Shlomo Raz1
(1)
Division of Pelvic Medicine and Reconstructive Surgery, UCLA School of Medicine, Los Angeles, CA, USA
Electronic supplementary material
The online version of this chapter (doi:10.1007/978-1-4939-2941-2_5) contains supplementary material, which is available to authorized users.
Keywords
FistulaVesicovaginal fistulaUrethrovaginal fistulaPeritoneal flapRotational pudendal artery-based inner thigh flapMartius flapRectovaginal fistulaRectoperineal fistula
There are three sections in the chapter on fistulae: vesicovaginal and urethrovaginal fistula, rectovaginal fistula, and repair of perineal fistula.
5.1 Vesicovaginal Fistula and Urethrovaginal Fistula
The appearance of a urinary fistula to the vagina is one of the most devastating complications after surgery. The emotional distress of the patient and the surgeon is high because of the little hope that conservative therapy offers and the need (in the majority of cases) for a second surgery to correct the problem. In developed countries, the most common cause of vesicovaginal fistulae is gynecologic surgery, specifically hysterectomy. Other causes include urologic surgery or manipulation, trauma, gastrointestinal surgery, and radiation therapy for pelvic malignancies. Obstetric trauma resulting in fistulae is more common in underdeveloped nations.
5.1.1 Diagnosis
The suspicion of a fistula should lead one to diagnostic maneuvers to confirm or deny its presence. The clinical history and pelvic examination will aid in diagnosis. An unaccounted increase in vaginal drainage and the occurrence of bloody urine immediately after hysterectomy may be suggestive of fistula formation. Most vesicovaginal fistulae classically present as continuous day and night incontinence following a recent pelvic operation, but a watery vaginal discharge accompanied by normal voiding may be the only sign of a small fistula.
Fistulae related to radiation therapy may develop months after therapy or up to 20 years later, and recurrent tumor must be considered. Ureterovaginal fistulae may present as both continuous leakage from the vagina and normal voiding.
Distending the bladder with saline dyed with methylene blue or indigo carmine can identify the site of leakage in the vagina. Cystoscopy and vaginoscopy may demonstrate the size, location, and relation to the ureteric orifices, as well as collateral fistulae. A biopsy of the site is mandatory in any patient with a history of pelvic neoplasm. Performing these studies under anesthesia may aid in diagnosis.
Up to 10 % of patients with a vesicovaginal fistula may have a second fistula, especially a ureterovaginal fistula. CT urogram may demonstrate partial or complete obstruction suggesting a ureterovaginal fistula, but many times it can be perfectly normal. Retrograde pyelograms are more likely to demonstrate the exact location of a ureterovaginal fistula.
A cystogram or voiding cystourethrogram may demonstrate the extent of the fistula, the presence of vesicoureteral reflux, and associated urethral or vesical prolapse or stress incontinence, which may require simultaneous repair (Fig. 5.1).

Fig. 5.1
(a) Lateral cystogram of a patient suffering from a high vesicovaginal fistula after hysterectomy. (b) Cystoscopic findings in a patient with a recurrent vesicovaginal fistula at the vaginal cuff
5.1.2 Indications for Surgery
As soon as the diagnosis of fistula is made, a trial of conservative therapy should be started. These measures include proper and undisturbed bladder drainage and antibiotics when indicated. If the fistula is extremely small (1 mm), coagulation of the fistulous tract may occasionally be successful. Only 5–10 % of small iatrogenic fistulae will close spontaneously with continuous bladder drainage and antibiotics. If the fistula has not closed after 3 weeks of catheter drainage, it is unlikely that it will close without surgical intervention. When conservative treatment has failed and the fistula is large enough that most of the urine passes per vagina in spite of continuous bladder drainage, immediate surgical repair is indicated.
5.1.3 Preoperative Considerations
Sound surgical principles should be used in the treatment of this condition. The blood supply to the tissues must be adequate to support the area of repair. The tissues must be in optimal condition for repair. That is, tissue should be free of infection, excessive inflammation, and cancer. A layered, tension-free closure, and tissue interposition should be used, avoiding overlapping suture lines. Suture material must be absorbable and cause little tissue reaction.
Continuous, uninterrupted postoperative urinary drainage is critical to prevent extravasations and distention with breakdown of suture lines. A urethral catheter and, if required, suprapubic catheter drainage are highly recommended.
In planning the treatment of vesicovaginal fistula, several important issues should be addressed: the timing of surgery; the surgical approach (abdominal, vaginal, or combined); the use of estrogens or antibiotics; the decision whether to excise the fistulous tract; and the use of adjuvant surgical measures to improve the repair, such as flaps from the labia (Martius fibro-fatty flaps), vagina, peritoneum, or skin.
5.1.3.1 Timing of Surgery
The timing of surgery remains controversial. It is obvious that infection of the vaginal cuff or pelvic infection after abdominal hysterectomy requires prolonged antibiotic therapy before any attempted repair is made. The classic opinion regarding timing of repair is to wait 3–6 months to allow the surgical inflammatory reaction to subside, particularly if an abdominal approach is contemplated and the etiology of the fistula was a complicated abdominal hysterectomy. Shortening the waiting period is socially very important for these “totally wet” patients, who are already distressed; early surgery would be of great psychological help. Nevertheless, one should not trade social convenience for compromised surgical success.
We favor early repair in the properly selected patient. Early surgery is not indicated for patients with ischemia (obstetric fistula), cuff infection, or poor general condition. For others, we do not feel that the short wait period has added extra risks to the surgical results. The patients are very satisfied with this early transvaginal repair, which prevents the emotional distress of long months of constant wetting.
5.1.3.2 Abdominal or Vaginal Approach?
The selected route of repair depends mostly on the surgeon’s training and experience. The best approach is probably the one with which the surgeon feels most experienced and comfortable. We personally favor the vaginal approach because we can avoid a laparotomy and the splitting of the bladder. With the advances in robotic and laparoscopy surgery, repair of vesico vaginal fistulae can be done also in a minimal invasive approach. The vaginal repair is done as an outpatient procedure, providing a quicker recovery with less morbidity. We reserve the abdominal approach for rare patients with intra-abdominal pathology that requires simultaneous care. The most common case is radiation cystitis and fistula with a small contracted bladder capacity requiring cystoplasty and fistula repair. Radiation fistula per se does not preclude the vaginal approach if the bladder capacity is appropriate.
5.1.3.3 Use of Estrogens and Antibiotics
Estrogen replacement in the postmenopausal or post-hysterectomy patient may improve the tissue quality and vascular supply of the vaginal wall, aiding in healing, so it is strongly recommended. Broad-spectrum antibiotics are required in any fistula repair, particularly if an early approach is used.
5.1.3.4 Excision of the Fistulous Tract
The classic approach to the repair of vesicovaginal fistula or any other fistula includes the wide excision of the fistulous tract in order to freshen the margins and provide a better repair. We have challenged this dogma and have not excised the fistulous tract in our last 210 cases of early transvaginal repair of vesicovaginal fistula, without apparent adverse effects. In our view, not excising the fistulous tract has many advantages: (1) During repair, a small fistula stays small if it is not excised, whereas excising a fistulous tract makes a small opening into one that is very large. (2) Bleeding of the freshly excised margins may require coagulation of the edges and compromise the closure. (3) The fistulous tract provides a ring of protection against postsurgical bladder spasms that, if severe, may compromise the healing. If the fistulous tract is removed, this protective ring is lost and the fresh repair may be at higher risk from severe bladder spasms. (4) Excision of a fistulous tract that is very close to the ureteric orifices will necessitate open surgery and reimplantation of the ureters, whereas if the fistula is not excised, the ureter can be catheterized with a stent, and a safe transvaginal closure of the fistula can be performed under direct vision of the ureteric catheter, avoiding ureteric reimplantation. We have found this to be the greatest advantage of not excising the fistulous tract.
5.1.3.5 Adjuvant Measures
In most patients, uncomplicated vesicovaginal fistula requires only multilayer tension-free repair of the fistula. But when complicating factors such as prior radiation, prior failed surgery, or poor quality of tissues are present, adjuvant measures are required, including the techniques listed below.
In short, our technique of transvaginal repair of vesicovaginal fistula includes no excision of the fistulous tract, multilayer tension-free closure of the fistula, and the advancement of a vaginal flap to cover the area of the repair with healthy tissue. Very often, however, it is desirable or necessary to reinforce a routine closure. Several measures are available in these cases:
· The use of fibro-fatty tissue from the labia (Martius flap), used mainly in cases of urethral or trigonal fistula
· The use of rotation flaps of the entire labia, inner thigh skin, or both to cover large vaginal wall defects
· The use of myocutaneous gracilis muscle flaps for unusual radiation injury
· The use of peritoneum flaps in the repair of high fistulae (repair of a high vesicovaginal fistula after hysterectomy is the most common repair)
5.1.3.6 Repair of Post-hysterectomy Vesicovaginal Fistula Using a Peritoneal Flap
The patient is placed in the lithotomy position. In a narrowed vaginal vault, increased exposure can be obtained by posterolateral relaxing incisions prior to insertion of the posterior-weighted vaginal retractor. If the fistula is close to the ureteral orifices, cystoscopy and ureteral catheterization should be performed. A urethral catheter and optionally a suprapubic catheter can be placed by means of a Lowsley retractor. Figures 5.2, 5.3, 5.4, 5.5, 5.6, 5.7, 5.8, 5.9, and 5.10 illustrate the technique for transvaginal repair of the fistulous tract using a flap of vaginal tissue.

