Peter D. Peng
Timothy M. Pawlik
Presentation
A 62-year-old man is referred to your office 2 years after a right hemicolectomy for T3N1M0 colon cancer detected on screening colonoscopy. Following surgery, the patient received adjuvant chemotherapy with 5-fluorouracil, leucovorin, and oxaliplatin (FOLFOX). Now, 2 years later, he presents with an elevated carcinoembryonic antigen (CEA) of 80 ng/mL and a CT scan demonstrating four new masses in his right hemiliver.
Differential Diagnosis
The differential diagnosis of a liver mass includes both benign (cyst, hemangioma, focal nodular hyperplasia, hepatic adenoma) and malignant (metastatic disease, primary hepatocellular carcinoma, intrahepatic cholangiocarcinoma, gallbladder cancer) disease processes. However, in a patient with a history of colon cancer and a rising CEA, metastatic colon cancer is the most likely diagnosis. Approximately one-half of patients with a history of colon cancer will either present with synchronous hepatic metastasis or develop them during the course of their disease. In general, about 15% to 25% of patients will present with synchronous disease, while 20% to 25% will develop metachronous colorectal liver metastasis. In about 30% to 50% of patients with colorectal liver metastasis, the liver will be the only site of metastatic disease, and these patients will be candidates for local surgical therapy.
Workup
Patients being considered for resection of colorectal liver metastasis should have preoperative cross-sectional imaging. A helical triphasic CT scan is the most commonly employed imaging modality. On CT imaging, metastases from colorectal cancer are visualized best on the venous phase and appear as hypointense, low-attenuating masses within the liver if the scan is obtained when the contrast is in the portal circulation (Figure 1). With multidetector helical CT, the sensitivity of identifying liver metastasis is about 80% to 90%. MRI is another imaging modality that can be utilized. On contrast-enhanced MRI with agents such as gadolinium, colorectal liver metastases are best seen on the T2-weighted images and give MRI an 80% to 90% sensitivity in detecting colorectal liver metastasis. Chest imaging should be considered before resection of colorectal liver metastasis to rule out pulmonary metastasis. The use of positron emission tomography (PET), either alone or in combination with CT (PET/CT), may also be useful and should, in general, be obtained prior to surgery to assess the extent of metastatic disease.
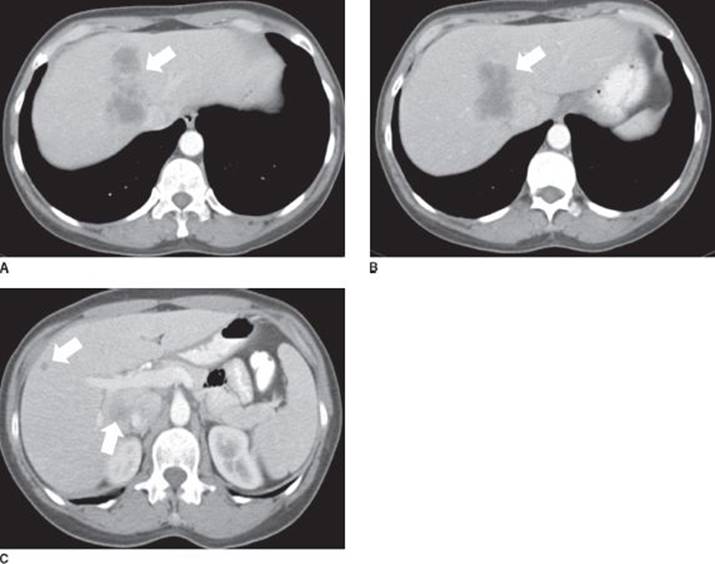
FIGURE 1 • Select CT axial images of colorectal liver metastases (arrows), venous phase. Note that the metastases are hypointense and low attenuating. The larger mass at the dome of the liver abuts the right and the middle hepatic vein.
Some studies have reported that PET may change clinical management in up to 20% of patients with colorectal liver metastasis being considered for surgical resection. Identification of extrahepatic metastases is a relative contraindication to hepatic metastectomy, especially when it is non-pulmonary in location and multi-focal in nature.
Resectable lesions should not be routinely biopsied. In most cases, biopsy is unnecessary to make the diagnosis in the appropriate clinical context, associated with elevation of serum CEA and pathognomonic radiologic features on cross-sectional imaging/PET (Figure 2). Although the risk is small, biopsy can result in tumor dissemination and therefore should be reserved only for those situations when there is diagnostic uncertainty or when the biopsy will provide needed data to allow for the administration of chemotherapy or other nonresectional treatment options. Finally, depending on the situation and timing of the last colonoscopy, a repeat colonoscopy should be considered in the patient with a history of colon cancer.
