John Heesakkers1 , Frank Van der Aa2 and Tufan Tarcan3
(1)
Department of Urology, Radboud University Medical Center, Nijmegen, The Netherlands
(2)
Department of Urology, University Hospitals, Leuven, Leuven, Belgium
(3)
Department of Urology, Marmara University School of Medicine, Istanbul, Turkey
John Heesakkers (Corresponding author)
Email: john.heesakkers@radboudumc.nl
Frank Van der Aa
Email: frank.vanderaa@uzleuven.be
Tufan Tarcan (Corresponding author)
Email: bilgi@tufantarcan.com
Stress Urinary Incontinence In Women
John Heesakkers4 , Frank Van der Aa5 and Tufan Tarcan6
(4)
Radboud University Medical Center, Nijmegen, The Netherlands
(5)
University Hospitals, Leuven, Leuven, Belgium
(6)
Marmara University School of Medicine, Istanbul, Turkey
John Heesakkers
Email: J.Heesakkers@uro.umcn.nl
Frank Van der Aa
Email: frank.vanderaa@uzleuven.be
Background
Stress urinary incontinence (SUI) is defined as the loss of urine in response to sudden increase in intra-abdominal pressure (‘stress’) in the absence of a detrusor contraction. It occurs primarily in women who have had vaginal deliveries. The prevalence of incontinence is about 13 %, and half of the women have complaints of SUI. The lifelong risk of women to undergo surgery for SUI or pelvic organ prolapse is about 10 %.
Stress urinary incontinence (SUI) is defined as the loss of urine in response to sudden increase in intra-abdominal pressure (‘stress’) in the absence of a detrusor contraction. It occurs primarily in women after vaginal delivery.
The diagnosis is based on patient history and physical examination. The severity can be assessed with pad testing. Urodynamic investigation has been the cornerstone for many years, but the importance is questioned lately. Well performed RCT’s demonstrated that in clear cut cases the value of urodynamic investigation is limited. Therefore in most guidelines urodynamic investigation is only recommended in selected, non- clear cut cases.
Treatment options re conservative treatment with life style interventions, physical therapy and pelvic floor muscle therapy. The classical options for surgical intervention are bulking agents, bladderneck colposuspensions and pubovaginal slings. Since the end of the 20st century retropubic and transobturator tapes were introduced. Single incision slings have been developed since the beginning of this century.
Special attention should be paid to obese and elderly patients, patients with mixed incontinence and secondary procedures. Intraoperative, early and long term postoperative complications occur and should be dealt with carefully.
Clinical Practice
Diagnosis
After having a patient with the complaint of SUI, the diagnosis of SUI should be made. This consists of patient history, physical examination and additional tests like voiding diaries, pad testing and urodynamic investigation. The assessment should be based on existing guidelines.
Patient History
Patient history should focus on the confirmation of SUI, the onset and duration and the severity of incontinence. Since SUI is defined as the loss of urine in response to sudden increase in intra-abdominal pressure, one should focus on those individual circumstances that increase the intra-abdominal pressure. For one patient, this means playing volleyball, for the other, gardening or playing golf. The onset can also confirm the type of incontinence. If the complaints started directly after delivery, it is very likely that the incontinence type is SUI. The severity is indicative of the contribution of the ISD part to the SUI. A higher severity meaning severe urine loss during intra-abdominal pressure rise or also urinary loss under relatively mild circumstances implies a higher grade of ISD. This can help choosing the right therapy.
Other lower urinary tract symptoms and accompanying comorbidities should also be asked for.
Physical Examination
In this era of evidence-based medicine, it is fashionable to only perform those diagnostic and treatment activities that have proven value and are recommended by guideline committee. There is no evidence for the necessity of clinical examination, but the agreement is that clinical examination is essential. The physical examination should include the assessment of genital prolapse and oestrogen status of the vaginal mucosa. Pelvic organ prolapsed should be assessed with an objective measure like the POPQ system. The examination of the vagina should focus on the amount of mobility of the bladder neck and urethra during a provoked Valsalva and cough manoeuvre. If the bladder neck and urethra is fixed, e.g. after a colposuspension or after pelvic radiotherapy, the choice of surgical procedure is more difficult and the outcome is lower than with a so-called hypermobile urethra. In patients with a hypermobile urethra, which is the majority of female patients with SUI, any kind of tape will have good results and patients’ expectations can easier be met. It is also important to observe the loss of urine during coughing or Valsalva to confirm patient’s complaint of SUI. This can be done in the supine position, but if no urine loss is observed in this position, it should be done in the standing position. If urinary loss is observed in with intra-abdominal pressure increase, then the sign of SUI is confirmed. The combination with urodynamic SUI gives the condition of SUI. Contraction, symmetry and coordination of the pelvic floor muscles are assessed by digital vaginal examination.
Assessing Urethral Hypermobility
Classically the amount of movement of the urethra during coughing or Valsalva is assessed by the Q-tip test. Here, a Q-tip swab is positioned in the urethra during physical examination in the supine position. The patient is asked to squeeze or cough, and the deviating angle from the resting position is determined. If the angle is more than 30°, urethral hypermobility is present. In clinical practice, the Q-tip test is not used frequently. Mostly eyeball diagnosis is used in the same way as doing the Q-tip test but without the swab. If one observes a freely rotating descent of the urethra around the symphysis pubis, the conclusion can be drawn that urethral (hyper)mobility is present and that a tension-free application of a midurethral tape can be done successfully. If at the same time with coughing or Valsalva urine loss is observed, the sign SUI is established. Of course, one has to be sure that the bladder is not empty during the provocative procedure.
Voiding Diaries
It is possible to reliably quantify the amount of urinary loss by noting down events of the voiding cycle. The best way to do this is to use the so-called voiding diaries. During optimally 3 days, patients note down how much they drink every time and when. They note how much they void per void and also when. They have to mark the time they go to bed and when they wake up. What is very important is that the moment is noted when urine loss occurs and how much this is. This should be noted with scores like 1= drop, 2 = dash and 3 = complete void. The occurrence of urge and/or urgency can also be noted. With these data, it is possible for the experienced caregiver to distinguish SUI from OAB wet. Drops of urine loss during the day and not at night and without urgency occurring at the same moment most likely SUI. If the number of voids is normal, meaning less than eight times per day, and the functional capacity is normal, this strengthens the likelihood of SUI.
Diaries can also be used to monitor treatment response and are widely used in clinical trials as a semi-objective measure of treatment outcome.
Pad Testing
Quantification of the amount of urine loss allows objectivation of the incontinence. From the available options like the Stamey grading, the standardised 1-h pad test and the 24-h pad test, the last one is the most reliable. The standardised 1-h pad test has limited validity and a high false-negative rate. Moreover, it often does not reflect the situation in which the patient experiences urine loss. The 24-h pad test is sensitive and has a low false-negative rate. It is not needed to extend the testing period, and it is representative for patient’s daily situation. The only requirement is to have a scale that can measure grams.
Urodynamic Investigation
Urodynamic investigation (UDI) is one of the cornerstones for diagnosing SUI. The urodynamic principles have been described by the International Continence Society in ‘The Good Urodynamic Practice Guidelines’. This means that SUI can be demonstrated and other abnormalities like detrusor overactivity and incomplete bladder emptying diagnosed. UDI is therefore used to demonstrate SUI and to predict postoperative problems like OAB or post-void residual. For a long time, urodynamics have been used to distinguish intrinsic sphincter deficiency (ISD) from urethral hypermobility. This can be done by urethral pressure measurement (UPP) or by abdominal leak point pressures. The indicators for ISD with UPP are a low maximal urethral closing pressure (MUCP). The cut-off value is put at 20 cmH2O. If the MUCP is below 20 cmH2O, ISD exists, above 20 cmH2O, this is not clear. Urethral hypermobility is diagnosed with pressure transmission ratios (PTR). If the PTR is lower than approximately 80 %, urethral hypermobility can be present.
