Patricia C. Alves-Ferreira1 and Brooke Gurland1
(1)
Department of Colorectal Surgery A30, Cleveland Clinic, 9500 Euclid Avenue, Cleveland, OH 44195, USA
Brooke Gurland
Email: gurlanb@ccf.org
Abstract
Direct sphincter trauma or neuropathic injuries from vaginal deliveries are the principal causative factors in the development of fecal incontinence in women less than 40 years old [1]. Treatment options for the incontinent woman include anal sphincteroplasty, sacral nerve stimulation (SNS), artificial anal sphincter, posterior anal repair, and dynamic graciloplasty. Anal sphincteroplasty has been the preferred surgical treatment for the symptomatic female with an anatomically disrupted external anal sphincter (EAS) muscle. Short-term results report improved bowel incontinence as high as 90% [2, 3] with decreasing continence on long-term follow-up studies. Over the past decade, SNS has been utilized as a treatment modality with good results for patients with fecal incontinence with or without an anal sphincter defect [4–7], but SNS has only recently approved in the USA for this indication. The artificial anal sphincter, a silastic band surgically placed around the lower rectum, has been shown to improve bowel control. However, infection rates are reported at 33% [8, 9] leading to device failure or extrusion. Posterior anal repair is indicated for neuropathic incontinence and not very popular as its earlier results could not be duplicated. At best, only about 30% of the patients report improvement [10–12]. However, several long-term studies have shown favorable results [13, 14]. Dynamic graciloplasty is a complex procedure that involves gracilious muscle transposition and stimulation. This requires expertise; it is associated with a high morbidity and it is expensive [15–20]. This procedure is not an option in the United States since the stimulator used for muscle contraction is not commercially available. Other options include injectable bulking agents [21–24] into the anal sphincter which have shown to be effective in some studies, but are not presently approved in the USA for treatment of fecal incontinence. Transobturator insertion of a rectal sling has been reported with some success [25] and ongoing USA studies are in progress.
Introduction
Direct sphincter trauma or neuropathic injuries from vaginal deliveries are the principal causative factors in the development of fecal incontinence in women less than 40 years old [1]. Treatment options for the incontinent woman include anal sphincteroplasty, sacral nerve stimulation (SNS), artificial anal sphincter, posterior anal repair, and dynamic graciloplasty. Anal sphincteroplasty has been the preferred surgical treatment for the symptomatic female with an anatomically disrupted external anal sphincter (EAS) muscle. Short-term results report improved bowel incontinence as high as 90% [2, 3] with decreasing continence on long-term follow-up studies. Over the past decade, SNS has been utilized as a treatment modality with good results for patients with fecal incontinence with or without an anal sphincter defect [4–7], but SNS has only recently approved in the USA for this indication. The artificial anal sphincter, a silastic band surgically placed around the lower rectum, has been shown to improve bowel control. However, infection rates are reported at 33% [8, 9] leading to device failure or extrusion. Posterior anal repair is indicated for neuropathic incontinence and not very popular as its earlier results could not be duplicated. At best, only about 30% of the patients report improvement [10–12]. However, several long-term studies have shown favorable results [13, 14]. Dynamic graciloplasty is a complex procedure that involves gracilious muscle transposition and stimulation. This requires expertise; it is associated with a high morbidity and it is expensive [15–20]. This procedure is not an option in the United States since the stimulator used for muscle contraction is not commercially available. Other options include injectable bulking agents [21–24] into the anal sphincter which have shown to be effective in some studies, but are not presently approved in the USA for treatment of fecal incontinence. Transobturator insertion of a rectal sling has been reported with some success [25] and ongoing USA studies are in progress.
Unlike many of the alternatives mentioned, anal sphincteroplasty does not require expensive devices or postoperative maintenance required with implantable devices.
Sphincteroplasty can be performed in conjunction with other pelvic organ prolapse and urinary incontinence procedures without additional morbidity [26].
