Adriana Laser
Guillermo A. Escobar
Gilbert R. Upchurch Jr.
Presentation
A 65-year-old male smoker presents to the emergency room complaining of a sharp, continuous pain in his left back and groin starting earlier in the evening. His vital signs are significant for tachycardia and a decreased mental status. There was no associated trauma. He has a history of coronary artery disease and hyperlipidemia. He is taking aspirin and an HMG CoA reductase inhibitor daily. He smokes approximately one pack per day. On physical examination, the patient is neurologically intact but lethargic. He is in sinus tachycardia by EKG. On abdominal examination, he is obese with guarding, but no rebound tenderness. A pulsatile midabdominal mass is noted, and no hernias are identifiable. He has palpable femoral pulses, but decreased popliteal and dorsalis pedis pulses with livedo reticularis of bilateral lower extremities.
Differential Diagnosis
The incidence of ruptured abdominal aortic aneurysm (rAAA) in the United States is 1-3/100,000. Mortality from rAAA repair remains high despite advances in screening, medical therapy, operative technique, and postoperative management. The mean age for a patient with rAAA is 70.6 years in males and 77.3 years in females.
Presentation and therefore differential diagnosis of an rAAA are varied. If the AAA ruptures intraperitoneally, presentation is usually acute with hemodynamic instability. Cardiovascular collapse often ensues. An AAA can also rupture retroperitoneally, and the patient can present hemodynamically stable. Retroperitoneal rupture is most often posterior and can at least temporarily be contained via clotting, plugging by the aneurysm’s mural thrombus being ejected, and tamponade by the retroperitoneal periaortic and perivertebral tissues. One study of 226 patients found that rAAA bleed into the retroperitoneum 85% of the time, the peritoneum 7%, the inferior vena cava (IVC) 6%, and enterically in 2% of cases.
Some studies report that up to three-quarters of patients are asymptomatic before rupture. Presentation, either with or without preceding symptoms, includes 45% of patients with hypotension, 72% with back and abdominal pain, and 83% with a pulsatile abdominal mass. Less than 50% of patients present with the classic triad of hypotension, abdominal pain, and pulsatile abdominal mass. Symptoms of rAAA can include those resulting from hematoma on adjacent structures or signs of hypovolemic shock, such as diaphoresis, emesis, syncope, pallor, flank ecchymosis, and vital sign abnormalities. Contained or sealed rupture can even exist chronically before being discovered. Chronic ruptures can present with chronic lower back pain, lower-extremity neuropathy, or can be asymptomatic. Another complication of chronic rAAAs is IVC fistulae. These can occur in as many as 2% to 6% of patients with rAAA and can present as lower-extremity swelling, congestive heart failure, or a left varicocele. Atypical presentations of rAAA can include pain radiating to the groin, acute femoral neuropathy or thigh ecchymosis from femoral nerve compression, partial upper gastrointestinal (GI) obstruction (third part of duodenum), lower-extremity ischemia from emboli of mural thrombi or aortic thrombosis, visceral thromboembolism, aortic enteric fistula, trauma, and gross hematuria. The differential diagnosis of rAAA can be seen in Table 1.
TABLE 1. Differential Diagnosis for rAAA
Largely because many AAA patients are asymptomatic, it is believed that one-third to one-half of patients with rAAA die before arriving at the hospital. In hospitals, mortality can reach 40% including patients who die before repair or perioperatively. This brings the overall mortality of patients with rAAA to a reported range of 50% to 94% and essentially 100% for untreated true rAAA.
Predisposition to rupture of an existing AAA includes female gender, with a three- to fourfold higher risk when compared with males. Current smoking predisposes to rupture with a 2.7 odds ratio; current smoking is an independent predictor of rupture and greater than formerly smoking. Aortic morphology, such as an eccentric or a saccular shape leading to increased wall stress, less tortuosity, greater cross-sectional diameter asymmetry, and increased aortic compliance also predispose to rupture. Other factors that have been associated with an increased probability of rupturing an existing AAA include (Table 2)large size at initial diagnosis, rapidly progressing in size with an expansion rate >1 cm/y, chronic obstructive pulmonary disease (COPD) and a lower forced expiratory volume in one second (FEV1) (independent risk factor), hypertension (independent risk factor), pain upon manual palpation of aneurysm, a mycotic AAA, family history, and uninsured status. It was also noted that more ruptures occur in winter, likely due to lower atmospheric pressure. Diabetes has been found to be associated with a greater risk of rupture of a small AAA, although it is negatively associated with the development of AAAs.
