12 different antispasmodics, including dicyclomine, patients allocated to the antispasmodic group had improvement in global symptoms of IBS or abdominal pain compared to the placebo group (39 % vs. 56 %). Adverse events occurred in 14 % of patients [63] that most commonly included dry mouth, dizziness, and blurred vision. Dicyclomine hydrochloride, an anticholinergic agent, appears to improve tenderness and overall symptoms in patients with IBS but side effects have been noted in up to 69 % of patients at doses of 160 mg/day [62].
There is limited information of dicyclomine (category B) during pregnancy use. Animal studies have not demonstrated fetal harm, and epidemiologic studies in pregnant women taking dicyclomine up to 40 mg/day during the first trimester have not shown fetal abnormalities [64]. Dicyclomine is categorized as compatible with pregnancy [53]. Hyoscyamine (class C) crosses the placenta and little is known on its effects on the fetus.
TCAs have been used to treat functional bowel symptoms and likely work by modulating pain centrally and peripherally. Controlled studies in the nonpregnant population are limited by few numbers of patients and short follow-ups. A meta-analysis showed improvement in abdominal pain scores and clinical response when pooling several TCAs for the use of IBS patients. There are no studies for their use in the pregnant population. Desipramine and amitriptyline are category C, and nortriptyline is category D. TCAs cross the placenta, and their use has been associated with preterm birth and possible complications such as jitteriness, irritability, respiratory distress, and endocrine and metabolic disturbances in the newborn [65, 66]. Few studies have looked at the teratogenic effects on a population level. While Swedish and US studies demonstrated an increased risk of congenital abnormalities in neonates exposed to TCA use during pregnancy [66, 67], another UK study did not find a significant association with their use during the first trimester [68]. The AGA recommends avoidance of TCAs during pregnancy given the limited efficacy data for IBS [17], though risk and benefit should be discussed in women requiring this medication for management of their symptoms.
Peppermint oil in animal models has been shown to reduce calcium influx resulting in the relaxation of gastrointestinal smooth muscle [69]. This mechanism of action may be responsible for its use in the treatment of IBS and abdominal pain. A meta-analysis, pooling the results of four trials comparing peppermint oil to placebo, demonstrated that the use of peppermint oil (ranging from cumulative daily doses of 450 to 600 mg) improved abdominal pain and/or global symptoms. There are few studies looking at the use of herbal supplements during pregnancy and birth outcomes. Low birth weight and preterm delivery do not seem to be increased in women taking peppermint during pregnancy [70, 71], and in a study of the use of peppermint oil for treatment of pruritus in pregnant women, no side effects were noted in patients [72].
Bloating is a common complaint reported by patients with IBS, and these patients often report associated abdominal pain. Impaired gas transit and malabsorption of short-chain carbohydrates may contribute to the symptoms of abdominal bloating and distension. Dietary modifications, such as limitation of FODMAPs (fermentable oligo-, di-, and monosaccharides and polyols), and reduction of fiber intake may improve symptoms of bloating. FODMAP short-chain carbohydrates are poorly absorbed and fermented in the intestinal lumen, causing bloating and abdominal pain. A low FODMAP diet therefore restricts a large number of foods that contain lactose, gluten, as well as many artificial sugars, and some vegetables and fruits in which there is an excess of fructose over glucose. While the exact mechanism of efficacy is not clearly understood, probiotics appear to alter immune response, reducing inflammation, and alter the gut flora composition, resulting in improved IBS symptoms as well. Bifidobacterium infantis and Bifidobacterium bifidum have been shown to improve symptoms of abdominal pain and bloating [73, 74]. VSL#3 also appears to reduce colonic transit and improve flatulence in IBS patients and bloating in patients with diarrhea-predominant IBS [75, 76]. In a randomized controlled trial of pregnant women receiving probiotics at 2–4 weeks before expected delivery, there were no congenital malformations reported in the probiotic group. A meta-analysis failed to show difference in birth weight, gestational age, and Cesarean section for maternal probiotic use during pregnancy [77].
Antibiotics may also play a role in the treatment of IBS symptoms. There are potentially important differences in enteric flora of the IBS patient compared to the general population. Lactobacilli and bifidobacilli have been found in lower amounts in stool samples of patients with IBS [78, 79]. Reduction in these beneficial bacteria, which produce short-chain fatty acids that inhibit adherence of invasive bacteria, may favor colonization of the intestine with pathogenic bacteria. Increased hydrogen release during carbohydrate fermentation is associated with bloating and gaseous symptoms, and methane production has been associated with chronic constipation [80]. Rifaximin improves global symptoms of IBS, bloating, and abdominal pain [81–83]. There is limited oral absorption of rifaximin and the exposure to the fetus is expected to be low. Rifaximin is category C and has been associated with teratogenicity in some but not all animal studies [84, 85]. There are no adequate clinical studies in pregnant women.
Inflammatory Bowel Disease
Case 4
S.M. is a 32-year-old female with a history of Crohn’s disease (CD) diagnosed 8 years ago, who presents for preconception counseling. She was diagnosed at the age of 24, while in law school, when she developed abdominal pain and bloating. A colonoscopy and cross-sectional imaging showed ileitis and biopsies were consistent with Crohn’s disease. She was started on a thiopurine, azathioprine (AZA), and a prednisone taper at diagnosis. Four years after diagnosis, she had recurrence of her symptoms and persistent ileitis, necessitating a second prednisone course as an outpatient and dose increase of her AZA. With the increased dose in AZA, she was noted to have elevations in her liver function tests. AZA was ultimately stopped and prednisone was continued. After discussion with her gastroenterologist, she was started on infliximab (IFX) 5 mg/kg. She has been in clinical and endoscopic remission since then. Prior to starting IFX and while off of AZA, she had one miscarriage at gestation week 8. At the time of her miscarriage, she was in clinical remission and managed with oral prednisone therapy.
Now she is contemplating pregnancy and has been on a stable dose of 5 mg/kg of IFX every 8 weeks for the past 2 years. She has never had any extraintestinal manifestations of CD and has not had any abdominal surgeries. She is a former smoker, but quit after she was diagnosed with CD. Currently she is asymptomatic from the perspective of her Crohn’s disease. She is on daily iron supplements and monthly vitamin B12 injections. She recently stopped her oral contraceptive therapy. She has a cousin with ulcerative colitis (UC) who had a proctocolectomy in her twenties and had a difficult time conceiving, but was ultimately successful after in vitro fertilization. She asks you whether her CD affects her ability to conceive given her family and personal history as well as the course of CD during pregnancy.
Discussion
Inflammatory bowel disease (IBD), CD and UC, often affects women during their childbearing years. Many women with IBD express concern over conception and pregnancy. A multidisciplinary approach between the obstetrician, gastroenterologist, surgeon, and nutritionist is encouraged during preconception and pregnancy (Fig. 2). Women with quiescent disease have similar fertility rates as the general population [86, 87]. While overall fertility is likely to be normal, women with IBD report voluntary childlessness and sexual dysfunction, both of which affect fertility [88]. Active disease is thought to affect fertility and previous pelvic surgery, including restorative proctocolectomy (RPC) and ileal pouch/anal anastomosis (IPAA) which appear to impair conception and reduce fertility [89, 90]. Patients appear to have more spontaneous pregnancies and shorter time to conception and are less likely to undergo in vitro fertilization pre-RPC compared to post-RPC [89].

