Systemic Circulation
The systemic circulation supplies blood to all the tissues of the body except the lungs. Important considerations in understanding the physiology of the systemic circulation include the anatomic components of the systemic circulation, physical characteristics of the systemic circulation and of blood, determinants and control of tissue blood flow, regulation of systemic blood pressure, and regulation of cardiac output and venous return. In addition, the fetal circulation possesses many unique features, which distinguish it from the systemic circulation after birth.
Endothelial Function
The entire vascular system is lined by endothelial cells. Thus, the endothelium is a large and widely distributed structure. Indeed, it is estimated that the adult endothelium is composed of 10 trillion cells and weighs approximately 1 kg.1 It is now widely appreciated that the endothelium is not simply an inert lining layer of the circulation but is an important “organ” that is involved in many physiologic processes in health and disease (Tables 14-1 and 14-2). The luminal side of the endothelium is lined with a “glycocalyx,” a web of membrane-bound glycoproteins and proteoglycans, which plays an important role in transcapillary flow.2 The healthy endothelium promotes vasodilation and confers antithrombotic and antiadhesive properties to the vessel wall; damage to the glycocalyx and endothelium result in increased vascular permeability and adherence of inflammatory mediators and cells. The endothelium also regulates smooth muscle proliferation and has an important role in the regulation of glucose and lipid metabolism. Endothelial dysfunction is an important element of cardiovascular disease and aging. Cardiovascular risk factors including smoking, diabetes mellitus, hyperlipidemia, obesity, and systemic hypertension are related to their adverse effects upon endothelial function.


Endothelial Function and Regulation of Vascular Tone
Endothelial synthesis and release of vasoactive mediators are important elements in the regulation of vascular tone. Substances are released by the endothelium in response to both mechanical and humoral stimuli and generally have an immediate effect upon the adjacent vascular smooth muscle tone. However, there may also be endothelium-induced long-term effects from vascular remodeling and smooth muscle hypertrophy. Under physiologic conditions, local vascular pressure and flow are the primary stimuli for endothelial vasoactive substance release. Nitric oxide and prostacyclin are powerful vasodilators released by endothelial cells and both also inhibit platelet aggregation and thrombosis. Continuous nitric oxide production maintains vascular tone in a normally low state. This minute-to-minute regulation of local vascular tone is controlled by type 3 constitutive nitric oxide synthase (cNOS). cNOS is a rapidly responding endothelial enzyme that catalyses the local conversion of L-arginine into small quantities of nitric oxide in response to endothelial shear stress during normal pulsatile flow. On the other hand, type 2 inducible NOS (iNOS) is a relatively slow-responding enzyme that catalyses the production of large amounts of nitric oxide in response to inflammatory cytokines. The widespread generation of large amounts of nitric oxide via iNOS is responsible for the low systemic vascular resistance and hypotension encountered in septic shock.3 Endothelin-1 (ET-1) is a potent vasoactive compound released by the endothelium. Its predominant effect is vasoconstriction via smooth muscle ETA receptors. However, ET-1 can also cause vasodilation through its effect on endothelial ETB receptors. ET-1 stimulates smooth muscle proliferation and is an important factor in the development of vascular structural changes in systemic and pulmonary hypertension.
Components of the Systemic Circulation
The components of the systemic circulation are the arteries, arterioles, capillaries, venules, and veins.
Arteries
The function of the arteries is to transport blood under high pressure to tissues. Therefore, arteries have strong vascular walls and blood flows rapidly through their lumens.
Arterioles
Arterioles are the last small branches of the arterial system, having diameters of less than 200 µm. Arterioles have strong muscular walls, which are capable of dilating or contracting and thus controlling blood flow into the capillaries. Indeed, blood flow to each tissue is controlled almost entirely by resistance to flow in the arterioles. Metarterioles arise at right angles from arterioles and branch several times, forming 10 to 100 capillaries which in turn connect with venules.
Capillaries
Capillaries are the sites for transfer of oxygen and nutrients to tissues and receipt of metabolic byproducts.
Venules and Veins
Venules collect blood from capillaries for delivery to veins, which act as conduits for transmitting blood to the right atrium. Because the pressure in the venous system is low, venous walls are thin. Nevertheless, walls of veins are muscular, which allows these vessels to contract or expand and thus store varying amounts of blood, depending on physiologic needs. As a result, veins serve an important storage function as well as being conduits to return blood to the right atrium. A venous pump mechanism is important for propelling blood forward to the heart.
Physical Characteristics of the Systemic Circulation
The systemic circulation contains about 80% of the blood volume, with the remainder present in the pulmonary circulation and heart (Fig. 14-1).4 Of the blood volume in the systemic circulation, about 64% is in veins and 7% is in the cardiac chambers. The heart ejects blood intermittently into the aorta such that blood pressure in the aorta fluctuates between a systolic level of about 120 mm Hg and a diastolic level of about 80 mm Hg (Table 14-3) (Fig. 14-2).4


One of the primary responsibilities of the anesthesiologist is to maintain organ perfusion with oxygenated blood. Our standard physiologic monitors (heart rate, blood pressure, pulse oximetry, capnography) all serve as surrogate markers of organ perfusion and oxygenation. Currently, our standard monitoring techniques do not allow us to directly monitor the level of perfusion in specific organs or tissues. However, various techniques for monitoring end-organ perfusion are being explored and will likely be increasingly used in the years to come.
Progressive Declines in Systemic Blood Pressure
As blood flows through the systemic circulation, perfusion pressure decreases progressively to nearly 0 mm Hg by the time blood reaches the right atrium (see Fig. 14-2).4 The decrease in systemic blood pressure in each portion of the systemic circulation is directly proportional to the resistance to flow in the vessels. Resistance to blood flow in the aorta is minimal, and mean arterial pressure decreases only 3 to 5 mm Hg as blood travels into arteries as small as 3 mm in diameter. Resistance to blood flow begins to increase rapidly in small arteries, causing the mean arterial pressure to decrease to about 85 mm Hg at the beginning of the arterioles. It is in the arterioles that resistance to blood flow is the highest, accounting for about 50% of the resistance in the entire systemic circulation. As a result, systemic blood pressure decreases to about 30 mm Hg at the point where blood enters the capillaries. At the venous end of the capillaries, the intravascular pressure has decreased to about 10 mm Hg. The decrease in systemic blood pressure from 10 mm Hg to nearly 0 mm Hg as blood traverses veins indicates that these vessels impart far more resistance to blood flow than would be expected for vessels of their large sizes. This resistance to blood flow is caused by compression of the veins by external forces that keep many of them, especially the vena cava, partially collapsed.
Pulse Pressure in Arteries
Pulse pressure reflects the intermittent ejection of blood into the aorta by the heart (see Table 14-3). The difference between systolic and diastolic blood pressure is the pulse pressure. A typical systemic blood pressure curve recorded from a large artery is characterized by a rapid increase in pressure during ventricular systole followed by a maintained high level of blood pressure for 0.2 to 0.3 second (Fig. 14-3). This plateau is followed by the dicrotic notch (incisura) at the end of systole and a subsequent, more gradual decrease of pressure back to the diastolic level. The dicrotic notch reflects a decrease in the intraventricular pressure and a backflow of blood in the aorta, which causes the aortic valve to close.

Factors that Alter Pulse Pressure
The principal factors that alter pulse pressure in the arteries are the left ventricular stroke volume, velocity of blood flow, and compliance of the arterial tree. The larger the stroke volume, the greater the volume of blood that must be accommodated in the arterial vessels with each contraction resulting in an increased pulse pressure. Pulse pressure also increases when capacitance increases for outflow. When systemic vascular resistance decreases, flow of blood from arteries to veins is accelerated. Pulse pressure is also increased in the presence of patent ductus arteriosus and aortic regurgitation, reflecting rapid runoff of blood into the pulmonary circulation or left ventricle, respectively. In this regard, attempts have been made to predict systemic vascular resistance by the position of the dicrotic notch relative to the diastolic pressure. A controlled study, however, failed to confirm a correlation between the position of the dicrotic notch and the calculated systemic vascular resistance (Fig. 14-4).5 An increase in heart rate while the cardiac output remains constant causes the stroke volume and pulse pressure to decrease. Pulse pressure is inversely proportional to the compliance (distensibility) of the arterial system. For example, with aging, the distensibility of the arterial walls often decreases (elastic and muscular tissues are replaced by fibrous tissue) and pulse pressure increases.

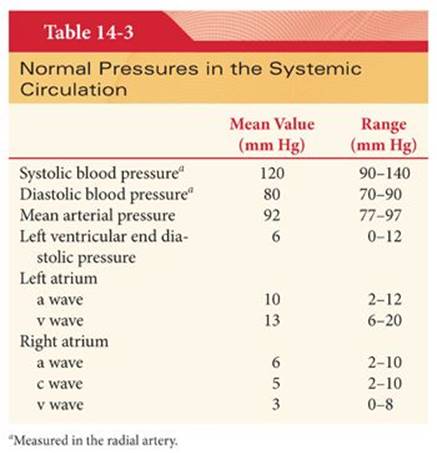
Transmission of the Pulse Pressure
There is often enhancement of the pulse pressure as the pressure wave is transmitted peripherally (Fig. 14-5).4 Part of this augmentation results from the progressive decrease in compliance of the more distal portions of the large arteries. Second, pressure waves are reflected to some extent by the peripheral arteries. Specifically, when a pulsatile pressure wave enters the peripheral arteries and distends them, the pressure on these peripheral arteries causes the pulse wave to begin traveling backward. If the returning pulse wave strikes an oncoming wave, the two summate, causing a much higher pressure than would otherwise occur.
Augmentation of the peripheral pulse pressure must be identified whenever systemic blood pressure measurements are made in peripheral arteries. For example, systolic pressure in the radial artery is sometimes as much as 20% to 30% higher than that pressure present in the central aorta, and diastolic pressure is often decreased as much as 10% to 15%. Mean arterial pressures are similar regardless of the site of blood pressure measurement in a peripheral artery.
Pulse pressure becomes progressively less as blood passes through small arteries and arterioles until it becomes almost absent in capillaries (see Fig. 14-2).4 This reflects the extreme distensibility of small vessels such that the small amount of blood that is caused to flow during a pulsatile pressure wave produces progressively less pressure increase in the more distal vessels. Furthermore, resistance to blood flow in these small vessels is such that flow of blood and, consequently, the transmission of pressure are greatly impeded.
Systemic Blood Pressure Measurement during and after Cardiopulmonary Bypass
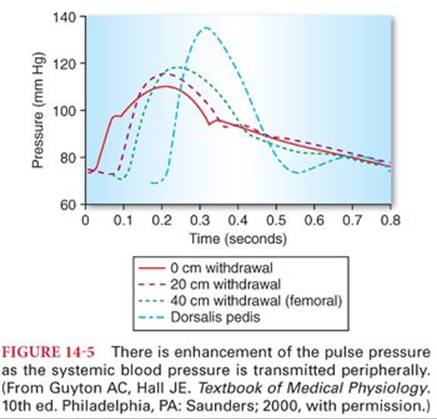
Reversal of the usual relationship between aortic and radial artery blood pressures can occur during the late period of hypothermic cardiopulmonary bypass and in the early period after termination of cardiopulmonary bypass (Fig. 14-6).6,7 One mechanism proposed for this unpredictable and transient disparity (usually persists for 10 to 60 minutes after discontinuation of cardiopulmonary bypass) is a high blood flow in the forearm and hand after rewarming on cardiopulmonary bypass, causing an increased pressure drop along the normal resistance pathway provided by the arteries leading to the radial site. Conversely, others describe the appearance of this gradient with initiation of cardiopulmonary bypass, suggesting that the etiology is associated with events such as cross-clamping of the aorta occurring during initiation of cardiopulmonary bypass rather than rewarming or discontinuing cardiopulmonary bypass (Fig. 14-7).8,9 Failure to recognize this disparity may lead to an erroneous diagnosis and unnecessary treatment. Systemic blood pressure measured in the brachial artery is more accurate and reliable during the periods surrounding cardiopulmonary bypass, which are most likely to be associated with disparities between the aortic and radial artery blood pressures.10

