Frank C. Detterbeck and Andrew T. Arndt
Introduction
This chapter addresses the clinical approach to and management of posterior mediastinal tumors in adults. The majority of these are neurogenic tumors, and conversely most neurogenic tumors are in the posterior mediastinum. Various benign systemic conditions can also cause posterior mediastinal abnormalities, such as tuberculous vertebral infections and hematopoietic disorders (e.g., sickle cell anemia, thalassemia). These conditions are generally obvious from the clinical presentation—the posterior mediastinal/vertebral lesions are usually noted secondarily when the diagnosis of an infection or hematopoietic disorder is already clear. Therefore, the features and management of these conditions are not addressed here; interested readers are referred to internal medicine textbooks or other specialty books.
This chapter provides a structure for how to approach the assessment and management of a patient with a posterior neurogenic lesion presenting for evaluation. The approach starts with making a clinical diagnosis and assessing the likelihood that this is correct. These factors guide the need for further investigations and eventually management of the condition.
We have taken a clinically oriented approach, using readily available information (e.g., age, gender, symptoms), leading to a presumptive diagnosis and eventually to a final diagnosis and management strategy. This is opposite to most publications, which start with a known specific diagnosis and use this to estimate the age, gender, etc. of a cohort of such patients.
The most fundamental issue is whether a posterior mediastinal mass is benign or malignant. A more detailed question is which additional tests (imaging, biopsy) are needed to efficiently and sufficiently confirm the initial diagnosis. This includes deciding how certain and detailed one must be in to develop an appropriate management plan.
Details of a management plan may require investigation of particular aspects of the condition (e.g., Is there extension through a neural foramen?). While some imaging tests can help solidify a diagnosis as well as identify features that impact details of management, it is better to maintain a structured approach to patient evaluation—namely, to separate thinking through what the likely clinical diagnosis is, what (if anything) is needed to sufficiently confirm this, and what details need to be defined to carry out the management plan. Because posterior mediastinal masses are uncommon, most physicians have not evolved to an internalized “routine” in how they approach patients. Therefore, a structure for how to think through the approach to patients is needed. This prevents getting sidetracked by details and questions that are not germane to a particular patient at a particular point in the evaluation process.
Background
Definition of Posterior Mediastinum
Dividing the mediastinum into compartments has proven useful in the evaluation of mediastinal masses, but these have been defined in many ways. The International Thymic Malignancy Interest Group (ITMIG) has recently developed a modern, computed tomography (CT)-based international standard.1 This schema defines the paravertebral (posterior) compartment as bounded by the thoracic inlet, the diaphragm, the chest wall posteriorly, the tip of the transverse processes laterally, and anteriorly a line 1 cm behind the anterior border of the vertebral bodies. The contents of the posterior mediastinum include sympathetic nerves, intercostal nerves and ganglia, azygos and hemiazygos veins, and posterior intercostal arteries and veins. Tumors may arise from any of these structures, but the most common primary tumors of the posterior mediastinum are neurogenic tumors, arising from the sympathetic chain, paraganglionic cells, intercostal nerves, or nerve sheath cells.
Classification
Posterior neurogenic tumors are often classified by tissue of origin and as benign or malignant, thus defining four groups (Table 29.1). In adults, most posterior neurogenic tumors are of nerve sheath origin (Fig. 29.1); however, the cell of origin has no clinical importance. The key feature is that the vast majority are benign. In children, most posterior neurogenic tumors are malignant, especially in younger age groups (Fig. 29.2). Because general thoracic surgeons deal largely with adults, this chapter does not address the distinct group of tumors encountered in children.
Natural History
An understanding of the natural history (i.e., course without treatment) of posterior neurogenic tumors is important in defining a management algorithm. There is limited direct data because these tumors are rare and immediate resection has been the traditional approach. Recurrence and survival data from case series of treated patients with adequate long-term follow-up can also provide some insight.
Malignant Lesions
Malignant posterior neurogenic tumors in adults carry a very poor prognosis if left untreated. The more common malignant neurogenic tumors, such as malignant schwannomas, are typically quite aggressive. The natural history of untreated patients has not been reported. The survival after treatment attempts is generally short with patients developing a recurrence and/or dying in 2 to 18 months.2–4 Only 14 cases of adult mediastinal neuroblastoma have been reported, with only three of these occurring in the posterior mediastinum.5 The time to recurrence is generally only a few months.6 Inference drawn from this limited data is that untreated lesions portend a progressive course with early mortality, likely within 1 year.