Fig. 2
Management of IBD in patients of reproductive age
Disease activity at conception is predictive of disease activity during pregnancy. Patients in remission at conception have a milder disease course during pregnancy and are less likely to relapse postpartum, compared to those with active disease [91–93]. In a study from 12 European countries, 81 % of pregnant women with CD in remission remained in remission by the end of pregnancy, while 19 % relapsed. For those who had inactive disease at conception, the risk of relapse during pregnancy and in the postpartum period was the same as the nonpregnant patient with CD, and the cumulative probability of remaining in remission during pregnancy was the same as that in nonpregnant controls. Disease duration was a risk factor for relapse during pregnancy and postpartum period. However, in this same study, UC pregnant women demonstrated a tendency to relapse during pregnancy. Sixty-five percent of women with UC in remission remained in remission, while 35 % relapsed. There was an increased risk of relapse during the first and second trimester compared to the nonpregnant UC women [93]. In a meta-analysis, 55 % of patients with active UC at the time of pregnancy remained active during pregnancy, while only 29 % of patients with quiescent UC relapsed during pregnancy. Similarly, 46 % of patients with active CD at conception remained active during pregnancy, and 23 % of patients who were in remission prior to pregnancy relapsed during pregnancy [92]. These findings highlight the goal of achieving and maintaining remission prior to conception.
Women with IBD appear to be at an increased risk of adverse pregnancy outcomes, with an increased risk of preterm birth and low birth weight compared to women in the general population [94–97]. In a large community-based study from Northern California, IBD patients compared to pregnant women without IBD were less likely to have live birth (60 % vs. 68 %) and more likely to have adverse conception outcome, such as spontaneous abortion (23 % vs. 17 %) and adverse pregnancy outcomes, such as preterm birth, small for gestational age and stillbirth (25 % vs. 19 %). Pregnant women with IBD were also more likely to have pregnancy complications, such as abruptio placenta, premature rupture of membrane and infection, when compared to controls (25 % vs. 16 %). There was no increased risk of congenital abnormalities [98]. Therefore, in clinical practice it is recommended to first achieve and maintain quiescent disease at conception and during pregnancy. The general consensus is to wait until patients are in remission prior to conception [88].
Obstetric need should determine the mode of delivery. The European Crohn’s and Colitis Organization (ECCO) recommends that active perianal disease or rectal involvement is indication for Cesarean delivery, while IPAA and ileorectal anastomosis are relative indications [99]. Vaginal delivery carries the concern for lacerations and the risk of recurrent Crohn’s in the rectovaginal septum and fistula formation. Patients with inactive perianal disease may consider vaginal delivery [88]. The sphincter and pelvic floor integrity is key for the maintenance of fecal continence in patients with IPAA and vaginal delivery may increase the risk of pudendal nerve damage and sphincter injury.
While UC patients with IPAA may report worsening incontinence and increases in stool frequency during pregnancy, these symptoms appear to resolve in the postpartum period. A study of 29 women with UC who delivered after IPAA procedure reported the mean incontinence score increased during pregnancy and subsequently improved postpartum. Thirty-five percent of patients had worsening of daytime continence during the third trimester of pregnancy; 21 % of patients experienced transient changes in continence (reported as seepage) that improved after delivery and 14 % experienced permanent changes (reported as seepage and soilage in one patient). Stool frequency increased in 55 % of patients toward the end of the second trimester and returned to normal in most patients within a few weeks of delivery (permanent increase in stool frequency was reported by 7 % of women) [100]. Another study of 43 UC patients with IPAA reported daytime and nighttime stool frequency increased during pregnancy 5.5–6.6 stools/day and 0.9–1.4 stools/night, respectively. Stool frequency resolved at longer follow-up (greater than 3 months) postpartum. Daytime and nighttime incontinence also worsened during pregnancy, but improved postpartum [100, 101].
Nevertheless, it is not clear if patients with IPAA necessarily benefit from Cesarean delivery. In one study of 82 patients with UC and IPAA, anal manometry and anal endosonography demonstrated lower mean squeeze pressure and greater sphincter defects, respectively, in those who had vaginal delivery compared to Cesarean delivery [102]. However, stool frequency after pregnancy compared to prepregnancy was not different for women undergoing Cesarean section compared to vaginal delivery in this study and in other studies [100–102]. The presence of colostomy or ileostomy is not considered an indication for Cesarean sections, unless indicated for other reasons.
Therefore, in this patient who is in remission and has not had previous abdominal surgeries, it is unlikely that her CD will affect her fertility. Her previous miscarriage appears to have been during a time when her disease was more active and may have been unrelated to her disease. Her disease course is likely to remain inactive as she is currently in remission on her current regimen.
Case 4 Continued
She continues to express concerns regarding medication use during pregnancy. She is concerned about the potential effects of IFX on the fetus during pregnancy as well as while breastfeeding. She asks if this medication should be stopped prior to conception or during pregnancy.
Discussion
Disease activity at conception and during pregnancy appears to be an important predictor of adverse pregnancy outcomes based on multiple studies. Disease activity has been associated with preterm births, low birth weight, fetal loss, and greater risk of adverse perinatal outcomes [103–107]. In clinical practice, it is recommended that patients achieve and maintain remission prior to conception. Patients are advised to continue medical therapy for IBD with the goal of preventing both relapses during pregnancy as well as improving pregnancy and conception outcomes. Therefore, it is recommended that this patient remain on IFX at conception and during pregnancy. It is important to have a thorough discussion regarding the risks of medication (Table 6) use during pregnancy and the risks of noncompliance through a multidisciplinary approach.
Table 6
Safety of medications used for the management of IBD during pregnancy and lactation
|
Drug |
FDA classification |
Pregnancy |
Lactation |
|
5-ASA |
B/C |
Low risk Mesalamine can be continued in both oral and topical forms Consider switching Asacol HD (category C due to DPH coating) |
Low risk |
|
Sulfasalazine |
B |
Low risk and can be continued in pregnancy Supplement with folic acid 2g/day |
Low risk |
|
Corticosteroids |
C or D |
Likely low risk, has been associated with cleft lip with or without cleft palate and decreased infant birth weight Prednisone and prednisolone oral solution, immediate release prednisone, and methylprednisolone (category C) Prednisone oral delayed-release tablet (D) |
Low risk, consider 4-h delay before breastfeeding |
|
Budesonide |
C |
Limited information on use during pregnancy, likely low risk |
Safety in lactation not known |
|
AZA/6-MP |
D |
Low risk; recent studies suggest no increase in adverse pregnancy outcomes. Recommend continuing during pregnancy if necessary to maintain remission |
Low risk, consider 4-h delay before breastfeeding |
|
Anti-TNF agents |
B |
Low risk based on limited data. IFX and ADA cross the placenta, while CTP does not No live vaccinations in infants for at least the first 7 months |
Low risk |
|
Natalizumab |
C |
Limited data, safety not known |
Safety in lactation not known |
|
Vedolizumab |
B |
Limited data, safety not known |
Safety in lactation not known |
|
Methotrexate |
X |
Contraindicated |
Contraindicated |
Aminosalicylates are compounds containing 5-aminosalicylic acid (5-ASA). 5-ASAs are category B, except for olsalazine and Asacol HD, which are category C. These medications are considered to be low risk for use during pregnancy by the AGA and ECCO [17, 99]. Mesalamine crosses the placenta and has not demonstrated fetal toxicity in animal studies [108]. There is one report of increased stillbirth and preterm birth for mothers prescribed 5-ASA drugs, but there was no accounting for disease activity [109]. However, in other population-based studies as well as a meta-analysis of over 2,000 pregnant women, no significant association of adverse outcomes such as congenital abnormalities, stillbirth, spontaneous abortion, preterm delivery, and low birth weight occurred [110–112]. The reason that Asacol HD is category C is due to the coating of the tablet. Dibutyl phthalate, an ingredient in Asacol’s enteric coating, in animal studies has been associated with skeletal malformations and adverse effects on the male reproductive system. A switch from Asacol to another 5-ASA medication should be considered during pregnancy. Sulfasalazine, composed of a 5-ASA joined to sulfapyridine, is category B and is also considered low risk for use during pregnancy. Sulfapyridine crosses the placenta. A report of hemolytic anemia in infants born to a mother taking sulfasalazine has been noted [113]. However, in an epidemiologic study and in smaller case-control studies of sulfasalazine use during pregnancy, sulfasalazine was not associated with pregnancy complications or congenital abnormalities [114–116]. Sulfapyridine can inhibit folate absorption; therefore, it is recommended that women supplement with folate 2 mg/day. Sulfapyridine and low levels of mesalamine can be detected in breast milk [117, 118]. 5-ASA and sulfasalazine are considered low risk for use in breastfeeding mothers by the AGA and ECCO, though the American Academy of Pediatrics (AAP) reports an incident of bloody diarrhea in an infant exposed to sulfasalazine via breast milk [17, 99, 119, 120].