Pulsus Paradoxus
Pulsus paradoxus is an exaggerated decrease in systolic blood pressure (>10 mm Hg) during inspiration in the presence of increased intrapericardial pressures (cardiac tamponade). During normal inspiration, the decrease in intrathoracic pressure increases the compliance of the pulmonary vasculature, which leads to a relative decrease in pulmonary venous return to the left ventricle. The resultant reduction in left ventricular preload decreases the stroke volume, which manifests as a mildly decreased systolic blood pressure during inspiration (<10 mm Hg). Cardiac tamponade causes an exaggeration of this change in blood pressure with respiration.
Pulsus Alternans
Pulsus alternans is alternating weak and strong cardiac contractions causing a similar alteration in the strength of the peripheral pulse. A variety of physiologic conditions are associated with pulsus alternans. Digitalis toxicity, varying degrees of atrioventricular heart block, and left ventricular dysfunction are commonly associated with pulsus alternans. In the setting of left ventricular dysfunction, pulsus alternans is caused by cyclic alterations in the contractile state of the heart. A reduced stroke volume increases end diastolic volume, which results in increased myocardial contraction and therefore increased ventricular emptying and blood pressure (per the Frank-Starling law). During the subsequent cardiac cycle, the lower filling pressures in the left ventricle result in a decreased stroke volume and therefore decreased ventricular emptying and blood pressure.
Electrical Alternans
Electrical alternans is a phenomenon where the amplitude of the QRS complex changes between heart beats. This electrocardiographic finding is seen in cardiac tamponade and pericardial effusion, where the heart essentially moves within the fluid-filled pericardial sac during contraction.
Pulse Deficit
In the presence of atrial fibrillation or ectopic ventricular beats, two beats of the heart may occur so close together that the ventricle does not fill adequately and the second cardiac contraction ejects an insufficient volume of blood to create a peripheral pulse. In this circumstance, a second heart beat is audible with a stethoscope applied on the chest directly over the heart, but a corresponding pulsation in the radial artery cannot be palpated. This phenomenon is called a pulse deficit.
Measurement of Blood Pressure by Auscultation
Measurement of blood pressure by auscultation uses the principle that blood flow in large arteries is laminar and not audible. If blood flow is arrested by an inflated cuff and the pressure in the cuff is released slowly, audible tapping sounds (Korotkoff’s sounds) can be heard when the pressure of the cuff decreases just below systolic blood pressure and blood starts flowing in the brachial artery. These tapping sounds occur because flow velocity through the constricted portion of the blood vessel is increased, resulting in turbulence and vibrations that are heard through the stethoscope. Diastolic blood pressure correlates with the onset of muffled auscultatory sounds. The auscultatory method for determining systolic and diastolic blood pressure usually gives values within 10% of those determined by direct measurement from the arteries.
The width of the blood pressure cuff will affect measurements; ideally, the width of the blood pressure cuff should be 20% to 50% greater than the diameter of the patient’s extremity. If the cuff is too narrow, the blood pressure will be overestimated. If the cuff is too large, the blood pressure may be underestimated.
Right Atrial Pressure
Right atrial pressure is regulated by a balance between venous return and the ability of the right ventricle to eject blood. Normal right atrial pressure is about 5 mm Hg, with a lower limit of about −5 mm Hg, which corresponds to the pressure in the pericardial and intrapleural spaces that surround the heart. Right atrial pressure approaches these low values when right ventricular contractility is increased or venous return to the heart is decreased by hemorrhage. Poor right ventricular contractility or any event that increases venous return (hypervolemia, venoconstriction) tends to increase right atrial pressure. Pressure in the right atrium is commonly designated the central venous pressure(CVP). Other factors that increase CVP include tension pneumothorax, heart failure, tamponade, pleural effusion, mechanical ventilation, positive end-expiratory pressure, Valsalva, pulmonary hypertension, and pulmonary embolism.
Jugular Venous Pressure
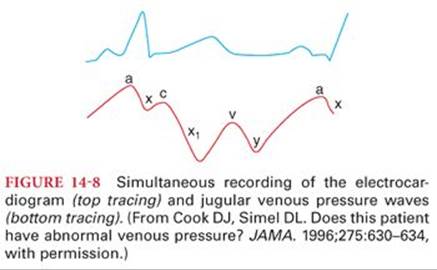
Jugular venous pressure or the pressure in the internal jugular vein mirrors the CVP. The normal jugular venous pressure reflects phasic changes in the right atrium and consists of three positive waves and three negative troughs (Fig. 14-8).11 Abnormalities of these venous waveforms may be useful in the diagnosis of various cardiac conditions (Table 14-4).11

Peripheral Venous Pressure
Large veins offer little resistance to blood flow when they are distended. Most large veins, however, are compressed at multiple extrathoracic sites. For example, pressure in the external jugular vein is often so low that atmospheric pressure on the outside of the neck causes it to collapse. Veins coursing through the abdomen are compressed by intraabdominal pressure, which may increase 15 to 20 mm Hg as a result of pregnancy or ascites. When this occurs, pressure in leg veins must increase above abdominal pressure. It is important to recognize that veins inside the thorax are not collapsed because of the distending effect of negative intrathoracic pressure.
Effect of Hydrostatic Pressure
Pressure in veins below the heart is increased and that in veins above the heart is decreased by the effect of gravity (Fig. 14-9).4 As a guideline, pressure changes 0.77 mm Hg for every centimeter the vessel is above or below the heart. For example, in a standing human, pressure in the veins of the feet is 90 mm Hg because of the distance from the heart to the feet. Conversely, veins above the heart tend to collapse, with the exception being veins inside the skull, where they are held open by surrounding bone. As a result, negative pressure can exist in the dural sinuses and air can be entrained immediately if these sinuses are entered during surgery.

Hydrostatic pressure affects peripheral pressure in arteries and capillaries as well as veins. For example, a standing human who has a systemic blood pressure of 100 mm Hg at the level of the heart has a blood pressure of about 190 mm Hg in the feet.
Venous Valves and the Pump Mechanism
Valves in veins are arranged so that the direction of blood flow can be only toward the heart. In a standing human, movement of the legs compresses skeletal muscles and veins so blood is directed toward the heart. This venous pump or skeletal muscle pump is usually sufficient to maintain venous pressure below 25 mm Hg in a walking human. If an individual stands immobile, the venous pump does not function. As a result, pressures in the veins and capillaries of the legs can increase rapidly, resulting in leakage of fluid from the intravascular space. Indeed, as much as 15% of the blood volume can be lost from the intravascular space in the first 15 minutes of quiet standing.
Varicose Veins
Valves of the venous system can be destroyed when the veins are chronically distended by increased venous pressure as occurs during pregnancy or in an individual who stands most of the day. The end result is varicose veins characterized by bulbous protrusions of the veins beneath the skin of the legs. Venous and capillary pressures remain increased because of the incompetent venous pump, and this causes constant edema in the legs of these individuals. Edema interferes with diffusion of nutrients from the capillaries to tissues, so there is often skeletal muscle discomfort and the skin may ulcerate.
Reference Level for Measuring Venous Pressure
Hydrostatic pressure does not alter venous or arterial pressures that are measured at the level of the tricuspid valve. As a result, the reference point for pressure measurement is considered to be the level of the tricuspid valve. An appropriate external reference point for the level of the tricuspid valve in a supine individual is 5 cm posterior to the sternum at the level of the 4th intercostal space. A precise hydrostatic point to which pressures are referenced is essential for accurate interpretation of venous pressure measurements. For example, each centimeter below the hydrostatic point adds 0.77 mm Hg to the measured pressure, whereas 0.77 mm Hg is subtracted for each centimeter above this point. The potential error introduced by measuring pressures above or below the tricuspid valve is greatest with venous pressures that are normally low. For example, an error introduced by 5 cm of hydrostatic pressure has a much greater influence on the clinical interpretation of CVP than arterial pressure.
The reason for lack of hydrostatic effects at the tricuspid valve is the ability of the right ventricle to act as a regulator of pressure at this site. For example, if the pressure at the tricuspid valve increases, the right ventricle fills to a greater extent, thereby decreasing the pressure at the tricuspid valve toward normal. Conversely, if the pressure decreases at the tricuspid valve, the right ventricle does not fill optimally and blood pools in the veins until pressure at the tricuspid valve again increases to a normal value.
Measurement of right atrial pressure is accomplished by using a transducer or a fluid-filled manometer referenced to the level of the tricuspid valve. A venous pressure measurement in mm Hg can be converted to cm H2O by multiplying the pressure by 1.36, which adjusts for the density of mercury relative to water (10 mm Hg equals 13.6 cm H2O). Conversely, dividing the CVP measurement in cm H2O by 1.36 converts this value to an equivalent pressure in mm Hg.
Blood Viscosity
Blood is a viscous fluid composed of cells and plasma. More than 99% of the cells in plasma are erythrocytes. As a result, leukocytes exert a minimal influence on the physical characteristics of blood. The percentage of blood comprising erythrocytes is the hematocrit, which to a large extent determines the viscosity of blood (Fig. 14-10).4 When the hematocrit increases to 60% to 70%, viscosity of blood is increased about 10-fold compared with water, and flow through blood vessels is greatly decreased. Plasma protein concentrations influence blood viscosity only minimally.

Viscosity exerts fewer effects on blood flow in capillaries than in larger vessels. This most likely reflects alignment of erythrocytes as they pass through small blood vessels rather than the random arrangement characteristic of flow through larger vessels. This alignment of erythrocytes, which greatly decreases the viscous resistance that occurs normally between cells, is largely offset by a decreased velocity of flow that greatly increases viscosity. The net effect may be that viscous effects in small blood vessels are similar to those that occur in large blood vessels.
Plasma is considered extracellular fluid that is identical to interstitial fluid except for the greater concentrations of proteins (albumin, globulin, fibrinogen) in plasma. These greater concentrations reflect the inability of plasma proteins to pass easily through capillaries into the interstitial spaces. The presence of albumin creates colloid osmotic pressure, which prevents fluid from leaving the capillaries.
Determinants of Tissue Blood Flow
Tissue blood flow is directly proportional to the pressure difference between two points (not absolute pressure) and inversely proportional to resistance to flow through the vessel. This relationship between flow, pressure, and resistance can be expressed mathematically as a variant of Ohm’s law, in which blood flow (amperes) is directly proportional to the pressure drop across two points (voltage) and inversely proportional to resistance (Fig. 14-11). Rearrangement of this formula emphasizes that pressure is directly proportional to flow times resistance. Likewise, resistance is directly proportional to pressure and inversely proportional to flow. Furthermore, resistance is directly proportional to viscosity of blood and the length of the vessel and inversely proportional to the fourth power of the radius of the vessel (doubling the radius of the vessel or intravenous catheter size decreases resistance to flow 16-fold [Poiseuille’s law]).

