W. Greg Leader
LEARNING OBJECTIVES
Upon completion of the chapter, the reader will be able to:
1. Discuss the economic and health burden caused by asthma.
2. Explain the pathophysiology of asthma.
3. Describe the clinical presentation of acute and chronic asthma.
4. Identify factors that affect asthma severity.
5. Identify the goals of asthma management.
6. Classify asthma severity based on impairment due to asthma and future risk for negative outcomes due to asthma.
7. Recommend environmental control strategies for patients with identified allergies.
8. Educate patients on the use of inhaled drug delivery devices, peak flow meters, and asthma education plans.
9. Develop a therapeutic plan for patients with chronic asthma that maximizes patient response while minimizing adverse drug events and other drug-related problems.
10. Evaluate current asthma control and make therapeutic changes when necessary.
11. Develop a therapeutic plan for treating patients with acute asthma.
KEY CONCEPTS
![]() Asthma is a complex disease that presents in a heterogeneous manner.
Asthma is a complex disease that presents in a heterogeneous manner.
![]() Asthma is the most prevalent chronic disease of childhood, and it causes significant morbidity and mortality in both adults and children.
Asthma is the most prevalent chronic disease of childhood, and it causes significant morbidity and mortality in both adults and children.
![]() Asthma is characterized by inflammation, airway hyper-responsiveness (AHR), and airway obstruction.
Asthma is characterized by inflammation, airway hyper-responsiveness (AHR), and airway obstruction.
![]() In chronic asthma, initial classification of asthma severity is based on current disease impairment and future risk.
In chronic asthma, initial classification of asthma severity is based on current disease impairment and future risk.
![]() Direct airway administration of asthma medications through inhalation is the most efficient route and minimizes systemic adverse effects.
Direct airway administration of asthma medications through inhalation is the most efficient route and minimizes systemic adverse effects.
![]() Short-acting-inhaled β2-agonists are the most effective agents for reversing acute airway obstruction caused by bronchoconstriction and are the drugs of choice for treating acute severe asthma and symptoms of chronic asthma.
Short-acting-inhaled β2-agonists are the most effective agents for reversing acute airway obstruction caused by bronchoconstriction and are the drugs of choice for treating acute severe asthma and symptoms of chronic asthma.
![]() Inhaled corticosteroids (ICS) are the preferred therapy for all forms of persistent asthma in all age groups.
Inhaled corticosteroids (ICS) are the preferred therapy for all forms of persistent asthma in all age groups.
![]() The intensity of pharmacotherapy for chronic asthma is based on disease severity for initial therapy and level of control for subsequent therapies.
The intensity of pharmacotherapy for chronic asthma is based on disease severity for initial therapy and level of control for subsequent therapies.
![]() In acute severe asthma, early and appropriate intensification of therapy is important to resolve the exacerbation and prevent relapse and severe airflow obstruction in the future.
In acute severe asthma, early and appropriate intensification of therapy is important to resolve the exacerbation and prevent relapse and severe airflow obstruction in the future.
INTRODUCTION
In 2007, the National Heart, Lung, and Blood Institute’s (NHLBI) National Asthma Education and Prevention Program (NAEPP) updated its Guidelines for the Diagnosis and Management of Asthma.1 In this update, the Expert Panel Report-3 (EPR-3) defines asthma as “... a common chronic disorder of the airways that is complex and characterized by variable and recurring symptoms, airflow obstruction, bronchial hyperresponsiveness and underlying inflammation.”1
![]() Asthma is a complex disease that presents in a heterogeneous manner. Severity of chronic disease ranges from mild intermittent symptoms to severe and disabling disease if left untreated. Despite variances in the underlying severity of chronic asthma, all asthmatics are at risk of acute severe disease when exposed to the appropriate trigger or if inadequately treated. The NAEPP guidelines1 emphasize the importance of treating underlying airway inflammation to control asthma and reduce asthma-associated risks.
Asthma is a complex disease that presents in a heterogeneous manner. Severity of chronic disease ranges from mild intermittent symptoms to severe and disabling disease if left untreated. Despite variances in the underlying severity of chronic asthma, all asthmatics are at risk of acute severe disease when exposed to the appropriate trigger or if inadequately treated. The NAEPP guidelines1 emphasize the importance of treating underlying airway inflammation to control asthma and reduce asthma-associated risks.
EPIDEMIOLOGY AND ETIOLOGY
![]() Asthma is the most prevalent chronic disease of childhood, and it causes significant morbidity and mortality in both adults and children. Approximately 22.9 million people in the United States carry the diagnosis of asthma, and nearly 6.8 million of these are younger than 18 years of age. The highest prevalence is in children 5 to 17 years of age.2 Puerto Ricans and non-Hispanic blacks have a higher prevalence than non-Hispanic whites.2,3
Asthma is the most prevalent chronic disease of childhood, and it causes significant morbidity and mortality in both adults and children. Approximately 22.9 million people in the United States carry the diagnosis of asthma, and nearly 6.8 million of these are younger than 18 years of age. The highest prevalence is in children 5 to 17 years of age.2 Puerto Ricans and non-Hispanic blacks have a higher prevalence than non-Hispanic whites.2,3
Approximately 10.1 million workdays and 12.8 million school days are missed every year due to asthma.2 In 2005, there were 1.77 million emergency department visits and 488,594 hospital discharges related to asthma.2Children have the highest rates of emergency department visits and hospitalizations.2,3 There were approximately 3,816 asthma-related deaths in 2004. The total number of asthma deaths have decreased every year since 1999.2
Asthma is also a significant economic burden in the United States, costing $19.7 billion in 2007. Prescription drugs are the single largest direct medical expenditure and account for 42% of direct medical costs.3 Costs increase with disease severity, and it has been suggested that less than 20% of asthma patients account for over 80% of direct medical expenditures.4
Asthma results from a complex interaction of genetic and environmental factors, but the underlying cause is not well understood. The onset of asthma occurs early in life for most patients.1 There appears to be an inherited component, as the presence of asthma in a parent is a strong risk factor for development of asthma in a child. This risk increases when a family history of atopy is also present.1 The presence of atopy is a strong prognostic factor for continued asthma as an adult. Furthermore, the severity of early childhood asthma is a predictor of adult asthma severity.5
Environmental exposure also appears to be an important factor in the etiology of asthma. Patients with occupational asthma develop the disease late in life upon exposure to specific allergens in the workplace. Exposure to second-hand smoke after birth increases the risk of childhood asthma.1 Adult-onset asthma may be related to atopy, nasal polyps, aspirin sensitivity, occupational exposure, or a recurrence of childhood asthma.
PATHOPHYSIOLOGY
![]() Asthma is characterized by inflammation, airway hyper-responsiveness (AHR), and airway obstruction. Inhaled antigens induce a type 2 T-helper CD4+ (TH2) response. Antigens are taken up by antigen-presenting cells, and presentation of antigens to T-lymphocytes causes activation of the TH2 type response. This leads to B-cell production of antigen-specific immunoglobulin E (IgE) as well as proinflammatory cytokines and chemokines that recruit and activate eosinophils, neutrophils, and alveolar macrophages.6,7 Further exposure to the antigen results in cross-linking of cell-bound IgE in mast cells and basophils, causing the release of preformed inflammatory mediators such as histamine or generation of new inflammatory mediators such as leukotriene C4 and prostaglandins.8 Activation and degranulation of mast cells and basophils result in an early-phase response involving acute bronchoconstriction that lasts approximately 1 hour after allergen exposure.6 This early-phase response can be blocked by pretreatment with inhaled short-acting β2-agonists (SABA) or cromolyn.
Asthma is characterized by inflammation, airway hyper-responsiveness (AHR), and airway obstruction. Inhaled antigens induce a type 2 T-helper CD4+ (TH2) response. Antigens are taken up by antigen-presenting cells, and presentation of antigens to T-lymphocytes causes activation of the TH2 type response. This leads to B-cell production of antigen-specific immunoglobulin E (IgE) as well as proinflammatory cytokines and chemokines that recruit and activate eosinophils, neutrophils, and alveolar macrophages.6,7 Further exposure to the antigen results in cross-linking of cell-bound IgE in mast cells and basophils, causing the release of preformed inflammatory mediators such as histamine or generation of new inflammatory mediators such as leukotriene C4 and prostaglandins.8 Activation and degranulation of mast cells and basophils result in an early-phase response involving acute bronchoconstriction that lasts approximately 1 hour after allergen exposure.6 This early-phase response can be blocked by pretreatment with inhaled short-acting β2-agonists (SABA) or cromolyn.
In the late-phase response, activated airway cells release inflammatory cytokines and chemokines, thereby recruiting more inflammatory cells into the lungs. The late-phase response occurs 4 to 6 hours after the initial allergen challenge and results in a less intense bronchoconstriction as well as increased AHR and airway inflammation.6
Airway Inflammation and Hyper-responsiveness
Although the symptoms of asthma are intermittent, airway inflammation is chronic.7 Considerable variations in the pattern of inflammation may exist, resulting in phenotypic differences.1 T-lymphocytes release cytokines that coordinate eosinophilic infiltration and IgE production by B-lymphocytes.9 Mast cells, eosinophils, macrophages, neutrophils, fibroblasts, and airway smooth muscle cells are also activated in asthma. Mast cells infiltrate airway smooth muscle and bronchial epithelium and may cause mucous gland hyperplasia. Proinflammatory mediators generated during mast cell degranulation propagate the inflammatory response and contribute to AHR and airway remodeling.10
AHR is the exaggerated ability of the airways to narrow in response to a variety of stimuli. AHR is a characteristic feature of asthma and is related to airway inflammation and structural changes in the airways.1 Treatment of airway inflammation with inhaled corticosteroids (ICS) attenuates AHR in asthma but does not eliminate it.1 Clinically, AHR manifests as increased variability of airway function. Although not commonly used to diagnose asthma, AHR can be evaluated clinically using a methacholine or histamine bronchoprovocation test.
