Gian Carlo Di Renzo1 , Irene Giardina1, Giulia Babucci1, Chiara Antonelli1, Sandro Gerli1 and Graziano Clerici1
(1)
Santa Maria della Misericordia University Hospital, San Sisto, Perugia, 06132, Italy
Gian Carlo Di Renzo
Email: giancarlo.direnzo@unipg.it
1 Introduction
Pre-term births (PTB) (below 37 weeks) is one of the major health challenges to-day. The incidence is high, accounting for 5–9 % of births in Europe [1]. In addition, the incidence is increasing. The incidence was 9.5 % in the U.S. in 1981, rising to 12.7 % in 2005 [2]. Perinatal mortality increases more than three times in women with PTB (51.7/1,000 births), and is a leading cause of long-term morbidity, including neurodevelopmental handicap, cerebral palsy, seizures, blindness, deafness and non-neurological disorders, such as bronchopulmonary dysplasia & retinopathy of prematurity. Additionally, initial hospital care of infants born at 25–27 weeks costs 28 times as much as for those born at term in the U.S.
The diverse etiology of preterm delivery makes prediction difficult. Previous PTB, cervical effacement or dilatation on clinical vaginal examination or shortening of the length of the cervical canal on ultrasound are predisposing factors. Similarly, the presence of contractions may indicate the start of preterm labour. In pre-term labor (PTL) various tocolytics have been used to stop contractions including: (1) β adrenergic stimulators, known to relax smooth muscle such as ritodrine, terbutalin etc. (2) Prostaglandin synthetase inhibitors such as indomethacin. (3) Calcium channel blockers such as nifedipine, and (4) oxytocin inhibitors such as atosiban. The efficacy of all of these is controversial.
A growing body of evidence suggests that progesterone plays a role in preventing labour. Progesterone decreases prostaglandin synthesis, reduces cervical stromal degradation, alters the barrier to ascending inflammation and or infection in the cervix. It reduces gap junction formation, decreases the conduction and frequency of myometrial contractions, increases the stimulation threshold of the myometrium and decreases spontaneous myometrial activity. Progesterone alters estrogen synthesis in fetal membranes and the placenta, alters fetal endocrine-mediated effects. It decreases the number of oxytocin receptors and suppresses the inflammatory cascade.
This chapter concentrates on the mechanisms whereby progestogens may prevent PTL, the optimal progestogens, and their route of administration, and the place of progestogens in current obstetric practice.
2 Role of Progesterone in Pregnancy
Progesterone is an essential hormone in the process of reproduction. It has been proposed and used in the treatment of different gynaecological pathologies, such as endometrial hyperplasia, dysfunctional uterine bleeding, amenorrhoea, luteal phase deficiency and premenstrual syndrome, as well as being used as a contraceptive tool alone or in combination with estrogens, in assisted reproductive technologies and in the maintenance of pregnancy [3]. The role of progesterone in the maintenance of pregnancy comprises: modulation of maternal immune response [4–6], suppression of inflammatory response [7], reduction of uterine contractility [8–10], and improvement of the utero-placental circulation [11, 12]. Some of the cytokine effects associated with pre-term births have been defined. Increased amniotic levels of the proinflammatory cytokines have such as IL-6, IL-8 and TNFα have been correlated with preterm births [13]. The physiological effects of progesterone are fully discussed in Chap. 1. The cytokine modulatory effects have been best studied in the synthetic progestogen dydrogesterone. Dydrogesterone inhibits IFNγ & TNFα, but increases IL-4 production [14]. Dydrogesterone induces the production of a progesterone induced blocking factor (PIBF). In Preterm labour, dydrogesterone is associated with significantly higher serum levels of IL-10, increased PIBF production & lower concentrations of IFNγ than controls [15]. Progesterone itself has been reported to up-regulate LIF mRNA expression in vitro [16].
All these effects have been used to explain the therapeutic application of progesterone during pregnancy, whether progestogens are targeted to the prevention and treatment of threatened miscarriage, recurrent miscarriage or preterm birth. Regarding the myometrial effect, progesterone has been shown to possess tocolytic action on the myometrium both in vitro and in vivo during pregnancy. It has been demonstrated, that adequate progesterone concentrations in the myometrium are able to counteract the stimulatory activity of both prostaglandins and oxytocin. Progesterone decreases the concentration of myometrial oxytocin receptors, (counteracting the effect of estrogens). Progesterone also decreases the number and activity of gap junctions [17]. Moreover, progesterone and its metabolites induce uterine quiescence through interactions between nuclear and membrane progesterone receptors [9, 18, 19].
The in vivo effect of progesterone has also been shown to be dose dependent. Only high-doses of progesterone are tocolytic in early pregnancy. High dosages are also effective in the maintenance of uterine quiescence during cervical cerclage (during the first trimester of pregnancy) and/or following abdominal surgery (e.g. appendectomy) [17]. However, the progestogen 17 alpha-hydroxyprogesterone caproate (17 OHP-C) does not directly inhibit human myometrial contractions in vitro. Surprisingly, in fact, 17 OHP-C has been shown to stimulate contractility in a dose dependent manner [20] (Fig. 6.1).
