Robert J. Straka, David Parra, and Kade T. Birkeland
LEARNING OBJECTIVES
Upon completion of the chapter, the reader will be able to:
1. Classify blood pressure (BP) levels and treatment goals.
2. Recognize the underlying causes and contributing factors in the development of hypertension.
3. Describe the appropriate measurement of BP.
4. Recommend appropriate lifestyle modifications and pharmacotherapy for patients with hypertension.
5. Identify populations requiring special consideration when designing a treatment plan.
6. Construct an appropriate monitoring plan to assess hypertension treatment.
KEY CONCEPTS
![]() Hypertension is widely prevalent and accounts for significant morbidity and mortality, as well as billions of dollars in direct and indirect costs.
Hypertension is widely prevalent and accounts for significant morbidity and mortality, as well as billions of dollars in direct and indirect costs.
![]() The cause of hypertension is unknown in the majority of cases (primary hypertension), but for patients with secondary hypertension, specific causes can be identified.
The cause of hypertension is unknown in the majority of cases (primary hypertension), but for patients with secondary hypertension, specific causes can be identified.
![]() Patients failing to achieve goal BP despite maximum doses of three antihypertensives including a diuretic should be carefully screened for resistant hypertension.
Patients failing to achieve goal BP despite maximum doses of three antihypertensives including a diuretic should be carefully screened for resistant hypertension.
![]() The pathophysiology of primary hypertension is heterogeneous, but ultimately exerts its effects through the two primary determinants of BP: cardiac output (CO) and peripheral resistance (PR).
The pathophysiology of primary hypertension is heterogeneous, but ultimately exerts its effects through the two primary determinants of BP: cardiac output (CO) and peripheral resistance (PR).
![]() Appropriate technique in measuring BP is a vital component to the diagnosis and continued management of hypertension in the outpatient setting.
Appropriate technique in measuring BP is a vital component to the diagnosis and continued management of hypertension in the outpatient setting.
![]() Drug selection for the management of patients with hypertension should be considered as adjunctive to nonpharmacologic approaches for BP lowering, and ultimately the attainment of target BP in many cases may be more important than the antihypertensive agent used.
Drug selection for the management of patients with hypertension should be considered as adjunctive to nonpharmacologic approaches for BP lowering, and ultimately the attainment of target BP in many cases may be more important than the antihypertensive agent used.
![]() Implementation of lifestyle modifications successfully lowers BP, often with results similar to those of therapy with a single antihypertensive agent.
Implementation of lifestyle modifications successfully lowers BP, often with results similar to those of therapy with a single antihypertensive agent.
![]() An approach to selection of drugs for the treatment of patients with hypertension should be evidence-based. Consideration should be given to the individual’s comorbidities, coprescribed medications, and practical patient-specific issues including costs.
An approach to selection of drugs for the treatment of patients with hypertension should be evidence-based. Consideration should be given to the individual’s comorbidities, coprescribed medications, and practical patient-specific issues including costs.
![]() Specific antihypertensive therapy is warranted for certain patients with comorbid conditions that may elevate their level of risk for cardiovascular disease (CVD).
Specific antihypertensive therapy is warranted for certain patients with comorbid conditions that may elevate their level of risk for cardiovascular disease (CVD).
![]() The frequency of follow-up visits for patients with hypertension will vary based on individual cases, but will be influenced by severity of hypertension, comorbidities, and choice of agent selected.
The frequency of follow-up visits for patients with hypertension will vary based on individual cases, but will be influenced by severity of hypertension, comorbidities, and choice of agent selected.
INTRODUCTION
Despite efforts to promote awareness, treatment, and the means available to aggressively manage high blood pressure (BP), trends over the past 15 years demonstrate only modest improvements in its treatment and control. National and international organizations continually refine their recommendations of how clinicians should approach the management of patients with high BP, and although approaches vary to some degree, there are clear themes that emerge regardless of which national or international organization’s algorithm is followed. The purpose of this chapter is to provide a summary of key issues associated with the management of patients with hypertension. We will discuss the basic approach to treating patients with hypertension and provide a functional summary of the currently prevailing themes of national guidelines, including their grounding in relevant landmark trials. Finally, we will summarize salient pharmacotherapeutic issues essential for clinicians to consider when managing patients with hypertension.
Various algorithms recommending nonpharmacologic and pharmacologic management for typical and atypical patients are proposed, with the underlying theme that achievement of BP targets mitigate end-organ damage, leading to substantial reductions in stroke, myocardial infarction (MI), end-stage renal disease, and heart failure. Although references to other algorithms will be mentioned, this chapter will focus primarily on the Seventh Report of the Joint National Committee on Prevention, Detection, Evaluation and Treatment of High Blood Pressure, more commonly referred to as the Joint National Committee Seventh Report (JNC 7) report,1 with additional reference to recent recommendations from the American Heart Association2 and European Society of Cardiology.3 It should be noted that an update of the JNC 7 report (JNC 8), as part of an integrated set of cardiovascular risk reduction guidelines, is expected to be released in 2010.3
The JNC 7 report describes four stages of BP classification and provides guidance on nonpharmacologic and pharmacologic approaches to managing patients with hypertension. The four stages of BP classification include normal, prehypertension, stage 1 hypertension, and stage 2 hypertension (Table 5–1). These stages are defined as such to connote a level of risk and thus the need for varying intensities of intervention with drug therapy (Fig. 5–1). With the exception of individuals with “compelling indications,” recommendations for drug therapy typically begin with one or two (in the case of stage 2) antihypertensive drugs as an initial step. Specific drug selection is guided by the presence of compelling indications—specific comorbid conditions. These compelling indications, such as heart failure, diabetes, and chronic kidney disease (CKD), represent specific conditions for which explicit evidence in the literature exists to document the utility of a particular agent or class of agents. Selection of drug therapy consequently involves an iterative process of considering multiple antihypertensive drugs as needed to achieve target BPs of less than 140/90 mm Hg for all patients, with more aggressive targets of less than 130/80 mm Hg for patients with diabetes or chronic) greater than 3 months (kidney disease) estimated glomerular filtration rate [GFR] less than 60 mL/min/1.73 m2 or the presence of albuminuria [300mg/day or 200 mg/g creatinine]).1 In addition, the AHA scientific statement expands those in whom a lower BP target of less than 130/80 mm Hg should be pursued to include patients with known coronary artery disease or coronary artery disease risk equivalents (carotid artery disease, peripheral arterial disease, abdominal aortic aneurysm), or a 10-year Framingham risk score of greater than 10%. See Dyslipidemias chapter. In patients with left ventricular dysfunction, additional BP lowering to a target of less than 120/80 mm Hg may also be considered.2
EPIDEMIOLOGY
![]() Hypertension is widely prevalent and accounts for significant morbidity and mortality, as well as billions of dollars in direct and indirect costs. Worldwide prevalence of hypertension is estimated to include 1 billion individuals. There are an estimated 7 million deaths per year that may be related to the diagnosis of hypertension.4 The prevalence of hypertension in the United States is among the highest in the world and is estimated to include 73 million individuals (1 in 3 adults) with an estimated 69.4 billion dollars spent annually in direct and indirect costs.5 Furthermore, it is estimated that 37.4% of the U.S. population older than 20 years of age has prehypertension.
Hypertension is widely prevalent and accounts for significant morbidity and mortality, as well as billions of dollars in direct and indirect costs. Worldwide prevalence of hypertension is estimated to include 1 billion individuals. There are an estimated 7 million deaths per year that may be related to the diagnosis of hypertension.4 The prevalence of hypertension in the United States is among the highest in the world and is estimated to include 73 million individuals (1 in 3 adults) with an estimated 69.4 billion dollars spent annually in direct and indirect costs.5 Furthermore, it is estimated that 37.4% of the U.S. population older than 20 years of age has prehypertension.
The prevalence of hypertension differs based on age, sex, and ethnicity. As individuals become older, their risk of systolic hypertension increases. Individuals 55 years of age who do not have hypertension are estimated to have a lifetime risk of 90% of eventually developing hypertension. In the United States, hypertension is slightly more prevalent in women (33.6%) than men (33.2%).5 In addition, age-adjusted prevalence of hypertension is highest in non-Hispanic blacks or African Americans (41%) when compared to non-Hispanic whites (28.1%) and Mexican Americans (22%). However, Mexican Americans have lower rates of treatment and control.6
Table 5–1 Classification of BP in Children, Adolescents, and Adultsa

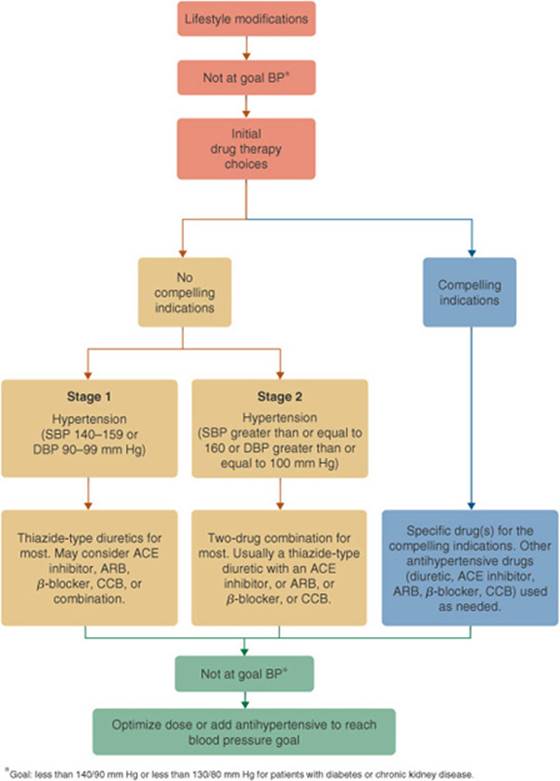
FIGURE 5–1. Algorithm for treatment of hypertension when patients are not at their goal BP. Compelling indications refer to specific indications where the selection of a particular antihypertensive drug class for a defined high-risk population is highly recommended. These recommendations are usually based on results from landmark randomized placebo-controlled outcome trials or consensus statements from clinical guidelines and are usually based on findings documenting superior outcomes in terms of morbidity and mortality. (ACE, angiotensin-converting enzyme; ARB, angiotensin receptor blocker; BP, blood pressure; CCB, calcium channel blocker; DBP, diastolic blood pressure; SBP, systolic blood pressure.) (From Ref. 1.)
Hypertension is strongly associated with type 2 diabetes.7 The added comorbidity of hypertension in diabetes leads to a higher risk of cardiovascular disease (CVD), stroke, renal disease, and diabetic retinopathy leading to greater health care costs.8
ETIOLOGY
![]() In the majority of patients (greater than 90%), the cause of hypertension is unknown and is referred to as essential, or more appropriately, as primary hypertension.9 However, in some patients there is an identifiable cause of which the most common are:2
In the majority of patients (greater than 90%), the cause of hypertension is unknown and is referred to as essential, or more appropriately, as primary hypertension.9 However, in some patients there is an identifiable cause of which the most common are:2
• CKD
• Coarctation of the aorta
• Cushing’s syndrome and other glucocorticoid excess states
• Drug induced/related (Table 5–2)
• Pheochromocytoma
• Primary aldosteronism and other mineralocorticoid excess states
• Renovascular hypertension
• Sleep apnea
• Thyroid or parathyroid disease
Hypertension caused by any of these conditions is referred to as secondary hypertension. Identification of a secondary cause of hypertension is often not initially pursued unless suggested by routine clinical and laboratory evaluation of the patient, or failure to achieve BP control.
Table 5–2 Causes of Resistant Hypertension
Apparent Resistance
Improper BP measurement
Failure to receive or take antihypertensive medication
Inadequate doses (subtherapeutic)
Improper antihypertensive selection or combination
White coat effect
True Resistance
Secondary hypertension
Medication effects and interactions
Nonsteroidal anti-inflammatory medications
Sympathomimetics (decongestants, anorectics, and stimulants)
Cocaine, amphetamines, and other illict drugs
Recent caffeine or nicotine intake
Oral contraceptive hormones
Adrenal steroid hormones
Erythropoietin
Natural licorice (including some chewing tobacco)
Cyclosporine and tacrolimus
Herbal products (ma huang, guarana, bitter orange)
Volume overload
Excess sodium intake
Inadequate diuretic therapy
Fluid retention from kidney disease or potent vasodilators (e.g., minoxidil)
Comorbidities
Obesity
Excess alcohol intake
Chronic pain syndromes
Intense vasoconstriction (arteritis)
Anxiety-induced hyperventilation/panic attacks
Genetic variation
Genetic differences in drug efficacy or metabolism
From Ref. 1 and Kaplan NM, Sica DA. Resistant Hypertension in Izzo JL Jr, Black HR eds. Hypertension Primer. The Essentials of High Blood Pressure. 4th ed. Philadelphia: Lippincott Williams & Wilkins; 2008: 348–350.
In addition to primary and secondary hypertension, the clinician may encounter what is referred to as resistant hypertension.![]() Patients failing to achieve goal BP despite maximum doses of three antihypertensives including a diuretic should be carefully screened for resistant hypertension. Several causes of resistant hypertension are listed in Table 5–2 and should be carefully considered in such patients.
