Kamran P. Sajadi1 and Sandip P. Vasavada2, 3
(1)
Division of Urology, Oregon Health and Science University, Portland, OR, USA
(2)
Center for Female Urology and Reconstructive Pelvic Surgery, Cleveland Clinic Main Campus, Mail Code Q10-1, 9500 Euclid Avenue, Cleveland, OH 44195, USA
(3)
Glickman Urological and Kidney Institute, Cleveland Clinic, Cleveland, OH, USA
Sandip P. Vasavada
Email: vasavas@ccf.org
Abstract
The transvaginal approach to the vaginal apex is commonly performed for pelvic organ prolapse, and offers a minimally invasive alternative to the transabdominal route. Nonetheless, as with any major surgical procedure, there are complications specific to these approaches. Complications common to all procedures, including urinary tract infection, wound infection, venous thrombosis, and neuropraxias, are discussed in Chap. 2. We will focus on major complications related to transvaginal approaches to apical prolapse repair including ureteral or bowel injury, hemorrhage, and peripheral nerve injury.
Introduction
The transvaginal approach to the vaginal apex is commonly performed for pelvic organ prolapse, and offers a minimally invasive alternative to the transabdominal route. Nonetheless, as with any major surgical procedure, there are complications specific to these approaches. Complications common to all procedures, including urinary tract infection, wound infection, venous thrombosis, and neuropraxias, are discussed in Chap. 2. We will focus on major complications related to transvaginal approaches to apical prolapse repair including ureteral or bowel injury, hemorrhage, and peripheral nerve injury.
Ureteral Injury and Obstruction
Ureteral obstruction is a known complication of uterosacral vaginal vault suspension (USVVS), usually related to kinking of the ureter during plication of the uterosacral ligament to the vaginal cuff. The distal uterosacral ligament is intimately involved with the cardinal ligament—which contains the uterine vessels—and lies in close proximity to the ureter. Anatomic studies of the ligament demonstrate that the middle and proximal segments may be ideal for use in apical suspension, with the mean ± SD distance from the ureter 0.9 ± 0.4 cm distally, 2.3 ± 0.9 cm in the middle segment, and 4.1 ± 0.6 cm proximally (Fig. 5.1) [1]. Obstruction can occur in up to 11% of procedures [2], but the incidence is markedly reduced by performing cystoscopy at the conclusion of the procedure. Indigo carmine is injected intravenously, and cystoscopy is performed to visualize efflux of blue dye from each ureter. If a strong ureteral jet is seen from both sides after the vault suspension has been completed, then ureteral obstruction is unlikely. A study of hysterectomies showed that cystoscopy is cost-effective when the rate of injury is at least 2% [3], and intraoperative as opposed to postoperative diagnosis of ureteral obstruction substantially reduces morbidity [4].
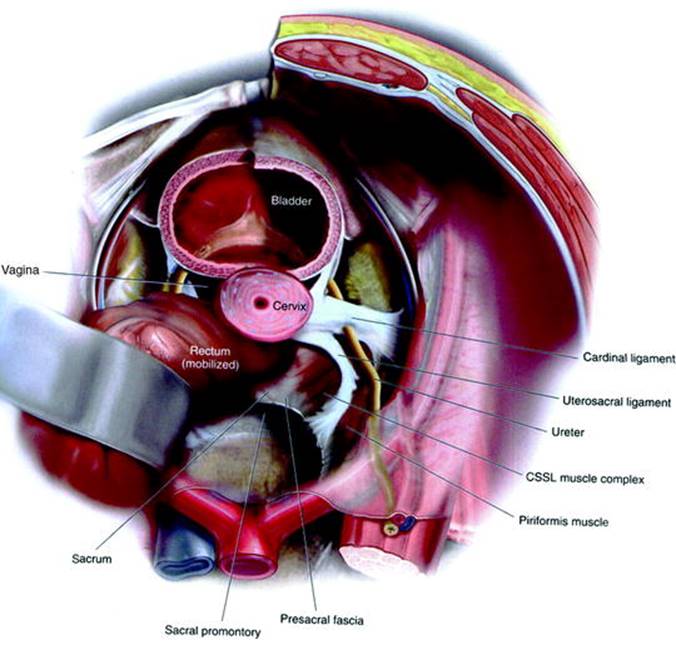
Fig. 5.1
Abdominal view illustrating the relationship between the ureter and the uterosacral ligament. Proceeding cephalad, the uterosacral ligament proceeds medially while the ureter proceeds laterally. Vault suspension to the proximal third therefore has the lowest rate of ureteral obstruction. Illustration from [38]
Ureteral Obstruction: Intraoperative Presentation
When there is no efflux from one or both sides, it is important to have a clear plan and algorithm in place for diagnosis and management. First, consider the patient scenario. Reevaluate the patient’s history to consider if she has had a prior nephrectomy or ureteral reimplant, in the latter case the ureter may efflux from a different position. If the patient has had any previous abdominal imaging, it can be helpful in identifying the occasional case of a prior nephrectomy or congenital absence of the ipsilateral kidney. In addition, confirm the time of administration of indigo carmine with the anesthesiologist or nurse, as early delivery may mean that all dye has been excreted, or late delivery may mean you have not watched the ureteral orifice long enough. Many different maneuvers have been attempted to promote more rapid excretion of the dye. Most commonly, ensuring adequate hydration by the anesthetist and/or administrating a diuretic such as furosemide may promote more rapid renal excretion of indigo. Resuming a level position or reverse Trendelenburg to encourage gravitational drainage has also been performed, although these reports are anecdotal.
