The management of acid–base disorders requires establishing the cause(s) of the disorder and then treating the underlying physiologic derangement. The treatment of acid–base disturbances is complex for a variety of reasons including the fact that the laboratory information on which we make decisions is rarely complete. This is because we often do not have access to all the data which influence a patient’s acid–base status (e.g., serum phosphate, sulfate, etc.). The acid–base disturbance is often evolving rapidly (e.g., ischemia, shock, etc.) and there is a delay getting laboratory results. The algorithms which have been designed to help us understand acid–base therapy are based on steady-state conditions and the terminology that we use to describe acid–base chemistry is not intuitive (Table 26-1). However, with just blood gas and common serum biochemistry data, we can manage the majority of clinical acid–base disorders.

The central focus of treating acid–base disturbances is the understanding of the biochemistry of the hydrogen ion. Hydrogen ion concentrations in the various body fluid compartments are precisely regulated in the face of enormous variations in local production and clearance. Deviations in hydrogen ion concentrations from the normal range can cause marked alterations in protein structure and function, enzyme activity, and cellular function. Although hydrogen ions are continuously produced in the hydrolysis of adenosine triphosphate, the largest contribution of metabolic acids arises from the oxidation of carbohydrates, principally glucose, to produce carbon dioxide (volatile acid, approximately 24,000 mEq per day). By comparison, the average net production of nonvolatile metabolic acid, such as lactate, is relatively small (approximately 60 mEq per day).
The hydrogen ion concentration is regulated to maintain the arterial blood pH between 7.35 and 7.45. However, expression of the hydrogen ion concentration as pH masks large variations in hydrogen ion concentration despite small changes in pH. For example, a pH range of 7.0 to 7.7 is associated with a fivefold change (100 nmol/L to 20 nmol/L) in hydrogen ion concentration. The pH of venous blood and interstitial fluid is lower than that of arterial blood (approximately 7.35).
Mechanisms for Regulation of Hydrogen Ion Concentration
Regulation of pH over a narrow range depends on (a) buffer systems, (b) ventilatory responses, and (c) renal responses. The buffer system mechanism is local and immediate, but incomplete. Ventilatory responses are slower (minutes) and usually incomplete. Renal responses develop very slowly (hours) but can produce nearly complete pH correction.
Buffer Systems
Body fluids contain acid–base buffer systems that immediately combine with acid or alkali to prevent excessive changes in the hydrogen ion concentration. This ability to neutralize excess protons maintains the local pH near 7.4 in the face of continuous acid generation. The most important buffer systems are (a) bicarbonate and carbonic acid in plasma, interstitial and intracellular fluid, and bone; (b) hemoglobin and other proteins in intracellular fluid; (c) plasma proteins; and (d) phosphates in intracellular and extracellular fluid and the kidney (Fig. 26-1).

Bicarbonate Buffering System
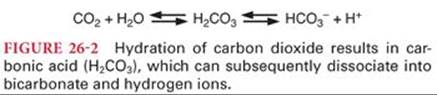
The bicarbonate buffering system consists of carbonic acid (H2CO3) and sodium bicarbonate (NaHCO3). Bicarbonate buffer is primarily a product of the approximately 200 mL of carbon dioxide produced per minute, of which considerably less than 1% dissolves to become carbonic acid. Carbonic acid is a weak acid because of its limited degree of dissociation (<5% at physiologic pH) into hydrogen and bicarbonate ions (Fig. 26-2). Most carbonic acid in solution almost immediately dissociates into carbon dioxide and water, the net result being a very high concentration of dissolved carbon dioxide compared to the concentration of bicarbonate ions. This relationship is described mathematically by the Henderson-Hasselbalch equation, which can be used to calculate the pH of a solution if the concentration of bicarbonate ions and dissolved carbon dioxide is known (Fig. 26-3).

The addition of a strong acid such as hydrochloric acid to the bicarbonate buffering system results in conversion of the strong acid to weak carbonic acid (Fig. 26-4). Therefore, a strong acid lowers the pH of body fluids only slightly. The addition of a strong base, such as potassium hydroxide, to the bicarbonate buffering system results in the formation of a weak base and water. Buffers are most effective when they operate at a pH that is close to their pKa(under these circumstances, the buffer system is approximately 50% dissociated). The bicarbonate buffering system is not a powerful buffer because its pKa of 6.1 differs greatly from the normal pH of 7.4. Physiologically, buffers are most effective when their pKa is equal to normal pH. However, the bicarbonate system is important because (a) bicarbonate is present in significant quantities in nearly all fluid compartments, (b) the concentration of its components is ultimately regulated by the lungs and kidneys, and (c) in severe acidosis the pH approaches the pKa of the bicarbonate system thus increasing its efficiency.

