Erica R. Gross
Robert A. Cowles
Presentation
A 4-week-old, full-term male infant is brought to the emergency room by his parents at the instruction of the pediatrician. The mother reports that the baby has had several days of progressively worsening vomiting. The child acts hungry and drinks from a bottle without difficulty, but then, 30 to 60 minutes after eating, throws up most of his formula. The vomit is described as forceful and resembling formula. The baby is still having wet diapers, but not as frequently, and he is less active than usual. The pregnancy and the delivery were uncomplicated, and the infant is otherwise healthy and had been developing normally. Vital signs are remarkable for tachycardia. On physical examination, the baby is sleeping, but easily aroused. The anterior fontanelle is depressed. His abdomen is soft, nondistended, and appears nontender. No masses are palpable, but gastric waves are visible on the abdomen. No inguinal hernias are detected. A complete blood count and chemistry panel are ordered, and the results are given in Table 1. The patient is admitted for treatment of dehydration and electrolyte imbalance.
TABLE 1. Laboratory Values
Differential Diagnosis
It is important to differentiate bilious from nonbilious emesis in a neonate. Bilious emesis raises suspicion for malrotation with volvulus, a true surgical emergency that must be ruled out. From the presentation above, however, this infant has nonbilious emesis, leading to the following differential diagnoses: formula intolerance, gastroesophageal reflux, pyloric stenosis, pylorospasm, antral or duodenal web, and gastroparesis. Recurrent emesis can also be associated with metabolic disorders, inborn errors of metabolism, or elevated intracranial pressure due to brain tumors; however, emesis is rarely the isolated symptom in these systemic or neurologic disorders. Based on the history provided by the caregiver, pyloric stenosis can often be identified by the report that the infant is hungry after emesis, that the emesis is forceful and nonbilious, and that the baby is otherwise well. As emesis persists over days, lethargy may develop secondary to hypovolemia.
Workup
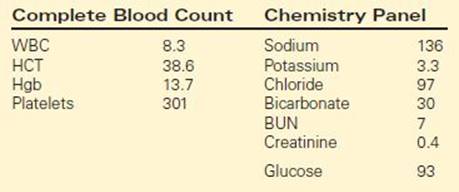
The clinical history and the physical exam alone are sufficient to make a diagnosis of pyloric stenosis in the majority of infants with the condition. Electrolytes and an abdominal ultrasound (Figure 1) confirm the diagnosis and evaluate the severity of the associated electrolyte imbalance. Ultrasound is very sensitive and specific for the diagnosis of pyloric stenosis. It is quick, available, inexpensive, noninvasive, and does not expose the child to radiation. A pylorus is considered hypertrophied if the thickness is ≥4 mm and if the pyloric channel length is ≥16 mm. In addition to these static measurements, the radiologist can often assess whether fluid is able to pass from the stomach into the duodenum. In the uncommon situation that the diagnosis is not clear after physical exam and ultrasound, an upper gastrointestinal series can show a distended stomach that cannot empty due to pyloric obstruction (“shoulder sign”) and a narrowed pyloric channel (“string sign”) (Figure 2).

FIGURE 1 • Abdominal ultrasound of hypertrophied pylorus. The patient has taken Pedialyte® by mouth, and the stomach appears the density of water (white asterisk). Mucosa is radiopaque and is seen outlining the pyloric channel (black asterisk). The hypertrophied pyloric muscle (M) measures 5 mm in thickness and 20 mm in length.
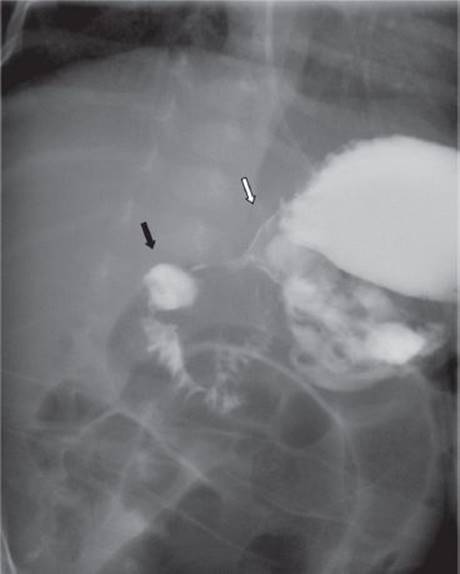
FIGURE 2 • Fluoroscopy after oral contrast. A string sign is seen between the antrum (white arrow) and the duodenal bulb (black arrow) indicating pyloric stenosis.
