Nicole D. Fleming, MD
Oliver Dorigo, MD, PhD
ENDOMETRIAL HYPERPLASIA & CARCINOMA
ESSENTIALS OF DIAGNOSIS
![]() Abnormal uterine bleeding: menorrhagia, metrorrhagia, or postmenopausal bleeding
Abnormal uterine bleeding: menorrhagia, metrorrhagia, or postmenopausal bleeding
![]() Risk factors: hyperestrogenism—long-term exposure to unopposed estrogens (polycystic ovarian syndrome, chronic anovulation, late menopause, and exogenous estrogens); metabolic syndrome including diabetes, hypertension, and obesity; nulliparity; increasing age; history of breast cancer; genetic predisposition (hereditary nonpolyposis colon cancer syndrome)
Risk factors: hyperestrogenism—long-term exposure to unopposed estrogens (polycystic ovarian syndrome, chronic anovulation, late menopause, and exogenous estrogens); metabolic syndrome including diabetes, hypertension, and obesity; nulliparity; increasing age; history of breast cancer; genetic predisposition (hereditary nonpolyposis colon cancer syndrome)
![]() Diagnosis: endometrial sampling, ultrasonography
Diagnosis: endometrial sampling, ultrasonography
Pathogenesis
Endometrial cancer is the most common gynecologic malignancy. The American Cancer Society estimates that over 43,000 new cases will be diagnosed in 2010, and over 7900 women will die from endometrial cancer. In the United States, white women have a lifetime risk of endometrial carcinoma of 2.4% compared with 1.3% for black women; however, survival for white women is about 8% greater at each stage of diagnosis compared to black women. The peak incidence of onset is in the seventh decade, but 25% of cancers occur in premenopausal women, and the disease has even been reported in women ages 20–30 years.
Most endometrial carcinomas arise on the background of endometrial hyperplasia and are well-differentiated tumors. There are 2 major types of endometrial cancer. Type I tumors are more common (85%) and tend to occur in younger women. These are associated with either endogenous or exogenous unopposed estrogen exposure and usually consist of a low-grade or well-differentiated tumor with a favorable prognosis. Type II tumors grow independent of estrogen, are associated with endometrial atrophy, and occur in an older population. Poorly differentiated endometrioid or nonendometrioid, such as papillary serous and clear cell, histologies are included in type II tumors and confer a high risk of relapse with poor prognosis. Gene expression profiles have also been shown to be different between type I and II tumors, with PTEN mutations more common in type I and p53 overexpression more common in type II tumors.
Estrogens and progesterone are the 2 main hormones that influence the metabolic and proliferative state of the endometrium. In general, estrogens stimulate the endometrium, unlike progesterone, which has an antiproliferative effect. Long-term exposure to estrogens can lead to endometrial hyperplasia and, subsequently, to hormone-driven atypical endometrial hyperplasia and endometrial cancer. Clinical circumstances with chronically high levels of estrogenic stimulation include obesity, metabolic syndrome, polycystic ovary syndrome, exogenous and unopposed estrogen replacement therapy, and chronic anovulation in the premenopausal women. Granulosa cell tumors of the ovary can produce high levels of estrogens and can be associated with endometrial hyperplasia or cancer. The selective estrogen receptor modulator (SERM) tamoxifen used for adjuvant therapy in breast cancer has a weak estrogenic effect on the endometrium and increases the incidence of endometrial cancer by about 2- to 3-fold. However, the benefit of tamoxifen therapy for breast cancer outweighed the potential increase in endometrial cancer reported with a 38% reduction in the 5-year cumulative hazard rate in the tamoxifen-treated group.
More than a dozen case-control studies indicate an association between estrogen administration and endometrial carcinoma. These studies report a 2- to 10-fold increase in the incidence of endometrial carcinoma in women receiving exogenous unopposed estrogens. The risk of cancer is related to both the dose and the duration of exposure and diminishes with cessation of estrogen use. The risk seems to be neutralized by the addition of cyclic progestin for 10 days at least every 1–3 months. In women without a hysterectomy, progestin should be added to the treatment to oppose the effect of estrogens on the endometrium. Endometrial biopsies to rule out endometrial hyperplasia or pelvic ultrasonography to evaluate the thickness of the endometrial stripe should be obtained if abnormal bleeding occurs.
About 5–6% of endometrial cancer cases develop on a genetic background. Women with a personal history of ovarian, colon, or breast cancer as well as those with a family history of endometrial cancer may be at higher risk. In hereditary nonpolyposis colorectal cancer (HNPCC) or Lynch syndrome, there is an autosomal dominant pattern of inheritance for colon and endometrial cancers most commonly, and rectal, ovarian, small bowel, and renal cancers less frequently. Patients with Lynch syndrome have an up to 70% lifetime risk of developing endometrial cancer, and a 10–12% risk of developing ovarian cancer. Most cases of HNPCC are a result of alterations in mismatch repair genes MSH2, MLH1, or MSH6.
Surgical Staging
In 1988, the Cancer Committee of the International Federation of Gynecology and Obstetrics (FIGO) introduced a surgical staging system for endometrial carcinoma based on abdominal exploration, pelvic washings, total hysterectomy with salpingo-oophorectomy, and selective pelvic and periaortic lymph node biopsies. This was revised by the FIGO Committee on Gynecologic Oncology in 2009 (Table 49–1). The grade of the tumors refers to the architecture and nuclear atypia on histology. The architecture of the tumor is judged by the percentage of differentiated (glandular) versus nondifferentiated (solid) elements within the tumor specimen. Grade 1 tumors consist of at least 95% glandular tissue and have <5% of a nonsquamous solid growth pattern. Areas of squamous differentiation are not considered to be solid tumor growth. Grade 2 tumors contain 6–50% of a nonsquamous solid growth pattern. Tumors with more than 50% of a solid pattern are classified as grade 3. The nuclear grade depends on the appearance of the nucleus (size of nucleus, chromatin pattern) and is more subjective. An architectural grade of 1 or 2 is raised by 1 point in the presence of significant nuclear atypia (nuclear grade 3).
Table 49–1. International Federation of Gynecology and Obstetrics (FIGO) surgical staging of carcinoma of the corpus uteri (2009).

Surgical stage I tumors account for 75% of all endometrial carcinomas, which explains the relatively good overall prognosis. Eleven percent of cancers are surgical stage II, and the remaining 11% and 3% are surgical stages III and IV, respectively.
A. Endometrial Hyperplasia
The glandular hyperplasias of the endometrium are benign conditions that can be classified as simple or complex and either with or without atypia. Because of their association with hyperestrogenic states, the atypical hyperplasias are considered premalignant lesions. Because endometrial hyperplasia and endometrial carcinoma present clinically as abnormal bleeding, thorough endometrial sampling or fractional curettage is always necessary when hyperplasia is present to rule out coexisting carcinoma.
1. Hyperplasia without atypia—Microscopically, this type of hyperplasia shows crowding of glands in the stroma without nuclear atypia. This type of hyperplasia is frequently asymptomatic and found incidentally in hysterectomy specimens. When followed without treatment over a 15-year period, approximately 1% progressed to endometrial cancer, whereas 80% spontaneously regressed.
Simple hyperplasia without atypia describes enlarged glands with an irregular outline. Long-term follow-up reveals a 1% risk of progression to carcinoma if untreated. Complex hyperplasia without atypia (previously designated “adenomatous hyperplasia”) describes a complex, crowded back-to-back glandular appearance with intraluminal papillae. Complex hyperplasia regresses under progestin therapy in approximately 85% of cases but progresses to cancer in 3–5% if untreated.
2. Hyperplasia with atypia—The histology of hyperplasia with atypia is characterized by endometrial glands that are lined with enlarged cells. An increased nuclear-to-cytoplasmic ratio is a sign of increased nuclear activity (eg, transcription). The nuclei may be irregular with coarse chromatin clumping and prominent nucleoli. These hyperplasias are generally considered premalignant. Progression to carcinoma occurs in 10% of simple atypical and in 30% of complex atypical hyperplasias. The majority of lesions regress with progestin therapy but have a higher rate of relapse when therapy is stopped compared to lesions without atypia. In peri- and postmenopausal patients with atypical hyperplasias who relapse after progestin therapy or who cannot tolerate the associated side effects, vaginal or abdominal hysterectomy is recommended.
A recent prospective cohort study by the Gynecologic Oncology Group (GOG) demonstrated that in patients with untreated atypical endometrial hyperplasia on preoperative biopsy, 42.6% had a concurrent endometrial carcinoma at hysterectomy. In the subset of women who had biopsies interpreted as less than atypical endometrial hyperplasia who underwent hysterectomy, 18.9% had cancer.
The term atypical endometrial hyperplasia should be applied to endometrial neoplasia without invasion. Severe atypical endometrial hyperplasia and adenocarcinoma in situ describe preinvasive histologies that are frequently difficult to distinguish from early invasive endometrial cancer. It is still a matter of debate whether the term adenocarcinoma in situ should be used for endometrial pathology. In contrast, the precursor lesion for serous carcinomas in endometrial intraepithelial carcinoma shows pleomorphic tumor cells in the epithelium of the endometrial surface and the underlying glands without stromal invasion.
In recent years, the term endometrial intraepithelial neoplasia (EIN) has been used to describe premalignant lesion of the uterine lining that predisposes to endometrioid endometrial adenocarcinoma. EIN lesions have been discovered by molecular, histologic, and clinical outcome studies and comprise a subset of endometrial hyperplasia lesions. EIN should not be confused with intraepithelial carcinoma (EIC), which is an early stage of papillary serous adenocarcinomas originating from the glandular endometrium.
B. Endometrial Carcinoma
Endometrial cancer is characterized by obvious hyperplasia and anaplasia of the glandular elements, with invasion of underlying stroma, myometrium, and vascular spaces. Although atypical complex hyperplasia is thought to be a precursor lesion, only approximately 25% of patients with endometrial carcinoma have a history of hyperplasia.
