Marcin Zielin’ski, Wojciech Czajkowski, Tomasz Nabiałek, Artur Szlubowski, and Juliusz Pankowski
![]() INDICATIONS/CONTRAINDICATIONS
INDICATIONS/CONTRAINDICATIONS
Transcervical extended mediastinal lymphadenectomy (TEMLA) was developed to improve a diagnostic yield of staging of the mediastinal nodes for nonsmall cell lung cancer (NSCLC).1,2 TEMLA is performed in patients with pathologically proven operable NSCLC in whom the results of endobronchial ultrasonography with transbronchial fine needle biopsy (EBUS/TBNA) and endoesophageal ultrasonography and fine needle aspiration (EUS/FNA) were negative, regardless of the results of computed tomography of the chest (chest CT) and positron emission tomography combined with CT (PET/CT). In such patients TEMLA is an alternative for a mediastinoscopy, regarded as a gold standard of the mediastinal staging according to the ESTS guidelines.3
The second aim of TEMLA is a possible improvement of results of surgical treatment of NSCLC due to the bilateral extensive lymphadenectomy.
TEMLA is applicable also for removal of the metastatic nodes of the other malignancies such as the thyroid cancer.
![]() PREOPERATIVE PLANNING
PREOPERATIVE PLANNING
No special preparations for TEMLA are necessary except the standard preparation for general anesthesia.
Figure 5.1 The Rochard frame. (Aesculap-Chifa, Nowy Tomysl, Poland)
![]() SURGERY
SURGERY
Positioning of a Patient
TEMLA is performed in a patient in a supine position with the role placed under the upper back area, the same for the standard mediastinoscopy. The modified Rochard frame (Aesculap-Chifa, Nowy Tomysl, Poland) is used for elevation of the sternum (Fig. 5.1).
Surgical Technique of TEMLA
The operation starts from the 5- to 8-cm collar incision in the neck.
Subplatysmal flaps are widely dissected for better exposure of the mediastinum during the procedure. Visualization of both laryngeal recurrent nerves is obligatory in every case.
The detailed technique of visualization of the laryngeal recurrent nerves was described elsewhere.4 In brief, visualization of the right laryngeal recurrent nerve is started first. The carotid sheath of the right common carotid artery is divided with scissors, reaching the clean wall of the artery. The thyroid gland, the trachea, and the thymus gland are retracted to the left side with fingers and the right common carotid artery is retracted to the right side. Due to division of the deep fascial layers the right paratracheal space is easily accessible. Using blunt dissection with a peanut sponge the right laryngeal recurrent nerve is almost instantly visible. It runs from the point of division of the innominate artery toward the larynx. The deepest fascial layer covering the nerve is preserved, so the nerve is visualized, but not dissected; therefore, the risk of injury to the nerve is minimalized.
Visualization of the left laryngeal recurrent nerve is started after the right upper pole of the thymus gland is dissected free, grasped with a clamp, and retracted to the left side. The carotid sheath of the left common carotid artery is divided with scissors, the same as on the right side. The left common carotid artery is retracted to the left side and the trachea is retracted with the fingers to the right side. Using blunt dissection with the peanut sponge, the left laryngeal nerve is easily shown, running parallelly to the trachea in the tracheoesophageal groove (Fig. 5.2).
The left recurrent nerve is especially endangered during the procedure, so it must be dissected very delicately and carefully protected from injury. Circumferential dissection of the nerve is not advisable, in our opinion it increases the risk of injury to the nerve.
The central step of the presented technique is the division of the deep cervical fascial layers covering and obscuring the laryngeal recurrent nerves. Preservation of the deepest layer of the fascia protects the nerves from injury. No anatomical variations of the lower portions of both nerves were noted in this series—the nerves were in exactly the same location in all patients.
