Geoffrey W. Krampitz
Jeffrey A. Norton
Presentation
A 22-year-old morbidly obese male with a history of peptic ulcer disease was referred to our clinic for epigastric pain. Over the past year, the pain has persisted despite a 3-month trial of proton pump inhibitor therapy. Prior esophagoduodenoscopy demonstrated a 1-cm nonhealing, solitary ulcer in the proximal duodenum with a workup that was negative for Helicobacter pylori infection. The patient’s also describes chronic diarrhea over this same time interval. In our clinic, in addition to the chief complaints of burning epigastric pain and four to five large-volume loose bowel movements on a daily basis, the patient also divulged that he had been feeling increasingly fatigued over the last 3 years and his mood had become progressively depressed. He also complained of persistent bone and muscle pain that he attributed to his increasing weight. He also indicated that he had a soft mobile mass in his right inguinal region, similar to his prior lipomas. He had no fevers, chills, night sweats, weight loss, headaches, syncope, visual changes, galactorrhea, palpitations, shortness of breath, flushing, nausea, vomiting, hematochezia, melena, hematuria, or dysuria. His complete review of systems was otherwise negative.
His past medical history was significant for gynecomastia at the age of 16, the workup of which revealed elevated prolactin level >280 ng/mL and a 2.1-cm pituitary macroadenoma. He underwent a surgical reduction mammoplasty, and he was started on cabergoline therapy after which his prolactin level normalized. A recent prolactin level was normal at 40 ng/mL. A recent MRI showed slight reduction in tumor size, and visual field testing suggested no progression of his disease. He also had multiple lipomas excised over the past 5 years. He was diagnosed with hypogonadotropic hypogonadism for which he was prescribed transdermal testosterone with little improvement in symptoms, morbid obesity for which he was attempting dietary changes without success, and insulin resistance not requiring medications.
Family history was significant for his mother, now aged 54, who had amenorrhea at age 35, the workup of which revealed a prolactinoma that was managed medically. Several years later, she was diagnosed with parathyroid hyperplasia for which she underwent a subtotal (3 and ½ gland) parathyroidectomy. More recently, she had progressive epigastric discomfort for which she had yet to be evaluated.
The patient’s physical exam was notable for morbid obesity (BMI, 56), undisturbed visual fields, nonfocal neurologic exam, and no expressible galactorrhea; heart exam revealed a regular rate and rhythm; lungs were clear to auscultation; abdomen was protuberant but soft, nontender, and nondistended; rectal exam showed a normal prostate and no masses or blood; normal external male genitalia; a 4-cm, soft, nontender, mobile soft tissue mass in the right inguinal region; and extremities with good pulses and without clubbing or cyanosis.
Differential Diagnosis
This patient has a constellation of symptoms classic for Zollinger-Ellison syndrome (ZES). The diagnosis of ZES is often delayed because it can mimic many other much more common conditions that result in peptic ulcers and/or hypergastrinemia. Ulcerogenic conditions with excessive gastric acid secretion include gastric outlet obstruction, retained gastric antrum after Bilroth II reconstruction, and G-cell hyperplasia. Nonulcerogenic conditions without excessive gastric acid secretion include postvagotomy, postgastric bypass, pernicious anemia, atrophic gastritis, short gut syndrome after significant intestinal resection, renal failure, Helicobacter pylori infection, VIPoma, and stomach irradiation. Many of these conditions are associated with achlorohydria, in which stomach acid production is absent, resulting in hypergastrinemia, mimicking ZES. However, our patient had no obstructive symptoms, prior intra-abdominal operations, irradiation, renal failure, gastritis, or anemia. He had endoscopically proven peptic ulcers without H. pylori infection refractory to a standard trial of PPI therapy and in the presence of chronic diarrhea raising our suspicion for ZES.
