Melinda M. Neuhauser and Susan L. Pendland
LEARNING OBJECTIVES
Upon completion of the chapter, the reader will be able to:
1. Discuss the pathophysiology of osteomyelitis.
2. List common risk factors for osteomyelitis.
3. Compare and contrast the classic signs and symptoms of acute and chronic osteomyelitis.
4. Evaluate microbiology culture data and other laboratory tests utilized for the diagnosis and treatment of bone infections.
5. List the most common pathogens isolated in acute and chronic osteomyelitis.
6. Develop a treatment plan for osteomyelitis.
7. Recommend parameters to monitor antimicrobial therapy for effectiveness and toxicity.
8. Educate patients regarding disease state and drug therapy.
KEY CONCEPTS
![]() Osteomyelitis, an infection of the bone, can be an acute or chronic process.
Osteomyelitis, an infection of the bone, can be an acute or chronic process.
![]() Osteomyelitis is most often classified by route of infection and duration of disease.
Osteomyelitis is most often classified by route of infection and duration of disease.
![]() Staphylococcus aureus is the predominant pathogen seen in all types of osteomyelitis. However, the spectrum of potential causative pathogens varies with patient-specific risk factors and route of infection.
Staphylococcus aureus is the predominant pathogen seen in all types of osteomyelitis. However, the spectrum of potential causative pathogens varies with patient-specific risk factors and route of infection.
![]() Typical signs and symptoms of osteomyelitis include local pain and tenderness over the affected bone, as well as inflammation, erythema, edema, and decreased range of motion. Patients with acute hematogenous osteomyelitis may also present with fever, chills, and malaise.
Typical signs and symptoms of osteomyelitis include local pain and tenderness over the affected bone, as well as inflammation, erythema, edema, and decreased range of motion. Patients with acute hematogenous osteomyelitis may also present with fever, chills, and malaise.
![]() The gold standard for diagnosis of osteomyelitis is a bone biopsy with isolation of microorganism(s) from culture and the presence of inflammatory cells and osteonecrosis on histological exam.1–3 Due to the invasive nature of the bone biopsy, the diagnosis of osteomyelitis is often based upon on clinical findings, laboratory tests, and imaging studies rather than bone biopsy.4 A thorough history and physical examination are especially important for diagnosis in patients with limited or atypical symptoms.
The gold standard for diagnosis of osteomyelitis is a bone biopsy with isolation of microorganism(s) from culture and the presence of inflammatory cells and osteonecrosis on histological exam.1–3 Due to the invasive nature of the bone biopsy, the diagnosis of osteomyelitis is often based upon on clinical findings, laboratory tests, and imaging studies rather than bone biopsy.4 A thorough history and physical examination are especially important for diagnosis in patients with limited or atypical symptoms.
![]() The treatment goals for acute and chronic osteomyelitis are to eradicate the infection and prevent recurrence. Higher cure rates are seen with acute compared to chronic osteomyelitis. Therefore, in chronic osteomyelitis, a common treatment goal for many patients is to prevent complications such as amputation.
The treatment goals for acute and chronic osteomyelitis are to eradicate the infection and prevent recurrence. Higher cure rates are seen with acute compared to chronic osteomyelitis. Therefore, in chronic osteomyelitis, a common treatment goal for many patients is to prevent complications such as amputation.
![]() Treatment of osteomyelitis is dependent on the extent of bone necrosis. For acute osteomyelitis with minimal bone destruction, an extended course of antimicrobial therapy should effectively treat the infection; however, in chronic osteomyelitis surgical intervention is also typically required.
Treatment of osteomyelitis is dependent on the extent of bone necrosis. For acute osteomyelitis with minimal bone destruction, an extended course of antimicrobial therapy should effectively treat the infection; however, in chronic osteomyelitis surgical intervention is also typically required.
![]() Empiric antimicrobial therapy should target likely causative pathogen(s) based on patient-specific risk factors and route of infection. However, therapy should be modified based on culture and sensitivity data.
Empiric antimicrobial therapy should target likely causative pathogen(s) based on patient-specific risk factors and route of infection. However, therapy should be modified based on culture and sensitivity data.
![]() The total duration of antimicrobial therapy is typically 4 to 6 weeks. Therapy is often administered IV for 1 or 2 weeks and then switched to the oral route.
The total duration of antimicrobial therapy is typically 4 to 6 weeks. Therapy is often administered IV for 1 or 2 weeks and then switched to the oral route.
![]() Patients should be monitored for clinical and laboratory response, development of adverse drug reactions, and potential drug–drug interactions. Patients should also be closely monitored for compliance in the outpatient setting.
Patients should be monitored for clinical and laboratory response, development of adverse drug reactions, and potential drug–drug interactions. Patients should also be closely monitored for compliance in the outpatient setting.
INTRODUCTION
![]() Osteomyelitis is an infection of the bone that is associated with high morbidity and increased health care costs. The inflammatory response associated with acute osteomyelitis can lead to bone necrosis and subsequently chronic infections. Bacterial pathogens, particularly Staphylococcus aureus, are the most common microorganisms implicated in these infections. Diagnosis and treatment are often difficult due to the heterogeneous nature of osteomyelitis. Medical management is the mainstay of treatment for acute infections; however, surgical intervention is necessary for chronic cases that involve bone necrosis. Outcomes may vary based on patient-specific risk factors, duration of disease, and site of infection.