The bicarbonate buffer system accounts for >50% of the total buffering capacity of blood. Approximately one-third of the bicarbonate buffering capacity of blood occurs within erythrocytes. The electrical charge of bicarbonate ions limits their diffusion into cells other than erythrocytes.
Hemoglobin Buffering System
Hemoglobin is a particularly effective buffer because it is localized in quantity in erythrocytes; it has a pKa of 6.8 and has a buffering capacity that varies with oxygenation. The imidazole ring of the amino acid histidine has a pKathat is close to the physiologic pH. Thus, hemoglobin and other histidine-containing proteins are excellent physiologic buffers. Furthermore, deoxygenated hemoglobin is a weaker acid (better proton acceptor) than oxyhemoglobin. Thus, in the systemic capillaries, dissociation of oxyhemoglobin to deoxyhemoglobin facilitates the binding of hydrogen ions produced by the dissociation of carbonic acid. This situation is reversed in the pulmonary circulation where the conversion of deoxyhemoglobin to oxyhemoglobin facilitates the release of hydrogen ions.
Protein Buffering System
Like hemoglobin, other histidine-containing proteins are important intracellular buffers. Proteins are localized in high concentrations within the cell where it is estimated that approximately 75% of all the buffering of body fluids occurs, mostly by proteins. Of particular importance is the local buffering of hydrogen ions by proteins in the mitochondria. Although the relatively low concentration of plasma proteins limits their role as extracellular buffers, hypoproteinemia will further reduce buffering capacity, especially in the critically ill patient.
Phosphate Buffering System
The phosphate buffering system is important in most fluid compartments but is especially important in renal tubules, where phosphate is concentrated. Renal tubular fluid is more acidic than extracellular fluid, bringing the pH of renal tubular fluid closer to the pKa (6.8) of the phosphate buffering system. Phosphate is a very important intracellular buffer because it is the most abundant intracellular anion. Furthermore, the relatively acidic pH of intracellular fluid is closer to the pKa of the phosphate buffering system than is the pH of extracellular fluid.
Intracellular pH Regulation
Although blood pH is commonly measured clinically, it is the intracellular pH (pHi) that is of functional importance. The routine measurement and manipulation of pHi is not possible in current practice. Indeed, during hypothermic cardiopulmonary bypass and hibernation, the pHi in heart and brain tissue appears to be highly regulated despite significant deviations in systemic pH. Cellular metabolism, transmembrane transport, membrane potential generation, cell growth and division, cytoskeletal structure, and contractile function are processes that are crucially dependent on pHi (Table 26-2). Furthermore, the optimal function of several organelles, including lysosomes and mitochondria, require that their local pH is significantly different from the general pHi. Thus there are highly regulated mechanisms to maintain local pHi including intracellular buffer systems and membrane-bound proton transporters. Indeed, the pHi (7.0) is higher than is predicted by the −90mV transmembrane potential (pH 6.8). As in the extracellular compartment, intracellular protons are rapidly bound to weak acids and bases resulting in a low free proton concentration.

Ventilatory Responses
Ventilation is quantitatively the most important mechanism of acid removal, given the enormous daily production of volatile acid compared to nonvolatile acid. Ventilatory responses cannot return pH to 7.4 when a metabolic abnormality is responsible for the acid–base disturbance. This reflects the fact that the intensity of the stimulus responsible for increases or decreases in alveolar ventilation will begin to diminish as pH returns toward 7.4. As a “buffer,” ventilatory responses are able to buffer up to twice the amount of acids or bases as all the chemical buffers combined. However, compensation for extreme metabolic acidosis imposes a significant respiratory burden. If the bicarbonate (HCO3−) is reduced to 10 mmol/L, the carbon dioxide tension must be reduced to 15 mm Hg in order to normalize the pH. Most patients cannot hyperventilate to below 20 mm Hg. Further, it is likely that the insult causing severe metabolic acidosis will also adversely affect respiratory muscle function, thus compromising the respiratory response.
