Cerebral or peripheral embolism is a frequent indication for echocardiographic study to rule out cardiac masses or any other source of embolism. Echocardiography is the method of choice in identifying the presence and extent of both intracavitary and intramural tumors, masses and infective vegetations. Masses can cause obstruction, infiltration, thromboembolism, arrhythmias and even death. Echocardiography answers key questions like size, shape, mobility, tissue characteristics, potential for embolization, hemodynamic consequences and extracardiac effects of masses. Echocardiographic contrast perfusion imaging differentiates the neovascularization of malignancies from the avascularity of thrombi and the sparse vascularity of stromal tumors. Compared with the adjacent myocardium, malignant and vascular tumors hyper-enhance, whereas stromal tumors and thrombi hypo-enhance.
There are a variety of other conditions which can present as cardiac masses like hydatid cysts, blood cysts, thrombi, intramyocardial hematomas. There are characteristic echocardiographic features to differentiate these. Perivalvular abscesses due to infective endocarditis are another important space-occupying lesions. Mobile and sessile masses on the valve leaflets in presence of fever may represent infective vegetations and these have typical features. This chapter is more of a descriptive and illustrative atlas of the masses rather than a detailed analysis.

Fig. 20.1: Mitral valve myxoma obstructing mitral inflow as well as the left ventricular outflow tract (parasternal long axis view).
(RV: Right ventricle; AO: Aorta; LV: Left ventricle; LA: Left atrium).
Two-dimensional (2D) echocardiography has a good sensitivity to detect intracardiac tumors, but a lower detection rate for pericardial lesions or paracardial lesions. It can easily be used to detect pericardial effusion due to metastasis in the pericardium. Smaller masses and greater details are better seen by transesophageal echocardiography (TEE).
CARDIAC MANIFESTATIONS OF MASSES
• Obstruction of circulation (Fig. 20.1).
• Interference with valve function (Fig. 20.2).
• Direct invasion (Fig. 20.3).

Fig. 20.2: Transesophageal echocardiographic (TEE) view. Doughnut shaped mass (arrows) obstructing the mitral orifice (detached thrombus).
(LAA: Left atrial appendage).

Fig. 20.3: Right atrial angiosarcoma producing obstruction at the orifice of the superior vena cava with peak instantaneous pressure gradients of 20 mm Hg during inspiration (right panel).

Fig. 20.4: Dilated atria with a doughnut thrombus (arrows) in the left atrium with spontaneous contrast.

Fig. 20.5: 3D echocardiography (3DE) longitudinal slice of the left ventricle (LV) cavity. Apical dyskinesis with large thrombus (arrows).
- Decreased myocardial contractility.
- Conduction disturbance (block and arrhythmias).
- Tamponade.
CARDIAC THROMBI AND SPONTANEOUS ECHO CONTRAST
Cardiac thrombi are one of the most important causes of stroke, peripheral and pulmonary embolism.1-3 the predisposing factors are:
• Dilated atria (Fig. 20.4).
• Atrial fibrillation.
• Segmental wall motion abnormality (Fig. 20.5).
• Severe ventricular hypokinesis (Figs 20.6A and B).
• Valvular heart disease (Fig. 20.7).
• ttrombophilia.
Atrial fibrillation and dilated atria usually have spontaneous echo contrast (Fig. 20.8). It usually represents aggregation of the cellular components of blood in conditions of stasis and low flow. It represents a high risk for thrombus formation (Fig. 20.9). Presence of spontaneous contrast should compel one to look for the thrombus. This contrast is best seen in TEE examination.
In suspected cardiac thrombi, echocardiography is used for:
• Detection of thrombi.
• Location and number.
• Attachment and mobility.

Figs 20.6A and B: Parasternal long-axis view in dilated cardiomyopathy. (A) Left panel shows large thrombus (arrow) and (B) the right panel shows dissolution of the thrombus after heparin.

Fig. 20.7: Rheumatic mitral stenosis in apical four-chamber view with large mobile thrombus with narrow stalk (arrows).