TABLE 2. Risk Factors for Rupture

Once an AAA is diagnosed, size becomes one of the most important determinants for planning surgical repair. The VA Cooperative Natural History of Large Abdominal Aortic Aneurysms Study determined the incidence of rupture in patients with large AAA > 5.5 cm. One-year incidence of rupture by initial aortic diameter was 9.4% for 5.5 to 5.9 cm, 20% for 6.5 to 6.9 cm, and 29.5% for ≥7.0 cm AAA. The annual rupture risk of observed small aneurysms (4 to 5.5 cm) was 0.6% per Aneurysm Detection and Management (ADAM) screening program and 3.2% per UK Small Aneurysm Trial (UKSAT). Other studies have also reported that one-third to one-half of all AAAs eventually rupture. Although the above factors describe who is at risk of rupturing an AAA, most are too prevalent and nonspecific to be used to identify patients for management or treatment. Risk of aneurysm rupture relates to hemodynamic stresses placed on a degenerative aortic wall and the capacity of the tissue to resist tensile stress. Berguer et al. numerically analyzed wall thickness, herniation of soft plaque through elastic coats of aneurysm, and local stress concentrators due to rigid calcium plaques using finite element analysis to determine that hemodynamic stresses are better than periodic diameter changes at predicting rupture. Although it is more time consuming, volume analysis is more sensitive than change in diameter alone in predicting rupture; however, diameter remains the predominantly used determinant of rupture risk clinically.
On a molecular level, the cause of AAA rupture involves many other complex processes. Choke et al. discovered increased angiogenesis at the rupture site and HIF-1-alpha up regulation with relative hypoxia at the aneurysm rupture edge. Other changes seen within the ruptured aortic wall are decreased elastin, changes in the extracellular matrix, such as increased collagen turnover, and an imbalance of matrix metalloproteinases and their inhibitors. Thrombosis-associated enzymes (tissue plasminogen activator), lipids (lysophosphatidic acid), and inflammatory mediators (c-reactive protein) may also be associated with AAA expansion and rupture.
Workup
If available, an unstable patient without a diagnosis can undergo ultrasound examination in the emergency room, especially when there is unclear etiology of hemodynamic instability. Contrast-enhanced computed tomography angiography (CTA) should be performed in all stable patients where rAAA is suspected to confirm the presence of an AAA and determine operative planning and suitability for endovascular aneurysm repair (EVAR). This will also make available evaluation of the iliac arteries and any venous anomalies, should open repair be undertaken. If ultrasound or CTA is unavailable, and the unstable patient has a history of an AAA or a current pulsatile abdominal mass, he should be taken to the endovascular hybrid suite directly for immediate endovascular or surgical repair based on angiography, recognizing that aortography often underestimates the true size of an AAA because laminated clot obscures the outer limit of the wall. If the receiving facility is unable to perform surgical repair, then immediate transfer should be undertaken.
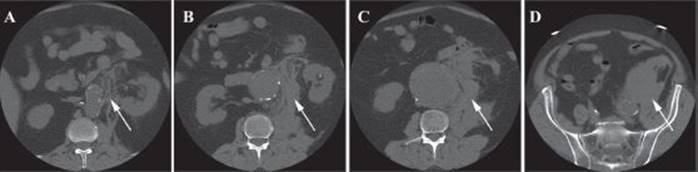
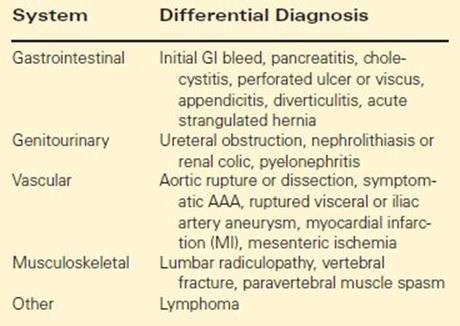
FIGURE 1 • Axial noncontrast CT image of a 60-year-old male with an rAAA preoperatively. A: Aneurysm is shown with left renal artery (arrow) and stranding. B: Further high attenuation stranding and retroperitoneal blood. C: Retroperitoneal blood contained. D: Extravasation of blood from the aorta.
