Nancy D. Ciesla & Jill D. Kuramoto
INTRODUCTION
Recent literature supports the need for early mobility in the intensive care unit (ICU), and that patients can be safely mobilized.1–6 Acute respiratory distress syndrome (ARDS) survivors report significant impairments in quality of life, including physical functioning, which may be more impaired than respiratory function.7,8 One year following mechanical ventilation for at least 48 hours more than half of the survivors required caregiver support at home.9
New evidence suggests 7 day per week physical therapy as part of a protocol-driven mobility team is associated with earlier mobilization out of bed, ambulation, and decreased ICU and hospital length of stay.4,5 Therefore, support and demand for physical therapy interventions are increasing in the ICU, particularly with mechanically ventilated patients.
Mechanically ventilated patients are often acutely ill, hospitalized in an ICU, and connected to a plethora of lines, tubes, and monitors to sustain life. Examining medically and surgically complex patients with all of this paraphernalia can be quite intimidating for both the novice and experienced physical therapist with little training in the critical care setting. This chapter provides a basic understanding of the physiological aspects of pulmonary function related to mechanical ventilation. Respiratory failure is defined, and the student is introduced to the criteria used to initiate and discontinue mechanical ventilatory support. Commonly utilized modes of mechanical ventilation are described to enable the entry-level therapist to examine and safely treat patients requiring artificial ventilation. This knowledge can be utilized not only in the acute care setting but also in rehabilitation, subacute, and home care settings, where greater numbers of patients are being discharged with a continued need for mechanical ventilation. Physical therapy examination and interventions are described in detail using a case demonstration of a monitored and mechanically ventilated tetraplegic patient with a complete lesion at the C5 level on the American Spinal Injury Association impairment scale, the standard neurological classification of spinal cord injury. Physical therapy interventions such as secretion clearance techniques, breathing exercises, therapeutic exercises, and functional mobility training may assist the patient in being weaned from a ventilator and improve functional outcomes. The risks of mechanical ventilation, ICU interventions, and immobility are discussed throughout this chapter, with an introduction to evidence-based practice and the future for physical therapists working with mechanically ventilated patients.
DESCRIPTION OF PATTERN 6F
A practice pattern has been developed by the American Physical Therapy Association for patients who are in respiratory failure. This Practice Pattern, Pattern 6F, Impaired Ventilation and Respiration/Gas Exchange Associated with Respiratory Failure, is the basis for this chapter (Fig. 19-1).10 Mechanical ventilation is frequently required until the cause of respiratory failure is improved, removed, or reversed. This chapter addresses patients who require mechanical ventilation 24 hours per day and who may require weaning to be liberated from mechanical ventilatory support. A description of the modes of mechanical ventilation, including continuous positive airway pressure and bilevel ventilation, is included.
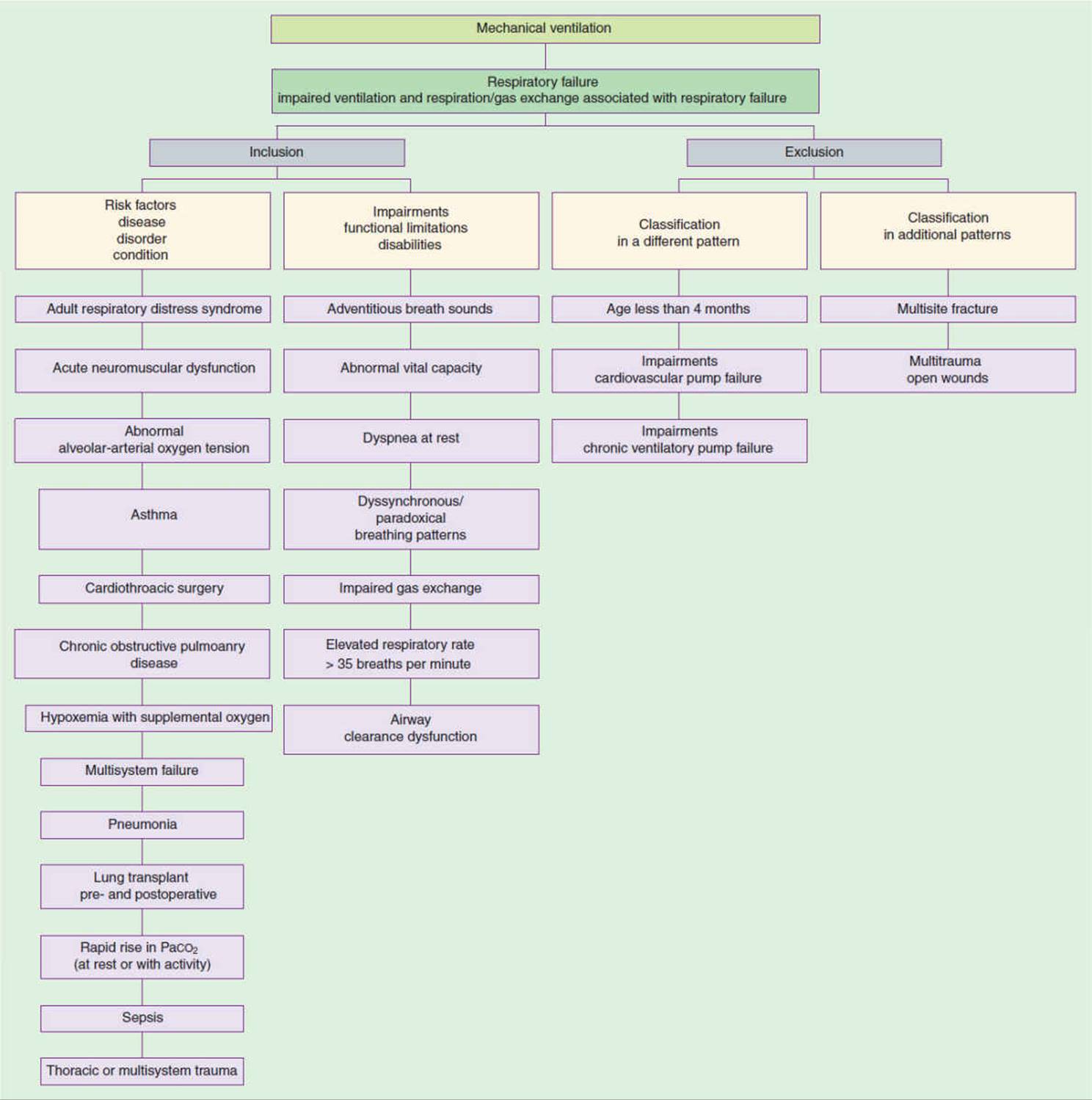
FIGURE 19-1 Patient/client diagnostic classification adapted from Practice Pattern 6F. (Reproduced with permission from American Physical Therapy Association. Guide to Physical Therapist Practice, 2nd ed. Phys Ther. 2001 Jan;81:539–553.)
RESPIRATORY PHYSIOLOGY: APPLICATION TO THE MECHANICALLY VENTILATED PATIENT
During normal breathing, inspired air passes from the mouth through the trachea and bronchial tree to the alveoli where most gas exchange takes place. Oxygen diffuses across the alveolar capillary membrane into the circulating blood where it binds with hemoglobin. Oxygen is then carried from the lungs to the capillary beds that are present in all metabolically active tissues. Oxygen then dissociates from hemoglobin and diffuses into the cells. (Refer to Chapter 5 for a description of oxyhemoglobin dissociation.) For adequate oxygenation, there must be sufficient oxygen carried in the blood, or oxygen-carrying capacity. The total oxygen concentration of a sample of blood (dissociated oxygen and the oxygen combined with hemoglobin) is determined using the formula11:
CaO2 = (1.39 × Hb × Sat/100) + 0.003 PO2
Hb refers to hemoglobin concentration in grams per 100 mL; Sat is the arterial hemoglobin saturation as a percentage; and PO2 is the partial pressure of oxygen dissolved in the blood.11,12 With this simple equation, note that nearly all the oxygen carried in the blood is bound to hemoglobin, and only a small clinically insignificant amount is transported and dissolved in the plasma (approximately 1%–2%). Hypoxemia results when either the lungs are unable to diffuse sufficient oxygen to saturate hemoglobin or the red blood cell count is insufficient.
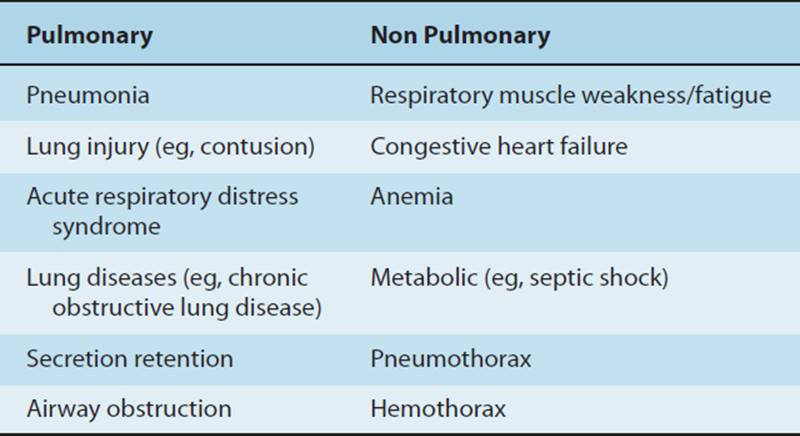
Normally, a healthy person breathing room air has a hemoglobin concentration of 15 g/100 mL of blood, an arterial saturation of 97.5%, and a PO2 of 100 mmHg. Therefore, a normal arterial oxygen concentration is approximately 20.8 mL of O2/100 mL of blood.11 Venous blood contains less saturated hemoglobin, about 15 mL of O2/100 mL of blood. When patient oxygen-carrying content is low due to low hemoglobin, a blood transfusion may be necessary, yet the patient may have normal lung function and normal oxygen saturation. However, if the oxygen-carrying content is low due to a low oxygen saturation and PO2, the appropriate treatment may be to deliver supplemental oxygen. Supplemental oxygen is defined as a percentage of oxygen greater than 21% (room air contains 21% oxygen). A patient may receive supplemental oxygen through a face mask, tracheostomy collar, or nasal prongs, which require the patient to breathe independently, or oxygen may be delivered via positive pressure using a mechanical ventilator. After administering supplemental oxygen, the medical team must determine the cause of the hypoxemia. The cause of hypoxemia may be either pulmonary (lung injury or disease) or nonpulmonary (respiratory muscle or metabolic) dysfunction. A patient with severe chronic obstructive pulmonary disease (COPD) or a lung contusion may be hypoxemic as a result of secretion retention, lung injury, lung disease, or an obstruction in the tracheobronchial tree, all pulmonary causes of hypoxemia. A patient who has a chest radiograph that denotes a severe hemothorax compressing the lung would have a nonpulmonary cause of hypoxemia. Once a chest tube is inserted and blood is drained from the pleural cavity, the patient’s PaO2 and SaO2 are likely to improve. A patient in respiratory failure as a result of septic shock would have a metabolic, nonpulmonary indication for mechanical ventilation (Table 19-1).
TABLE 19-1 Common Causes of Hypoxemia/Hypoxia
The matching of ventilation to perfusion (![]() ) is the principal determinant of PaO2.
) is the principal determinant of PaO2. ![]() is the matching of ventilation (respiratory gases) to perfusion (pulmonary blood). Shunt refers to the amount of blood entering the left heart without passing through ventilated areas of the lung.11,13 Ninety-five percent of the blood entering the pulmonary circulation passes through the alveoli and equilibrates with inspired respiratory gases. A shunt greater than 20% may indicate the need for mechanical ventilation.12 Arterial oxygen saturation and PaO2 may be decreased because of
is the matching of ventilation (respiratory gases) to perfusion (pulmonary blood). Shunt refers to the amount of blood entering the left heart without passing through ventilated areas of the lung.11,13 Ninety-five percent of the blood entering the pulmonary circulation passes through the alveoli and equilibrates with inspired respiratory gases. A shunt greater than 20% may indicate the need for mechanical ventilation.12 Arterial oxygen saturation and PaO2 may be decreased because of ![]() mismatching, alveolar hypoventilation, anatomic right to left shunt, decreased ambient oxygen, and a limitation in diffusion.11 Physical therapists working with mechanically ventilated patients should review the most recent arterial blood gases or oxygen saturation prior to physical therapy interventions. For example, a patient breathing room air with a
mismatching, alveolar hypoventilation, anatomic right to left shunt, decreased ambient oxygen, and a limitation in diffusion.11 Physical therapists working with mechanically ventilated patients should review the most recent arterial blood gases or oxygen saturation prior to physical therapy interventions. For example, a patient breathing room air with a ![]() mismatch exceeding 20% may have a PaO2 of only 50 mmHg. Mechanical ventilation may be necessary if increasing the FiO2 with spontaneous breathing is unsuccessful. Physical therapy may be indicated once the patient is adequately oxygenated.
mismatch exceeding 20% may have a PaO2 of only 50 mmHg. Mechanical ventilation may be necessary if increasing the FiO2 with spontaneous breathing is unsuccessful. Physical therapy may be indicated once the patient is adequately oxygenated.
Minute Ventilation
Normally, an adult patient’s minute ventilation is about 7.5 L/min. Total minute ventilation is the sum of alveolar ventilation (![]() A) and dead space ventilation (
A) and dead space ventilation (![]() D). Alveolar ventilation represents the volume of gas that reaches the respiratory zone, and is therefore available for gas exchange, and accounts for approximately two-thirds of normal minute ventilation. Dead space ventilation may be considered in two different ways, either anatomic or physiologic. Anatomic dead space represents the volume of the conducting airways, whereas physiologic dead space represents the volume of the lung that does not exchange carbon dioxide (CO2). Normally, anatomic and physiologic dead space are virtually the same, but in the patient with pulmonary dysfunction, physiologic dead space may be greater due to
D). Alveolar ventilation represents the volume of gas that reaches the respiratory zone, and is therefore available for gas exchange, and accounts for approximately two-thirds of normal minute ventilation. Dead space ventilation may be considered in two different ways, either anatomic or physiologic. Anatomic dead space represents the volume of the conducting airways, whereas physiologic dead space represents the volume of the lung that does not exchange carbon dioxide (CO2). Normally, anatomic and physiologic dead space are virtually the same, but in the patient with pulmonary dysfunction, physiologic dead space may be greater due to ![]() mismatch.11 The ventilator usually supplies a calculated minute ventilation, or the therapist may be able calculate it by multiplying the respiratory rate by the tidal volume if tidal volume has been set on the ventilator. For a patient receiving mechanical ventilation, the expired minute volume is a combination of mandatory machine breaths and patient-initiated breaths. During physical therapy interventions, the physical therapist should note any significant changes in respiratory rate, tidal volume, or minute ventilation.
mismatch.11 The ventilator usually supplies a calculated minute ventilation, or the therapist may be able calculate it by multiplying the respiratory rate by the tidal volume if tidal volume has been set on the ventilator. For a patient receiving mechanical ventilation, the expired minute volume is a combination of mandatory machine breaths and patient-initiated breaths. During physical therapy interventions, the physical therapist should note any significant changes in respiratory rate, tidal volume, or minute ventilation.
Carbon dioxide (CO2) is a by-product of cellular aerobic metabolism and diffuses from the tissues into venous capillary blood. The pulmonary capillary network surrounds the alveoli, and CO 2diffuses into the alveoli and is exhaled through the mouth and nose after passing through the tracheobronchial tree. The partial pressure of carbon dioxide (PaCO2) is determined by the balance between carbon dioxide production that occurs during cellular metabolism and the amount removed by the lungs during ventilation. Carbon dioxide is highly soluble and is not usually affected clinically by changes in ![]() . When a patient is at rest we can assume that CO 2tension is constant. Changes in PaCO2can be attributed to changes in
. When a patient is at rest we can assume that CO 2tension is constant. Changes in PaCO2can be attributed to changes in ![]() A. Increases in PaCO2 occur when changes in carbon dioxide production are not accompanied by a proportional change in minute ventilation. Exercise, fever, and an increase in dead space ventilation are conditions that may cause an increase in PaCO2.12 High levels of PaCO2 may not be as detrimental as originally thought.14 Therapists working in the intensive care unit may see mechanically ventilated patients with PaCO2 levels exceeding 80 mmHg without known deleterious side effects. An intentional mechanical ventilation strategy which incorporates an elevated PaCO2 is referred to as permissive hypercapnia.15
A. Increases in PaCO2 occur when changes in carbon dioxide production are not accompanied by a proportional change in minute ventilation. Exercise, fever, and an increase in dead space ventilation are conditions that may cause an increase in PaCO2.12 High levels of PaCO2 may not be as detrimental as originally thought.14 Therapists working in the intensive care unit may see mechanically ventilated patients with PaCO2 levels exceeding 80 mmHg without known deleterious side effects. An intentional mechanical ventilation strategy which incorporates an elevated PaCO2 is referred to as permissive hypercapnia.15
Work of Breathing
Work of breathing is defined as energy a patient must expend to move gases into and out of the lung. During normal ventilation, inspiration involves the contraction of the respiratory muscles, and expiration occurs passively, using the elastic recoil of the lung and chest wall. The normal work of breathing is around 3 mL/min or less than 2% of the total metabolic rate. Only 10% of the energy consumed during respiration is a result of contraction of the respiratory muscles to move gases against the resistance and compliance factors of the airways and lung tissues; 90% of the energy consumed during ventilation is wasted and used to generate heat. However, the respiratory work of breathing is also the result of both the airway pressures required to overcome airway resistance and the elastic forces of the lung and thorax. Therefore respiratory work increases during periods of high airway resistance and rapid respiratory rates, or with large lung volumes and low lung–thorax compliance. Asthmatic patients and patients with a tracheostomy or endotracheal tube may have increased airway resistance and breathe optimally with a slower respiratory rate at higher tidal volumes. Conversely, patients with low lung compliance, such as those with severe ARDS, pulmonary edema, pulmonary fibrosis, atelectasis, and pulmonary contusion, breathe more efficiently at faster respiratory rates using smaller tidal volumes.16,17 The ventilator can be adjusted to minimize the patient’s work of breathing. This will be explained later in this chapter in the section on Modes of Mechanical Ventilation. The optimal respiratory frequency is a balance between the resistive and the elastic forces of the lung. See Chapter 5 for more details regarding respiratory physiology.
RESPIRATORY FAILURE
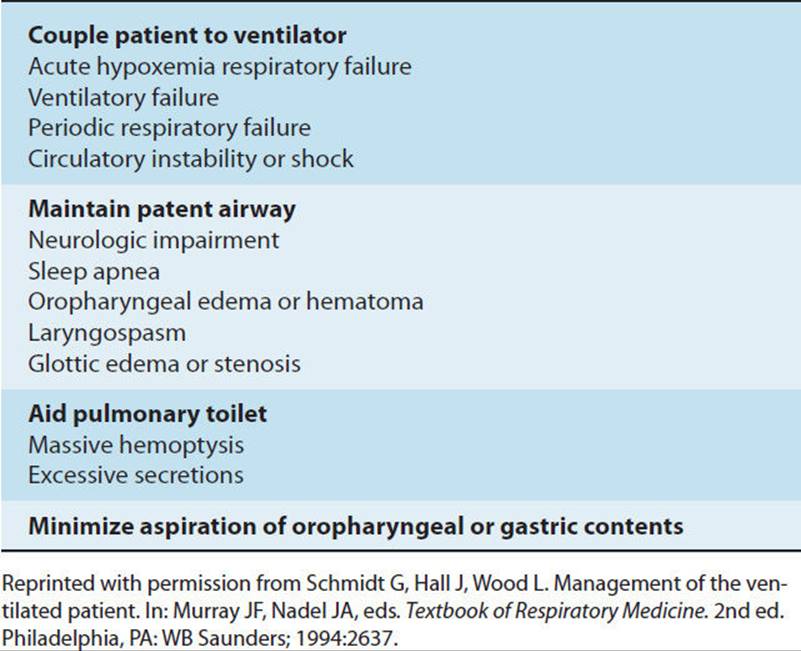
Respiratory failure occurs when the exchange of gases (oxygen and carbon dioxide) is inadequate to meet the patient’s metabolic needs. Acute respiratory failure is a life-threatening condition, diagnosed clinically, and the primary indication for mechanical ventilation. Breathing requires integration of the respiratory centers in the brainstem, the respiratory muscles, and connecting nerves. Injury or disease negatively impacting this integration, or the lung parenchyma, may lead to respiratory failure. Patients in respiratory failure usually require mechanical ventilation and therefore fall into physical therapy Practice Pattern 6F. A patient may present with hypoxemia, hypercarbia, acidosis, or a combination of these three conditions. This clinical presentation may result from the lung not being efficient as a gas-exchange membrane, from inadequate gas exchange, or from the patient being unable to support respiratory function because it requires too much work. Common clinical indications for intubation and mechanical ventilation that the physical therapist may encounter are listed in Table 19-2. The exact criteria used to intubate and mechanically ventilate a patient may vary depending on patient diagnosis, physician preference and training, and institutional guidelines.
TABLE 19-2 Indications for Endotracheal Intubation
MECHANICAL VENTILATION
The critical care physical therapist may be challenged by the variety of ventilators and ventilator settings they encounter while treating ICU patients. Ventilator preferences vary between facilities based on physician preference, patient population, funding, and staff knowledge. This section will describe frequently used types and modes of ventilators for adult patients receiving physical therapy interventions. Most are positive-pressure ventilators with a multitude of dials, digital readouts, lines, tubes, and alarms. Although there are many theories in practice about the pros and cons of each ventilator type and the modes of ventilation, it is vital to remember the most important factor related to the duration and success of mechanical ventilation: treatment of the patient’s underlying condition causing respiratory failure. This is far more important in influencing patient morbidity and mortality than the type of ventilator. Although ventilators from different manufacturers may have controls, dials, and digital readouts that appear very different, the principles and functions of most ventilators are similar.
While mechanical ventilation is a common medical intervention, 80% of patients are weaned without difficulty after only a short duration of mechanical ventilation.18 For the 20% of mechanically ventilated requiring more complex weaning, the newer more sophisticated ventilators may hold advantages for patients with resolving severe respiratory failure.
The first positive pressure ventilators were developed in the early part of the 20th century by anesthesiologists to deliver anesthetic agents via an endotracheal tube to patients having surgery, and to support breathing during thoracic surgery. In 1952, positive pressure ventilation was brought to the bedside, when medical students in Copenhagen were scheduled by Dr. Isben, an anesthetist, to manually inflate the lungs of patients with poliomyelitis using manual resuscitator bags, 24 hours per day.19 Manual resuscitator bags, often referred to as ambu bags, delivered a concentration of oxygen greater than 21% (oxygenation) and a larger than resting tidal volume (ventilation); the respiratory rate (work of breathing) was controlled by the medical student. The same goals, for example, improving oxygenation, ventilation, and the work of breathing, are the basis for mechanical ventilation today. In addition, recent technological advances allow some ventilators to automatically adjust parameters to the patient’s physiological response to the disease process and the demands associated with medical and physical therapy interventions. Figure 19-2 shows the common features of a mechanical ventilator.

FIGURE 19-2 Mechanical ventilator. (A) Ventilator and humidifier at the patient’s bedside. (B) Patient’s actual ventilation: note airway pressure (mean airway pressure 11), Peep, inspiratory—expiratory time, respiratory rate (ftot), and tidal volume. (C) Ventilator settings: note tidal volume, 600 mL, Fio240%, Peep 5. The patient is spontaneously breathing; there is no preset respiratory rate.
Oxygenation
Oxygenation is primarily controlled by the concentration (percentage) of oxygen in the inspired gas and the positive end-expiratory pressure (PEEP). The concentration of oxygen is referred to as the FiO2, the percentage or fraction of inspired oxygen. Additional oxygen is usually delivered by a ventilator after other methods of supplemental oxygen delivery have failed to achieve adequate oxygenation. In addition to increasing the FiO2, PEEP may be added. PEEP retards small airway and alveoli closure, thus preventing derecruitment of alveoli. Derecruited alveoli do not participate in gas exchange.20 Therefore, PEEP prevents early airway and alveolar collapse at the end of expiration by increasing functional residual capacity, increasing end-expiratory lung volumes, and improving the matching of ventilation to perfusion by decreasing intrapulmonary shunt (Table 19-3).21
TABLE 19-3 Interventions to Increase PaO2
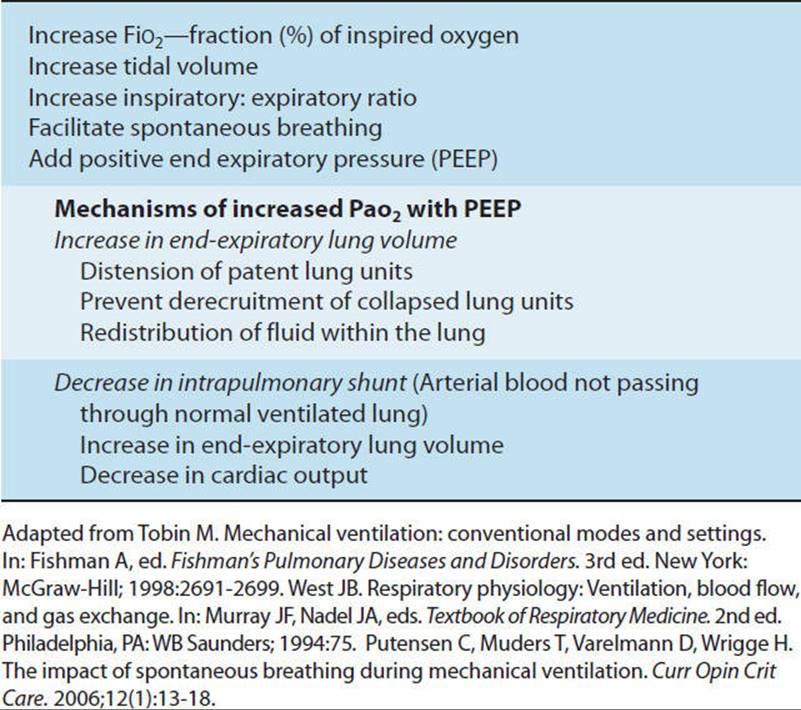
Oxygenation may also be enhanced by increasing tidal volume or increasing inspiratory versus expiratory (inverse I:E ratio) ventilation, providing increased time for gas exchange to occur in ventilated alveoli. Some advocate transient use of high levels of PEEP in conjunction with recruitment maneuvers to facilitate recruitment or reopening of collapsed alveoli. More recently, facilitation of spontaneous breathing has also been shown to improve oxygenation22 (see Table 19-3).
Functional residual capacity (FRC) may be negatively impacted in the sick lung and in patients lying in the supine or near supine position. A means of increasing FRC is to add PEEP or continuous positive airway pressure (CPAP). Some believe physiological PEEP, defined as positive pressure within the alveoli in the presence of a closed glottis, is lost with the presence of a tracheal tube. This is because the tracheal tube either passes through the glottis, which remains open, or the tube is placed below the glottis and vocal cords. However, evidence is lacking as to whether physiological PEEP actually exists.23
To restore FRC, 5 cmH2O of PEEP is usually applied during mechanical ventilation. A fixed resistance is applied to the expiratory limb of the ventilator circuit to maintain a positive pressure at the end of expiration. PEEP is increased by the physician as clinically indicated; levels as high as 15 to 20 cmH2O may be necessary with ARDS, severe pulmonary edema, and severe bilateral pneumonia when distal airways may be edematous and prone to collapse. PEEP is increased cautiously as it may decrease cardiac output and adversely affect blood pressure. Patients who require frequent adjustments of FiO2 and PEEP usually have continuous blood pressure monitoring via an arterial line and may have pulmonary artery catheters to measure pulmonary artery pressures and cardiac output as the level of PEEP is titrated. PEEP may be contraindicated for patients with untreated pneumothorax, or bronchopleural fistulas.24
As a general rule those patients requiring PEEP greater than 10 cmH2O pressure should not be routinely disconnected from the ventilator for turning, suctioning, and transfer activities. Disconnection, either intentional or inadvertent, may result in alveolar derecruitment with ensuing complications. High levels of PEEP alone usually do not preclude a patient from tolerating secretion clearance techniques and mobilization, including ambulation with a portable ventilator. It is important to maximally mobilize the patient and perform secretion clearance techniques, as immobility and secretion retention may be contributing to the hypoxemia for which high levels of PEEP are required. Functional mobility and range-of-motion exercises are performed to patient tolerance, while the physical therapist monitors the patient’s vital signs. A PEEP adapter or in-line suction catheter is strongly recommended for patients who require frequent suctioning and have a PEEP of 10 cmH2O or greater (see CD-ROM).
Ventilation
There are several controls or dials on the ventilator that regulate ventilation. These include controls for respiratory rate, tidal volume, inspiratory flow rate, and inspiratory/expiratory (I:E) ratio. Tidal volume and respiratory rate regulate PaCO2.. In volume targeted modes of ventilation the inspiratory flow rate is the speed with which inspired gas is delivered to the lungs and may be adjusted to meet the patient’s demand. Patients who require high respiratory rates require higher flow rates. Occasionally, during physical therapy interventions the patient may seem to be “bucking” or dyssynchronous with the ventilator due to higher minute ventilation requirements. This may be due to a flow rate that is not adequate and the patient subsequently breathes against a fixed resistance. The result is an increased work of breathing for the patient and increased inspiratory effort. In pressure targeted and dual targeting modes of ventilation, the inspiratory flow is variable and may be advantageous to meet varying patient demands. Critically ill patients may also have an increase in dead space ventilation as high as 70% of minute ventilation, secondary to increased ![]() mismatch or shunt related to their disease process.25 Thus, mechanically ventilated patients routinely require a higher than normal minute ventilation to meet their metabolic demand.
mismatch or shunt related to their disease process.25 Thus, mechanically ventilated patients routinely require a higher than normal minute ventilation to meet their metabolic demand.
Work of Breathing
Work of breathing will be influenced by the mode of ventilation used. A mode of ventilation is defined by the interaction between machine and patient. How the breath is delivered and how the patient participates or interacts with that delivery defines the mode.26 It is essential that the physical therapist understand the concepts of the different modes of mechanical ventilation if they are to independently deliver care. For example, a patient may have a resting respiratory rate of 21 breaths/min, with 8 mandatory breaths and 13 spontaneous breaths. During physical therapy treatment, without any ventilator adjustments, the respiratory rate may temporarily increase to 35 breaths/min. The therapist will note that the spontaneous respiratory rate is now 27 breaths/min and there is an increased work of breathing. The patient may need to rest before additional physical therapy interventions. Temporary changes in respiratory rate are normal and should not interfere with physical therapy interventions if they quickly return to baseline. However, the number of breaths per minute delivered by the ventilator can be increased (with a physician order) to allow a patient to tolerate more physical therapy and nursing interventions such as side-to-side turning, therapeutic exercises, and bed-to-chair transfers.
Physical therapists usually do not adjust ventilator settings. After consultation with the critical care personnel: physician, respiratory therapist and/or nurse, orders may be written that permit adjustments by appropriate staff to support physical therapy interventions.
CLINICAL CORRELATE
Mechanical ventilation should be a dynamic process, similar to changes in spontaneous breathing during changes in activity levels. It therefore may be necessary to change the mode of ventilation or increase the flow rate, respiratory rate, or FiO2 during all or part of a patient’s physical therapy treatment. Collaboration between the physical therapist and the ICU clinicians trained in the complexities of mechanical ventilation is recommended to determine the best ventilator settings during physical therapy interventions.
VENTILATOR ALARMS
Prior to treating ventilated patients, the physical therapist must have a basic understanding of the alarms generic to all ventilators. The therapist should be able to discriminate between those requiring emergent nursing or medical interventions and alarms which may be the result of normal changes in respiratory parameters/mechanics in response to the treatment intervention. The parameters for each alarm are selected by the bedside clinician and are typically set for the patient in a resting state. Therefore normal physiological responses to position changes, activity, coughing, suctioning, and therapeutic exercise may activate an alarm.
Alarms may be divided into three categories: those alarms resulting from oxygenation/system failure, pressure changes, and volume changes.
Oxygen/System Failure Alarms
Though infrequent, the alarm associated with one of the most serious consequences notifies the clinician of system failure or a low or nonexistent oxygen supply. This alarm usually has a piercing sound that calls immediate attention to the situation. Most hospitals have a backup generator system that will immediately take over in cases of electrical failure. When a ventilator becomes nonoperational, the therapist should immediately turn the wall oxygen flow rate up as high as possible (usually around 15 L/min) and begin ventilating the patient with a manual resuscitator bag, while calling for assistance. Whenever the therapist is treating a ventilated patient outside the ICU setting, he or she should check that an oxygen supply is readily available from either a portable tank or a wall oxygen supply. Manual resuscitator bags, oxygen tubing, an oxygen supply, and tracheal suction equipment and supplies should be in the work area and checked daily according to hospital/departmental standards.
Pressure Alarms
Pressure alarms notify the clinician that the ventilator is operating outside preset pressure ranges, either high or low. These alarms require that the clinician determine the cause of the alarm and whether any additional interventions are necessary.
High-pressure alarms indicate that higher than expected pressures are necessary to deliver the desired tidal volume. High-pressure alarms may be triggered by either an increase in airway resistance or a decrease in lung compliance. Increase in airway resistance occurs because of an obstruction in the tracheal tube, coughing, or agitation causing the patient to breathe in a way that is not in synchrony with the ventilator settings. The tracheal tube may be obstructed by a mucus plug, blood clot, or by a patient biting the endotracheal tube. When a mucus plug or blood clot is suspected, the therapist should immediately suction the patient. While suctioning, the therapist should note whether the catheter could reach the carina. Noting the distance from the opening of the tracheal tube to the carina will help the therapist determine whether there is still an obstruction in the airway after suctioning. The therapist should also auscultate the chest and note any changes in breath sounds since the beginning of the physical therapy intervention. If it is suspected that the patient is biting the endotracheal tube, or that the tube is kinked, the nurse should be notified and a bite block may be placed in the patient’s mouth or sedation administered. If the alarm persists after suctioning, placing a bite block, and sedation, medical attention may be necessary. The physician evaluates the situation and may need to change the tracheal tube.
High-pressure alarms may also be activated when a patient’s lungs are becoming less compliant due to a worsening medical condition such as ARDS or pneumothorax. In this situation, the ventilator must generate higher pressures in order to maintain the same tidal volume. The physician or respiratory therapist may change the mode of ventilation, targeted tidal volume, or increase the upper limit of the pressure alarm setting in order to resolve the situation. Changes in patient position may also decrease thoracic compliance, care should be taken with upper extremity positioning to facilitate versus inhibit chest wall expansion, particularly in the side-lying position.
Low-pressure alarms are the result of some type of leak in the respiratory circuit. This occurs when the endotracheal tube is disconnected, there is a break in the integrity of the tubing circuit, or there is an air leak around the tracheal tube. The nurse, patient, or therapist may inadvertently disconnect the ventilator tubing from the tracheal tube, or open an access port within the ventilator circuit. If consistent with institutional policies, therapists may briefly disconnect the tracheal tube from the ventilator tube for suctioning, when mobilizing a patient, or to remove excess water from condensation in the tubing. As disconnection will eliminate PEEP, the frequency and duration of disconnection will depend upon the level of PEEP the patient requires. Once the tube is reconnected, the alarm will no longer sound. While working with a patient who disconnects him or herself from the ventilator, the therapist should not hesitate to reattach the ventilator tubing to the tracheal tube and report this behavior to the bedside nurse. Such circumstances may make it appropriate to apply protective devices that restrain movement, for which a physician order is usually required. If a patient pulls out the tracheal tube (self-extubates), the therapist should immediately begin bagging the patient with a manual resuscitator bag, supplemental oxygen and a face mask while summoning help.
Volume Alarms
Volume alarms signal that the ventilator is operating outside the expected volume ranges set by the operator. These alarms may be set to monitor minute ventilation and/or tidal volume. It is expected that the inhaled and exhaled lung volumes of a mechanically ventilated patient are close. When the cuff on the tracheostomy or endotracheal tube ruptures, leaks, or is deflated, there will be a marked difference in inhaled and exhaled volumes causing the low-volume alarm to sound. The therapist should notify the nurse, physician, or respiratory therapist when this is suspected.
High-volume alarms alert the clinician that the patient is getting a higher than preset minute ventilation. This may occur with an increase in respiratory rate in response to agitation or to a change in the patient’s mental status. The high-volume alarm frequently sounds while turning a patient or moving a patient to sit on the edge of the bed or transfer. Once the activity is completed, the patient usually returns to the baseline ventilatory pattern, and the alarm stops. This is a normal response to physical therapy and does not require any special intervention. However, if the patient becomes agitated, and does not respond to activity or the therapist trying to calm the patient, the nurse should be notified. There may be a better time of day to see the patient, or the nurse may need to administer medication to enhance participation. It is recommended that the therapist anticipate the need for additional medication and speak with the nurse prior to physical therapy interventions.
It is notable that an increase in respiratory rate is a normal response to activity. Therefore, a marked increase in respiratory rate with an exercise program may necessitate a change in the ventilator settings, not medication.
CLINICAL CORRELATE
The decision to use sedation or antianxiety medication to facilitate therapy interventions must always take into account the detrimental effects the medication may have on the patient’s ability to interact and actively participate in the therapeutic intervention as well as potential detrimental long-term effects of the medication.