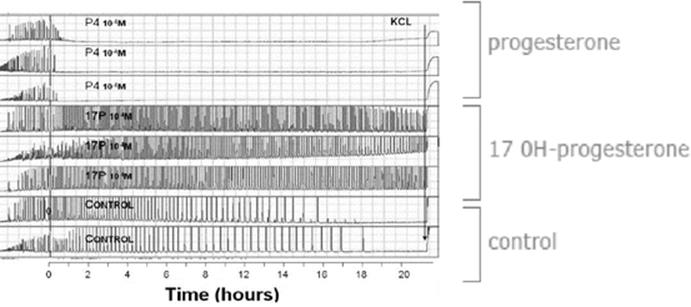
Fig. 6.1
Tocolytic effects of progesterone: P4 versus 17OHP: changes in contractility in progesterone and 17OHP treated myometrial strips. Modified from Chanrachakul B et al. Am J Obstet Gynecol 2005 and Ruddock NK et al., Am J Obstet Gynecol 2008 [10, 20]
Since the 1990s, progesterone has been studied for its possible role in preterm birth. Keirse in his meta-analysis of placebo-controlled trials, investigated the possible prophylactic use of 17 OHP-C in women at a high risk of pre-term birth. This analysis no effect in miscarriage, but did demonstrate a reduction in the incidence of preterm birth and the incidence of low birthwheight babies [21].
3 Key Differences in the Route of Administration
Although the pharmacokinetics and pharmacodynamics of progesterone have been well studied since 1935 when progesterone was first synthesized, its use in the pathophysiology of pregnancy remains controversial. One of these concerns is the optimal route of administration [22]. The rate of absorption is dependent on which pharmaceutical form is used, the blood flow at the site of administration and the solubility in the tissues into which the drug is administered. Progesterone can be administered by many different routes, but the main are oral, vaginal and intramuscular (Table 6.1).
Table 6.1
Routes of administration of progesterone
|
Different routes of administration |
|
|
Different pharmacokinetics and dynamics |
|
|
Intramuscular Supraphysiological plasma concentrations |
|
|
Oral • Rapid increase n plasma concentration followed by a gradual decrease • First liver pass effect with several biological active metabolites • Specific activity on different target organs (uterus, brain…) |
Metabolism – In the gut (bacteria with 5b-reductase activity) – In the intestinal wall (5a-reductase activity) – In the liver (5b-reductase, 3a- and 20a-hydrodylase activities) |
|
Vaginal • Stable plasma concentrations and consistent tissue levels • First uterine pass effect with targeted delivery into the endometrium • Minimal systemic effects |
Metabolism – Normal vaginal bacteria and mucosa seem devoid 5a- and 5b-reductases – After vaginal, only a small increase in 5a-pregnanolone observed and 5b-pregnanolone levels were not affected |
3.1 Oral Administration
Oral administration is associated with better patient compliance, but has several disadvantages, the main disadvantage being extreme variability in plasma concentrations due to individual variability in gastric filling and enteropathic circulation. Food may influence the rate and extent of drug absorption by reducing the rate of gastric emptying, decreasing gastrointestinal motility, increasing gastrointestinal secretions and increasing splanchnic blood flow. Oral administration has another major shortcoming: after absorption, progesterone is first passed to the liver by the portal vein. The metabolites produced by the liver interfere with progesterone action; as shown by the discrepancy between progesterone levels and endometrial histology [23].
A number of synthetic oral progestogens have been developed to overcome progesterone’s low oral bioavailability. Their pharmacologic effects, however, differ from progesterone itself, and may result in increased androgenic effects, fluid retention, alterations in high density lipoproteins, headaches, mood disturbances, and possible teratogenicity dependant on the individual progestogen. Dydrogesterone however, seems to have no androgenic or anti-androgenic effect (See Table 2 in Chap. 2). Oral progesterone may also have other side effects such as nausea, headache and sleepiness. In an attempt to improve the characteristics of oral administration, micronized progesterone has been introduced. It is manufactured from chemicals derived from plants (Mexican wild yams, Dioscorea barbasco), yet it has a molecular structure identical to human progesterone [24]. Micronization of progesterone into particle sizes of <10 μm increases the available surface area, enhances the aqueous dissolution rate and intestinal absorption of progesterone. Suspension in oil and packaging in a gelatin capsule has been shown to further enhance the intestinal absorption of micronized progesterone [25].
3.2 Vaginal Administration
The vaginal route results in higher concentrations in the uterus [17, 22]. Bulletti et al. [26] tried to verify the hypothesis of a “first uterine pass effect,” suggested by the evidence of higher than expected uterine tissue concentrations after vaginal administration of progesterone. Three different hypotheses have been reported to explain the first uterine pass: direct diffusion through or between cells of the vagina to cells of the uterus, portal-like arrangements of lymphatics linking the upper vagina to the uterus, and a counter circulation system, much like a portal system, with vein to artery diffusion between the upper vagina and uterus. Bulletti et al. [26] demonstrated that a “first uterine pass effect” occurs when drug is delivered vaginally, confirming that the vaginal route permits targeted drug delivery to the uterus, maximizing the desired effects while minimizing the potential for adverse systemic effects (Fig. 6.2).