Patients failing to achieve goal BP despite maximum doses of three antihypertensives including a diuretic should be carefully screened for resistant hypertension. Several causes of resistant hypertension are listed in Table 5–2 and should be carefully considered in such patients.
PATHOPHYSIOLOGY
![]() The pathophysiology of primary hypertension is heterogeneous, but ultimately exerts its effects through the two primary determinants of BP: cardiac output (CO) and peripheral resistance (PR). The processes influencing these two determinants are numerous and complex (Fig. 5–2).9 The underlying cause of primary hypertension is unknown and most likely multifactorial. Although numerous hypotheses exist, an in-depth review of these is beyond the scope of this text and the reader is referred to additional texts for more information including discussion on the pathophysiology behind secondary causes of hypertension.10
The pathophysiology of primary hypertension is heterogeneous, but ultimately exerts its effects through the two primary determinants of BP: cardiac output (CO) and peripheral resistance (PR). The processes influencing these two determinants are numerous and complex (Fig. 5–2).9 The underlying cause of primary hypertension is unknown and most likely multifactorial. Although numerous hypotheses exist, an in-depth review of these is beyond the scope of this text and the reader is referred to additional texts for more information including discussion on the pathophysiology behind secondary causes of hypertension.10
Genetics
Multiple genetic polymorphisms have been associated with hypertension. It is estimated that up to 30% to 50% of variability in BP may have a genetic basis.11 The majority of these polymorphisms appear to be involved directly or indirectly in renal sodium reabsorption, which may represent future therapeutic drug targets.12 In addition to the identification of genetic factors contributing to the development of hypertension, explorations into the genetic basis of variability in response to drug therapy are at early stages. Although advances are made on both fronts, replication of early findings identifying genetic variations from genome-wide association scans (GWAS)13 clearly require more comprehensive study before investigators, let alone clinicians, are in a position to utilize these findings.14
Cardiac Output
CO is an important determinant of BP. Factors which elevate CO may, in theory, contribute to the development of primary hypertension. Increases in CO and subsequently BP may arise from factors that increase preload (fluid volume) or myocardial contractility. Nonetheless, even if increased CO may be involved in the development of primary hypertension, these increases do not appear to persist over time. As a consequence, elevated CO is not considered a hemodynamic hallmark of established primary hypertension.
Sodium Regulation
The contribution of sodium to the development of primary hypertension is related to excess sodium intake and/or abnormal sodium excretion by the kidneys. Proposed mechanisms supporting either of these are numerous and complex. However, it is generally accepted that dietary salt is associated with increases in BP that can be lowered with reduction of sodium intake.1,15 There appears to be a threshold effect of sodium intake in the range of 50 to 100 mmol/day (1.2–2.4 g of sodium per day, which is equivalent to 3–6 g of sodium chloride per day [50–100 mmol/day]) and its impact on BP. The mean sodium intake per day is 175 mmol (4.1 g) for men and 120 mmol (2.7 g) for women in the United States, with the majority derived from processed foods.1 Adherence to sodium restriction is important as up to 50% of all individuals appear to be sodium-sensitive and thus susceptible to a high dietary sodium intake.9
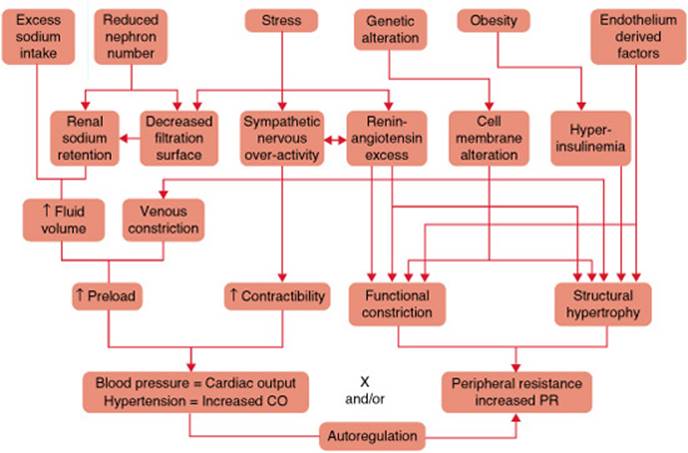
FIGURE 5–2. Factors involved in the pathogenesis of hypertension are summarized. Some of the factors involved in the control of BP affect the basic equation: BP = CO × PR. The figure depicts the complex nature of various factors that may play a role in the development of hypertension. Each of these factors may individually or collectively modulate BP through its actions on various physiologic systems at the cellular, organ, and organ system level. (BP, blood pressure; CO, cardiac output; PR, peripheral resistance.) (From Kaplan NM. Primary hypertension: Pathogenesis. In: Kaplan’s Clinical Hypertension. 9th ed. Philadelphia: Lippincott) Williams & Wilkins; 2006: 63, with permission.)
Renin-Angiotensin-Aldosterone System
Since the discovery of renin over 100 years ago, the renin-angiotensin-aldosterone system (RAAS) has been extensively studied as a prime target or site of action for many effective antihypertensives.16 Renin is produced and stored in the juxtaglomerular cells of the kidney, and its release is stimulated by impaired renal perfusion, salt depletion, β1-adrenergic stimulation. The release of renin is the rate-limiting step in the eventual formation of angiotensin II, which is primarily responsible for the pressor effects mediated by the RAAS (Fig. 5–3). Evidence indicates that renin’s pressor effects occur at the cellular level (autocrine), the local environment (paracrine), and throughout the systemic circulation (endocrine).17 The role of the RAAS in primary hypertension is supported by the presence of high levels of renin, suggesting that the system is inappropriately activated. Proposed mechanisms behind this inappropriate activation include increased sympathetic drive, defective regulation of the RAAS (nonmodulation), and the existence of a subpopulation of ischemic nephrons that release excess renin.9 However, there are also patients with primary hypertension and low levels of renin. This observation suggests that alternate mechanisms for hypertension unrelated to renin levels or activity may be in play.18 Ongoing studies utilizing the oral direct renin inhibitor (DRI) known as aliskiren may advance our understanding of the role this DRI may play when combined with other agents affecting the RAAS.19
Sympathetic Overactivity
Overactivation of the sympathetic nervous system (SNS) may also play a role in the development and maintenance of primary hypertension for some individuals. Among other effects, direct activation of the SNS may lead to enhanced sodium retention, insulin resistance, and baroreceptor dysfunction.9 Regardless of which mechanism(s) underlies the role the SNS may play in the development of primary hypertension, the SNS remains a target of many antihypertensive agents.
Peripheral Resistance
Elevated peripheral arterial resistance is the hemodynamic hallmark of primary hypertension. The increase in PR typically observed may be due to a reduction in the arterial lumen size as a result of vascular remodeling. This remodeling, or change in vascular tone, may be modulated by various endothelium-derived vasoactive substances, growth factors, and cytokines. This increase in arterial stiffness or reduced compliance results in the observed increase in systolic BP.9
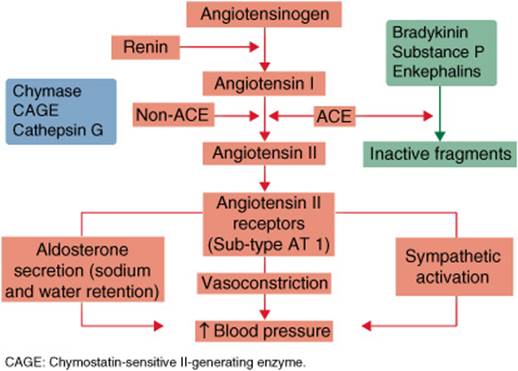
FIGURE 5–3. Diagram of the RAAS, a key system involved in the modulation of BP. The diagram depicts the pathways involved in the action of various antihypertensives including ACE inhibitors, ARBs, diuretics, and aldosterone antagonists. By inhibiting the action of angiotensin-converting enzymes, ACE inhibitors reduce both the formation of the vasoconstrictor angiotensin II, and the degradation of vasodilating substances including bradykinin. ARBs primarily act through inhibition of the action of angiotensin II on the angiotensin-1 receptors that modulate vasoconstriction. Aldosterone antagonists directly inhibit the actions of aldosterone, while diuretics affect sodium and water retention at a renal level. (ACE, angiotensin-converting enzyme; ARB, angiotensin receptor blockers; AT1, angiotensin-1; BP, blood pressure; RAAS, renin-anglotensin aldosterone system.)
Other Contributing Processes and Factors
Many other processes are proposed to contribute to the development of hypertension, including obesity, physical inactivity, insulin resistance, potassium and magnesium depletion, chronic moderate alcohol consumption, and transient effects of cigarette smoking and caffeine intake.9 The assessment of global cardiovascular risk in all hypertensive patients should be part of the management plan while also pursuing target BPs through nonpharmacologic and pharmacologic means. Regardless of the initiating process or processes leading to the development of hypertension, the ultimate goal is to reduce the risk of cardiovascular events and minimize target organ damage. This clearly requires the early identification of risk factors and treatment of patients with hypertension.
CLINICAL PRESENTATION AND CO-EXISTING RISK FACTORS
![]() Appropriate technique in measuring BP is a vital component to the diagnosis and continued management of hypertension in the outpatient setting. Accurate measurement of a patient’s BP identifies and controls for factors that may influence the variability in the measure. Failure to consider how each of these factors may influence BP measurement results in significant variation in measurements, leading to misclassification or inaccurate assessments of risk. Factors including body position, cuff size, device selection, auscultatory technique, and dietary intake prior to the clinic visit may contribute to such inaccuracies. Clinicians should instruct patients to avoid exercise, alcohol, caffeine, or nicotine consumption 30 minutes before BP measurement. Patients should be sitting comfortably with their back supported and arm free of constrictive clothing with legs uncrossed and feet flat on the floor for a minimum of 5 minutes before the first reading.
Appropriate technique in measuring BP is a vital component to the diagnosis and continued management of hypertension in the outpatient setting. Accurate measurement of a patient’s BP identifies and controls for factors that may influence the variability in the measure. Failure to consider how each of these factors may influence BP measurement results in significant variation in measurements, leading to misclassification or inaccurate assessments of risk. Factors including body position, cuff size, device selection, auscultatory technique, and dietary intake prior to the clinic visit may contribute to such inaccuracies. Clinicians should instruct patients to avoid exercise, alcohol, caffeine, or nicotine consumption 30 minutes before BP measurement. Patients should be sitting comfortably with their back supported and arm free of constrictive clothing with legs uncrossed and feet flat on the floor for a minimum of 5 minutes before the first reading.
Systolic and diastolic BP tend to increase when the cuff size is too small. Ideally, the cuff bladder should encircle at least 80% of the arm’s circumference to ensure a more accurate measurement of BP.
Mercury sphygmomanometers are recommended for routine office measurements, but concerns of patient exposure and environmental contamination of mercury has fostered the development of other devices to measure BP. However, there is no general consensus among health care providers as to an acceptable replacement for mercury sphygmomanometers.
To reduce deviations in BP measurement in the clinic, the patient and clinician should not talk during BP readings. The measurement arm is supported and positioned at heart level. If a mercury or aneroid device is used, then the palpatory method must be used first to estimate the systolic BP.20 If an automated device is used, this is not necessary. After the patient’s cuff is inflated above the systolic pressure, the mercury column should drop at a rate of 2 to 3 mm Hg/s. A stethoscope placed over the brachial artery in the antecubital fossa identifies the first and last audible Korotkoff sounds, which should be taken as systolic and diastolic pressure, respectively. A minimum of two readings at least 1 minute apart are then averaged. If measurements vary by more than 5 mm Hg between the two readings, then one or two additional BP measurements are collected and the multiple readings averaged. BP classification is based on the average of two or more properly measured, seated BP readings on each of two or more office visits. Details and further recommendations for accurate measurement of BP in special populations can be reviewed in the American College of Cardiology/American Heart Association (ACC/AHA) BP Measurement in Humans Statement for health care professionals.20
Finally, the measurement of clinic or office BPs is poorly correlated with assessments of BP in other settings. Consequently, under select circumstances, clinicians are increasingly using 24-hour ambulatory BP monitoring devices and home BP monitors. These tools are useful in identifying patients with white coat hypertension or with elevations of BP during nighttime. They may also aid in the management of refractory hypertensives with minor target organ damage, those with suspected autonomic neuropathy, those with hypotensive symptoms, and patients with large differences between home and clinic BP measurements. Benefits derived from alternative BP monitoring may be of greater prognostic significance than traditional office based measurements.20
Clinical Presentation and Diagnosis of Primary Hypertension
General
Age: Prevalence of hypertension is likely to be highest with middle-aged or older patients
Sex: In the United States, hypertension is slightly more prevalent in women (33.6%) than men (33.2%)
Symptoms
The patient with primary hypertension may be asymptomatic yet still have major CVD risk factors
Signs
Adult patients have an average of two or more BP readings (SBP and DBP) on two seperate occassions indicating either:

Laboratory Tests (not necessarily indicative of hypertension, but should be measured in patients with hypertension)
Fasting lipid panel:
• Low-density lipoprotein greater than 160 mg/dL (4.14 mmol/L)
• Total cholesterol greater than 240 mg/dL (6.22 mmol/L)
• HDL less than 40 mg/dL (1.04 mmol/L)
• Triglycerides greater than 200 mg/dL (2.26 mmol/L)
Fasting plasma glucose or hemoglobin A1c (does not need to be fasting):
• Impaired fasting glucose is a glucose of 100–125 mg/dL (5.55–6.94 mmol/L)
• Diagnosis of diabetes with fasting glucose greater than or equal to 126 mg/dL (6.99 mmol/L) or a hemoglobin A1c greater than or equal to 6.5% on two separate occasions, or a random plasma glucose reading of greater than or equal to 200 mg/L (11.1 mmol/L) with symptoms of diabetes
The following abnormal tests may indicate hypertension related damage
• Serum creatinine elevated (greater than 1.2 mg/dL [106 μmol/L])
• Microalbuminuria, which is diagnosed either from a 24-hour urine collection (20–200μg/min) or from elevated concentrations (30–300 mg/L) on at least two occassions. Use of the albumin-to-creatinine ratio (ACR) in a spot urine sample is becoming more common and microalbuminuria is defined by this measure as 30–300 mg/g creatinine
Common Comorbidities and Factors Contributing to CV Risk
Diabetes mellitus
Metabolic syndrome
Insulin resistance
Dyslipidemia
Microalbuminuria
Family history
Central obesity
Physical inactivity
Tobacco use
Target Organ Damage
Heart (left ventricular hypertrophy, angina, prior myocardial infarction, prior coronary revascularization, heart failure)
Brain (stroke or transient ischemic attack, dementia)
Chronic kidney disease
Peripheral arterial disease
Retinopathy
TREATMENT
Desired Outcomes
Hypertension management by nonpharmacologic and pharmacologic therapies has proven useful in reducing the risk of heart attack, heart failure, stroke, and kidney disease morbidity and mortality. For every 20 mm Hg systolic or 10 mm Hg diastolic increase in BP, there is a doubling of mortality for both ischemic heart disease and stroke.21 The goal of BP management is to reduce the risk of CVD and target organ damage. Targeting a specific BP is actually a surrogate goal that has been associated with reductions in CVD and target organ damage.