Low-exhaled-volume alarms are usually the result of the patient becoming disconnected from the ventilator or from a leak in the cuff of the tracheal tube or ventilator circuit, as previously discussed. However, a low-volume alarm may also sound when a spontaneously breathing patient receiving ventilatory support is given a narcotic, sedative, or paralytic medication, decreasing their respiratory drive or ability. Patients who require ventilatory support may benefit from a short-acting drug for some physical therapy interventions. For example, patients with abnormal muscle tone may require pharmacological intervention to optimize joint positioning prior to serial casting. Close monitoring is required. Ventilatory support may need to be increased for the procedure; yet, once the patient’s spontaneous respirations return, the ventilator is adjusted back to the original baseline settings.
MODES OF MECHANICAL VENTILATION
A mode of ventilation is a means to deliver a breath to a patient and is defined by the interaction between patient and machine. There are many different modes with specific characteristics. At times, these differences may appear subtle, but are sufficient to warrant distinction. Modes may be considered on a continuum, from the ventilator assuming all or the vast majority of the patient’s breathing, to the patient primarily breathing on his own.
There are numerous modes of ventilation utilized in clinical practice that are designed to improve oxygenation and subsequently support oxygen consumption. Oxygen consumption (![]() O2)) of the respiratory muscles depends on the patient’s clinical condition and ranges from 2% to 3% of total body
O2)) of the respiratory muscles depends on the patient’s clinical condition and ranges from 2% to 3% of total body ![]() O2 in healthy spontaneously breathing subjects, 5% to 10% in patients breathing on a ventilator during assisted modes, and up to 50% in patients with severe respiratory failure on mechanical ventilation.27,28 Patients with high oxygen consumption may require the more complex and sophisticated modes of ventilation, whereas patients with lower oxygen demands may only require continuous positive airway pressure (CPAP). Although medical centers and physicians have distinct preferences for the mode of ventilation used, research is limited regarding mortality, length of stay, and functional outcome. Most studies examine physiological variables during the time of mechanical ventilation. The most cost-effective modes that provide the best outcomes have yet to be substantiated. Recent studies advocate low tidal volumes and higher levels of PEEP for the management of acute respiratory failure.29,30 High tidal volumes and progressive ventilator-free breathing are advocated for weaning the tetraplegic patient when conventional weaning techniques have failed.31Figure 19-3 demonstrates pressure waveforms with spontaneous breathing and various forms of mechanical ventilation.
O2 in healthy spontaneously breathing subjects, 5% to 10% in patients breathing on a ventilator during assisted modes, and up to 50% in patients with severe respiratory failure on mechanical ventilation.27,28 Patients with high oxygen consumption may require the more complex and sophisticated modes of ventilation, whereas patients with lower oxygen demands may only require continuous positive airway pressure (CPAP). Although medical centers and physicians have distinct preferences for the mode of ventilation used, research is limited regarding mortality, length of stay, and functional outcome. Most studies examine physiological variables during the time of mechanical ventilation. The most cost-effective modes that provide the best outcomes have yet to be substantiated. Recent studies advocate low tidal volumes and higher levels of PEEP for the management of acute respiratory failure.29,30 High tidal volumes and progressive ventilator-free breathing are advocated for weaning the tetraplegic patient when conventional weaning techniques have failed.31Figure 19-3 demonstrates pressure waveforms with spontaneous breathing and various forms of mechanical ventilation.
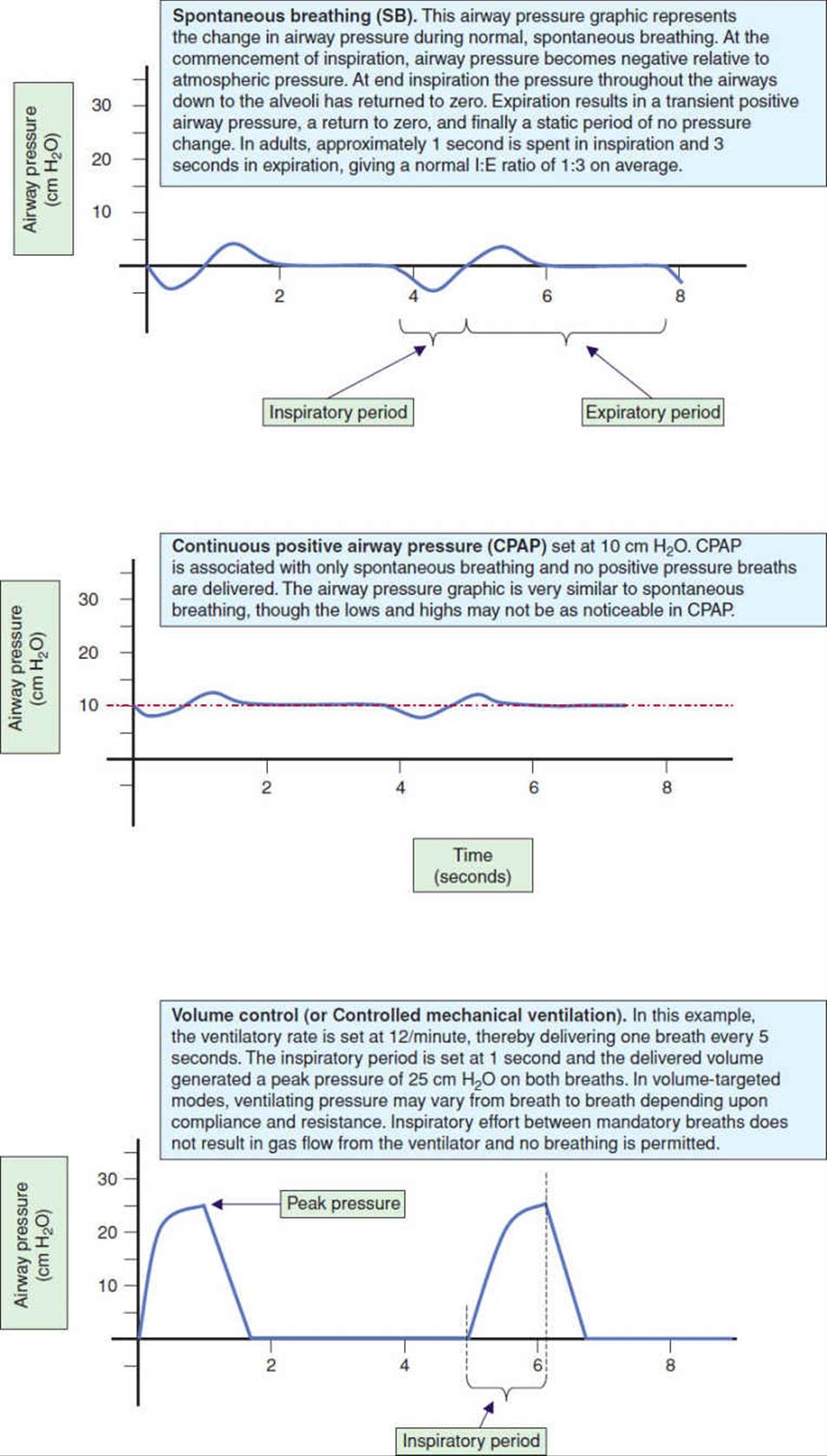
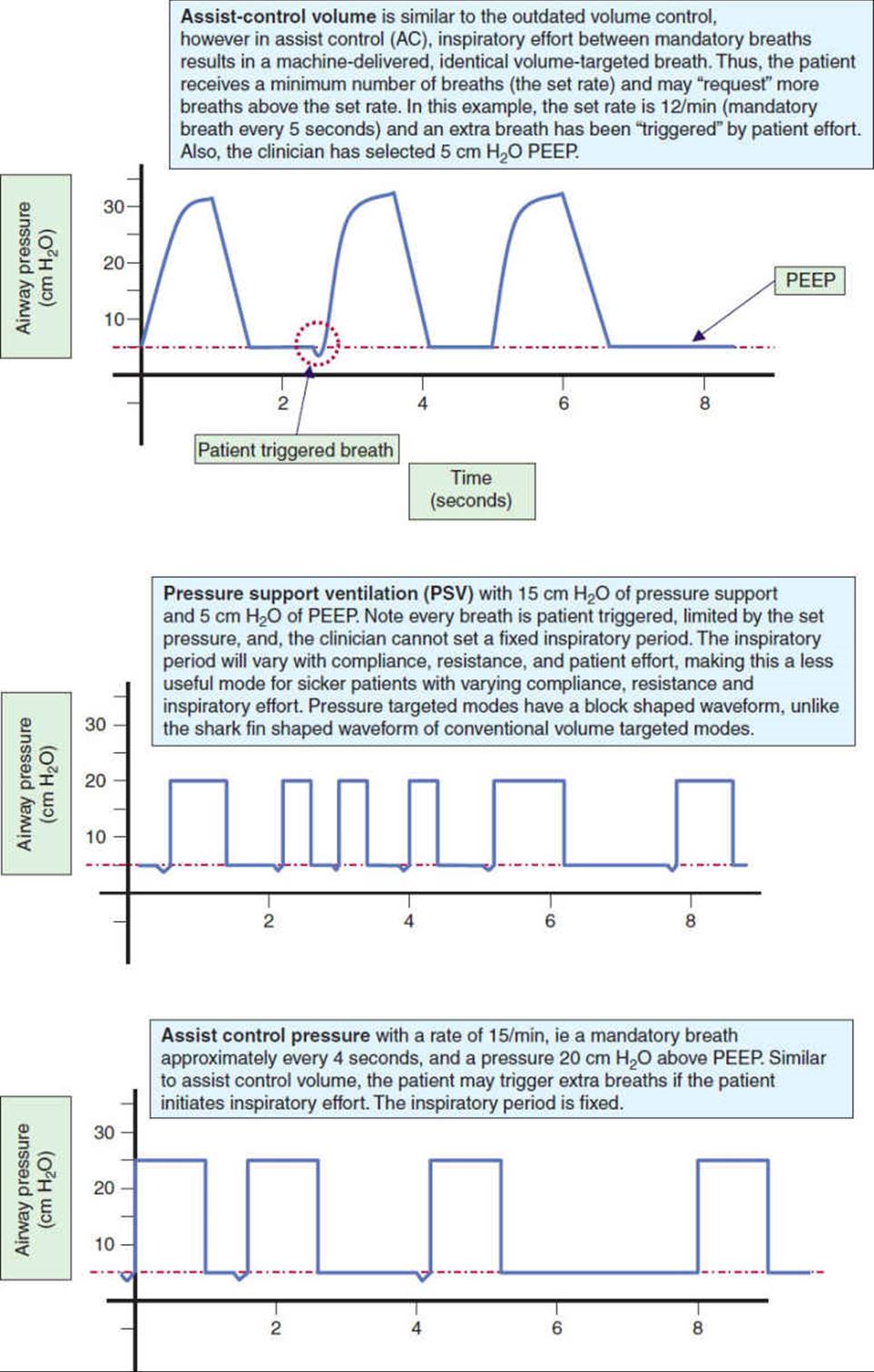
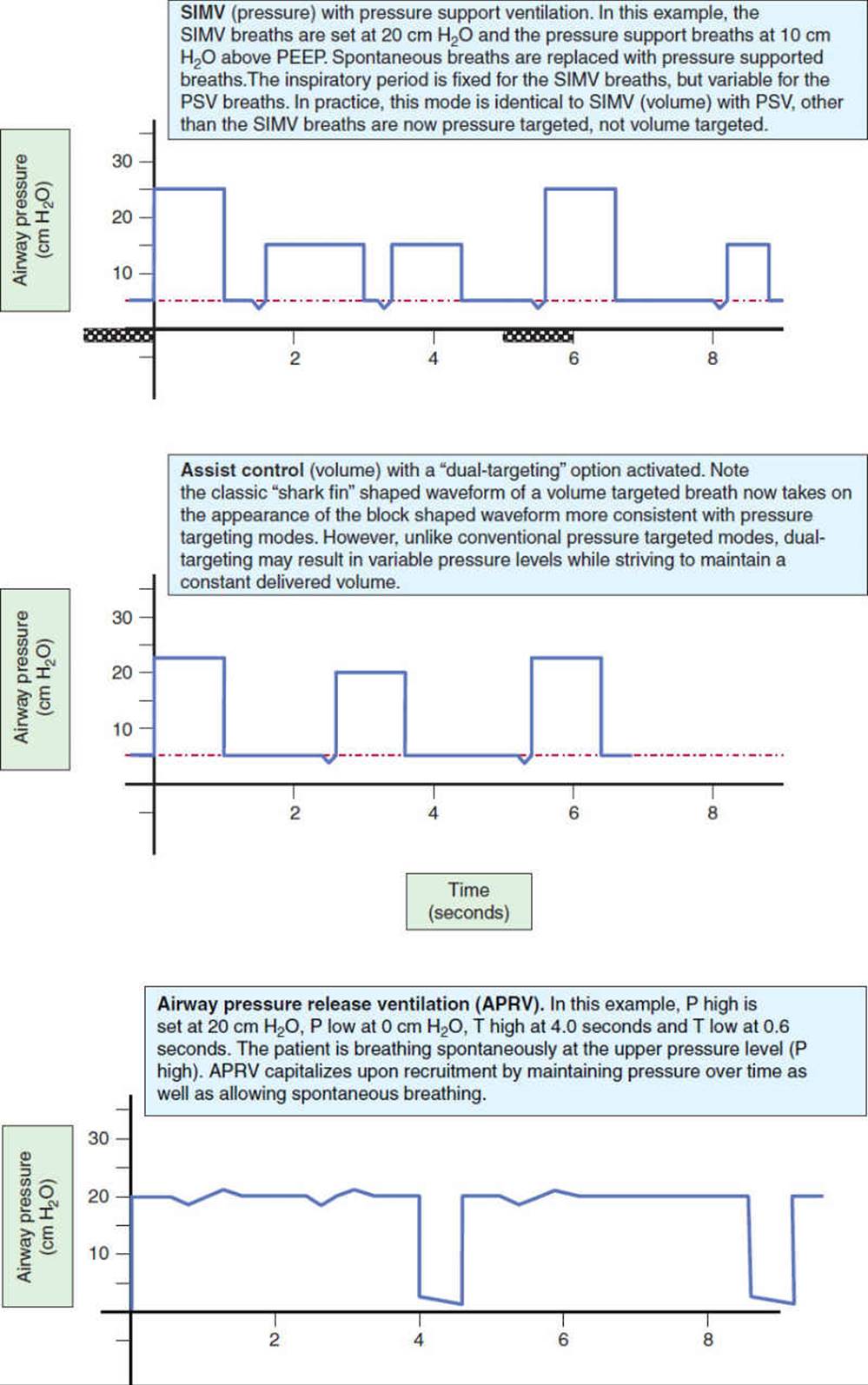
FIGURE 19-3 Pressure waveforms with spontaneous breathing and various forms of mechanical ventilation. (This article was published in Trauma nursing: from resuscitation through rehabilitation, 4th edition, McQuillan KA, Makic MB, Whalen E, eds. Thoracic Trauma, pp 614-677, Copyright Saunders Elsevier (2009).) (continued)
Ventilatory Support
Control Modes
Critically ill patients who are unable to maintain adequate oxygenation and/or carbon dioxide removal without support require a mode of ventilation that relieves the patient of the majority of the work of breathing while ensuring ventilation and oxygenation. Depending upon the mode selected and level of sedation utilized, the patient may initiate some breaths, and/or breathe spontaneously. Thus, even for the critically ill, there is a continuum of ventilatory support from maximal to minimal support.26 Historically, the disadvantages to maximum ventilatory support included the need for frequent sedation and possibly neuromuscular blockade. In turn, this led to decreased spontaneous respiratory efforts, muscle atrophy, increased atelectasis, and inspissated secretions. These disadvantages were reluctantly accepted as necessary to sustain life. However, technological advances in mechanical ventilation now offer options that allow more limited use of sedation, and particularly paralytics.
For many years, modes of ventilation were broadly categorized into either volume targeted or pressure targeted modes. The volume targeted modes required the clinician to select a tidal volume for the ventilator to deliver (or target) with each mandatory breath, while pressure targeted modes required selection of a fixed ventilating pressure for each mandatory breath. The incorporation of computer technology into the ventilator has allowed increasing sophistication in the delivery of breaths. Computer science, along with evolving clinical knowledge, has paved the way for a third classification of modes known as dual-targeting. In the 1990s, dual-targeting modes permitted the clinician to deliver a breath that combined features from both volume and pressure targeting ventilation.26
Volume-controlled ventilation—Volume-controlled ventilation is probably the simplest and earliest method of positive pressure ventilation. Developed in the 1950s, adults were the primary patient group. In this mode the patient is not allowed, nor required, to initiate a breath, and the work of breathing is primarily provided by the ventilator, as long as the patient’s respiratory cycle is synchronized with the mechanically delivered breaths. All breaths are initiated by the ventilator at the rate that has been set by the clinician. The tidal volume is preset for each breath, and the minute ventilation becomes the product of the set rate and the tidal volume. Respiratory muscle efforts and their contribution to oxygen consumption may be eliminated if the patient is chemically paralyzed. Some also believe that subsequent relaxation of the chest wall muscles may enhance recruitment of lung tissue.32 Eliminating patient effort may also relieve patient dyssynchrony, although anxiety can be high. Lung thorax compliance, airway resistance, and auto-PEEP are easily calculated for pressure and flow measurements. The generation of high tidal volumes from volume-controlled ventilation may increase the risk of volutrauma, a major facet of ventilator induced lung injury (VILI)33 (see page 612). It is recommended that tidal volumes and airway pressures be closely monitored to minimize the risk of alveolar over distension and VILI.
Pressure-controlled ventilation—Pressure-controlled ventilation applies a pressure that is preset by the clinician, as is the ventilatory rate. The clinician also sets a fixed inspiratory time. In pure control modes, the patient cannot trigger or initiate a breath. Inspiratory effort may appear as dyssynchrony between the patient and the machine, as the patient attempts to draw gas into their lungs, but the machine does not respond. Controlled breaths are delivered at a predictable interval, ie, a rate of 12/min results in a breath every 5 seconds. The inspiratory flow from the ventilator is high initially, the flow decelerates as the alveolar pressure rises with lung inflation. The delivered tidal volume varies depending on the inspiratory time, patient effort, as well as the patient’s lung compliance and airway resistance. Minute ventilation is not predetermined.34
Patients receiving ventilatory support frequently require postural drainage with or without manual techniques and suctioning for secretion retention. The physical therapist should be careful not to dislodge or pull on the tracheal tube when turning the patient. With careful positioning, manual techniques and most postural drainage positions, including the prone position, are possible (Fig. 19-4). Obstacles to the prone position include severe kyphosis or a pelvic external fixator. It is possible to place a patient prone while wearing a brace to stabilize spinal fractures, if the patient requires manual techniques, braces such as thoracolumbosacral orthoses (TLSOs) can often be opened once the patient is securely positioned. For patients with cervical bracing such as Halo vests or Yale braces, full prone positioning may be problematic. In these instances, the therapist should try to position the patient as close to one-fourth turn to prone from side-lying as possible. If a patient does not have adequate cervical rotation to lie prone, a towel roll can be placed both under the forehead and under the upper thorax. For patients with a tracheostomy a blanket roll, a sheet roll or wedge is carefully placed under the upper thorax to allow room for the tracheostomy tube and airway suctioning. A roll under the pelvis may also be helpful to allow for a shift in abdominal contents, particularly for patients with a large abdominal girth.
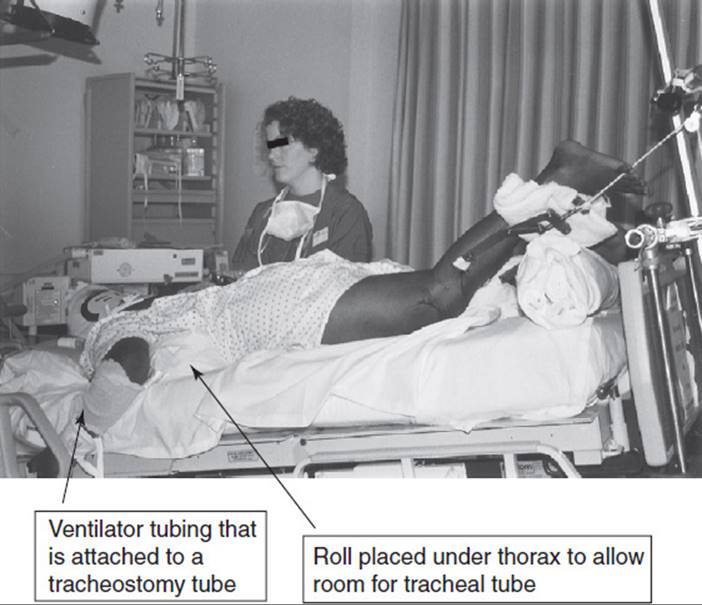
FIGURE 19-4 Prone positioning.
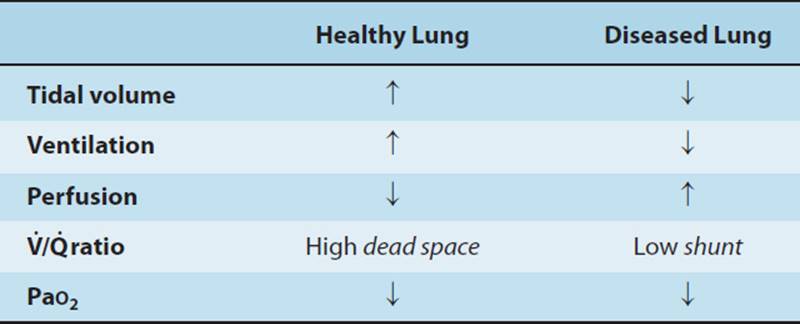
In summary, the disadvantages of control modes of mechanical ventilation include the need for heavy sedation and possibly neuromuscular blockade, decreased spontaneous respiratory efforts, respiratory alkalosis, and progressive atelectasis. Progressive infiltrates and atelectasis develop in dependent lung zones as a result of the gravitational redistribution of fluid, impaired secretion clearance, and poor inflation. The positive pressure breaths of mechanical ventilation result in ventilation along the path of least resistance, hence upper/anterior lung regions (when the patient is supine) are more readily ventilated than the posterior lung, while perfusion is primarily gravity dependent and greater in dependent/posterior regions, creating a ![]() mismatch.11 The decrease in spontaneous respiratory effort associated with control modes of ventilation is unfortunate, as it has potential to mitigate negative effects of positive pressure ventilation. Spontaneous breathing has been shown to decrease the development of atelectasis and reduce
mismatch.11 The decrease in spontaneous respiratory effort associated with control modes of ventilation is unfortunate, as it has potential to mitigate negative effects of positive pressure ventilation. Spontaneous breathing has been shown to decrease the development of atelectasis and reduce ![]() mismatch, as it improves ventilation of posterior/dependent lung regions based on diaphragmatic mechanics.35–38 Mechanically ventilated patients in the ICU frequently experience atelectasis and consolidation in the dorsal dependent lung regions. Therefore strategies which enhance ventilation and recruitment of these lung regions, such as facilitation of spontaneous breathing, are valuable. Recent studies, primarily in the animal model, have also shown significant decrement in diaphragmatic muscle force39,40 and diaphragm atrophy41 as a result of mechanical ventilation.
mismatch, as it improves ventilation of posterior/dependent lung regions based on diaphragmatic mechanics.35–38 Mechanically ventilated patients in the ICU frequently experience atelectasis and consolidation in the dorsal dependent lung regions. Therefore strategies which enhance ventilation and recruitment of these lung regions, such as facilitation of spontaneous breathing, are valuable. Recent studies, primarily in the animal model, have also shown significant decrement in diaphragmatic muscle force39,40 and diaphragm atrophy41 as a result of mechanical ventilation.
Controlled mechanical ventilation results in a greater decrease in diaphragmatic force than assist control ventilation.42 If paralytic agents are required to maintain compliance with a controlled mode of ventilation, inadvertent disconnection from the ventilator can be life-threatening. Despite these disadvantages, controlled mechanical ventilation with low tidal volumes and PEEP remains an option for ventilation of patients with severe respiratory failure and elevated intracranial pressure. Arguably, the simplicity, and immediate responsiveness, makes it an attractive choice for clinicians with less expertise in other modes.
Assist-Control Modes
Assist-control (AC) modes may be either volume or pressure targeted, and while similar to their precursors, the control modes, the AC modes are far more commonly used.26 The difference between control and assist-control is the ability of the patient to trigger or request breaths above the set ventilatory rate. All breaths continue to be of the same size and type as the mandatory breaths. The rate set by the clinician becomes the minimum number of breaths a patient will receive, however if the patient initiates additional breaths, the ventilator will reward the patient with a machine breath.
The goals of assist control modes are to allow and improve synchrony between the patient and the ventilator, reduce patient effort, and optimize comfort. However, how a particular mode of ventilation is used may be equally as important as the chosen mode of ventilation. Physical therapists should be familiar with the terms and general principles of AC, synchronized intermittent mandatory ventilation (SIMV), pressure support ventilation (PSV), pressure regulated volume control ventilation (PRVC), airway pressure release ventilation (APRV), and proportional assist ventilation (PAV) when working with mechanically ventilated patients.
Synchronized intermittent mandatory ventilation—Synchronized intermittent mandatory ventilation (SIMV) appears the same as AC ventilation when the patient is receiving only ventilator-assisted breaths (not taking any spontaneous breaths). The patient receives periodic positive-pressure breaths from the ventilator at a preset volume or pressure and rate. With SIMV, the patient can inhale with unassisted spontaneous breaths between mechanically assisted breaths. When a patient is able to breathe spontaneously, spontaneous efforts will be synchronized with the timing of the mandatory ventilator breaths. If spontaneous breaths are taken within the preset triggering period that a mechanical breath is scheduled to be delivered (usually about a 1-second zone), the ventilator will deliver the mandatory breath while synchronizing with the patient’s inspiratory effort.19 If the patient makes no effort during the triggering period, the ventilator waits until the end of the triggering period and delivers the targeted volume or pressure. The precursor to SIMV was intermittent mandatory ventilation (IMV), which was developed to facilitate weaning.43,44 SIMV evolved with the goal to avoid problems with dyssynchrony when weaning patients from the ventilator and to gradually decrease the number of mechanically assisted breaths to decrease the duration of mechanical ventilation. However, the use of SIMV to decrease weaning time has not been substantiated in clinical studies (see page 603). SIMV may actually contribute to respiratory muscle fatigue when the patient has a high respiratory rate and increased work of breathing.45,46 SIMV was historically a volume targeted mode until the 1990s with the mainstream introduction of SIMV as a pressure targeted mode as well. In clinical practice SIMV, whether volume or pressure targeted, is routinely used with pressure-support ventilation.
Pressure regulated volume control—Pressure regulated volume control (PRVC) is a combination of volume control and pressure regulation. In this mode, the ventilator initially delivers a volume-controlled breath, while measuring the plateau pressure. The next breath is delivered using the measured pressure of the previous breath. If subsequent breaths increase above the preset volume, the pressure level is incrementally decreased until the preset tidal volume is delivered. If measured tidal volumes fall below the preset volumes, pressure is increased incrementally to reach the preset volume, up to a preset maximum upper pressure limit. In this mode, the ventilator is set to deliver a guaranteed respiratory rate, however breaths may be either ventilator or patient initiated. An alarm sounds if the ventilator is unable to deliver the preset volume within the preset pressure limit. It has been theorized that PRVC may decrease work of breathing while being used in a lung protective strategy, however this has not yet been established.47
Pressure-support ventilation—Pressure-support ventilation (PSV) is a pressure targeted mode requiring the patient to trigger every breath. The clinician does not set a machine rate, so in the absence of patient effort, the ventilator will not deliver a breath. This, probably more than any other mode, gives the patient more freedom with breathing. With PSV the main setting is the pressure target. When the ventilator senses an inspiratory effort (dependent on the trigger sensitivity that has been set) it responds by delivering a decelerating gas flow which raises the pressure in the airways to the targeted pressure level and holds the pressure constant. Decelerating gas flow is a consistent feature of pressure-targeted modes, as compared to a fixed or constant gas flow, which is a consistent feature of volume targeted modes.26 The pressure is maintained throughout inspiration. The inspiratory period terminates when the gas flow rate decreases to a preset value (typically 5%–25% of the peak inspiratory flow rate). Alternate measures to terminate the inspiratory period are available but are either uncommon or included as a safety feature.48 The patient indirectly controls rate, tidal volume, minute ventilation, and I/E ratio. The two most notable features of PSV are that the patient must trigger every breath, and the clinician does not set a fixed inspiratory period (one of the only modes having this distinction).
In the stable lung, as pressure support increases, respiratory rate decreases and tidal volume increases. In most cases, minute ventilation is not significantly modified. Alveolar ventilation is increased and PaCO2 decreases. Conversely, when pressure support is decreased the tidal volume decreases, PSV assists respiratory muscle activity by improving the efficacy of spontaneously initiated breaths, reducing the demand on the inspiratory muscles, and increasing tidal volume, therefore reducing the workload on the respiratory muscles. PSV has been shown to reduce the work of breathing and oxygen consumption of the inspiratory muscles.28,49 Lower levels of PSV counteract the element of work of breathing incurred by the ventilator circuitry and, particularly, the endotracheal tube. However, resistance changes throughout inspiration, being greatest at the beginning of the breath and least at the end of the breath, but the pressure level remains fixed. Therefore, the support to overcome resistance initially under compensates and later overcompensates. Nonetheless, PSV has become a highly useful adjunct particularly in the stable ventilated patient and in the patient being weaned from ventilatory support.26
CLINICAL CORRELATE
Physical therapists should be aware that some physicians will use a pressure support of 5 cm of water pressure and a resting respiratory rate of <35 breaths/min as criteria for extubation.50–52
Airway-pressure-release ventilation—Airway-pressure-release ventilation (APRV) (also referred to as BiVent, BIPAP, DuoPAPBiLevel or Biphasic ventilation, depending upon the manufacturer) is simply a modified form of CPAP (continuous positive airway pressure) which uses two different levels of pressure.53 As the name CPAP suggests, CPAP utilizes a continuous positive airway pressure while the patient breathes spontaneously. APRV is CPAP with a periodic release in the airway pressure to a lower level. Typically, the release is very short (less than 1 second) and the release level is to zero cmH2O. The higher CPAP level of APRV allows the patient to breathe spontaneously, facilitating recruitment, and improving oxygenation. The release phase aids in the removal of CO2. Conceptually, lowering the airway pressure to zero may seem like a bad idea. However, as the release phase is quite short, not all the gas empties from the lungs before the higher airway pressure is reinstituted, thus alveoli tend not to derecruit54–56 (Fig. 19-3). APRV works well with ARDS where lung compliance is low and the respiratory muscles are intact. Proposed advantages of APRV include reducing the risk of VILI (see risks of mechanical ventilation) by limiting peak airway pressures, and a reduction in repetitive recruitment/derecruitment of alveoli, which results in atelectrauma. Studies have also shown a decreased need for patient sedation and neuromuscular blockade,22,57 as well as benefits associated with spontaneous breathing.58 It should be noted that with critically ill patients who are unable to initiate spontaneous breathing, APRV can be used essentially as a “Full Support” mode of ventilation until the patient is able to initiate spontaneous breaths. APRV’s standout feature is allowing patients to spontaneously breathe; therefore practices inhibiting breathing, such as heavy sedation and or paralytic use, limit the usefulness of this mode.
Proportional-assist ventilation—Proportional-assist ventilation (PAV) may have promise and replace other ventilatory modes though it has yet to gain widespread acceptance. PAV offers maximal patient autonomy; every breath is initiated and terminated by the patient. The ventilator essentially acts as an accessory muscle imposing no volume or pressure targets; the patient has total control over all aspects of breathing. The operator selects which portion of the work will be performed by the machine. Pressure assistance by the machine is proportional to a variable combination of the inspired volume (elastic assist) and the inspiratory flow rate (the resistive assist). Tidal volume and flow are totally controlled by the patient. When the patient pulls harder, the machine boosts its output, and as the patient relaxes, the machine cuts back.49,59–61 This is different from the patient ventilator interaction observed in conventional modes, where the ventilator-generated pressure is either constant or inversely related to effort. The advantage of PAV is that it yields to the patient’s own neuromuscular control mechanisms and is guided by motion of the respiratory system synchronizing the ventilator’s output with the patient’s continuously changing needs. Because tidal volume and flow rates are controlled by natural breathing with PAV they vary continuously; therefore, PAV requires backup in the event that the patient’s ventilatory effort ceases. PAV has the potential for providing appropriate ventilatory support in a variety of clinical settings, ranging from acute lung injury, to weaning from mechanical ventilation, to increasing exercise tolerance in patients with COPD for pulmonary rehabilitation when used noninvasively.62 PAV theoretically improves the physiological relationship between inspiratory effort and ventilatory return that often characterizes respiratory failure. PAV may require lower peak airway pressures than standard volume-targeted modes, improve patient comfort,63 and provide a better synchrony of breathing64, however, the clinical benefit has yet to be clearly established.
Dual-Targeting
In the 1990s, ventilator technology began incorporating a combination of both volume- and pressure-targeted features. In these new “modes”, the clinician sets a volume target for each breath, but unlike historical volume targeting modes, the ventilator utilizes a decelerating gas flow to deliver the breath. This requires the ventilator to initially do a series of test breaths to gauge lung compliance. The ventilator is then able to calculate how to deliver the breath with the lowest possible pressure. Calculations are performed on every breath. As lung compliance improves, the ventilator requires less pressure, as lung compliance worsens, the ventilator will increase pressure. Pressure changes are incremental, usually no more than 3 cms H2O at a time. The clinician sets a pressure limit not to be exceeded and the ventilator alarms as the pressure alarm level are reached. At that time, pressure will not increase, but volume will decrease until the clinician intervenes. Some ventilators have dual targeting designed modes, for example, PRVC, while other ventilators apply the feature of dual targeting to conventional volume modes, for example, SIMV with AutoFlow. It has been theorized that this technology may decrease work of breathing while being used in a lung protective strategy, however this has not yet been established.47
Noninvasive Ventilation
Noninvasive ventilation (NIV) is a term used to describe ventilatory support supplied via nasal prongs or some type of face mask to provide CPAP, BiPap or positive pressure ventilation to the nonintubated patient. Thus, NIV is not a mode of ventilation, rather a technique of delivering a mode. The machine used may be either a standard ventilator or a single purpose unit. Noninvasive positive pressure ventilation has been shown to have particular benefit for patients with COPD in avoiding intubation and failure of extubation, as well as facilitating weaning.65 Noninvasive ventilation may also be used to increase exercise tolerance for patients with COPD62 or for sleep apnea.
Continuous positive airway pressure—Continuous positive airway pressure (CPAP) is a form of ventilatory support that is simply PEEP delivered to a patient who is spontaneously breathing. No machine breaths, that is, positive pressure breaths, are delivered. CPAP is the terminology reserved for patients who are only spontaneously breathing, while PEEP is terminology used when patients are receiving some form of positive pressure breaths. Both CPAP and PEEP increase FRC and help prevent derecruitment of alveoli. In normal subjects, CPAP increases tidal volume by 25% and lowers respiratory rate by over 30%.66 In intubated patients CPAP may decrease the work of breathing by 50%. CPAP is frequently used as a method of weaning the patient from the previously described modes of ventilatory support. CPAP can also prevent the flail action of a paralyzed hemidiaphragm, thereby improving the efficiency of the remaining innervated respiratory muscles, and may also prevent atelectasis.
CPAP can be delivered with a mechanical ventilator or a separate device via a tracheal tube, nasal prongs, or face mask. Nasal CPAP has been shown to reduce the number of apneic episodes, arrhythmias, and hypoxic episodes during sleep and to reduce daytime sleepiness and improve neuropsychiatric function in patients with obstructive sleep apnea, which affects 2% to 4% of the population.67,68 Nasal CPAP provides a pneumatic splint for the airway, preventing airway collapse during sleep (when upper airway dilator muscle activity is low), and increases the airway caliber in the retropalatal and retroglossal regions. Nasal CPAP also increases the lateral dimensions of the airway and thins the lateral pharyngeal walls. Typical settings are 5 to 20 cm of H2O pressure. Poor patient compliance is noted with nasal prongs and face masks, which may be related to facial and skin discomfort, rhinitis, nasal irritation and dryness, difficulty exhaling, and claustrophobia. Full-face face masks have been associated with increased aspiration and are typically reserved for patients with persistent mouth leaks.68
Because CPAP can increase functional residual capacity and shorten inspiratory muscles placing them at a mechanical disadvantage, there is the potential to worsen inspiratory muscle weakness. However, patients with preexisting shortened inspiratory muscles because of COPD may benefit from the ventilatory assistance of CPAP.66–69 Some patients may require CPAP at night or while in bed to maintain adequate oxygenation, yet have adequate oxygenation while they are mobile. After consulting with the physician, the physical therapist can evaluate whether CPAP is required during mobility activities. CPAP can frequently be disconnected for short periods of time during ambulation and wheelchair mobility activities, or extension cords and battery packs can be used to provide adjunctive CPAP during functional tasks. Likewise, CPAP during physical therapy interventions such as aerobic exercise training or functional training may enhance patient tolerance, comfort, and compliance.