Osteomyelitis is an infection of the bone that is associated with high morbidity and increased health care costs. The inflammatory response associated with acute osteomyelitis can lead to bone necrosis and subsequently chronic infections. Bacterial pathogens, particularly Staphylococcus aureus, are the most common microorganisms implicated in these infections. Diagnosis and treatment are often difficult due to the heterogeneous nature of osteomyelitis. Medical management is the mainstay of treatment for acute infections; however, surgical intervention is necessary for chronic cases that involve bone necrosis. Outcomes may vary based on patient-specific risk factors, duration of disease, and site of infection.
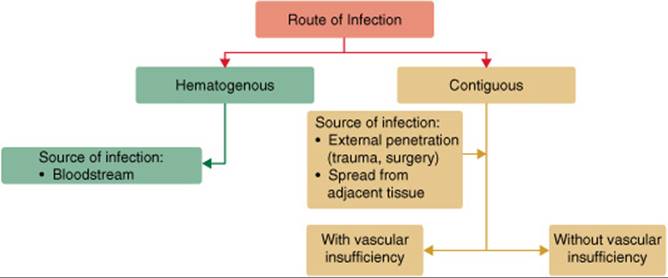
FIGURE 81–1. Classification by route of infection.
EPIDEMIOLOGY AND ETIOLOGY
![]() There are multiple classification schemes for osteomyelitis.5 Due to the heterogeneity of bone infections, no single classification system has been universally accepted.1 Two of the most common classification schemes are based onroute of infection and duration of disease.1,4–6 In the classification scheme developed by Waldvogel and colleagues,6 the route of infection is categorized as either hematogenous or contiguous (Fig. 81–1). Osteomyelitis secondary to a contiguous focus was further subdivided into infections with or without vascular insufficiency.
There are multiple classification schemes for osteomyelitis.5 Due to the heterogeneity of bone infections, no single classification system has been universally accepted.1 Two of the most common classification schemes are based onroute of infection and duration of disease.1,4–6 In the classification scheme developed by Waldvogel and colleagues,6 the route of infection is categorized as either hematogenous or contiguous (Fig. 81–1). Osteomyelitis secondary to a contiguous focus was further subdivided into infections with or without vascular insufficiency.
Historically, osteomyelitis has been classified as acute or chronic based on duration of disease (Fig. 81–2).1 However, there are no established definitions for acute and chronic infections.1,4 Acute infection has been defined as first episode or recent onset of symptoms (less than 1 week).4 Chronic osteomyelitis is generally defined as relapse of the disease or symptoms persisting beyond 4 weeks.4 Others describe chronic osteomyelitis as the presence of necrotic bone.4
An alternative classification, the Cierny-Mader staging system, is based on anatomic site and physiologic status of the patient.5 This classification scheme was developed for chronic osteomyelitis involving long bones, but has limited application for small bones and digits. The detailed stratification has the greatest utility in clinical trials since it permits comparison of treatment regimens in patients with diverse comorbidities and infection sites.
The epidemiology of osteomyelitis in adults has been changing over the past several decades.5 The frequency of contiguous osteomyelitis has been increasing. This trend may be related to the rising rates of diabetes and peripheral vascular disease (PVD), as well as the increased presence of prosthetic implants and surgical interventions.4,7,8 In contrast, the incidence of acute hematogenous osteomyelitis, which is most often seen in children, has been declining.7,9
![]() The etiology of osteomyelitis has remained relatively unchanged, with S. aureus being the predominant pathogen seen in all types of osteomyelitis.1,10 However, the susceptibility of S. aureus has been shifting from methicillin-sensitive to methicillin-resistant in both the health care and community settings.11–16 Risk factors for health care–associated methicillin-resistant S. aureus (MRSA) include previous antimicrobial therapy, prolonged hospitalization, hemodialysis, or presence of an indwelling catheter.17 In contrast, community-associated MRSA has emerged in patients with no prior health care exposure or apparent risk factors.14–17 While more common in skin and soft tissue infections, both health care-and community-associated MRSA have been increasingly reported in osteomyelitis.18,19
The etiology of osteomyelitis has remained relatively unchanged, with S. aureus being the predominant pathogen seen in all types of osteomyelitis.1,10 However, the susceptibility of S. aureus has been shifting from methicillin-sensitive to methicillin-resistant in both the health care and community settings.11–16 Risk factors for health care–associated methicillin-resistant S. aureus (MRSA) include previous antimicrobial therapy, prolonged hospitalization, hemodialysis, or presence of an indwelling catheter.17 In contrast, community-associated MRSA has emerged in patients with no prior health care exposure or apparent risk factors.14–17 While more common in skin and soft tissue infections, both health care-and community-associated MRSA have been increasingly reported in osteomyelitis.18,19
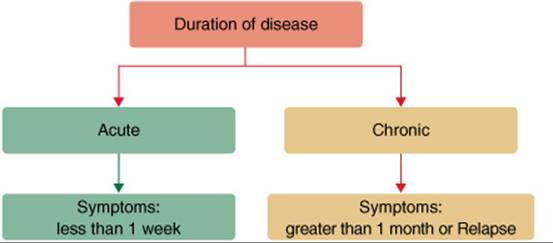
FIGURE 81–2. Classification by duration of disease.