Corticosteroids cross the placenta, and short-acting steroids such as prednisone, prednisolone, and methylprednisolone reach lower concentration in the fetus than longer-acting dexamethasone. Oral clefts have been reported in newborns exposed to prednisone use during the first trimester in animal studies and in a meta-analysis [121]. Recent population-based studies have failed to demonstrate an association with prednisone use and orofacial malformations and major congenital abnormalities [122–124]. Adrenal suppression in the newborn has been reported after corticosteroid use in pregnancy [125, 126]. There is sparse information on the use of budesonide during pregnancy. Budesonide undergoes extensive first-pass metabolism in the liver and has low systemic bioavailability. In one case series, there were no adverse fetal or maternal effects related to budesonide use during pregnancy [127]. Risks and benefits to the mother and fetus should be discussed when initiating corticosteroids. Corticosteroids are felt to be overall low risk when indicated during pregnancy for management of the IBD patient [17, 99]. Corticosteroids can be excreted in breast milk, with milk/serum concentration ratio of about 0.2 at prednisolone doses ≥ 30 mg and 0.1 at lower prednisolone doses. Peak concentrations occur about 1 h after the dose is given [128]. A 4-h delay before breastfeeding can be considered [99].
Thiopurines, 6-mercaptopurine (6-MP) and AZA, are categorized as category D, based on early experience and case reports. The placenta is a relative barrier to the metabolites of 6-MP and AZA, and metabolites are detected at lower levels in the newborn than that detected in mothers. Multiple epidemiologic studies in the IBD population have not demonstrated an increased risk in small for gestational age and congenital abnormalities in newborns exposed to thiopurines at conception or during pregnancy, while studies have shown mixed results in regard to the risk of low birth weight and preterm birth [129–136]. It is recommended that thiopurines be continued during pregnancy in patients requiring this medication to sustain remission. However, it is generally recommended that thiopurines not be started for the first time in pregnancy, given their lack of efficacy for induction therapy and the small risk of pancreatitis and bone marrow suppression [137]. In regard to lactation, the levels of metabolites in breast milk appear to be very low, and concentrations of 6-MP in breast milk peak within the first 4 h of drug intake [138–140]. Although the evidence is sparse, there does not appear to be an increased risk in infectious complications in infants exposed to thiopurines via breast milk [141]. Long-term data are not available. A 4-h delay before breastfeeding can be considered to minimize an infant’s exposure to thiopurines.
Infliximab (IFX), adalimumab (ADA), certolizumab pegol (CZP), and golimumab (GLB) are antitumor necrosis factors (anti-TNFs) used for the management of IBD. IFX, ADA, and GLB are IgG1 antibodies that are transported across the placenta during the second and third trimester. IFX levels in the newborn cord blood appear to be higher than maternal levels and have been detected in infants born to mothers taking IFX up to gestational week 31 [142, 143]. In a small case series, no short-term infectious complications were seen in infants exposed to IFX in utero [142]. Based on results from a safety registry and epidemiologic studies, IFX use does not appear to be associated with neonatal complications such as low birth weight, spontaneous abortions, and congenital abnormalities [107, 144–147]. Preliminary data from the PIANO study, a prospective registry of pregnancy outcomes in IBD women, demonstrated an increased risk of infections in infants born to mothers on combination therapy of an anti-TNF and immunomodulator at 12 months of age [148]. Data on long-term effects of IFX exposure in utero are not available. ADA like IFX can be detected in the newborn cord blood [143], and based on limited safety data in pregnancy, ADA does not appear to be associated with adverse pregnancy outcomes [146]. CZP is a pegulated Fab fragment that passively diffuses across the placenta. The levels of CZP in the newborn are detected at minimal levels, though clinical data of its use during pregnancy is scarce. In one series, there were no reported congenital abnormalities or infectious complications in 12 newborns exposed to CZP in utero [149]. There are no available data on the use of GLM, which was approved for the treatment of UC by the FDA in 2013, in the pregnant IBD patient.
There are differing opinions about whether anti-TNF therapy should be held late in pregnancy [150–152]. In patients with active disease, it is generally recommended to continue anti-TNFs throughout pregnancy. Some suggest discontinuing anti-TNF therapy in the early third trimester, to minimize infant exposure [153]. However, other providers advocate continuing anti-TNF throughout pregnancy as it appears that levels are present in newborns even when anti-TNFs are stopped prior to gestation week 30 and to minimize the risk of disease relapse in mothers. In one study, 12 patients with IBD in remission on IFX who discontinued IFX at gestational weeks 18–27 did not experience relapse in pregnancy [154]. Since levels of anti-TNF are detected in infants born to mothers on these medications, live vaccines should be avoided for at least the first 7 months However, there is recent data from a prospective multicenter study that anti-TNF levels can be detected in infants up to 12 months. Therefore, delaying live vaccines up to 1 year unless anti-TNF clearance has been documented should be considered. (Reference: Julsgarrd M, Christensen LA, Gibson PR, Gearry RB, Fallingborg J, Hvas CL, Bibby BM, Uldbjerg N, Connell WR, Rosella O, Grosen A, Brown SJ, Kjeldsen J, Wildt S, Svenningsen L, Sparrow MP, Walsh A, Conner SJ, Radford-Smith G, Lawrance IC, Andrews JM, Ellard K, Bell SJ. Concentrations of adalimumab and infliximab in mothers and newborns and effects on infection. Gastroenterology. 2016; 151(1): 110-9.. In regard to lactation, there are data from small studies on the compatibility of anti-TNF therapy and lactation. The data suggest that little to no IFX is detected in breast milk and is likely compatible with breastfeeding [153, 155, 156].
Monoclonal antibodies to integrin such as natalizumab (NAT) and vedolizumab (VDZ) can be used in patients with IBD. NAT is a monoclonal antibody against the alpha-4 subunit integrin. It is approved for the treatment of Crohn’s disease and is category C. There are scant data on the use of NAT during pregnancy. In animal studies on guinea pigs treated with NAT, attributable fetal toxicity and teratogenic effects were not observed [157]; however, reduction in pregnancy rates in female guinea pigs treated with high-dose NAT was observed [158]. In a study looking at 35 women with multiple sclerosis, NAT did not show adverse pregnancy outcomes compared to women with the disease not on disease-modifying therapy [159]. VDZ is a gut-selective monoclonal antibody to alpha-4 beta-7 integrin and is approved for treatment of CD and UC. VDZ is category B. Animal studies of VDZ in rabbits and monkeys at 20 times the dose levels recommended in human did not demonstrate fetal harm. There are no studies on the use of VDZ in pregnant women. The review of VDZ clinical development program revealed that 24 VDZ-treated IBD females became pregnant during the clinical study. Two of a total of 11 live births were premature, and there was one report of congenital defect (agenesis of the corpus callosum) in a mother with a history of spontaneous abortions and ectopic pregnancy who received one dose of VDZ [160]. Methotrexate is contraindicated in pregnancy due to teratogenic effects. It is recommended that MTX be stopped in prospective mothers 3–6 months before trying to conceive and high-dose folic acid be initiated [99].
References
1.
Ramu B, Mohan P, Rajasekaran MS, Jayanthi V. Prevalence and risk factors for gastroesophageal reflux in pregnancy. Indian J Gastroenterol. 2011;30(3):144–7.PubMed
2.