Renal Responses
The day-to-day renal contribution to acid–base regulation is directed toward the conservation of bicarbonate and the excretion of hydrogen ions. Plasma bicarbonate is freely filtered at the glomerulus. Almost all filtered bicarbonate must be reabsorbed from the glomerular filtrate to maintain the normal plasma bicarbonate concentration (25 mEq/L) and plasma pH. Most bicarbonate reabsorption occurs in the proximal convoluted tubule and is facilitated by the presence of carbonic anhydrase in the luminal fluid and is driven by the sodium-potassium-ATPase pump in the peritubular cell membrane. Active sodium ion extrusion from the renal tubular cell into the peritubular circulation favors sodium diffusion from the tubular lumen into the tubular cell in exchange for hydrogen ions. Hydrogen in the renal tubular fluid then combines with filtered bicarbonate to form carbonic acid. Carbonic anhydrase facilitates the dissociation of carbonic acid into water and carbon dioxide that both enter the renal tubular cell. Carbon dioxide and water generate bicarbonate, which enters the peritubular circulation accompanied by sodium. The remaining hydrogen ions are secreted into the lumen in exchange for sodium (Fig. 26-5). Inhibition of carbonic anhydrase by acetazolamide interferes with the reabsorption of bicarbonate ions from renal tubular fluid. As a result, excess bicarbonate ions are lost in the urine and the plasma bicarbonate concentration is decreased.

Hydrogen ions are secreted into renal tubules by epithelial cells lining proximal renal tubules, distal renal tubules, and collecting ducts. At the same time, sodium ions are reabsorbed in exchange for the secreted hydrogen ions and combine with bicarbonate ions in the peritubular capillaries. This process is facilitated by aldosterone. As a result, the amount of sodium bicarbonate in the plasma is increased during the secretion of hydrogen ions into renal tubules. Active hydrogen ion transport is inhibited when the urinary pH drops below 4.0. Thus, hydrogen ions must combine with ammonia and phosphate buffers in the renal tubular lumen to prevent the pH from decreasing below this critical level. Ammonia is generated in the mitochondria of the proximal tubule. Ammonia (NH3) combines with hydrogen ions to form ammonium (NH4+), which is excreted in the urine in combination with chloride ions as the weak acid ammonium chloride (Fig. 26-6). In renal insufficiency, the capacity to generate urinary ammonia is impaired, thus reducing hydrogen ion excretion.

Renal responses that regulate hydrogen ion concentrations do so by acidification or alkalinization of the urine. In the presence of acidosis, the rate of hydrogen ion secretion exceeds the net loss of bicarbonate ion into the renal tubules. As a result, an excess of hydrogen ions is excreted into the urine. In the presence of alkalosis, the effect of the titration process in the renal tubules is to increase the number of bicarbonate ions filtered into the renal tubules relative to the secretion of hydrogen ions. Excess bicarbonate ions are excreted into the urine accompanied by cations, most often sodium.
Extracellular fluid is electroneutral such that the sum of the positive charges of all cations must equal the sum of negative charges of all anions. In the process of altering the plasma concentration of bicarbonate ions, it is mandatory to remove some other anion each time the concentration of bicarbonate ions is increased or to increase some other anion when the bicarbonate concentration is decreased. Typically, the anion that follows changes in the concentration of bicarbonate ions is chloride. As the most abundant extracellular anion, physiologic manipulation of chloride appears to be an important element of pH control. Conceptually, when bicarbonate ions are replaced by chloride ions, the pH will generally tend to decrease as a weak acid (carbonic acid) is replaced by a strong acid (hydrochloric acid).
The value of renal regulation of hydrogen ion concentration is not its rapidity but instead its ability to nearly completely neutralize any excess acid or alkali that enters the body fluids. Ordinarily, the kidneys can remove up to 500 mmol of acid or alkali each day. If greater quantities than this are generated, the kidneys are unable to maintain normal acid–base balance, and acidosis or alkalosis occurs. Even when the plasma pH is 7.4, a small amount of acid is still lost each minute. This reflects the daily production of 50 to 80 mmol of more acid than alkali. Indeed, the normal urine pH of approximately 6.4 is due to the presence of this excess acid in the urine.
Classification of Acid–Base Disturbances
Acid–base disturbances are categorized as respiratory or metabolic acidosis (pH <7.35) or alkalosis (pH >7.45) (Table 26-3).1 An acid–base disturbance that results primarily from changes in alveolar ventilation is described as respiratory acidosis or alkalosis. An acid–base disturbance unrelated to changes in alveolar ventilation is designated as metabolic acidosis or alkalosis. Compensation describes the secondary renal or ventilatory responses that occur as a result of the primary acid–base disturbance.