Once sufficient time has passed to confirm a lack of excretion from one or both sides, there are a few ways to proceed. One option is to cut the more distal (i.e., more lateral) uterosacral plication suture (the uterosacral ligament is closest to the ureter distally) out of the vaginal cuff, and observe if efflux then occurs. With an assistant, it is possible to cut this suture while the cystoscope is still in place. If this suture was the cause, brisk efflux will usually immediately ensue and most pelvic reconstructive surgeons would not attempt to replace the suture in this situation, believing the remaining suspension sutures to be adequate. If efflux does not ensue, remove the remaining sutures on that side, one at a time, proceeding from the most lateral and caudad to the most cranial and medial. It is important to remember, however, that if a concomitant anterior colporrhaphy was performed, that procedure also carries a risk of ureteral obstruction, and it may be prudent to remove those sutures first, because it is easier to repeat an anterior colporrhaphy than an apical suspension.
Occasionally, there will still be a lack of efflux even after removal of all potentially offending sutures. If the patient lacks preoperative upper urinary tract imaging or sufficient historical reason to explain the lack of efflux, a urologic consultation may be prudent. The most common obstacle to performing retrograde ureterography in such cases is that these patients are often not positioned appropriately on the bed or on an appropriate operative table for pelvic fluoroscopy. Therefore, many urologists will attempt blind passage of a wire or ureteral catheter into the ureter to assure patency. If this is done, a flexible tipped, soft hydrophilic wire should be used, and even then there is risk of converting a ureteral kink or obstruction into a ureteral perforation. Making the extra effort to obtain a C-arm and repositioning the patient can significantly improve patient safety. With retrograde ureteropyelography, the urologist can accurately assess the patency of the ureter and make a decision whether or not a stent should be placed. If there is a suspicion of injury and a stent can be passed, it should be left in place for a minimum of 4–6 weeks [4].
Ureteral Obstruction: Postoperative Presentation
Ureteral injury is a potential complication of uterosacral colpopexy even when intraoperative cystoscopy reveals bilateral ureteral efflux. So-called “delayed obstruction” may occur due to excessive scarring between the uterosacral plication and the distal ureter, due to compromise of the ureteral blood supply or perhaps because of inadequate intraoperative examination for efflux. Ureteral obstruction presents in the acute postoperative period with flank pain, nausea and vomiting, and potentially fever. The diagnosis should be confirmed with imaging, and the study of choice in patients with normal renal function is CT Urography (CTU, see Fig. 5.2). The severity of hydronephrosis, site of ureteral obstruction, presence and location of any extravasation, presence or size of a potential urinoma, and the status of the contralateral kidney can all be assessed with a CTU. Once identified, in the acute postoperative period (up to 7 days), cutting the offending colpopexy sutures may be sufficient to relieve the obstruction. It is usually ideal to perform this in the operating room for several reasons. Aside from patient comfort, under anesthesia cystoscopy and retrograde ureteropyelography can be performed at the same time, to confirm patency of the ureter following removal of the suture(s). In addition, given the potential for ureteral edema and the severity of the obstruction, many urologists would choose to place an indwelling ureteral stent after relief of the obstruction. With further delay in presentation or failure to unobstruct in this manner, open abdominal or laparoscopic ureterolysis and reimplant is often necessary, although transvaginal ureterolysis and retrograde stenting has also been reported [5]. In a meta-analysis of USVVS, there was a 1.8% rate of ureteral obstruction, of which 2/3 resolved with suture removal, and the remainder required ureteral reimplantation [6].