The complications associated with anal sphincteroplasty are low and will be discussed in this chapter.
Patient Evaluation
The following considerations are important when evaluating a patient with fecal incontinence for sphincteroplasty:
1.
Bowel habits
2.
Age
3.
Obesity
4.
Severity of symptoms
5.
Local physical findings
6.
Anal physiology and endoanal ultrasound
1.
Bowel habits: Loose or watery stools may results in fecal incontinence. Bulking agents and antidiarrheals to thicken and decrease frequency of bowel movements remain the first line therapies. Sphincteroplasty will not be effective in patients with loose and irregular stools.
2.
Age of the patient: Aging tissues are less likely to recover and maintain good quality over time. Several retrospective analyses suggest that older women have anorectal function that deteriorates over time [27, 28]. Advancing age may be associated with other pelvic floor defects including increased fibrosis and collagen deposition [29]. Other studies have found that age does not affect outcomes [2, 30]. Each case should individually take into consideration factors such as tissue quality and anal muscle contractility rather than biologic age alone.
3.
Obesity: A high body mass index has been associated with poorer outcome after sphincteroplasty [27]. Obese women may have other factors that can contribute to the incontinence such as excessive pelvic floor descent and diabetes.
4.
Severity of symptoms: Mild fecal symptoms or gas incontinence may persist even after successful sphincter repair. Patients should be counseled preoperatively regarding realistic postsurgical expectations. Nikiteas et al. [27] found that patients with severe symptoms undergoing primary repair reported the best outcomes.
5.
Local physical findings: Lax anal sphincter muscles or a patulous anus may be associated with mucosal or full thickness rectal prolapse. Decreased or no anal sphincter contractility noted on physical examination is a poor prognostic sign for sphincter repair as it represents a poorly functioning anal sphincter. Good muscle bulk was reported by Vaizey et al. [31] as an important selection criterion for best results.
6.
Anal physiologic testing: includes endoanal ultrasound and anal manometry. Low-squeeze pressure on anal manometry in conjunction with an anterior sphincter defect on endoanal ultrasound is the primary indication for sphincteroplasty. Other sonographic findings may include a variegated appearance of the EAS-indicating atrophic muscles, a very thin internal anal sphincter (IAS), or a large (more than 120°) defect of the EAS muscle. Pudendal nerve terminal latencies (PNTML) have also been used to evaluate the neurologic function of the anal sphincters, but the significance of prolonged PNTML are debated. In some studies, bilateral prolonged pudendal nerve latencies have been shown to be an important prognostic factor in patients undergoing anal sphincter repair [32].
Preoperative Management
Preoperative management includes:
· Appropriate patient selection
· Setting realistic postoperative continence expectations
· Mechanical bowel preparation 24 h before the onset of surgery
· A single dose of intravenous antibiotics administered prior to the surgery
Fecal diversion prior to sphincteroplasty has not been shown to improve outcomes and is not recommended. Hasegawa et al. [33] demonstrated equivalent sphincter reported outcomes between groups randomized to sphincteroplasty with or without diverting stoma. Patients in the stoma group suffered stoma-related complications.
Postoperative Management
Postoperative management requires keeping the stools soft, the area clean, and pain tolerable. Patients are kept overnight and discharged the following morning. There is no consensus on the routine administration of postoperative oral antibiotics at discharge. The patient is discharged on stool softeners.
Operative Management
Operative Technique and Results
A number of techniques have been described for sphincteroplasty and the choice is operator-dependant. Some authors advocate direct muscle apposition verses overlapping sphincteroplasty. Tjandra et al. [34] studied 36 patients with fecal incontinence caused by obstetric injuries, 12 underwent direct repair and 11 overlapping sphincter repair. At a median follow-up of 18 months, the functional results were significantly improved in both groups irrespective of the technique with improvement in incontinence in 75% and 73%, respectively.