The principal manifestation of severe respiratory or metabolic acidosis is depression of the central nervous system. For example, coma is a characteristic of severe diabetic acidosis or renal dysfunction leading to uremia. The principal manifestation of respiratory or metabolic alkalosis is increased excitability of the peripheral nervous system and central nervous system. As a result, there may be repetitive stimulation, causing skeletal muscles to undergo sustained contraction known as tetany. Tetany of respiratory muscles may interfere with adequate ventilation of the lungs. Central nervous system excitability may manifest as seizures.
Respiratory Acidosis
Any event (drug or disease) that decreases alveolar ventilation results in an increased concentration of dissolved carbon dioxide in the blood (increased PaCO2), which in turn leads to formation of carbonic acid and hydrogen ions. By convention, carbonic acid resulting from dissolved carbon dioxide is considered a respiratory acid, and respiratory acidosis is present when the pH is <7.35 and PaCO2 is >45 mm Hg. It is important to note that although an increase in dissolved carbon dioxide generates an equivalent increase in both the hydrogen ion and bicarbonate ion concentrations, the pH will fall. This is because the relative increase in hydrogen ions is significantly greater than the relative increase in bicarbonate ions since the plasma concentration of H+ is far lower than the concentration of HCO3−. An increase in carbon dioxide sufficient to reduce the pH from 7.4 to 7.1 will essentially double the hydrogen ion concentration from 40 to 79 nmol/L, compared to an increase in the bicarbonate ion concentration only from 24.000000 to 24.000039 mmol/L.
Acidosis, respiratory or metabolic, often has profound effects on many drug and enzyme interactions in the body, which function optimally only within normal pH ranges. Of particular importance to the anesthesiologist is the clinical scenario of increasing respiratory acidosis due to inadequate reversal of muscle relaxants and the interaction between anticholinesterases and the enzyme acetlycholinesterase. The commonest method to reverse the muscle relaxant effects of nondepolarizing neuromuscular blocker agents (NMBAs) is by administering an anticholinesterase, such as neostigmine. Anticholinesterases inhibit normal acetylcholinesterase, increasing the concentration of acetylcholine in the synaptic clefts of the neuromuscular junction and thus antagonizing the neuromuscular block. The concentration of NMBAs remains essentially unchanged at the time of reversal. If the reversal is inadequate due to an excess of NMBAs or due to the limited duration of action of the anticholinesterase (e.g., 20 to 30 minutes for neostigmine) then inadequate alveolar minute ventilation can lead to progressive respiratory acidosis, which will potentiate the NMBAs and weaken the effects of the anticholinesterase. This clinical picture of increasing muscle weakness after a seemingly appropriately reversed neuromuscular block has been termed “recurarization.”2 Although this was more of a clinical problem with the older longer acting NMBAs (e.g., pancuronium), it can still occur with the newer shorter acting drugs (e.g., rocuronium).
Respiratory Alkalosis
Respiratory alkalosis is present when increased alveolar ventilation removes sufficient carbon dioxide from the body to decrease the hydrogen ion concentration to the extent that pH becomes >7.45. A physiologic cause of respiratory alkalosis is hyperventilation due to stimulation of chemoreceptors by a low Po2 associated with ascent to altitude. Kidneys compensate with time for this loss of carbon dioxide by excreting bicarbonate ions in association with sodium and potassium ions. This renal compensation is evident in individuals residing at altitude who have a nearly normal pH despite a low Paco2. A frequent cause of acute respiratory alkalosis is iatrogenic hyperventilation of the lungs as during anesthesia. Tetany that accompanies alkalosis reflects hypocalcemia due to the greater affinity of plasma proteins for calcium ions in an alkaline, compared with an acidic, solution.
Metabolic Acidosis
The most common and most confusing acid–base disorder that clinicians are required to manage is metabolic acidosis. Any acid formed in the body other than carbonic acid from carbon dioxide is considered a metabolic acid, and its accumulation results in metabolic acidosis. Acidosis impairs myocardial contractility and the responses to endogenous or exogenous catecholamines.3 Hemodynamic deterioration is usually minimal (in the awake state) when the pH remains >7.2 due to compensatory increases in sympathetic nervous system activity. Of great clinical importance are the accentuated detrimental effects of metabolic acidosis in individuals with underlying left ventricular dysfunction or myocardial ischemia or in those in whom sympathetic nervous system activity may be impaired, as by drug-induced β-adrenergic blockade or general anesthesia. Respiratory acidosis may produce more rapid and profound myocardial dysfunction than does metabolic acidosis, reflecting the ability of carbon dioxide to freely diffuse across cell membranes and exacerbate intracellular acidosis.