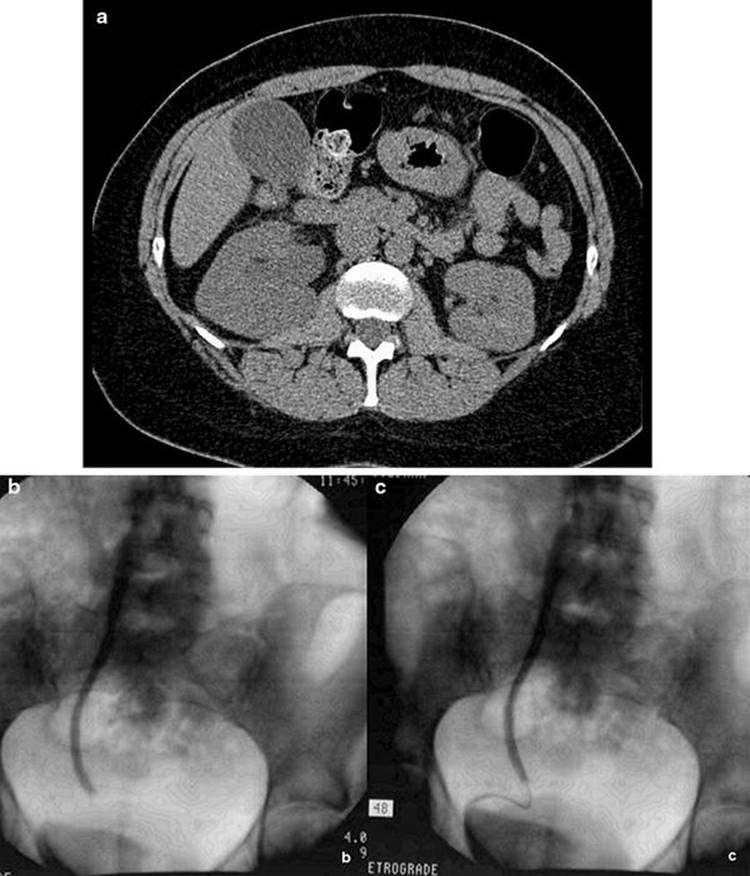
Fig. 5.2
A woman with postoperative suspicion of ureteral injury is found to have right hydronephrosis on a CT (a). Right retrograde ureterography demonstrates medial deviation of the distal ureter, and the distal ureter is not opacified (b). A wire was successfully passed (c), over which a stent was then placed (photograph courtesy of Howard Goldman, MD, Cleveland Clinic, OH)
Other Apical Suspensions
The sacrospinous ligament is fairly posterior to the path of the ureter, and so sacrospinous ligament fixation (SSLF) is rarely associated with ureteral obstruction. A cohort of women undergoing SSLF found a 3.5% rate of temporary ureteral obstruction [7]. Although 88 of the 200 women studied underwent simultaneous anterior colporrhaphy, all of the ureteral obstructions were right-sided, as were the apical suspensions, implicating the latter. Although no interventions were required, the study does not explain the presentation or management of these patients. The iliococcygeus suspension has recently gained popularity, with one of the purported benefits being the lack of vital structures in the immediate vicinity of the iliococcygeal fascia. The risk of ureteral injury and obstruction in this procedure is therefore theoretically low, and to our knowledge none have been reported [8].
Hemorrhage
Sacrospinous Ligament Colpopexy or Hysteropexy
Significant hemorrhage is more common with sacrospinous colpopexy or hysteropexy than with other transvaginal apical repairs, largely due to the ligament’s delicate anatomic location. The sacrospinous ligament, running for about 5 or 6 cm [9] between the ischial spine laterally and the lateral border of the sacrum medially, forms the inferior border of the greater sciatic foramen, through which pass the superior gluteal vessels above the piriformis muscle, and the inferior gluteal and internal pudendal vessels below. The internal pudendal neurovascular bundle runs just inferior and medial to the ischial spine, and therefore suspension sutures should be sufficiently medial and superficial to these structures (Fig. 5.3). Similarly, dissection and suture placement should avoid the cephalad border of the sacrospinous ligament in order to minimize risk to the gluteal vessels and sciatic nerve. Although there is variation in the distance between the ischial spine and pudendal nerve and vessels and the sciatic nerve [10], in general these structures can be avoided by placing suspension sutures in the middle or medial third of the ligament, avoiding the cephalad border of the ligament, and placing sutures through the ligament alone, and not deep through the underlying coccygeus muscle.