Host factors such as age, comorbidities, medication, and presence of foreign devices can influence the spectrum of infection (i.e., bone and pathogen involvement) (Table 81–1).1 For example, patients with diabetes and PVD have poor wound healing and often present with nonhealing skin ulcers. These wounds are typically colonized with a mixture of aerobic and anaerobic microorganisms, which can lead to polymicrobial osteomyelitis. Therefore, if a wound is deep or extensive, these patients should be evaluated for underlying osteomyelitis.2 Other special populations that have a varied pathogen spectrum include intravenous drug abusers (IVDA) (Pseudomonas aeruginosa and MRSA), sickle cell patients (Salmonella), and individuals with prosthetic implants (coagulase-negative staphylococci).4 Recently, rare cases of osteomyelitis of the jaw have been reported in patients receiving bisphosphonate therapy.20,21 Risk factors include cancer, chemotherapy, corticosteroids, tooth extraction and poor oral hygiene. Bisphosphates may induce osteoclast apoptosis and slow wound healing, leaving oral lesions susceptible to infection from oral microflora.21
Acute Hematogenous Osteomyelitis
• Single pathogen most often isolated
• Staphylococcus aureus is the predominant pathogen
• Other pathogens based on risk factors:
• Neonates: Eschericia coli or group B streptococci
• Elderly: E. coli (secondary to urinary tract infections)
Contiguous Focus Osteomyelitis With Vascular Insufficiency
• Multiple pathogens often isolated
• Mixture of aerobic and anaerobic organisms
• Staphylococcus aureus, Enterococcus spp., Enterobacteriaceae, Pseudomonas aeruginosa, anaerobes
Table 81–1 Empiric Antimicrobial Therapy for Osteomyelitis
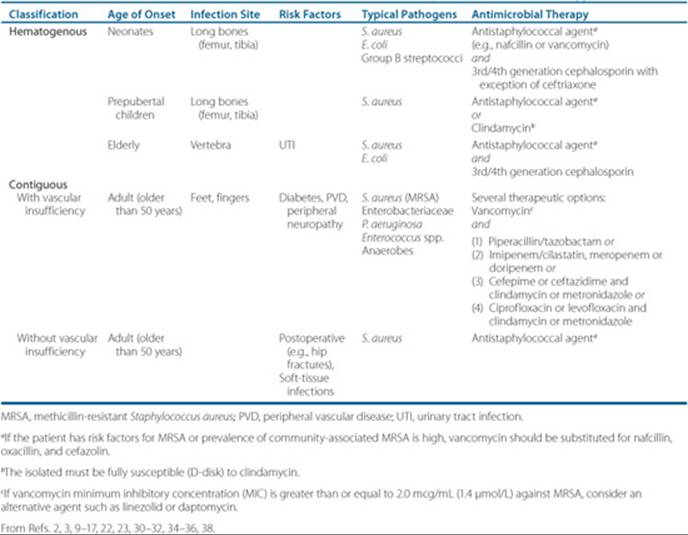
Contiguous Focus Osteomyelitis Without Vascular Insufficiency
• Single or multiple pathogens isolated
• S. aureus is predominant pathogen
• Other pathogens based on source of infection:
• Mandibular osteomyelitis (mixture of aerobic and anaerobic oral flora)
PATHOPHYSIOLOGY
Both microbial and host factors are important determinants in the development of osteomyelitis.1,3,4 Healthy bone tissue is normally resistant to infection but may become susceptible under certain conditions.1,3 Bone can become infected: (a) via the presence of bacteria in the bloodstream, (b) by direct inoculation from trauma or surgery, and (c) by spread from an adjacent site (e.g., soft-tissue infection).1,3 The latter is particularly problematic in patients with foreign body implants (e.g., hip replacement) and chronic skin ulcers.2,4 Staphylococcus species possess bacterial adhesins, which promote their attachment to tissues and foreign devices.3 Microbial adherence to bone elicits an inflammatory response.4 The subsequent release of leukocytes and cytokines leads to edema and ischemia. In some cases, these processes can lead to bone necrosis.3 Pieces of dead bone may become separated forming sequestra.3,4These areas typically cannot be penetrated by antimicrobials and phagocytic cells and thus require surgical intervention to eradicate the bacterial nidus (Fig. 81–3).4
CLINICAL PRESENTATION AND DIAGNOSIS
The clinical presentation of osteomyelitis may vary depending on route and duration of infection, as well as patient-specific factors such as infection site, age, and comorbidities. ![]() In hematogenous osteomyelitis, the patient typically experiences systemic and localized signs and symptoms.4,9,22,23 In comparison, patients with chronic infection typically present with only localized signs and symptoms.7 A cardinal sign of chronic osteomyelitis is the formation of sinus tracts with purulent drainage.4,7
In hematogenous osteomyelitis, the patient typically experiences systemic and localized signs and symptoms.4,9,22,23 In comparison, patients with chronic infection typically present with only localized signs and symptoms.7 A cardinal sign of chronic osteomyelitis is the formation of sinus tracts with purulent drainage.4,7
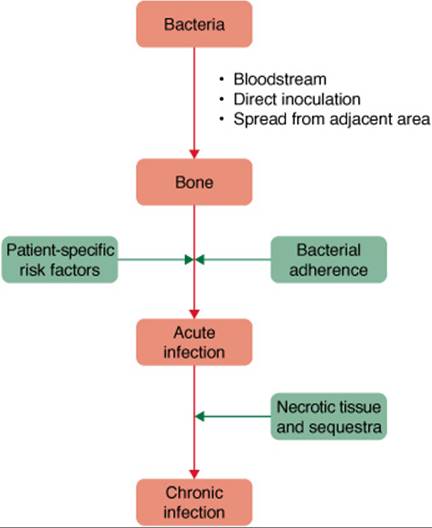
FIGURE 81–3. Pathogenesis of osteomyelitis.
Patient Encounter, Part 1
A 62-year-old male with history of diabetes, hypertension, and PVD comes to the emergency department complaining of a “painful sore” on his left lower leg. After questioning him, you determine that the wound has been present for months and has not responded to oral antibiotic therapy. On physical examination, a large, deep wound with purulent drainage is seen.