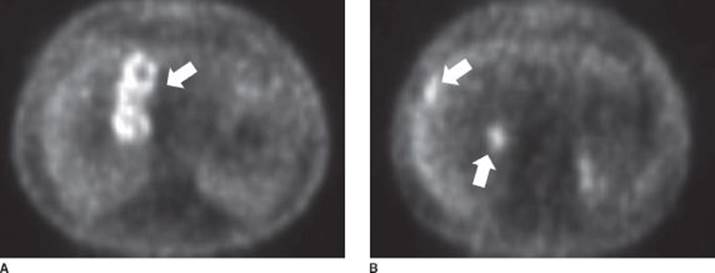
FIGURE 2 • PET scan demonstrating multiple FDG-avid lesions (arrows) in the liver that correlate with the metastases identified on cross-sectional CT imaging.
Diagnosis and Treatment
Surgical therapy for liver metastasis remains the only therapy that promises a potential cure. Long-term survival after surgery for colorectal liver metastasis has been demonstrated in numerous studies, with the overall 5-year survival reported to range from 35% to 58%. In addition to resection, ablative techniques, such as radiofrequency or microwave ablation, can also be utilized to destroy the tumor via the application of heat. In general, tumor ablation should not be viewed as a replacement for resection, but more as a supplement or an extension of localized therapy for those patients with extensive disease or those patients who otherwise are not resection candidates. Ablation should not be used for lesions near the hilum due to the risk of bile duct injury and stricture.
In the past, tumor number ≥4, bilobar disease, metastasis within 1 cm of the planned transection margin, or extrahepatic disease were all considered strong relative contraindications to liver-directed surgery. However, with improvements in operative technique and systemic chemotherapy, more recent data have demonstrated that these clinicopathologic factors should not be considered absolute contraindications to surgery. Rather, decisions regarding resectability have shifted in focus away from criteria based on the metastatic disease (tumor size, number, margin width, etc.). Decisions regarding resectability now are more based on whether all the metastatic disease can be removed (R0 resection) while preserving enough volume of liver after the resection to avoid liver insufficiency/failure. In general, following liver resection at least two contiguous hepatic segments with adequate inflow, outflow, and biliary drainage need to be preserved with a functional liver remnant (FLR) volume of at least 20%.
Given this new therapeutic approach, there has been a shift to try to expand the pool of patients who might be candidates for surgical therapy. Over the past two decades, chemotherapeutic and biologic agents have been found that have significant increased activity against colon cancer. The improved efficacy of chemotherapy agents has not only increased patient survival in the noncurative setting, but has also allowed a subset of previously unresectable patients to undergo potentially curative liver surgery after tumor downsizing. The use of chemotherapy in the preoperative setting for patients with resectable colorectal liver metastasis is more controversial. The rationale for using neoadjuvant chemotherapy is supported, in part, by the better prognosis obtained with preoperative chemotherapy and surgery among patients with multifocal colorectal liver metastasis compared with immediate surgery alone. The decision to give chemotherapy before or after surgery needs, however, to be individualized and be based on the specific clinical situation (Figure 3).
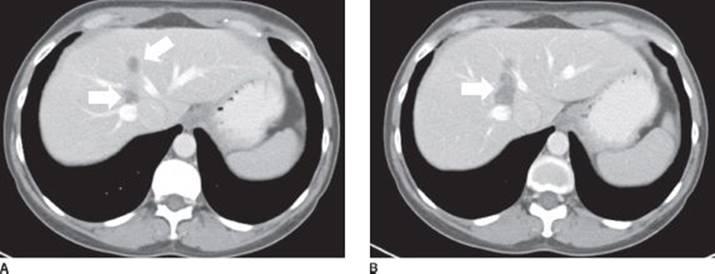
FIGURE 3 • Repeat CT axial imaging following treatment with neoadjuvant treatment with four cycles of FOLFOX plus bevacizumab chemotherapy. Note the cytoreduction in the size of the liver metastases (arrows).
In a small subset of patients with extensive disease, portal vein embolization (PVE) or a two-stage hepatectomy may be warranted. Some patients with extensive intrahepatic disease will require an extended hepatectomy (right hepatectomy plus segment 4) to extirpate all disease. To avoid operating on patients with low-volume FLR, PVE can be considered to induce hypertrophy. In PVE, the portal vein to the tumor-bearing side of the liver is embolized inducing contralateral liver hypertrophy. The selective use of PVE may enable the performance of an extended hepatectomy in a subset of patients who otherwise would not be considered candidates for surgery. In those patients with multiple metastases with a bilobar distribution, a sequential—or two-stage—hepatectomy may be more appropriate. For example, the left hemiliver can be cleared of disease during the first operation. A right portal vein ligation can be performed intraoperatively or a right PVE can be performed postoperatively. The remnant left liver is then allowed to hypertrophy and a right or an extended right hepatectomy is performed as the second stage (Figure 4).