Airway Obstruction
Symptoms of airway obstruction include chest tightness, cough, and wheezing. Airway obstruction can be caused by multiple factors including airway smooth muscle constriction, airway edema, mucus hypersecretion, and airway remodeling. Airway smooth muscle tone is maintained by an interaction between sympathetic, parasympathetic, and nonadrenergic mechanisms. Acute bronchoconstriction usually results from mediators such as histamine, cysteinyl leukotrienes, prostaglandins, and tryptase released or generated during degranulation of mast cells and basophils.1 Inflammatory mediators such as histamine, leukotrienes, and bradykinin increase microvascular permeability leading to mucosal edema, which causes the airways to become more rigid and limits airflow.11 These changes exaggerate the consequences of acute bronchoconstriction. In asthmatics, there is an increased number and volume of mucous glands, with increased mucus secretion.12 Extensive mucus plugging may be a cause of persistent airway obstruction in acute severe attacks.
Although airway obstruction in asthma is generally reversible, some asthmatics have an irreversible or fixed obstruction. Airway remodeling is the term used to describe the process that produces the structural airway changes leading to this fixed obstruction; it is characterized by airway epithelial damage, subepithelial fibrosis, airway smooth muscle hypertrophy, increased mucus production, and increased vascularity of the airways.1,12 These changes increase airflow obstruction and airway responsiveness and may decrease patient responsiveness to therapy.1
CLINICAL PRESENTATION AND DIAGNOSIS
The diagnosis of asthma is based on a detailed medical history, a physical examination of the upper respiratory tract and skin, and spirometry. The clinician should determine that episodic symptoms of airflow obstruction are present, airflow obstruction is at least partially reversible, and alternative diagnoses are excluded.1 Spirometry is required for diagnosing asthma because the medical history and physical examination are not reliable for characterizing the status of lung impairment or excluding other diagnoses.1
Factors Affecting Asthma Severity
Major factors that may contribute to the severity of asthma include allergens, environmental chemical exposures or pollution, and exposure to tobacco smoke. Up to 80% of asthmatics have symptoms of rhinitis, and treatment of rhinitis with intranasal corticosteroids may relieve the symptoms.13 Gastroesophageal reflux has been associated with increased asthma symptoms, especially nighttime symptoms. Obesity has been associated with asthma persistence and severity.
Nonselective β-blockers, including those in ophthalmic preparations, may cause asthma symptoms, and these agents should be avoided in asthmatics unless the benefits of therapy outweigh the risks.1 In asthmatic patients requiring β-blocker therapy, a β1-selective agent should be chosen. Because selectivity is dose related, the lowest effective dose should be used. β-blockers may inhibit β-agonist reversal of bronchospasm, and a larger dose of β-agonist or the use of an anticholinergic agent may be necessary to reverse bronchospasm.
Clinical Presentation and Diagnosis of Chronic Asthma
General
Asthma severity ranges from normal pulmonary function and symptoms with only acute exacerbations to significantly decreased pulmonary function with continuous symptoms.
Symptoms
• Symptoms may include dyspnea, cough, wheezing, and chest tightness that may be continual, episodic, seasonal, or occur in association with known triggers.
• Symptoms may occur more often at night, early in the morning, or with exercise.
• Patients with intermittent asthma may be symptom-free and have normal pulmonary function between exacerbations.
Signs
• Patients may have end-expiratory wheezing and dry cough.
Laboratory Tests
• Increased serum concentrations of IgE or eosinophils may help confirm the diagnosis of asthma but are not diagnostic for asthma.
Other Diagnostic Tests
• Spirometry, an objective measure of pulmonary function, can be used to assist in confirming the diagnosis of asthma.
• Useful pulmonary function tests include the forced expiratory volume in 1 second (FEV1) and forced vital capacity (FVC). The following values support a diagnosis of asthma:
• Decreased FEV1/FVC relative to predicted values demonstrates airway obstruction. The ratio may be normal between exacerbations.
• 12% or greater (at least 200 mL) improvement in FEV1, after an inhaled bronchodilator demonstrates a reversible obstruction. A 2-to 3-week course of oral corticosteroids may be necessary to demonstrate reversibility in airway obstruction.
• A decrease in FEV1, of 15% or more after an exercise test is diagnostic for exercise-induced asthma.
• Assessment of diurnal variation in peak expiratory flow (PEF) may be useful in patients who have asthma symptoms and normal spirometry.
• When spirometry is equivocal, a 20% or greater decrease in FEV1 after the administration of methacholine is diagnostic for asthma. A negative bronchoprovocation test with methacholine may help rule out asthma.
• A positive allergen test may help guide nonpharmacologic therapy but is not diagnostic for asthma.
Clinical Presentation and Diagnosis of Acute Asthma
General
Acute asthma can present rapidly (within 3 to 6 hours) but more commonly, deterioration occurs over several hours, days, or even weeks. Typically, there is gradual deterioration over several days followed by a more rapid decline over 2 to 3 days. Acute asthma can be a life-threatening event, and severity does not correspond to severity of the chronic disease.
Symptoms
• The patient usually presents with complaints of dyspnea, cough, shortness of breath, and chest tightness.
• Because of their inability to breathe, patients are generally anxious and may be agitated. In acute severe asthma, patients may be unable to communicate in complete sentences.
• Mental status changes may indicate impending respiratory failure.
Signs
• The patient usually has tachypnea and may have tachycardia.
• Wheezing may vary from end-expiratory wheezing in mild exacerbations to wheezing throughout inspiration and expiration in severe exacerbations.
• Bradycardia and absence of wheezing may indicate impending respiratory failure.
• Patients may also present with hyperinflation, use of accessory muscles to breathe, pulsus paradoxus, diaphoresis, and cyanosis.
Laboratory Tests
• Arterial blood gases for evaluating partial arterial pressure of carbon dioxide (Pco2) should be considered for patients in severe distress, suspected hypoventilation, or when PEF or FEV1 is 30% or less after initial treatment.
• A CBC with differential should be obtained in patients with fever or purulent sputum.
• Serum electrolytes should be obtained, because frequent β2-agonist administration may decrease serum potassium, magnesium, and phosphate.
Other Diagnostic Tests
• Patients may present with PEF rates ranging from greater than 80% and oxygen saturation greater than 95% in mild exacerbations to PEF rates less than 50%, oxygen saturations less than 91%, partial arterial oxygen pressures (Pao2) less than 50 mm Hg (less than 6.65 kPa), and Pco2 greater than 42 mm Hg (greater than 5.59 kPa) in severe exacerbations.
• A chest x-ray should be performed when pneumonia is suspected.
Patient Encounter, Part 1
RB is a 13-year-old African American female who presents with complaints of shortness of breath when she exercises. She recently joined the cross-country team. She finishes her training runs, but she is having trouble keeping up with the other girls on the team because she gets extremely short of breath 5 or 10 minutes into her run, her chest begins to feel tight, and she coughs. The symptoms usually go away 30 minutes to an hour after she stops running. She also wakes up at night about once a week because she is having trouble catching her breath.
What information is suggestive of asthma?
Based on the information presented, how would you classify this patient’s asthma severity?
What additional information do you need to know before creating a treatment plan for this patient?
TREATMENT OF ASTHMA
Desired Outcomes
Chronic Asthma
Therapy for chronic asthma is directed at maintaining long-term control of asthma using the least amount of medications and minimizing adverse effects.1 Treatment goals are to: (a) prevent chronic and troublesome symptoms; (b) require infrequent use (2 or fewer days/week) of SABA for quick relief of symptoms; (c) maintain normal or near-normal pulmonary function; (d) maintain normal activity levels; (e) meet patients’ and families’ expectations of satisfaction with asthma care; (f) prevent exacerbations of asthma and the need for emergency department visits or hospitalizations; (g) prevent progressive loss of lung function; and (h) provide optimal pharmacotherapy with minimal or no adverse effects.
Acute Severe Asthma
Acute or worsening asthma can be a life-threatening situation and requires rapid assessment and appropriate intensification of therapy. Mortality associated with asthma exacerbations is usually related to an inappropriate assessment of the severity of the exacerbation resulting in insufficient treatment or referral for medical care.14 The goals of therapy are to: (a correct significant hypoxemia; (b) reverse airflow obstruction rapidly; and (c) reduce the likelihood of exacerbation relapse or recurrence of severe airflow obstruction in the future.1
General Approach to Treatment
Chronic Asthma
Chronic asthma is classified as: (a) intermittent asthma or (b) persistent asthma that may be graded as mild, moderate, or severe. ![]() In chronic asthma, initial classification of asthma severity is based on current disease impairment and future risk. The term impairment refers to the frequency and severity of symptoms, use of SABA for quick relief of symptoms, pulmonary function, and impact on normal activity and quality of life. Risk refers to the potential for future severe exacerbations and asthma-related death, progressive loss of lung function (adults) or reduced lung growth (children), and the occurrence of drug-related adverse effects. Initial assessment of severity is made at the time of diagnosis, and initial therapy is based on this assessment. Future therapy decisions are based on ongoing assessments of asthma control.
In chronic asthma, initial classification of asthma severity is based on current disease impairment and future risk. The term impairment refers to the frequency and severity of symptoms, use of SABA for quick relief of symptoms, pulmonary function, and impact on normal activity and quality of life. Risk refers to the potential for future severe exacerbations and asthma-related death, progressive loss of lung function (adults) or reduced lung growth (children), and the occurrence of drug-related adverse effects. Initial assessment of severity is made at the time of diagnosis, and initial therapy is based on this assessment. Future therapy decisions are based on ongoing assessments of asthma control.