Rey E, Rodriguez-Artalejo F, Herraiz MA, Sanchez P, Alvarez-Sanchez A, Escudero M, et al. Gastroesophageal reflux symptoms during and after pregnancy: a longitudinal study. Am J Gastroenterol. 2007;102(11):2395–400.PubMed
3.
Malfertheiner SF, Malfertheiner MV, Kropf S, Costa SD, Malfertheiner P. A prospective longitudinal cohort study: evolution of GERD symptoms during the course of pregnancy. BMC Gastroenterol. 2012;12:131.PubMedPubMedCentral
4.
Madanick RDK, Philip O. GERD and pregnancy. Pract Gastroenterol. 2006;XXX(6):30–9.
5.
Fisher RS, Roberts GS, Grabowski CJ, Cohen S. Altered lower esophageal sphincter function during early pregnancy. Gastroenterology. 1978;74(6):1233–7.PubMed
6.
Richter JE. Review article: the management of heartburn in pregnancy. Aliment Pharmacol Ther. 2005;22(9):749–57.PubMed
7.
Al-Amri SM. Twenty-four hour pH monitoring during pregnancy and at postpartum: a preliminary study. Eur J Obstet Gynecol Reprod Biol. 2002;102(2):127–30.PubMed
8.
Van Thiel DH, Gavaler JS, Joshi SN, Sara RK, Stremple J. Heartburn of pregnancy. Gastroenterology. 1977;72(4 Pt 1):666–8.PubMed
9.
Marrero JM, Goggin PM, de Caestecker JS, Pearce JM, Maxwell JD. Determinants of pregnancy heartburn. Br J Obstet Gynaecol. 1992;99(9):731–4.PubMed
10.
Naumann CR, Zelig C, Napolitano PG, Ko CW. Nausea, vomiting, and heartburn in pregnancy: a prospective look at risk, treatment, and outcome. J Matern Fetal Neonatal Med. 2012;25(8):1488–93.PubMed
11.
Bassey OO. Pregnancy heartburn in Nigerians and Caucasians with theories about aetiology based on manometric recordings from the oesophagus and stomach. Br J Obstet Gynaecol. 1977;84(6):439–43.PubMed
12.
Tytgat GN, Heading RC, Muller-Lissner S, Kamm MA, Scholmerich J, Berstad A, et al. Contemporary understanding and management of reflux and constipation in the general population and pregnancy: a consensus meeting. Aliment Pharmacol Ther. 2003;18(3):291–301.PubMed
13.
Mahadevan U. Gastrointestinal medications in pregnancy. Best Pract Res Clin Gastroenterol. 2007;21(5):849–77.PubMed
14.
Hofmeyr GJ, Lawrie TA, Atallah AN, Duley L, Torloni MR. Calcium supplementation during pregnancy for preventing hypertensive disorders and related problems. Cochrane Database Syst Rev. 2014;6:CD001059.
15.
Bullarbo M, Odman N, Nestler A, Nielsen T, Kolisek M, Vormann J, et al. Magnesium supplementation to prevent high blood pressure in pregnancy: a randomised placebo control trial. Arch Gynecol Obstet. 2013;288(6):1269–74.PubMed
16.
Rudnicki M, Frolich A, Rasmussen WF, McNair P. The effect of magnesium on maternal blood pressure in pregnancy-induced hypertension. A randomized double-blind placebo-controlled trial. Acta Obstet Gynecol Scand. 1991;70(6):445–50.PubMed
17.
Mahadevan U, Kane S. American gastroenterological association institute medical position statement on the use of gastrointestinal medications in pregnancy. Gastroenterology. 2006;131(1):278–82.PubMed
18.
Ruigomez A, Garcia Rodriguez LA, Cattaruzzi C, Troncon MG, Agostinis L, Wallander MA, et al. Use of cimetidine, omeprazole, and ranitidine in pregnant women and pregnancy outcomes. Am J Epidemiol. 1999;150(5):476–81.PubMed
19.
Magee LA, Inocencion G, Kamboj L, Rosetti F, Koren G. Safety of first trimester exposure to histamine H2 blockers. A prospective cohort study. Dig Dis Sci. 1996;41(6):1145–9.PubMed
20.
Matok I, Gorodischer R, Koren G, Sheiner E, Wiznitzer A, Uziel E, et al. The safety of H(2)-blockers use during pregnancy. J Clin Pharmacol. 2010;50(1):81–7.PubMed
21.
Prilosec (Omeprazole). Whitehouse Station: Merc & Co., Inc.; 2006.
22.
Anderka M, Mitchell AA, Louik C, Werler MM, Hernandez-Diaz S, Rasmussen SA. Medications used to treat nausea and vomiting of pregnancy and the risk of selected birth defects. Birth Defects Res A Clin Mol Teratol. 2012;94(1):22–30.PubMed
23.
Erichsen R, Mikkelsen E, Pedersen L, Sorensen HT. Maternal use of proton pump inhibitors during early pregnancy and the prevalence of hypospadias in male offspring. Am J Ther. 2014;21(4):254–9.PubMed
24.
Gill SK, O’Brien L, Einarson TR, Koren G. The safety of proton pump inhibitors (PPIs) in pregnancy: a meta-analysis. Am J Gastroenterol. 2009;104(6):1541–5; quiz 0, 6.PubMed
25.
Diav-Citrin O, Arnon J, Shechtman S, Schaefer C, van Tonningen MR, Clementi M, et al. The safety of proton pump inhibitors in pregnancy: a multicentre prospective controlled study. Aliment Pharmacol Ther. 2005;21(3):269–75.PubMed
26.
Nielsen GL, Sorensen HT, Thulstrup AM, Tage-Jensen U, Olesen C, Ekbom A. The safety of proton pump inhibitors in pregnancy. Aliment Pharmacol Ther. 1999;13(8):1085–9.PubMed
27.
Kallen B. Delivery outcome after the use of acid-suppressing drugs in early pregnancy with special reference to omeprazole. Br J Obstet Gynaecol. 1998;105(8):877–81.PubMed
28.
Lalkin A, Loebstein R, Addis A, Ramezani-Namin F, Mastroiacovo P, Mazzone T, et al. The safety of omeprazole during pregnancy: a multicenter prospective controlled study. Am J Obstet Gynecol. 1998;179(3 Pt 1):727–30.PubMed
29.
Pasternak B, Hviid A. Use of proton-pump inhibitors in early pregnancy and the risk of birth defects. N Engl J Med. 2010;363(22):2114–23.PubMed
30.
Matok I, Levy A, Wiznitzer A, Uziel E, Koren G, Gorodischer R. The safety of fetal exposure to proton-pump inhibitors during pregnancy. Dig Dis Sci. 2012;57(3):699–705.PubMed
31.
Gerson LB. Treatment of gastroesophageal reflux disease during pregnancy. Gastroenterol Hepatol. 2012;8(11):763–4.
32.
Ali RA, Egan LJ. Gastroesophageal reflux disease in pregnancy. Best Pract Res Clin Gastroenterol. 2007;21(5):793–806.PubMed
33.
Matok I, Gorodischer R, Koren G, Sheiner E, Wiznitzer A, Levy A. The safety of metoclopramide use in the first trimester of pregnancy. N Engl J Med. 2009;360(24):2528–35.PubMed
34.
Pasternak B, Svanstrom H, Molgaard-Nielsen D, Melbye M, Hviid A. Metoclopramide in pregnancy and risk of major congenital malformations and fetal death. JAMA. 2013;310(15):1601–11.PubMed
35.
Shergill AK, Ben-Menachem T, Chandrasekhara V, Chathadi K, Decker GA, Evans JA, et al. Guidelines for endoscopy in pregnant and lactating women. Gastrointest Endosc. 2012;76(1):18–24.PubMed
36.
Savas N. Gastrointestinal endoscopy in pregnancy. World J Gastroenterol. 2014;20(41):15241–52.PubMedPubMedCentral
37.