TABLE 29.1 Classification of Posterior Neurogenic Tumors

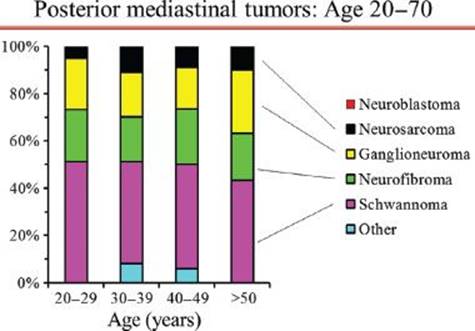
Figure 29.1 Proportion of posterior mediastinal tumor types by decades of age in adults. Legend: N/GN’blastoma, neuroblastoma/ganglioneuroblastoma; PNET, primitive neuroectodermal tumor. Reproduced with permission from Detterbeck: Mediastinal Tumors.32
Benign Lesions
Limited data corroborates the conventional wisdom that benign neurogenic posterior mediastinal lesions are very indolent. Benign tumors have a very low recurrence rate—even if incompletely resected; for example, one recurrence occurring 4 years after an incomplete resection was reported out of 87 resected benign posterior neurogenic tumors.2 Four other incompletely resected benign lesions in this series did not recur during 10 years of follow-up.2 Experts in these tumors have remarked that resection of benign lesions has no influence on patient survival although they recommend resection of benign lesions.2,7
There is limited data on observation of posterior neurogenic tumors in patients deemed to be too high risk for resection. No progression was noted over 5 years of observation in a patient with chronic rejection of a transplanted kidney with foraminal extension of a posterior neurogenic tumor.8 In another series of neurogenic tumors (in various locations, not only thoracic), six tumors that were considered benign based on positron emission tomography (PET) activity were observed and were all found to be unchanged at a median follow-up of 17 months.3
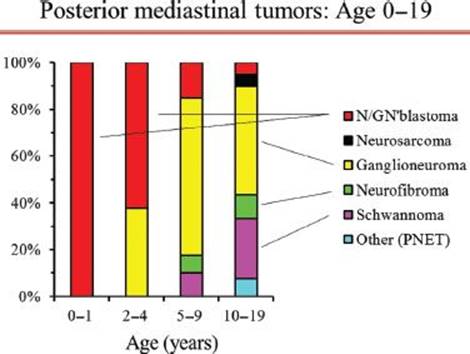
Figure 29.2 Proportion of posterior mediastinal tumor types by age in children and adolescents. Legend: N/GN’blastoma, neuroblastoma/ganglioneuroblastoma; PNET, primitive neuroectodermal tumor. Reproduced with permission from Detterbeck: Mediastinal Tumors.32
In conclusion, anecdotal data suggests that growth occurs in only a minority of benign neurogenic tumors during several years of observation.
Specific Clinical Questions
The traditional recommendation has been to resect all posterior mediastinal neurogenic tumors—even if benign—because of concern about malignant degeneration or complications due to growth. However, to appropriately apply these arguments it is necessary to define how high the risk of these events actually is.
What Is the Risk of Malignant Degeneration?
Papers justifying resection of benign neurogenic tumors because of a risk of malignant degeneration invariably provide no data or references. An extensive literature search revealed no well-documented benign neurogenic tumor that became malignant in adults without neurofibromatosis. Several case reports of malignant degeneration of neurogenic tumors involve either children, tumors of other areas of the body, or tumors not arising spontaneously—for example, after prior irradiation of a neuroblastoma or in an immunocompromised patient.9–11 One case report12 of a thoracic neurogenic tumor in an adult speculated about “malignant degeneration” because it involved a composite tumor with features of ganglioneuroma as well as neuroblastoma; however, this could also be a manifestation of the heterogeneity of tumors or spontaneous maturation from neuroblastoma to ganglioneuroma, as has been described.13
Another case14 involved a malignant neuroblastoma that developed at the site of a previously resected benign retroperitoneal ganglioneuroma 11 years earlier. Local control was achieved with partial reresection and radiation, but eight years later a metastasis at a separate site was found. It was argued that this might represent malignant transformation because it would be unlikely to have had two distinct tumors, and the typically rapid growth of neuroblastoma makes it unlikely, this was a present but missed component in the original tumor. However, this case is very atypical because the indolent behavior (metastasis after 8 years) and the age at diagnosis (age 32) are highly unusual for a neuroblastoma.
Therefore, malignant degeneration of a benign neurogenic tumor is not clearly documented in the literature in patients without neurofibromatosis, and such speculation rests on anecdotal cases of composite tumors and one potential case report in which the malignant tumor exhibited unusual (indolent) behavior. The argument for resection of benign neurogenic tumors because of potential malignant degeneration must be viewed as not being clearly substantiated.
What Is the Risk of Development of Symptoms/Clinical Problems?
An extensive literature search revealed no series of prolonged observation of benign neurogenic tumors that allow estimation of a rate of growth and subsequent development of complications. The lack of case reports of patients eventually resected after initial observation suggests that the rate of growth and development of symptoms is rare. Thus, the potential for clinically significant growth is not well defined but appears likely to be rather low.
A rational argument can be made to resect asymptomatic lesions that involve the spinal canal, given the potentially major consequences if one waits for symptoms to develop. Whether careful surveillance with regular imaging is a safe alternative is not documented in the literature.
What Is the Risk of Malignant Neurogenic Tumor in Patients With Neurofibromatosis?
Patients with familial neurofibromatosis who have intrathoracic tumors present a unique clinical challenge. The lifetime incidence of a malignant peripheral nerve sheath tumor is 2% to 5% in this population, as compared to 0.001% in the general population.15 Nordback et al. suggest a 10% risk of malignancy in intrathoracic neurogenic tumors in patients with neurofibromatosis16; they additionally surmise that surgical trauma may induce malignant transformation based on cases of early appearance of malignant tumors at the site of previously completely resected benign tumors.16 Finally, malignant neurogenic tumors in these patients are said to have higher rates of local recurrence, distant metastases and worse overall survival.17
Making a Clinical Diagnosis
The first step in approaching patients is to establish a presumptive clinical diagnosis and an assessment of how confident one can be of this. The primary clinical issue is knowing when to suspect malignancy. Specific characterization of benign lesions has no clinical relevance. Potential factors to help identify malignancy are reviewed in the next sections.