Patient Encounter 1
A 62-year-old Caucasian man comes to your clinic with results from a health fair he attended earlier this month. He was concerned as his BP at that time was 170/86 mm Hg, and when repeated 184/96 mm Hg. Upon examination, seated BP in the left arm is 144/82 mm Hg and 148/80 mm Hg in the right arm. Repeat measurements 5 minutes later are 142/84 mm Hg in the left arm and 144/76 mm Hg in the right. Much relieved, he declines any further evaluation or intervention, but promises to return in 1 week. BP at this 1 week follow-up is 146/84 as averaged from two readings in his right arm. The physical exam and past medical history are unremarkable, and all laboratory measurements are within normal limits with the exception of a high-density lipoprotein cholesterol of 45 mg/dL (1.17 mmol/L), triglyceride level of 155 mg/dL (1.75 mmol/L), total cholesterol of 180 mg/dL (4.66 mmol/L) and calculated LDL cholesterol of 104 mg/dL (2.69 mmol/L). Of note, he does not smoke.
Based on above information should this patient be classified as having hypertension?
What is this patient’s BP target using recommendations from JNC 7?
Does the BP target differ using recommendations from the 2007 American Heart Association Statement on the treatment of hypertension in the prevention and management of ischemic heart disease?
What factors may have contributed to the discrepancy between the health fair and office based BP readings?
General Approach to Treatment
![]() As is the case with dyslipidemia and other cardiovascular conditions, drug selection for the management of patients with hypertension should be considered as adjunctive to nonpharmacologic approaches for BP lowering. Previous clinical research has established the relative value of using individual antihypertensive drugs versus placebo to achieve reduction in morbidity and mortality by lowering BP. However, as newer antihypertensive agents are developed, it is difficult to justify the comparison of newer agents to placebo on ethical grounds. Consequently, contemporary large outcome-based, multicenter trials have been designed to compare one specific agent-based therapy (along with options to add others) versus another agent-based therapy (along with options to add others of a different class). These attempts at “head-to-head” comparisons and meta-analyses of multidrug regimen trials have, in general, provided evidence supporting the position that the main benefits of pharmacologic therapy are related to the achievement of BP lowering and are generally largely independent of the selection of an individual drug regimen.Inherent in this position is the realization that nonpharmacologic approaches alone are rarely successful in attaining target BPs, and multidrug therapy (sometimes as many as three or more agents) is necessary for most patients with hypertension.22 While JNC 7 focuses on utilizing BP levels in determining the threshold and target for treatment, the European Society of Cardiology also incorporates total cardiovascular risk in determining thresholds for treatment. This approach results in a “flexible” definition of hypertension which is dependent on an individual’s total cardiovascular risk.3 While acknowledging that there are several approaches currently employed to manage patients with hypertension, this chapter will use as its basis the JNC 7 guidelines while recognizing important changes outlined within the American Heart Association Guidelines.
As is the case with dyslipidemia and other cardiovascular conditions, drug selection for the management of patients with hypertension should be considered as adjunctive to nonpharmacologic approaches for BP lowering. Previous clinical research has established the relative value of using individual antihypertensive drugs versus placebo to achieve reduction in morbidity and mortality by lowering BP. However, as newer antihypertensive agents are developed, it is difficult to justify the comparison of newer agents to placebo on ethical grounds. Consequently, contemporary large outcome-based, multicenter trials have been designed to compare one specific agent-based therapy (along with options to add others) versus another agent-based therapy (along with options to add others of a different class). These attempts at “head-to-head” comparisons and meta-analyses of multidrug regimen trials have, in general, provided evidence supporting the position that the main benefits of pharmacologic therapy are related to the achievement of BP lowering and are generally largely independent of the selection of an individual drug regimen.Inherent in this position is the realization that nonpharmacologic approaches alone are rarely successful in attaining target BPs, and multidrug therapy (sometimes as many as three or more agents) is necessary for most patients with hypertension.22 While JNC 7 focuses on utilizing BP levels in determining the threshold and target for treatment, the European Society of Cardiology also incorporates total cardiovascular risk in determining thresholds for treatment. This approach results in a “flexible” definition of hypertension which is dependent on an individual’s total cardiovascular risk.3 While acknowledging that there are several approaches currently employed to manage patients with hypertension, this chapter will use as its basis the JNC 7 guidelines while recognizing important changes outlined within the American Heart Association Guidelines.
Nonpharmacologic Treatment: Lifestyle Modifications
Therapeutic lifestyle modifications consisting of nonpharmacologic approaches to BP reduction should be an active part of all treatment plans for patients with hypertension. The most widely studied interventions demonstrating effectiveness include:
• Weight reduction in overweight or obese individuals
• Adoption of a diet rich in potassium and calcium
• Dietary sodium restriction
• Physical activity
• Moderation of alcohol consumption
![]() Implementation of these lifestyle modifications successfully lowers BP (Table 5–3), often with results similar to those of therapy with a single antihypertensive agent.23 Combinations of two or more lifestyle modifications can have even greater effects with BP lowering. BP lowering in overweight patients may be seen by a weight loss of as few as 4.5 kg (10 lb). The Dietary Approaches to Stop Hypertension (DASH) trial demonstrated that a diet high in fruits, vegetables, and low-fat dairy products, along with a reduced intake of total and saturated fat, significantly reduced BP in as little as 8 weeks.23 Sodium restriction in moderate amounts lowers BP, is generally well-accepted, and is free of adverse effects. Restriction of sodium intake to 2.4 g (100 mmol) of elemental sodium (6 g of sodium chloride [100 mmol] or 1 teaspoon of table salt) should be easily achievable in most patients simply by avoidance of highly salted processed foods.24 Simple dietary advice and instructions on reading packaging labels should be introduced to the patient initially and assessed and reinforced at subsequent office visits. As is the case with weight loss, changes in physical activity do not need to be profound in order to have a significant effect on BP. It is generally accepted that 30 minutes of moderately intense aerobic activity (e.g., brisk walking) most days of the week will lower BP.25 While many patients can safely engage in moderately intense aerobic activity, individuals with known CVD, multiple risk factors with symptoms, or selected diabetic patients should undergo medical examination, possibly including exercise testing, prior to participation.26,27 The effects of alcohol on BP are variable. Initially, acute ingestion leads to a fall in BP followed by a rise several hours later,28 and binge drinking is associated with a higher risk of stroke. Furthermore, abstinence from alcohol in heavy drinkers leads to a reduction in BP.29 Alcohol also attenuates the effects of antihypertensive therapy, which is mostly reversible within 1 to 2 weeks with moderation of intake.
Implementation of these lifestyle modifications successfully lowers BP (Table 5–3), often with results similar to those of therapy with a single antihypertensive agent.23 Combinations of two or more lifestyle modifications can have even greater effects with BP lowering. BP lowering in overweight patients may be seen by a weight loss of as few as 4.5 kg (10 lb). The Dietary Approaches to Stop Hypertension (DASH) trial demonstrated that a diet high in fruits, vegetables, and low-fat dairy products, along with a reduced intake of total and saturated fat, significantly reduced BP in as little as 8 weeks.23 Sodium restriction in moderate amounts lowers BP, is generally well-accepted, and is free of adverse effects. Restriction of sodium intake to 2.4 g (100 mmol) of elemental sodium (6 g of sodium chloride [100 mmol] or 1 teaspoon of table salt) should be easily achievable in most patients simply by avoidance of highly salted processed foods.24 Simple dietary advice and instructions on reading packaging labels should be introduced to the patient initially and assessed and reinforced at subsequent office visits. As is the case with weight loss, changes in physical activity do not need to be profound in order to have a significant effect on BP. It is generally accepted that 30 minutes of moderately intense aerobic activity (e.g., brisk walking) most days of the week will lower BP.25 While many patients can safely engage in moderately intense aerobic activity, individuals with known CVD, multiple risk factors with symptoms, or selected diabetic patients should undergo medical examination, possibly including exercise testing, prior to participation.26,27 The effects of alcohol on BP are variable. Initially, acute ingestion leads to a fall in BP followed by a rise several hours later,28 and binge drinking is associated with a higher risk of stroke. Furthermore, abstinence from alcohol in heavy drinkers leads to a reduction in BP.29 Alcohol also attenuates the effects of antihypertensive therapy, which is mostly reversible within 1 to 2 weeks with moderation of intake.
Table 5–3 Lifestyle Modifications to Manage Hypertensiona

In addition to their beneficial effects on lowering BP, lifestyle modifications also have a favorable effect on other risk factors such as dyslipidemia and insulin resistance, which are commonly encountered in the hypertensive population, and lifestyle modifications should be encouraged for this reason as well. Smoking cessation should also be encouraged for overall cardiovascular health despite its lack of chronic effects on BP.30,31 Although lifestyle modifications have never been documented to reduce cardiovascular morbidity and mortality in patients with hypertension, they do effectively lower BP to some extent in most hypertensive patients. This may obviate the need for drug therapy in those with mild elevations in BP or minimize the doses or number of antihypertensive agents required in those with greater elevations in BP.
Pharmacologic Treatment
![]() An approach to selection of drugs for the treatment of patients with hypertension should be evidence-based with considerations regarding the individual’s coexisting disease states, coprescribed medications, and practical patient-specific issues including cost. The JNC 7 report and statements from other global organizations recommend drug therapy that is largely grounded in the best available evidence for superiority in outcomes—specifically morbidity and mortality.1 The approach is often tempered with practical considerations related to competing options for specific comorbidities and issues regarding a patient’s experience or tolerance for side effects, and in some cases, the cost of medications.
An approach to selection of drugs for the treatment of patients with hypertension should be evidence-based with considerations regarding the individual’s coexisting disease states, coprescribed medications, and practical patient-specific issues including cost. The JNC 7 report and statements from other global organizations recommend drug therapy that is largely grounded in the best available evidence for superiority in outcomes—specifically morbidity and mortality.1 The approach is often tempered with practical considerations related to competing options for specific comorbidities and issues regarding a patient’s experience or tolerance for side effects, and in some cases, the cost of medications.
Although landmark trials, such as the Antihypertensive and Lipid-Lowering Treatment to Prevent Heart Attack Trial (ALLHAT), have provided some objective basis for comparisons between initiating antihypertensive drug therapy with one class of antihypertensives versus another, there is room for criticism of these studies.22,32,33 Consequently, practical interpretations of their conclusions must always leave room for individualization based on clinical judgment. Overall current clinical guidelines provide a reasonable basis for guiding the selection of drug classes for individuals based on their stage of hypertension, comorbidities, and special circumstances. The following section will summarize key features of specific drug classes and guideline recommendations for patients with hypertension. Finally, an overview of the specific oral antihypertensive drug classes in common use is summarized in Table 5–4.