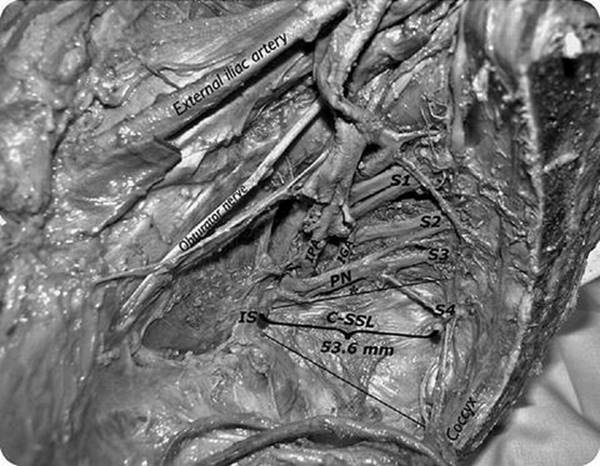
Fig. 5.3
Sagittal cadaveric dissection demonstrating the relationship of the coccygeus-sacrospinous ligament (C-SSL) to the sacral nerve roots and pudendal nerve (PN). Important vascular structures include the internal pudendal artery (IPA) and the more medial inferior gluteal artery (IGA). Source: Roshanravan et al. [28]
Sacrospinous ligament colpopexy or hysteropexy can be performed unilaterally (usually to the right, to avoid the sigmoid colon) or bilaterally, and is approached through either the anterior or posterior vaginal wall. In either case, copious hydrodistension of the vaginal mucosa, especially with epinephrine-containing solutions, can provide not only hemostasis, but also better anatomic delineation of the plane between the vaginal mucosa and muscularis. Meticulous attention to this plane reduces the likelihood of significant bleeding and injury to adjacent viscera (the bladder anteriorly or the rectum posteriorly). SSLF is associated with deviation of vaginal axis posteriorly (and laterally, in the case of unilateral fixation), and therefore increased risk of anterior vaginal wall prolapse recurrence, and many surgeons feel the anterior approach reduces this deviation. In a retrospective cohort study comparing both approaches, there was improved apical and anterior vaginal wall support in the anterior group, and a lower reoperation rate [11]. No randomized controlled studies have compared the two.
We believe the posterior approach offers better control of bleeding because of an increased space in which to place hand-held retractors (Breisky-Navratil) for optimal visualization of the ligament and surrounding structures. With direct visualization, the judicious placement of small surgical clips on bleeding vessels can prevent significant hemorrhage. A comparison of suture placement methods found increased complications with suture placement by palpation as opposed to direct visualization, especially with blood loss and nerve-related complications [12]. Of note, the group criticized the use of a Deschamps needle driver by palpation as opposed to standard needle driver under direct vision, however we use the Deschamps needle driver under direct visualization, and feel these differences are due to visualization as opposed to the type of driver.
Bleeding Management
With appropriate judgment, small, slow venous bleeding where the source cannot be directly visualized can be managed by completing the operation, closing, and sufficiently packing the vagina so that pressure can be maintained at least overnight. Alternatively, when significant bleeding occurs, the anesthetist should be notified to monitor hemodynamics, and obtain a blood type and screen or cross-match if not done preoperatively. The extraperitoneal approach of this operation offers the advantage of easy packing with sponges and application of pressure transvaginally to slow the bleeding and allow appropriate time for decision-making and definitive management. After packing the vagina and holding pressure for sufficient time (at least 5 min if bleeding is significant), slowly reexpose the area to assess if the source is visible and amenable to suture or clip application. Arterial bleeding is easier to identify in this situation, but occasionally significant venous ooze makes source identification extremely difficult.
When bleeding is considerable and/or there is question of hemodynamic compromise, then definitive vascular control is necessary. If suspension sutures have been placed or can be placed quickly, then vault suspension may be completed as rapidly as possible. Attempting to control the bleeding via an open retropubic or abdominal route is invasive, time-consuming, and often unsuccessful, as the expanding retropubic hematoma makes identification of the source, and preservation of important surrounding structures, difficult. Moreover, extensive “surgically significant” collateral circulation exists, and therefore control of the internal iliac artery may be insufficient to stop bleeding [9, 13]. The best definitive management in this case is angiography, either via an interventional radiologist or, if an interventionalist is not readily available, a vascular surgeon. While awaiting the consultation, adequate resuscitation with crystalloid and, once available, packed red cells is prudent. Continue to pack the vagina and apply pressure until arrangements for definitive management have been made.