Fig. 5.2
To facilitate the exposure (in particular, exposure of a high fistula after hysterectomy), the fistulous tract is dilated to 12 Fr and a small 8–10-Fr Foley catheter is inserted into the fistula. This catheter will facilitate retraction and exposure at the time of initial preparation of the vaginal flaps. A green line marks the area of the circumferential incision around the fistula

Fig. 5.3
Diagram of the vaginal incisions. An inverted J shaped incision is made in the anterior vaginal wall circumscribing the fistula. The long end of the incision is extended toward the apex of the vagina. The convex portion of the incision circumscribes the fistulous tract. This asymmetric incision allows for the later advancement and rotation of the posterior flap over the fistula repair

Fig. 5.4
Diagram of the two flaps created anterior and posterior to the fistula

Fig. 5.5
(a) A U-shaped anterior vaginal wall flap is prepared. Creation of the flaps is begun in healthy tissue away from the fistulous tract. This maneuver aids in the dissection of proper tissue planes without perforating the bladder or increasing the size of the fistulous tract. The scarred margin of the fistula is left in place, and no attempt is made to dissect the thin layer of the vaginal wall overlying the fistula. The green line indicate the outline of the anterior flap preparation. (b) The flap of the vaginal wall is retracted superiorly. The dissection of the flaps is extended at least 2–4 cm from the fistulous tract, exposing the perivesical fascia

Fig. 5.6
A wide posterior vaginal flap is prepared. The dissection is carried out for a distance of at least 4–5 cm from the fistula. It is directed toward the bladder base without incorporating the margins of the fistula. The flap is retracted posterior

Fig. 5.7
Exposure of the fistulous tract with the catheter in place is seen. Further lateral dissection has been performed to expose the fistulous tract for at least a distance of 3–4 cm around the fistula. The cuff of the vagina is often involved and can be excised

Fig. 5.8
Diagram of the closure of the fistulous tract with the superior and inferior vaginal flaps retracted

Fig. 5.9
(a) Interrupted sutures of #2-0 delayed absorbable sutures are used to close the first layer of the repair. Included in this suture line is the whole thickness of the bladder and the vaginal wall, which has remained connected to the fistulous tract. A strong bite of tissue 2–3 mm from the margin of the fistula is obtained. (b) After application of the sutures, the intra-fistula catheter is removed and the sutures are tied, closing the fistulous tract. The bladder is irrigated with diluted indigo carmine solution to ensure that no extravasations are seen after the sutures are tied

Fig. 5.10
(a) The perivesical fascia is dissected free and mobilized around the fistulous tract. (b) A second layer of interrupted absorbable sutures is used to invert and cover the prior layer. These sutures include the perivesical fascia and deep musculature of the bladder. The sutures should be applied at least 1 cm from the prior line of sutures and should be tied free of tension. The first layer of the repair remains invaginated and is covered by the second layer of sutures. At this time, the bladder again is filled with diluted indigo carmine solution to test the integrity of the repair. In a simple repair, the operation can now be completed. The previously raised anterior flap is partially resected, allowing the posterior (proximal) flap to rotate and extend beyond the closure of the fistula in order to cover the fistulous tract with fresh vaginal tissue and to avoid adjacent and overlapping suture lines. A running suture of #2-0 absorbable material is used to complete this layer
Figure 5.11 shows the use of omentum.

Fig. 5.11
In cases of prior failed surgery with the use of omentum, a peritoneal window is made. The omentum is freed and advanced with interrupted sutures to cover the fistula repair
In patients with high fistula after hysterectomy, we routinely use a peritoneal flap to cover the fistulous tract. This simple maneuver (Figs. 5.12, 5.13, 5.14, 5.15, 5.16, and 5.17) will result in a multilayer closure of the fistula with tissue that is easy to harvest and is found in the vicinity of the surgical area.

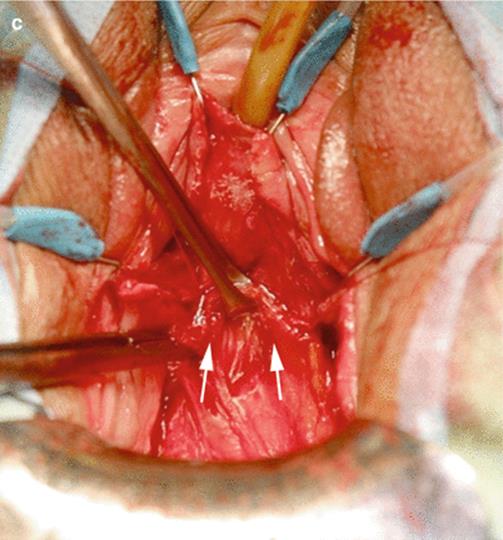
Fig. 5.12
(a) Diagram of the dissection of the perivesical fascia, extended more proximally toward the prerectal fascia to expose the peritoneal fold. (b) Allis clamps retract the double-fold peritoneal flap, helping its dissection and mobilization. (c) The peritoneal flap is advanced 2 cm distal to the fistula repair so a double layer of peritoneum covers the fistula repair. If the peritoneum is open during this maneuver, it is not necessary to close because the advancement of the flap against the posterior bladder wall will close it. The peritoneal flap is extensively mobilized to allow a tension free advancement to cover the fistula repair

Fig. 5.13
Diagram of the anatomical location of a high vesicovaginal fistula and its relationship to the surrounding peritoneum

Fig. 5.14
Diagram of the interposition of the peritoneal flap between the vagina and bladder