American Society of Anesthesiologists (ASA) and the American College of Obstetricians and Gynecologists (ACOG) Statement on non obstetric surgery during pregnancy. 2009. Report No.
38.
Kuhnert BR, Kuhnert PM, Philipson EH, Syracuse CD. Disposition of meperidine and normeperidine following multiple doses during labor. II. Fetus and neonate. Am J Obstet Gynecol. 1985;151(3):410–5.PubMed
39.
Morrison JC, Martin JN, Christie RJ, Martin RW, Hess LW, Wiser WL, et al. Meperidine and normeperidine distribution in the rhesus monkey. J Perinatol. 1988;8(1):24–6.PubMed
40.
Cooper J, Jauniaux E, Gulbis B, Quick D, Bromley L. Placental transfer of fentanyl in early human pregnancy and its detection in fetal brain. Br J Anaesth. 1999;82(6):929–31.PubMed
41.
Dolovich LR, Addis A, Vaillancourt JM, Power JD, Koren G, Einarson TR. Benzodiazepine use in pregnancy and major malformations or oral cleft: meta-analysis of cohort and case–control studies. BMJ. 1998;317(7162):839–43.PubMedPubMedCentral
42.
Rosenberg L, Mitchell AA, Parsells JL, Pashayan H, Louik C, Shapiro S. Lack of relation of oral clefts to diazepam use during pregnancy. N Engl J Med. 1983;309(21):1282–5.PubMed
43.
Safra MJ, Oakley Jr GP. Association between cleft lip with or without cleft palate and prenatal exposure to diazepam. Lancet. 1975;2(7933):478–80.PubMed
44.
Bellantuono C, Tofani S, Di Sciascio G, Santone G. Benzodiazepine exposure in pregnancy and risk of major malformations: a critical overview. Gen Hosp Psychiatry. 2013;35(1):3–8.PubMed
45.
McElhatton PR. The effects of benzodiazepine use during pregnancy and lactation. Reprod Toxicol. 1994;8(6):461–75.PubMed
46.
Nitsun M, Szokol JW, Saleh HJ, Murphy GS, Vender JS, Luong L, et al. Pharmacokinetics of midazolam, propofol, and fentanyl transfer to human breast milk. Clin Pharmacol Ther. 2006;79(6):549–57.PubMed
47.
Johnson P, Mount K, Graziano S. Functional bowel disorders in pregnancy: effect on quality of life, evaluation and management. Acta Obstet Gynecol Scand. 2014;93(9):874–9.PubMed
48.
Ponce J, Martinez B, Fernandez A, Ponce M, Bastida G, Pla E, et al. Constipation during pregnancy: a longitudinal survey based on self-reported symptoms and the Rome II criteria. Eur J Gastroenterol Hepatol. 2008;20(1):56–61.PubMed
49.
Bradley CS, Kennedy CM, Turcea AM, Rao SS, Nygaard IE. Constipation in pregnancy: prevalence, symptoms, and risk factors. Obstet Gynecol. 2007;110(6):1351–7.PubMed
50.
Derbyshire EJ, Davies J, Detmar P. Changes in bowel function: pregnancy and the puerperium. Dig Dis Sci. 2007;52(2):324–8.PubMed
51.
Anderson AS, Whichelow MJ. Constipation during pregnancy: dietary fibre intake and the effect of fibre supplementation. Hum Nutr Appl Nutr. 1985;39(3):202–7.PubMed
52.
Jick H, Holmes LB, Hunter JR, Madsen S, Stergachis A. First-trimester drug use and congenital disorders. JAMA. 1981;246(4):343–6.PubMed
53.
Briggs GG, Freeman R, Yaffe S. Drugs in pregnancy and lactation: a reference guide to fetal and neonatal risk. 9th ed. Philadelphia/Baltimore/New York/London: Lippincott Williams & Wilkins; 2011.
54.
Schindler AM. Isolated neonatal hypomagnesaemia associated with maternal overuse of stool softener. Lancet. 1984;2(8406):822.PubMed
55.
Mengs U. Reproductive toxicological investigations with sennosides. Arzneimittelforschung. 1986;36(9):1355–8.PubMed
56.
Acs N, Banhidy F, Puho EH, Czeizel AE. No association between severe constipation with related drug treatment in pregnant women and congenital abnormalities in their offspring: a population-based case–control study. Congenit Anom. 2010;50(1):15–20.
57.
Cascara (Cascara Sagrada). Bethesda: U.S. National Library of Medicine, National Institutes of Health; 2015 [cited 2015 April 8, 2015].
58.
Garcia-Cortes M, Borraz Y, Lucena MI, Pelaez G, Salmeron J, Diago M, et al. Liver injury induced by “natural remedies”: an analysis of cases submitted to the Spanish Liver Toxicity Registry. Rev Esp Enferm Dig. 2008;100(11):688–95.PubMed
59.
Prather CM. Pregnancy-related constipation. Curr Gastroenterol Rep. 2004;6(5):402–4.PubMed
60.
Thomas RH, Allmond K. Linaclotide (linzess) for irritable bowel syndrome with constipation and for chronic idiopathic constipation. P T. 2013;38(3):154–60.PubMedPubMedCentral
61.
Crawford DF, Perentesis GP, Engelke KJ. Effects of lubiprostone, a novel GI chloride channel activator, on reproductive and developmental toxicity endpoints in rats. Am J Gastroenterol. 2005;100:S331.
62.
Page JG, Dirnberger GM. Treatment of the irritable bowel syndrome with Bentyl (dicyclomine hydrochloride). J Clin Gastroenterol. 1981;3(2):153–6.PubMed
63.
Ford AC, Talley NJ, Spiegel BM, Foxx-Orenstein AE, Schiller L, Quigley EM, et al. Effect of fibre, antispasmodics, and peppermint oil in the treatment of irritable bowel syndrome: systematic review and meta-analysis. BMJ. 2008;337:a2313.PubMedPubMedCentral
64.
Bentyl (dicyclomine HCl, USP) US Package Insert. Axcan Pharma Inc; 2006.
65.
Yonkers KA, Wisner KL, Stewart DE, Oberlander TF, Dell DL, Stotland N, et al. The management of depression during pregnancy: a report from the American Psychiatric Association and the American College of Obstetricians and Gynecologists. Obstet Gynecol. 2009;114(3):703–13.PubMedPubMedCentral
66.
Davis RL, Rubanowice D, McPhillips H, Raebel MA, Andrade SE, Smith D, et al. Risks of congenital malformations and perinatal events among infants exposed to antidepressant medications during pregnancy. Pharmacoepidemiol Drug Saf. 2007;16(10):1086–94.PubMed
67.
Reis M, Kallen B. Delivery outcome after maternal use of antidepressant drugs in pregnancy: an update using Swedish data. Psychol Med. 2010;40(10):1723–33.PubMed
68.
Ban L, Gibson JE, West J, Fiaschi L, Sokal R, Smeeth L, et al. Maternal depression, antidepressant prescriptions, and congenital anomaly risk in offspring: a population-based cohort study. BJOG. 2014;121(12):1471–81.PubMedPubMedCentral
69.
Hills JM, Aaronson PI. The mechanism of action of peppermint oil on gastrointestinal smooth muscle. An analysis using patch clamp electrophysiology and isolated tissue pharmacology in rabbit and guinea pig. Gastroenterology. 1991;101(1):55–65.PubMed
70.
Moussally K, Berard A. Exposure to herbal products during pregnancy and the risk of preterm birth. Eur J Obstet Gynecol Reprod Biol. 2010;150(1):107–8.PubMed
71.
Moussally K, Berard A. Exposure to specific herbal products during pregnancy and the risk of low birth weight. Altern Ther Health Med. 2012;18(2):36–43.PubMed
72.
