Dominique Fabre and Philippe Dartevelle
![]() INDICATIONS
INDICATIONS
It is generally agreed that the maximum length of tracheal resection that can be repaired by end-to-end anastomosis is 6 cm. Primary tracheal neoplasms (including adenoid cystic carcinomas [ACCs], squamous cell carcinomas [SCCs]) and other tracheal diseases can be usually managed by tracheal resection with primary anastomoses. However, there are diseases that require resection of segments of trachea longer than 6 cm also requiring reconstruction with a tracheal substitute.
Tracheal replacement for disease invading 6 to 12 cm of the trachea can be performed with the use of reliable autologous free fasciocutaneous flaps in combination with autologous cartilage struts. Previously, circumferential resection with release procedures (suprathyroid or suprahyoid laryngeal release) and direct anastomosis must be considered before this procedure. The most reliable flap for this reconstruction is the forearm free flap that could replace the entire membranous wall of the trachea for extended tracheoesophageal fistulae (TOF). Reinforced with cartilage ribs this autologous tracheal substitution (ATS) using the forearm free flap can replace the entire trachea from the cricoid cartilage to the carina. Indications are primary tracheal neoplasm (including ACC and SCC), secondary tracheal neoplasm (thyroid carcinoma, thymic carcinoma, etc.) and extended TOF (postintubation tracheal destruction, tracheal necrosis after lymphoma, etc.). Benign and malignant neoplasms have to be discussed by a multidisciplinary team before surgery.
![]() CONTRAINDICATIONS
CONTRAINDICATIONS
Determination of Resectability
Tracheal resection was considered when complete resection of gross airway disease appeared feasible. An Assessment of locoregional and distant metastatic disease has to be performed before resection. Preoperative radiation therapy is associated with a higher incidence of complications but is not a contraindication for ATS. Bronchoscopy has to be performed before resection to assess the presence and extent of luminal invasion. Patients with SCC and N2 disease should be contraindicated for ATS. N2 disease is assessed using preferentially EBUS, EUS, or mediastinoscopy. Bilateral recurrent nerve involvement is most of the time an indication for laryngectomy. Involvement up to the cricoid or thyroid cartilage and extended esophageal involvement are considered as contraindications to this extensive and difficult surgery. But a partial laryngotracheal resection could be performed in few cases. On bronchoscopy, the lengths of involved airway define the length for tracheal reconstruction that is limited to 12 cm. Tracheal tumor extension up to the carina and extended esophageal invasion are currently a contraindication for ATS. Indeed, the main limitation of the neotrachea is the absence of mucociliary clearance because its inner aspect is covered by a squamous epithelium. The quality of the mucociliary clearance is correlated to the resection length. We currently do not recommend this technique to treat lesions that extend to the main bronchi and for patients with pulmonary and diaphragmatic dysfunction of sufficient magnitude to interfere with effective coughing. The other limits of this technique are also chronic respiratory insufficiency and cartilage calcifications (risk of cartilage fracture).
![]() PREOPERATIVE PLANNING
PREOPERATIVE PLANNING
Patient Selection
Because of the high risks of this surgery a careful preoperative assessment of each patient is required. Ethics approval should be granted by the Ethics Board and individual patient consent should be obtained. For neoplasms, a multidisciplinary consultation (including thoracic oncologist and thoracic surgeon) before surgery is needed to confirm the indication for ATS. Patients with an extended primary tracheal neoplasm, a secondary tracheal neoplasm, or an extended tracheal destruction are the main indication for this ATS. Indication for ATS in case of ACC with lung metastasis should be carefully weighted by the actual results of radiation therapy.