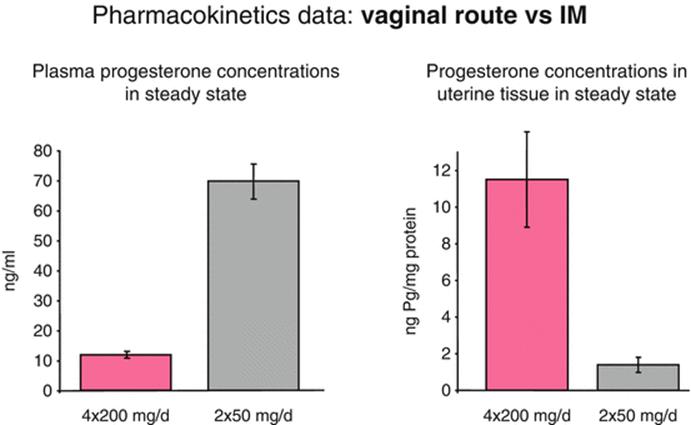
Fig. 6.2
Comparison between vaginal and intramuscular route of administration. Modified from Miles A et al. Fertil Steril 1994 [45]
After vaginal administration of progesterone, uterine tissue concentration have been found to exceed the levels achieved by systemic administration by more than tenfold; but the plasma levels are more than seven times higher after systemic administration compared to vaginal administration. The time to peak concentration is generally slightly less than after oral administration of micronized preparation. Moreover, after vaginal administration, plasma concentrations display a plateau-like profile, with a more constant concentration over time. Peak plasma levels of progesterone seem to be variable and not consistently greater or less than corresponding peak plasma values obtained after oral administration of micronized progesterone [25]. Cicinelli et al. demonstrated the direct transport of progesterone from vagina to uterus comparing progesterone concentrations in serum and endometrial tissue from hysterectomy specimens after vaginal or intramuscular administration of progesterone gel. They demonstrated that the ratios of endometrial to serum progesterone concentrations were markedly higher in women who received vaginal progesterone [27] (Fig. 6.3).
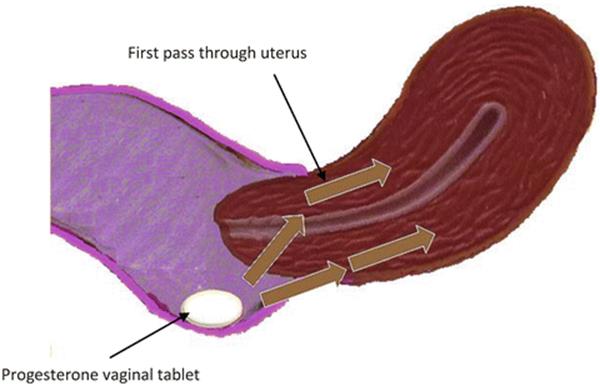
Fig. 6.3
Vaginal administration of progesterone: first uterine pass effect
3.3 Intramuscular Administration
Historically, the most common delivery method of progesterone has been intramuscular [25]. Progesterone administered intramuscularly can lead to local discomfort and extreme pain, and occasionally induces non-septic abscesses at the injection site. A large number of other side effects have been described including: hypersensitivity reactions, cough, dyspnea, tiredness, dizziness, genital itching, & increased risk of gestational diabetes, mood swings, headaches, bloating, abdominal pain, perineal pain, constipation, diarrhea, nausea, vomiting, joint pain, depression, decreased sex drive, nervousness, sleepiness, breast enlargement, breast pain, dysuria, polyuria, UTI, vaginal discharge, fever, flu-like symptoms, back pain, leg pain, sleep disorder, upper respiratory infection, asthma, acne and pruritus.
However, intramuscular administration is the route that results in optimal blood levels, can be administered in patient with vaginal bleeding and needs only one single daily dose [17, 22].
3.4 Other Routes of Administration
Other routes have been described. Progesterone can be administered by a transdermal patch as progesterone can penetrate the skin but it is rapidly metabolized by the 5-α-reductase enzyme, which converts it to 5-α-dihydro-progesterone, thereby lowering plasma progesterone levels. Rectal administration, is associated with variable absorption and there is insufficient scientific evidence concerning effects on the endometrium after rectal administration.
4 Progesterone and Preterm Birth
As stated above, PTB is the leading cause of perinatal mortality and morbidity. Its incidence has not declined (12 % of all birth) over the last 20 years, and, due to its long-term neuro-developmental sequelae, it is one of the major financial drains on health and educational resources [10, 28]. It is the major challenge in Obstetrics to-day to reduce the number of PTB’s or at least prolong pregnancy until fetal maturity is sufficient to ensure survival without excess morbidity.