The patient was deemed stable for transport to CT. Figure 1 shows an rAAA with an indistinct border on the left side and a likely site of rupture. There is left perinephric region stranding and a collection of increased attenuation in the retroperitoneum. The patient is believed to have suitable anatomy for EVAR.
Diagnosis and Treatment
Arrival to the endovascular suite or operating room should occur immediately because it is better to resuscitate there than in the emergency room. Fluids should be minimized, allowing for permissive hypotension, as blood pressure needs only to maintain cerebral and end-organ perfusion. Resuscitation beyond this can increase bleeding, and crystalloid dilutes the coagulation factors. Patient should have blood products type and crossed, labs drawn for a CBC and creatinine, placement of two large-bore peripheral intravenous catheters and an arterial blood pressure monitoring line, bladder catheter placement, and antibiotics given. A dedicated vascular operating room (OR) team should be involved from beginning to end of the case. The patient should be prepped and draped from chest to toes, and the surgical team should be ready to make an incision before anesthesia is induced since sudden hypotension can occur due to reversal of the tamponade, vasodilatation from the anesthetic agents, abdominal wall muscle relaxation, and decompression by the incision. Traditionally, open rAAA repair has been the only option with its attendant high mortality (44%) and morbidity (56%). EVAR is now becoming the preferred option for repair of rAAA at facilities where it is available on an emergency basis. Although only 8.8% of rAAA were repaired endovascular in 2003 (vs. 43% for unruptured AAA), a recent meta-analysis illustrated that 34% to 100% of patients presenting with rAAA met criteria for EVAR via CT. A 2010 study found that implementing an algorithm favoring endovascular repair over open repair for rAAA significantly improved mortality. A significant mortality advantage, a reduction by 25%, was also found in another study of EVAR-suitable patients undergoing EVAR as compared with open repair, though follow-up was a short 6 months.
Surgical Approach
For open repair, a transperitoneal approach via midline incision is most commonly undertaken. The primary goal is to first control the inflow and limit hemorrhage, so rapid supraceliac aortic control can be undertaken by manual (or sponge-stick) aortic compression at the level of the diaphragm. This is complemented by dividing the gastrohepatic ligament and the left crus of the diaphragm and then bluntly dissecting through the crus and around the aorta to place a clamp. Then, the third and fourth portions of the duodenum are rotated to the right to expose the perirenal aorta for assessment of infrarenal clamping. Heparin is often omitted if the patient is actively bleeding, but lasix and mannitol are given. Distal control of the aorta is obtained by dissecting the iliac arteries free and clamping them. Then, the aneurysm can then be opened and any lumbar and inferior mesenteric arteries that are bleeding into the sac are ligated. If at all possible, a tube graft is selected to serve as the repair conduit as this configuration requires the least length of anastomosis (when compared to a bifurcated graft). The graft is anastomosed proximally with 3-0 mono-filament suture. After checking this anastomosis, the distal anastomosis is created in an end-to-end configuration as well. Iliac clamps should be removed after informing the anesthesiologists and done one at a time to decrease hypotension from sudden perfusion to the lower extremities. The aneurysm sac is closed over the graft to decrease the risk of graft-enteric fistulae later on. Typically, the retroperitoneal hematoma is not decompressed. Distal pulses are checked before abdominal closure. There should be a low threshold for considering the patient at risk for abdominal compartment syndrome (ACS) and the abdomen may need to be left open.