Patients should be carefully screened from a general medical point of view. The age, sex, histology, preoperative medical history, pulmonary function test, performance, laboratory tests, tumor location, vocal cords function, and cardiac function should be carefully assessed. The diagnostic staging modalities included bronchoscopy, computed tomography (CT) scanning, and positron emission tomography-computed tomography (PET-CT). Echocardiography and stress thallium are used when indicated. Angiography of supra-aortic arteries should be performed to warrant the patency of the donor vessel for microvascular anastomosis of the free flap. Furthermore, involvement and patency of the supra-aortic arteries has to be assessed before surgery. Allen’s test must be performed on both sides (the color of the hand should return to normal in 7 seconds) to confirm the possibility for forearm free flap harvesting. Predicted postoperative forced expiratory volume in 1 second should be more than 70%. Indeed, respiratory failure is a contraindication for ATS due to the increasing pressure on the neotracheal wall during inspiratory depression. Indeed, respiratory insufficiency can lead to late cartilage fracture inserted between the dermal layers of the free flap.
![]() SURGERY
SURGERY
The patient is positioned supine, anesthetized and intubated with a single-lumen endotracheal tube and then prepped and draped in the usual fashion. With a warming system, a pillow is placed transversally in the back at the middle of the sternum. An inflatable bag beneath the patient’s shoulders (“thyroid bag”) is very useful and could be deflated, giving a measure of cervical flexion. For this long operation two teams of surgeons are working together. The entire chest and entire neck is prepped with a chlorhexidine–alcohol solution and then draped with sterile towels and sheets. For the forearm free flap harvesting, the chosen arm should be placed on an arm table usually placed at 90 degrees. ATS is performed by an open approach through a transverse cervicotomy with a median vertical sternotomy. The groin and one thigh should also be placed in the operative field. The groin could be used for cannulation and one of the thighs should be prepared for skin graft on the harvested arm. Cartilage harvesting is usually performed on the opposite site from the free flap harvesting.
The first part of the surgery is the tracheal resection including firstly determination of local resectability. After this part performed by the thoracic team, the reconstruction team can start the flap harvesting. During this, cartilages could be harvested on the contralateral side. Usually, the operating thoracic surgeon stands on the right side of the patient while the assistant is on the contralateral side. The operating plastic surgeon stands on the opposite side if the forearm free flap is performed on the left side.
Tumor Resection
Before completing the construction of the neotracheal conduit, the damaged trachea is approached by cervicotomy and median sternotomy. The diseased trachea is resected on healthy margins and the autologous conduit sutured to the native tracheal stumps (i.e., to the tracheobronchial bifurcation below and to the larynx above). If necessary, in addition to removing the trachea, partial resection of the esophagus and removal of one of the recurrent nerves and possibly of the adjacent vascular structures may be needed. During this stage of the procedure, respiratory gas exchange is provided by ventilation with an intubation tube inserted into the bronchi through the surgical field. Alternatively, extracorporeal circulation (ECC) between the right atrium and the ascending aorta can be instituted.
ATS Construction
The neotracheal conduit is constructed from a large, rectangular fasciocutaneous flap harvested from the patient’s forearm (Fig. 42.1). The skin of this flap is rotated around a silicone tube, the diameter of which is approximately slightly larger than that of the normal trachea. The tubular conduit, supplied by the radial artery and vein, is made of skin along its inner aspect and of fascia in its outer aspect. Before transforming the fasciocutaneous rectangle of the forearm free flap into a tube by suturing the main lengths together, several costal cartilage segments obtained from the patient’s rib cage have to be inserted between skin and fascia in the subcutaneous tissue to ensure the transverse rigidity of the tube. Through a transverse latero-sternal approach, the cartilage junction of the fifth, sixth, and seventh ribs are exposed. Typically, six or seven cartilaginous segments (5 mm in width, 2 mm in thickness, and about 9 cm long) are elevated from the most caudal ribs and then slid in place taking care at not injuring the skin vascular supply from the perforators. Transillumination should be used to locate the perforating vessels. A tunnel is created for each cartilage just under the skin with Metzenbaum Scissors. Halstead forceps are used to insert the cartilage ribs in all the different tunnels. The flap is then rolled around a silicone stent (Tracheobronxane Dumon silicone stent Novatech). We recommend to use a simple straight tube or a Y tube when the distal tracheal suture is close to the carina. The caliber should be preoperatively sized up based on the CT scan. The structural rigidity of the neotracheal “cylinder” is then obtained by suturing the skin around the stent and the ends of each costal cartilage between each other (using a polypropylene monofilament 4/0 [Prolene]) so that this framework is included in the wall of the conduit without coming into contact with the lumen of the trachea. During the phase of construction of this totally autologous neotrachea, the flap remains vascularized by its radial pedicle dissected up to the level of the elbow. This pedicle will be divided only immediately before its final implantation in the chest cavity to minimize the flap ischemic time (Fig. 42.2). The last step is to suture the fascia of the forearm free flap around the cartilages. This step is major to avoid mediastinal structures’ erosion. The fascia should be harvested to be enough large to ensure this cartilage coverage (1 cm on each side of the flap).