Important prognostic factors include stage, histologic grade and cell type, depth of myometrial invasion, presence of lymphovascular space involvement (LVSI), lymph node status, involvement of the lower uterine segment, and size of tumor.
Endometrial cancers of endometrioid histology of any grade with no myometrial invasion are almost never associated with lymph node metastases. The depth of myometrial invasion and histologic grade are correlated with the incidence of pelvic and aortic lymph node metastases. In the surgical pathology study GOG-33, nodal disease was more frequent with increasing grade (3% grade 1, 9% grade 2, 18% grade 3), depth of invasion (1% endometrium only, 5% inner one-third, 6% middle one-third, 25% outer one-third myometrial invasion), and LVSI (27% with LVSI, 7% without LVSI). Patients with poorly differentiated deeply invasive cancers have about a 35% incidence of involved pelvic nodes and a 10–20% incidence of aortic node metastases. Because patients with lymph node metastases are at very high risk for recurrence, these pathologic features have serious implications for treatment planning.
Endometrial cancer can spread by 4 possible routes: direct extension, lymphatic metastases, peritoneal implants after transtubal spread, and hematogenous spread. Undifferentiated lesions (grade 3) may spread to the pelvic and aortic nodes while still confined to the superficial myometrium. In serous and clear cell subtypes, the spread pattern is similar to that of ovarian cancer, and upper abdominal metastases are common. Hematogenous metastases to the lungs are uncommon with primary tumors limited to the uterus but do occur with recurrent or disseminated disease. Endometrial cancer spreads via a dual pathway to the pelvic and aortic lymph nodes (Fig. 49–1). The aortic nodes are rarely involved when the pelvic nodes are free of metastases. The lymph nodes most commonly involved in endometrial cancer are found in the obturator space.
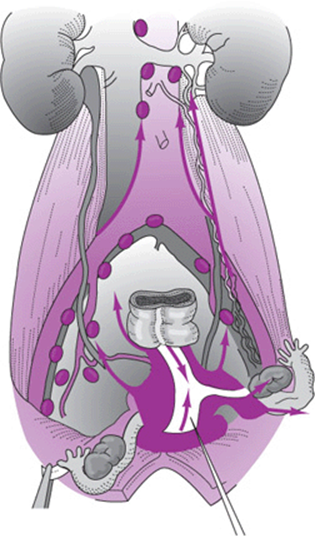
Figure 49–1. Dual lymphatic spread pattern of endometrial carcinoma.
Vaginal metastases occur by submucosal lymphatic or vascular metastases in approximately 3–8% of patients with clinical stage I disease. The concept that these metastases occur by spillage of tumor through the cervix at the time of surgery lacks convincing support. However, vaginal metastases are more common with higher histologic grade and with lower uterine segment or cervical involvement.
Pathologists recognize various histologic types of endometrial carcinoma. Approximately 80% of all endometrial cancers are of the endometrioid type with several variants: villoglandular, secretory, with squamous differentiation, and with ciliated cells. These types have similar presenting symptoms and signs, patterns of spread, and general clinical behavior. For this reason, they can be considered collectively for purposes of clinical workup, differential diagnosis, and treatment. Endometrial adenocarcinomas of the nonendometrioid phenotype show mucinous, serous, clear cell, squamous, small cell, mixed, or transitional cell differentiation.
1. Adenocarcinoma—The most common type of endometrial carcinoma is adenocarcinoma, composed of malignant glands that range from well-differentiated (grade 1) to anaplastic carcinoma (grade 3). To determine stage and prognosis, the tumor is usually graded by the most undifferentiated area visible under the microscope (Fig. 49–2). In the United States, adenocarcinoma comprises 80% of endometrial carcinomas.

Figure 49–2. Adenocarcinoma of the endometrium. Note the sharp demarcation of the tumor at the isthmus.
2. Adenocarcinoma with squamous differentiation—Approximately 25% of endometrioid carcinomas contain focal to extensive squamous elements, ranging from bland squamous cells to foci that could be viewed as squamous carcinoma. The behavior of the tumors with squamous differentiation is dependent on the grade of the glandular component.
3. Serous carcinoma—Histologically, this cancer is identical to the complex papillary architecture seen in serous carcinomas of the ovary. Serous carcinoma represents approximately 10% of endometrial carcinomas. Women with serous carcinoma are more likely to be older and less likely to have hyperestrogenic states. These tumors account for 50% of all relapses in stage I tumors. Serous tumors spread early and involve peritoneal surfaces of the pelvis and abdomen. The tumors also have a propensity for myometrial and lymphatic invasion. The prognosis is unfavorable, and patients with serous tumors should be treated in a manner similar to that of patients with ovarian tumors.
4. Clear cell carcinoma—This subtype is not associated with clear cell carcinomas of the cervix and vagina that are seen in young women with diethylstilbestrol exposure. Clear cell carcinomas encompass approximately 1–4% of all endometrial carcinomas. Its microscopic appearance is significant for clear cells or hobnail cells. Solid, papillary, tubular, and cystic patterns are possible. Clear cell carcinoma is commonly high grade and aggressive with deep invasion and is seen at an advanced stage. The mean age at diagnosis is approximately 67 years, which is similar to the serous subtype, and it is not associated with a hyperestrogenic state.
5. Miscellaneous subtypes—Mucinous carcinomas make up 1–9% of endometrial adenocarcinomas. The cytoplasm is positive for mucin, carcinoembryonic antigen, and periodic acid-Schiff stain. Secretory carcinoma, present in 1–2% of cases, exhibits subnuclear or supranuclear vacuoles resembling early secretory endometrium. These rare cancers behave in a manner similar to that of typical endometrial carcinomas. Pure squamous cell carcinomas are extremely rare (<1%) and are associated with cervical stenosis, pyometra, and chronic inflammation.
Prevention
A doubling of the incidence of endometrial cancer in the 1970s correlated with unopposed estrogen use in hormone replacement and sequential oral contraceptives over the previous 10 years. The declining incidence in the 1980s paralleled progesterone use in hormone replacement regimens and low-dose estrogen combination birth control pills.
Estrogens are implicated as a causative factor in endometrial carcinoma based mainly on the high incidence of this disease in patients with presumed alterations in estrogen metabolism and in those who take exogenous estrogens. Furthermore, patients with anovulatory cycles are at higher risk of developing endometrial cancer because of prolonged periods of estrogenic stimulation of the endometrium without the opposing effects of progesterone. Progesterone has an antiproliferative effect on the endometrium and can induce apoptosis of endometrial cells.
Classically, endometrial carcinoma affects the obese, nulliparous, infertile, hypertensive, and diabetic white woman, but it can occur in the absence of all these factors. Unlike cervical cancer, it is not related to sexual history.
Prophylactic hysterectomy and bilateral salpingo-oophorectomy have been shown to be effective strategies for preventing endometrial and ovarian cancer in these high-risk patients.
Several modifiable risk factors for endometrial carcinoma have been described, including obesity, diabetes, hypertension, and nulliparity. Prevention of endometrial cancer is primarily based on weight control, physical exercise, adequate control of diabetes and hypertension, and increased surveillance of women at high risk. In addition, a careful family history of each patient will help identify patients with a genetic predisposition for endometrial cancer, for example, as part of the HNPCC syndrome. If appropriate, these patients should undergo genetic counseling and genetic testing. A hysterectomy after the completion of childbearing is appropriate for patients with HNPCC syndrome given the lifetime risk for endometrial cancer of up to 70%. Hormone therapy in postmenopausal patients without hysterectomy should always include a progestational agent to oppose the action of estrogens on the endometrium. Estrogens should be administered either continuously or cyclically using the lowest dose that controls symptoms. Progesterone (10 mg of medroxyprogesterone acetate or 200 mg of micronized progesterone) should be added for the last 10–14 days of the cycle to neutralize the risk of endometrial carcinoma. Alternatively, if estrogen and progesterone are administered continuously, 2.5 mg of medroxyprogesterone acetate is given daily.
Clinical Findings
The onset of endometrial bleeding facilitates detection in the earlier stages of disease. The most common presenting symptom is abnormal vaginal bleeding, particularly post-menopausal bleeding. Less frequently, severe cramps from hematometra or pyometra caused by an obliterated endocervical canal in elderly patients may be the presenting symptom.
A. Symptoms & Signs
Abnormal bleeding occurs in approximately 80% of patients and is the most important and early symptom of endometrial carcinoma. An abnormal vaginal discharge, especially after menopause or intermittent spotting, is reported by some patients. During the premenopausal years, the bleeding is usually described as excessive flow at the time of menstruation. However, bleeding may occur as intermenstrual spotting or premenstrual and postmenstrual bleeding. Approximately 5–10% of patients with postmenopausal bleeding have underlying cancer, but the probability increases with age and depends on underlying risk factors. Approximately 10% of patients complain of lower abdominal cramps and pain secondary to uterine contractions caused by detritus and blood trapped behind a stenotic cervical os (hematometra). If the uterine contents become infected, an abscess develops and sepsis may occur.
Physical examination is usually unremarkable but may reveal medical problems associated with advanced age. Speculum examination may confirm the presence of bleeding, but because it may be minimal and intermittent, blood might not be present. Atrophic vaginitis is frequently identified in these elderly women, but postmenopausal bleeding should never be ascribed to atrophy without a histologic sampling of the endometrium to rule out endometrial carcinoma. Bimanual and rectovaginal examination of the uterus in the early stages of the disease will be normal unless hematometra or pyometra is present. If the cancer is extensive at the time of presentation, the uterus may be enlarged and may be misdiagnosed as a benign condition such as leiomyomata. In advanced cases, the uterus may be fixed and immobile from parametrial extension.
Vaginal, vulvar, or inguinal–femoral lymph node metastases are rarely identified in early disease but are not uncommon in advanced cases or with recurrence following treatment. Ovarian metastases may cause marked enlargement of these organs.