Abdominal leak point pressure (ALPP) determines the lowest intravesical pressure at which urine loss occurs as observed by the urodynamicist or by the flowmeter. If the ALPP is low (<40 cmH2O), this indicates ISD. If it is high (>80 cmH2O), no ISD should be present. UPP and ALPP have been used to make therapy choices in older times. If urethral hypermobility and no ISD was present, a colposuspension was the choice. If a lot of ISD was present, a pubovaginal sling was the treatment of choice. As well UPP and ALPP do not correlate well with clinical practice, and their role in diagnosing ISD or choosing the type of surgery is limited.
Since the introduction of the polypropylene midurethral slings, things have changed. The assessment of urethral hypermobility by physical examination is the most important diagnostic tool to choose therapy or to predict the outcome of tape surgery. If substantial urethral hypermobility is present, any tape (retropubic tape, transobturator tape or single incision slings) has a good outcome with respect to the cure of SUI. If urethral hypermobility is not or hardly present, it is more difficult to cure SUI with a tape. Since the retropubic tapes give more support to the urethra and are easier to comply in a compressive manner, they give better results and they are the tapes of choice. This is especially so in redo surgery where the movement of the urethra is mostly limited.
Recently, the role of urodynamics has been questioned and this debate is still ongoing. It appears that in women with primary SUI complaints, with urethral hypermobility and without complicating factors, a urodynamic examination might not be mandatory. Certainly urodynamics are not needed when only conservative therapy is being considered. When surgery is being planned, use urodynamics if the findings might change the choice of invasive options. There is only limited evidence that performing urodynamics can predict the outcome of the treatment. Urethral pressure measurements such as MUCP and ALPP have not been shown to be able to grade the severity of the incontinence nor to be able to predict the outcome of treatment.
Conservative Treatment of SUI
Although SUI can be worsened by underlying diseases such as diabetes, COPD, neurological disorders, cardiac failure, etc., there is little evidence that correcting these underlying conditions will improve the incontinence significantly.
Medication reviews should be taken in all patients with urinary incontinence since medication. Alpha-blockers, drugs acting on the CNS, might have an impact on the urinary continence. Diuretics do not seem to have a significant impact on incontinence, while systemic oestrogens will increase the risk of developing incontinence or can worsen pre-existing incontinence.
Lifestyle Interventions
Despite the fact that there is an association between constipation and urinary incontinence, there is limited evidence that treatment of constipation will improve SUI.
In the elderly, however, multimodal behavioural therapy including the treatment of constipation showed to improve the urinary continence.
Often lifestyle interventions are promoted to improve continence: weight reduction, smoking cessation, increased level of physical activity and avoiding caffeine-containing drinks, etc.
Many patients will restrict their fluid intake in an attempt to reduce the amount of urine loss. Patients should be advised that the 24-h diuresis should be between 1500 and 3000 cc and that reduction of fluid intake has not been shown to improve the incontinence significantly. A micturition diary is an indispensable tool in the diagnosis and the management of fluid intake problems, and patients should be taught to manage their fluid intake and diuresis using this tool.
Caffeinated drinks and fizzy drinks are often blamed for aggravating incontinence, but there seems to be little evidence that reduction of caffeine-containing drinks will reduce urinary incontinence. However, reducing caffeine might improve symptoms of urgency and frequency.
Obesity certainly is a risk factor for UI. The prevalence SUI increases proportionately with rising body mass index. A significant proportion of patients who undergo surgery for incontinence are overweight or obese.
Weight reduction of 5 % or more will improve UI. Even bariatric surgery in obese women can significantly improve or cure incontinence. Therefore, obese patients should always be stimulated to lose weight first, before undergoing surgical treatment for SUI.
Smoking by itself is a health hazard and every health professional should stimulate smokers to quit. In relation to continence, the role of smoking is less clear. The cessation of smoking has no clear effect on UI except maybe for those with severe incontinence.
Behavioural Therapy and Bladder Training
While behavioural therapy prompted voiding and bladder training might have a role in the treatment of urgency/frequency and urgency incontinence, their role in the treatment of SUI is limited. Only in the elderly – mostly institutionalised and frail – these forms of continence management can be useful.
Pelvic Floor Muscle Therapies
The pelvic floor musculature weakens with increasing age. At the same time, the proprioception of the pelvic floor deteriorates. This process is negatively influenced by vaginal deliveries, pelvic trauma and menopause. The weakened muscles can however be retrained to a certain level. This will improve the position and support of the urethra and will increase the closure pressure.
Patients are taught to identify their pelvic muscles, usually by vaginal examination by a specialised physiotherapist or trained nurse. Once they learn which muscles to contract, the strength of these muscles is enhanced by several types of muscle exercises. There is a large variability in techniques, methods of delivery, etc. of these exercises. In some cases, just some paper instructions are given to women; in others, women are following individual or group sessions with trained professionals. Typically all these exercises have to be repeated several times a day for several weeks.
The exercise regimes can be enhanced by the use of electrical stimulation, biofeedback, etc.
The effect of PFMT in women with SUI does not seem to decrease with increased age: in trials with older women with SUI, it appeared that both primary and secondary outcome measures were comparable to those in trials focused on younger women.
The literature about pelvic floor muscle therapy (PFMT) is difficult to interpret because of poor design of the trails, the use of different techniques that are often not well described and the use of a variety of outcome measures.
However, systematic reviews and meta-analysis were able to show that PFMT is effective for curing and improving UI.
A Cochrane review comparing different approaches to delivery of PFMT concluded that increased intensity of delivery of the therapy improves response and that there is no consistent difference between group therapy and individualised treatment sessions. Nor was there a significant difference between techniques.
Long-term adherence to PFMT is low and 50 % will end up having surgery anyway.
Women with SUI or mixed incontinence should be offered supervised intensive PFMT for at least 3 months. The PFMT program should be as intensive as possible. Biofeedback can be helpful to motivate the patient. Electrical stimulation (skin, vaginal or anal) alone is not helpful and should be combined with a multimodal exercise program.
Magnetic extracorporeal stimulation has not been proven to improve or cure SUI.
Medical Treatment
For SUI only duloxetine is being considered at this moment. Several studies have shown a positive effect of duloxetine 2 × 40 mg/day on the leakage of urine and on the quality of life. Duloxetine however does not cure SUI, and patients must be willing to take this drug for a long period. Usually the drug is used for patients on waiting lists or for those who want to delay surgery.
Duloxetine has significant gastrointestinal and CNS side effects. There is a high incidence of nausea, vomiting, dry mouth, constipation, insomnia, somnolence, fatigue and dizziness. These lead to a high rate of treatment discontinuation: even in trial conditions, up to 40 % of the patients withdrew, while in long-term studies, up to 90 % stopped the drug.
Evidence-Based EAU Guidelines on Incontinence 2014
Surgery for SUI should as much be based on evidence as possible. This means that the results of the described techniques have been published in order to appreciate its value. This does not mean that the described operations are prefect but that the surgeon is able to consult the patients about expected successes and risks of the procedure and that the patient can make a choice based on facts. These facts about the surgical procedures as regarded by the EAU in 2014 are listed hereunder.
