Nishant Patel1 and T. Mike Hsieh1
(1)
Department of Urology, University of California, San Diego, San Diego, CA, USA
T. Mike Hsieh
Email: tmikehsieh@ucsd.edu
Keywords
Erectile dysfunctionPDE-5 inhibitorsVacuum erection deviceIntraurethral suppositoryIntracavernosal injectionsPenile prosthesisAcupuncture
7.1 Introduction
The treatment of erectile dysfunction (ED) has advanced over the past century. Prior to the 1960s, when ED was thought to be of psychogenic or unknown etiology, treatments consisted of testosterone supplementation or psychiatric evaluation and treatment. With advancements in the understanding of vascular, neurologic, and hormonal physiology of erectile function, a multitude of pharmacologic and surgical treatments have emerged (Fig. 7.1) [1].
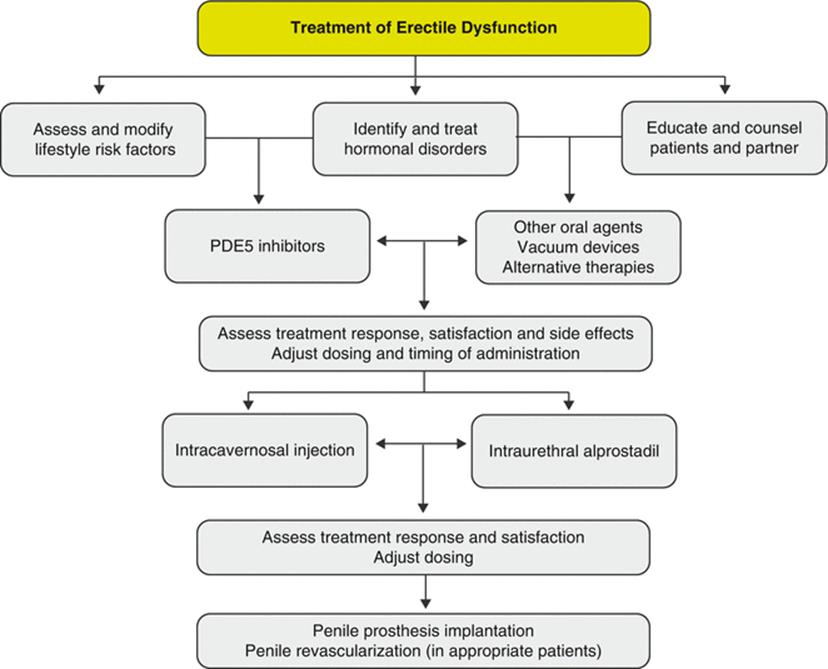
Fig. 7.1
Algorithm for the clinical treatment of erectile (dysfunction PDE5—phospho-diesterase type 5)
Overall, the use of oral phosphodiesterase type 5 inhibitors (PDE5i’s) is the most common treatment for ED. Other treatment options include lifestyle modifications to address comorbid conditions, testosterone supplementation for androgen-deficient men, intracavernosal injection (ICI) therapy, intraurethral medications, vacuum and constriction devices, penile prosthetic surgery, alternative therapies, and psychotherapy. Combining psychosocial and medical treatment will offer individualized therapy and assist patients in understanding their individual conditions.
7.2 Lifestyle Modification
Normal erectile function relies on the interaction of vascular, neurologic, hormonal, and psychologic mechanisms. Comorbidities such as coronary artery disease, peripheral vascular disease, hypertension, diabetes, and the metabolic syndrome are implicated as risk factors in developing ED. Additionally, a number of lifestyle risk factors have been associated with ED, including reduced physical activity, tobacco use, alcohol consumption, and obesity [2]. Thus, physician-directed counseling and modification of these risk factors can positively impact erectile function. For instance, in a group of patients where a strong association between cigarette smoking and degree of ED was observed, smoking cessation improved ED in up to a quarter of the population [3]. In a randomized study of obese patients, those men who adopted lifestyle changes including reduced caloric intake and increased physical activity reported an increase of three points on the International Index of Erectile Function (IIEF) , a validated instrument for the assessment of erectile function [4].
Several mechanisms explaining the beneficial effect of physical activity and weight reduction on erectile function have been proposed, including improvement in endothelial cell function, decreased inflammation, increased serum testosterone, and improvements in mood and affect [5]. However, definitive clinical and basic science evidence is often lacking to confirm the favorable association between lifestyle changes and improvement in erectile function. A recent meta-analysis demonstrated improved IIEF scores after only 6 weeks of lifestyle modification and pharmacotherapy for cardiovascular risk factors [6]. However, other reports suggest lifestyle changes may take 12–24 months to achieve maximal clinical results, and proven pharmacotherapy should not be withheld pending these changes [4]. Sensible lifestyle goals for the man with ED include increasing exercise, reducing weight to achieve a BMI less than 30 kg/m2, instituting a “heart-healthy” diet, and tobacco cessation [5, 7].
