Peter D. Lichty, MD, MOH
This chapter discusses health effects of occupational exposure to extreme temperatures (cold and heat), electricity, radiation, atmospheric pressure changes, vibration, and high-pressure injection.
HYPOTHERMIA (COLD INJURY)
Cold injuries are classified as systemic or localized and as freezing (eg, frostbite) or nonfreezing (eg, immersion foot). Factors influencing the risk for these injuries include the atmospheric or water temperature, humidity, wind velocity, duration of exposure, type of protective equipment or clothing, type of work being performed and associated energy expenditure, and age and health status of the worker.
Workers at risk include both indoor and outdoor workers exposed to cold, such as meat packers and others who work inside freezers, construction workers, cold-room personnel, fishermen, woodsmen, divers, mail carriers, firefighters, utility repair workers, search and rescue staff, and road maintenance workers. The risk of hypothermia increases with age and also is increased if the employee is intoxicated with drugs or alcohol; is receiving medications such as barbiturates, antipsychotics, or reserpine; smokes; or has adrenal insufficiency, diabetes, myxedema, neurologic disease affecting hypothalamic or pituitary function or causing peripheral sensory impairment, peripheral vascular disease, or cardiovascular disease causing diminished cardiac output.
1. Systemic Hypothermia
![]() Pathogenesis
Pathogenesis
Systemic hypothermia is reduction of the body’s core temperature below 35°C (95°F). Hypothermia can occur at air temperatures up to 18.3°C (65°F) or in water up to 22.2°C (72°F).
When the body is exposed to cold environments, it has two types of normal physiologic reactions: (1) constriction of superficial blood vessels in the skin and subcutaneous tissue, resulting in heat conservation, and (2) increase in metabolic heat production through voluntary movement and by shivering. In cases of systemic hypothermia, cellular and physiologic functions are diminished. Oxygen consumption is decreased by approximately 7% per degree Celsius, myocardial repolarization is slowed, and ventricular fibrillation is a major hazard.
![]() Clinical Findings
Clinical Findings
The medical history should address the circumstances under which the patient was found, the probable duration of exposure, associated injuries or frostbite, preexisting medical conditions, alcohol or drug use, and recent changes in the level of consciousness. Because body heat is lost more quickly when a person is wet, immersed in water, or exhausted, these factors should be considered.
The onset of hypothermia often is insidious, without any specific characteristics. With profound hypothermia, there is often diminished memory, a decrease in or absence of shivering, and combativeness. Initial findings may include drowsiness, slurred speech, irritability, impaired coordination, general weakness and lethargy, recent diuresis, and puffy and cool skin and face.
Physical examination often reveals diminished neurologic reflexes, slow mental and muscular reactions, weak or nonpalpable pulse, arrhythmia, low blood pressure, and increased blood viscosity. Shivering and peripheral vasoconstriction begin with the core temperature at 35°C (95°F). Heart rate, respiratory rate, and blood pressure decrease with reduced temperature. With mild hypothermia (33–35°C [91.4–95°F]), there is extensive shivering, which decreases as temperature drops below 33°C (91.4°F), wherein joint and muscle stiffness becomes more predominant.
Core temperature should be taken with a thermometer or thermocouple capable of measuring temperatures as low as 28°C (82.4°F), and esophageal or deep rectal measurement (15 cm) is best. The temperature may range from 25–35°C (77–95°F). Below 35°C (95°F), consciousness becomes dulled, causing disorientation, irrational thinking, forgetfulness, and hallucinations. Below 30°C (86°F), semiconsciousness and confusion may occur. Nerve conduction is slowed, although the central nervous system is protected from ischemic damage. The respiratory rate falls to 7–12 breaths per minute, and gastrointestinal motility slows or ceases. There may be hemoconcentration as a result of diuresis and loss of plasma volume. The latter occurs because of subcutaneous edema, which is accompanied by an elevation in corticosteroid levels. Loss of consciousness seldom occurs at temperatures above 28°C (82.4°F).
Evaluation should include a complete blood count; measurement of blood glucose, renal and liver function tests, electrolytes, amylase, and alcohol and drug levels; urinalysis; urine volume; cagulation screen; sputum and blood cultures; thyroid function tests; arterial blood gas measurements with pH corrected for temperature (add 0.0147 pH unit for each degree less than 37°C [98.6°F]); chest radiograph; and electrocardiograph (ECG). There may be evidence of metabolic acidosis, hypovolemia, elevation or depression of the blood glucose level, and renal failure. The ECG may show a pathognomonic J wave at the QRS-ST junction. The level of consciousness may worsen, and death may result from ventricular fibrillation or cardiac arrest.
![]() Prevention
Prevention
The risk of hypothermia is directly related to the wind chill index, which assesses the effects of both ambient temperature and wind velocity. Cold-stress guidelines are based on the wind velocity and temperature and are intended to prevent the core temperature from falling below 36°C (96.8°F) (Table 14–1).
Table 14–1. Work warm-up schedule for outdoor 4-hour period.

The wind chill index (WCI) can be calculated from T and V, where T is the air temperature in Celsius (°C) and V is the wind speed at 10 meters height in kilometers per hour.
![]()
The equivalent chill temperature tch, the temperature equivalent to that under calm wind (1.8 m/s), is given from
![]()
When tch drops below –30°C, exposed flesh may freeze, and tch below –60°C may freeze skin within 1–2 minutes.
Ambient temperature is measured with a dry-bulb thermometer; wind velocity is measured with a standard wind gauge. Work and break schedules should take into account the expected wind velocity and temperature. Under high-risk weather conditions, workers should be under constant protective observation.
The required clothing insulation level (IREQ), duration limited exposure (DLE), and required recovery time (RT) can be calculated by the mathematical equation proposed in ISO/CD 11079 using physical activity, ambient temperature, radiant temperature, wind velocity, etc. Adequate insulating dry clothing to maintain core temperatures above 36°C (96.8°F) must be provided to workers if work is performed in ambient temperatures below 4°C (40°F).
Hypothermia can be prevented by wearing clothing specially designed to resist wind and rain but that also allows water vapor generated by perspiration to escape. Overheating when strenuous work is required in extreme cold can be prevented by wearing a number of thin layers of clothing that can be removed or donned as necessary. Wet garments should be replaced as soon as possible with dry ones, and constrictive garments should not be worn.
Jobs should be designed so that workers remain relatively active when exposed to cold environments and provided with dry, wind-protected, heated shelters for tasks involving stationary work positions. Outdoor workers should have heated rest facilities and hot food and hot drinks available. Workers should be trained to “keep warm, keep moving, and keep dry.”
Workers exposed to the cold should be physically fit, without underlying vascular, metabolic, or neurologic diseases that place them at increased risk for hypothermia. They should be cautioned to avoid smoking and drug or alcohol use. New workers should be introduced into the work schedule slowly and instructed in the use of protective clothing, recognition of impending frostbite and early signs and symptoms of hypothermia, proper warming procedures, and first-aid treatment.
![]() Treatment & Prognosis
Treatment & Prognosis
In cases of mild hypothermia (rectal temperatures >33°C [91.4°F]), patients who are young and otherwise healthy should be treated by rewarming in a warm bed or bath or with warm packs and blankets and with oral rehydration with warmed fluids (caffeine-free and nonalcoholic). Mildly hypothermic elderly or debilitated patients should be treated conservatively, using an electric blanket heated to 37°C (98.6°F). Treatment should increase in aggressiveness with decreasing core temperature, which in severe cases may call for both selected internal and external techniques (Figure 14–1).

![]() Figure 14–1. Hypothermia treatment algorithm. AED = automated external defibrillator; CPR = cardiopulmonary resuscitation; IV = intravenous; J = joules; VF = ventricular fibrillation; VT = ventricular tachycardia.
Figure 14–1. Hypothermia treatment algorithm. AED = automated external defibrillator; CPR = cardiopulmonary resuscitation; IV = intravenous; J = joules; VF = ventricular fibrillation; VT = ventricular tachycardia.
Cardiac rhythm and rate should be monitored. Because the risk of death from ventricular fibrillation is high with severe hypothermia (<32°C [89.6°F]), treatment methods that may trigger fibrillation (eg, central catheters, cannulas, or tubes) should be avoided unless their use is essential. However, patients who are comatose or in respiratory failure should be tracheally intubated. If cardiopulmonary resuscitation (CPR) is instituted, it should be continued until the patient has been rewarmed to at least 36°C (96.8°F). Evaluation for and treatment of localized areas of trauma and frostbite should be undertaken.
Measures should be instituted to correct acid-base deficiencies, normalize the serum potassium and blood glucose levels, increase the blood volume, maintain cardiac output and blood pressure, and provide adequate ventilation. Adequate cardiovascular support, acid-base balance, arterial oxygenation, and intravascular volume should be established as quickly as possible to minimize the risk of organ infarction during rewarming. Oxygen administration should begin prior to rewarming. Because most arrhythmias revert spontaneously to normal sinus rhythm as the patient rewarms, it is usually unnecessary to give antiarrhythmic agents unless there is a preexisting cardiac condition. Ventricular arrhythmias, however, should be treated as they are treated in a euthermic patient. Blood volume expansion with 5% dextrose–normal saline solution is recommended. Potassium-containing expanders should be avoided until the serum potassium levels are stable. If myxedema is an underlying factor, or if drug intoxication is present, appropriate treatment should be given. Localized areas of frostbite should be evaluated and managed as outlined in the section “Hypothermia of the Extremities.”
Use of steroids or antibiotics is not recommended unless otherwise clinically indicated. Core temperature should be monitored frequently during and after initial rewarming because of the potential for delayed, repeat hypothermia.
Active internal rewarming for severe hypothermia is more effective than external rewarming.
A. Active External Rewarming Methods
Although relatively simple and generally available, active external warming methods may cause marked peripheral dilatation that predisposes to ventricular fibrillation and hypovolemic shock. Either heated blankets or warm baths may be used for active external rewarming. Rewarming in a warm bath is most effective and performed in a tub of stirred water at 40–42°C (104–107.6°F), with a rate of rewarming of about 1–2°C (1.8–3.6°F) per hour. It is easier, however, to monitor the patient and to carry out diagnostic and therapeutic procedures when heated blankets are used for active rewarming. Forced-air rewarming (38–43°C [100.4–109.4°F]) is recommended when extracorporeal rewarming is not available; heated blankets are recommended for transport.
B. Active Internal (Core) Rewarming Methods
Internal rewarming is essential for patients with severe hypothermia; extracorporeal blood rewarming (cardiopulmonary, arteriovenous [femorofemoral], or venovenous bypass) is the treatment of choice. If extracorporeal rewarming is not feasible, left-sided thoracotomy followed by pericardial cavity irrigation with warmed saline has been effective in patients with systemic hypothermia of less than 28°C. Repeated peritoneal dialysis may be employed with 2 L of warm (43°C [109.4°F]) potassium-free dialysate solution exchanged at intervals of 10–12 minutes until the core temperature is raised to about 35°C (95°F). Parenteral fluids (5% dextrose in normal saline) should be warmed to 43°C (109°F) before administration. Heated, humidified air warmed to 42°C (107.6°F) should be administered through a facemask or endotracheal tube. Warm colonic and gastrointestinal irrigations are of less value.
Passive rewarming (insulation from cold) is of value only for mildly hypothermic patients or as first-aid management on the scene. Hypothermia victims without vital signs should not be pronounced dead until they have been rewarmed to a core temperature of 36°C (96.8°F) and are found to be unresponsive to continued CPR at that temperature.