EVAR can be performed under local, regional, or general anesthesia, depending on patient comfort, hemodynamic status, respiratory status, and level of consciousness. The patient should be placed supine on angio table, preferably in a hybrid OR. Prep should be from chest to toes. Bilateral femoral artery cut-downs are done before giving systemic heparin. Bilateral iliofemoral sheaths are placed, and wires are placed into the thoracic aorta along with a marking catheter. An angiogram of the aorta is performed using either iodinated contrast dye or carbon dioxide (to minimize injury to the kidneys) in order to identify the anatomy and location of the renal arteries. At this time, a decision is made regarding whether to use an aortouni-iliac (tube) or a modular, bifurcated endograft. Once this is done, the endograft body is inserted over a stiff guide-wire and deployed below the renal arteries. Assuming a bifurcated graft is chosen, the contralateral gate is opened, cannulated, and a contralateral limb is docked into the endograft and deployed. Balloon angioplasty is performed at the proximal and distal fixation sites, as well as in the gate area. Completion angiogram is performed to confirm there are no leaks and ensure exclusion of the ruptured aneurysm. All wires and sheaths are removed and the femoral arteriotomies are closed. Flow is confirmed distal to the arteriotomy closures by handheld Doppler. Protamine is given to reverse the heparin if heparin was used. Groin incisions are closed in multiple layers, and lastly pulses are checked prior to leaving the endovascular suite. These steps are highlighted in Table 3. Conversion to open repair may be necessary due to continuing blood loss, difficult access, graft migration, and other anatomic challenges.
TABLE 3. Key Technical Steps and Potential Pitfalls for EVAR for rAAAA

The patient in this scenario underwent endovascular repair under general anesthesia. The postprocedure angiogram was undertaken with CO2 angiography and no leaks were detected (Figure 2). He was then transported to ICU in stable condition for recovery.
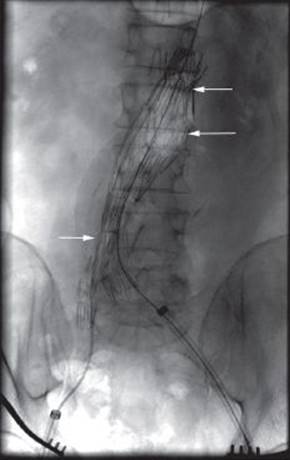
FIGURE 2 • Intraoperative angiography with CO2 at completion of endograft placement for rAAA. Endograft successfully excluding aneurysm with no leaks (arrow).
Special Intraoperative Considerations
If the patient is bleeding and hypotensive, rather than depend on open control of the aorta, an endovascularly placed aortic occlusion balloon catheter (inserted either transfemoral or transbrachial) can be utilized as an alternative for rapid proximal aortic control. It can also be used to minimize the acute drop in blood pressure during general anesthesia. Some institutions find that balloon occlusion is necessary in up to one-third of rAAAs treated. Care must be taken to minimize renal, mesenteric, and spinal ischemic time during all occlusions of the proximal aorta. Other scenarios that may be encountered intraoperatively are an infection that preceded the rupture, an aortoenteric fistula, or an aortic-IVC fistula. These are rare occurrences and in the acute setting may be treated as other rAAA, although higher morbidity and mortality are expected.
Endoleaks occur after elective and ruptured AAA repairs, and are the primary indication for reintervention. Type I, II, or III is seen in up to one-half of patients by 1 year after AAA repair. Endoleaks appear to be more common following rAAA repair; therefore, long-term follow up is mandatory.
Postoperative Management
Morbidity after rAAA repair has been documented at 61%, including respiratory failure, tracheostomy, renal failure, sepsis, MI, congestive heart failure, and bleeding. Less commonly seen postoperative complications are stroke, ischemic colitis, lower-extremity ischemia, and paraplegia. Late vascular complications are also higher after rAAA, 17% as compared with 8% after elective AAA repair. As well there is a high rate (20%) of secondary operations: 50% get laparotomy and 50% undergo other procedure. Complications, notably the prevalent respiratory and renal failure, are seen more frequently in open as compared with endovascular rAAA repair.