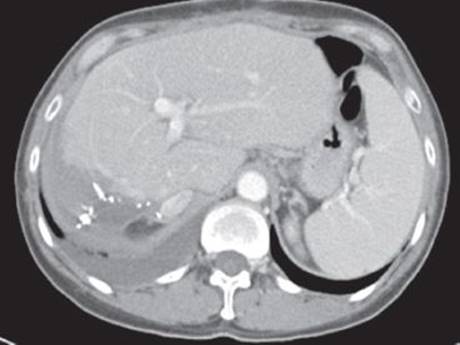
FIGURE 4 • Postoperative CT axial imaging. Note the hypertrophy of the remnant left hemiliver, especially segments 2 and 3.
In general, the goals of surgical therapy are to extirpate all disease (R0) with the use of either resection alone or, if necessary, in combination with ablation. While not supported with robust level one data, adjuvant chemotherapy is usually employed given that the majority of patients with hepatic colorectal metastasis will experience a recurrence after surgery.
Surgical Approach
Liver resection can be performed using both a traditional open or a laparoscopic approach (see Table 1). While there are increasing data emerging about the use of laparoscopic liver resection to treat malignancies, most major liver resections are still performed using an open technique. For an open approach, either a midline or a right subcostal incision can be used. Upon entering the abdomen, the ligamentum teres and falciform ligament are taken down and a full evaluation of the liver is performed visually, by palpation, and with intraoperative ultrasound (IOUS). IOUS is an important tool for accurately staging liver tumors, assessing the true extent of disease, and making intraoperative decisions. IOUS is usually performed using a midfrequency (5.0 and 7.5 MHz) transducer probe, with the 7.5-MHz probe able to penetrate 6 to 8 cm. IOUS needs to always be performed in a systematic manner in both the transverse and the sagittal planes to avoid missing any small occult lesions. The hepatic vasculature should also be examined with IOUS to identify possible anatomic variants and to aid in the planning of the resection.
TABLE 1. Key Technical Steps and Potential Pitfalls to Open Major Hepatectomy

Inflow control through an intermittent Pringle maneuver of the porta hepatis can be utilized to help decrease blood loss. In the case of a formal hemihepatectomy, the hepatic inflow pedicle is often dissected out at the level of the hilum. With an extrahepatic pedicle dissection, the ipsilateral hepatic artery and portal vein are divided outside the substance of the liver after lowering the hilar plate. Following division of the hepatic artery and the portal vein, the liver usually demarcates along the principal plane of the liver. Depending on the location of the lesion (i.e., for those lesions not near the hilum), an intrahepatic technique can alternatively be utilized to obtain pedicle control. The intrahepatic technique has the potential advantages of being faster and more unlikely to cause injury to the biliary or vascular structures to the contralateral liver.
The extent of liver mobilization is dictated by the extent and type of procedure to be performed. The coronary ligament at the top of the liver is taken down with cautery to expose the suprahepatic vena cava and hepatic veins. In the case of a right hemihepatectomy, the right hemiliver is mobilized by taking down the right triangular ligament, exposing the bare area of the liver along the right diaphragm. The right hemiliver is further mobilized by taking down the retroperitoneal attachments along the inferior aspect of the right liver and the attachments along the right adrenal gland. With the right liver rotated medially and superiorly, the retrohepatic vena cava is exposed and several small venous branches traveling directly from the cava to the liver are ligated and divided. The hepatocaval ligament (Makuuchi’s ligament) is dissected free and divided, often with an endovascular stapler. The right hepatic vein is circumferentially dissected free and can be divided between clamps or with an endovascular stapling device.
For a left hemihepatectomy, the left hemiliver is mobilized by taking down the left triangular ligament. The ligamentum venosum is dissected free and divided to better expose the left hepatic vein. For a left hepatectomy, the left hepatic vein is often not dissected out at this point, but instead is divided later within the substance of the liver during the hepatic parenchymal transection. The specifics of the dissection are, however, dictated by the size and location of the lesion relative to the hepatic vein.
A number of techniques can be utilized for parenchymal transection. Some of these techniques include, but are not limited to, crush-clamp, bipolar or monopolar cautery, radiofrequency ablation (Habib device), harmonic scalpel, as well as ultrasonic aspirator or hydrojet devices. While different surgeons advocate different transection techniques, no technique has clearly been shown to be superior to another. As such, the most important factor in choosing a method of liver transection should be the surgeon’s comfort and familiarity with the technique so as to ensure minimal blood loss and an R0 surgical margin. In general, major liver surgery should be performed in the setting of a low central venous pressure (CVP) (<5 mm Hg) before and during parenchymal transection. A low CVP helps ensure less back-bleeding from the cava/hepatic veins and, therefore, less blood loss. Larger bleeding structures are controlled surgically with sutures. Hemostasis of the transected liver margin may also require topical hemostatic agents such as methylcellulose, collagen sponges, thrombin sealants, and argon beam coagulation. The liver margin should then be carefully inspected for bile leakage, which can be oversewn.