For surgeons preferring overlapping sphincteroplasty, this can be performed en bloc thus avoiding separating the internal and external sphincters [35]. An anterior 120° curvilinear incision is made along the perineum to allow dissection and mobilization of the sphincter muscle and scar. It is important to preserve all scar tissue in order to anchor the sutures. En bloc repair involves mobilization of the EAS and IAS as a unit with overlap of the muscles complex. Other authors advocate anterior levatorplasty, IAS imbrication, and overlapping EAS repair [30]. The EAS is overlapped and mattress sutures are used to approximate the ends 2.0 PDS (Figs. 17.1, 17.2 and 17.3).
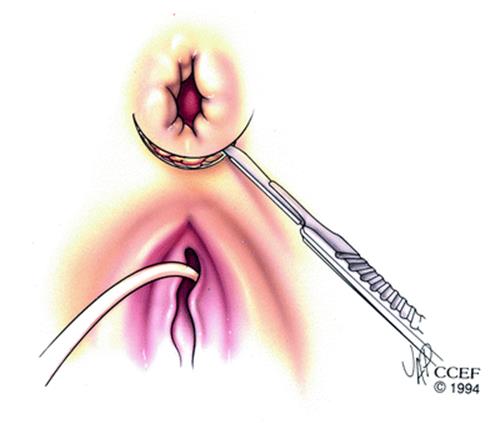
Fig. 17.1
A transverse incision along the perineum
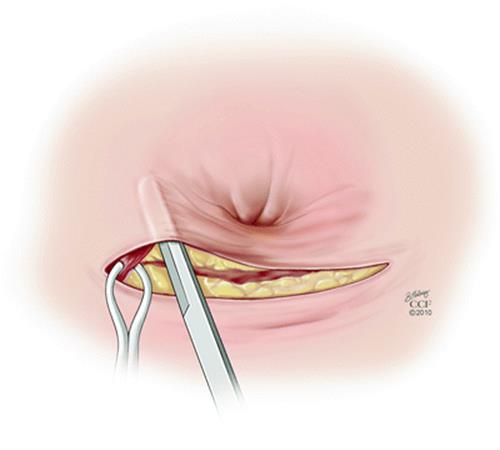
Fig. 17.2
The external sphincter is identified and grasped with the Allis clamp

Fig. 17.3
The external sphincter is overlapped and sutured into place
The edges of the wound are approximated in a V shape or longitudinally with interrupted 3.0 Vicryl mattress sutures. The center of the wound can be left open, a small drain inserted, or the wound can be closed. There are no studies that compare the functional results of en bloc compared to layered sphincteroplasty.
For patients with recurrent fecal incontinence after sphincteroplasty reevaluation and repeat repair is an alternative.
The rate of acceptance of the repeat sphincter repair is the same as that after a primary repair [31] and hence should be considered for selected patients with failed primary repairs.
Complications
Complications that can occur in the early postoperative period include hematoma or seroma formation. This can be treated by opening the wound and evacuating the contents. Warm soaks in a bathtub or sitz bath for 5–10 min help with pain relief by promoting relaxation of the pelvic floor muscles. Directing a handheld shower or peri-bottle at the wound facilitates hygiene and gently debrides the perineum. Nonsteroidal medications are encouraged over narcotics for pain relief to avoid the constipating side effects associated with narcotics. Antibiotics with gram positive, negative, and anaerobic coverage are selectively prescribed in the setting of wound cellulitis.
Late complications include abscess formation and wound dehiscence. Abscesses require drainage; wound breakdown usually heals secondarily and rarely requires secondary suturing. The patient’s main complaint after surgery is pain from the perineal wound. Table 17.1 reports previous studies and complications after sphincteroplasty surgery. Among the studies analyzed, the overall complications rate ranged from 9 to 31%. The outcomes are reported using different endpoints making comparisons difficult.