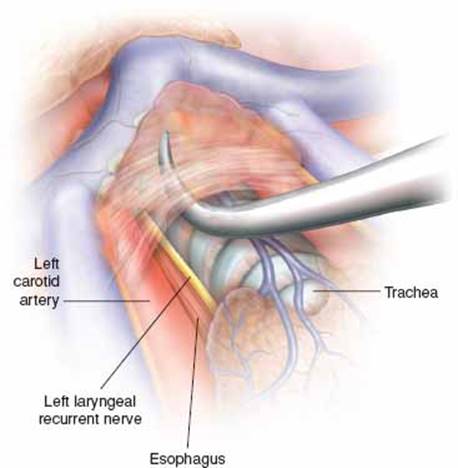
Figure 5.2 Dissection of the left laryngeal recurrent nerve. To expose the nerve, the vascularized fascial layers covering the nerve must be divided.
Both vagus nerves must also be found. The vagus nerve lies between the carotid arteries and internal jugular veins. The vagus nerves are the important landmarks for further dissection in the mediastinum.
Elevation of the sternal manubrium with a hook connected to the frame mounted on the operative table is a critically important part of the procedure (Rochard frame, Aesculap-Chifa, Nowy Tomysl, Poland) (Fig. 5.1). Elevation of the sternum widens access to the mediastinum from the neck and enables reaching the deeply located mediastinal structures.
During TEMLA all mediastinal nodal stations except for the pulmonary ligament nodes (station 9 according to the recent International Association for the Study of Lung Cancer (IASLC) map are sampled.5Generally, most part of TEMLA is an open procedure, with exception of dissection of the subcarinal (station 7), periesophageal (station 8), and the left lower paratracheal (station 4L) nodes, which are dissected in the mediastinoscopy-assisted fashion with the aid of Linder–Dahan two-blade mediastinoscope (Richard Wolf, Knittlingen, Germany) (Fig. 5.3). The para-aortic, station 6 and aorta-pulmonary window, station 5 nodes are sometimes dissected with the aid of videothoracoscope introduced to the mediastinum through the operative wound. An attempt should be made to remove en-bloc the nodes with the surrounding fatty tissue. Only the left paratracheal (station 4L) nodes are removed individually, due to close proximity of the left recurrent nerve.
Bilateral supraclavicular lymphadenectomy and even deep cervical lymph node dissection is possible during TEMLA through the same incision.
The mediastinal dissection begins from removal of the highest mediastinal, station 1 lymph nodes lying above the left innominate vein. Resection of the upper poles of the thymus at the level of the left innominate vein improves access to the mediastinum (Fig. 5.4).6
The next step is a dissection of the right paratracheal space, which proceeds along the posterior wall of the superior vena cava until the azygos vein becomes visible. Right upper paratracheal, station 2R nodes located above the apex of the aortic arch and below the left innominate vein are dissected and removed.
The right lower paratracheal, station 4R nodes are dissected from the trachea, right main bronchus, superior vena cava, the azygos vein, the ascending aorta, and the right mediastinal pleura.
Very occasionally, retrotracheal, station 3P nodes are found and removed. We never found any metastatic lymph node in this station.

Figure 5.3 The Linder–Dahan two-blade mediastinoscope. (Richard Wolf, Knittlingen, Germany)
In the right-sided tumors the next step is a dissection of the prevascular, station 3A nodes, located in front of the superior vena cava, right to the ascending aorta. This station is never involved in the left-sided tumors.
In our opinion, dissection of the left paratracheal nodes is the most difficult and risky part of TEMLA because these nodes lie in close proximity to the left recurrent nerve (Fig. 5.5). Generally, the left upper paratracheal, station 2L nodes lie in front of the nerve and the left lower, station 4L nodes lie behind the nerve (Fig. 5.6). The subcarinal and periesophageal nodes (stations 7 and 8) are dissected with the aid of Linder–Dahan mediastinoscope.
During dissection of the subcarinal, station 7 nodes the fascial layer covering the nodes must be divided. Usually, one or several prominent bronchial arteries crossing the operating field must be coagulated with harmonic knife or bipolar coagulation or clipped and divided because they hamper further dissection.