Acute metabolic acidosis has been treated with intravenous administration of an exogenous buffer, usually sodium bicarbonate, in the hope that normalizing pH will attenuate the detrimental effects of acidosis. The effectiveness of the use of sodium bicarbonate to treat metabolic acidosis is debatable.4 Sodium bicarbonate administration increases the carbon dioxide load to the lungs, leading to further increases in arterial and intracellular PCO2 if alveolar ventilation is not concomitantly increased. It is estimated that 1 mEq/kg sodium bicarbonate, given intravenously, produces approximately 180 mL of carbon dioxide and necessitates a transient doubling of alveolar ventilation to prevent hypercarbia. In the presence of increased dead space ventilation, even greater increases in alveolar ventilation are required for carbon dioxide elimination to equal production. Even if PaCO2 is maintained normal, it is possible that tissue pHi and the risk of ventricular fibrillation will not be altered by administration of sodium bicarbonate during cardiopulmonary resuscitation. Also, the standard formulation of sodium bicarbonate, 8.4%, is hypertonic and this will have a plasma-expanding effect that contributes to dilutional acidosis (see the following text). However, if alveolar ventilation can be increased to deal with the increased carbon dioxide load from administration of sodium bicarbonate (initial bolus dose 0.5 to 1 mEq/kg), then it can be useful as a temporizing measure to help restore hemodynamic stability in shock combined with severe metabolic acidosis.5
Lactic Acidosis
Under normal circumstances, lactate is produced at a rate of approximately 1 mmol/kg per hour. Normal clearance of lactate maintains its serum concentration between 0.5 and 1.0 mmol/L. Most lactate is cleared by the liver, where it undergoes oxidation, gluconeogenesis, and eventual conversion to bicarbonate. Lactate undergoes both passive diffusion and active transport into the liver via a monocarboxylate transporter. However, active transport becomes saturated at serum lactate concentrations that exceed 2.5 mmol/L. Severe reductions in hepatic blood flow, which occur during shock, will decrease hepatic lactate clearance. Lactic acid is a strong acid and therefore dissociates almost completely under physiologic conditions into the lactate anion and a hydrogen ion. Although lactate accumulation has classically been taught to occur mainly during anaerobic glycolysis, it is now clear that significant lactate generation occurs under normoxic conditions. Indeed, lactate is an important gluconeogenic precursor and is involved in cell-to-cell signaling. However, in the critically ill patient, lactate production may increase while lactate clearance is impaired and lactic acidosis may occur. Point-of-care testing allows almost instantaneous lactate determinations to be performed in the operating room and intensive care unit. A serum lactate >1.5 mmol/L upon admission is an independent predictor of mortality in critically ill patients. Furthermore, failure to decrease lactate concentration to ≤1.0 mmol/L 24 hours after admission is also associated with significant mortality.
The investigational drug dichloroacetate (DCA) decreases lactate concentration in cardiogenic shock, burns, diabetic ketoacidosis, and malaria. DCA activates the mitochondrial pyruvate dehydrogenase complex, thus accelerating the irreversible oxidation of lactate via pyruvate to acetyl CoA, which then enters the Krebs cycle. The buffer tris(hydroxymethyl)aminomethane (THAM) can be used to treat metabolic acidosis and does not generate carbon dioxide. It may be particularly useful, as an alternative to sodium bicarbonate, to treat metabolic acidosis in patients who are hypernatremic.6
Dilutional Acidosis
Because the pH of water at 37°C is 6.8, any increase in the free-water volume of the body will contribute to acidosis (e.g., administration of 5% dextrose). Dilutional acidosis also occurs when the plasma pH is decreased by extracellular volume expansion with chloride-containing solutions such as normal saline. Clinically, a hyperchloremic metabolic acidosis may accompany large-volume infusion of isotonic saline.7 Normal saline is commonly thought of as being a “physiologic” solution because it has an osmolarity close to plasma and does not lyse red blood cells. However, it has a pH (5.7) that is more acidic than plasma and contains significantly more chloride (154 vs. 100 mmol/L) and slightly more sodium (154 vs. 140 mmol/L) (Table 26-4). Thus, infusion of a large volume of normal saline will increase plasma chloride concentration to a relatively greater degree than sodium concentration. Chloride can be thought of as a strong acid, (hydrochloric acid-proton donor), just as sodium can be thought of as a strong base (sodium hydroxide-hydroxyl donor).8

Other Causes of Metabolic Acidosis
Renal failure prevents excretion of acids formed by normal metabolic processes, and metabolic acidosis occurs. Severe diarrhea and associated loss of sodium bicarbonate rapidly leads to metabolic acidosis, especially in the pediatric age group. Lack of insulin secretion (diabetes mellitus) or starvation impairs glucose utilization, forcing tissues to metabolize fat to meet energy needs. As a result, the plasma concentration of ketones such as acetoacetic acid may increase sufficiently to cause metabolic acidosis.