B. Laboratory Findings
Routine laboratory findings are normal in most patients with endometrial carcinoma. If bleeding has been prolonged or profuse, anemia may be present. Cytologic study of specimens taken from the endocervix and posterior vaginal fornix can reveal adenocarcinoma in symptomatic patients. More important, endometrial carcinoma will be missed in 40% of symptomatic patients by routine cytologic examination. Accuracy has been greatly increased by aspiration cytologic study or biopsy (discussed under Special Examinations). Nevertheless, the Papanicolaou (Pap) smear is an integral part of the examination of all patients, because it identifies a small but definite percentage of patients with asymptomatic disease. Furthermore, the presence of benign endometrial cells in the cervical or vaginal smear of a menopausal or postmenopausal woman is associated with occult endometrial carcinoma in 2–6% of cases and in up to 25% with postmenopausal bleeding. Thus, any postmenopausal woman who shows endometrial cells on a routine cervical Pap smear requires evaluation for endometrial cancer, including endometrial sampling.
Routine blood counts, urinalysis, endocervical and vaginal pool cytology, chest radiography, stool guaiac, and sigmoidoscopy are useful ancillary diagnostic tests in patients with endometrial carcinoma. Liver function tests, blood urea nitrogen, serum creatinine, and a blood glucose test (because of the known relationship to diabetes) are considered routine. Serum CA-125 (cancer antigen-125), a well-established tumor marker for epithelial ovarian cancer, might be useful for endometrial cancer. Approximately 20% of patients with clinical stage I disease (preoperatively, the tumor appears to be confined to the uterus) have an elevated CA-125. In cases with extensive intraperitoneal spread or enlarged uterus, the tumor marker CA-125 may be markedly elevated. However, in contrast to patients with ovarian cancer, the value of CA-125 in the management of patients with endometrial cancer is limited.
C. Imaging Studies
Chest radiography might reveal metastases in patients with advanced disease but is rarely positive in the early stages. Colonoscopy is usually unnecessary in a patient with a negative stool guaiac test and normal sigmoidoscopic examination but should always be performed in the patient with gross or occult gastrointestinal bleeding or symptoms. In patients from families with HNPCC, a colonoscopy should be performed preoperatively, particularly if the patient screens positive for the HNPCC-associated DNA mismatch repair gene mutations.
Hysteroscopy can increase the diagnostic accuracy over office endometrial biopsy or dilatation and curettage. Hysteroscopy promotes the transtubal spread of tumor cells into the peritoneal cavity. However, the presence of a positive peritoneal cytology after hysteroscopy does not seem to alter the prognosis. Computed tomography is useful in assessing pelvic anatomy, visualizing enlarged lymph nodes in the pelvis and periaortic areas and diagnosing distant metastasis in the liver and lungs. Magnetic resonance imaging (MRI) is particularly helpful in identifying myometrial invasion and lower uterine segment or cervical involvement.
D. Special Examinations
1. Fractional curettage—Dilatation and fractional curettage (D&C) is the definitive procedure for diagnosis of endometrial carcinoma. It should be performed with the patient under anesthesia to provide an opportunity for a thorough and more accurate pelvic examination. It is carried out by careful and complete curettage of the endocervical canal followed by dilatation of the canal and circumferential curettage of the endometrial cavity. When obvious cancer is present with the first passes of the curette, the procedure should be terminated as long as sufficient tissue for analysis has been obtained from the endocervix and endometrium. Perforation of the uterus followed by intraperitoneal contamination with malignant cells, blood, and bacteria is a common complication in patients with endometrial carcinoma and can usually be avoided by gentle surgical technique and limitation of the procedure to the extent necessary for accurate diagnosis and staging. D&C is never considered curative in these circumstances and should not be performed with the same vigor as therapeutic curettage.
2. Endometrial biopsy—This procedure is attractive because it can be performed in an outpatient setting, resulting in a substantial savings in cost. It can usually be done without anesthesia, although paracervical block is effective when necessary. Results of endometrial biopsies (EMB) correlate well with endometrial curettings with the accuracy to detect cancer between 91% and 95%. The accuracy of identifying cancers with EMB is higher in postmenopausal patients than in premenopausal patients. There is a false-negative rate of approximately 10%, and all symptomatic patients with a negative EMB need to undergo a formal D&C. There are many types of office biopsy techniques including a Pipelle, Novak curet (Fig. 49–3), and Vabra aspirator (Fig. 49–4). All types of EMB are notoriously inaccurate for diagnosing polyps and will miss a significant number of cases of endometrial hyperplasia as well.
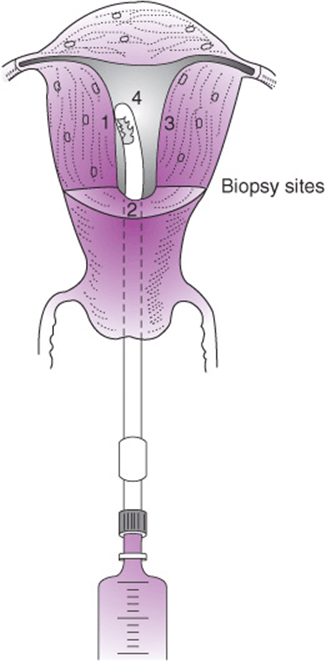
Figure 49–3. Technique of endometrial biopsy with Novak curet.
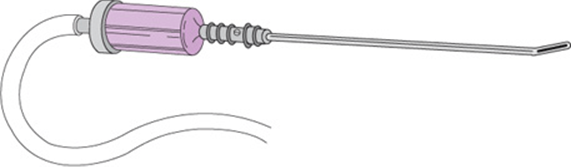
Figure 49–4. Vabra aspirator.
3. Pelvic ultrasonography—Ultrasonography can be helpful in the surveillance of asymptomatic, high-risk patients (eg, breast cancer patients on tamoxifen and women with strong family histories of endometrial cancer). Pelvic and transvaginal ultrasonography yield information about the size and shape of the uterus, as well as the thickness and surface contour of the endometrium. Transvaginal ultrasound measuring the lining thickness of the endometrium has an excellent negative predictive value for ruling out endometrial cancer or hyperplasia when the thickness is <5 mm, but provides less information when >5 mm. In postmenopausal women, an endometrial thickness of more than 5 mm is considered to be suspicious for hyperplasia or malignancy and should be further evaluated with an EMB. Transvaginal ultrasound, however, can yield a high false-positive rate in women who have been on tamoxifen for more than 2 years. The subendometrial edema that develops from tamoxifen use is indistinguishable from a thickened endometrial stripe. A sonohysterogram, which involves instilling sterile saline into the endometrial cavity prior to transvaginal ultrasound, can reduce false-positive results and better delineate the endometrial cavity.
4. Estrogen and Progesterone Receptor Assays—Estrogen and progesterone receptor assays should be obtained from the neoplastic tissue. This information helps in planning adjuvant or subsequent hormone therapy. Estrogen and progesterone receptor content are inversely proportional to histologic grade. In general, patients with tumors positive for 1 or 2 receptors have longer survival than patients with receptor-negative tumors. Furthermore, patients with receptor-positive tumors might be candidates for hormone-based therapy of recurrent tumor disease.
Differential Diagnosis
In the asymptomatic patient, a diagnosis may be made incidentally from an abnormal Pap smear, but cytologic discovery of endometrial cancer is not consistent and should not be relied on for early diagnosis. Screening for endometrial cancer in the general population is not recommended but should be performed for patients with a Lynch or HNPCC syndrome.
Clinically, the differential diagnosis of endometrial carcinoma includes all the various causes of abnormal uterine bleeding. In the premenopausal patient, complications of early pregnancy, such as threatened or incomplete abortion, must be considered initially. Other causes of bleeding in premenopausal patients are leiomyomata, endometrial hyperplasia and polyps, cervical polyps, an intrauterine device, and various genital or metastatic cancers. Cervical, endometrial, tubal, and ovarian neoplasms all can cause abnormal uterine bleeding. Although rare, metastatic cancers from the bowel, bladder, and breast have also been reported to cause abnormal uterine bleeding. After exclusion of anatomic causes for vaginal bleeding, a workup for hemophilias should be performed. In the postmenopausal age group, the differential diagnosis includes atrophic vaginitis, exogenous estrogens, endometrial hyperplasia and polyps, and various genital neoplasms. The likelihood of cancer increases with age. In the patient with a normal pelvic examination and recurrent postmenopausal bleeding following a recent negative D&C, tubal and ovarian cancer must be strongly considered. Patients with recurrent unexplained episodes of postmenopausal uterine bleeding should be considered for total hysterectomy and bilateral salpingo-oophorectomy.
Complications
Patients with advanced disease and deep myometrial invasion may present with severe anemia secondary to chronic blood loss or acute hemorrhage. If bleeding is significant and continuous, a short-term boost of radiation therapy is usually effective in slowing the hemorrhage.
The presence of a hematometra can be confirmed by sounding the uterus under anesthesia, followed by dilatation of the cervix to allow adequate drainage. When a pyometra is present, the patient may present with peritonitis or generalized sepsis, with all the consequent complications.
Perforation of the uterus at the time of D&C or EMB is not an uncommon problem. If the perforating instrument is large, loops of small bowel may be inadvertently retrieved through the cervical canal. A large perforation warrants laparoscopy or laparotomy to evaluate and repair the damage. If significant contamination of the peritoneal cavity with blood or necrotic tumor has occurred, the patient should be treated with broad-spectrum antibiotics to prevent peritonitis. Perforation in the patient with endometrial cancer should be viewed as a serious complication, as spillage of tumor into the peritoneal cavity may alter her prognosis.
Treatment
The mainstay of treatment is surgery, including a total hysterectomy with bilateral salpingo-oophorectomy and staging with pelvic and periaortic lymphadenectomy. Further postoperative therapy depends on the particular histologic characteristics and the extent of the tumor.
