Stephen R. Broderick and Traves D. Crabtree
The thoracic duct is the main conduit for the transit of lymphatic drainage through the chest. The normal thoracic duct is a thin-walled, valved, 2 to 5 mm structure. It originates at the cisterna chyli and enters the chest through the aortic hiatus. The duct travels cephalad between the azygous vein and the aorta (Fig. 24.1) until approximately the fifth thoracic vertebra where it crosses over to the left side of the vertebral column, behind the aortic arch and terminates at the junction of the left internal jugular and subclavian veins. The anatomy of the thoracic duct is quite variable. The most frequent anatomic variation is a duplication of the lower thoracic duct due to persistence of the left embryonic trunk in approximately 40% of individuals.
Ligation of the thoracic duct is indicated in any patient with a thoracic duct leak that has failed conservative measures, which include a trial of low-fat diet, total parenteral nutrition, and octreotide. The manifestation of a thoracic duct leak is variable, but patients most commonly present with chylothorax. Alternative presentations include chylopericardium and rarely chyloptysis. Most clinically significant chylothoraces are the result of traumatic or iatrogenic injury of the thoracic duct such as may occur during esophagectomy. Chylothoraces associated with pulmonary resection generally result from injury to a branch duct during nodal dissection and resolve with conservative management. Mediastinal lymphoma is the most common cause of spontaneous chylothorax and may require duct ligation.
High-volume chylothorax following esophagectomy mandates reoperation for ligation of the thoracic duct. While a critical volume of chest tube effluent has not been defined, persistent drainage post esophagectomy can lead to volume depletion, leukopenia, and nutritional compromise. Prior to initiation of enteral nutrition, effluent will be serous. Debate remains regarding what constitutes high-output versus low-output chyle leak. However, daily drainage over 800 mL has historically prompted consideration of duct ligation. The literature supports early intervention in postesophagectomy chylothorax.
Prophylactic ligation of the thoracic duct during esophagectomy remains controversial. A single randomized controlled study supports mass ligation during transthoracic esophagectomy. Results from retrospective reports are mixed. The authors support prophylactic thoracic duct ligation during transthoracic esophagectomy as reoperation for duct ligation requires mobilization of the gastric conduit, which may result in tension on the esophagogastric anastomosis.
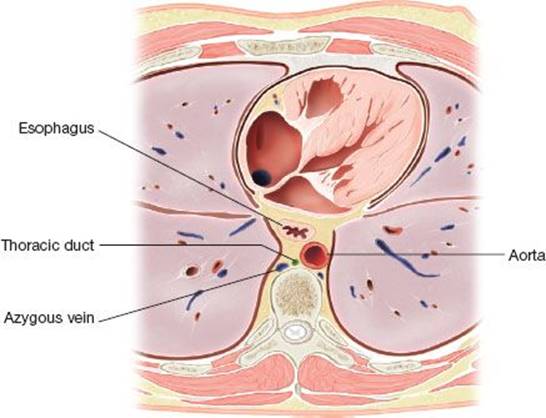
Figure 24.1 The thoracic duct usually lies in the posterior mediastinum, between the azygous vein, aorta, and esophagus.
![]() PREOPERATIVE PLANNING
PREOPERATIVE PLANNING
![]() Lymphangiography is a difficult study to perform and its application is limited by local expertise. However, if available, this study can be a useful adjunct to preoperative planning. Lymphangiography can clarify the anatomy of the thoracic duct and potentially identify duplicate or aberrant anatomy. The study may also identify the leak (Fig. 24.2). If the expertise is available, an attempt at embolization of the thoracic duct is reasonable. Lymphangiography is most useful in complicated chyle leaks where the etiology or source of the leak is unclear or in situations where mass ligation fails to control the leak.
Lymphangiography is a difficult study to perform and its application is limited by local expertise. However, if available, this study can be a useful adjunct to preoperative planning. Lymphangiography can clarify the anatomy of the thoracic duct and potentially identify duplicate or aberrant anatomy. The study may also identify the leak (Fig. 24.2). If the expertise is available, an attempt at embolization of the thoracic duct is reasonable. Lymphangiography is most useful in complicated chyle leaks where the etiology or source of the leak is unclear or in situations where mass ligation fails to control the leak.
![]() Enteral administration of cream may aid in visualization of the chyle leak. Cream should be given 30 to 60 minutes before operation to enhance the flow of chyle during inspection of the chest. This can be administered through a nasogastric or orogastric tube after induction of anesthesia.
Enteral administration of cream may aid in visualization of the chyle leak. Cream should be given 30 to 60 minutes before operation to enhance the flow of chyle during inspection of the chest. This can be administered through a nasogastric or orogastric tube after induction of anesthesia.