Differential Diagnosis of Metabolic Acidosis
Several different methods have been developed over the past 60 years to help clinicians in the differential diagnosis and treatment of acid–base disturbances, particularly relating to metabolic acidosis. All of these methods have their strengths and their weaknesses because they are based on theoretical stable states. In clinical practice, these methods have been made less relevant by the ability to get rapid laboratory or point-of-care results for several plasma values such as lactate.
Base Excess
The concept of base excess (BE) and its converse base-deficit were developed in the 1940s. This is defined as the amount of strong acid or base to return the plasma pH to 7.4 assuming PaCO2 40 mm Hg and normothermia. The BE is calculated (not measured) by modern blood gas analyzers from an algorithm based on measured HCO3− and pH. In isolated acute respiratory acidosis or alkalosis, the BE should not change (normal value = 0). BE remains useful to alert the clinician to the presence of a concurrent metabolic acidosis (BE < 0) in the presence of a respiratory acidosis (pH <7.35, PaCO2 >45 mm Hg) or to the presence of an underlying metabolic alkalosis (BE > 0). Mixed respiratory and metabolic acidosis is a common clinical problem. Also, it alerts the physician to the severity of the metabolic derangement, which can then be used as a guide to the initial therapy. The weakness of the BE measurement is that it does not distinguish among the possible causes of metabolic acidosis.
Anion Gap
Calculation of the anion gap may assist in the evaluation of acid–base disorders. The anion gap is a derived value based on the principle of electrochemical neutrality such that the sum of the positive (cationic) charges in a solution must equal the sum of the negative (anionic) charges. The major extracellular anions are chloride and bicarbonate. Other significant anions include proteins, phosphate, sulfate, and organic acids (including lactate). The latter are less commonly measured in routine practice and are referred to as “unmeasured anions.” The predominant extracellular cation is sodium. Although potassium is now routinely measured, its inclusion in the anion gap calculation is inconsistent and varies from institution to institution. Potassium is often grouped with the other “unmeasured cations,” calcium, and magnesium. Under normal circumstances, the concentration of the predominant cation (sodium) exceeds that of the combined predominant anions (chloride and bicarbonate) [anion gap = Na+ − (HCO3− + Cl−)] by 9 to 13 mEq/L. For electroneutrality to occur, the concentration of the combined unmeasured anions must therefore exceed that of the unmeasured cations by the same amount. The term “anion gap” refers solely to the difference in concentration between the traditionally measured anions and cations. The anion gap does not imply a true discrepancy between the total positive and negative charges in physiologic solution where the total anion charge must equal the total cation charge.
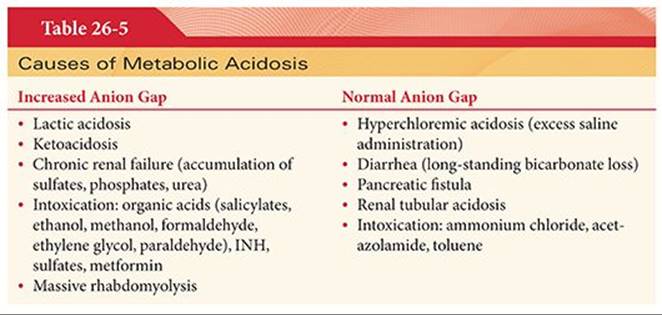
Metabolic acidosis is most often associated with an increase in the anion gap (Table 26-5). An increase in the concentration of unmeasured anions (or a decrease in the concentration of unmeasured cations) will increase the anion gap. Lactic acidosis, ketoacidosis, and renal failure increase the concentration of unmeasured endogenous anions. Exogenous anions will also increase the concentration of unmeasured anions (salicylate toxicity, ethylene glycol, and methanol ingestion). Hyperchloremic acidosis and renal tubular acidosis (bicarbonate loss) will have a normal anion gap. The weaknesses of the anion gap concept include that it does not differentiate between the causes of increased anion gap metabolic acidosis; it does not correct for pH changes due to free-water volume increase or decreases; and it does not correct for changes in serum albumin and phosphate, which have an effect on acid–base balance.