The majority of endometrial cancer cases are diagnosed at an early stage and can be treated with high cure rates. The most important treatment modality is surgery with total hysterectomy, bilateral salpingo-oophorectomy, and staging, including pelvic and periaortic lymphadenectomy. Primary radiation therapy is used only in patients with medical contraindications for surgery or advanced pelvic disease. It has been repeatedly demonstrated that radiation therapy can cure endometrial carcinoma in some patients. However, radiation therapy averages about a 20% lower cure rate compared to surgery in stage I disease. Primary chemotherapy is used infrequently and mostly in patients with metastatic disease. High-dose progesterone therapy, commonly with medroxyprogesterone acetate or megestrol acetate, may be used for patients who are inoperable or in younger patients who elect for fertility preservation. The overall response to high-dose progesterone therapy is up to 75% in grade 1 endometrial cancer cases limited to the endometrium. To verify that the patient is responding to therapy, regular endometrial sampling needs to be performed.
Adjuvant treatment is dependent on the results of surgical staging and histology. For example, adjuvant radiation therapy is frequently used in high-risk endometrial cancers of endometrioid histology to prevent pelvic recurrences. Advanced pelvic disease may be treated with radiation followed by systemic chemotherapy. Serous cancers of the endometrium behave biologically similar to ovarian cancer and are treated with adjuvant platinum-based chemotherapy possibly in conjunction with radiation.
A. Emergency Measures
Patients with endometrial adenocarcinoma may present with severe anemia after prolonged periods of vaginal bleeding. Acute and massive blood loss may lead to hypovolemic shock. The management of these patients includes stabilization of vital signs with volume substitution and blood transfusion. A tamponade of the uterus using vaginal packing might be useful, particularly in the presence of a bleeding cervical or vaginal tumor. Monsel’s solution or silver nitrate can further aid in obtaining hemostasis. An emergency D&C might help to control the bleeding but has to be performed with great caution to avoid perforation. If bleeding does not subside, a high-dose radiation boost to the whole pelvis is usually the treatment of choice to acutely control uterine bleeding in this situation. Rarely, in the face of very advanced lesions, embolization of the hypogastric arteries via percutaneous selective angiography may be required to control hemorrhage before treatment can be initiated. Hysterectomy should always be considered if it can be accomplished safely without jeopardizing curative therapy.
Elderly patients may present with severe lower abdominal pain and cramping secondary to hematometra or pyometra; these complications result from endometrial carcinoma in more than 50% of cases. When adequate blood levels of broad-spectrum antibiotics are established, the cervix should be dilated and the endometrial cavity adequately drained. In this setting, vigorous D&C is contraindicated because of the high risk of uterine perforation. If the cervix is well dilated, an indwelling drain is usually unnecessary, but if sepsis is not controlled within 24–48 hours, the patient should be re-examined to ascertain cervical patency. Once the infection has completely subsided and the patient has been afebrile for 7–10 days, gentle fractional curettage should be performed if the diagnosis was not confirmed at the initial procedure.
B. Radiation Therapy
Radiation therapy is used as primary therapy in patients considered too medically unstable for laparotomy. Adjuvant preoperative radiation is no longer used unless the patient presents with gross cervical involvement. In this situation, after preoperative whole-pelvic radiation and an intracavitary implant, an extrafascial hysterectomy is performed. Relative contraindications to preoperative radiation therapy include the presence of a pelvic mass, a pelvic kidney, pyometra, history of a pelvic abscess, prior pelvic radiation, and previous multiple laparotomies (see Chapter 52).
Adjuvant radiation therapy has been shown to significantly improve locoregional control in early-stage, high-risk endometrial cancer. Two randomized controlled clinical trials, one conducted in the United States by the GOG (GOG-99) and the other one conducted in Europe (PORTEC trial), demonstrated that the addition of postoperative external-beam radiation therapy (EBRT) in early-stage, high-risk endometrial cancers decreased the rate of vaginal and pelvic recurrences versus surgery alone. However, local radiation alone did not result in improvement of overall survival.
Accordingly, in the presence of extrauterine extension, lower uterine segment or cervical involvement, poor histologic differentiation, papillary serous or clear cell histology, or myometrial invasion greater than one-third of the full thickness, adjuvant radiation therapy is recommended. In the absence of these findings, it is difficult to justify the risk and morbidity of any additional treatment beyond simple total abdominal hysterectomy and bilateral salpingo-oophorectomy. In stage III and IV disease, the optimal adjuvant therapy has also been of much debate. Management options in advanced-stage disease can include systemic chemotherapy alone or in addition to radiation therapy to improve locoregional control. Overall, adjuvant therapy in patients with early-stage, high-risk endometrioid endometrial cancers should be individualized based on stage and grade, on whether surgical lymph node staging was performed, and on the risk of nodal versus vaginal recurrence.
C. Surgical Treatment
Because bleeding is usually an early sign of endometrial carcinoma, most patients present with early disease and can be adequately and completely treated by simple hysterectomy. Staging includes a bilateral salpingo-oophorectomy, peritoneal washings for cytology, and removal of pelvic and periaortic lymph nodes. Recently, minimally invasive methods, including laparoscopic-assisted and robotic-assisted endometrial cancer staging procedures, have been successfully performed. Laparoscopic surgery is currently the preferred management for patients with endometrial cancer, because patient outcome is equivalent to open surgery. In addition, length of hospital stay and recovery time are both shorter.
Pelvic and para-aortic lymphadenectomy play an important role in the surgical staging of endometrial cancer. A gross pathologic assessment of the uterus should be performed during surgery to determine the need for surgical staging in patients with grade 1 or 2 endometrioid adenocarcinomas. Patients who require surgical staging are patients with stage I disease with grade 3 lesions, tumors >2 cm in maximum dimension, tumors with >50% myometrial invasion, cervical extension, and evidence of extrauterine spread. Furthermore, staging should be performed in clear cell and papillary serous carcinomas in all cases because of a high incidence of lymphatic spread. However, the criteria for lymphadenectomy are not universally accepted and are under constant investigation. The therapeutic role of lymphadenectomy is still under investigation. Several studies have suggested that EBRT may be omitted or the radiation field reduced to the central pelvis if the lymph nodes are negative. Bulky, positive nodes, which are unlikely to respond to EBRT, should be removed during surgery.
Radical hysterectomy for stage II tumors is an accepted procedure that has the potential to omit adjuvant radiation therapy. A radical hysterectomy can also be an effective treatment for patients with recurrence following treatment with radiation therapy alone or for those who have previously received therapeutic doses of pelvic radiation therapy for other pelvic cancers. The increased risk of bowel or urinary tract injury in this setting must be understood and accepted by both patient and physician.
Patients who present with significant cervical involvement or vaginal and parametrial involvement should receive initial pelvic radiation. Exploratory laparotomy should then be considered in patients whose disease seems resectable. Hormonal therapy or chemotherapy is most appropriate for patients with clinical evidence of extrapelvic metastases. Palliative radiation to bone or brain metastases is beneficial for symptomatic relief. Pelvic radiation can be helpful for local tumor control and alleviation of bleeding.
D. Hormone Therapy
Progesterone has shown some efficacy in the treatment of recurrent endometrial carcinoma not amenable to irradiation or surgery. This type of therapy can be administered orally or parenterally. Oral megestrol, parenteral medroxyprogesterone acetate suspension, and parenteral hydroxyprogesterone caproate appear to have similar effectiveness with response rates of approximately 25%. Overall, approximately 13% of patients with recurrent disease appear to achieve long-term remission with progesterone therapy. The average duration of response is 20 months, and up to 30% of responders survive for 5 years. In general, the clinical response is better in patients with localized recurrence, well-differentiated tumors, long disease-free intervals, and positive estrogen or progesterone receptor status. Because some patients do not achieve remission until after 10–12 weeks of therapy, the minimum duration of treatment should be longer than 3 months. Although progesterones have a somewhat encouraging record in the treatment of recurrent endometrial adenocarcinoma, they are disappointing as prophylactic agents. They have not improved survival or decreased recurrence when used following definitive treatment of early-stage disease.
Tamoxifen, either alone or in combination with progesterone, has been used in advanced or recurrent endometrial cancer. Patients with well-differentiated, estrogen receptor– positive tumors and long disease-free intervals tend to have a better response to tamoxifen. Tamoxifen is administered orally at 10–20 mg twice daily. For single-agent tamoxifen, the overall response rate is approximately 15–20%. Studies using combination tamoxifen–progestin therapy suggest a possibly better clinical response of up to 40%.
E. Chemotherapy
Doxorubicin and cisplatin are the 2 most active agents in the treatment of advanced or recurrent endometrial cancer. Doxorubicin used as a single agent has an overall response rate of 38%, with 26% of the patients achieving a complete response. The combination of cisplatin and doxorubicin shows slightly longer survival than either agent alone. The addition of paclitaxel to cisplatin and doxorubicin shows an overall response rate of 57% with improved long-term survival compared to the same regimen without paclitaxel. More recently, the combination of paclitaxel and carboplatin has shown comparable response rates and less side effects. Other agents with antitumor activity against endometrial cancer include cyclophosphamide, hexamethylmelamine, and 5-fluorouracil.
Prognosis
The overall prognosis is considerably better than for the other major gynecologic cancers with 5-year survival rates of 96%, 67%, and 17% for local, regional, and distant disease at diagnosis, respectively.
The most important prognostic factors for endometrial cancer are stage, histologic type, grade, myometrial invasion, and the presence of lymphovascular space invasion. Identification of these risk factors is crucial for treatment decisions, surveillance, and counseling of the patient. The prognosis is worse with increasing age, higher pathologic grade, advanced-stage disease, increasing depth of myome-trial invasion, and presence of lymphovascular space invasion. Because the prognosis for each patient is dependent on a variety of factors, overall 5-year survival stratified by stage is indicated as a range of percentages. The overall 5-year survival rates are 81–95% for surgical stage I, 67–77% for stage II, 31–60% for stage III, and 5–20% for stage IV.