![]() The surgeon should ensure adequate volume resuscitation preoperatively.
The surgeon should ensure adequate volume resuscitation preoperatively.
![]() An epidural catheter should be placed if the initial catheter has been removed.
An epidural catheter should be placed if the initial catheter has been removed.
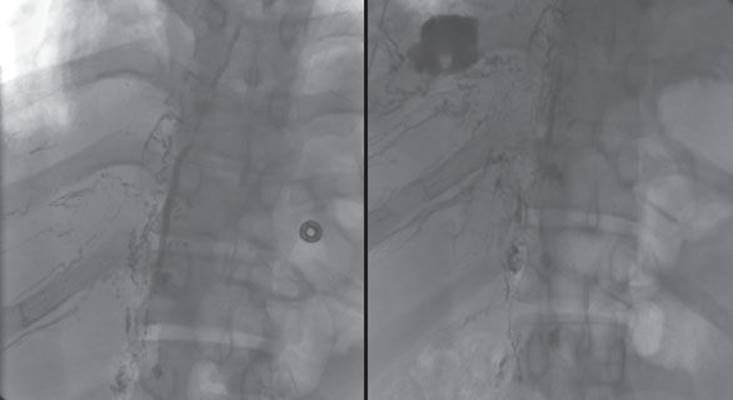
Figure 24.2 Lymphangiography can demonstrate the anatomy of the thoracic duct as well as the location of a thoracic duct injury. This lymphangiogram demonstrates the main thoracic duct in its normal anatomical location and a chyle leak into the right pleural space.
![]() The drainage tube for the gastric conduit (retrograde gastrostomy or nasogastric tube) should remain to intermittent suction to drain the conduit and reduce the risk of aspiration with induction of anesthesia.
The drainage tube for the gastric conduit (retrograde gastrostomy or nasogastric tube) should remain to intermittent suction to drain the conduit and reduce the risk of aspiration with induction of anesthesia.
![]() A double-lumen endotracheal tube is mandatory for visualization.
A double-lumen endotracheal tube is mandatory for visualization.
![]() Preoperative planning is similar for open or thoracoscopic visualization of the thoracic duct.
Preoperative planning is similar for open or thoracoscopic visualization of the thoracic duct.
![]() SURGERY
SURGERY
The technique of thoracic duct ligation is discussed for two scenarios: Ligation following transthoracic esophagectomy and VATS thoracic duct ligation for spontaneous chylothorax. The approaches and technical steps discussed apply to most indications for duct ligation.
Thoracic Duct Ligation Following Transthoracic Esophagectomy
Positioning
The patient is positioned in the left lateral decubitus position, rotated slightly forward. This positioning is identical to that used for Ivor Lewis esophagectomy. If the esophagectomy was performed in a minimally invasive fashion, the patient should be positioned as for that procedure.
Technique
If the patient underwent a recent thoracotomy then the thoracotomy is reopened and any residual effusion drained from the hemithorax. If no previous thoracotomy has been performed then the incision can be made slightly lower through the seventh or eighth intercostal space. For exposure of the thoracic duct, a complete muscle sparing posterior thoracotomy can be made with elevation and preservation of the latissimus muscle. The conduit is mobilized from the chest wall and posterior mediastinum and the pedicle identified. This is facilitated by early reoperation but can be more tedious if reoperation is delayed. Throughout the procedure care must be taken by the assistant retracting the conduit to avoid tension on the anastomosis and to prevent trauma to the right gastroepiploic artery. Once the conduit is mobilized the mediastinum is inspected for leaks. If identified, a leak should be closed by suture ligation or clipping. So me authors recommend the use of pledgeted sutures. This has not been our practice, but is certainly reasonable. Regardless of whether a leak is visualized or not we perform a mass ligation of the thoracic duct as inferior as possible.
A figure-of-eight heavy polypropylene suture is placed in the dome of the diaphragm and brought through a chest tube incision to retract the diaphragm. The visceral pleura overlying the vertebral body adjacent to the aortic hiatus is incised with electrocautery. A large right-angle dissector is then used to elevate all of the soft tissue between the vertebral column and the medial aspect of the aorta. This tissue is then doubly ligated with heavy silk sutures (Fig. 24.3). Mass ligation of the duct as low as possible is important to control leaks that are not identified visually at the time of surgery. After ligation, some surgeons advocate mechanical pleurodesis as an adjunct. This is followed by placement of chest tubes to widely drain the hemithorax. The chest is closed in the standard fashion.
For patients in whom minimally invasive esophagectomy was performed, the same approach to ligation of the thoracic duct can be performed through the minimally invasive esophagectomy (MIE) incisions. We routinely perform prophylactic ligation of the thoracic duct during transthoracic esophagectomy using the technique described.