Fig. 5.15
(a) Diagram of the peritoneal flap advancement a few centimeters distal to the suture line of the fistula repair. (b) Interrupted absorbable sutures are used to advance the peritoneal flap. The sutures are applied over the perivesical fascia several centimeters distal of the original line of closure of the fistulous tract. Green line depicts the peritoneal flap advancement. Arrowsindicated the line of sutures of the peritoneal flap to cover the area of the fistula repair. The white dot marks the area of the fistula closure

Fig. 5.16
(a) The anterior vaginal wall flap created at the beginning of the surgery is exposed. (b) Diagram of the excision of the distal vaginal flap that will allow to cover the fistula repair with new tissue from the posterior flap

Fig. 5.17
(a) Diagram of the advancement of the vaginal wall to cover the area of the reconstruction. (b) Final closure of the vagina: #2-0 absorbable sutures are used to advance the posterior flap over the freshly excised margin of the anterior flap to cover far distal the area of the repair of the fistula (white dot)
5.1.3.7 Repair of Urethrovaginal Fistula Due to Radiation: Use of a Martius Flap
The patient shown in Figs. 5.18, 5.19, 5.20, 5.21, 5.22, 5.23, 5.24, 5.25, 5.26, and 5.27 presented with a recurrent vesicovaginal fistula located at the proximal urethra, 1 cm distal to the bladder neck. There was no hydronephrosis.
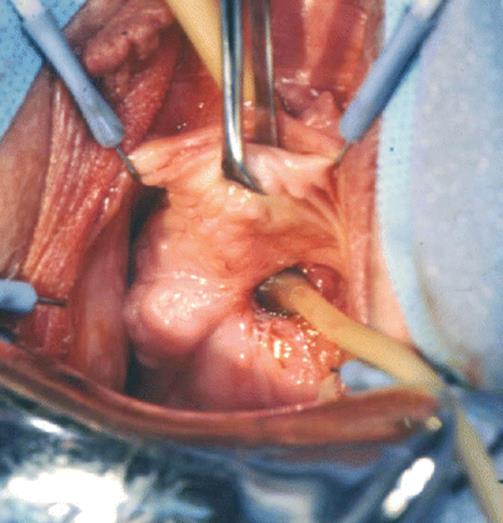
Fig. 5.18
A Foley catheter is inserted in the large fistulous tract

Fig. 5.19
A circular incision is made around the fistula, and superior and inferior flaps of the vaginal wall are created. The fistulous tract itself is not excised

Fig. 5.20
Two layers of delayed absorbable sutures are used to close the fistulous tract. The first layer includes the whole fistulous tract, and the second layer uses the perivesical fascia to cover the first layer

Fig. 5.21
The sutures over the perivesical fascia have been tied. The Foley catheter is irrigated with diluted indigo carmine solution to ensure proper closure of the fistula. Tying the sutures over the periurethral fascia will imbricate and cover the first line of sutures

Fig. 5.22
The left labia majora is exposed

Fig. 5.23
The fibro-fatty tissue of the labia is dissected free from the underlying adductor fascia and is isolated over a retractor. A Penrose drain will be placed under the flap to facilitate the dissection

Fig. 5.24
The Martius flap is dissected superiorly toward the anterior pubic bone and transected. The flap is mobilized proximal toward the base of the labia majora. A tunnel will be made under the labia toward the left vaginal wall incision

Fig. 5.25
Diagram of the transfer of the Martius flap under the labia toward the anterior vaginal wall

Fig. 5.26
The flap has been transferred and interrupted sutures are used to expand the flap and widely cover the area of the repair

Fig. 5.27
The anterior vaginal wall is advanced distally to cover the fistula repair and the underlying Martius flap
5.1.3.8 Repair of Urethrovaginal Fistula After Anterior Repair: Use of a Martius Flap
The etiology of a urethrovaginal fistula and the principles of its repair are slightly different from those of a vesicovaginal fistula. The etiology of urethral fistula is mainly obstetric or surgical, especially as a complication of anterior colporrhaphy, repair of urethral diverticula, or transvaginal surgery for incontinence using mesh. The urethra is a high-pressure system during voiding, and with the inherent weakness of the traumatized urethral wall, a fibro-fatty flap of the labia (Martius flap) is strongly recommended in most cases.
The patient shown in Figs. 5.28, 5.29, 5.30, 5.31, 5.32, and 5.33 presented with a large urethral defect after complications of the excision of a urethral diverticulum.

Fig. 5.28
Voiding cystogram of the patient. Urine leaks into the vagina through the midurethral fistula
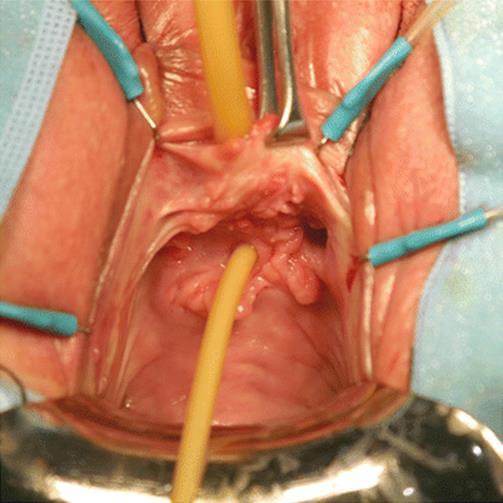
Fig. 5.29
Exposure of the anterior vaginal wall shows the urethral defect. A small Foley catheter is inserted into the fistula

Fig. 5.30
(a) An inverted U incision is made posterior to the fistula, and an anterior vaginal wall flap is created. It is extended toward the bladder neck. A distal vaginal wall is also created to expose the length of the urethra. (b) The anterior vaginal flap is retracted inferiorly to expose the urethral fistula and a few centimeters around it

Fig. 5.31
(a) A transverse incision is made over the periurethral fascia, and two flaps of periurethral fascia (arrows) are dissected superior and inferior to the fistula. (b) The urethral wall is closed with running, locking #3-0 delayed absorbable sutures and a second layer of interrupted figure-of-eight sutures to reinforce the closure. (c) The urethral wall is closed and the integrity of the closure is tested by the injection of diluted indigo carmine solution through the urethral meatus. Arrows indicate the line of sutures closing the urethral wall

Fig. 5.32
(a) The inferior margin of the transversely incised periurethral fascia (arrow) is elevated and mobilized as required to reach the distal segment of the incision. (b) The inferior margin of the periurethral fascia is advanced distally to cover the area of the fistula. (c) The periurethral fascia is closed in a transverse fashion with interrupted figure-of-eight #3-0 delayed absorbable sutures, providing support and cover to the area of the fistula

Fig. 5.33
(a) A Martius flap is prepared and mobilized in the right labia. It will be transferred toward the anterior vaginal wall, covering the reconstructed periurethral fascia. Sutures are applied to spread the flap a few centimeters distal, proximal, and lateral to the line of sutures. (b) The flap of the anterior vaginal wall is advanced distally, covering the Martius flap and the reconstructed urethra
5.1.3.9 Repair of Urethrovaginal and Urethral Reconstruction After Mesh and Urethral Transection: Use of an in-situ Martius Flap
Four months after an obturator sling procedure, the patient in Figs. 5.34, 5.35, 5.36, 5.37, 5.38, 5.39, 5.40, 5.41, 5.42, and 5.43 presented with pain, urinary infections, and severe incontinence due to a urethrovaginal fistula. The arms of the mesh were already removed in another surgery.