Fig. 20.8: Cloud-like spontaneous echo contrast in the right atrium in presence of atrial fibrillation and unguarded tricuspid valve.
• Delineation of focal echolucent areas indicative of clot lysis (Fig. 20.10).
Real-time three-dimensional echocardiography (3DE) provides a direct view of the left atrial appendage and hence is superior to 2D transthoracic echocardiography (TTE) as well as 2D TEE imaging (Figs 20.11 and 20.12) Real-time 3DE also improves diagnostic yield of thrombi at other locations (Fig. 20.13).
Echocardiography is now the principle method for the diagnosis of cardiac tumors. There is, however, nothing
specific about the echocardiographic appearance to identify the tumor.
Metastatic tumors are most commonly intramyocardial and may manifest as localized thickening within the myocardium. Such tumors can affect contractility or they may impinge upon the various cavities of the heart and obstruct flow.
Certain extracardiac tumors may affect the heart and can be noted by echocardiography (Fig. 20.14). Intrathoracic tumors of any origin can compress the cardiac chambers from outside the heart.
Renal cell carcinoma may grow into the renal vein and thus into the inferior vena cava, obstructing venous inflow.
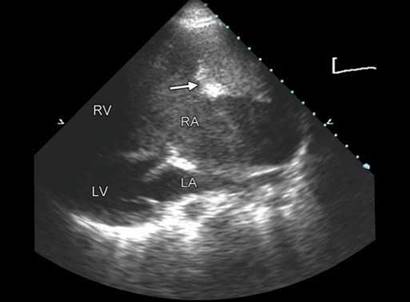
Fig. 20.9: Parasternal long-axis view with clockwise tilt of the transducer to show thrombus in the right atrium (arrow) in presence of spontaneous echo contrast.
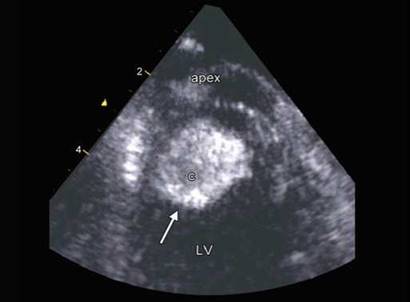
Fig. 20.10: Echolucency in the center of the apical left ventricle (LV) thrombus (c)

Fig. 20.11: 3D TEE images of the left atrial appendage. Note the lobular structure on the right side that can hide thrombi.

Fig. 20.12: 3D TEE image of a wall-hugging thrombus in the left atrial appendage (arrows) that was not picked up by 2D TEE. Note the spontaneous echo contrast in the left atrium.

Fig. 20.13: 3DE image showing a thrombus at bifurcation of the pulmonary artery (PA).

Fig. 20.14: Extracardiac mass with cystic change compressing the right ventricular outflow tract (RVOT) and the pulmonary arteries. (MPA: Main pulmonary artery; RPA: Right pulmonary artery).

Fig. 20.15: Apical four-chamber view showing a variable shape myxoma protruding through the mitral orifice and attached to the limbus of oval fossa.

Fig. 20.16: Real-time 3DE image in four-chamber view showing broad-based relatively sessile left atrial myxoma.

Fig. 20.17: 2D TEE image. Pedunculated left atrial myxoma protruding through the mitral orifice (arrows).
Seventy-five percent of the primary cardiac tumors are benign.4-6 Amongst, benign tumors, myxomas, fibroela- stomas and lipomas predominate in adults while rhabdomyoma, fibroma and myxoma are more frequent in children.5Primary malignant tumors are mostly sarcomas.7
ECHOCARDIOGRAPHIC FEATURES OF MYXOMA (FIGS 20.15 TO 20.17)
• Endocardium-based tumors that do not inflitrate.
• Predilection for fossa ovalis.
• Mobile in 75%.
• Broad-based or pedunculated stalk.
• Lobular, irregular with variegated echo texture.
• Marked fluidity.
Seventy-five percent of the myxomas occur in the left atrium, 20% in the right atrium and rest 5% at different places (Figs 20.15 to 20.17).8
Complex cardiac myxomas are those that occur in families in combination with two or more of the following conditions: skin myxomas, cutaneous lentiginosis, myxoid fibroadenomas of the breast, pituitary adenomas, primary adrenocortical micronodular dysplasia with Cushing's syndrome and testicular tumors. the Carney complex is a syndrome of myxoma, endocrine hyperfunction and skin pigmentation. Symptoms in 25-50% of patients

Fig. 20.18: Parasternal short-axis view showing right atrial myxoma (T) with attachment to the interatrial septum (arrow).

Fig. 20.19: Fibroelastoma of the aortic valve. The right panel is a 3DE image.

