Hari R. Kumar
Judiann Miskulin
Presentation
A 53-year-old obese female presents with a diagnosis of new-onset diabetes that has been difficult to control, requiring multiple oral medications and insulin. She notes an overall change in appearance including thinning of her skin and striations, increased facial hair, and a redistribution of her body habitus with more central adiposity. A workup initiated by her primary care physician reveals an elevated serum cortisol with a suppressed adrenocorticotropic hormone (ACTH) level.
Differential Diagnosis
The most common cause of hypercortisolism is administration of exogenous glucocorticoids. Steroid medications, such as prednisone, are often used to treat a variety of ailments due to their anti-inflammatory and immune-modulating effects. Once synthetic sources have been eliminated, endogenous causes can then be explored. Among endogenous causes, excess secretion of ACTH from the pituitary (Cushing’s disease) accounts for over half of cases, with adrenal tumors, bilateral adrenal hyperplasia, and ectopic ACTH-producing tumors accounting for most of the remaining cases.
Discussion
Cortisol is a steroid hormone manufactured in the zona fasciculata layer of the adrenal cortex. As a member of the glucocorticoid family, cortisol mediates the stress-induced “fight or flight” response whose effects involve the cardiovascular, metabolic, immunologic, and other systems.
Serum levels of cortisol demonstrate diurnal variation with the peak occurring around 8 am and the nadir at midnight. The overall daily production of cortisol ranges from 10 to 30 mg a day but in times of stress may exceed 300 mg a day.
Cortisol synthesis is tightly regulated by a feedback loop consisting of the hypothalamus, pituitary, and adrenal gland (HPA axis). Corticotropin-releasing hormone is secreted by the hypothalamus, which stimulates release of ACTH from the anterior pituitary. ACTH then precipitates the release of various products from the adrenal cortex, including cortisol. Cortisol, in turn, inhibits further production from the hypothalamus and the pituitary.
Elevated levels of cortisol produce a constellation of symptoms known as Cushing’s syndrome, named after the American neurosurgeon who first described the case in 1912. Symptoms include asymmetrical weight gain (truncal obesity, “buffalo hump,” “moon facies”), hyperglycemia, hypertension, skin changes (striae, fragility, impaired wound healing), osteoporosis, mood lability, and sexual hormone imbalance (hirsutism, menstrual irregularities, decreased libido/impotence).
Workup
The initial step in diagnosing Cushing’s syndrome is to establish the presence of hypercortisolism. Conventional methods include 24-hour urine cortisol measurements or failure to suppress endogenous cortisol with a low-dose (1 mg) dexamethasone test. Emerging methods include measurement of midnight plasma or salivary cortisol levels.
Once the presence of excess cortisol has been confirmed, the next step is to measure serum ACTH level to determine whether this production is ACTH-dependent or ACTH-independent. Normal to high levels of ACTH suggest a pituitary or an ectopic etiology. Low levels of ACTH suggest an adrenal source. Our patient’s ACTH level was significantly below the normal level, which would indicate an adrenal source.
Imaging of the adrenal gland will then help to differentiate an adrenal tumor from bilateral adrenal hyperplasia. If on CT a discrete tumor is visualized, specific CT protocols can be used to further distinguish a benign adenoma from a malignant adrenocortical carcinoma. Benign adrenal tumors tend to have low attenuation on unenhanced images due to high lipid content. Both benign and malignant tumor will enhance with IV contrast; however, after a 10-minute post-contrast administration period, washout in excess of 50% is seen in benign lesions. In our patient, CT scan of the abdomen demonstrated a 5.3-cm contrast enhancing mass in the right adrenal gland (Figure 1).
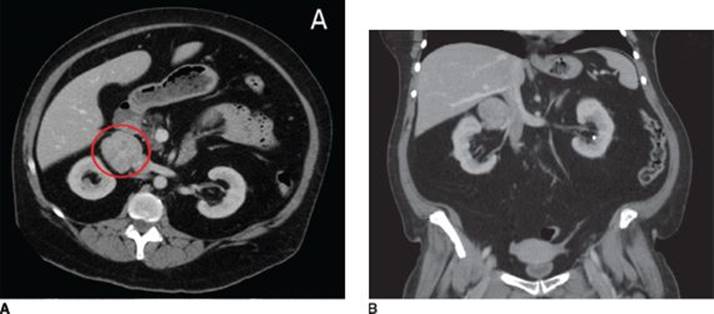
FIGURE 1 • A: CT scan of the abdomen demonstrating a contrast-enhancing mass in the right adrenal gland (circled). B: Coronal reformatting depicting the relationship of the mass to the right renal vein and the inferior vena cava.