Table 17.1
Previous studies and complications after sphincteroplasty surgery
|
References |
N |
Age, mean (ranges) |
Repair |
Complications |
FU, mean (ranges) |
Outcomes, good/excellent N(%) |
|
Gibbs (1993) |
36 |
47 (20–74) |
OSR |
Total: 11 (31%) Urinary alteration: 8 Anal stenosis: 3 Congestive heart failure: 1 |
43 (4–114) |
24 (73) |
|
Rothbarth (2000) |
39 |
51 (29–74) |
Anterior sphincter repair |
Total: 5 (12.8%) UTI: 1 Pneumonia: 1 Wound infection: 3 |
39.3 (12–114) |
24 (62%) |
|
Halverson and Hull [26] |
44 |
38.5 (22–80)a |
OSR |
Wound infection: 4 (9%) |
62.5 (47–141)a |
30 (68) |
|
Mevik (2009) |
29 |
45 (6–77) |
Anterior sphincteroplasty |
Total: 7 (24%) Wound complications: 4 UTI: 2 Pudendal damage: 1 |
84 (74–185)a |
9 (53%) |
|
Oom et al. [39] |
172 |
58 (30–85)a |
OSR |
Total: 39 (23%) Wound complications: 35 Ileus: 2 DVT: 1 Lung embolism: 1 |
111 (12–207)a |
44 (37) |
|
Johnson et al. [40] |
33 |
36 (22–75)a |
OSR |
Wound infection: 6 (18%) |
103 (62–162)a |
21 (64) |
OSR overlapping sphincter repair; UTI urinary tract infection
aResults reported as median
Early symptom improvement is noted after sphincteroplasty [2, 3, 30, 36]. However, there is a deterioration of fecal incontinence over time with return to baseline in 10 years. Long-term 5 and 10 year follow-up reveals [3] a decline in continence and increasing fecal accidents [28, 37]. Barisic et al. [38] reported that 48% of their patients had good or excellent results, with patients totally continent or continent to solid and liquid feces after a median follow-up of 6.7 years, while Oom et al. [39] reported 37% after 9 years. Johnson et al. [40] reported improved results in 55% of patients and excellent results in just 9% of patients after 8.6 years. Halverson and Hull [26] reported 17% of patients totally continent after 5 years and 41% continent to liquid and solid stools.
Conclusion
Despite criticism regarding long-term functional results, sphincteroplasty is a viable option for women with sphincter trauma and associated fecal incontinence. Improvement after sphincteroplasty is noted but it is not to the level that it was before the sphincter injury. Complication rates are low and this procedure can be offered with limited morbidity.
References
1.
Sultan AH, Kamm MA, Bartram CI, Hudson CN. Anal sphincter trauma during instrumental delivery. Int J Gynaecol Obstet. 1993;43(3):263–70.PubMedCrossRef
2.
Simmang C, Birnbaum EH, Kodner IJ, Fry RD, Fleshman JW. Anal sphincter reconstruction in the elderly: does advancing age affect outcome? Dis Colon Rectum. 1994;37(11):1065–9.PubMedCrossRef
3.
Grey BR, Sheldon RR, Telford KJ, Kiff ES. Anterior anal sphincter repair can be of long term benefit: a 12-year case cohort from a single surgeon. BMC Surg. 2007;7:1.PubMedCrossRef
4.
Jarrett ME, Dudding TC, Nicholls RJ, Vaizey CJ, Cohen CR, Kamm MA. Sacral nerve stimulation for fecal incontinence related to obstetric anal sphincter damage. Dis Colon Rectum. 2008;51(5):531–7.PubMedCrossRef
5.
Chan MK, Tjandra JJ. Sacral nerve stimulation for fecal incontinence: external anal sphincter defect vs. intact anal sphincter. Dis Colon Rectum. 2008;51(7):1015–24; discussion 1024–5.
6.
Leroi AM, Parc Y, Lehur PA, et al. Efficacy of sacral nerve stimulation for fecal incontinence: results of a multicenter double-blind crossover study. Ann Surg. 2005;242(5):662–9.PubMedCrossRef
7.