Fig. 20.20: Papillary fibroelastoma attached to the chords of the tricuspid valve. Note the papillary fronds on the surface.
are caused by systemic embolization, or inflow or outflow obstruction. These include transient ischemic attacks, dyspnea, syncope, sudden death or palpitations (Fig. 20.18).
PAPILLARY FIBROELASTOMA
• Papillary fibroelastomas are rare, primary, benign cardiac tumors most frequently located in the heart valves. They are a potential cause of systemic emboli, stroke, myocardial infarction and sudden death.
• Even rarer are multiple fibroelastomas, which show an echocardiographic picture of multiple pedunculated formations most commonly located in the atrioventricular valves.
• Most common sites are aortic and mitral valves followed by tricuspid valve, LVOT and interventricular septum and rarely, the eustachian valve.9,10
• Papillary fibroelastoma represents 75% of all valvular tumors.
• Second most common primary cardiac tumor of adults.
• Some synonyms:
- Giant Lambl excrescence
- Papilloma of valves
- Myxofibroma
- Myxoma of valves
- Hyaline fibroma
- Fibroma of valves
Pattern of Papillary Fibroelastoma
• Attachment to a cardiac valve.
• Presence of a pedicle.
• Papillariferous surface and papillary fronds detectable by echocardiography (Figs 20.19 to 20.22).
MALIGNANT TuMORS
The most common primary cardiac malignant tumor is a cardiac sarcoma.7 Subtypes include angiosarcoma (most common), undifferentiated sarcoma (second commonest), malignant fibrous histiocytoma (11-24%), leiomyosarcoma (8-9%), and osteosarcoma (3-9%).
An angiosarcoma arises preferentially in the right atrium of middle-aged men and can be very bulky (Figs 20.23 and 20.24). Obstruction of the right side's blood flow may cause right-sided heart failure or may mimic pericardial tamponade.

Fig. 20.21: 3DE image in cross-section showing fibroelastoma attached to P2 segment of the mitral valve.
(AML: Anterior mitral leaflet ).

Fig. 20.22: Complete atrioventricular canal defect with papillary fibroelastoma of the mitral valve (arrow).

Fig. 20.23: Angiosarcoma of the right atrium attached to its free wall and extending into the right ventricle (RV).

Fig. 20.24: Mobile mass in the left ventricle (LV) apex with normal wall motion abnormality. Histologically, it was rhabdomyosarcoma.
differential diagnosis of cardiac tumors
Following differential diagnoses need to considered (Figs 20.25 to 20.33):
• Thrombi
• Anomalous muscle
• Mural hematoma
• Cysts
• Infective masses
• Abscess
Hydatid cysts are frequent in certain regions of the world and present with myriad features resembling tumors.11
infective endocarditis
Invasion of endocardium or biofilm of the intravascular devices by microbes is called infective endocarditis (IE). Microbes produce destruction but local area swells up due to defense mechanisms mounted by host as well as organism. These localized swellings produce variable

Fig. 20.25: Cardiac masses (arrows) in the left ventricle (LV) that may be confused with multicentric tumor. Their disappearance (right lower panel) after sometimes, suggests these were thrombi.

Fig. 20.26: Anomalous muscle bundle (arrow) in the left ventrical (LV) cavity that can be mistaken for a tumor.

Fig. 20.27: Abscess between aortic root and the left atrium in a patient with infective endocarditis of the aortic valve.
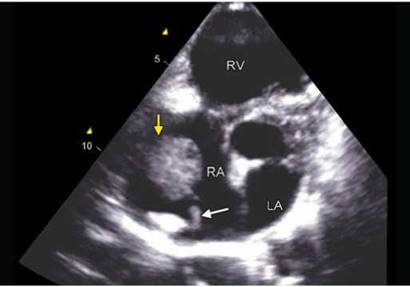
Fig. 20.28: Tumor-like infective mass attached to the pacing lead (white arrow) in a patient with arrhythmogenic right ventricle (RV) dysplasia.

Fig. 20.29: Localized pericardial effusion (PE) posterior to the left ventricle (LV).

Fig. 20.30: Caseous degeneration of the calcific mitral annulus mimicking a tumor (arrow). Outer calcified shell with echolucency in the center is characteristic.

Figs 20.31A to C: Myocardial dissection flap (arrow) showing movement during various phases in cardiac cycle (A to C) that can mimic a tumor.