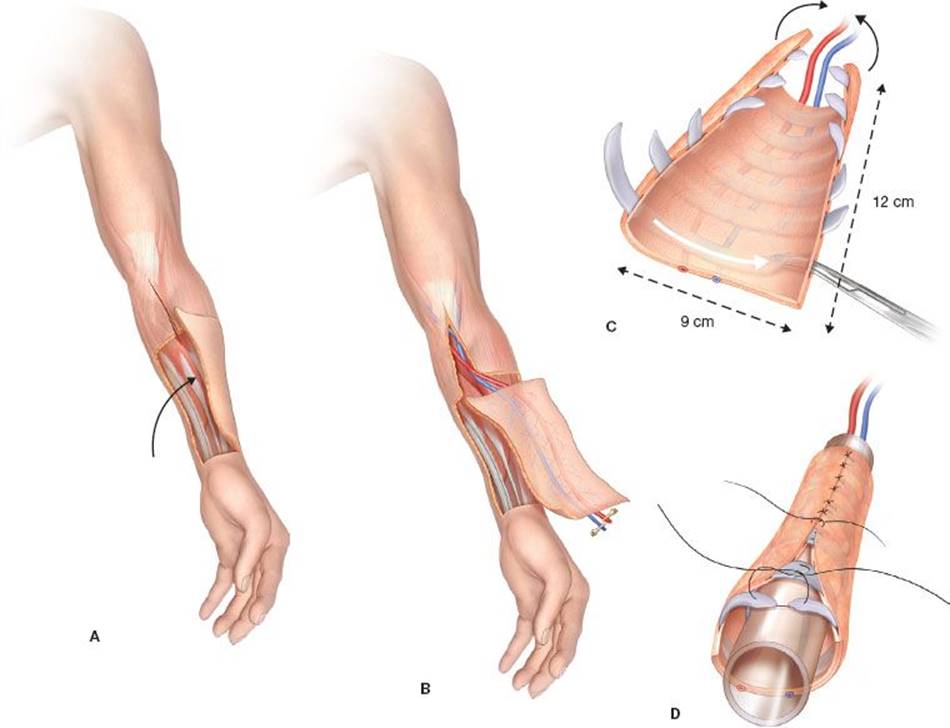
Figure 42.1 Surgical procedure for autologous tracheal replacement: A: First step: Flap harvesting; B: Second step: Vessels dissection; C: Third step: Cartilage rib insertion; D: Fourth step: Construction of the neotrachea conduit.
Tracheal Replacement
Revascularization of the flap is provided by microanastomoses performed with a 4 × 0 magnification microscope between the radial vessels of the flap and small neck vessels (with polypropylene monofilament 9/0 [Prolene] or 10/0 Nylon) (Fig. 42.3). The most frequently used arteries are the internal thoracic artery or branches of the subclavian artery or external carotid artery. The duration of microvascular anastomosis must be the shortest. Technically, the distal tracheal anastomosis has to be done firstly before the microvascular anastomosis. For this tracheal anastomosis, a silicone stent (preoperatively sized) is placed before performing the anastomosis and cut just to cover the level of the anastomosis. Both tracheal anastomosis are performed with a posterior continuous suture and an anterior interrupted suture (4/0 PDS or 4/0 synthetic polyglycolic acid sutures [Vicryl, coated]). Careful calibration of the conduit proved necessary to counteract narrowing of the neotracheal lumen due to flap edema and high inspiratory negative pressures generated by bronchial congestion and laryngeal edema.