It is important to understand that resistance to blood flow cannot be measured but rather is a calculated value based on measurement of driving pressures and the cardiac output. For example, systemic vascular resistance is calculated as the difference between mean arterial pressure and right atrial pressure divided by cardiac output. Pulmonary vascular resistance is calculated as the difference between mean pulmonary artery pressure and left atrial pressure divided by the cardiac output. Resistance is expressed in dynes/s/cm−5 and is calculated by multiplying the equation for either systemic vascular resistance or pulmonary vascular resistance just described by a conversion factor of 80. Conductance is the reciprocal of resistance and is a measure of the amount of blood flow that can pass through a blood vessel in a given time for a given pressure gradient.
Vascular Distensibility
Blood vessels are distensible such that increases in systemic blood pressure cause the vascular diameter to increase, which in turn decreases resistance to blood flow. Conversely, decreases in intravascular pressure increase the resistance to blood flow. The ability of blood vessels to distend as intravascular pressure increases varies greatly in different parts of the circulation. Anatomically, the walls of arteries are stronger than those of veins. As a result, veins are 6 to 10 times as distensible as arteries. Systemic blood pressure can eventually decrease to a level where intravascular pressure is no longer capable of keeping the vessel open. This pressure averages 20 mm Hg and is defined as the critical closing pressure. When the heart is abruptly stopped, the pressure in the entire circulatory system (mean circulatory pressure) equilibrates at about 7 mm Hg.
Vascular Compliance
Vascular compliance is defined as the increase in volume (capacitance) of a vessel produced by an increase in intravascular pressure. The compliance of the entire circulatory system is estimated to be 100 mL for each 1 mm Hg increase in intravascular pressure.4 The compliance of veins is much greater than that of arteries. For example, the volume of blood normally present in all veins is about 2,500 mL, whereas the arterial system contains only about 750 mL of blood when the mean arterial pressure is 100 mm Hg. Sympathetic nervous system activity can greatly alter the distribution of blood volume. Enhancement of sympathetic nervous outflow to the blood vessels, especially the veins, decreases the dimensions to the circulatory system, and the circulation continues to function almost normally even when as much as 25% of the total blood volume has been lost. Vasoconstriction or vasodilation refers to resistance changes in arterioles, whereas changes in the caliber of veins are described as venoconstriction or venodilation.
Control of Tissue Blood Flow
Control of blood flow to different tissues includes local mechanisms, autonomic nervous system responses, and release of hormones. Total tissue blood flow or cardiac output is about 5 L per minute, with large amounts being delivered to the heart, brain, liver, and kidneys (Table 14-5).4 In contrast, skeletal muscles represent 35% to 40% of body mass but receive only about 15% of the total cardiac output, reflecting the low metabolic rate of inactive skeletal muscles.

Local Control of Blood Flow
Local control of blood flow is most often based on the need for delivery of oxygen or other nutrients such as glucose or fatty acids to the tissues. The response to decreased oxygen delivery may reflect the local release of vasodilatory substances (adenosine, lactic acid, carbon dioxide, potassium ions), which results in increased tissue blood flow and oxygen delivery.
Autoregulation of Blood Flow
Autoregulation is a local mechanism that controls blood flow in which a specific tissue is able to maintain a relatively constant blood flow over a wide range of mean arterial pressures. When the mean arterial pressure increases, the associated increase in tissue blood flow causes the blood vessels to constrict, thereby limiting any increase in blood flow. Conversely, decreases in mean arterial pressure result in vasodilation, which maintains tissue blood flow. Autoregulatory responses to sudden changes in mean arterial pressure occur within 60 to 120 seconds. The ability of autoregulation to return local tissue blood flow to normal is incomplete.
Long-term Control of Blood Flow
Long-term regulatory mechanisms that return local tissue blood flow to normal involve a change in vascularity of tissues. For example, sustained increases in mean arterial pressure to specific tissues, as occurs above a coarctation of the aorta, is accompanied by a decrease in the size and number of blood vessels. Likewise, if metabolism in a tissue becomes chronically increased, vascularity increases, or, if metabolism is decreased, vascularity decreases. Indeed, inadequate delivery of oxygen to a tissue is the stimulus for the development of collateral vessels. Neonates exposed to increased concentrations of oxygen can manifest cessation of new vascular growth in the retina. Subsequent removal of the neonate from a high-oxygen environment causes an overgrowth of new vessels to offset the abrupt decrease in availability of oxygen. There may be so much overgrowth that the new vessels cause blindness (retrolental fibroplasia).
Autonomic Nervous System Control of Blood Flow
Autonomic nervous system control of blood flow is characterized by a rapid response time (within 1 second) and an ability to regulate blood flow to certain tissues at the expense of other tissues. The sympathetic nervous system is the most important component of the autonomic nervous system in the regulation of blood flow; sympathetic stimulation causes release of norepinephrine, which stimulates α-adrenergic receptors to produce vasoconstriction. Constriction of small arteries influences resistance to blood flow through tissues, whereas venoconstriction alters vascular capacitance and distribution of blood in the peripheral circulation. Sympathetic nervous system innervation is prominent in the kidneys and skin and minimal in the cerebral circulation.
Vasomotor Center
The vasomotor center, which is located in the pons and medulla, transmits sympathetic nervous system impulses through the spinal cord to all blood vessels. Evidence for a continuous, sustained state of partial vasoconstriction (vasomotor tone) is the abrupt decrease in systemic blood pressure that occurs when sympathetic nervous system innervation to the vasculature is abruptly interrupted, as by traumatic spinal cord transection or regional anesthesia. Activity of the vasomotor center can be influenced by impulses from a number of sites, including diffuse areas of the reticular activating system, hypothalamus, and cerebral cortex. Sympathetic nervous system impulses are transmitted to the adrenal medulla at the same time they are transmitted to the peripheral vasculature. These impulses stimulate the adrenal medulla to secrete epinephrine and norepinephrine into the circulation, where they act directly on adrenergic receptors in the walls of vascular smooth muscle.
The medial and lower portions of the vasomotor center do not participate in transmission of vasoconstrictor impulses but rather function as an inhibitor of sympathetic nervous system activity, which allows blood vessels to dilate. Conceptually, this portion of the vasomotor center is functioning as the parasympathetic nervous system.
Mass Reflex
The mass reflex is characterized by stimulation of all portions of the vasomotor center, resulting in generalized vasoconstriction and an increase in cardiac output in an attempt to maintain tissue blood flow. The alarm reaction resembles the mass reflex, but associated skeletal muscle vasodilation and psychic excitement are intended to prepare the individual to confront a life-threatening situation.
Syncope
Emotional fainting (vasovagal syncope) may reflect profound skeletal muscle vasodilation such that systemic blood pressure decreases abruptly and syncope occurs. Associated vagal stimulation results in bradycardia. This phenomenon may occur in patients who have an intense fear of needles, resulting in syncope during placement of an intravenous catheter.
Hormone Control of Blood Flow
Vasoconstrictor hormones that may influence local tissue blood flow include epinephrine, norepinephrine, angiotensin, and arginine vasopressin (formerly known as antidiuretic hormone). Bradykinin, serotonin, histamine, prostaglandins, and low circulating concentrations of epinephrine are vasodilating substances. Local chemical factors, such as accumulation of hydrogen ions, potassium ions, and carbon dioxide, relax vascular smooth muscle and cause vasodilation. Carbon dioxide also has an indirect vasoconstrictor effect because it stimulates the outflow of sympathetic nervous system impulses from the vasomotor center.
Regulation of Systemic Blood Pressure
Systemic blood pressure is maintained over a narrow range by reciprocal changes in cardiac output and systemic vascular resistance. The autonomic nervous system and baroreceptors play a key role in moment-to-moment regulation of systemic blood pressure. Long-term regulation of blood pressure depends on control of fluid balance by the kidneys, adrenal cortex, and central nervous system.
Systolic, diastolic, and mean arterial pressure tends to increase progressively with age. Because a greater portion of the cardiac cycle is nearer the diastolic blood pressure, it follows that mean arterial pressure is not the arithmetic average of the systolic and diastolic blood pressures. Mean arterial blood pressure is the most important determinant of tissue blood flow because it is the average, tending to drive blood through the systemic circulation.
Rapid-Acting Mechanisms for the Regulation of Systemic Blood Pressure
Rapid-acting mechanisms for regulation of systemic blood pressure involve nervous system responses as reflected by the baroreceptor reflexes, chemoreceptor reflexes, atrial reflexes, and central nervous system ischemic reflex. These reflex mechanisms respond almost immediately to changes in systemic blood pressure. Furthermore, within about 30 minutes, these nervous system reflex responses are further supplemented by activation of hormonal mechanisms and shift of fluid into the circulation to readjust the blood volume. These short-term mechanisms can return systemic blood pressure toward but never entirely back to normal. Indeed, the impact of many of the rapid-acting regulatory mechanisms, such as the baroreceptor reflexes, diminishes with time as these mechanisms adapt to the new level of systemic blood pressure.
Baroreceptor Reflexes
Baroreceptors are nerve endings in the walls of large arteries in the neck and thorax, especially in the internal carotid arteries just above the carotid bifurcation and in the arch of the aorta (Fig. 14-12).12 These nerve endings respond rapidly to changes in systemic blood pressure and are crucial for maintaining normal blood pressure when an individual changes from the supine to standing position. An increase in mean arterial pressure produces stretch of baroreceptor nerve endings, and increased numbers of nerve impulses are transmitted to the depressor portion of the vasomotor center, leading to a relative decrease in the central nervous system outflow of sympathetic nervous system (vasoconstrictive) impulses (Fig. 14-13).12 The net effects are vasodilation throughout the peripheral circulation, decreased heart rate, and decreased myocardial contractility, which all act to decrease systemic blood pressure back toward normal. Conversely, decreases in systemic blood pressure reflexively produce changes likely to increase blood pressure. Baroreceptors adapt in 1 to 3 days to sustained changes in systemic blood pressure, emphasizing that these reflexes are probably of no importance in long-term regulation of blood pressure. Volatile anesthetics, particularly halothane, inhibit the heart rate response portion of the baroreceptor reflex that occurs in response to changes in systemic blood pressure (see Chapter 4).