Prognosis is directly related to the severity of metabolic acidosis; with low pH (6.6), elevated Paco2 (8.0), and/or elevated potassium (4 mEq/L), the prognosis is poor. The prognosis is good for otherwise healthy patients but worsens with the presence of underlying predisposing problems or a delay in treatment.
2. Hypothermia of the Extremities
The cheeks, nose, earlobes, fingers, toes, hands, and feet are the areas most likely to develop ice crystals within the tissue, resulting in localized hypothermic injury. As skin temperature falls below 25°C (77°F), tissue metabolism slows, although oxygen demand increases if work continues. There may be tissue damage at 15°C (59°F) as a consequence of ischemia and thrombosis and at –3°C (26.6°F) as a consequence of actual freezing of the tissue.
Immersion foot (trench foot) is caused by a combination of cold temperature and exposure to water. This problem and chilblains (pernio) are nonfreezing injuries, whereas frostbite is a freezing injury. Predisposing factors for non-freezing injuries include inadequate clothing and constricting garments. Risk factors for frostbite include prior cold injuries, smoking, Raynaud phenomenon, and collagen-vascular disease.
![]() Clinical Findings
Clinical Findings
A. Chilblains (Pernio)
Chilblains, also called acute pernio, consist of painful, erythematous, pruritic skin lesions caused by inflammation as a result of cold or dampness with cold. With prolonged exposure, this condition can progress to chronic pernio or “blue toes,” characterized by erythematous, edematous, and ulcerating lesions of the acral parts of the toes. Scarring, fibrosis, and atrophy can follow.
B. Immersion Foot
There are three clinical stages of recovery after removal from the cold: an ischemic stage, a hyperemic stage, and a posthyperemic recovery stage. Initially, feet are cold, numb, swollen, and waxy white or cyanotic. Between 2 and 3 days following removal from the cold, hyperemia occurs, along with intense pain, additional swelling, redness, heat, blistering, hemorrhage, lymphangitis, ecchymoses, and in some cases, sequelae such as cellulitis, gangrene, or thrombophlebitis. After 10–30 days, intense paresthesias sometimes occur and are accompanied by cold sensitivity and hyperhidrosis, which may persist for years. Tropical immersion foot occurring at higher temperatures is similar but usually has less intense symptoms with faster recovery.
C. Frostbite
In frostbite, freezing of superficial tissues (skin, subcutaneous) usually causes symptoms of numbness, prickling, and itching; skin is gray-white and hard. In severe cases, there may be paresthesias and stiffness, as well as injury to deeper tissues—bone, muscle, and nerve. Skin is often white and edematous. Deep frostbite may be followed by ulceration, necrosis, or gangrene.
![]() Prevention
Prevention
Keep skin dry and wear moisture-resistant hats, facemasks, earmuffs, scarves, gloves, mittens, socks, and boots. Wet or constrictive socks should be replaced as soon as possible to prevent immersion foot. Pocket hand warmers may be used to warm extremities. Additional prevention guidelines are the same as for systemic hypothermia (see above).
![]() Treatment
Treatment
A. Chilblains (Pernio) and Immersion Foot
Treatment is intended to improve capillary circulation and includes elevating the extremities, gradually rewarming them by exposure to air at room temperature, and protecting pressure sites from trauma. Prazosin hydrochloride, 1 mg at bedtime, has been recommended for treatment and prophylaxis of pernio. Massage, ice, heat, and immersion should be avoided. Antibiotics are given if infection develops.
B. Frostbite
At the site of exposure, extremities can be rewarmed by removing wet gloves, socks, and shoes; drying the extremities and covering them again with dry clothing; and either elevating them or placing them next to a warmer part of the body (eg, placing the hands in the armpits). Caution: Rewarming should not be attempted if refreezing is likely prior to definitive therapy.
In cases of severe frostbite, hospitalization is recommended until the extent of tissue damage has been determined. The patient should be evaluated and treated, if necessary, for systemic hypothermia (see the section “Systemic Hypothermia”).
Rapid rewarming of the frostbitten parts of the body can be accomplished by placing them in a moving water bath heated to 40–42°C (104–107.6°F) and leaving them there until thawing is complete but no longer (often 30 minutes). Dry heat is not recommended, and external heat should be discontinued once normal temperature has been reached. The patient should remain in bed with the affected parts elevated and uncovered at room temperature. Frostbitten parts should not be exercised, rubbed, or exposed to pressure. Dressings and bandages should not be applied. Whirlpool therapy at 37–40°C (98.6–104°F) twice daily for 15–30 minutes for 3 or more weeks helps to cleanse the skin and débrides superficial tissue. A combination of ibuprofen 200 mg four times daily and aloe vera can be used to prevent dermal ischemia.
Infection can be treated with povidone-iodine soaks, water soaks, whirlpool therapy, systemic antibiotics, or a combination of these methods. Tetanus antitoxin or a tetanus toxoid booster may be indicated.
Surgery generally should be avoided and amputation not considered until it is certain that the tissue is dead. Gangrenous and necrotic tissue should be treated by specialists.
Physical therapy can be instituted as healing progresses. The patient should be instructed to avoid exposure to the cold for several months and be advised of future hypersusceptibility to frostbite.
![]() Disorders Caused by HEAT
Disorders Caused by HEAT
Five medical disorders can result from excessive exposure to hot environments (in order of decreasing severity): heat stroke, heat exhaustion, heat cramps, heat syncope, and skin disorders. Among the many types of workers at risk are steel workers, oven and furnace operators, glassblowers, farmers, ranchers, fishermen, and construction workers.
A stable internal body temperature requires maintenance of a balance between heat production and loss, which the hypothalamus regulates by triggering changes in thirst, muscle tone, vascular tone, and sweat gland function. Production and evaporation of sweat are a major mechanism of heat removal (however, sweating causes loss of body water and sodium). The transfer of heat from the skin to surrounding air or liquid (convection) or between two solids in direct contact (conduction) also may occur, but this decreases in efficiency as ambient temperature increases. The passive transfer of heat via infrared rays from a warmer to a cooler object (radiation) accounts for 65% of body heat loss under normal conditions. Radiant heat loss also decreases as temperature increases up to 37.2°C (99°F), at which point heat transfer reverses. At normal temperatures, evaporation accounts for approximately 20% of body heat loss, but at excessive temperatures, it becomes the most important means for heat dissipation. It, too, is limited as humidity increases and is ineffective at 100% relative humidity.
The gradual, controlled exposure to heated environments of increasing intensity and duration (acclimatization) allows the body to adjust to heat by beginning to sweat at lower body temperatures, increasing the quantity of sweat produced, reducing the salt content of sweat, and increasing the plasma volume, cardiac output, and stroke volume while the heart rate decreases.
Health conditions that inhibit sweat production or evaporation and increase susceptibility to heat injury include obesity; skin disease; decreased cutaneous blood flow; dehydration; hypotension; cardiac disease resulting in reduced cardiac output; use of alcohol or medications that inhibit sweating, reduce cutaneous blood flow, or cause dehydration (eg, atropine, antipsychotics, tricyclic antidepressants, diuretics, laxatives, anticholinergics, antihistamines, monoamine oxidase inhibitors, vasoconstrictors, and beta blockers); and use of drugs that increase muscle activity and thereby increase the generation of body heat (eg, phencyclidine [PCP], lysergic acid diethylamide [LSD], amphetamines, cocaine, and lithium carbonate). Infections, cancer, malnutrition, thyroid dysfunction, and other debilitating medical conditions can reduce the effectiveness of the sweating mechanism and circulatory response to heat. Age and sex also affect susceptibility to heat injury. Older people do not acclimatize as easily because of their reduced sweating efficiency, and women generally generate more internal heat than men when performing the same task.
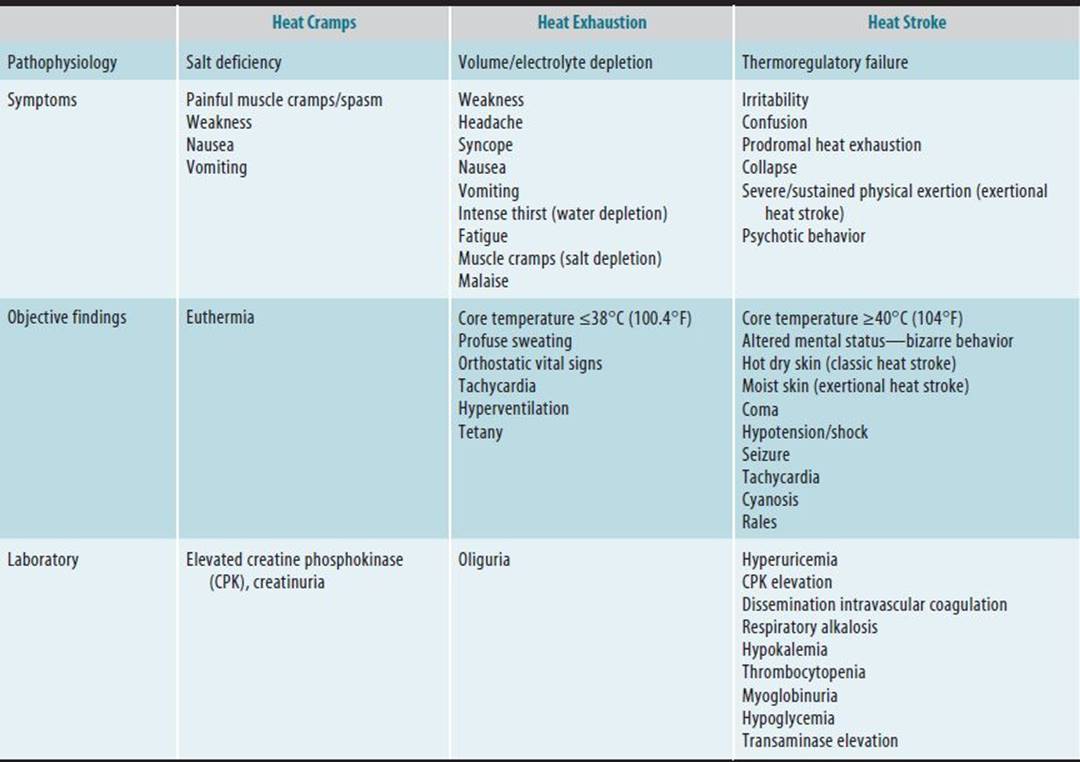
1. Heat Stroke
Heat stroke is a life-threatening medical emergency caused by thermal regulatory failure manifested by cerebral dysfunction with altered mental status, hyperpyrexia, abnormal vital signs, and usually, hot, dry skin. Heat stroke becomes imminent as the core (rectal) temperature approaches 41.1°C (106°F). It is most apt to occur following excessive exposure to heat; it occurs in one of two forms: classic or exertional. The classic form occurs under conditions of extreme heat among those with compromised heat-dissipation capability (elderly individuals, infants, and chronically ill or debilitated patients). Exertional heat stroke results from strenuous exertion in hot environments, often in unacclimatized individuals. Morbidity or mortality can result from cerebral, cardiovascular, hepatic, or renal damage.
![]() Clinical Findings
Clinical Findings
Thermal regulatory failure is characterized by dizziness, weakness, nausea, vomiting, confusion, delirium, and visual disturbances; changes in mental status are its hallmark. Convulsions, collapse, or unconsciousness may occur. The skin is hot and initially covered with perspiration; later it dries. Blood pressure may initially be slightly elevated but later becomes hypotensive. Core temperatures usually exceed 41°C (105.8°F). As with heat exhaustion, hyperventilation can occur and lead to respiratory alkalosis and compensatory metabolic acidosis. There also may be abnormal bleeding, renal failure, or arrhythmias.