7.3 Testosterone Supplementation
Testosterone has well-defined roles in maintaining libido, supporting fertility, and sustaining adequate erectile function, through the stimulation of genes that increase nitric oxide synthase (NOS) expression [8]. Testosterone supplementation is recommended only in those men with ED who have androgen deficiency and are no longer interested in reproduction. Most clinicians agree that men who will likely benefit from testosterone therapy have serum testosterone levels of <300 ng/dL. However, calculating bioavailable or free testosterone might provide a more accurate measurement [9]. Testosterone monotherapy in men with ED and a serum total testosterone concentration <300 ng/dL has been shown to improve mean IIEF scores by six points after 1 year of therapy [10]. In men aged >65 years with androgen deficiency, combination therapy with PDE5i’s and testosterone supplementation has been successful when PDE5i’s alone were ineffective [11].
Testosterone is available in several preparations and can be taken orally, intramuscularly, transdermally (patch or gel), or buccally or implanted subcutaneously [12]. Prior to initiating testosterone supplementation, several baseline parameters should be assessed including serum prostate-specific antigen (PSA), complete blood count (CBC), lipid profile, and liver function tests, and a digital rectal examination performed. According to manufacturer’s product inserts, testosterone supplementation has the potential to exacerbate certain conditions such as untreated prostate or breast cancer, uncontrolled congestive heart failure, polycythemia (hematocrit >52 %), severe sleep apnea, severe dyslipidemia, or severe prostatic bladder outlet obstruction and can affect patients at risk for pulmonary edema. Therefore, testosterone supplementation in these patients warrants extra attention and should be implemented after thorough evaluation and treatment of underlying conditions. Repeat serum testosterone levels after initiating therapy are necessary to assess for potential over- and undertreatment, together with assessment of the patient’s hypogonadal symptoms. A patient’s clinical response may not correlate with serum testosterone levels, and attempting to achieve supratherapeutic testosterone levels is not recommended. Some authors suggest discontinuing testosterone replacement if no clinical benefit is seen 3 months into therapy [13]. Potential adverse effects should be identified including acne, gynecomastia, progression of urinary symptoms, prostate cancer progression in both treated and untreated men, polycythemia, dyslipidemia, and hepatitis. Therefore, monitoring of PSA, CBC, lipid profile, and liver function tests is requisite at six-month intervals.
7.4 Oral Phosphodiesterase Type 5 Inhibitors
Per American Urologic Association (AUA) guidelines, unless contraindicated, oral PDE5i’s constitute first-line therapy for the treatment of ED [14]. The US Food and Drug Administration (FDA) has approved five PDE5i’s for the treatment of ED to date: sildenafil citrate tablets (Viagra®, Pfizer, Inc), vardenafil hydrochloride tablets (Levitra®, Bayer Pharma AG), vardenafil hydrochloride orally disintegrating tablets (Staxyn®, Bayer Pharma AG), tadalafil tablets (Cialis®, Lilly LLC), and, recently, avanafil tablets (Stendra®, Vivus, Inc.) (Table 7.1). The mechanism of action of PDE5i’s is to inhibit phosphodiesterase 5, the enzyme responsible for metabolizing cGMP, which is produced by guanylate cyclase under the influence of nitric oxide. Increased levels of cGMP result in relaxation of erectogenic smooth muscle in the corpora cavernosa, improving cavernosal blood flow and resulting in penile erections [15]. PDE5i’s do not result in spontaneous erections and require sexual stimulation for efficacy, as this generates 3′5′-cGMP, the substrate for PDE5.
Table 7.1
Food and Drug Administration (FDA) —approved oral PDE5-inhibitor drugs
|
Sildenafil |
Vardenafil |
Tadalafil |
Avanafil |
|
|
Dosage |
25, 50, and 100 mg. Start with 50 mg |
2.5, 5, 10, and 20 mg. Start with 10 mg 10 mg—orally disintegrating |
2.5, 5, 10, and 20 mg. 2.5 and 5 mg daily dosing. Start with 10 mg |
50, 100, and 200 mg. Start with 100 mg |
|
Onset of action (min) |
30–60 |
30 |
45 |
15 |
|
Duration (h) |
4–8 |
4–8 |
Up to 36 |
6 |
|
Efficacy (%) |
>65 |
>65 |
>65 |
>65 |
|
Side effects |
Headache, flushing, dyspepsia, nasal congestion, abnormal vision |
Flushing, nasal congestion, headache, abnormal vision |
Flushing, back pain, myalgia, headache, dyspepsia, facial flushing |
Flushing, nasal congestion, back pain |
|
Contraindications |
Nitrates, recent cardiovascular events. Caution with alpha-blockers |
Nitrates, alpha-blockers, type 1 and 3 antiarrhythmics, prolonged QT interval |
Nitrates, recent cardiovascular events. Caution with alpha-blockers |
Nitrates, recent cardiovascular events. Caution with alpha-blockers |
|
Fatty food |
Reduced absorption |
Reduced absorption |
No effect |
Reduced absorption |
Overall, PDE5i’s are effective in 65 % of patients with efficacy defined as resulting in an erection sufficient for vaginal penetration, of 65 % [16]. The choice of PDE5i depends on the patient’s frequency of intercourse while balancing efficacy with individual side effect profiles. An assessment of a couple’s “sexual script” can help determine which drug is given based on timing and need [17]. For patients engaging in occasional sexual activity at a defined time, on-demand dosing is ideal, whereas those men engaging in frequent activity might benefit from daily PDE5i use. The various PDE5i’s vary in terms of onset of action, duration of effect, absorption efficacy, and side effect profiles.