Demographic Features
Demographic features are of little benefit in defining the nature of a posterior mediastinal mass in adults. Posterior neurogenic tumors occur with similar frequency in all ages from infancy to the eighth decade of life.8 While the probability of malignancy is high in children (∼40% overall, ∼100% if under age 2 years; Fig. 29.2),8 it is 1% to 6% in adults and is relatively constant across all adult ages (Fig. 29.1).7,17
Posterior neurogenic tumors occur equally in men and women; this is true of malignant tumors as well.8 Small differences exist in the incidence of specific benign tumors, but this has no clinical impact (e.g., ganglioneuromas are slightly more common in men and nerve sheath tumors slightly more common in women).2
Medical History
In patients with a posterior mediastinal mass one should specifically ask about a history of neurofibromatosis and look for superficial signs such as café au lait spots or subcutaneous neurofibromas. While neurofibromatosis is associated with 14% of posterior neurogenic tumors overall, it is present in roughly one-half of all cases with malignant tumors.2 In addition, neurofibromas in the setting of neurofibromatosis classically carry a risk of malignant transformation (to be discussed in further detail). However, these rates still imply that in patients with neurofibromatosis the vast majority (∼85%) of posterior mediastinal tumors will be benign.
Symptoms
Posterior mediastinal masses in adults are often detected incidentally on imaging for other reasons. Only 12% to 37% of patients present with symptoms,2,4,8,18 typically related either to neurologic compromise of the spinal cord or local intrathoracic mass effect (chest pain, Horner syndrome, cough, dyspnea, or dysphagia).
Symptoms Suggestive of Malignancy
The presence of symptoms in general is not a significant predictor of malignancy in adults, nor does the absence of symptoms guarantee benignity. (In children, absence of symptoms is a statistically significant predictor of benignity). However, pain or dyspnea specifically was found to be highly indicative of a malignant tumor in adults.2
Symptoms Suggestive of Spinal Involvement
The presence of neurologic symptoms correlates with the likelihood of intraspinal tumor involvement (so-called dumbbell tumors connected by a thin isthmus extending through a neural foramen). However, about 40% of patients with intraspinal tumors have no neurologic symptoms.19,20

Figure 29.3 Benign and malignant neurogenic tumors. A: Benign neurogenic tumor with bony erosion, widening of the foramen, and extension into the spinal canal. None of these signs are associated with malignancy. B: Ganglioneuroblastoma in an adult. The large size is associated with a likelihood of malignancy.
Imaging
CT
A study of CT imaging of posterior neurogenic tumors in 68 adults identified four imaging features that were associated with malignancy: (a) Size >10 cm (malignancy in almost all patients), (b) bone destruction (four out of four patients), (c) pleural effusion (two out of two patients), and (d) mediastinal displacement (one out of two patients).2 However, while bone destruction was strongly predictive of malignancy, some degree of bony abnormality was noted in 32% (22/68); these findings include erosions, splaying of the ribs, foraminal enlargement, or localized scoliosis and were not associated with malignancy (Fig. 29.3A,B).2
MRI
MRI is an accurate test to assess for a dumbbell tumor.21 Because CT may miss an intraspinal component,20 MRI is indicated whenever the tumor is contiguous with a neural foramen, there is a widened intervertebral foramen or erosion of a vertebral body or pedicle (Fig. 29.4A,B).21 MRI is more sensitive than traditional myelography.20 In addition, magnetic resonance angiography (MRA) can be useful in lower posterior mediastinal tumors to locate the artery of Adamkiewicz instead of arteriography, which has a 2% complication rate.20
MRI features suggestive of malignancy include size >5 cm, prominent vascularity or enhancement, infiltrative margins, marked heterogeneity with central necrosis, rapid growth from a recent imaging study, and increased uptake of Ga-67 citrate.22 However, how well these features predict malignancy is not defined, and MRI is not recommended to categorize posterior mediastinal tumors as benign or malignant.
PET
The role of PET and how to interpret results in posterior neurogenic tumors has not been defined. A retrospective analysis of PET in adult patients found a statistically significantly higher standard uptake value (SUV) in malignant versus benign tumors (median SUV of 2.9 [range 1.8 to 12.3] vs. 1.1 [range 0.5 to 1.8]). A retrospectively applied SUV cutoff of 1.8 yielded a sensitivity of 100% and a specificity of 83%.3 However, in another study of benign tumors in a woman with neurofibromatosis type 1 the SUV ranged from 1.8 to 5.3.23 Further validation is needed to define a specific cutpoint and the false-positive and false-negative rates associated with it. Nevertheless, a strongly positive or negative PET may help confirm a clinical diagnosis and define the extent of disease in some patients.