Figure 42.2 Free antebrachial fasciocutaneous flap reinforced with cartilage ribs before mediastinal implantation.

Figure 42.3 Extended tracheal autologous reconstruction after tumor resection, microvascular anastomosis, and tracheal anastomosis.
Intercricothyroid transitory tracheostomy has to be performed at the end of the procedure after closure of all incisions. A simple skin graft is harvested on the thigh with a dermatome to cover the forearm. A forearm splint/cast is then applied for 21 days.
![]() POSTOPERATIVE MANAGEMENT
POSTOPERATIVE MANAGEMENT
Immediately postoperatively, patients are transported intubated to the ICU for constant monitoring. Follow-up visits may include the following: Complete physical examination, chest x-ray, blood gas, and daily bronchoscopy through tracheostomy.
Prior to extubation, patients undergo bronchoscopy to ensure adequate clearance of secretions. Following extubation, the chest tubes are removed in the absence of an air leak, commonly within 48 hours postoperatively. The cervical drainage is removed after negative bacteriologic culture of the daily sample.
Temporary tracheotomy is recommended for all patients with a complete tracheal replacement. In addition to daily bronchoscopies, bacteriologic sampling, and postural drainage, we also believe that the postoperative management should be supervised by an experienced surgeon in tracheal surgery.
As an average, the airway stent has to be removed bronchoscopically usually 1 week after surgery. In all instances, the flap should be endoscopically checked once daily for satisfactory healing of the anastomoses and viability. Frequent bronchoscopies are needed to clear retained secretions. Transitory tracheotomy is associated due to absence of mucociliary clearance and a medical humidifier must be used.
Patients should be followed and controlled with bronchoscopy at 1 month, 6 months, 1 year, and every year. An adjuvant treatment has to be associated in case of R1 resection for ACC on the proximal and distal anastomosis.
Viability and Collapsibility of the Flap
Viability is usually easily assessed by bronchoscopy: Color of the flap is the best indicator of viability. The anastomotic integrity is directly checked. All flaps should remain viable; the cartilaginous framework prevents inspiratory collapse. There is a late contrast enhancement of the cartilages and of the skin. Pedicles are controlled. CT reconstructions are performed to check the cartilages in the skin flap. In one patient, a late fracture of the cartilages appears with the need for tracheostomy.
![]() COMPLICATIONS
COMPLICATIONS
The mean length of hospital stay is around 1 month.
Major complications are:
![]() Arterial thrombosis of the free flap: Usually checked under bronchoscopy (ischemic “white” flap) but can be followed using portable Doppler in the ICU. Thrombosis of the arterial anastomosis is usually signaled by loss of cutaneous capillary refill, a decrease in the temperature of the skin, and a loss of flap turgor. Those parameters are difficult to check inside the tracheal lumen through a silicone stent. Color changes are also the best indicator for arterial thrombosis and should be followed by a CT scan. When recognized early and managed promptly (<6 hours) the patient must be reoperated for anastomosis revision. Streptokinase may be used in relevant flap vessels for flap salvage in anastomotic thrombosis.
Arterial thrombosis of the free flap: Usually checked under bronchoscopy (ischemic “white” flap) but can be followed using portable Doppler in the ICU. Thrombosis of the arterial anastomosis is usually signaled by loss of cutaneous capillary refill, a decrease in the temperature of the skin, and a loss of flap turgor. Those parameters are difficult to check inside the tracheal lumen through a silicone stent. Color changes are also the best indicator for arterial thrombosis and should be followed by a CT scan. When recognized early and managed promptly (<6 hours) the patient must be reoperated for anastomosis revision. Streptokinase may be used in relevant flap vessels for flap salvage in anastomotic thrombosis.
![]() Venous thrombosis of the free flap: Usually checked under bronchoscopy (ischemic “purple” flap) and very difficult to check with Doppler. This can be managed with a novel ATS reconstruction.
Venous thrombosis of the free flap: Usually checked under bronchoscopy (ischemic “purple” flap) and very difficult to check with Doppler. This can be managed with a novel ATS reconstruction.