The mechanism of human parturition is the expression of anatomic, biochemical, physiologic and clinical events that occur in the mother and in the fetus in both term and preterm labour. This pathway comprises: decidual/fetal membrane activation, increased uterine contractility and cervical ripening (dilatation and effacement). Preterm labor is the consequence of the pathologic activation of one or more of these elements. The apparent loss of progesterone sensitivity at term could be a consequence of several different mechanisms including: alterations in progesterone receptors (PR) isoform ratios, the anti-inflammatory function of progesterone, the catabolism of progesterone in the uterus into inactive compounds, changes in cofactor protein levels affecting PR transactivation and inflammation-induced trans-repression of PR [17].
Before undertaking any therapeutic strategy, careful identification is needed, so as to detect manageable conditions and fetal and/or maternal contraindication. The real challenge is to develop sensitive and specific tests that reliably detect these pregnancy changes before they became irreversible, and to find effective interventions capable of arresting the process of preterm labor and to enhance the effectiveness of current available interventions.
Regarding the management of threatened preterm labor, tocolysis and administration of corticosteroids to induce lung maturation are the first therapeutic tools; additionally bed rest and hydration are usually recommended in the management of these patients, although none have been shown to be clearly effective. Progesterone, other related synthetic compounds such as 17 OHP-C and other progestogens, have been tested in clinical trials for the prevention of preterm birth [29]. The three main groups of patients considered at particularly high risk include: patients with a previous preterm birth, patients with short cervix and patients with multiple pregnancy. Studies using synthetic progestogens to reduce the incidence of preterm delivery have been reported. However, there are mixed results. Progesterone itself however, has been demonstrated to be beneficial, especially in view of cost, availability and biological safety [30, 31].
The vast majority of clinical trials have been performed with various formulations of either microized progesterone or 17 OHP-C. The first has been administered through the vaginal route and the latter by weekly intramuscular injection. The use of both micronized progesterone and 17 OHP-C has been advised in asymptomatic women with a prior history of preterm birth as early prophylaxis for preterm, birth and in single pregnant, nulliparous women with asymptomatic cervical shortening (15 mm) detected with trans-vaginal ultrasound at midgestation. The tocolytic effect of oral micronized progesterone was first reported in a 1986 report by Erny et al. [32]. These investigators evaluated the effects of 400 mg of orally absorbed micronized progesterone or placebo in women at risk for premature labor; 88 % of the patients who were treated with oral micronized progesterone had decreased uterine activity compared with 42 % of the patients who received placebo. Furthermore Istwan et al. demonstrated that in patients with a history of one prior spontaneous preterm delivery prophylactic 17 OHP-C, confers a reduction in risk of preterm labour compared to untreated patients [33].
One of the main studies concerning the prophylactic use of vaginal progesterone to decrease the incidence of preterm birth in patients at a high risk, has been performed by Fonseca et al. [34]. Fonseca et al. demonstrated that the daily use of 100 mg vaginal progesterone is able to reduce the frequency of uterine contractions and the incidence of preterm birth. More recently it has also been demonstrated that the use of vaginal progesterone in asymptomatic women with a sonographic short cervix not only reduces the risk of preterm birth, but it is also able to reduce neonatal morbidity and mortality (Fig. 6.4, Table 6.2) [35]. In nulliparous women with a midtrimester cervical length <30 mm, the use of weekly intramuscular 17 OHP-C did not show a reduced incidence of preterm birth [36]. Comparing the use of vaginal progesterone with the intramuscular route of administration, a statistically significant decrease of the number of admission to the Neonatal Intensive Care Unit and a significant deficiency of severe side effects has been demonstrated [37].
