Upendra P. Hegde and Sanjiv S. Agarwala
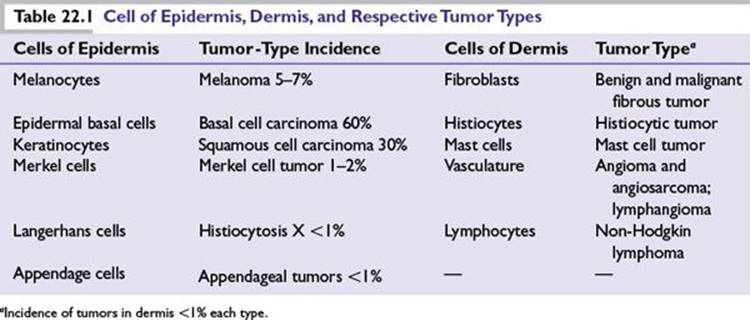
The skin is the largest organ of the human body that is embryologically derived from the neuroectoderm and the mesoderm to be organized into three layers namely epidermis, dermis, and subcutis. Cancer of the skin arises from the cell types of structures in all the three layers (Table 22.1). The direct exposure to sun’s ultraviolet radiation and a wide variety of environmental carcinogens predisposes skin cells to genetic damage and increased risk of cancer. The skin cancers are best divided into melanoma and nonmelanoma.
MELANOMA
Melanoma arises from the melanocyte, a neural crest–derived cell that migrates during embryogenesis predominantly to the basal layer of the epidermal skin and less commonly to the other tissues in the body such as mucosa of the upper aerodigestive and the lower genitourinary tract, the meninges, and the ocular choroid, where melanoma is rarely encountered.
Epidemiology
■Melanoma ranks as the fifth and seventh leading type of cancer in men and women, respectively, in the United States.
■The estimated lifetime risk of developing melanoma in US whites is about 1 in 50.
■In the United States 76,250 new cases of melanoma were expected to be diagnosed in 2012, with projected deaths of 9,180.
■The incidence of melanoma is more than 10 times greater in whites than in blacks.
■The incidence of melanoma among US whites is over 20 cases per 100,000 population although some geographic areas have higher rates.
■The rate of increase in melanoma incidence has decreased from 6% a year in the 1970s to 3% a year between 1980 and 2000 and stabilized after that period in younger subjects.
■In white males over 50 years of age, the incidence continues to climb at the fastest rate.
■Australia has the highest incidence of melanoma in the world, approximately 40 cases per 100,000 populations per year.
Etiology
■Ultraviolet rays: Exposure to sun’s ultraviolet rays in the electromagnetic radiation spectrum is a major risk factor for melanoma development and is related to (Fig. 22.1)
–Intermittent intense exposure
–Exposure at a young age
–Exposure in individuals with fair skin, blue eyes, blonde or red hair, propensity for sunburns, and inability to tan (poor tanners)
■Age: High incidence in young and middle-aged adults as well as in older subjects.
■Sex: Slightly more common in male subjects than in females.
■Ethnicity: Higher incidence in Northern than in Eastern and Southern Europeans.
Familial Melanoma
■About 5% to 10% of melanomas are familial and up to 40% have hereditary basis.
■A tumor suppressor gene cyclin-dependent kinase inhibitor 2A (CDKN2A) is the most commonly mutated gene located on the short arm of chromosome 9.
■The protective effect of CDKN2A is mediated by encoded protein p16INK4A.
■Other candidate genes in this category include cyclin-dependent kinase 4 and CDKN2A/p14 alternate reading frame CDKN2A/ARF.
■A high-risk variant of the α-melanocyte-stimulating hormone receptor gene (MC1R) located on chromosome 16q24 and associated with red hair and freckles confer high risk of familial melanoma in families segregating the CDKN2A gene.