Laboratory evaluation may reveal an increase in leukocytes because of dehydration; decreased serum potassium, calcium, and phosphorus levels; increased blood urea nitrogen levels; hemoconcentration; decreased blood coagulation; and concentrated urine with proteinuria, tubular casts, and myoglobinuria. Thrombocytopenia, increased bleeding and clotting times, fibrinolysis, and consumptive coagulopathy may be present. Myocardial, liver, or renal damage may be reflected in laboratory tests (Table 14–2).
Table 14–2. Accidental hyperthermia—clinical differential.
![]() Prevention
Prevention
The American Conference of Governmental Industrial Hygienists (ACGIH) has developed an index of threshold limit values for exposure to heat in occupational settings. The values (wet-bulb globe temperature [WBGT]) are based on a formula (below) that includes the natural wet-bulb temperature Tnwb, the shielded dry-bulb temperature Tdb, and black-globe temperature Tg, which are measurements that account for effects caused by solar radiant heat, air velocity, relative humidity, and ambient temperature. With direct exposure to sunlight:
![]()
Without direct exposure to sunlight:
![]()
Exposure limits take into account the type of work-rest regimen and the workload, including body position, movement, acclimatization, and limb use. These determine the heat load or metabolic rate, which is then related to the index to arrive at a recommended exposure standard for workers in a particular situation. In the absence of WBGT data, heat-index guidelines developed by the National Weather Service predict exposure risks according to ambient temperature and humidity (Figure 14–2). The standards are based on the assumption that workers are acclimatized and physically fit, are wearing appropriate clothing, and are supplied with adequate water and food. If these conditions are not met or the work environment cannot be controlled within the appropriate limits, calculation of the recommended sweat rate provided in International Standards Organization (ISO) 7933 or physiologic measurements of heart rate and core temperature provided in ISO 9886 should be performed by experienced personnel. Occupational heat exposure can be minimized with engineering controls such as air conditioning/cooling, fans, hot-air venting, reflective shielding, and spot cooling. Administrative controls such as limiting exposure duration may be necessary. Special cooled suits have been designed for hot environments.

![]() Figure 14–2. Heat index chart showing associated heat disorders.
Figure 14–2. Heat index chart showing associated heat disorders.
In occupations in which workers are exposed to excessive heat, medical evaluation is recommended to identify individuals at increased risk for heat disorders caused by preexisting medical conditions or use of medications. Exposed workers should be trained to recognize early signs and symptoms of heat disorders and should be advised of the importance of proper attire, nutrition, and fluid intake. Employers should provide cool drinking water or electrolyte-carbohydrate solutions and should ensure that there are shaded rest areas close to the work site. For workers unacclimatized to heat, balanced electrolyte-carbohydrate solutions or 1% saline drinking water should be made available. Salt tablets are not recommended because their use may exacerbate or cause electrolyte imbalance. Organized athletic events should be managed with attention to thermoregulation; the WBGT index should be monitored, water consumption should be encouraged, and medical care should be immediately accessible.
![]() Treatment
Treatment
Treatment is aimed at rapid (within 1 hour) reduction of the core temperature and control of secondary effects. Evaporative cooling provides rapid and effective lowering of temperature and is accomplished easily in most emergency settings. Until medical care becomes available, the patient should be moved to a shady, cool place. Clothing should be removed, and the entire body should be sprayed with cool water (15°C [59°F]); cooled or ambient air should be blown across the patient at high velocity (100 ft/min). The patient should be placed in the lateral recumbent position or supported in the hands-to-knees position to expose more skin surface to the air.
The cooling process should continue in the hospital with use of wet sheets accompanied by fanning. Immersion in an iced-water or cool water bath is effective for rapid cooling down to 39°C (102.2°F) (then stop immersion), but it has a greater potential for complications of hypotension and shivering and may impede other interventions. Other treatment alternatives include ice packs (groin, axilla, and neck) and iced gastric lavage, although these are much less effective than evaporative cooling. Treatment should continue until the core temperature drops to 39°C (102.2°F). Because of the risks of hypoxia and aspiration, intubation should be considered and 100% oxygen administered until the patient is cooled. The core temperature should continue to be monitored, although it usually remains stable after it has returned to normal. Chlorpromazine, 25–50 mg intravenously, or diazepam, 5–10 mg intravenously, can be used to control shivering and thus prevent an increase in heat. Antipyretics are contraindicated (Figure 14–3).

![]() Figure 14–3. Algorithm for the management of heat emergencies. ARDS = acute respiratory distress syndrome; CT = computed tomography; DIC = disseminated intravascular coagulation; PA = pulmonary arterial; PEEP = positive end-expiratory pressure; Po2 = partial pressure of oxygen.
Figure 14–3. Algorithm for the management of heat emergencies. ARDS = acute respiratory distress syndrome; CT = computed tomography; DIC = disseminated intravascular coagulation; PA = pulmonary arterial; PEEP = positive end-expiratory pressure; Po2 = partial pressure of oxygen.
Patients should be monitored for hypovolemic and cardiogenic shock, either or both of which may occur. Attention should be paid to maintaining a patent airway, providing oxygen, correcting fluid and electrolyte imbalances, and supporting vital processes. Central venous or pulmonary artery wedge pressure should be assessed and intravenous fluids administered if indicated. If hypovolemic shock is suspected, 500–1000 mL of 5% dextrose in normal or half-normal saline solution may be given intravenously without overloading the circulation. Other medications appropriate for cardiovascular support should be considered.
Fluid output should be monitored with an indwelling urinary catheter, and fluid administration should maintain a urine output of more than 50 mL/h. The patient should be monitored for complications, including renal failure (caused by dehydration and rhabdomyolysis), hepatic failure, or cardiac failure, respiratory distress, hypotension, electrolyte imbalance (hypokalemia), and coagulopathy. Elevated creatine phosphokinase (CPK), elevated liver enzymes, and metabolic acidosis are predictors of multiorgan dysfunction.
Because hypersensitivity to heat continues in some patients for prolonged periods following heat stroke, they should be advised to avoid reexposure to heat for at least 4 weeks.
2. Heat Exhaustion
In individuals performing strenuous work, prolonged exposure to heat and insufficient salt and water intake can cause heat exhaustion, dehydration, and sodium depletion or isotonic fluid loss with accompanying cardiovascular changes. Symptoms and signs may include intense thirst, weakness, nausea, fatigue, headache, confusion, a core (rectal) temperature exceeding 38°C (100.4°F), increased pulse rate, and moist skin. Symptoms associated with both heat syncope and heat cramps (see below) also may be present. Hyperventilation sometimes occurs secondary to heat exhaustion and can lead to respiratory alkalosis. Progression to heat stroke is indicated by a rise in temperature or a decrease in sweating.
Treatment consists of placing the patient in a cool and shaded environment and providing hydration (1–2 L over 2–4 hours) and salt replenishment—orally if the patient is able to swallow. Physiologic saline or isotonic glucose solution should be administered intravenously in more severe cases. At least 24 hours’ rest is recommended.
3. Heat Cramps
Heat cramps result from dilutional hyponatremia caused by replacement of sweat losses with water alone. They are usually characterized by slow and painful muscle contractions and severe muscle spasms that last from 1 to 3 minutes and involve the muscles employed in strenuous work.
The skin is moist and cool, and the involved muscle groups feel like hard, stony lumps similar to billiard balls. The temperature may be normal or slightly increased, and blood tests may show low sodium levels and hemoconcentration. Because the thirst mechanism is intact, blood volume is not diminished significantly.
The patient should be moved to a cool environment and given a balanced salt solution or an oral saline solution consisting of 4 tsp of salt per gallon of water. Salt tablets are not recommended. Rest for 1–3 days with continued salt supplementation in the diet may be necessary before returning to work.
4. Heat Syncope
In heat syncope, sudden unconsciousness results from volume depletion and cutaneous vasodilatation with consequent systemic and cerebral hypotension. Episodes occur commonly following strenuous work for at least 2 hours.
The skin is cool and moist and the pulse weak. Systolic blood pressure is usually below 100 mm Hg. Treatment consists of recumbency, cooling, and rehydration. Preexisting medical conditions should be monitored and treated if necessary.
5. Skin Disorders Caused by Heat
Miliaria (heat rash) is caused by sweat retention resulting from obstruction of the sweat gland duct. There are three forms (listed here in increasing order of severity): miliaria crystallina, miliaria rubra, and miliaria profunda. As the site of duct obstruction becomes deeper in the skin, the severity increases and presentation varies (eg, vesicles, erythema, desquamation, macules).
Erythema ab igne (“from fire”) is characterized by the appearance of hyperkeratotic nodules following direct contact with heat that is insufficient to cause a burn. Intertrigo results from excessive sweating and often is seen in obese individuals. Skin in the body folds (eg, the groin and axillas) is erythematous and macerated. Heat urticaria (cholinergic urticaria) can be localized or generalized and is characterized by the presence of wheals with surrounding erythema (“hives”).
Treatment for these disorders consists of reduction or removal of heat exposure, reduction of sweating, and control of symptoms. Antihistamines may help to relieve pruritus in patients with urticaria. Corticosteroids are not beneficial.
ELECTRICAL INJURIES
Electrical accidents comprise up to 4% of all fatal industrial accidents. Electrical power-line installers and repairmen, electricians, operators of high-power electric equipment, and maintenance personnel are at greatest risk for electric shock.
Physical contact with an energized electric circuit provides a pathway for electricity to traverse the body as it seeks a ground. Conductivity to the body is affected by skin moisture, as well as moisture on contacting surfaces (eg, floors). Factors influencing the severity of electrical injury include the voltage (electrical force), amperage (current intensity), current type (alternating or direct current), duration of contact, area of contact, pathway of the current through the body, and amount of tissue resistance.
Electricity from alternating currents is more dangerous than that from direct currents. The alternating currents usually cause muscle tetanization and sweating, whereas the direct currents cause electrolytic changes in tissue. Most tissue damage is related to the heat produced by the electric current, and tissue resistance is largely influenced by the water content of the tissue. The vascular system and muscles are good conductors of electricity, whereas the bones, peripheral nerves, and dry skin have higher resistance.
A sudden exposure to intense electrical energy can cause not only tissue destruction and necrosis from heat and burning but also depolarization of electrically sensitive tissues such as nerve and heart. Alternating currents with voltages and frequencies as low as domestic circuits (100 V and 60 Hz) can produce ventricular fibrillation. High voltages (>1000 V) can cause respiratory paralysis. Most shocks involving currents exceeding 10,000 V are of such magnitude that the electrical force knocks the victim away from the power source, which reduces the electrical injury potential but often causes blunt trauma.
A tetanizing effect of voluntary muscles is greatest at frequencies between 15 and 150 Hz. Sustained grasp of the conductor does not usually occur at high voltages because the circuit probably arcs before contact with the victim, who is thrown back instead. Current above 20 mA can cause sustained contraction of chest respiratory muscles; alternating currents above 30–40 mA can induce ventricular fibrillation, whereas direct current is more likely to cause asystole. Lightning injuries differ from high-voltage electric shock injuries in that lightning usually involves higher voltage, briefer duration of contact, asystole rather than ventricular fibrillation, nervous system injury, a shock-wave characteristic, and multisystem pathologic involvement.
![]() Clinical Findings
Clinical Findings
Exposure to electric current can cause shock, flash burns, flame burns, or direct tissue necrosis. Surface wounds covering heat-induced tissue necrosis are usually round or oval and well demarcated, and they may have a relatively innocuous yellow-brown appearance. A search must be made for both the entry and exit wounds to determine the electrical pathway through the body. Depending on the contact site and the pathway, there may be damage to nerves, muscles, or major organs such as the heart, brain, eye, kidney, and gastrointestinal tract.