Sildenafil was the first PDE5i introduced for the treatment of ED in 1998. It is effective 30–60 min after administration and has a mean duration of action of 4–8 h, although efficacy may extend up to 12 h. Dosing is available in 25, 50, and 100 mg pills, with the usual starting dose at 50 mg [1]. If taken with a high fat meal (>57 % fat), sildenafil’s rate of absorption is reduced, translating to prolonged onset times or decreased efficacy. Therefore, administration while fasting is recommended. The most common side effects of sildenafil include headache (16 %), flushing (10 %), and dyspepsia (7 %). Other less common adverse reactions include nasal congestion, vision changes, and diarrhea [18].
Vardenafil , introduced in 2003, is effective within 30 min of administration and is available in 2.5, 5, 10, and 20 mg doses, with 10 mg representing the most common starting dose. In vitro, vardenafil inhibits PDE5 at concentrations tenfold lower than does sildenafil, although this may not correlate with clinical response. Vardenafil shares the same decreased absorption as sildenafil when taken with high fat meals. Flushing and nasal congestion are more common with vardenafil (9–11 %) [19]. The orally disintegrating preparation of vardenafil (Staxyn) is available in a 10 mg dose, can be taken with a high fat meal with no impact on absorption, and attains higher plasma concentrations than film-coated tablets. Neither form of vardenafil should be administered to patients taking type 1 or 3 antiarrhythmic drugs or those with congenital QT interval prolongation [20].
Tadalafil , introduced in 2003, may be administered in an on-demand fashion or daily. Available in 2.5, 5, 10, and 20 mg doses, on-demand dosing should begin at 10 mg, with a maximum of 20 mg. While tadalafil has a longer time to onset than sildenafil or vardenafil (45 min), its effects may last up to 36 h and are not impacted by the fat content of food [21]. In 2008, tadalafil was approved for daily use for ED, with a dose of 2.5–5 mg. This regimen, ideal for couples desiring frequent spontaneous sexual activity, is well tolerated, with improvements in IIEF scores [22]. Tadalafil has also recently been approved for treating men with lower urinary tract symptoms secondary to BPH at a dose of 5 mg daily [23]. More than the other PDE5i’s, tadalafil exhibits the higher incidence of back pain and myalgia, thought to be secondary to cross-reactivity with PDE11, found in skeletal muscle.
Lastly, avanafil , introduced in 2013, has shown greater selectivity for PDE5 than the other agents and can be effective within 15 min of administration. The absorption of avanafil is reduced with a high fat meal, and its duration of action is short—6 h. Avanafil displays a low prevalence (<6 %) of common PDE5i adverse effects [24]. Other PDE5i’s, namely, udenafil and mirodenafil, are approved for use in Korea, while lodenafil is in clinical trials.
An absolute contraindication to the use of all PDE5i’s is in patients taking organic nitrates or nitric oxide donors (nitroglycerin, isosorbide dinitrate, amyl nitrates) for angina within a period of 2 weeks, as concomitant use may result in severe and life-threatening hypotension. Men taking alpha-blockers for bladder outlet obstruction are also at risk for hypotension when taking PDE5i’s, and combination therapy of alpha-blockers and PDE5i’s should be used with caution. Interestingly, low-dose daily tadalafil (2.5–5 mg) is also FDA approved for the treatment of lower urinary tract symptoms secondary to benign prostatic hyperplasia. Sildenafil and vardenafil have a risk of blurry vision from PDE6 cross-reactivity, and rare sudden loss of vision secondary to nonarteritic anterior optic neuropathy (NAION) may occur, although this pathology has not been directly linked to PDE5i’s [25]. The pharmacokinetics of PDE5i’s are not greatly affected by alcohol, although refraining from alcohol is suggested given the link between alcohol consumption and ED. However, if alcohol is taken, studies have shown that PDE5i’s do not potentiate alcohol’s hypotensive effects. A lower dose of PDE5i may be needed in patients taking steroid production inhibitors (ketoconazole), macrolide antibiotics, and HIV protease inhibitors (ritonavir) or those with history of liver and renal dysfunction [1].
The major reasons for nonresponse to PDE5i are poor patient understanding of medication usage and a lack of PDE5i efficacy. Patients require adequate counseling on the timing of administration related to food intake, need for sexual stimulation, timing of onset of each agent, and an understanding of potential side effects. Men should attempt a single PDE5i agent at least 4 times prior to dose escalation. No convincing data exists to support switching PDE5i after failure with one correctly administered agent, although changing agents due to drug intolerance is likely to be beneficial [26].