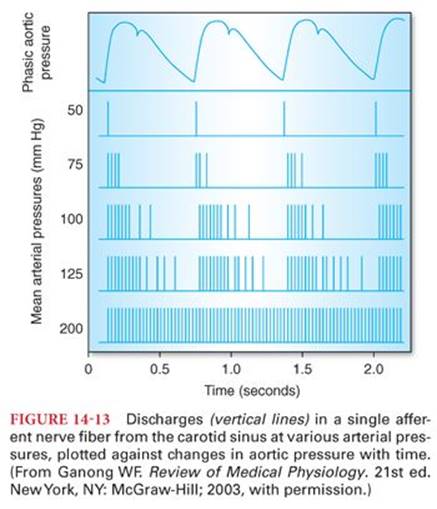
Chemoreceptor Reflexes
Chemoreceptors are cells that transduce chemical signals into nerve impulses. There are chemoreceptors located in the carotid bodies and aortic body (see Fig. 14-12).12 Each carotid or aortic body is supplied with an abundant blood flow through a nutrient artery so that the chemoreceptors are always exposed to oxygenated blood. When the systemic blood pressure, and thus the blood flow, decrease below a critical level, the chemoreceptors in the carotid body are stimulated by decreased availability of oxygen and also because of excess carbon dioxide and hydrogen ions that are not removed by the sluggish blood flow. Impulses from the chemoreceptors are transmitted to the vasomotor center, which results in reflex changes that tend to increase systemic blood pressure back toward the normal. Chemoreceptors do not respond strongly until systemic blood pressure decreases below 80 mm Hg. Chemoreceptors are more important in stimulating breathing when the PaO2 decreases below 60 mm Hg (ventilatory response to arterial hypoxemia). The ventilatory response to arterial hypoxemia is inhibited by subanesthetic concentrations of most of the volatile anesthetics (0.1 minimum alveolar concentration) as well as injected drugs such as barbiturates and opioids (see Chapters 4, 5, and 7).
Bezold-Jarisch Reflex
The Bezold-Jarisch reflex is a circulatory response whereby a decrease in left ventricular volume activates receptors that cause a paradoxical bradycardia. This compensatory decrease in heart rate allows for increased ventricular filling but may also exacerbate hypotension. The bradycardia and hypotension that can occur during spinal or epidural anesthesia have been attributed to this reflex.
Atrial Reflexes
The atria contain low-pressure atrial stretch receptors similar to baroreceptors in large arteries. Stretching of the atria evokes reflex vasodilation and decreases the systemic blood pressure back toward the normal level. An increase in atrial pressure also causes an increase in heart rate (Bainbridge reflex) due to a direct effect of the increased atrial volume on stretch of baroreceptors located in both atria at the venoatrial junctions. The increase in heart rate evoked by stretching of the atria prevents accumulation of blood in the atria, veins, or pulmonary circulation.
Central Nervous System Ischemic Reflex
The central nervous system ischemic reflex response occurs when blood flow to the medullary vasomotor center is decreased to the extent that ischemia of this vital center occurs. As a result of this ischemia, there is an intense outpouring of sympathetic nervous system activity, resulting in profound increases in systemic blood pressure. It is believed that this reflex response is caused by failure of slowly flowing blood to remove carbon dioxide from the vasomotor center. The central nervous system reflex response does not become highly active until mean arterial pressure decreases to less than 50 mm Hg and reaches its greatest degree of stimulation at systemic blood pressures of 15 to 20 mm Hg. This reflex response is not useful for regulation of normal blood pressure but rather acts as an emergency control system to prevent further decreases in systemic blood pressure when cerebral blood flow is dangerously decreased.
Cushing Reflex
The Cushing reflex is a central nervous system ischemic reflex response that results from increased intracranial pressure. When intracranial pressure increases to equal arterial pressure, the Cushing reflex acts to increase systemic blood pressure above intracranial pressure. Cushing’s triad is defined as having (a) hypertension, (b) bradycardia, and (c) irregular respirations (due to brainstem dysfunction). The latter is not often seen in this era, as most patients with severe intracranial hypertension are now mechanically ventilated.
Respiratory Variations in Systemic Blood Pressure
Systemic blood pressure normally varies by 4 to 6 mm Hg in a wavelike manner during quiet spontaneous breathing. Systemic blood pressure is increased during end-inspiration and the beginning of exhalation and decreased during the remainder of the breathing cycle. This is due to increased venous return to the right heart during inspiration, which takes a few cardiac cycles to be transmitted to the left heart. Positive pressure ventilation of the lungs produces a reversed sequence of blood pressure change because the initial positive airway pressure simultaneously pushes more blood toward the left ventricle causing an increase in pressure; this is followed by a decrease in left ventricular filling due to the decrease in venous return to the right heart caused by the positive intrathoracic pressure.
Continuous or beat-to-beat monitoring of the changes in arterial blood pressure, pulse pressure, and stroke volume occurring during mechanical ventilation may provide an indication of the patient’s ability to respond to volume administration with an increase in cardiac output or “fluid responsiveness.” Respiratory variation in these parameters of more than 12% to 15% generally indicates fluid responsiveness. When the chest is closed and an adequate tidal volume is employed, “goal-directed” fluid resuscitation can be guided by such measurements.
Heart Rate Variability
Variations in heart rate occur during normal respiration, whereby inspiration increases heart rate and expiration decreases it. High-frequency heart rate variability is controlled by autonomic reflexes mediated by neural input to the heart supplied by the vagus nerve. Low-frequency heart rate variability results from the interaction between parasympathetic and sympathetic tone. Analysis of heart rate variability provides information regarding the integrity of the autonomic nervous system. High heart rate variability is a sign of good health and conversely, low heart rate variability can be a manifestation of disease (myocardial infarction, heart failure, neuropathy) and occurs universally following the denervation that occurs during cardiac transplantation.13
Systemic Blood Pressure Vasomotor Waves
Cyclic increases and decreases in systemic blood pressure lasting 7 to 10 seconds are referred to as vasomotor or Traube-Hering waves. The presumed cause of vasomotor waves is oscillation in the reflex activity of baroreceptors. For example, increased systemic blood pressure stimulates baroreceptors, which then inhibit the sympathetic nervous system, causing a decrease in systemic blood pressure. Decreased systemic blood pressure decreases baroreceptor activity and allows the vasomotor center to become active once again, increasing the systemic blood pressure to a higher value.
Moderately Rapid-Acting Mechanisms for the Regulation of Systemic Blood Pressure
There are at least three hormonal mechanisms that provide either rapid or moderately rapid control of systemic blood pressure. These hormonal mechanisms are catecholamine-induced vasoconstriction, renin-angiotensin–induced vasoconstriction, and vasoconstriction induced by arginine vasopressin, all of which increase systemic blood pressure by increasing systemic vascular resistance. Circulating catecholamines may even reach parts of the circulation that are devoid of sympathetic nervous system innervation, such as metarterioles. Renin-angiotensin–induced vasoconstriction manifests to a greater degree on arterioles than veins and requires about 20 minutes to become fully active.
In addition to hormonal mechanisms, there are two intrinsic mechanisms (capillary fluid shift and stress-relaxation of blood vessels), which begin to react within minutes of changes in systemic blood pressure. For example, changes in systemic blood pressure produce corresponding changes in capillary pressure, thus allowing fluid to enter or leave the capillaries to maintain a constant blood volume. Stress-relaxation is the gradual change in blood vessel size to adapt to changes in systemic blood pressure and the amount of blood that is available. The stress-relaxation mechanism has definite limitations such that increases in blood volume greater than about 30% or decreases of more than about 15% cannot be corrected by this mechanism alone.
Long-term Mechanisms for the Regulation of Systemic Blood Pressure
Long-term mechanisms for the regulation of systemic blood pressure, unlike the short-term regulatory mechanisms, have a delayed onset but do not adapt, providing a sustained regulatory effect on systemic blood pressure. The renal–body fluid system plays a predominant role in long-term control of systemic blood pressure because it controls both the cardiac output and systemic vascular resistance. This crucial role is supplemented by accessory mechanisms, including the renin-angiotensin-aldosterone system and arginine vasopressin regulation.
Renal–Body Fluid System
Increased systemic blood pressure, as provoked by modest increases in blood volume, results in sodium ion and water excretion by the kidneys. The resultant decrease in blood volume leads to decreases in cardiac output and systemic blood pressure. After several weeks, the cardiac output returns toward normal, and systemic vascular resistance decreases to maintain the lower but more acceptable blood pressure. Conversely, a decrease in systemic blood pressure stimulates the kidneys to retain fluid. A special feature of this regulatory mechanism is its ability to return systemic blood pressure completely back to normal values. This contrasts with rapid-acting to moderately rapid-acting mechanisms, which cannot return systemic blood pressure entirely back to normal.
Renin-Angiotensin System
Aldosterone secretion that results from the action of angiotensin II on the adrenal cortex exerts a long-term effect on systemic blood pressure by stimulating the kidneys to retain sodium and water. The resulting increase in extracellular fluid volume causes cardiac output, and subsequently systemic blood pressure, to increase.
Regulation of Cardiac Output and Venous Return
Cardiac output is the amount of blood pumped by the left ventricle into the aorta each minute (product of stroke volume and heart rate), and venous return is the amount of blood flowing from the veins into the right atrium each minute. Because the circulation is a closed circuit, the cardiac output must equal venous return. Cardiac output for the average person weighing 70 kg and with a body surface area of 1.7 m2 is about 5 L per minute. This value is about 10% less in women.
Determinants of Cardiac Output
Venous return is the main determinant of cardiac output. The metabolic requirements of tissues control cardiac output through alterations in resistance to tissue blood flow. For example, increased local metabolic needs lead to regional vasodilation, with a resulting increase in tissue blood flow and thus venous return. Cardiac output is increased by an amount equivalent to the venous return.
Any factor that interferes with venous return can lead to decreased cardiac output. Hemorrhage decreases blood volume such that venous return decreases and cardiac output decreases. Acute venodilation, such as that produced by spinal anesthesia and accompanying sympathetic nervous system blockade, can so increase the capacitance of peripheral vessels that venous return is reduced and cardiac output declines. Restoration of venous tone (with a vasoconstrictor) and/or administration of fluid can restore cardiac output. Positive pressure ventilation of the lungs, particularly in the presence of a decreased blood volume, causes a decrease in venous return and cardiac output.
Factors that increase cardiac output are associated with decreases in systemic vascular resistance. For example, anemia decreases the viscosity of blood, leading to a decrease in systemic vascular resistance and increase in venous return. An increased blood volume increases cardiac output by increasing the gradient for flow to the right atrium and by distending blood vessels, which decreases resistance to blood flow. Increased cardiac output caused by an increased blood volume lasts only 20 to 40 minutes because increased capillary pressures cause intravascular fluid to enter tissues, thereby returning blood volume to normal. Furthermore, increased pressure in veins caused by the increased blood volume causes the veins to distend (stress-relaxation). Cardiac output increases during exercise, in hyperthyroidism, and in the presence of arteriovenous shunts associated with hemodialysis, reflecting decreases in systemic vascular resistance.
Sympathetic nervous system stimulation increases myocardial contractility and heart rate to increase cardiac output beyond that possible from venous return alone. Maximal stimulation by the sympathetic nervous system can double cardiac output. Nevertheless, this sympathetic nervous system–induced increase of cardiac output is only transient, despite sustained increases in nervous system activity. A reason for this transient effect is autoregulation of tissue blood flow, which manifests as vasoconstriction to decrease venous return and thus decrease cardiac output back toward normal. In addition, increased systemic blood pressure associated with increases in the cardiac output causes fluid to leave the capillaries, thereby decreasing blood volume, venous return, and cardiac output.
An increase in myocardial contractility or inotropy can increase the stroke volume and thereby the cardiac output. Cardiac muscle is sensitive to calcium; most hormones and drugs which increase contractility augment intracellular calcium use. One newer agent available outside of the United States that is used to provide inotropic support in severe heart failure (levosimendan) sensitizes the myofibrils to calcium. Decreased contractility is caused by many anesthetic agents. Strictly speaking, changes in inotropic state should be defined in the absence of changes in preload and afterload; because many inotropic drugs affect these parameters as well, changes in cardiac output are not always due only to the inotropic activity.
Ventricular Function Curves
Ventricular function curves (Frank-Starling curves) depict the cardiac output at different atrial (ventricular end diastolic) filling pressures (Fig. 14-14). Clinically, ventricular function curves are used to estimate myocardial contractility. Improved cardiac function (sympathetic nervous system stimulation) is characterized by a shift of the cardiac output curve to the left of the normal curve (greater cardiac output for a given filling pressure), whereas a shift of the curve to the right of normal (myocardial infarction, cardiomyopathy) reflects decreased cardiac function. Even with a normal ventricular function curve, as preload is increased, a point is reached where further stretching of the cardiac muscle results in no further increase and eventually to a decrease in cardiac output.

Pressure–Volume Loops
Pressure–volume loops describe the dynamic characteristics of cardiac function (Fig. 14-15). If ventricular pressure is plotted against ventricular volume, each cardiac cycle can be depicted by a pressure–volume loop.