Fig. 20.32: 3DE image. Hydatid cyst (arrows) in the left ventricular free wall that is abnormally thickened and with distorted cavity.

Fig. 20.33: Tumor-like invasion of the left ventricle (LV) free wall due to hydatid cyst (arrow).

Fig. 20.34: 3DE image. Pedunculated mass attached to the anterior mitral leaflet (arrow) in a patient with fever. Vegetation needs differentiation from fibroelastoma. Right lower panel shows gross appearance on surgery.

Fig. 20.35: Irregular mass along the right ventricular free wall (arrows) in a patient with prior pacemaker implantation and fever (transesophageal echocardiographic view).
infective masses called vegetations.12 Predispositions are and intracardiac devices, rheumatic heart disease and congenital heart disease, degenerated valves, prostheses so on.12

Fig. 20.36: Dehiscence of the ventricular septal defect (VSD) patch due to infective endocarditis (arrow) with vegetations on the surface.

Fig. 20.37: Perforation in the anterior mitral leaflet seen in transesophageal echocardiography 110° view (arrow).

Fig. 20.38: 3D TEE image. A hole in the anterior mitral leaflet (arrow) with a mass along margins is seen.

Fig. 20.39: 2D TEE four-chamber view showing flow by color Doppler through the hole in anterior mitral leaflet.
Endocardial involvement is a major criterion amongst the modified Duke criteria for diagnosis of IE.1314 Endocardial involvement can be of the following type:
• Infective vegetation attached to endocardium (Fig. 20.34).
• Masses attached to the intravascular or intracardiac devices (Figs 20.35 and 20.36).
• Abscess.
• Valvular perforation (Figs 20.37 to 20.39).
• Aneurysm or Fistula formation due to infection (Figs 20.40 and 20.41).
Vegetation: An oscillating polypoid mass with independent motion attached to an endocardial surface in a patient with clinically suspected IE (Fig. 20.34).
Clinical presentation of IE is showing a change over time due to graying of the population, greater use of
intracardiac devices, wide spread use of immuno- supressive drugs and changing microbes.15,16
Aneurysm formation is a compelling indication for
surgery1718
Transesophageal echocardiography is the preferred imaging technique for the diagnosis and management of IE in adults with either high risk for IE or moderate to high clinical suspicion of IE or in patients in whom imaging by transthoracic echocardiography is difficult.19
Fungal infections produce large masses on the valves and should be differentiated from the tumors (Fig. 20.42).
points to remember
• With amelioration of infection, the vegetations can become smaller or larger and can disappear as well.

Fig. 20.40: 3D TEE image showing aneurysm of the anterior mitral leaflet (arrows). A vegetation is seen at the leaflet tip.

Fig. 20.41: Large pseudoaneurysm (P) of sinus of Valsalva obstructing the RV outflow tract secondary to infective endocarditis (IE) of the aortic valve. Small vegetations are seen on the aortic valve.

Fig. 20.42: Candida ball attached to mitral and aortic valves in an infant (arrows).

Fig. 20.43: Healed bright echoes on the aortic valve in the parasternal long-axis view.
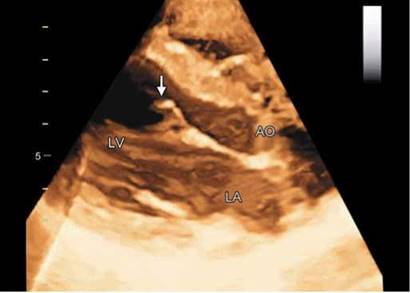
Fig. 20.44: Broken chord attached to the anterior mitral leaflet (arrow).
• Vegetations that are greater than 10 mm or are pedunculated have high predilection for embolization.
• Healed vegetations may become more echogenic and still retain their embolic potential (Fig. 20.43).
• The infective vegetations require to be differentiated from:
- Lambl's excrescences
- Fibroma of the valve
- Arantius nodules
- Degenerated and rheumatic nodules
- Ruptured chord (Fig. 20.44)
- Lupus vegetations20
- Myxomatous valves (Fig. 20.45)
• Presence of obstructive vegetations, fistula or abscess formation or embolization portend bad prognosis.

Fig. 20.45: Myxomatous posterior mitral leaflet with thickening of the distal half of the cusp (arrow).