Uncomplicated SUI Surgery in Women
|
Evidence summary |
LE |
|
Anterior colporrhaphy has lower rates of cure for UI especially in the longer term. |
1a |
|
Open colposuspension and autologous fascial sling are similarly effective for cure of SUI in women. |
1b |
|
Laparoscopic colposuspension has similar efficacy to open colposuspension for cure of SUI and a similar risk of voiding difficulty or de novo urgency. |
1a |
|
Laparoscopic colposuspension has a lower risk of other complications and shorter hospital stay than open colposuspension. |
1a |
|
Autologous fascial sling has a higher risk of operative complications than open colposuspension, particularly voiding dysfunction and postoperative UTI. |
1b |
|
Evidence summary |
LE |
|
Compared to colposuspension, the retropubic insertion of a midurethral synthetic sling gives equivalent patient-reported cure of SUI and superior clinician-reported cure of SUI at 12 months. |
1a |
|
Compared to colposuspension, the transobturator insertion of a midurethral synthetic sling gives equivalent patient-reported and clinician-reported cure of SUI at 12 months. |
2 |
|
Insertion of a midurethral synthetic sling by the transobturator route gives equivalent patient-reported and clinician-reported cure rates at 12 months compared to retropubic insertion. |
1a |
|
The skin-to-vagina direction of retropubic insertion of midurethral sling is less effective than a vagina-to-skin direction. |
1a |
|
Midurethral sling insertion is associated with a lower rate of a new symptom of urgency, and voiding dysfunction, compared to colposuspension. |
1a |
|
The retropubic route of insertion is associated with a higher intraoperative risk of bladder perforation and a higher rate of voiding dysfunction than the transobturator route. |
1a |
|
The transobturator route of insertion is associated with a higher risk of chronic perineal pain at 12 months than the retropubic route. |
1a |
|
The skin-to-vagina direction of both retropubic and transobturator insertion is associated with a higher risk of postoperative voiding dysfunction. |
1b |
|
Evidence summary |
LE |
|
|
Single-incision midurethral slings are effective in curing SUI in women in the short term. |
1b |
|
|
Operation times for insertion of single-incision midurethral slings are shorter than for standard retropubic slings. |
1b |
|
|
Blood loss and immediate postoperative pain are lower for insertion of single-incision slings compared with standard midurethral slings. |
1b |
|
|
Single-incision slings are less effective than other midurethral slings at medium-term follow-up*. |
1b |
|
|
There is no evidence that other adverse outcomes from surgery are more or less likely with single-incision slings than with standard midurethral slings. |
1b |
|
|
Evidence summary |
LE |
|
|
Adjustable midurethral synthetic sling device may be effective for cure or improvement of SUI in women |
3 |
|
|
There is no evidence that adjustable slings are superior to standard midurethral slings. |
4 |
|
|
Evidence summary |
LE |
|
Periurethral injection of bulking agent may provide short-term improvement in symptoms (3 months), but not cure, in women with SUI. |
2a |
|
Repeat injections to achieve therapeutic effect are very common. |
2a |
|
Bulking agents are less effective than colposuspension or autologous sling for cure of SUI. |
2a |
|
Adverse effect rates are lower compared to open surgery. |
2a |
|
There is no evidence that one type of bulking agent is better than another type. |
1b |
|
Periurethral route of injection may be associated with a higher risk of urinary retention compared to transurethral route. |
2b |
|
Recommendations for surgery for uncomplicated stress urinary incontinence in women |
GR |
|
Offer the midurethral sling to women with uncomplicated stress urinary incontinence as the preferred surgical intervention whenever available. |
A |
|
Offer colposuspension (open or laparoscopic) or autologous fascial sling to women with stress urinary incontinence if midurethral sling cannot be considered. |
A |
|
Warn women who are being offered a retropubic insertion synthetic sling about the relatively higher risk of peri-operative complications compared to transobturator insertion. |
A |
|
Warn women who are being offered transobturator insertion of midurethral sling about the higher risk of pain and dyspareunia in the longer term. |
A |
|
Warn women undergoing autologous fascial sling that there is a high risk of voiding difficulty and the need to perform clean intermittent self-catheterisation; ensure they are willing and able to do so. |
A |
|
Do a cystoscopy as part of retropubic insertion of a midurethral sling, or if difficulty is encountered during transobturator sling insertion, or if there is a significant cystocoele. |
C |
|
Women being offered a single-incision sling device for which an evidence base exists, should be warned that short-term efficacy is inferior to standard midurethral slings and that long-term efficacy remains uncertain. |
C |
|
Only offer single-incision sling devices, for which there is no level 1 evidence base, as part of a structured research programme. |
A |
|
Only offer adjustable midurethral sling as a primary surgical treatment for stress urinary incontinence as part of a structured research programme. |
C |
|
Do not offer bulking agents to women who are seeking a permanent cure for stress urinary incontinence. |
A |
Complicated SUI Surgery in Women
|
Evidence summary |
LE |
|
The risk of treatment failure from surgery for SUI is higher in women who have had prior surgery for incontinence or prolapse. |
1b |
|
Open colposuspension and autologous fascial sling appear to be as effective for first-time repeat surgery as for primary surgery. |
1b |
|
The midurethral sling is less effective as a second-line procedure than for primary surgery. |
2 |
|
Recommendations for surgery for complicated stress urinary incontinence in women |
GR |
|
The choice of surgery for recurrent stress urinary incontinence should be based on careful evaluation of the individual patient. |
C |
|
Women should be warned that the outcome of second-line surgical procedures is likely to be inferior to first-line treatment, both in terms of reduced benefit and increased risk of harm. |
C |
|
Offer implantation of AUS or ACT as an option for women with complicated stress urinary incontinence if they are available and appropriate monitoring of outcome is in place. |
C |
|
Warn women receiving AUS or ACT that there is a high risk of mechanical failure or a need for explantation. |
C |
Surgical Treatment
Bulking Agents
The application of bulking agents has been performed for many years as the treatment for SUI and especially ISD. Various materials have been used like autologous fat, silicone particles, collagen or polyacrylamide hydrogel. The working mechanism is thought to be coaptation of the urethra. Since bulking agents are mostly applied in cases of ISD, they should enforce the urethra in order to compensate for the intrinsic sphincteric defect. The concept is not to keep the urethra in the proper position during intra-abdominal pressure increase like tapes, but to reinforce the sphincter itself by injecting material somewhere in the sphincteric complex.
Indications
Bulking agents are applied in cases where no urethral mobility is present, e.g. after an earlier procedure for SUI. Since bulk can be applied locally, it is also frequently applied in those patients that can’t stand extensive operations. Other indications are minor to mild SUI and poor bladder emptying and those who can’t stand polypropylene tapes.
Technique
It is not known what the best position and the best way to apply bulk is. Bulk can be injected transurethrally, paraurethrally, at the bladder neck and midurethrally. Injection of bulk can be done under cystoscopic vision or blindly with standardised application device. If the bulk is injected blindly, it is possible to standardise the procedure and to combine results from various surgeons. The cystoscopic procedure allows better visual control and is preferred by many experienced surgeons. Normally, the bulk is injected at three to four locations opposite each other in order to be capable to coapt the urethral lumen.
Points of Interest
There are some important factors that have to be taken into account when injecting bulk:
· When the injection is done under local anaesthesia, enough time should be allowed between applying the local anaesthesia and the injection of bulk.
· The anatomy of innervation of the anterior vaginal wall and the urethra should be known in order to inject regionally effectively and not to close to the urethra in order to avoid extra oedema because of the local anaesthesia.
· The bulk should be injected very slowly in order to inject the bulk between the tissue layers of the urethra without rupturing them.
· Backflow of the material should be avoided by not withdrawing the needle too quickly.
· After having finished the procedure, the bladder should preferably not be emptied or emptied with a small calibre catheter to avoid the bulking agent to be forced into the external layers of the urethra.
Bladder Neck Colposuspension
Background
Bladder neck colposuspension was first described by Burch in 1961. After having firstly described the attachment of the paravaginal fascia to the arcus tendineus, he later improved his technique by attaching the paravaginal fascia to Cooper’s ligament, which allowed for more firm fixation points and less chance of infection as compared to the Marshall-Marchetti-Krantz (MMK) procedure, which is another type of retropubic colposuspension. The goal of these procedures is to suspend and stabilise the urethra so that the bladder neck and proximal urethra are replaced intra-abdominally. This anatomic placement allows normal pressure transmission during periods of increased intra-abdominal pressure restoring continence. Although its durability has been proven, the open repair is less commonly performed due to the advent of less invasive procedures. Laparoscopic surgeons have also demonstrated that the repair can be performed via a laparoscopic approach, decreasing morbidity, while still providing a satisfactory outcome.
Technique
A catheter is inserted into the bladder. The incision is either a Pfannenstiel or lower midline abdominal incision to access the Retzius space. After incision of the rectus fascia, the rectus muscles are separated in the midline. Downward pressure behind and lateral to the symphysis pubis gives access to the lateral walls of the pelvis and to the endopelvic fascia. The peritoneal reflection is swept of the bladder. After dissection of the retropubic space, the bladder neck, the anterior vaginal wall and urethra are exposed.
The bladder neck is identified with palpation of the balloon of the catheter. The pubocervical fascia may be identified by sweeping away the overlying fat. To identify the anterior vaginal wall and distinguish this from the urethra, a gloved and protected finger can be inserted intravaginally in the vagina to tent the vagina laterally.