What information is suggestive of osteomyelitis?
What risk factors, if any, does he have for osteomyelitis?
Common signs and symptoms of osteomyelitis include:
Systemic: Fever, chills, malaise
Localized: Pain or tenderness, edema, erythema, inflammation, decreased range of motion of infected area
![]() The gold standard for diagnosis of osteomyelitis is a bone biopsy with isolation of microorganism(s) from culture and the presence of inflammatory cells and osteonecrosis on histological exam.1–3 Due to the invasive nature of the bone biopsy, the diagnosis of osteomyelitis is often based upon clinical findings, laboratory tests, and imaging studies rather than bone biopsy.4 A thorough history and physical examination are especially important for diagnosis in patients with limited or atypical symptoms.
The gold standard for diagnosis of osteomyelitis is a bone biopsy with isolation of microorganism(s) from culture and the presence of inflammatory cells and osteonecrosis on histological exam.1–3 Due to the invasive nature of the bone biopsy, the diagnosis of osteomyelitis is often based upon clinical findings, laboratory tests, and imaging studies rather than bone biopsy.4 A thorough history and physical examination are especially important for diagnosis in patients with limited or atypical symptoms.
No single noninvasive laboratory test is currently available for the diagnosis of osteomyelitis. However, despite their low specificity, several tests are commonly used to aid in the diagnosis and to monitor response to therapy. Nonspecific inflammatory markers for infection include white blood cell count (WBC), erythrocyte sedimentation rate (ESR), and C-reactive protein (CRP):4,7,22
• WBC, ESR and CRP are often elevated, but may also be within normal limits. An elevated WBC is mostly seen in patients with acute osteomyelitis.
• CRP rises faster than ESR during early stages of infection and also returns to normal levels more quickly than ESR. This makes CRP a more useful tool for both diagnosis and monitoring of therapeutic response.
A number of different imaging tests are used to assist in the diagnosis of osteomyelitis. These include plain film radiographs, MRI, CT scans and nuclear medicine scans.1–4,7,8
• Plain film radiographs. Most commonly used for initial screening. Bone abnormalities are not apparent for 10 to 21 days, so early infection may be missed.
• MRI. Most accurate for diagnosing bone infection and can detect early infection. However, it is more expensive than radiographs.
• CT scan. Less specific than MRI, but superior to MRI in detection of sequestra. CT is useful for monitoring clinical improvement. It cannot be used to diagnose infections with metallic implants.
• Nuclear medicine scans. Useful for early detection (24–48 hours after onset of symptoms). Radionuclide scans are more sensitive than radiographs, but lack specificity and are expensive. Bone scans demonstrate low specificity in diagnoses involving trauma, surgery, orthopedic implants and diabetes. Leukocyte scans have increased specificity compared to bone scans, but have limited resolution in some anatomical sites.
Microbiologic evaluation.2,4,7,22
• Isolation of causative pathogen is essential for targeted antimicrobial therapy
Patient Encounter, Part 2
The medical resident suspected osteomyelitis and ordered plain film radiographs. He also swabbed the open wound and sent the swab to the microbiology laboratory for culture and sensitivity. The following result was reported:
Radiograph: Lytic changes consistent with bone destruction
How would you classify the infection in this patient?
How should the results of the wound swab culture be utilized to target the patient’s antimicrobial therapy?
• Bone biopsy can provide definitive diagnosis: Samples should be submitted for culture and histology (rarely performed due to invasive nature)
• Blood cultures may be positive in patients with hematogenous osteomyelitis
• Superficial swabs should have a limited role in directing therapy as they may represent colonization rather than infecting organism(s)
TREATMENT
Desired Outcomes
![]() The treatment goals for osteomyelitis are to eradicate the infection and prevent recurrence. Cure rates of greater than 85% have been reported for acute hematogenous osteomyelitis.9,23 In contrast, chronic osteomyelitis is associated with higher failure rates largely due to the presence of necrotic bone.1 These patients typically require surgical intervention to remove the necrotic bone and tissue, and if applicable, to replace infected hardware.4,24Comorbidities such as vascular insufficiency can further contribute to the poor outcomes seen with chronic osteomyelitis. Due to the high failure rates, treatment in this patient population may require prolonged therapy with the primary goal of preventing amputation of infected areas.2,4,7,25