Discussion
Pyloric stenosis is the most common cause of gastric outlet obstruction in the 1 to 2 month old infant and has an incidence of 1/500 live births. This disorder is most common in male Caucasian infants. No single etiology has been identified, but more than one family member can be affected.
Diagnosis and Treatment
The ultrasound of this patient showed that the pyloric muscle is 5 mm thick and the channel length is 20 mm. After receiving these results, the diagnosis of pyloric stenosis is confirmed. Pyloric stenosis is not a surgical emergency. What should be addressed urgently, however, are the infant’s electrolyte abnormalities and hypovolemia. This child has a hypokalemic, hypochloremic metabolic alkalosis. Prolonged emesis results in dehydration, metabolic alkalosis, and hypochloremia. As dehydration worsens, aldosterone release stimulates the absorption of sodium and excretion of potassium in the urine in an attempt to maintain blood volume. Hypokalemia then causes the excretion of hydrogen and a paradoxical aciduria. In pyloric stenosis, the longer the duration of symptoms, the worse the electrolyte abnormalities become.
It is important to re-hydrate these patients aggressively and correct their electrolyte derangement prior to surgery. All infants diagnosed with pyloric stenosis should receive a 20 mL/kg bolus of normal saline (NS). Closely monitor urine output and administer additional fluid boluses until adequate urine output is achieved (1.5 to 2 mL/kg/h). Intravenous (IV) fluids, D5 ½ NS, should be infused at 1.5 times maintenance rate. Potassium supplementation can be given after urine output is re-established. Emesis should cease once the infant is taking nothing by mouth and, therefore, a nasogastric tube (NGT) is not necessary.
Surgical Approach to Open Pyloromyotomy
After the electrolyte imbalances have been corrected, the surgeon must relieve the gastric outlet obstruction. Medical treatment with atropine and endoscopic dilation has been attempted, but these therapies are inferior to surgical treatment. The standard of care is a Fredet-Ramstedt pyloromyotomy. Classically, the procedure was performed though a transverse right upper quadrant incision. A supraumbilical curvilinear incision is currently the preferred approach and will be described here (Table 2).
TABLE 2. Surgical Approach to Open Pyloromyotomy

The infant is placed in a supine position on the operating table, an NGT is placed, and general anesthesia is induced. After the infant is relaxed, the pylorus should be palpable in the epigastric region. An incision is made over this area in the right upper quadrant or above the umbilicus and is carried down through the muscle and fascia. The omentum is retracted downward to lift the transverse colon and reveal the stomach. Then, the antrum and the pylorus are identified and brought through the incision. Holding the duodenum with the left index finger supporting the pylorus, the serosa is incised approximately 2 mm proximal to the pyloric vein extending onto the gastric antrum (Figure 3). A blunt instrument is then used to divide the remaining circular muscle fibers without injuring, or violating, the underlying mucosa. When separation is complete, bulging of the mucosa is often seen, and the two sides of the pyloric muscle can be moved independently. The anesthesiologist can then fill the stomach with air to assess for perforation. The pylorus is then placed back into the abdomen, and the fascia and the skin are closed.
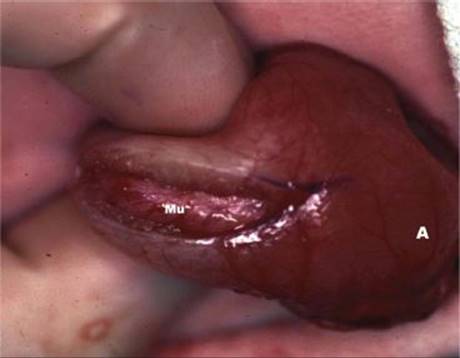
FIGURE 3 • Externalization of pylorus during open pyloromyotomy. The duodenum is being held, and the pyloromyotomy has been extended onto the gastric antrum. Mucosa (Mu) is seen ballooning through the completed incision.
If the pyloric incision is not deep enough or long enough, the child may show signs of persistent pyloric obstruction and require re-operation. Incomplete myotomy commonly occurs proximally, close to the gastric antrum. If the incision is too deep, through the mucosa, leakage of gastric or duodenal contents will occur. This most commonly occurs at the distal aspect of the incision, on the duodenal bulb. If this is identified intraoperatively, the mucosa should be closed with interrupted sutures and covered by omentum. In the case of perforation, the NGT can be left for 24 hours after repair to ensure gastric decompression.
Surgical Approach to Laparoscopic Pyloromyotomy
The first laparoscopic pyloromyotomy was described by Alain in 1991. Studies have shown that the laparoscopic approach is safe and effective and offers shorter operative times and hospital stays (Table 3).