The subcarinal nodes must be dissected from the trachea, main bronchi (laterally), the esophagus (posteriorly), and the main pulmonary arteries (anteriorly). After completion of this part of the operation the tracheal bifurcation, both main bronchi, both pulmonary arteries, pericardium covering the left atrium, the right superior pulmonary vein, and the right upper lobe bronchus become clearly visible (Fig. 5.7).
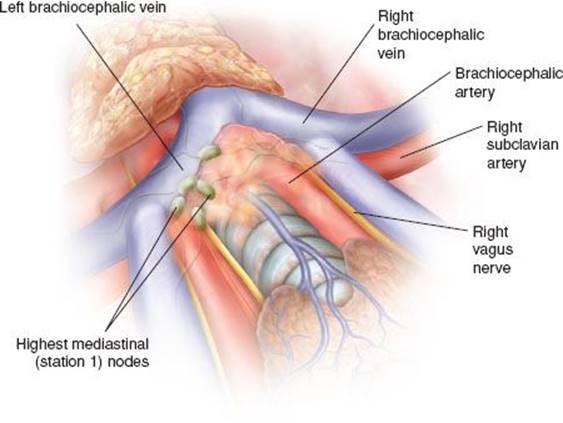
Figure 5.4 Dissection of the highest mediastinal nodes (station 1).
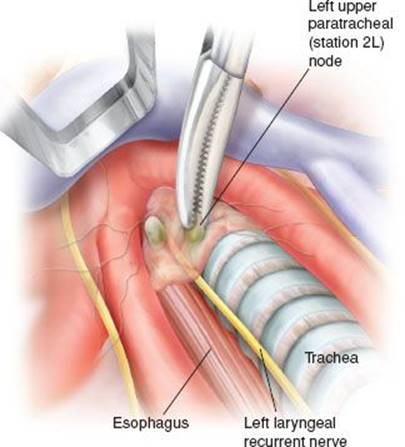
Figure 5.5 Dissection of the left upper paratracheal nodes (station 2L).
In majority of patients, but not in all patients, the periesophageal, station 8 nodes are found and removed. The limited length of the Linder–Dahan mediastinoscope precludes the possibility of removal of the pulmonary ligament (station 9) nodes.
Dissection of the station 4L nodes is performed in the mediastinoscopy-assisted fashion after completion of the dissection of stations 7 and 8, with the mediastinoscope withdrawn from the subcarinal area and introduced along the left main bronchus.
The entrance to the aorta-pulmonary window containing stations 6 and 5 lies between the left innominate vein and the left carotid artery. The left vagus nerve is a landmark of dissection. Division of the fascial layer between the left carotid artery and the left innominate vein facilitates the entrance to the aorta-pulmonary window.
At first, the adipose tissue is dissected from the wall of the ascending aorta down to the level of the left pulmonary artery with a peanut sponge. Medial retraction of the ascending aorta is a safe maneuver facilitating access to the aorta-pulmonary window.
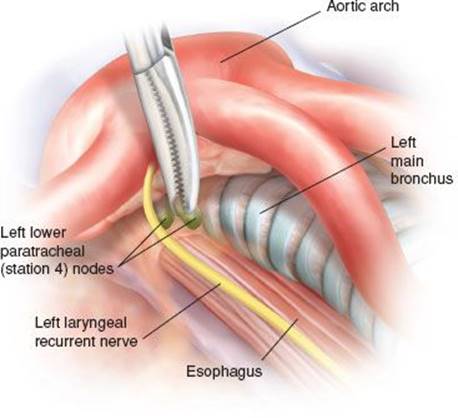
Figure 5.6 Dissection and removal of the left lower paratracheal nodes (station 4L).