The treatment goals for osteomyelitis are to eradicate the infection and prevent recurrence. Cure rates of greater than 85% have been reported for acute hematogenous osteomyelitis.9,23 In contrast, chronic osteomyelitis is associated with higher failure rates largely due to the presence of necrotic bone.1 These patients typically require surgical intervention to remove the necrotic bone and tissue, and if applicable, to replace infected hardware.4,24Comorbidities such as vascular insufficiency can further contribute to the poor outcomes seen with chronic osteomyelitis. Due to the high failure rates, treatment in this patient population may require prolonged therapy with the primary goal of preventing amputation of infected areas.2,4,7,25
General Approach to Treatment
![]() Antimicrobial therapy alone is the mainstay of treatment for acute osteomyelitis.9,22 In comparison, treatment for chronic osteomyelitis typically requires a combination of antimicrobial therapy and surgical intervention.2–4,7,24,25If the patient is not a candidate for surgical intervention, prolonged antimicrobial therapy is generally necessary.2,7,26
Antimicrobial therapy alone is the mainstay of treatment for acute osteomyelitis.9,22 In comparison, treatment for chronic osteomyelitis typically requires a combination of antimicrobial therapy and surgical intervention.2–4,7,24,25If the patient is not a candidate for surgical intervention, prolonged antimicrobial therapy is generally necessary.2,7,26
Pharmacologic Therapy
![]() Empiric antimicrobial therapy should target likely causative pathogen(s) based on patient-specific risk factors and route of infection (Table 81–1). Empiric antimicrobial coverage against S. aureus should be considered for all classifications of osteomyelitis. Specific recommendations may vary based on factors such as patient allergies, potential for harboring a resistant organism, institution formulary, and cost considerations. Antimicrobial therapy should be modified based on culture and sensitivity data of appropriately collected specimens (Table 81–2).27
Empiric antimicrobial therapy should target likely causative pathogen(s) based on patient-specific risk factors and route of infection (Table 81–1). Empiric antimicrobial coverage against S. aureus should be considered for all classifications of osteomyelitis. Specific recommendations may vary based on factors such as patient allergies, potential for harboring a resistant organism, institution formulary, and cost considerations. Antimicrobial therapy should be modified based on culture and sensitivity data of appropriately collected specimens (Table 81–2).27
![]() Typically, treatment is initiated with IV antimicrobials to ensure that therapeutic drug concentrations will be achieved in the bone.26 IV therapy can be administered in the inpatient or outpatient setting.25,28 Following 1 to 2 weeks of IV therapy, a switch to oral antibiotics may be considered in patients with good adherence and outpatient follow-up.22,25,29 Oral agents should possess such characteristics as high bioavailability, good bone penetration, and long half-life (i.e., extended dosing interval).26,29 Antimicrobials commonly used as oral therapy for osteomyelitis include fluoroquinolones, clindamycin, linezolid, and trimethoprim-sulfamethox azole.1,7,10,23,27,29,30 Additionally, oral rifampin may be used in combination with another antibiotic in the treatment of chronic osteomyelitis, particularly in patients with foreign devices.25,29
Typically, treatment is initiated with IV antimicrobials to ensure that therapeutic drug concentrations will be achieved in the bone.26 IV therapy can be administered in the inpatient or outpatient setting.25,28 Following 1 to 2 weeks of IV therapy, a switch to oral antibiotics may be considered in patients with good adherence and outpatient follow-up.22,25,29 Oral agents should possess such characteristics as high bioavailability, good bone penetration, and long half-life (i.e., extended dosing interval).26,29 Antimicrobials commonly used as oral therapy for osteomyelitis include fluoroquinolones, clindamycin, linezolid, and trimethoprim-sulfamethox azole.1,7,10,23,27,29,30 Additionally, oral rifampin may be used in combination with another antibiotic in the treatment of chronic osteomyelitis, particularly in patients with foreign devices.25,29
Table 81–2 Pathogen-Targeted Antimicrobial Therapy and Dosing Recommendations in Pediatric and Adult Patients
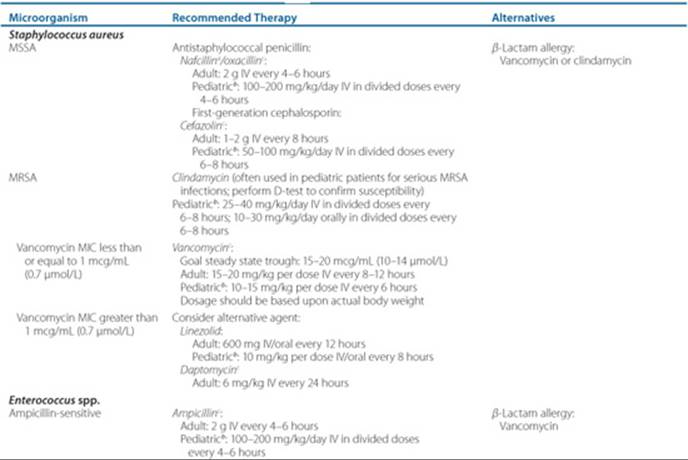
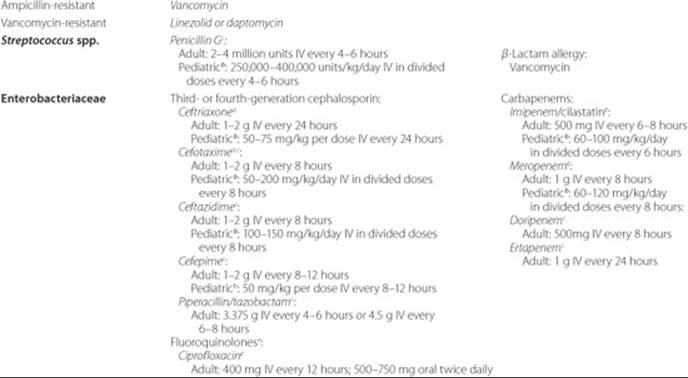