In all cases, an ECG with a rhythm strip and a urine dipstick for blood and protein should be obtained, and the respiratory rhythm and rate should be checked. If organ, muscle, or nerve damage is suspected, appropriate diagnostic tests should be ordered such as urine myoglobin; CPK should be monitored for at least 24 hours if muscle symptoms occur or muscle injury is otherwise suspected. With muscle injury, the CPK level can be elevated significantly (>1000 U/L) but the MB fraction will be below 3% if there is no cardiac muscle injury. Occult fractures may occur following muscle tetany or blunt trauma. Patients should be observed for several days because some develop posttraumatic myositis with rhabdomyolysis.
Electrical injury causes increased vascular permeability, which may result in reduced intravascular volume and fluid extravasation in the area of internal injury. Hematocrit, plasma volume, and urine output should be monitored closely.
Acute-and delayed-onset central and peripheral nervous system complications are the most common sequelae of electrical injury. Cardiac complications usually consist of rhythm and conduction abnormalities, with rare infarction. Sepsis and psychiatric complications also occur.
![]() Prevention
Prevention
Electrical injuries can be prevented in industrial settings by making sure that electrical workers are properly qualified and trained to follow safety procedures involving the installation, grounding, and disconnection of power sources. Locking out and tagging out electrical shutoff switches along with verifying the lack of voltage are effective work practices for preventing electrical injuries. Particular attention should be given to work requiring equipment manipulation during “live” operation. Nonconducting tools and clothing should be used whenever possible. Barricades and warning signs should be placed around high-voltage areas, and procedures to exclude other employees from these areas should be strictly enforced.
Workers should be instructed in the proper measures to free a victim from contact with electric current. If possible, the power should be turned off. If not, a nonconducting object such as a rope, a broom or other wooden instrument, or an article of clothing can be used to pull the victim away from the current and protect the rescuer from injury.
![]() Treatment
Treatment
Prior to CPR, first aid, or treatment, the patient must be separated from the “live” electric current. Power should be turned off and/or nonconductive devices should be used to separate the rescuer and patient from the current. The rescuer must be protected during this procedure. If necessary, CPR (including automated external defibrillator [AED] use) should be instituted until medical help arrives. Because the victim may have suffered spinal injury, extreme care must be taken during handling or transport.
If major electrical injuries are suspected, the patient should be hospitalized and observed for secondary organ damage, impaired renal function, hemorrhage, acidosis, and myoglobinuria. Indications for hospitalization include significant arrhythmia or ECG changes, large burns, loss of consciousness, neurologic findings, pulmonary or cardiac symptoms, or evidence of significant deep-tissue/organ damage. A tetanus booster or antitoxin should be administered if indicated.
Superficial tissue damage and burns should be addressed. If major soft-tissue damage is suspected, surgical exploration, fasciotomy, or both must be considered. Gross myoglobinuria may indicate the need for fasciotomy and/or amputation.
Lactated Ringer solution should be administered intravenously at a rate sufficient to maintain urine output at between 50 and 100 mL/h. Continuous monitoring and prompt correction of acid-base or electrolyte imbalance are necessary if rhabdomyolysis occurs.
NONIONIZING RADIATION INJURIES
1. Injuries Caused by Radiofrequency & Microwave Radiation
![]() Exposure
Exposure
Injuries can be caused by the thermal effects of acute exposure to high levels of radiofrequency (RF) and microwave. As with other thermal injuries, these injuries are characterized by protein denaturation and tissue necrosis at the site of tissue heating, with an accompanying inflammatory reaction and subsequent scar formation. Nonthermal effects of low-level exposure have been demonstrated in some laboratory studies, but their significance in humans is not clear.
RF radiation and microwave radiation consist of energy in wave form traveling in free space at the speed of light. The radiation is defined in terms of frequency and intensity, with the frequency portion of the electromagnetic spectrum extending from 0–1000 GHz (1 Hz equals 1 wave or cycle per second [cps]). Microwaves occupy only the portion of this frequency spectrum between 300 MHz and 300 GHz (Figure 14–4).

![]() Figure 14–4. The electromagnetic radiation spectrum. GHz = gigahertz; IR = infrared radiation; kHz = kilohertz; MHz = megahertz; THz = terahertz; UV = ultraviolet light; VR = visible radiation (light).
Figure 14–4. The electromagnetic radiation spectrum. GHz = gigahertz; IR = infrared radiation; kHz = kilohertz; MHz = megahertz; THz = terahertz; UV = ultraviolet light; VR = visible radiation (light).
RF radiation has insufficient energy to cause molecular ionization, but it does cause vibration and rotation of molecules, particularly molecules that have an asymmetric charge distribution or are polar in structure. The radiation is composed of separate electric and magnetic field vectors, each perpendicular to the other and both perpendicular to the direction of the resulting electromagnetic wave (Figure 14–5). The electric field component is measured in volts per meter, the magnetic component in amperes per meter, and the resulting power density in watts per square meter.

![]() Figure 14–5. Electric field (E) and magnetic field (H) components of radiofrequency radiation.
Figure 14–5. Electric field (E) and magnetic field (H) components of radiofrequency radiation.
Absorption of RF radiation depends partly upon the orientation of the body in relation to the direction of the electromagnetic wave. Radiation at frequencies below 15 MHz and above 25 GHz are poorly absorbed by human tissues and unlikely to cause significant thermal damage. Factors affecting conduction of RF radiation within the body include the thickness, distribution, and water content of the various tissues. As the water content increases, energy absorption and thermal effects increase. RF radiation can be modulated according to amplitude (AM) and frequency (FM) and can be generated in pulsed or continuous form. Pulsed waves are considered more dangerous.
The risk of thermal injury increases with higher intensities of radiation and closer proximity to the radiation source. Other factors that affect human susceptibility to RF radiation injury include environmental humidity and temperature, grounding, reflecting medium, tissue vascularity, increased temperature sensitivity of tissues (eg, the testes), and lack of anatomic barriers to external radiation (eg, the eye).
Occupational exposures are likely in any workplace where employees are near equipment that generates RF radiation, particularly equipment for dielectric heating (used in sealing of plastics and drying of wood), physiotherapy, radio communications, and maintenance of antennae and high-power electrical equipment (Table 14–3). Injuries have been documented for acute exposure to energy levels exceeding 10 mW/cm2. In most cases, the levels were greater than 100 mW/cm2. Most studies of RF radiation effects in animals and other biologic test systems have not demonstrated thermally induced effects at energy levels below 10 mW/cm2. In animal studies, thermal effects include superficial and deep tissue destruction, cataract, and testicular damage.
Table 14–3. Occupational radiofrequency and microwave exposures.
Sealing and heating equipment
Automotive trades
Furniture and woodworking
Glass-fiber production
Paper production
Plastics manufacturing and fabrication
Rubber product heating
Textile manufacturing
Electrical equipment maintenance
Radar
Radio: AM, FM, CB
Television: UHF and VHF
Satellite
Radio navigation
Microwave generators and heat sources
RF applications
Microwave tube testing and aging
RF laser
RF welding
Medical diathermy and healing promotion
Power transmission line workers
Generally, acute high-level or long-term low-level exposures are not thought to cause cancer, but there is evidence for carcinogenesis in association with exposure to extremely low-frequency (ELF) radiation magnetic fields (<200 Hz). Current data indicate that ELF radiation causes childhood leukemia; accordingly, the International Agency for Research on Cancer has classified ELF radiation as a category 2B carcinogen (“possibly carcinogenic to humans”). Although some studies have found an increased incidence of brain tumors, breast cancer (male), or leukemia in workers exposed to ELF radiation, most recent data regarding adult and occupational ELF radiation exposure do not indicate carcinogenic effects at any RF frequency. Similarly, teratogenicity has been questioned on the basis of findings of chromosomal changes in workers and an increase in the incidence of anomalies found in offspring of male physical therapists. However, because of conflicting study results, confounding exposures, and a lack of a verified biologic mechanism, the hypotheses of magnetic field–induced reproductive effects or teratogenicity have not been verified.
Although a number of health effects have been attributed to nonthermal and chronic RF exposures (including microwave and ELF radiation), current data do not support an association with cardiovascular, neurologic, or reproductive outcomes, with the possible exception of amyotrophic lateral sclerosis.
Literature is emerging regarding possible health effects associated with use of cell phones or mobile phones. Research in this area is hampered by the rapid expansion of cell phone usage across the world, which generates confounding with all simultaneous health trends. In addition, exposures have changed over time with the introduction of new electronic communication technologies. Most of the recent authoritative reviews indicate that data are inadequate to draw any conclusions about health effects in relation to the use of cell phones.
![]() Clinical Findings
Clinical Findings
Acute high-dose exposure usually is associated with a feeling of warmth on the exposed body part, followed by the feeling of hot or burning skin. The sensation of clicking or buzzing also may be present during the exposure. Other symptoms include irritability, headache or light-headedness, vertigo, pain at the site of exposure, watery eyes and a gritty eye sensation, dysphagia, anorexia, abdominal cramps, and nausea. Localized thermally induced masses may appear within days of exposure and consist of interstitial edema and coagulation necrosis.
The exposed skin has a sunburned appearance, with erythema and slight induration. There may be vesiculations or bullae. Blood pressure may be increased, and CPK levels may be elevated. Hematologic values, electroencephalographic and brain scan findings, sedimentation rate, and electrolyte values usually are within normal limits.
Beyond the immediately evident thermal injury, no further structural injury would be anticipated. Symptoms of posttraumatic stress disorder have occurred, with emotional lability and insomnia persisting for as long as 1 year.
![]() Differential Diagnosis
Differential Diagnosis
High-power equipment capable of generating RF and microwave radiation also might generate other forms of non-ionizing radiation that should be considered. Chemical reactions caused by heat sources in the workplace should be investigated because thermal decomposition products of a heated hydrocarbon can cause the acute onset of similar symptoms, although blood pressure and CPK levels would not be expected to rise in such circumstances. Fear and anxiety resulting from the knowledge of a possibly damaging exposure also may cause many of the functional symptoms described earlier, although objective evidence of thermal injury and elevated CPK levels would not be expected.
![]() Prevention
Prevention
Exposure assessment should include the following factors: the distance between the power source and exposed workers, the peak power density at the time of exposure, the frequency and type of radiation wave (pulsed or continuous), and the duration of exposure (in minutes). Metal barriers around the energy source can be used to contain RF radiation. Intensity is proportional to 1/d2, where d is distance from the source. Accordingly, there is a rapid decrease in power density over distance, and specification of a “Personnel not allowed” area can provide an effective barrier. Procedures to deenergize equipment are recommended when employees are working close to exposed sources. Protective clothing generally is ineffective. Periodic environmental RF radiation measurements for equipment exposures are essential.
![]() Treatment & Prognosis
Treatment & Prognosis
Treatment is the same as for other thermal injuries. Thermal injuries usually heal without problems. If hypertension develops, it usually will resolve following a short course of antihypertensive therapy. Posttraumatic stress disorder and other psychological sequelae generally are responsive to short-term therapy.
2. Injuries Caused by Infrared Radiation
Infrared (IR) radiation covers the portion of the electromagnetic spectrum between visible and RF radiation (see Figure 14–4). It has wavelengths between 750 and 3 × 106 nm and is composed of three spectral bands—A, B, and C—that begin at 750, 1400, and 3000 nm, respectively. IR radiation is given off by any object having a temperature greater than absolute zero. Occupational exposures—in addition to sunlight—include processes in which thermal energy from IR radiation is used, such as heating and dehydrating processes, welding, glassmaking, and the drying and baking of coatings on consumer products. In addition, workers near molten metals and glass are exposed to high levels of infrared radiation. In recent years, the use of infrared lasers, such as neodymium-doped yttrium aluminum garnet (nd:YAG) has increased the potential for accidental high-power exposure to infrared radiation.