Diuretics
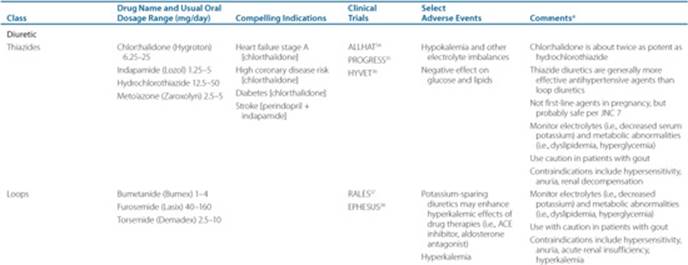
Many authorities recognize the value of diuretics as first-line agents for the majority of patients with hypertension. Among others, the basis for endorsement of diuretics as choice initial drug therapy for a variety of patient types includes their practical attributes (acquisition cost and availability as combination agents), extent of experience and favorable outcomes in placebo-controlled trials.1,2 These virtues are further supported by results of studies such as ALLHAT.22
Table 5–4 Commonly Used Oral Antihypertensive Drugs by Pharmacologic Class






This landmark double-blind study tested the hypothesis that newer antihypertensive agents would outperform thiazide-type diuretics when selected as initial drug therapy. After 4.9 years of follow-up in over 42,000 patients, the primary endpoint of fatal coronary heart disease and nonfatal MI was indistinguishable between chlorthalidone versus either amlodipine or lisinopril. A fourth arm examining doxazosin was terminated early based on a higher risk of heart failure for doxazosin compared with chlorthalidone.58 In spite of these findings for the primary endpoint, differences in outcomes for select secondary endpoints demonstrated superiority of chlorthalidone over either of the two remaining comparison groups. These observations, along with perceived cost-effectiveness (which was not specifically evaluated in this study), led the authors of JNC 7 to endorse diuretics as initial drug therapy for most patients with hypertension. Nonetheless, substantial criticism of this trial has undermined the enthusiasm for diuretics as first-line therapy in the minds of some clinicians’ as well as members of international guideline committees.3 Criticism of the differential BPs achieved in the various treatment groups, the artificial construct guiding the use of add-on drugs to base therapy, and the over-representation of African Americans exhibiting select endpoints have weakened the interpretability of the authors’ conclusions. Furthermore, other contemporary studies32,59 have also challenged the status of diuretics as ideal baseline choices for initial antihypertensive drug therapy for all patients. Specifically, the Australian-New Zealand Blood Pressure-2 (ANBP2) Study59 seemingly demonstrated (in particular for the male cohort) a superior outcome for angiotensin-converting enzyme (ACE) inhibitor-based therapy versus diuretic-based therapy in over 6,000 relatively older patients treated for over 4 years. Similarly, the Anglo-Scandinavian Cardiac Outcomes Trial-Blood Pressure Lowering Arm (ASCOT-BPLA)32 study demonstrated outcomes which seemingly favored the calcium channel blocker agent (CCBA)/ACE inhibitor-based approach versus a β-blocker/diuretic-based approach in over 19,000 patients treated for approximately 5 years. Needless to say, all three of these major trials are subject to significant criticism including heterogeneity of achievement in BP targets between treatment arms for both the ALLHAT and ASCOT-BPLA studies.60 In addition, the recently completed Avoiding Cardiovascular Events in Combination Therapy in Patients Living with Systolic Hypertension (ACCOMPLISH) trial failed to demonstrate superiority of a low-dose diuretic-ACE inhibitor combination versus a calcium channel blocker-ACE inhibitor combination in hypertensive patients. This has refueled the debate over whether the means by which BP is lowered (drug selection) is more or less important than the extent and or time taken to lower BP.61 Furthermore, controversy has ignited as to whether the choice and dose of diuretic (chlorthalidone, ALLHAT) versus hydrochlorothiazide (ANBP2, ACCOMPLISH) was partly responsible for the difference in study results, and raised the question whether chlorthalidone should be the preferred thiazide type diuretic.62,63Nonetheless, diuretics remain supported by most as baseline initial therapy for the majority of hypertensive patients without compelling indications.60
Key features of diuretics that must be kept in mind, along with evidence from outcome-based studies, include the diversity between the subtypes of diuretics and their corresponding diversity of pharmacologic actions. The four subtypes include thiazides, loop diuretics, potassium-sparing agents, and aldosterone antagonists. The latter will be discussed as a separate entity. Each subtype has clinically based properties which distinguish their roles in select patient populations. Thiazide diuretics are by far the most commonly prescribed subtype with the greatest number of outcome-based studies supporting their use. In the United States, hydrochlorothiazide and chlorthalidone represent the most commonly prescribed thiazide-type diuretics and have been the subject of the majority of large outcome-based studies. Although subtle differences in pharmacokinetics between these agents exist, practical differences are limited to their relative diuretic potency. Chlorthalidone is considered approximately 1.5 to 2 times more potent than hydrochlorothiazide for BP reduction.64 Since the relationship between antihypertensive efficacy and metabolic/electrolyte-related side effects of thiazide diuretics is considered to be dose-related, attention to this differential in potency may be important. Specifically, select metabolic effects (hyperlipidemic and hyperglycemic) and electrolyte-related effects (hypokalemic, hypomagnesemic, hyperuricemic, and hypercalcemic) seem to increase with higher doses. This has led to national guidelines1 recommending doses not exceed 6.25 to 25 mg/day for chlorthalidone or 12.5 to 50 mg/day for hydrochlorothiazide. These metabolic effects may clearly complicate the management of higher-risk patients with common comorbidities such as dyslipidemia or diabetes, or even those likely to be sensitive to the potassium-or magnesium-wasting effects of diuretics (patients with dysrhythmias or those taking digoxin). While rates of diabetes are higher following administration of thiazides, there is evidence that this can be greatly minimized by keeping potassium in the high normal range (i.e., above 4.0 mEq/L [4 mmol/L]).65 Furthermore, whether the development of new-onset diabetes in association with thiazide diuretic is of clinical significance is in question as the large ALLHAT and the Systolic Hypertension in the Elderly Program [SHEP]) trials showed no significant adverse CV events from new diuretic-associated diabetes whereas a smaller trial did.66 Nonetheless, clinicians should rarely approach the upper limits of these dosage ranges without careful assessment of their metabolic effects or potential to induce electrolyte disturbances. In this way, optimization of BP lowering potential may be achieved while minimizing potential adverse outcomes.
Another key feature of the thiazide-type diuretics is their limited efficacy in patients whose renal function is reduced simply by age. Estimated by a calculated GFR, as renal function declines with age to less than 30 mL/min, loop type diuretics such as furosemide emerge as superior to thiazide type diuretics for lowering BP. Clinicians often fail to reevaluate the use of thiazide diuretics prescribed to individuals whose renal function has been declining with age.
The loop diuretics, such as furosemide, bumetanide, torsemide, and ethacrynic acid, have a common site of action in the thick ascending limb of the loop of Henle.67 Responsible for reabsorption of over 65% of the filtered sodium, their diuretic efficacy is clearly superior to that of the thiazides, potassium-sparing diuretics, and mineralocorticoids. Practically speaking, furosemide is the most common agent used as an alternative to thiazide type agents for patients whose renal function has been compromised. With the exception of torsemide, which has a longer half-life, the loop diuretics should be administered twice daily versus once when utilized primarily for their antihypertensive (versus diuretic) effect. The most significant adverse effects related to loop diuretic use concerns their potential for excessive diuresis leading to hyponatremia or hypotension. Additionally, hypokalemia, hypomagnesemia, and hypocalcemia may develop over time and contribute to the potential for cardiac arrhythmias. Overall relevance of drug-drug interactions and potential for aggravating select conditions (hyperglycemia, dyslipidemias, and hyperuricemia) should be routinely considered in the monitoring plan for those taking loop diuretics for extended periods of time.
Potassium-sparing diuretics that do not act through mineralocorticoid receptors include triamterene and amiloride. These agents are often prescribed with potassium-wasting diuretics in an attempt to mitigate the loss of potassium. When administered as a single entity or as one component of a combination product, these agents result in moderate diuresis. Potassium-sparing diuretics act on the late distal tubule and collecting duct, and thereby have limited ability to affect sodium reabsorption, which translates into modest diuresis. The most important adverse effects associated with these agents are their potential to contribute to hyperkalemia. This is especially relevant in the context of those patients receiving other agents with potassium-sparing properties, such as ACE inhibitors, angiotensin receptor blockers (ARBs), and potassium supplements, as well nonsteroidal anti-inflammatory drugs (NSAIDs). It is also relevant in those with more than mild renal impairment.
Aldosterone Antagonists
Aldosterone antagonists such as spironolactone, and eplerenone (Fig. 5–3) modulate vascular tone through a variety of mechanisms besides diuresis. Their potassium-sparing effects mediated through aldosterone antagonism, complement the potassium-wasting effects of more potent diuretics such as thiazide or loop diuretics. Patients with resistant hypertension (with or without primary aldosteronism) experience significant BP reductions with the addition of low-dose spironolactone (12.5–50 mg/day) to diuretics, ACE inhibitors, and ARBs.68 Although functional in this circumstance, it is important to recognize their potential to enhance the risk for hyperkalemia when used in conjunction with ACE inhibitors, ARBs, and now potentially DRIs. This is particularly relevant for individuals with comorbidities associated with reduced renal function or those receiving either potassium supplements or NSAIDs. The most commonly used potassium-sparing diuretic is spironolactone; however, eplerenone has been used with increasing frequency in patients with heart failure following acute myocardial infarction (AMI).38 Although spironolactone is commonly associated with gynecomastia, eplerenone rarely causes this complication.69 The risk of hyperkalemia is also more commonly reported with patients on spironolactone.70
β-Blockers
Controversy surrounds the JNC 7 recommended role of βblockers as first-line antihypertensive agents. For example, a meta-analysis by Lindholm et al.71 demonstrated a higher risk of stroke in patients treated for primary hypertension with β-blockers compared to other antihypertensives. Moreover, Messerli et al.72 showed that β-blockers were ineffective in preventing coronary heart disease, cardiovascular mortality, and all-cause mortality compared to diuretics for elderly patients (60 years of age or older) treated for primary hypertension. Consequently, current evidence suggests patients with uncomplicated hypertension may not benefit as much, if at all, from β-blocker therapy relative to other antihypertensives.2 This may be in part due to their tendency to reduce central aortic pressure and cardiac afterload to a lesser degree relative to other agents.73 It must be noted that all of these analyses were conducted with a limited number of β-blockers such as atenolol and metoprolol tartrate. Whether newer formulations such as metoprolol succinate, or agents with unique properties such as carvedilol would be more efficacious in reducing morbidity and mortality is unknown. The drug with the most prominent difference in the increased risk of stroke between the three β-blocker use subgroups was atenolol. The concern regarding β-blocker use in hypertensive patients without compelling indications is reflected in American Heart Association’s recommendations and the European Society of Cardiology’s abandonment of β-blockers as first-line antihypertensive agents. On the other hand, the role of β-blockers in patients with specific select comorbidities is well established (Table 5–5). Specific outcome-based studies conducted in patients with comorbidities such as heart failure and recent MI have clearly demonstrated a benefit from β-blocker use.74 Their hemodynamic effects and antiarrhythmic properties make them desirable agents for hypertensive patients who suffer from ischemic conditions including AMI.67 When used judiciously in hypertensive patients with heart failure, β-blocker inhibition of neurohormonal mediated cardiac remodeling reduces morbidity and mortality relative to standard heart failure therapies. The mechanisms through which β-blockers affect BP are complex, but most certainly include their modulation of renin (Fig. 5–3) which appears to result in a reduction in CO and/or reduction in PR along with their negative inotropic/chronotropic actions.
The specific pharmacologic properties of β-blockers are varied and diverse. An understanding of these properties may assist in the selection of one agent over others given a patient’s specific condition(s). One of these properties is cardioselectivity—the property of some β-blockers that preferentially block β1-versus β2-receptors. Another property exhibited by some β-blockers is membrane stabilization activity, which relates to the propensity of the β-blocker to possess some capacity for antiarrhythmic properties, in addition to β-receptor blocking properties. Some β-blockers (Fig. 5–4) possess properties referred to as intrinsic sympathomimetic activity (ISA). β-Blockers possessing this property effectively block the β-receptor at higher circulating catecholamine levels, such as during exercise, while having modest β-blocking activity at times of lower catecholamine levels, such as at rest.75
Table 5–5 Compelling Indications for Individual Drug Classes
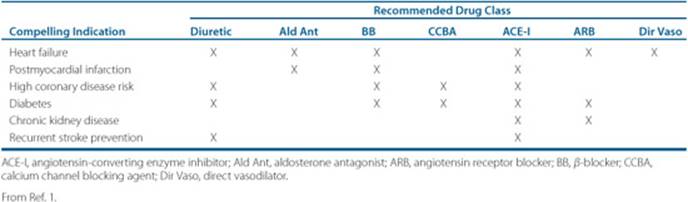
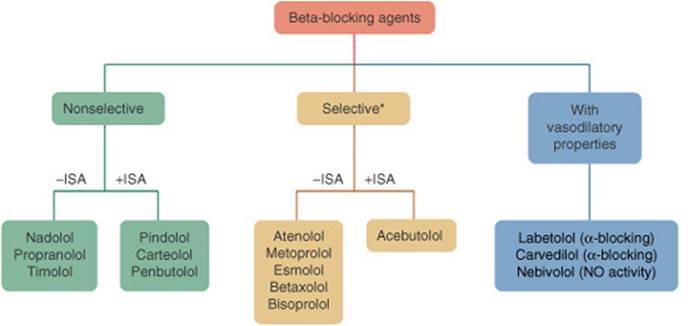
FIGURE 5–4. Flowchart listing β-blocking agenvarious β-receptor activity and intrinsic sympathomimetic activty.*β-1 Cardioselective. (ISA, intrinsic sympathomimetic activity; NO, nitrous oxide.)