Shock Syndromes
Circulatory shock is characterized by inadequate tissue blood flow and oxygen delivery to cells resulting in generalized deterioration of cellular and organ function. Inadequate tissue flow is due to inadequate cardiac output and can result from decreased venous return, myocardial depression, or both. Cellular metabolism is depressed, and the amount of heat liberated is decreased resulting in a decreased body temperature particularly when the ambient environment is cold. In the early stages of shock, consciousness is usually maintained, although mental clarity may be impaired. Consciousness is likely to be lost as shock progresses. Low cardiac output greatly decreases urine output, eventually leading to anuria as glomerular pressure decreases below the critical value required for filtration of fluid into Bowman’s capsule. Furthermore, the kidneys have such a high rate of metabolism that decreased renal blood flow may cause acute tubular necrosis (see Chapter 16). An important feature of persistent shock is eventual progressive deterioration of the heart. In addition to myocardial depression caused by decreased coronary artery blood flow, myocardial function can also be depressed by lactic acid, bacterial endotoxins, and myocardial depressant factor released from an ischemic pancreas.
Hemorrhagic Shock
Hemorrhage is the most common cause of shock due to decreased venous return. Any decrease in systemic blood pressure initiates powerful baroreceptor-mediated increases in sympathetic nervous system activity, manifesting as arterial constriction, venoconstriction, and direct myocardial stimulation. Venoconstriction is particularly important for sustaining venous return to the heart and, thus, maintaining cardiac output. Arterial constriction is responsible for initially maintaining systemic blood pressure despite decreases in cardiac output. This maintenance of systemic blood pressure sustains cerebral and coronary artery blood flow as significant vasoconstriction does not occur in these organs. In other organs, such as the kidneys, intense sympathetic nervous system–mediated vasoconstriction may decrease blood flow dramatically.
Nonhemorrhagic Hypovolemic Shock
Loss of plasma volume from the circulation can result in shock similar to that produced by hemorrhage. Intestinal obstruction results in fluid loss into the gastrointestinal tract and reduction in plasma volume. Severe burns may also be associated with sufficient loss of plasma volume to result in shock. Severe dehydration from any cause can also lead to hypovolemic shock due to reduction in plasma volume. Hypovolemic shock that results from a reduction in plasma volume has the same clinical characteristics as hemorrhagic shock except that selective reduction of the plasma volume greatly increases the viscosity of blood and exacerbates sluggish blood flow.
Neurogenic Shock
Neurogenic shock occurs in the absence of blood loss when vascular capacity increases so greatly that even a normal blood volume is not capable of maintaining venous return and cardiac output. Common causes of loss of vasomotor tone and subsequent neurogenic shock are traumatic transection of the spinal cord and acute blockade of the peripheral sympathetic nervous system by spinal or epidural anesthesia.
Septic Shock
Septic shock is characterized by profound peripheral vasodilation, increased cardiac output secondary to decreased systemic vascular resistance, increased vascular permeability with fluid loss from the vascular compartment, and development of disseminated intravascular coagulation. Septic shock is most commonly caused by gram-positive bacteria and less commonly by endotoxin-producing gram-negative bacteria. Endotoxins are bacterial membrane lipopolysaccharides that are made up of a toxic fatty acid (lipid A) core and a complex polysaccharide coat. Analogous molecules in the walls of gram-positive bacteria and fungi can also cause septic shock. The septic response is likely to reflect a systemic inflammatory response produced by exposure to bacterial cell products that ultimately lead to a progressively dysfunctional host response and multisystem organ failure. Elderly patients and those with immunosuppression are vulnerable to the development of sepsis and associated septic shock. The end stages of septic shock are not greatly different from the end stages of hemorrhagic shock, even though the initiating factors are markedly different. Mortality approaches 50% in septic shock despite significant improvements in supportive care.14
Measurement of Cardiac Output
The management of patients in the operating room and intensive care unit involves therapeutic interventions intended to optimize tissue oxygen delivery. Common interventions include fluid administration, blood transfusion, inotrope and vasoactive pharmacotherapy, heart rate and rhythm manipulation, mechanical assist devices, and mechanical ventilation. Ideally, these interventions should be guided by the measurement of cardiac output. However, cardiac output determination has traditionally required invasive techniques such as the insertion of pulmonary artery catheters. Recent advances in microprocessor technology, a greater awareness of the limitations and hazards of pulmonary artery catheter insertion, and a need to measure cardiac output in out of operating room settings such as the emergency department, have led to the development of newer monitoring techniques. The general trend has been to develop techniques that are less invasive and allow more frequent, or even continuous, measurement of cardiac output.15 Nevertheless, the pulmonary artery thermodilution technique represents the clinical standard against which new techniques are compared.16 Currently used methods of cardiac output measurement include Fick methods, indicator dilution methods, thermodilution, echocardiographic techniques, impedance cardiography, and pulse contour analysis.
Fick Method
Adolf Fick first described use of the “Fick principle” to estimate cardiac output in 1870. Cardiac output is estimated by dividing the oxygen consumption by the arteriovenous difference for oxygen (Fig. 14-16).4 Oxygen consumption is usually measured by a respirometer containing a known oxygen mixture. The patient’s exhaled gases are collected in a large inflatable reservoir (Douglas bag). The volume and oxygen concentrations of the exhaled gases are measured, allowing calculation of oxygen consumption. Venous blood used for calculation of oxygen content must be obtained from the right ventricle, or, ideally, the pulmonary artery, to ensure adequate mixing. Blood from the right atrium may not yet be adequately mixed to provide a true mixed venous sample. Blood used for determining the oxygen saturation in arterial blood can be obtained from any artery because all arterial blood is thoroughly mixed before it leaves the heart and therefore has the same concentration of oxygen.
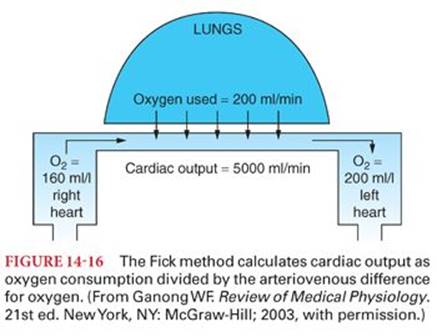
The Fick principle can also be applied to carbon dioxide elimination from the lung.17 The NICO (Novametrix Medical Systems, Inc., Wallingford, CT) is a continuous noninvasive monitor of cardiac output. By intermittently adding an additional dead space to the breathing circuit, CO2 elimination is reduced thus increasing end-tidal CO2. The change in end-tidal CO2 is used to estimate the change in arterial CO2content. This proprietary system uses these data in a modified Fick equation to calculate cardiac output.
Indicator Dilution Method
In measuring the cardiac output by the indicator dilution method, a nondiffusible dye (indocyanine green) is injected into the right atrium (or central venous circulation), and the concentration of dye is subsequently measured continuously in the arterial circulation by a spectrophotometer. The area under the resulting time-concentration curve before recirculation of the dye occurs, combined with knowing the amount of dye injected, allows calculation of the pulmonary blood flow, which is the same as the cardiac output. It is necessary to extrapolate the dye curve to zero because recirculation of the dye occurs before the down slope of the curve reaches baseline. Early recirculation of the dye may indicate the presence of a right-to-left intracardiac shunt (foramen ovale), permitting direct passage of a portion of the dye to the left side of the heart without first passing through the lungs.
Thermodilution Method
A bolus of cold fluid may also be considered an indicator and used in the indicator dilution technique. A pulmonary artery catheter with ports in the right atrium and pulmonary artery and a temperature sensor on the distal port is used to measure thermodilution cardiac outputs. Thermodilution cardiac outputs are determined by measuring the change in blood temperature between two points (right atrium and pulmonary artery) after injection of a known volume of cold saline solution at the proximal right atrial port. The change in blood temperature as measured at the distal pulmonary artery port is inversely proportional to pulmonary blood flow (the extent to which the cold saline solution is diluted by blood), which is equivalent to cardiac output. The area under the temperature-time curve is converted to its equivalent in cardiac output. Advantages of this technique compared with the indicator dilution method include dissipation of cold in tissues so recirculation is not a problem, and safety of repeated and frequent measurements because saline is innocuous. Recent advances in thermodilution techniques include semicontinuous cardiac output determination using a heat-generating thermal filament incorporated into the pulmonary artery catheter. It is necessary to use sophisticated signal processing strategies to measure the downstream temperature because the thermal filament must generate only a modest level of heat for safety.
Echocardiographic Techniques
Echocardiography can be used to estimate cardiac output by combining the Doppler principle to determine the velocity of blood in the aorta with two-dimensional views to determine aortic diameter.15Conventional transesophageal or transthoracic echocardiographic techniques have the advantage that systolic and diastolic function, volume status, regional wall abnormalities, valve function, and the presence of pericardial effusion may also be evaluated. However, this technique requires significant operator expertise. More recently, Doppler techniques have been developed using transesophageal probes designed solely for the purpose of estimating cardiac output. Aortic dimensions are not measured but are estimated from age-, sex-, and body size–specific nomograms derived from large population studies. Transesophageal Doppler estimates of cardiac output require minimal operator training and allow rapid cardiac output estimation.
Impedance Cardiography
The thorax is a conductor whose impedance is altered by changes in blood volume and velocity with each cardiac cycle. Impedance cardiography is based on the principle of thoracic electrical bioimpedance and involves the placement of electrodes to allow the transmission of current and measurement of voltage across the chest.18 Thus, thoracic electrical bioimpedance techniques can be used to noninvasively estimate cardiac output. However, the reliability of this technique is limited under several circumstances including patient movement, poor electrocardiogram signal quality, cardiac tachydysrhythmias, excessive thoracic fluid, and open chest wounds with metal retractors.
Pulse Contour Analysis
The first attempt to determine cardiac output from analysis of the pulse contour was made in 1904. The aortic pressure waveform is a function of the stroke volume and its interaction with the vascular tree. The arterial pulse contour can be modeled in a manner analogous to an electrical circuit that has specific values for resistance, compliance, and impedance.17 Thus, the flow within that system can be estimated from the pressure waveform that is generated. The validity of this system is improved by calibration using a separate cardiac output estimation technique. However, in clinical practice, reliable use of this technique requires frequent recalibration because the peripheral circulation undergoes significant changes in arteriolar tone in response to physiologic and pharmacologic stimuli.
Microcirculation
The circulation exists to supply tissues with blood in amounts commensurate with their needs for oxygen and nutrients.4 The microcirculation is defined as the circulation of blood through the smallest vessels of the body—arterioles, capillaries, and venules. Capillaries, whose walls consist of a single layer of endothelial cells, serve as the site for the rapid transfer of oxygen and nutrients to tissues and receipt of metabolic byproducts. There are an estimated 10 billion capillaries providing a total surface area that exceeds 6,300 m2 for nutrient exchange. Capillary density varies from tissue to tissue. Capillaries are numerous in metabolically active tissues, such as cardiac and skeletal muscles, whereas in less active tissues, capillary density is low. Nevertheless, it is unlikely that any functional cell is greater than 50 µm away from a capillary. The muscular arterioles serve as the major resistance vessels and regulate regional blood flow to the capillary beds. Venules act primarily as collecting channels and storage vessels.
Anatomy of the Microcirculation
Arterioles give rise to metarterioles, which give rise to capillaries (Table 14-6) (Fig. 14-17).12 Other metarterioles serve as thoroughfare channels to the venules, bypassing the capillary bed. Capillaries drain via short collecting venules to the venules. Blood flow through capillaries is regulated by muscular precapillary sphincters present at the capillary opening. The arterioles, metarterioles, and venules contain smooth muscle. As a result, the arterioles serve as the major resistance vessels and regulate regional blood flow to the capillary beds, whereas the venules and veins serve primarily as collecting channels and storage or capacitance vessels.


Capillary walls are about 1 µm thick, consisting of a single layer of endothelial cells surrounded by a thin basement membrane on the outside (Fig. 14-18).12 The structure of the capillary wall varies from tissue to tissue, but in many organs, including those in skeletal, cardiac, and smooth muscle, the interdigitated junction between endothelial cells allows passage of molecules up to 10 nm in diameter. In addition, the cytoplasm of endothelial cells is attenuated to form gaps or pores that are 20 to 100 nm in diameter. These pores permit the passage of relatively large molecules. It also appears that plasma and its dissolved proteins are taken up by endocytosis, transported across endothelial cells, and discharged by exocytosis into the interstitial fluid. In the brain, the capillaries resemble those in skeletal muscles, except the interdigitated junctions between endothelial cells are tighter (blood–brain barrier), permitting passage of only small molecules.
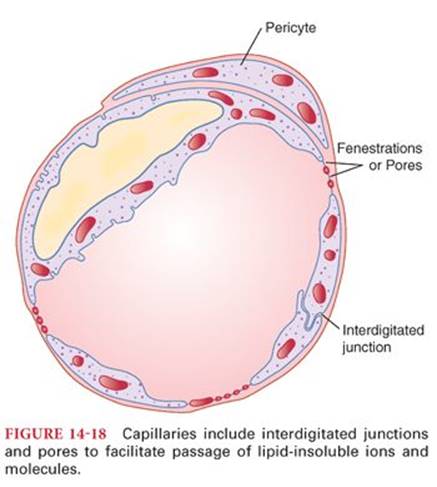
The diameter of capillary pores is about 25 times the diameter of water molecules (0.3 nm), which are the smallest molecules that normally pass through capillary channels. Plasma proteins have diameters that exceed the width of capillary pores. Other substances, such as sodium, potassium, and chloride ions and glucose, have intermediate diameters (0.39 to 0.86 nm) such that permeability of capillary pores for different substances varies according to their molecular weights (Table 14-7). Oxygen and carbon dioxide are both lipid soluble and readily pass through endothelial cells.