The bladder should be displaced medially and cranially away from the site of suture placement using a small swab. Haemostasis at this point of the procedure may be necessary. It is important to identify the white-coloured pubocervical fascia. This is the place where the suspending sutures must be placed.
Two to four sutures are placed on each side of the bladder neck through the pubocervical fascia. It is important to take good bites of overlying fascia and anterior vaginal wall. If nonabsorbable sutures are used, it is important not to incorporate the vaginal mucosa. If (slow) absorbable sutures are used, this is less critical. Double bites of tissue to lessen the risk of the suture pulling through can be applied.
The most distal suture is placed just distally or at the level of the bladder neck and approximately 2 cm laterally. The sutures are placed in the pubocervical fascia and anterior vaginal wall proximal to the bladder neck about 1 cm apart. The sutures are placed into the Cooper’s ligament at the same level. Care must be taken to leave a suture bridge and do not apply excessive tension. Sometimes the most distal sutures need to be placed in the periosteum and in the fibrous insertion of the rectus muscle instead of in Cooper’s ligament. Elevation of the anterior vaginal wall with a gloved and protected finger in the vagina helps with tying the sutures without tension. The goal is to approximate the anterior vaginal wall to the lateral wall of the pelvis. Tension may lead to pulling through of the sutures.
Points of Interest
· Avoid entry into the peritoneal cavity.
· Identify the white-coloured pubocervical fascia to place the suspending sutures properly.
· When using nonabsorbable sutures, care must be taken not to perforate the vaginal mucosa.
· Leave a suture bridge and do not apply excessive tension in order to overcorrect the descended bladder neck.
· Cystoscopy can be performed to rule out injury to the bladder.
Pubovaginal Slings
Background
Many types of pubovaginal slings have been described more than 100 years ago. Only after numerous improvements and reintroduction by McGuire in 1978, the pubovaginal sling became more popular. Traditionally the sling has been used only when other incontinence procedures, like especially the Burch colposuspension, have failed.
Indication
The most common indications for a pubovaginal sling are intrinsic sphincter deficiency with or without urethral hypermobility and a prior failed incontinence procedure. Also certain patients with SUI due to urethral hypermobility may be better served with a sling procedure, because of the long-term success and durability of the pubovaginal sling.
Technique
It is beneficial to teach the patient clean intermittent catheterisation before surgery because incomplete emptying is common for a short while postoperatively. One dose of intravenous antibiotics can be given preoperatively. General or regional anaesthesia can both be used.
The procedure is performed in the low lithotomy position. The legs should only be moderately flexed at the hips to allow simultaneous exposure to the vagina and the lower abdomen. A Foley catheter is placed and the balloon inflated to allow palpation of the bladder neck and urethra. A vaginal speculum is placed ad good view on the anterior vaginal wall should be possible. A pfannenstiel incision is made. The rectus fascia is cleared, and a fascial area is selected that looks strong and is scar free. A fascial sling of about 10 cm length and 2 cm width is obtained by incising parallel to the fibres. This can be done lengthwise or widthwise. It is possible to make a so-called sling-on-a-string in this way. If you want to leave one end fixed to the body, you should leave the part of the sling close to the pubic bone intact and cut the other end of the sling in order to use that part to wrap it around the urethra. The sutures on the sling may be placed before or after transection. Make a choice for the type and size of suture based on personal preference. The suture should be strong and durable like 1-0 or 2-0 vicryl absorbable suture. The sutures are placed perpendicular to the direction of the fibres at 1.0 cm from the ends incorporating all of the fibres in the bites.
The vaginal part starts by placing an Allis clamp midway between the bladder neck and the urethral meatus. Hydrodisection by injecting saline over the urethra can be used to facilitate the dissection. A midline incision of 3 cm is made over the proximal urethra. The vaginal dissection is performed in the proper plane, superficial to the white-coloured periurethral fascia. The dissection is continued to the lateral area. Afterwards the retropubic space is entered at bladder neck level, inferiorly to the ischium. The endopelvic fascia is perforated with curved Metzenbaum scissors. Blunt finger dissection should not be used in order not to cause bladder damage.
Through the abdominal incision a lateral defect is created at the level where the rectus muscle inserts onto the symphysis. With gentle dissection, easy access is obtained to the retropubic space. This can be done by finger dissection or with scissors over the posterior pubic bone. This can be helpful after prior procedures. After completion no tissue should be palpable between fingers inserted from above and from the vaginal incision. Make sure that the bladder is not between the pubic bone and the dissection plane. A similar procedure is performed on the opposite side.
After having developed the plane left and right, two clamps can be passed on both sides. Cystoscopy can be performed to look for urethral or bladder damage. The sling sutures are pulled into the abdominal incision and the sling is placed under the proximal urethra. It is critical that a good portion of the sling extend into the retropubic space to allow good fixation. Some absorbable sutures are placed through the edge of the sling and superficially through the periurethral fascia to secure its place. The sling sutures are passed through the rectus fascia. The vagina is closed. The sling sutures are pulled up and tied over the rectus. A clamp can be used to hold tension on the untied sutures until the appropriate tension is obtained. The appropriate tension is when one or two fingers can easily slide under the suture knot. In the situation where the patient does not void and permanent urinary retention is desired, increased tension can be applied. The skin is closed and a vaginal pack can be placed.
Points of Interest
· Teach patients how to perform clean intermittent catheterisation before surgery.
· Avoid extensive retropubic dissection when developing a plane for the sling.
· Take care not to incorporate the bladder or urethra into the retropubic dissection route for sling placement.
· Perform urethrocystoscopy after dissecting the retropubic plane to rule out bladder injury.
· Allow one to two fingers to be put between rectus fascia and the sling sutures to avoid a too tight sling placement.
Retropubic Tapes
Some developments have paved the way to the introduction of the revolutionary retropubic tension-free vaginal tape (TVT) in 1996. The first was that it was recognised to restore the function of the urethral closing mechanism instead of correcting anatomy. The second was the importance of the midurethra in the continence mechanism. This was supported by the discovery of the pubourethral ligaments, the awareness that the maximum urethral closing pressure is located at the midurethral level and that in continent women the urethra closes in the middle part. Therefore, the new concept of the integral theory by Petros and Ulmsten stated that SUI was caused by a lacking midurethral closing mechanism. This could be caused by failure of the pubourethral ligaments, lacking support of the anterior vaginal wall to the midurethra or by defective functioning of the pubococcygeal muscles that support the adjacent part of the urethra. Since connective tissue is regarded very important in the integral theory, surgical treatments are aiming at restoring these connective tissue structures (ligaments).
Success rates of retropubic slings in prospective studies show 80 % and higher success rates after 12 months.
Retropubic tape is the technique with the most longstanding experience of all midurethral tape procedures. Follow-up data up to 17 years have been published, showing that the technique has a long time performance.
Technique
The TVT procedure is a tape procedure that uses a polypropylene mesh with a pore size of 75–150 micrometres that allows ingrowth of connective tissue cells and cells of the immune system. This prevents colonisation of the tape with bacteria. The tape size is 11 mm by 40 cm. It is covered with a plastic sheath that allows easy passage retropubically and protects the tape for potential contamination with bacteria.
Most surgeons will give prophylactic antibiotics at the beginning of the procedure.
The tape originally was inserted under local anaesthesia that was injected in the operating region. Nowadays most procedures are done under regional anaesthesia, but it can be performed under general anaesthesia as well. The advantage of local anaesthesia is that it is possible to test the effect of the TVT during the procedure with Valsalva manoeuvres of the patient in order to position it with the appropriate tension.
The patient is positioned in the lithotomy position with hip flexion. After inserting a catheter, an incision is made at the anterior vaginal wall of approximately 2 cm length starting 0.5–1 cm from the urethral meatus. After having made the midline incision, the space between the anterior vaginal wall and the pubocervical fascia is prepared bluntly with Metzenbaum scissors. It is very important to be in the right plane. This makes dissection easy. If the preparation of the space is too deep, the urethral sphincter complex is unnecessarily damaged; if the plane is too superficial, healing problems and subsequently erosion may occur. The paraurethral space is prepared up to the inferior rim of the pubic bone. Afterwards two suprapubic skin incisions are made. The incisions are approximately 1 cm long, starting 3 cm from the midline and 2.5–3 cm cranially of the pubic bone. Then the tape is mounted to the placement needle.