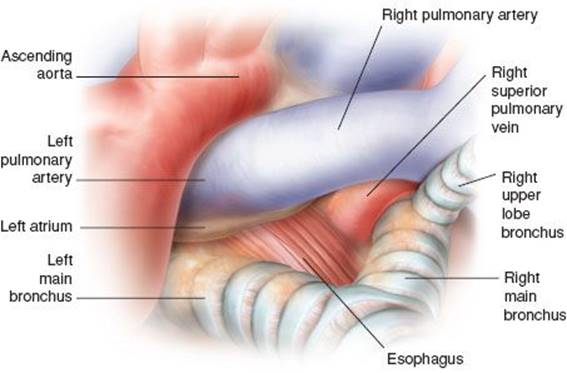
Figure 5.7 View of the subcarinal region after removal of the subcarinal (station 7) and the periesophageal (station 8) nodes.
The whole fatty tissue from the aorta-pulmonary window space is removed. Any blood vessel crossing the space is clipped and divided.
Dissection proceeds along the left vagus nerve. The fatty tissue is then dissected from the left mediastinal pleura until the clear pleura and the underlying left lung become well visible and removed en-bloc with the surrounding fatty tissue. Left pulmonary artery, the left phrenic nerve, and the left superior pulmonary vein are well visible after completion of dissection (Fig. 5.8). Coagulation and division of the hemiazygos accessory vein crossing the operative field is sometimes necessary. In case of opening of the mediastinal pleura there is no need for drainage of the mediastinum. Insertion of the piece of fibrin sponge and hyperinflation of the lungs during closure of the wound with high end-expiratory pressures is all what is necessary in such patients, without any need of drainage of the mediastinum and/or the pleural cavity.
The detailed description of the pitfall of TEMLA and how to avoid intraoperative complications are presented elsewhere.7
The use of TEMLA for patients who underwent neoadjuvant chemotherapy or a chemoradiotherapy (even with the use of doses 60 to 70 Gy) is the same as for the primary surgical treatment of NSCLC. Despite some fibrosis of the mediastinum after induction therapy, especially in patients who underwent chemoradiotherapy, technical difficulties of an operation could be managed without any special problems. Full range of nodal dissection was performed in all patients and there were no intraoperative or postoperative complications.
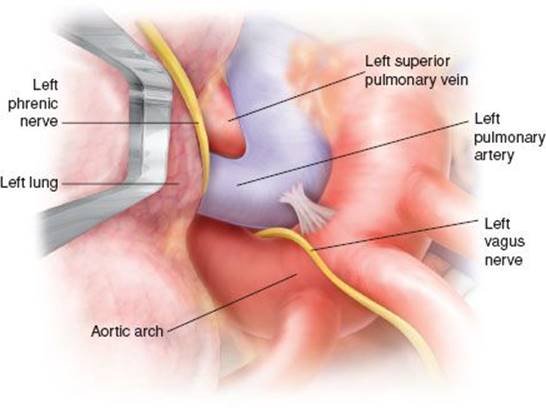
Figure 5.8 View of the aorta-pulmonary window area after dissection of the station 5 and station 6 nodes.
![]() POSTOPERATIVE MANAGEMENT
POSTOPERATIVE MANAGEMENT
Patients are extubated immediately after procedure. No opioid drugs are usually necessary for pain control. Patients are generally discharged from hospital on the second-third postoperative day. In case of negative result of TEMLA patients are admitted again for a subsequent pulmonary resection after 14 days, due to Polish insurance financial regulations.
![]() COMPLICATIONS
COMPLICATIONS
Complications of TEMLA occurred in 68/928 of patients (7.3%) (Table 5.1). The major intraoperative complications included a small injury of the right main bronchus managed with fibrin sponge packing, and the injury of the right segment 1 bronchus managed with sutures placed through cervicotomy supplemented with fibrin sponge packing. Both patients did not necessitate conversion to sternotomy/thoracotomy and both healed uneventfully. There was injury of the upper trunk of the right pulmonary artery controlled with tangential placement of two vascular clips supplemented with fibrin sponge packing. This patient also did not necessitate conversion to sternotomy/thoracotomy and healed uneventfully. Two patients required revision for bleeding of the minor arteries in the mediastinum. The most prevalent complication was the recurrent laryngeal nerve palsy with temporary laryngeal nerve palsy in 20/928 patients (2.2%) and permanent nerve palsy in 5/928 patients (0.5%).