True capillaries are devoid of smooth muscle and are therefore incapable of active constriction. Nevertheless, the endothelial cells that form them contain actin and myosin and can alter their shape in response to certain chemical stimuli. The diameter of capillaries (7 to 9 µm) is just sufficient to permit erythrocytes to squeeze through in single file. The thin walls of capillaries are able to withstand high intraluminal pressures because their small diameter prevents excessive wall tension (Laplace law).
Blood Flow in Capillaries
Blood flow in capillaries is approximately 1 mm per second and is intermittent rather than continuous. This intermittent blood flow reflects contraction and relaxation of metarterioles and precapillary sphincters in alternating cycles 6 to 12 times per minute.19 The phenomenon of alternating contraction and relaxation is known as vasomotion. Oxygen is the most important determinant of the degree of opening and closing of metarterioles and precapillary sphincters. A low PO2 allows more blood to flow through capillaries to supply tissues. In this regard, the impact of oxygen on capillary blood flow provides a form of autoregulation of tissue blood flow.
In addition to nutritive blood flow through tissues that is regulated by oxygen, there is also nonnutritive (shunt) blood flow regulated by the autonomic nervous system. The nonnutritive blood flow is characterized by direct vascular connections between arterioles and venules. Some of these arteriovenous connections have muscular coverings so blood flow can be altered over a wide range. In some parts of the skin, these arteriovenous anastomoses provide a mechanism to permit rapid inflow of arterial blood to warm the skin and dissipate the heat.
Vasoactive Role of the Capillary Endothelium
The notion that the endothelium of capillaries is an inert single layer of cells serving only as a passive filter to permit passage of water and small molecules across the blood vessel wall is no longer considered valid.20 Rather, the endothelium is now recognized as an important source of substances that cause contraction or relaxation of vascular smooth muscle.
One of these substances is prostacyclin that can relax vascular smooth muscle via an increase in cyclic adenosine monophosphate concentration. Prostacyclin is formed in the endothelium from arachidonic acid and the reaction is catalyzed by prostacyclin synthase. The principal function of prostacyclin is to inhibit platelet adherence to the endothelium and platelet aggregation and thus prevent intravascular clot formation.
The formation and release of nitric oxide (NO) is also important in the endothelium-mediated vascular dilation.21 NO is released when endothelial cells are stimulated by acetylcholine or other vasodilator substances (adenosine triphosphate, bradykinin, serotonin, substance P, and histamine). NO release can be stimulated by the shear stress of blood flow on the endothelium. A vasoconstrictor peptide synthesized by the capillary endothelium is endothelin. Endothelin may affect vascular tone and blood pressure.
Fluid Movement across Capillary Membranes
Solvent and solute movement across capillary endothelial cells occurs by filtration, diffusion, and pinocytosis via endothelial vesicles.22 Diffusion is the most important process for transcapillary exchange and pinocytosis is the least important. It is important to distinguish between filtration and diffusion through capillary membranes. Filtration is the net outward movement of fluid at the arterial end of capillaries. Diffusion of fluid occurs in both directions across capillary membranes.
Filtration
The four pressures that determine whether fluid will move outward across capillary membranes (filtration) or inward across capillary membranes (reabsorption) are capillary pressure, interstitial fluid pressure, plasma colloid osmotic pressure, and interstitial fluid colloid osmotic pressure. The net effect of these four pressures is a positive filtration pressure at the arterial end of capillaries, causing fluid to move outward across cell membranes into interstitial fluid spaces (Table 14-8). At the venous end of capillaries, the net effect of these four pressures is a positive reabsorption pressure causing fluid to move inward across capillary membranes into capillaries (Table 14-9). Overall, the mean values of the four pressures acting across capillary membranes are nearly identical such that the amount of fluid filtered nearly equals the amount reabsorbed (Table 14-10). Any fluid that is not reabsorbed enters the lymph vessels.
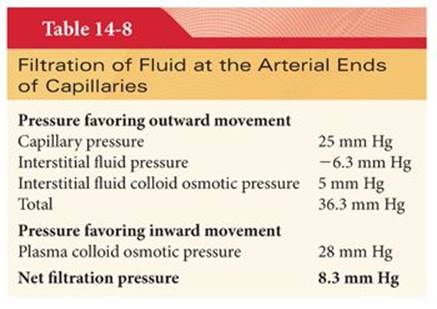


Traditionally, filtration has been considered to occur at the arterial end of the capillary and absorption to occur at the venous end because of the gradient of hydrostatic pressure along the capillary. Nevertheless, many capillaries only filter, whereas others only absorb. In some vascular beds such as the renal glomerulus, hydrostatic pressure in the capillary is high enough to cause filtration along the entire length of the capillary.
Capillary Pressure
Capillary pressure tends to move fluid outward across the arterial ends of capillary membranes. It is estimated that capillary pressure at the arterial end of capillaries is 25 mm Hg, whereas pressure at the venous end of capillaries is 10 mm Hg, corresponding to the pressure in venules. The mean capillary pressure is about 17 mm Hg. Changes in arterial pressure have little effect on capillary pressure and flow because of adjustments of precapillary resistance vessels. Autoregulation describes the maintenance of unchanged tissue blood flow despite changes in perfusion pressure.
Interstitial Fluid Pressure
Interstitial fluid pressure tends to move fluid outward across capillary membranes. It is estimated that average interstitial fluid pressure is −6.3 mm Hg. This negative pressure acts as a vacuum to hold tissues together and maintain a minimal distance for diffusion of nutrients. Under normal conditions, almost all of the interstitial fluid is held in a gel that fills the spaces between cells. This gel contains large quantities of mucopolysaccharides, the most abundant of which is hyaluronic acid. Loss of negative interstitial fluid pressure allows fluid to accumulate in tissue spaces as edema.
Plasma Colloid Osmotic Pressure
Plasma proteins are principally responsible for the plasma colloid osmotic (oncotic) pressure that tends to cause movement of fluid inward through capillary membranes. Each gram of albumin exerts twice the colloid osmotic pressure of a gram of globulin. Because there is about twice as much albumin as globulin in the plasma, about 70% of the total colloid osmotic pressure results from albumin and only about 30% from globulin and fibrinogen.
A special phenomenon known as Donnan equilibrium causes the colloid osmotic pressure to be about 50% greater than that caused by proteins alone. This reflects the negative charge characteristic of proteins that necessitates the presence of an equal number of positively charged ions, mainly sodium ions, on the same side of the capillary membrane as the proteins. These extra positive ions increase the number of osmotically active substances and thus increase the colloid osmotic pressure. Indeed, about one-third of the normal plasma colloid osmotic pressure of 28 mm Hg is caused by positively charged ions held in the plasma by proteins. This is the reason that plasma proteins cannot be replaced by inert substances, such as dextran, without some decrease in plasma colloid osmotic pressure.
Interstitial Fluid Colloid Osmotic Pressure
Proteins present in the interstitial fluid are principally responsible for the interstitial fluid colloid osmotic pressure of about 5 mm Hg, which tends to cause movement of fluid outward across capillary membranes. Albumin, because of its small size, normally leaks 1.6 times as readily as globulins through capillaries, causing the proteins in interstitial fluids to have a disproportionately high albumin to globulin ratio. The total protein content of interstitial fluid is similar to the total protein content of plasma, but because the volume of the interstitial fluid is four times the volume of plasma, the average interstitial fluid protein content is only one-fourth that in plasma or about 1.8 g/dL. Interstitial fluid protein content also remains low because proteins cannot readily diffuse across capillary membranes, and any that crosses is likely to be removed by lymph vessels.
Diffusion
Diffusion is the most important mechanism for transfer of nutrients between the plasma and the interstitial fluid. Oxygen, carbon dioxide, and anesthetic gases are examples of lipid-soluble molecules that can diffuse directly through capillary membranes independently of pores. Sodium, potassium, and chloride ions and glucose are insoluble in lipid capillary membranes and therefore must pass through pores to gain access to interstitial fluids. The diffusion rate of lipid-soluble molecules across capillary membranes in either direction is proportional to the concentration difference between the two sides of the membrane. For this reason, large amounts of oxygen move from capillaries toward tissues, whereas carbon dioxide moves in the opposite direction. Typically, only slight partial pressure differences suffice to maintain adequate transport of oxygen between the plasma and interstitial fluid.
Pinocytosis
Pinocytosis is the process by which capillary endothelial cells ingest small amounts of plasma or interstitial fluid followed by migration to the opposite surface where the fluid is released. Transport of high-molecular-weight substances such as plasma proteins, glycoproteins, and polysaccharides (dextran) most likely occurs principally by pinocytosis.
Lymphatics
Lymph vessels represent an alternate route by which fluids flow from interstitial spaces into the blood. The most important function of the lymphatic system is return of proteins into the circulation and maintenance of a low-protein concentration in the interstitial fluid. The small amount of protein that escapes from the arterial end of the capillary cannot undergo reabsorption at the venous end of the capillary. If lymph vessels were not available, this protein would be progressively concentrated in the interstitial fluid, resulting in increases in interstitial fluid colloid osmotic pressure that, within a few hours, would produce life-threatening edema. Only cartilage, bone, epithelium, and tissues of the central nervous system are devoid of lymphatic vessels.
Anatomy
The major terminal lymph vessels are the thoracic duct and the right lymphatic duct (Fig. 14-19). The thoracic duct is the larger of the two (2 mm in diameter), entering the venous system in the angle of the junction of the left internal jugular and subclavian veins. The right lymphatic duct is not always present, and if it is, it rarely exists as such because the three vessels that occasionally unite to form it usually open separately into the right internal jugular, subclavian, and innominate veins. Damage (surgical or traumatic) to a thoracic duct can cause intrathoracic fluid accumulation.

Peripheral lymph vessels are small, difficult to identify, and contain flaplike valves between endothelial cells that open toward the interior, allowing the unimpeded entrance of interstitial fluid and proteins but preventing backflow.
Formation and Flow of Lymph
Lymph is interstitial fluid that flows into lymphatic vessels. The protein concentration of peripheral lymph is about 1.8 g/dL, whereas lymph from the gastrointestinal tract and liver contains two to three times this concentration of protein. The lymphatic system is one of the major channels for absorption of nutrients, especially fat, from the gastrointestinal tract. Bacteria that enter lymph vessels are removed and destroyed by lymph nodes.
Flow of lymph through the thoracic duct is about 100 mL per hour. A decrease in the normally negative value of interstitial fluid pressure increases the flow of interstitial fluid into terminal lymph vessels and consequently increases the rate of lymph flow. For example, at 0 mm Hg interstitial fluid pressure, the rate of lymph flow is increased 10 to 50 times compared with flow at an average interstitial fluid pressure of −6.3 mm Hg. Skeletal muscle contraction and passive movements of the extremities facilitate flow of lymph. For example, during exercise, lymph flow is increased up to 14 times that present at rest. Elevations in CVP can impair the return of lymph to the central circulation.
Edema
Edema is the presence of excess interstitial fluid in peripheral tissues that results from the inability of lymph vessels to adequately transport fluid. This may occur due to damage or obstruction of lymphatic vessels, excessive fluid administration, capillary fluid leakage due to increased permeability, low serum protein concentration, or elevated CVP. Peripheral edema most common in dependent areas may be accompanied by accumulation of fluid in potential spaces such as the pleural cavity, pericardial space, peritoneal cavity, and synovial spaces. Excessive fluid in the peritoneal space—one of the spaces most prone to develop edema fluid—is called ascites. The peritoneal cavity is susceptible to the development of edema fluid because any increased pressure in the liver, such as occurs in cirrhosis or cardiac failure, causes transudation of protein-containing fluids from the surface of the liver into the peritoneal cavity.
Pulmonary Circulation
The pulmonary circulation is a low-pressure, low-resistance system in series with the systemic circulation. The volume of blood flowing through the lungs and systemic circulation is essentially identical. Blood passes through pulmonary capillaries in about 1 second, during which time it is oxygenated and carbon dioxide is removed.
Anatomy
Anatomically, the right ventricle is semilunar in shape, wrapped around the medial aspect of the left ventricle. The thickness of the right ventricle is one-third that of the left ventricle, as it normally generates pressures approximately 25% that of the left side.
The pulmonary artery extends only about 4 cm beyond the apex of the right ventricle before division into the right and left main pulmonary arteries. The pulmonary artery is a thin structure with a wall thickness about twice that of the vena cava and one-third that of the aorta. The large diameter and distensibility of the pulmonary arteries allows the pulmonary circulation to easily accommodate the stroke volume of the right ventricle. Pulmonary veins, like pulmonary arteries, are large in diameter and highly distensible. Pulmonary capillaries supply the estimated 300 million alveoli, providing a gas exchange surface of 70 m2.
Pulmonary blood vessels are innervated by the sympathetic nervous system, but the density of these fibers is less than in systemic vessels. α-Adrenergic stimulation from norepinephrine produces vasoconstriction of the pulmonary vessels, whereas β-adrenergic stimulation, as produced by isoproterenol, results in vasodilation. Parasympathetic nervous system fibers from the vagus nerves release acetylcholine, which produces vasodilation of pulmonary vessels. Despite the presence of autonomic nervous system innervation, the resting vasomotor tone is minimal, and the larger pulmonary vessels are almost maximally dilated in the normal resting state. Indeed, overall regulation of pulmonary blood flow is passive, with local adjustments of perfusion relative to ventilation being determined by local oxygen tension.
The diameter of thin-walled alveolar vessels changes in response to alterations in the transmural pressure (intravascular pressure minus alveolar pressure). If alveolar pressure exceeds intravascular pressure as occurs in nondependent regions of the lungs during positive pressure ventilation, pulmonary capillaries collapse and blood flow ceases. The size of larger vessels embedded in the lung parenchyma (extraalveolar vessels) largely depends on lung volume. For example, resistance to flow through these vessels decreases as lung volumes increase because these vessels are tethered to the surrounding tissue (Fig. 14-20A,B). However, the largest pulmonary vessels, those at the hilum of the lung, vary in size in response to changes in intrapleural pressure.