Fig. 5.34
Exposure of the anterior vaginal wall showing a midurethral fistula (arrow). A Foley catheter is in place

Fig. 5.35
An inverted U anterior vaginal flap is created and dissected toward the bladder neck area. The mesh surrounding the urethra has been excised in the right urethral wall

Fig. 5.36
The mesh is dissected free from the urethral wall and removed lateral to the urethra toward the obturator fossa

Fig. 5.37
The mesh is removed and the large circular defect of the urethra is seen
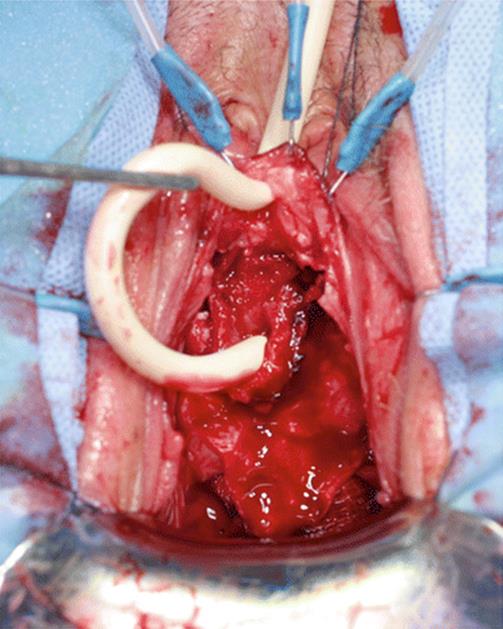
Fig. 5.38
The Foley catheter is retracted to expose the large urethral defect and the small bridge of posterior urethral wall

Fig. 5.39
An end-to-end anastomosis of the urethra has been performed in two layers of #3-0 delayed absorbable sutures

Fig. 5.40
A tunnel is made under the right labia to expose the fibro-fatty tissue of the labia. An in situ Martius flap will be used to cover the urethral defect

Fig. 5.41
Exposure of the inner aspect of the labia showing the fibro-fatty tissue that will be used for the in situ Martius flap

Fig. 5.42
After dissection and exposure of the right labial fatty tissue, the upper segment is freed and transected. The Martius flap is transferred toward the anterior urethral wall and sutured to the periurethral fascia, forming a wide area of cover

Fig. 5.43
The anterior vaginal wall flap is advanced to cover the area of the reconstruction
5.1.3.10 Repair of Urethral Fistula with Total Urethral Transection as Complication of Hymeneal Excision: Use of a Martius Flap
An 18-year-old woman had a surgical procedure to excise the hymeneal ring. As a complication, the whole urethra was transected from the external meatus to the bladder neck, resulting in total urinary incontinence. Figures 5.44, 5.45, 5.46, 5.47, 5.48, and 5.49 illustrate the reconstructive procedure.

Fig. 5.44
Exposure of the anterior vaginal wall shows the open urethral wall from the meatus to the bladder neck. The tissues have healed at the margin of the incision

Fig. 5.45
An inverted U flap of the anterior vaginal wall is created proximal to the fistula. The flap is mobilized to reach the external meatus

Fig. 5.46
Two parallel incisions are made between the open urethral mucosa and the vaginal wall. The lateral vaginal flaps and the medial urethral wall are mobilized

Fig. 5.47
The mucosa and the whole urethral wall are approximated using two layers of #3-0 delayed absorbable sutures

Fig. 5.48
An in situ Martius flap is prepared and transferred to cover the urethral defect. Multiple sutures are used to anchor the flap over the area of urethral reconstruction

Fig. 5.49
The anterior vaginal flap is advanced to cover the Martius flap. The Foley catheter was left indwelling for 3 weeks. The patient regained full continence after the procedure because the midline 6 o’clock surgical incision did not damage the lateral or anterior segments of the urethra
5.1.3.11 Repair of Recurrent Radiation Vesicovaginal Fistula in a Patient with Prior Colpocleisis: Use of a Rotational Labial Flap
The patient shown in Figs. 5.50, 5.51, 5.52, 5.53, 5.54, 5.55, and 5.56 developed a vesicovaginal fistula of the bladder base after pelvic radiation. Three prior surgeries, including colpocleisis, failed to correct the problem.

Fig. 5.50
Voiding cystogram showing the recurrent fistula of the bladder next to the trigone

Fig. 5.51
Exposure of the anterior vaginal wall shows the short vaginal canal, with a Foley catheter in the bladder and a second one in the fistulous tract. A circular incision is made over the fistula. The incision is extended as an inverted U incision of the left labia. The skin and subcutaneous tissues are incised, elevated, and dissected free to allow rotation of the labial flap. The vascular supply of the labia will be from the superior pudendal pedicle, coming from the femoral artery

Fig. 5.52
After exposure of the anterior bladder wall, a catheter is inserted into the fistulous tract

Fig. 5.53
The labial flap was rotated toward the left, exposing the bladder wall and the fistula. The fistula is closed in two layers of delayed absorbable sutures. The bladder is irrigated with diluted indigo carmine solution to ensure the integrity of the fistula closure

Fig. 5.54
After closure of the fistula, the labial flap is rotated medially. The medial segment of the flap covers the area of the fistula closure so new, thick fat pad and vascularized tissue cover the fistula repair

Fig. 5.55
The rotational flap transfer has been completed and new labial flap tissue covers the area of the fistula. After removal of the catheter 4 weeks after surgery, the fistula was cured

Fig. 5.56
Exposure of the vaginal wall shows a large defect after closure of a recurrent radiation vesicovaginal fistula. The fistula was closed in layers. The patient had a wide introitus. Two layers of delayed absorbable sutures were used to close the fistula. After closure of the fistula the inner thigh is exposed in the left side
5.1.3.12 Repair of Recurrent Radiation Vesicovaginal Fistula: Use of a Pudendal Artery-Based Inner Thigh Island Flap
Inner thigh flaps provide great versatility and excellent vascularized skin and fatty tissue to cover large vaginal defects. For patients with a wide introitus and radiation fistula, this island flap is our preferred technique (Figs. 5.57, 5.58, 5.59, 5.60, 5.61, 5.62, 5.63, 5.64, 5.65, 5.66, 5.67, and 5.68). In patients with a narrow introitus due to prior surgery or radiation, a rotational inner thigh flap will be used after a lateral episiotomy, as demonstrated in the next case.

Fig. 5.57
The area of the inner thigh lateral to the left labia is exposed

Fig. 5.58
The area of the incision is marked. It will be an inverted U flap with a wide base just lateral to the labia majora

Fig. 5.59
The incision has been made and the flap is mobilized from the underlying adductor fascia

Fig. 5.60
A tunnel is made under the left labia toward the vaginal canal and the area of the closed fistula. A wide tunnel is created to allow the passage of the inner thigh flap

Fig. 5.61
A Penrose drain is inserted at the labial flap for retraction

Fig. 5.62
An incision will be made at the distal flap and a rectangle of skin will be isolated. The size of the rectangle will depend on the size of the vaginal defect

Fig. 5.63
The skin at the base of the inner thigh flap is excised, taking care to remain superficial to avoid damaging the vascular supply to the flap