■Hereditary basis of melanoma should be suspected in the following circumstances:
•Individuals with three or more primary cutaneous melanomas
•Melanoma at young age and family history of melanoma (mean age between 30 and 40)
•Individuals with cutaneous melanoma and a family history of at least one invasive melanoma and two or more other diagnoses of melanoma and/or pancreatic cancer among first- or second-degree relatives on the same side of the family
•Melanoma associated in patients with dysplastic nevi and atypical nevi
■Precursor lesions of melanoma include
•Dysplastic nevi locus of which resides on short arm of chromosome 1
•Congenital nevi and acquired melanocytic nevi (Table 22.2)
Risk Factors for Melanoma
■Xeroderma pigmentosum
■Familial atypical mole melanoma syndrome (FAMMS)
■Advanced age and immune-suppressive states
■Sun exposure and sun-sensitive phenotype
■Melanoma in a first-degree relative and previous history of melanoma
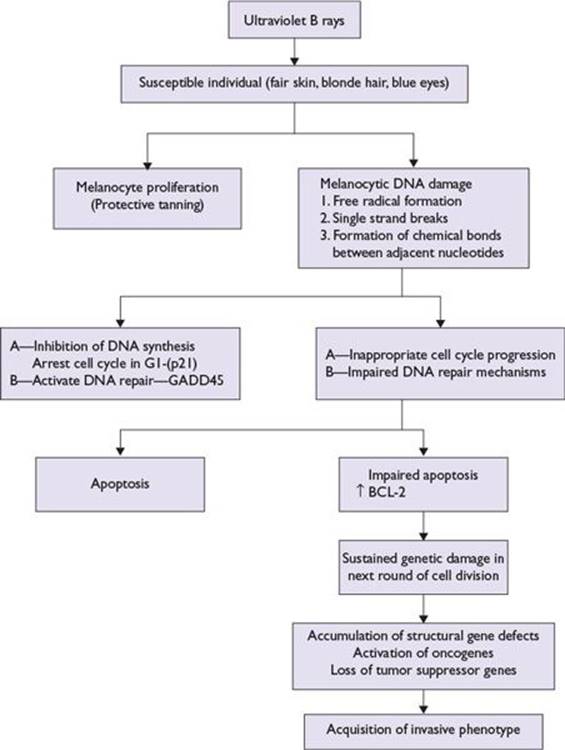
FIGURE 22.1 Model of ultraviolet B light-mediated pathogenesis of cutaneous melanoma.
Common Chromosomal Abnormalities in Melanoma
■Early chromosomal abnormalities:
•Loss of 10q
•Loss of 9p
■Late chromosomal abnormalities:
•Deletion of 6q
•Loss of terminal part of 1p
•Duplication of chromosome 7
•Deletion of 11q23

Clinical Features of Melanoma (abcde)
■Most cutaneous melanoma lesions are pigmented and display asymmetry, irregular borders, variegate colors with shades of brown, black or pink, white, red or blue, diameter of at least 6 mm; progressive change in size, nodularity, ulceration, or bleeding with or without pain and or itching and evolving in size, shape, or color.
■Less than 1% of cutaneous melanomas lack pigment. These are called amelanotic melanomas and may pose diagnostic challenges.
■Cutaneous melanoma can occur anywhere in the body. Cutaneous melanoma is more common in the lower extremities in women, the trunk in men, and head and neck in the elderly subjects.
Pathologic Diagnosis of Cutaneous Melanoma
Melanoma In Situ
Biopsy of a suspicious skin lesion for melanoma reveals characteristic tumor cell morphology in the basal epidermal layer verified by the tumor cells staining of melanoma-specific antigens such as S-100, premelanosomal protein HMB-45, nerve growth factor receptor, tyrosinase-related protein-1 (MEL-5). Melanoma tumor cells stain positive for vimentin and negative for cytokeratin stains.