Acute, high-intensity exposure to wavelengths shorter than 2000 nm can cause thermal damage to the cornea, iris, or lens. Thermal injury to the skin can also occur, but it is usually self-limited by withdrawal from the pain and results in an acute skin burn with increased pigmentation. Injuries can be prevented by shielding heat sources, using protective eye and skin wear, and monitoring exposure levels. Threshold limit values for exposure intensity are frequency-dependent in the biologically active wavelength spectrum of 750–2000 nm. Wavelengths in this range cause molecular excitation and vibration, resulting in heat that is absorbed by tissues and can cause thermal injury. In contrast, wavelengths exceeding 2000 nm are absorbed by water and are not biologically active because of the high water content of tissues.
3. Injuries Caused by Visible Radiation
Visible radiation (light) covers the portion of the electromagnetic spectrum between infrared and ultraviolet radiation (see Figure 14–4) and the wavelengths between 400 and 750 nm. The eye is the most sensitive target organ, with damage resulting from structural, thermal, or photochemical light-induced reactions. Workers at risk are those with prolonged or repeated exposure to intense light sources, including sunlight, high-intensity lamps, lasers, flashbulbs, spotlights, and welding arcs. Extremely intense light sources such as lasers also can cause pressure-induced (mechanical) retinal damage.
The retina is the usual site of injury and is most sensitive to the wavelengths of 440–500 nm (blue light), which cause a destructive photochemical reaction. Blue light is responsible for solar retinitis (eclipse blindness) and may contribute to retinal aging and to senile macular degeneration, which can result in visual field defects. Because the lens normally filters out wavelengths between 320 and 500 nm, it provides some protection of the retina from blue light. Individuals with aphakia (absence of the lens), who are more susceptible to retinal damage, should be cautioned against looking into the midday sun and other intense light sources and should be urged to wear spectacle filters when working in bright environments. Short bursts of high-intensity light can cause flash blindness in which the temporary visual loss and afterimage are a result of bleaching of visual pigments. As the light intensity and exposure duration increase, the afterimage persists longer. With mild to moderate exposures, symptoms of flash blindness resolve quickly.
Insufficient lighting or reflected light (glare) can cause asthenopia (eyestrain), visual fatigue, headache, and eye irritation. These problems are more likely to occur in people older than age 40. Symptoms are transient, and there is no indication that repeated episodes lead to ocular damage.
Contrast from surrounding light sources on areas of lesser intensity has led to complaints of asthenopia associated with video-display terminal use. This usually can be corrected by decreasing surrounding light intensities, using antiglare filters, and adjusting the contrast of the characters on the screen.
Measures to prevent injury in workers at risk include preplacement evaluations for individuals with aphakia or a history of light sensitivity and medical surveillance to detect changes in visual acuity or early signs of ocular damage, use of goggles or face shields by welders, proper illumination of the workplace to reduce glare (see Chapter 12), and use of filters on intense light sources to eliminate blue-light wavelengths.
4. Injuries Caused by Ultraviolet Radiation
Ultraviolet (UV) radiation covers the portion of the electromagnetic spectrum between visible radiation and ionizing radiation (see Figure 14–4) and has wavelengths between 100 and 400 nm. The wavelengths are divided into three spectral bands—A, B, and C—with the A and B bands representing the longer wavelengths and producing the greatest biologic effects (Table 14–4). Wavelengths shorter than 200 nm are absorbed over extremely short distances in air, preventing health effects. Wavelengths of 200–290 nm are absorbed primarily in the stratum corneum of the skin or the cornea of the eye, whereas the longer wavelengths can affect the dermis, lens, iris, or retina.
Table 14–4. Ultraviolet light spectrum: ultraviolet A (UVA) and ultraviolet B (UVB) comparison.

Because UV radiation has relatively poor penetration, the only organs it affects are the eye and skin. Eye injury is caused by thermal action from pulsed or brief high-power exposures, and skin damage is caused more commonly by photochemical reactions (including toxic and hypersensitivity reactions) from brief high- or extended low-power exposures. The thermal effects of protein coagulation and tissue necrosis are rapid in onset. The effects of chronic exposure include accelerated aging of the skin, characterized by loss of elasticity, hyperpigmentation, wrinkling, and telangiectasia.
UV injuries occur in occupations involving drying and curing processes, arc welding, or use of lasers or germicidal UV lights (Table 14–5), but by far the greatest proportion of injuries result from occupations that expose workers to natural sunlight during the peak time of UV energy dissemination, 10 AM to 3 PM. Factors affecting the severity of injury include exposure duration, radiation intensity, distance from the radiation source, altitude, and orientation of the exposed individual relative to the source and its wave-propagation plane. UV reflections from water and snow or their surrounding surfaces may increase exposure intensity.
Table 14–5. Workers potentially exposed to ultraviolet radiation.

![]() Clinical Findings & Treatment
Clinical Findings & Treatment
A. Photokeratoconjunctivitis (Welder’s Flash)
Ocular exposure to UV wavelengths shorter than 315 nm (especially wavelengths of 270 nm, to which the eye is most sensitive) can cause photokeratoconjunctivitis. Symptoms occur 6–12 hours after exposure and include severe pain, photophobia, a sensation of a foreign body or sand in the eyes, and tearing. After a latency period that varies inversely with the severity of the exposure, conjunctivitis appears, sometimes accompanied by erythema and swelling of the eyelids and facial skin. Fluorescein examination may reveal diffuse punctate staining of both corneas.
Treatment consists of providing symptomatic relief, which may include ice packs, systemic analgesics, eye patches, and mild sedation. Local anesthetics should not be used because of the risk of further injury to the anesthetized eye. Symptoms usually resolve within 48 hours. Permanent sequelae are rare, and the eye does not develop tolerance to repeated exposure.
B. Cataracts
Cataractogenesis (cortical) is attributed to both photochemical and thermal effects of intense exposure to UV wavelengths of 295–320 nm and usually appears within 24 hours. Cataract formation following repeated exposures to UV wavelengths longer than 324 nm has been reported but is not well documented. Treatment is by corrective surgery. Intraocular lenses implanted after cataract removal often contain UV filters to protect the retina.
C. Other Eye Injuries
The cornea and lens protect the retina from the effects of UV wavelengths shorter than 300 nm, but damage to the iris and retina is possible if individuals with aphakia are exposed to these wavelengths. In others, damage is possible with exposure to longer wavelengths or to high-power UV lasers. Treatment is supportive (Table 14–6). Two lesions of the bulbar conjunctiva have been associated with repeated exposures to UV radiation: pterygium (a benign hyperplasia) and epidermoid carcinoma.
Table 14–6. Eye injuries caused by ultraviolet light.

D. Erythema
Absorbed UV radiation reacts with photoactive substances present in the skin and 2–24 hours later causes erythema (sunburn), the most common acute UV effect. Erythema is most severe following exposure to wavelengths of 290–320 nm and may be accompanied by edema, blistering, desquamation, chills, fever, nausea, and rarely, circulatory collapse.
Treatment of acute sunburn and any blistering that occurs is supportive and symptomatic and may include topical and mild systemic analgesics. Most symptoms subside within 48 hours. The resulting scaling, darkening of the skin (caused by increased melanin production), and thickening of the stratum corneum provide increased protection against subsequent exposures.
E. Photosensitivity Reactions
Two types of acute photosensitivity reactions of the skin can occur following exposure to UV radiation: phototoxic (nonallergic) and photoallergic reactions. Phototoxic reactions are much more common and frequently occur in association with use of medications such as griseofulvin, tetracycline, sulfonamides, thiazides, and preparations containing coal tar or psoralens. Phototoxicity may exaggerate or aggravate the effects of some systemic diseases, including lupus erythematosus, dermatomyositis, congenital erythropoietic porphyria, porphyria cutanea tarda symptomatica, pellagra, actinic reticuloid, herpes simplex, and pemphigus foliaceus. Photosensitivity reactions may be characterized by blisters, bullae, and other skin manifestations.
Exposure to UV wavelengths above 320 nm after skin contact with furocoumarin-producing plants such as celery can cause phytophotodermatitis. A mild phototoxic reaction causes pigmentary changes along the pattern of points of contact, whereas bullae may result from a more severe inflammatory reaction. Photoallergic reactions to UV radiation occur in association with bacteriostatic agents and perfume ingredients, which cause skin irritation, erythema, and blistering. Treatment of photosensitivity reactions depends on the particular underlying or associative cause and ranges from symptomatic care in mild cases to hospitalization and use of systemic corticosteroids in cases of severe reactions.
F. Premalignant and Malignant Skin Lesions
Premalignant lesions associated with chronic exposure to UV radiation include actinic keratosis, keratoacanthoma, and Hutchinson melanosis. Malignant lesions associated with exposure are basal cell carcinoma (the most common), squamous cell carcinoma, and malignant melanoma. Hazardous UV wavelengths are thought to be between 256 and 320 nm. UV radiation also promotes carcinogenesis following exposure to some chemicals, including those found in tar and pitch. Increased risk for premalignant and malignant lesions occurs in fair-skinned individuals and in those who have repeated sunburns or tan poorly. Patients with a history of xeroderma pigmentosum are at greater risk for malignant melanoma.
Patients should be referred to a dermatologist for definitive diagnosis and treatment. Premalignant lesions may be treated by removal or use of topical medication. Treatment of malignant lesions may involve simple excision, radiation, or major surgery.
![]() Prevention
Prevention
Exposure guidelines are based on wavelength and irradiance. Exposed individuals should be counseled concerning photosensitizing agents. Welders should be required to wear goggles or face shields to protect their eyes. Outdoor workers should be instructed to use sunscreen (broad spectrum with skin protective factor [SPF] 30) and protective clothing, and persons at increased risk because of preexisting medical conditions or excessive exposure should be examined periodically for the presence of premalignant or malignant lesions.
Ionizing Radiation Injuries
The two most significant health responses to ionizing radiation are the acute radiation syndrome that follows a brief but massive exposure and the chronic effects that are caused by a brief high-dose exposure or to excessive cumulative exposures. More than 200 significant radiation incidents have occurred since 1940 as a result of exposure to radioisotopes, x-ray generators and accelerators, radar generators, and similar sources of ionizing radiation. Because of the ubiquity of ionizing radiation in our environment, the effects of long-term incremental low-dose exposures are difficult to pinpoint. Workers at risk, based on their history of exposures and resulting injury, include radiologists, uranium miners, radium dial painters, nuclear power plant operators, and military personnel. Table 14–7 lists other workers at risk based on the potential for exposure.
Table 14–7. Potential occupational exposures to ionizing radiation.