7.5 Other Oral Treatments
Dopamine within the periventricular nucleus of the brain plays a role in the central control of erections. Apomorphine is a centrally acting dopaminergic agent that is taken sublingually on demand in doses of 2, 4, and 6 mg and is of particular interest in treating psychogenic ED. Side effects include nausea, headache, dizziness, and, rarely, syncopal events. Apomorphine has shown the best efficacy in patients with mild to moderate ED [27]. Bremelanotide is a synthetic analogue of alpha-melanocyte stimulating hormone, which activates melanocortin receptors 3 and 4. This activation is thought to regulate sexual behavior and erectile function. Studies of bremelanotide administered subcutaneously appear to increase libido and initiate erections, but the drug is not currently approved for commercial use [28].
Oral phentolamine mesylate is an alpha-adrenergic receptor antagonist. In erectile physiology, the alpha-1 adrenoreceptor’s downstream effects result in contraction of smooth muscle and detumescence via phospholipase C. One study demonstrated 40 mg of phentolamine to be efficacious in men with mild to moderate ED, with side effects including headache, facial flushing, and nasal congestion [29]. Yohimbine hydrochloride, a derivative from the bark of the yohimbe tree, acts as an alpha-2 adrenoreceptor antagonist and is administered at 5.4 mg three times daily. Adverse reactions include hypertension, tachycardia, and anxiety, and yohimbe has no proven efficacy in men with organic ED compared to placebo [30]. Acting via serotonin receptors, trazodone, with a known side effect of priapism, has been studied in the setting of ED, but clinical evidence demonstrating efficacy is lacking.
7.6 Intraurethral Alprostadil
After failure of oral therapies, second-line agents such as ICIs and intraurethral suppositories are indicated. Alprostadil is a synthetic vasodilator that mimics naturally occurring prostaglandin E1. Alprostadil binds to adenylate cyclase and increases cyclic AMP, which results in relaxation of erectogenic smooth muscle. MUSE® (Medicated Urethral System for Erections, Meda Pharmaceuticals, Inc.) is an intraurethral alprostadil suppository placed into the distal urethra (Fig. 7.2). The drug is absorbed through the urethral mucosa and into the corpora cavernosa and acts within 20 min of administration. The suppositories are available in 125, 250, 500, and 1000 mcg, and recent literature supports a starting dose of 500 mcg, as this has shown higher efficacy than lower doses without a significant increase in adverse events [31]. Men with neurogenic ED should be started on the lowest possible dose. Approximately 50 % of men respond to MUSE, and among those, 70 % have erections sufficient for penetration [32]. Adverse effects include priapism, penile/urethral pain, headache, dizziness, and syncope. Since a 3 % risk of hypotension and syncope has been reported, especially in men with a venous leak component to their ED, it is recommended that the first dose of MUSE be administered under supervision of a healthcare provider [32]. If the female partner is pregnant, a condom is recommended when using MUSE. Studies have demonstrated that intraurethral alprostadil, in combination with a PDE5i or penile constriction device, is more effective than alprostadil alone [33].
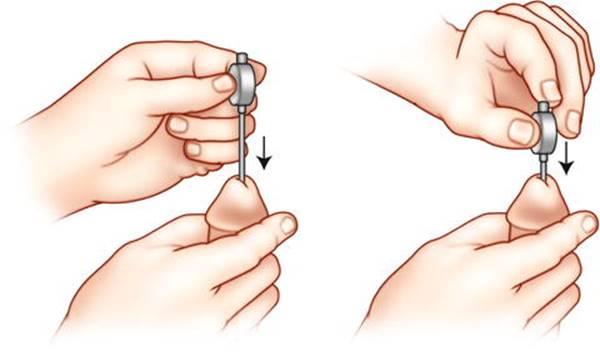
Fig. 7.2
Insertion method for MUSE® intraurethral alprostadil pellet.
7.7 Intracavernosal Injections
As another second-line therapy, ICI of vasoactive agents allows for on-demand erections within 5–15 min (Fig. 7.3). Efficacy rates range from 70 to 90 %, with satisfaction rates ranging from 87 to 93.5 % [1]. Several agents are used alone or in combination to achieve an optimal response. Injectable alprostadil is available as Edex (Auxilium Pharmaceuticals, LLC) and Caverject (Pfizer, Inc.) as monotherapy. Side effects include penile pain (11 %), local hematoma, priapism (2 %), and penile fibrosis (1 %) [34]. Papaverine is a nonspecific phosphodiesterase inhibitor, resulting in increased intracellular levels of cAMP and cGMP. Papaverine has a lower risk of penile pain than alprostadil, but higher risk of priapism and fibrosis (10–12 %). Phentolamine inhibits alpha-adrenergic receptors and inhibits detumescence. Its side effects include hypotension, nasal congestion, reflex tachycardia, and dyspepsia. Bi-Mix, a mixture of papaverine and phentolamine; Tri-Mix, a mixture of papaverine, phentolamine, papaverine, and prostaglandin E1; and Quad-mix, consisting of Tri-Mix with the addition of atropine or forskolin, all result in efficacy rates of 80–90 % [35]. The distinct advantage of the above mixtures includes decreased volume for injection and potential superiority versus prostaglandin E1, alone in treating ED due to venous leak. Vasoactive intestinal polypeptide (VIP) has been studied for ICI, but has poor results as monotherapy. A mixture of VIP with phentolamine (Invicorp) exhibits greater efficacy (80 %) than VIP alone and is currently approved in other countries [36].