Intraoperative pathologic confirmation of an R0 margin is critical after removal of the specimen. While attaining an R0-negative margin is important, the width of the negative margin has not been demonstrated to correlate with recurrence rates.
Routine placement of closed suction drains is unnecessary following major liver resections. Placement of close suction drain should, however, be placed in select situations (e.g., diaphragm or bile duct resection/reconstruction performed in conjunction with liver resection, etc.).
Special Intraoperative Considerations
The surgeon needs to be aware of aberrant arterial anatomy. Hepatic artery variants should be identified prior to the initiation of hepatic resection. The lesser omentum should be examined to identify a replaced/accessory left hepatic artery that would normally course through the middle of the lesser omentum as it travels into the base of the umbilical fissure. In contrast, a replaced/accessory right hepatic artery originating from the superior mesenteric artery usually courses posterior and lateral to the common bile duct.
Not infrequently, IOUS may identify disease not recognized on preoperative imaging. New findings may necessitate a change in the operative plan, including either a revision in the type of planned hepatic resection or the addition of ablation. When unsuspected extrahepatic disease is encountered, in general, hepatic resection is not warranted. Patients with unsuspected peritoneal disease or gross hilar adenopathy probably do not derive long-term survival benefit from hepatic resection. Rather, this subset of patients is probably best treated with systemic chemotherapy and should only be considered for liver-directed surgery after restaging and careful multidisciplinary consideration.
Postoperative Management
Following major hepatic resection, most patients are monitored in the intensive or intermediate care unit overnight. During this time, standard crystalloid resuscitation and postoperative care is instituted. Electrolytes, liver function tests, hemoglobin, and prothrombin (PT) are checked after the operation and then daily. Hypophosphatemia is common after a major hepatic resection; phosphate levels should therefore be checked and repeated as necessary. Following an extended hepatic resection (removal of 70% to 80%), liver insufficiency/failure can sometimes occur. In these circumstances, the PT may be elevated and is treated with fresh frozen plasma when the PT is longer than 17 seconds. In most circumstances, a regular diet can be started on postoperative day 2 or 3, with an anticipated discharge on day 4 or 5.
Although uncommon, bile leaks can be a potential complication of liver resection. Usually, patients have a fever and an elevated bilirubin level in the setting of a normal alkaline phosphatase. A CT scan should be obtained and can easily identify a biloma. Intra-abdominal collections can usually be adequately drained using percutaneous techniques. Most bilomas will subsequently resolve following drainage with expectant management.
Case Conclusion
The patient received four cycles of FOLFOX plus bevacizumab with a measureable decrease in the size of the intrahepatic metastases. The patient underwent an extended right hemihepatectomy with complete extirpation of all intrahepatic disease. Postoperatively, the patient received adjuvant chemotherapy and remains disease free.
TAKE HOME POINTS
· Resection of solitary colorectal hepatic metastasis is associated with approximately 50% 5-year survival.
· Resectability is now defined as the ability to achieve an R0 resection while preserving two or more contiguous segments with vascular inflow, outflow, biliary drainage, as well as an adequate liver volume (~20%).
· Chemotherapy for colorectal liver metastasis can result in response rates >50%; chemotherapy should often be integrated into the overall therapeutic plan (i.e., neoadjuvant, conversion, or adjuvant depending on the situation).
· Resection resulting in R0-negative margins is the most important operative goal.
SUGGESTED READINGS
Abdalla EK, Adam R, et al. Improving resectability of hepatic colorectal metastases: expert consensus statement. Ann Surg Oncol. 2006;13(10):1271–1280.
Charnsangavej C, Clary B, et al. Selection of patients for resection of hepatic colorectal metastases: expert consensus statement. Ann Surg Oncol. 2006;13(10):1261–1268.
Nordlinger B, Sorbye H, et al. Perioperative chemotherapy with FOLFOX4 and surgery versus surgery alone for resectable liver metastases from colorectal cancer (EORTC Intergroup trial 40983): a randomised controlled trial. Lancet. 2008;371(9617):1007–1016.
Pawlik TM, Choti MA. Surgical therapy for colorectal metastases to the liver. J Gastrointest Surg. 2007;11(8):1057–1077.
Pawlik TM, Schulick RD, et al. Expanding criteria for resectability of colorectal liver metastases. Oncologist. 2008;13(1):51–64.
Poston GJ, Figueras J, et al. Urgent need for a new staging system in advanced colorectal cancer. J Clin Oncol. 2008;26(29):4828–4833.