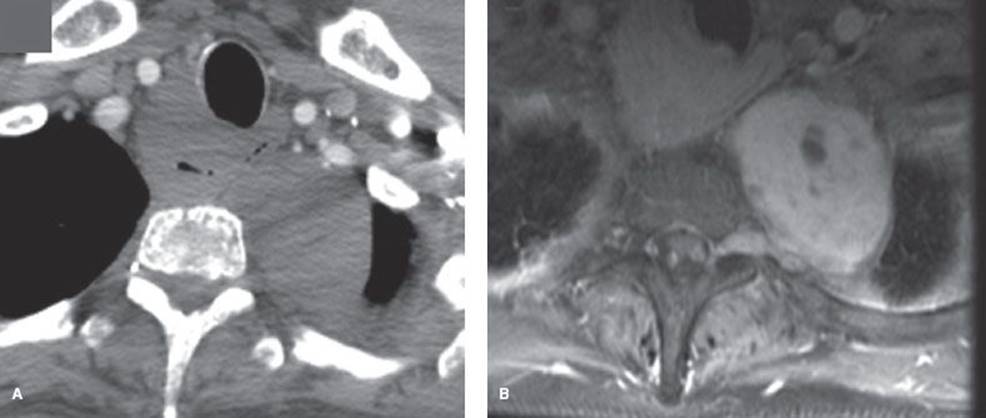
Figure 29.4 Schwannoma in a woman with neurofibromatosis. A: CT with IV contrast does not clearly show invasion through the foramen. B: MRI (T1 postcontrast) image shows invasion through the foramen into the spinal canal.
Laboratory Studies
In children (especially under 5 years) the incidence of neuroblastoma or ganglioneuroblastoma is quite high. Serum vanillylmandelic acid (VMA) or hydroxyl-vanillylmandelic acid (HVMA) was elevated in approximately 30% of malignant neuroblastomas or ganglioneuroblastomas in children.2 The combination of a typical presentation in young children with an elevated serum VMA or HVMA is essentially diagnostic without a biopsy,2 and measurement of these markers in children suspected of having such tumors is clearly justified.
Posterior mediastinal neurogenic tumors in adults are typically nonfunctional.24 No posterior neurogenic tumor in adults showed an elevation of VMA or HVMA in one series.2 Routinely checking VMA or HVMA levels in an adult with a posterior mediastinal tumor is not indicated, as these markers are potentially useful only when neuroblastoma or ganglioneuroblastoma is present, which is extremely rare in adults. These tests are not useful to predict malignancy. These laboratory studies may be useful if there are clinical manifestations of a secretory tumor, but not indicated if such symptoms are absent.
Clinical Approach to Patients
Patients have typically had a chest CT before being referred to a specialist such as a thoracic surgeon. The initial clinical evaluation and the CT almost always provide enough information to develop a treatment plan, which hinges on whether the lesion is likely or unlikely to cause health issues if left alone. Patients can be grouped into four categories: (a) Asymptomatic and CT not “concerning,” (b) symptomatic but CT not “concerning,” (c) spine symptoms or spine concern on CT, or (d) concern for malignancy (Fig. 29.5).
Asymptomatic Patients with Benign CT Findings
Most adult patients with a posterior mediastinal tumor are asymptomatic (∼75%) and have a simple well-defined mass with no concerning features. The preceding discussion suggests that in such patients (without neurofibromatosis), a diagnosis of a benign tumor can be made with a high degree of certainty (∼99%).
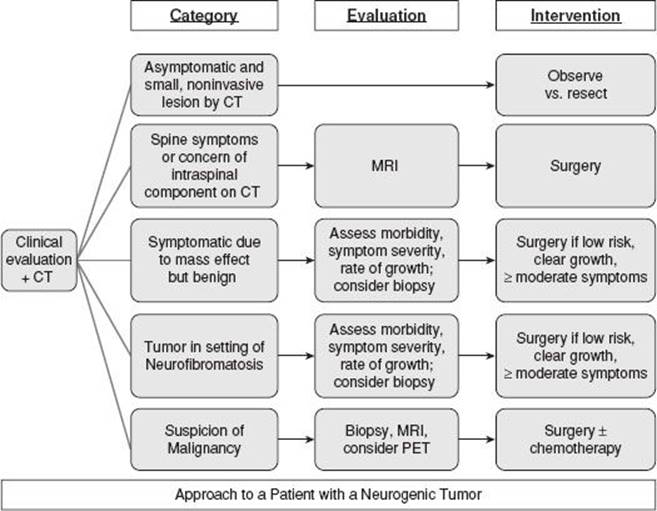
Figure 29.5 Clinical approach to patients with a posterior mediastinal tumor.
Routine biopsy of posterior mediastinal lesions is rarely useful.25,26 A needle biopsy of posterior mediastinal tumors in adults was concordant with the final pathology in only about 50% or less in several older studies (involving eight and 16 patients who had benign lesions).25,26 Furthermore, the histologic type of benign tumor has no impact on clinical management. Pneumothorax occurred in 18% (one patient required a chest tube) and one patient developed a pericardial effusion with tamponade.25 There is also no justification in adults to obtain routine serum markers for neuroblastoma or ganglioneuroblastoma. Unless the CT or clinical presentation is unusual, further imaging or tests are not indicated.
In asymptomatic patients with a smooth, homogeneous mass without bony changes or contiguity to the neural foramen, observation is a reasonable management policy. Although the data is limited, whether growth, symptom development, or malignant degeneration (outside of neurofibromatosis) actually occurs is questionable at best. On the other hand, resection of such lesions is generally easy (usually by thoracoscopy). Therefore, while arguments for resection are dogmatic and unfounded, the argument against resection is also weak.
In situations in which the evidence for benefit and for harm is very weak, patient preferences should play a significant role. Some may prefer resection to prolonged follow-up; others may prefer the opposite. Patients should be informed that both observation and resection are reasonable, without strong evidence of benefit or harm in either case. (Note: This discussion applies to patients without major comorbidities.)