Treatment of chronic asthma involves avoidance of triggers known to precipitate or worsen asthma and the use of long-term control and quick-relief medications. Long-term control medications include ICS, inhaled long-acting β2-agonists (LABA), oral theophylline, oral leukotriene modifying agents, and omalizumab (Table 14–1). In patients with severe asthma, systemic corticosteroids may be used as a long-term control medication. Quick-relief medications include SABA, anticholinergics, and systemic corticosteroids. A stepwise approach to therapy is recommended to achieve the treatment goals.1
Acute Severe Asthma
In acute severe asthma, the severity of an exacerbation is not dependent upon the classification of the patient’s chronic asthma because even patients with intermittent asthma can have life-threatening acute exacerbations. Treatment of acute or worsening asthma primarily involves pharmacologic therapy. Early and aggressive treatment is necessary for quick resolution.1
Important elements of an early treatment plan include: (a) a written action plan; (b) recognition of early indicators of an acute exacerbation (e.g., asthma symptoms or worsening PEF or FEV1) and taking prompt action; (c) appropriate intensification of pharmacotherapy by increasing inhaled SABA, and in some cases adding a short course of oral corticosteroids; (d) removal of triggers or irritants that may be contributing to the acute exacerbation; and (e) timely communication between patient and clinician about worsening symptoms, declining PEF, and decreased responsiveness to SABA.1
Initial treatment of acute severe asthma includes use of oxygen for rapid reversal of hypoxemia, a SABA and perhaps inhaled ipratropium bromide to reverse airway constriction, and a systemic corticosteroid to attenuate the inflammatory response. Close monitoring of objective measures such as FEV1 or PEF is important to quantify the response to therapy. Because recovery from exacerbations is often gradual, intensified therapy should be continued for several days.
Nonpharmacologic Therapy
Patients should play an active role in their therapy, and an active partnership should be developed with the patient and family. Goals for asthma treatment should be shared, and the patient and health care provider should jointly agree on the patient’s personal treatment goals.
Nonpharmacologic therapy should be incorporated into each step of therapy, and patient education should occur at all points where health care professionals interact with patients. Patient education should begin at the time of diagnosis and be tailored to meet individual patient needs.1 Patients should understand: the difference between the asthmatic and normal lung, what happens to the lung during an asthma attack, differences between controller and relief medications, how to take inhaled medications correctly, and environmental control measures. Patients should also learn: self-management of asthma, including assessing level of control, recognizing signs and symptoms of worsening asthma, skills for self-monitoring of pulmonary function, when and how to take rescue actions, and when to seek medical care.
The importance of understanding asthma as a chronic disease and the need for daily treatment with long-term control medications should be stressed. The importance of proper use of medication-delivery devices should be reinforced.1
Risk Factor Avoidance
Patients who smoke should be strongly encouraged to quit; cigarette smoking decreases the efficacy of ICS and can trigger an acute asthmatic response.1 All patients should also avoid second-hand smoke. Patients should avoid outdoor activities when air quality is poor and avoid exposure to other irritants such as hairspray, paint, exhaust fumes, and smoke from any fire.
Patients sensitive to specific allergens should be educated on ways to avoid them. Environmental controls to reduce the allergen load in the patient’s home may reduce asthma symptoms, school absences because of asthma, and unscheduled clinic and emergency visits for asthma.15 Patients allergic to warm-blooded pets should remove them from home if possible or at least keep them out of the bedroom. Allergies to cockroach antigens and dust mites should be identified and appropriate measures taken to reduce or eliminate them.
Table 14–1 Usual Dosages for Quick-Relief Medications in Asthma
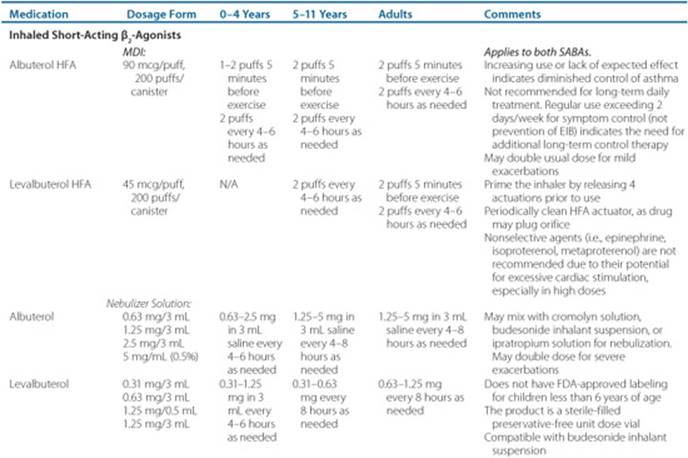


The inactivated influenza vaccine should be considered in patients having asthma to decrease their risk of complications from influenza.1 The pneumococcal vaccine may decrease the risk of invasive pneumococcal disease in asthmatics, but current guidelines do not include routine administration to asthma patients.1,16
Drug Delivery Devices
![]() Direct airway administration of asthma medications through inhalation is the most efficient route and minimizes systemic adverse effects. Poor inhaler technique can result in increasedoropharyngeal deposition of the drug with decreased efficacy and increased adverse effects. Figure 14–1 provides steps for the appropriate use of inhaled delivery devices. Inhaled asthma medications are available in metered-dose inhalers (MDIs), dry powder inhalers (DPIs), and nebulized solutions. Because inhaler technique deteriorates over time, health care providers should take every opportunity to reinforce appropriate inhaler technique. Although nebulizers have often been used for drug delivery in children, their use is expensive and time consuming.
Direct airway administration of asthma medications through inhalation is the most efficient route and minimizes systemic adverse effects. Poor inhaler technique can result in increasedoropharyngeal deposition of the drug with decreased efficacy and increased adverse effects. Figure 14–1 provides steps for the appropriate use of inhaled delivery devices. Inhaled asthma medications are available in metered-dose inhalers (MDIs), dry powder inhalers (DPIs), and nebulized solutions. Because inhaler technique deteriorates over time, health care providers should take every opportunity to reinforce appropriate inhaler technique. Although nebulizers have often been used for drug delivery in children, their use is expensive and time consuming.
Patients should be educated to keep track of inhaler use. Some inhalers have a built-in counter or device to notify the patient of how many doses are remaining (e.g., Ventolin, hydrofluoroalkane [HFA], and Twisthaler).
Spacers or holding chambers with valves decrease the need for coordination of actuation of MDI devices with inhalation, decrease oropharyngeal deposition of drug, and increase pulmonary drug delivery.17,18Patients using a spacer or holding chamber should be counseled to place only one puff of the drug into the chamber at a time, because actuating the MDI more than once into the chamber before inhalation decreases drug delivery. However, taking multiple breaths after a single actuation is appropriate and does not decrease drug efficacy.17 Spacers or holding chambers with valves are equipped with a mouthpiece or a facemask allowing the use of an MDI in children younger than 5 years of age.
Asthma Self-Management
Asthma self-management plans give patients the freedom to adjust therapy based on personal assessment of disease severity and a predetermined action plan. These plans reduce morbidity and the need for medical services.19 For self-management plans to be effective, patients should be given a written action plan that is part of a global educational program.1 The plan should include instructions on daily management and how to recognize and handle worsening asthma.1
Asthma control is assessed by evaluating signs and symptoms of worsening asthma and/or monitoring PEF. Early signs of deterioration include increasing nocturnal symptoms, increasing use of inhaled SABA, or symptoms that do not respond to increased use of inhaled SABAs. Measurement of PEF should be considered for patients with moderate to severe asthma, a poor perception of worsening asthma or airflow obstruction, and those with an unexplained response to environmental or occupational exposures.1 If PEF measurements are used to assess control, the patient must be able to use a peak flow meter properly. PEF should be measured daily in the morning on waking, before using a bronchodilator. For PEF-based asthma action plans, the patient’s personal best PEF should be established over a 2-to 3-week period using established methods when the patient is receiving optimal treatment.1Subsequent PEF measurements are evaluated in relation to their variability from the patient’s best.19 PEF measurements in the range of 80% to 100% of personal best (green zone) indicate that current therapy is acceptable. A PEF in the range of 50% to 79% of personal best (yellow zone) may indicate an impending exacerbation, and therapy should be intensified based on the self-management plan. A PEF less than 50% (red zone) signals a medical alert; patients should use their SABA immediately and consult their asthma action plan.

FIGURE 14–1. Instructions for using an inhaler. (From Ref. 20.)
Pharmacologic Therapy
β2-Adrenergic Agonists
β2-Agonists relax airway smooth muscle by directly stimulating β2-adrenergic receptors.20 They also increase mucociliary clearance and stabilize mast cell membranes. Inhalation dosage forms are most commonly used, but oral and injectable dosage forms are also available. β2-Agonists have significantly better bronchodilating activity in acute asthma than theophylline or anticholinergic agents.
Adverse effects include tachycardia, tremor, and hypokalemia, which are usually not troublesome with inhaled dosage forms. Oralβ2-agonists have increased adverse effects and should be avoided in patients who are able to use inhaled medications. Oral β2-agonists should not be used in acute asthma because of a delayed onset of action compared to the inhaled route. Inhaled β2-agonists are classified as either short-or long-acting based on duration of action.