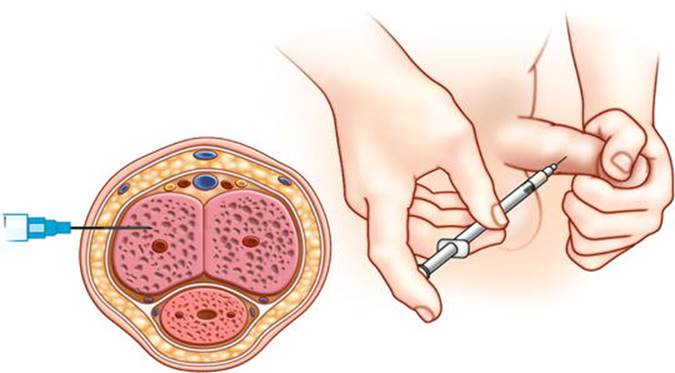
Fig. 7.3
Injection method for intracavernosal injection . The patient performs self-injection of a vasoactive agent directly into the penile corpora cavernosa.
Dose titration should begin at the lowest possible dose, especially in men with neurogenic ED. Initial dosing under a healthcare provider’s supervision is recommended to ensure proper technique (including partner education) and determine appropriate dose. Contraindications for ICI include men at risk for priapism (prior events, sickle cell anemia), poor manual dexterity, significant coagulopathy, and the use of monoamine oxidase inhibitors (to prevent severe hypertension if an alpha agonist is necessary during a priapism event) [37]. Men with recurring priapism secondary to ICI are managed with dose reduction and a phenylephrine kit for self-treatment.
7.8 Vacuum Erection Device
In men who fail, cannot tolerate, or have contraindications to PDE5i or ICI, a vacuum erection device (VED) offers a good noninvasive option and represents another second-line ED treatment option. A VED works by creating a vacuum around the penis, resulting in expansion of the corpora cavernosa and increased inflow of blood. Placement of a penile constriction ring diminishes outflow of blood and prevents penile detumescence. The VED consists of a plastic cylinder and a pump source to generate negative pressure (Fig. 7.4). After an erection is achieved, the constriction band is placed at the penile base, and the device removed. Recent reviews suggest efficacy rates range from 75 to 91 % in achieving erection sufficient for penetration and satisfaction rates ranging from 65 to 80 % [38].
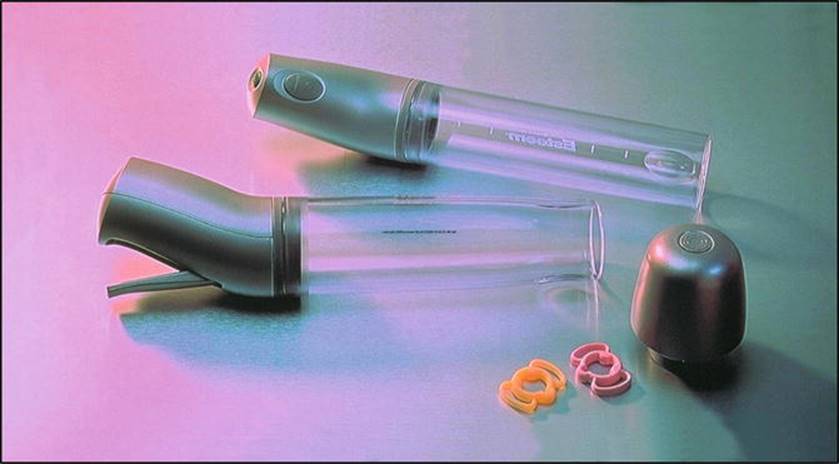
Fig. 7.4
Commercially available vacuum erection device with either manual or electric pump and constriction bands. With permission from Timm Medical Technologies, Inc.
VEDs display a low rate of complications, including bruising, interference with ejaculation secondary to the constrictive ring, numbness, and penile pain. Men on anticoagulants may experience increased ecchymosis and should use caution. The device is of particular interest in post-prostatectomy penile rehabilitation, as early VED use may improve cavernosal tissue oxygenation and erections and facilitate earlier sexual intercourse postoperatively [39]. Additionally, combination of a VED with PDE5i, ICI, or MUSE has been shown to increase efficacy and patient-partner satisfaction [40, 41].