Bilevel positive airway pressure—Bilevel positive airway pressure (often referred to as BiPap) may be used with noninvasive ventilation for ventilatory support. As with CPAP, intubation is not required. Sleep apnea is a common indication, along with exacerbations of COPD, congestive heart failure, and cystic fibrosis. BiPap is sometimes referred to as bilevel CPAP because it adds the advantage of an inspiratory positive airway pressure to CPAP. As with CPAP, BiPap may be used exclusively at night or intermittently throughout the day depending on the patient’s condition. Successful treatment can be predicted by improvement in pH, PaCO2, PaO2, and functional status.18,69
CLINICAL CORRELATE
Physical therapy interventions are used to assist in strengthening the respiratory muscles and provide general conditioning to assist in weaning the patient from the ventilator. The physical therapist should closely monitor oxygen saturation, respiratory rate, and the patient’s tolerance to activity. The physical therapist should notify the physician when a patient has markedly abnormal signs and symptoms during treatment. It may be necessary to add invasive or noninvasive mechanical ventilation to allow the patient to tolerate physical therapy and nursing interventions.
IMPROVING OXYGENATION FOR THE DIFFICULT TO VENTILATE PATIENT
Patients with acute lung injury (ALI), ARDS, or other disease processes may reach a point in their management when they cannot be adequately oxygenated despite optimal traditional ventilation strategies. The physician may have tried several different modes of mechanical ventilation, and is now faced with a dilemma of how to best oxygenate the patient. The use of prone positioning, extracorporeal membrane oxygenation, nitric oxide, and independent lung ventilation are all options to improve oxygenation in patients with severe respiratory failure.70–73
Prone Positioning
Prone positioning has been used since the 1970s to improve oxygenation of patients with acute respiratory failure. The mechanism of action appears to be a reduction of compressed lung segments by the heart, recruitment of collapsed lung tissue by reexpansion of dependent consolidation as it shifts from dependent to nondependent positions, and improved gravity-related drainage from previously dependent consolidated lung tissue, all resulting in improved matching of ventilation to perfusion.70,73 Turning a patient into the prone position for 7 to 20 hours per day has been shown to improve oxygenation within 2 hours to 10 days of implementing the procedure. It is also thought to decrease the incidence of ventilator-associated pneumonia.70,74,75 In one study, 30 minutes of prone positioning recruited more edematous lung than adjusting ventilator settings to optimize PEEP, as noted by computed tomography. The results were most pronounced in patients with lobar ALI.76 Turning a patient prone is considered safe without an increased incidence of displacement of the tracheal tube or accidental extubation, however; an increase in pressure sores without significant sequelae has been noted.75,77 Introducing prone positioning earlier in a patient’s care may prevent the need for 100% inspired oxygen.
Physical therapists may be consulted to assist the nursing staff in turning a patient with neuromuscular or musculoskeletal impairments into the prone position, or for airway clearance of the most involved dependent lung segments (Fig. 19-4). A good rule of thumb is to turn the patient from supine, onto the least involved side, and then prone. Range-of-motion exercises can also be performed. Nurses may look to physical therapists for guidance and instruction on how best to provide range of motion exercises and move limbs while in the prone position, as well as positioning of limbs during turning.
Extracorporeal Life Support
Extracorporeal life support (ECLS), also commonly referred to as: extracorporeal membrane oxygenation (ECMO), extracorporeal carbon dioxide removal (ECCO2R), or extracorporeal lung assist (ECLA), is a supportive therapy that may be used in patients with severe, but reversible cardiorespiratory failure. Studies regarding the efficacy of ECLS in adults with respiratory failure are not conclusive, though its use in neonates and pediatric patients is well established.78,79 Its use with adults continues in primarily large, academic centers of excellence, where clinical experience has supported its efficacy. ECLS, when used for respiratory failure in adults, most commonly utilizes a venovenous circuit. A common femoral venous cannula drains large volumes of deoxygenated blood, which is pumped through an oxygenating device and a heat exchanger prior to being infused back into the patient through a right internal jugular cannula.80 The extracorporeal blood flow is titrated according to the patient’s required level of support, generally beginning with full support, with flow rates of approximately 100 mL/kg/min.81 Gas exchange takes place in the extracorporeal support system, allowing ventilator management to focus on lung protection, while aggressive measures to treat the underlying disease process continue. Patients on ECLS are often sedated (the degree of sedation may be minimal depending on institutional practice).
ECLS is an invasive and often intimidating form of life support, particularly for clinicians who have not been exposed to this modality. Although the cannulas are large, and the extracorporeal blood volume quite significant, these patients frequently require physical therapy interventions including airway clearance techniques, contracture prevention, and positioning. Patients requiring ECLS may also be positioned prone as an adjunctive therapy. As with most invasive monitoring lines, the cannulas are sutured, reducing the risk of dislodgement. They are also wire reinforced, significantly reducing the risk of kinking. Blood flow through the system is also closely monitored and tracked, another safeguard which allows judicious patient care. While studies examining the limits of ROM are lacking, it is the personal experience of these authors that hip flexion to 90 degrees, abduction to 45 degrees, internal and external rotation to 30 degrees, full shoulder flexion and abduction, and three-fourth prone positioning toward and away from the cannulated side can be performed without complications.
Inhaled Nitric Oxide
Inhaled nitric oxide (INO) is another adjunctive therapy for patients with ARDS/ALI and life-threatening hypoxemia, who have failed conventional lung protective ventilation. NO improves hypoxemia by causing vasodilation to areas of well-ventilated lung while diverting blood away from poorly ventilated lung, thus improving ventilation/perfusion matching.70,71 Significant improvement in PaO2/FiO2 ratios, while decreasing pulmonary vascular resistance occurs with low doses of INO.82
NO appears to be a safe, yet expensive intervention; however, its effect on mortality, ICU stay and functional outcomes is not yet known. The precautions are the same as with all mechanically ventilated ICU patients; physical therapists should be careful not to displace any of the NO equipment when working with these patients and discuss any concerns with the nurse or physician. (See Chapters 6 and 8 for further discussion of NO.)
Synchronous Independent Lung Ventilation
Synchronous independent lung ventilation (SILV) may be required for patients who have failed conventional methods of full ventilatory support, because the injury or disease to one lung is so severe that it prevents both lungs from being adequately ventilated. Independent lung ventilation may be necessary with unilateral, or asymmetrical lung disease as a result of severe pulmonary trauma (blunt or penetrating), aspiration pneumonitis, or bronchopleural fistula. These patients usually do not tolerate position changes with conventional ventilation and ultimately may require resection of part or all of one lung (see Chapter 7). Pulmonary congestion and the loss or inactivation of surfactant in the diseased lung may lead to a decrease in tissue elasticity and decreased lung compliance. Therefore, the gases delivered to the lung follow the path of least resistance and the “healthy” or “better” lung receives the majority of the tidal volume delivered by the ventilator. Higher inspiratory pressures are required in attempts to ventilate the more diseased lung, which may have detrimental effects on the lung tissue and potentially a greater potential for VILI (see potential risks associated with mechanical ventilation section). Hyperinflation of the healthy lung may also occur, diverting blood flow to the affected lung, which results in a large dead space. Furthermore, overdistension of healthy alveoli may often result in volutrauma leading to ALI. Ventilation to the healthy lung continues, but pulmonary perfusion is either decreased or absent, resulting in poor gas exchange (Table 19-4).
TABLE 19-4 Effects of Conventional Mechanical Ventilation on Alveoli in Unilateral Lung Disease
The advantage of independent lung ventilation is that each lung can be ventilated separately; overdistension of the good lung can be prevented while adequately ventilating the “sick lung.” Two ventilators are required. The patient is intubated with a double-lumen endotracheal tube. Intrapulmonary cross-contamination is prevented by the presence of a distal second cuff on the endotracheal tube, which is inflated in the mainstem bronchus, usually the left. The end of the tube has an attachment for each ventilator.83 Computerized assessment techniques allow synchronous mechanical ventilation to each lung with a different FiO2, different tidal volumes, and PEEP levels. Different modes of mechanical ventilation, tailored to the pathophysiology of each lung may be required.73,83 Typically, oxygenation improves within hours after SILV is initiated.
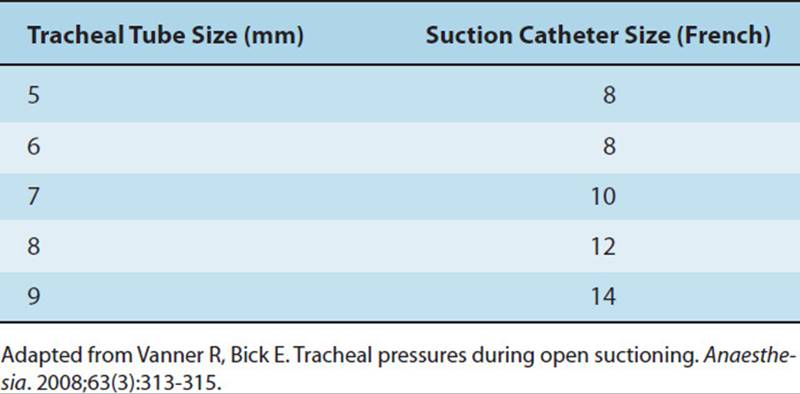
Patients on SILV are typically on bed rest. Standard postural drainage positions and manual techniques can be administered. Because the patient is intubated with a double-lumen tube (which prevents transbronchial aspiration), each lung can be suctioned separately, as clinically indicated. The suction catheter is changed between passes to each lumen to prevent contamination from one lung to the other. Ideally, an in-line catheter is in use. The therapist should also note the size of the internal diameter of each port of the endotracheal tube because smaller suction catheters are usually necessary. The suction catheter should not exceed half the diameter of the airway (Table 19-5).
TABLE 19-5 Recommended Suction Catheter Sizes with Tracheal Tube Sizes
When deciding which intervention or combination of interventions is optimal to improve oxygenation for a specific patient, resource availability, physician preference, and the expertise of health care practitioners must be taken into consideration. The effect of these four interventions on long-term outcomes, including mortality, is unknown. Improvement in oxygenation may lead to a decrease in inflation pressures and could be associated with improved outcomes. However the mortality for patients with ventilator-associated lung injury and severe ARDS/ALI remains high.
LIBERATING THE PATIENT FROM MECHANICAL VENTILATION
1.Weaning is the term used when trying to liberate a patient from mechanical ventilation. Discontinuation from mechanical ventilation is easily achieved in the majority (70%–80%) of patients.18 Weaning should not be initiated until the patient can maintain alveolar ventilation with less ventilator support without causing excessive stress, which may lead to respiratory muscle fatigue. It is advised that weaning not be initiated until the pathophysiology for weaning failure and any imbalance between energy supply and demand for the respiratory muscles is corrected.52 Metabolic disturbances and circulatory disturbances are easier for the physician to correct than neuromuscular incompetence, where a program of exercise interspersed with rest may be necessary. The decision of how to wean depends partly on the type of ventilator and primarily on the physiologic response of the patient to the weaning process. To wean, a patient may be changed from assist-controlled ventilation to SIMV with pressure support, or from APRV to CPAP. In addition, the degree of pressure support, FiO2, and PEEP may be lowered as the patient’s condition improves. Once the PEEP and FiO2 have been reduced, there are several methods to further reduce ventilatory support and assess the patient for extubation. Recent research strongly supports the use of daily spontaneous weaning trials (SBTs) to aid in determining a patient’s readiness for ventilator discontinuance.52,84 SBTs are recommended for patients who demonstrate reversal of the underlying cause of respiratory failure, adequate oxygenation, hemodynamic stability, and the ability to initiate an inspiratory effort52(Table 19-6A). SBTs are typically performed on low levels of pressure support, CPAP or a T-piece and lead to liberating the patient from the ventilator (Table 19-6B). Failure of a SBT requires that the medical team reevaluate the cause of respiratory failure, and attempt to rectify any reversible causes, after returning the patient to a stable, nonfatiguing mode of ventilation.52 The physical therapist should be aware of the physician’s or ICU’s weaning protocol and different weaning modes to appropriately modify treatment interventions. The three most commonly used techniques are T-piece weaning, synchronized intermittent mandatory ventilation with pressure support, and pressure-support ventilation. All of these methods lead to removal of the mechanical ventilator and independent patient breathing. Daily physical therapy interventions may need to be timed to accommodate SBTs.
TABLE 19-6A Clinical Considerations (Adult) “Weaning” from Mechanical Ventilation Parameter Consider Weaning Normal Value
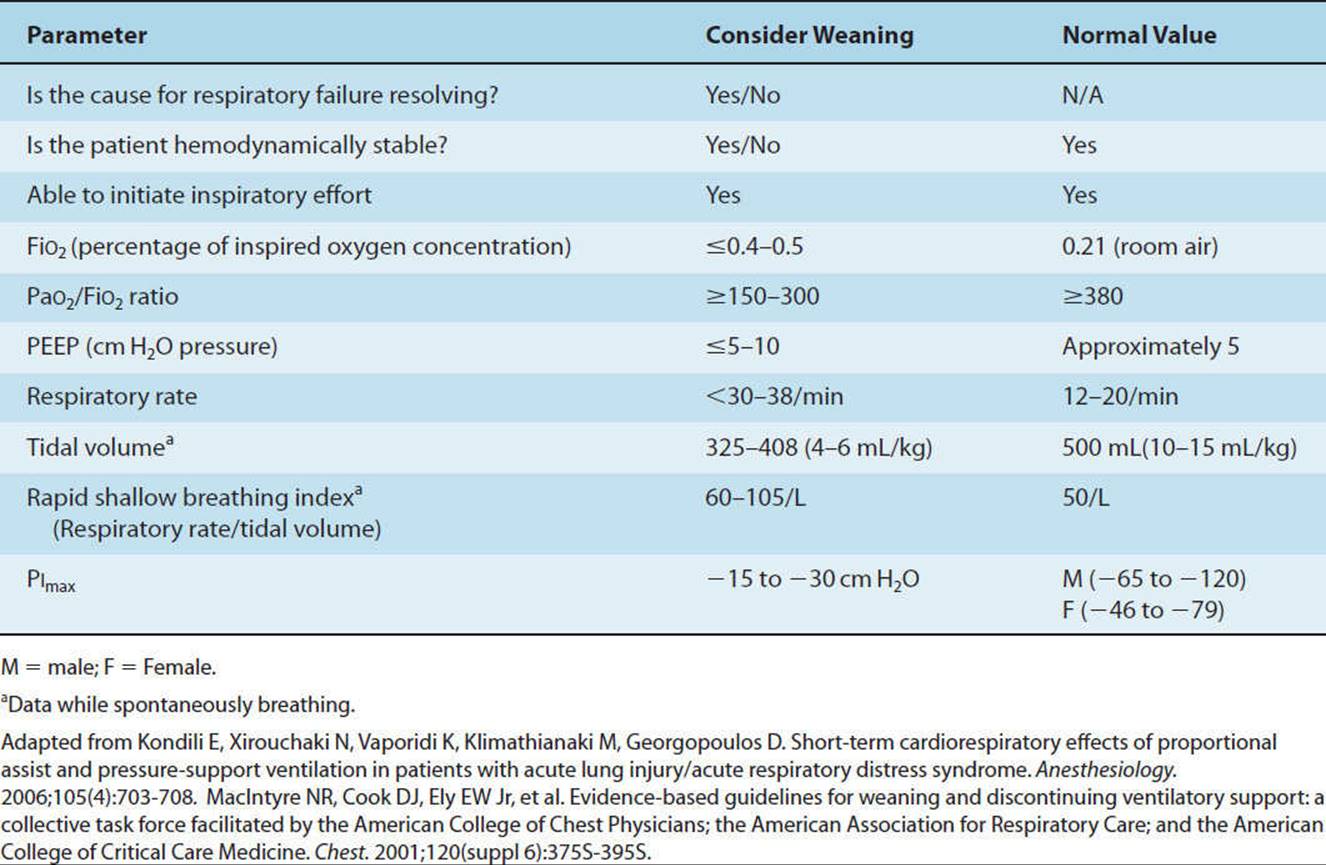
TABLE 19-6B Criteria for a Spontaneous
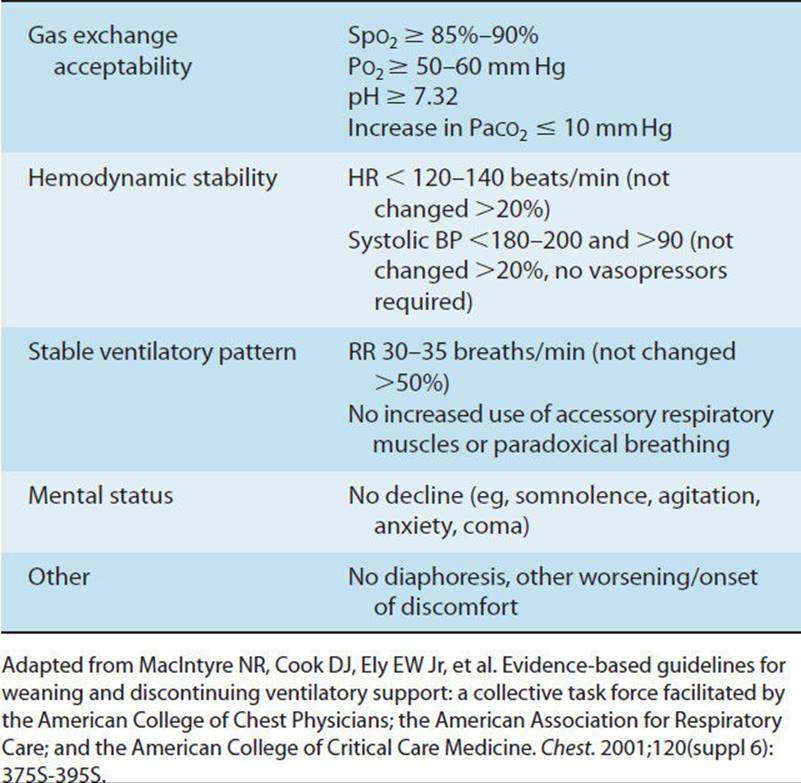
T-Piece Weaning
T-Piece weaning refers to removing the patient from the ventilator and placing a plastic adapter shaped like a T on the patient’s tracheostomy or endotracheal tube. The patient is still intubated yet able to breathe on his or her own with humidified supplemental oxygen. If the patient maintains good vital signs, blood gases, oxygen saturation, and an appropriate breathing pattern, extubation usually occurs within hours. Supplemental oxygen is usually still required and is delivered through a face mask or nasal cannula. This is the oldest method of weaning and is associated with outcomes similar to other methods. Patients may receive airway secretion clearance techniques during T-piece weaning. This may enable them to better tolerate extubation. If a patient fails a T-piece trial, they will be put back on their previous mode of mechanical ventilation and physical therapy interventions can be continued. This decision is usually made within a few hours.
Synchronized Intermittent Mandatory Ventilation and Pressure Support
Synchronized intermittent mandatory ventilation can be used as a weaning mode by gradually reducing the number of breaths per minute delivered by the ventilator over several days or weeks. SIMV weaning was initially thought to be a weaning mode that would decrease the duration of mechanical ventilation. However, there is some evidence that this mode of ventilation increases weaning time rather than decreasing the time necessary for mechanical ventilatory support.59,85 When mechanical breaths are decreased, there is a gradual increase in the mean inspiratory effort. When weaning with pressure support ventilation unloading of the respiratory muscles is more gradual. Pressure support can also be added to spontaneous breaths to reduce the additional work of breathing during SIMV weaning.
MacIntyre suggests that patients who remain mechanically ventilated for greater than 21 days “should not be considered permanently ventilator dependent until three months of weaning attempts have failed.”52 Ventilator support should be gradually reduced, and self-breathing trials of gradually increasing duration be employed.52 Upper arm strength was a predictor of weaning time in the chronically ventilated patients, giving support to the role of physical therapy in this population.86
CLINICAL CORRELATE
When a patient is being weaned and does not tolerate physical therapy interventions during weaning trials, the physical therapist should confer with the physician to determine if the ventilator can be adjusted back to preweaning settings which may facilitate the provision of physical therapy interventions. As a general guideline, physical therapy treatments should be performed when the patient is not being weaned.
PNEUMONIA AND ATELECTASIS
Pneumonia is an inflammatory response of the bronchioles and alveolar spaces to an infective agent.87 These infections may be bacterial, fungal, or viral. Exudates lead to lung consolidation, dyspnea, tachypnea, and adventitious breath sounds on clinical examination of the involved lung lobe or segment. Pneumonia is the leading cause of death in patients with spinal cord injury (SCI).88
Ventilator-associated pneumonia (VAP), the most frequent type of nosocomial infection in the ICU, occurs in up to 30% of intubated patients.89 VAP consists of a pulmonary inflammatory reaction and sepsis in patients who are mechanically ventilated for a minimum of 48 hours. It is difficult to accurately diagnose, and because the underlying illness of critically ill patients often has a high mortality rate, it is difficult to differentiate to what extent mechanical ventilation contributes to the risk of nosocomial pneumonia.90 Historically, clinical findings have been used to diagnose pneumonia yet have not been shown to have sufficient accuracy, leading to the overuse of antimicrobial therapy (Table 19-7). Quantitative cultures are recommended to determine the bacterial load in a tracheal sample to differentiate colonization from infection and avoid overutilization of antibiotics. Clinicians may also see the use of biomarkers to assist in the diagnosis of VAP in the future.89
TABLE 19-7 Criteria for the Diagnosis of Pneumonia
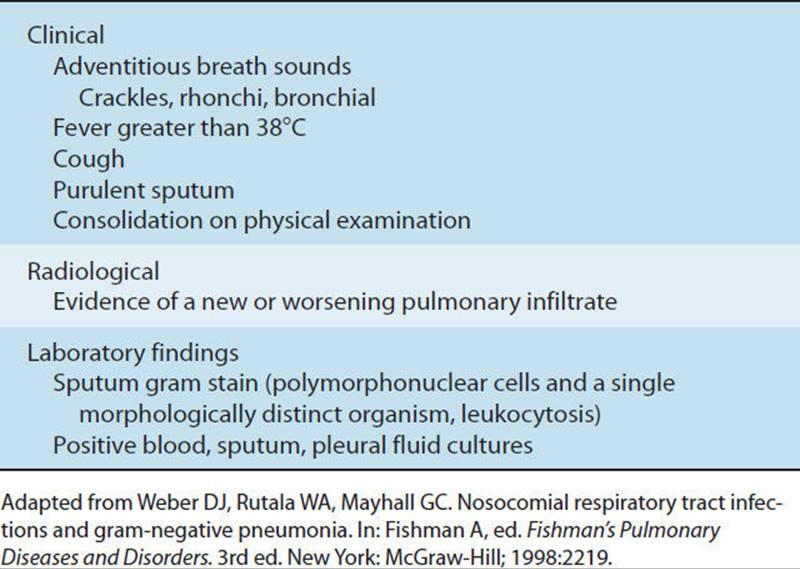
Placing mechanically ventilated patients in a semirecumbent position of 45 degrees has been demonstrated to reduce the incidence of VAP. Physical therapists can assist the nursing staff in moving patients with abnormal muscle tone or orthopedic injuries, which make positioning supine, side-lying, and sitting a challenge. VAP has a documented mortality rate from 20% to as high as 70% with multi resistant pathogens, despite treatment with antibiotics.91Consequently, the treatment of pneumonia with antibiotics is controversial for the mechanically ventilated patient.92–94 Antibiotic usage has its own adverse effects: allergic reactions, acute renal failure, development of antimicrobial resistance, superinfections, and death. Therefore antibiotics should be prescribed cautiously with consideration as to the cost and adverse effects. Pneumonia in patients who have received previous antibiotic therapies is associated with a higher mortality.92 Antibiotics are given to patients who do not need them as much as 77% of the time, with some evidence that mortality is greater in those patients receiving antibiotics.90
Although a placebo-controlled trial for antibiotic treatment of VAP has not been conducted, there is some evidence that chest physical therapy may be utilized to help in the diagnosis of pneumonia prior to administering antibiotics. Joshi and colleagues studied 39 trauma patients, 32 of whom were intubated, mechanically ventilated, and diagnosed with pneumonia. Chest physical therapy was initiated within 72 hours. Thirty-one of the 39 patients showed complete or partial clearing of the pulmonary infiltrate without antimicrobial therapy.95 Patients who present with the traditional signs of pneumonia may have atelectasis rather than a true infection of the bronchioles and alveoli. Atelectasis is defined as an incomplete expansion, or collapse, of the lung and can result from secretion retention for which airway clearance techniques are beneficial, even when the chest radiograph shows an infiltrate. Atelectasis without secretion retention may be the result of compression of the lung (eg, pneumothorax, hemothorax, pleural effusion, or lung tumor) and would therefore not be responsive to secretion removal techniques. When an infiltrate noted on the chest radiograph improves within 24 to 48 hours, it is not indicative of pneumonia. Therefore, chest physical therapy interventions that clear an infiltrate within hours or days may assist the physician in determining whether the patient truly has pneumonia and needs antibiotics. This is an example of an intervention that also aids in a diagnosis.
Newer techniques such as bronchoalveolar lavage and quantitative cultures of protected sputum brushings are reported to have better sensitivity and specificity in the diagnosis of pneumonia, yet there is controversy as to which technique is most effective. It is suggested that consideration be given to these examinations to better diagnose pneumonia and potential pathogens for immunocompromised patients who present with a broad range of potential pathogens (eg heart or lung transplant patients, critically ill patients with severe VAP) and when the patient’s condition is not improving with empiric antimicrobial therapy. Invasive diagnostic testing may lower mortality and provides more antibiotic free days.94
TRACHEAL, BRONCHIAL, AND PULMONARY SECRETION REMOVAL
Tracheal Suctioning
Physical therapy interventions frequently mobilize pulmonary secretions. Tracheal suctioning may, therefore, be necessary to remove secretions from the upper airway. If a patient is unable to either cough or huff secretions to the proximal portion of the tracheal tube, deep suctioning is indicated. Suctioning is a sterile procedure. Eye protection, a mask, and sterile gloves should be worn when not using an inline catheter, as the physical therapist is at risk for exposure to blood and body fluids. Deep suctioning is required for many mechanically ventilated patients and requires that the catheter be inserted until an obstruction is felt (usually the carina or wall of the right mainstem bronchus), then slightly withdrawn, suction is continuously applied while the catheter is withdrawn from the airway (Table 19-8).
TABLE 19-8 Recommended Procedure—Tracheal Suctioning
1. Check baseline vital signs (eg heart rate, blood pressure, Spo2) and arterial blood gases.
2. Check equipment to make sure it is operational (vacuum pressure < 120 mmHg).
3. Note ventilator settings, increase FiO2 if baseline SpO2<92% to 95%.
4. Check with medical staff regarding need for a PEEP adapter if PEEP ≥ 10 cmH2O pressure, if not using an inline catheter.
5. Explain procedure to patient.
6. Don mask, eye protection, gloves.
7. Attach suction catheter to suction tubing.
8. Discontinue patient from oxygen source.
9. Increase ventilation by giving 3–5 breaths with a manual resuscitator bag if the patient is breathing spontaneously.
10. Insert suction catheter into the airway until resistance is felt. Apply suction after withdrawing the catheter a few centimeters. Suction should be continuous, and catheter rotated while withdrawing. Assess vital signs during the procedure.
11. Ventilate with 3–5 breaths from a manual resuscitator bag if indicated. Reattach the patient to the oxygen source, recheck vital signs, if SpO2 has dropped wait until it returns to baseline. Increase FiO2 if indicated.
12. Repeat steps 9–11 as indicated for 2–4 suction catheter passes. Change suction catheter if the patient requires more than 3–4 passes of the suction catheter.
13. Suction the mouth.
14. Return FiO2 to baseline setting and reassess vital signs.
15. Dispose of suction supplies.
The recommended features of tracheal suction catheters and complications of tracheal suctioning are listed in Tables 19-5 and 19-9. One of the complications of tracheal suctioning that contributes to tracheolaryngeal ciliary dysfunction is irritation of the airway mucosa. The greatest mucosal damage has been noted with vacuum pressures greater than 120 mmHg, although pressures as high as 170 mmHg are used. Clinicians should use the lowest pressure that is effective.96 Inline tracheal suction catheters were initially thought to be more beneficial than using a port adapter with a standard catheter to maintain PEEP during the suctioning procedure. Currently, there is no evidence to support a reduction in ventilator-associated pneumonia, decrease in hospital stay, or a decrease in mortality with the inline devices. The inline devices have been shown to have more colonization of the respiratory tract and should be changed every 24 to 48 hours to minimize pulmonary infection.97–100 The physical therapist may observe a clinician instilling saline in the tracheal tube to “loosen” secretions. However, saline does not reach the peripheral airways or change the rheological properties of the mucus. Saline instillation may also cause nosocomial infection, and the fluid instilled is not all retrieved.101–103 No studies have found that instillation of saline is beneficial, current recommendations are that instillation of normal saline should not be performed as a routine step with endotracheal suctioning.104 Patients should receive adequate hydration and airway humidification rather than resort to instillation of saline.
TABLE 19-9 Tracheal Suctioning: Catheter Features and Complications
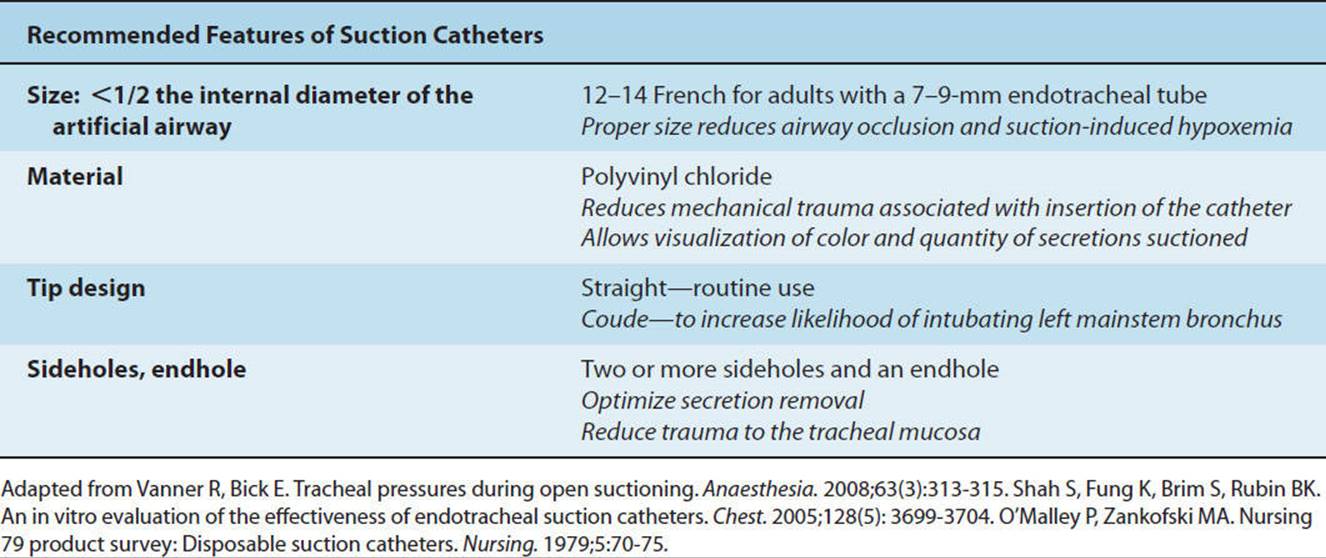
Prior to decannulation (removal of the tracheal tube), some physicians prefer to insert a fenestrated tracheostomy tube. The fenestrated tube can be plugged and requires that the patient breathe though the oropharynx (Fig. 19-5A). If the tracheostomy tube has a cuff, the cuff is deflated, and inspired gases pass through the fenestration and around the tube. Patients with fenestrated tubes who are in need of suctioning should have the plug and inner cannula of the fenestrated tube removed and replaced with a nonfenestrated cannula to minimize the chances of irritating or puncturing the posterior tracheal wall (Fig. 19-5A). When the patient cannot take deep enough breaths to maintain adequate oxygenation during the suctioning procedure, a manual resuscitator bag (MRB) is used to provide greater lung volumes, or for preoxygenation. Use of an MRB requires inflation of the tracheal cuff.
FIGURE 19-5 (A) Fenestrated tracheostomy tubes. (B) Standard endotracheal tube. (C) Bivona tracheostomy tube. (D) Variable length Bivona tracheostomy tubes.
In summary, suctioning through an artificial airway of a patient with adequate oxygenation and stable vital signs has relatively few contraindications. Suctioning should not be performed routinely, only when clinical indications for suctioning exist (rhonchi, increased airway pressure, decreased lung volumes, increased work of breathing, visible secretions) and with close monitoring of vital signs and vacuum pressures. Based upon the current literature, it is up to the clinician’s discretion whether to use an open or closed suction technique.
To Suction or Not to Suction—That Is the Question!
Prior to suctioning a patient with unstable vital signs or a low SpO2, the therapist should discuss the benefit of suctioning versus the risk of causing additional arrhythmias or desaturation with the medical and nursing staff. However, suctioning should not be withheld when indicated because retained secretions may result in airway occlusion and hypoxemia.
Nasotracheal suctioning (suctioning through a catheter inserted from the nose to the trachea without an artificial airway) is recommended only when vigorous airway clearance techniques, including prolonged postural drainage, cough stimulation techniques, and suctioning the oropharynx, are ineffective and the medical team does not plan to intubate the patient. See Table 19-10 for the complications associated with nasotracheal suctioning.
TABLE 19-10 Complications Associated with Tracheal Suctioning
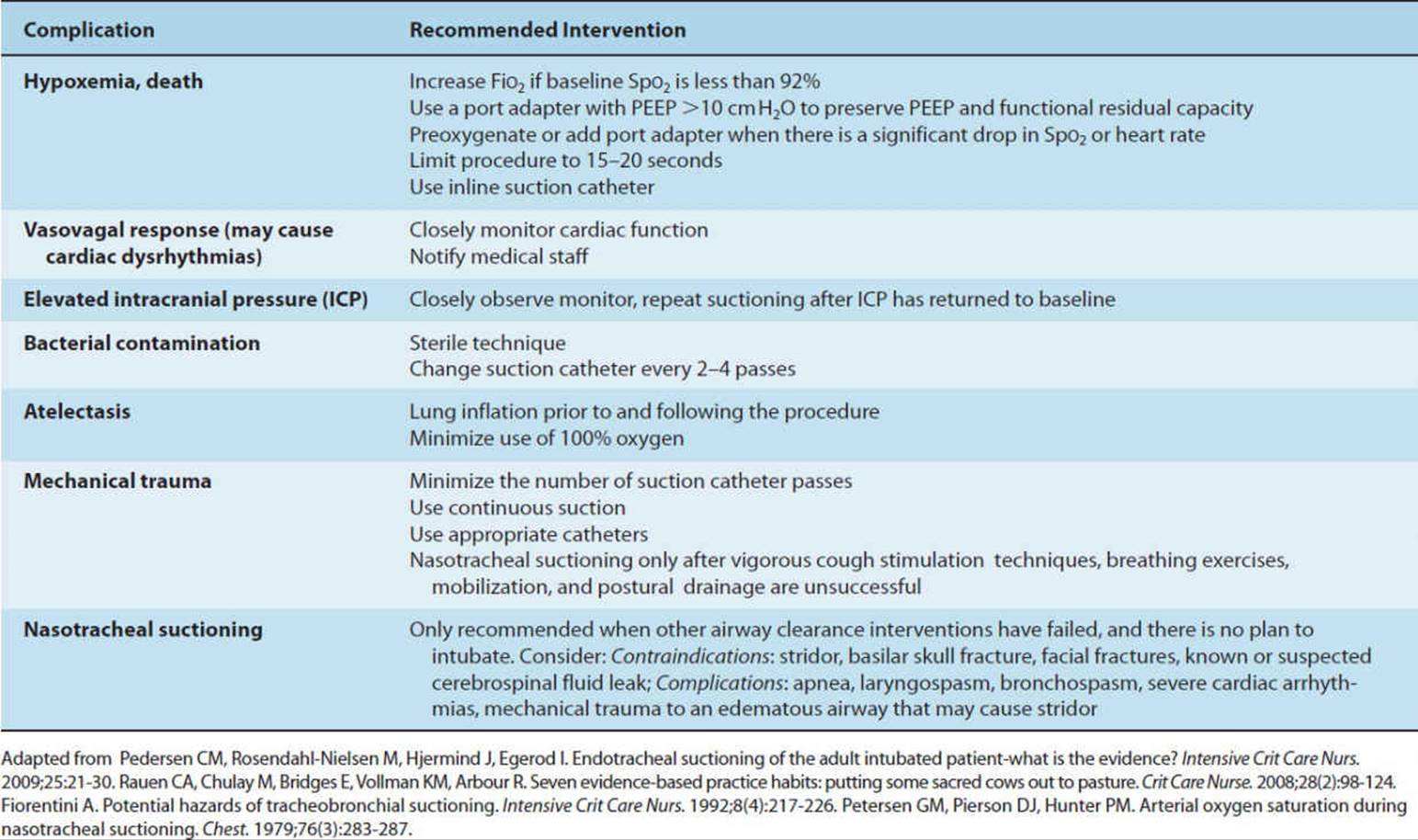
Refer to the CD-ROM for a detailed description of tracheal suctioning including a patient demonstration.