Discussion
In 1955, Zollinger and Ellison first described a syndrome of severe peptic ulcers associated with pancreatic islet cell tumors that were refractory to conventional acid-reduction surgery. We now know that these tumors were gastrinomas causing gastrin hypersecretion and excessive gastric acid production, which in turn leads to intractable peptic ulcer disease. Gastrinoma has a yearly incidence of approximately 0.1 to 3 cases per million people, making it the second most common pancreatic neuroendocrine tumor overall. It is also the causative factor in approximately 0.1% to 1% of patients with peptic ulcer disease. In 80% of cases, ZES occurs sporadically. However, approximately 20% of patients with ZES have the familial form associated with multiple endocrine neoplasia type 1 (MEN-1). MEN-1 is a syndrome first described in 1954 by Wermer caused by mutations in a gene located on chromosome 11q13 encoding a tumor suppressor protein called menin. Patients with MEN-1 have asymmetrical parathyroid hyperplasia, duodenal and pancreatic neuroendocrine tumors and anterior pituitary tumors, lipomas, as well as thyroid and adrenocortical adenomas. Fifty percent of patients with MEN-1 have ZES, making gastrinoma the most common functional pancreatic or duodenal neuroendocrine tumor in MEN-1. Despite increased awareness of ZES and improvements in diagnostic methodologies, the mean time from symptom onset to diagnosis is 8 years in many studies, so that improvements in detection and awareness are still needed.
Workup
Any patient with peptic ulcer disease that is refractory, recurrent, atypical, requiring surgery, or in the absence H. pylori should undergo a workup for ZES. The presence of hyperparathyroidism, nephrolithiasis, or family history suggestive of MEN-1 should also raise suspicion for ZES. We begin our workup by obtaining a fasting serum concentration of gastrin. Hypergastrinemia occurs in almost all patients with ZES (99% sensitivity) and is defined as a serum gastrin concentration >100 pg/mL. Because PPI therapy can induce hypergastrinemia, we checked the patient’s PPI for 1 week prior to the test. In this case, our patient’s gastrin level was significantly elevated at 1,210 pg/mL. Basal acid output (BAO) was also elevated at 39 mEq/h (normal ≤15 mEq/h or <5 mEq/h in patients who have undergone previous acid-reducing operations). In addition, a gastric pH measured at 1.7 also indicated acid hypersecretion. Although less accurate than BAO, a gastric pH >3 essentially excludes ZES, whereas a pH ≤2 is consistent with ZES. However, many patients with ZES have gastric acid hypersecretion and minimally increased fasting serum gastrin concentrations (100 to 1,000 pg/mL). For these patients, the secretin stimulation test is the provocative test of choice to establish the diagnosis of ZES.
Twenty percent of ZES cases occur in association with MEN-1, and ZES is the presenting symptom in 40% of cases of MEN-1. Thus during the initial workup for ZES, MEN-1 must always be excluded. This was particularly relevant in our patient who exhibited other signs and symptoms consistent with MEN-1, including pituitary adenoma, lipomas, as well as fatigue, depressed mood, and musculoskeletal pain. Our initial screen was a serum calcium measurement, exploiting the high penetrance of hyperparathyroidism in MEN-1, followed by serum parathyroid hormone (PTH) concentration, if necessary. In this case, our patient’s serum calcium and PTH levels were elevated at 12 mg/dL and 146 pg/mL, respectively.
With a biochemical diagnosis of ZES established, the next step in the workup was to localize and characterize the gastrinoma to determine resectability and the best operative approach. Approximately 80% of gastrinomas are found within the gastrinoma triangle, the apices of which are bounded by the junction of the cystic and common bile ducts superiorly, the junction of the second and third portions of the duodenum laterally, and the neck of the pancreas medially. Gastrinomas associated with MEN-1 tend to be multiple, small, and usually originate in the duodenum.
In our case, a pancreatic protocol CT scan including PO and IV contrast with 5-mm cuts demonstrated a dominant 3-cm tumor in the superiomedial aspect of the head of the pancreas abutting the superior mesenteric vein (Figure 1). The sensitivity of CT is directly related to the size of the tumor. Tumors >3 cm are detected in 83% to 95% of cases, tumor 1 to 3 cm are detected in 30% of cases, whereas tumors <1 cm are not detectable. Another limitation of CT scanning in the setting of gastrinoma is that only 50% of liver metastases are detected.