These figures underline the increasing risk for treatment failure and recurrence with increasing bulk and extension of tumor. In the absence of risk factors, a simple total abdominal hysterectomy and bilateral salpingo-oophorectomy should result in survival >95% at 5 years. However, in the presence of risk factors, a more aggressive surgical approach and using adjuvant radiation and chemotherapy may be warranted.
Amant F, Moerman P, Neven P, et al. Endometrial cancer. Lancet 2005;366(9484):491–505. PMID: 16084259.
American Cancer Society. American Cancer Society Facts and Figures 2010. Available at: http://www.cancer.org/Research/CancerFactsFigures/CancerFactsFigures/cancer-facts-and-figures-2010.
Cao QJ, Belbin T, Socci N, et al. Distinctive gene expression profiles by cDNA microarrays in endometrioid and serous carcinomas of the endometrium. Int J Gynecol Pathol 2004;23:321–329. PMID: 15389101.
Creutzberg C, van Putten WL, Koper PC, et al. Surgery and postoperative radiotherapy versus surgery alone for patients with stage 1 endometrial carcinoma: multicentre randomized trial. Lancet2000;355:1404–1411. PMID: 10791524.
Dijkhuizen FP, Mol BW, Brölmann HA, Heintz AP. The accuracy of endometrial sampling in the diagnosis of patients with endometrial carcinoma and hyperplasia: a meta-analysis. Cancer 2000;89:1765–1772. PMID: 11042572.
Duska LR, Berkowitz R, Matulonis U, et al. Pilot trial of TAC (paclitaxel, doxorubicin, and carboplatin) chemotherapy with filgrastim (r-metHuG-CSF) support followed by radiotherapy in patients with “high-risk” endometrial cancer. Gynecol Oncol 2005;96:198–203. PMID: 15589601.
FIGO Committee on Gynecologic Oncology. Revised FIGO staging for carcinoma of the vulva, cervix, and endometrium. Int J Gynecol Obstet 2009;105:103–104. PMID: 19367689.
Fung MFK, Reid A, Faught W, et al. Prospective longitudinal study of ultrasound screening for endometrial abnormalities in women with breast cancer receiving tamoxifen. Gynecol Oncol 2003;91:154–159. PMID: 14529676.
Karamursel BS, Guven S, Tulunay G, et al. Which surgical procedure for patients with atypical endometrial hyperplasia? Int J Gynecol Cancer 2005;15:127–131. PMID: 15670307.
Keys HM, Roberts JA, Brunetto VL et al. A phase III trial of surgery with or without adjunctive external pelvic radiation therapy in intermediate risk endometrial adenocarcinoma: a Gynecologic Oncology Group study. Gynecol Oncol 2004; 92:744–751. PMID: 14984936.
Koh WJ, Tran AB, Douglas JG, Stelzer KJ. Radiation therapy in endometrial cancer. Baillieres Best Pract Res Clin Obstet Gynaecol 2001;15:417–432. PMID: 11476563.
Lalloo F, Evans G. Molecular genetics and endometrial cancer. Baillieres Best Pract Res Clin Obstet Gynaecol 2001;15:355–363. PMID: 11476568.
Lu K, Dinh M, Kohlman W, et al. Gynecologic cancer as a “sentinel cancer” for women with hereditary nonpolyposis colorectal cancer syndrome. Obstet Gynecol 2005;105:569–574. PMID: 15738026.
Mariani A, Webb MJ, Keeney GL, Podratz KC. Routes of lymphatic spread: a study of 112 consecutive patients with endometrial cancer. Gynecol Oncol 2001;81:100–104. PMID: 11277658.
Montz FJ. Significance of “normal” endometrial cells in cervical cytology from asymptomatic postmenopausal women receiving hormone replacement therapy. Gynecol Oncol 2001;81:33–39. PMID: 11277646.
Nout RA, Smit VT, Putter H, et al. Vaginal brachytherapy versus pelvic external beam radiotherapy for patients with endometrial cancer of high-intermediate risk (PORTEC-2): an open-label non-inferiority, randomized trial. Lancet 2010;375:816–823. PMID: 20206777.
Pothuri B, Ramondetta L, Martino M, et al. Development of endometrial cancer after radiation treatment for cervical carcinoma. Obstet Gynecol 2003;101:941–945. PMID: 12738155.
Ramirez P, Frumovitz M, Bodurka DC, et al. Hormonal therapy for the management of grade 1 endometrial adenocarcinoma: a literature review. Gynecol Oncol 2004;95:133–138. PMID: 15385122.
Sakuragi N, Hareyama H, Todo Y, et al. Prognostic significance of serous and clear cell adenocarcinoma in surgically staged endometrial carcinoma. Acta Obstet Gynecol Scand 2000;79:311–316. PMID: 10746868.
Schmeler K, Lynch H, Chen L, et al. Prophylactic surgery to reduce the risk of gynecologic cancers in the Lynch syndrome. N Engl J Med 2006;354:261–269. PMID: 16421367.
Takeshima N, Nishida H, Tabata T, et al. Positive peritoneal cytology in endometrial cancer: enhancement of other prognostic indicators. Gynecol Oncol 2001;82:470–473. PMID: 11520142.
Thigpen JT, Brady MF, Homesley HD, et al. Phase III trial of doxorubicin with or without cisplatin in advanced endometrial carcinoma: a Gynecologic Oncology Group study. J Clin Oncol 2004;22:3902–3908. PMID: 15459211.
Trimble CL, Kauderer J, Zaino R, et al. Concurrent endometrial carcinoma in women with a biopsy diagnosis of atypical endometrial hyperplasia: a Gynecologic Oncology Group study. Cancer2006;106:812–819. PMID: 16400639.
Walker JL, Piedmonte MR, Spirtos NM, et al. Laparoscopy compared with laparotomy for comprehensive surgical staging of uterine cancer: a Gynecologic Oncology Group Study Lap 2. J Clin Oncol2009;27:5331–5336. PMID: 19805679.
SARCOMA OF THE UTERUS (LEIOMYOSARCOMA, ENDOMETRIAL SARCOMAS)
ESSENTIALS OF DIAGNOSIS
![]() Bleeding: metrorrhagia, menorrhagia, postmenopausal or preadolescent bleeding
Bleeding: metrorrhagia, menorrhagia, postmenopausal or preadolescent bleeding
![]() Mass: rapid enlargement of the uterus or a leiomyoma
Mass: rapid enlargement of the uterus or a leiomyoma
![]() Pain: pelvic discomfort as a result of mass effect from the enlarged uterus
Pain: pelvic discomfort as a result of mass effect from the enlarged uterus
![]() Malignant tissue: histology confirmed by D&C or in hysterectomy specimen
Malignant tissue: histology confirmed by D&C or in hysterectomy specimen
Pathogenesis
The uterine sarcomas are mesodermally derived highly malignant tumors and account for approximately 3–4% of all uterine malignancies. No common etiology has been identified in uterine sarcomas, but prior pelvic radiation therapy is associated with the mixed forms of uterine sarcoma.
Sarcomas can occur at any age but are most prevalent after age 40. A bimodal age distribution has been seen with uterine sarcomas, with a premenopausal or perimenopausal peak for patients with leiomyosarcoma and a postmenopausal peak for patients with carcinosarcoma. Sarcomas are well known as a source of hematogenous metastases, but with the exception of leiomyosarcomas, lymphatic permeation and contiguous spread are probably the most common methods of extension. Endometrial sarcomas can usually be diagnosed by EMB or D&C, but the sarcomas derived from the myometrium (leiomyosarcoma) frequently require hysterectomy to obtain adequate tissue for analysis.
In general, uterine sarcomas follow a very aggressive growth pattern with early metastasis to the abdomen, liver, and lung. There is no universal agreement on the histologic features that determine outcome, but most authorities agree that the number of mitotic figures per high-power field, vascular and lymphatic invasion, serosal extension, and degree of anaplasia are all helpful. Surgery is the most common primary treatment approach, followed by radiation and chemotherapy. Chemotherapeutic agents reported to be active against sarcomas include doxorubicin, cisplatin, ifosfamide, gemcitabine, and the taxanes. Clinical response rates for combination chemotherapy in recurrent and advanced disease are reported to be as high as 54%. However, most responses are partial and only temporary.
Histogenesis, Classification, & Staging
Although several classification systems exist for uterine sarcomas, they can be separated into 4 major categories: leiomyosarcomas (LMSs), endometrial stromal sarcomas (ESSs), malignant mixed mesodermal tumors (MMMTs), and adenosarcomas. LMSs are thought to arise from the myometrial smooth muscle cell or a similar cell lining blood vessels within the myometrium. ESS and MMMT arise from undifferentiated endometrial stromal cells, which retain the potential to differentiate into malignant cell lines that histologically appear native (homologous) or foreign (heterologous) to the human uterus. Because the undifferentiated stromal cells of the endometrium arise from specialized mesenchymal cells of the müllerian apparatus in the genital ridge, and ultimately from the mesoderm during embryogenesis, endometrial sarcomas have been variously termed “mesodermal,” “müllerian,” or “mesenchymal” sarcomas. The prognoses of patients with homologous and heterologous tumors are similar stage for stage, and this terminology has limited clinical usefulness. ESSs have been categorized in the older literature as “pure” and homologous endometrial sarcomas because they are composed of a single cell line. MMMTs, previously designated as “mixed” because they contain 2 or more cell lines, arise from an undifferentiated malignant stem cell. MMMTs contain both a carcinomatous or epithelium-derived element and a sarcomatous or mesenchymal element and have also been called “carcinosarcomas.” The carcinomatous element is usually an undifferentiated adenocarcinoma. The concept of this terminology is better understood by study of Figure 49–5, which graphically represents the histogenesis of uterine sarcomas. Table 49–2 combines the prevailing histogenetic terminology for endometrial sarcomas and depicts the various possibilities in each category.