Fig. 5.64
Before transferring the flap, interrupted sutures are applied to the margins of the vaginal defect. The needles are left in place and will be used to suture the vaginal margins to the island flap
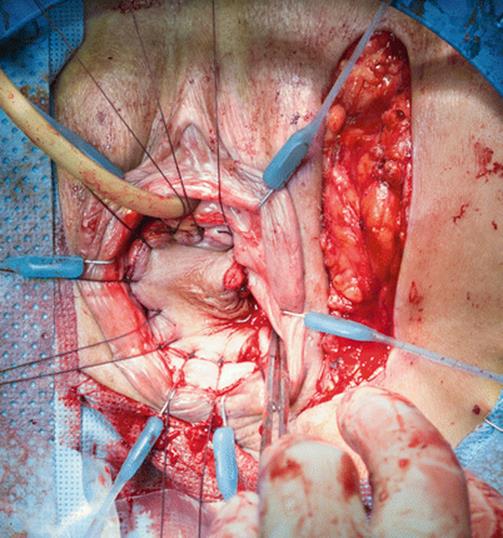
Fig. 5.65
The rectangular flap has been transferred, and the preplaced vaginal sutures are incorporated to the flap in a sequential fashion

Fig. 5.66
The vaginal sutures are sequentially tension-free. The fistula is now covered by the flap of well-vascularized skin and fatty tissue

Fig. 5.67
The skin defect of the inner thigh is closed with interrupted sutures after undermining the medial and lateral margins of the donor site

Fig. 5.68
Voiding cystogram in a patient suffering from recurrent radiation vesicovaginal fistula. The fistula is at the cuff of the hysterectomy, in a very high position. The vaginal canal is narrow and stenotic
5.1.3.13 Repair of Recurrent Radiation Vesicovaginal Fistula: Use of Rotational Inner Thigh Flap in a Patient with Vaginal Stenosis
The patient in Figs. 5.69, 5.70, 5.71, 5.72, 5.73, 5.74, 5.75, 5.76, 5.77, 5.78, and 5.79 suffers from a recurrent vesicovaginal fistula after radiation for cancer of the uterus. The introitus is very narrow and fibrotic, making access to the vaginal cuff practically impossible. A lateral episiotomy at the 5 o’clock position will be done to obtain access to the fistula. A rotational inner thigh flap will be used to widen the vaginal canal and cover the fistula repair with a flap of well-vascularized skin and fatty tissue.
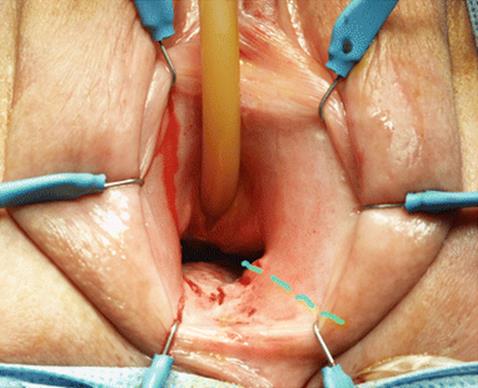
Fig. 5.69
Exposure of the vaginal area shows a very narrow, fibrotic introitus not allowing the passage of deep instruments or retractors. A line marks the site of the lateral episiotomy

Fig. 5.70
A lateral 5 o’clock episiotomy is performed by transecting the lateral vaginal wall, levator hiatus, and labial skin. The dissection is carried out toward the pararectal space, which is dissected free and reaches the vaginal cuff at the area of the fistula. The incisions have widened the vaginal canal, creating a superior incision at the 2 o’clock position and a second incision at the 7 o’clock position. The fistula will be repaired in multiple layers, and diluted indigo carmine solution will be used to irrigate the bladder to ensure the integrity of the closure. Green line shows the margins of the vagina after the incision
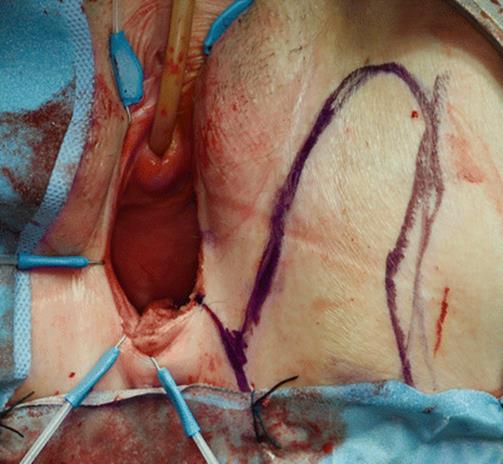
Fig. 5.71
An inverted U flap with a wide base will be created lateral to the labia, reaching the anterior pubic bone

Fig. 5.72
The inverted U flap is created deep to the adductor fascia medial and lateral. The flap is extended to the line of incision of the labia at the site of the episiotomy

Fig. 5.73
The tight flap has been mobilized and retracted inferiorly

Fig. 5.74
The base of the labia is grasped with Allis clamps, elevated, and detached from the lateral vaginal fascia and the underlying pubic bone and obturator fascia. The labial flap is mobilized lateral toward the left

Fig. 5.75
The flaps will be crossed over. The inner thigh flap is rotated medially toward the vaginal canal and the labial flap is rotated laterally toward the lateral thigh incision

Fig. 5.76
The inner thigh flap is transferred to the vaginal cuff to cover the area of the fistula repair. Preplaced sutures facilitate the advancement and suturing of the flap to the vaginal wall margins

Fig. 5.77
Interrupted delay absorbable sutures have completed the anastomosis of the flap to the vaginal incision at the 2 o’clock position

Fig. 5.78
(a) The anastomosis of the medial segment of the flap and the medial vaginal incision at the 7 o’clock position have been completed. The incision of the inner thigh is also completed, using interrupted delay absorbable sutures. The lateral aspect of the labia flap is easily approximated to the lateral donor site incision. (b) Postoperative picture 5 months after surgery, showing good take of the flap. The patient has good vaginal depth, the fistula was cured, and the patient was able to resume sexual relations

Fig. 5.79
Exposure of the vaginal wall showing a short, fibrotic vagina with a high vesicovaginal fistula
5.1.3.14 Repair of Recurrent Radiation Vesicovaginal Fistula: Use of a Myocutaneous Gracilis Flap
The patient in Figs. 5.80, 5.81, and 5.82 presented with a recurrent vesicovaginal fistula due to pelvic radiation. She was treated with the use of a myocutaneous gracilis flap, which is used less frequently in very selected cases as an alternative to an inner thigh flap.
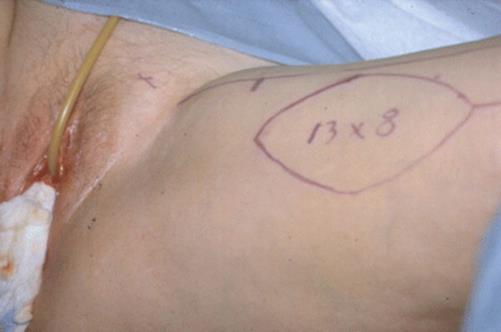
Fig. 5.80
The inner thigh and inner aspect of the leg are exposed. The size and location of the donor site are marked

Fig. 5.81
The skin, the subcutaneous tissue, and the detached gracilis muscle are mobilized free