CLINICAL CORRELATE
The therapist should note the type of tracheal tube the patient has in place (endotracheal vs tracheostomy) and if the tracheostomy tube has a fenestration. The length of the tracheal tube will give the therapist a gross assessment of how far the catheter may be inserted into the airway. Suctioning is recommended only when cough instruction and breathing exercises are ineffective. Deep suctioning is reserved for patients who cannot mobilize secretions into the tracheal tube. Vital signs and airway pressures are monitored throughout the procedure. Following suctioning, the therapist may note an improvement in breath sounds and lower airway pressures or increased volumes on the ventilator.
Therapeutic Bronchoscopy Versus Chest Physical Therapy
Therapeutic Bronchoscopy
A bronchoscope is a multipurpose instrument that is inserted into the upper airway to perform diagnostic and therapeutic procedures. The procedure is performed by a physician and referred to as a bronchoscopy, a common therapeutic modality in the management of critically ill patients. Flexible bronchoscopes are used in conjunction with a swivel adaptor and rubber diaphragm to allow mechanical ventilation throughout the procedure.105 Because of the associated risk of hypoxemia, the FiO2 is increased to 100% during the procedure. The diagnostic indications for bronchoscopy include visualization of the airway for tracheal or bronchial tears, burns, foreign bodies, tumors, and placement of airway stents. Qualitative cultures obtained via bronchoscopy may assist in the diagnosis of pneumonia and prescription of an appropriate antimicrobial agent (see Chapter 8). Bronchoscopic needle aspiration is used to stage lung cancer. The therapeutic indications include aspiration, lung contusion, lung abscess, atelectasis from secretion retention, and the removal of foreign bodies. These are the same therapeutic indications for airway clearance techniques performed by physical therapists, nurses, and respiratory therapists.
The mechanically ventilated patient usually presents with borderline cardiopulmonary function where even small changes in ventilation and hemodynamics during bronchoscopy can have critical effects. Therefore, this procedure should be scrutinized, performed with caution, and utilized therapeutically when chest physical therapy treatment is unsuccessful.105,106 The complications of therapeutic bronchoscopy with a comparison to chest physical therapy interventions are listed in Table 19-11. Although research comparing the advantages and disadvantages of chest physical therapy to therapeutic bronchoscopy is limited, there is some evidence that one chest physical therapy treatment is equally or more effective than therapeutic bronchoscopy in the treatment of lobar collapse from secretion retention.107–112
TABLE 19-11 Complications of Bronchoscopy
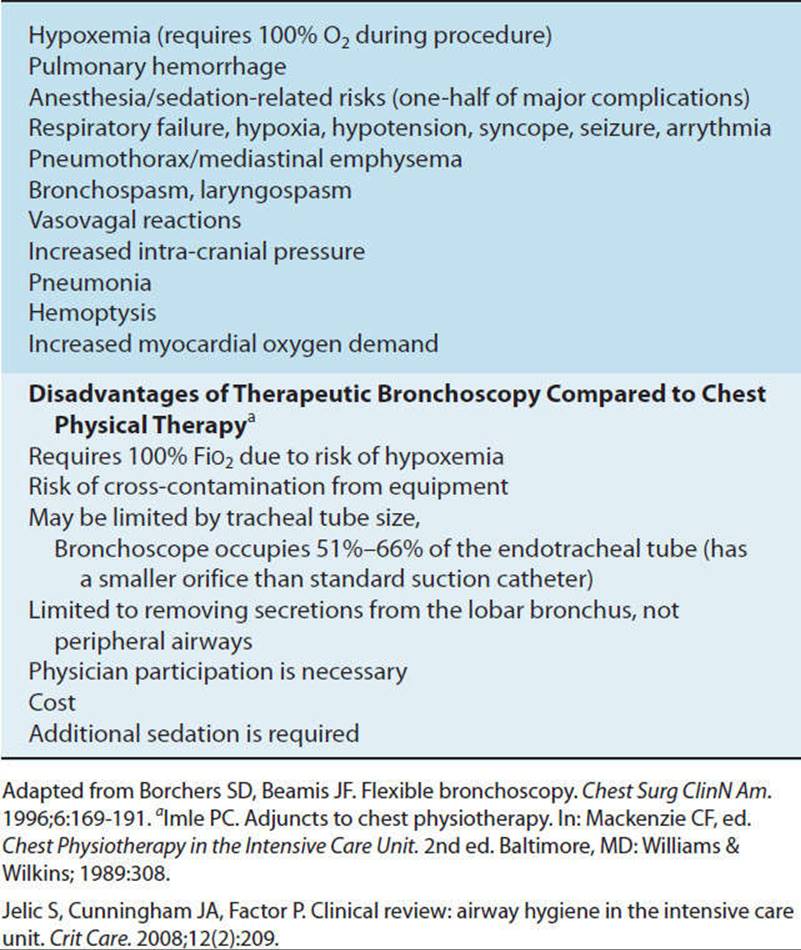
Chest Physical Therapy
Several case studies have demonstrated improved oxygenation and a decrease in the FiO2 after one chest physical therapy intervention.107,110,113 In these studies patients had a clear indication for chest physical therapy treatment.
CLINICAL CORRELATE
The indications for chest physical therapy include radiological evidence of atelectasis or infiltrate associated with secretion retention, adventitious breath sounds coupled with secretion retention, poor cough, and a decrease in arterial oxygenation thought to be a result of secretion retention.
Chest physical therapy consisted of postural drainage positioning to the areas of radiological involvement, manual percussion, manual vibration, airway suctioning and breathing exercises with assistive cough techniques for spontaneously breathing patients. Treatment duration was not predetermined. Patients were treated as long as they were productive of secretions; and breath sounds were improving, suggesting that pulmonary secretions were being mobilized.107,110,114 Patients who are mobile and able to expectorate secretions would not require these airway clearance interventions. Patients with lobar collapse who are unable to be mobilized or optimally positioned for postural drainage to an area of radiologically proven pathology may be responsive to bronchoscopy if they remain symptomatic after 24 hours of aggressive chest physical therapy.106 The primary problem for secretion retention should be resolved and not considered an indication for repeated invasive intervention of the airways.106
CLINICAL CORRELATE
With optimal positioning for postural drainage, percussion, and vibration, it may take 20 to 30 minutes to mobilize secretions from the lung parenchyma and alveolar areas. Evidence of successful secretion mobilization via positioning and manual techniques is typically characterized by diminished or absent breath sounds prior to positioning which become crackles, rhonchi, or vesicular during treatment.
POTENTIAL RISKS ASSOCIATED WITH MECHANICAL VENTILATION
Risk Factors Associated with Intubation
Patients requiring prolonged mechanical ventilation usually require tracheal intubation. A tube is placed in the trachea through the nose (nasotracheal tube), mouth (orotracheal tube), or an incision in the trachea (tracheostomy tube) (Fig. 19-5). The proximal/external end of the tracheal tube is connected to tubing and attached to the mechanical ventilator. In adults, tubes with an inflated cuff are used. Indications for tracheal intubation are included in Table 19-2. With a tracheal tube, the normal cough mechanism, which includes glottic closure, is interrupted and the inflated cuff inhibits mucociliary function. Complications of intubation include translocation of organisms from the oropharynx to the lung, intubation of the right mainstem bronchus, laryngeal or tracheal injury (perforation of the trachea is rare), and aspiration. After the patient is intubated by the physician (or in some institutions the respiratory therapist) the position of the tube is confirmed by a chest radiograph (distal end 2–3 cm above the carina) before the initial physical therapy intervention.21 Airway clearance techniques may be indicated shortly after intubation if aspiration is suspected. Physical therapists working with intubated patients should check that the tube is secured in place and that the patient is appropriately restrained if at high risk for self-extubation. During treatment interventions the physical therapist should note if there is a marked difference in inhaled and exhaled tidal volumes, which may denote an air leak around the tracheal tube, or a cuff that is not properly inflated or may be damaged. This finding should be reported immediately to the nurse or physician. If inflating the cuff does not resolve the problem, the tracheal tube may need to be changed by the physician prior to any further physical therapy interventions.
It has been reported that the majority of patients intubated for greater than 3 days have some type of tracheal or laryngeal injury ranging from mild mucosal erythema to ulceration, granuloma formation, or vocal cord immobility.115Laryngotracheal injury or edema narrow the upper airway, predisposing the patient to stridor (a high pitched inspiratory wheeze located in the trachea or larynx) once the tube is removed. Stridor is a serious complication, which may require that the patient be reintubated because of upper airway occlusion when the lungs themselves are functioning normally. The highest risk of stridor is when a patient extubates himself or herself without the cuff being deflated, causing the inflated cuff to be pulled through the vocal cords. Stridor does not appear to be related to the number of intubations and usually occurs within the first few hours after extubation.116,117
The initial treatment for stridor is inhalation of racemic epinephrine and intravenous corticosteroids, or inhalation of a helium–oxygen gas mixture (heliox). Inhaling heliox reduces the large pressure drop associated with turbulent flow across the obstruction.118,119 Secretion removal techniques, including head-down positioning, may be necessary to assist in the removal of pulmonary secretions.
CLINICAL CORRELATE
Head-down positioning with close monitoring of vital signs is less invasive in assisting secretions to drain to the oropharynx than is nasotracheal suctioning. Nasotracheal suctioning is contraindicated with stridor due to the mechanical injury it may cause to the soft tissues. Reintubation is necessary if stridor does not respond to the treatment interventions previously described. Physical therapists should closely monitor patients postextubation and report any suspicion of stridor to the nurse or physician.
Endotracheal or tracheal tube cuffs may also cause pressure necrosis of the trachea resulting in tracheal stenosis and tracheal malacia. Using tracheal tubes with high-volume, low-pressure cuffs and monitoring cuff pressures reduces the complication of tracheal ischemia. Cuff pressures should be maintained below the pressure that occludes capillary blood flow to the tracheal tissues that are compressed by the cuff. Cuff pressures between 20 and 30 cmH2O pressure are recommended.120 Mean tracheal capillary perfusion pressure is approximately 30 mmHg. The lowest pressure that prevents leakage around the cuff is used. A minimal air leak at the moment in the ventilatory cycle when the tracheal diameter is greatest will also act to minimize tracheal ischemia. Patients who have developed tracheal injury related to the cuff may require a tracheal tube with a foam cuff (Fig. 19-5C).121
Nursing, speech–language pathology, and respiratory therapy personnel are usually capable of inflating and deflating the cuff on the tracheal tube. However, it has been shown that periodic cuff deflation is not beneficial. Deflating the cuff 5 minutes each hour does not restore blood flow to the tracheal mucosa and can create periods of inadequate ventilation, potential cardiovascular instability, and potential aspiration. Erosion of the trachea may also result in a fistula forming between the trachea and the esophagus (tracheal-esophageal fistula) or into the innominate artery (tracheal-innominate fistula). These complications are rare yet life threatening when they occur, requiring immediate surgical repair.
Impaired Mucociliary Function and Lack of Humidification
The presence of an endotracheal tube bypasses the naso-, oro-, laryngo-pharynx that humidifies inspired gases to 100% relative humidity and protects the lung from particulate, chemical and microbiologic matter.122 Inhalation of dry respiratory gases that bypass the normal mechanisms of warming and humidification can irritate the respiratory mucosa, reduce mucociliary transport, and cause drying and retention of lung secretions. Therefore, mechanically ventilated patients require some type of airway humidification.
The most common methods of airway humidification for mechanically ventilated patients are use of a heated humidifier or a heat and moisture exchanger.123 The humidifier allows gas to pass over a water reservoir and water is evaporated into the gas, increasing the humidity level of the gas delivered to the patient (Fig. 19-6A). The humidified gas then passes through a delivery tube to the patient. Most systems can deliver near 100% humidity at 37°C.123Water temperature is closely monitored to minimize condensation and prevent burning of the airway. Because condensation is often present in the tubing, humidifiers and ventilator tubing should be positioned lower than the patient to prevent the water from spilling back into the patient’s airway. When turning or mobilizing a patient, the physical therapist may need to maneuver the tubing to prevent backflow of water into the patient’s airway.
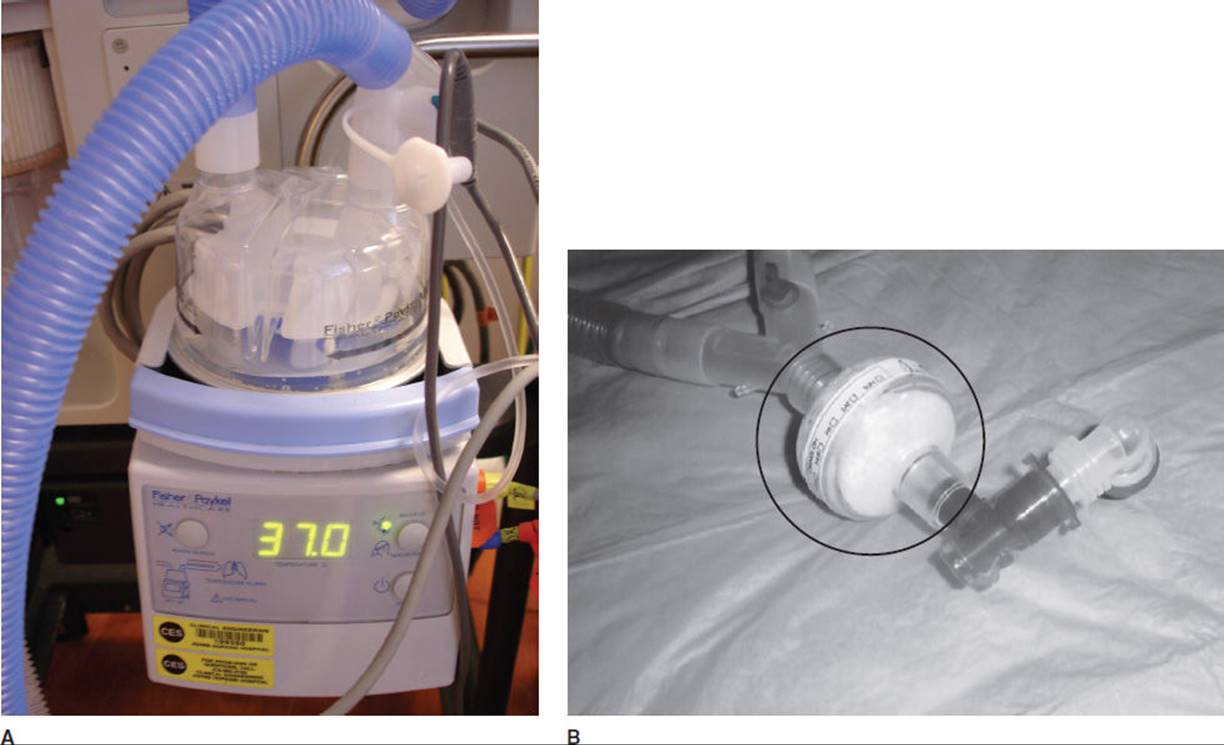
FIGURE 19-6 (A) Heated humidifier. (B) Heat and moisture exchanger—“artificial nose.”
Heat and moisture exchangers (HME) are placed on the distal end of the tracheal tube to minimize the loss of heat and humidity from the upper respiratory tract. They are commonly referred to as an artificial nose (Fig. 19-6B). This device recovers part of the heat and moisture contained in the expired air. Humidified expired gases pass through a sponge or similar material with low thermal conductivity, which conserves heat and causes condensation of moisture and heat retention. The retained heat and moisture are added to the inspired gases. The humidifying process is possible because of the condensation of water vapor during expiration and its subsequent evaporation during inspiration.123 HMEs are thought to require less maintenance than a heated humidifier.
HMEs may be sufficient for patients ventilated for a short duration, and patient transport, however for patients with long-term mechanical ventilation, haemoptysis, tenacious secretions, increased airway resistance and hypothermia, a heated humidifier is recommended.124,125 In the ICU setting, an increased viscosity of bronchial secretions has been noted during prolonged mechanical ventilation with an HME. Thick secretions can increase airway resistance due to clogging of the endotracheal tube or HME and may lead to increased respiratory effort by the patient, air trapping, airway obstruction, and pulmonary infection. The physical therapist working in the ICU should evaluate the consistency of the patient’s secretions and be aware of both the type and presence of humidification. Institutional protocols will determine which type of humidification is routinely used. Patients with bloody, copious, or viscous secretions with a HME may require increased systemic hydration or another type of humidification. The therapist should discuss this with the patient’s physician who may decide to increase the patient’s fluid intake or change the method of humidification.
Respiratory Muscle Weakness and Fatigue
It is generally accepted that patients requiring prolonged mechanical ventilation are subject to respiratory muscle weakness. This may be the result of inadequate neuromuscular function, atrophy of the respiratory muscles, paralysis due to administration of a neuromuscular blockade, electrolyte imbalance, inadequate nutritional support, fibers remodeling, oxidative stress, and/or steroid myopathy.60,126 Future research may assist physical therapists to determine the type and timing of inspiratory, and expiratory (including diaphragmatic) muscle strength training, which may assist the weaning process. There is also interest in the potential for newer modes of mechanical ventilation, which allow unassisted spontaneous breathing, for example, APRV, which may limit development of diaphragmatic weakness.
The degree of support offered to the respiratory muscles will vary with the chosen mode of mechanical ventilation. When pharmacological paralysis of the respiratory muscles is necessary to sustain life (see Chapter 8), and where a higher incidence of respiratory muscle atrophy is suspected, modes of ventilation such as volume control or pressure control are used. However, it is difficult to precisely determine the volume or pressure required. Offering more support than is necessary may contribute to respiratory muscle atrophy, whereas not providing enough support may lead to respiratory muscle fatigue. Modes utilized for weaning, such as PSV and SIMV, as well as APRV or similar modes of ventilation allow for greater activity of the respiratory muscles. If muscles fatigue, the recommended treatment is rest, which is usually accomplished by either increasing the number of breaths offered by the ventilator per minute or increasing the pressure associated with each breath or cycle. For example, increasing inspiratory pressure support increases tidal volume, decreases respiratory rate, decreases PaCO2, and decreases oxygen consumption and the work of the inspiratory muscles.49 Reducing PSV, a shorter inflation time and lower tidal volumes may decrease the work of breathing with patients requiring long-term ventilation.127 Pressure support ventilation is therefore a mode of ventilation commonly used to assist weaning and prevent respiratory muscle fatigue. Unfortunately, the precise intensity and duration of mechanical ventilation needed to rest the inspiratory muscles without causing fatigue is unknown and probably varies from patient to patient.
Weaning is not attempted until the cause of respiratory muscle fatigue is reversed. Further research will determine the optimal modes of ventilation required to minimize respiratory muscle weakness and fatigue and enhance the patient’s chances of successful liberation from a ventilator. Evidence suggesting that inspiratory muscle training (IMT) improves the strength and/or endurance of the respiratory muscles and enhances weaning from mechanical ventilation is limited to small sample sizes and case reports.128–131 Although the mechanism for rapid increases in maximal inspiratory pressure (MIP) with training is unknown, it is speculated that neural adaptations rather than muscle hypertrophy occur because the duration of training is probably insufficient to elicit microscopic changes in muscle fibers.132 Physical therapists usually do not provide inspiratory muscle training exercises to assist weaning unless the patient has a neuromuscular injury or disease affecting the inspiratory muscles, or the patient has not responded to prior attempts to discontinue mechanical ventilation.
Inspiratory muscle training devices are usually either pressure (nonlinear) or resistive (linear) threshold devices with an adapter to attach the device to the tracheal tube (Fig. 19-7). A nose clip is not required when the cuff on the tracheal tube is inflated. The patient is trained once to twice daily in a position that optimizes vital capacity (sitting for nontetraplegic patients with the head of bed elevated to at least 30 degrees) for up to 30 minutes.133Mechanically ventilated patients may better tolerate morning training sessions.
FIGURE 19-7 Inspiratory muscle trainers. (A) Resistive, linear device (B) Pressure, nonlinear device (C) Noseclip (Courtesy of Mary Massery, PT, DPT).
With pressure threshold devices the patient breathes though a set of adjustable orifices and the resistance is increased as the size of the orifice is decreased, however the resistance is dependent upon airflow which can be modulated by the patient adjusting their tidal volume and respiratory rate. Resistive threshold devices are preferred as they can be adjusted to a specific workload to provide a constant pressure through the entire inspiration, independent of airflow. Depending upon the device used, the resistance is adjusted by assessing the rate of perceived exertion (RPE) or MIP (linear devices) or the patient’s tolerance to the size of the orifice of the device (nonlinear devices). Based upon the limited evidence of the efficacy of inspiratory muscle training, it should be implemented on a case-by-case basis, when the physician has determined the patient is unable to wean by conventional methods, including maximal mobilization (page 601). Vital capacity, inspiratory mouth pressures, tidal volume, ventilator settings, resistance, training time, oxygen saturation, and heart rate should be monitored and recorded by the physical therapist during each training session and the resistance adjusted based upon the patient’s response to treatment.
Tetraplegic patients are notoriously difficult to wean due to a reduction in lung volumes and inability to cough effectively due to the loss of abdominal muscle strength133 (Fig. 19-8). Lesions above C5 result in impairment or loss of diaphragmatic muscle strength. Respiratory muscle strengthening exercises are therefore advocated. Inspiratory muscle training is often used however current evidence is inconclusive and primarily restricted to patients who have been weaned from the ventilator.134–138 Training is initiated in the supine position during CPAP or during interruptions from mechanical ventilation twice daily 5 to 7 days per week.
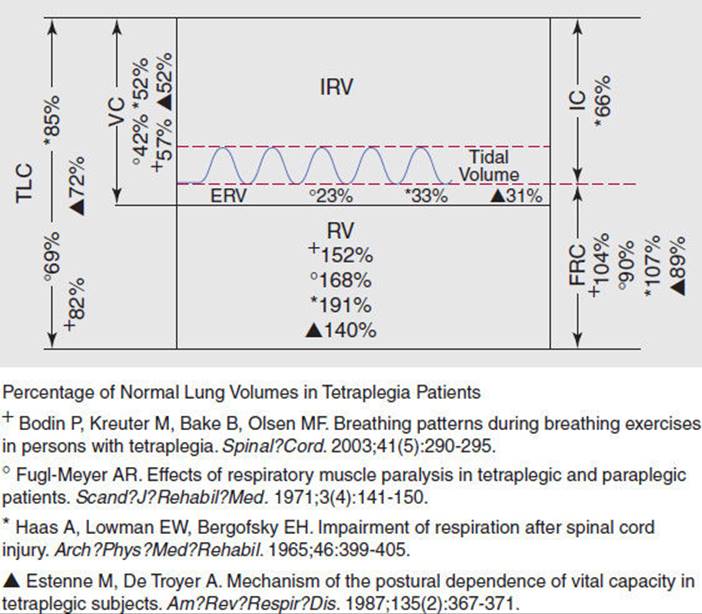
FIGURE 19-8 Lung volumes.
A second type of respiratory muscle strengthening, abdominal weight training has been used with tetraplegic patients during periods of spontaneous breathing both with and without CPAP.136,139 Theoretically, abdominal weight training, where cuff or dish weights are placed on the abdomen, would appear to provide more strength training than endurance training of the inspiratory muscles. During abdominal weight training the patient lies supine and the therapist determines a training load (Fig. 19-9). The inspiratory load can be determined by choosing the maximum weight with which a subject can breathe for 6 minutes, or by adding weights until the inspiratory capacity (grossly assessed using an incentive spirometer) decreases. Training protocols include a rate of 6 to 40 breaths per session or abdominal weight training for a predetermined time.
FIGURE 19-9 Abdominal weight training—tetraplegic patient.
It is advisable to reevaluate the inspiratory mouth pressures and the training load at least weekly during either IMT or abdominal weight training. Literature is scant regarding which training method is optimal during weaning from mechanical ventilation. Some tetraplegic patients prefer abdominal weight training over inspiratory muscle training stating it is easier to learn. Pulmonary secretions may clog the diaphragm of the IMT device and require frequent cleaning. Conversely, IMT is easily performed in the sitting position once the patient’s respiratory condition improves and can be performed more independently than abdominal weight training.
Other respiratory interventions advocated for the tetraplegic patient include strengthening the latissimus dorsi and the clavicular portion of the pectoralis major, glossopharyngeal breathing, summed breathing, incentive spirometry, and strengthening the cervical accessory muscles of respiration. Strengthening the clavicular portion of the pectorals major, the latissimus dorsi, and cervical accessory muscles should be included in the physical therapy regimen starting while the patient is mechanically ventilated, as these muscles have been shown to increase peak expiratory flow rates and thus are likely to enhance cough efficacy.140–142 Studies evaluating breathing exercises and glossopharyngeal breathing when the patient with tetraplegia is free from mechanical ventilation are covered in detail in Chapter 20. Objective assessment of the efficacy of breathing exercises requires electromyography and the insertion of gastric and esophageal balloons to measure gastric and subdiaphragmatic pressures and is therefore impractical in the ICU setting. However, examination of mouth pressures via manometers is simple and clinically useful, and can be taken when a patient has a tracheal tube (see Chapter 9).
Ventilator-Induced Lung Injury
Experimental studies have shown that excessive mechanical stresses associated with mechanical ventilation inflict injury on normal and acutely injured lungs. Repetitive application of excessive stress or strain to the lung by mechanical ventilation leads to diffuse alveolar damage, referred to as ventilator induced lung injury (VILI).143 VILI may result from one of four different mechanisms—Barotrauma, volutrauma, atelectrauma, biotrauma144,145—and is thought to result in the release of inflammatory mediators and proteinaceous edema entering the alveolar space. This is very similar to the macroscopic and microscopic lung injury observed in ARDS.
Barotrauma is induced by high ventilating pressures and can describe alveolar rupture and extra-alveolar air present in abnormal locations. Risk factors thought to predispose a patient to ventilator-associated barotrauma include diminished lung integrity from necrotizing pneumonia, aspiration, alveolar distension from high tidal volumes, high airway pressures, and possibly PEEP. Extra-alveolar air can be present in many locations including the pleural space, soft tissue, abdominal cavity, and pericardium. Extra-alveolar air occurs after alveolar rupture as air dissects along the vascular sheaths back to the mediastinum. It may then dissect the pleural space and cause a pneumothorax. When the air diffuses into the soft tissues, it is known as subcutaneous emphysema. Air in the abdominal cavity is termed pneumoperitoneum, while pneumopericardium refers to air in the pericardial space.
Subcutaneous emphysema is a frequent complication of barotrauma that has implications for physical therapy interventions. Subcutaneous emphysema is diagnosed by palpating crepitation in the neck, face, chest, axilla, or abdomen. The crepitus of subcutaneous emphysema feels like crisped rice cereal. The physical therapist may be the first health care practitioner to pick up this clinical sign during an examination or treatment intervention. Subcutaneous emphysema can be an important symptom as it may be a precursor to a pneumothorax, particularly in a patient who has elevated airway pressures and diminished or absent breath sounds in one lung. If the patient has not had a recent chest radiograph and been evaluated for pneumothoraces the physician will usually order a chest radiograph. Once a pneumothorax has been ruled out or treated, physical therapy interventions including mobility, manual techniques for secretion clearance and cough assistance techniques can be performed if clinically indicated. A sudden elevation in airway pressures and/or drop in tidal volume and decreased breath sounds in the absence of subcutaneous emphysema should also be brought to the physician’s attention, as they may be indicative of a pneumothorax.
Volutrauma describes damage caused by overdistension of the alveoli occurring as a result of high volume ventilation. Overdistension of the alveoli results in diffuse alveolar damage, pulmonary edema, increased fluid filtration, epithelial permeability, and microvascular permeability and mostly affects healthy or highly compliant alveoli.144 Atelectrauma is lung injury related to repetitive opening and closing of alveoli, and is theoretically prevented by the use of PEEP to prevent derecruitment or collapsing of alveoli.33 Biotrauma describes inflammatory injury to the alveoli due to cytokine release in response to mechanical injury.144 Ventilation at high tidal volumes, high pressures, or without peep can exacerbate VILI, and recent studies have demonstrated a reduced mortality with mechanical ventilation using low tidal volumes.29,146,147 Plateau pressures should be monitored and ideally not exceed 30 cmH2O pressure. The literature is inconclusive as to whether the tidal volume or plateau pressure is the best target for therapeutic interventions.148 It is recommended that plateau pressure should be monitored during mechanical ventilation. Plateau pressure is a more accurate indicator of lung compliance than peak inspiratory pressure. As lung compliance falls, plateau pressure rises. Peak inspiratory pressures are influenced by decreasing lung compliance, but also influenced by any increase in airway resistance. Plateau pressures in the first 48 hours of ALI may reflect the severity of disease and mortality.149
Physical therapists should be aware that ventilatory management that emphasizes lower alveolar pressures and avoids hyperinflation may result in an intentional elevation of PaCO2, which is usually well tolerated in critically ill patients, as long as the increases are gradual.150
COMPLICATIONS OF IMMOBILITY AND ICU INTERVENTIONS
The following quotation made by Dr. Henry Bendixon in 1974 remains true today and is worth reiterating.151
The importance of immobilization has been part of medicine’s body of knowledge for many, many, years and can be accepted as fact. As with any fact, if not restated from time to time it tends to be forgotten. Our advanced knowledge and technology by themselves cannot save the patient. Instead, something so simple as turning the patient … at least hourly, may make the difference between living and dying for the intensive care patient.
Patients receiving mechanical ventilation are at risk for all the complications associated with immobility. The hazards of immobility alter the normal physiological function of most, if not all body systems. Interventions to prevent these complications are often overlooked, due to medical prioritization of the life-saving aspects of the patient’s plan of care. Physical therapists working in the critical care unit often bring this necessary perspective of patient management to the ICU team. Once aware of the principles of mechanical ventilation, and the life-sustaining equipment these patients require, the physical therapist can develop a treatment plan to enhance respiratory, cardiovascular, neurological, and musculoskeletal function. Attention to respiratory function and mobility may decrease the risk of integumentary complications. The medical team is usually receptive to the physical therapist attending rounds and taking an active role as part of the interdisciplinary team.
EFFECTS OF BODY POSITION AND THE USE OF ABDOMINAL BINDERS FOR PATIENTS WITH TETRAPLEGIA
Changes in respiratory function occur during the initial period of adjustment from the sitting to the supine position. In normal subjects, total lung capacity, vital capacity, functional residual capacity, residual volume, and forced expiratory volume may all be reduced simply by lying down.152–154 A reduction in rib cage compliance, decreased anterio-posterior diameter, and increased lateral diameter of the rib cage are also noted in the supine compared to the sitting position. Alveolar size decreases, and small airway closure increases in dependent lung zones with a decrease in PaO2.153 The normal subject may have a 7.5% decrease in vital capacity when assuming the supine position.155,156 Similarly, a reduction in the FRC occurs consistently in changing from the sitting to supine position.157 Therefore, patients with normal innervation of the respiratory muscles usually tolerate weaning and functional activities better while sitting out of bed or in bed with the head of the bed elevated.
The tetraplegic patient may also have dramatic changes in respiratory function when moving from the upright or sitting position to the supine position. However, these changes are quite different from those noted in patients who have normal innervation of the respiratory muscles (Fig. 19-8). With tetraplegia, inspiratory muscle weakness results in a 15% to 31% reduction in total lung capacity, 48% to 58% loss of vital capacity, and a 34% loss of inspiratory capacity. Up to a 77% reduction in expiratory reserve volume and a 50% increase in residual volume are attributed to a lack of expiratory muscle function.158–161
The tetraplegic patient has a decreased tidal volume, vital capacity, inspiratory capacity, and decreased ventilation in the lung bases when assuming the sitting position from the supine position.156,160 With tetraplegia an increase in vital capacity has also been noted in the 20-degree head-down position with supine positioning.162 Therefore, the tetraplegic patient may better tolerate weaning trials, respiratory muscle strengthening exercises, and early rehabilitative interventions, while supine. Vital capacity may be greater during postural drainage positioning for the lower and middle lobes than in the upright position.
Abdominal binders are advocated for the spontaneously breathing or weaning tetraplegic patient in order to align the abdominal contents under the diaphragm, thus improving the length–tension relationship of the diaphragm. The effect of an abdominal binder is most evident in the seated position. Increases in inspiratory capacity, vital capacity, tidal volume, total lung capacity, and decreases in functional residual capacity have been documented.163–165 These improved lung volumes assist the tetraplegic patient’s ability to cough. Maximum expiratory airflow increases with an abdominal binder when manual lung inflation and chest compressions are utilized as an assistive cough maneuver.166
Physical therapists working with mechanically ventilated tetraplegic patients should recommend the use of an abdominal binder once the patient is spontaneously breathing during part of the ventilatory cycle, and prior to raising the head of the bed to initiate sitting. Elastic binders are recommended and can usually be obtained from the hospital medical supply department. Elastic binders usually come with 3 to 4 circumferential elastic panels (Fig. 19-10). The therapist should measure the distance from a few inches below the xiphoid process to the level of the anterior superior iliac spine (ASIS). Clinically, it has been noted that skin breakdown is greater when the binder extends below the ASIS. If the elastic binder is too wide, one or two panels can be cut away. Binders that are too tight or extend too high on the thorax may hinder inspiration. Although binders are recommended as a standard of care in acute rehabilitation the overall benefits are not known. A decrease in functional residual capacity with the use of a binder has the potential to impair gas exchange and abdominal strapping alone is unlikely to improve expiratory flow rates sufficient to improve the efficiency of cough in tetraplegic patients 6 to 200 months after injury.167,168 However, abdominal binders appear to improve vital capacity and enhance the patient’s ability to cough by providing abdominal pressure, which may be assisted by the clinician during expiration.
FIGURE 19-10 Abdominal binder.
CLINICAL CORRELATE
If the therapist is in doubt as to whether a binder is beneficial for a particular patient, vital capacity measurements can be taken with a respirometer both with and without the binder to determine if the binder is advantageous.
Some advocate that abdominal binders be worn until a plateau in pulmonary function or cough effectiveness/assistance is observed. When patients are observed to be stable and no further improvement in pulmonary function or cough is noted, abdominal binders are frequently removed. However, chronic abdominal binder use after spinal cord injury has been suggested to maintain and possibly improve pulmonary function, cardiovascular status, and posture. This will be presented in the following chapter where the patient in the case study of this chapter is progressed to a subacute facility and the physical therapist must consider the chronic ramifications of spinal cord injury.
Central Nervous System
Cognitive Impairment
Cognitive function can be dramatically altered by bedrest and particularly in mechanically ventilated ICU patients. Seventy-eight percent of ICU survivors of ARDS may have cognitive impairment at one year, which is severely underestimated by rehabilitation professionals.169,170 College students placed on bedrest were noted to have slowing of electroencephalogram activity, emotional and behavioral changes, decreased psychomotor performance, and changes in sleep patterns.171,172 Decreased psychomotor performance in areas of intellect, perception, and coordination accompanied by visual and auditory changes have also been noted.173
Once mechanically ventilated, a patient is at a high risk for delirium due to acute multisystem illness, comorbidities, and medications.174 Delirium is defined as an acute onset of impaired cognitive functioning which impacts the patient’s ability to receive, process, store, and recall information.174 Delirium can result in increased morbidity, including more time to liberate the patient from mechanical ventilation, higher rates of nosocomial pneumonia, delayed functional recovery, and a prolonged hospital stay.175 ICU patients also experience a loss of circadian light patterns, electrolyte imbalances, fever, and hypoxia, and are subjected to monitoring alarms and numerous caregivers approaching the bedside to provide interventions around the clock, all of which disrupt their cognitive function.
It is not uncommon for mechanically ventilated patients to receive various forms of analgesia, sedation, and even neuromuscular blockade for surgical procedures and management while in the ICU. Sedation and delirium can have a dramatic impact on the ability of the patient to participate with physical therapy interventions. Recent literature recommends at least daily monitoring of sedation and delirium by nursing and other health care providers.176 The Richmond Agitation—Sedation Scale (RAAS) assists physicians and nurses in determining appropriate doses of sedative and analgesic medications.177 The Confusion Assessment Method for ICU patients (CAM-ICU) alerts the clinician to disorganized thinking, inattention, and altered level of consciousness, and is used to determine delirium.174
Most physical therapists are familiar with and can utilize the Glasgow Coma Scale (GCS), a 3- to 15-point scale rating eye opening, verbal, and motor responses to determine level of consciousness.178 Despite its widespread use for neurological assessment in the ICU setting, it was developed for assessment of level of consciousness after head injury, and its use has not been validated in the general ICU population. Unfortunately, few physical therapists are aware of the RAAS and ICU–CAM, two highly reliable and valid tools for ICU patients that take 1 to 2 minutes to administer and are used to assess the patient’s level of sedation and delerium.174,176,179 It is therefore recommended that physical therapists working in the ICU learn the meaning of the scores and how to administer these tools to enhance communication with the medical team. Improved teamwork may lead to a change in sedation or analgesia and the physical therapist may gain a better understanding of the patient’s mental status, therefore optimizing the patient’s ability to participate in therapy.