TABLE 5.1 Complications of 928 TEMLA Procedures
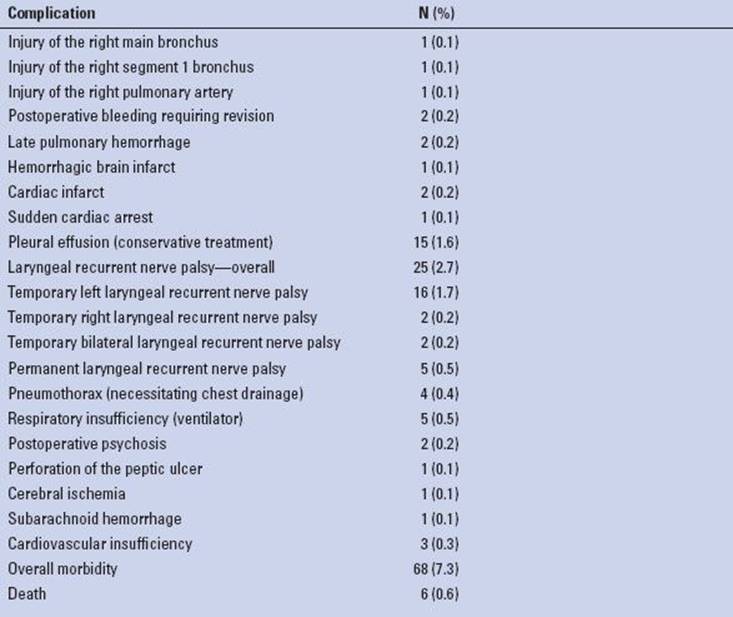
TABLE 5.2 Causes of Death After TEMLA
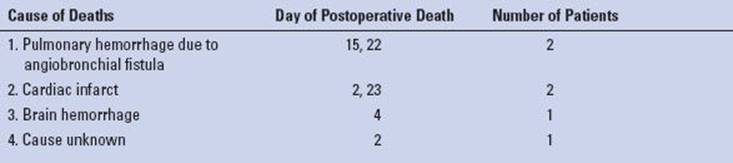
Pneumothorax necessitating pleural drainage occurred in 4/928 patients (0.4%), and pleural effusion in 15/928 patients (1.6%). Asymptomatic widening of the mediastinum was noted in about 40% of patients. It necessitated no treatment, subsided after several days, and was not regarded a postoperative complication. There were six postoperative deaths unrelated to the procedure (mortality 0.6%) (Table 5.2). There were no intraoperative injuries necessitating conversion to sternotomy/thoracotomy in any patient.
![]() RESULTS
RESULTS
TEMLA was performed on 928 patients with NSCLC, including 755 men and 173 women of age 41 to 79 (mean age 62.8) from 1.1.2004 to 31.12.2012. There were 588 squamous cell carcinomas, 214 adenocarcinomas, 25 large cell carcinomas, 19 mixed histology cancers, and 82 others (described as NSCLC in most of cases). Time of the operation was 35 to 330 minutes (mean 107 minutes). In the last 100 patients mean time of the operation was 91.5 minutes.
The number of dissected nodes during TEMLA was 15 to 85 (mean 33.9). Metastatic N2 nodes were found in 190/928 patients (20.5%) and N3 nodes were found in 39/928 patients (4.2%). Metastatic nodes were most prevalent in station 7, station 4R, station 2R, station 5, and station 4L. Subsequent thoracotomy was performed on 656 patients including 53 patients operated on after neoadjuvant therapy after previous positive TEMLA. Overall, 603/699 patients (86.3%) underwent primary surgery after negative result of TEMLA and 97 patients did not undergo operation after negative TEMLA. The reasons for omission of pulmonary resection included deaths after TEMLA in six patients, discovery of tumor progression/dissemination in 16 patients, lack of consent in 31 patients. In the other 44 patients we decided after TEMLA that pulmonary resection was an excessive risk, especially in the light of necessity of pneumonectomy. TEMLA was regarded a sort of biologic test in these high-risk patients.