Aircraft workers
Atomic energy plant workers
Biologists
Cathode ray tube makers
Chemists
Dental workers
Drug makers and sterilizers
Electron microscope makers and operators
Electrostatic eliminator operators
Embalmers
Fire alarm makers
Food preservers and sterilizers
Gas mantle makers
High-voltage electron, x-ray, vacuum, radar, klystron or television tube makers, users, repairmen
Industrial radiographers and fluoroscopists
Inspectors using—and workers in proximity to—sealed gamma ray sources (cesium 137, cobalt 60, iridium 192) and x-rays
Liquid-level gauge painters
Luminous dial painters
Military personnel
Oil well loggers
Ore assayers
Petroleum refinery workers
Physicians and nurses
Plasma torch operators
Plastics technicians
Prospectors
Radium refinery workers
Research workers, chemists, biologists, physicists
Thickness gauge operators
Thorium ore and alloy workers
Tile glazers
Uranium workers and miners
Veterinarians
X-ray aides and technicians
X-ray diffraction apparatus operators
Ionizing radiation is emitted from radioactive atomic structures as energized particles (alpha, beta, proton, and neutron particles) that impart energy through collision with other structures or as high-energy electromagnetic x-rays or gamma rays. The different forms of ionizing radiation vary in natural source, energy, frequency, and penetrability, but they all share the ability to ionize incident materials and exist at the highest energies and frequencies of the electromagnetic spectrum (see Figure 14–4). Dislocation of an electron from an incident atom and the resulting biomolecular chemical reactions and instability can cause tissue damage. Table 14–8 summarizes the clinical effects of ionizing radiation.
Table 14–8. Summary of clinical effects of ionizing radiation dosages.

External biologic exposure to x-rays, gamma rays, and proton and neutron radiation results in high absorption, whereas beta particles penetrate skin poorly and alpha particles do not penetrate at all. Internal exposure to alpha or beta particles by radioisotope inhalation, implantation, or ingestion can result in serious acute or delayed injury. If radioactive contamination is suspected, contamination control procedures should be followed scrupulously during all phases of patient management.
As an emergency resource, the Oak Ridge Institute for Science and Education maintains 24-hour phone access to consultation regarding medical and health physics problems associated with radiation accidents (865-576-1005).
1. Acute Radiation Syndrome
Acute radiation syndrome is a consequence of brief but heavy exposure of all or part of the body to ionizing radiation. The radiation disrupts chemical bonds, which causes molecular excitation and free-radical formation. Highly reactive free radicals react with other essential molecules such as nucleic acids and enzymes; in turn, this disrupts cellular function. The clinical presentation and severity of illness are determined by the dosage, body part(s) exposed, and duration of exposure. Tissues with the most rapid cellular turnover are the most radiosensitive: reproductive, hematopoietic, and gastrointestinal tissues.
![]() Clinical Findings
Clinical Findings
Although symptoms are unlikely with exposure to doses less than 100 cGy, abnormal laboratory findings may be seen at any dose over 25 cGy. For doses of 100–400 cGy, symptoms begin within 2–6 hours and may last up to 48 hours. For doses of 600–1000 cGy, symptoms begin within 2 hours and later merge into the illness phase. For those at Chernobyl who received more than 600 cGy, headache, fever, and vomiting developed within the first half hour. Within 6 days, severe lymphopenia developed, followed by severe gastroenteritis, granulocytopenia, and thrombocytopenia. For those in the lowest exposure group (80–210 cGy), slight lymphopenia occurred within a few days, followed by mild granulocytopenia and thrombocytopenia at 4 weeks.
Doses of 1000–3000 cGy can cause immediate gastrointestinal symptoms and massive fluid, blood, and electrolyte loss resulting from denudation of the gastrointestinal mucosa. Doses exceeding 3000 cGy are lethal. They cause progressive neurologic incapacitation associated with ataxia, lethargy, tremor, and convulsions. Death is almost immediate with the highest doses.
Some patients with acute radiation syndrome pass through four phases: prodrome, latent phase, illness, and recovery.
A. Prodrome
Symptoms and signs may include anorexia, nausea, vomiting, diarrhea, intestinal cramps, salivation, dehydration, fatigue, apathy, prostration, arrhythmia, fever, respiratory distress, hyperexcitability, ataxia, headache, and hypotension. Gastrointestinal and central nervous system findings predominate.
B. Latent Phase
The prodrome is sometimes followed by a period of relative well-being prior to the onset of illness. In cases of exposure to higher doses of radiation, the latent period is shortened or eliminated, and central nervous system or gastrointestinal effects predominate.
C. Illness Phase
Symptoms and signs in this phase may include fatigue, weakness, fever, diarrhea, anorexia, weight loss, hair loss, arrhythmia, ileus, ataxia, disorientation, convulsions, coma, and shock. Effects are primarily hematopoietic and a result of inhibition of hematopoietic stem cells. There may be a sequential decrease in lymphocytes, granulocytes, platelets, and erythrocytes. Leukopenia and thrombocytopenia may occur with secondary infection, hemorrhagic diathesis, or anemia. Cardiovascular collapse, pericarditis, and myocarditis have been reported. With doses exceeding 200 cGy, there may be reproductive system effects, including sterility, aspermatogenesis, and cessation of menses. Fetal and embryo toxicity or death also can occur.
D. Recovery Phase
The prognosis for recovery from exposures of up to 600 cGy is good when appropriate therapy is given. For higher exposures, the prognosis worsens as the dose increases. Infection and sepsis are the major causes of morbidity and mortality in cases involving exposures below 1000 cGy, in which the major impact is hematopoietic.
Prevention
Occupational exposure to ionizing radiation should be monitored. The technology varies with the type of radiation and the target site. Personal exposure measurement devices include film badges (x-rays, gamma, and beta) or nuclear emulsion monitors (x-rays, gamma, beta, and neutrons), thermoluminescent dosimeters (beta, gamma, and neutron), and ionization dosimeters. A scintillation counter can be used to measure some radioisotopes in urine specimens or in tissue from target organs (eg, urine tritium or 32P, thyroid scintillation scan for 125I). Environment- or area-monitoring devices include the Geiger-Müller counter, ionization chamber, and scintillation detector. Where an exposure potential occurs, shielding with lead or other effective barrier can contain emissions.
To quantify risk from radiation exposure, a system of units has been created and revised many times. The International Commission on Radiological Units and Measurements (ICRU) has recommended that the older CGS (centimeter-gram-second) units be replaced by the equivalent SI (International System of Units) units, as shown in Table 14–9. Table 14–10 lists the recommended external exposure limits. The basis for the limits is what is referred to as the acceptable risk of adverse health effects. This is thought to be 1 in 10,000 effects per year for workers with occupational exposures and 1 in 10,000–1,000,000 effects per year for the general public, based on estimated radiation-induced fatal cancers and serious hereditary disorders. Exposures can be easily prevented by the use of lead or other high-density material, which can enclose the source and/or shield the work area (eg, lead blocks, cement, and leaded glass).
Table 14–9. Radiation units.

Table 14–10. External radiation exposure limits.

![]() Treatment
Treatment
The patient should be decontaminated, hospitalized, and placed under the care of hematologists and infectious disease specialists. Vital signs; fluid and electrolyte balance; and hematopoietic, gastrointestinal, and central nervous system functions should be monitored closely. Consultation with a health physicist should be expedited to attempt to measure the radiation dose.
If the granulocyte counts fall below 1000/μL, prophylactic antibacterial agents, acyclovir, and antifungals are recommended. If there is fever or suspected sepsis, antimicrobial agents by intravenous infusion should be started immediately. The choice of agents also should depend on the endemic pathogens at the particular hospital. Antimicrobial therapy should be continued until the granulocyte count exceeds 500/μL or until the patient has been afebrile for 5 consecutive days without evidence of infection. Reverse isolation should be maintained.
Granulocyte, platelet, and red cell transfusions may be necessary. Lymphocytes should be obtained immediately for HLA typing. Transfusions are recommended if the platelet count falls below 20,000/μL, the granulocyte count below 200/μL, or the hematocrit below 25%. Hematopoietic growth factors (filgrastim, sargramostim) have been effective in accelerating hematopoietic recovery. Bone marrow transplants have been used with questionable success in combating intractable hemorrhage and infection. They should be considered for patients exposed to 600–2000 cGy, and the decision about whether to use them should be made within a week of the radiation exposure.
Patients should receive supportive therapy as necessary for control of nausea, dehydration, and other symptoms. Ondansetron hydrochloride, 8 mg orally two or three times daily, has been recommended for nausea; chlorpromazine, 25–50 mg given deeply intramuscularly every 4–6 hours, is an alternative.
2. Acute Localized Radiation Injuries
Localized exposure of skin and body parts to ionizing radiation will result in hair loss (doses above 300 cGy), erythema (above 600 cGy), dry desquamation (radionecrosis) (above 1000 cGy), and wet desquamation (above 2000 cGy). Pain and itching occur shortly after exposure and are followed by erythema and blister formation. In cases of severe localized burns, there may be tissue ischemia and necrosis.
Prevention is the same as acute radiation syndrome (see above). Treatment is conservative and should not include surgery unless dictated by secondary complications. To conserve joint motion and prevent contractures, splinting and physical therapy may be required during convalescence. Injuries should be followed closely because the extent of tissue damage often is not readily apparent. Subsequent fibrosis, ulceration, infection, necrosis, or gangrene may occur and require surgery or more radical medical treatment.
3. Radionuclide Contamination
Skin contamination with radionuclides is rarely life-threatening. Immediate decontamination measures consist of gently scrubbing the skin with soap and warm water and, if necessary, cutting the hair. Hair clippings, material removed by scrubbing, swabs of the nares and mouth, clothing, and personal effects should be saved for radioactivity analysis and dosage calculation
For contaminated open wounds, gentle surgical debridement should be performed and wound irrigation considered. Depending on the type of radionuclide causing the injury, administration of a chelating agent may be indicated. For plutonium and alpha emitters, diethylenetriaminepentaacetic acid (DTPA) is effective and can be administered systemically, as well as in the wound irrigation solution. Blocking agents may also be considered, as in the case of radioiodines. Uranium and its associated radon daughter emissions are associated with lung cancer and probably reproductive effects, nonmalignant pulmonary disease, and nephritis. To assist with dose measurement, all urine and feces for the first 24 hours (or longer) should be collected.
4. Delayed Effects of High-Dose Radiation
Radiodermatitis often occurs in association with ionizing radiation therapy. The skin is dry, smooth, shiny, thin, pruritic, and sensitive, and there are signs of telangiectasia, atrophy, and diffuse pigmentation. The nails are brittle and striated.
Scarring in other tissues following high-dose exposure has led to endarteritis obliterans, dry-eye syndrome, myelopathy, pericarditis, hepatitis, nephritis, coronary artery disease, chromosomal injury, intestinal stenosis, pulmonary fibrosis, and cataracts.
Systemic exposure because of nuclear blast and/or fallout is associated with increases in thyroid neoplasms and leukemia. Cancers related to localized radioactivity include bone cancer from localized radioisotopes, thyroid cancer following childhood thymus irradiation, liver cancer associated with thorium dioxide, and lung cancer associated with radon decay products (radon daughters) in uranium miners. Leukemia has been reported in patients receiving radiotherapy for ankylosing spondylitis. Additional cancers associated with exposure to ionizing radiation include skin and breast.
Other effects of high-dose exposure include premature aging, shortening of the life span, and teratogenic (central nervous system deficit, mental retardation, and microcephaly) and reproductive abnormalities.
5. Effects of Low-Dose Radiation
Controversy continues over whether the risk for somatic and genetic disorders is increased significantly by cumulative low-dose exposures. The dose-response curve in the low-dose range cannot be determined at present, so most estimates of risk continue to be based on mathematical extrapolations from experiences with higher doses. Although developmental abnormalities are associated with doses as low as 10 cGy, and cancers are associated with levels below 100 cGy, the practical relevance of low-dose phenomena is extremely difficult to establish not only because of inconsistencies in the literature but also because the cumulative average exposure for people in the United States is approximately 8–10 cGy per lifetime (Table 14–11). Current data suggest that low cumulative exposure (50–100 mSv) is carcinogenic, but its risk depends on the total dose, cancer type, and age at exposure.