When selecting a β-blockers, some of these properties may be of practical value while others, such as membrane stabilization activity and ISA, are of theoretical interest only. Because neither membrane stablization activity nor ISA has been directly proven value in the clinical setting, they will not be discussed further other than to point out that β-blockers with ISA are not recommended for use in the post-MI patient.76 With regards to cardioselectivity, consider a patient with mild asthma, chronic obstructive pulmonary disease, or peripheral vascular disease (intermittent claudication). A β-blocker with relative cardioselectivity to block β1-receptors may be more desirable in such a patient, while a nonselective β-blocker (Fig. 5–4) may be potentially disadvantageous. In such a patient, low doses of cardioselective β-blockers may achieve adequate blockade of β1-receptor in the heart and kidneys while minimizing the undesirable effects of β2-blockade on the smooth muscle lining the bronchioles. In doing so, hypertension may be managed while avoiding complications of the coexisting reactive airway disease, which is mediated by β-receptor stimulation. Similarly, either because of a reduction in the β-mediated vascular blood flow or by enhanced unopposed α-agonist–mediated vasoconstriction, a patient with peripheral vascular disease (intermittent claudication) may experience a worsening of symptoms with use of a nonselective β-blocker (Fig. 5–4). It is important to remember that cardioselectivity is dependent upon dose, with diminished selectivity exhibited with higher doses.
A limited number of β-blockers also possess vasodilatory properties that are either mediated through α1-receptor blockade (carvedilol, labetalol) or via L-arginine/nitric oxide-induced release from endothelial cells with subsequent increased nitric oxide bioavailability in the endothelium (nebivolol; Fig. 5–4). Reductions in PR through α1-receptor mediated blockade or via L-arginine/nitric oxide-induced release, in addition to β-blockade, may benefit patients with hypertension. Such combinations should theoretically contribute to enhanced reductions in vascular tone. Nonetheless, there has been no proven evidence of superior outcomes from the use of β-blockers with vasodilatory properties through either means compared to those with only β-blocking activity.
The adverse effects of β-blockers logically follow their pharmacology. Initiating β-blockers to treat patients with hypertension may have the potential to precipitate bradycardia, various degrees of heart block, or signs and symptoms of heart failure. The latter is usually limited to those with a subclinical diagnosis and should be considered in the elderly or those with documented reductions in left ventricular function. Conversely, abrupt discontinuation of β-blockers has been cited as a precipitating factor in the development of ischemic syndromes—especially for those patients in whom β-blockers were used for extended periods of time, at higher doses, or who had underlying ischemic heart disease. In such cases, the dose of these agents should be reduced (tapered) over a period of several days to perhaps 1 or even 2 weeks depending on patient-related factors.
Calcium Channel Blocking Agents
Exhibiting considerable interclass diversity, CCBAs are recognized as effective antihypertensives, particularly in the elderly. Patients with isolated systolic hypertension who were taking CCBAs experienced reduced CVD endpoints during several clinical trials. Several trials comparing a CCBA-based approach versus non-CCBA-based approach have been published. Specifically, the Valsartan Antihypertensive Long-term Use Evaluation (VALUE) trial compared valsartan-based therapy to amlodipine-based therapy in over 15,000 patients who were at high-risk for cardiac events. In spite of an attempt to achieve identical BP reductions, differences were noted early and sustained throughout the 4.2-year length of the study.32 Overall the primary endpoint (composite cardiac mortality and morbidity) was not statistically significantly different between the groups, but cause-specific outcomes did favor the regimen affording the achievement of lower BPs—namely the amlodipine-based therapy. This theme of unequal reductions in BP accounting for differences in cause-specific outcomes was shared by the findings of the ASCOT-BPLA study, which compared amlodipine-based therapy versus atenolol-based therapy in over 19,000 hypertensives for 5.5 years. Again although no statistically significant reduction in the primary endpoint was observed, the study was stopped prematurely. The basis for this early termination was grounded in the association of a higher mortality rate and worse outcome for those allocated to the atenolol-based regimen compared to those receiving the amlodipine-based regimen. These observations support the argument that regardless of the agents used, the evidence appears to indicate that the amount of BP lowering achieved has more to do with event reduction than with the agents or combinations of agents used to achieve them. Primary endpoints aside, certain secondary endpoints demonstrated differences between regimens. Protection from the development of new-onset diabetes over the duration of the study was noted for the amlodipine-based therapy in the ASCOT-BPLA study and favored the ARB-based regimen over the CCBA-based regimen in the VALUE study.
The diversity of pharmacologic properties among the subclasses of CCBAs is significant. A clinician’s familiarity with these differences among subclasses helps categorize their expected effects on the cardiovascular system and potential risk of toxicities. Dihydropyridine CCBAs such as amlodipine are commonly associated with edema, especially when used at higher doses. Phenylalkylamine-verapamil and benzothiazepine-diltiazem are more commonly recognized for their electrophysiological effects, negative chronotropic and negative inotropic effects. Many of these pharmacologic properties are exploited for their specific clinical utility. Given that verapamil and diltiazem (both are nondihydropyridine CCBAs) effectively block cardiac conduction through the atrioventricular node, their value in the management of patients with atrial fibrillation in addition to hypertension is obvious. In contrast, the dihydropyridine subclass of agents has no utility in managing atrial dysrhythmias. Similarly, all three subclasses of CCBAs possess some coronary vasodilating properties and hence may be used in select patients for the management of patients with angina, in addition to their antihypertensive benefits.
ACE Inhibitors
ACE inhibitors are widely used for the treatment of hypertension in a variety of patients with or without comorbidities and/or cardiovascular risk factors. Supported by the findings of numerous outcome trials, they have been extensively studied. This broad utility extends to the list of compelling indications (Table 5–5) for patients as described in JNC 7. These compelling indications include their qualified role in managing patients with hypertension who have type 1 diabetes,33 heart failure,77 post-MI,76 type 2 diabetes,78 CKD,79,80 or recurrent stroke prevention.35 Comparative trials between ACE inhibitors and various other agents as initial drug therapy have also demonstrated some differences in outcomes for this class. In ALLHAT,22 ACE inhibitors appeared to perform less well than diuretics in terms of incidence of combined CVD and heart failure. On the other hand, the ANBP2 trial59 seemed to suggest that ACE inhibitors may be equivalent to diuretics in terms of overall outcomes. Because there are legitimate criticisms of both these trials (including the smaller number of overall events and choice of hydrochlorothiazide as the diuretic in ANBP2), it may only be safe to conclude that both diuretics and ACE inhibitors represent formidable agents as either first-or second-line hypertensive therapies that effectively achieve a target BP goal for most patients with or without comorbidities.
Although generally well-tolerated, classic side effects associated with ACE inhibitors include their potential to cause hyperkalemia and a persistent dry cough. Modest elevations in serum potassium should be anticipated, particularly in patients with compromised renal function, those receiving concurrent NSAIDs, or those taking potassium supplementation or using a potassium containing salt substitute. The elevations in potassium should be anticipated, if not prospectively considered, when starting or increasing the dose of an ACE inhibitor. Hyperkalemia is rarely a reason for discontinuation of therapy. Nonetheless, periodic monitoring of serum potassium is prudent for patients receiving ACE inhibitors. The dry cough associated with ACE inhibitors is thought to be caused by accumulation of bradykinin resulting from a direct effect of inhibiting ACE. Although mild forms are tolerable, should cough jeopardize compliance with the agent, ARBs should be considered as possible alternative agents since there is less incidence of cough.
In general, the effects of ACE inhibitors on diminished renal function and potassium can be predicted given an understanding of their pharmacologic actions (Fig. 5–3). Inhibition of the generation of angiotensin II through ACE inhibition (or direct blockage of the angiotensin II receptor by angiotensin II receptor blockers) naturally would reduce the efferent renal artery tone thereby changing the intraglomerular pressure. Although changes in the afferent renal artery tone also occur, the overall effects usually translate into a reduction in GFR80 with resulting elevations of up to 30% in serum creatinine values. It is important to recognize that such elevations in serum creatinine are not usually indications to discontinue use of the ACE inhibitor. Rather, possible dose reduction and continued monitoring for further increases in serum creatinine remains prudent. Alternatively, should elevations in serum creatinine exceed 30%, discontinuation is prudent until further evaluation can be made.
More rare forms of adverse effects of ACE inhibitors include blood dyscrasias, angioedema, and more serious effects of ACE inhibitors on renal function. The latter include acute renal failure in those with pre-existing kidney dysfunction, bilateral renal artery stenosis, or unilateral renal artery stenosis in a patient with one functioning kidney.
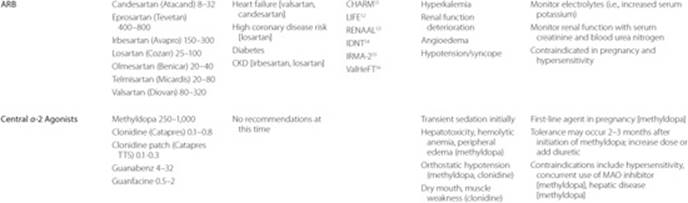
Angiotensin Receptor Blockers
ARBs are another key class of agents whose role in managing patients with hypertension has been further defined by recently completed studies. ARBs are inhibitors of the angiotensin-1 (AT1) receptors (Fig. 5–3). AT1 receptor stimulation evokes a pressor response via a host of accompanying effects on catecholamines, aldosterone, and thirst.67 Consequently, inhibition of AT1 receptors directly prevents this pressor response and results in up-regulation of the RAAS. Up-regulation of the RAAS results in elevated levels of angiotensin II, which have the added effect of stimulating the angiotensin-2 (AT2) receptors. AT2-receptor stimulation is generally associated with antihypertensive activity; however, long-term effects of AT2-receptor stimulation that involve cellular growth and repair are relatively unknown. What is clear is that ARBs differ from ACE inhibitors in that the former causes up-regulation of the RAAS while the latter blocks the breakdown of bradykinin. The therapeutic relevance resulting from these pharmacologic differences has yet to be fully evaluated through long-term clinical comparative trials in hypertensive patients. However, data from other patient populations (heart failure, high-risk coronary artery disease patients) suggest that the clinical benefits of ARBs are less robust than those of ACE inhibitors.81,82
At this point, ARBs have emerged as an effective class of antihypertensives whose low incidence of side effects and demonstrated clinical role in patients with specific comorbidities have afforded them an attractive position in the antihypertensive armamentarium. Like ACE inhibitors, the antihypertensive effectiveness of ARBs is greatly enhanced by combining them with diuretics. Furthermore, they have proven their value as well-tolerated alternatives to ACE inhibitors for patients with CKD, diabetes mellitus, and post-AMI (Table 5–5). As of late, the addition of ARBs to standard therapy for patients with heart failure (HF), including ACE inhibitors, have demonstrated additional incremental benefits for patients with systolic dysfunction83 or diastolic dysfunction84 or as alternatives to ACE inhibitors when ACE inhibitors are not tolerated.85 Comparative studies with alternate (non-ACE inhibitors) antihypertensive regimens in patients with type 2 diabetes84 and left ventricular hypertrophy52 have demonstrated their usefulness as effective antihypertensives in these special populations. Studies (the Irbesartan Diabetic Nephropathy Trial [IDNT] and Reduction of Endpoints in NIDDM with the Angiotensin II Antagonist Losartan [RENAAL; NIDDM refers to noninsulin-dependent diabetes mellitus]) have demonstrated superiority of delaying progression toward renal dysfunction for ARBs relative to alternative antihypertensives in type 2 diabetics.84 Although better tolerated than ACE inhibitors, ARBs have not been shown to demonstrate superiority of outcomes relative to ACE inhibitors. This key observation, in addition to their relatively higher acquisition cost, has mitigated the growth of ARB use relative to ACE inhibitors.
Renin Inhibitors
Aliskiren is the first agent in the newest class of antihypertensive agents. While similar to ACE inhibitors and ARBs in that it acts within the RAAS, it is unique in that it directly blocks renin thereby reducing plasma renin activity (PRA), and subsequently angiotensin I and angiotensin II, with a resultant reduction in BP. This disruption of the negative feedback loop results in a compensatory increase in renin levels—the significance of which is not yet known. Aliskiren has been shown to be well tolerated and effective in reducing BP when used as monotherapy and in combination with hydrochlorothiazide or valsartan for patients with mild to moderate HTN. However, long-term clinical trials evaluating efficacy and safety have yet to be completed, and thus the effects of aliskiren on morbidity and mortality are as of yet unknown. Because of aliskiren’s role in the RAAS, recommendations and precautions for monitoring serum potassium and kidney function should be similar to those of ACE inhibitors and ARBs.