Strong Ion Gap
The strong ion gap (SIG) method is also based on the concept of electroneutrality of plasma.9 The SIG compares the excess measured serum concentrations of strong cations (Na+, K+, Mg2+, Ca2+) to the calculated total of measurable anions (Cl−, HCO3−, albumin, phosphate); the normal gap is 6 to 10 mEq/L. The SIG can be corrected for changes in plasma free-water volume and may be more useful in combined causes of metabolic acidosis, which are common in clinical practice. Measurement of the plasma lactic acid concentration and calculation of the anion gap from sodium, chloride, and bicarbonate permits differentiation of dilutional acidosis from acidosis due to tissue hypoperfusion.
Simplified Approach to Metabolic Acidosis of Uncertain Etiology
When the cause of a metabolic acidosis is unclear, measure the serum lactate, blood urea nitrogen (BUN), creatinine, and glucose. If this does not identify the etiology of the acidosis, then send serum for toxicology to measure salicylates, methanol, ethylene glycol, etc.
Metabolic Alkalosis
Metabolic alkalosis is commonly iatrogenic. Causes include vomiting with excess loss of hydrochloric acid, nasogastric suction, chronic administration of diuretics, hypoalbuminemia, and excess secretion of aldosterone. Excess administration of sodium bicarbonate may be an iatrogenic cause of metabolic alkalosis. A loss of free water (pH 6.8) will cause a volume-contraction alkalosis. Treatment involves treating the underlying cause.
Compensation for Acid–Base Disturbances
Respiratory acidosis is compensated for within 6 to 12 hours by increased renal secretion of hydrogen ions, with a resulting increase in the plasma bicarbonate concentration. After a few days, the pH will be normal despite persistence of an increased PaCO2. Sudden correction of chronic respiratory acidosis, by iatrogenic hyperventilation, may result in acute metabolic alkalosis because increased plasma bicarbonate is not promptly eliminated by the kidneys.
Respiratory alkalosis is compensated for by decreased reabsorption of bicarbonate ions from renal tubules. As a result, more bicarbonate ions are excreted in the urine, which decreases the plasma concentration of bicarbonate and returns the pH toward normal despite persistence of a decreased PaCO2.
Metabolic acidosis stimulates alveolar ventilation, which causes rapid removal of carbon dioxide from the body and decreases the hydrogen ion concentration toward normal. This respiratory compensation for metabolic acidosis, however, is only partial because pH remains somewhat below normal.
Metabolic alkalosis diminishes alveolar ventilation, which in turn causes accumulation of carbon dioxide and a subsequent increase in hydrogen ion concentration. As with metabolic acidosis, the respiratory compensation for metabolic alkalosis is only partial. Renal compensation for metabolic alkalosis is increased by reabsorption of hydrogen ions. This metabolic compensation is limited by the availability of sodium, potassium, and chloride ions. During prolonged vomiting, there may be excessive loss of chloride ions along with sodium and potassium. When this occurs, the kidneys preferentially conserve sodium and potassium ions and the urine becomes paradoxically acidic. Indeed, the presence of paradoxical aciduria indicates electrolyte depletion.
Effects of Temperature on Acid–Base Status
Temperature changes have several effects on blood and tissue pH and PCO2. As blood is cooled, carbon dioxide becomes more soluble. Therefore, for a given carbon dioxide content, the partial pressure will decrease as the temperature falls. The magnitude of this change is approximately 4.5% per degree Celsius and will tend to increase the pH. The blood pH is further increased as the dissociation of water into protons and hydroxyl ions decreases with cooling, thus decreasing hydrogen ion concentration. In addition, proton buffering by hemoglobin α-imidazole groups is enhanced by hypothermia. The sum of these effects is an increase of 0.015 pH units per degree Celsius decrease in temperature. These changes are probably insignificant within the physiologic temperature range but are important when interpreting blood-gas and acid–base data during induced cooling during cardiopulmonary bypass. If the blood temperature is decreased by 10°C to 27°C, the pH will increase to 7.6. Two alternate blood-gas management strategies, “α-stat” and “pH-stat” are utilized during hypothermia in the operating room (Table 26-6).