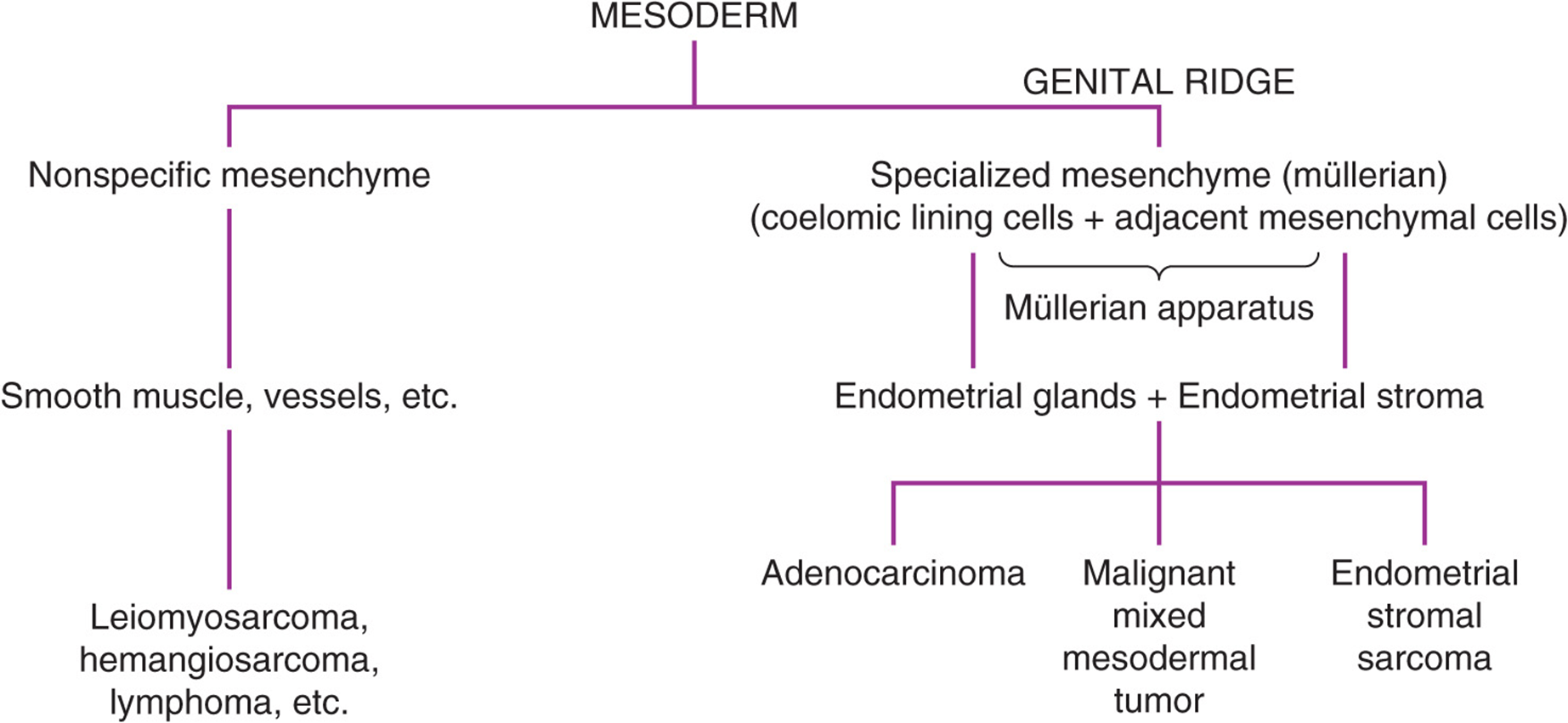
Figure 49–5. Histogenesis of uterine sarcomas.
Table 49–2. Classification of uterine sarcomas.
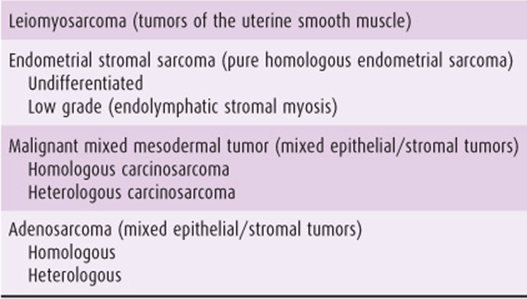
Pure heterologous sarcomas, such as rhabdomyosarcoma, chondrosarcoma, osteosarcoma, and liposarcoma, are extremely rare. Other uterine sarcomas, like hemangiosarcomas, fibrosarcomas, reticulum cell sarcomas, and lymphosarcomas, are indistinguishable from identical sarcomas elsewhere in the body and are therefore not considered specialized tumors of the uterus.
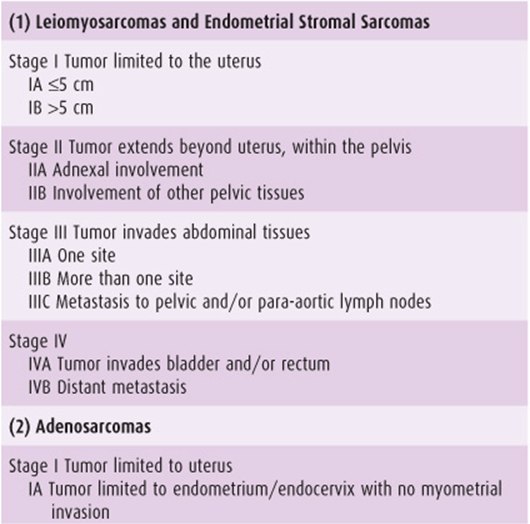
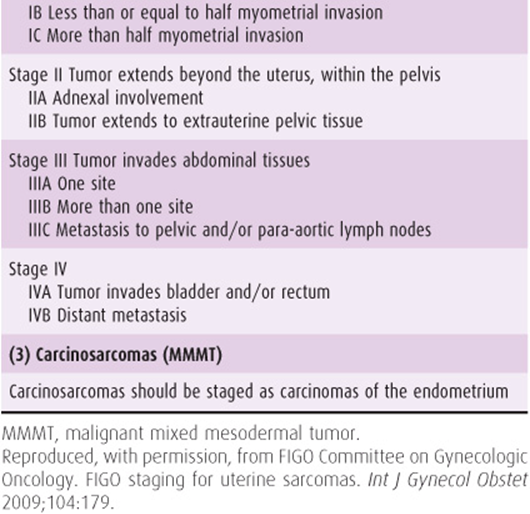
Recently, a new FIGO classification and staging system has been specifically designed for uterine sarcomas in an attempt to reflect their different biologic behavior (Table 49–3). Carcinosarcomas should continue to be staged as carcinomas of the endometrium.
Table 49–3. International Federation of Gynecology and Obstetrics (FIGO) staging of uterine sarcomas (2009).
Major Types of Sarcomas of the Uterus
A. Leiomyosarcomas
LMSs make up 35–40% of all uterine sarcomas and 1–2% of all uterine cancers. LMS usually occurs between ages 25 and 75 years, with a mean incidence at about age 50 years. Younger patients with this disease seem to have a more favorable outcome than postmenopausal women. Like the benign leiomyomas, LMSs are 1.5 times more common in African American patients than in the white population. Leiomyomas are commonly identified in uteri containing LMSs, but the incidence of malignant transformation of a leiomyoma is only 0.1–0.5%. Only approximately 5–10% of LMSs are reported to originate in a leiomyoma.
Abnormal uterine bleeding is the most common symptom of LMS, occurring in approximately 60% of patients; pelvic or abdominal pain and discomfort are reported by approximately 50% of all patients. Only approximately 10% of patients are aware of an abdominal mass. The deeply situated intramural position of most tumors impedes diagnosis by D&C, which is accurate in only 25% of cases. Abnormal cells might be identified on Pap smear. The diagnosis is more commonly made after pathologic analysis of a hysterectomy specimen.
LMSs spread by contiguous growth, invading the myometrium, cervix, and surrounding supporting tissues. Lymphatic dissemination is common in the late stages. Pelvic recurrence and peritoneal dissemination following resection are also common. In the more malignant types, hematogenous metastasis to the lungs, liver, kidney, brain, and bones probably occurs early but is clinically evident only in the lungs until the advanced stages.
The clinical behavior of the tumor is generally aggressive, with some correlation with the number of mitotic figures identified on microscopic examination. Low-grade LMSs are those with <5 mitoses per 10 high-power fields, with pushing rather than infiltrating margins. LMSs with 5–10 mitoses per 10 high-power fields are considered to be of intermediate grade, and tumors with mitotic counts >10 per 10 high-power fields are highly malignant and usually lethal; <20% of these patients are alive at 5 years. Tumor size and mitotic index are useful prognostic indicators, but other factors have to be taken into account. An invasive pattern, particularly into the blood and lymphatic vessels and the surrounding smooth muscle, is important. By contrast, cellular characteristics, such as atypia, anaplasia, and giant cells, are not accurate prognosticators of aggressive behavior. Clinically, the most reliable prognostic feature of LMS is stage. The prognosis of patients with extrauterine disease is much worse compared to patients with disease confined to the uterus. Patients with LMS mostly present at stage I. A pelvic MRI can help delineate the extent of uterine involvement and can help in the preoperative determination of a benign versus a malignant smooth muscle tumor by evidence of scattered foci of hemorrhage and necrosis and absence of calcifications. Benign leiomyomata have sharp boundaries, whereas sarcomas have infiltrating borders on MRI imaging.