Wexner SD, Coller JA, Devroede G, et al. Sacral nerve stimulation for fecal incontinence: results of a 120-patient prospective multicenter study. Ann Surg. 2010;251(3):441–9.PubMedCrossRef
8.
Wong WD, Jensen LL, Bartolo DC, Rothenberger DA. Artificial anal sphincter. Dis Colon Rectum. 1996;39(12):1345–51.PubMedCrossRef
9.
Christiansen J, Rasmussen OO, Lindorff-Larsen K. Long-term results of artificial anal sphincter implantation for severe anal incontinence. Ann Surg. 1999;230(1):45–8.PubMedCrossRef
10.
Engel AF, van Baal SJ, Brummelkamp WH. Late results of postanal repair for idiopathic faecal incontinence. Eur J Surg. 1994;160(11):637–40.PubMed
11.
Jameson JS, Speakman CT, Darzi A, Chia YW, Henry MM. Audit of postanal repair in the treatment of fecal incontinence. Dis Colon Rectum. 1994;37(4): 369–72.PubMedCrossRef
12.
Setti Carraro P, Kamm MA, Nicholls RJ. Long-term results of postanal repair for neurogenic faecal incontinence. Br J Surg. 1994;81(1):140–4.PubMedCrossRef
13.
Abbas SM, Bissett IP, Neill ME, Parry BR. Long-term outcome of postanal repair in the treatment of faecal incontinence. ANZ J Surg. 2005;75(9):783–6.PubMedCrossRef
14.
Mackey P, Mackey L, Kennedy ML, et al. Postanal repair—do the long-term results justify the procedure? Colorectal Dis. 2010;12(4):367–72.PubMedCrossRef
15.
Sielezneff I, Malouf AJ, Bartolo DC, Pryde A, Douglas S. Dynamic graciloplasty in the treatment of patients with faecal incontinence. Br J Surg. 1999;86(1):61–5.PubMedCrossRef
16.
Eccersley AJ, Williams NS. Dynamic graciloplasty for severe anal incontinence. Br J Surg. 1998;85(8): 1158–9.PubMed
17.
Rosen HR, Novi G, Zoech G, Feil W, Urbarz C, Schiessel R. Restoration of anal sphincter function by single-stage dynamic graciloplasty with a modified (split sling) technique. Am J Surg. 1998;175(3):187–93.PubMedCrossRef
18.
Christiansen J, Rasmussen OO, Lindorff-Larsen K. Dynamic graciloplasty for severe anal incontinence. Br J Surg. 1998;85(1):88–91.PubMedCrossRef
19.
Baeten CG, Geerdes BP, Adang EM, et al. Anal dynamic graciloplasty in the treatment of intractable fecal incontinence. N Engl J Med. 1995;332(24):1600–5.PubMedCrossRef
20.
Mander BJ, Wexner SD, Williams NS, et al. Preliminary results of a multicentre trial of the electrically stimulated gracilis neoanal sphincter. Br J Surg. 1999;86(12):1543–8.PubMedCrossRef
21.
Chan MK, Tjandra JJ. Injectable silicone biomaterial (PTQ) to treat fecal incontinence after hemorrhoidectomy. Dis Colon Rectum. 2006;49(4):433–9.PubMedCrossRef
22.
Davis K, Kumar D, Poloniecki J. Preliminary evaluation of an injectable anal sphincter bulking agent (Durasphere) in the management of faecal incontinence. Aliment Pharmacol Ther. 2003;18(2):237–43.PubMedCrossRef
23.
Kenefick NJ, Vaizey CJ, Malouf AJ, Norton CS, Marshall M, Kamm MA. Injectable silicone biomaterial for faecal incontinence due to internal anal sphincter dysfunction. Gut. 2002;51(2):225–8.PubMedCrossRef
24.