A biopsy of the adrenal to provide a tissue diagnosis is not warranted unless the patient has a history of cancer in some other organ system. In that case, a biopsy can help rule out the presence of metastatic disease. Otherwise, once the diagnosis of a functional adrenal lesion has been made, the patient should proceed toward surgical treatment.
Surgical Approach
The location of the adrenal gland within the retroperitoneum allows it to be accessed by multiple surgical routes. Open anterior, posterior, flank, and thoracoabdominal approaches have all been described. The enthusiasm for laparoscopic surgery has enabled for the conversion of several of these approaches to minimally invasive procedures.
For most patients with adrenal tumors, a laparoscopic adrenalectomy is the preferred method of resection. It is associated with shorter hospitalization, less pain and morbidity, quicker recovery, and fewer incisional hernias. An absolute contraindication to laparoscopic adrenalectomy is extension of the tumor into nearby structures. A relative contraindication is tumor size as controversy exists as to whether tumors >6 cm should be resected laparoscopically due to increased chance of local recurrence. From a practical standpoint, tumors of this size can be difficult to mobilize and manipulate and more often result in conversion to an open procedure. In patients with local invasion, and perhaps those with large tumors, an open approach should be undertaken.
The following sections describe the most common methods of adrenalectomy: the laparoscopic transabdominal technique and the open transabdominal technique.
Laparoscopic Transabdominal Technique
The patient is placed on a beanbag in the lateral decubitus position. The table is flexed in order to open up the space between the costal margin and the pubis. Careful attention must be paid to safely padding the arms and positioning them in gentle anterior flexion to avoid nerve injury. The ports are placed in a line that approximates the standard subcostal incision. A 12-mm camera port is placed in the middle with two 5-mm working ports placed on either side. Some surgeons advocate moving the camera port off the subcostal line and more toward the umbilicus in order to reduce instrument collision during the operation.
For right-sided lesions, a fourth port is placed medially in order to facilitate retraction of the liver by an assistant. In order to mobilize the liver, the right triangular ligament is taken down using cautery or ultrasonic shears. The dissection begins laterally and proceeds medially until the inferior vena cava is identified. Retraction of the liver in an anterior and lateral fashion will appropriately expose the retroperitoneal space where the adrenal gland lies so that Gerota’s fascia can then be opened. Blunt dissection between inferior vena cava and the medial aspect of the adrenal gland will identify the central adrenal vein that is then divided with either endoscopic clips or a laparoscopic stapling device. The inferior phrenic pedicle is then divided in a similar fashion. The remaining small vessels can usually be controlled with cautery or ultrasonic shears. Once the adrenal gland is freed from the surrounding structures, it is placed in an endoscopic specimen bag and removed.
For left-sided lesions, the port placement is similar, though a fourth port may not be necessary. The spleen and splenic flexure of the colon are taken down from their lateral attachments using cautery or ultrasonic shears. Once the spleen has been mobilized medially, the plane of dissection continues posterior to the pancreatic tail until the left adrenal is identified in proximity to the aorta. The dissection should begin between the junction of the adrenal and left renal vein. The left central adrenal vein empties into the left renal vein, and although this junction does not necessarily need to be identified, the central vein should be ligated close to the adrenal to avoid injuring the left renal vein. The central vein and inferior phrenic pedicle can be divided using clips or a laparoscopic stapling device. The gland can then be mobilized circumferentially with cautery or ultrasonic shears and removed in an endoscopic specimen bag.
Open Anterior Technique
The patient can be placed in the supine position or with the flank slightly elevated to provide exposure. Either a subcostal or a midline incision can be utilized. The anatomic details and step of the procedure mirror the laparoscopic approach (Table 1). The key difference is that additional exposure is often needed in the open approach due to the large nature of the tumors or invasion of nearby structures. Any structure grossly involved with the tumor must be removed en bloc.