Short-Acting Inhaled β2 -Agonists
![]() Inhaled SABAs are the most effective agents for reversing acute airway obstruction caused by bronchoconstriction and are the drugs of choice for treating acute severe asthma and symptoms of chronic asthma as well as preventing exercise-induced bronchospasm.1 Inhaled SABA have an onset of action of less than 5 minutes and a duration of action of 4 to 6 hours. Using an MDI with a spacer is quicker and at least as effective as administration by nebulization.
Inhaled SABAs are the most effective agents for reversing acute airway obstruction caused by bronchoconstriction and are the drugs of choice for treating acute severe asthma and symptoms of chronic asthma as well as preventing exercise-induced bronchospasm.1 Inhaled SABA have an onset of action of less than 5 minutes and a duration of action of 4 to 6 hours. Using an MDI with a spacer is quicker and at least as effective as administration by nebulization.
Albuterol (known as salbutamol outside the United States), the most commonly used inhaled SABA, is a racemic mixture (50:50) of albuterol enantiomers. The R-enantiomer is the active component, whereas the S-enantiomer is inactive and may be associated with unwanted effects. Levalbuterol, the pure R-enantiomer of albuterol (and referred to as R-salbutamol outside the United States), is available as an MDI and solution for nebulization. Levalbuterol and albuterol are similar in efficacy, but the acquisition cost of levalbuterol is substantially higher.21 Nonselective β2-agonists (e.g., metaproterenol) are not used commonly due to the potential for increased adverse effects.
Doses used for quick relief in chronic asthma are provided in Table 14–1. Usual rescue doses may be doubled for mild exacerbations. The regular use of inhaled SABAs is not recommended.1
Long-Acting Inhaled β2-Agonists
Salmeterol and formoterol are LABA that provide up to 12 hours of bronchodilation after a single dose. Both agents are approved for chronic prevention of asthma symptoms. Salmeterol is a partial agonist with an onset of action of approximately 30 minutes. Because of this delayed onset, patients should be cautioned not to use salmeterol as a quick-relief medication. Formoterol is a full agonist that has an onset of action similar to that of albuterol, but it is not currently approved for the treatment of acute bronchospasm.
Inhaled LABA are indicated for add-on therapy for asthma not controlled on low to medium doses of ICS. Adding an LABA is at least as effective in improving symptoms and decreasing asthma exacerbations as doubling the dose of an ICS.22,23 Adding an LABA to ICS therapy also reduces the amount of ICS necessary for asthma control.24
Although both formoterol and salmeterol are effective as add-on therapy for moderate persistent asthma, neither agent should be used as monotherapy for chronic asthma. There may be an increased risk of severe asthma exacerbations and asthma-related deaths when LABA are used alone or added to standard therapy.25,26 The labeling for all drugs containing LABA contains a “black-box” warning against their use without an ICS. The risk of increased severe asthma exacerbations does not appear to be increased in adults receiving both an LABA and ICS.27,28
Salmeterol and formoterol are available in fixed ratio combination products containing fluticas one and budesonide, respectively. Combination products may increase adherence because of the need for fewer inhalers and inhalations. However, they offer less flexibility in dosage adjustment of individual ingredients when that is considered necessary. Doses used for long-term control of chronic asthma are provided in Table 14–2.
Corticosteroids
Corticosteroids are the most potent anti-inflammatory agents available for the treatment of asthma and are available in inhaled, oral, and injectable dosage forms. They decrease airway inflammation, AHR, and mucus production and secretion. Corticosteroids also improve the response to β2-agonists.20
Inhaled Corticosteroids
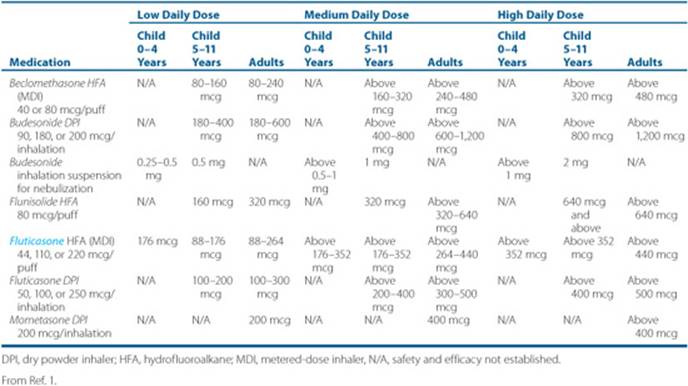
![]() ICS are the preferred therapy for all forms of persistent asthma in all age groups.1 ICS are more effective than cromolyn, leukotriene modifiers, nedocromil, and theophylline in improving lung function and preventing emergency department visits and hospitalizations due to asthma exacerbations.1,22 The primary advantage of using ICS compared to systemic corticosteroids is the targeted drug delivery to the lungs, which decreases the risk of systemic adverse effects. All ICS are equally effective if given in equipotent doses (Table 14–3). Product selection should be based on preference for dosage form, delivery device, and cost.
ICS are the preferred therapy for all forms of persistent asthma in all age groups.1 ICS are more effective than cromolyn, leukotriene modifiers, nedocromil, and theophylline in improving lung function and preventing emergency department visits and hospitalizations due to asthma exacerbations.1,22 The primary advantage of using ICS compared to systemic corticosteroids is the targeted drug delivery to the lungs, which decreases the risk of systemic adverse effects. All ICS are equally effective if given in equipotent doses (Table 14–3). Product selection should be based on preference for dosage form, delivery device, and cost.
Although some beneficial effect is seen within 12 hours of administration, 2 weeks of therapy is necessary to see significant clinical effects. Longer treatment may be necessary to realize the full effects on airway inflammation and remodeling.
ICS have a flat dose-response curve; doubling the dose has a limited additional effect on asthma control.1,29 Considerable variability in response to ICS exists,30,31 and increasing the doses may be of greater benefit in severe asthma than in mild-to-moderate asthma.32 The ICS are effective when given twice daily and may be effective when given once daily for mild asthma.
Local adverse effects of ICS include oral candidiasis, cough, hoarse voice, and dysphonia. The incidence of local adverse effects can be reduced by using a spacer or valved holding chamber and by having the patient rinse the mouth with water and expectorate after using the ICS. Decreasing the dose reduces the incidence of hoarseness. For most delivery devices, the majority of the drug is deposited in the mouth and throat and swallowed. Systemic absorption occurs via the pulmonary and oral routes. Although only a fraction of the drug is delivered to the lungs, 100% of the drug reaching the lungs is absorbed systemically.1
Systemic adverse effects are dose dependent and rare at low to medium doses. However, high-dose ICS have been associated with adrenal suppression, decreased bone mineral density, skin thinning, cataracts, and easy bruising.1Growth suppression in children occurs primarily in the first year of treatment and may be due to delayed growth with the potential of future catch-up growth.33
Systemic Corticosteroids
Systemic corticosteroids are effective as both long-term control and rescue medications. Because of serious potential adverse effects, systemic corticosteroids should be used for long-term asthma control only in patients who have failed other therapies. If systemic therapy is necessary, once-daily or every-other-day therapy should be used with repeated attempts to decrease the dose or discontinue the drug.
Systemic corticosteroids are the cornerstone of treatment for worsening asthma not responding to bronchodilators and for acute severe asthma. For patients with nonresponsive worsening asthma, a short course or “burst” of systemic corticosteroids is effective for gaining control and preventing progression.1
In acute severe asthma, systemic corticosteroids should be given to all patients who have moderate to severe exacerbations or who do not respond to initial bronchodilator therapy. Corticosteroids reduce inflammation, increase the response to β2-agonists, hasten recovery, decrease hospital admissions, and reduce relapse rates. The onset of action is delayed, and a clinical response may not be seen for 4 to 12 hours.20 For this reason, systemic corticosteroids should be started early in the course of acute exacerbations or worsening asthma. The oral route is preferred in acute severe asthma; there is no evidence that IV corticosteroid administration is more effective.1 Recommended doses for acute asthma exacerbations are shown in Table 14–4.
Table 14–2 Usual Dosages for Long-Term Control Medications in Asthma


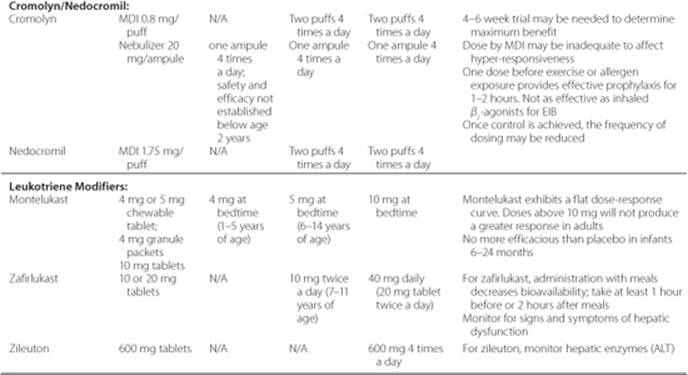

Table 14–3 Estimated Comparative Daily Dosages for Inhaled Corticosteroids for Asthma
Therapy with systemic corticosteroids should generally be continued until the PEF is 70% or more of the predicted value or personal best. The duration of therapy usually ranges from 3 to 10 days, but longer therapy may be necessary for severe exacerbations. Tapering the corticosteroid dose in patients receiving short bursts (up to 10 days) is not necessary because any adrenal suppression is transient and rapidly reversible.1,20
Anticholinergics
Anticholinergic agents (see Tables 14–1 and 14-4) act by inhibiting the effects of acetylcholine on muscarinic receptors in the airways. They only protect against cholinergic-mediated bronchoconstriction and are not as effective as β2-agonists in asthma.20 Anticholinergic drugs may cause bothersome adverse effects such as blurred vision, dry mouth, urinary retention, and constipation. However, the inhaled anticholinergic agents are quaternary amines that are not absorbed systemically and have limited adverse effects.