Akhavan Amjadi M, Mojab F, Kamranpour SB. The effect of peppermint oil on symptomatic treatment of pruritus in pregnant women. Iranian J Pharmaceut Res. 2012;11(4):1073–7.
73.
Whorwell PJ, Altringer L, Morel J, Bond Y, Charbonneau D, O’Mahony L, et al. Efficacy of an encapsulated probiotic Bifidobacterium infantis 35624 in women with irritable bowel syndrome. Am J Gastroenterol. 2006;101(7):1581–90.PubMed
74.
Guglielmetti S, Mora D, Gschwender M, Popp K. Randomised clinical trial: Bifidobacterium bifidum MIMBb75 significantly alleviates irritable bowel syndrome and improves quality of life--a double-blind, placebo-controlled study. Aliment Pharmacol Ther. 2011;33(10):1123–32.PubMed
75.
Kim HJ, Vazquez Roque MI, Camilleri M, Stephens D, Burton DD, Baxter K, et al. A randomized controlled trial of a probiotic combination VSL# 3 and placebo in irritable bowel syndrome with bloating. Neurogastroenterol Motil. 2005;17(5):687–96.PubMed
76.
Kim HJ, Camilleri M, McKinzie S, Lempke MB, Burton DD, Thomforde GM, et al. A randomized controlled trial of a probiotic, VSL#3, on gut transit and symptoms in diarrhoea-predominant irritable bowel syndrome. Aliment Pharmacol Ther. 2003;17(7):895–904.PubMed
77.
Dugoua JJ, Machado M, Zhu X, Chen X, Koren G, Einarson TR. Probiotic safety in pregnancy: a systematic review and meta-analysis of randomized controlled trials of Lactobacillus, Bifidobacterium, and Saccharomyces spp. J Obstet Gynaecol Can. 2009;31(6):542–52.PubMed
78.
Malinen E, Rinttila T, Kajander K, Matto J, Kassinen A, Krogius L, et al. Analysis of the fecal microbiota of irritable bowel syndrome patients and healthy controls with real-time PCR. Am J Gastroenterol. 2005;100(2):373–82.PubMed
79.
Shukla R, Ghoshal U, Dhole TN, Ghoshal UC. Fecal microbiota in patients with irritable bowel syndrome compared with healthy controls using real-time polymerase chain reaction: an evidence of dysbiosis. Dig Dis Sci. 2015.
80.
Furnari M, Savarino E, Bruzzone L, Moscatelli A, Gemignani L, Giannini EG, et al. Reassessment of the role of methane production between irritable bowel syndrome and functional constipation. J Gastrointestin Liver Dis. 2012;21(2):157–63.PubMed
81.
Pimentel M, Lembo A, Chey WD, Zakko S, Ringel Y, Yu J, et al. Rifaximin therapy for patients with irritable bowel syndrome without constipation. N Engl J Med. 2011;364(1):22–32.PubMed
82.
Menees SB, Maneerattannaporn M, Kim HM, Chey WD. The efficacy and safety of rifaximin for the irritable bowel syndrome: a systematic review and meta-analysis. Am J Gastroenterol. 2012;107(1):28–35; quiz 6.PubMed
83.
Pimentel M, Park S, Mirocha J, Kane SV, Kong Y. The effect of a nonabsorbed oral antibiotic (rifaximin) on the symptoms of the irritable bowel syndrome: a randomized trial. Ann Intern Med. 2006;145(8):557–63.PubMed
84.
Bertoli D, Borelli G. Teratogenic action of Rifaximin in the rat and rabbit and its effect on perinatal development in the rat. Boll Soc Ital Biol Sper. 1984;60(5):1079–85.PubMed
85.
Bertoli D, Borelli G. Fertility study of rifaximin (L/105) in rats. Chemioterapia. 1986;5(3):204–7.PubMed
86.
Baird DD, Narendranathan M, Sandler RS. Increased risk of preterm birth for women with inflammatory bowel disease. Gastroenterology. 1990;99(4):987–94.PubMed
87.
Hudson M, Flett G, Sinclair TS, Brunt PW, Templeton A, Mowat NA. Fertility and pregnancy in inflammatory bowel disease. Int J Gynaecol Obstet. 1997;58(2):229–37.PubMed
88.
Friedman S, McElrath TF, Wolf JL. Management of fertility and pregnancy in women with inflammatory bowel disease: a practical guide. Inflamm Bowel Dis. 2013;19(13):2937–48.PubMed
89.
Tulchinsky H, Averboukh F, Horowitz N, Rabau M, Klausner JM, Halpern Z, et al. Restorative proctocolectomy impairs fertility and pregnancy outcomes in women with ulcerative colitis. Colorectal Dis. 2013;15(7):842–7.PubMed
90.
Lepisto A, Sarna S, Tiitinen A, Jarvinen HJ. Female fertility and childbirth after ileal pouch-anal anastomosis for ulcerative colitis. Br J Surg. 2007;94(4):478–82.PubMed
91.
Mogadam M, Korelitz BI, Ahmed SW, Dobbins 3rd WO, Baiocco PJ. The course of inflammatory bowel disease during pregnancy and postpartum. Am J Gastroenterol. 1981;75(4):265–9.PubMed
92.
Abhyankar A, Ham M, Moss AC. Meta-analysis: the impact of disease activity at conception on disease activity during pregnancy in patients with inflammatory bowel disease. Aliment Pharmacol Ther. 2013;38(5):460–6.PubMedPubMedCentral
93.
Pedersen N, Bortoli A, Duricova D, D Inca R, Panelli MR, Gisbert JP, et al. The course of inflammatory bowel disease during pregnancy and postpartum: a prospective European ECCO-EpiCom Study of 209 pregnant women. Aliment Pharmacol Ther. 2013;38(5):501–12.PubMed
94.
Dominitz JA, Young JC, Boyko EJ. Outcomes of infants born to mothers with inflammatory bowel disease: a population-based cohort study. Am J Gastroenterol. 2002;97(3):641–8.PubMed
95.
Kornfeld D, Cnattingius S, Ekbom A. Pregnancy outcomes in women with inflammatory bowel disease--a population-based cohort study. Am J Obstet Gynecol. 1997;177(4):942–6.PubMed
96.
Lin HC, Chiu CC, Chen SF, Lou HY, Chiu WT, Chen YH. Ulcerative colitis and pregnancy outcomes in an Asian population. Am J Gastroenterol. 2010;105(2):387–94.PubMed
97.
Nguyen GC, Boudreau H, Harris ML, Maxwell CV. Outcomes of obstetric hospitalizations among women with inflammatory bowel disease in the United States. Clin Gastroenterol Hepatol. 2009;7(3):329–34.PubMed
98.
Mahadevan U, Sandborn WJ, Li DK, Hakimian S, Kane S, Corley DA. Pregnancy outcomes in women with inflammatory bowel disease: a large community-based study from Northern California. Gastroenterology. 2007;133(4):1106–12.PubMed
99.
van der Woude CJ, Ardizzone S, Bengtson MB, Fiorino G, Fraser G, Katsanos K, et al. The second European evidenced-based consensus on reproduction and pregnancy in inflammatory bowel disease. J Crohns Colitis. 2015;9(2):107–24.PubMed
100.
Ravid A, Richard CS, Spencer LM, O’Connor BI, Kennedy ED, MacRae HM, et al. Pregnancy, delivery, and pouch function after ileal pouch-anal anastomosis for ulcerative colitis. Dis Colon Rectum. 2002;45(10):1283–8.PubMed
101.
Juhasz ES, Fozard B, Dozois RR, Ilstrup DM, Nelson H. Ileal pouch-anal anastomosis function following childbirth. An extended evaluation. Dis Colon Rectum. 1995;38(2):159–65.PubMed
102.
Remzi FH, Gorgun E, Bast J, Schroeder T, Hammel J, Philipson E, et al. Vaginal delivery after ileal pouch-anal anastomosis: a word of caution. Dis Colon Rectum. 2005;48(9):1691–9.PubMed
103.