Concern of Spinal Cord Compression
It is crucial to identify patients who are at risk for spinal cord compression. Retrospectively, approximately 40% of patients with confirmed intraspinal tumors had no neurologic symptoms.19 Furthermore, CT fails to show the intraspinal involvement in a substantial minority of patients.20 Therefore, an absence of symptoms is not sufficient and recognition of radiographic features that elevate or diminish the suspicion is important. If a posterior mediastinal tumor is not contiguous with the neural foramina by CT, clinical experience and common sense suggest this is sufficiently reliable. CT findings of neuroforaminal enlargement or erosion of a vertebral body, spinous process, or pedicle have been found to correlate with the presence of a dumbbell tumor.20 MRI should be done in all patients with such findings or with a tumor adjacent to the neural foramina.20
If there is symptomatic cord compression, the need for surgical intervention is obvious. In asymptomatic patients with an intraspinal tumor, concern about cord compression creates strong pressure to intervene although the incidence of cord compression with observation is not well defined but appears anecdotally to be low. In patients without an intraspinal component but with neural foraminal involvement, the same arguments apply. In this case it may be easier to consider observation with subsequent intervention should intraspinal growth develop, but this is countered by the argument that resection can often be accomplished easily (by thoracoscopy) when there is only limited neural foraminal involvement. For all practical purposes, however, resection is undertaken in all patients with symptoms, imaging evidence of cord compression, or with a lesion that is asymptomatic but is close to the spinal canal (unless there are major comorbidities).
Symptoms Due to Intrathoracic Mass Effect
If patients are symptomatic due to intrathoracic mass effect, there is strong pressure to intervene. However, in these cases the size of the lesion and the structures involved typically make this a difficult undertaking. In addition, the symptoms have generally been present for a long time, making it difficult to determine when to intervene—that is, when symptoms are only mild and the procedure might be easier, or when they are more severe, but intervention is quite difficult.
Neurogenic tumors causing symptoms due to intrathoracic mass effect are usually benign tumors. Nevertheless, usually malignancy must be conclusively ruled out. Furthermore, often these tumors occur in patients with neurofibromatosis, which raises specific considerations (discussed in the next section). A needle biopsy may be sufficient, but often a surgical biopsy is needed (i.e., via video-assisted thoracoscopic surgery [VATS]). It is generally better to obtain a biopsy first as a separate procedure rather than rely on a frozen section at the time of resection. These tumors are rare and therefore often require careful review by the pathologist before a conclusive diagnosis can be made. In addition, the difference in operative planning for a benign or malignant lesion makes it difficult in most cases to adequately plan for either situation. PET need not be obtained before biopsy but should be done if malignancy is confirmed.
Intrathoracic Tumors in Patients with Neurofibromatosis
Patients with neurofibromatosis present a particular challenge. The effectiveness of prophylactic resection of benign neurofibromas in preventing malignant schwannomas is undefined. This population has a 2% to 5% lifetime risk of a malignant neurofibroma,15 and a 10% risk for intrathoracic tumor has been reported.16 However, it is unclear if this is due to the transformation of a preexisting neurofibroma, spontaneous development of a malignant tumor, or somehow induced by surgical intervention.16 Thus, whether surgical intervention is beneficial, has no impact, or should be avoided is unclear. No data defining the incidence of malignant tumors in various settings (resection, observation) is available.
It certainly seems appropriate to resect a lesion causing symptoms. The presence of neurofibromatosis provides a reasonable argument to remove a single easily resectable lesion even without symptoms. A suspicion of malignancy should be confirmed or ruled out (biopsy, possibly PET imaging) and treated accordingly.
However, patients with neurofibromatosis often present with many lesions, making it relatively difficult or morbid to resect all of them. Furthermore, these patients often present because of a large lesion that is difficult to resect. In the absence of symptoms, intraspinal involvement, or data that resection diminishes the development of malignant tumors, it seems better to avoid surgery. The argument against surgery is strong in the face of difficult resection whereas arguments of benefit are weak at best.
If a decision is made to observe, surveillance should be carried out to rule out growth over time. How often this happens, the rate at which this happens, or even whether this happens is not defined. Furthermore, there are no known patient or lesion characteristics that predict a greater likelihood that a malignant tumor will develop. We suggest MRI is the best imaging test because it avoids radiation, with surveillance at 6- to 12-month intervals. Evidence of growth supports a decision to proceed with resection.
Concern of Malignancy
It is crucial to identify the small subset of patients who have a malignant posterior mediastinal tumor. Certain specific symptoms tend to correlate with malignancy, such as pain or dyspnea.2 Certain specific CT findings have also been found to correlate with malignancy: Size >10 cm, bone destruction, pleural effusion, and mediastinal displacement.2 However, other CT findings—such as erosions, rib splaying, foraminal enlargement, or scoliosis—are common and not suggestive of malignancy.
A clinical or radiographic concern for malignancy necessitates further investigation to confirm the diagnosis (biopsy) and define the extent of disease (PET, MRI). A tissue diagnosis is almost always needed because preoperative chemotherapy is often considered. If a needle biopsy is not diagnostic, a limited surgical biopsy (e.g., VATS) is indicated.
![]() SURGERY
SURGERY
Simple Benign Neurogenic Tumors
Small benign tumors in the asymptomatic patient may be safely observed. If resection is chosen, a VATS approach is recommended. VATS is safe, associated with shorter hospital stays (2.6 vs. 4.5 days) and a more rapid return to work (4.3 vs. 7.7 weeks) compared with thoracotomy.4,27 Operative times are reported as either being similar4 or slightly longer27 with VATS. There is no absolute size limit, and even large tumors may be thoracoscopically mobilized and then removed through a subxiphoid incision without incurring the morbidity of a thoracotomy.