Vaizey CJ, Kamm MA. Injectable bulking agents for treating faecal incontinence. Br J Surg. 2005;92(5): 521–7.PubMedCrossRef
25.
Yamana T, Takahashi T, Iwadare J. Perineal puborectalis sling operation for fecal incontinence: preliminary report. Dis Colon Rectum. 2004;47(11): 1982–9.PubMedCrossRef
26.
Halverson AL, Hull TL. Long-term outcome of overlapping anal sphincter repair. Dis Colon Rectum. 2002;45(3):345–8.PubMedCrossRef
27.
Nikiteas N, Korsgen S, Kumar D, Keighley MR. Audit of sphincter repair. Factors associated with poor outcome. Dis Colon Rectum. 1996;39(10):1164–70.PubMedCrossRef
28.
Zutshi M, Tracey TH, Bast J, Halverson A, Na J. Ten-year outcome after anal sphincter repair for fecal incontinence. Dis Colon Rectum. 2009;52(6):1089–94.PubMedCrossRef
29.
Keighley M, Williams N. Fecal incontinence. In: Keighley M, editor. Surgery of the anus, colon and rectum, vol. 1. Philadelphia: WB Saunders; 2001.
30.
Evans C, Davis K, Kumar D. Overlapping anal sphincter repair and anterior levatorplasty: effect of patient’s age and duration of follow-up. Int J Colorectal Dis. 2006;21(8):795–801.PubMedCrossRef
31.
Vaizey CJ, Norton C, Thornton MJ, Nicholls RJ, Kamm MA. Long-term results of repeat anterior anal sphincter repair. Dis Colon Rectum. 2004;47(6): 858–63.PubMedCrossRef
32.
Gilliland R, Altomare DF, Moreira Jr H, Oliveira L, Gilliland JE, Wexner SD. Pudendal neuropathy is predictive of failure following anterior overlapping sphincteroplasty. Dis Colon Rectum. 1998;41(12): 1516–22.PubMedCrossRef
33.
Hasegawa H, Yoshioka K, Keighley MR. Randomized trial of fecal diversion for sphincter repair. Dis Colon Rectum. 2000;43(7):961–4; discussion 964–5.
34.
Tjandra JJ, Han WR, Goh J, Carey M, Dwyer P. Direct repair vs. overlapping sphincter repair: a randomized, controlled trial. Dis Colon Rectum. 2003;46(7):937–42; discussion 942–3.
35.
Galandiuk S, Roth LA, Greene QJ. Anal incontinence-sphincter ani repair: indications, techniques, outcome. Langenbecks Arch Surg. 2009;394(3):425–33.PubMedCrossRef
36.
Karoui S, Leroi AM, Koning E, Menard JF, Michot F, Denis P. Results of sphincteroplasty in 86 patients with anal incontinence. Dis Colon Rectum. 2000; 43(6):813–20.PubMedCrossRef
37.
Bravo Gutierrez A, Madoff RD, Lowry AC, Parker SC, Buie WD, Baxter NN. Long-term results of anterior sphincteroplasty. Dis Colon Rectum. 2004;47(5): 727–31; discussion 731–2.
38.
Barisic GI, Krivokapic ZV, Markovic VA, Popovic MA. Outcome of overlapping anal sphincter repair after 3 months and after a mean of 80 months. Int J Colorectal Dis. 2006;21(1):52–6.PubMedCrossRef
39.
Oom DM, Gosselink MP, Schouten WR. Anterior sphincteroplasty for fecal incontinence: a single center experience in the era of sacral neuromodulation. Dis Colon Rectum. 2009;52(10):1681–7.PubMedCrossRef
40.
Johnson E, Carlsen E, Steen TB, Backer Hjorthaug JO, Eriksen MT, Johannessen HO. Short- and long-term results of secondary anterior sphincteroplasty in 33 patients with obstetric injury. Acta Obstet Gynecol Scand. 2010;89(11):1466–72.PubMedCrossRef