![]() Deep cervical abscess: Usually checked by CT scan control. All abscesses should be drained mostly through a surgical approach.
Deep cervical abscess: Usually checked by CT scan control. All abscesses should be drained mostly through a surgical approach.
![]() Mediastinal fistulae of the ATS: The management is different for patient with mechanical ventilation or without.
Mediastinal fistulae of the ATS: The management is different for patient with mechanical ventilation or without.
![]() Pulmonary infection and acute respiratory distress syndrome: All patients operated on for ATS have to be early extubated but they all present at least one pulmonary infection leading in ARDS (58% of cases). Thereby daily bronchoscopic aspiration should be performed. Patients usually do not need any mechanical ventilation and breathe through the tracheostomy tube during the first days. But in case of ARDS, mechanical ventilation is necessary. Those complications are usually managed with antibiotics but extracorporeal membrane oxygenator (ECMO) could be discussed for severe ARDS to avoid high-pressure ventilation. Indeed high-pressure mechanical ventilation could lead to mediastinal fistulae.
Pulmonary infection and acute respiratory distress syndrome: All patients operated on for ATS have to be early extubated but they all present at least one pulmonary infection leading in ARDS (58% of cases). Thereby daily bronchoscopic aspiration should be performed. Patients usually do not need any mechanical ventilation and breathe through the tracheostomy tube during the first days. But in case of ARDS, mechanical ventilation is necessary. Those complications are usually managed with antibiotics but extracorporeal membrane oxygenator (ECMO) could be discussed for severe ARDS to avoid high-pressure ventilation. Indeed high-pressure mechanical ventilation could lead to mediastinal fistulae.
![]() Brachiocephalic artery rupture: This complication has to be surgically and quickly managed. Erosion of the posterior part of the innominate artery by the neotrachea has to be prevented by interposition of tissue.
Brachiocephalic artery rupture: This complication has to be surgically and quickly managed. Erosion of the posterior part of the innominate artery by the neotrachea has to be prevented by interposition of tissue.
![]() Hand ischemia: Usually, the nondominant arm is selected as the donor site. Negative Allen’s test results are prerequisite for forearm free flap. If hand ischemia is a concern based on the results of a preoperative Allen’s test, alternative flaps may be necessary.
Hand ischemia: Usually, the nondominant arm is selected as the donor site. Negative Allen’s test results are prerequisite for forearm free flap. If hand ischemia is a concern based on the results of a preoperative Allen’s test, alternative flaps may be necessary.
![]() RESULTS
RESULTS
Between August 2004 and December 2012, we treated 12 consecutive patients who required ATS after resection of the trachea. They included six men and six women, with a mean age of 50.6 years (range from 37 to 68 years). The lengths of the tracheal involvement lead to mean length of tracheal resection of 10.5 cm (range from 8 to 12 cm). Indications were eight primary tracheal neoplasms (including five ACCs and three SCCs), three secondary tracheal neoplasms (including one thyroid carcinoma and two tracheal lymphomas), and one postintubation-extended tracheal destruction after long history of stenting. Full-length resection from the first cartilaginous ring to the carina was required in five patients upon 12 with an extended circular tracheal resection (41.6%). When locally invaded, recurrent laryngeal nerve was resected but we did not observe any recurrent laryngeal nerve injury in our experience. For two patients, tracheal resection included the carina and both main bronchi; one of these patients also needed a right upper lobectomy (RUL). Total laryngectomy with ATS was performed in three cases with end stomas. Cardiopulmonary bypass (CPB) was used in six cases upon 12 to avoid intermittent ventilation of the left main bronchus in patients with extension to the carina. CPB is started before dividing the distal trachea and stopped just after performing distal tracheal anastomosis.
After ATS, two patients suffered pneumonia and ARDS after replacement of the trachea and the main bronchi. One of them was treated with a venovenous ECMO. In both instances, the flaps remained viable and functional until the patients died from respiratory infection and excessive bronchial congestion.