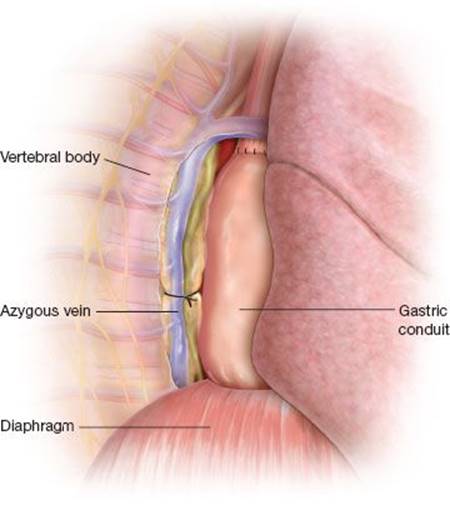
Figure 24.3 Mass ligation of the thoracic duct performed during transthoracic (Ivor Lewis) esophagectomy. All of the soft tissue between the vertebral body and aorta is elevated and mass ligated with a heavy silk suture.
![]() POSTOPERATIVE MANAGEMENT
POSTOPERATIVE MANAGEMENT
Postoperatively, the patient can usually be returned to the surgical ward or step-down unit. Chest tube drainage is monitored closely. Enteral nutrition is generally resumed 24 to 48 hours after reoperation if there is no other contraindication. Chest tubes are removed when the output is minimal and an esophagram has demonstrated the absence of anastomotic leak.
Postoperatively, the patient is given a clear liquid diet for 24 hours. After 24 to 48 hours the patient may be advanced to a regular diet. The quality of the chest tube output is monitored and the absence of chylous drainage suggests successful ligation of the duct. Removal of the chest tube is at the discretion of the surgeon, usually when drainage is less than 200 cc per day.
Vats Thoracic Duct Ligation
Positioning
The patient is positioned in the left lateral decubitus position, rotated slightly forward, and flexed at the hip.
Technique
The camera port for VATS thoracic duct ligation is placed in the seventh or eighth intercostal space in the posterior axillary line. A retraction port is placed in the seventh or eighth space in the midscapular line. Finally, a working incision is created in the fifth intercostal space anteriorly.
The diaphragm is gently depressed with a sponge stick while the lower lobe is retracted cephalad, exposing the inferior pulmonary ligament. The ligament is divided to the level of the inferior pulmonary vein with a combination of electrocautery and blunt dissection. The lung is then retracted anteriorly, exposing the posterior mediastinum. A heavy prolene suture is placed in the dome of the diaphragm in a figure-of-eight fashion and brought out through a stab incision low in the hemithorax to allow for continued retraction of the diaphragm. The mediastinal pleura is opened longitudinally, lateral to the azygous vein (Fig. 24.4). The azygous vein is dissected free from surrounding structures and retracted laterally; branches of the vein may be clipped and divided as necessary. Next the mediastinal pleura overlying the esophagus is opened and the esophagus retracted medially. Blunt dissection between the azygous vein and the esophagus will reveal the thoracic duct overlying the aorta (Fig. 24.5). Application of several 10-mm clips is sufficient for ligation (Fig. 24.6). Several centimeters of the duct should be identified to allow sufficient room for ligation. A small incision can be made in the wall of the duct with an endoscopic scissor; the presence of chylous fluid confirms the identity of the structure being ligated. Alternatively, a small portion of the wall of the duct can be removed for pathologic confirmation. If the duct is not able to be identified directly, which is most the case, mass ligation is performed by encircling all of the soft tissue between the azygous vein and the esophagus with a large right angle and doubly ligating it with heavy silk ligatures.
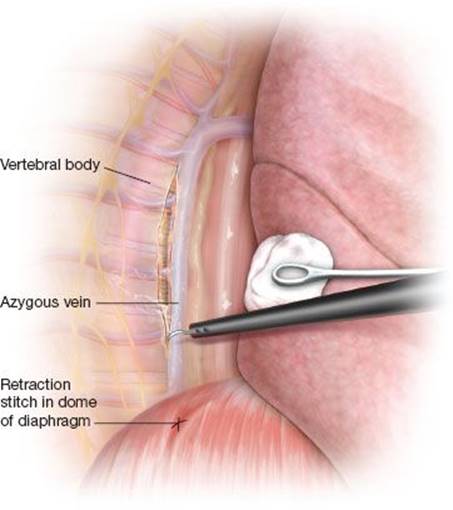
Figure 24.4 A retraction stitch placed in the dome of the diaphragm aids in exposure of the posterior mediastinal during thoracoscopic thoracic duct ligation. The mediastinal pleura is incised with electrocautery lateral to the azygous vein.