Bush MC, Patel S, Lapinski RH, Stone JL. Perinatal outcomes in inflammatory bowel disease. J Matern Fetal Neonatal Med. 2004;15(4):237–41.PubMed
104.
Fedorkow DM, Persaud D, Nimrod CA. Inflammatory bowel disease: a controlled study of late pregnancy outcome. Am J Obstet Gynecol. 1989;160(4):998–1001.PubMed
105.
Morales M, Berney T, Jenny A, Morel P, Extermann P. Crohn’s disease as a risk factor for the outcome of pregnancy. Hepatogastroenterology. 2000;47(36):1595–8.PubMed
106.
Reddy D, Murphy SJ, Kane SV, Present DH, Kornbluth AA. Relapses of inflammatory bowel disease during pregnancy: in-hospital management and birth outcomes. Am J Gastroenterol. 2008;103(5):1203–9.PubMed
107.
Seirafi M, de Vroey B, Amiot A, Seksik P, Roblin X, Allez M, et al. Factors associated with pregnancy outcome in anti-TNF treated women with inflammatory bowel disease. Aliment Pharmacol Ther. 2014;40(4):363–73.PubMed
108.
Asacol (mesalamine). 2010. http://www.accessdata.fda.gov/drugsatfda_docs/label/2010/019651s023lbl.pdf.
109.
Norgard B, Fonager K, Pedersen L, Jacobsen BA, Sorensen HT. Birth outcome in women exposed to 5-aminosalicylic acid during pregnancy: a Danish cohort study. Gut. 2003;52(2):243–7.PubMedPubMedCentral
110.
Rahimi R, Nikfar S, Rezaie A, Abdollahi M. Pregnancy outcome in women with inflammatory bowel disease following exposure to 5-aminosalicylic acid drugs: a meta-analysis. Reprod Toxicol. 2008;25(2):271–5.PubMed
111.
Ban L, Tata LJ, Fiaschi L, Card T. Limited risks of major congenital anomalies in children of mothers with IBD and effects of medications. Gastroenterology. 2014;146(1):76–84.PubMed
112.
Moskovitz DN, Bodian C, Chapman ML, Marion JF, Rubin PH, Scherl E, et al. The effect on the fetus of medications used to treat pregnant inflammatory bowel-disease patients. Am J Gastroenterol. 2004;99(4):656–61.PubMed
113.
Bokstrom H, Holst RM, Hafstrom O, Swolin B, Johansson ML, Brunlof G, et al. Fetal hemolytic anemia associated with maternal sulfasalazine therapy during pregnancy. Acta Obstet Gynecol Scand. 2006;85(1):118–21.PubMed
114.
Baiocco PJ, Korelitz BI. The influence of inflammatory bowel disease and its treatment on pregnancy and fetal outcome. J Clin Gastroenterol. 1984;6(3):211–6.PubMed
115.
Norgard B, Czeizel AE, Rockenbauer M, Olsen J, Sorensen HT. Population-based case control study of the safety of sulfasalazine use during pregnancy. Aliment Pharmacol Ther. 2001;15(4):483–6.PubMed
116.
Mogadam M, Dobbins 3rd WO, Korelitz BI, Ahmed SW. Pregnancy in inflammatory bowel disease: effect of sulfasalazine and corticosteroids on fetal outcome. Gastroenterology. 1981;80(1):72–6.PubMed
117.
Ambrosius Christensen L, Rasmussen SN, Hansen SH, Bondesen S, Hvidberg EF. Salazosulfapyridine and metabolites in fetal and maternal body fluids with special reference to 5-aminosalicylic acid. Acta Obstet Gynecol Scand. 1987;66(5):433–5.PubMed
118.
Esbjorner E, Jarnerot G, Wranne L. Sulphasalazine and sulphapyridine serum levels in children to mothers treated with sulphasalazine during pregnancy and lactation. Acta Paediatr Scand. 1987;76(1):137–42.PubMed
119.
Branski D, Kerem E, Gross-Kieselstein E, Hurvitz H, Litt R, Abrahamov A. Bloody diarrhea--a possible complication of sulfasalazine transferred through human breast milk. J Pediatr Gastroenterol Nutr. 1986;5(2):316–7.PubMed
120.
Transfer of drugs and other chemicals into human milk. Pediatrics. 2001;108(3):776–89.
121.
Park-Wyllie L, Mazzotta P, Pastuszak A, Moretti ME, Beique L, Hunnisett L, et al. Birth defects after maternal exposure to corticosteroids: prospective cohort study and meta-analysis of epidemiological studies. Teratology. 2000;62(6):385–92.PubMed
122.
Skuladottir H, Wilcox AJ, Ma C, Lammer EJ, Rasmussen SA, Werler MM, et al. Corticosteroid use and risk of orofacial clefts. Birth Defects Res A Clin Mol Teratol. 2014;100(6):499–506.PubMedPubMedCentral
123.
Hviid A, Molgaard-Nielsen D. Corticosteroid use during pregnancy and risk of orofacial clefts. CMAJ. 2011;183(7):796–804.PubMedPubMedCentral
124.
Bay Bjorn AM, Ehrenstein V, Hundborg HH, Nohr EA, Sorensen HT, Norgaard M. Use of corticosteroids in early pregnancy is not associated with risk of oral clefts and other congenital malformations in offspring. Am J Ther. 2014;21(2):73–80.PubMed
125.
Kurtoglu S, Sarici D, Akin MA, Daar G, Korkmaz L, Memur S. Fetal adrenal suppression due to maternal corticosteroid use: case report. J Clin Res Pediatr Endocrinol. 2011;3(3):160–2.PubMedPubMedCentral
126.
Homar V, Grosek S, Battelino T. High-dose methylprednisolone in a pregnant woman with Crohn’s disease and adrenal suppression in her newborn. Neonatology. 2008;94(4):306–9.PubMed
127.
Beaulieu DB, Ananthakrishnan AN, Issa M, Rosenbaum L, Skaros S, Newcomer JR, et al. Budesonide induction and maintenance therapy for Crohn’s disease during pregnancy. Inflamm Bowel Dis. 2009;15(1):25–8.PubMed
128.
Ost L, Wettrell G, Bjorkhem I, Rane A. Prednisolone excretion in human milk. J Pediatr. 1985;106(6):1008–11.PubMed
129.
Cleary BJ, Kallen B. Early pregnancy azathioprine use and pregnancy outcomes. Birth Defects Res A Clin Mol Teratol. 2009;85(7):647–54.PubMed
130.
Coelho J, Beaugerie L, Colombel JF, Hebuterne X, Lerebours E, Lemann M, et al. Pregnancy outcome in patients with inflammatory bowel disease treated with thiopurines: cohort from the CESAME Study. Gut. 2011;60(2):198–203.PubMed
131.
de Boer NK, Jarbandhan SV, de Graaf P, Mulder CJ, van Elburg RM, van Bodegraven AA. Azathioprine use during pregnancy: unexpected intrauterine exposure to metabolites. Am J Gastroenterol. 2006;101(6):1390–2.PubMed
132.
Jharap B, de Boer NK, Stokkers P, Hommes DW, Oldenburg B, Dijkstra G, et al. Intrauterine exposure and pharmacology of conventional thiopurine therapy in pregnant patients with inflammatory bowel disease. Gut. 2014;63(3):451–7.PubMed
133.
Norgard B, Pedersen L, Christensen LA, Sorensen HT. Therapeutic drug use in women with Crohn’s disease and birth outcomes: a Danish nationwide cohort study. Am J Gastroenterol. 2007;102(7):1406–13.PubMed
134.
Shim L, Eslick GD, Simring AA, Murray H, Weltman MD. The effects of azathioprine on birth outcomes in women with inflammatory bowel disease (IBD). J Crohns Colitis. 2011;5(3):234–8.PubMed
135.