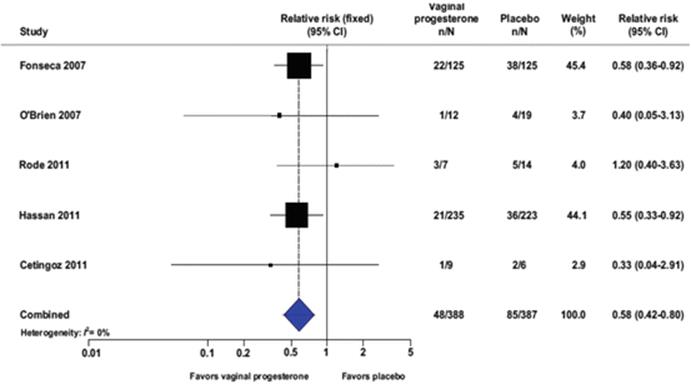
Fig. 6.4
Vaginal progesterone and reduction of preterm birth before 33 weeks of gestation: a metanalysis. From Romero R et al. Am J Obstet Gynecol 2012 [35]
Table 6.2
Vaginal progesterone in women with an aymptomatic short cervix in the midtrimester ultrasound decreases PTD and improves neonatal outcome
|
No. of events/total no. |
||||||
|
Outcome |
No. of trials |
Vaginal progesterone |
Placebo |
Pooled RR (95% CI) |
I 2(%) |
NNT (95% CI) |
|
Preterm birth <37 wk |
5 |
144/388 |
165/387 |
0.89 (0.75–1.06) |
0 |
– |
|
Preterm birth <36 wk |
5 |
108/388 |
136/387 |
0.82 (0.67–1.00) |
0 |
– |
|
Preterm birth <35 wk |
5 |
79/388 |
118/387 |
0.69 (0.55–0.88) |
0 |
11 (7–27) |
|
Preterm birth <34 wk |
5 |
62/388 |
105/387 |
0.61 (0.47–0.81) |
0 |
9 (7–19) |
|
Preterm birth <30 wk |
5 |
29/388 |
51/387 |
0.58 (0.38–0.89) |
0 |
18 (12–69) |
|
Preterm birth <28 wk |
5 |
21/388 |
43/387 |
0.50 (0.30–0.81) |
0 |
18 (13–47) |
|
Spontaneous preterm birth <33 wk |
5 |
39/388 |
71/387 |
0.57 (0.40–0.81) |
0 |
13 (9–29) |
|
Spontaneous preterm birth <34 wk |
5 |
51/388 |
87/387 |
0.62 (0.46–0.84) |
0 |
12 (8–28) |
|
Respiratory distress syndrome |
5 |
25/411 |
52/416 |
0.48 (0.30–0.76) |
0 |
15 (11–33) |
|
Neonatal death |
5 |
8/411 |
15/416 |
0.55 (0.26–1.19) |
43 |
– |
|
Admission to NICU |
5 |
85/411 |
121/416 |
0.75 (0.59–0.94) |
0 |
14 (8–57) |
|
Mechanical ventilation |
5 |
35/411 |
51/416 |
0.66 (0.44–0.98) |
0 |
24 (15–408) |
|
Congenital anomaly |
7 |
30/1967 |
34/1,954 |
0.89 (0.55–1.44) |
0 |
– |
|
Any maternal adverse event |
3 |
86/624 |
80/595 |
1.04 (0.79–1.38) |
0 |
– |
|
Intraventricular hemorrhage |
5 |
6/411 |
9/416 |
0.74 (0.27–2.05) |
0 |
– |
Modified from Romero et al. Am J Obstet Gynecol 2012 [35]
In both twin and triplet pregnancy, neither micronized progesterone nor 17 OHP-C have been shown to prevent preterm birth [29]. As long ago as 1980, Hartikainen-Sorri et al. reported a placebo controlled trial of 77 twin pregnancies treated during the last trimester until the 37th gestational week with weekly injections of either 17 OHP-C. There were no differences in the gestational length, birth weight or outcome of the neonates [38]. More recently, Combs et al. demonstrated that the prophylactic administration of 17 OH-C to mothers with twin pregnancy does not reduce the incidence of preterm delivery or neonatal morbidity [39].
4.1 Progesterone as a Possible Tocolytic Agent
The administration of high-dosage progesterone has been advocated as a possible tocolytic agent. Progesterone itself has been documented to inhibit uterine contractions, whereas 17 OHP-C seems to have no effect on uterine contractions (Fig. 6.1) [29]. The action of progesterone is slow and it can be used for acute tocolysis only in conjunction with tocolytic agents such as β-agonists [33, 40]. The combination of the two drugs has shown synergistic effects by decreasing the need for high concentrations of β-agonists, which have potentially dangerous side effects (Table 6.3) [17, 22].
Table 6.3
Use of progesterone in association with tocolytic agents
|
Patients |
47 |
42 |
|
Gestational age |
30.5 ws |
30.3 ws |
|
Treatment |
– Ritodrine (100 mg in saline 0.1–0.3 mg/min) |
– Ritodrine (50 mg in saline 0.1–0.3 mg/min) – Progesterone (200 mg die) |
|
Outcome |
– Deliveries after 48 h: 87 % – Deliveries After 7 days: 65 % |
– Deliveries after 48 h: 85 % – Deliveries After 7 days: 68 % |
|
Side effects |
– Mat. tachicardia: 97 % – Nausea & vomiting: 28 % – Tremblings: 26 % – Palpitations: 32 % – Chest pain: 15 % – Hyperglycemia: 47 % – Hypokaliemia: 92 % |
– Mat tachicardia: 42 % – Nausea & vomiting: 6 % – Tremblings: 12 % – Palpitations: 12 % – Chest pain: 8 % – Hyperglycemia: 28 % – Hypokaliemia: 23 % |
Modified from Di Renzo et al. Curr Opin Obstet Gynecol 2005 [22]
A recent study of Baumbach et al. demonstrates that progesterone significantly inhibits uterine contractility at relatively high concentrations and its combination with nifedipine and indomethacin increases this effect [41] (Fig. 6.5). Also Chanrachakul et al. recently demonstrated this synergetic effect of progesterone with β-mimetics. They concluded that progesterone is able to increase the relaxant effect of ritodrine, most likely through non-genic action [10].

