Definition
• Precursor lesion of adenocarcinoma, ≤3 cm but more than 0.5 cm, with pure lepidic growth of neoplastic cells along preexisting alveolar septa without stromal, vascular, or pleural invasion
Clinical features
Presentation
• Often asymptomatic; incidental finding on radiology
Prognosis and treatment
• Excellent prognosis after complete surgical resection (100% tumor-free survival)
Pathology
Gross
• Single peripheral nodule that is soft and poorly circumscribed; rarely mucinous
Histology
• Tumor cells grow along preexisting alveolar septae with no evidence of stromal, vascular, or pleural invasion
• Histological variants
• Nonmucinous AIS
– Most common variant
– More often associated with septal sclerosis than mucinous variant
– Tumor cells have Clara cell or type II pneumocyte differentiation
– Clara cell differentiation: columnar cells with apical nuclei and pale eosinophilic cytoplasm with cytoplasmic snouts
– Type II pneumocyte differentiation: cuboidal to dome-shaped, clear to foamy cytoplasm with fine cytoplasmic vacuoles, or intranuclear eosinophilic inclusions
– No clinical significance in distinguishing Clara cell from type II pneumocyte differentiation
• Mucinous AIS
– Very rare
– Tumor cells are tall and columnar with basally located nuclei, pale cytoplasm, variable amounts of intracytoplasmic mucin, and minimal cytological atypia
– Usually with mucin accumulation in the surrounding alveolar spaces associated with numerous macrophages engulfing mucin
– The tumor formerly called mucinous BAC is now classified as mucinous adenocarcinoma if multifocal, >3 cm or with invasive component >0.5 cm
• Mixed mucinous and nonmucinous; rare
Immunopathology/special stains
• Nonmucinous AIS has similar immunoprofile as adenocarcinoma of lung with positive TTF-1, CK7, and negative CK20
Main differential diagnoses
• Type II pneumocytic hyperplasia: usually occurs in reactive process
• Atypical adenomatous hyperplasia: ≤5 mm, cells less crowded than AIS, less nuclear atypia
• Minimally invasive adenocarcinoma of the lung: peripheral lepidic growth with central invasive focus ≤0.5 cm; thus, AIS can only be diagnosed in a resection specimen
• Metastatic carcinoma with predominant lepidic growth: rare; clinical history is important
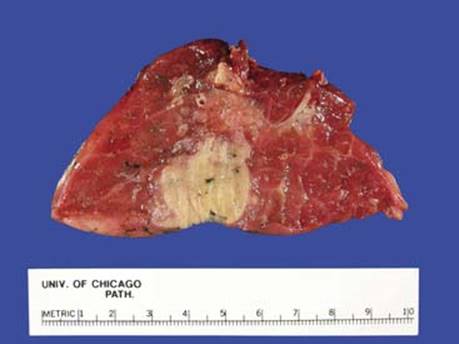
Fig 1 Adenocarcinoma in situ. Gross picture of cut surface of peripherally located AIS, wedge resection. The tumor is more spongy and softer than adenocarcinoma.
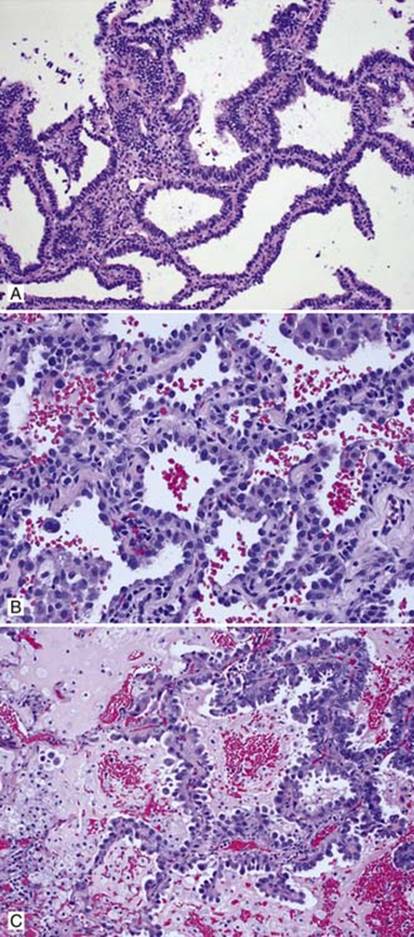
Fig 2 Adenocarcinoma in situ. Nonmucinous AIS: A, columnar cells growing along alveolar septae; B, cuboidal tumor cells; and C, nonmucinous AIS with associated obstructive pneumonitis leading to accumulation of intraalveolar mucin and muciphages.