The RAAS is a 10-point scale with four positive levels for agitation and five negative levels. The RAAS separates the patient’s responses to verbal and physical stimulation. The ICU–CAM is recommended when a patient’s RAAS score indicates the patient has some response to voice. Each visit the therapist should also orient the patient to place and time, work with the nursing staff to display pictures of family members, and procure audiotapes to assist with cognitive orientation. A calendar noting the day displayed within the patient’s line of vision is helpful. The therapist should speak to the patient throughout the treatment session and never underestimate the patient’s ability to understand the activity and environment around him or her. Upright positioning out of bed is recommended to enhance the patient’s orientation to, and interaction with, their environment, as well as minimize the other associated complications of immobility.
Many clinicians (physicians, nurses, and therapists) currently defer getting out of bed for patients with a low level of consciousness, due to a perceived lack of ability to participate or benefit. Sitting in a chair out of bed, the patient may be stimulated to an increased level of consciousness, and demonstrate greater than expected participation, when attempts are made to begin mobility within days of admission to the ICU. It is the clinical experience of the authors that patients often demonstrate automatic responses to rolling, moving from supine to sitting, sitting to standing and transferring, despite the inability to follow commands or effectively participate in activities while supine in bed. Activities out of bed should not be discounted solely due to a patient’s perceived inability to participate with physical therapy interventions.
Cardiovascular
Cardiac
While mechanically ventilated, most patients spend the majority of their day in bed. It is therefore assumed that the same risks associated with immobility that occur within the cardiovascular system with bedrest occur with mechanically ventilated patients. Decreased blood volume, decreased plasma and red blood cell mass, decreased hemoglobin concentration, increased basal and maximal heart rate, decreased maximum oxygen uptake, and a decreased transverse diameter of the heart have all been reported.180,181 Critically ill patients without previous cardiac disease may also be susceptible to reversible myocardial dysfunction including systolic dysfunction, segmental contractility disturbances, and electrocardiographic changes.182 Immobilized patients develop a decreased ability to perform aerobic work. Oxygen uptake during exercise decreases, whereas oxygen debt and blood lactate concentrations increase. Pharmacological agents are frequently required to maximize cardiac output. Detailed descriptions of the cardiac risks of immobility are described in Chapter 14.
Orthostatic or postural hypotension is the term used to describe a decrease in blood pressure upon assuming the erect position accompanied by symptoms of dizziness or syncope. Circulating blood volume is reduced with bed rest and vascular tone is decreased. Medical management will attempt to maximize circulating blood volume; however, sitting the patient up may result in decreased cardiac filling pressures and decreased cardiac output because of vasodilation of the lower extremity blood vessels. The resulting decreased blood flow to the brain may result in mild cerebral hypoxemia. The alert, oriented, and spontaneously breathing patient can report to the caretaker that they are feeling “lightheaded” or “dizzy” after sitting up. The mechanically ventilated patient usually cannot speak because of intubation and may have cognitive impairment, muscular weakness, or lines and tubes making it difficult to display discomfort with body language.
CLINICAL CORRELATE
Although augmentative communication devices may assist the patient with speaking, the therapist should be able to identify subtle changes in the patient’s body language, report of symptoms, cognitive status, and changes in vital signs (heart rate, blood pressure, and oxygen saturation) from baseline when placing the patient in the upright position. Pallor and diaphoresis may also be present.
The therapist should be aware of whether the patient is taking any medications such as β-blockers that blunt the body’s normal response to activity (see Chapter 8). The most common response to upright positioning is a drop in blood pressure with a compensatory increase in heart rate. Oxygen saturation may drop and may prompt the physician to increase the FiO2 delivered by the ventilator.
The patient with an acute cervical spinal cord injury presents an additional challenge for the physical therapist. Rapid changes in body position for the acute tetraplegic patient in spinal shock may have a more dramatic adverse affect on cardiac function. Head elevation of greater than 20 degrees may cause a sudden decrease in cardiac filling pressures, resulting in a decrease in cardiac output, and even cardiac arrest. Similarly, sudden head-down positioning, as is necessary for postural drainage of the middle and lower lobes, may cause a rise in cardiac filling pressures. Because of the loss of sympathetic cardiac innervation associated with lesions above T1, the steep head-down position may precipitate acute myocardial failure with pulmonary edema. Therefore, in the early stages of acute tetraplegia, head elevation and head-down positioning should be performed with careful monitoring of arterial and venous pressures. Most mechanically ventilated tetraplegic patients tolerate head-down positioning when experienced personnel move the patient cautiously.
The lower extremities of a patient with acute tetraplegia should be wrapped with elastic bandages or elastic stockings that cover the entire lower extremity prior to sitting the patient upright, particularly after prolonged bedrest. Compression of the lower extremity veins will assist the return of venous blood to the heart. The benefits of using elastic bandages versus compression stockings are not known. However, some clinicians have noted that patients who do not tolerate upright position changes with elastic stockings are better able to tolerate these position changes with elastic bandages. Bandages are more time-consuming to don and require special training of the personnel who apply them. They should be wrapped in a spiral or figure-eight fashion. The greatest tension is applied distally over the foot and gradually reduced as the extremity is wrapped to the groin. If applied incorrectly circumferential pressure may impair blood flow and increase edema below the circumferential wrap. An abdominal binder is also recommended for patients with acute tetraplegia to prevent pooling of blood in the abdominal cavity.
Venous Thromboembolic Disease
Medical and surgical patients have a 10% to 30% incidence of deep venous thrombosis (DVT) within the first week of ICU admission.183 The risk of DVT with acute spinal cord injury is extremely high at 60% to 80%.184
Venous thrombi are intravascular deposits composed mainly of fibrin and red blood cells with a variable platelet and leukocyte component, usually located in the deep veins. It is important that they are recognized early and treated effectively. DVTs occur in regions of slow or disturbed blood flow (vascular stasis) in the lower extremity, or with loss of vessel wall integrity as a result of trauma or disease. The majority are confined to the calf and are asymptomatic, small, and not associated with major complications.185 However, venous thrombi, particularly in the proximal veins (popliteal, femoral, iliofemoral), may break off and form a pulmonary embolus (PE) which is life threatening.
Mechanical ventilation and the associated immobility causing venous stasis put the patient at high risk for DVT and subsequent PE. Cancer, autoimmune disorders, lower extremity fractures, trauma or surgery to the veins, congestive heart failure, neurological disorders, lack of skeletal muscle activity, and postoperative sepsis all increase the risk of DVT and PE. Patients on bedrest have impaired venous return from the lower extremities and abnormal coagulation factors. It is thought that the decreased circulating blood volume that occurs with immobility is due more to the loss of plasma than to a decrease in red blood cell mass. With loss of endothelial lining cells along a vein, platelets attach to the underlying collagen and release factors that cause more platelets to adhere and aggregate to underlying collagen. A fibrin mesh attaches to the platelets and eventually traps erythrocytes and leucocytes. The result is an intravascular clot or thrombus.185
Patients with a decreased level of consciousness and sensory deficits may not perceive pain or tenderness along the track of the deep venous system, making DVT even more difficult to diagnose. More than 50% of DVTs occur without signs or symptoms.185 When a physical therapist suspects a patient may have a DVT due to the presence of new clinical signs, the physician should be notified and further diagnostic testing may be necessary. Scoring for probability of deep vein thrombosis may be accomplished by use of the scoring method as described by Anand et al.186Ultrasonography and venography are most commonly used to diagnose DVT in critically ill patients. Once a diagnosis is made the therapist should be aware of whether the DVT is from the calf, which is most common, or from a more proximal vein such as the popliteal, femoral, iliac, or inferior vena cava where there is a higher incidence of complications including pulmonary embolus.
Anticoagulation therapy is effective in the prevention of extension, embolization, and recurrence of DVT. However, although anticoagulation therapy prevents enlargement of the thrombus and allows for further attachment of the thrombus to the vessel wall to prevent a pulmonary embolus, it does not dissolve the DVT.187 The treatment of choice for acute DVT, PE, or while awaiting the results of diagnostic tests when the clinician has a high index of suspicion for thromboembolitic disease is anticoagulation therapy. Options include subcutaneous low-molecular-weight heparin (Lovenox, Fragmin), monitored IV or subcutaneous unfractionated heparin (Heparin Sodium), or fondaparinux (Arixtra) a Factor Xa inhibitor in the coagulation cascade.187 A treatment regimen is recommended for most ICU patients at risk of DVT, and for most major trauma and spinal cord injury patients.184,188,189 The efficacy of unfractionated heparin is measured by clotting times such as the activated partial thromboplastin time (APTT or PTT). Because of the mechanism of action for the low-molecular-weight heparins and fondaparinux, monitoring bleeding times is not required. The International Normalized Ratio for Prothrombin time (PT INR) is used to monitor warfarin (Coumadin) (Table 19-12).
TABLE 19-12 Laboratory Tests to Monitor Anticoagulant Therapy
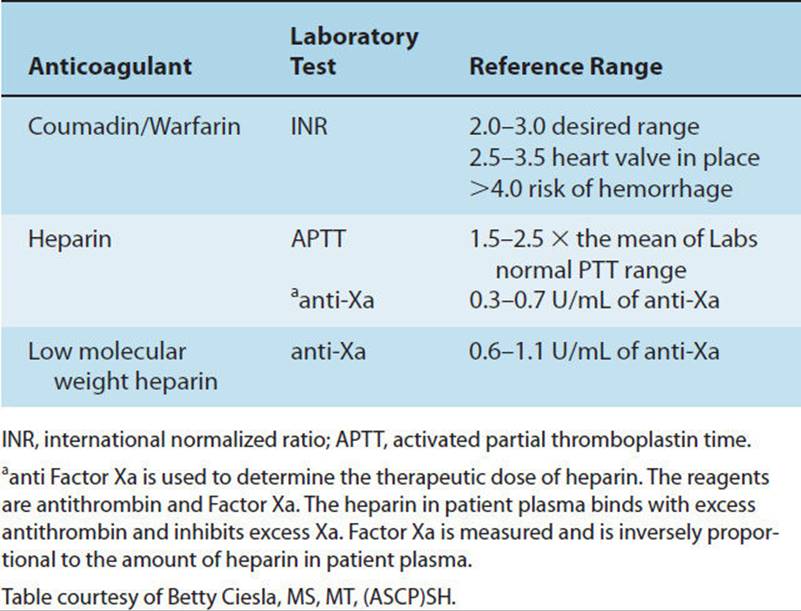
When anticoagulation is contraindicated because of a high risk of bleeding, elastic compression stockings (CS) with a pressure gradient of 30 to 40 mmHg and/or intermittent pneumatic compression (IPC) is recommended until pharmacologic thromboprophylaxis can be started. The advantages of IPC and CS are that they do not increase the risk of bleeding, may reduce leg swelling, and may enhance the effectiveness of anticoagulant thromboprophylaxis. They also have a greater effect in reducing distal versus proximal DVT. However, they are generally less efficacious than anticoagulation as a prophylactic measure, staff compliance is low and there are no standards for size, pressure, or physiologic features.184,190 Studies have shown that CS may reduce postthrombotic sequelae by 50%, and therefore should be worn for at least 2 years in patients who have had a symptomatic proximal DVT.191–193
Placing a filter in the inferior vena cava is a treatment modality for DVT and PE and is used for PE prophylaxis. Currently vena cava filters are not recommended except when a patient has a PE or acute proximal DVT and anticoagulant therapy is not indicated because of the risk of bleeding.191,194 Anticoagulant therapy is recommended once the risk of bleeding subsides.191 Physical therapists working in the ICU may see filters being placed at the bedside or in the interventional radiology department.195 The physical therapist should be aware of any postprocedure precautions, such as immobilization of the hip for several hours (filters are usually placed via the femoral vein). However, by the day following the procedure routine physical therapy interventions such as range-of-motion exercises and bed-chair transfers can usually be resumed. Once the filter is in place there is no contraindication to range-of-motion exercises to the limb with the DVT.
The medical recommendations for mobilizing patients with a DVT have dramatically changed over the past several years. Historically, bed rest was advocated. It is now generally accepted that bed rest is associated with a slower resolution of leg pain and swelling from a DVT, and does not lower the risk of PE.187 Early ambulation and CS are recommended for patients with acute DVT and may provide even faster improvement with less pain, swelling, and minimize the extension of a DVT.187,189,196–199 The physical therapist should treat the mechanically ventilated patient with a DVT, the same as a nonmechanically ventilated patient with a DVT or suspected DVT. If the clinical examination and chart review reveal that the patient can be mobilized and transferred out of bed, the therapist removes the IPC device, applies elastic stockings or elastic bandages to the lower extremities if the patient is having symptoms of orthostatic hypotension, and begins mobility training activities. For patients in whom an IPC device is prescribed the device is reapplied after transfer training or ambulation. A patient with an untreated DVT in one extremity can receive secretion removal techniques and range-of-motion exercises to the noninvolved extremities.
A patient with a DVT who develops unexplained breathlessness, desaturation, hemoptysis, pleuritic pain, arrhythmia, or fever is suspected of having developed a PE.200 However, the mechanically ventilated patient may not demonstrate breathlessness, may have hemoptysis from tracheal irritation with suctioning or a lung contusion, and may have arrhythmias or fever unrelated to a PE. Therefore, the diagnosis of PE is more difficult. The nurse, physical therapist, or respiratory therapist may note a critical drop in PaO2 or oxygen saturation that leads the physician to suspect a PE. The most frequent diagnostic testing for PE is serial computed tomography, followed by ventilation/perfusion scans, or pulmonary angiography.201 A PE usually does not preclude airway clearance interventions with manual techniques. To date there are no documented cases of PE dislodgement, with manual chest physical therapy techniques, coughing, or suctioning. Should an embolus be dislodged, it would move peripherally into the pulmonary vasculature, where there is less rather than more occlusion of pulmonary blood flow. Anatomically it is impossible for a pulmonary embolus to cause a cerebral vascular accident or occlusion of an extremity vessel; yet this is a common concern of many practicing physical therapists.
The patient with acute tetraplegia has both a high risk of pulmonary embolus and a high risk for hypoxemia due to pulmonary secretion retention. Figure 19-11 demonstrates a ventilation perfusion scan of a patient with tetraplegia suspected of having developed a pulmonary embolus accompanied by a drop of PaO2 to 50 torr. Following the scan, intensive chest physical therapy was instituted and the patient’s oxygenation status significantly improved. In this case the patient was ruled out for a PE and was diagnosed with a ventilation versus a perfusion abnormality for which physical therapy was indicated for secretion clearance. When in doubt as to the appropriateness of secretion clearance interventions for a suspected or confirmed PE, the therapist should discuss their concerns with the referral source.
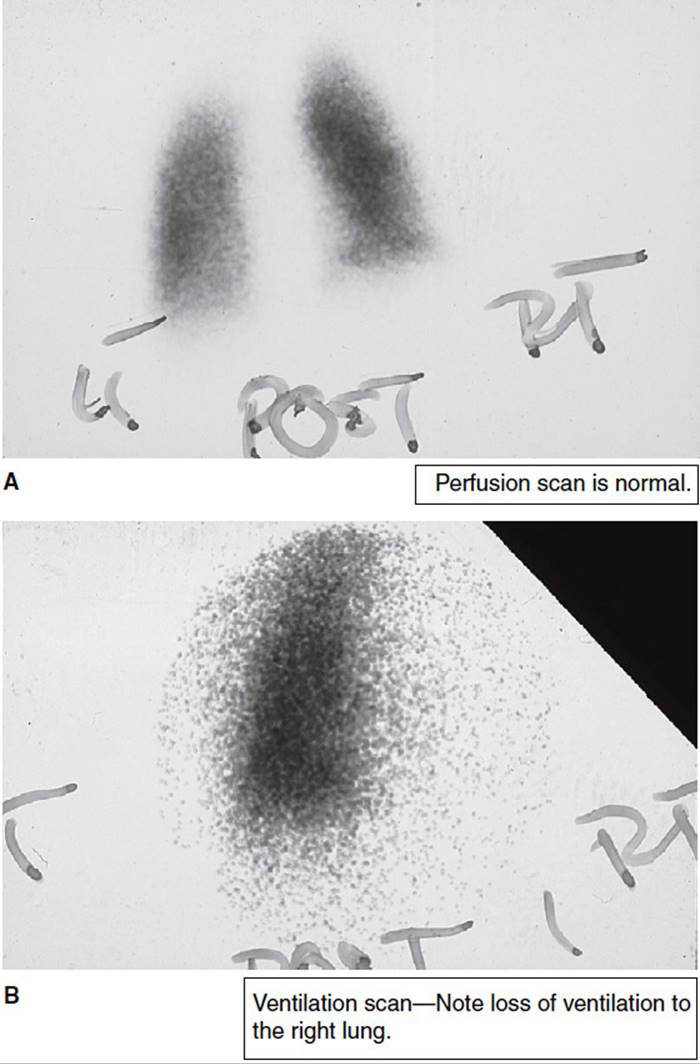
FIGURE 19-11 Ventilation–perfusion scans.
Neuro-musculoskeletal
The mechanically ventilated patient is subject to the same risks of contractures, muscle atrophy, and weakness as any immobilized, malnourished, or deconditioned patient. Because of the ventilator and the plethora of lines and tubes required for physiological monitoring, there may be an even greater risk of muscle wasting and limitations to joint mobility. Fourteen days of bedrest alone results in a 4.1% decrease in lean thigh mass, with a 15% to 30% decrease in quadriceps strength after 6 weeks.202,203 Significant decrements in bone mineral density occur which may not be fully reversed after 6 months of normal weightbearing activities.204 Heterotopic ossification, which increases the risk of contracture formation, has been documented in a variety of ICU populations, including patients with traumatic brain and/or spinal cord injuries, thermal burns, multitrauma without head trauma or simply long-term sedation.205–209Sedation and medical paralysis may be necessary to manage respiratory failure, placing some patients at an even higher risk of neuromusculoskeletal dysfunction.
Contractures
While research looking specifically at the relationship between contractures and mechanical ventilation is limited, there is some evidence that 34% of patients with ICU stays of greater than 14 days develop a functionally significant contracture in at least one joint at the time of ICU discharge, with 23% of the contractures present at the time of discharge to home.210 In addition, clinical experience has noted contractures in the cervical spine, most likely due to the limitation of spontaneous cervical rotation by the ventilator tubing, and cervical extension contractures leading to a forward head from prolonged supine positioning with the head on pillows.
The incidence of contractures has been more clearly documented with acute SCI patients (who are usually admitted to an ICU) and may require mechanical ventilation. A study of 181 SCI patients demonstrated that those who are not admitted to an SCI center with early physical therapy developed more contractures.211 Fifty-one percent of elbows examined in patients with complete C5 or C6 traumatic spinal cord injuries demonstrated flexion contractures. Sixteen percent of the upper extremities had elbow flexion contractures of 50 degrees or greater.212 A patient with C6 tetraplegia who develops a 25-degree elbow flexion contracture frequently loses one critical functional neurological level, resulting in the mobility of a patient with C5 tetraplegia, who requires the physical assistance of another person for most functional mobility.213 In addition, delaying shoulder range-of-motion exercises for 2 weeks is associated with a higher incidence of shoulder pain in the SCI population.
To preserve cervical ROM, the physical therapist can show the nursing staff how to position and maintain the head in the neutral position while the patient is supine. Outdated IV bags or rolled up towels placed on either side of the face will help to maintain the head in the neutral position. Active, active-assistive, and passive cervical ROM exercises (supine, side-lying, and prone as indicated) are also recommended. The physical therapist and nursing staff may move the ventilator and ventilator tubing to provide enough slack for the patient to actively rotate the cervical spine. The alert patient should be periodically reminded to actively turn their head both toward and away from the ventilator while staff stabilizes the tubing. Alert and oriented patients who watch television may benefit from having the television placed on the side of the bed opposite the ventilator tubing. In rare cases, when a severe limitation of cervical motion rotation has developed, the ventilator can be moved to the other side of the bed (space permitting) to encourage spontaneous motion toward the most involved side.
The ICU physical therapist has a major role in preventing extremity contractures. Hip and knee flexion contractures, limited hip internal rotation, loss of ankle dorsiflexion; and limitations in scapular abduction, shoulder flexion, abduction, and external rotation are frequently noted in mechanically ventilated patients. ICU patients are at risk for losing motion in joints adjacent to lines and tubes due to discomfort and reluctance to spontaneously move the joint/extremity. No accidental removals or adverse effects have been found with physical therapy interventions for patients with arterial catheters.4,5
Many mechanically ventilated patients in acute respiratory failure present with subclavian and femoral lines. Central lines inserted in the subclavian vein with tubing extending perpendicular to the thorax contribute to decreased scapular mobility and limited shoulder motion. While some institutions advocate limiting shoulder ROM in the presence of subclavian lines, it should be noted that full-shoulder ROM can usually be accomplished without compromising the line. These lines are sutured in place and confirmed radiologically. The therapist carefully monitors the line for patency as the shoulder is moved to determine limitations in specific joint motions. When a central line is limiting shoulder motion, the therapist can speak with the physician and request that it be repositioned so that it does not cross the shoulder joint at the time of the next line change. If the line cannot be repositioned, side-to-side turning may pose greater challenges, requiring time-consuming positioning utilizing towels or outdated IV bags under the axilla to minimize the risk of occluding the line.
Therapists are sometimes fearful of moving the hip joint of a patient who has a femoral line. However, when therapists are working closely with the medical team this is usually not a problem, and the risks and benefits of hip flexion exercises and functional activities which incorporate hip flexion can be discussed on a case-by-case basis. Hip-extension contractures are rare; therefore, aggressive hip flexion exercises are usually not necessary. The presence of a femoral line does not preclude hip extension, abduction, adduction, and internal and external rotation unless the patient has an unusual acute condition where ROM exercises may increase bleeding at the site or the line is not sutured in properly. Transfers out of bed and even ambulation should not be aborted solely because a patient has a femoral line if it is adequately sutured in place. Early mobility including ambulation has not been shown to adversely affect or dislodge arterial lines.
Many mechanically ventilated patients have chest tubes. These tubes are also sutured in place, and full shoulder ROM is not contraindicated, though care should be taken to assess the chest tube for drainage and possible air leaks both prior to and after intervention. Any change may indicate that the tube is not securely placed, and may need evaluation. Chest tubes placed for hemothorax or pneumothorax may well be accompanied by rib fractures, and early intervention to encourage full shoulder mobility and encourage deep breathing will reduce the risk of complications.
When possible, prone positioning is an ideal intervention to prevent hip flexion and shoulder internal rotation contractures. The prone or side-lying position facilitates hip extension and knee flexion exercises (Fig. 19-4).
In summary, contracture prevention for the mechanically ventilated patient should include the following: side-to-side turning to at least 90 degrees (a foam wedge or roll of several sheets or blankets is effective in keeping patients turned), ROM exercises, mobilization from supine to sit, bed to chair, and sitting the patient on the edge of the bed as soon as tolerated (Table 19-13). Early ambulation is also recommended (Fig. 19-12).
TABLE 19-13 Physical Therapy Interventions That May Be Utilized During Mechanical Ventilation to Prevent Contractures
Side-to-side turning to 90 degrees or greater (Place a roll or foam wedge behind the patient’s back, or turn patient onto the roll, so anterior trunk rests on the roll)
Dependent hip in neutral position
Uppermost shoulder flexed, scapula protracted
Move TV or ventilator side to side (increase rotation of the cervical spine)
Maintain cervical spine in neutral position while supine (place small towel rolls or expired IV bags on either side of the patient’s neck)
Bed mobility exercises
Extremity therapeutic exercises
Splinting, serial casting
Sitting balance activities on edge of bed
Transfer patient to upright sitting position out of bed
Wheelchair propulsion or ambulation using a manual resuscitator bag or portable ventilator
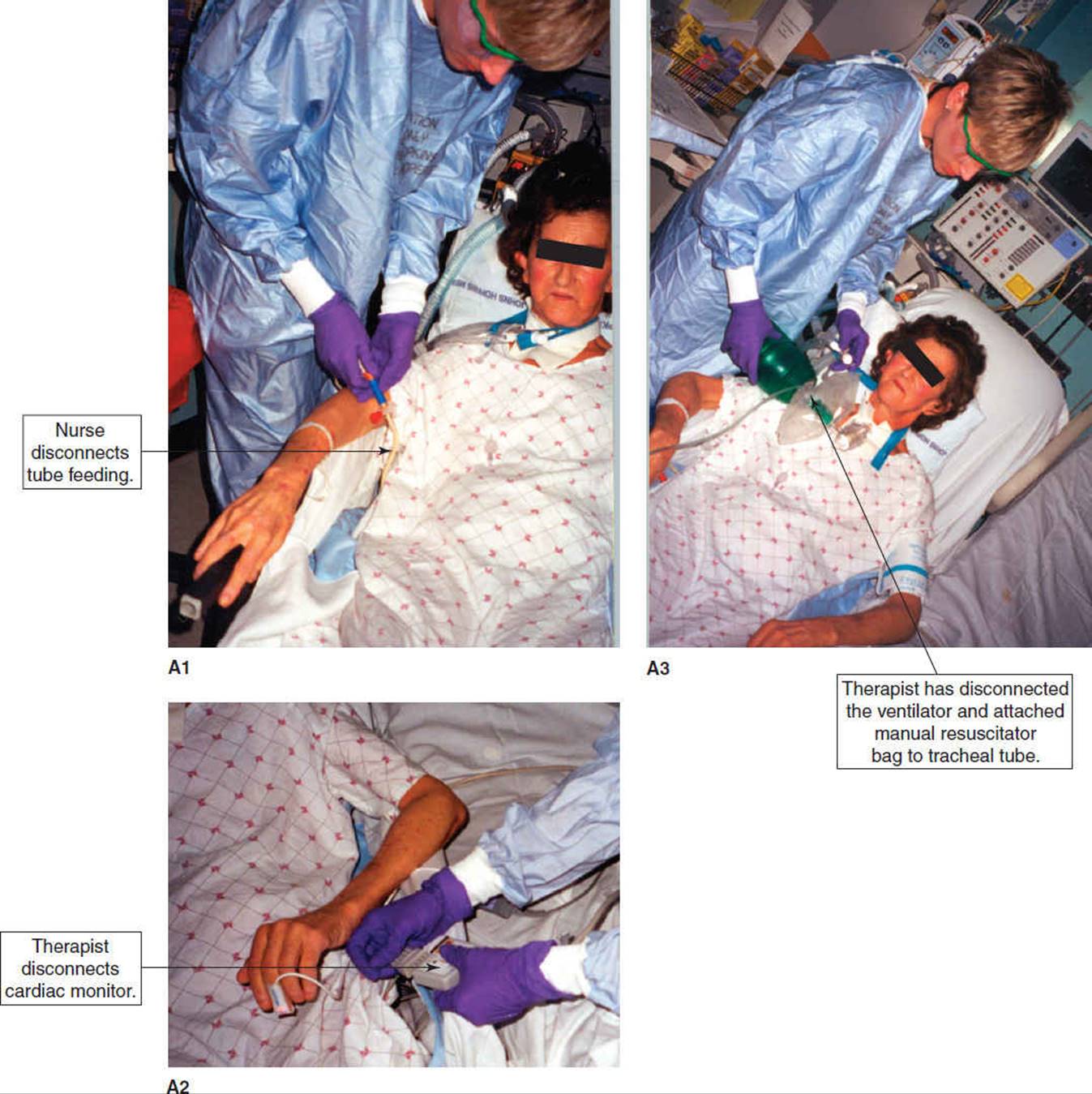
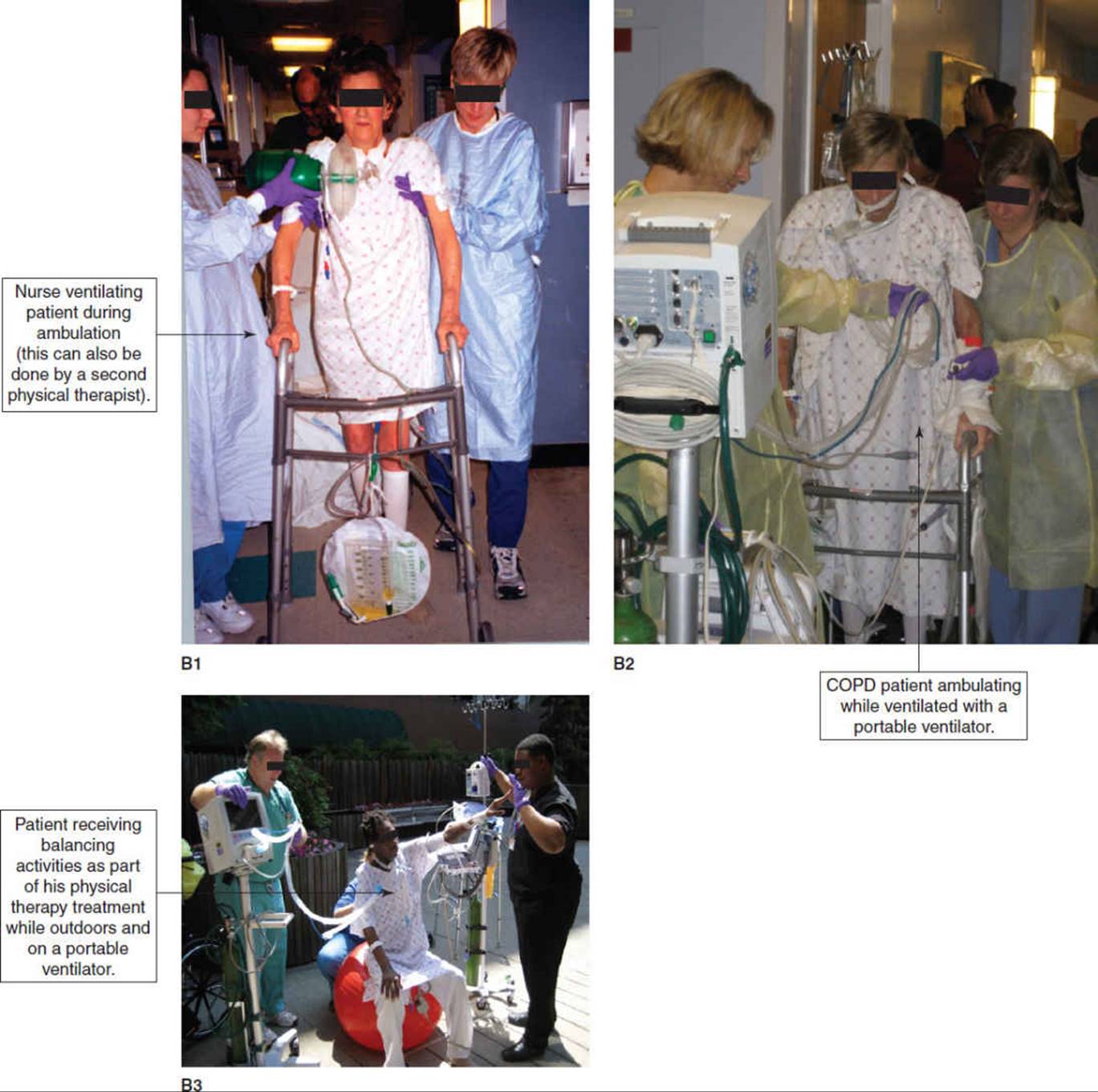
FIGURE 19-12 Examples of mobilizing mechanically ventilated patients. (A) Preparation: A1. Nurse disconnects tube feeding. A2. Therapist disconnects cardiac monitor. A3. Therapist has disconnected the ventilator and attached manual resuscitator bag to tracheal tube.
Splinting or serial casting (for patients who demonstrate increased muscle tone) can be applied to prevent contractures while patients are receiving mechanical ventilation. Short-acting pharmacological agents may be necessary to temporarily reduce spasticity for optimal joint positioning prior to serial casting (see Chapter 8). These drugs may temporarily decrease the patient’s respiratory function necessitating an increase in ventilator support just prior to, during, and for a short time following the casting procedure.
CLINICAL CORRELATE
Early motion is recommended for most mechanically ventilated patients, particularly those with SCI or orthopedic injuries stabilized with internal and external fixation devices. The therapist should consult with the orthopedic or neurosurgeon for clarification of any precautions or restrictions to range-of-motion exercises, sitting upright, or weight bearing and instruct the nursing staff and family in ROM exercises that may enhance physical therapy sessions.
Critical Illness Neuromyopathy
Muscle weakness and atrophy are also significant complications for patients who require mechanical ventilation and ICU care. This weakness may be attributed to a variety of factors, including critical illness neuropathy and/or myopathy, corticosteroid use, hyperglycemia, immobility and deconditioning.214,215 Critical illness neuromyopathy (CINM) has been defined as an illness that develops during a patient’s ICU stay, involving peripheral nerves, muscles or the neuromuscular junction. Several risk factors have been identified, including sepsis, multiple organ failure and hyperglycemia.216 While corticosteroid use has been suspected of increasing the risk of CINM, limited evidence suggests that corticosteroid use is not an independent risk factor for development of CINM.217 Use of neuromuscular blockers has long been associated with muscle weakness, especially after prolonged use.218,219 However, a clear causative role has not been established, as other known risk factors such as sepsis and hyperglycemia were not excluded. The typical patient with CINM presents with grossly symmetrical weakness without a known neuromuscular disease process predating ICU admission. Reflexes are typically depressed, yet may be normal, and muscle atrophy is often present. Facial musculature and sensation are generally spared.215,220
Quantification of muscle weakness is often difficult in the ICU patient who is sedated or delirious. Decreased motor response to noxious stimulus in the presence of facial grimacing may suggest CINM. Manual muscle testing is advocated for patients who can follow simple commands. For the critically ill patient who is unable to be turned 90 degrees side-lying, the therapist can raise the head of the bed as high as possible and with simple adaptations test the major muscle groups of the shoulder, wrist and hand as well as hip flexion, knee extension and ankle dorsiflexion and assign a 1-to 5- muscle grade.
Asymmetrical weakness, particularly in the peroneal or ulnar nerve distributions, should prompt consideration of compressive neuropathy, which is also common in the ICU patient population.221 Respiratory muscles are also affected, and likely contribute to prolonged weaning from mechanical ventilation, based on studies which compared duration of mechanical ventilation between patients with and without CINM.222–224
In clinical practice, diagnosis is typically made based on suggestive presentation, in the presence of known risk factors. Electroneuromyogram testing and neuroimaging may be used to confirm or rule out the CINM in the presence of other possible diagnoses.220 Electroneuromyogram testing typically shows a reduced compound muscle action potential with normal conduction velocity, and spontaneous electrical activity on muscle needle recording.220To date, the only established preventative measure for CINM is intensive insulin control to prevent hyperglycemia. Intensive insulin therapy has been shown to decrease the incidence of CINM, and reduce prolonged mechanical ventilation in medical ICU patients.216 An increasing attention to minimizing the use of sedation and analgesia in the ICU setting, while not initiated with this goal in mind, may aid in earlier diagnosis of CINM. Decreased use of sedation also has the potential to reduce drug-induced immobility. The value of early exercise and mobility in treating CINM has yet to be established, although these interventions are safe for patients with respiratory failure.2,4,6
Disconnecting the patient from the ventilator and ambulating the patient using a manual resuscitator bag (MRB), or ambulation using a portable ventilator (many of the ventilators found in standard use in the ICU have this capability) are also recommended for difficult-to-wean patients (Fig. 19-12). If a patient is unable to ambulate due to injury or disease affecting the lower extremity, yet is having difficulty being weaned from the ventilator, the therapist can use an MRB or portable ventilator to assist with ventilation while the patient is trained in wheelchair transfers and propulsion. Research regarding this type of early intervention including mobilization and exercise in the ICU patient population is extremely limited. In a study of 103 patients admitted to a respiratory ICU, Bailey and colleagues demonstrated that early activity including transfers and ambulation was both safe and feasible, with a less than 1% adverse event rate. Additionally, 69% of their patients were able to ambulate > 100 feet at the time of discharge from the RICU.6
Integumentary
The details of integumentary complications are discussed in Chapter 13. Rotating beds, bariatric beds, low-air-loss beds, air-fluidized beds, and mattresses that inflate at specified times to turn the patient 20 to 30 degrees to either side all assist in relieving capillary pressure on the skin to prevent pressure sores. Placing a patient in a bed that minimizes skin capillary pressure may prevent a pressure sore but does not prevent the other cardiopulmonary, musculoskeletal, and neurological hazards of immobility. Therapists should learn how to operate these beds and utilize any features that will assist with mobility such as deflating the mattress to achieve a firm surface for bed mobility and transfer activities.
When a patient is in a specialty bed, part of the therapists’ assessment should include whether the bed is optimal for patient mobility and pulmonary care. If it is suspected that a standard bed will enhance mobility and pulmonary care, this information should be relayed to the medical and nursing staff as a recommendation to change the bed. Newer standard beds allow mechanically ventilated patients to be positioned in the sitting position and begin sitting balance and standing activities without transferring to a chair.
In summary, patients who are mechanically ventilated and subsequently immobile benefit from the same interventions of cognitive orientation, positioning, therapeutic exercise, functional mobility, splinting, and serial casting as the patient who is not in respiratory failure. The same orthopedic precautions are followed regardless of whether the patient is mechanically ventilated. Special attention is paid to joints adjacent to intravascular lines and cervical ROM that may be limited because of bulky ventilator tubing.