Fig. 6.5
Effects of nifedipine (left) or indomethacin (right) with and without P4 on myometrial contractility. AUC area under contractions curve, Nife nifedipina, P4 progesterone. Modified from Baumbach J et al. Am J Obstet Gynecol 2012 [41]
Progesterone can be used for the maintenance of uterine quiescence in patients previously treated for an episode of threatened preterm labour. In comparison with nifedipine, progesterone has a significantly greater effect in prolonging pregnancies of women with arrested preterm labour and, moreover, it results in a better neonatal outcome with less side effects [28]. Borna et al. have studied the role of progesterone in the maintenance of tocolysis after threatened preterm labour treated with intravenous magnesium sulphate. A longer latency period until delivery, a higher gestational age at delivery and lower frequency of low birth weight infants was found [42]. Moreover there is only evidence that micronized progesterone reduces the rate of preterm birth in women successfully treated for a previous episode of preterm labour [29].
5 Conclusions
In conclusion, progesterone is a molecule that has been thoroughly studied and evaluated. According to guidelines published in recent years by various societies (RCOG, ACOG, EAPM, SOGC) concerning the management of preterm labor, the use of progesterone can be recommended as follows: (1) early prophylaxis (from 12 to 36 weeks of gestation) with either micronized progesterone (100 mg vaginally daily) or 17 OHP-C (injection of 250 mg weekly) to prevent a recurrence in case of prior history of PTB [29]. (2) Vaginal micronized progesterone in singleton gestation, with and without history of prior PTB, and a short cervical length (<25 mm) at 18–23 weeks of gestation, since it has been found to be associated with reduction in the rate of PTB and perinatal morbidity and mortality (200 mg vaginal daily) [43, 44]. In this respect recently FIGO has published a best practice advice where it is recommended that women with a sonographic short cervix (< 25 mm) diagnosed in the mid-trimester should be offered daily vaginal micronized progesterone treatment for the prevention of preterm birth and neonatal morbidity. The progesterone formulation to be used is vaginal micronized progesterone (200 mg vaginal soft capsules) nightly or vaginal micronized progesterone gel (90 mg) each morning .Universal cervical length screening and vaginal progesterone treatment (90 mg vaginal gel or 200 mg micronized vaginal soft capsules) is a cost-effective model for the prevention of preterm birth [45]. (3) Vaginal micronised progesterone after one episode of threatened preterm labor (200 mg vaginal daily). (4) Vaginal progesterone in acute tocolysis (400 mg vaginal daily for 48 h) in conjunction with other tocolytic drugs usually used for tocolysis.
Paraphrasing Marcel Proust, “the real voyage of discovery consists not in seeking new landscapes, but in having new eyes,” we can say that the rediscovery of progesterone activities place the hormone in a key role in the prevention of preterm birth.
References
1.
Goldenberg RL, Culhane JF, Iams JD, Romero R. Epidemiology and causes of preterm birth. Lancet. 2008;371:75–84.CrossRefPubMed
2.
Haas DM. Preterm birth. Clin Evid. 2011, pii: 1404.
3.
Di Renzo GC, Cutuli A, Liotta L, Burnelli L, Luzi G. Management of preterm labor: pharmacological and nonpharmacological aspects. In: Textbook of perinatal medicine. 2nd eds, Kurjak A, Chervenak FA. Informa Healthcare; 2006. Cap. 130, pp. 1394–1400.
4.
Druckmann R, Druckmann MA. Progesterone and the immunology of pregnancy. J Steroid Biochem Mol Biol. 2005;97:389–96.CrossRefPubMed
5.
Szekeres-Bartho J, Barakonyi A, Par G, Polgar B, Palkovics T, Szereday L. Progesterone as an immunomodulatory molecule. Int Immunopharmacol. 2001;1:1037–48.CrossRefPubMed
6.
Di Renzo GC, Giardina I, Clerici G, Mattei A, Alajmi AH, Gerli S. The role of progesterone in maternal and fetal medicine. Gynecol Endocrinol. 2012;28:925–32.CrossRefPubMed
7.
Schwartz N, Xue X, Elovitz MA, Dowling O, Metz CN. Progesterone suppresses the fetal inflammatory response ex vivo. Am J Obstet Gynecol. 2009;201:211.PubMed
8.
Fanchin R, Ayoubi JM, Olivennes F, Righini C, de Ziegler D, Frydman R. Hormonal influence on the uterine contractility during ovarian stimulation. Hum Reprod. 2000;15 Suppl 1:90–100.CrossRefPubMed
9.
Perusquía M, Jasso-Kamel J. Influence of 5alpha- and 5beta-reduced progestins on the contractility of isolated human myometrium at term. Life Sci. 2001;68:2933–44.CrossRefPubMed
10.