7.9 Surgical Treatments
For patients with ED who fail first- and second-line treatments, a third-line treatment is penile prosthetic surgery . After replacing the normal erectile tissue with the prosthesis, the corporal tissue is altered and the potential for smooth muscle relaxation is eliminated, making “natural” erections impossible after prosthesis placement. Adequate counseling is necessary regarding penile prosthesis options, risk of erosion or infection, need for subsequent operations, penile shortening, and proper use of the device. Satisfaction rates as high as 85 % for the patient and 76 % for his partner have been reported [42]. The two main types of prostheses are non-inflatable (malleable) and inflatable. Non-inflatable devices are semirigid and manipulate to an erect position for sexual activity. The advantages of a non-inflatable device include a low rate of mechanical failure and less need for manual dexterity. However, malleable prostheses carry an increased risk of erosion and chronic pain [43]. Inflatable prostheses are available in two- or three-piece configurations. The three-piece inflatable prosthesis is most commonly used and consists of paired corporal cylinders, an abdominal fluid reservoir, and a scrotal pump. It provides a flaccid state at baseline, and activating the scrotal pump allows fluid to transfer from the abdominal reservoir to the cylinders, resulting in an increase in penile girth and rigidity. A release valve on the pump then transmits fluid back to the reservoir (Fig. 7.5). Compared to malleable devices, inflatable prostheses have a higher rate of mechanical failure (6–15 % at 5 years) [44] yet more lifelike function.
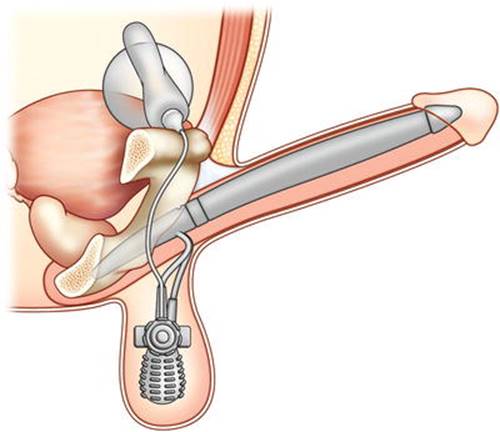
Fig. 7.5
The three-piece inflatable penile prosthesis consisting of paired corporal cylinders, an abdominal fluid reservoir, and a scrotal pump.
Complications of penile prosthesis placement include bleeding, infection (2–4 % of cases), mechanical failure, injury to vascular or bowel structures, erosion, and penile shortening. Erosion and infection usually require explantation of some or all of the prosthesis. Reimplantation of another device after infection may be performed after the infection resolves or during the explantation procedure to preserve penile length [45].
Surgeries to either limit venous outflow or increase arterial inflow are appealing in theory, but currently have limited clinical utility. The AUA recommends against surgeries to restrict venous outflow and recommends penile arterial reconstructive surgery for men <55 years old, nonsmokers, nondiabetics, without venous leak, and with a focal stenosis of the internal pudendal artery [46]. Complications of revascularization include glans hyperemia, thrombosis of the anastomoses, scarring, and hernia [47].
7.10 Alternative Therapies
Throughout history, a myriad of alternative therapies have been employed to treat ED, including herbal formulations (gingko, ginseng), L-arginine, hypnotherapy, pelvic floor physiotherapy, biofeedback, and acupuncture [48]. Unfortunately, randomized controlled trials are lacking in comparing herbal therapies to well-established pharmacotherapy. A small study randomizing men with ED to acupuncture and hypnosis versus placebo demonstrated improvements in sexual function in the treatment group, but these were not significantly different from the control group [49]. Since most of these therapies are patient driven, they are best employed in conjunction with evidence-based pharmacotherapy.
7.11 Psychotherapy
While medical therapies are aimed at treating the underlying pathophysiologic mechanisms of ED, a psychogenic component exists for all types of ED. Effectively addressing these psychogenic issues is essential in maximizing sexual outcomes for the couple. A Japanese study demonstrated that ED was significantly associated with depression and anxiety status [50]. The pharmacologic treatment of depression and anxiety often carries undesired sexual side effects. Psychotherapy can aid in overcoming psychosocial barriers, making clinical treatments for ED more effective. The goals of psychotherapy include identifying and addressing resistances to medical intervention, reducing or eliminating performance anxiety, understanding the context for a couple’s sexual activity, and implementing education and modification of sexual scripts [51]. A meta-analysis evaluating the effectiveness of psychological interventions for the treatment of ED compared to the clinical treatments described above demonstrated that group therapy improves ED and that psychotherapy in addition to PDE5i’s was more effective than pharmacotherapy alone [52]. As the clinical treatment of ED advances, physicians can maximize effectiveness by addressing the patient as a whole with appropriate psychosocial interventions.
FUTURE THERAPIES—addressed in Chap. 12 (A. Burnett).
7.12 Summary Statement
The physician treating erectile dysfunction has a large armamentarium of clinical treatments to utilize in a step-wise fashion if initial therapies fail. A thorough assessment of psychosocial factors will aid in maximizing the benefit of these therapies.
References
1.
Hatzimouratidis K, Amar E, Eardley I, et al. Guidelines on male sexual dysfunction: erectile dysfunction and premature ejaculation. Eur Urol. 2010;57:804–14.CrossRefPubMed
2.
Lewis RW, Fugl-Meyer KS, Corona G, et al. Definitions/epidemiology/risk factors for sexual dysfunction. J Sex Med. 2010;7:1598–607.CrossRefPubMed
3.
Pourmand G, Alidaee MR, Rasuli S, et al. Do cigarette smokers with erectile dysfunction benefit from stopping?: a prospective study. BJU Int. 2004;94:1310–3.CrossRefPubMed
4.