Occasionally, a patient will present following SSLF with significant postoperative hemorrhage, in which case a similar protocol is usually effective. Again, transvaginal or transabdominal exploration to control bleeding is often difficult and ineffective. Placement of a vaginal packing and attention to hemodynamic monitoring and resuscitation, if necessary, are the next steps while arrangements are made for angiographic control of hemorrhage.
Iliococcygeus Vaginal Vault Suspension
As the iliococcygeus lies distal and anterior to the sacrospinous ligament, and requires less dissection and exposure for placement of suspension sutures, many feel there is less hemorrhage reported with this approach. One comparative study of iliococcygeal vs. sacrospinous fixation found similar rates of hemorrhage and transfusion between the two approaches [14]. Another study of iliococcygeal suspension found a mean estimated blood loss of 358 mL, and 3 of 110 patients had hemorrhage >750 mL [15]. To our knowledge, however, there have been fewer surgical series, anatomical studies, and no randomized controlled trials regarding the iliococcygeal suspension and therefore, much of our knowledge is anecdotal.
Uterosacral Vaginal Vault Suspension
Hemorrhage requiring blood transfusion during USVVS is about 1.3% [6]. Prompt attention to bleeding is necessary because this procedure is intraperitoneal and therefore it can be difficult to control by tamponade alone. In addition, when bleeding is encountered during USVVS, it is important to remember that the most common sources may be the vascular pedicles if a concomitant vaginal hysterectomy was performed. For this reason, leaving suture tags on the pedicles for easy retrieval and examination can be invaluable. Again, the distal uterosacral ligament lies close to the uterine vessels, and therefore targeting suspension sutures towards the middle or proximal uterosacral ligament can minimize bleeding. Minor to moderate bleeding from placement of the uterosacral ligament suture can be controlled by applying tension to the suture until the end of the operation, at which point it can be tied down to stop the bleeding.
Bowel Injury
Sacrospinous Ligament Fixation and Iliococcygeal Suspension
As extraperitoneal operations, the SSLF and iliococcygeal suspension offer the potential advantage of less small bowel and colonic injury. However, the procedures themselves involve perforation into and dissection of the pararectal space and so rectal injury is a known risk that patients should be counseled about preoperatively. Unilateral right-sided SSLF is preferred because this avoids the recto-sigmoid junction, and makes retraction of the rectum easier. Through the posterior approach, copious hydrodissection in the proper plane between the vaginal mucosa and muscularis makes dissection of the vaginal wall off the rectum easier. After beginning the dissection sharply, with adequate hydrodissection the rectum can usually be bluntly swept off the vagina in patients who have not had previous repairs. Caution is advised when a previous posterior colporrhaphy has been performed, as there is an increased risk of rectal injury in that setting. When there is uncertainty about the plane, keeping a finger behind the vaginal mucosa is helpful on the vaginal aspect, and occasionally a second glove can be worn and a finger placed in the rectum to guide dissection.
After the rectum has been swept off the vagina, when perforating into the pararectal space, the sacrospinous ligament can be exposed by palpating the ischial spine and sweeping the finger medially. When using Breisky-Navratil retractors to expose the ligament, great care is needed in placing these retractors to avoid rectal laceration. First, manual dissection must ensure there is adequate space for the first (thinner) retractor. Second, place the retractor laterally, against the pelvic sidewall, and then rotate it 180° so that it retracts the rectum, rather than inserting it alongside the rectum, which may result in a tear.
Rectal laceration or perforation has been reported in 0.4–4% of SSLF [16–20]. Most rectal injuries occur in the distal anterior rectum upon initial dissection [17, 19]. These injuries are usually small (<2 cm) and can managed with primary closure in 2–3 layers, copious irrigation, and postoperative bowel rest for 2 or 3 days [17]. Many vaginal reconstructive surgeons may repair these injuries themselves, but obtaining a colorectal surgery consult intraoperatively can be helpful for technical and medicolegal reasons. If the injury is readily identifiable, easily accessible, and can be closed in multiple layers in a tension-free manner in a healthy patient without a history of irradiation, then diverting colostomy is usually not necessary.
For sutures that are not placed under direct vision, such as with a suture-capturing device, inadvertent placement of the suture through the rectum can occur. Usually this can be identified intraoperatively with a careful digital rectal examination. If suture is palpable, remove it and replace the suture, and usually there are no sequelae. Identifying rectal placement of suture is essential; however, as there are scattered case reports of significant pararectal infectious complications associated with SSLF [21, 22].