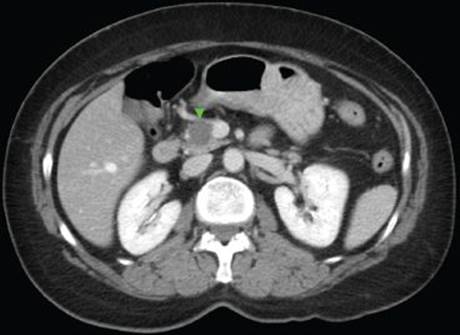
FIGURE 1 • Computed tomography scan demonstrating 3-cm gastrinoma at the head of the pancreas and abutting the superior mesenteric vein.
We then obtained a somatostatin receptor scintigraphy (SRS, also called octreoscan) that revealed focal, intense tracer uptake in the region of the head of the pancreas corresponding to the 3-cm lesion seen on CT scan (Figure 2).In addition, SRS also demonstrated a 1-cm tumor in the neck of the pancreas on the anterior wall, a third 8-mm tumor in the inferior portion of the head of the pancreas abutting the duodenum, and no scintigraphic evidence of distant metastases. The additional findings seen on SRS demonstrate the increased sensitivity of this modality over CT. SRS has a sensitivity that surpasses all other imaging modalities combined, and in the setting of ZES has an overall sensitivity of about 90%, specificity of 100%, and positive predictive value near 100%. Nevertheless, the sensitivity of SRS is still limited when interrogating for very small tumors. The sensitivity of SRS for tumors >2.2 cm is 96%, for tumors 1.1 to 2.2 cm is 64%, but for tumors <1.1 cm is only 30%. Because duodenal gastrinomas are usually subcentimeter in size, SRS fails to detect about 50% of these tumors.
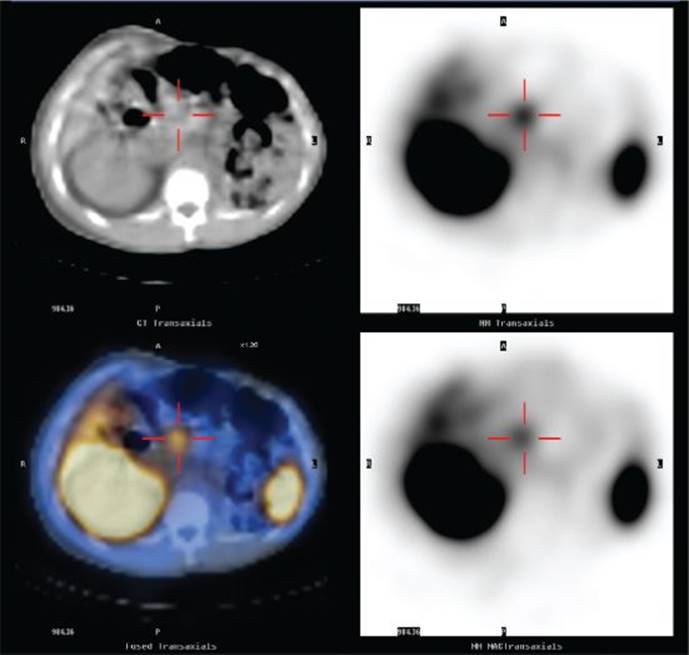
FIGURE 2 • Octreoscan demonstrating focal intense tracer uptake in the pancreatic head region corresponding to the 3-cm hypervascular lesion seen on CT.
The patient’s constellation of signs including pituitary adenoma, hyperparathyroidism, multiple pancreatic gastrinomas, and lipomas, as well as similar finding of prolactinoma and hyperparathyroidism in his mother was strongly suggestive of MEN-1. The patient, his mother, and his two brothers underwent genetic testing for MENIN gene mutations. This testing revealed a T-278 mutation in the patient and the mother, but not in the siblings, thus establishing a genetic diagnosis of MEN-1.
Diagnosis and Treatment
The first principle of treating ZES associated with MEN-1 is to control symptoms medically. Following our diagnostic workup for ZES, we reinitiated PPI therapy for our patient and titrated the dose to obtain a BAO below 15 mEq/h, ultimately requiring 80 mg of pantoprazole orally twice per day.