Akbari M, Shah S, Velayos FS, Mahadevan U, Cheifetz AS. Systematic review and meta-analysis on the effects of thiopurines on birth outcomes from female and male patients with inflammatory bowel disease. Inflamm Bowel Dis. 2013;19(1):15–22.PubMed
136.
Broms G, Granath F, Linder M, Stephansson O, Elmberg M, Kieler H. Birth outcomes in women with inflammatory bowel disease: effects of disease activity and drug exposure. Inflamm Bowel Dis. 2014;20(6):1091–8.PubMed
137.
Ng SW, Mahadevan U. My treatment approach to management of the pregnant patient with inflammatory bowel disease. Mayo Clin Proc. 2014;89(3):355–60.PubMed
138.
Christensen LA, Dahlerup JF, Nielsen MJ, Fallingborg JF, Schmiegelow K. Azathioprine treatment during lactation. Aliment Pharmacol Ther. 2008;28(10):1209–13.PubMed
139.
Gardiner SJ, Gearry RB, Roberts RL, Zhang M, Barclay ML, Begg EJ. Exposure to thiopurine drugs through breast milk is low based on metabolite concentrations in mother-infant pairs. Br J Clin Pharmacol. 2006;62(4):453–6.PubMedPubMedCentral
140.
Sau A, Clarke S, Bass J, Kaiser A, Marinaki A, Nelson-Piercy C. Azathioprine and breastfeeding: is it safe? BJOG. 2007;114(4):498–501.PubMed
141.
Angelberger S, Reinisch W, Messerschmidt A, Miehsler W, Novacek G, Vogelsang H, et al. Long-term follow-up of babies exposed to azathioprine in utero and via breastfeeding. J Crohns Colitis. 2011;5(2):95–100.PubMed
142.
Zelinkova Z, de Haar C, de Ridder L, Pierik MJ, Kuipers EJ, Peppelenbosch MP, et al. High intra-uterine exposure to infliximab following maternal anti-TNF treatment during pregnancy. Aliment Pharmacol Ther. 2011;33(9):1053–8.PubMed
143.
Bortlik M, Machkova N, Duricova D, Malickova K, Hrdlicka L, Lukas M, et al. Pregnancy and newborn outcome of mothers with inflammatory bowel diseases exposed to anti-TNF-alpha therapy during pregnancy: three-center study. Scand J Gastroenterol. 2013;48(8):951–8.PubMed
144.
Casanova MJ, Chaparro M, Domenech E, Barreiro-de Acosta M, Bermejo F, Iglesias E, et al. Safety of thiopurines and anti-TNF-alpha drugs during pregnancy in patients with inflammatory bowel disease. Am J Gastroenterol. 2013;108(3):433–40.PubMed
145.
Katz JA, Antoni C, Keenan GF, Smith DE, Jacobs SJ, Lichtenstein GR. Outcome of pregnancy in women receiving infliximab for the treatment of Crohn’s disease and rheumatoid arthritis. Am J Gastroenterol. 2004;99(12):2385–92.PubMed
146.
Nielsen OH, Loftus Jr EV, Jess T. Safety of TNF-alpha inhibitors during IBD pregnancy: a systematic review. BMC Med. 2013;11:174.PubMedPubMedCentral
147.
Schnitzler F, Fidder H, Ferrante M, Ballet V, Noman M, Van Assche G, et al. Outcome of pregnancy in women with inflammatory bowel disease treated with antitumor necrosis factor therapy. Inflamm Bowel Dis. 2011;17(9):1846–54.PubMed
148.
A Special Meeting Review Edition: Highlights in Crohn’s Disease and Ulcerative Colitis: Digestive Disease Week 2012 May 19–22, 2012 * San Diego, CaliforniaSpecial Reporting on:* Safety and Efficacy of Subcutaneous Golimumab Induction Therapy in Patients with Moderately to Severely Active UC: PURSUIT-SC* The Future of IBD Therapy: Individualized and Optimized Therapy and Novel Mechanisms* Infliximab Concentration and Clinical Outcome in Patients with UC* Vedolizumab Induction Therapy for UC: Results of GEMINI I, A Randomized, Placebo-Controlled, Double-Blind, Multicenter, Phase III Trial* Novel Infliximab and Antibody-to-lnfliximab Assays Are Predictive of Disease Activity in Patients with CD* Accelerated Step-Care Therapy with Early Azathioprine Versus Conventional Step-Care Therapy in CD* PIANO: A 1,000-Patient Prospective Registry of Pregnancy Outcomes in Women with IBD Exposed to Immunomodulators and Biologic TherapyPLUS Meeting Abstract Summaries With Expert Commentary by: William J. Sandborn, MDChief of the Division of Gastroenterology Director of the UCSD IBD Center UC San Diego Health System La Jolla, California. Gastroenterol Hepatol. 2012;8(8 Suppl 5):1–24.
149.
Mahadevan U, Wolf DC, Dubinsky M, Cortot A, Lee SD, Siegel CA, et al. Placental transfer of anti-tumor necrosis factor agents in pregnant patients with inflammatory bowel disease. Clin Gastroenterol Hepatol. 2013;11(3):286–92; quiz e24.PubMed
150.
Gisbert JP, Chaparro M. Safety of anti-TNF agents during pregnancy and breastfeeding in women with inflammatory bowel disease. Am J Gastroenterol. 2013;108(9):1426–38.PubMed
151.
Kane S. Anti-tumor necrosis factor agents and placental transfer: relevant clinical data for rational decision-making. Clin Gastroenterol Hepatol. 2013;11(3):293–4.PubMed
152.
Nielsen OH, Jess T. IBD: Can TNF inhibitors be administered during the third trimester? Nat Rev Gastroenterol Hepatol. 2013;10(3):130–1.PubMed
153.
Mahadevan U, Cucchiara S, Hyams JS, Steinwurz F, Nuti F, Travis SP, et al. The London Position Statement of the World Congress of Gastroenterology on Biological Therapy for IBD with the European Crohn’s and Colitis Organisation: pregnancy and pediatrics. Am J Gastroenterol. 2011;106(2):214–23; quiz 24.PubMed
154.
Zelinkova Z, van der Ent C, Bruin KF, van Baalen O, Vermeulen HG, Smalbraak HJ, et al. Effects of discontinuing anti-tumor necrosis factor therapy during pregnancy on the course of inflammatory bowel disease and neonatal exposure. Clin Gastroenterol Hepatol. 2013;11(3):318–21.PubMed
155.
Ben-Horin S, Yavzori M, Kopylov U, Picard O, Fudim E, Eliakim R, et al. Detection of infliximab in breast milk of nursing mothers with inflammatory bowel disease. J Crohns Colitis. 2011;5(6):555–8.PubMed
156.
Kane S, Ford J, Cohen R, Wagner C. Absence of infliximab in infants and breast milk from nursing mothers receiving therapy for Crohn’s disease before and after delivery. J Clin Gastroenterol. 2009;43(7):613–6.PubMed
157.
Wehner NG, Shopp G, Rocca MS, Clarke J. Effects of natalizumab, an alpha4 integrin inhibitor, on the development of Hartley guinea pigs. Birth Defects Res B Dev Reprod Toxicol. 2009;86(2):98–107.PubMed
158.
Wehner NG, Skov M, Shopp G, Rocca MS, Clarke J. Effects of natalizumab, an alpha4 integrin inhibitor, on fertility in male and female guinea pigs. Birth Defects Res B Dev Reprod Toxicol. 2009;86(2):108–16.PubMed
159.
Hellwig K, Haghikia A, Gold R. Pregnancy and natalizumab: results of an observational study in 35 accidental pregnancies during natalizumab treatment. Mult Scler. 2011;17(8):958–63.PubMed
160.
P563. Vedolizumab exposure in pregnancy: outcomes from clinical studies in inflammatory bowel disease. J Crohns Colitis. 2015;9 Suppl 1:S361–2.