The goal of surgery should be complete excision of the tumor. A margin of normal tissue is not needed. The overlying pleura is incised, followed by primarily blunt dissection in the plane of the capsule of the lesion. These tumors are generally easy to separate from the surrounding tissues. If technical difficulties are anticipated leading to an incomplete resection, it is questionable whether surgery should be undertaken for an asymptomatic simple benign lesion without neural foraminal involvement or mass effect.
Recurrence after complete resection of a benign neurogenic tumor is extremely rare. Even among incompletely resected lesions recurrences are quite low.2
Tumors with Intraspinal or Neural Foraminal Involvement
It is critical to fully assess the presence of and degree of intraspinal involvement whenever there is any suspicion that this might be present. Lack of awareness of a spinal extension may lead to undue traction on the spinal cord, leading to permanent damage. In addition, it could lead to an incomplete resection, leaving an intraspinal component, which is the real concern in the first place. Finally, hemorrhage at the vertebral foramen is particularly difficult to control from a thoracic approach.19 Therefore, preoperative planning and collaboration of surgeons with the appropriate skills is needed.
Older approaches to dumbbell tumors involved two stages—first a laminectomy through a posterior approach followed later by a standard thoracotomy. Single-stage operations have since been described, including a two-position procedure consisting of a posterior vertical midline incision in the prone position followed by thoracotomy in the lateral decubitus position,28 and a single-position (lateral decubitus) approach involving a posterior midline vertical incision starting 5 cm above the foramen of interest and extending 5 cm caudally before curving to meet a standard posterolateral thoracotomy. This approach may provide superior visualization.19 Regardless of positioning, laminectomy and thoracic mobilization of the tumor should take place during the same procedure. No specific intraoperative complications have been reported with either single-stage approach.19,28
Other single-field, posterior-only techniques have been described for dumbbell tumors, with laminectomy for access to the intraspinal component and rib resection for access to the mediastinal component; both components may be performed from a vertical posterior midline incision. The primary advantages of this approach are avoiding a thoracotomy, an extrapleural dissection, and greater stability of the laminectomized spine.29
Several centers use a two-field thoracoscopic approach that combines a posterior laminectomy with VATS mobilization of the intrathoracic component.30 No specific major perioperative complications are common with this approach.
Perioperative and long-term results of resection of tumors with intraspinal involvement are very good regardless of which technique is used. No deaths and no neurologic or other major complications occurred in one series of 16 dumbbell tumors resected using a posterolateral thoracotomy either alone or with a posterior midline extension. No recurrences were seen at a mean follow-up of 7.5 years, including a patient with a malignant schwannoma treated with adjuvant radiotherapy and a patient with lymphoma.20
Large Benign Neurogenic Tumors
Neurogenic tumors that are benign but cause symptoms due to size present a difficult problem. While the presence of symptoms favors resection, one must weigh the symptoms against the complexity and risks associated with resection of large tumors. Because the rate of growth (if any) appears to be quite small, observation is reasonable if symptoms are mild. A fear of malignant degeneration is so poorly substantiated that it should probably not play into decision making. Whether to resect is a judgment call, based on weighing the risks and morbidity of resection, the severity of symptoms, and the rate of growth (as assessed by the progression of symptoms or serial imaging).
A resection should be complete, provided this does not pose excessive risk. These tumors generally push against adjacent structures rather than invading them. However, the morbidity posed by an extensive but complete resection in some cases makes it reasonable to undertake an incomplete resection. There is very little data specifically on large tumors; however, after an incomplete resection recurrence occurs in only a minority of patients and then typically after several years.2,8
Patients with Neurofibromatosis
Patients with neurofibromatosis present a particular conundrum. Arguments both for and against resection are usually stronger but not more clear. Malignant tumors can develop, but it is unclear if this represents de novo development of a malignant tumor or transformation of an existing lesion. Usually there are many tumors, so resection of all of them is not possible. Larger tumors usually are the most concerning but are difficult to resect. There should be greater hesitation to accept an incomplete resection given the possibility of development of a malignant tumor.
The decision to resect is usually driven by the morbidity of resection. If it is easy, it should probably be done, especially if the lesion is causing symptoms. If it is complex, it is probably better to avoid surgery unless there is documented evidence of growth or a suspicion of malignancy.
Malignant Tumors
Malignant posterior neurogenic tumors are treated primarily by surgery (i.e., complete resection). Because these tumors invade surrounding tissues, this includes en bloc removal of adjacent structures. Many centers advocate postoperative radiation for malignant tumors,8 although it is not clear that chemotherapy or radiation is effective.2,7 Survival after treatment is generally short with patients developing a recurrence and/or dying in 2 to 18 months,2–4although rare cases of long-term recurrence-free survival with adjuvant radiotherapy are described.20 There is no evidence suggesting that the specific tumor type matters for the treatment strategy or outcome.
Adult patients with neuroblastoma and ganglioneuroblastoma are exceedingly rare; as of 2011, there were 14 case reports of mediastinal neuroblastoma in adults, only three of which occurred in the posterior mediastinum.5 A 36% 5-year survival rate with treatment has been reported for adult neuroblastoma in any location although the survival appears shorter for mediastinal lesions.6 The optimal therapy for these rare tumors is undefined, but a multimodality approach seems to be warranted.