The original technique is to apply the tape tension free. This means that urethral hypermobility is blocked during intra-abdominal pressure increase. The second effect is that at the place of the tape, kinking of the urethra occurs that enhances the closing effect. However, after gaining experience with the tape, experienced surgeons make in some patients the choice to put some tension on the tape especially when there is suspicion of ISD. This can be based on urodynamic testing with ALPP or UPP. Another indication for a non-tension-free tape is SUI without urethral mobility, e.g. after a previous colposuspension. If necessary, the bladder can be filled with 300 ml of saline and Valsalva manoeuvres performed to check for residual SUI.
Points of Interest
· Empty the bladder at the start of the procedure.
· After having made the midline incision, get into the right plane between vaginal mucosa and the pubocervical fascia.
· Divert directly from the midline, and dissect parallel to the vaginal mucosa.
· Put your finger between urethra and needle in order to avoid urethral damage.
· Do cystoscopy after having passed the needle with a 30 or 70° optical.
· Adjust the tape tension free or tension it with a partially filled bladder (300 ml) and Valsalva of the patient
· Keep the tape on the proper width and positioned at the midurethral level.
Transobturator Tapes
The new era of stress incontinence surgery started with the introduction of TVT. This new classic implied the retropubic technique to give support to the midurethra and to prevent dislocation of the midurethra during intra-abdominal pressure rise. The midurethral tape is U-shaped and runs retropubically. The most frequent occurring complication is bladder perforation when the needles with the connected tapes pass the pubic bone dorsally. Since the bladder is in the vicinity, it is easily possible to perforate it. Therefore, urethrocystoscopy is recommended during the procedure. In order to avoid bladder perforation and urethrocystoscopy during the procedure, the transobturator approach was introduced by Delorme in 2001. With this technique, the tape runs from one obturator foramen along the anterior vaginal wall via the midurethra to the opposite obturator foramen in a V-shaped fashion. There are two ways of introducing the tapes.
Inside-Out Technique
The patient is placed in the lithotomy position with thighs in flexion. A catheter is inserted into the bladder. The anterior vaginal wall is incised at a length of 1–2 cm and starting 1 cm proximally to the urethral meatus. Now the dissection path is developed towards the upper part of the ischiopubic ramus on with a 45° angle to the urethral sagittal plane. It is important to start in the good layer between the vaginal mucosa and the pubocervical fascia for developing a proper dissection plane. Once the upper part of the ischiopubic ramus is reached, the obturator membrane can be perforated with the tip of the scissors. A needle introducer, with the open side of its gutter facing the surgeon, is pushed along the developed dissection canal up to the obturator membrane. The distal end of the tube that is mounted on the spiral needle is slipped along the gutter of the introducer in order to pass through the obturator foramen. Then, the needle introducer is removed. Now the tube with the needle has passed through the obturator foramen and has appeared at the previously determined skin exit point. The tube is removed from the supporting needle, which is removed and the tube and the tape is pulled through the whole tract. The same technique is applied at the other side.
Next, the ends of the tape are cut; the tape is aligned tension free under the middle of the urethra. The plastic sheaths are removed and the tape ends are cut in the subcutaneous layer and the incisions are closed. The technique was first described by De Leval in 2003.
Outside-In Technique
This is the classic technique that was used as a first transobturator route. It implies the tape is going from outside to inside. The same plane is developed from the incision on the anterior vaginal wall under the urethral meatus. The insertion points in the thighs are determined, and the spiral-shaped needle is inserted through the skin. The correct insertion points are easily found when palpating the origin of the adductor muscles on the pubic bone. The incisions are made 1 cm under this origin and medial from the bone, which is directly at the upper inner corner of the obturator foramen.
Points of Interest
· After having made the midline incision, get into the right plane between vaginal mucosa and the pubocervical fascia.
· Divert directly from the midline, and dissect parallel to the vaginal mucosa.
· Consider doing cystoscopy after having passed the needle with a 30 or 70° optical.
· Pay special attention to the vaginal fornix at both sides for mucosal perforation.
· Keep the tape on the proper width and at the midurethral level.
Single Incision Slings
The idea behind single incision slings is to avoid the drawbacks of the transobturator route as well as the retropubic route. The theoretical advantage of the SIS is the avoidance of the retropubic space and obturator fossa and the lack of necessary thigh or suprapubic incisions. The effect of the tape for SUI is that it fixes the midurethra. This restores the hammock effect which restores proper sphincter functioning. It also induces kinking of the urethra on the spot of the tape which increases bladder outlet resistance and restores continence. Fixation of the two ends of the tape is in the pubocervical fascia. It creates a U-shaped look in cases of the retropubic approach and a V-shaped in case of the transobturator approach. The fixation of the tape ending in the pubocervical fascia also means that parts of the tape beyond that point are redundant. This means for retropubic tapes that those ends running lateral to the bladder neck and through the rectus abdominus muscle and the rest of the abdominal wall are redundant. If those ends would not be there, bladder perforation could be avoided. In cases of transobturator tapes, nerve and vascular injury in the obturator foramen and groin pain can be avoided. This should theoretically result in an easier procedure with shorter operating times and less postoperative pain.
Technique
The technique can vary according to the specific device that is being used. One technique has been described by Kennelly et al. A 1.5–2 cm single incision in the midline over the anterior vaginal wall, followed by subsequent periurethral dissection. The sling mounted to the needle is advanced behind the ischiopubic ramus in a dissection area towards the obturator space on both sides. The needle is removed after fixation of the sling with some kind of anchoring device into the obturator internus muscle after penetration of the obturator internus fascia. At this moment, the sling is fixed on one side. The tensioning of the sling now depends on the fixation of the opposite anchoring system at the contralateral transobturator foramen. This implies that the surgeon needs to decide with how much tension the sling is fixed before release of the needle. Adjustment after insertion of the tape and release of the needle is not easy, although some other devices allow for determining the tension before definite fixation of the sling. The clinical success therefore determines the dexterity and experience of the surgeon and the robust anchoring mechanism to the obturator complex with a strong post-insertion pull-out force.
Points of Interest
· Determine beforehand the fixation strength of the tape.
· Make sure that anchoring of the tapes at both sides is robust.
· Perform urethrocystoscopy when in doubt.
· Pay special attention to the vaginal fornix at both sides for mucosal perforation.
· Use a tape that has proven its efficacy and safety.
Tips and Tricks/Dos and Don’ts
Obese
Obesity and overweight are often seen as risk factors for developing stress incontinence and vaginal prolapse as well as for undergoing surgery. Indeed a relationship between BMI and continence has been demonstrated in several studies, where in morbidly obese women the prevalence of incontinence ranged between 66 and 71 % compared to 4–50 % in the nonobese population.
It has been shown that lifestyle adaptations that lead to weight loss as well as bariatric surgery in the obese leads to significant improvements or even cure of incontinence.
Concerning the outcome of surgical interventions for SUI in obese women, the literature is less clear. Most studies are retrospective and of poor quality. Some studies claim to have similar results in normal and obese women, while others showed less favourable objective cure rates in obese women. In one prospective long-term study on TVT, obesity was an independent risk factor for recurrence with a hazard ratio of 17!
Practically, surgeons planning incontinence surgery in the obese should counsel the patient thoroughly about the anaesthesiological and surgical risks related to obesity. Although there is no clear evidence or guidance on this subject, an appropriate approach should be chosen since a retropubic approach might be too risky in obese women. A transobturator approach might be more feasible. Surgeons should be aware that some kits only provide standard needles that might be too short in some women.
Good postoperative care with early mobilisation is mandatory.
Mixed Incontinence
Careful history taking is of utmost importance in the preoperative workout of urinary incontinence. In 20–36 % of cases, a concomitant history of urgency with or without urgency incontinence will be present. When this is the case, it is important to determine the predominant complaint of the patient. A micturition diary is an essential part of this workout. Whenever in doubt, a urodynamic study should be performed. All these investigations aim to determine the chief complaint of the patient.