One of the most serious complications patients have for an increased risk of developing following rAAA repair, as compared with elective AAA repair, is sigmoid colon ischemia (presumable from ligation or covering of the inferior mesenteric artery during the repair). Ischemic colitis may present as hypotension, thrombocytopenia, bloody diarrhea, or metabolic acidosis. Sigmoidoscopy is performed at the bedside to evaluate for transmural ischemia. Mild cases may be treated with antibiotics and supportive care alone; however, severe cases may require resection. Bowel ischemia is described as occurring in 42% of patients after open rAAA repair and 22% after endovascular rAAA repair.
Another complication, abdominal compartment syndrome (ACS), can result if the abdomen is closed under excessive tension, or due to massive resuscitation attempts in the setting of EVAR or open repair for rAAA. ACS is defined as bladder pressure >25 when bladder volume is 50 to 100 mL. ACS may occur in the setting of an rAAA as a result of an expanding retroperitoneal hematoma, but it is not clearly defined if risk is greater from endovascular or open repair. Presumably, the risk of ACS after rAAA would be higher following EVAR as compared with open repair due to unligated aortic branch vessels causing an enlarging hematoma. However, a recent study found that rAAA treated with open repair had significantly higher postoperative intraabdominal pressures than those undergoing EVAR. The state of shock associated with AAA-free rupture and the insult of an open repair also contribute to tissue edema via microvascular permeability alterations. Since increased abdominal pressure leads to bowel ischemia and respiratory, cardiac, and renal dysfunction, immediate intervention is required. This consists of intraabdominal pressure monitoring, early recognition, and abdominal decompression, at the bedside, if the patient is unstable. In a recent study of rAAA, factors associated with increased risk of developing ACS included need for occlusion balloon, greater transfusion requirement, higher partial activated thromboplastin time, and higher use of aortouni-iliac grafts. These patients, and those with a massive bowel edema or a large retroperitoneal hematoma, should be considered intraoperatively for temporary abdominal closure with vacuum-assisted closure or mesh if undergoing open repair.
Mortality rates after rAAA open repair have been shown to be 35% to 65% (95% if present in extremis) and are not decreasing like the mortality rates of unruptured AAA. In contrast, elective AAA repair mortality is 2% to 5%. Consecutive patient series have found rAAA mortality to be associated with age >80 years, history of hypertension, angina, or MI, APACHE II score, low hematocrit, preoperative cardiac arrest or loss of consciousness, pre- or intraoperative hypotension, estimated blood loss ≥6 L or resuscitation with ≥12 L, and postoperative renal or respiratory failure. Traditionally, the majority of postoperative mortality is attributed to other cardiovascular diseases, such as coronary artery disease.
However, EVAR has become the new gold standard for rAAA. Veith et al. took patients with rAAA and, utilizing hypotensive hemostasis, performed arteriography. EVAR was undertaken if anatomy was deemed suitable. Supraceliac balloon occlusion was used if circulatory collapse ensued (10 of 29 patients required it). Operative mortality was only 13%. More recently, mortality was shown to be 33% for rAAA patients undergoing EVAR versus 41% for open repair. The advantage for rEVAR remained significant for patients >70 years old (36% vs. 47%, p < 0.001). Elevated operative risk for patients undergoing open repair of rAAA as compared with rEVAR has been attributed to aspects of the procedure. Both general anesthesia induction and sudden decompression of the aorta may lead to hypo-tension. Increased hypothermia and blood loss leading to coagulopathy are also often involved. Cost analysis for rAAA repair has been estimated at double that of elective repair, but more information is still needed contrasting open and endovascular repairs of rAAA.
Case Conclusion
The patient recovered uneventfully from his surgery except for mild renal dysfunction. He developed azotemia but did not require dialysis. He underwent CT imaging on postoperative day 2, which showed no endoleaks (Figure 3). He had no neurologic sequelae, was eating a regular diet, had no signs of infection, and had adequate blood pressure control. He was able to be discharged home on postoperative day 5 with smoking cessation information and a follow-up visit in 1 month with CT.