Esposito K, Giugliano F, Di Palo C, et al. Effect of lifestyle changes on erectile dysfunction in obese men: a randomized controlled trial. JAMA. 2004;291:2978–84.CrossRefPubMed
5.
Glina S, Sharlip ID, Hellstrom WJ. Modifying risk factors to prevent and treat erectile dysfunction. J Sex Med. 2013;10:115–9.CrossRefPubMed
6.
Gupta BP, Murad MH, Clifton MM, et al. The effect of lifestyle modification and cardiovascular risk factor reduction on erectile dysfunction: a systematic review and meta-analysis. Arch Intern Med. 2011;171:1797–803.CrossRefPubMed
7.
Mozaffarian D, Wilson PW, Kannel WB. Beyond established and novel risk factors: lifestyle risk factors for cardiovascular disease. Circulation. 2008;117:3031–8.CrossRefPubMed
8.
Jacob BC. Testosterone replacement therapy in males with erectile dysfunction. J Pharm Pract. 2011;24:298–306.CrossRefPubMed
9.
Bhasin S, Cunningham GR, Hayes FJ, et al. Testosterone therapy in men with androgen deficiency syndromes: an endocrine society clinical practice guideline. J Clin Endocrinol Metab. 2010;95:2536–59.CrossRefPubMed
10.
Karazindiyanoglu S, Cayan S. The effect of testosterone therapy on lower urinary tract symptoms/bladder and sexual functions in men with symptomatic late-onset hypogonadism. Aging Male. 2008;11:146–9.CrossRefPubMed
11.
Shamloul R, Ghanem H, Fahmy I, et al. Testosterone therapy can enhance erectile function response to sildenafil in patients with PADAM: a pilot study. J Sex Med. 2005;2:559–64.CrossRefPubMed
12.
Fabbri A, Giannetta E, Lenzi A, et al. Testosterone treatment to mimic hormone physiology in androgen replacement therapy. A view on testosterone gel and other preparations available. Expert Opin Biol Ther. 2007;7:1093–106.CrossRefPubMed
13.
Wang C, Nieschlag E, Swerdloff R, et al. Investigation, treatment, and monitoring of late-onset hypogonadism in males: ISA, ISSAM, EAU, EAA, and ASA recommendations. Eur Urol. 2009;55:121–30.CrossRefPubMed
14.
Montague DK, Jarow JP, Broderick GA, et al. Chapter 1: The management of erectile dysfunction: an AUA update. J Urol. 2005;174:230–9.CrossRefPubMed
15.
Goldstein I, Lue TF, Padma-Nathan H, et al. Oral sildenafil in the treatment of erectile dysfunction. Sildenafil study group. N Engl J Med. 1998;338:1397–404.CrossRefPubMed
16.
Carson CC. Phosphodiesterase type 5 inhibitors: state of the therapeutic class. Urol Clin North Am. 2007;34:507–15.CrossRefPubMed
17.
Dunn ME, Althof SE, Perelman MA. Phosphodiesterase type 5 inhibitors’ extended duration of response as a variable in the treatment of erectile dysfunction. Int J Impot Res. 2007;19:119–23.CrossRefPubMed
18.
Viagra (sildenafil) package insert. 2013.
19.
Vardenafil (Levitra) package insert. 2013.
20.
Staxyn (vardenafil ODT) package insert. 2013.
21.
Tadalafil (Cialis) package insert. 2013.
22.
Rajfer J, Aliotta PJ, Steidle CP, et al. Tadalafil dosed once a day in men with erectile dysfunction: a randomized, double-blind, placebo-controlled study in the US. Int J Impot Res. 2007;19:95–103.CrossRefPubMed
23.
Porst H, Kim ED, Casabe AR, et al. Efficacy and safety of tadalafil once daily in the treatment of men with lower urinary tract symptoms suggestive of benign prostatic hyperplasia: results of an international randomized, double-blind, placebo-controlled trial. Eur Urol. 2011;60:1105–13.CrossRefPubMed
24.
Burke RM, Evans JD. Avanafil for treatment of erectile dysfunction: review of its potential. Vasc Health Risk Manag. 2012;8:517–23.PubMedPubMedCentral
25.
Bella AJ, Brant WO, Lue TF, et al. Non-arteritic anterior ischemic optic neuropathy (NAION) and phosphodiesterase type-5 inhibitors. Can J Urol. 2006;13:3233–8.PubMed
26.
Kendirci M, Tanriverdi O, Trost L, et al. Management of sildenafil treatment failures. Curr Opin Urol. 2006;16:449–59.CrossRefPubMed
27.
Mohee A, Bretsztajn L, Eardley I. The evaluation of apomorphine for the treatment of erectile dysfunction. Expert Opin Drug Metab Toxicol. 2012;8:1447–53.CrossRefPubMed
28.
Eardley I, Donatucci C, Corbin J, et al. Pharmacotherapy for erectile dysfunction. J Sex Med. 2010;7:524–40.CrossRefPubMed
29.
Padma-Nathan H, Goldstein I, Klimberg I, et al. Long-term safety and efficacy of oral phentolamine mesylate (Vasomax) in men with mild to moderate erectile dysfunction. Int J Impot Res. 2002;14:266–70.CrossRefPubMed
30.