There were 161 pneumonectomies, 48 sleeve lobectomies, 417 lobectomies/bilobectomies, 5 sublobar resections, and 23 explorations (3.4%). During thoracotomy, omitted N2 was found in 9/603 (1.5%) patients and omitted normal mediastinal nodes were found in 61/603 patients (10.1%). Omitted metastatic N2 nodes were found in the station 5 (four patients), station 7 (one patient), station 4R (one patient), station 8 (one patient), and station 9 (two patients).
Sensitivity of TEMLA in the discovery of N2–3 nodes was 96.2%, specificity was 100%, accuracy was 99.0%, negative predictive value (NPV) was 98.7%, and positive predictive value (PPV) was 100%. The postoperative mortality after pulmonary resection preceded by TEMLA was 1.5%. There were 53/229 patients (23.1%) who underwent subsequent pulmonary resection after neoadjuvant therapy (after positive TEMLA).
The results of restaging with TEMLA for patients after neoadjuvant treatment were no different than for the patients undergoing primary surgery. The diagnostic sensitivities 95.7% to 100% and NPV of 97.6% to 100% of TEMLA in restaging of the mediastinal nodes were reported in our previous studies.8,9
![]() CONCLUSIONS
CONCLUSIONS
TEMLA is a new minimally invasive surgical technique enabling unique possibility to perform very extensive bilateral lymphadenectomy of the mediastinum with very high diagnostic yield.
Recommended References and Readings
1. Kuzdzal J, Zielinski M, Papla B, et al. Transcervical extended mediastinal lymphadenectomy–the new operative technique and early results in lung cancer staging. Eur J Cardiothorac Surg. 2005;27:384–390.
2. Zielin´ski M. Transcervical extended mediastinal lymphadenectomy: Results of staging in two hundred fifty-six patients with non-small cell lung cancer. J Thorac Oncol. 2007;2:370–372.
3. De Leyn P, Lardinois D, Van Schil PE, et al. ESTS guidelines for preoperative lymph node staging for non-small cell lung cancer. Eur J Cardiothorac Surg. 2007;32:1–8.
4. Zielin´ski M, Kuz·dz·ał J, Szlubowski A, et al. A safe and reliable technique for visualization of the laryngeal recurrent nerves in the neck. Am J Surg. 2005;189:200–202.
5. Rusch VW, Crowley J, Giroux DJ, et al. The IASLC Lung Cancer Staging Project: Proposals for the revision of the N descriptors in the forthcoming seventh edition of the TNM classification for lung cancer. J Thorac Oncol.2007;2:603–612.
6. Zielinski M, Kuzdzal J, Nabialek T, et al. Transcervical extended mediastinal lymphadenectomy. Multimed Man Cardiothorac Surg. 2006;2006(1009):mmcts. 2005.001693. doi:10.1510/mmcts. 2005.001693.
7. Zielin´ski M. Technical pitfalls of transcervical extended mediastinal lymphadenectomy – how to avoid them and to manage intraoperative complications. Semin Thorac Cardiovasc Surg. 2010;22:236–243.
8. Zielin´ski M, Hauer L, Hauer J, et al. Non-small-cell lung cancer restaging with transcervical extended mediastinal lymphadenectomy. Eur J Cardiothorac Surg. 2010;37:776–780.
9. Zielinski M, Szlubowski A, Kołodziej M, et al. Comparison of endobronchial ultrasound and/or endoesophageal ultrasound with transcervical extended mediastinal lymphadenectomy for staging and restaging of non-small-cell lung cancer. J Thorac Oncol. 2013;8(5):630–636.