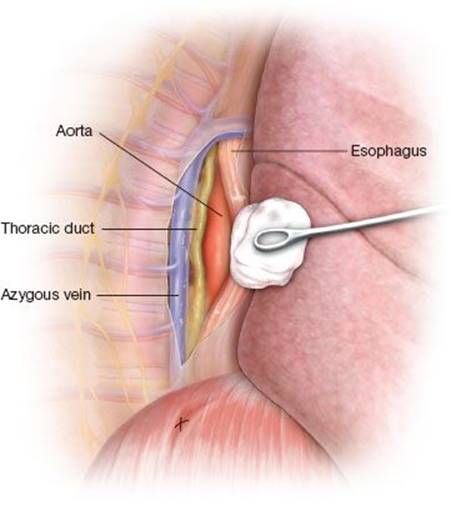
Figure 24.5 Dissection between the azygous vein and esophagus reveals the thoracic duct overlying the aorta during thoracoscopic duct ligation.
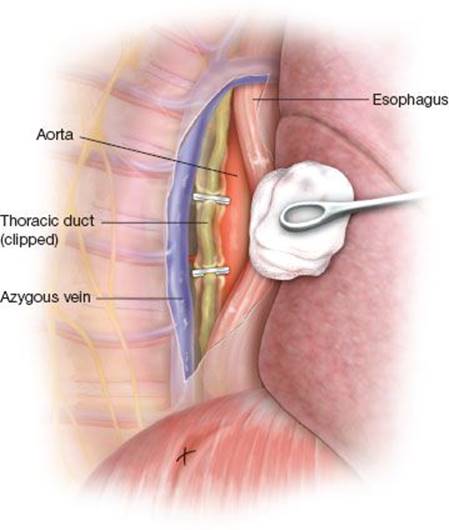
Figure 24.6 Ten-millimeter endoscopic clips are used to ligate the thoracic duct.
Before closing, the thoracic duct is inspected for leakage of chylous material and hemostasis is ensured. A mechanical pleurodesis is performed and a single chest tube is placed in a posterior location. In the absence of an epidural catheter an intercostal nerve block is performed with 0.5% bupivacaine. The lung is reinflated and the incisions closed in the standard fashion.
![]() RESULTS
RESULTS
The results of transthoracic ligation of the thoracic duct are excellent. For postesophagectomy chylothorax, reoperation with ligation of the thoracic duct results in cessation of the chyle leak in over 90% of patients as reported by two large-volume centers.
![]() CONCLUSIONS
CONCLUSIONS
Thoracic duct ligation is an effective means of managing the various manifestations of chylous fistula. Following esophagectomy we recommend early intervention through mass ligation of the thoracic duct. Chyle leak in this setting is usually the result of traumatic injury to the main thoracic duct and nonoperative management is unlikely to be effective. For other indications, VATS thoracic duct ligation is effective and can be accomplished with minimal morbidity.
Recommended References and Readings
Cerfolio R. Ligation of the thoracic duct for chylothorax. CTSNet Inc. http://www.ctsnet.org/sections/clinicalresources/thoracic/expert_tech-19.html posted 6 October, 2009.
Cerfolio RJ, Allen MS. Postoperative chylothorax. J Thorac Cardiovasc Surg. 1996;112:1361–1366.
Hayden JD, Sue-Ling HM, Sarela AI, et al. Minimally invasive management of chylous fistula after esophagectomy. Dis Esophagus. 2007;20(3):251–255.
Lai FC, Chen L, Tu YR, et al. Prevention of chylothorax complicating extensive esophageal resection by mass ligation of thoracic duct: A random control study. Ann Thorac Surg. 2011;91(6):1770–1774.
Schuchert MJ, Luketich JD, Fernando HC. Complications of minimally invasive esophagectomy. Semin Thorac Cardiovasc Surg. 2004;16:133–141.
Shah RD, Luketich JD, Schuchert MJ, et al. Postesophagectomy chylothorax: Incidence, risk factors, and outcomes. Ann Thorac Surg. 2012;93(3):897–903.
Sieczka EM, Harvey JC. Early thoracic duct ligation for postoperative chylothorax. J Surg Oncol. 1996;61:56–60.
Sukumar M, Schipper P, Komanapalli C. Thoracoscopic ligation of the thoracic duct. CTSNet Inc. http://www.ctsnet.org/sections/clinincalresources/thoracic/expert_tech-30.html posted 6 October, 2009.
Wurnig PN, Hollaus PH, Ohtsuka T, et al. Thoracoscopic direct clipping of the thoracic duct for chylopericardium and chylothorax. Ann Thorac Surg. 2000;70:1662–1665.