In all cases, conservative treatment remains the first-line treatment. In patient with mixed incontinence, pelvic floor exercises with bladder training can significantly improve both symptoms.
When predominant SUI is present, surgical treatment of stress incontinence should be offered. It is hard to predict in individual cases how the pre-existing urgency will evolve after the treatment of SUI. As a rule of thumb, it will be improved to even absent in 1/3th of patients, unchanged in 1/3th and worsened in 1/3th. As some patients will develop preventive frequency (to avoid SUI when the bladder gets filled), this symptom will improve after sling surgery.
When predominant urgency incontinence is present, one should not directly offer surgery for SUI. It is likely that the patient will not be satisfied after the surgery. At first, classical approaches to treat urgency and urgency incontinence should be offered, such as antimuscarinics/botulinum toxin or sacral neuromodulation. Only when the urgency complaints are substantially improved, one should offer sling surgery to patients with persistent SUI. Again, it is hard to predict in individual cases how the pre-existing urgency will evolve after the treatment of SUI. Similar is stated above; as a rule of thumb, it will improve to even disappear in 1/3th of patients, remain unchanged in 1/3th and worsen in 1/3th.
Second Operation
As described before, most incontinence procedures have very high success rates. Nevertheless, due to the very high numbers of incontinence procedure performed all over the world, the relative low failure rates do constitute a large number of patients that will have persistent or recurrent SUI. In the literature, there is conflicting evidence whether repeat surgery for SUI results in inferior outcomes as compared to primary surgery.
Recurrent incontinence is in general defined by the presence of urinary incontinence that is preceded by a period of total continence of at least 6 weeks after the first surgery. Persistent incontinence is usually defined by urinary incontinence that has never disappeared after the treatment.
When a patient has persistent or recurrent urinary incontinence, a new thorough workout has to be performed. We refer to the diagnostics section of this manuscript for details regarding preoperative workout. In the setting of recurrent or persisting incontinence, it is of utmost importance to clearly distinguish between (de novo) urgency urinary incontinence and persistent or recurrent SUI. Of course, mixed incontinence is also possible. Whenever mixed incontinence is present, the predominant complaint will be treated first as in the setting of primary mixed urinary incontinence (see previous section).
In the setting of urgency urinary incontinence after previous surgery for SUI, treatment modalities are similar to the treatment modalities of urgency urinary incontinence in primary setting. One major difference is that the treating physician has to address the possibility of obstructive voiding after the first sling surgery. Evaluating obstruction after previous sling surgery is not easy. A history of straining, Valsalva voiding, micturition in several times, post-void residual urine and frank urinary retention that emerged after surgery for SUI are suggestive for obstructive voiding. Several authors have tried to define urodynamic parameters to determine the presence of obstructive voiding in females. Until now, there exists no generally accepted and validated nomogram for female voiding function. Most of the nomograms are developed using pressure flow analysis with or without free flow. Others use radiographic imaging to define obstruction. In clinical practice, it is important to realise that these proposed nomograms have their strengths and weaknesses. They are an important aid in medical diagnosis when they are interpreted in the light of the complaints and the history of the specific patient. They are however not an exact scientific tool to objectively determine the presence or absence of voiding obstruction.
Whenever clinical and/or urodynamic findings are suggestive of obstructive voiding after anti-incontinence surgery, desobstructive surgery has to be considered. In the case of previous sling surgery, this surgery can vary between simple sling transection over partial sling excision to complete excision of the sling. In the case of previous colposuspension, a complete urethrolysis will have to be performed. This procedure has a high success rate with absence of obstructive voiding in more than 80 %. The absence however of obstructive voiding is not always a synonym for satisfied patients. A proportion of these patients will still suffer overactive bladder complaints or SUI mandating further treatment.
In the case of persistent or recurrent SUI, the usefulness of repeat surgery has to be estimated. Again, there exists no accurate, scientific or objective tool to guide our decisions. A good clinical examination followed by urodynamic study is the mainstay of these patients’ work out. During clinical examination, one of the key elements is to evaluate urethral mobility. In the presence of urethral mobility, repeat sling surgery has proven its efficacy in several nonrandomised studies.
In all these studies, repeat sling surgery was technically easy, associated with low blood loss and with short surgery times and hospital stays. Complication rates were higher as compared to primary surgery, but acceptable. Most complications were mild and easy to treat.
On urodynamics, the presence or absence of ‘intrinsic sphincter deficiency’ (ISD) can be judged. In literature, a Valsalva leak point pressure of 60 cmH2O or less and a mean urethral closing pressure of 20 cmH2O or less are considered to determine ISD. The prevalence of ISD in literature varies between 10 and 100 % of reported series. It is clear that the real prevalence is not known and that this wide variety reflects a selection bias.
In the presence of ISD or a fixed urethra, subsequent surgery for SUI will obviously be much more difficult. Sling surgery will, by definition, consist of a non-physiological obstructive sling with high postoperative rates of voiding dysfunction. There is some evidence in literature that cure rates for retropubic slings are higher than cure rates for transobturator slings. Some case series where adjustable sling systems were used also show high cure rates. Most of these studies are small case series with variable definitions of cure and satisfaction. One has to be cautious to interpret these results. When the use of synthetic sling material is undesirable, also pubovaginal slings have shown high success rates. As in the synthetic slings, these slings will be obstructive in nature and the patient has to be warned for the possibility of postoperative retention and subsequent need for intermittent catheterization.
In selected cases, also the use of in artificial urinary sphincter (AMS 800) should be considered. The use of the device in females is less known and less widespread as compared to its use in males. Nevertheless, some large series have been published with good results. A high revision rate is an inevitable consequence of the use of the device. The surgery is more invasive as compared to the different sling solutions. Previous anti-incontinence surgery and higher age are independent predictors of failure. This surgery should only be performed in tertiary centres.
In summary, a second operation can offer a solution for recurrent or persistent incontinence in selected cases. The indication for a second operation should always be made with great caution and most energy should be put in the preoperative diagnosis of the problem. With the right diagnosis, a second operation will improve patients’ quality of life and satisfaction.
Elderly
There are limited specific data on SUI treatment in the elderly.
Concerning conservative therapy, it has been shown that elderly patients benefit as well as younger women.
There are no direct comparative trials on surgical treatment in older versus younger women, but subgroup data have been published.
Elderly women benefit from surgical treatment of SUI, and age by itself is not a contraindication for surgery. It has been shown that cure rates decrease with age. Elderly women are also prone to have persisting incontinence and have a higher risk for de novo urgency. There are no data showing that one or another type of surgery or sling would have better results in elderly patients.
Complications of Female Stress Incontinence Surgery
Introduction and General Recommendations
This section will focus on the prevention and management of the complications related to tension-free midurethral synthetic slings (MUSS) based on the classification of the 5th International Consultation on Incontinence (Table 5.1). It is noteworthy that most of the recommendations in this section are derived from level four studies or ‘majority evidence’ from level 2/3 studies or Delphi processed expert opinion with the recommendation grade of C according to Oxford system.
Table 5.1
Classification of complications of MUSS surgery [6]
|
Intraoperative complications |
Immediate postoperative complications |
Chronic problems |
|
Vessel injury Bladder injury Urethral injury Visceral injury |
Infection Urinary retention and emptying difficulty Groin pain |
De novo urgency Sling erosion/extrusion |
The basic principles of the management of MUSS complications are summarised as follows:
1.
2.
3.
4.
5.
6.
Dealing with Intraoperative Complications
Vessel Injury
Minor to Moderate Haemorrhage
Haemorrhage during surgery should be managed first by local compression to decrease blood loss and prevent postoperative haematoma. Local compression can be continued postoperatively by intravaginal tamponade with surgical sponges. Intraoperative blood loss more than 100 mL may occur twice as frequently in the retropubic (RP) compared to transobturator (TO) route. Due to the route of needle passage, haematoma and intraoperative haemorrhage rarely occur in TO MUSS procedures. Inserting redon drains in patients with increasing bleeding during RP MUSS may reduce the risk of retropubic haematoma that is a risk factor for infection, voiding difficulty and mesh problems.