Uterosacral Vaginal Vault Suspension
Despite the intraperitoneal nature of the operation, bowel injury is rare with USVVS, and is reported in less than 1% of cases [6]. Small bowel obstruction (SBO) in particular is very rare, and was first reported in a series in 2006 [23]. Three patients presented with significant nausea and vomiting on postoperative days 1–14 and were found to have possible SBO [23]. After failing conservative management, all subsequently underwent laparoscopy. The source of the obstruction was adhesions in two of the patients, and a prolene suture in the third. One of the patients requiring significant adhesiolysis underwent small bowel resection and enteroenterostomy due to enterotomies during dissection.
Despite the low reported rate of small bowel injury or obstruction, there are several important technical considerations required to keep this rate low. When exposing the uterosacral ligaments, packing of the bowel with tagged, counted laparotomy sponges is usually necessary. The peritoneum should be carefully inspected for abdominal adhesions, the sponges advanced slowly and gently to avoid enterotomies, and gentle retraction on the sponges to minimize trauma. Similarly, these packs should be removed slowly and carefully, and counted, after placing suspension sutures. If performing culdoplasty, care in closing the parietal peritoneum can avoid capturing bowel in the closure.
Evisceration
Small bowel evisceration has been reported following vaginal hysterectomy [24, 25], as well as following transvaginal enterocele and SSLF [26]. Evisceration is a surgical emergency, and although some have had success through a transvaginal route alone, usually a transabdominal route is helpful to assess the viability of the small bowel involved [27].
Neurologic and Pain Complications
Sacrospinous Ligament Fixation
Knowledge and understanding of the nearby anatomy is essential. Suture placement in the middle third of the sacrospinous ligament avoids the region of the pudendal nerve, and staying caudad to the greater sciatic foramen avoids the sciatic nerve [10]. Anatomic studies also reveal, however, that nerves to the coccygeus and levator ani actually pass ventral to the ligament in the middle segment, where sutures are typically placed, and may be encountered during dissection and suture placement [28]. Histologic studies of the ligament itself confirm nerves within the substance of the ligament, especially in the middle segment [29], which explains the potential for pain after this operation. In fact, another cadaveric study found the only nerve-free region of the ligament to be the medial third [30], which is more medial than is often described in the operation.
Buttock or tailbone pain, which may be due to involvement of peripheral nervous branches or to the tension on the ligament, occurs in around 6–14% of patients after SSLF [7, 31, 32]. The majority of cases of postoperative buttock pain resolve spontaneously or with medical management, although in one report 3 of 18 patients with postoperative pain subsequently had chronic pain [32]. Persistent pelvic and perineal pain should raise suspicion of potential pudendal nerve entrapment. History and physical exam will indicate onset of symptoms coincident with surgery, in the sensory distribution of the pudendal nerve. Although removal of the offending suture should not be delayed, a report of a patient who presented 2 years after SSLF noted that they still had complete relief after suture removal even after that length of time from surgery [33].
Uterosacral Vaginal Vault Suspension
The intraperitoneal nature of this operation makes direct visualization of retroperitoneal vasculature and nerves difficult, and therefore a thorough anatomic understanding is necessary. Assessing the position of the ischial spine allows avoidance of the pudendal nerve, which is usually sufficiently far from the uterosacral ligaments [34]. On the other hand, the sacral nerve routes are susceptible during USVVS. A cadaveric study demonstrated that by tenting the uterosacral ligaments distally and ventrally using an Allis clamp before suture placement, the sacral nerve roots can be avoided [34]. Although tension on the ligament is also distributed to the ureter, this effect is seen most dramatically distally, and can be avoided by proximal suture placement [1]. The sacral nerve roots as well as the intrapelvic portion of the sciatic nerve are vulnerable to entrapment during uterosacral suspension, which can explain postoperative pain in some patients [35]. Sensory neuropathies have been reported in 3.8% of patients [36]. Pain tends to present in the acute postoperative period, in the distribution of the S1 through S3 nerve roots, and can be successfully managed by removal of the ipsilateral suspension suture, or with medical management [36, 37].
Summary
Transvaginal apical suspensions can have serious complications, including hemorrhage, visceral injury, and neurologic sequelae. Intimate knowledge of the relevant anatomy can, however, reduce these risks, and the majority of complications can be successfully treated with proper identification and management.