Fig. 5.82
The myocutaneous gracilis flap is transferred through a tunnel under the thigh to the vaginal canal, where it is anastomosed to the margins of the excised vaginal wall after closure of the fistula
5.1.4 Intraoperative Complications
Bleeding can be a problem during the dissection of the vaginal flaps and should be controlled with fine absorbable sutures. Apical fistulae generally do not require ureteric catheterization, but this maneuver is recommended for fistulae close to the trigonal area because of the higher risk of ureteric injury. If there is doubt, cystoscopy after injection of indigo carmine and ureteric catheterization should be performed to ensure the integrity of the ureters. The strength of the first and second lines of closure is crucial for a successful outcome. Excessive dissection of the fistulous tract may turn a small opening in a large tract and compromise the quality of the repair, so we advocate not excising the fistulous tract, avoiding the thinning of the surgical margins, and providing a strong anchor to the line of suture. When in doubt, adjunctive procedures such as the fibro-fatty labial flap (Martius flap) or the other techniques described should be used.
5.1.5 Postoperative Care
The vagina is packed with an antibiotic-impregnated gauze, which may be removed after several hours. The suprapubic and urethral catheters are joined to a Y connector and left in place for 10 days. An oral cephalosporin or fluoroquinolone is continued until the catheters are discontinued, and cholinolytics are given to minimize bladder spasms. Prior to catheter removal, a voiding cystogram is performed to evaluate the integrity of the repair. Sexual relations may resume after 12 weeks.
Postoperatively, the patient receives antibiotics until the Foley catheter is removed and anticholinergics to avoid bladder spasms. Most of the procedures described above can be performed as outpatient surgery. The patient is discharged after surgery, with the only restriction being instructions to avoid strenuous exercise. The most important aspect of postoperative care is uninterrupted catheter drainage. The urethral catheter is removed on days 10–14 after surgery and a suprapubic cystogram is performed. If the outpatient cystogram documents vesical integrity, the suprapubic catheter is removed. Patients should abstain from sexual intercourse for 3 months.
5.1.6 Postoperative Complications
Immediate complications such as vaginal infection, bladder spasms, or vaginal bleeding should be treated aggressively to avoid fistula recurrence. Perioperative antibiotics are important to avoid vaginal wall infection and should be continued as required. Bladder spasms may lead to breakage of the repair and should be treated with cholinolytic agents, as well as local anesthetics if required. Secondary vaginal bleeding should be treated with bed rest and vaginal packing if necessary.
Vaginal stenosis and shortening may result from unnecessary excessive resection of the vaginal wall and may require secondary vaginoplasty. Unrecognized ureteric injury (leak or, more commonly, obstruction) may require percutaneous nephrostomy and a cooling-off period. Endoscopy procedures such as retrograde catheterization or transurethral ureteroscopy should be avoided in the immediate postoperative period; antegrade procedures are more acceptable.
The most important complication is recurrence of the fistula. The repair will succeed in more than 95 % of cases with adherence to the basic principles of wide mobilization and tension-free closure, with multiple non-crossing layers. If the fistula does recur, it can be repaired through the vagina again after a proper waiting period, but a flap of fibro-fatty labial tissue or peritoneum should be used.
5.2 Repair of Rectovaginal Fistula
Rectovaginal fistula may occur spontaneously (as in immune-suppressed patients), but most commonly they result from radiation, perirectal or intra-abdominal infections, or traumatic injury of the rectovaginal area (obstetric, instrumental, or external).
5.2.1 Indications
Corrective surgery is indicated in medically fit patients. Diverting colostomy is required in some cases, depending on the size and etiology of the fistula and whether the patient has a history of prior failures or inflammatory conditions such as ulcerative colitis. Colostomy may be avoided if the fistula is small, well defined, and distal.
5.2.2 Diagnosis
The diagnosis is obvious, as stool material is seen flowing into the vagina. Physical examination reveals the location, size, and number of fistulous tracts. It is important to determine the function and tone of the anal sphincter and the integrity of the rest of the rectal wall and distal colon. Rectosigmoidoscopy, pelvic CT scans, and radiological evaluation of the gastrointestinal tract should be performed when indicated.
5.2.3 Preoperative Considerations
The use of adjuvant measures like proper antibiotic cover, estrogen replacement, and complete bowel preparation are important. The decision to perform simultaneous or initial diverting colostomy is based on the extent of the fistula, its size, its etiology (radiation fistula requires colostomy), and the general condition of the patient.
Tissue flaps to cover the fistulous area with fresh vascularized tissue should be used routinely. The most common alternatives are fibro-fatty tissue of the labia (Martius flap), labial skin, inner thigh skin, or gracilis muscular flaps. Prior to preparation and draping, copious irrigation of the rectum is performed to clear any residual stool matter.
5.2.4 Surgical Technique
Figures 5.83, 5.84, 5.85, 5.86, 5.87, 5.88, 5.89, 5.90, 5.91, 5.92, 5.93, 5.94, 5.95, and 5.96 illustrate the surgical treatment of a patient with a small, distal rectovaginal fistula.

Fig. 5.83
Vaginal examination showing a small, distal rectovaginal fistula (arrow)

Fig. 5.84
A Foley catheter is inserted into the fistulous tract. The catheter will facilitate the dissection and isolation of the fistulous tract

Fig. 5.85
A U incision in the posterior vaginal wall is created with the distal tip in the area of the fistula. The flap is extended 5–8 cm toward the proximal vagina. A flap to the prerectal fascia (if present) will be developed and used later to reinforce the closure of the fistula. A flap of the vaginal wall is created distally to the fistula, reaching the posterior fourchette or distal vagina 3–4 cm from the fistula
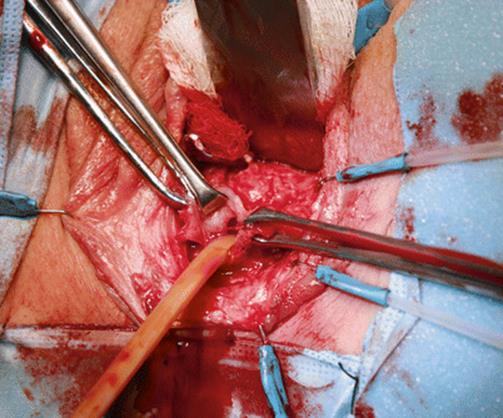
Fig. 5.86
The fistulous tract and the anterior rectal wall are exposed while the Foley catheter is in place
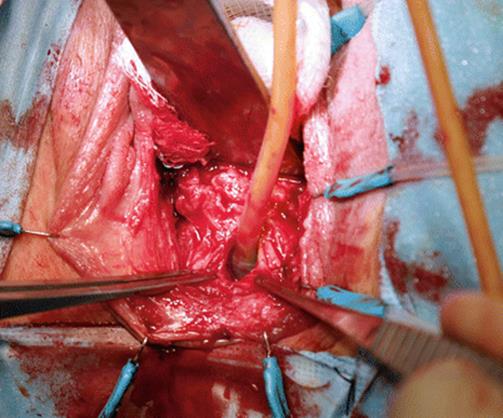
Fig. 5.87
The fistulous tract has been leaving a small ring of the fistulous tract. Wide exposure of the fistula is obtained for several centimeters around the fistula

Fig. 5.88
The fistulous tract is closed, incorporating the whole rectal wall and the margins of the fistulous tract

Fig. 5.89
The fistulous tract (arrow) is closed with two layers of delayed absorbable sutures. Irrigation of the rectum with a diluted iodine solution confirms that there is no extravasation

Fig. 5.90
The prerectal flap created at the beginning of the posterior flap creation is advanced distally to cover the area of the fistula repair

Fig. 5.91
A vertical incision is made over the left labia majora. The fibro-fatty (Martius) flap is isolated, preserving its inferior pedicle

Fig. 5.92
The Martius flap has been mobilized and the superior segment tied and cut anterior to the pubic bone