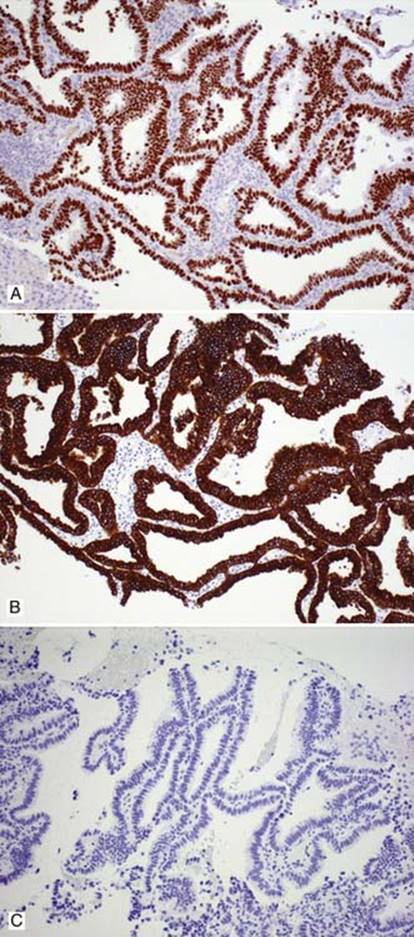
Fig 3 Adenocarcinoma in situ. Nonmucinous AIS with positive TTF-1 (A), positive CK7 (B), and negative CK20 (C).
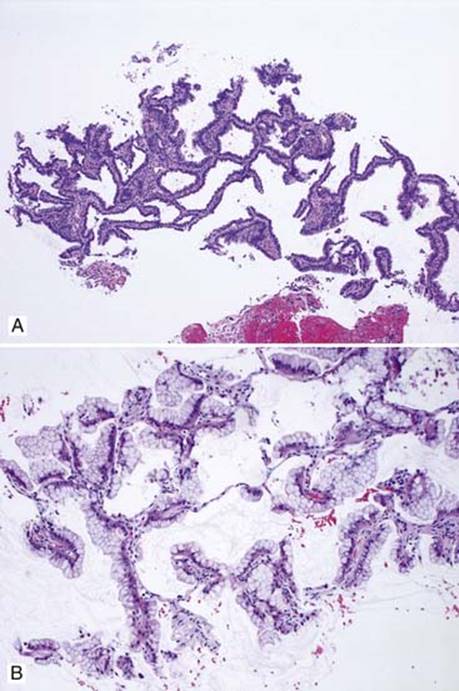
Fig 4 Adenocarcinoma in situ. Needle biopsy with lepidic pattern: nonmucinous (A) and mucinous (B). Definite diagnosis of AIS should not be made on biopsy.
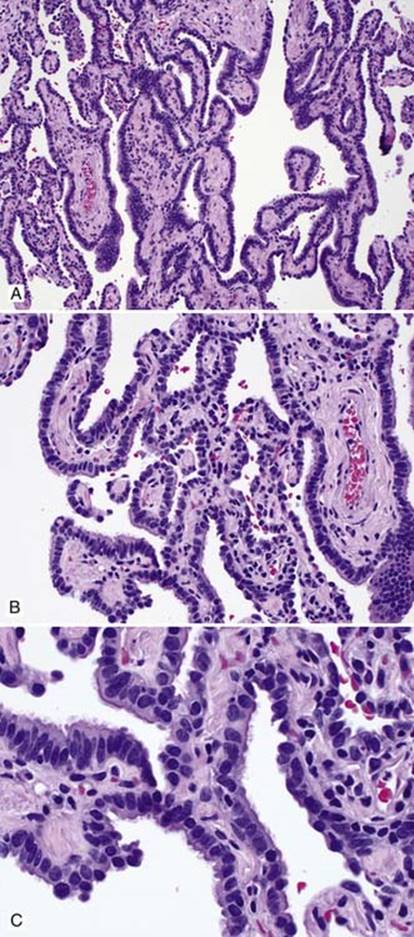
Fig 5 Adenocarcinoma in situ. Extensive bronchiolar metaplasia mimics AIS: A, low; B, medium; and C, high powers. Note the uniformity and cilia of the bronchial epithelial cells.