Chanrachakul B, Broughton Pipkin F, Warren AY, Arulkumaran S, Khan RN. Progesterone enhances the tocolytic effect of ritodrine in isolated pregnant human myometrium. Am J Obstet Gynecol. 2005;192:458–63.CrossRefPubMed
11.
Liu J, Matsuo H, Laoag-Fernandez JB, Xu Q, Maruo T. The effects of progesterone on apoptosis in the human trophoblast-derived HTR-8/SV neo cells. Mol Hum Reprod. 2007;13:869–74.CrossRefPubMed
12.
Czajkowski K, Sienko J, Mogilinski M, Bros M, Szczecina R, Czajkowska A. Uteroplacental circulation in early pregnancy complicated by threatened abortion supplemented with vaginal micronized progesterone or oral dydrogesterone. Fertil Steril. 2007;87:613–8.CrossRefPubMed
13.
Jacobsson B, Mattsby-Baltzer I, Andersch B, Bokström H, Holst RM, Wennerholm UB, Hagberg H. Microbial invasion and cytokine response in amniotic fluid in a Swedish population of women inpreterm labor. Acta Obstet Gynecol Scand. 2003;82:120–8.CrossRefPubMed
14.
Raghupathy R, Al Mutawa E, Makhseed M, Azizieh F, Szekeres-Bartho J. Modulation of cytokine production by dydrogesterone in lymphocytes from women with recurrent miscarriage. BJOG. 2005;112:1096–101.CrossRefPubMed
15.
Hudic I, Szekeres-Bartho J, Fatusic Z, et al. Dydrogesterone supplementation in women with threatened preterm delivery – the impact on cytokine profile, hormone profile, and progesterone-induced blocking factor. J Reprod Immunol. 2011;92:103–7.CrossRefPubMed
16.
Aisemberg J, Vercelli CA, Bariani MV, Billi SC, Wolfson ML, Franchi AM. Progesterone is essential for protecting against LPS-induced pregnancy loss. LIF as a potential mediator of the anti-inflammatory effect of progesterone. PLoS One. 2013;8:e56161. Epub 2013 Feb 7.PubMedCentralCrossRefPubMed
17.
Di Renzo GC, Rosati A, Mattei A, Gojnic M, Gerli S. The changing role of progesterone in preterm labour. BJOG. 2005;112 Suppl 1:57–60.CrossRefPubMed
18.
Karteris E, Zervou S, Pang Y, Dong J, Hillhouse EW, Randeva HS, Thomas P. Progesterone signaling in human myometrium through two novel membrane G protein- coupled receptors: potential role in functional progesterone withdrawal at term. Mol Endocrinol. 2006;20:1519–34.CrossRefPubMed
19.
Merlino AA, Welsh TN, Tan H, Yi LJ, Cannon V, Mercer BM, Mesiano S. Nuclear progesterone receptors in the human pregnancy myometrium: evidence that parturition involves functional progesterone withdrawal mediated by increased expression of progesterone receptor-A. J Clin Endocrinol Metab. 2007;92:1927–33.CrossRefPubMed
20.
Ruddok NK, Shi SQ, Jain S, Moore G, Hankins GDV, Romero R, Garfield RE. Progesterone, but not 17-alpha-hydroxyprogesterone caproate, inhibits human myometrial contractions. Am J Obstet Gynecol. 2008;199(391):e1–7.
21.
Keirse MJ. Progestogen administration in pregnancy may prevent preterm delivery. Br J Obstet Gynaecol. 1990;97:149–54.CrossRefPubMed
22.
Di Renzo GC, Mattei A, Gojnic M, Gerli S. Progesterone and pregnancy. Curr Opin Obstet Gynecol. 2005;17:598–600.CrossRefPubMed
23.
Nahoul K, Dehennin L, Scholler R. Radioimmunoassay of plasma progesterone after oral administration of micronized progesterone. J Steroid Biochem. 1987;26:241–9.CrossRefPubMed
24.
Zava DT, Dollbaum CM, Blen M. Estrogen and progestin bioactivity of foods, herbs, and spices. Proc Soc Exp Biol Med. 1998;217:369–78.CrossRefPubMed
25.
Fitzpatrick LA, Good A. Micronized progesterone: clinical indications and comparison with current treatments. Fertil Steril. 1999;72:389–97.CrossRefPubMed
26.
Bulletti C, de Ziegler D, Flamigni C, Giacomucci E, Polli V, Bolelli G, Franceschetti F. Targeted drug delivery in gynaecology: the first uterine pass effect. Hum Reprod. 1997;12:1073–9.CrossRefPubMed
27.
Cicinelli E, de Ziegler D, Bulletti C, Matteo MG, Schonauer LM, Galantino P. Direct transport of progesterone from vagina to uterus. Obstet Gynecol. 2000;95:403–6.CrossRefPubMed
28.
Kamat S, Veena P, Rani R. Comparison of nifedipine and progesterone for maintenance tocolysis after arrested preterm labour. J Obstet Gynaecol. 2014;34(4):322–5. Epub ahead of print.CrossRefPubMed
29.