Major Haemorrhage Due to Major Vessel Injury
Seven deaths were reported in the database of the US FDA from 1998 to 2005 due to major vessel injury during trocar passage in RP procedures, whereas major vascular injury has not been reported in TO procedures. During surgery, haemodynamic instability, growing hematoma or excessive bleeding at the operative site may indicate major vessel injury that necessitates urgent primary vascular repair to prevent mortality. Prompt intervention can save the life of the patient as it was reported in a case of external iliac artery injury during an RP MUSS. The low risk for large vessel injury reported in TO procedures – given the anatomy of TO trocar passage – does not mean that it is impossible, considering the few centimetres distance of the trocar from obturator vessels.
Injury to the Bladder and Urethra
Injury to the lower urinary tract may occur at the level of the urethra and bladder during dissection or more commonly during trocar passage. RP procedures are more commonly associated with this complication compared to TO route where top-to-bottom or bottom-to-top passage does not affect the bladder perforation rates. Violation of the urinary tract can be managed successfully without causing significant morbidity unless it is not recognised intraoperatively. Missing an injury to the urinary tract may cause fistula, stone formation, haematuria, infection and even progressive cellulitis in the postoperative period and will necessitate additional surgical intervention(s). If severe progressing cellulitis occurs in a patient despite broad-spectrum antibiotic coverage unrecognised bladder perforation and nonbacterial causes of infection should be ruled out.
Traditionally, cystourethroscopy with a 70° optic lens is advised to rule out any violation to the urinary tract in all RP MUSS procedures. The decision to perform cystourethroscopy in TO procedures is usually made by the surgeon according to his experience and preference where any suspicion mandates cystourethroscopy. A simple method to predict urinary tract damage may be the intraoperative irrigation of the bladder with saline through the Foley catheter after insertion of the trocars to reveal blood in the bladder or extravasation from the urinary tract. It should be also kept in mind that bladder perforation can occasionally be missed during cystourethroscopy where perforation may be determined by suprapubic incisional fluid leakage after removing the trocars.
In the presence of a urethral injury, the urethra should be repaired appropriately without placing any suburethral synthetic sling since this would lead to urethral erosion. Utilising a Martius flap and continuing the surgery with autologous fascial graft depends on the decision of the surgeon and prior consent of the patient. When there is a bladder perforation by the trocar, the trocar should be gently pulled out and then correctly reinserted without the need for any repair of the bladder. After completing MUSS placement, the bladder should be drained 2 days by a urethral catheter. Some authors even discharge patients with bladder violation the same day of MUSS surgery without catheter drainage.
Visceral Injury
Bowel injury is an extremely rare complication of MUSS surgery exclusively seen after RP procedures especially in patients with previous history of pelvic operations. It may manifest postoperatively with persistent low abdominal pain resistant to analgesics, fever and sepsis in late cases. Immediate laparoscopy or laparotomy should be performed whenever bowel injury is suspected. Subsequent repair of bowel peroration site is indicated to solve the problem.
Dealing with Immediate Postoperative Complications
Infections
Infections after MUSS include a spectrum from uncomplicated urinary tract infections reaching necrotising fasciitis and pelvic or thigh abscess formation. The presence of positive urine culture, vaginal discharge, pelvic discomfort or unexplained fever mandates a careful clinical assessment to rule out increased post-voiding residual urine (PVR)/obstruction, mesh erosion or extrusion and pelvic infections. Infected pelvic hematoma, diabetes and morbid obesity are risk factors for necrotising infections. Infection-related complications after TO route have included thigh abscess requiring drainage and an infected obturator hematoma also requiring exploration and drainage.
Urinary Retention and Emptying Difficulty
Voiding problems after MUSS’s may vary in terms of the type and the severity in a range between urinary frequency and urinary retention (Fig. 5.1). The pathophysiology of de novo voiding problems is not well understood, but it is linked to the mesh-related urethral obstruction or irritation. Other causes such as bladder perforation, pelvic haematoma, urethral erosion or vaginal extrusion of the mesh should always be considered in the differential diagnosis.
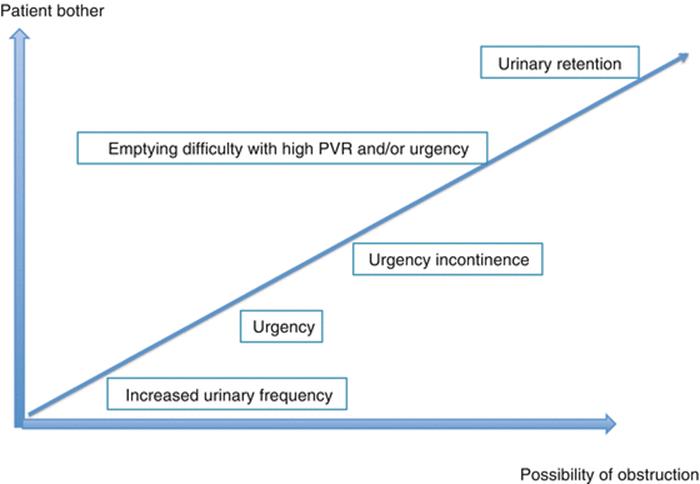
Fig. 5.1
Spectrum of de novo voiding symptoms after MUSS
Urinary retention stands at the edge of the voiding symptom spectrum after MUSS as the most annoying one both for the patient and the surgeon (Fig. 5.1). However, its treatment is more straightforward than the other voiding problems since it is obviously caused by urethral obstruction. Urinary retention may be defined as catheter dependency for at least 28 days where there is no consensus about the cutoff level of PVR that necessitates catheterisation. It is not only the PVR volume but also the discomfort of the patient that necessitates catheterisation.
Urinary retention may be first recognised when the patient fails to empty her bladder at the first voiding trial after surgery. Women are more likely to empty their bladders effectively if they are evaluated with a backfill-assisted voiding trial compared to spontaneous natural bladder filling and emptying. Therefore, the bladder should not be overfilled for the initial voiding trials after surgery. Emptying difficulty is common in the early postoperative period but may be transient and associated with the immediate voiding conditions such as increased fluid load and bladder overdistention. Among patients who fail the initial voiding trial, about 1/3 successfully can void on subsequent trials. Therefore, multiple voiding trials are advised before making any decision. Interestingly, 16 % of patients who pass the initial voiding trial may fail on the second necessitating at least two successful voiding trials before discharge.
The conventional approach to urinary retention includes indwelling catheterisation up to 1 week (3–7 days) and retesting the patient after catheter removal (Fig. 5.2). Some authors prefer tape release in the early postoperative period that is possible up to 10 days. Retention lasting longer than 1 week may be managed by an early tape cut or clean intermittent catheterisation up to 1 month. There is no benefit in waiting more than 4 weeks since symptoms persisting beyond that time rarely resolve spontaneously. Waiting period of at least 14 days but optimally 4 weeks prior to tape cut is usually recommended to allow suburethral fibrosis to prevent recurrent incontinence.
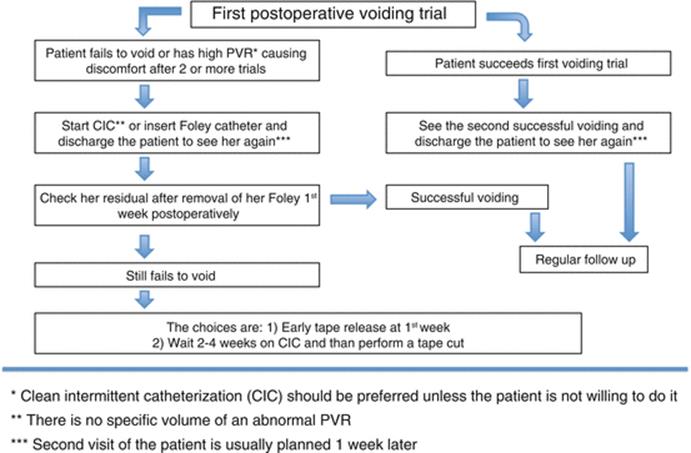
Fig. 5.2
A suggested algorithm for the management of urinary retention after MUSS
Groin Pain and Other Neurological Symptoms
Neurologic adverse effects such as numbness and/or weakness in the legs or pelvic area and dyspareunia are more common after TO compared to RP procedures. Transient groin pain occurs in about 2.3–15.9 % of patients after TO MUSS and generally responds to nonsteroidal anti-inflammatories. The aetiology of the pain is likely to be due to either subclinical hematoma or a transient neuropathic phenomenon. In patients who present with neurological symptoms, dyspareunia or pain, the physician should try to locate the origin of the pain. Persistent leg pain should raise the suspicion of urethral erosion or vaginal extrusion.