Bronchial Circulation
Bronchial arteries from the thoracic aorta supply oxygenated blood to supporting tissues of the lungs, including connective tissue and airways. After bronchial arterial blood has passed through supporting tissues, the majority of it empties into pulmonary veins and enters the left atrium rather than passing back to the right atrium. The entrance of deoxygenated blood into the left atrium dilutes oxygenated blood and accounts for an anatomic shunt that is equivalent to an estimated 1% to 2% of the cardiac output. This anatomic shunt plus a part of coronary blood flow which drains directly into the left side of the heart are the reasons the cardiac output of the left ventricle slightly exceeds that of the right ventricle.
Pulmonary Lymph Vessels
Pulmonary lymph vessels extend from all the supportive tissues of the lung to the hilum of the lung and then to the thoracic duct. Pulmonary lymphatic flow facilitates the removal of edema fluid from alveolar spaces. Particulate matter entering the alveoli is also eventually removed by lymph vessels.
Pulmonary Vascular Pressure
The normal pressure in the pulmonary artery is about 22/8 mm Hg, with a mean pulmonary artery pressure of 13 mm Hg (Fig. 14-21).4 The mean pulmonary capillary pressure is about 10 mm Hg, and the mean pressure in the pulmonary veins is about 4 mm Hg, such that the pressure gradient across the pulmonary circulation is only 9 mm Hg. The resistance to blood flow in the pulmonary circulation is about one-tenth the resistance in the systemic circulation.

Pulmonary artery pressure is not typically influenced by left atrial pressures of less than 7 mm Hg. However, when left atrial pressure exceeds approximately 7 mm Hg, previously collapsed pulmonary veins are expanded, and pulmonary artery pressure increases in parallel with increases in left atrial pressure. In the absence of left ventricular failure, even marked increases in systemic vascular resistance do not cause the left atrial pressure to increase significantly. Consequently, the right ventricle continues to eject its stroke volume against a normal pulmonary artery pressure despite increased workloads imposed on the left ventricle. Accordingly, the right ventricular stroke volume is not measurably altered by changes in systemic vascular resistance unless the left ventricle fails.
Should the left ventricle fail, left atrial pressures can increase to greater than 15 mm Hg. Mean pulmonary artery pressures also increase, placing an increased workload on the right ventricle. If this occurs acutely, the right ventricle may also fail as it may not be able to generate an adequate stroke volume due to its structure (see earlier discussion). If pulmonary artery pressures rise gradually over time, the right ventricle may adapt with remodeling and dilation but will eventually begin to fail.
Measurement of Left Atrial Pressure
The left atrial pressure can be estimated by inserting a balloon-tipped catheter into a small pulmonary artery. When the balloon is temporarily inflated and the vessel is completely occluded, a stationary column of blood is created distal to the catheter tip. As a result, the pressure measured immediately distal to the balloon is equivalent to that downstream in the pulmonary veins. This measurement is termed the pulmonary artery occlusion pressure (“wedge” pressure although the catheter is not truly wedged) and is usually 2 to 3 mm Hg higher than left atrial pressure. If the balloon is deflated, flow will resume and the pulmonary artery end diastolic pressure can be measured. This measurement correlates with the pulmonary artery occlusion pressure in the absence of pulmonary hypertension.
Interstitial Fluid Space
The interstitial fluid space in the lung is minimal, and a continual negative pulmonary interstitial pressure of about −8 mm Hg dehydrates interstitial fluid spaces of the lungs and keeps the alveolar epithelial membrane in close approximation to the capillary membranes. As a result, the diffusion distance between gas in the alveoli and the capillary blood is minimal, averaging about 0.4 µm. Negative pressure in pulmonary interstitial spaces draws fluid from alveoli through alveolar membranes and into the interstitium, keeping the alveoli dry. Mean pulmonary capillary pressure is about 10 mm Hg, whereas plasma colloid osmotic pressure is about 28 mm Hg. This net pressure gradient of about 18 mm Hg discourages the movement of fluid out of capillaries, decreasing the likelihood of pulmonary edema.
Pulmonary Blood Volume
Blood volume in the lungs is about 450 mL. Of this amount, about 70 mL is in capillaries and the remainder is divided equally between pulmonary arteries and veins. Cardiac failure or increased resistance to flow through the mitral valve causes pulmonary blood volume to increase.
Cardiac output can increase nearly four times before pulmonary artery pressure becomes increased (Fig. 14-22).4 This reflects the distensibility of the pulmonary arteries and opening of previously collapsed pulmonary capillaries. The ability of the lungs to accept greatly increased amounts of pulmonary blood flow, as during exercise, without excessive increases in pulmonary artery pressures is important in preventing development of pulmonary edema or right ventricular failure.

Pulmonary blood volume can increase up to 40% when an individual changes from the standing to the supine position. This sudden shift of blood from the systemic circulation to pulmonary circulation is responsible for the decrease in vital capacity in the supine position and the occurrence of orthopnea in the presence of left ventricular failure.
Pulmonary Blood Flow and Distribution
Optimal oxygenation depends on matching ventilation to pulmonary blood flow.23 Shunt occurs in lung areas that are perfused but inadequately ventilated, whereas dead space ventilation occurs in lung areas that are ventilated but inadequately perfused (Fig. 14-23). Although the lungs are innervated by the autonomic nervous system, it is doubtful that neural influences exert a major effect in the normal control of pulmonary blood flow. There is no doubt, however, that decreases in PaO2 cause increases in pulmonary artery and right ventricular pressures.

Clinically, segmental pulmonary blood flow can be studied by intravenous injection of radioactive xenon while monitoring is performed externally over the chest with radiation detectors. Xenon rapidly diffuses from capillaries into alveoli and radioactivity is detected early in well-perfused regions of the lung.
Endothelial Regulation of Pulmonary Blood Flow
Active vasodilation is crucial to the maintenance of the low resting tone of the normal pulmonary circulation. The pulmonary vascular endothelium is responsible for the synthesis and secretion of various compounds that regulate smooth muscle activity in the pulmonary circulation. The primary vasodilatory compounds are NO and prostacyclin. The predominant influence of a healthy pulmonary endothelium is to reduce pulmonary vascular tone. Endothelin is also released by the pulmonary endothelium and may have both a constrictive or dilatory effect depending on the circumstances. There are several negative feedback mechanisms in effect that regulate vascular tone. The synthesis and release of NO, prostacyclin, ET-1, and other vasoactive compounds are integrated into a system that optimizes pulmonary vascular tone and facilitates local control of ventilation to perfusion (V/Q) matching. Therapeutically, inhaled NO, intravenous and inhaled prostacyclin, and oral endothelin antagonists are used to treat pulmonary hypertension.
Nitric Oxide
NO is synthesized in endothelial cells by NO synthase (NOS). There are several isoforms of NOS, but it is likely that cNOS (also termed endothelial NOS or eNOS) is responsible for the normal regulation of pulmonary vascular tone. The activity of cNOS is rapidly up- or downregulated and produces picomolar amounts of NO in response to stimuli such as shear stress and local oxygen tension. In pathologic states, another NOS isoform, iNOS, produces considerably larger quantities of NO but takes longer to upregulate and downregulate. iNOS is located in vascular smooth muscle cells and inflammatory macrophages. Inflammatory mediators are potent upregulators of iNOS activity. NO diffuses from its site of production, usually endothelial cells, into adjacent pulmonary vascular smooth muscle cells. NO induces vasorelaxation by stimulating the production of cyclic guanosine monophosphate (cGMP) by the enzyme guanylate cyclase. cGMP is promptly metabolized by phosphodiesterases. Increased cGMP production appears to stimulate phosphodiesterase upregulation, thus causing accelerating cGMP clearance. Indeed, phosphodiesterase upregulation may be partly responsible for suboptimal responses to inhaled NO and for rebound pulmonary hypertension after NO withdrawal. Type V phosphodiesterase is the predominant enzyme type in the pulmonary circulation. Selective inhibitors of type V phosphodiesterase such as sildenafil can both selectively decrease pulmonary vascular resistance when given alone and significantly potentiate the effects of iNOS and prostacyclin.
Endothelin
ET-1 is a potent endogenous peptide vasoconstrictor that also promotes smooth muscle proliferation. Endothelin has been implicated in the pathogenesis of some types of pulmonary hypertension because some patients have both increased expression and reduced clearance of ET-1 in lung tissue and plasma. There are two endothelin receptors: vascular smooth muscle endothelin A and endothelial endothelin B. Smooth muscle endothelin A receptor stimulation causes vasoconstriction, whereas endothelial endothelin B receptor stimulation causes both vasoconstriction and relaxation. ET-1 release is increased as part of the inflammatory response to cardiopulmonary bypass, thereby increasing the potential for pulmonary hypertension after cardiovascular surgery. Exposure to inhaled NO significantly increases ET-1 levels, suggesting that ET-1 may be partly responsible for rebound pulmonary hypertension after withdrawal of inhaled NO. Endothelin antagonists may prove to be useful in the treatment of pulmonary hypertension. Bosentan is an oral, nonselective endothelin antagonist that is used in the treatment of pulmonary hypertension.
Prostacyclin
Prostacyclin is a potent vasodilatory prostaglandin released by the pulmonary endothelium. Other prostaglandins, such as thromboxane A2, have potent vasoconstrictive effects. Pulsatile flow and local shear stress are important stimuli for prostacyclin release. Prostacyclin receptor binding causes the activation of adenylate cyclase, thereby increasing local adenosine-3,5 cyclic monophosphate release. The adenyl cyclase pathway, regulated by prostacyclin, and the guanylate cyclase pathway, regulated by NO, are parallel pathways that converge to reduce vascular smooth muscle tone. Prostacyclin is also an extremely potent inhibitor of platelet aggregation and vascular smooth muscle proliferation. The chronic intravenous administration of prostacyclin is an established treatment for pulmonary hypertension and was first described more than 20 years ago. However, intravenous prostacyclin is not a selective pulmonary vasodilator and can cause significant systemic side effects, including hypotension, and will worsen V/Q matching in the pulmonary circulation. Both the positive and negative hemodynamic effects of prostacyclin are potentiated by the coadministration of phosphodiesterase inhibitors. Of particular interest to anesthesiologists are the encouraging findings with the inhaled aerosolized prostacyclin analogues iloprost and treprostinil. These drugs have physical characteristics that make them better suited to nebulization and have longer duration of action than intravenous prostacyclin.
Hypoxic Pulmonary Vasoconstriction
Alveolar hypoxia (PaO2 <70 mm Hg) evokes vasoconstriction in the pulmonary arterioles supplying these alveoli. The net effect is to divert blood flow away from poorly ventilated alveoli. As a result, the shunt effect is minimized, and the resulting PaO2 is maximized. The mechanism for hypoxic pulmonary vasoconstriction is presumed to be locally mediated, as this response occurs in isolated and denervated lungs as well as intact lungs. Indeed, this vasoconstrictor response is apparent in isolated pulmonary artery strips and isolated smooth muscle cells. There are probably multiple mechanisms responsible for hypoxic pulmonary vasoconstriction and these mechanisms likely differ during acute and chronic responses. The suppression of endothelial release of the potent vasodilator NO is likely an important element of both the acute and chronic response. A crucial component of the acute response is the inhibition of potassium channels that leads to membrane depolarization. Membrane depolarization increases calcium influx, which in turn activates the intracellular contractile response. Chronic vascular responses to hypoxia may be, in part, mediated by endothelin release and eventually involve vascular remodeling, eventually leading to irreversible increases in pulmonary vascular resistance (pulmonary hypertension).
Drug-induced inhibition of hypoxic pulmonary vasoconstriction could result in unexpected decreases in Pao2 in the presence of lung disease. Potent vasodilating drugs such as nitroprusside and nitroglycerin may be accompanied by decreases in Pao2 attributed to this effect.24 Although animal studies suggest that potent inhaled anesthetic agents inhibit hypoxic vasoconstriction in a dose-dependent manner, in clinically relevant concentrations, these findings have not been supported by clinical studies in patients (Fig. 14-24).25,26 The present consensus is that potent volatile anesthetics are acceptable choices for thoracic surgery requiring one-lung ventilation, particularly in view of the beneficial effects of these drugs on bronchomotor tone and their high potency that permits delivery of maximal concentrations of oxygen.27