The second principle of treating ZES associated with MEN-1 is to address parathyroid hyperplasia. Because ZES symptoms can be exacerbated by hypercalcemia resulting from hyperparathyroidism, the patient underwent a subtotal (3 and ½ gland) parathyroidectomy with a transcervical thyrmectomy. This allowed for decreased end-organ effect of hypergastrinemia and better medical control of ZES symptoms.
The third principle of treating ZES associated with MEN-1 is to determine if the patient is a candidate for operative intervention. Gastrinomas associated with MEN-1 have a propensity to spread to peripancreatic lymph nodes but are usually more indolent and less likely to metastasize to the liver when compared to sporadic tumors. Because hepatic involvement is the most important predictor of survival, ZES patients with MEN-1 have a more favorable long-term prognosis. Although surgery for MEN-1-associated disease is rarely curative (0% to 10%), resection may prevent liver metastases and thus affect long-term survival. Because tumor size >2 cm is predictive of progression to liver metastasis, surgery for ZES associated with MEN-1 is recommended only if there is an identifiable tumor larger than 2 cm. This is relevant to our patient because his main tumor was 3 cm in size.
Surgical Approach
Because of tumor multifocality (both in the pancreas and the duodenum) in MEN-1 patients, there is no biochemical cure. The goal of surgery is to prevent liver metastases and thus decrease tumor-related mortality. As such, we recommend operative approaches that focus on resecting tumor while preserving as much of the normal tissue as possible. In general, the operation should include resection of body and tail pancreatic tumors, enucleation of palpable pancreatic head tumors, duodenotomy with excision of duodenal tumors, and peripancreatic lymph node sampling. We do not favor routinely performing Whipple pancreaticoduodenectomy for attempted cure because long-term survival is excellent with the surgical approach described above, which has much less morbidity. Nevertheless, Whipple pancreaticoduodenectomy may be necessary with bulky tumors at the head of the pancreas, tumors involving the ampulla, or tumor invasion into major ductal or vascular structures. Our patient had multiple pancreatic tumors, the largest of which was a bulky tumor in the head of the pancreas abutting the superior mesenteric vein. Thus, we obtained informed consent for a possible Whipple procedure.
We entered the abdomen via bilateral subcostal incision and performed a full abdominal exploration. We inspected and palpated the liver without finding any evidence of hepatic metastases. In addition, there was no evidence of carcinomatosis or distant metastases. We then established exposure by placing a Thomson retractor and performed a cholecystectomy.
The next step was the portal dissection. The common hepatic artery was identified and protected. We identified the common bile duct that appeared normal in size and caliber. We also dissected out the common hepatic duct and placed a right-angled clamp and a Vessel loop around it. In between the common hepatic artery and the bile duct, we identified the portal. At the superior border of the pancreas, we identified the gastroduodenal artery, placed two 2-0 silk sutures around it, a 3-0 silk suture ligature on the side of the common hepatic artery, and divided it with a 15-blade. We were then able to dissect underneath the neck of the pancreas toward the superior mesenteric vein.
In order to obtain adequate exposure of the pancreas, we mobilized the right colon and performed a Kocher maneuver. It was at this time we could clearly palpate the 1-cm tumor in the neck of the pancreas on the anterior wall, and as we did the Kocher maneuver, we could feel the 8-mm tumor in the more inferior aspect of the head of the pancreas abutting the pancreas and duodenum, as well as the larger 3-cm tumor in superiomedial aspect of the head of the pancreas, abutting the sidewall of the SMV. The remainder of the pancreas appeared and palpated normally.
We opened the lesser sac widely dividing the gastrocolic ligament and dissected along the inferior border of the body and tail of the pancreas from lateral to medial toward the superior mesenteric vein. We identified the superior mesenteric vein, the right sidewall of which was abutting the tumor. However, we were able to dissect underneath the neck of the pancreas and follow the anterior surface of the SMV toward the portal vein.