Dealing with Chronic Problems
De Novo Urgency
Storage symptoms that were not present preoperatively but appear after surgery should be considered as mesh or surgery related. However, these symptoms may sometimes have a gradual onset and may not be temporarily related to the previous MUSS surgery. Furthermore, mild symptoms may be underdiagnosed and under-reported. Cystoscopy is mandatory in patients with haematuria, bladder pain or recurrent cystitis especially when bladder perforation or urethral mesh erosion is suspected (Fig. 5.3). Urethrocystoscopy may further reveal urethral kinking.
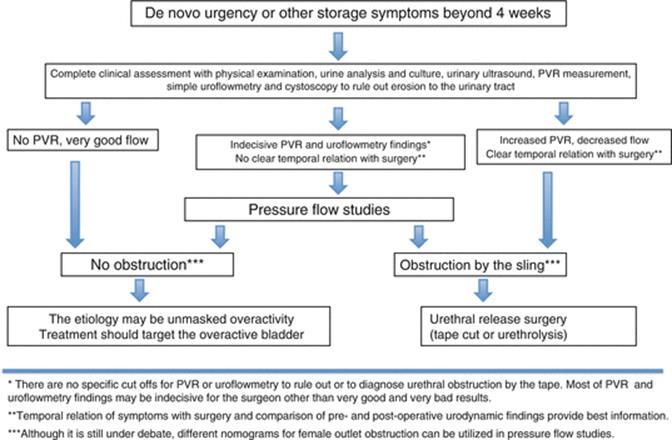
Fig. 5.3
A suggested algorithm for the management of de novo urgency after MUSS
Urodynamic evaluation may include noninvasive tests such as PVR measurement and uroflowmetry and invasive tests such as cystometry and pressure flow studies. Diagnosis of obstruction in women lacks well-defined urodynamic criteria since women empty their bladder by relaxing the pelvic floor without a strong detrusor contraction. Furthermore, there are no predicting factors including urodynamics to show which patients would benefit from urethrolysis. Comparison of the pre- and postoperative uroflowmetric studies may help better in the diagnosis of mesh-related obstruction than a single postoperative pressure flow study (Fig. 5.3). The temporal relationship correlating symptoms with an antecedent surgical procedure should be the primary criterion in selecting patients for urethral release. Midline transvaginal tape cut without urethrolysis is the most often performed surgical treatment, and this treatment provides resolution of symptoms with maintenance of continence in the majority of patients. A part of the sling should be excised for pathological confirmation. Antimuscarinic treatment may be utilised after urethral obstruction by the mesh is ruled out or managed.
Sling Erosion/Extrusion
Vaginal Extrusion
Wound infection, impaired wound healing, improper vaginal dissection plane and vaginal atrophy may be causative factors. Presenting symptoms may include vaginal discharge, palpable rough surface in the vagina, sexual discomfort (usually partner related) and lower urinary tract symptoms including haematuria. The management options are not standardised and include local oestrogens, partial and complete tape excision and reapproximation of the vaginal mucosa over the exposed tape. Initial observation of small vaginal erosions with topical oestrogen creams is recommended. Excision should be reserved for conservative treatment failures or for larger defects. Partial mesh excision with reapproximation of the vaginal mucosa will not cause incontinence in the majority of patients.
Urethral Erosion
Surgeon-related factors play the most important role in the aetiology of urethral erosion such as entering wrong plane during dissection beneath the urethra, compromising thickness of suburethral tissue or even causing an unrecognised urethral perforation and finally applying excess sling tension on the urethra. Urethral erosion may present itself with storage or emptying lower urinary tract symptoms or urinary tract infection or haematuria. Observing the tape within the urethral lumen during cystoscopy makes the diagnosis. Conservative observational treatment is not an option, and endoscopic tape transection or transvaginal excision of the tape with closure of the urethrotomy is needed. A Martius fat pad graft may be used in case of extensive urethrotomies. A sling preferentially autologous can be placed at the time of surgery or in a delayed stage to treat possible SUI.
Intravesical Erosion
This complication almost exclusively occurs secondary to an unrecognised bladder perforation during trocar placement. Therefore, intravesical tape erosion rate is lower for TO procedures compared to RP route. Patients may present with storage symptoms, haematuria, recurrent urinary tract infections and pelvic pain. Observational treatment is not an option. The sling material inside the bladder must be removed either endoscopically or by open surgery. Only patients with complete tape removal develop recurrent stress incontinence. Reduced pore size is associated with higher erosion rates and type 1 synthetic slings with larger pore size are favourable since they facilitate vascular and tissue ingrowth optimising mesh incorporation.
Tips and Tricks
1.
2.
3.
4.
5.
Further reading
1.
Hay-Smith EJC, Herderschee R, Dumoulin C, Herbison GP. Comparisons of approaches to pelvic floor muscle training for urinary incontinence in women. [Internet]. Cochrane Database Syst Rev (Online). 2011; p. 149. Available from: http://www.ncbi.nlm.nih.gov/pubmed/22161451.
2.
Richter HE, Albo ME, Zyczynski H, Kenton K. Retropubic versus transobturator midurethral slings for stress incontinence. N Engl J Med [Internet]. 2010;362(22):2066–76. Available from: http://www.ncbi.nlm.nih.gov/pubmed/22153438.
3.
Nilsson CG, Palva K, Aarnio R, Morcos E, Falconer C. Seventeen years’ follow-up of the tension-free vaginal tape procedure for female stress urinary incontinence. Int Urogynecol J [Internet]. 2013;24(8):1265–9. [cited 29 Jun 2014]. Available from: http://www.ncbi.nlm.nih.gov/pubmed/23563892.
4.
Mostafa A, Phay C, Hopper L, Madhuvrata P. Single-incision mini-slings versus standard midurethral slings in surgical management of female stress urinary incontinence: an updated systematic review and meta-analysis of effectiveness and complications. Eur Urol [Internet]. 2014;65(2):402–27. European Association of Urology. Available from: http://dx.doi.org/10.1016/j.eururo.2013.08.032.
5.
Costa P, Poinas G, Ben Naoum K, Bouzoubaa K, Wagner L, Soustelle L, et al. Long-term results of artificial urinary sphincter for women with type III stress urinary incontinence. Eur Urol. 2013;63(4):753–8.CrossRefPubMed
6.
Dmochowski R, Athanasiou S, Reid F, Kraus S, Nitti V, Gomelsky A, Robinson D, Smith ARB. Surgery for urinary incontinence in women. In: Abrams P, Cardozo L, Khoury S, Wein A, editors, Incontinence, 5th ed. 5th international consultation on incontinence, Paris February 2012. 2013.
7.
Çetinel B, Tarcan T. Management of complications after tension-free mid-urethral slings. Korean J Urol. 2013;54(10):651–9.PubMedCentralCrossRefPubMed
8.
Brubaker L, Norton PA, Albo ME, et al. Adverse events over two years after retropubic or transobturator midurethral sling surgery: findings from the Trial of Midurethral Slings (TOMUS) study. Am J Obstet Gynecol. 2011;205:498.e1–6.CrossRef
9.
Dmochowski R, Scarpero H, Starkman J. Chap 68: Tension-free vaginal tape procedures. In: Wein AJ, editor. Campbell-Walsh urology. 9th ed. Philadelphia: Saunders Elsevier; 2007. p. 2251–72.
10.
Carr LK, Webster GD. Voiding dysfunction following incontinence surgery: diagnosis and treatment with retropubic or vaginal urethrolysis. J Urol. 1997;157(3):821–3.CrossRefPubMed