Di Renzo GC, Cabero Roura L, Facchinetti F, The European Association of Perinatal Medicine-Study Group on “Preterm Birth”. Guidelines for the management of spontaneous preterm labour. Identification of spontaneous preterm labor, diagnosis of preterm premature rupture of membranes and preventive tools for preterm birth. J Matern Fetal Neonatal Med. 2011;24:659–67.PubMedCentralCrossRefPubMed
30.
Goldstein P, Berrier J, Rosen S, Sacks HS, Chalmers TC. A meta-analysis of randomized control trials of progestational agents in pregnancy. Br J Obstet Gynaecol. 1989;96:265–74.CrossRefPubMed
31.
Daya S. Efficacy of progesterone support for pregnancy in women with recurrent miscarriage. A meta-analysis of controlled trials. Br J Obstet Gynaecol. 1989;96:275–80.CrossRefPubMed
32.
Erny R, Pigne A, Prouvost C, Gamerre M, Malet C, Serment H, Barrat J. The effects of oral administration of progesterone for premature labor. Am J Obstet Gynecol. 1986;154:525–9.CrossRefPubMed
33.
Barton JR, Barton LA, Istwan NB, Rhea DJ, Desch CN, Sibai BM. 17-Alphahydroxyprogesterone caproate in women with previous spontaneous preterm delivery: does a previous term delivery affect the rate of recurrence? Am J Obstet Gynecol. 2011;205:269.e1–6.CrossRef
34.
da Fonseca EB, Bittar RE, Carvalho MH, Zugaib M. Prophylactic administration of progesterone by vaginal suppository to reduce the incidence of spontaneous preterm birth in women at increased risk: a randomized placebo-controlled double-blind study. Am J Obstet Gynecol. 2003;188:419–24.CrossRefPubMed
35.
Romero R, Nicolaides K, Conde-Agudelo A, Tabor A, O’Brien JM, Cetingoz E, et al. Vaginal progesterone in women with an asymptomatic sonographic short cervix in the midtrimester decreases preterm delivery and neonatal morbidity: a systematic review and metaanalysis of individual patient data. Am J Obstet Gynecol. 2012;206:124.e1–19.CrossRef
36.
Grobman WA, Thom EA, Spong CY, Iams JD, Saade GR, Mercer BM, Eunice Kennedy Shriver National Institute of Child Health and Human Development Maternal-Fetal Medicine Units (MFMU) Network, et al. 17 Alpha-hydroxyprogesterone caproate to prevent prematurity in nulliparas with cervical length less than 30 mm. Am J Obstet Gynecol. 2012;207:390.e1–8.CrossRef
37.
Maher MA, Abdelaziz A, Ellaithy M, Bazeed MF. Prevention of preterm birth: a randomized trial of vaginal compared with intramuscularprogesterone. Acta Obstet Gynecol Scand. 2013;92:215–22.CrossRefPubMed
38.
Hartikainen-Sorri AL, Kauppila A. Tuimala R Inefficacy of 17 alpha-hydroxyprogesterone caproate in the prevention of prematurity in twin pregnancy. Obstet Gynecol. 1980;56:692–5.PubMed
39.
Combs CA, Garite T, Maurel K, Das A, Porto M, Obstetrix Collaborative Research Network. 17-Hydroxyprogesterone caproate for twin pregnancy: a double-blind, randomized clinical trial. Am J Obstet Gynecol. 2011;204:221.e1–8.CrossRef
40.
Kurjak A, Chervenak FA. Management of preterm labor: pharmacological and non-pharmacological aspects. In: Textbook of perinatal medicine. 2nd ed. Informa Healthcare; 2006. Cap. 130, pp. 1394–1400.
41.
Baumbach J, Shi SQ, Shi L, Balducci J, Coonrod DV, Garfield RE. Inhibition of uterine contractility with various tocolytics with and without progesterone: in vitro studies. Am J Obstet Gynecol. 2012;206:254.e1–5.
42.
Borna S, Sahabi N. Progesterone for maintenance tocolytic therapy after threatened preterm labour: a randomised controlled trial. Aust N Z J Obstet Gynaecol. 2008;48:58–63.CrossRefPubMed
43.
Society for Maternal-Fetal Medicine Publications Committee, with assistance of Vincenzo Berghella. Progesterone and preterm birth prevention: translating clinical trials data into clinical practice. Am J Obstet Gynecol. 2012;206:376–86.CrossRef
44.
Farine D, Mundle WR, Dodd J, Basso M, Delisle MF, Farine D, Maternal Fetal Medicine Committee of the Society of Obstetricians and Gynaecologists of Canada, et al. The use of progesterone for prevention of preterm birth. J Obstet Gynaecol Can. 2008;30:67–77.PubMed
45.
Miles RA, Paulson RJ, Lobo RA, Press MF, Dahmoush L, Sauer MV. Pharmacokinetics and endometrial tissue levels of progesterone after administration by intramuscular and vaginal routes: a comparative study. Fertil Steril. 1994;62:485–90.PubMed