Ipratropium bromide (Atrovent) is available as an MDI and solution for nebulization. It has an onset of action of approximately 30 minutes and a duration of action of 4 to 8 hours. Care should be taken to avoid contact of the spray or nebulized solution with the eyes, as it can cause mydriasis and blurred vision.
The addition of ipratropium bromide to inhaled β2-agonist therapy in acute severe asthma improves pulmonary function and decreases hospitalization rates in both adult and pediatric patients.34 The benefit of combining ipratropium and albuterol appears to be greatest in moderate to severe exacerbations, and the combination should be considered first-line therapy in severe exacerbations.
Tiotropium bromide (Spiriva) is a long-acting inhaled anticholinergic available in a DPI; it has an onset of action of approximately 30 minutes and a duration of action longer than 24 hours. There is little evidence supporting the use of tiotropium bromide in asthma.
Leukotriene Modifiers
Leukotriene modifiers (see Table 14–2) either inhibit 5-lipoxygenase (zileuton) or competitively antagonize the effects of leukotriene D4 (montelukast and zafirlukast). These agents improve FEV1 and decrease asthma symptoms, rescue drug use, and exacerbations due to asthma. Although these agents offer the convenience of oral therapy for asthma, they are significantly less effective than low doses of ICS.1,35 Combining a leukotriene receptor antagonist with an ICS or LABA is not as effective as an ICS plus an LABA.1
Table 14–4 Dosages of Selected Drugs for Asthma Exacerbations


The leukotriene receptor antagonists zafirlukast (Accolate) and montelukast (Singulair) are generally well tolerated and dosed twice daily and once daily, respectively. Significant increases in hepatic enzymes have been reported in postmarketing studies for zafirlukast but not montelukast. Zafirlukast also inhibits the CYP2C9 and CYP3A4 isoenzymes and may increase prothrombin time in patients receiving warfarin; the International Normalized Ratio should be monitored if warfarin and zafirlukast are used concomitantly. Montelukast does not appear to inhibit the cytochrome P-450 enzymes.
Zileuton (Zyflo) is not commonly used because of the need for dosing four times daily, potential drug interactions, and potential hepatotoxicity with the resulting need for frequent monitoring of hepatic enzymes.
Cromolyn and Nedocromil
Cromolyn sodium (Intal) and nedocromil sodium (Tilade) are inhaled anti-inflammatory agents that block both the early-and late-phase response possibly by inhibiting release of mediators from mast cells. Both agents are alternatives to ICS for treatment of mild persistent asthma, but they are significantly less effective than low doses of ICS (see Table 14–2).1 Cromolyn and nedocromil are similar in efficacy to the leukotriene antagonists and theophylline for persistent asthma.20 Both drugs require dosing four times daily until symptoms stabilize, after which the dosage frequency can be reduced to three times a day for cromolyn and twice daily for nedocromil.20 Patients may notice improvement in 1 to 2 weeks, but maximal benefit may not be seen for 4 to 6 weeks.
One dose of cromolyn or nedocromil prior to exercise or allergen exposure will provide prophylaxis for 1 to 2 hours. These agents are not as effective as albuterol for prophylaxis of exercise-induced asthma.
Both agents are well tolerated with adverse effects limited to cough and wheezing. Bad taste and headache have also been reported with nedocromil.
Methylxanthines
Theophylline (see Table 14–2) causes bronchodilation by inhibiting phosphodiesterase and antagonizing adeno-sine. It may also have mild anti-inflammatory and immu-nomodulatory properties.36 Its use is limited because of inferior efficacy as a controller medication compared to ICS, a narrow therapeutic index with potentially life-threatening toxicity, and multiple clinically important drug interactions.
Target serum theophylline concentrations are 5 to 15 mg/L (28–83 μmol/L); an increased risk of adverse effects outweighs the increased bronchodilation in most patients above 15 mg/L (83 μmol/L).37Headache, nausea, vomiting, and irritability may occur at serum concentrations less than 20 mg/L (110 μmol/L) but are rare when the dose is started low and increased slowly. More serious adverse effects, including cardiac arrhythmias, seizures, toxic encephalopathy, and death can occur at higher concentrations.20
Theophylline is primarily metabolized by CYP1A2 and CYP3A4 and is involved in a large number of disease and drug interactions. Theophylline exhibits nonlinear pharmacokinetics in the therapeutic range; therefore, serum concentration changes due to dosage adjustments and drug interactions may not always be predictable.20 Theophylline also exhibits interpatient variability in hepatic clearance; consequently, serum theophylline concentrations should be monitored.
Omalizumab
Omalizumab (Xolair) is a recombinant humanized monoclonal anti-IgE antibody that inhibits binding of IgE to receptors on mast cells and basophils, resulting in inhibition of mediator release and attenuation of the early-and late-phase allergic response. It is indicated for treatment of moderate to severe persistent asthma in patients 12 years of age or older whose asthma is not controlled by ICS and who have a positive skin test or in vitro reactivity to perennial allergens.38 Omalizumab significantly decreases ICS use, reduces the number and length of exacerbations, and increases asthma-related quality of life. It is also effective in improving asthma control in severe asthmatics receiving combination therapy with high-dose ICS and LABAs.1 However, its place in therapy is limited by its high cost.39
Omalizumab is given as a subcutaneous injection every 2 to 4 weeks, and the initial dose is based on the patient’s weight and initial total IgE serum concentration. The dosage should not be adjusted based on subsequent total serum IgE measurements (see Table 14–2). Drug clearance depends on patient weight, and dosage should be adjusted if there is a significant change in body weight. Doses greater than 150 mg should be administered as separate injections at multiple sites.
The most common adverse effects are injection site reactions and include bruising, redness, pain, stinging, itching, and burning. Anaphylactic reactions are rare but may be delayed 2 hours or more after drug administration.40Reports of delayed anaphylactic reactions have led to a “black box” warning in the labeling and a medication guide warning of this risk.
Treatment of Chronic Asthma
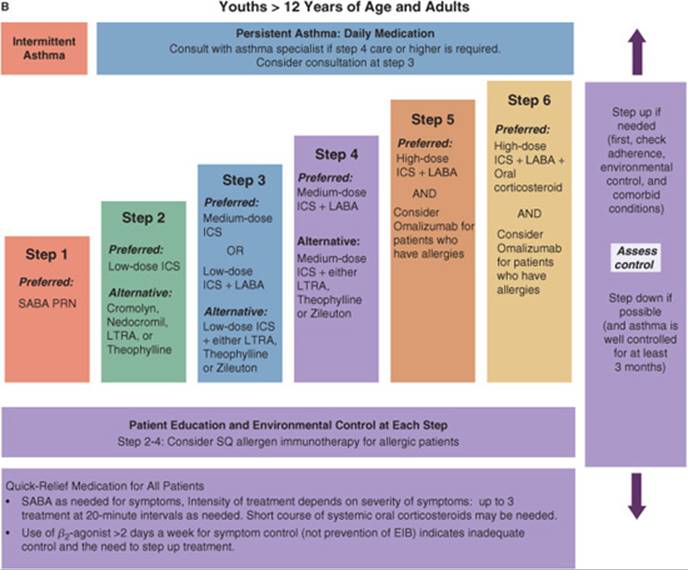
![]() The intensity of pharmacotherapy for chronic asthma is based on disease severity for initial therapy and level of control for subsequent therapies. The least amount of medications necessary to meet the goals of asthma therapy should be used.1 Current recommendations for stepwise therapy for chronic asthma are shown in Figure 14–2. However, because of varying asthma presentations, the therapeutic plan must be individualized. The EPR-3 separates treatment recommendations into three categories based on patient age: (a) children younger than 5 years of age, (b) childrenbetween the ages of 5 and 11 years, and (c) individuals 12 years of age and older. Refer to Ref. 1 for more information on assessing asthma control and adjusting therapy in these three categories.1
The intensity of pharmacotherapy for chronic asthma is based on disease severity for initial therapy and level of control for subsequent therapies. The least amount of medications necessary to meet the goals of asthma therapy should be used.1 Current recommendations for stepwise therapy for chronic asthma are shown in Figure 14–2. However, because of varying asthma presentations, the therapeutic plan must be individualized. The EPR-3 separates treatment recommendations into three categories based on patient age: (a) children younger than 5 years of age, (b) childrenbetween the ages of 5 and 11 years, and (c) individuals 12 years of age and older. Refer to Ref. 1 for more information on assessing asthma control and adjusting therapy in these three categories.1

FIGURE 14–2. Stepwise approach for managing asthma in children (A) and adults (B).
(ICS, inhaled corticosteroids; LABA, long-acting β-agonists; LTRA, leukotriene receptor antagonist; PRN, as needed; SABA, short-acting β-agonist.) (From Ref. 20.)
Intermittent Asthma
Long-term control medications are not necessary in patients with intermittent asthma, and patients should use an SABA to prevent or treat symptoms.1 This classification includes patients with exercise-induced asthma, seasonal asthma, or asthma symptoms associated with infrequent trigger exposure. Patients can pretreat with two puffs of albuterol, cromolyn, or nedocromil prior to exposure to a known trigger.
Persistent Asthma
Patients who use an SABA more than twice a week for symptom control (not exercise-induced bronchospasm) should be treated as persistent asthma. Patients who experienced two or more exacerbations requiring oral corticosteroids in the past year may also be categorized as having persistent asthma.1 Patients with persistent asthma require daily long-term control therapy (see Tables 14–2 and 14-3). However, daily therapy limited to a predefined period of risk (e.g., seasonal asthma) may be considered when these periods are identified by history. ICS are the long-term control medication of choice at all levels of severity and in all age groups.1 SABAs should be prescribed for all patients with chronic asthma for use on an as-needed basis.