pH-Stat Management
During hypothermic conditions, blood pH is increased and PCO2 is decreased. The pH-stat strategy seeks to return the pH and PCO2 of hypothermic blood to normal. During hypothermic cardiopulmonary bypass, this strategy usually involves the addition of carbon dioxide via the oxygenator. A purported advantage of this strategy is that cerebral blood flow will be increased because carbon dioxide is a potent cerebral vasodilator. However, delivery of microemboli to the brain may also be increased. Temperature correction of blood gas samples is required to interpret the values obtained from a hypothermic patient but measured at 37°C. The pH-stat strategy is used more often in surgery for pediatric congenital heart disease, especially during cooling and deep hypothermic circulatory arrest.10 Under these circumstances, enhanced cerebral perfusion that facilitates brain cooling is thought to be desirable. Cerebral injury secondary to global hypoperfusion is thought to be a greater threat than delivery of microemboli in this patient population. Hypothermia, hypocarbia (via the Bohr effect), and alkalosis, all shift the oxyhemoglobin dissociation curve to the left and impair tissue oxygen delivery. The addition of carbon dioxide during pH-stat management will counter these effects and facilitate oxygen unloading from hemoglobin.
α-Stat Management
The α-stat strategy seeks to replicate the alkalinization of blood that occurs during cooling in poikilothermic mammals (e.g., naked mole rat). This strategy seeks to optimize enzyme function during hypothermia. The α of α-stat refers to the charged portion of the histidine imidazole residue. The objective is to maintain biologic neutrality by preserving the α-imidazole and protein charge state, the OH−/H+ratio, and therefore enzyme function, even though the pH will increase. This strategy is most often used during adult cardiopulmonary bypass and does not generally encourage the delivery of microemboli to the brain because supplemental carbon dioxide is not generally administered. This strategy does not require temperature correction of blood gas results.
There are examples of both strategies in nature. Homeotherms (e.g., humans) have homeostatic mechanisms for maintaining the temperature of the internal environment within very narrow limits. Homeotherms and hibernating animals hypoventilate in order to maintain blood pH at 7.4 as their body temperature decreases (pH-stat). pHi is low in most tissues under these circumstances and suppresses metabolism and conserves energy stores in nonfunctioning tissues. However, the brain and heart of these animals employ α-stat strategies to maintain pHi at α-stat values and to maintain near normal function. Poikilotherms (e.g., snakes) have not developed mechanisms for regulating the temperature of their internal environment that changes with that of the external environment. Poikilotherms use the α-stat strategy and allow their blood pH to increase and PCO2 to decrease with cooling in order to preserve cellular and enzyme function over wide temperature ranges.
References
1. Black RM. Disorders of acid-base and potassium balance. In: ACP Medicine. Danbury, CT: WebMD Professional Publishing; 2001.
2. Srivastava A, Hunter J. Reversal of neuromuscular blockade. Br J Anaesth. 2009;103:115–129.
3. Hindman BJ. Sodium bicarbonate in the treatment of subtypes of acute lactic acidosis: physiologic considerations. Anesthesiology. 1990;72:1064–1066.
4. Graf H, Leach W, Arieff AI. Metabolic effects of sodium bicarbonate in hypoxic lactic acidosis in dogs. Am J Physiol. 1985;249:F630–F635.
5. Forsythe SM, Schnmidt GM. Sodium bicarbonate for treatment of lactic acidosis. Chest. 2000;117:260–267.
6. Hoste EA, Colpaert K, Vanholder RC, et al. Sodium bicarbonate versus THAM in ICU patients with mild metabolic acidosis. J Nephrol. 2005;18:303–307.
7. Moritz ML, Ayus JC. Water water everywhere: standardized postoperative fluid therapy with 0.9% normal saline. Anesth Analg. 2010;110:293–295.
8. Levetown M. Saline-induced hyperchloremic metabolic acidosis. Crit Care Med. 2002;30:259–261.
9. Story DA, Morimatsu H, Bellomo R. Strong ions, weak ions and base excess. Br J Anaesth. 2004;92:54–60.
10. Abdul Aziz KA, Meduoye A. Is pH-stat or alpha-stat the best technique to follow in patients undergoing deep hypothermic circulatory arrest? Interact Cardiovasc Thorac Surg. 2010;10:271–282.