Effect of Breathing
During spontaneous respiration, venous return to the heart is increased due to contraction of the diaphragm and abdominal muscles, which decreases intrathoracic pressure. The resulting augmented blood flow to the right atrium increases right ventricular stroke volume. In contrast to spontaneous breathing, positive pressure ventilation increases intrathoracic pressure and thus impedes venous return to the heart and decreases right ventricular stroke volume.
Regional Blood Flow in the Lungs
Although traditional teaching has focused on the gravitational effect of blood flow within the lungs, more recent work has demonstrated that the gravitational “zone”28 (Fig. 14-25) has a relatively minor role in blood flow distribution. On average, pulmonary blood flow is greater in areas of the lung below the heart as compared to those above the heart,29 but measurements have demonstrated greater variation from spot to spot in any isogravitational plane than there is on average from the top of the lung to the bottom. Indeed, only 25% of the variability in pulmonary blood flow is accounted for by gravitational effects. Furthermore, the local differences in flow at any isogravitational level are fairly constant over time, suggesting that approximately 75% of the distribution of pulmonary blood flow is determined by the branching structure of the pulmonary vascular tree.

Pulmonary Circulatory Pathology
Pulmonary Edema
Pulmonary edema is present when there are excessive quantities of fluid either in pulmonary interstitial spaces or in alveoli. Mild degrees of pulmonary edema may be limited to only an increase in the interstitial fluid volume. The alveolar epithelium, however, is not able to withstand more than a modest increase in interstitial fluid pressure before fluid spills into alveoli. Dehydrating forces of the colloid osmotic pressure of the blood in the lungs provide a large safety factor against development of pulmonary edema. In humans, plasma colloid osmotic pressure is about 28 mm Hg, so pulmonary edema rarely develops below a pulmonary capillary pressure of 30 mm Hg. The most common cause of acute pulmonary edema is greatly increased pulmonary capillary pressure resulting from left ventricular failure, and the lymphatic flow cannot adequately remove the increased fluid.
During chronic increases of left atrial pressure, pulmonary edema may not occur despite pulmonary capillary pressures as high as 45 mm Hg. Enlargement of the pulmonary lymph vessels allowing lymph flow to increase up to 20 times is the most likely reason pulmonary edema does not occur in the presence of chronically increased left atrial pressures. At the other end of the spectrum, a transplanted lung does not have any lymphatic drainage and is acutely sensitive to fluid overload or increased in left atrial pressure.
Pulmonary edema can also result from local capillary damage that occurs with inhalation of acidic gastric fluid or irritant gases, such as smoke. The result is rapid transudation of fluid and proteins into alveoli and interstitial spaces. This is called permeability pulmonary edema to distinguish it from “hydrostatic” pulmonary edema, which is due to increased pulmonary capillary pressure. In either case, increased interstitial fluid and fluid-filled alveoli interfere with gas exchange, decrease lung compliance, and result in an increase in the work of breathing.
Pulmonary Embolism
Embolism of venous clots to the lungs may be chronic with small emboli or acute with massive embolus. Total blockage of a major branch of a pulmonary artery by an embolus is usually not immediately fatal because other lung regions may be able to accommodate all the pulmonary blood flow, but major emboli may result in right ventricular strain and failure. Tachypnea and dyspnea are characteristic responses in awake patients experiencing pulmonary embolism; in the anesthetized patient, an acute decrease in end-tidal CO2 may occur due to a sudden increase in dead space (loss of perfusion but not ventilation to major part of the lung). Depending on the circumstance, anticoagulation, thrombolysis, or pulmonary embolectomy may be indicated.
Pulmonary Hypertension
The World Health Organization has classified pulmonary hypertension according to etiology (Table 14-11)30 but the hemodynamic diagnosis in all causes is made when the sustained mean pulmonary artery pressure is above 25 mm Hg. The cause may be “primary” or idiopathic (type 1) or in association with other disease (other types). As mentioned earlier, gradual increases in pulmonary artery pressure may be tolerated by the right heart, but with progressive increases in pressure, right ventricular failure will eventually occur. In addition, many of the causes of pulmonary hypertension are associated with the development of hypoxemia. Treatment of the underlying cause, where possible, is of course more effective than treatment of the result, that is, oxygen for hypoxemia and pulmonary vasodilators; the pulmonary dilators mentioned earlier may not be effective or be associated with intolerable side effects. Lung transplant or in the case of right ventricular failure, heart–lung transplant may be required.
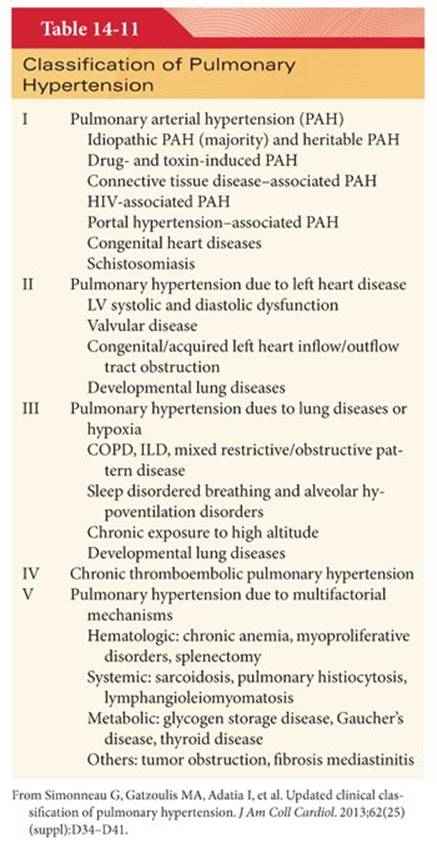
References
1. Galley HF, Webster NR. Physiology of the endothelium. Br J Anaesth. 2004;93:105–113.
2. Myburgh JA, Mythen MG. Resuscitation fluids. N Engl J Med. 2013;369:1243–1251.
3. Young JD. The heart and circulation in severe sepsis. Br J Anaesth. 2004;93:114–120.
4. Guyton AC, Hall JE. Textbook of Medical Physiology. 10th ed. Philadelphia, PA: Saunders; 2000.
5. Gerber MJ, Hines RL, Barash PG. Arterial waveforms and systemic vascular resistance: is there a correlation? Anesthesiology. 1987;66:823–825.
6. Pauca AL, Hudspeth AS, Wallenhaupt SL, et al. Radial artery-to-aorta pressure difference after discontinuation of cardiopulmonary bypass. Anesthesiology. 1989;70:935–941.
7. Stern DH, Gerson JI, Allen FB, et al. Can we trust the direct radial artery pressure immediately following cardiopulmonary bypass? Anesthesiology. 1985;62:557–561.
8. Baba T, Goto T, Yoshitake A, et al. Radial artery diameter decreases with increased femoral to radial arterial pressure gradient during cardiopulmonary bypass. Anesth Analg. 1997;85:252–258.
9. Rich GF, Lubanski RE, McLoughlin TM. Differences between aortic and radial artery pressure associated with cardiopulmonary bypass. Anesthesiology. 1992;77:63–66.
10. Bazaral MG, Welch M, Golding LAR, et al. Comparison of brachial and radial arterial pressure monitoring in patients undergoing coronary artery bypass surgery. Anesthesiology. 1990;73:38–45.
11. Cook DJ, Simel DL. Does this patient have abnormal central venous pressure? JAMA. 1996;275:630–634.
12. Ganong WF. Review of Medical Physiology. 21st ed. New York, NY: McGraw-Hill; 2003.
13. Cohen MA, Taylor JA. Short-term cardiovascular oscillations in man: measuring and modeling the physiologies. J Physiol. 2002;542(pt 3):669–683.
14. Kaukonen KM, Bailey M, Suzuki S, et al. Mortality related to severe sepsis and septic shock among critically ill patients in Australia and New Zealand, 2000-2012. JAMA. 2014;311(13):1308–1316.
15. Tibby SM, Murdoch IA. Measurement of cardiac output and tissue perfusion. Curr Opin Pediatr. 2002;14:303–309.
16. Moise SF, Sinclair CJ, Scott DHT. Pulmonary artery blood temperature and the measurement of cardiac output by thermodilution. Anaesthesia. 2002;57:562–566.
17. Berton C, Cholley B. Equipment review: new techniques for cardiac output measurement–oesophageal Doppler, Fick principle using carbon dioxide, and pulse contour analysis. Crit Care. 2002;6:216–221.
18. Summers RL, Shoemaker WC, Peacock WF, et al. Bench to bedside: electrophysiologic and clinical principles of noninvasive hemodynamic monitoring using impedance cardiography. Acad Emerg Med. 2003;10:669–680.
19. Welsch DG, Segal SS. Endothelial and smooth muscle cell conduction in arterioles controlling blood flow. Am J Physiol. 1998;274:H323–H328.
20. Berne RM, Levy MN, Koeppen BM. Physiology. 5th ed. St Louis, MO: Mosby; 2004.
21. Feng Q, Hedner T. Endothelium-derived relaxing factor (EDRF) and nitric oxide. II. Physiology, pharmacology, and pathophysiological implications. Clin Physiol. 1990;10:503–510.
22. Michel CC, Neal CR. Openings through endothelial cells associated with increased permeability. Microcirculation. 1999;6:45–51.
23. West JB. Blood flow to the lung and gas exchange. Anesthesiology. 1974;41:124–138.
24. Colley PS, Cheney FW, Hlastala MP. Ventilation-perfusion and gas exchange effects of sodium nitroprusside in dogs with normal and edematous lungs. Anesthesiology. 1979;50:489–495.
25. Rogers SN, Benumof JL. Halothane and isoflurane do not decrease Pao2 during one-lung ventilation in intravenously anesthetized patients. Anesth Analg. 1985;64:946–954.
26. Carlsson AJ, Bindsley L, Hedenstierna G. Hypoxia-induced pulmonary vasoconstriction in the human lung: the effect of isoflurane anesthesia. Anesthesiology. 1987;66:312–316.
27. Eisenkraft JB. Effects of anaesthetics on the pulmonary circulation. Br J Anaesth. 1990;65:63–78.
28. West JB, Dollery CT, Naimark A. Distribution of blood flow in isolated lung: relation to vascular and alveolar pressures. J Appl Physiol. 1964;19:713–718.
29. Glenny RW, Bernard S, Robertson HT, et al. Gravity is an important but secondary determinant of regional pulmonary blood flow in upright primates. J Appl Physiol. 1999;86:623–632.
30. Simonneau G, Gatzoulis MA, Adatia I, et al. Updated clinical classification of pulmonary hypertension. J Am Coll Cardiol. 2013;62(25)(suppl):D34–D41.