After initiating therapy, patients should be monitored within 2 to 6 weeks to ensure that asthma control has been achieved. Before increasing therapy, the patient’s inhaler technique and adherence to therapy should be evaluated.1Patients with controlled asthma should be monitored at 1-to 6-month intervals to ensure control is maintained. A gradual stepdown in control therapy should be initiated when possible, usually once control has been maintained for at least 3 months.1
Patient Encounter, Part 2: The Medical History, Physical Exam, and Diagnostic Tests
HPI: As presented in Part 1. In addition, the patient does not report any daytime symptoms and states that she does not get short of breath walking up the stairs at school.
PMH: Allergic rhinitis for 6 years (allergic to dust mites per skin testing; no other allergies positive on the skin testing panel). Bronchitis 3 times in the last 6 years (all treated with azithromycin and Robitussin DM); last episode 6 months ago. Hospitalized with viral lower respiratory tract infections twice at ages 1 and 2.
FH: Mother had asthma as a child, but “outgrew it” and has had no problems with it since she was 12 or 13 years old; both her mother and father have allergic rhinitis.
SH: Only child who lives at home with her mother and father in a two-bedroom duplex built on a concrete slab. Neither the patient nor her parents smoke or drink alcohol. They have no animals inside or outside the home.
Meds: Cetirizine 10 mg by mouth daily
ROS: Unremarkable except as described above.
PE:
Gen: Small for her age but appears to be well nourished and healthy
VS: BP 112/68 mm Hg, P 78 bpm, RR 18 bpm, T 37.0°C (98.7°F), ht 4’6” (137 cm), wt 28 kg (62 lb)
Chest: CTA bilaterally, no wheezing
CV: RRR; S1 and S2 normal; no rubs, gallops, or murmurs
Ext: No clubbing, cyanosis, or edema
Labs: Normal except for WBC differential with 7% eosinophils
Pulmonary Function Tests
FEV1: 1.5 L (84% predicted)
FVC: 1.75 L (92% predicted)
FEV1/FVC: 0.857
Postbronchodilator FEV1: 1.70 L (13.3% increase)
FEV1 after exercise: 1.23 L (23.1% decrease)
Given this additional information, what is your assessment of the patient’s asthma severity?
Identify your treatment goals for this patient.
What nonpharmacologic and pharmacologic alternatives are feasible for this patient?
Outline a treatment plan for this patient that includes nonpharmacologic therapy, pharmacologic therapy, and a monitoring plan. Justify your therapeutic selections.
Children Up to 4 Years of Age (Fig. 14–2)
Long-term control medications should be initiated in patients who have had: (a) four or more episodes within the last year that have lasted for a day or longer and affected sleep and (b) have one major or two minor risk factors for developing persistent asthma. Major risk factors include a parental history of asthma, diagnosis of atopic dermatitis, and evidence of sensitization to aeroallergens. Minor risk factors include sensitization to food, 4% or more eosinophils in peripheral blood, and wheezing apart from colds. In addition, controller therapy should be considered if the patient requires symptomatic treatment for more than 2 days a week for more than 4 weeks or has two asthma exacerbations requiring systemic corticosteroids within 6 months.1,33
Daily ICS are the preferred long-term control therapy in all steps, and nonpreferred alternatives are cromolyn or a leukotriene receptor antagonist. Patients not controlled on low doses of ICS should be increased to medium doses before adding other therapies. Because high-dose ICS may be associated with significant adverse effects, addition of a leukotriene receptor antagonist or LABA to medium-dose therapy is preferred before increasing the ICS dose further. Theophylline is not recommended as an alternative at any step in this age group.1
Children 5 to 11 Years of Age (Fig. 14–2)
Daily ICS are the preferred long-term control therapy in all steps. Nonpreferred alternatives are cromolyn, a leukotriene receptor antagonist, nedocromil, or theophylline. For patients not controlled on low-dose ICS, the addition of an LABA, leukotriene receptor antagonist, or theophylline to current therapy or increasing to medium-dose ICS are equivalent options.1 In patients not controlled on medium-dose ICS or low-dose ICS plus adjunctive therapy, the addition of an LABA to medium-dose ICS is preferred over other adjunctive therapies.1
Individuals 12 Years of Age and Older (Fig. 14–2)
Daily ICS are the preferred long-term control therapy in all steps. Nonpreferred alternatives include cromolyn, a leukotriene receptor antagonist, nedocromil or sustained-release theophylline. For patients not controlled on low doses of ICS, the addition of a LABA, or increasing to medium-dose ICS are equivalent options.1 The addition of other add-on therapies (leukotriene receptor antagonist, sustained-release theophylline, or zileuton) are nonpreferred options.1 Omalizumab maybe considered in patients not controlled on high-dose ICS and an LABA.
Treatment of Acute Severe Asthma
The optimal treatment of acute severe asthma depends on the severity of the exacerbation. The patient’s condition usually deteriorates over several hours, days, or weeks. Gradual deterioration may indicate failure of long-term controller therapy. However, rapid deterioration can occur in some patients; these patients usually respond well to bronchodilator therapy.41 Severity at the time of the evaluation can be estimated by signs and symptoms or presenting PEF or FEV1 but patient response 30 minutes after inhalation of a bronchodilator is the best predictor of outcome.14
![]() In acute severe asthma, early and appropriate intensification of therapy is important to resolve the exacerbation, prevent relapse, and prevent severe airflow obstruction in the future. Starting therapy at home allows for rapid initiation and early assessment of response (see Fig. 14–3). Patients should follow their written action plan as symptoms intensify or lung function deteriorates. Based on the initial response to β2-agonist therapy, the severity of the exacerbation can be assessed, and treatment can be appropriately intensified.1
In acute severe asthma, early and appropriate intensification of therapy is important to resolve the exacerbation, prevent relapse, and prevent severe airflow obstruction in the future. Starting therapy at home allows for rapid initiation and early assessment of response (see Fig. 14–3). Patients should follow their written action plan as symptoms intensify or lung function deteriorates. Based on the initial response to β2-agonist therapy, the severity of the exacerbation can be assessed, and treatment can be appropriately intensified.1
In patients with a good response to therapy, doubling the dose of ICS is no longer recommended, and a short course of oral corticosteroids should be considered. All patients with an incomplete response or whose response to an inhaled SABA lasts less than 1 hour should receive a short course of systemic corticosteorids.1 Corticosteroid therapy should continue until PEF is at least 70% of predicted or personal best.1 The SABA therapy can be continued at two to four inhalations every 3 to 4 hours for 24 to 48 hours until symptoms resolve. Continued reliance on an SABA for prolonged periods indicates a need to seek medical care.
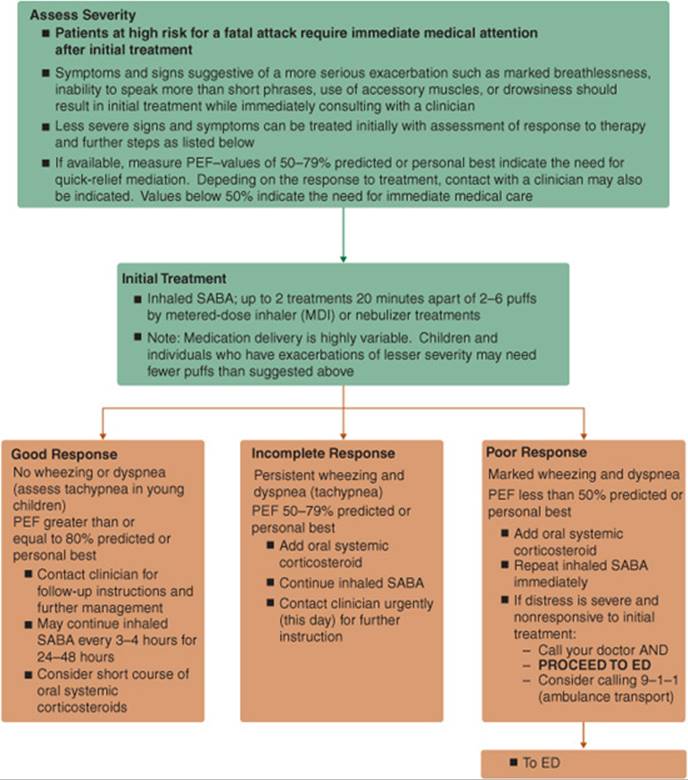
FIGURE 14–3. Management of Asthma Exacerbations: Home Treatment. (ED, emergency department; MDI, metered-dose inhaler; PEF, peak expiratory flow; SABA, short-acting β2-agonist.) (From Ref. 1.)
Patient Encounter, Part 3
Follow-up: Six months later, RB’s mother brings her back for a checkup. The patient has been using her albuterol inhaler 3 to 4 days a week to treat symptoms, and she is waking up about one to two times a month with shortness of breath. She has developed a sore mouth. White patches on the back of her tongue and the inside of her cheek are observed on examination.
Meds: Pulmicort Flexhaler 180 mcg/puff, one puff twice a day; proventil HFA two puffs as needed for symptoms; fluticasone nasal spray one puffin each nostril daily
What further information do you need to assess this patient’s asthma control?
How would you counsel this patient to prevent further adverse reactions?
Assuming that the patient is using her medications appropriately, how would you adjust this patient’s medication?