At this point, we felt that a Whipple operation was possible, but part of the sidewall of the SMV at the confluence of the SMV, splenic vein, and portal vein would have to be resected. We divided the duodenum 2 cm distal to the pylorus with a GIA 55-mm stapler. We elected to divide the pancreas initially rather than ligate the vessels from the head of the pancreas to the SMV and portal vein. We used the electrocautery to divide the neck of the pancreas with care to remove the second 1-cm pancreatic neuroendocrine tumor with the specimen. This exposed the confluence where the inferior mesenteric vein, superior mesenteric vein, and splenic vein combined to form the portal vein. We divided the bile duct with electrocautery and dissected the more superior aspect of the head of the pancreas and the bile duct off the portal vein. The superior pancreaticoduodenal vein was then identified and ligated in continuity with 2-0 silk ties. We were able to identify and dissect the inferior pancreaticoduodenal followed by the right gastric vein and ligated them in a similar fashion.
We divided the jejunum approximately 20 cm distal to the ligament of Treitz with a GIA 55-mm stapler. We took down the proximal jejunal mesentery with the LigaSure device. We widely opened the ligament of Treitz and passed the proximal jejunum to the right side of the abdomen. We dissected the head of the pancreas off the SMA, feeling the SMA pulse, and using a right-angle clamp and the LigaSure device. This was posterior to the SMV, but we were able to completely mobilize the head of the pancreas off the SMA.
The head of the pancreas was completely freed off the SMV except for approximately 8-mm aspect of the tumor that was adherent to the sidewall of the SMV. We used a TA-30-mm stapler with a vascular load and resected partially the sidewall of the SMV with the specimen. Once we placed the staple line, we divided it with the 15-blade and delivered the specimen to pathology. We did narrow the SMV slightly, but there was good flow within the vessel. Further, the IMV and splenic vein were widely patent, and there was a large portal vein. We dissected the neck of the pancreas off the splenic vein and IMV in order to mobilize it for the pancreaticojejunal anastamosis.
Next, we completed anastomoses of the jejunum to the pancreas, the common hepatic duct, and the duodenum. Because the pancreatic duct was not dilated (2 mm in diameter), we did a dunking anastomosis with 3-0 silk sutures, full thickness on the jejunum and capsule of pancreas such that 2 to 3 cm of the body of the pancreas was inverted into this area of jejunum. Approximately 6 cm distal to that anastomosis, we connected the common hepatic duct to the same limb of jejunum with interrupted 4-0 PDS sutures, such that the posterior row had the knots on the inside of the anastomosis while the anterior row had the knots on the outside. Next, we restored GI continuity by anastomosing the duodenum to the jejunum approximately 20 cm distal to the hepaticojejunostomy. This was done with a 3-0 silk seromuscular layer and a running full-thickness 3-0 PDS layer.
We irrigated saline solution and placed 10 mL of Tisseel fibrin glue on the pancreaticojejunostomy and the hepaticojejunostomy. We placed two No. 15 round JP drains to drain the right upper quadrant. We closed the fascia in two layers with looped No. 1-PDS. Finally, the skin was closed with a subcuticular 4-0 Monocryl. There were some lipomas in the subcutaneous tissue of the abdominal wall, and these were resected and sent to pathology (Table 1).
TABLE 1. Key Technical Steps and Potential Pitfalls for a Whipple Procedure

Special Intraoperative Considerations
The first intraoperative consideration is to confirm the presence or absence of tumor metastases seen on preoperative imaging. Hepatic metastases are the primary determinant of survival in ZES patients. Thus, tumors involving the liver would require appropriate debulking via wedge resection, sementectomy, or lobectomy depending on the extent and respectability of disease.
The second intraoperative consideration is to determine if the tumors seen on preoperative imaging indeed require a Whipple procedure or whether a more limited resection would be sufficient. As mentioned previously in this case, the tumor in the head of the pancreas was of a significant size that required a Whipple procedure.
The third intraoperative consideration is to determine whether involvement of vascular structures necessitates vascular reconstruction. Since the goal of the operation was not curative, but rather to prevent liver metastasis, microscopically negative margins are not as significant a consideration as in sporadic tumors. Thus, in this case, we decided to partially resect the sidewall of the SMV with the specimen since doing so would not compromise blood flow through the vessel and would achieve the goal of the operation without the risks of vascular reconstruction.