Table 14–11. Average annual effective dose equivalent of ionizing radiations to a member of the US population.

LASER INJURIES
The energy of the laser source is transformed through atomic excitation into a coherent, collimated, monochromatic beam of radiation. Lasers operate at one wavelength, usually in the ultraviolet, visible, or infrared portion of the electromagnetic spectrum. They may emit radiation in continuous or pulsed waves.
The list of industrial laser applications that may result in human exposures is a long one and is growing. Familiar uses of low power lasers using visible light include bar code scanning, laser pointers, and laser leveling. Moderate power lasers can perform material cutting, soldering, and brazing operations. Medical applications include skin treatment, tattoo removal, and retinal surgery. Intense laser sources are used to cut hard metals and diamonds.
Lasers have been categorized by their ability to cause injury in the ANSI standard Z136.1, “For the Safe Use of Lasers.” The basic categories are Class 1, 2, 3a, 3b, and 4. These laser categories provide some indication of relative hazard, but many other variables in the laser environment and application contribute to the overall risk. Laser Safety Officers are specially trained in the more sophisticated risk analysis, and should be consulted in the event of a laser injury.
Class 1 lasers are generally regarded as safe, barring the use of optical concentration with 1M lasers. Class 2 laser beams are not expected to ever cause injuries due to the blink reflex, again barring optical concentration of the 2M beam. Class 3 lasers can cause eye injuries, with class 3b lasers likely to cause retinal injury even from reflections. One should be aware that not every laser is classified correctly: green lasers labeled class 1 have recently been found to exceed the power level of that classification.
Biologic effects occur at low intensities, although there is some variation with wavelength. Permanent injuries have not been associated with repeated low-intensity exposures. At high intensities, thermal or pressure-induced damage to the skin or eyes can occur. These injuries are more likely with lasers that have a high-intensity beam outside the visible light spectrum because the worker’s proximity to the beam may not be apparent. Because of the thermal mechanism, any damage that occurs would be expected to be manifested immediately with the symptoms and signs of a corneal, retinal, or cutaneous burn. Exposure to ultraviolet (UV) lasers is more likely to cause corneal damage, whereas infrared and visible-light lasers are more likely to injure the retina because of their ocular penetration. Eye symptoms of accidental high-intensity laser exposure include photophobias or a sudden visual flash followed by scotoma or shadow of unusual size and color. Visual acuity or fields may be reduced. Retinal changes, including edema, coagulation, hemorrhage, and opaque vitreous, can occur. Prompt treatment for laser injuries is important, and often includes corticosteroids.
To prevent injuries, proper engineering controls and personal protective equipment matched to the laser wavelength should be used. A qualified laser safety officer should review controls, especially for class 3b and 4 lasers. Evaluation of workers following laser exposures should include an assessment of exposure intensity, wavelength, duration, viewing angle, and ANSI laser hazard classification.
Individuals working in proximity to high-power lasers should be instructed in proper operating procedures and provided with protective eyewear designed for the specific wavelength of the laser. In some cases, the eyewear does not by itself offer sufficient protection owing to possible laser reflections, or ultrashort laser pulses. Other devices, including door interlocks, barriers, and where possible, remote viewing equipment, should be considered. Skin protection is also important when high-power lasers are used. If feasible, systems should be designed with the beam line totally enclosed.
Preplacement examinations are recommended for individuals who will work with class 3B and 4 lasers and should consist of, at a minimum, the medical history, tests for visual acuity (near and far), refractive errors, visual fields, and inspection of the outer eye and skin. Baseline retinal photographs can be useful in evaluating patients after they have experienced a laser flash sensation. Periodic evaluation is not recommended for laser operators unless an acute high-intensity exposure occurs.
ATMOSPHERIC PRESSURE DISORDERS (DYSBARISM)
Sudden shift to an environment of lower ambient pressure, as occurs with rapid ascension to the surface from deep-sea diving or with loss of cabin pressure while flying at high altitudes, causes decompression sickness. Compression sickness can also occur following movement to an environment of higher ambient pressure, but the only common example of this is barotitis.
1. Decompression Sickness (Caisson Disease)
Decompression sickness results from mechanical and physiologic effects of expanding gases and bubbles in blood and tissue. When the body is exposed to an environment of higher than atmospheric gas pressure, as in tunneling or diving, it absorbs more of the inhaled gases than it does at sea level. Aided by its fat solubility, nitrogen concentrations increase in tissues, particularly those of the nervous system, bone marrow, and fat. Because the blood supply is poor in bone marrow and fat, nitrogen enters and leaves these tissues more slowly than oxygen or carbon dioxide does. As the surrounding pressure decreases (decompression), nitrogen expands and will form gas bubbles if there is insufficient time for its dissolution from tissues. Because oxygen and carbon dioxide have greater fluid solubility and move more easily between tissue compartments, their tendency for bubble formation is reduced. Remaining nitrogen gas bubbles are more destructive, especially in less elastic structures or tissues (eg, joints and central nervous system).
Most cases of decompression sickness have occurred after rapid ascension from sea depths in excess of 9 m (29.5 ft) or after sudden cabin pressure loss at altitudes in excess of 7000 m (22,966 ft).
![]() Clinical Findings
Clinical Findings
A complete evaluation of the systems affected—as determined by the history and physical examination—should be performed with appropriate x-rays and other diagnostic procedures. Anyone exhibiting signs or symptoms of decompression sickness within 48 hours of a high-pressure exposure should be given a compression test in which 100% oxygen at 3 atm is administered for 20 minutes in a hyperbaric chamber.
There are three types of decompression sickness, as described below. The type and severity of symptoms will depend on the age, weight/body fat, smoking status and physical condition of the patient, the degree of physical exertion, the depth or altitude before decompression, duration of compression, and rate and duration of decompression.
A. Type 1 Decompression Sickness
This type, which has the best prognosis, affects the limbs and skin. Acute pain, usually around a frequently used joint, may be incapacitating and cause the patient to assume a stooped posture (the “bends”). Pain may begin immediately after decompression or up to 12 hours later and sometimes is accompanied by urticarial and bluish red mottling and itching of the skin (“diver’s lice”).
B. Type 2 Decompression Sickness
Type 2 is more severe than type 1. Symptoms and signs of central and peripheral nerve damage may include vertigo, “pins-and-needles” paresthesias, hypoesthesia, ataxic gait, hyperreflexia, Babinski sign, paralysis or weakness of the limbs, headache, seizures, vomiting, visual loss or visual field defects, incontinence, impaired speech, tremor, and coma. Pulmonary manifestations (the “chokes”) may include substernal pain, chest tightness, severe coughing, dyspnea, pulmonary edema, and shallow respirations. Cardiovascular findings include arrhythmia and hypertension.
Type 2 sickness, which is probably caused by gas bubbles in the central nervous system and spinal cord, may have significant sequelae, such as vascular obstruction and tissue infarction (which sometimes are accompanied by hemoconcentration, changes in osmotic pressure or lipid emboli, hemorrhagic infarcts of the lungs, ulcers of the colon, multifocal degeneration of white matter, and hypercoagulation of blood).
Pulmonary barotrauma and gas expansion in other tissues can cause arterial gas embolism, which is the second leading cause of death in divers (drowning is first). Types 1 and 2 decompression sickness also can occur with unpressurized ascent to high altitude or with loss of cabin pressurization (decompression) at high altitude. Severity depends on the initial altitude, the final altitude, and the rate of ascent or the speed of depressurization.
C. Type 3 Decompression Sickness
The third type is characterized by aseptic necrosis of bone (osteonecrosis), which frequently involves the head or shaft of the humerus and less often involves the lower end of the femur and the tibial head. Osteonecrosis usually occurs 6–60 months following decompression and is asymptomatic unless there is joint involvement, which can cause permanent impairment. Radiographic examination may show bone sclerosis and mottling. Lesions are often symmetric.
Osteonecrosis may be the result of nitrogen bubbles obstructing the capillaries and has been reported in up to 50% of divers and underwater workers, although disability occurs in fewer than 3%. An increased incidence of memory deficits, retrograde amnesia, emotional instability, and other neurologic and psychiatric symptoms has been observed in divers with a history of multiple episodes of decompression sickness.
![]() Prevention
Prevention
Divers, underwater workers, and pilots should be screened to make that sure they are in good physical condition—not overweight and with no other conditions imposing an increased risk for dysbarism, such as vascular disorders, hypercoagulopathy, obstructive airways disease, pneumothorax, sinusitis, otitis media, dehydration, substance abuse, or recent bone fractures. Workers should receive training and education in proper compression and decompression procedures and in recognizing the symptoms and signs of decompression sickness.
![]() Treatment
Treatment
A. Types 1 and 2 Decompression Sickness
The patient should be placed in a supine position. For immediate first aid, 100% oxygen should be administered, and aspirin may be given for analgesia. The patient should be transported rapidly to an emergency facility that has a hyperbaric chamber for recompression and decompression. Information about the nearest facility and advice about recompression can be obtained 24 hours a day by calling the National Divers Alert Network (DAN) at 919-684-8111.
In the hyperbaric chamber, the patient is placed in an atmosphere of raised pressure. The pressure then is reduced at a slow rate, with decompression pressures and schedules determined on the basis of the duration and pressure exposure of the inciting incident. Compression protocols vary by provider; the U.S. Navy treatment schedules are commonly followed in the United States. Breathing 100% oxygen by mask, alternating with breathing normal air should shorten the period of decompression. Some centers use oxygen-helium mixtures as an alternative to protocols requiring 100% oxygen in an effort to speed decompression without causing oxygen toxicity.
Corticosteroids, diuretics, or both can be used for cerebral or spinal edema. Volume depletion should be corrected with oral or parenteral fluids (normal saline or lactated Ringer solution). In severe cases, anticoagulation with heparin or plasma volume expansion with low-molecular-weight dextran 40 is effective. Diazepam is used for treatment of confusional states and oxygen toxicity if oxygen is administered during treatment.
In cases of type 2 sickness, decompression may take several days. Careful monitoring should be maintained to guard against oxygen toxicity of the lungs and central nervous system.
B. Type 3 Decompression Sickness
1Osteonecrosis and sequelae that are a result of chronic decompression sickness are treated in the same manner as those conditions arising from other causes.
2. Compression Sickness
When atmospheric pressure is increased, internal gases become compressed, usually with little effect. The only common form of compression sickness is barotitis. This can occur with descent of an aircraft from a high altitude, under water during diving descent, or during hyperbaric oxygen therapy, any of which causes a relative vacuum in the middle ear space if the auditory tube is already obstructed because of allergies or upper respiratory tract infection. Symptoms may include pain or a foggy feeling in the ears, dizziness, nausea, and vertigo. In more severe cases, the tympanic membrane may appear inflamed and retracted or ruptured.
Barotitis can be prevented in people at risk by avoiding high-pressure exposures or, for short exposures, by using decongestants. Barotitis is usually self-limiting but can be treated with decongestant nose drops, a nasal vasoconstrictor inhaler, or use of a Valsalva maneuver.
DISORDERS CAUSED BY VIBRATION
Vibration occurs when mechanical energy from an oscillating source is transmitted to another structure. Every structure has its own natural vibration level, including the human body as a whole and each of its parts. When vibration of the same frequency is applied, resonance (amplification) of that vibration occurs, often with adverse effects. For example, at a frequency of 5 Hz, whole-body resonance occurs, and the body acts in concert with externally generated vibration and amplifies that effect.