Patients with incomplete responses should contact their health care provider immediately for instructions. Those with a poor response should proceed directly to the emergency department.1 In the emergency department, baseline PEF measurements and oxygen saturation should be monitored. PEF should be monitored before and 15 to 20 minutes after bronchodilator administration. Treatment should be initiated as soon as lung function is assessed (Fig. 14–4). Multiple doses of inhaled ipratropium should be added to SABA therapy in patients with severe airflow obstruction.1 Dosages for emergency department and hospital use of quick-relief medications are shown in Table 14–4.
Patients with oxygen saturation less than 90% (less than 95% in children, pregnant women, and patients with coexisting heart disease) should receive oxygen with the dose adjusted to keep oxygen saturation above these levels.14Administration of low concentrations of oxygen (less than 30% of the fraction of inspired air) by nasal cannula or facemask is usually sufficient to reverse hypoxemia in most patients.
Routine antibiotic use is not warranted because the primary infectious agents associated with asthma exacerbations are viruses.1 Antibiotics should be reserved for situations when bacterial infection is strongly suspected (e.g., fever and purulent sputum, pneumonia, and suspected sinusitis).
In patients with impending respiratory failure, IV magnesium and heliox, a mixture of helium and oxygen that results in a lower density of inspired air and improved oxygen delivery, should be considered.1Theophylline is not recommended for treatment of acute asthma.1
Patients responding to therapy in the emergency department with a sustained response to inhaled β2- agonists (PEF greater than 70%) can be discharged home.1 Patients should have an inhaled SABA, be restarted on maintenance medications, and receive a 3-to 10-day course of oral corticosteroids. Patients who do not respond adequately to intensive therapy in the emergency department within 3 to 4 hours should be admitted to the hospital.
Special Populations
Pregnancy
Approximately 4% to 8% of pregnant women are affected by asthma with about one-third of them experiencing worsening asthma during pregnancy.42 Because uncontrolled asthma is a greater risk to the fetus than the risk of asthma medication use, it is safer for pregnant women to have asthma treated with medications than to experience worsening asthma. Consequently, asthma exacerbations should be managed aggressively with pharmacotherapy. The stepwise approach to asthma therapy in pregnancy is similar to that for the general population.
Budesonide has the most safety data in humans and is the preferred ICS; it is the only ICS classified as pregnancy category B. However, there are no data indicating that other ICS contribute to increased risk to the mother or fetus. Albuterol is the drug of choice for the treatment of asthma symptoms and exacerbations in pregnancy.42
Exercise-Induced Asthma
Exercise is one of the most common precipitants of asthma symptoms. Shortness of breath, wheezing, or chest tightness usually occur during or shortly after vigorous exercise, peak 5 to 10 minutes after stopping the activity, and resolve within 20 to 30 minutes.
Patients with exercise-induced asthma should warm up prior to vigorous exercise and cover the mouth and nose with a scarf or mask during cold weather. Increased physical conditioning and gradually decreasing the intensity of exercise prior to stopping may also help prevent bronchospasm. Pretreatment with albuterol 5 minutes prior to exercise is the treatment of choice and will protect against bronchospasm for 2 to 3 hours.1 Pretreatment with cromolyn sodium or nedocromil may also be effective in preventing bronchospasm but is not as effective as albuterol.20 Pretreatment with a leukotriene modifier and regular treatment with ICS also prevents bronchospasm associated with exercise. Consideration should be given to initiating long-term control therapy in mild intermittent asthmatics using multiple weekly doses of a bronchodilator to prevent asthma symptoms associated with exercise.

FIGURE 14–4. Management of asthma exacerbations: Emergency department and hospital-based care. (FEV1 forced expiratory volume in 1 second; ICS, inhaled corticosteroid; MDI, metered-dose inhaler, Pco2, partial arterial pressure of carbon dioxide; PEF, peak expiratory flow; SABA, short-acting β1-agonist [(quick-relief inhaler]; SaO2 oxygen saturation.) (From Ref. 1.)
Patient Encounter, Part 4: Emergency Department Visit
RB is brought to the emergency department short of breath and unable to speak in complete sentences. The symptoms started approximately 1½ hours ago, and she has already used four puffs of albuterol every 20 minutes for three doses. She has never been hospitalized for asthma previously. On exam, she has inspiratory and expiratory wheezes and appears to be in distress. She is leaning forward to breathe, pursing her lips, and has intercostal and supraclavicular retractions. Her heart rate is 120 bpm and her respiratory rate is 26 breaths per minute. A PEF measurement is 35% of predicted value and her O2saturation is 87%.
HomeMeds: Symbicort 160 mcg/4.5 mcg, one inhalation twice a day; proventil HFA two puffs as needed for symptoms; fluticasone nasal spray one puff in each nostril daily
Based on the information presented above, what initial treatment would you recommend for this patient?
What are the goals of treatment in this patient?
IfRB does not respond to therapy, what adjunctive therapies should be considered?
Aspirin Sensitivity
Patients with aspirin-sensitive asthma are usually adults and often present with the triad of rhinitis, nasal polyps, and asthma. In these patients, acute asthma may occur within minutes of ingesting aspirin or another nonsteroidal anti-inflammatory drug (NSAID). These patients should be counseled against using NS AIDs.1 Although acetaminophen is generally safe in this population, doses larger than 1 gram may cause acute asthmatic reactions in some patients.43 Patients with aspirin-sensitive asthma may tolerate cyclooxygenase-2 inhibitors; however, given the potentially serious adverse events that could occur in aspirin-sensitive asthmatics, the first dose of a cyclooxygenase-2 inhibitor should be given under the observation of a health care provider with rescue drugs available.44
OUTCOME EVALUATION
Chronic Asthma
• Assess the patient s inhaler technique frequently and always assess technique before stepping up therapy. Reeducate the patient on appropriate inhaler technique at every visit.
• Monitor symptoms such as wheezing, shortness of breath, chest tightness, cough, and nocturnal awakenings. Daytime symptoms should occur no more than twice a week, and nocturnal symptoms should occur no more than twice a month in adolescents and adults and no more than once a month in children younger than 12 years of age. Patients with more frequent symptoms should have their long-term control medications increased.
• In persons 12 years of age and older, monitor lung function. FEV1 or PEF should remain above 80% of predicted or personal best. Patients with PEF rates consistently greater than 80% over several months should be evaluated for a stepdown in long-term control therapy. Patients with a PEF less than 80% of personal best should begin to monitor PEF twice daily and consult their asthma action plan. Patients with a PEF less than 50% of personal best should immediately use their SABA and consult their asthma action plan.
• Monitor patient activity levels. Inability of a patient to perform routine physical activities indicates inappropriate therapy, and long-term control medications should be increased.
• In individuals 12 years of age and older, monitor asthma impairment using a validated questionnaire to ensure asthma is well controlled.
• Monitor frequency of patient exacerbations. Frequent exacerbations, unscheduled clinic visits, emergency department visits, and hospitalizations due to asthma may indicate a nonadherent patient or the need to step up long-term control medications.
• Monitor use of long-term control medications to ensure adherence. Reeducate nonadherent patients on the importance of these medications for asthma control.
• Monitor use of inhaled SABAs. Their use more than twice a week in intermittent asthma may indicate the need to initiate long-term control therapy. Use of more than one canister per month indicates the need to step up long-term control therapy.
• Monitor for adverse events from medications, including candidiasis and dysphonia from ICS.
• Monitor the patient’s immunization status and provide an annual influenza vaccination if warranted.
Acute Severe Asthma
• Monitor PEF, which should increase to greater than 70% of personal best or predicted after the first three doses of an inhaled SABA.
• Monitor patients for hypoxemia. Oxygen saturation should be greater than 90% in adults and greater than 95% in children, pregnant women, and patients with coexisting cardiovascular disease.
• In patients with severe exacerbations, monitoring of Pco2 should be considered. Patients with acute asthma usually have a respiratory alkalosis, and a normal or increased Pco2 indicates the potential for respiratory failure.
• Monitor serum potassium in patients receiving high-dose or continuous nebulization of SABA. Serum potassium concentrations should be obtained upon hospital admission, and if the patient is hypokalemic, every 4 hours (after each 30 to 40 mEq [mmol] of replacement) until the serum potassium is stable. Serum potassium should be monitored every 3 to 6 months after discharge.
Patient Care and Monitoring
Chronic Asthma
1. Obtain a thorough medical history focusing on disease states that may worsen asthma severity.
2. Ask the patient about the frequency and severity of symptoms, when symptoms occur, and whether or not symptoms are associated with exposure to known allergens. Ask about previous emergency department visits and hospitalizations due to asthma.
3. Use the patient’s level of impairment and risk of future adverse outcomes to classify disease severity.
4. Explain the goals of therapy and ask whether the patient has any personal treatment goals.
5. Develop a patient education plan that fits the patient’s needs. Educate about the differences between the asthmatic and normal lung and what happens to the lung during an asthma attack. Counsel the patient on how their medications work and differentiate between long-term control and quick relief medications.
6. Provide a specific allergen avoidance plan and counsel all patients to avoid second-hand tobacco smoke.
7. Demonstrate the appropriate use of drug delivery devices and peak flow meters; then have the patient perform these activities for you. If the task is performed incorrectly, demonstrate the skill again, emphasizing the incorrect step and have the patient redemonstrate the skill.
8. Prepare a patient-specific self-monitoring plan and review it with the patient. Educate the patient on the signs and symptoms of asthma deterioration and when and how to take rescue actions.
9. Assess the patient’s adherence to long-term control therapy. Stress the importance of adherence if necessary. Evaluate the complexity of the treatment plan and simplify it if possible.
10. Assess the patient for adverse effects such as candidiasis and dysphonia associated with ICS.
11. Evaluate therapy on a regular basis. Assess the patient’s control of asthma by evaluating the patient’s impairment due to asthma and their risk for future adverse events due to asthma. Step long-term control therapy up or down based on these parameters. Before stepping up therapy, reassess the patient’s inhaler technique to assure appropriate drug delivery.
