Haggi Mazeh
Rebecca S. Sippel
Presentation
A 42-year-old female, without any previous medical or surgical history, presents for routine physical examination. Her vital signs are within normal limits. On neck examination, she is noted to have a palpable nodule in the front of her neck. The nodule is located two finger breadth inferior to the cricoid cartilage and 1 cm to the right of the midline. The nodule measures about 2 cm in its greatest diameter; it is firm, mobile, and non-tender. When the patient swallows, the nodule moves up and down with the thyroid cartilage. There are no palpable nodules on the left side of the neck, nor is there cervical of supraclavicular lymphadenopathy. The remaining physical examination is normal.
Differential Diagnosis
The description of the nodule suggests that it is located within the thyroid gland. Thyroid nodules may be of benign nature such as colloid-containing cysts, thyroid adenoma, hyperplastic nodules, or thyroiditis. Thyroid nodules may harbor malignancy including papillary, follicular, medullary, Hürthle cell, or anaplastic thyroid cancer. In rare cases, thyroid nodules may represent lymphoma, squamous cell carcinoma, or metastasis of other origin. The differential diagnosis of other neck masses is broad and is beyond the scope of this chapter.
Workup
At this point, it is important to identify whether the patient has any risk factors for malignancy. The two most important risk factors for thyroid malignancy are a history of neck radiation and a family history of thyroid cancer or other endocrine tumors. Specific attention must be paid to symptoms associated with local compression or invasion such as hoarseness, cough, dysphagia, or airway compressive symptoms. Rapid growth and new onset of hoarseness raise the suspicion for malignancy.
Thyroid function testing with a thyroid-stimulating hormone (TSH) level is recommended. If the TSH is abnormal, additional testing of thyroid function is warranted. If the TSH is elevated, serum concentrations of thyroperoxidase antibody should be checked. If the TSH is suppressed, a thyroid scintigraphy scan should be obtained to distinguish between Graves’ disease and a toxic nodule. Currently, routine calcitonin testing is not recommended.
Ultrasound (U/S) is the imaging of choice for a newly diagnosed thyroid nodule. Ultrasound may assess nodule size, location, and other concomitant thyroid pathologies as well as the cervical lymph nodes. Ultrasonic features suspicious for malignancy include hypoechogenicity, microcalcification, irregular margins, chaotic vascular patterns, as well as extracapsular invasion and lymph node involvement. Figure 1 demonstrates the appearance of a malignant nodule on U/S.
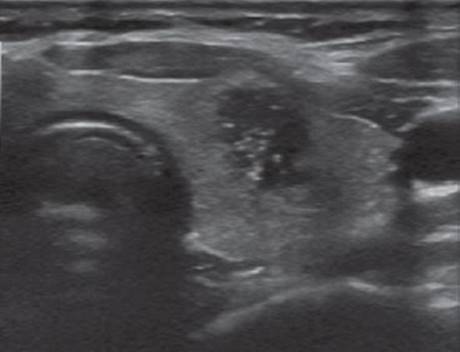
FIGURE 1 • A malignant thyroid nodule. The nodule appears heterogeneous, cystic, hypoechoic, irregular margins, and microcalcifications are present.
Ultrasound-guided fine needle aspiration biopsy (FNAB) is the most important tool for evaluation of thyroid nodules. Possible FNAB results are nondiagnostic, benign, atypia or follicular lesion of undetermined significance, follicular neoplasm, suspicious for malignancy, and malignant (see Figure 2).

FIGURE 2 • An algorithm for the evaluation of thyroid nodules. The six possible of FNAB results are given with their associated risk of malignancy and the appropriate management. TSH, thyroid-stimulating hormone; FNAB, fine needle aspiration.
The patient’s U/S demonstrated a 3.2-cm right thyroid lobe nodule. The nodule was hypoechoic and complex with some microcalcifications. In this patient, FNAB identified a follicular neoplasm.