With increasing methicillin resistance, clinicians will need to be aware of changing treatment strategies for infections that are often staphylococcal in origin. Historically, IV vancomycin has been first-line therapy for serious MRSA infections. Based upon pharmacokinetic/pharmacodynamic principles, higher vancomycin trough levels (15–20 mcg/mL; 10–14 μmol/L) have been recommended for serious infections including osteomyelitis.31,32 Although susceptibility of MRSA to vancomycin remains almost 100%,32 vancomycin displays reduced activity against MRSA with minimum inhibitory concentrations (MICs) at the high end of the susceptible range (MIC 1–2 mcg/mL [0.7–1.4 μmol/L]).32–37 As a result, clinicians should know the specific MIC of vancomycin for MRSA. If the MIC is greater than or equal to 2 mcg/mL (1.4 μmol/L), switching to another anti-MRSA agent may be advisable, despite lack of FDA approval.38 Clinical effectiveness has been reported with linezolid and daptomycin in several small, nonrandomized studies against MRSA osteomyelitis (Table 81–3).39–41 However, some in vitro studies have shown reduced susceptibility to daptomycin in vancomycin-intermediate or -resistant S. aureusstrains;42–44 therefore, daptomycin susceptibility should be established prior to switching from vancomycin to daptomycin. Clinical trials being conducted with newer agents include daptomycin versus vancomycin for treatment of osteomyelitis related to prosthetic infections, and tigecycline versus ertapenem for treatment of diabetic foot infections.45 In children, clindamycin is an effective antibiotic against susceptible strains of MRSA.10,30 However, micro-biology laboratories must screen with a disk diffusion test (D-test) for inducible resistance via the macrolide-lincosamide-streptogramin (MLS) gene as clindamycin failures have been associated with infections caused by these isolates.10,30 Oral anti-MRSA agents often utilized for treatment of osteomyelitis include linezolid, trimethoprim-sulfamethoxazole and clindamycin (mostly in pediatrics).27,29,30
![]() The duration of treatment is typically 4 to 6 weeks for acute osteomyelitis.25 Chronic osteomyelitis also requires 4 to 6 weeks of therapy. In patients who have undergone surgical intervention, the total length of therapy should be counted after the last major surgical intervention.25 Therapy should be continued until the infection has resolved. Prolonged therapy may be necessary for certain populations such as patients with vascular insufficiency or patients with recalcitrant infections that do not respond to 4 to 6 weeks of therapy.2,4,7,26
The duration of treatment is typically 4 to 6 weeks for acute osteomyelitis.25 Chronic osteomyelitis also requires 4 to 6 weeks of therapy. In patients who have undergone surgical intervention, the total length of therapy should be counted after the last major surgical intervention.25 Therapy should be continued until the infection has resolved. Prolonged therapy may be necessary for certain populations such as patients with vascular insufficiency or patients with recalcitrant infections that do not respond to 4 to 6 weeks of therapy.2,4,7,26
Patient Encounter, Part 3: The Medical History, Physical Exam, and Diagnostic Tests
PMH: Diabetes mellitus, PVD, hypertension
SH: Tobacco smoker (2 packs per day for the past 30 years), social drinker (“3 cans of beer per day”), nonemployed with medical disability
Allergies: NKDA
Meds: Aspirin 81 mg orally once daily; atorvastatin 40 mg orally once daily; clopidogrel 75 mg orally once daily; lisinopril 10 mg orally once daily; insulin glargine 20 units every day at bedtime; insulin lispro 7 units before breakfast and lunch; insulin lispro 9 units before dinner
PE:
Gen: His general appearance is that of an obese male with tenderness and pain in the region of the nonhealing skin ulcer
Skin: Large, deep ulcer with purulent drainage on his left lower leg
VS: BP 145/87 mm Hg, P 80 bpm, RR 18/min, T 36.0°C (96.8°F), ht 5’6”(168 cm), wt 93 kg (205 lb)
Labs: WBC 13 × 103/mm3 (13 × 109/L), BUN 19 mg/dL (6.8 mmol/L), serum creatinine (Scr) 1.6 mg/dL (141 μmol/L), fasting blood glucose 156 mg/dL (8.7 mmol/L), ESR 80 mm/h, CRP 49 mg/dL (490 mg/L or 0.49 g/L)
Microbiology: Culture from wound swab: Enterococcus, coagulase-negative staphylococci, and P. aeruginosa; culture from bone biopsy during débridement: MRSA and Bacteroides fragilis
Against which organisms should the antimicrobial therapy be targeted?
Based on the information presented, create a care plan for this patient’s osteomyelitis. Your plan should include: (a) goals of therapy, (b) patient-specific detailed therapeutic plan, (c) nonpharmacologic interventions, and (d) follow-up plan to determine if outcomes have been achieved.
In addition to medical and surgical management, nonpharmacologic interventions that reduce risk factors for developing osteomyelitis should be communicated to the patient. Examples include smoking cessation, weight-control, exercise, and good nutrition. Additionally, a diabetic patient should be counseled regarding the necessity of controlled blood glucose, routine care and self-examination of lower extremities, and aggressive wound care.2
Table 81–3 Monitoring Considerations for Select Parenteral Antistaphylococcal Agents

Patient Encounter, Part 4
The patient received 4 weeks of IV antimicrobial therapy following débridement. Due to clinical improvement, the physician contacts you for a recommendation for an oral antibiotic to complete a total of 6 weeks of therapy.
What antimicrobial therapy would you recommend for this patient?
Evaluate the patient’s medication profile for drug-drug interactions.
Counsel the patient regarding this drug therapy.