α-Blockers
Generally, α1-blockers are considered as inferior agents and should not be used as monotherapy. The ALLHAT trial had an α1-blocker arm that was discontinued early as terazosin was associated with an increase in cardiovascular events. α1Blockers may be considered as add-on therapy to other agents (i.e., 3rd or 4th line) when hypertension is not adequately controlled. In addition, they may have a specific role in the antihypertensive regimen for elderly males with prostatism; however, their use is often curtailed by complaints of syncope, dizziness, or palpitations following the first dose and orthostatic hypotension with chronic use. The roles of doxazosin, terazosin, and prazosin in the management of patients with hypertension are limited due to the paucity of outcome data and the absence of a unique role for special populations or compelling indications from JNC 7.
Patient Encounter 2
A 55-year-old African American woman comes to your clinic with a recent diagnosis of hypertension. She is 5’5” (165 cm) tall and weighs 73 kg (160 lb; body mass index 26.6 kg/m2). She reports that she does not use tobacco or drink alcohol, and exercises about once a week. Physical exam was unremarkable, but an ECG revealed left ventricular hypertrophy. Baseline laboratory tests were significant for fasting blood glucose of 124 mg/dL (6.88 mmol/L), serum creatinine of 1.5 mg/dL (133μmol/L), potassium of 4.8 mEq/L (4.8 mmol/L), total cholesterol of 200 mg/dL (5.18 mmol/L), high-density lipoprotein cholesterol of 40 mg/dL (1.04 mmol/L), triglycerides of 200 mg/dL (2.26 mmol/L), and low-density lipoprotein cholesterol of 120 mg/dL (3.11 mmol/L). She has a history of an elevated serum creatinine ranging from 1.3 to 1.5 mg/dL (115–133μmol/L) over the past 6 months. Urinalysis was positive for microalbuminuria. BP today was 165/86 mm Hg with a pulse of 55 bpm.
What signs of target-organ damage does this patient exhibit?
Is more extensive testing for identifiable causes of hypertension indicated at this time?
Based on the information presented, create a care plan for this patient’s hypertension. This should include (a) goals of therapy, (b) a patient-specific therapeutic plan, and (c) a plan for appropriate monitoring to achieve goals and avoid adverse effects.
Central α2-Agonists
Limited by their tendency to cause orthostasis, sedation, dry mouth, and vision disturbances, clonidine, methyldopa, guanfacine, and guanabenz represent rare choices in contemporary treatment of patients with hypertension. Their central α2-adrenergic stimulation is thought to reduce sympathetic outflow and enhance parasympathetic activity thereby reducing heart rate, CO, and total PR. Occasionally used for cases of resistant hypertension, these agents may have a role when other more conventional therapies appear ineffective. The availability of a transdermal clonidine patch that is applied once weekly may offer an alternative to hypertensive patients with adherence problems. Of particular importance is the issue of severe rebound hypertension when clonidine is abruptly discontinued. The dose of this agent should be tapered when discontinuation is considered. If the patient is also receiving concomitant β-blocker therapy, the β-blocker should be tapered to discontinuation ideally several days before discontinuation of clonidine is initiated.
Other Agents
Direct vasodilators such as hydralazine and minoxidil represent additional alternative agents used for patients with resistant hypertension. Primarily acting to relax smooth muscles in arterioles and activate baroreceptors, their use in the absence of concurrently administered β-blockers and diuretics is uncommon. This is due to the need to offset their tendency to cause reflex tachycardia and fluid retention. Other, more rare adverse effects include hydralazine-induced lupus-like syndrome and hypertrichosis from minoxidil. Finally, reserpine, although slow to act, represents another rarely used alternative agent for those who are recalcitrant to more standard therapy. This agent is a long-acting depleter of the catecholamine norepinephrine from sympathetic nerve endings and via blockade of norepinephrine transport into its storage granules which causes reduced sympathetic tone leading to reductions in PR. Reserpine’s association with numerous side effects including gastric ulceration, depression, and sexual side effects has limited its perception as a useful agent, but in low doses it can be well tolerated. In fact, the SHEP trial86demonstrated the BP lowering effectiveness of low-dose reserpine (0.05 mg/day) when combined with a diuretic, with similar cardiopulmonary and psychosocial side effects between the treatment and placebo groups. In addition, low-dose reserpine was also used as add-on therapy in the ALLHAT trial. Two additional agents, guanethidine and guanadrel, act as postganglionic sympathetic inhibitors inhibiting the release of norepinephrine as well as depleting norepinephrine from these nerve terminals. However, these agents have little role in the management of hypertension because of significant adverse effects.
SPECIAL PATIENT POPULATIONS
Compelling Indications and Special Considerations
![]() Specific antihypertensive therapy is warranted for certain patients with comorbid conditions that may elevate their level of risk for CVD. Clinical conditions for which there is compelling evidence supporting one or more classes of drug therapy include:1
Specific antihypertensive therapy is warranted for certain patients with comorbid conditions that may elevate their level of risk for CVD. Clinical conditions for which there is compelling evidence supporting one or more classes of drug therapy include:1
• Ischemic heart disease
• Heart failure
• Diabetes
• CKD
• Cerebrovascular disease
Compelling indications for specific drug therapies are summarized in Table 5–5.1,87 In patients with hypertension and angina, β-blockers and long-acting CCBAs are indicated due to their antihypertensive and antianginal effects.1,26 In addition, patients may benefit from the potential atherosclerotic plaque stabilizing effects of amlodipine.2 In patients at high-risk of ischemic heart disease, such as diabetic patients with additional cardiovascular risk factors or chronic coronary artery or vascular disease, ACE inhibitors are particularly useful in reducing the risk of cardiovascular events regardless of whether the patient carries a concurrent diagnosis of hypertension.1,50,88 In patients intolerant to an ACE inhibitor, an ARB may be substituted given the findings of the recently completed ONgoing Telmisartan Alone and in combination with Ramipril Global Endpoint Trial (ONTARGET).89
β-Blockers and ACE inhibitors are indicated for post-MI patients due to their proven reduction of cardiovascular morbidity and mortality in this population. Aldosterone antagonists are also indicated for the post-MI patient with reduced left ventricular systolic function and diabetes or signs and symptoms of heart failure.1,76
Patients with asymptomatic left ventricular systolic dysfunction and hypertension should be treated with β-blockers and ACE inhibitors. Those with heart failure secondary to left ventricular dysfunction and hypertension should be treated with drugs proven to also reduce the morbidity and mortality of heart failure, including β-blockers, ACE inhibitors, ARBs, aldosterone antagonists, and diuretics for symptom control as well as antihypertensive effect. In African Americans with heart failure and left ventricular systolic dysfunction, combination therapy with nitrates and hydralazine not only affords a morbidity and mortality benefit, but may also be useful as antihypertensive therapy if needed.57 The dihydropyridine CCBAs amlodipine or felodipine may also be used in patients with heart failure and left ventricular systolic dysfunction for uncontrolled BP, although they have no effect on heart failure morbidity and mortality in these patients.77 For patients with heart failure and preserved ejection fraction, antihypertensive therapies that should be considered include diuretics, β-blockers, ACE inhibitors, ARBs, CCBAs (including nondihydropyridine agents), and others as needed to control BP.1,77
Patients with diabetes and hypertension should initially be treated with either ACE inhibitors, ARBs, β-blockers, diuretics, or CCBAs. There is a general consensus that therapy focused on RAAS inhibition by ACE inhibitors or ARBs may be optimal if the patient has additional cardiovascular risk factors such as left ventricular hypertrophy or CKD.1,78,79,52
In patients with CKD and hypertension, ACE inhibitors and ARBs are preferred, usually in combination with a diuretic.79 ACE inhibitors in combination with a thiazide diuretic are also preferred in patients with a history of prior stroke or transient ischemic attack. This therapy reduces the risk of recurrent stroke, making it particularly attractive in these patients for BP control.83
There are several situations in the management of hypertension requiring special considerations including, but not limited to:
• Hypertensive crisis
• Elderly populations
• Isolated systolic hypertension
• Minority populations
• Pregnancy
• Pediatrics
Hypertensive crisis can be divided into hypertensive emergencies and hypertensive urgencies. A hypertensive emergency occurs when severe elevations in BP are accompanied by acute or life-threatening target organ damage such as AMI, unstable angina, encephalopathy, intracerebral hemorrhage, acute left ventricular failure with pulmonary edema, dissecting aortic aneurysm, rapidly progressive renal failure, accelerated malignant hypertension with papilledema, and eclampsia, among others. BP is generally greater than 220/140 mm Hg, although a hypertensive emergency can occur at lower levels, particularly in individ-uals without previous hypertension. The goal in a hypertensive emergency is to reduce mean arterial pressure by up to 25% to the range of 160/100 to 110 mm Hg in minutes to hours.1,90 IV therapy is generally required and may consist of the agents listed in Table 5–6.87 A hypertensive urgency is manifested as a severe elevation in BP without evidence of acute or life-threatening target organ damage. In these individuals, BP can usually be managed with orally administered short-acting medications (i.e., captopril, clonidine, or labetalol) and observation in the emergency department over several hours, with subsequent discharge on oral medications and follow-up in the outpatient setting within 24 hours.1,87
The treatment of elderly patients with hypertension, as well as those with isolated systolic hypertension, should follow the same approach as with other populations with the exception that lower starting doses may be warranted to avoid symptoms. Special attention should be paid to postural hypotension. This should include a careful assessment of orthostatic symptoms, measurement of BP in the upright position, and caution to avoid volume depletion and rapid titration of antihypertensive therapy.1 In individuals with isolated systolic hypertension, the optimal level of diastolic pressure is not known, and although treated patients who achieve diastolic pressures less than 60 to 70 mm Hg had poorer outcomes in a landmark trial, their cardiovascular event rate was still lower than those receiving placebo.90 In addition, a recently completed trial (HYVET) documented the benefits of antihypertensive therapy in patients over the age of 80 as they experienced a significant reduction in all-cause mortality, fatal stroke and heart failure when treated with a diuretic (indapamide) with or without an ACE inhibitor (Perindopril).36
Table 5–6 Parenteral Antihypertensive Agents for Hypertensive Emergencya


Patient Encounter 3
A 78-year-old Caucasian man presents to the emergency department with complaints of a headache persisting over the last 3 days. Repeated BP measurements average 200/110 mm Hg. He reports no other symptoms; physical examination and laboratory tests are unremarkable as is his past medical history with exception of hypertension diagnosed in his early 60s. He reports he is struggling on a fixed retirement income with no prescription coverage and takes “what I can afford.” BP medications are metoprolol succinate (Toprol XL) 200mg once daily, amlodipine 10mg (Norvasc) once daily, torsemide (Demadex) 10 mg once daily, and valsartan (Diovan) 320 mg once daily.
What type of hypertensive crisis is this patient experiencing?
What are likely causes of this patient’s loss of BP control?
Create a care plan for this patient’s hypertensive crisis. This should include (a) acute goals of therapy, (b) a patient-specific therapeutic plan to achieve this, and (c) a plan for appropriate outpatient follow-up.