Infants with neuroblastoma are classified as low, intermediate, or high risk, according to several factors: International Neuroblastoma Staging System (INSS) stage, age, International Neuroblastoma Pathologic Classification (INPC), ploidy, and presence of MYCN oncogene amplification.31 Treatment is generally resection for low-risk, chemotherapy followed by surgery for intermediate-risk, and chemotherapy, surgery, radiation, stem cell transplantation, and immunotherapy for high-risk tumors.31
Complications and their Management
In general, simple benign posterior mediastinal tumors can be resected with minimal morbidity. The complications that arise are mostly general (e.g., wound infection, incisional pain, atelectasis) and will not be discussed. However, resection of extensive tumors can be associated with significant morbidity. This is quite varied and depends on the additional structures involved (e.g., chest wall, aorta, esophagus, heart). More information can be found in other chapters dealing specifically with surgery involving these structures. Finally, complications associated with the spine component of some larger resections (e.g., stabilization) lie beyond the scope of this chapter in the purview of the spine surgeon.
A specific issue relevant to the thoracic surgeon is unrecognized tumor extension into the spinal foramen or canal. The liberal use of MRI whenever the possibility of extension exists (e.g., tumor abutting the foramen) should prevent this. If intraoperative unsuspected extension is found, it is best to abort the procedure and enlist the help of a spine surgeon. There is a danger of incomplete resection, retraction of the nerve root into the spinal canal, and intracanalicular bleeding.
Proximal division of a nerve root (i.e., within the foramen) can result in leakage of cerebrospinal fluid (CSF). It is best to proximally ligate or clip the nerve root to prevent a CSF leak. Intraoperative recognition and involvement of a spine surgeon allows the hole to be closed. Postoperative ongoing leak is heralded by postural headache. Although the leak may resolve with several days of bed rest, a pseudomeningocele may form due to the negative intrathoracic pressure. If the leak persists, reoperation with suture, sealant, or muscle transposition is needed.
Dissection around a nerve root must be done with care. Excessive traction can cause spinal cord damage. Unipolar electrocautery should be avoided to prevent cord injury. Prevention is the key—treatment once it has occurred has little impact.
![]() CONCLUSIONS
CONCLUSIONS
Posterior mediastinal neurogenic tumors are infrequent, and even busy centers encounter only several per year. Therefore, it is difficult to develop a comfortable routine in how to approach these patients. The traditional management has been to resect all posterior mediastinal tumors once identified. However, this approach is largely based on dogma and speculation; with an increase in incidentally detected lesions a more evidence-based approach is needed. Most of these tumors (∼75%) do not cause symptoms and the vast majority (∼95%) are benign. There is no clear evidence that observed tumors will grow, become symptomatic, or develop malignant transformation (except in patients with neurofibromatosis).
Patients can be grouped into five categories. The largest group is asymptomatic with a simple, well-defined, benign lesion. If the tumor is contiguous with the neural foramen or there are bony changes, an MRI should be performed to rule out an intraspinal component. Either observation or resection is reasonable for this group, depending on patient preferences. If undertaken, surgery should involve complete resection (enucleation), ideally by VATS. With either approach, long-term outcomes are excellent.
A complete resection is suggested for patients with an intraspinal tumor, typically involving both a thoracic and a spine surgeon. Although the incidence of growth or development of symptoms in asymptomatic patients appears low, resection is reasonable. Surgical techniques vary but have generally shifted to single-stage and less-invasive approaches; the intraspinal component is usually mobilized first.
Patients with large, benign neurogenic tumors should probably be resected if there are significant symptoms due to intrathoracic organ compression and observed if symptoms are mild, growth appears to be limited, or resection involves major risk. A complete resection is ideal but an incomplete resection is reasonable if it avoids major morbidity.
Patients with neurofibromatosis are challenging. They usually present with multiple tumors, large lesions that are difficult to resect, and a risk (albeit low) of developing a malignant tumor. Whether resection decreases or increases the risk of malignancy is unclear. In general it appears that observation is reasonable unless there are major symptoms, growth, or rarely only limited tumors that are simple to resect.
A careful evaluation and preoperative diagnosis is needed when a malignant posterior mediastinal tumor is suspected. The primary treatment is complete en bloc resection, but outcomes are poor, despite multimodality therapy.
A structured approach to evaluating patients with posterior mediastinal tumor, such as is provided here, can help clinical management. The clinical presentation and CT usually allows patients to be placed in one of five categories. This guides the need for further evaluation and subsequent patient management.
Recommended References and Readings
1. Carter B, Tomiyama N, Bhora F, et al. A modern definition of mediastinal compartments. J Thorac Oncol. 9(9):S97-S101, September 2014.
2. Ribet ME, Cardot GR. Neurogenic tumors of the thorax. Ann Thorac Surg. 1994;58:1091–1095.
3. Cardona S, Schwarzbach M, Hinz U, et al. Evaluation of F18-deoxyglucose positron emission tomography (FDG-PET) to assess the nature of neurogenic tumours. Eur J Surg Oncol. 2003;29:536–541.
4. Yamaguchi M, Yoshino I, Fukuyama S, et al. Surgical treatment of neurogenic tumors of the chest. Ann Thorac Cardiovac Surg. 2004;10:148–151.