OUTCOME EVALUATION
Therapeutic success is measured by the extent to which the care plan (a) resolves signs and symptoms, (b) eradicates the microorganism(s), (c) prevents relapses, and (d) prevents complications such as amputation. Patients should be evaluated for resolution of clinical signs and symptoms and normalization of laboratory tests (WBC, CRP, ESR, and cultures). Hospitalized patients should be examined daily. Improvement in clinical manifestations should be seen within 48 to 72 hours of initiation of IV antimicrobial therapy.2 In the outpatient setting, patients should be evaluated weekly during the initial 4 to 6 weeks of therapy. A reduction in CRP should be seen within 1 week of therapy and should be monitored weekly throughout therapy for a continued downward trend. ESR can also be monitored weekly although normalization will be slower than for CRP. Patients should also be monitored for antimicrobial tolerability and toxicity (see Table 81–3). If poor response is noted, the following should be evaluated: (a) patient compliance, (b) significant drug–drug or drug–food interactions, (c) appropriate dosage to achieve therapeutic concentrations, (d) development of antimicrobial resistance necessitating a change in the treatment regimen, (e) need for additional imaging studies, and (f) diagnostic reevaluation.2 Treatment is considered successful if all clinical signs and symptoms are resolved and all laboratory tests have returned to normal following 4 to 6 weeks of appropriate treatment. Due to high rates of relapse, patients should have medical follow-up for at least 1 year following resolution of symptoms.2 Patients should be evaluated at 3- to 6-month intervals for any clinical manifestations of recurring infection and continued normalization of laboratory tests. Follow-up imaging studies at 1 to 2 years may be useful in some patients to confirm therapeutic success.
Patient Care and Monitoring
1. ![]() Assess the patient’s symptoms and laboratory test results to determine if patient-directed therapy is appropriate.
Assess the patient’s symptoms and laboratory test results to determine if patient-directed therapy is appropriate.
2. Obtain a thorough history of prescription, nonprescription, and natural drug product use.
3. Educate the patient on lifestyle modifications that will reduce the risk of recurrent infections.
4. Develop a plan to assess the effectiveness of antimicrobial therapy.
5. Evaluate the patient for the presence of adverse drug reactions, drug allergies, and drug interactions (Table 81–3).
6. Stress the importance of adherence to the therapeutic regimen.
7. Provide patient education with regard to disease state and drug therapy. Explain the following:
• Causes of osteomyelitis
• Complications associated with osteomyelitis
• The drug, dose, duration, and route of administration of the patient’s antimicrobial regimen
• Optimal medication administration time for the patient’s lifestyle and other concurrent medications
• Available options for missed dose(s)
• Adverse effects that may occur
• Provide patient education monographs for antibiotic(s)
Abbreviations Introduced in This Chapter


Self-assessment questions and answers are available at http://www.mhpharmacotherapy.com/pp.html.
REFERENCES
1. Calhoun JH, Manring MM. Adult osteomyelitis. Infect Dis Clin N Am 2005;19:765–786.
2. Lipsky BA, Berendt AR, Deery HG, et al. Diagnosis and treatment of diabetic foot infections. Clin Infect Dis 2004;39:885–910.
3. Hartemann-Heurtier A, Senneville E. Diabetic foot osteomyelitis. Diabetes Metab 2008;34:87–95.
4. Lew DP, Waldvogel FA. Osteomyelitis. Lancet 2004;364:369–378.
5. Mader JT, Shirtliff M, Calhoun JH. Staging and staging application in osteomyelitis. Clin Infect Dis 1997;25:1303–1309.
6. Waldvogel FA, Medoff G, Swartz MN. Osteomyelitis: A review of clinical features, therapeutic considerations and unusual aspects. N Engl J Med 1970;282:198–206.
7. Lazzarini L, Mader JT, Calhoun JH. Osteomyelitis in long bones. J Bone Joint Surg Am 2004;86:2305–2318.
8. Mandracchia VJ, Sanders SM, Jaeger AJ, Nickles WA. Management of osteomyelitis. Clin Podiatr Med Surg 2004;21:335–351.
9. Vazquez M. Osteomyelitis in children. Curr Opin Pediatr 2002;14:112–115.
10. Kaplan SL. Osteomyelitis in children. Infect Dis Clin N Am 2005;19:787–797.
11. Gafur OA, Copley LA, Hollmig ST, et al. The impact of the current epidemiology of pediatric musculoskeletal infection on evaluation and treatment guidelines. J Pediatr Orthop 2008;28:777–785.
12. Saavedra-Lozano J, Mejías A, Ahmad N, et al. Changing trends in acute osteomyelitis in children: Impact of methicillin-resistant Staphylococcus aureus infections. J Pediatr Orthop 2008;28:569–575.
13. Klevens RM, Edwards JR, Tenover FC, et al. Changes in the epidemiology of methicillin-resistant Staphylococcus aureus in intensive care units in US hospitals, 1992–2003. Clin Infect Dis 2006 Feb 1;42:389–391.
14. Boucher HW, Corey GR. Epidemiology of methicillin-resistant Staphylococcus aureus. Clin Infect Dis 2008;46(suppl 5):S344–S349.
15. King MD, Humphrey BJ, Wang YF, Kourbatova EV, Ray SM, Blumberg HM. Emergence of community-acquired methicillin-resistant Staphylococcus aureus USA 300 clone as the predominant cause of skin and soft-tissue infections. Ann Intern Med 2006;144:309–317.
16. Moran GJ, Krishnadasan A, Gorwitz RJ, et al. Methicillin-resistant S. aureus infections among patients in the emergency department. N Eng J Med 2006;355:666–674.
17. Klevens RM, Morrison MA, Nadle J, et al. Invasive methicillin-resistant Staphylococcus aureus infection in the United States. JAMA 2007;298:1763–1771.
18. Game F, Jeffcoate W. MRSA and osteomyelitis of the foot in diabetes. Diabet Med 2004;21(suppl 4):16–19.
19. Naimi TS, LeDell KH, Como-Sabetti K, et al. Comparison of community-and health care-associated methicillin-resistant Staphylococcus aureus infection. JAMA 2003;290:2976–2984.