While the treatment approach of hypertension in minority populations is similar, special consideration should be paid to socioeconomic and lifestyle factors that may be important barriers to BP control. In addition, in patients of African origin, diminished BP responses have been seen with ACE inhibitors and ARBs compared to diuretics or calcium channel blockers.1 Hypertension in pregnancy is a major cause of maternal, fetal, and neonatal morbidity and mortality. There are many categories of hypertension in pregnancy; however, pre-existing hypertension and pre-eclampsia are treated differently. The therapeutic selection of an oral antihypertensive agent (Table 5–7) in a pregnant patient with chronic hypertension is determined with regard to fetal safety. Therapeutic options for acute severe hypertension in pre-eclampsia may be reviewed in JNC 7.1
Similar to the JNC 7 criteria (which has four stages for BP classification in adults), the measurement of three or more BPs in children and adolescents are compared to tables listing the 90th-, 95th-, and 99th-percentile BPs based on age, height, and gender that classify BP as normal, prehypertension, and stage 1 and stage 2 hypertension (Table 5–1).91 The prevalence of hypertension in adolescent populations is increasing and is associated with obesity, sedentary lifestyle, or a positive family history, which increases the risk of CVD. The clinician should be aware that secondary causes are common in adolescent hypertensives, and the identification and aggressive modification of risk factors with nonpharma-cologic and pharmacologic interventions is paramount for risk reduction of target organ damage. The 2004 National High Blood Pressure Education Program (NHBPEP) Working Group Report on Hypertension in Children and Adolescents provides specific recommendations to modify and treat risk factors in this population of patients. In addition, the American Heart Association has recently released a source guiding the role of ambulatory BP measurements in children and adolescents.91,92 The Nelson Textbook of Pediatrics is also recommended for a comprehensive review of treatment of congenital and pediatric hypertension.93
Table 5–7 Treatment of Chronic Hypertension in Pregnancy

PATIENT CARE AND MONITORING
![]() The frequency of follow-up visits for patients with hypertension will vary based on individual cases, but will be influenced by severity of hypertension, comorbidities, and choice of agent selected. A recent analysis of the VALUE trial found that achieving BP control within 6 months reduced CV events compared to a longer time to achieve goal. In addition, another recent analysis of the INSIGHT trial found that the more times a patient is below goal, the lower the risk.94,95 These studies suggest that there is an urgency to achieve and maintain BP control, and this may necessitate frequent assessment and adjustments (i.e., 2 weeks). At a minimum, assessment of response to medications should be done at 1-month intervals.1 In patients with stage 2 hypertension or those with comorbidities (e.g., diabetes, vascular disease, CHF, or CKD), shorter time frames of 2 weeks or less are more appropriate.79 Once BP is controlled, annual or semiannual monitoring for changes in serum biochemistries such as serum creatinine or potassium are recommended.1 However, for patients with CHF, CKD, or diabetes, more frequent monitoring will be necessary to adequately control comorbid conditions. Another aspect to monitoring relates to the importance of medication adherence. Confirmation of continued use of antihypertensive medications should be considered in the routine monitoring of patients on numerous medications for hypertension. Evaluation of side effects, lab abnormalities, and/or progression to target organ damage should also be considered at appropriate intervals. Given the generally asymptomatic nature of hypertension, patient motivation to adhere to prescribed medications becomes a key tool in controlling hypertension. 2
The frequency of follow-up visits for patients with hypertension will vary based on individual cases, but will be influenced by severity of hypertension, comorbidities, and choice of agent selected. A recent analysis of the VALUE trial found that achieving BP control within 6 months reduced CV events compared to a longer time to achieve goal. In addition, another recent analysis of the INSIGHT trial found that the more times a patient is below goal, the lower the risk.94,95 These studies suggest that there is an urgency to achieve and maintain BP control, and this may necessitate frequent assessment and adjustments (i.e., 2 weeks). At a minimum, assessment of response to medications should be done at 1-month intervals.1 In patients with stage 2 hypertension or those with comorbidities (e.g., diabetes, vascular disease, CHF, or CKD), shorter time frames of 2 weeks or less are more appropriate.79 Once BP is controlled, annual or semiannual monitoring for changes in serum biochemistries such as serum creatinine or potassium are recommended.1 However, for patients with CHF, CKD, or diabetes, more frequent monitoring will be necessary to adequately control comorbid conditions. Another aspect to monitoring relates to the importance of medication adherence. Confirmation of continued use of antihypertensive medications should be considered in the routine monitoring of patients on numerous medications for hypertension. Evaluation of side effects, lab abnormalities, and/or progression to target organ damage should also be considered at appropriate intervals. Given the generally asymptomatic nature of hypertension, patient motivation to adhere to prescribed medications becomes a key tool in controlling hypertension. 2
Given the chronic nature of hypertension, parsimony of medication regimens is a virtue of a good therapeutic plan. Minimizing the number of medications a patient is required to take has the potential to enhance adherence and mitigate cost. Often, control of BP is achieved by use of two or even three or more BP lowering medications.34,96 Many combination products contain a diuretic as one of their active components. However, combination therapy may limit the ability of the clinician to titrate the dose of a specific agent. As such, the number of medications (“pill count”) may often be reduced through use of combination products. This inherently simplifies the number of medications and copays a patient may have to endure to achieve effective BP control. These practicalities, although obvious, go a long way to optimize adherence, another challenge to maximizing therapy effectiveness.
Patient Care and Monitoring
1. Measure patient BP twice, at least 1 minute apart in a sitting position, and then average the readings to determine if BP is adequately controlled.
2. Conduct a medical history. Does the patient have any compelling indications? Is the patient pregnant?
3. Conduct a medication history (prescription, over-the-counter, and dietary supplements) to determine conditions or causes of hypertension. Does the patient take any medications, supplements, herbal products, or foods that may elevate SBP or DBP? Does the patient have drug allergies?
4. Review available laboratory tests to examine electrolyte balance and renal function.
5. Discuss lifestyle modifications that may reduce BP with the patient. Determine what nonpharmacologic approaches might be or have been helpful to the patient.
6. Evaluate the patient if pharmacologic treatment has reached the target BP goal. If the patient is at the goal, skip to step 9.
7. If patient is not at goal BP, assess efficacy, safety, and compliance of the antihypertensive regimen to determine if a dose increase or additional antihypertensive agent (step 8) is needed to achieve goal BP.
8. Select an agent to minimize adverse drug reactions and interactions when additional drug therapy is needed. Does the patient have prescription coverage or is the recommended agent in the formulary?
9. Open a dialogue to address patient concerns about hypertension and management of the condition.
10. Provide a plan to assess effectiveness and safety of therapy. Follow-up at monthly intervals or less until BP is achieved, otherwise semiannual or annual clinic visits to assess electrolyte balance and renal function once BP target is achieved. Presence of comorbidities (i.e., CKD) may require more frequent follow-up.
OUTCOME EVALUATION
• Short-term goals are to safely achieve reduction in BP through the iterative process of employing drug therapy, along with nondrug therapy or lifestyle changes.
• Lifestyle changes should address other risk factors for CVD including obesity, physical inactivity, insulin resistance, dyslipidemia, smoking cessation, and others.
• Monitoring for efficacy, adverse events, and adherence to therapy is key to achieving the long-term goals of reducing the risk of morbidity and mortality associated with CVD.
Abbreviations Introduced in This Chapter

 Self-assessment questions and answers are available at http://www.mhpharmacotherapy.com/pp.html.
Self-assessment questions and answers are available at http://www.mhpharmacotherapy.com/pp.html.
REFERENCES
1. Chobanian AV, Bakris GL, Black HR, et al. Seventh report of the Joint National Committee on Prevention, Detection, Evaluation, and Treatment of High Blood Pressure. Hypertension 2003;42(6):1206–1252.
2. Rosendorff C, Black HR, Cannon CP, et al. Treatment of hypertension in the prevention and management of ischemic heart disease: A scientific statement from the American Heart Association Council for High Blood Pressure Research and the Councils on Clinical Cardiology and Epidemiology and Prevention. Circulation 2007 29;115(21):2761–2788.
3. Mancia G, De Backer G, Dominiczak A, et al. 2007 Guidelines for the management of arterial hypertension: The Task Force for the Management of Arterial Hypertension of the European Society of Hypertension (ESH) and of the European Society of Cardiology (ESC). Eur Heart J 28 2007;(12):1462–1536.
4. WHO. The world health report 2002-reducing risks, promoting healthy life.. Available at: http://www.who.int/whr/2002/en/whr02_en.pdf
5. Rosamond W, Flegal K, Furie K, et al. Heart disease and stroke statistics—2008 update: A report from the American Heart Association Statistics Committee and Stroke Statistics Subcommittee. Circulation 29 2008;117(4):e25–e146.
6. Ostchega Y, Hughes JP, Wright JD, McDowell MA, Louis T. Are demographic characteristics, health care access and utilization, and comorbid conditions associated with hypertension among US adults? Am J Hypertens 2008;(2):159–165.
7. Sowers JR, Haffner S. Treatment of cardiovascular and renal risk factors in the diabetic hypertensive. Hypertension Dec 2002;40(6):781–788.
8. Arauz-Pacheco C, Parrott MA, Raskin P. Hypertension management in adults with diabetes. Diabetes Care 2004;27(Suppl 1):S65–S67.
9. Kaplan NM, Flynn JT. Kapalan’s Clinical Hypertension, 9th ed. Philadelphia, PA: Lippincott Williams & Wilkins, 2006.
10. Izzo JL, Sica DA, Black HR. Council for High Blood Pressure Research (American Heart Association). Hypertension Primer, 4th ed. Philadelphia, PA: Lippincott Williams & Wilkins, 2008.
11. Dominiczak AF, Negrin DC, Clark JS, Brosnan MJ, McBride MW, Alexander MY. Genes and hypertension: From gene mapping in experimental models to vascular gene transfer strategies. Hypertension 2000;35(1 Pt 2):164–172.
12. Meneton PG, Warnock D. Involvement of renal apical Na transport systems in the control of blood pressure. Am J Kidney Dis 2001;37 (1 Suppl 2):S39–S47.
13. Genome-wide association study of 14,000 cases of seven common diseases and 3,000 shared controls. Nature 2007;447(7145): 661–678.
14. Ehret GB, Morrison AC, O’Connor AA, et al. Replication of the Wellcome Trust genome-wide association study of essential hypertension: The Family Blood Pressure Program. Eur J Hum Genet 2008;16:1507–1511.
15. Chobanian AV, Hill M. National Heart, Lung, and Blood Institute Workshop on Sodium and Blood Pressure: A critical review of current scientific evidence. Hypertension 2000;35(4):858–863.
16. Phillips MI, Schmidt-Ott KM. The Discovery of Renin 100 Years Ago. News Physiol Sci 1999;14:271–274.
17. Lavoie JL, Sigmund CD. Minireview: Overview of the renin-angiotensin system—An endocrine and paracrine system. Endocrinology 2003; 144(6):2179–2183.
18. Laragh J. Laragh’s lessons in pathophysiology and clinical pearls for treating hypertension. Am J Hypertens 2001;14(1):84–89.
19. Brown MJ. Aliskiren. Circulation 2008;118(7):773–784.
20. Pickering TG, Hall JE, Appel LJ, et al. Recommendations for blood pressure measurement in humans and experimental animals: Part 1: Blood pressure measurement in humans: A statement for professionals from the subcommittee of professional and public education of the American heart association council on high blood pressure research. Circulation 2005;111(5):697–716.
21. Lewington S, Clarke R, Qizilbash N, Peto R, Collins R. Age-specific relevance of usual blood pressure to vascular mortality: A meta-analysis of individual data for one million adults in 61 prospective studies. Lancet 2002;360(9349):1903–1913.
22. Major outcomes in high-risk hypertensive patients randomized to angiotensin-converting enzyme inhibitor or calcium channel blocker vs diuretic: The Antihypertensive and Lipid-Lowering Treatment to Prevent Heart Attack Trial (ALLHAT). JAMA 2002;288(23): 2981–2997.
23. Sacks FM, Svetkey LP, Vollmer WM, et al. Effects on blood pressure of reduced dietary sodium and the Dietary Approaches to Stop Hypertension (DASH) diet. DASH-Sodium Collaborative Research Group. N Engl J Med 2001;344(1):3–10.
24. Beard TC. The bread of the 21st century. Aust J Nutr Diet 1997;54:198–203.
25. Whelton SP, Chin A, Xin X, He J. Effect of aerobic exercise on blood pressure: A meta-analysis of randomized, controlled trials. Ann Intern Med 2002;136(7):493–503.
26. Gibbons RJ, Abrams J, Chatterjee K, et al. ACC/AHA 2002 guideline update for the management of patients with chronic stable angina—summary article: A report of the American College of Cardiology/American Heart Association Task Force on practice guidelines (Committee on the Management of Patients With Chronic Stable Angina). J Am Coll Cardiol 2003;41(1):159–168.
27. Simmons-Morton DG. Exercise Therapy. In: Izzo JL, Jr, Black HR, eds. Hypertension Primer: The Essentials of High Blood Pressure, 3rd ed. Philadelphia: Lippincott Williams & Wilkins, 2003:388–389.
28. Rosito GA, Fuchs FD, Duncan BB. Dose-dependent biphasic effect of ethanol on 24-h blood pressure in normotensive subjects. Am J Hypertens 1999;12)2 Pt 1):236–240.
29. Xin X, He J, Frontini MG, Ogden LG, Motsamai OI, Whelton PK. Effects of alcohol reduction on blood pressure: A meta-analysis of randomized controlled trials. Hypertension 2001;38(5):1112–1117.
30. Omvik P. How smoking affects blood pressure. Blood Press 1996;5(2): 71–77.
31. Primatesta P, Falaschetti E, Gupta S, Marmot MG, Poulter NR. Association between smoking and blood pressure: Evidence from the health survey for England. Hypertension 2001;37(2):187–193.
32. Dahlof B, Sever PS, Poulter NR, et al. Prevention of cardiovascular events with an antihypertensive regimen of amlodipine adding perindopril as required versus atenolol adding bendroflumethiazide as required, in the Anglo-Scandinavian Cardiac Outcomes Trial-Blood Pressure Lowering Arm (ASCOT-BPLA): A multicentre randomised controlled trial. Lancet 2005;366(9489):895–906.
33. Julius S, Kjeldsen SE, Weber M, et al. Outcomes in hypertensive patients at high cardiovascular risk treated with regimens based on valsartan or amlodipine: The VALUE randomised trial. Lancet 2004;363(9426):2022–2031.
34. ALLHAT Collaborative Research Group. Major cardiovascular events in hypertensive patients randomized to doxazosin vs chlorthalidone: The antihypertensive and lipid-lowering treatment to prevent heart attack trial (ALLHAT). JAMA 2000;283(15):1967–1975.
35. Randomised trial of a perindopril-based blood-pressure-lowering regimen among 6,105 individuals with previous stroke or transient ischaemic attack. Lancet 2001;358(9287):1033–1041.
36. Beckett NS, Peters R, Fletcher AE, et al. Treatment of hypertension in patients 80 years of age or older. N Engl J Med 2008;358(18):1887–1898.
37. Pitt B, Zannad F, Remme WJ, et al. The effect of spironolactone on morbidity and mortality in patients with severe heart failure. Randomized Aldactone Evaluation Study Investigators. N Engl J Med 1999;341(10):709–717.