Summary of Potential Risks Associated with Mechanical Ventilation
One may wonder why patients are intubated and mechanically ventilated after reviewing all of the associated risks. However, it is important to remember that the primary clinical concern is to provide adequate oxygenation for recovery from the underlying cause of respiratory failure. Given the inevitability of having patients who require mechanical ventilation, clear understanding of the potential risks will aid in their prevention. Therapists working with mechanically ventilated patients should always be cautious and aware of the modes and complications of mechanical ventilation. This will enable them to appropriately perform secretion clearance techniques and mobilize patients despite minute-by-minute changes in ventilatory and hemodynamic status.
CASE STUDY
Patient with C5 Tetraplegia
The intent of this case example is to familiarize the physical therapist with the medical, and physical therapy interventions pertinent to treating an ICU patient with tetraplegia. This multitrauma patient sustained a spinal cord injury following a motor vehicle collision (MVC). The patient fits the following inclusion criteria for Practice Pattern 6F. The patient has acute neuromuscular dysfunction, abnormal alveolar-arterial oxygen tension, and multisystem trauma. The patient also has adventitious breath sounds, abnormal vital capacity, elevated respiratory rate, dyspnea at rest and dyssynchronous (paradoxical) breathing patterns (without mechanical ventilation), and impaired gas exchange (Fig. 19-1).
The case presented is a 38-year-old Caucasian postal worker named Lee who sustained a C4-5 fracture dislocation in a motor vehicle crash, resulting in C5 motor complete tetraplegia. The case presentation follows the Guide to Physical Therapist Practice as applicable to the utilization of physical therapy in an ICU/trauma center. This case study is written as a physical therapist would realistically collect relevant data from the medical record, examine the patient, evaluate data, and determine a prognosis and intervention strategy within 24 hours of the patient’s admission. Intervention strategies are modified as the patient’s clinical condition changes. For the ICU patient the medical status can change hourly. Therefore, the physical therapist must continually modify the treatment plan and intervention strategies based on the patient’s examination at each treatment session.
Initial Examination
When entering the ICU to examine a patient, the physical therapist must efficiently extract the most pertinent and relevant information from the medical record and team members. It is most important to obtain general demographic information, a detailed history of the patient’s current condition, medications pertinent to physical therapy interventions, a pertinent past medical history including social habits, and the patient’s functional status at the time of the initial examination. Some centers utilize a paper form or computerized database to assure consistency of data collection at the initial and subsequent visits. Details regarding social history, employment, growth and development, living environments, family history, and the patient’s general health status are frequently not available at the time of the first visit. This information can be obtained later in the hospitalization through discussion with team members and the patient’s family, friends, and/or a significant other. Patients who are intubated have difficulty communicating and may be receiving medications or have injuries that make it difficult and sometimes impossible to obtain a complete history. Augmentative communication devices may be recommended by a speech–language pathologist, which enhance the physical therapist’s ability to communicate with the patient. The patient history (Table 19-14) was obtained within Lee’s first 10 days of hospitalization after attending rounds; meeting with the family in a family conference; reviewing the medical record daily prior to each visit; and speaking with the patient’s nurse, physician, social worker, and occupational therapist.
TABLE 19-14 Lee’s Patient History
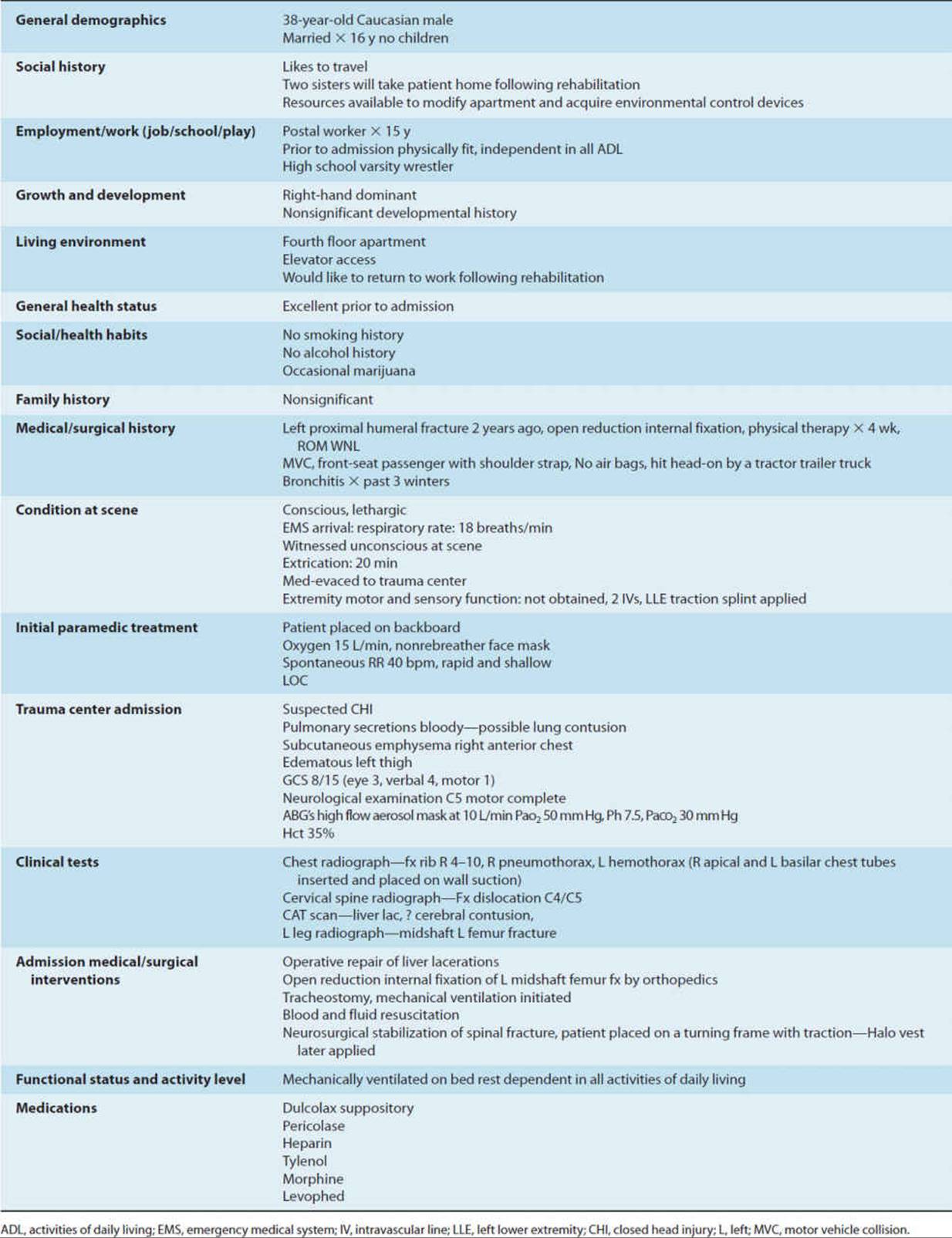
Lee was admitted to the critical care unit 12 hours after reaching the trauma center. The physical therapy service had standing orders to evaluate and treat any patient admitted with suspected or diagnosed spinal cord injury. The physical therapist provided an initial examination 18 hours after the patient’s hospital admission.
A chest radiograph was taken when Lee was admitted to the ICU. The report noted right rib fractures, clear lung fields, correct placement of the pulmonary artery catheter in the pulmonary artery, and the presence of electrocardiogram electrodes. Chest tubes and a feeding tube that passed through the stomach to the small intestine were also seen in proper position on the chest and abdominal radiographs. It was too early for an infiltrate from a pulmonary contusion to appear on the chest film.
Tests and Measures
Upon entering Lee’s cubicle, the therapist noted that the patient was connected to numerous lines and tubes and mechanically ventilated via a tracheostomy tube. Table 19-15 shows the ventilator settings and arterial blood gas results. Humidification was delivered through a heated humidifier (Fig. 19-6A). The therapist also noted that the patient had a pulmonary artery catheter to measure central venous and pulmonary artery pressures. Pulmonary artery (PA) catheters can be inserted through the jugular, subclavian, brachial, or femoral veins. This patient’s PA line was inserted through the subclavian vein, the most common insertion site. Figure 19-13 shows a quadruple lumen PA line and the waveforms associated with the line passing though the different chambers of the heart and into the pulmonary artery. The normal values for pulmonary artery pressures and other monitoring devices are noted in Table 19-16. Lee had chest leads connected to the cardiac monitor with a continuous readout of heart rate and rhythm, a pulse oximeter probe on the right index finger, a peripheral IV for fluid administration, and a catheter inserted in the bladder to facilitate and monitor urine output. The patient’s blood pressure was monitored through a peripheral radial arterial line.
TABLE 19-15 Ventilator Settings and Arterial Blood Gas Measurements
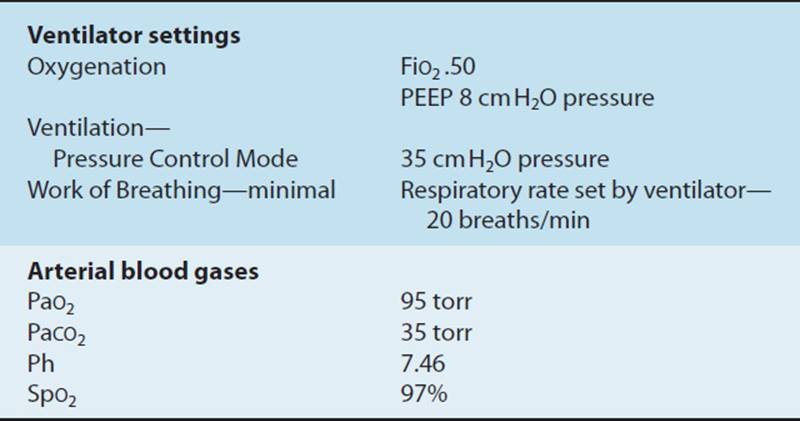
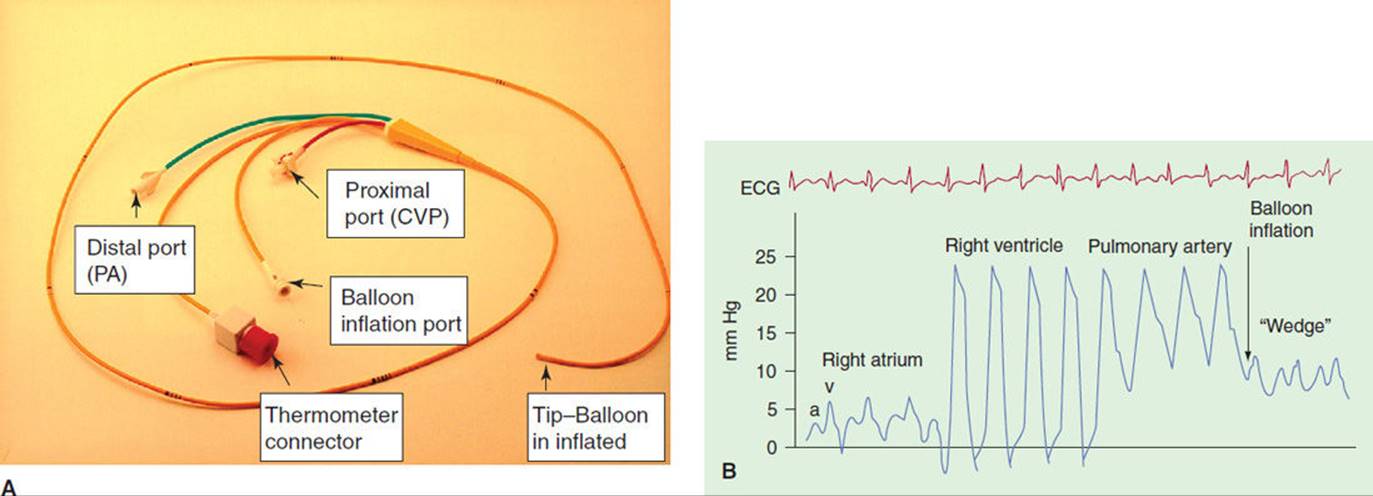
FIGURE 19-13 (A) Pulmonary artery line. (B) Waveforms seen on cardiac monitor as a pulmonary artery catheter passes through the chambers of the heart.
TABLE 19-16 Lines, Tubes, and Catheters: Summary of Precautions and Physical Therapy Implications
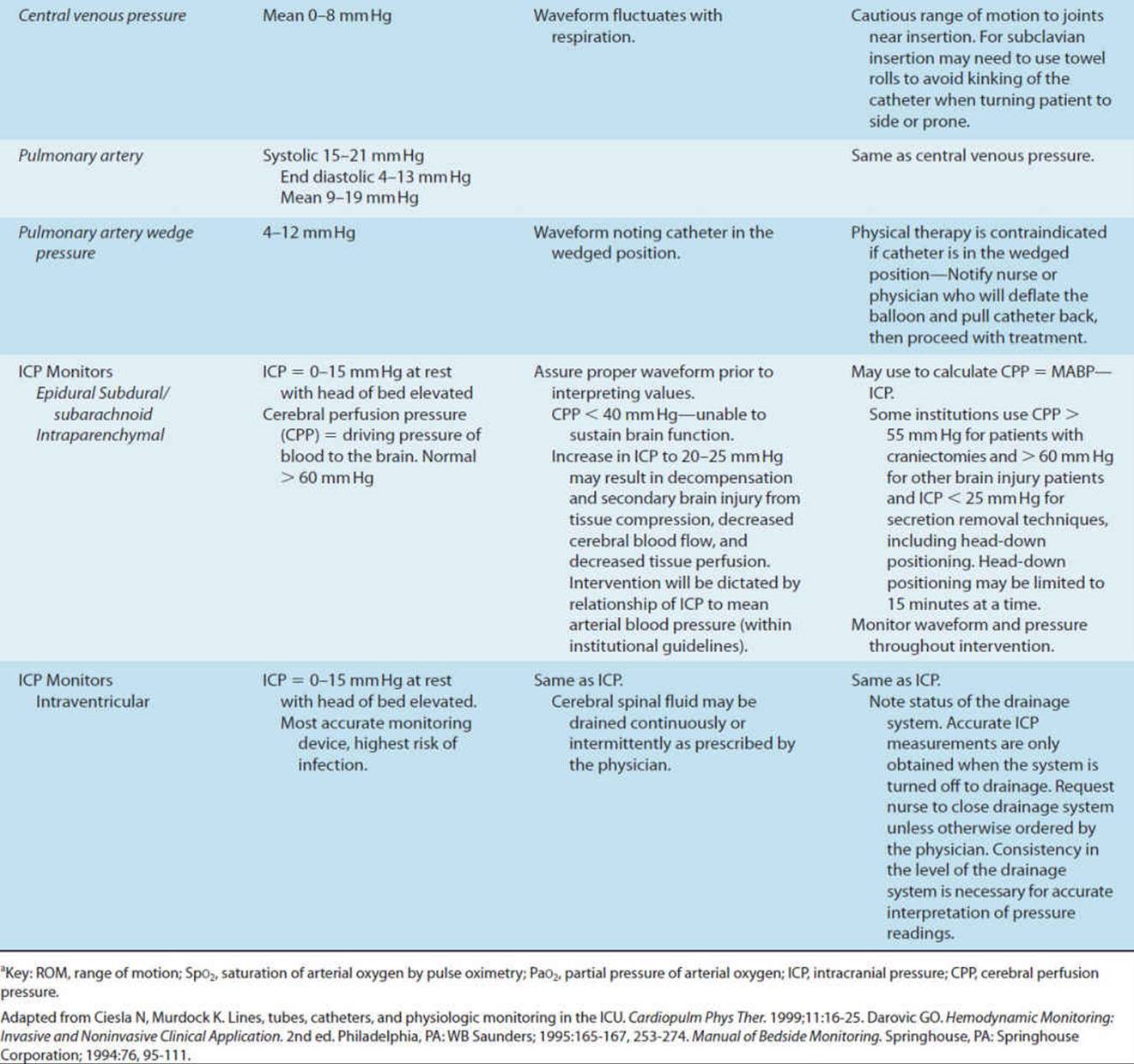
Arterial pressure monitoring via an indwelling peripheral catheter (arterial line) is the most common mode of invasive hemodynamic monitoring. It is used to draw blood samples, and to monitor arterial blood pressure when rapid fluctuations are expected. The radial artery is the most common site for placement of a peripheral artery catheter. Radial arterial lines have the least discomfort and do not require total joint immobilization. They are associated with a low risk of ischemic injury to the hand, as there is usually adequate collateral circulation through the ulnar artery. Other sites for monitoring arterial pressures include the dorsalis pedis, femoral, and brachial arteries. The femoral artery is the preferred site for emergency cannulation and is easily catheterized with a low risk of thrombosis. Care should be taken not to dislodge an arterial line, and not to kink or disconnect the tubing. The transducer is positioned at the level of the right atrium when blood pressure measurements are taken.
The patient also had an oral feeding tube that passed through the stomach and terminated in the small intestine, an incision on the left proximal lateral thigh from insertion of an intramedullary rod to stabilize the femur fracture, and an abdominal incision from the surgical repair of liver lacerations. The patient was wearing sequential pneumatic compression devices for prevention of venous thromboembolism. Skin was intact. There was some bruising on the anterior chest from the seatbelt.
The therapist completed a neurological assessment of the patient’s level of function at the initial visit. Lee’s Glasgow Coma Scale was 11T (eye 4, motor 6, verbal 1T) and he was following simple commands (Table 19-17). “T” indicates that the patient was intubated and could not speak. Richmond agitation scale was 0 and CAM-ICU was administered by having the patient stick out his tongue as he did not have adequate hand function to use hand gestures. It was determined that Lee was not delirious at this time and would most likely be able to participate in the initial physical therapy examination. Lee luckily did not have a severe brain injury and therefore did not require intracranial pressure monitoring. However, concomitant brain injury with cervical spine injury is as high as 60%, 43% of these patients having severe brain injuries.225 Had he sustained a severe brain injury in addition to his other injuries, he would most likely have had an intracranial pressure monitor inserted. Intracranial pressure monitoring alone is not a contraindication to physical therapy. Severe brain injury increases the risk of contractures, pulmonary infection, and hypoxemia and is therefore a strong indicator for physical therapy interventions. The therapist is responsible for monitoring the intracranial pressure and cerebral perfusion pressure during physical therapy interventions following institutional guidelines (see Table 19-16).
TABLE 19-17 Lee’s Physical Therapy Initial and Discharge Examinations
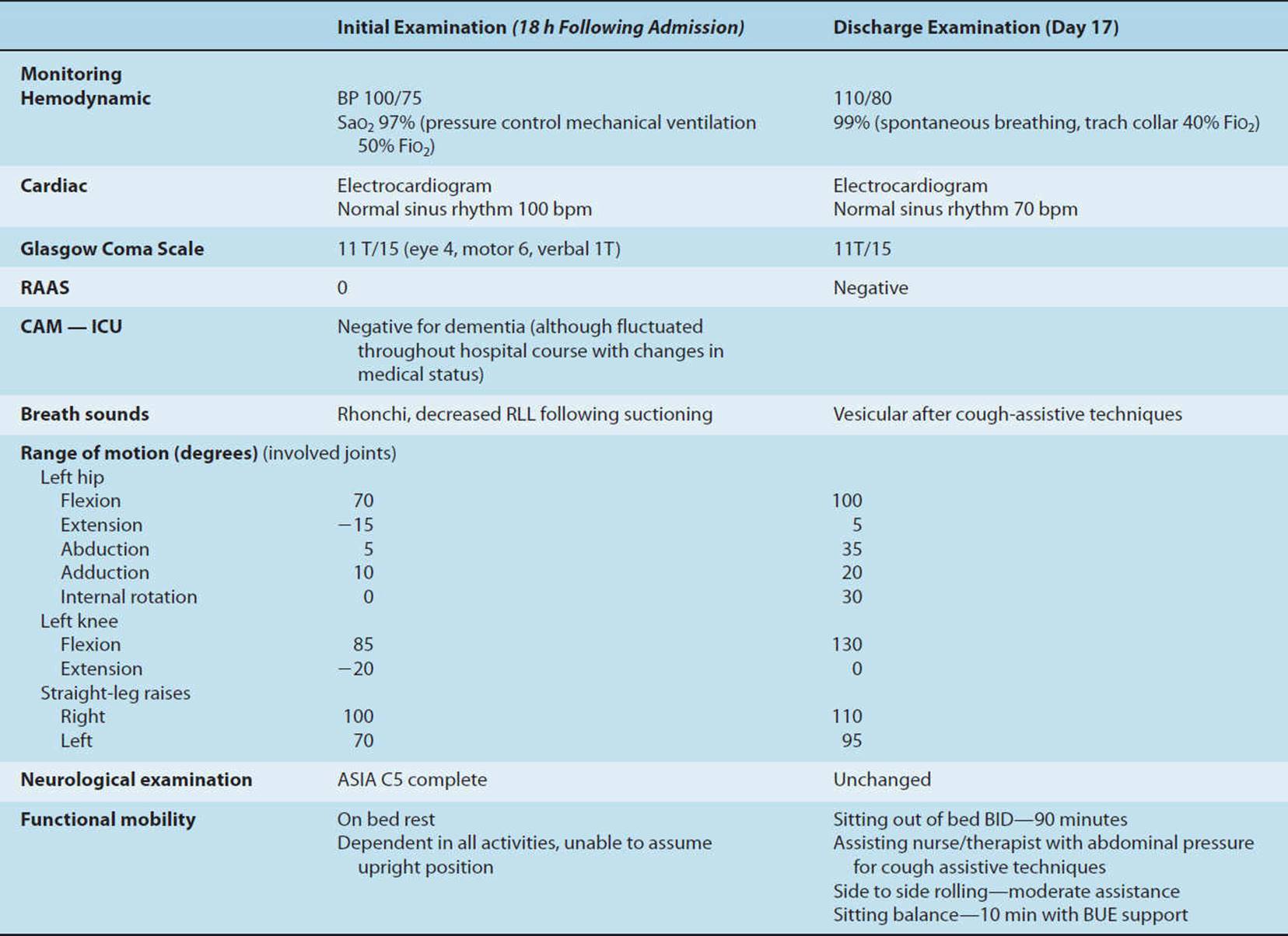
The therapist evaluated the patient’s range of motion (ROM) (Table 19-17). Passive ROM in both lower extremities (LEs) was WNL except for some limitations in left hip and knee motions. Because of early fracture fixation, complete assessment of the left hip and knee was possible after consulting with the orthopedic surgeon. ROM was within normal limits in both upper extremities, and no pain was noted with any ROM exercises. The spine had been stabilized with the Halo vest, and therefore there were no restrictions to assessing shoulder strength and providing shoulder ROM exercises to the patient’s tolerance.
The therapist then auscultated the patient’s chest and noted upper-airway secretions (rhonchi). Lee was unable to effectively cough or mobilize his secretions, so the therapist suctioned the patient through the tracheostomy tube with a 14 French suction catheter (the patient had a size 9 tracheostomy tube) and reexamined the chest. Decreased breath sounds were noted over the right lower lobe (adventitious breath sounds may precede a change in chest radiograph). While palpating the right anterior thorax, the therapist also noticed that the patient had subcutaneous emphysema. The therapist checked the chest tubes for an air leak (there was none) and discussed this finding with the nurse. The nurse reported that both chest tubes had been functioning to this point without an air leak. The nurse and therapist checked the patient’s airway pressures on the ventilator. They had not changed within the last few hours. After discussion and assessment, the therapist determined that the subcutaneous emphysema was likely residual from his admission pneumothorax, as resolution can sometimes take hours to several days.
Evaluation
Following the examination, the therapist determined that Lee was at high risk for additional pulmonary complications and musculoskeletal impairments due to his injury. Table 19-18 outlines the physical therapy tests and measures that were appropriate to administer during this patient’s hospital stay. The examination at each physical therapy visit determined which test was administered. Table 19-19 outlines the critical components of a physical therapy examination and evaluation for the mechanically ventilated patient.
TABLE 19-18 Lee’s Physical Therapy Tests and Measures
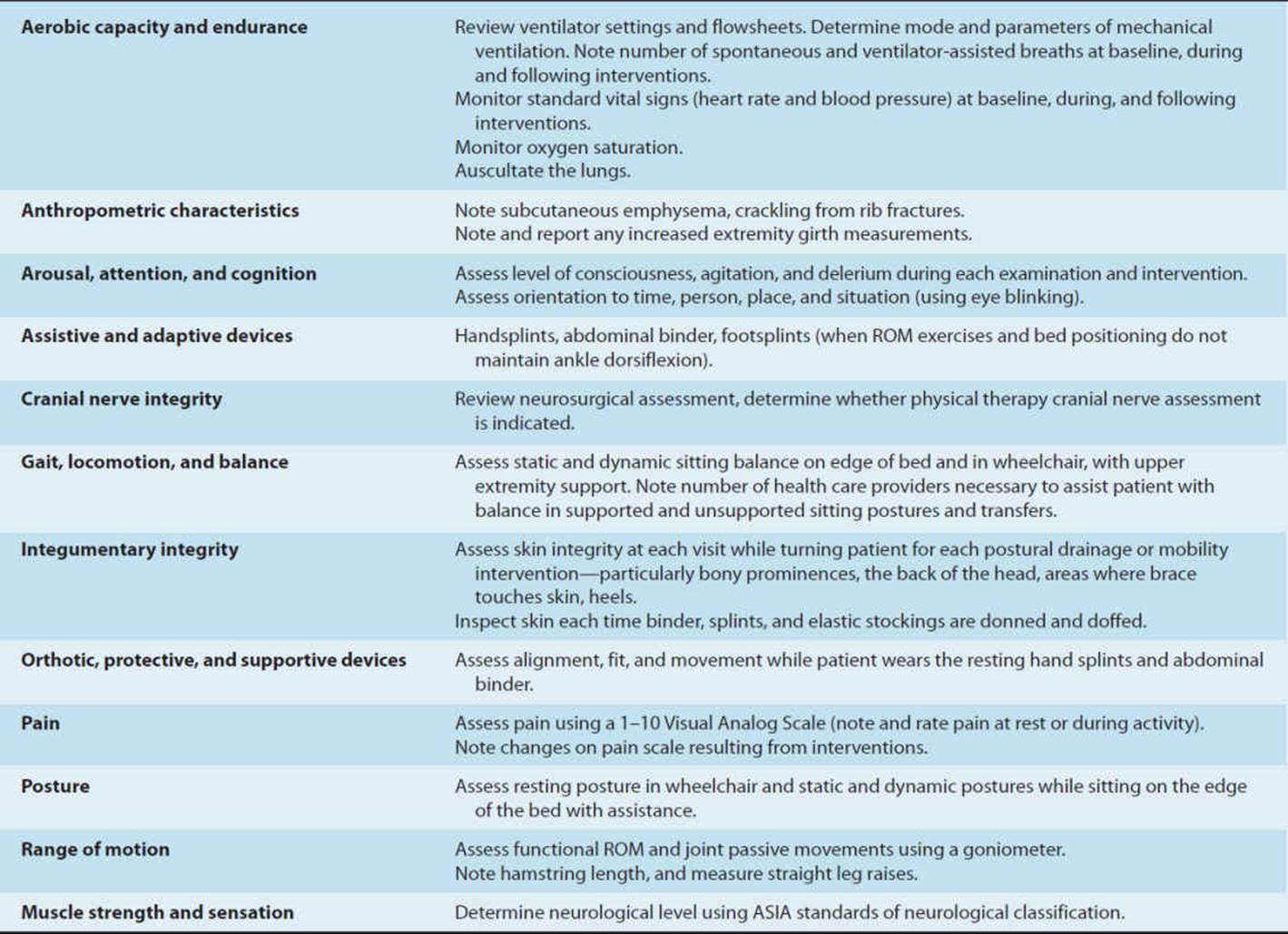
TABLE 19-19 Mechanically Ventilated and Monitored Patient Components of Physical Therapist Examination and Evaluation During Physical Therapy Sessionsa
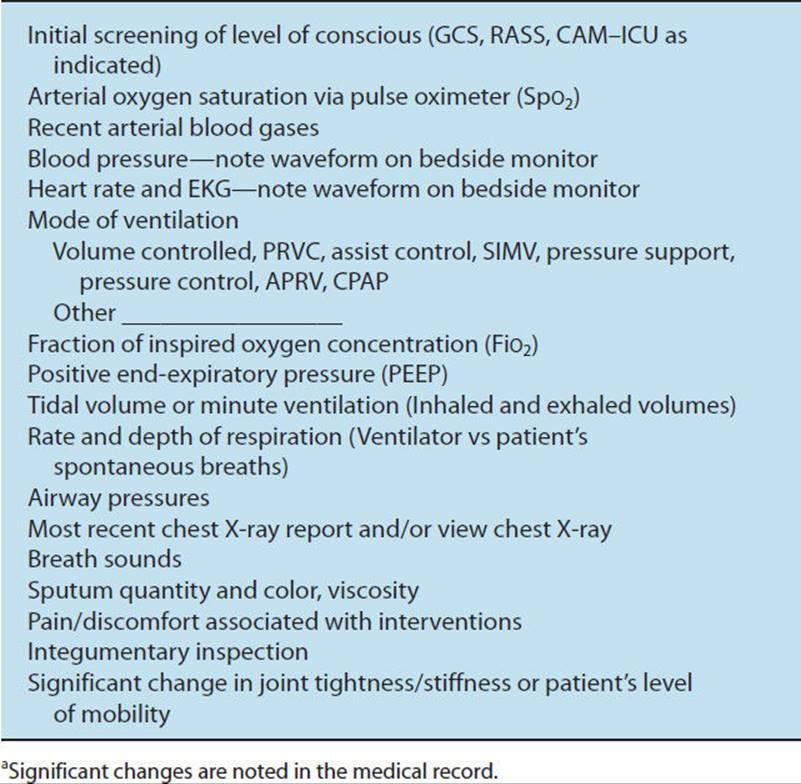
Diagnosis
The therapist determined that Lee had impairments in airway clearance, ROM, muscle strength, sensation, and mobility that would benefit from physical therapy interventions.
Prognosis
The therapist estimated Lee’s prognosis and outcome at the initial visit. The therapist expected the patient would leave the trauma center and enter a spinal cord injury rehabilitation center within 10 to 15 days, timing dependent upon the patient’s medical complications such as developing an infection, DVT, PE or skin breakdown. The patient would be spontaneously breathing through a tracheostomy tube while receiving supplemental oxygen and have functional passive range of motion of all four extremities. The patient would also have an understanding of how to assist himself with deep breathing and coughing exercises and instruct a caretaker in pressure relief. It was anticipated that the patient would tolerate sitting on the edge of the bed with moderate assistance of one individual and sitting in a wheelchair with the legs dependent for 2 to 4 hours per day. It was hoped that the patient would not develop any skin breakdown or thromboemboli.
Physical Therapy Interventions During Mechanical Ventilation
The first physical therapy priority was to clear Lee’s airway secretions, as a right lower lobe atelectasis due to secretion retention was suspected. The therapist and nurse positioned the patient in the left side-lying position, one-fourth turn from prone. They noted that there was no excessive drainage from either chest tube. There was also no air bubbling in the waterseal chamber of either chest tube, so it was unlikely that the patient had developed a bronchopleural fistula. The therapist and nurse monitored the patient’s vital signs, lines, and tubes and positioned the bed in the head-down position. Aspiration was less of a concern, because the feeding tube passed through the stomach and extended to the small intestine; however, the tube feeding was turned off prior to head-down positioning. The uppermost lateral strap of the vest was opened and taped to the bedrails to allow the therapist access to the patient’s chest for percussion and vibration. The therapist noted and marked which hole in the strap was used to secure the vest. The therapist manually percussed the right lateral chest below the axilla and posterior chest wall from thoracic vertebrae T3 to T10 (Fig. 19-14). Although the patient had right rib fractures, which were noted as a precaution, it was necessary to administer manual techniques to assist in secretion clearance on the right side because the patient could not be mobilized or cough sufficiently to clear retained pulmonary secretions. It should be noted that manual percussion may be performed judiciously over rib fractures without any known complications. When properly performed, there is no known increased risk in the development of extrapleural hematomas, hemo- and/or pneumothorax.226,227 Properly performed, percussion should cause less pressure over the thorax and ribs than coughing or lying on the involved side. A gentle form of vibration is used over rib fractures by some physical therapists.
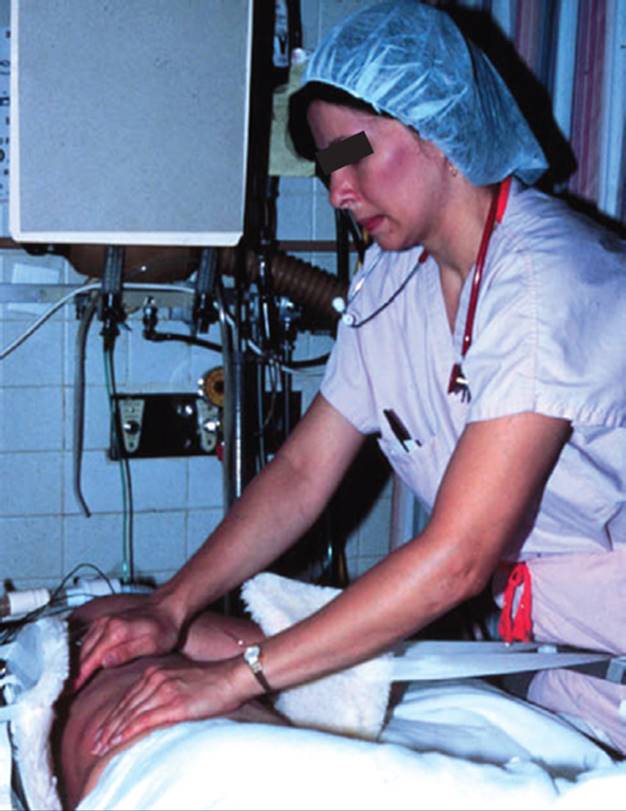
FIGURE 19-14 Physical therapist performing chest physical therapy to the right lower lobe of a patient with tetraplegia.
After 10 minutes of percussion to the right lower lobe, the therapist noted crackles with auscultation, which was indicative of secretions loosened in the small airways. The therapist suctioned the trachea. A cough was stimulated with the suction catheter, but no secretions were suctioned. This is most likely because the secretions remained in the lower airways as the cough was ineffective, and secretions were unable to be suctioned into the suction catheter that most likely was at the carina (level of the second rib), or upper level of the right mainstem bronchus. The therapist continued with percussion over the right lower lobe for another 10 minutes. Subsequent auscultation of breath sounds revealed diffuse rhonchi and the patient was suctioned again. After three passes of the suction catheter 5 cc of bloody sputum was obtained. Bloody secretions are to be expected after a lung contusion. Upon auscultation, the therapist noted the breath sounds had improved; there was increased air entry in the right lung base with some crackles. The therapist continued the treatment for 15 minutes with percussion, vibration, and suctioning after which vesicular or normal breath sounds were heard. The therapist closed the vest, placed the bed flat, and with the nurse turned the patient to the supine position. The therapist once again auscultated the chest and discovered that the breath sounds were normal in all lung zones, indicating that transbronchial aspiration of secretions was unlikely. It was therefore not necessary to turn the patient and treat the lung lobes and segments that were dependent during this portion of the treatment.
While Lee was supine, the therapist performed passive ROM exercises to both lower extremities with emphasis on straight-leg raises, ankle dorsiflexion, and the limited motion in the left hip and knee. The therapist was cautious while performing ROM exercises to the left hip and knee. Because of the patient’s neurological injury, he was unable to perceive pain in the lower extremities. The therapist was cautious not to overstretch the lumbosacral musculature that would impair the patient’s stability in sitting as he progressed through rehabilitation. To stretch the hamstrings, the therapist extended the knee while the hip was flexed to 90 degrees. The patient became exhausted and did not participate in any active or active-assistive ROM exercises of the innervated upper extremity muscles; therefore, passive ROM exercises were given. Emphasis was placed on achieving full wrist extension with elbow extension and shoulder external rotation to facilitate sitting at the edge of the bed. Care was taken not to stretch the finger flexors during wrist extension to allow a tenodesis grip to develop, given the level of Lee’s injury. Finger flexors were only stretched with the wrist in a neutral position. The therapist documented the physical therapy examination and interventions in the medical record. It was noted that the patient’s vital signs were stable during each intervention and that breath sounds improved over the right lower lobe; 5 cc of blood-tinged secretions was suctioned, and the patient tolerated 65 minutes of physical therapy examination and treatment.
The therapist returned in the afternoon for a second assessment of the need for airway clearance techniques. Upon auscultation, the therapist noted that the patient had crackles over the lingula (comparable to the right middle lobe). Treatment was given as in the morning in the postural drainage position for the lingula: Lee was positioned one-fourth turn to the right in the head-down position. The vest was opened again after Lee was placed and stabilized in the head-down position, and closed prior to moving him. The therapist added vibration to the patient’s tolerance. There were no documented rib fractures on the left side; therefore, vibration was added to mobilize secretions in the larger bronchi. During bronchoscopy, vibration has been shown to mobilize secretions into the upper airway, which in theory will decrease the duration of postural drainage.228 A moderate quantity of clear secretions were suctioned. (Precise measurements of sputum volume are obtained only when a sputum trap is attached to the suction catheter.)