Invasive Melanoma
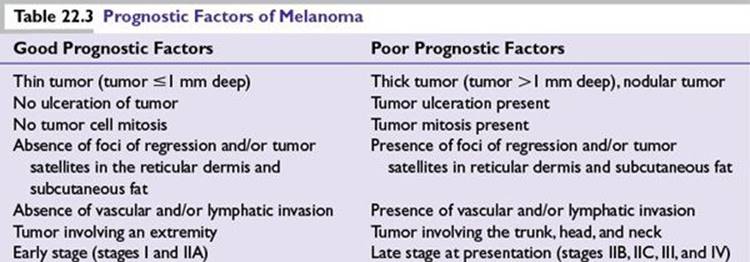
Tumor invasion into the dermis makes it an invasive melanoma quantified by two methods described by Clark et al. and Breslow see below. Additional histologic information provides important prognostic information and includes morphologic variants such as spindle cell morphology, ulceration, mitosis counted as number/mm2, lymphocytic infiltration, regression if any, vascular invasion, perineural invasion, and solar elastosis (Table 22.3).
Clinicohistologic Types of Melanoma: Microstaging
Clark Levels
Clark et al. subdivided melanoma invasion of the papillary dermis into a deep group in which tumor cells accumulate at the junction of the papillary and reticular dermis and a superficial group in which tumor cells did not invade deeper layers (Fig. 22.2).
Breslow Thickness
Breslow used an ocular micrometer to measure the vertical depth of penetration of tumor from the granular layer of the epidermis or from the base of the ulcerated melanoma to the deepest identifiable contiguous melanoma cell (Breslow thickness).
Principles of American Joint Committee on Cancer Melanoma Staging
Melanoma is staged based on information derived from three key categories (TNM):
■(T) Tumor characteristics on microscopic examination
■(N) Nodes—status of the regional lymph node metastasis
■(M) Distant metastasis—either present or absent
In American Joint Committee on Cancer (AJCC) staging, melanoma is divided into four stages:
■Stage I—thin melanoma (subdivided into IA and IB)
■Stage II—deeper melanoma without lymph node metastasis (subdivided into IIA, IIB, and IIC)
■Stage III—melanoma spread to regional lymph nodes (subdivided into IIIA, IIIB, and IIIC)
■Stage IV—distant metastasis (subdivided into M1a, M1b, and M1c)
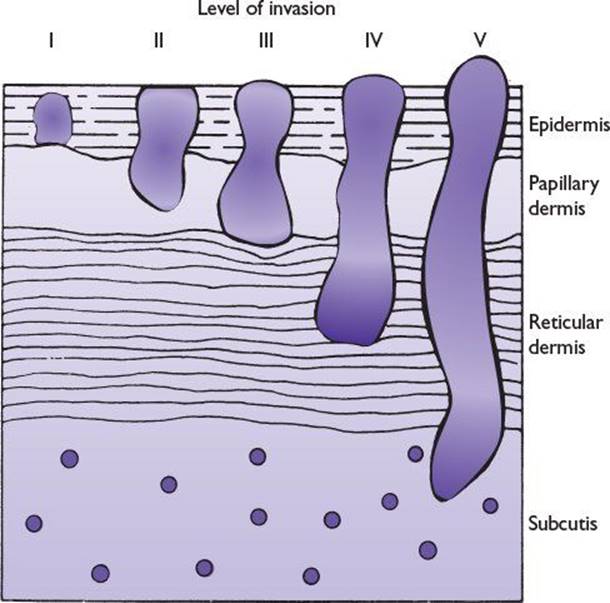
FIGURE 22.2 Schematic diagram of Clark levels of invasion.
Factors taken into consideration for subdividing each stage into subcategories A, B, or C include the following:
■Stages IA and IB: depth of invasion, ulceration, and mitosis
■Stages IIA, IIB, IIC: depth of invasion, presence or absence of ulceration
■Stages IIIA, IIIB, IIIC: Number of lymph node metastases, microscopic versus clinically palpable macroscopic lymph nodes, intralymphatic metastases (in transit metastasis) or satellites lesions (microscopic or macroscopic)
•A sentinel lymph node biopsy procedure is prerequisite to identify occult lymph node metastasis when involved lymph nodes are not palpable on clinical examination. Only exception is melanoma 1or less than 1 mm in depth without ulceration or mitosis.