FIGURE 3 • Axial CT image with contrast enhancement of a 60-year-old male with an rAAA post-EVAR. A: Aneurysm is shown with endograft in place, stranding (arrow). B: Aneurysm sac around endograft with residual blood in the retroperitoneum (arrow). C: Retroperitoneal blood (arrow) with possible site of rupture. D: Residual blood in the pelvis (arrow).
TAKE HOME POINTS
· Rupture increases with age, female gender, and increased aortic diameter.
· Presentation is varied. However, patients uncommonly present with the classic triad of hypotension, abdominal pain, and pulsatile abdominal mass.
· CT imaging for stable patients, ultrasound or OR for unstable patients
· ACS and bowel ischemia are severe complications following rAAA repair.
· Endovascular repair shows morbidity and mortality advantage over open repair for rAAA.
SUGGESTED READINGS
Berceli SA. Ruptured Infrarenal Abdominal Aortic Aneurysm. Clinical Scenarios in Vascular Surgery. In: Upchurch GR, Henke PK, eds. Philadelphia, PA: Lippincott Williams & Wilkins, 2005.
Berguer R, Bull JL, Khanafer K. Refinements in mathematical models to predict aneurysm growth and rupture. Ann N Y Acad Sci. 2006;1085:110–116.
Bosch JAT, Teijink JAW, Willigendael EM, et al. Endovascular aneurysm repair is superior to open surgery for ruptured abdominal aortic aneurysms in EVAR-suitable patients. J Vasc Surg. 2010;52(1):13–18.
Boxer LK, Dimick JB, Wainess RM, et al. Payer status is related to differences in access and outcomes of abdominal aortic aneurysm repair in the United States. Surgery. 2003;134:142–145.
Brewster DC, Cronenwett JL, Hallett JW, et al. Guidelines for the treatment of abdominal aortic aneurysms. Report of a subcommittee of the Joint Council of the American Association for Vascular Surgery and Society for Vascular Surgery. J Vasc Surg. 2003;37:1106–1117.
Brown MJ, McCarthy MJ, Bell PR, et al. Low atmospheric pressure is associated with rupture of abdominal aortic aneurysms. Eur J Vasc Endovasc Surg. 2003;25(1):68–71.
Champagne BJ, Darling RC, Daneshmand M, et al. Outcome of aggressive surveillance colonoscopy in ruptured abdominal aortic aneurysm. J Vasc Surg. 2004;39(4):792–796.
Champagne BJ, Lee EC, Valerian B, et al. Incidence of colonic ischemia after repair of ruptured abdominal aortic aneurysm with endograft. J Am Coll Surg. 2007;204(4):597–602.
Cho JS, Gloviczki P, Martelli E, et al. Long-term survival and late complications after repair of ruptured abdominal aortic aneurysms. J Vasc Surg. 1998;27(5):813–819.
Choke E, Cockerill GW, Dawson J, et al. Hypoxia at the site of abdominal aortic aneurysm rupture is not associated with increased lactate. Ann N Y Acad Sci. 2006;1085:306–310.
Choke E, Cockerill GW, Dawson J, et al. Increased angiogenesis at the site of abdominal aortic aneurysm rupture. Ann N Y Acad Sci. 2006;1085:315–319.
Cowan JA Jr, Dimick JB, Henke PK, et al. Epidemiology of aortic aneurysm repair in the United States from 1993– 2003. Ann N Y Acad Sci. 2006;1085:1–10.
Duong C, Atkinson N. Review of aortoiliac aneurysms with spontaneous large vein fistula. ANZ J Surg. 2001;71: 52–55.
Fillinger MF, Marra SP, Raghavan ML, et al. Prediction of rupture risk in abdominal aortic aneurysm during observation: wall stress vs diameter. J Vasc Surg. 2003;37:724–732.
Fillinger MF, Racusin J, Baker RK, et al. Anatomic characteristics of ruptured abdominal aortic aneurysm on conventional CT scans: implications for rupture risk. J Vasc Surg. 2004;39:1243–1252.
Fillinger MF, Raghavan ML, Marra SP, et al. In vivo analysis of mechanical wall stress and abdominal aortic aneurysm rupture risk. J Vasc Surg. 2002;36:589–597.