References
1.
Buller JL, Thompson JR, Cundiff GW, et al. Uterosacral ligament: description of anatomic relationships to optimize surgical safety. Obstet Gynecol. 2001;97:873–9.PubMedCrossRef
2.
Gustilo-Ashby AM, Jelovsek JE, Barber MD, et al. The incidence of ureteral obstruction and the value of intraoperative cystoscopy during vaginal surgery for pelvic organ prolapse. Am J Obstet Gynecol. 2006;194:1478–85.PubMedCrossRef
3.
Visco AG, Taber KH, Weidner AC, et al. Cost-effectiveness of universal cystoscopy to identify ureteral injury at hysterectomy. Obstet Gynecol. 2001;97(5 Pt 1):685–92.PubMedCrossRef
4.
Kim JH, Moore C, Jones JS, et al. Management of ureteral injuries associated with vaginal surgery for pelvic organ prolapse. Int Urogynecol J Pelvic Floor Dysfunct. 2006;17:531–5.PubMedCrossRef
5.
Siddighi S, Yandell PM, Karram MM. Delayed presentation of complete ureteral obstruction deligated transvaginally. Int Urogynecol J Pelvic Floor Dysfunct. 2011;22:251–3.CrossRef
6.
Margulies RU, Rogers MA, Morgan DM. Outcomes of transvaginal uterosacral ligament suspension: systematic review and metaanalysis. Am J Obstet Gynecol. 2010;202:124–34.PubMedCrossRef
7.
Lantzsch T, Goepel C, Wolters M, et al. Sacrospinous ligament fixation for vaginal vault prolapse. Arch Gynecol Obstet. 2001;265:21–5.PubMedCrossRef
8.
Koyama M, Yoshida S, Koyama S, et al. Surgical reinforcement of support for the vagina in pelvic organ prolapse: concurrent iliococcygeus fascia colpopexy (Inmon technique). Int Urogynecol J Pelvic Floor Dysfunct. 2005;16:197–202.PubMedCrossRef
9.
Rane A, Frazer M, Jain A, et al. The sacrospinous ligament: conveniently effective or effectively convenient? J Obstet Gynaecol. 2011;31:366–70.PubMedCrossRef
10.
Verdeja AM, Elkins TE, Odoi A, et al. Transvaginal sacrospinous colpopexy: anatomic landmarks to be aware of to minimize complications. Am J Obstet Gynecol. 1995;173:1468–9.PubMedCrossRef
11.
Goldberg RP, Tomezsko JE, Winkler HA, et al. Anterior or posterior sacrospinous vaginal vault suspension: long-term anatomic and functional evaluation. Obstet Gynecol. 2001;98:199–204.PubMedCrossRef
12.
Pollak J, Takacs P, Medina C. Complications of three sacrospinous ligament fixation techniques. Int J Gynaecol Obstet. 2007;99:18–22.PubMedCrossRef
13.
Barksdale PA, Elkins TE, Sanders CK, et al. An anatomic approach to pelvic hemorrhage during sacrospinous ligament fixation of the vaginal vault. Obstet Gynecol. 1998;91:715–8.PubMedCrossRef
14.
Maher CF, Murray CJ, Carey MP, et al. Iliococcygeus or sacrospinous fixation for vaginal vault prolapse. Obstet Gynecol. 2001;981:40–4.CrossRef
15.
Meeks GR, Washburne JF, McGehee RP, Wiser WL. Repair of vaginal vault prolapse by suspension of the vagina to iliococcygeus (prespinous) fascia. Am J Obstet Gynecol. 1994;171:1444–52.PubMed
16.
Hoffman MS, Harris MS, Bouis PJ. Sacrospinous colpopexy in the management of uterovaginal prolapse. J Reprod Med. 1996;41:299–303.PubMed
17.
Hoffman MS, Lynch C, Lockhart J, Knapp R. Injury of the rectum during vaginal surgery. Am J Obstet Gynecol. 1999;181:274–7.PubMedCrossRef
18.
Beer M, Kuhn A. Surgical techniques for vault prolapse: a review of the literature. Eur J Obstet Gynecol Reprod Biol. 2005;119:144–55.PubMedCrossRef
19.
Demirci F, Ozdemir I, Somunkiran A, et al. Perioperative complications in abdominal sacrocolpopexy and vaginal sacrospinous ligament fixation procedures. Int Urogynecol J Pelvic Floor Dysfunct. 2007;18:257–61.PubMedCrossRef
20.