Postoperative Management
The major complications of Whipple pancreaticoduodenectomy are death, anastomotic leaks, intraabdominal abscesses, and delayed gastric emptying. With improvements in pancreatic surgery and postoperative care, the mortality for pancreaticoduodenectomy at most high-volume is reported as 2% to 6%. Anastomotic leaks, primarily at the pancreaticojejunostomy, occur in approximately 8% of patients and usually resolve with adequate drainage. Intraabdominal abscesses occur in 5% to 10% of cases. Delayed gastric emptying occurs in approximately 30% of patients and usually resolves within a couple of weeks postoperatively.
Case Conclusion
Our patient tolerated the procedure well and had an uncomplicated postoperative course. He was transferred to the ICU on the operative evening as is the standard practice at our institution. The following day he was transferred to a nonmonitored surgical unit. He tolerated a clear liquid diet on postoperative day 3 and had return of bowel function the following day. At that time, the epidural was removed and the patient’s pain was well controlled on oral pain medications. On postoperative day 5, the anterior Jackson-Pratt drain was removed, and the posterior drain was removed the following day. The patient was discharged home on postoperative day 7.
The pathology showed multiple well-differentiated pancreatic neuroendocrine neoplasms, the largest measuring 3.2 cm, arising in a background of diffuse islet cell hyperplasia. The surgical margins were not involved with tumor, but there were multiple (4/12) lymph nodes with metastatic disease that gave the clinical appearance of multifocal disease. Although careful pathologic examination did not show any multifocal duodenal microtumors, these are difficult to detect. As a result, for patients with ZES not undergoing a pancreaticoduodenectomy, we routinely perform a duodenotomy for direct inspection and exploration of the duodenal mucosa. Duodenotomy was particularly important in the detection of small duodenal tumors, allowing localization of 90% of subcentimeter tumors versus only 50% discovered on preoperative imaging. A recent prospective study of patients with sporadic ZES who underwent surgical exploration revealed a significantly higher cure rate following duodenotomy, both immediately and long term.
TAKE HOME POINTS
· ZES is a syndrome caused by gastrinoma usually located within the gastrinoma triangle and associated with symptoms of peptic ulcer disease and diarrhea.
· The diagnosis of ZES is achieved by measuring fasting levels of serum gastrin, basal acid output, and postsecretin challenge testing.
· Due to the high association of ZES with MEN-1, hyperparathyroidism must be excluded by obtaining a serum calcium and parathyroid hormone level.
· Treatment of ZES consists of medical control of symptoms with PPIs and evaluation for surgical intervention.
· Noninvasive imaging studies including SRS, CT, and MRI should be performed initially to evaluate for metastases and identify resectable disease. Invasive imaging modalities, such as EUS, may be performed to further evaluate primary tumors, if necessary. IOUS, palpation, and duodenotomy are used for intraoperative localization of gastrinomas.
· In patients with MEN-1, surgical resection should be pursued only if there is an identifiable tumor larger than 2 cm in contrast to resectable sporadic gastrinoma, for which all patients should undergo surgical exploration.
· In patients with liver metastases, cytoreductive surgery should be performed if more than 90% of the visible tumor can be safely removed. Figure 3 summarizes the workup, medical management, and surgical approach to ZES.

FIGURE 3 • Summary algorithm
SUGGESTED READINGS
Alexander HR, Fraker DL, Norton JA, et al. Prospective study of somatostatin receptor scintigraphy and its effect on operative outcome in patients with Zollinger-Ellison syndrome. Ann Surg. 1998;228(2):228–238.
Béhé M, Gotthardt M, Behr TM. Imaging of gastrinomas by nuclear medicine methods. Wien Klin Wochenschr. 2007;119(19–20):593–596. Review.
Birkmeyer JD, Finlayson SR, Tosteson AN, et al. Effect of hospital volume on in-hospital mortality with pancreaticoduodenectomy. Surgery. 1999;125:250–256.