1. Effects of Whole-Body Vibration
Truck and bus drivers, heavy-equipment operators, miners, and others exposed to long-term whole-body vibration have been reported to have a higher incidence of musculoskeletal, neurologic, circulatory, and digestive system disorders than does the general population. Low-back pain, inter-vertebral disk damage, and spinal degeneration are found frequently. European studies have found associated bony abnormalities (intervertebral osteochondrosis and calcification of intervertebral disks) and adverse reproductive effects (spontaneous abortion, congenital malformations, and menstrual changes). “Vibration sickness,” characterized by gastrointestinal problems, decreased visual acuity, labyrinthine disorders, and intense musculoskeletal pain, also has been reported in these workers. Despite these reports, a relationship between exposure intensity or quantity and the disorders found in occupationally-exposed groups has not been clearly defined. Although many questions remain unanswered regarding the effects of long-term whole-body vibration exposure, neurologic and spinal effects appear likely.
Although almost all clinical and experimental effects of whole-body vibration have occurred at frequencies less than 20 Hz, they also have been reported to occur at frequencies as high as 100 Hz, depending on other factors such as the amplitude, acceleration, duration, and direction (vertical or lateral) of the vibrating force. The International Standards Organization (ISO) has established guidelines for whole-body vertical vibration exposure times to various frequencies and accelerations, as shown in Figure 15–16. Not all investigators agree with existing exposure standards because of the many inconsistencies in the literature; however, prudence suggests that employers should try to minimize whole-body vibration exposures of their employees whenever possible by limiting the duration of exposure and choosing well-designed equipment that insulates workers from vibration.
2. Vibration-Induced White-Finger Disease (Hand-Arm Vibration Syndrome)
Vibration-induced white-finger disease (hand-arm vibration syndrome [HAVS]) is an occupational injury caused by segmental vibration of the hands. In the United States, more than 1 million workers are estimated to have significant exposure to vibration from hand tools such as power saws, grinders, sanders, pneumatic drills, jackhammers, and other equipment used in construction, foundry work, machining, and mining. Although segmental vibration injury can occur with frequencies ranging from 5 to 1500 Hz, it usually occurs with frequencies of 125–300 Hz. Other factors affecting risk include the amplitude and acceleration of the equipment used and the duration of use. Cumulative trauma occurs most often with a work history of at least 2000 hours of exposure and usually over 8000 hours.
HAVS is characterized by spasms of the digital arteries (Raynaud phenomenon) caused by vibration-induced damage of the peripheral nerve and vascular tissue, subcutaneous tissue, bones, and joints of the hands and fingers. The pathologic process also may involve arterial muscle wall hypertrophy; demyelinating peripheral neuropathy; excess connective-tissue deposition in perivascular, perineural, and subcutaneous tissues; and microvascular occlusion. Attacks of vasospasm can last for minutes to hours and are more likely to occur with exposure to the cold and with strenuous physical exertion. The worker often is standing erect, with the hand held lower than the heart and maintained in a contracted position.
![]() Clinical Findings
Clinical Findings
Attacks in severe cases can last from 15 minutes to 2 hours. They are usually easily reversible if the individual is removed from vibration exposure. Early symptoms consist of tingling followed by numbness of the fingers. The fingers later begin to turn white in a cold environment or when cold objects are touched. Intermittent blanching often starts with the tip of one finger but extends progressively to other fingertips and eventually to the tips and bases of all fingers on the exposed hands. With increasing severity of disease, blanching or cyanosis of the fingers may extend into the summer season. Return of blood circulation (reactive hyperemia, or “red flush”) following each episode is accompanied by redness and swelling, acute pain, throbbing, and paresthesias.
In more advanced cases, there may be degeneration of bone and cartilage, with resulting joint stiffness, restriction of motion, and arthralgia. Manual dexterity may decrease and clumsiness increase. With greater intensities of vibration, the period between exposure to vibration and the appearance of “white finger” is shorter.
Diagnosis is based on exposure history and response to cold. Specific diagnostic tests can include finger systolic pressure response to cold stress, finger temperature response to cold, vibrotactile sensitivity and perception threshold measurements, ultrasonic measurements of blood flow, plethysmography, thermography, and other tests of localized vascular and neurologic function. Diagnostic staging is based on the Stockholm Workshop Scales, which consider both vascular and neurologic effects.
![]() Differential Diagnosis
Differential Diagnosis
The diagnosis of HAVS is based on the occupational history of vibration exposures, the association of these exposures with episodes of Raynaud phenomenon (digital vasospasms), and exclusion of idiopathic Raynaud disease and other causes of Raynaud phenomenon, including trauma of the fingers and hands, frostbite, occlusive vascular disease, connective-tissue disorders, neurogenic disorders, drug intoxication, and exposure to vinyl chloride monomer.
![]() Prevention
Prevention
Segmental vibration can be prevented by using well-designed tools (see Chapter 15), wearing gloves to minimize vibration and keep the hands warm, and following a work-rest schedule that prevents long periods of exposure to vibration. Workers should be instructed about the early symptoms and signs of HAVS and advised of factors that may place them at higher risk, such as the use of vasoactive drugs and cigarette smoking. Exposure limits developed by the ACGIH rely on measurements of the acceleration of the tool in each of three directional axes.
![]() Treatment
Treatment
In most cases, symptoms and signs disappear when the worker is removed from exposure to vibration. In other cases, attacks can be reduced in severity or stopped by massaging, shaking, or swinging the hands or by placing them in warm water or warm air.
For more intractable episodes, nifedipine, 30–40 mg/d, is effective; thymoxamine is an alternative. For more severe cases, stanozolol or prostaglandin E may be useful. Biofeedback training also has been suggested. Surgical sympathectomy may be considered for irreversible cases. For medical or surgical therapy, the patient should be referred to the appropriate vascular specialist or hand surgeon.
HIGH-PRESSURE INJECTION INJURIES
Use of pressurized tools and systems in manufacturing and service industries occasionally results in severe injection injury. Common types of materials injected include hydraulic fluid, paint and paint thinner, grease, and fuel. The nondominant hand is the most common injury location, but other sites such as the orbit have been reported.
![]() Pathogenesis
Pathogenesis
The type and amount of material injected, the anatomic location, and the velocity of injection determine the extent of injury. As an example of the effect of material type, paint and paint thinners often incite a large inflammatory response on a chemical basis, as well as having antibacterial properties. The amount of material injected determines the amount of localized tissue distension, which, in turn, determines the extent of vascular compromise. The site and velocity of injection affect the dispersion of the material injected along tissue layers as well as tissue penetration.
Pathologic response occurs in three stages. The first stage involves acute inflammation associated with vascular compromise from tissue distension. Gangrene and/or infection often complicate the first stage. The second stage involves chemically induced inflammation and foreign-body granuloma formation. The late stage involves tissue fibrosis and breakdown of skin overlying granulomas, resulting in ulceration and subcutaneous sinus formation.
![]() Clinical Findings
Clinical Findings
With the initial event, the patient may feel a momentary stinging sensation; numbness and swelling are the initial symptoms. In fact, the initial appearance of the injury often does not reflect the severity of the injury. Also present will be a small puncture wound, from which some of the injected material may be oozing. Pressure exerted around the puncture may increase the amount of oozing.
Within a few hours, throbbing pain and pallor or cyanosis may develop. The pain sometimes is described as a burning sensation. Patients who do not seek immediate evaluation may present hours to several days later with leukocytosis and evidence of lymphangitis. Laboratory tests include radiography for evidence of radiopaque materials such as metals and xeroradiography for grease. CT scanning has been used to demonstrate localized edema, gas pockets, and globe distortion involved in orbital injection injuries.
![]() Treatment
Treatment
The goal of treatment is preservation of neurovascular structures. Aggressive decompression, debridement, and irrigation are recommended. Incision and debridement of devitalized tissues and removal of as much of the injected material should be done as soon as possible. Open debridement of all contaminated structures including tendon sheaths is recommended. Amputation may be required and is most frequent in injections involving paint or solvents. Pulsed-lavage irrigation, drainage, and open packing techniques have been used successfully. Delayed wound closure or closure by secondary intention is recommended.
Broad-spectrum antibiotic and tetanus prophylaxis, if indicated, should be provided. Early range-of-motion and intense physical therapy should be provided; twice-daily hand soaks in povidone-iodine and daily whirlpool treatments have been used successfully.
Initially, analgesics will be required, but local digital blocks should be avoided because of the risk of further vascular compromise. The value of steroid or dextran use has not been demonstrated for injection injuries, with the possible exception of steroids for high-pressure orbital injections, for which surgical debridement is more difficult or may not be appropriate.
REFERENCES
Brown DJA: Accidental hypothermia. New Engl J Med 2012;367:1930 [PMID: 23150960].
Divers Alert Network. www.diversalertnetwork.org.
Donnelly EH: Acute radiation syndrome: assessment and management. South Med J 2010;103:541 [PMID: 20710137].
Fish JS: Diagnosis of long-term sequelae after low voltage electrical injury. J Burn Care Res 2012;33:199 [PMID: 21979842].
Heaver C: Hand-arm vibration syndrome. J Hand Surg Eur 2011;36:354 [PMID: 21310765].
ICNIRP statement—Protection of workers against ultraviolet radiation. Health Phys 2010;99:66 [PMID: 20539126].
Kasunori K: Radiation effects on cancer risks in the Life Span Study cohort. Radiat Prot Dosimetry 2012;151:674 [PMID: 22908358].
Pappou PP: High-pressure injection injuries. J Hand Surg Am 2012;37:2404 [PMID: 22999384].
Radiation Emergency Assistance Center/Training Site (REACTS). www.orau.gov/reacts.
Vann RD: Decompression illness. Lancet 2011;377:153 [PMID: 21215883].
![]() SELF-ASSESSMENT QUESTIONS
SELF-ASSESSMENT QUESTIONS
Select the one correct answer for each question.
Question 1: Systemic hypothermia
a. is reduction of the body’s core temperature below 35°C (95°F)
b. onset is reliably symptomatic
c. may result in hemoconcentration as a result of internal bleeding
d. loss of consciousness is irreversible
Question 2: Heat stroke
a. presents with increased alertness
b. presents with cold, dry skin
c. stabilizes as the core (rectal) temperature approaches 41.1°C (106°F)
d. may cause cerebral, cardiovascular, hepatic, or renal damage
Question 3: Electrical injuries
a. comprise up to 10% of all fatal industrial accidents
b. from alternating currents are less dangerous than those from direct currents
c. can cause not only tissue destruction and necrosis from heat and burning but also depolarization of electrically sensitive tissues such as nerve and heart
d. involving currents exceeding 10,000 V freeze the worker to the source
Question 4: Acute radiation syndrome
a. largely spares tissues with the most rapid cellular turnover
b. primarily affects reproductive, hematopoietic, and renal tissues
c. from doses exceeding 1000 cGy are uniformly lethal
d. patients may pass through four phases: prodrome, latent phase, illness, and recovery
Question 5: Decompression sickness (Caisson disease)
a. results from mechanical and physiologic effects of toxic gases
b. reflects nitrogen concentration in the nervous system, bone marrow, and liver
c. symptoms are related to oxygen and carbon dioxide bubble formation
d. occurs after rapid ascension from sea depths in excess of 9 m (29.5 ft) or after sudden cabin pressure loss at altitudes in excess of 7000 m (22,966 ft)
Question 6: Laser eye injuries
a. only occur when looking directly into a laser beam
b. are not related to the laser’s power
c. only occur with visible light lasers
d. are much more likely when exposed to Class 3b or stronger lasers
Question 7: High-pressure injection injuries
a. should be treated conservatively and allowed to drain spontaneously
b. usually occur in the dominant hand
c. often require extensive debridement with delayed closure
d. tend to recover without physical or occupational therapy