Fig. 5.93
A tunnel is made under the labia toward the anterior rectal wall

Fig. 5.94
The fibro-fatty tissue (Martius flap) is transferred over the anterior rectal wall in the vaginal canal. Interrupted absorbable sutures are used to fix the flap to the rectal wall and prevent displacement. The flaps should cover at least 3 cm around the closed fistulous tract
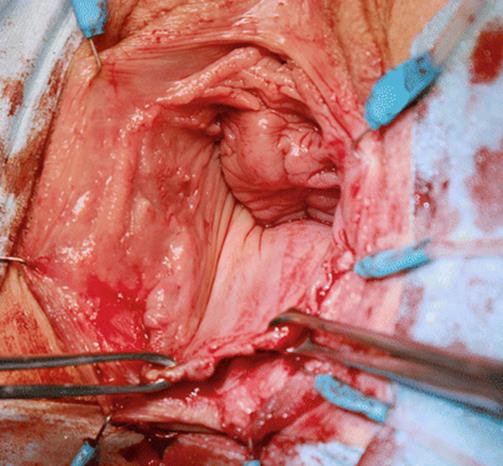
Fig. 5.95
The posterior vaginal wall flap is advanced distally so new tissue covers the area of the reconstruction

Fig. 5.96
Final view of the posterior vaginal wall. The posterior vaginal wall flap extends to the introitus
5.2.5 Intraoperative Complications
Complications are rare and common to other rectal surgery. An inability to bring together the edges of the fistula may be caused by poor quality of the tissues owing to radiation or infection. In these cases, the surgery should be performed with a concomitant diverting colostomy.
5.2.6 Postoperative Care
The vaginal packing is removed after a few hours. Regular diet can be resumed immediately. Broad-spectrum antibiotic coverage should be continued for at least 1 week. If a diverting colostomy is performed, a waiting period of 3–6 months is recommended before a takedown. Direct and radiological examination should be performed to confirm the healing of the fistula.
5.2.7 Postoperative Complications
Recurrent fistula requires re-exploration and a diverting colostomy, if it was not performed in the first surgery. Contributing to this complication is undue tension of the suture line or wound infection. Abscess formation requires incision and drainage. Vaginal or rectal stenosis is a rare complication.
5.3 Repair of Rectoperineal Fistula
Traumatic delivery, perineal surgery, or direct trauma to the vaginal area may cause a fistula from the rectum to the perineum. On occasion, a perirectal or perineal abscess may drain into the rectum and create a fistulous tract to the perineum.
5.3.1 Diagnosis
This condition is clinically manifested with the appearance of stool contents in the perineal or vaginal area. On physical examination, the open fistula is seen in the perineum or vagina. The rectal examination will help to characterize the size and location of the communication. A probe or catheter in the fistula will help to better define the fistulous tract. Assessment of anal function and sphincter tone should be documented preoperatively. Radiological or endoscopic examinations are usually not necessary; they are performed only to rule out conditions like cancer, ulcerative colitis, or Crohn’s disease.
5.3.2 Preoperative Considerations
A 48-h full bowel preparation should be performed prior to any surgery. Diverting colostomy is recommended in most cases but is not always necessary. Rotational flaps of fibro-fatty tissue from the labia or the gluteal or perineal area are usually performed to provide a thick, vascular layer of tissue interposition.
5.3.3 Surgical Technique
This section describes the surgical technique for three cases. First is a simpler case of rectoperineal fistula due to ulcerative colitis, in which a subcutaneous flap of fat from the perineum is used for the reconstruction (Figs. 5.97, 5.98, 5.99, 5.100, 5.101, 5.102, 5.103, 5.104, 5.105, and 5.106). The second case has stool incontinence due to poor sphincter function and a rectoperineal fistula requiring anal sphincter repair and repair of the fistula (Figs. 5.107, 5.108, 5.109, 5.110, 5.111, 5.112, 5.113, 5.114, 5.115, 5.116, 5.117, 5.118, and 5.119). The third, more complex case had a rectoperineal fistula, rectovaginal fistula, and anal sphincter incompetence due to traumatic delivery. After multiple failed repairs, this patient required repair of the rectovaginal fistula, reconstruction of the anal sphincter (internal and external), and repair of the rectoperineal fistula (Figs. 5.120, 5.121, 5.122, 5.123, 5.124, and 5.125).

Fig. 5.97
Exposure of the perineum is obtained. A small Foley catheter is inserted through the fistula into the rectum and placed under slight traction
5.3.3.1 Repair of Recurrent Rectoperineal Fistula with Interposition of Perineal Fat
Prior to preparation and draping, the rectum is irrigated with a copious amount of antibiotic solution. Broad-spectrum antibiotics are given intravenously. A Foley catheter is inserted and a ring retractor with hooks is used to facilitate the surgical exposure. Figures 5.98, 5.99, 5.100, 5.101, 5.102, 5.103, 5.104, and 5.105 show the technique used.

Fig. 5.98
A circular incision is made in the perineum around the fistula, and lateral dissection is done at least 3–4 cm from the fistula

Fig. 5.99
After the dissection, the trans-anal fistulous tract is isolated

Fig. 5.100
Under finger control in the rectum, multiple figure-of-eight delayed absorbable sutures are used to approximate the fistulous tract

Fig. 5.101
A second layer of mattress sutures 5 mm from the fistula is applied to cover the first layer of closure. Rectal irrigation with a diluted iodine solution will confirm that there is no leakage from the fistula

Fig. 5.102
On the left side, the skin is undermined and the perineal fat is mobilized. The dissection will often include the incision of the lower margin of the fat flap to allow mobility and rotation. The flap must reach the contralateral side of the skin incision and cover the rectal repair without tension

Fig. 5.103
The free margins of the perineal fat flap are transferred to the contralateral side and sutured medial, superior, and inferior to the perineal tissues, covering the area of the fistula repair

Fig. 5.104
A strip of the superficial skin of the left lateral incision is excised, exposing the subcutaneous tissue
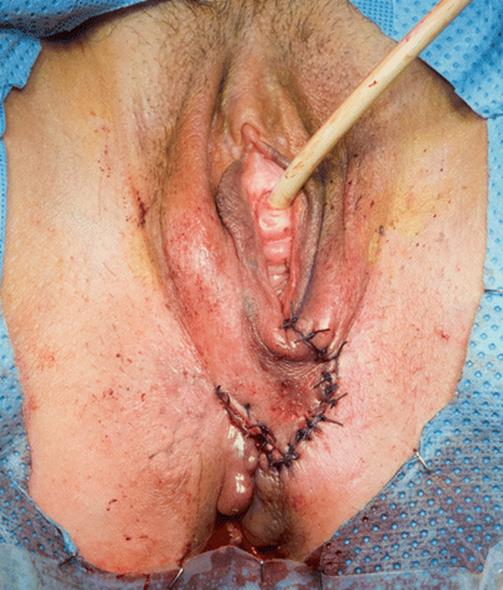
Fig. 5.105
The lower segment of the labia majora (with its subcutaneous fat) and the right skin incision are transferred to the left and sutured to the left skin margin. This segment of skin and subcutaneous tissue covers the reconstructed area, preventing overlapping lines of sutures

Fig. 5.106
Perineal examination of the patient who has a perineal fistula (arrow). A finger in the rectum is seen through one of the multiple fistulous tracts
5.3.3.2 Repair of Rectoperineal and Rectovaginal Fistula with Concomitant Anal Sphincter Reconstruction
The patient in Figs. 5.106, 5.107, 5.108, 5.109, 5.110, 5.111, 5.112, 5.113, 5.114, 5.115, 5.116, 5.117, and 5.118 had a traumatic delivery resulting in an extensive rectal and perineal tear. Multiple repairs have failed, and the patient remains with a perineal rectal fistula, rectovaginal fistula, and an open, incompetent anal sphincter. Most stools come from the vagina and perineum.