Teloken C, Rhoden EL, Sogari P, et al. Therapeutic effects of high dose yohimbine hydrochloride on organic erectile dysfunction. J Urol. 1998;159:122–4.CrossRefPubMed
31.
Costa P, Potempa AJ. Intraurethral alprostadil for erectile dysfunction: a review of the literature. Drugs. 2012;72:2243–54.CrossRefPubMed
32.
Padma-Nathan H, Hellstrom WJ, Kaiser FE, et al. Treatment of men with erectile dysfunction with transurethral alprostadil. Medicated urethral system for erection (MUSE) study group. N Engl J Med. 1997;336:1–7.CrossRefPubMed
33.
Mydlo JH, Volpe MA, Macchia RJ. Initial results utilizing combination therapy for patients with a suboptimal response to either alprostadil or sildenafil monotherapy. Eur Urol. 2000;38:30–4.CrossRefPubMed
34.
Linet OI, Ogrinc FG. Efficacy and safety of intracavernosal alprostadil in men with erectile dysfunction. The Alprostadil Study Group. N Engl J Med. 1996;334:873–7.CrossRefPubMed
35.
Perimenis P, Konstantinopoulos A, Perimeni PP, et al. Long-term treatment with intracavernosal injections in diabetic men with erectile dysfunction. Asian J Androl. 2006;8:219–24.CrossRefPubMed
36.
Dinsmore WW, Wyllie MG. Vasoactive intestinal polypeptide/phentolamine for intracavernosal injection in erectile dysfunction. BJU Int. 2008;102:933–7.CrossRefPubMed
37.
Porst H. Current perspectives on intracavernosal pharmacotherapy for erectile dysfunction. Int J Impot Res. 2000;12 Suppl 4:S91–100.CrossRefPubMed
38.
Brison D, Seftel A, Sadeghi-Nejad H. The resurgence of the vacuum erection device (VED) for treatment of erectile dysfunction. J Sex Med. 2013;10:1124–35.CrossRefPubMed
39.
Raina R, Agarwal A, Ausmundson S, et al. Early use of vacuum constriction device following radical prostatectomy facilitates early sexual activity and potentially earlier return of erectile function. Int J Impot Res. 2006;18:77–81.CrossRefPubMed
40.
Canguven O, Bailen J, Fredriksson W, et al. Combination of vacuum erection device and PDE5 inhibitors as salvage therapy in PDE5 inhibitor nonresponders with erectile dysfunction. J Sex Med. 2009;6:2561–7.CrossRefPubMed
41.
John H, Lehmann K, Hauri D. Intraurethral prostaglandin improves quality of vacuum erection therapy. Eur Urol. 1996;29:224–6.PubMed
42.
Holloway FB, Farah RN. Intermediate term assessment of the reliability, function and patient satisfaction with the AMS700 Ultrex penile prosthesis. J Urol. 1997;157:1687–91.CrossRefPubMed
43.
Zermann DH, Kutzenberger J, Sauerwein D, et al. Penile prosthetic surgery in neurologically impaired patients: long-term followup. J Urol. 2006;175:1041–4 (discussion 1044).CrossRefPubMed
44.
Montague DK. Penile prosthesis implantation in the era of medical treatment for erectile dysfunction. Urol Clin North Am. 2011;38:217–25.CrossRefPubMed
45.
Mulcahy JJ. Penile prosthesis infection: progress in prevention and treatment. Curr Urol Rep. 2010;11:400–4.CrossRefPubMed
46.
Hellstrom WJ, Montague DK, Moncada I, et al. Implants, mechanical devices, and vascular surgery for erectile dysfunction. J Sex Med. 2010;7:501–23.CrossRefPubMed
47.
Kawanishi Y, Kimura K, Nakanishi R, et al. Penile revascularization surgery for arteriogenic erectile dysfunction: the long-term efficacy rate calculated by survival analysis. BJU Int. 2004;94:361–8.CrossRefPubMed
48.
Aung HH, Dey L, Rand V, et al. Alternative therapies for male and female sexual dysfunction. Am J Chin Med. 2004;32:161–73.CrossRefPubMed
49.
Aydin S, Ercan M, Caskurlu T, et al. Acupuncture and hypnotic suggestions in the treatment of non-organic male sexual dysfunction. Scand J Urol Nephrol. 1997;31:271–4.CrossRefPubMed
50.
Sugimori H, Yoshida K, Tanaka T, et al. Relationships between erectile dysfunction, depression, and anxiety in Japanese subjects. J Sex Med. 2005;2:390–6.CrossRefPubMed
51.
Althof SE, Wieder M. Psychotherapy for erectile dysfunction: now more relevant than ever. Endocrine. 2004;23:131–4.CrossRefPubMed
52.
Melnik T, Soares BG, Nasello AG. The effectiveness of psychological interventions for the treatment of erectile dysfunction: systematic review and meta-analysis, including comparisons to sildenafil treatment, intracavernosal injection, and vacuum devices. J Sex Med. 2008;5:2562–74.CrossRefPubMed