5. Ohtaki Y, Ishii G, Hasegawa T, et al. Adult neuroblastoma arising in the superior mediastinum. Interact Cardiovasc Thorac Surg. 2011;13:220–222.
6. Esiashvili N, Goodman M, Ward K, et al. Neuroblastoma in adults: Incidence and survival analysis based on SEER data. Pediatr Blood Cancer. 2007;49:41–46.
7. Bicakcioglu P, Demirag F, Yazicioglu A, et al. Intrathoracic neurogenic tumors. Thorac Cardiovasc Surg. 2013;62(2):147–152.
8. Takeda S, Miyoshi S, Minami M, et al. Intrathoracic neurogenic tumors–50 years’ experience in a Japanese institution. Eur J Cardiothorac Surg. 2004;26:807–812.
9. Banks E, Yum M, Brodhecker C, et al. A malignant peripheral nerve sheath tumor in association with a paratesticular ganglioneuroma. Cancer. 1989;64:1738–1742.
10. Chandrasoma P, Shibata D, Radin R, et al. Malignant peripheral nerve sheath tumor arising in an adrenal ganglioneuroma in an adult male homosexual. Cancer. 1986;57:2022–2025.
11. Ghali V, Gold J, Vincent R, et al. Malignant peripheral nerve sheath tumor arising spontaneously from retroperitoneal ganglioneuroma: A case report, review of the literature, and immunohistochemical study. Hum Pathol.1992;23:72–75.
12. Fletcher C, Fernando I, Braimbridge M, et al. Malignant nerve sheath tumour arising in a ganglioneuroma. Hitopathology. 1988;12:445–454.
13. Rozmus J, Langer M, Murphy J, et al. Multiple persistent ganglioneuromas likely arising from the spontaneous maturation of metastatic neuroblastoma. J Pediatr Hematol Oncol. 2012;34:151–153.
14. Kulkarni A, Bilbao J, Cusimano M, et al. Malignant transformation of ganglioneuroma into spinal neuroblastoma in an adult. J Neurosurg. 1998;88:324–327.
15. Ferner RE, Gutmann DH. International consensus statement on malignant peripheral nerve sheath tumors in neurofibromatosis. Cancer Res. 2002;62:1573–1577.
16. Nordback P, Halkic N, Boumghar M. Intrathoracic tumours in van Recklinghausen’s neurofibromatosis. Schweiz Med Wochenschr. 2000;130:1105–1111.
17. Strollo DC, Rosado-de-Christenson ML, Jett JR. Primary mediastinal tumors: Part II. Tumors of the middle and posterior mediastinum. Chest. 1997;112:1344–1357.
18. Davidson KG, Walbaum PR, McCormack RJ. Intrathoracic neural tumours. Thorax. 1978;33:359–367.
19. Grillo HC, Ojemann RG, Scannell JG, et al. Combined approach to “dumbbell” intrathoracic and intraspinal neurogenic tumors. Ann Thorac Surg. 1983;36:402–407.
20. Shadmehr MB, Gaissert HA, Wain JC, et al. The surgical approach to “dumbbell tumors” of the mediastinum. Ann Thorac Surg. 2003;76:1650–1654.
21. Ricci C, Rendina EA, Venuta F, et al. Surgical approach to isolated mediastinal lymphoma. J Thorac Cardiovasc Surg. 1990; 99:691–695.
22. Murphey MD, Smith WS, Smith SE, et al. From the archives of the AFIP. Radiographics. 1999;19:1253–1280.
23. Son J, Ahn M, Cho K, et al. Varying degrees of FDG uptake in multiple benign neurofibromas on PET/CT. Br J Radiol. 2007;80:e222–e226.
24. Shields TW. Primary tumors and cysts of the mediastinum. In: Shields TW, LoCicero J, Ponn RB, eds. General Thoracic Surgery. Philadelphia, PA: Lea & Febiger; 1983:927–954.
25. Weisbrod G, Lyons D, Tao L, et al. Percutaneous fine-needle aspiration biopsy of mediastinal lesions. AJR Am J Roentgenol. 1984;143:525–529.
26. Dahlgren S, Ovenfors C. Aspiration biopsy diagnosis of neurogenous mediastinum tumors. Acta Radiol. 1970;10:289–296.
27. Bousamra M, Haasler G, Patterson G, et al. A comparative study of thoracoscopic vs open removal of benign neurogenic mediastinal tumors. Chest. 1996;109:1461–1465.
28. Akwari O, Payne W, Onofrio B, et al. Dumbbell neurogenic tumors of the mediastinum. Diagnosis and management. May Clin Proc. 1978;53:353–358.
29. Osada H, Aoki H, Yokote K, et al. Dumbbell neurogenic tumor of the mediastinum: A report of three cases undergoing single-staged complete removal without thoracotomy. Jpn J Surg. 1991; 21:224–228.
30. Citow J, MacDonald R, Ferguson M. Combined laminectomy and thoracoscopic resection of dumbbell neurofibroma: Technical case report. Neurosurg. 1999;45:1263–1266.
31. Health NCIaNIo. Treatment option overview for neuroblastoma. In: Neuroblastoma Treatment (PDQ (R)). Washington, DC: National Institutes of Health; 2013.
32. Detterbeck F. Clinical approach to mediastinal masses. In: Kuzdzal J, Muller M, Papagiannopoulos K, et al., eds. ESTS Textbook of Thoracic Surgery. European Society of Thoracic Surgeons; 2013.