The therapist continued the same treatment regimen for the next 2 days. During each treatment session the therapist noted the patient’s breath sounds and determined which lung/lobes or segments required intervention. Table 19-20lists the treatment interventions that were administered throughout the patient’s hospital stay. Active, active-assistive, and passive ROM exercises were carried out to patient tolerance. The nursing staff supplemented physical therapy sessions with PROM exercises. Hip and shoulder extension exercises were administered in the side-lying position. Shoulder extension, shoulder external rotation, elbow extension, and wrist extension exercises with the fingers in a position of tenodesis were emphasized to enhance sitting balance. Strengthening of the pectoralis muscles was added and emphasized to further assist the patient with coughing.140 Strengthening of serratus anterior was also included, to promote scapular stability, and facilitate functional use of the upper extremity. Occupational therapy made resting hand splints for the patient. The patient maintained ankle dorsiflexion with passive ROM exercises, had no increased plantarflexor muscle tone. Therefore, it was decided that ankle splints were not indicated.
TABLE 19-20 Lee’s Physical Therapy Interventions
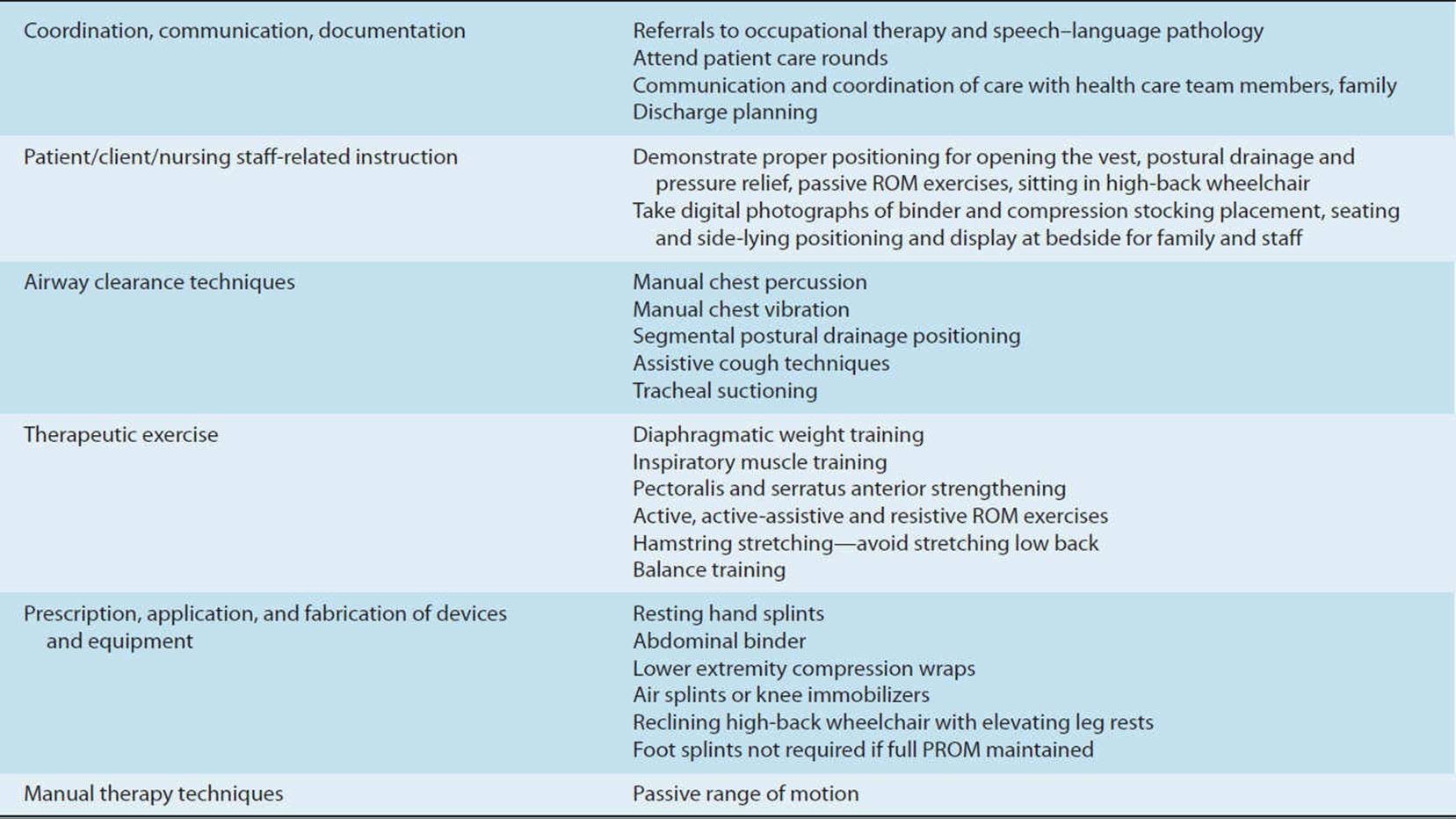
The fourth day following admission, secretion clearance and early rehabilitative interventions continued as previously described. The therapist noted that the humidifier had been changed to a heat and moisture exchanger. Secretions were viscid and dry with suctioning. The therapist spoke with the physician and respiratory therapist, the heated humidifier was placed back on the ventilator, and the HME was removed. The physician also decided to increase the patient’s fluid intake through the IV. Secretions were noted to be less viscid during suctioning throughout the day.
In addition, the therapist and nurse decided to transfer Lee from bed to chair, as the standing orders included progressing the patient’s activity as tolerated. The patient’s respiratory status had improved and he was no longer on pressure-controlled ventilation. However, pressure-controlled ventilation is not a contraindication for getting patients out of bed. Lee was on an SIMV of 12 breaths/min, pressure support of 15 cmH2O pressure, FiO2 = 0.45, and remained on a PEEP of 8 cmH2O pressure. He was taking 10 spontaneous breaths per minute and therefore had a respiratory rate of 22 breaths/min. An abdominal binder was applied to aide in venous return and improve the biomechanics of breathing. The IPCs were removed from the patient’s legs, and groin high compression stockings were applied to enhance venous return. The therapist examined the monitor and noted that the patient’s blood pressure was 100/60 mmHg while supine, and the heart rate was 90 bpm with a regular rhythm. The head of the bed was elevated 30 degrees for 15 minutes, and the patient’s blood pressure was noted to be 110/95 mmHg. The head of the bed was raised to 70 degrees, and the patient became diaphoretic, with a spontaneous respiratory rate of 21 breaths/min, and when questioned, he confirmed feeling dizzy. At this time, the patient’s blood pressure was 80/60 mmHg and the heart rate was 120 bpm. The patient’s head was lowered to 50 degrees. Lee no longer felt dizzy and the blood pressure had returned to baseline. Because of the increase in heart rate, decrease in blood pressure, and the patient’s complaints of dizziness the nurse increased his vasopressors (often used in the first 7 days to maintain mean arterial pressure and promote spinal cord perfusion), which allowed him to sit upright in bed and transfer out of bed that day. Because of numerous monitoring and therapeutic lines as well as to the patient’s inability to assist, three caregivers were needed to transfer Lee out of bed. Each caregiver was careful not to dislodge the IV or any of the central monitoring lines. The ventilator was disconnected from the tracheostomy tube during the transfer and reconnected as soon as the patient was in the wheelchair. The patient sat out of bed for 30 minutes with pressure relief every 15 minutes. The hips were flexed to 90 degrees, and the leg rests were kept elevated to aide in venous return. Two pictures were taken and placed at the patient’s bedside for the nursing staff. One picture showed the patient sitting in the high-back wheelchair, the other demonstrated the staff providing pressure relief. The nursing staff began getting the patient out of bed daily using the photographs to assure continuity in positioning, and inspecting the skin after each sitting intervention.
In a situation where the nurse does not have the ability to immediately titrate vasopressors the therapist would develop a plan with the nurse to monitor blood pressure and gradually increase the patient’s sitting position in bed at least two times daily, time permitting. The nurse would inspect the skin and document the time the patient tolerated sitting up in bed with each session. Once the patient could tolerate sitting up in bed for 30 minutes transfers out of bed would be initiated.
Over the next 48 hours, Lee’s physical therapy interventions included secretion removal techniques, active and active-assistive ROM exercises to the pectoralis and innervated cervical and upper extremity muscle groups, and passive ROM to the lower extremities. Patient education regarding the rationale as well as specific ROM exercises was incorporated in the treatment plan. He continued sitting out of bed in a highback reclining wheelchair with a pressure distributing cushion and a full sheet for rapid return to bed if necessary. Vital signs remained stable with a blood pressure of 100/80 mmHg and heart rate of 85 bpm while wearing elastic bandages and an abdominal binder, with his legs dependent on the footrests of the wheelchair. On day 7, the therapist decided to attempt sitting balancing activities on the side of the bed with a therapy aide. Lee continued to improve and was now mechanically ventilated with an FiO2 of 0.40, pressure support 12 cm of H2O pressure, SIMV 8, and PEEP of 5 cmH2O pressure. His respiratory rate was 18 breaths/min. Lee required maximal assistance to assume the sitting position, yet was able to tolerate sitting upright with the binder, leg wraps, and moderate assistance from the therapist (Fig. 19-15). During the treatment session Lee used his upper extremities for support for 10 minutes. The ankles were supported in a neutral position on a footstool. This treatment was continued throughout Lee’s hospital stay with the goal of getting the patient to support his upper body with shoulders externally rotated and elbows and wrists extended, while the fingers remained flexed. After ensuring adequate scapular stability, the therapist also encouraged Lee to assist as much as possible with rolling in bed, and transfers from the side-lying to sitting position. Small knee immobilizers (air splints may also be used) were placed over the elbows during deltoid and serratus anterior strengthening exercises to give the patient a longer lever arm by maintaining the elbows in extension (Fig. 19-16).
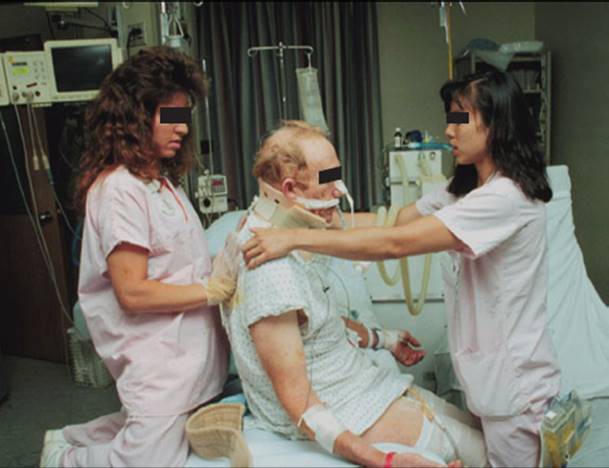
FIGURE 19-15 Patient with C5 tetraplegia—initiating sitting balancing activities.
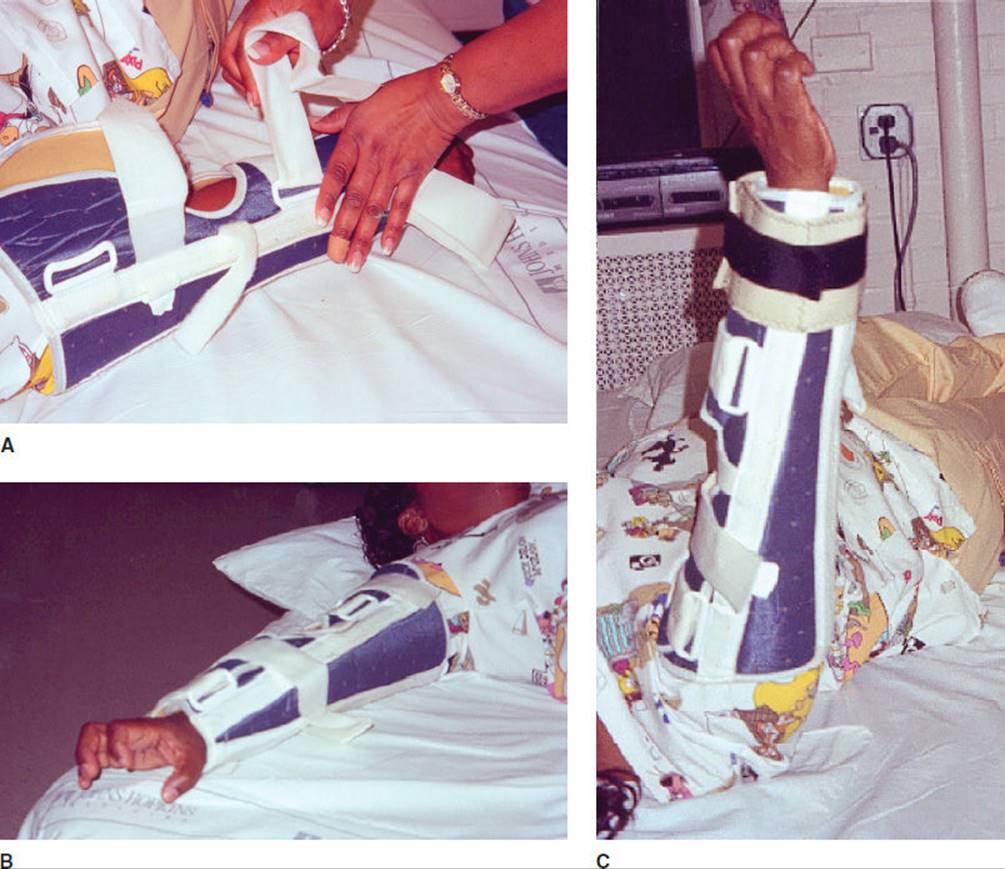
FIGURE 19-16 Use of small knee immobilizers to maintain elbow extension with deltoid exercises.
On day 9, Lee developed a fever of 104°F (40.5°C) and was noted to have positive blood cultures. The source was thought to be pulmonary; yet this was never confirmed. Lee could no longer tolerate getting out of bed, and refused to turn onto his left side because of left shoulder pain. Although it is a routine procedure to turn a tetraplegic patient side to side every 2 hours and position them as comfortably as possible, this patient had a new nurse who felt badly about having to impose discomfort on the patient. Therefore, he was not turned onto his left side for 12 hours. Lee’s oxygen saturation decreased to 85%, and upon clinical examination, the physician noted no air entry into the right lung. A chest radiograph was ordered and is shown in Figure 19-17. The physician decided a therapeutic bronchoscopy was indicated because of the significant right middle and lower lobe collapse noted on the chest radiograph. The FiO2 was increased to 1.00, as is standard procedure during a bronchoscopy. Following the bronchoscopy the patient’s chest radiograph was only minimally improved as seen in Figure 19-18. Physical therapy was immediately paged and asked to treat the patient. Immediate chest physical therapy treatment was given to the right lower and middle lobes for 35 minutes. Fifteen cc of whitish sputum was obtained, and breath sounds became audible over the right lower and middle lobes. During the treatment the breath sounds progressed from barely audible to rhonchi and slightly diminished with a few crackles. Following the treatment, the therapist auscultated both sides of the patient’s chest. After returning the patient to the supine position, the right lower lobe sounded clear and crackles were noted over the left lower lobe. Chest physical therapy was then provided to the left lower lobe for 25 minutes as it was suspected that the patient had aspirated secretions from the right to the left lung. A repeat chest X-ray was taken, and the right lower lobe atelectasis was markedly improved as shown in Figure 19-19. Lee tolerated having his FiO2decreased to 45% with a SpO2 of 93% 30 minutes after physical therapy treatment.
FIGURE 19-17 Chest radiograph before therapeutic bronchoscopy.
FIGURE 19-18 Chest radiograph following therapeutic bronchoscopy.
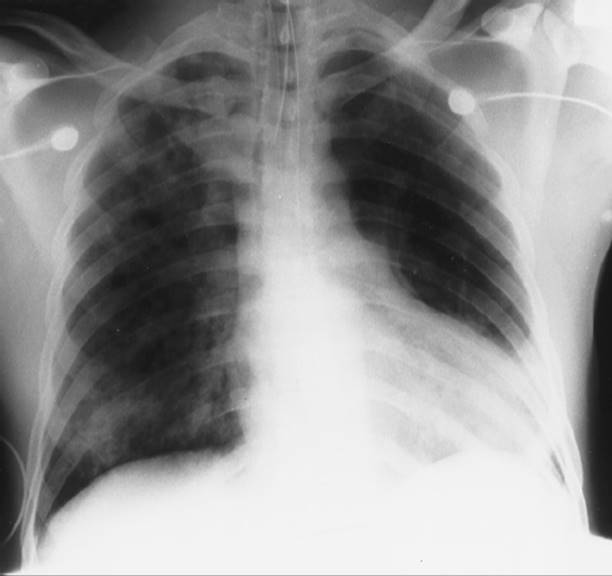
FIGURE 19-19 Chest radiograph following chest physical therapy.
Physical Therapy Interventions with Liberating the Patient from Mechanical Ventilation
On day 11, Lee’s blood cultures were available and found to be negative. It was therefore believed that the patient was free of systemic infection. The physician decided to begin liberating Lee from the ventilator. Lee’s temperature was 99°F, 37.2°C. The weaning procedure consisted of pressure support and SIMV ventilation for the majority of the day interspersed with periods CPAP. During CPAP the physical therapist was asked to provide respiratory muscle strengthening exercises and instruct the patient in how to assist with “quad coughing” (see Chapter 20). The therapist had two available options for respiratory muscle strengthening exercises—either abdominal weight training or inspiratory muscle training (IMT). The therapist chose to use abdominal weight training because the patient had difficulty understanding how to use the inspiratory muscle trainer, and each time the trainer was attached to the tracheostomy tube a cough was stimulated and secretions occluded the trainer. Greater patient education in IMT and frequent cleaning of the IMT device were needed to enhance the effectiveness of IMT in this patient, which was difficult during this acute period. Inspiratory muscle training was used later during the patient’s hospital stay (see Chapter 20).
The therapist therefore used the method of diaphragmatic strength training described by Derrickson et al.139 With the patient supine, the maximal inhaled volume without weights was recorded using an incentive spirometer. This was a gross assessment of the patient’s inspiratory capacity (IC). Standard dish weights were placed on the patient’s upper abdomen, just below the xiphoid process in 1/2- to 5-lb increments (Fig. 19-9).
The patient’s baseline inspiratory capacity was 800 mL, and 1,000 mL after adding 15 lb. With 20 lb, the inspiratory capacity was 759 mL. The therapist decided to begin training with 20 lb twice per day. During each training session, the patient was disconnected from mechanical ventilation and performed 10 maximal inspirations, holding each breath for several seconds. This sequence was repeated three times for a total of 40 breaths per session. The patient continued with abdominal weight training twice daily for 5 days in conjunction with weaning. Initially therapeutic exercises, functional mobility activities, and postural drainage with manual techniques were continued while the patient was mechanically ventilated. During weaning it is important to prevent respiratory muscle fatigue; therefore, initially it was necessary to perform physical therapy interventions (with the exception of abdominal weight training) during periods of mechanical ventilation when the respiratory muscles were rested. The therapist monitored the patient’s vital signs and used clinical judgment to determine whether the treatment interventions were best performed with mechanical ventilation or CPAP. The patient was able to tolerate all physical therapy interventions during CPAP on day 15. Suctioning was frequently necessary during diaphragmatic weight training, postural drainage, and functional mobility sessions because the patient mobilized secretions but could not adequately expectorate (cough) on his own. The patients need for postural drainage with manual techniques decreased as breathing exercises and functional mobility activities were better tolerated. (When adventitious breath sounds were improved by breathing exercises and mobility activities.)
On day 16, the patient’s CPAP time had increased to 18 hours per day, with only 6 hours of mechanical ventilation during sleep. The physicians decided to monitor the patient during the night while on a CPAP of 8 cmH2O pressure. The patient tolerated this final step in weaning through the next 48 hours and the patient was liberated from mechanical ventilation (Table 19-21).
TABLE 19-21 Respiratory Parameters, Day 16 Clinical Considerations (Adult)—“Weaning” from Mechanical Ventilation
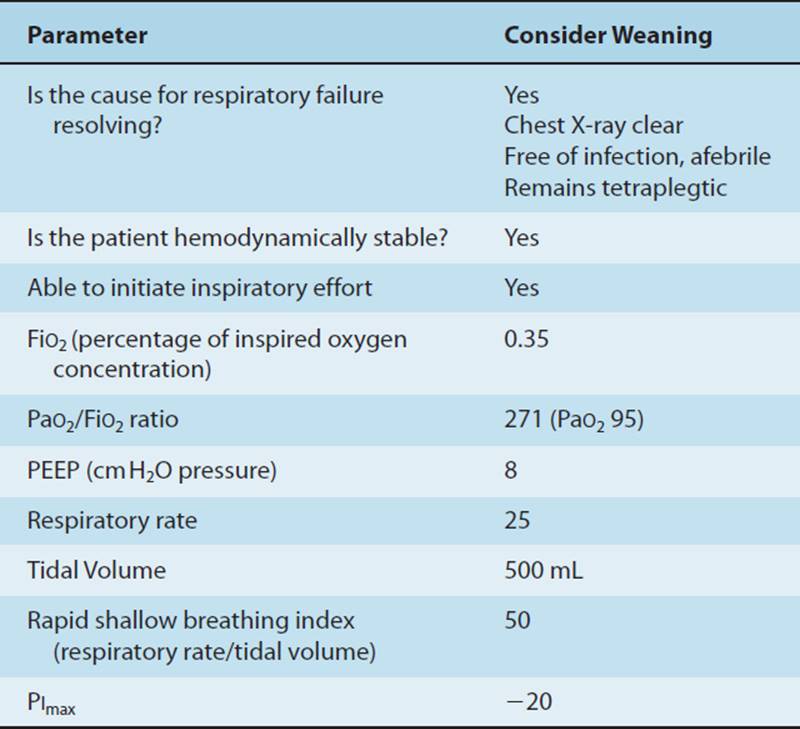
When Lee was weaned from the ventilator, he was tolerating CPAP sitting out of bed in a wheelchair twice daily with his legs dependent for 90-minute sessions, wearing the abdominal binder and elastic bandages. The nursing and physical therapy staff assisted the patient with forward and side bending for pressure relief every 15 to 30 minutes while sitting. He was participating in active and active-assistive ROM exercises to the upper extremities and able to sit on the edge of the bed supported by his upper extremities for 10 minutes. He was able to assist the nurses and therapists while they applied abdominal pressure to enable him to have a productive cough. Although he lacked independence in bed mobility, he could roll side to side and transfer from supine to sitting with moderate assistance from a caregiver. The patient was discontinued from this practice pattern because of successful separation from the mechanical ventilator. The patient’s physical therapy program while spontaneously breathing is described in Chapter 20 where he was assigned to Practice Pattern E.
At the time of the initial physical therapy examination, Lee was dependent in all mobility and required mechanical ventilation 24 hours per day. After receiving physical therapy for 16 days, Lee demonstrated improvement in bed mobility, sitting balance, tolerance to the upright position, the ability to participate in active ROM exercises, and an improved cough. Nursing and physical therapy interventions prevented pressure sores and ROM was either maintained or improved in all extremity joints.
DISABLEMENT MODEL
The Disablement Model is described in detail in Chapter 2. The domain of physical therapy practice is primarily at the impairment and functional limitation levels. However, physical therapy airway clearance techniques for the mechanically ventilated patient with pulmonary secretion retention may also impact the pathology and pathophysiology of pneumonia and atelectasis. When performing secretion clearance techniques, an atelectasis can be completely reversed as noted in the case study. Treatment interventions that prevent pulmonary infection may reverse the pathophysiology of a developing pneumonia. Physical therapy interventions are also provided at the impairment level (to prevent or improve limitations in ROM or muscle strength, deconditioning); and at the level of functional limitation (when bed mobility activities, transfer training, and ambulation or wheelchair propulsion using a manual resuscitator bag or portable ventilator may be indicated). Medical interventions may take priority, yet early mobility can enhance the medical interventions. With our current level of knowledge, determining a long-term prognosis and predicting a level of disability for the patient with acute respiratory failure is usually premature and better determined in the rehabilitation or home setting after the patient’s medical condition has plateaued. The patient’s social situation, including family support and educational and vocational interests and aptitudes, can be better assessed outside the acute hospital setting. The patient’s level of disability will vary depending on how the patient utilizes his or her inherent abilities.
The Limits of Our Knowledge
Despite physical therapists’ role in mobilizing and providing airway clearance interventions to mechanically ventilated patients for the past 35 years, there is only scant evidence supporting the benefit of these interventions.2,4–6,114,229–233 However, the need for compelling evidence to support physical therapy interventions is great since 57% of ARDS/ALI patients are unable to return to normal activity 12 months after hospitalization, and physical domains of survivors’ quality of life are more adversely affected than mental health domains.234,235 Moreover, after only 1 week of mechanical ventilation, 25% of conscious patients develop muscle weakness, and the risk of critical illness neuromuscular abnormalities is nearly 50% in ICU patients with sepsis, prolonged mechanical ventilation or multiorgan failure.215,236,237 ICU-acquired weakness is also associated with a longer duration of mechanical ventilation.214,215,220,237,238 Many believe that physical therapy interventions can have a positive effect on ventilator-associated pneumonia, muscle strength, functional mobility, and long-term functional outcomes. However, strong evidence to support these beliefs is lacking. One recent study demonstrated the safety and feasibility of physical therapy 48 hours after initiating mechanical ventilation with an associated decrease in hospital stay and cost for survivors.4 More are needed.
Patient outcomes vary depending upon patient comorbidities, the nature of their critical illness and the associated ICU management. The use of corticosteroids and neuromuscular blocking agents, glycemic control, and sedation all may impact the duration of the ICU stay and functional outcomes.214,215,236 Further research is necessary to establish the optimal type, timing, duration, and frequency of physical therapy services for patients who require mechanical ventilation. The most beneficial and cost-effective physical therapy interventions with the best outcome are also yet to be determined.
The Future
With advances in the management of critically ill patients, more patients may survive critical illness emphasizing the importance of optimizing ICU care to minimize patients’ long-term sequelae. As the population ages, the frail elderly may have even greater representation within ICU patient populations. In this population, even a small decrement in physical function may move the patient from independent living to requiring care in a nursing facility. Although heavy sedation and immobility have been commonly associated with mechanical ventilation, this paradigm of ICU care is changing. The need for physical therapists to be comfortable with providing interventions to mechanically ventilated patients may grow. Thus, the need for airway clearance techniques, ROM exercises, and functional mobility training may expand within the intensive care setting and beyond, as patients move on to lower levels of care while continuing to require mechanical ventilation.
Support for physical therapists to work closely with the ICU clinicians and researchers is vital for developing evidence-based assessment tools and protocols regarding the utilization of rehabilitative care in the ICU. Physical therapy education needs to provide more time and clinical experience in caring for mechanically ventilated patients. All too often, patient care in the ICU is deprioritized in favor of less acutely ill patients, when in fact, theses patients may have the greatest need for physical therapy interventions. More training in assessing cognitive function is also needed.169 Tapping into the resources of experienced, enthusiastic ICU physical therapists, and utilizing these experts when placing students at clinical education sites is essential. Training should emphasize the need for therapists to directly interact with ICU nurses and physicians in evaluating and encouraging safe and appropriate physical therapy interventions for critically ill patients.
Thoughts for the Future
What are the best type, timing, and duration, of physical therapy interventions?
How can physical therapy best be administered in an ICU when patients are frequently sedated and undergoing medical and nursing procedures?
Are outcomes better and costs reduced when therapists are assigned specifically to the ICU as an integral part of the ICU healthcare team?
Does inspiratory muscle training enhance weaning for patients who are optimally mobilized versus those who are unable to gain independence in functional mobility?
Do patients with pneumonia or atelectasis respond better to chest physical therapy interventions including directed postural drainage, percussion, and vibration than breathing exercises and mobilization?
What is the best timing for physical therapy interventions in conjunction with evidence-based daily spontaneous breathing trials to evaluate liberation from mechanical ventilation?
How can physical therapy interventions be best coordinated with evidence-based daily interruptions of continuous sedation?
What are optimal changes, if any, in mechanical ventilator settings for physical therapy interventions versus bed rest?
ACKNOWLEDGMENT
The authors would like to thank the following for their critical review and assistance with this chapter: Roy Brower, MD, for his review and expertise with mechanical ventilation; P Milo Frawley, RN, MS, ACNP, CCNS, for his critical review and assistance with the modes of mechanical ventilation; and Dale Needham, MD, PhD, for his review, photographs, references, and never-ending availability.
REFERENCES
1.Morris PE. Moving our critically ill patients: mobility barriers and benefits. Crit Care Clin. 2007;23(1):1-20.
2.Stiller K. Safety issues that should be considered when mobilizing critically ill patients. Crit Care Clin. 2007;23(1):35-53.
3.Perme CS, Southard RE, Joyce DL, Noon GP, Loebe M. Early mobilization of LVAD recipients who require prolonged mechanical ventilation. Tex Heart Inst J. 2006;33(2):130-133.
4.Morris PE, Goad A, Thompson C, et al. Early intensive care unit mobility therapy in the treatment of acute respiratory failure. Crit Care Med. 2008;36(8):2238-2243.
5.Thomsen GE, Snow GL, Rodriguez L, Hopkins RO. Patients with respiratory failure increase ambulation after transfer to an intensive care unit where early activity is a priority. Crit Care Med. 2008;36(4):1119-1124.
6.Bailey P, Thomsen GE, Spuhler VJ, et al. Early activity is feasible and safe in respiratory failure patients. Crit Care Med. 2007;35(1):139-145.
7.Dowdy DW, Needham DM, Mendez-Tellez PA, Herridge MS, Pronovost PJ. Studying outcomes of intensive care unit survivors: the role of the cohort study. Intensive Care Med. 2005;31(7):914-921.
8.Herridge M, Cheung AM, Tansey CM, Martyn AM, Diaz-Granados N. One-year outcomes in survivors of the acute respiratory distress syndrome. N Engl J Med. 2003;348(8):683-693.
9.Chelluri L, Im KA, Belle SH, et al. Long-term mortality and quality of life after prolonged mechanical ventilation. Crit Care Med. 2004;32(1):61-69.
10.American Physical Therapy Association. Guide to Physical Therapist Practice, 2nd ed. Phys Ther. 2001 Jan;81:539-553.
11.West JB. Respiratory Physiology—The Essentials. 7th ed. Philadelphia, PA: Lippincott Williams & Wilkins; 2005:186.
12.Gerold K, Nussbaum E. Understanding mechanical ventilation. Phys Ther Pract. 1994;3:81-91.
13.Shaffer T, Wolfson M, Gault JH. Respiratory physiology. In: Shaffer T, Wolfson M, Gault JH, eds. Cardiopulmonary Physical Therapy. 3rd ed. Baltimore, MD: Mosby; 1995:260.
14.Kacmarek R, Hickling K. Permissive hypercapnia. Respir Care. 1993;38:373-386.
15.Hickling KG, Henderson SJ, Jackson R. Low mortality associated with low volume pressure limited ventilation with permissive hypercapnia in severe adult respiratory distress syndrome. Intensive Care Med. 1990;16(6):372-377.
16.Wolthuis EK, Choi G, Dessing MC, et al. Mechanical ventilation with lower tidal volumes and positive end-expiratory pressure prevents pulmonary inflammation in patients without preexisting lung injury. Anesthesiology. 2008;108(1):46-54.
17.Marini JJ, Brower RG. Auto-peep with low tidal volume. Am J Respir Crit Care Med. 2003;167(8):1150-1151; author reply 1151.
18.Rossi A, De Ninno G, Mergoni M. Respiratory muscles in intensive care medicine. Monaldi Arch Chest Dis. 1999;54(6):532-538.
19.Tobin M. Mechanical ventilation: conventional modes and settings. In: Fishman A, ed. Fishman’s Pulmonary Diseases and Disorders. 3rd ed. New York: McGraw-Hill; 1998:2691-2699.
20.Kumar A, Falke KJ, Geffin B, et al. Continuous positive-pressure ventilation in acute respiratory failure. N Engl J Med. 1970;283(26):1430-1436.
21.Levitzky M, Cairo J, Hall S. Introduction to Respiratory Care. Philadelphia, PA: WB Saunders; 1990:525-527,534.
22.Putensen C, Zech S, Wrigge H, et al. Long-term effects of spontaneous breathing during ventilatory support in patients with acute lung injury. Am J Respir Crit Care Med. 2001;164(1):43-49.
23.Deem S, Bishop MJ. Physiological consequences of intubation. In: Marini JJ, Slutsky AS, eds. Physiologic Basis of Ventilatory Support. New York: Marcel Dekker Inc; 1998:623-654.
24.Acosta P, Santisbon E. The use of positive end-expiratory pressure in mechanical ventilation. Crit Care Clin. 2007;23(2):251-261.
25.Adams AB, Lim S. Monitoring and management of the patient in the intensive care unit. In: Wilkins RL, Stoller JK, Scanlan CL, eds. Egan’s Fundamentals of Respiratory Care. 8th ed. St Louis, MO: Mosby; 2003:1081-1119.
26.Frawley PM. Thoracic trauma. In: McQuillan KA, Makic MB, Whalen E, eds. Trauma Nursing: from Resuscitation Through Rehabilitation. 4th ed. St Louis, MO: Saunders Elsevier; 2009:614-677.
27.Staudinger T, Kordova H, Roggla M, et al. Comparison of oxygen cost of breathing with pressure-support ventilation and biphasic intermittent positive airway pressure ventilation. Crit Care Med. 1998;26(9):1518-1522.
28.Brochard L, Harf A, Lorino H, Lemaire F. Inspiratory pressure support prevents diaphragmatic fatigue during weaning from mechanical ventilation. Am Rev Respir Dis. 1989;139(2):513-521.
29.Oba Y, Salzman GA. Ventilation with lower tidal volumes as compared with traditional tidal volumes for acute lung injury and the acute respiratory distress syndrome. N Engl J Med. 2000;343(11):812-814.
30.Brower RG, Ware LB, Berthiaume Y, Matthay MA. Treatment of ARDS. Chest. 2001;120(4):1347-1367.
31.Peterson WP, Barbalata L, Brooks CA, Gerhart KA, Mellick DC, Whiteneck GG. The effect of tidal volumes on the time to wean persons with high tetraplegia from ventilators. Spinal Cord. 1999;37(4):284-288.
32.Marcy T. Full ventilatory support. In: Marini J, Slutsky A, eds. Physiological Basis of Ventilatory Support. New York: Dekker; 1998:783-792,807.
33.Pinhu L, Whitehead T, Evans T, Griffiths M. Ventilator-associated lung injury. Lancet. 2003;361(9354):332-340.
34.Pocket Guide: Servo i modes of ventilation. Maquet – Products – Servo-i Adult Web site. http://www.maquet.com/productPage.aspx?m1=112599774495&m2=112808545902&m3=105584076919&productGroupID=112808545902&productConfigID=105584076919&productConfigViewID=3&imageViewCategoryID=112799157632&languageID=1&subID=Sales%20Flyer. Published 2003. Accessed July 26, 2008.
35.Wrigge H, Zinserling J, Neumann P, et al. Spontaneous breathing improves lung aeration in oleic acid-induced lung injury. Anesthesiology. 2003;99(2):376-384.
36.Froese AB, Bryan AC. Effects of anesthesia and paralysis on diaphragmatic mechanics in man. Anesthesiology. 1974;41(3):242-255.
37.Putensen C, Muders T, Varelmann D, Wrigge H. The impact of spontaneous breathing during mechanical ventilation. Curr Opin Crit Care. 2006;12(1):13-18.
38.Putensen C, Hering R, Muders T, Wrigge H. Assisted breathing is better in acute respiratory failure. Curr Opin Crit Care. 2005;11(1):63-68.
39.Le Bourdelles G, Viires N, Boczkowski J, Seta N, Pavlovic D, Aubier M. Effects of mechanical ventilation on diaphragmatic contractile properties in rats. Am J Respir Crit Care Med. 1994;149(6):1539-1544.
40.Powers SK, Shanely RA, Coombes JS, et al. Mechanical ventilation results in progressive contractile dysfunction in the diaphragm. J Appl Physiol. 2002;92(5):1851-1858.
41.Shanely RA, Zergeroglu MA, Lennon SL, et al. Mechanical ventilation-induced diaphragmatic atrophy is associated with oxidative injury and increased proteolytic activity. Am J Respir Crit Care Med. 2002;166(10):1369-1374.
42.Sassoon CS, Zhu E, Caiozzo VJ. Assist-control mechanical ventilation attenuates ventilator-induced diaphragmatic dysfunction. Am J Respir Crit Care Med. 2004;170(6):626-632.
43.Downs JB, Perkins HM, Modell JH. Intermittent mandatory ventilation. An evaluation. Arch Surg. 1974;109(4):519-523.
44.Downs JB, Klein EFJ, Desautels D, Modell JH, Kirby RR. Intermittent mandatory ventilation: a new approach to weaning patients from mechanical ventilators. Chest. 1973;64(3):331-335.
45.Marini JJ, Smith TC, Lamb VJ. External work output and force generation during synchronized intermittent mechanical ventilation. Effect of machine assistance on breathing effort. Am Rev Respir Dis. 1988;138(5):1169-1179.