Fig. 20.46: Infective vegetations on the tricuspid valve leaflets (arrows) in a subject with fever and on maintenance hemodialysis. (IAS: Inter-atrial septum).
• Vegetations on the tricuspid valve occur more often in patients with central venous catheters, intravenous drug abusers, patients on hemodialysis and those with pacemakers (Fig. 20.46).
references
1. Egolum UO, Stover DG, Anthony R, et al. Intracardiac thrombus: diagnosis, complications and management. Am J Med Sci. 2013;345(5):391-5.
2. Bakalli A, Georgievska-Ismail L, Koginaj D, et al. Prevalence of left chamber cardiac thrombi in patients with dilated left ventricle at sinus rhythm: the role of transesophageal echocardiography. J Clin Ultrasound. 2013;41(1):38-45.
3. Lee JM, Park JJ, Jung HW, et al. Left ventricular thrombus and subsequent thromboembolism, comparison of anticoagulation, surgical removal, and antiplatelet agents. J Atheroscler ttromb. 2013;20(1):73-93.
4. Edwards FH, Hale D, Cohen A, et al. Primary cardiac valve tumors. Ann ttorac Surg. 1991;52(5):1127-31.
5. Nield LE, Mendelson M, Ahmad N, et al. Clinical review of obstructive primary cardiac tumors in childhood. Congenit Heart Dis. 2013.
6. Barreiro M, Renilla A, Jimenez JM, et al. Primary cardiac tumors: 32 years of experience from a Spanish tertiary surgical center. Cardiovasc Pathol. 2013.
7. Burnside N, MacGowan SW. Malignant primary cardiac tumours. Interact Cardiovasc ttorac Surg. 2012; 15(6): 1004-6.
8. Ha JW, Kang WC, Chung N, et al. Echocardiographic and morphologic characteristics of left atrial myxoma and their relation to systemic embolism. Am J Cardiol. 1999;83(11):1579-82, A8.
9. Harling L, Athanasiou T, Ashrafian H, et al. Minimal access excision of aortic valve fibroelastoma: a case report and review of the literature. J Cardiothorac Surg. 2012;7:80.
10. Klarich KW, Enriquez-Sarano M, Gura GM, et al. Papillary fibroelastoma: echocardiographic characteristics for diagnosis and pathologic correlation. J Am Coll Cardiol. 1997;30(3):784-90.
11. Abhishek V Avinash V. Cardiac hydatid disease: literature review. Asian Cardiovasc ttorac Ann. 2012;20(6):747-50.
12. Cecchi E, Chirillo F, Faggiano P, et al. For the Italian Registry on Infective Endocarditis (RIEI) Investigators. the Diagnostic Utility of Transthoracic Echocardiography for the Diagnosis of Infective Endocarditis in the Real World of the Italian Registry on Infective Endocarditis. Echocardiography. 2013;30(8):871-9.
13. Martos-Perez F, Reguera JM, Colmenero JD. Comparable sensitivity of the Duke criteria and the modified Beth Israel criteria for diagnosing infective endocarditis. Clin Infect Dis. 1996;23(2):410-11.
14. Tissieres P, Gervaix A, Beghetti M, et al. Value and limitations of the von Reyn, Duke, and modified Duke criteria for the diagnosis of infective endocarditis in children. Pediatrics. 2003;112(6 Pt 1):e467.
15. Nakagawa T, Wada H, Sakakura K, et al. Clinical features of infective endocarditis: Comparison between the 1990s and 2000s. J Cardiol. 2013;pii:50914-5087.
16. Hoen B, Duval X. Clinical practice. Infective endocarditis. N Engl J Med. 2013;368(15):1425-33.
17. Silbiger JJ, Krasner A, Chikwe J, et al. Pseudoaneurysm formation in infective endocarditis. Echocardiography. 2013.
18. Edwards NC, Smith NL, Steeds RP. Beyond the artery and ventricle-an aneurysm of the mitral valve leaflet. Echocardiography. 2013;30(8):E258-9.
19. Bruun NE, Habib G, ttuny F, et al. Cardiac imaging in infectious endocarditis. Eur Heart J. 2013.
20. Contractor T, Bell A, Khasnis A, et al. Antiphospholipid antibody-associated non-infective mitral valve endocarditis successfully treated with medical therapy. J Heart Valve Dis. 2013;22(1):36-8.