20. Hess LM, Jeter JM, Benham-Hutchins M, Alberts DS. Factors associated with osteonecrosis of the jaw among bisphosphonate users. Am J Med 2008;121:475–483.
21. Bertoldo F, Santini D, Lo Cascio V. Bisphosphates and osteomyelitis of the jaw: A pathogenic puzzle. Nat Clin Pract Oncol 2007;4(12):711–721.
22. Steer AC, Carapetis JR. Acute hematogenous osteomyelitis in children: Recognition and management. Pediatric Drugs 2004;6:333–346.
23. Gutierrez K. Bone and joint infections in children. Pediatr Clin North Am 2005;52:779–794.
24. Parsons B, Strauss E. Surgical management of chronic osteomyelitis. Am J Surg 2005;188:S57–S66.
25. Darley ES, MacGowan AP. Antibiotic treatment of gram-positive bone and joint infections. J Antimicrob Chemother 2004;53:928–935.
26. Lazzarini L, Lipsly BA, Mader JT. Antibiotic treatment of osteomyelitis; what have we learned from 30 years of clinical trials? Int J Infect Dis 2005;9:127–138.
27. Rayner CR, Baddour LM, Birmingham MC, et al. Linezolid in the treatment of osteomyelitis: Results of compassionate use experience. Infection 2004;32:8–14.
28. Tice AD, Hoaglund PA, Shoultz DA. Outcomes of osteomyelitis among patients treated with outpatient parenteral antimicrobial therapy. Am J Med 2003;114:723–728.
29. Shuford JA, Steckelberg JM. Role of oral antimicrobial therapy in the management of osteomyelitis. Curr Opin Infect Dis 2003;16:515–519.
30. Moellering Jr RC. Current treatment options for community-acquired methicillin-resistant Staphylococcus aureus infection. Clin Infect Dis 2008;46:1032–1037.
31. Toma MB, Smith KM, Martin CA, Rapp RP. Pharmacokinetic considerations in the treatment of methicillin-resistant Staphylococcus aureus osteomyelitis. Orthopedics 2006;29(6):497–501.
32. Rybak M, Lomaestro B, Rotschafer JC, et al. Therapeutic monitoring of vancomycin in adult patients: A consensus review of the American Society of Health-System Pharmacists, the Infectious Diseases Society of America, and the Society of Infectious Diseases Pharmacists. Am J Health-Syst Pharm 2009;66:82–98.
33. Jones RN. Microbiological features of vancomycin in the 21st century: Minimum inhibitory concentration creep, bactericidal/static activity, and applied breakpoints to predict clinical outcomes or detect resistant strains. Clin Infect Dis 2006;42(Suppl 1):S13–S24.
34. Sakoulas G, Moise-Broder PA, Schentag J, et al. Relationship of MIC and bactericidal activity to efficacy of vancomycin for treatment of methicillin-resistant Staphylococcus aureus bacteremia. J Clin Microbiol 2004;42:2398–2402.
35. Moise PA, Sakoulas G, Forrest A et al. Vancomycin in vitro bactericidal activity and its relationship to efficacy in clearance of methicillin-resistant Staphylococcus aureus bacteremia. Antimicrob Agents Chemother 2007;51:2582–2588.
36. Hidayat LK, Hsu DI, Quist R, et al. High-dose vancomycin therapy for methicillin-resistant Staphylococcus aureus infections: Efficacy and toxicity. Arch Intern Med 2006;166:2138–2144.
37. Steinkraus G, White R, Friedrich L. Vancomycin MIC creep in non-vancomycin-intermediate Staphylococcus aureus (VISA), vancomycin-susceptible clinical methicillin-resistant S. aureus (MRSA) blood isolates from 2001–2005. J Antimicrob Chemother 2007;60:788–794.
38. Awad SS, Elhabash SI, Lee L, Farrow B, Berger DH. Increasing incidence of methicillin-resistant Staphylococcus skin and soft-tissue infections: Reconsideration of empiric antimicrobial therapy. Am J Surg 2007;194:606–610.
39. Lamp KC, Friedrich LV, Mendez-Vigo L, Russo R. Clinical experience with daptomycin for the treatment of osteomyelitis. Am J Med 2007;120: S13–S20.
40. Falagas ME, Giannopoulou KP, Ntziora F, Papagelopoulous PJ. Daptomycin for treatment of patients with bone and joint infections: A systemic review of the clinical evidence. Int J Antimicrob Agents 2007;30:202–209.
41. Falagas ME, Siempos II, Papagelopoulos P, Vardakas KZ. Linezolid for the treatment of adults with bone and joint infections. Int J Antimicrob Agents 2007;29:233–239.
42. Sakoulas G, Alder J, Thauvin-Eliopoulos C, et al. Induction of daptomycin heterogeneous susceptibility in Staphylococcus aureus by exposure to vancomycin. Antimicrob Agents Chemother 2006;50:1581–1585.
43. Pfaller MA, Sader HS, Jones RN. Evaluation of the in vitro activity of daptomycin against 19,615 clincial isolates of Gram-positive cocci collected in North American hospitals (2002–2005). Diagn Microbiol Infect 2007;57:459–465.
44. Clinicaltrials.gov. A Service of National Institutes of Health. Osteomyelitis.www.clinicaltrials.gov/ct2/results?term=osteomyelitis.
44. Graber CJ, Wong MK, Carleton HA, Perdreau-Remington F, Haller BL, Chamber HF. Intermediate vancomycin susceptibility in a community-associated MRSA clone. Emerg Infect Dis 2007;13:491–493.
45. Lexi-Drugs Online. www.crlonline.com.