Diagnosis and Treatment
Although the prevalence of thyroid nodules on ultrasound may exceed 50%, palpable thyroid nodules can be detected in 10% of women and 2% of men. Fortunately, most nodules are of benign nature and only 5% harbor malignancy. In the last two decades, FNAB of thyroid nodules has become the “gold standard” of thyroid nodule workup. In an attempt to develop uniform terminology of FNAB reports, six categories were defined as mentioned above.
Inadequate or nondiagnostic aspirates constitute 4% to 16% of FNAB results and should be reaspirated. If the repeated aspiration is not diagnostic, surgical excision is recommended. Benign results on FNAB are the most common finding (up to 70%) and may be treated expectantly. A follow up US should be obtained in 6 to 12 months to evaluate for interval growth. Malignant FNAB results should be treated with total thyroidectomy. Follicular neoplasms (FN) are identified in 15% to 20% of all FNAB and about 15% to 30% prove to be malignant. FNAB is unable to distinguish benign from malignant FN; hence, surgical removal is required. Figure 2 provides an algorithm for the evaluation of thyroid nodules.
Surgical Approach
The surgical approach to a patient with a follicular neoplasm result on FNAB requires an educated decision by the patient. The appropriate surgical approach for a FN is a diagnostic lobectomy. If the final pathology reveals malignancy, then a completion thyroidectomy should be performed. Frozen section evaluation is usually not informative and is unnecessary unless there is a suspicion for papillary thyroid cancer. The advantages of thyroid lobectomy include avoiding possible injury to the contralateral recurrent laryngeal nerve (RLN) and parathyroid glands as well as avoiding the need for lifelong thyroid hormone replacement in the majority of patients. A major disadvantage of this approach is it involves the need for completion thyroidectomy if malignancy is identified on final pathology. This second procedure involves another admission and anesthetic. Patients with a history of radiation therapy to the neck, a positive family history for thyroid cancer, multinodular goiter, and those already on thyroid hormone replacement therapy may be best served with total thyroidectomy as the index procedure.
The patient in our scenario decided to undergo total thyroidectomy (Table 1).
TABLE 1. Key Technical Steps and Potential Pitfalls of Thyroidectomy

The procedure is usually performed under general anesthesia. The patient is positioned in a beach chair position and the neck is extended. A horizontal neck (Kocher) incision is performed and should be located just inferior to the cricoid cartilage, ideally in a neck crease. The platysma muscle is divided and subplatysmal flaps are elevated. The strap muscles are separated at the midline to offer access to the thyroid gland. At this point, it is preferable to begin the dissection on the side with the suspected lesion or tumor. It is important to dissect and retract the strap muscles off the thyroid gland in order to be in the correct plane. The middle thyroid vein should be divided. The upper pole of the thyroid is divided as close as possible to the thyroid gland in order to avoid injury to the superior laryngeal nerve and the upper parathyroid gland. Once the upper pole is divided, the thyroid lobe may be retracted medially and upward to assist further dissection. At this point, it is crucial to identify the recurrent laryngeal nerve (RLN) in the tracheoesophageal groove along its course to the cricothyroid membrane.
Parathyroid glands should be preserved with their native blood supply whenever possible. During the dissection, vessels can be divided with cautery, harmonic scalpel, clips, or ties according to surgeon preference. Once the lobe is mobilized, the isthmus is dissected off the trachea with cautery and the contralateral lobe is resected in a similar fashion. After hemostasis is assured, the strap muscles are reapproximated at the midline. The platysma muscle is sutured and the skin closed.
Special Intraoperative Considerations
· In cases of suspected malignancy or proven malignancy, special attention must be paid to the lymph nodes in the central compartment. Lymph nodes that are grossly involved should be resected and in such cases formal central lymph node dissection should be performed.
· Failure to identify and resect a pyramidal lobe can be the source of a significant thyroid remnant with radioactive iodine treatment. The pyramidal lobe may be elongated reaching above the thyroid cartilage. Special care must be taken to correctly identify and resect it as part of the thyroid gland.
· In order to identify and preserve the parathyroid glands, it is important to understand their anatomic location, especially in relationship to the recurrent laryngeal nerve. The upper parathyroid gland is located posterior and lateral to the RLN and the lower glands are located anterior and medial to the RLN. In cases when parathyroid glands are inadvertently resected, they should be autotransplanted in the sternocleidomastoid muscle.