38. Pitt B, Remme W, Zannad F, et al. Eplerenone, a selective aldosterone blocker, in patients with left ventricular dysfunction after myocardial infarction. N Engl J Med 2003;348(14):1309–1321. Epub 2003 Mar 1331.
39. Effect of metoprolol CR/XL in chronic heart failure: Metoprolol CR/XL Randomised Intervention Trial in Congestive Heart Failure (MERIT-HF). Lancet 1999;353(9169):2001–2007.
40. Krum H, Roecker EB, Mohacsi P, et al. Effects of initiating carvedilol in patients with severe chronic heart failure: Results from the COPERNICUS Study. JAMA 2003;289(6):712–718.
41. Dargie HJ. Effect of carvedilol on outcome after myocardial infarction in patients with left-ventricular dysfunction: The CAPRICORN randomised trial. Lancet 2001;357(9266):1385–1390.
42. Lampert R, Ickovics JR, Viscoli CJ, Horwitz RI, Lee FA. Effects of propranolol on recovery of heart rate variability following acute myocardial infarction and relation to outcome in the Beta-Blocker Heart Attack Trial. Am J Cardiol 2003;91(2):137–142.
43. The Cardiac Insufficiency Bisoprolol Study II (CIBIS-II): A randomised trial. Lancet 1999;353(9146):9–13.
44. Pepine CJ, Handberg EM, Cooper-DeHoff RM, et al. A calcium antagonist vs a non-calcium antagonist hypertension treatment strategy for patients with coronary artery disease. The International Verapamil-Trandolapril Study (INVEST): A randomized controlled trial. JAMA 2003;290(21):2805–2816.
45. Effect of enalapril on mortality and the development of heart failure in asymptomatic patients with reduced left ventricular ejection fractions. The SOLVD Investigattors. N Engl J Med 1992;327(10):685–691.
46. Effect of ramipril on mortality and morbidity of survivors of acute myocardial infarction with clinical evidence of heart failure. The Acute Infarction Ramipril Efficacy (AIRE) Study Investigators. Lancet 1993;342(8875):821–828.
47. Kober L, Torp-Pedersen C, Carlsen JE, et al. A clinical trial of the angiotensin-converting-enzyme inhibitor trandolapril in patients with left ventricular dysfunction after myocardial infarction. Trandolapril Cardiac Evaluation (TRACE) Study Group. N Engl J Med 1995;333(25):1670–1676.
48. Pfeffer MA, Braunwald E, Moye LA, et al. Effect of captopril on mortality and morbidity in patients with left ventricular dysfunction after myocardial infarction. Results of the survival and ventricular enlargement trial. The SAVE Investigators. N Engl J Med 1992;327(10):669–677.
49. Lewis EJ, Hunsicker LG, Bain RP, Rohde RD. The effect of angiotensin-converting-enzyme inhibition on diabetic nephropathy. The Collaborative Study Group. N Engl J Med 1993;329(20):1456–1462.
50. Fox KM. Efficacy of perindopril in reduction of cardiovascular events among patients with stable coronary artery disease: Randomised, double-blind, placebo-controlled, multicentre trial (the EUROPA study). Lancet 2003;362(9386):782–788.
51. Pfeffer MA, Swedberg K, Granger CB, et al. Effects of candesartan on mortality and morbidity in patients with chronic heart failure: The CHARM-Overall programme. Lancet 2003;362(9386):759–766.
52. Dahlof B, Devereux RB, Kjeldsen SE, et al. Cardiovascular morbidity and mortality in the Losartan Intervention For Endpoint reduction in hypertension study (LIFE): A randomised trial against atenolol. Lancet 2002;359(9311):995–1003.
53. Brenner BM, Cooper ME, de Zeeuw D, et al. Effects of losartan on renal and cardiovascular outcomes in patients with type 2 diabetes and nephropathy. N Engl J Med 2001;345(12):861–869.
54. Lewis EJ, Hunsicker LG, Clarke WR, et al. Renoprotective effect of the angiotensin-receptor antagonist irbesartan in patients with nephropathy due to type 2 diabetes. N EnglJ Med 2001;345(12): 851–860.
55. Parving HH, Lehnert H, Brochner-Mortensen J, Gomis R, Andersen S, Arner P. The effect of irbesartan on the development of diabetic nephropathy in patients with type 2 diabetes. N Engl J Med 2001;345(12):870–878.
56. Cohn JN, Tognoni G. A randomized trial of the angiotensin-receptor blocker valsartan in chronic heart failure. N Engl J Med 2001;345(23):1667–1675.
57. Taylor AL, Ziesche S, Yancy C, et al. Combination of isosorbide dinitrate and hydralazine in blacks with heart failure. N Engl J Med 2004;351(20):2049–2057.
58. Diuretic versus alpha-blocker as first-step antihypertensive therapy: Final results from the Antihypertensive and Lipid-Lowering Treatment to Prevent Heart Attack Trial (ALLHAT). Hypertension 2003; 42(3): 239–246.
59. Wing LM, Reid CM, Ryan P, et al. A comparison of outcomes with angiotensin-converting—enzyme inhibitors and diuretics for hypertension in the elderly. N Engl J Med 2003;348(7):583–592.
60. Psaty BM, Lumley T, Furberg CD, et al. Health outcomes associated with various antihypertensive therapies used as first-line agents: A network meta-analysis. JAMA 2003;289(19):2534–2544.
61. Jamerson K, Weber MA, Bakris GL, et al. Benazepril plus amlodipine or hydrochlorothiazide for hypertension in high-risk patients. N Engl J Med 2008;359:2417–2428.
62. Ernst ME, Carter BL, Goerdt CJ, et al. Comparative antihypertensive effects of hydrochlorothiazide and chlorthalidone on ambulatory and office blood pressure. Hypertension 2006;47(3):352–358.
63. Sica DA. Chlorthalidone: Has it always been the best thiazide-type diuretic? Hypertension 2006;47(3):321–322.
64. Carter BL, Ernst ME, Cohen JD. Hydrochlorothiazide versus chlorthalidone: Evidence supporting their interchangeability. Hypertension 2004;43(1):4–9.
65. Zillich AJ, Garg J, Basu S, Bakris GL, Carter BL. Thiazide diuretics, potassium, and the development of diabetes: A quantitative review. Hypertension 2006;48(2):219–224.
66. Carter BL, Einhorn PT, Brands M, et al. Thiazide-induced dysglycemia: Call for research from a working group from the national heart, lung, and blood institute. Hypertension 2008;52(1):30–36.
67. Goodman LS, Hardman JG, Limbird LE, Gilman AG. Goodman & Gilman’s the pharmacological Basis of Therapeutics, 10th ed. New York: McGraw-Hill, 2001.
68. Nishizaka MK, Zaman MA, Calhoun DA. Efficacy of low-dose spironolactone in subjects with resistant hypertension. Am J Hypertens 2003;16(11 Pt 1):925–930.
69. Menard J. The 45-year story of the development of an anti-aldosterone more specific than spironolactone. Mol Cell Endocrinol 2004;217(1–2): 45–52.
70. Sica DA. Pharmacokinetics and pharmacodynamics of mineralocor-ticoid blocking agents and their effects on potassium homeostasis. Heart Fail Rev 2005;10(1):23–29.
71. Lindholm LH, Carlberg B, Samuelsson O. Should beta blockers remain first choice in the treatment of primary hypertension? A meta-analysis. Lancet 2005;366(9496):1545–1553.
72. Messerli FH, Grossman E, Goldbourt U. Are beta-blockers efficacious as first-line therapy for hypertension in the elderly? A systematic review. JAMA 1998;279(23):1903–1907.
73. Williams B, Lacy PS, Thom SM, et al. Differential impact of blood pressure-lowering drugs on central aortic pressure and clinical outcomes: Principal results of the Conduit Artery Function Evaluation (CAFE) study. Circulation 2006;113(9):1213–1225.
74. Bangalore S, Messerli FH, Kostis JB, Pepine CJ. Cardiovascular protection using beta-blockers: A critical review of the evidence. J Am Coll Cardiol 2007;50(7):563–572.
75. Weber MA. The role of the new beta-blockers in treating cardiovascular disease. Am J Hypertens 2005;18(12 Pt 2):169S–176S.
76. Antman EM, Anbe DT, Armstrong PW, et al. ACC/AHA guidelines for the management of patients with ST-elevation myocardial infarction--executive summary: A report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines (Writing Committee to Revise the 1999 Guidelines for the Management of Patients With Acute Myocardial Infarction). Circulation 2004; 110(5):588–636.
77. Hunt SA, Abraham WT, Chin MH, et al. ACC/AHA 2005 Guideline Update for the Diagnosis and Management of Chronic Heart Failure in the Adult—Summary Article: A Report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines (Writing Committee to Update the 2001 Guidelines for the Evaluation and Management of Heart Failure): Developed in Collaboration With the American College of Chest Physicians and the International Society for Heart and Lung Transplantation: Endorsed by the Heart Rhythm Society. Circulation Sep 20, 2005;112(12):1825–1852.
78. Standards of medical care in diabetes. Diabetes Care 2005;28(Suppl 1): S4–S 36.
79. K/DOQI clinical practice guidelines on hypertension and antihypertensive agents in chronic kidney disease. Am J Kidney Dis 2004;43(5 Suppl 1):S1–S 290.
80. Bakris GL, Williams M, Dworkin L, et al. Preserving renal function in adults with hypertension and diabetes: A consensus approach. National Kidney Foundation Hypertension and Diabetes Executive Committees Working Group. Am J Kidney Dis 2000;36(3):646–661.
81. The Telmisartan Randomised AssesmeNt Study in ACE iNtolerant subject with cardiovascular Disease (TRANSCEND) investigator. Effects of the angiotensin-receptor blocker telmisartan on cardiovascular events in high-risk patients intolerant to angiotensin-converting enzyme inhibitors: a randomised controlled trial. Lancet 2008; published online Aug 31. DOI: 10.1016/S 0140-6736(08)61242-8.
82. Ripley TL, Harrison D. The power to TRANSCEND. Lancet 2008.
83. McMurray JJ, Ostergren J, Swedberg K, et al. Effects of candesartan in patients with chronic heart failure and reduced left-ventricular systolic function taking angiotensin-converting-enzyme inhibitors: The CHARM-Added trial. Lancet 2003;362(9386):767–771.
84. Yusuf S, Pfeffer MA, Swedberg K, et al. Effects of candesartan in patients with chronic heart failure and preserved left-ventricular ejection fraction: the CHARM-Preserved Trial. Lancet 2003;362(9386):777–781.
85. Granger CB, McMurray JJ, Yusuf S, et al. Effects of candesartan in patients with chronic heart failure and reduced left-ventricular systolic function intolerant to angiotensin-converting-enzyme inhibitors: The CHARM-Alternative trial. Lancet 2003;362(9386):772–776.
86. Prevention of stroke by antihypertensive drug treatment in older persons with isolated systolic hypertension. Final results of the Systolic Hypertension in the Elderly Program (SHEP). SHEP Cooperative Research Group. JAMA 1991;265(24):3255–3264.
87. DiPiro JT. Pharmacotherapy: A pathophysiologic Approach, 7th ed. New York: McGraw-Hill Medical, 2008.
88. Yusuf S, Sleight P, Pogue J, Bosch J, Davies R, Dagenais G. Effects of an angiotensin-converting-enzyme inhibitor, ramipril, on cardiovascular events in high-risk patients. The Heart Outcomes Prevention Evaluation Study Investigators. N Engl J Med 2000;342(3):145–153.
89. Yusuf S, Teo KK, Pogue J, et al. Telmisartan, ramipril, or both in patients at high risk for vascular events. N Engl J Med 2008;358(15):1547–1559.
90. Somes GW, Pahor M, Shorr RI, Cushman WC, Applegate WB. The role of diastolic blood pressure when treating isolated systolic hypertension. Arch Intern Med 1999;159(17):2004–2009.
91. The fourth report on the diagnosis, evaluation, and treatment of high blood pressure in children and adolescents. Pediatrics 2004;114(2 Suppl 4th Report):555–576.
92. Urbina E, Alpert B, Flynn J, et al. Ambulatory blood pressure monitoring in children and adolescents: Recommendations for standard assessment: A scientific statement from the American Heart Association Atherosclerosis, Hypertension, and Obesity in Youth Committee of the Council on Cardiovascular Disease in the Young and the Council for High Blood Pressure Research. Hypertension 2008;52(3):433–451.
93. Kliegman R, Nelson WE. Nelson Textbook of Pediatrics, 18th ed. Philadelphia: Saunders, 2007.
94. Mancia G, Messerli F, Bakris G, Zhou Q, Champion A, Pepine CJ. Blood pressure control and improved cardiovascular outcomes in the International Verapamil SR-Trandolapril Study. Hypertension 2007;50(2):299–305.
95. Weber MA, Julius S, Kjeldsen SE, et al. Blood pressure dependent and independent effects of antihypertensive treatment on clinical events in the VALUE Trial. Lancet 2004;363(9426):2049–2051.
96. Hansson L, Zanchetti A, Carruthers SG, et al. Effects of intensive blood-pressure lowering and low-dose aspirin in patients with hypertension: Principal results of the Hypertension Optimal Treatment (HOT) randomised trial. HOT Study Group. Lancet 1998;351(9118):1755–1762.