Fig. 5.107
A vertical incision is made from the distal vagina to the perineum. The perineal and vaginal skin is excised, exposing the fistulous area

Fig. 5.108
After excision of the perineal and vaginal scar, the distal rectum and the anal canal are fully exposed. The anal sphincter was incompetent and damaged, and the musculature retracted laterally

Fig. 5.109
The rectal wall and the mucosa of the anal canal are closed with running and interrupted #3-0 delay absorbable sutures

Fig. 5.110
With a finger in the rectum, the complete closure of the recto-anal mucosa and internal sphincter can be seen

Fig. 5.111
The external anal sphincter and perineal musculature are approximated with figure-of-eight delay absorbable sutures to rebuild the anal canal and the perineal musculature
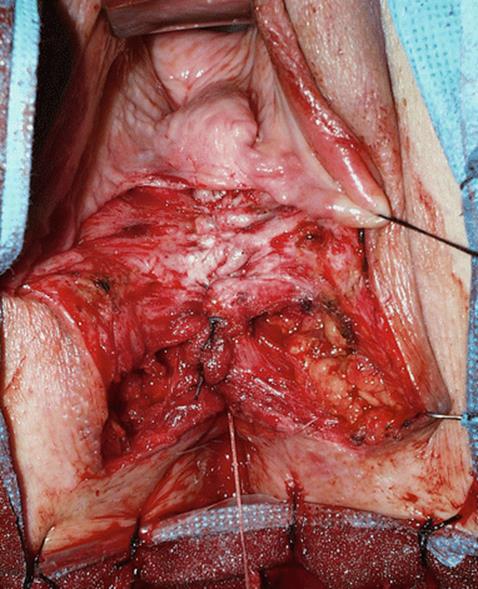
Fig. 5.112
Exposure of the posterior vaginal wall after the reconstruction of the anterior rectal wall and the anal sphincter

Fig. 5.113
The posterior vaginal wall is closed with running #2-0 delay absorbable sutures

Fig. 5.114
A rectangle of superficial skin incision is made on the right margin of the perineal incision

Fig. 5.115
The rectangle of skin is excised to expose the subcutaneous fatty tissue of the perineum on the right. The skin lateral to the right margin of the incision is mobilized

Fig. 5.116
An incision of subcutaneous fat is made at the inferior and lateral segments (arrows) to help mobilize a large flap of subcutaneous fat

Fig. 5.117
The right subcutaneous flap of fat is rotated, transferred toward the left side of the perineum, and anchored with delayed absorbable sutures. The flap completely covers the perineal reconstruction

Fig. 5.118
The skin closure is done with multiple delayed absorbable sutures. The line of suture is asymmetric and crosses toward the right, where the rectangular skin flap was excised
5.3.3.3 Repair of Rectoperineal Fistula and Anal Sphincter Reconstruction
The patient in Figs. 5.119, 5.120, 5.121, 5.122, 5.123, 5.124, and 5.125 had a traumatic delivery resulting in a low-output perineal fistula and severe stool incontinence due to anterior anal sphincter damage.

Fig. 5.119
With exposure of the perineum, a small probe inserted into the fistula can be seen coming out from the rectum

Fig. 5.120
An inverted U incision is made in the perineum, extending to the ischial tuberosities and distally toward the fistulous tract

Fig. 5.121
The posterior perineum is exposed by entering the ischial fossa and exposing the levator (puborectalis and pubococcygeus). The perineal fistula will be closed in two layers of delayed absorbable sutures

Fig. 5.122
Multiple figure-of-eight #0 delayed absorbable sutures are used to approximate the external anal sphincter musculature to the midline. The sutures are placed with a finger in the rectum to prevent mucosal penetration of the sutures. The sutures should avoid the inverted U flap of the perineum in order to allow for maximal mobility of the flap

Fig. 5.123
The sutures are tied and the reconstructed perineum is seen

Fig. 5.124
The perineal skin flap is mobile and will be advanced distally to cover the area of reconstruction

Fig. 5.125
The perineal flap is advanced distally, covering with new skin the fistula and the reconstructed anal sphincter
5.3.4 Intraoperative Complications
Intraoperative complications are rare. They include bleeding or an inability to close the rectal or perineal wounds. Wide tissue mobilization may be required in these cases. A temporary diversion of stool is indicated in recurrent cases and in patients with poor-quality tissues, a history of radiation, or other inflammatory conditions.
5.3.5 Postoperative Care
The packing is removed the next day after surgery. The Foley catheter is removed as soon as the patient is ambulating freely. Broad-spectrum antibiotics should be used in the perioperative area. If a diverting colostomy is performed, it should be closed 3–6 months after the closure of the fistula is ensured.
5.3.6 Postoperative Complications
Infection of the perineal and vaginal wound or tension of the suture lines may lead to recurrent fistula formation. Stool incontinence may be the result of surgery, or it may have existed prior to surgery but could not be demonstrated at that time. Wound infection and perineal abscess may require incision and drainage.
Suggested Reading
Arrowsmith S, Hamlin EC, Wall LL. Obstructed labor injury complex: obstetric fistula formation the multifaceted morbidity of maternal birth trauma in the developing world. Obstet Gynecol Surv. 1996;51(9):568–74.CrossRefPubMed
Blaivas JG, Heritz DM, Romanzi LJ. Early versus late repair of vesicovaginal fistulae: vaginal abdominal approaches. J Urol. 1995;153(4):1110–2.CrossRefPubMed
Blandy JP, Badenoch DF, Fowler CG, Jenkins BJ, Thomas NW. Early repair of iatrogenic injury to the ureter or bladder after gynecological surgery. J Urol. 1991;146(3):761–5.PubMed
Cass AS, Odl M. Ureteroarterial fistula: case report review of literature. J Urol. 1990;143(3):582–3.PubMed
Eilber KS, Kavaler E, Rodríguez LV, Rosenblum N, Raz S. Ten-year experience with transvaginal vesicovaginal fistula repair using tissue interposition. J Urol. 2003;169(3):1033–6.CrossRefPubMed
Falk HC, Tancer ML. Vesicovaginal fistula; an historical survey. Obstet Gynecol. 1954;3(3):337–41.PubMed
Gerber GS, Schoenberg HW. Female urinary tract fistulae. J Urol. 1993;149(2):229–36.PubMed
Kim J, Smith A, Raz S. Urinary fistulae: what does the evidence say? Curr Bladder Dysfunct Rep. 2008;3:208–13.CrossRef
Lee D, Dillon BE, Zimmern PE. Long-term morbidity of Martius labial fat pad graft in vaginal reconstruction surgery. Urology. 2013;82(6):1261–6.CrossRefPubMed
Lee RA, Symmonds RE, Williams TJ. Current status of genitourinary fistula. Obstet Gynecol. 1988;72(3 Pt. 1):313–9.PubMed
Margolis T, Mercer LJ. Vesicovaginal fistula. Obstet Gynecol Surv. 1994;49(12):840–7.CrossRefPubMed
McConnell DB, Sasaki TM, Vetto RM. Experience with colovesical fistula. Am J Surg. 1980;140(1):80–4.CrossRefPubMed
O’Conor Jr VJ, Sokol JK, Bulkley GJ, Nanninga JB. Suprapubic closure of vesicovaginal fistula. J Urol. 1973;109(1):51–4.PubMed
Tenggardjaja CF, Goldman HB. Advances in minimally invasive repair of vesicovaginal fistulas. Curr Urol Rep. 2013;14(3):253–61.CrossRefPubMed