· Identification of the RLN during thyroidectomy is essential to avoid injury. After dividing the upper pole of the thyroid gland, it is important to avoid further dissection inferiorly prior to the nerve identification because most nerve injuries occur close to the nerve entrance to the trachea. In rare cases, the nerve is entrapped within tumor tissue, and in such cases, the nerve should be sacrificed. Nerve-monitoring devices have a role in confirming the nerve is intact, especially in complicated cases.
· The role of intraoperative frozen section is controversial. Some surgeons use it to assist in deciding on the extent of surgery for lesions with no definite malignancy on FNAB. Frozen section is most helpful in cases that have an FNAB of suspicious papillary thyroid cancer. Frozen section is rarely able to distinguish follicular adenoma from follicular carcinoma, so it is probably not useful in most cases of FN.
Postoperative Management
Thyroid surgery may be performed in an outpatient setting or with a short admission (24 hours). Patients should be monitored for several hours after surgery to evaluate for the development of neck hematoma that may require emergent evacuation. Symptomatic transient postoperative hypocalcemia occurs in 10% to 20% of the patients that undergo total thyroidectomy and may be minimized with the use of oral calcium supplements. Measuring postoperative calcium and parathyroid hormone levels is used to identify patients that may require higher doses of calcium as well as calcitriol supplements. All patients that have the entire gland removed require thyroid hormone replacement therapy.
Follow-up varies according to the final histopathology results. Postoperative I ablation is administered for high-risk patients with differentiated thyroid carcinomas, especially those with gross residual disease, metastatic disease, or nodal involvement. In these patients, administration of levothyroxine at a suppressive dose (TSH < 0.1 mU) has been shown to improve disease-free survival. Serum thyrogobulin levels should be measured every 6 to 12 months for patients with differentiated thyroid cancer that underwent total thyroidectomy.
Case Conclusion
The patient undergoes uncomplicated total thyroidectomy as an outpatient. Postoperatively, she is treated with levothyroxine and calcium supplements, the calcium is discontinued after a week. On final pathology follicular thyroid carcinoma is identified, measuring 3.2 cm at greatest diameter with capsular invasion. The patient is treated with radioactive iodine ablation four weeks after surgery. Five years later, the patient is noted to have elevated thyroglobulin. Workup identifies a local recurrence that is removed surgically. At ten-year follow-up, she is free of disease.
TAKE HOME POINTS
· Palpable thyroid nodules are very common in up to 10% of the population.
· Most thyroid nodules are benign.
· The single most important test for palpable thyroid nodules is FNAB with an accuracy of up to 95%.
· Thyroid nodules should be managed according to FNAB results. Accurate terminology facilitates proper treatment.
· Follicular lesions on FNAB carry a 15% to 30% malignancy and require surgical intervention.
· The appropriate treatment for patients with a follicular neoplasm is at a minimum a thyroid lobectomy
SUGGESTED READINGS
American Thyroid Association (ATA) Guidelines Taskforce on Thyroid Nodules and Differentiated Thyroid Cancer, Cooper DS, Doherty GM, Haugen BR, Kloos RT, Lee SL, Mandel SJ, Mazzaferri EL, McIver B, Pacini F, Schlumberger M, Sherman SI, Steward DL, Tuttle RM. Revised American Thyroid Association management guidelines for patients with thyroid nodules and differentiated thyroid cancer. Thyroid. 2009;19(11):1167–1214.
Cibas ES, Ali SZ. The Bethesda system for reporting thyroid cytopathology. Thyroid. 2009;19(11):1159–1165.
Dean DS, Gharib H. Epidemiology of thyroid nodules. Best Pract Res Clin Endocrinol Metab. 2008;22:901–911.
Gharib H, Papini E, Valcavi R, et al. American Association of Clinical Endocrinologists and Associazione Medici Endocrinologi medical guidelines for clinical practice for the diagnosis and management of thyroid nodules. Endocr Pract. 2006;12:63–102.