Cisco RM, Norton JA. Surgery for gastrinoma. Adv Surg. 2007;41:165–176.
Diener MK, Fitzmaurice C, Schwarzer G, et al. Pylorus-preserving pancreaticoduodenectomy (pp Whipple) versus pancreaticoduodenectomy (classic Whipple) for surgical treatment of periampullary and pancreatic carcinoma. Cochrane Database Syst Rev. 2011;5:CD006053.
Eriksson B, Oberg K, Skogseid B. Neuro-endocrine pancreatic tumors: clinical findings in a prospective study of 84 patients. Acta Oncol. 1989;28:373–377.
Gibril F, Reynolds JC, Doppman JL, et al. Somatostatin receptor scintigraphy: its sensitivity compared with that of other imaging methods in detecting primary and metastatic gastrinomas. A prospective study (see comments). Ann Intern Med. 1996;125(1):26–34.
Howard TJ, Zinner MJ, Stabile BE, et al. Gastrinoma excision for cure. Ann Surg. 1990;211:9–14.
Imamura M, Komoto I, Ota S. Changing treatment strategy for gastrinoma in patients with Zollinger-Ellison syndrome. World J Surg. 2006;30(1):1–11. Review.
Li ML, Norton JA. Gastrinoma. Curr Treat Options Oncol. 2001;2(4):337–346.
Meko JB, Norton JA. Management of patients with Zollinger-Ellison syndrome. Annu Rev Med. 1995;46:395–411.
Norton JA. Gastrinoma: advances in localization and treatments. Surg Oncol Clin N Am. 1998;7(4):845–861.
Norton, JA. Neuroendocrine tumors of the pancreas and duodenum. Curr Probl Surg. 1994;31:77–164.
Norton, JA. Surgical treatment and prognosis of gastrinoma. Best Pract Res Clin Gastroenterol. 2005;19:799–805.
Norton JA, Fang TD, Jensen RT. Surgery for gastrinoma and insulinoma in multiple endocrine neoplasia type 1. J Natl Compr Canc Netw. 2006;4(2):148–153.
Norton JA, Fraker DL, Alexander HR, et al. Surgery to cure the Zollinger-Ellison syndrome. N Engl J Med. 1999;341:635–644.
Norton JA, Jensen RT. Resolved and unresolved controversies in the surgical management of patients with Zollinger-Ellison syndrome. Ann Surg. 2004;240(5):757–773. Review.
Norton JA, Venzon DJ, Berna MJ, et al. Prospective study of surgery for primary hyperparathyroidism (HPT) in multiple. Ann Surg. 2008;247(3):501–510.
Peterson DA, Dolan JP, Norton JA. Neuroendocrine tumors of the pancrease and gastrointestinal tract and carcinoid disease. In: Norton JA et al, eds. Surgery: Basic Science and Clinical Evidence. 2nd ed. New York, NY: Springer, 2008.
Pipeleers-Marichal M, Somers G, Willems G, et al. Gastrinomas in the duodenums of patients with multiple endocrine neoplasia type 1. N Engl J Med. 1990;322(11):723–727.
Pisegna JR, Norton JA, Slimak GG, et al. Effects of curative gastrinoma resection on gastric secretory function and. Gastroenterology. 1992;102:767–778.
Thompson NW, Vinik AI, Eckhauser FE. Micro-gastrinomas of the duodenum. Ann Surg. 1989;209:396–404.
Wank SA, Doppman HL, Miller DL, et al. Prospective study of the ability of computerized axial tomography to localize gastrinomas in patients with Zollinger-Ellison syndrome. Gastroenterology. 1987;92:905–912.
Wermer P. Endocrine adenomatosis and peptic ulcer in a large kindred: inherited multiple tumors and mosaic pleiotropism in man. Am J Med. 1963;35:205–212.
Wermer P. Genetic aspects of adenomatosis of endocrine glands. Am J Med. 1954;16(3):363–371.
Zollinger RM, Ellison EH. Primary peptic ulceration of the jejunum associated with islet cell tumors of the pancreas. Ann Surg. 1955;142:708–728.