Other unusual smooth muscle tumors of the uterus such as benign metastasizing leiomyoma and intravenous leiomyomatosis should be considered low-grade LMS or smooth muscle tumors of uncertain malignant potential (STUMP). Although they are histologically benign, they are notorious for local recurrence and can cause death by compression of contiguous or distant vital structures. Intravenous leiomyomatosis has been known to grow up the vena cava into the right atrium, impeding venous return and precipitating congestive heart failure. Because of their slow growth, they can frequently be controlled by repeated local excision. The metastatic lung lesions of benign metastasizing leiomyoma have disappeared following resection of the primary lesion in some cases, perhaps indicating hormone dependency.
B. Endometrial Sarcomas
1. Endometrial stromal sarcomas—ESSs make up 8% of all sarcomas. They occur predominantly in postmenopausal women. Patients with these tumors most commonly present with bleeding or lower abdominal discomfort and pain. The diagnosis can be made accurately by D&C in approximately 75% of cases. Although no etiologic relationship to hormones has been established, a small number of metastatic lesions have responded to progesterone therapy.
ESSs can be divided into 3 distinct subtypes: low-grade ESS, endometrial stromal nodules, and undifferentiated endometrial sarcomas previously known as high-grade ESS. The indolent low-grade ESSs—also called endolymphatic stromal myosis—have fewer than 10 mitoses per 10 high-power fields, with infiltrating margins and myometrial invasion. A benign form, the stromal nodule, contains pushing rather than infiltrating margins and fewer than 3 mitoses per 10 high-power fields, with no vascular or myometrial invasion.
The mean age at onset for low-grade ESS is 5–10 years earlier than for undifferentiated endometrial sarcomas. This tumor infiltrates surrounding structures and is characterized by indolent growth and a propensity to vascular invasion. Patients frequently present with yellowish wormlike extensions into the periuterine vascular spaces. Under such circumstances, it may be confused grossly with intravenous leiomyomatosis, as previously described. It tends to recur late, sometimes after 5–10 years, and can often be controlled by repeated local excisions. Low-grade ESSs frequently express estrogen and progesterone receptors. Adjuvant targeted hormonal treatment can be considered to reduce recurrence.
The undifferentiated endometrial sarcoma displays infiltrating margins and vascular and myometrial invasion and contains more than 10 mitoses per 10 high-power fields. These tumors are highly malignant and are associated with a poor prognosis, particularly when they extend beyond the uterus at the time of diagnosis. They spread by contiguous growth via the serosal uterine surface and lymphatic metastasis. Distant hematogenous metastases to the lungs and liver are usually a late event.
2. Carcinosarcoma (malignant mixed mesodermal tumors)—MMMTs account for 50% of all uterine sarcomas and 3–6% of all uterine tumors. They characteristically occur in postmenopausal women, with the exception of embryonal rhabdomyosarcoma of the cervix or vagina (sarcoma botryoides), which occurs also in infants and children. The incidence is about 3 times greater in black than in white women. Radiation therapy may be a predisposing cause, but the etiology of MMMTs is unknown. Many published series are available containing a significant number of patients with a history of pelvic radiation for benign or malignant conditions (Fig. 49–6).
Figure 49–6. Mixed sarcoma of the uterine fundus. Prior full-pelvic radiation therapy had little effect on the tumor.
As with the other types, the presenting symptom of MMMT is usually bleeding. Abdominal discomfort and pain or a large, bulky polypoid mass filling the uterine cavity and prolapsing through the cervical os also occurs. Because the tumors are endometrial in origin, approximately 75% can be diagnosed accurately by D&C. Histologically, MMMTs are usually highly anaplastic, with many bizarre nuclei and mitotic figures. They contain a carcinomatous or epithelial component and a sarcomatous component, hence the term malignant mixed mesodermal tumor, or carcinosarcoma. The carcinomatous component is usually serous (two-thirds) or endometrioid (one-third), and rarely clear cell, mucinous, or squamous cell carcinoma. If the sarcomatous component is derived from the smooth muscle tissue of the uterus, they are called homologous MMMTs. If the sarcomatous component contains bone, striated muscle, cartilage, or fat, the term heterologous MMMT is applied. The most common heterologous elements are malignant skeletal muscle or cartilage resembling either pleomorphic rhabdomyosarcoma or embryonal rhabdomyosarcoma. Presence of the heterologous elements is a poor prognostic factor in stage I patients.
MMMTs spread by contiguous infiltration of the surrounding tissues and by early hematogenous and lymphatic dissemination. The metastatic deposits are usually composed of the epithelial malignant glands, but sarcomatous elements have been identified in some cases. The prognosis depends mainly on the extent of the tumor at the time of primary surgery; there are virtually no long-term survivors among those whose tumor has extended beyond the confines of the uterus at the time of diagnosis. Treatment includes total abdominal hysterectomy with bilateral salpingo-oophorectomy, lymphadenectomy, and tumor debulking if necessary and technically feasible. Active chemotherapeutic agents include cisplatin, ifosfamide, doxorubicin, epirubicin, carboplatin, paclitaxel, and gemcitabine.
C. Adenosarcomas
Adenosarcoma is a distinctive mixed müllerian tumor of low malignant potential that accounts for 1–2% of uterine sarcomas. It arises from the endometrium and is composed of a combination of benign-appearing glands and a stromal sarcoma or fibrosarcoma. Adenosarcomas usually occur in the postmenopausal age group but have been reported in adolescents and women of reproductive age. Bleeding is the most common symptom and has been associated with prior tamoxifen or radiation therapy. Recurrence occurs in 25% of patients and is usually late. The primary treatment is total abdominal hysterectomy and bilateral salpingo-oophorectomy. Postoperative radiation therapy is recommended for those tumors with deep myometrial invasion.
D. Other Uterine Sarcomas
Embryonal rhabdomyosarcoma of the cervix (sarcoma botryoides), which occurs in infants and children, was previously lethal. However, combination therapy using surgery, radiation, and chemotherapy has considerably improved the outlook for these patients.
Fibrosarcoma, hemangiosarcoma, reticulum cell sarcoma, hemangiopericytoma, and other esoteric and bizarre uterine sarcomas are rare. In general, these sarcomas behave like the other intermediate-grade uterine sarcomas, but treatment must be individualized according to age, histologic type, and the patient’s state of health.
Prevention
Indiscriminate use of radiation therapy for benign conditions in the pelvis should be avoided, as several clinical studies have suggested an etiologic role of pelvic radiation in the development of MMMT.
Clinical Findings
A. Symptoms & Signs
Abnormal uterine bleeding is the most common manifestation of uterine sarcoma. Other recurring complaints include pelvic discomfort or pain, constipation, urinary frequency and urgency, and the presence of a mass low in the abdomen. Uterine sarcoma should be suspected in any nonpregnant woman with a rapidly enlarging uterus. Severe uterine cramps may exist if the tumor has prolapsed into the endometrial cavity or through the cervix. Pelvic examination may reveal the characteristic grapelike structures of sarcoma botryoides protruding from the cervix or the presence of velvety fronds of ESS in the cervical canal. A necrotic fungating mass at the vaginal apex should suggest an infarcted myoma, LMS, or MMMT. The uterus is usually enlarged and often soft and globular. If the cancer has involved the cervix, cul-de-sac, or cardinal ligaments, fixation or asymmetry of the parametria may be found. In advanced cases, inguinal or supraclavicular node metastases may be evident. Patients with advanced uterine sarcomas may present with a large omental mass or ascites secondary to abdominal carcinomatosis.
B. Laboratory Findings
Standard laboratory evaluation of patients with uterine sarcoma should include a complete blood count and urinalysis, liver function studies (especially serum alkaline phosphatase, prothrombin time, and serum lactic dehydrogenase), blood urea nitrogen, and serum creatinine. CA-125 may be elevated. Estrogen and progesterone receptor analysis may indicate which patients are likely to respond to hormone therapy. Office EMB or punch biopsy of a prolapsed vaginal mass is helpful only if positive.
C. Radiograph Findings
The chest radiography may contain metastatic coin lesions characteristic of uterine sarcomas. Because uterine sarcomas commonly metastasize to the lung, a chest computed tomography (CT) scan should be considered when the routine films are negative, particularly before any radical extirpative surgery in the pelvis is performed. CT scan of the abdomen and pelvis is helpful in assessing the extent of abdominal disease, evaluating the kidneys for hydronephrosis, identifying enlarged retroperitoneal nodes, and identifying liver metastases. MRI scans are not routinely performed but may provide an accurate preoperative assessment of uterine size and degree of involvement.
D. Special Examinations
Pelvic ultrasonography may confirm the presence of a pelvic mass or help to differentiate an adnexal from a uterine mass in the obese patient. Sigmoidoscopy should always be performed in older women, and in young women if gastrointestinal bleeding or masses suspected of being malignant are present. Cystoscopy is indicated in locally advanced disease or in the presence of gross or microscopic hematuria.
Differential Diagnosis
The clinical diagnosis of uterine sarcoma is frequently overlooked. Diagnostic accuracy can be increased if the physician keeps these tumors in mind while investigating any pelvic mass. The tumor frequently does not present the classic picture of abnormal bleeding accompanied by a symmetrically enlarged soft globular uterus. It can masquerade as any condition causing uterine enlargement or a pelvic mass; of these, pregnancy, leiomyoma, adenomyosis, and adherent ovarian neoplasms or pelvic inflammatory disease are most likely to cause misinterpretation. When cytologic studies, EMB, or D&C fail to provide the diagnosis—a situation not uncommon with LMS—laparotomy is necessary. At laparotomy, thorough evaluation is critical to the future management of the patient with uterine sarcoma and must include inspection (where possible) and palpation of all abdominal viscera, peritoneal and mesenteric surfaces, liver, both diaphragms, and retroperitoneal structures, especially the pelvic and aortic lymph nodes. Cytologic examination of peritoneal exudate is indispensable for treatment planning; if no free fluid is present, samples may be obtained by instilling 50–100 mL of normal saline into the abdominal cavity (pelvic washings). If a sarcoma is identified on frozen section of the hysterectomy specimen, suspicious lymph nodes should be removed. This information, gathered at the time of the initial exploration and carefully documented in the operative records, is critical for identification and staging of the neoplasm and for predicting outcome.