Eight patients are currently alive, without respiratory dysfunction, living normal lives 101, 75, 66, 41, 34, 20, 18, and 4 months postoperatively. Two patients died of cancer recurrence at 6 months and of lung metastases at 16 months after surgery.
One patient with a chronic severe respiratory insufficiency required a distal and short stent and two others have a permanent tracheostomy. Tracheostomy was required for severe respiratory insufficiency related to lumen collapse from a break of the calcified rings of costal cartilage. The other one was at the level of the postoperative tracheostomy which was performed through the upper airway anastomosis.
The two patients who complained of dysphagia before the procedure due to extensive esophageotracheal fistulas were able to resume oral feeding after surgery. Repeat endoscopy and dynamic CT scan demonstrated satisfactory patency of the neotrachea without inspiratory collapse during the immediate postoperative period, when the respiratory cycle increases the endoluminal negative pressure, as well as at several years after the procedure. The Kaplan–Meier survival analysis for 11 patients with cancer indicates a 64.8% of survival at 5 years.
![]() CONCLUSIONS
CONCLUSIONS
ATS is a reliable tracheal substitute, totally autologous, well vascularized, and resistant to infection. Transversally rigid is enough to resist respiratory pressure variations. On the other hand because of the absence of a mucociliary clearance of the skin epithelium, transitory tracheostomy and postural drainage are mandatory. We have shown that extensive tracheal diseases previously not amenable to tracheal replacement can now be successfully managed with the use of totally autologous conduits made of fasciocutaneous free flaps reinforced by costal cartilages. This ATS has nearly all the qualities of the ideal tracheal substitute. The absence of a ciliated epithelial lining to provide mucociliary clearance remains a limiting aspect of this method. Whereas excellent results can be achieved with autologous neotracheal, fasciocutaneous cartilage-supported tube grafts, we currently do not recommend this method to treat lesions that extend to the main bronchi or for patients with pulmonary and diaphragmatic dysfunction of sufficient magnitude to interfere with effective coughing. Research to develop a method for lining the neotrachea with ciliated respiratory epithelium is needed.
Recommended References and Readings
Beldholm BR, Wilson MK, Gallagher RM, et al. Reconstruction of the trachea with a tubed radial forearm free flap. J Thorac Cardiovasc Surg. 2003;126:545–550.
Davidson MB, Mustafa K, Girdwood RW. Tracheal replacement with an aortic homograft. Ann Thorac Surg. 2009;88:1006–1008.
Delaere P, Vranckx J, Verleden G, et al. Tracheal allotransplantation after withdrawal of immunosuppressive therapy. N Engl J Med. 2009;362:138–145.
Fabre D, Kolb F, Fadel E, et al. Successful tracheal replacement in humans using autologous tissues: An 8-year experience. Ann Thorac Surg. 2013;96:1146–1155.
Fabre D, Singhal S, De Montpreville V, et al. Composite cervical skin and cartilage flap provides a novel large airway substitute after long-segment tracheal resection. J Thorac Cardiovasc Surg. 2009;138:32–39.
Grillo HC. Tracheal replacement: A critical review. Ann Thorac Surg. 2002;73:1995–2004.
Lenot B, Macchiarini P, Dulmet E, et al. Tracheal allograft replacement. An unsuccessful method. Eur J Cardiothorac Surg. 1993;7:648–652.
Macchiarini P, Jungebluth P, Go T, et al. Clinical transplantation of a tissue-engineered airway. Lancet. 2008;372:2023–2030.
Macedo A, Fadel E, Mazmanian GM, et al. Heterotopic en bloc tracheobronchial transplantation with direct revascularization in pigs. J Thorac Cardiovasc Surg. 2004;127:1593–1601.
Pearson FG, Thompson DW, Weissberg D, et al. Adenoid cystic carcinoma of the trachea. Experience with 16 patients managed by tracheal resection. Ann Thorac Surg. 1974;18:16–29.
Wurtz A, Porte H, Conti M, et al. Surgical technique and results of tracheal and carinal replacement with aortic allografts for salivary gland-type carcinoma. J Thorac Cardiovasc Surg. 2010;140:387–393.