Acute Severe Asthma
1. Assess the patient’s PER.
2. Assess whether or not the patient can use an MDI with a spacer or holding chamber. If the patient cannot use the device, determine whether someone can assist the patient with the inhaler device, or whether a nebulizer is necessary.
3. Initiate therapy with an SABA and 2 to 6 L/min of oxygen if needed.
4. Perform a brief medical history to determine the time of symptom onset, symptom severity, symptom severity in relation to previous exacerbations, current medications, previous emergency department visits or hospitalizations due to asthma, previous history of respiratory failure, and psychiatric or psychological disorders.
5. Assess the patient’s general appearance, use of accessory muscles, respiratory rate, heart rate, lung sounds, presence of pulsus paradoxus, PEF, and oxygen saturation.
6. Reassess pulmonary function every 20 to 30 minutes. If there was not an immediate response to the inhaled SABA, initiate systemic corticosteroid therapy. If the patient is not improving, add ipratropium to the patient’s therapy and continue with a high-dose inhaled SABA.
7. Assess the patient for hospitalization or discharge home.
8. If the patient is discharged home, ensure that the patient has an SABA, review the appropriate technique for inhaler use, and ensure that the patient has a prescription for 3 to 10 days of an oral corticosteroid. Consider starting ICS and providing the patient with a 1 to 2 month supply of the medication.
9. Restart the patient on maintenance therapy. Instruct the patient on what to do if asthma worsens and to follow-up with his/her health care provider in 1 to 4 weeks.
Abbreviations Introduced in This Chapter


 Self-assessment questions and answers are available at http://www.mhpharmacotherapy.com/pp.html.
Self-assessment questions and answers are available at http://www.mhpharmacotherapy.com/pp.html.
REFERENCES
1. NHLBI National Asthma Education and Prevention Program, Expert Panel Report-3. Guidelines for the Diagnosis and Management of Asthma. NIH Publication No. 07-4051. Bethesda, MD: U.S. Department of Health and Human Services, 2007, http://www.nhlbi.nih.gov/guidelines/asthma
2. American Lung Association. Trends in asthma morbidity and mortality. American Lung Association Epidemiology & Statistics Unit Research and Program Services. November 2007, http://www.lungusa.org.
3. Moorman JE, Rudd RA, Johnson CA, et al. National surveillance for asthma—United States, 1980-2004. MMWR Morb Mortal Wkly Rep 2007;56(08):1-14, 18-54.
4. Weiss KB, Sullivan SD. The health economics of asthma and rhinitis. I. Assessing the economic impact. J Allergy Clin Immunol 2001;107:3-8.
5. Reed CE. The natural history of asthma. J Allergy Clin Immunol 2006;110:543-548.
6. Busse WW, Lemanske RF Jr. Advances in immunology: Asthma. N Engl J Med 2001;344:350-362.
7. Larché M, Robinson DS, Kay AB. The role of T lymphocytes in the pathogenesis of asthma. J Allergy Clin Immunol 2003;111:450-463.
8. Robinson DS. The role of mast cells in asthma: Induction of airway hyperresponsiveness by interaction with smooth muscle? J Allergy Clin Immunol 2004;114:58-65.
9. Cohn L, Elias JA, Chupp GL. Asthma: Mechanisms of disease persistence and progression. Annu Rev Immunol 2004;22:789-815.
10. Bradding P, Walls AF, Holgate ST. The role of the mast cell in the pathophysiology of asthma. J Allergy Clin Immunol 2006;117:1277-1284.
11. Lemanske RF Jr, Busse WW. Asthma. J Allergy Clin Immunol 2003;111:S502-S519.
12. Beckett PA, Howarth PH. Pharmacotherapy and airway remodeling in asthma? Thorax 2003;58:163-174.
13. Bousquet J, Khaltaev N, Cruz AA, et al. Allergic rhinitis and its impact on asthma (ARIA) 2008 update (in collaboration with the World Health Organization, GA(2) LEN and AllerGen). Allergy 2008;63(Suppl 86):8-160.
14. Rodrigo GJ, Rodrigo C, Hall JB. Acute asthma in adults: A review. Chest 2004;125:1081-1102.
15. O’Connor GT. Allergen avoidance in asthma: What do we do now? J Allergy Clin Immunol 2005;116:26-30.
16. Talbot TR, Hartert TV, Mitchel E, et al. Asthma as a risk factor for invasive pneumococcal disease. N Engl J Med 2005;352:2082-2090.
17. Newman SP. Spacer devices for metered dose inhalers. Clin Pharmacokinet 2004;43:349-360.
18. de Benedictis FM, Selvaggio D. Use of inhaler devices in pediatric asthma. Paediatr Drugs 2003;5:629-638.
19. Gibson PG, Powell H. Written action plans for asthma: An evidence-based review of the key components. Thorax 2004;59:94-99.
20. Kelly HW, Sorkness CA. Asthma. In: Dipiro JT, Talbert RL, Yee GC, et al., eds. Pharmacotherapy: A Pathophysiologic Approach. 7th ed. New York: McGraw-Hill, 2008:464-493.
21. Nowak RM, Emerman CL, Shaefer K, et al. Levalbuterol compared with racemic albuterol in the treatment of acute asthma: Results of a pilot study. Am J Emerg Med 2004;22:29-36.
22. Sin DD, Man J, Sharpe H, et al. Pharmacological management to reduce exacerbations in adults with asthma: A systematic review and meta-analysis. JAMA 2004;292:367-376.
23. Bateman ED, Boushey HA, Bousquet J, et al. Can guideline-defined asthma control be achieved? The gaining optimal asthma control study. Am J Respir Crit Care Med 2004;170:836-844.
24. Masoli M, Weatherall M, Holt S. Moderate dose inhaled corticosteroids plus salmeterol versus higher doses of inhaled corticosteroids in symptomatic asthma. Thorax 2005;60:730-734.
25. Nelson HS, Weiss ST, Bleeker ER, et al., and the Smart Study Group. The salmeterol multicenter asthma research trial: A comparison of usual pharmacotherapy for asthma or usual pharmacotherapy plus salmeterol. Chest 2006;129:15-26.
26. Kelly HW. Rationale for the major changes in the pharmacotherapy section of the National Asthma Education and Prevention Program Guidelines. J Allergy Clin Immunol 2007;120:989-994.
27. Nelson HS. Is there a problem with inhaled long-acting β-adrenergic agonists? J Allergy Clin Immunol 2006;117:3-16.
28. Bateman E, Neslon H, Bousquet J, et al. Meta-analysis: Effects of adding salmeterol to inhaled corticosteroids on serious asthma-related events. Ann Intern Med 2008;149:33-42.
29. Currie GP, Devereux GS, Lee DKC, et al. Recent developments in asthma management. BMJ 2005;330:585-589.
30. Szefler SJ, Martin RJ, King TS, et al. Significant variability in response to inhaled corticosteroids for persistent asthma. J Allergy Clin Immunol 2002;109:410-418.
31. Martin RJ, Szefler SJ, Chinchilli VM, et al. Systemic effect comparisons of six inhaled corticosteroid preparations. Am J Respir Crit Care Med 2002;165:1377-1383.
32. Masoli M, Weatherall M, Holt S, et al. Clinical dose response relationship of fluticasone propionate in adults with asthma. Thorax 2004;59:16-20.
33. Guilbert TW, Morgan WJ, Zeiger RS, et al. Long-term inhaled corticosteroids in preschool children at high risk for asthma. N Engl J Med 2006;354:1985-1997.
34. Rodrigo GJ, Castro-Rodrigo JA. Anticholinergics in the treatment of children and adults with acute asthma: A systematic review with meta-analysis. Thorax 2005;60:740-746.
35. Sorkness CA, Lemanske RF Jr, Mauger DT, et al. Long-term comparison of 3 controller regiments for mild-moderate persistent childhood asthma: The pediatric asthma controller trial. J Allergy Clin Immunol 2007;119:64-72.
36. Barnes PJ. Theophylline. New perspective on an old drug. Am J Respir Crit Care Med 2003;167:813-818.
37. Barnes PJ. Theophylline: New perspective on an old drug. Am J Resp Crit Care Med 2003;167:813-818.
38. Davis LA. Omalizumab: A novel therapy for allergic asthma. Ann Pharmacother 2004;38:1236-1242.
39. Wu AC, Paltiel D, Kuntz KM, et al. Cost-effectiveness of omaluzimab in adults with severe asthma: Results from the asthma policy model. J Allergy Clin Immunol 2007;120:1146-1152.
40. Limb SL, Starke PR, Lee CE, et al. Delayed onset and protracted progression of anaphylaxis after omalizumab administration in patients with asthma. J Allergy Clin Immunol 2007;120:1378-1381.
41. McFadden ER Jr. Acute severe asthma. Am J Respir Crit Care Med 2003;168:740-759.
42. National Heart Lung and Blood Institute, National Asthma Education and Prevention Program Asthma and Pregnancy Working Group. NAEPP expert panel report. Managing asthma during pregnancy: Recommendations for pharmacologic treatment—2004 update. J Allergy Clin Immunol 2005;115:34-46.
43. Eneli I, Sadri K, Camargo C, et al. Acetaminophen and the risk of asthma: The epidemiologic and pathophysiologic evidence. Chest 2005;127:604-612.
44. West PM, Fernández C. Safety of Cox-2 inhibitors in asthma patients with aspirin hypersensitivity. Ann Pharmacother 2003;37:1497-1501.