David-Montefiore E, Barranger E, Dubernard G, et al. Functional results and quality-of-life after bilateral sacrospinous ligament fixation for genital prolapse. Eur J Obstet Gynecol Reprod Biol. 2007;132:209–13.PubMedCrossRef
21.
Hibner M, Cornella JL, Magrina JF, Heppel JP. Ischiorectal abscess after sacrospinous ligament suspension. Am J Obstet Gynecol. 2005;193:1740–2.PubMedCrossRef
22.
Gafni-Kane A, Goldberg RP, Spitz JS, Sand PK. Extrasphincteric perianal fistulae after sacrospinous fixation for apical prolapse. Obstet Gynecol. 2011;117:438–40.PubMedCrossRef
23.
Ridgeway B, Barber MD, Walters MD, Paraiso MF. Small bowel obstruction after vaginal vault suspension: a series of three cases. Int Urogynecol J Pelvic Floor Dysfunct. 2007;18:1237–41.PubMedCrossRef
24.
Moen MD, Desai M, Sulkowski R. Vaginal evisceration managed by transvaginal bowel resection and vaginal repair. Int Urogynecol J Pelvic Floor Dysfunct. 2003;14:218–20.PubMedCrossRef
25.
Patravali N, Kulkarni T. Bowel evisceration through the vaginal vault: a delayed complication following hysterectomy. J Obstet Gynaecol. 2007;27:211.PubMedCrossRef
26.
Farrell SA, Scotti RJ, Ostergard DR, Bent AE. Massive evisceration: a complication following sacrospinous vaginal vault fixation. Obstet Gynecol. 1991;78:560–2.PubMed
27.
Rollinson D, Brodman ML, Friedman Jr F, Sperling R. Transvaginal small-bowel evisceration: a case report. Mt Sinai J Med. 1995;62:235–8.PubMed
28.
Roshanravan SM, Wieslander CK, Schaffer JI, Corton MM. Neurovascular anatomy of the sacrospinous ligament region in female cadavers: implications in sacrospinous ligament fixation. Am J Obstet Gynecol. 2007;197:660.e1–6.
29.
Barksdale PA, Gasser RF, Gauthier CM, et al. Intraligamentous nerves as a potential source of pain after sacrospinous ligament fixation of the vaginal apex. Int Urogynecol J Pelvic Floor Dysfunct. 1997;8:121–5.PubMedCrossRef
30.
Lazarou G, Grigorescu BA, Olson TR, et al. Anatomic variations in the pelvic floor nerves adjacent to the sacrospinous ligament: a female cadaver study. Int Urogynecol J Pelvic Floor Dysfunct. 2008;19:649–54.PubMedCrossRef
31.
Meschia M, Bruschi F, Amicarelli F, et al. The sacrospinous vaginal vault suspension: critical analysis of outcomes. Int Urogynecol J Pelvic Floor Dysfunct. 1999;10:155–9.PubMedCrossRef
32.
Lovatsis D, Drutz HP. Safety and efficacy of sacrospinous vault suspension. Int Urogynecol J Pelvic Floor Dysfunct. 2002;13:308–13.PubMedCrossRef
33.
Alevizon SJ, Finan MA. Sacrospinous colpopexy: management of postoperative pudendal nerve entrapment. Obstet Gynecol. 1996;88:713–5.PubMedCrossRef
34.
Siddiqui NY, Mitchell TR, Bentley RC, Weidner AC. Neural entrapment during uterosacral ligament suspension: an anatomic study of female cadavers. Obstet Gynecol. 2010;116:708–13.PubMedCrossRef
35.
Schön Ybarra MA, Gutman RE, Rini D, Handa VL. Etiology of post-uterosacral suspension neuropathies. Int Urogynecol J Pelvic Floor Dysfunct. 2009;20:1067–71.PubMedCrossRef
36.
Flynn M, Weidner AC, Amundsen CL. Sensory nerve injury after uterosacral ligament suspension. Am J Obstet Gynecol. 2006;19:1869–72.CrossRef
37.
Lowenstein L, Dooley Y, Kenton K, et al. Neural pain after uterosacral ligament vaginal suspension. Int Urogynecol J Pelvic Floor Dysfunct. 2007;18:109–10.PubMedCrossRef
38.
Vaginal repair of vaginal vault prolapse. In: Baggish, Karram, editors. Atlas of pelvic anatomy and gynecologic surgery. 3rd ed. Elsevier-Saunders; 2011;709.PubMedCrossRef