TABLE 3. Surgical Approach to Laparoscopic Pyloromyotomy

General anesthesia is induced and the infant is placed transversely on the operating table. The monitor is placed at the baby’s head, across from the operating surgeon, who stands at the infant’s feet. An incision is made through the umbilicus or inferior to it. The Veress needle is then placed into the peritoneal cavity through this incision. A 4-mm trocar is inserted after inflation and a 4-mm, 30° scope is inserted. The abdomen is insufflated to 8 to 10 mm Hg. Two 3-mm trocars are then inserted in the right and left epigastrum under direct visualization. The proximal duodenum is grasped with the left hand and an arthrotomy knife is used to incise the serosa of the pylorus (Figure 4). The same landmarks are used for the pyloric incision. A laparoscopic pyloric spreader is used to spread the deep muscular layers (Figure 5). Again, the stomach should be insufflated with the duodenum occluded to evaluate for mucosal perforation. The carbon dioxide is then evacuated from the abdominal cavity, and the umbilical fascia and the skin are closed. The epigastric incisions can be closed with steri-strips or Dermabond.
FIGURE 4 • Serosal incision on pylorus (dashed line) during laparoscopic pyloromyotomy, carried proximally onto the antrum of the stomach (A).

FIGURE 5 • The gastric mucosa (Mu) is seen ballooning through the completed laparoscopic pyloromyotomy (pyloric muscle, M).
Special Intraoperative Considerations
While diagnostic error is unusual, a normal pylorus can be an unexpected intraoperative finding. This error in diagnosis is likely due to pylorspasm, a condition expected to resolve with bowel rest. When this occurs, patency of the stomach, pylorus, and proximal duodenum should be confirmed. This can be achieved by passage of an orogastric tube across the pylorus or by performing an intraoperative fluoroscopic gastric emptying study. In addition, other etiologies of upper gastrointestinal obstruction should also be ruled out.
Postoperative Management
Oral acetaminophen should provide adequate pain control. Oral intake can begin 4 to 6 hours postoperatively with small volumes of Pedialyte® and increased as tolerated. Once the infant is able to tolerate an adequate volume of Pedialyte®, he or she can be transitioned to breast milk or formula. It is common for an infant to vomit after surgery. If the infant vomits a feeding, allow 3 hours and re-attempt the same volume. Continue maintenance IV fluids until the infant is tolerating oral feedings well. Most patients are discharged 24 to 48 hours after surgery. Very few patients have long-term complications, such as gastric dysmotility. Any infant with persistent emesis 1 week after surgery should be evaluated for an incomplete myotomy or for severe gastroesophageal reflux.
Case Conclusion
The baby is adequately rehydrated, and electrolytes normalize over the first 36 hours. On hospital day 2, a laparoscopic pyloromyotomy is performed. Oral feeding with Pedialyte® is started 4 hours postoperatively and administered in increasing volumes every 3 hours. The baby has one episode of emesis, but does well, and is discharged home on the first postoperative day. At a 4-week follow-up appointment, the child is gaining weight appropriately according to growth curves.
TAKE HOME POINTS
· In any newborn, differentiate bilious from nonbilious vomiting. Bilious emesis is intestinal obstruction secondary to volvulus and a surgical emergency, unless proven otherwise.
· The morbidity associated with pyloric stenosis comes from the dehydration and electrolyte disturbances rather than the gastric outlet obstruction alone. The dehydration and hypokalemic, hypochloremic alkalosis require treatment with IV fluids and monitoring for correction prior to surgery.
· Postoperative vomiting is usually self-limited and does not indicate surgical failure unless it becomes persistent with failure to gain weight.
· Incomplete myotomy occurs proximally (gastric antrum). Perforation occurs distally (duodenal bulb).
SUGGESTED READINGS
Alain JL, Grousseau, Terrier G. Extramucosal pyloromyotomy by laparoscopy. Surg Endosc. 1991;5:174–175.
Aldridge RD, MacKinlay GA, Aldridge RB. Choice of incision: the experience and evolution of surgical management of infantile hypertrophic pyloric stenosis. J Laparoendosc Adv Surg Tech A. 2007;17(1):131–136.
Aspelund G, Langer JC. Current management of hypertrophic pyloric stenosis. Semin Pediatr Surg. 2007;16(1):27–33.
Leclair MD, Plattner V, Mirallie E, et al. Laparoscopic pyloromyotomy for hypertrophic pyloric stenosis: a prospective, randomized controlled trial. J Pediatr Surg. 2007;42(4):692–698.