Giles KA, Hamdan AD, Pomposelli FB, et al. Population-based outcomes following endovascular and open repair of ruptured abdominal aortic aneurysms. J Endovasc Ther. 2009;16(5):554–564.
Harkin DW, Dillon M, Blair PH, et al. Endovascular ruptured abdominal aortic aneurysm repair (EVRAR): a systematic review. Eur J Vasc Endovasc Surg. 2007;34(6):673–681.
Harris LM, Faggioli GL, Fiedler R, et al. Ruptured abdominal aortic aneurysms: factors affecting mortality rates. J Vasc Surg. 1991;14(6):812–818.
Hechelhammer L, Lachat ML, Wildermuth S, et al. Midterm outcome of endovascular repair of ruptured abdominal aortic aneurysms. J Vasc Surg. 2005;41(5):752–757.
Kimball EJ, Adams DM, Kinikini DV, et al. Delayed abdominal closure in the management of ruptured abdominal aortic aneurysm. Vascular. 2009;17(6):309–315.
Lambert ME, Baguley P, Charlesworth D. Ruptured abdominal aortic aneurysms. J Cardiovasc Surg (Torino). 1986;27(3):256–261.
Lederle FA, Johnson GR, Wilson SE, et al. Prevalence and associations of abdominal aortic aneurysm detected through screening. Aneurysm Detection and Management (ADAM) Veterans Affairs Cooperative Study Group. Ann Intern Med. 1997;126(6):441–449.
Lederle FA, Johnson GR, Wilson SE, et al. Rupture rate of large abdominal aortic aneurysms in patients refusing or unfit for elective repair. JAMA. 2002;287:2968–2972.
Markar RR, Badger SA, O’Donnell ME, et al. The effects of abdominal compartment hypertension after open and endovascular repair of a ruptured abdominal aortic aneurysm. J Vasc Surg. 2009;49(4):866–872.
Mehta M, Darling RC, Roddy SP, et al. Factors associated with abdominal compartment syndrome complicating endovascular repair of abdominal aortic aneurysms. J Vasc Surg. 2005;42(6):1047–1051.
Miani S, Mingazzini P, Piglionica R, et al. Influence of the rupture site of abdominal aortic aneurysms with regard to postoperative survival. J Cardiovasc Surg (Torino). 1984;25(5):414–419.
Nicholls SC, Gardner JB, Meissner MH, et al. Rupture in small abdominal aortic aneurysms. J Vasc Surg. 1998; 28(5):884–888.
Noel AA, Gloviczki P, Cherry KJ Jr, et al. Ruptured abdominal aortic aneurysms: the excessive mortality rate of conventional repair. J Vasc Surg. 2001;34(1):41–46.
Riesenman PJ, Farber MA. Endovascular repair of ruptured abdominal aortic aneurysm. In: Upchurch G, Criado E, eds. Aortic Aneurysms, Contemporary Cardiology. New York, NY: Humana Press, 2009.
Starnes BW, Quiroga E, Hutter C, et al. Management of ruptured abdominal aortic aneurysm in the endovascular era. J Vasc Surg. 2010;51:9–18.
The UK Small Aneurysm Trial Participants, Brown LC, Powell JT. Risk factors for aneurysm rupture in patients kept under ultrasound surveillance. Ann Surg. 1999;230:289–296.
Thomas PRS, Stewart RD. Abdominal aortic aneurysm. Br J Surg. 1988;75:733–736.
Veith FJ, Ohki T, Lipsitz EC, et al. Treatment of ruptured abdominal aneurysms with stent grafts: a new gold standard? Semin Vasc Surg. 2003;16:171–175.
Visser JJ, van Sambeek HM, Hamza TH, et al. Ruptured abdominal aortic aneurysms: endovascular repair versus open surgery—systematic review. Radiology. 2007;245(1):122–129.
Wakefield TW, Whitehouse WM Jr, Wu SC, et al. Abdominal aortic aneurysm rupture: statistical analysis of factors affecting outcome of surgical treatment. Surgery. 1982;91:586–596.