The pathologic diagnosis of uterine sarcoma is often extremely difficult and may require consultation with a gynecologic pathologist familiar with these tumors. As each cancer becomes more anaplastic, the parent cell or tissue becomes more difficult to identify histologically. Because proper treatment is predicated on accurate histologic diagnosis, every effort should be expended to identify the cell of origin.
Complications
Severe anemia from chronic blood loss or acute hemorrhage may be present. The severity and extent of other complications caused by uterine sarcomas are directly related to the size and virulence of the primary tumor. A pedunculated mass may protrude into the uterine cavity or prolapse through the cervix, causing bleeding or uterine cramps as the uterus attempts to expel the tumor. Infarction with subsequent infection and sepsis may ensue. Rupture of the uterus as a consequence of rapidly growing uterine sarcomas has been reported. Obstructed labor and postpartum uterine inversion secondary to endometrial sarcomas have also been noted. Extensive pulmonary metastases can produce hemoptysis and respiratory failure. Ascites is common in advanced disease with peritoneal metastases.
A wide variety of complications has been reported secondary to pressure or compression of a neighboring viscus or resulting from extension or metastases to other vital structures. Urethral elongation caused by stretching of the bladder over a rapidly growing mass can simultaneously produce obstruction and loss of sphincter control, with subsequent overflow incontinence. Colon compression may result in ribbon stools and, eventually, complete bowel obstruction. Ureteral obstruction is common, especially with recurrent pelvic sarcomas. Urinary diversion or colostomy may be required prior to treatment if life-threatening viscus obstruction is present in an untreated patient, but urinary diversion should not be performed unless there is some hope for cure or meaningful palliation, because it precludes a painless death from uremia.
Treatment
A. Emergency Measures
Hemorrhage from uterine sarcomas can be severe and requires prompt attention. In acute hemorrhage, blood volume should be replaced rapidly, using packed red blood cells, crystalloid solutions, volume expanders, and fresh-frozen plasma.
Emergency D&C should be used only to obtain tissue for analysis. Vigorous curettage is likely to aggravate or provoke bleeding. High-dose bolus radiation is a more reliable and safe method of controlling bleeding. A dose of 400–500 cGy administered daily to the whole pelvis over 2–3 days usually controls acute hemorrhage; this does not appreciably interfere with future management. If these measures are not successful, emergency embolization or ligation of the hypogastric arteries sometimes controls hemorrhage when hysterectomy is not indicated or technically feasible.
B. Surgical Measures
Extirpative surgery provides the best chance for long-term palliation or cure for patients with uterine sarcomas. Surgery is the cornerstone of the treatment plan and should be the central focus of attack against these cancers.
Because low-grade uterine sarcomas (some LMSs, endolymphatic stromal myosis, intravenous leiomyomatosis) have a propensity for isolated local spread and central pelvic recurrence, such patients should be considered for radical hysterectomy and bilateral salpingo-oophorectomy. The benefits of this type of therapy have not been conclusively shown, but, theoretically, the problem of local recurrence should be improved by more radical excision of the primary tumor. Lymph node metastases in these low-grade tumors are negligible; consequently, pelvic lymphadenectomy can be reserved for patients with enlarged or suspicious nodes. Pelvic recurrences of low-grade uterine sarcomas have been successfully treated by repeated excisions of all resectable tumor. Patients have been known to survive for many years following this type of conservative treatment. Partial or complete pelvic exenteration may occasionally be useful for recurrence of indolent tumors.
The high-grade uterine sarcomas (some LMSs, ESSs, all MMMTs) display early lymphatic, local, and hematogenous metastases, even when apparently confined to the uterus. For this reason, radical surgery has been abandoned in favor of simple total abdominal hysterectomy and bilateral salpingo-oophorectomy preceded or followed by adjunctive radiation therapy. At the time of surgical exploration, a thorough examination and evaluation of the abdominal contents must be performed and documented. Cytologic specimens and omental tissue should be obtained, and suspicious papillations, excrescences, and adhesions should be excised for pathologic analysis. A thorough staging procedure is important for prognosis and a postoperative treatment plan.
When uterine sarcomas recur in the lung and the metastatic survey is negative, unilateral isolated metastases can be excised after a chest CT scan has ruled out other lesions not apparent on the routine chest radiograph. Considering all sources, resection of isolated sarcoma metastases to the lung carries about a 25% 5-year cure rate.
C. Chemotherapy
Adjuvant single-agent doxorubicin has been shown to have a 25% response rate for LMS. Recent data suggest that use of combination gemcitabine and docetaxel in LMS improves progression-free survival and reduces risk of recurrence.
Because of the high hormone receptor content in ESS, adjuvant progestin or aromatase inhibitors are recommended. For receptor-negative tumors, doxorubicin- or ifosfamide-based chemotherapy is used. Tamoxifen and estrogen replacement therapy should be avoided in patients with ESS.
Doxorubicin, cisplatin, carboplatin, paclitaxel, gemcitabine, and ifosfamide display significant activity against MMMTs. Cyclophosphamide and vincristine also show activity. Some data suggest that combination chemotherapy is more effective than single-agent therapy. In advanced or metastatic disease, adjuvant combination chemotherapy is recommended. Trabectedin, a new agent, has been actively evaluated in sarcomas and appears to show some modest promise. Treatment with tyrosine kinase inhibitors like imatinib or sorafenib has shown response in some patients.
D. Radiation Therapy
When used as the only modality of treatment for uterine sarcomas, radiation has produced dismal results—very few survivors are reported in the literature following treatment with radiation therapy alone for any of the uterine sarcomas. Radiation therapy does provide local tumor control and reduces local recurrences when used in combination with surgery for the treatment of some endometrial sarcomas. However, it is unclear whether a combined surgical and radiation approach changes overall survival. Collected data indicate that adjuvant radiation therapy improves the 2-year survival rate in patients with ESS by approximately 20% and may also improve survival for those with MMMTs, although less convincingly. Although an occasional 5-year survivor with LMS has been reported following radiation therapy alone, analysis of large numbers of patients from different institutions does not support its use for these tumors. Nevertheless, in advanced forms of LMS, radiation may prove useful for palliation and control of pelvic symptoms such as massive bleeding or pain.
Prognosis
In determining the prognosis for patients with uterine sarcomas, a constellation of factors must be examined simultaneously. Such considerations as the patient’s age, state of health, and ability to withstand major surgery or radiation therapy (or both) must be evaluated. The most important clinical characteristic—and probably the overriding prognostic feature affecting the prognosis of these patients—is the stage of the disease at the time of diagnosis. In the high-grade sarcomas (LMS and mixed endometrial sarcoma), the presence of tumor outside the uterus at the time of diagnosis is a clear prognostic omen: Fewer than 10% of patients survive 2 years. Even when the disease is apparently limited to the uterus, the prognosis is poor: 10–50% survive 5 years. In intermediate-grade LMS and undifferentiated endometrial sarcoma, the outcome is improved, with up to 80–90% of patients surviving 5 years if the disease is clinically limited to the uterus at the time of surgery. Low-grade ESS and low-grade LMS have a generally favorable outcome: 80–100% of patients survive 5 years following complete excision of the uterus. Low-grade stromal tumors have been known to recur locally after 10–20 years; this confuses the survival statistics. Undoubtedly, these patients must be followed closely for life.
Brooks SE, Zhan M, Cote T, Baquet CR. Surveillance, epidemiology, and end results analysis of 2677 cases of uterine sarcoma 1989–1999. Gynecol Oncol 2004;93:204–208. PMID: 15047237.
D’Angelo E, Prat J. Uterine sarcomas: a review. Gynecol Oncol 2010;116:131–139. PMID: 19853898.
Demetri GD. ET-743: the US experience in sarcomas of the soft tissues. Anticancer Drugs 2002;13:S7. PMID: 12173492.
Dinh TA, Oliva EA, Fuller AF Jr, et al. The treatment of uterine leiomyosarcoma. Results from a 10-year experience (1990–1999) at Massachusetts General Hospital. Gynecol Oncol 2004;92: 648–652. PMID: 14766261.
Ferguson SE, Tornos C, Hummer A, et al. Prognostic features of surgical stage I uterine carcinosarcoma. Am J Surg Pathol 2007;31:1653–1661. PMID: 18059221.
Giuntoli RL, Metzinger DS, DiMarco CS, et al. Retrospective review of 208 patients with leiomyosarcoma of the uterus: prognostic indicators, surgical management, and adjuvant therapy. Gynecol Oncol2003;89:460–467. PMID: 12798172.
Hensley ML, Ishill N, Soslow R, et al. Adjuvant gemcitabine plus docetaxel for completely resected stages I-IV high grade leiomyosarcoma: results of a prospective study. Gynecol Oncol 2009:112;563–567. PMID: 19135708.
Kushner D, Webster KD, Belinson JL, et al. Safety and efficacy of adjuvant single-agent ifosfamide in uterine sarcoma. Gynecol Oncol 2000;78:221–227. PMID: 10926807.
Look KY, Sandler A, Blessing JA, et al. Phase II trial of gemcitabine as second line chemotherapy of uterine leiomyosarcoma: an Gynecologic Oncology Group (GOG) study. Gynecol Oncol 2004;92:644–647. PMID: 14766260.
Manolitsas TP, Wain GV, Williams KE, et al. Multimodality therapy for patients with clinical stage I and II malignant mixed müllerian tumors of the uterus. Cancer 2001;15:1437–1443. PMID: 11301390.
O’Meara AT. Uterine sarcomas: have we made any progress? Curr Opin Obstet Gynecol 2004;16:1–4. PMID: 15128000.