TABLE 1. Key Technical Steps and Potential Pitfalls

On the right side, additional exposure can be gained by further mobilization of the duodenum through a Kocher maneuver. If invasion through the liver is found, a nonanatomic partial hepatectomy can be performed. If the inferior vena cava is involved, it may be resected and reconstructed using a synthetic graft.
On the left side, a splenectomy can be performed to gain additional exposure. If the adrenal gland cannot be approached from the lateral aspect, then an anterior approach can be used. The gastrocolic ligament is divided and the lesser sac is entered. The inferior border of the pancreas can be elevated to find the left adrenal, which lies just posterior.
Postoperative Management
Adrenalectomy for hypercortisolism is a well-tolerated procedure with most patients leaving the hospital within a few days. In addition to the routine infectious complications associated with most every surgery, when taking care of these patients, clinicians should specifically be monitoring for an Addisonian crisis. Symptoms to be wary of include weakness, mental status changes, hypotension, and hypoglycemia. These symptoms occur because chronic suppression of the HPA axis leads to a state of relative hypocortisolism. The day of operation and then postoperatively, patients must be supplemented with stress doses of steroids (hydrocortisone 100 mg three times daily) followed by a taper regimen to maintenance doses. This steroid replacement regimen should be continued until the HPA axis can recover.
While physical recovery from surgery is usually limited to a few weeks, resolution of symptoms and biochemical recovery often takes much longer. The median interval to discontinuation of steroid replacement therapy is between 12 and 30 months after a unilateral adrenalectomy. In a similar fashion, most of the physical and physiologic effects of hypercortisolism do improve after surgery, but the progression takes several months to years. Around three-quarters of all patients experience dramatic improvement in their physical features as well as significant decreases in their hypertension and diabetes.
Case Conclusion
The patient was taken to the operating room where a laparoscopic right adrenalectomy was attempted. The tumor was not able to be separated from the right renal vein safely and the case was converted to an open approach in order to complete the resection. Pathologic examination of the tumor revealed a large adenoma (Figure 2). Postoperative testing showed resolution of the patient’s hypercortisolism, but the steroid weaning and improvement in symptoms took place gradually over 12 months.
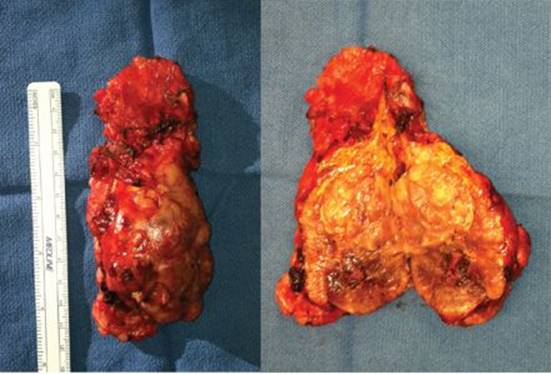
FIGURE 2 • The excised right adrenal gland (left) which was subsequently opened on the back table of the operating room (right) to reveal the large adrenal tumor.
TAKE HOME POINTS
· Elevated with cortisol with suppression of ACTH production indicates an adrenal source of hypercortisolism.
· All biochemically functional adrenal masses should be resected.
· A laparoscopic transabdominal approach is the preferred method of resection unless the tumor is large or involves nearby structures, which then mandates an open approach.
· Postoperative management must include a steroid supplementation regimen due to suppression of the HPA axis.
· Biochemical recovery and resolution of symptoms is gradual.
SUGGESTED READINGS
Brunt LM, Doherty GM, Norton JA, et al. Laparoscopic adrenalectomy compared to open adrenalectomy for benign adrenal neoplasms. J Am Coll Surg. 1996;183:1–10.
Doherty GM, Nieman LK, Cutler GB Jr, et al. Time to recovery of the hypothalamic-pituitary-adrenal axis after curative resection of adrenal tumors with Cushing’s syndrome. Surgery. 1990;108:1085–1090.
Findling JW, Raff H. Diagnosis and differential diagnosis of Cushing syndrome. Endocrinol Metab Clin North Am. 2001;30:729–747.
Shen WT, Sturgeon C, Duh QY. From incidentaloma to adrenocortical carcinoma: the surgical management of adrenal tumors. J Surg Oncol. 2005;89:186–192.