46.Imsand C, Feihl F, Perret C, Fitting JW. Regulation of inspiratory neuromuscular output during synchronized intermittent mechanical ventilation. Anesthesiology. 1994;80(1):13-22.
47.Kallet RH, Campbell AR, Dicker RA, Katz JA, Mackersie RC. Work of breathing during lung-protective ventilation in patients with acute lung injury and acute respiratory distress syndrome: a comparison between volume and pressure-regulated breathing modes. Respir Care. 2005;50(12):1623-1631.
48.Shelledy DC, Peters JI. Initiating and adjusting ventilatory support. In: Wilkins RL, Stoller JK, Scanlan CL, eds. Egan’s Fundamentals of Respiratory Care. 8th ed. St Louis, MO: Mosby; 2003:1026.
49.Brochard L, Pluskwa F, Lemaire F. Improved efficacy of spontaneous breathing with inspiratory pressure support. Am Rev Respir Dis. 1987;136(2):411-415.
50.Esteban A, Alia I, Tobin MJ, et al. Effect of spontaneous breathing trial duration on outcome of attempts to discontinue mechanical ventilation. Spanish Lung Failure Collaborative Group. Am J Respir Crit Care Med. 1999;159(2):512-518.
51.Kollef MH, Shapiro SD, Silver P, et al. A randomized, controlled trial of protocol-directed versus physician-directed weaning from mechanical ventilation. Crit Care Med. 1997;25(4):567-574.
52.MacIntyre NR, Cook DJ, Ely EW Jr, et al. Evidence-based guidelines for weaning and discontinuing ventilatory support: a collective task force facilitated by the American College of Chest Physicians; the American Association for Respiratory Care; and the American College of Critical Care Medicine. Chest. 2001;120(suppl 6):375S-395S.
53.Rose L, Hawkins M. Airway pressure release ventilation and biphasic positive airway pressure: a systematic review of definitional criteria. Intensive Care Med. 2008;34(10):1766-1773.
54.Frawley PM, Habashi NM. Airway pressure release ventilation: theory and practice. AACN Clin Issues. 2001;12(2):234-246.
55.Frawley PM, Habashi NM. Airway pressure release ventilation and pediatrics: theory and practice. Crit Care Nurs Clin North Am. 2004;16(3):337-348.
56.Habashi NM. Other approaches to open-lung ventilation: airway pressure release ventilation. Crit Care Med. 2005;33(suppl 3):S228-S240.
57.Rathgeber J, Schorn B, Falk V, Kazmaier S, Spiegel T, Burchardi H. The influence of controlled mandatory ventilation (CMV), intermittent mandatory ventilation (IMV) and biphasic intermittent positive airway pressure (BIPAP) on duration of intubation and consumption of analgesics and sedatives. A prospective analysis in 596 patients following adult cardiac surgery. Eur J Anaesthesiol. 1997;14(6):576-582.
58.Putensen C, Mutz NJ, Putensen-Himmer G, Zinserling J. Spontaneous breathing during ventilatory support improves ventilation-perfusion distributions in patients with acute respiratory distress syndrome. Am J Respir Crit Care Med. 1999;159(4, pt 1):1241-1248.
59.Esteban A, Frutos F, Tobin MJ, et al. A comparison of four methods of weaning patients from mechanical ventilation. Spanish Lung Failure Collaborative Group. N Engl J Med. 1995;332(6):345-350.
60.Hendra KP, Celli BR. Weaning from mechanical ventilation. Int Anesthesiol Clin. 1999;37(3):127-143.
61.Marini J. Mechanical ventilation: physiological considerations and newer ventilatory techniques. In: Fishman A, ed. Fishman’s pulmonary diseases and disorders. 3rd ed. New York: McGraw-Hill; 1998:2714.
62.Bianchi L, Foglio K, Pagani M, Vitacca M, Rossi A, Ambrosino N. Effects of proportional assist ventilation on exercise tolerance in COPD patients with chronic hypercapnia. Eur Respir J. 1998;11(2):422-427.
63.Grasso S, Puntillo F, Mascia L, et al. Compensation for increase in respiratory workload during mechanical ventilation. Pressure-support versus proportional-assist ventilation. Am J Respir Crit Care Med. 2000;161(3, pt 1):819-826.
64.Giannouli E, Webster K, Roberts D, Younes M. Response of ventilator-dependent patients to different levels of pressure support and proportional assist. Am J Respir Crit Care Med. 1999;159(6):1716-1725.
65.Liesching T, Kwok H, Hill NS. Acute applications of noninvasive positive pressure ventilation. Chest. 2003;124(2):699-713.
66.Aldrich T, Rochester D. The lungs and neuromuscular diseases. In: Murray J, Nadel J, eds. Textbook of Respiratory Medicine. 2nd ed. Philadelphia: WB Saunders; 1994:2648.
67.Kotloff R. Acute respiratory failure in the surgical patient. In: Fishman A, ed. Fishman’s Pulmonary Diseases and Disorders. 3rd ed. New York: McGraw-Hill; 1998:2603.
68.Schwab R, Goldberg A, Poack A. Sleep apnea syndromes. In: Fishman A, ed. Fishman’s Pulmonary Diseases and Disorders. 3rd ed. New York: Mc Graw-Hill; 1998:1627.
69.Criner GJ, Brennan K, Travaline JM, Kreimer D. Efficacy and compliance with noninvasive positive pressure ventilation in patients with chronic respiratory failure. Chest. 1999;116(3):667-675.
70.Dellinger RP. Inhaled nitric oxide versus prone positioning in acute respiratory distress syndrome. Crit Care Med. 2000;28(2):572-574.
71.Dupont H, Mentec H, Cheval C, Moine P, Fierobe L, Timsit JF. Short-term effect of inhaled nitric oxide and prone positioning on gas exchange in patients with severe acute respiratory distress syndrome. Crit Care Med. 2000;28(2):304-308.
72.Deja M, Hommel M, Weber-Carstens S, et al. Evidence-based therapy of severe acute respiratory distress syndrome: an algorithm-guided approach. J Int Med Res. 2008;36(2):211-221.
73.Rico FR, Cheng JD, Gestring ML, Piotrowski ES. Mechanical ventilation strategies in massive chest trauma. Crit Care Clin. 2007;23(2):299-315, xi.
74.Alsaghir AH, Martin CM. Effect of prone positioning in patients with acute respiratory distress syndrome: a meta-analysis. Crit Care Med. 2008;36(2):603-609.
75.Abroug F, Ouanes-Besbes L, Elatrous S, Brochard L. The effect of prone positioning in acute respiratory distress syndrome or acute lung injury: a meta-analysis. Areas of uncertainty and recommendations for research. Intensive Care Med. 2008;34(6):1002-1011.
76.Galiatsou E, Kostanti E, Svarna E, et al. Prone position augments recruitment and prevents alveolar overinflation in acute lung injury. Am J Respir Crit Care Med. 2006;174(2):187-197.
77.Fernandez R, Trenchs X, Klamburg J, et al. Prone positioning in acute respiratory distress syndrome: a multicenter randomized clinical trial. Intensive Care Med. 2008:;34(8):1487-1491.
78.Morris AH, Wallace CJ, Menlove RL, et al. Randomized clinical trial of pressure-controlled inverse ratio ventilation and extracorporeal CO2 removal for adult respiratory distress syndrome. Am J Respir Crit Care Med. 1994;149(2, Pt 1):295-305.
79.Zapol WM, Snider MT, Hill JD, et al. Extracorporeal membrane oxygenation in severe acute respiratory failure. A randomized prospective study. JAMA. 1979;242(20):2193-2196.
80.Rich PB, Awad SS, Crotti S, Hirschl RB, Bartlett RH, Schreiner RJ. A prospective comparison of atrio-femoral and femoro-atrial flow in adult venovenous extracorporeal life support. J Thorac Cardiovasc Surg. 1998;116(4):628-632.
81.Rich PB, Rock P. Extracorporeal life suppport for severe adult respiratory failure. Curr Opin Anaesthesiol. 2003;16:105-111.
82.Hsu CW, Lee DL, Lin SL, Sun SF, Chang HW. The initial response to inhaled nitric oxide treatment for intensive care unit patients with acute respiratory distress syndrome. Respiration. 2008;75(3):288-295.
83.Mays LC, Eckert S. Synchronous independent lung ventilation. Dimens Crit Care Nurs. 1994;13(5):249-255.
84.Ely EW, Baker AM, Evans GW, Haponik EF. The prognostic significance of passing a daily screen of weaning parameters. Intensive Care Med. 1999;25(6):581-587.
85.Brochard L, Rauss A, Benito S, et al. Comparison of three methods of gradual withdrawal from ventilatory support during weaning from mechanical ventilation. Am J Respir Crit Care Med. 1994;150(4):896-903.
86.Martin UJ, Hincapie L, Nimchuk M, Gaughan J, Criner GJ. Impact of whole-body rehabilitation in patients receiving chronic mechanical ventilation. Crit Care Med. 2005;33(10):2259-2265.
87.Seidel H, Ball J, Dains J, Bendeict G. Mosby’s Guide to Physical Examination. 3rd ed. Baltimore, MD: Mosby; 1995:358.
88.DeVivo MJ, Black KJ, Stover SL. Causes of death during the first 12 years after spinal cord injury. Arch Phys Med Rehabil. 1993;74(3):248-254.
89.Lisboa T, Rello J. Diagnosis of ventilator-associated pneumonia: is there a gold standard and a simple approach? Curr Opin Infect Dis. 2008;21:174-178.
90.Wunderink RG. Mortality and the diagnosis of ventilator-associated pneumonia: a new direction. Am J Respir Crit Care Med. 1998;157(2):349-350.
91.Sterling T, Ho E, Brehm W. Diagnosis and treatment of ventilator-associated pneumonia—Impact on survival. Chest. 1996;110:1025-1034.
92.Rello J, Ausina V, Ricart M, Castella J, Prats G. Impact of previous antimicrobial therapy on the etiology and outcome of ventilator-associated pneumonia. Chest. 1993;104(4):1230-1235.
93.Rello J, Quintana E, Ausina V, et al. Incidence, etiology, and outcome of nosocomial pneumonia in mechanically ventilated patients. Chest. 1991;100(2):439-444.
94.Fagon J, Chastre J, Hance A. Nosocomial pneumonia in ventilated patients: a cohort study evaluating attributable mortality and hospital stay. Am J Med. 1993;94:281.
95.Joshi M, Ciesla N, Caplan E. Diagnosis of pneumonia in critically ill patients. Chest. 1988;94:4s.
96.Donald KJ, Robertson VL, Tsebelis K. Setting safe and effective suction pressure: the effect of using a manometer in the suction circuit. Intensive Care Med. 2000;26(1):15.
97.Subirana M, Sola I. Closed tracheal suction systems versus open tracheal suction systems for mechanically ventilated adult patients. Cochrane Datanbase Syst Rev. 2007: Oct 17;(4):CD004581. Review
98.Siempos II, Vardakas KZ, Falagas ME. Closed tracheal suction systems for prevention of ventilator-associated pneumonia. Br J Anaesth. 2008;100(3):299-306.
99.Subirana M, Sola I, Benito S. Closed tracheal suction systems versus open tracheal suction systems for mechanically ventilated adult patients. Anesth Analg. 2008;106(4):1326.
100.Darvas JA, Leesa G Hawkins LG. The closed tracheal suction catheter: 24 hour or 48 hour change? Aust Crit Care. 2003:86-92.
101.Celik SA, Kanan N. A current conflict: use of isotonic sodium chloride solution on endotracheal suctioning in critically ill patients. Dimens Crit Care Nurs. 2006:11-14.
102.Raymond J. Normal saline instillation before suctioning: helpful or harmful? A review of the literature. Am J Crit Care. 1995;4(4):267-271.
103.Akqui S, Akyolcu N. Effects of normal saline on endotracheal suctioning. J Clin Nurs. 2002;11(1):826-830.
104.Rauen CA, Chulay M, Bridges E, Vollman KM, Arbour R. Seven evidence-based practice habits: putting some sacred cows out to pasture. Crit Care Nurse. 2008;28(2):98-124.
105.Golden J, Wank K, Keith F. Bronchoscopy, lung biopsy, and other diagnostic procedures. In: Murray J, Nadel J, eds. Textbook of Respiratory Medicine. 2nd ed. Philadelphia, PA: WB Saunders; 1994:738-739.
106.Jelic S, Cunningham JA, Factor P. Clinical review: airway hygiene in the intensive care unit. Crit Care. 2008;12(2):209.
107.Ciesla N. Chest physical therapy for the adult intensive care unit trauma patient. Phys Ther Pract. 1994;3:102-105.
108.Ciesla N, Klemic N, Imle PC. Chest physical therapy to the patient with multiple trauma. Two case studies. Phys Ther. 1981;61(2):202-205.
109.Ciesla ND. Chest physical therapy for patients in the intensive care unit. Phys Ther. 1996;76(6):609-625.
110.Mars M, Ciesla N. Chest physical therapy may have prevented bronchoscopy and exploratory laparotomy: a case report. Cardiopulm Phys Ther. 1993;4:4-5.
111.Marini J, Pierson D, Hudson L. Comparison of fiberoptic bronchoscopy and respiratory therapy [letter]. Am Rev Respir Dis. 1984;126:368.
112.Marini JJ, Pierson DJ, Hudson LD. Acute lobar atelectasis: a prospective comparison of fiberoptic bronchoscopy and respiratory therapy. Am Rev Respir Dis. 1979;119(6):971-978.
113.Mackenzie CF, Shin B, McAslan TC. Chest physiotherapy: the effect on arterial oxygenation. Anesth Analg. 1978;57(1):28-30.
114.Mackenzie CF, Imle PC, Ciesla N. Chest Physiotherapy in the Intensive Care Unit. 2nd ed. Baltimore, MD: Williams & Wilkins; 1989.
115.Santos PM, Afrassiabi A, Weymuller EA Jr. Risk factors associated with prolonged intubation and laryngeal injury. Otolaryngol Head Neck Surg. 1994;111(4):453-459.
116.Maury E, Guglielminotti J, Alzieu M, Qureshi T, Guidet B, Offenstadt G. How to identify patients with no risk for postextubation stridor? J Crit Care. 2004;19(1):23-28.
117.Miller RL, Cole RP. Association between reduced cuff leak volume and postextubation stridor. Chest. 1996;110(4):1035-1040.
118.Houck JR, Keamy MF III, McDonough JM. Effect of helium concentration on experimental upper airway obstruction. Ann Otol Rhinol Laryngol. 1990;99(7, Pt 1):556-561.
119.Schmidt G, Hall J, Wood L. Management of the ventilated patient. In: Murray J, Nadel J, eds. Textbook of Respiratory Medicine. 2nd ed. Philadelphia, PA: WB Saunders; 1994:2640-2644,2648.
120.Sengupta P, Sessler DI, Maglinger P, et al. Endotracheal tube cuff pressure in three hospitals, and the volume required to produce an appropriate cuff pressure. BMC Anesthesiol. 2004;4(1):8.
121.Hess DR. Tracheostomy tubes and related appliances. Respir Care. 2005;50(4):497-510.
122.Shelly MP. The humidification and filtration functions of the airways. Respir Care Clin N Am. 2006;12(2):139-148.
123.Rathgeber J. Devices used to humidify respired gases. Respir Care Clin N Am. 2006;12(2):165-182.
124.Kola A, Eckmanns T, Gastmeier P. Efficacy of heat and moisture exchangers in preventing ventilator-associated pneumonia: meta-analysis of randomized controlled trials. Intensive Care Med. 2005;31(1):5-11.
125.Lorente L, Lecuona M, Jimenez A, Mora ML, Sierra A. Ventilator-associated pneumonia using a heated humidifier or a heat and moisture exchanger: a randomized controlled trial [ISRCTN88724583]. Crit Care. 2006;10(4):R116.
126.Jubran A. Critical illness and mechanical ventilation: effects on the diaphragm. Respir Care. 2006;51(9):1054-1061; discussion 1062-1064.
127.Thille AW, Cabello B, Galia F, Lyazidi A, Brochard L. Reduction of patient-ventilator asynchrony by reducing tidal volume during pressure-support ventilation. Intensive Care Med. 2008;34(8):1477-1486.
128.Bissett B, Leditschke IA. Inspiratory muscle training to enhance weaning from mechanical ventilation. Anaesth Intensive Care. 2007;35(5):776-779.
129.Sprague SS, Hopkins PD. Use of inspiratory strength training to wean six patients who were ventilator-dependent. Phys Ther. 2003;83(2):171-181.
130.Martin AD, Davenport PD, Franceschi AC, Harman E. Use of inspiratory muscle strength training to facilitate ventilator weaning: a series of 10 consecutive patients. Chest. 2002;122(1):192-196.
131.Abelson H, Brewer, K. Inspiratory muscle training in the mechanically ventilated patient. Physiother Can. 1987;39:305-307.
132.Caruso P, Denari SD, Ruiz SA, et al. Inspiratory muscle training is ineffective in mechanically ventilated critically ill patients. Clinics. 2005;60(6):479-484.
133.Lin KH, Lai YL, Wu HD, Wang TQ, Wang YH. Cough threshold in people with spinal cord injuries. Phys Ther. 1999;79(11):1026-1031.
134.Liaw MY, Lin MC, Cheng PT, Wong MK, Tang FT. Resistive inspiratory muscle training: its effectiveness in patients with acute complete cervical cord injury. Arch Phys Med Rehabil. 2000;81(6):752-756.
135.Brooks D, O’Brien K, Geddes EL, Crowe J, Reid WD. Is inspiratory muscle training effective for individuals with cervical spinal cord injury? A qualitative systematic review. Clin Rehabil. 2005;19(3):237-246.
136.Lin KH, Chuang CC, Wu HD, Chang CW, Kou YR. Abdominal weight and inspiratory resistance: their immediate effects on inspiratory muscle functions during maximal voluntary breathing in chronic tetraplegic patients. Arch Phys Med Rehabil. 1999;80(7):741-745.
137.Rutchik A, Weissman AR, Almenoff PL, Spungen AM, Bauman WA, Grimm DR. Resistive inspiratory muscle training in subjects with chronic cervical spinal cord injury. Arch Phys Med Rehabil. 1998;79(3):293-297.
138.Mueller G, Perret C, Spengler CM. Optimal intensity for respiratory muscle endurance training in patients with spinal cord injury. J Rehabil Med. 2006;38(6):381-386.
139.Derrickson J, Ciesla N, Simpson N, Imle PC. A comparison of two breathing exercise programs for patients with quadriplegia. Phys Ther. 1992;72(11):763-769.
140.Estenne M, Knoop C, Vanvaerenbergh J, Heilporn A, De Troyer A. The effect of pectoralis muscle training in tetraplegic subjects. Am Rev Respir Dis. 1989;139(5):1218-1222.
141.Fujiwara T, Hara Y, Chino N. Expiratory function in complete tetraplegics: study of spirometry, maximal expiratory pressure, and muscle activity of pectoralis major and latissimus dorsi muscles. Am J Phys Med Rehabil. 1999;78(5):464-469.
142.Kang SW, Shin JC, Park CI, Moon JH, Rha DW, Cho DH. Relationship between inspiratory muscle strength and cough capacity in cervical spinal cord injured patients. Spinal Cord. 2006;44(4):242-248.
143.Marini JJ, Gattinoni L. Ventilatory management of acute respiratory distress syndrome: a consensus of two. Crit Care Med. 2004;32:250-255.
144.Slutsky AS. Lung injury caused by mechanical ventilation. Chest. 1999;116(suppl 1):9S-15S.
145.Carney D, DiRocco J, Nieman G. Dynamic alveolar mechanics and ventilator-induced lung injury. Crit Care Med. 2005;33(suppl 3):S122-S128.
146.Halter JM, Steinberg JM, Gatto LA, et al. Effect of positive end-expiratory pressure and tidal volume on lung injury induced by alveolar instability. Crit Care. 2007;11(1):R20.
147.Yilmaz M, Gajic O. Optimal ventilator settings in acute lung injury and acute respiratory distress syndrome. Eur J Anaesthesiol. 2008;25(2):89-96.
148.Steinberg KP, Kacmarek RM. Respiratory controversies in the critical care setting. Should tidal volume be 6 mL/kg predicted body weight in virtually all patients with acute respiratory failure? Respir Care. 2007;52(5):556-564; discussion 565-567.
149.Checkley W, Brower R, Korpak A, Thompson BT; Acute Respiratory Distress Syndrome Network Investigators. Effects of a clinical trial on mechanical ventilation practices in patients with acute lung injury. Am J Respir Crit Care Med. 2008;177(11): 1215-1222.
150.Hickling KG, Walsh J, Henderson S, Jackson R. Low mortality rate in adult respiratory distress syndrome using low-volume, pressure-limited ventilation with permissive hypercapnia: a prospective study. Crit Care Med. 1994;22(10):1568-1578.
151.Bendixen H. Editorial comment. Arch Surg. 1974:541.
152.Vilke GM, Chan TC, Neuman T, Clausen JL. Spirometry in normal subjects in sitting, prone, and supine positions. Respir Care. 2000;45(4):407-410.
153.Behrakis PK, Baydur A, Jaeger MJ, Milic-Emili J. Lung mechanics in sitting and horizontal body positions. Chest. 1983;83(4):643-646.
154.Vellody VP, Nassery M, Druz WS, Sharp JT. Effects of body position change on thoracoabdominal motion. J Appl Physiol. 1978;45(4):581-589.
155.Allen SM, Hunt B, Green M. Fall in vital capacity with posture. Br J Dis Chest. 1985;79(3):267-271.
156.Loveridge BM, Dubo HI. Breathing pattern in chronic quadriplegia. Arch Phys Med Rehabil. 1990;71(7):495-499.
157.Ibanez J, Raurich JM. Normal values of functional residual capacity in the sitting and supine positions. Intensive Care Med. 1982;8(4):173-177.
158.Anke A, Aksnes AK, Stanghelle JK, Hjeltnes N. Lung volumes in tetraplegic patients according to cervical spinal cord injury level. Scand J Rehabil Med. 1993;25(2):73-77.
159.Haas A, Lowman EW, Bergofsky EH. Impairment of respiration after spinal cord injury. Arch Phys Med Rehabil. 1965;46:399-405.
160.Estenne M, De Troyer A. Mechanism of the postural dependence of vital capacity in tetraplegic subjects. Am Rev Respir Dis. 1987;135(2):367-371.
161.Fugl-Meyer AR. Effects of respiratory muscle paralysis in tetraplegic and paraplegic patients. Scand J Rehabil Med. 1971;3(4):141-150.
162.Cameron GS, Scott JW, Jousse AT, Botterell EH. Diaphragmatic respiration in the quadriplegic patient and the effect of position on his vital capacity. Ann Surg. 1955;141(4):451-456.
163.Maloney FP. Pulmonary function in quadriplegia: effects of a corset. Arch Phys Med Rehabil. 1979;60(6):261-265.
164.McCool FD, Pichurko BM, Slutsky AS, Sarkarati M, Rossier A, Brown R. Changes in lung volume and rib cage configuration with abdominal binding in quadriplegia. J Appl Physiol. 1986;60(4):1198-1202.
165.Goldman JM, Rose LS, Williams SJ, Silver JR, Denison DM. Effect of abdominal binders on breathing in tetraplegic patients. Thorax. 1986;41(12):940-945.
166.MacLean D, Drummond G, Macpherson C, McLaren G, Prescott R. Maximum expiratory airflow during chest physiotherapy on ventilated patients before and after the application of an abdominal binder. Intensive Care Med. 1989;15(6):396-399.
167.Bodin P, Fagevik Olsen M, Bake B, Kreuter M. Effects of abdominal binding on breathing patterns during breathing exercises in persons with tetraplegia. Spinal Cord. 2005;43(2):117-122.
168.Estenne M, Pinet C, De Troyer A. Abdominal muscle strength in patients with tetraplegia. Am J Respir Crit Care Med. 2000; 161(3, Pt 1):707-712.
169.Hopkins RO, Jackson JC. Long-term neurocognitive function after critical illness. Chest. 2006;130(3):869-878.
170.Hopkins RO, Weaver LK, Pope D, Orme JF, Bigler ED, Larson-LOHR V. Neuropsychological sequelae and impaired health status in survivors of severe acute respiratory distress syndrome. Am J Respir Crit Care Med. 1999;160(1):50-56.
171.Zubeck J, Wilgosh L. Prolonged immobilization of the body: changes in performance and in the electroencephalogram. Science. 1963;140:306.
172.Hammer R, Kenan E. The psychological aspects of immobilization. In: Steinberg F, ed. The immobilized Patient: Functional Pathology and Management. New York: Plenum Medical Company; 1980:123-149.
173.Greenleaf J, Kozlowski S. Psychological consequences of reduced physical activity during bedrest. Exerc Sport Sci Rev. 1982;10:84.
174.Ely EW, Inouye SK, Bernard GR, et al. Delirium in mechanically ventilated patients: validity and reliability of the confusion assessment method for the intensive care unit (CAM-ICU). JAMA. 2001;286(21):2703-2710.
175.Plaschke K, von Haken R, Scholz M, et al. Comparison of the confusion assessment method for the intensive care unit (CAM-ICU) with the Intensive Care Delirium Screening Checklist (ICDSC) for delirium in critical care patients gives high agreement rate(s). Intensive Care Med. 2008;34(3):431-436.
176.Soja SL, Pandharipande PP, Fleming SB, et al. Implementation, reliability testing, and compliance monitoring of the Confusion Assessment Method for the intensive care unit in trauma patients. Intensive Care Med. 2008;34(7):1263-1268.
177.Ely EW, Truman B, Shintani A, et al. Monitoring sedation status over time in ICU patients: reliability and validity of the Richmond Agitation-Sedation Scale (RASS). JAMA. 2003;289(22):2983-2991.
178.Teasdale G, Jennette B. Assessment of coma and impaired consciousness. A practical scale. Lancet. 1974;2:81-83.
179.Sessler CN, Jo Grap M, Ramsay MA. Evaluating and monitoring analgesia and sedation in the intensive care unit. Crit Care. 2008;12(suppl 3):S2.
180.Friman G. Effect of clinical bed rest for seven days on physical performance. Acta Med Scand. 1979;205(5):389-393.
181.Bassey EJ, Fentem PH. Extent of deterioration in physical condition during postoperative bed rest and its reversal by rehabilitation. Br Med J. 1974;4(5938):194-196.
182.Ruiz Bailen M, Aguayo de Hoyos E, Lopez Martnez A, et al. Reversible myocardial dysfunction, a possible complication in critically ill patients without heart disease. J Crit Care. 2003;18(4):245-252.
183.Attia J, Ray JG, Cook DJ, Douketis J, Ginsberg JS, Geerts WH. Deep vein thrombosis and its prevention in critically ill adults. Arch Intern Med. 2001;161(10):1268-1279.
184.Geerts WH, Bergqvisr D, Pineo GF, et al. Prevention of deep venous thromboembolism: ACCP evidence-based clinical practice guidelines. Chest. 2008;133(6):381s-453s.
185.Tepper S, McKeough D. Deep vein thrombosis: risks, diagnosis, treatment interventions and prevention. Acute Care Perspect. 2000;9-11.
186.Anand S, Wells P, Hunt D, et al. Does this patient have deep vein thrombosis? JAMA. 1998;279:1094-1099.
187.Kearon C, Kahn SR, Giancardo A, Goldhaber S, Raskob GE, Comerota AJ. Antithrombotic therapy for venous thromboemboloc disease: ACCP evidence-based clinical practice guidelines. Chest. 2008;133:454S-545S.
188.Junger M, Diehm C, Storiko H, et al. Mobilization versus immobilization in the treatment of acute proximal deep venous thrombosis: a prospective, randomized, open, multicentre trial. Curr Med Res Opin. 2006;22(3):593-602.
189.Partsch H. Immediate ambulation and leg compression in the treatment of deep vein thrombosis. Dis Mon. 2005;51(2–3):135-140.
190.Stannard JP, Lopez-Ben RR, Volgas DA, et al. Prophylaxis against deep-vein thrombosis following trauma: a prospective, randomized comparison of mechanical and pharmacologic prophylaxis. J Bone Joint Surg Am. 2006;88(2):261-266.
191.Hirsh J, Guyatt G, Albers G, Harrington R, Schunemann HJ. Executive summary: ACCP evidence-based clinical practice guidelines. Chest. 2008;133(6):71S.
192.Prandoni P, Lensing AW, Prins MH, et al. Below-knee elastic compression stockings to prevent the post-thrombotic syndrome: a randomized, controlled trial. Ann Intern Med. 2004;141(4):249-256.
193.Segal JB, Streiff MB, Hofmann LV, Thornton K, Bass EB. Management of venous thromboembolism: a systematic review for a practice guideline. Ann Intern Med. 2007;146(3):211-222.
194.Giannoudis PV, Pountos I, Pape HC, Patel JV. Safety and efficacy of vena cava filters in trauma patients. Injury. 2007;38(1):7-18.
195.Paton BL, Jacobs DG, Heniford BT, Kercher KW, Zerey M, Sing RF. Nine-year experience with insertion of vena cava filters in the intensive care unit. Am J Surg. 2006;192(6):795-800.
196.Aschwanden M, Labs KH, Engel H, et al. Acute deep vein thrombosis: early mobilization does not increase the frequency of pulmonary embolism. Thromb Haemost. 2001;85(1):42-46.
197.Kahn SR, Shrier I, Kearon C. Physical activity in patients with deep venous thrombosis: a systematic review. Thromb Res. 2008:122(6):763-73.
198.Trujillo-Santos J, Perea-Milla E, Jimenez-Puente A, et al. Bed rest or ambulation in the initial treatment of patients with acute deep vein thrombosis or pulmonary embolism: findings from the RIETE registry. Chest. 2005;127(5):1631-1636.
199.Phillips SM, Gallagher M, Buchan H. Use graduated compression stockings postoperatively to prevent deep vein thrombosis. BMJ. 2008;336(7650):943-944.
200.Palevsky H, Kelley M, Fishman A. Pulmonary thromboembolotic disease. In: Fishman A, ed. Fishmans Pulmonary Diseases and Disorders. 3rd ed. New York: McGraw-Hill; 1998:1310-1325.
201.Cook D, McMullin J, Hodder R, et al. Prevention and diagnosis of venous thromboembolism in critically ill patients: a Canadian survey. Crit Care. 2001;5(6):336-342.
202.Ferrando AA, Lane HW, Stuart CA, Davis-Street J, Wolfe RR. Prolonged bed rest decreases skeletal muscle and whole body protein synthesis. Am J Physiol. 1996;270(4, Pt 1):E627-E633.
203.Berg HE, Larsson L, Tesch PA. Lower limb skeletal muscle function after 6 wk of bed rest. J Appl Physiol. 1997;82(1):182-188.
204.Bloomfield SA. Changes in musculoskeletal structure and function with prolonged bed rest. Med Sci Sports Exerc. 1997;29(2):197-206.
205.Dellestable F, Voltz C, Mariot J, Perrier JF, Gaucher A. Heterotopic ossification complicating long-term sedation. Br J Rheumatol. 1996;35(7):700-701.
206.Evans EB. Heterotopic bone formation in thermal burns. Clin Orthop Relat Res. 1991(263):94-101.
207.Garland DE. Clinical observations on fractures and heterotopic ossification in the spinal cord and traumatic brain injured populations. Clin Orthop Relat Res. 1988(233):86-101.
208.Pape HC, Lehmann U, van Griensven M, Gansslen A, von Glinski S, Krettek C. Heterotopic ossifications in patients after severe blunt trauma with and without head trauma: incidence and patterns of distribution. J Orthop Trauma. 2001;15(4):229-237.
209.Sugita A, Hashimoto J, Maeda A, et al. Heterotopic ossification in bilateral knee and hip joints after long-term sedation. J Bone Miner Metab. 2005;23(4):329-332.
210.Clavet H, Hebert PC, Fergusson D, Doucette S, Trudel G. Joint contracture following prolonged stay in the intensive care unit. CMAJ. 2008;178(6):691-697.
211.Yarkony GM, Bass LM, Keenan V III, Meyer PR Jr. Contractures complicating spinal cord injury: incidence and comparison between spinal cord centre and general hospital acute care. Paraplegia. 1985;23(5):265-271.
212.Bryden AM, Kilgore KL, Lind BB, Yu DT. Triceps denervation as a predictor of elbow flexion contractures in C5 and C6 tetraplegia. Arch Phys Med Rehabil. 2004;85(11):1880-1885.
213.Grover J, Gellman H, Waters RL. The effect of a flexion contracture of the elbow on the ability to transfer in patients who have quadriplegia at the sixth cervical level. J Bone Joint Surg Am. 1996;78(9):1397-1400.
214.De Jonghe B, Sharshar T, Lefaucheur JP, et al. Paresis acquired in the intensive care unit: a prospective multicenter study. JAMA. 2002;288(22):2859-2867.
215.Schweickert WD, Hall J. ICU-acquired weakness. Chest. 2007;131(5):1541-1549.
216.Hermans G, Wilmer A, Meersseman W, et al. Impact of intensive insulin therapy on neuromuscular complications and ventilator dependency in the medical intensive care unit. Am J Respir Crit Care Med. 2007;175(5):480-489.
217.Hermans G, De Jonghe B, Bruyninckx F, Van den Berghe G. Interventions for preventing critical illness polyneuropathy and critical illness myopathy. Cochrane Database Syst Rev. 2009;1:CD006832. 10.1002/14651858.CD006832.pub2.
218.Leatherman JW, Fluegel WL, David WS, Davies SF, Iber C. Muscle weakness in mechanically ventilated patients with severe asthma. Am J Respir Crit Care Med. 1996;153(5):1686-1690.
219.Segredo V, Caldwell JE, Matthay MA, Sharma ML, Gruenke LD, Miller RD. Persistent paralysis in critically ill patients after long-term administration of vecuronium. N Engl J Med. 1992;327(8):524-528.
220.De Jonghe B, Lacherade JC, Durand MC, Sharshar T. Critical illness neuromuscular syndromes. Crit Care Clin. 2007;23(1):55-69.
221.Angel MJ, Bril V, Shannon P, Herridge MS. Neuromuscular function in survivors of the acute respiratory distress syndrome. Can J Neurol Sci. 2007;34(4):427-432.
222.Garnacho-Montero J, Amaya-Villar R, Garcia-Garmendia JL, Madrazo-Osuna J, Ortiz-Leyba C. Effect of critical illness polyneuropathy on the withdrawal from mechanical ventilation and the length of stay in septic patients. Crit Care Med. 2005;33(2):349-354.
223.De Jonghe B, Bastuji-Garin S, Sharshar T, Outin H, Brochard L. Does ICU-acquired paresis lengthen weaning from mechanical ventilation? Intensive Care Med. 2004;30(6):1117-1121.
224.Latronico N, Shehu I, Seghelini E. Neuromuscular sequelae of critical illness. Curr Opin Crit Care. 2005;11(4):381-390.
225.Macciocchi S, Seel RT, Thompson N, Byams R, Bowman B. Spinal cord injury and co-occurring traumatic brain injury: assessment and incidence. Arch Phys Med Rehabil. 2008;89(7):1350-1357.
226.Ciesla N, Ridriequez A, Anderson P, Norton B. The incidence of extrapleural hematomas in patients with rib fractures. Phys Ther. 1987;67:766.
227.Imle P. Percussion and vibration. In: Mackenzie C, ed. Chest Physiotherapy in the Intensive Care Unit. 2nd ed. Baltimore, MD: Williams & Wilkins; 1989:141.
228.Kigin C. Advances in chest physical therapy. In: O’Donohue WJ, ed. Current Advances in Respiratory Care. Park Ridge, IL: American College of Chest Physicians; 1984:145.
229.Hopkins RO, Spuhler VJ, Thomsen GE. Transforming ICU culture to facilitate early mobility. Crit Care Clin. 2007;23(1):81-96.
230.Burns JR, Jones FL. Letter: Early ambulation of patients requiring ventilatory assistance. Chest. 1975;68(4):608.
231.King J, Crowe J. Mobilization practices in Canadian critical care units. Physiotherapy Canada. 1998(Summer):206-211.
232.Morris PE, Herridge MS. Early intensive care unit mobility: future directions. Crit Care Clin. 2007;23(1):97-110.
233.Stiller K. Physiotherapy in intensive care: towards an evidence-based practice. Chest. 2000;118(6):1801-1813.
234.Heyland DK, Groll D, Caeser M. Survivors of acute respiratory distress syndrome: relationship between pulmonary dysfunction and long-term health-related quality of life. Crit Care Med. 2005;33(7):1549-1556.
235.Dowdy DW, Eid MP, Dennison CR, et al. Quality of life after acute respiratory distress syndrome: a meta-analysis. Intensive Care Med. 2006;32(8):1115-1124.
236.Stevens RD, Dowdy DW, Michaels RK, Mendez-Tellez PA, Pronovost PJ, Needham DM. Neuromuscular dysfunction acquired in critical illness: a systematic review. Intensive Care Med. 2007;33(11):1876-1891.
237.MacIntyre N. Evidence-based guidelines for weaning and discontinuing ventilatory support. Chest. 2001;120(6):375S-395S.
238.De Jonghe B, Lacherade JC, Durand MC, Sharshar T. Critical illness neuromuscular syndromes. Crit Care Clin. 2006;22(4): 805-818.