•Immunohistochemical staining of melanoma-associated antigens is used to confirm morphologically identified cluster of few melanoma cells in the lymph node.
■Stage IV—M1a, M1b, M1c:
•M1a—Metastasis to the distant lymph node and subcutaneous tissues
•M1b—Lung metastasis
•M1c—Non–lung visceral metastasis that includes liver, bone, brain, and other organs
•Note: Serum enzyme lactate dehydrogenase if elevated upgrades M1a and M1b to M1c
Prediction of Patient Outcome Based on AJCC Melanoma Staging (Fig. 22.3)
■Low risk: Stages I and IIA (melanoma-specific mortality less than 25% at 20 years)
■Medium to high risk: Stages IIB, IIC, and III (melanoma-specific mortality between 55% and 75% at 20 years)
■Poor risk: Stage IV (melanoma-specific mortality more than 90% at 5 years)
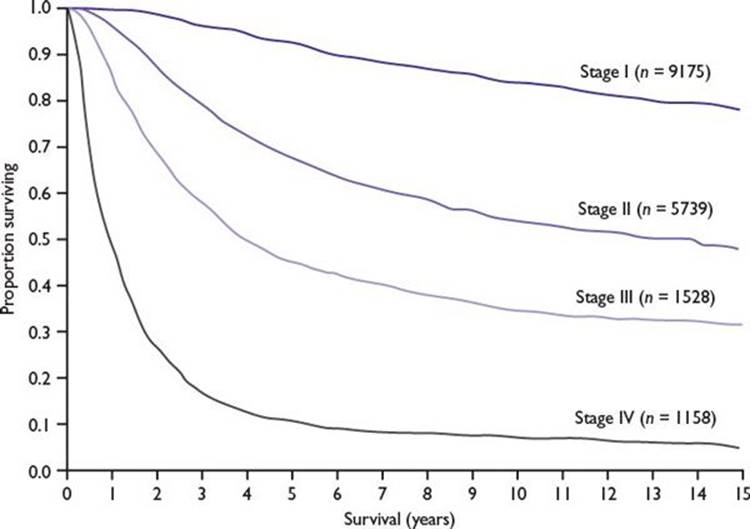
FIGURE 22.3 Relationship between the stage of melanoma and survival (20-year follow-up). (Kaplan-Meier survival curves adapted from Balch CM, Gershenwald JE, Soong S-J, et al. Final version of 2009 AJCC melanoma staging and classification. J Clin Oncol. 2009;27:6199-6206.)
Cutaneous Melanoma: Prevention and Early Diagnosis
■Patient education: increasing awareness of melanoma as a serious cancer, its risk factors, self-skin examination (SSE), sun avoidance, light clothing, and effective use of sunscreens.
■Close surveillance: Total body skin examination (TBSE) performed by a dermatologist provides close surveillance to identify suspicious skin lesions for biopsy and early diagnosis.
Selected patients at high risk of melanoma might benefit from the following two techniques:
■Digital photography is used for tracking suspicious skin lesions over time in patients with multiple nevi or dysplastic nevus syndrome.
■Dermoscopy (epiluminescence microscopy), performed by a trained operator, utilizes either a dermatoscope or 10× ocular scope (microscope ocular eyepiece held upside down) to visualize a variety of structures and patterns in pigmented skin lesions that are not discernible to the naked eye. The procedure has a potential to improve diagnostic sensitivity.
Melanoma Management: General Surgical Treatment Principles
An algorithm for melanoma management is presented in Figure 22.4.
Principle: Complete surgical excision of primary melanoma confirmed by comprehensive histologic examination of the entire excised specimen forms the basis of surgical treatment.
Risk of local recurrence: Relates to completeness of resection of the primary tumor, but is not significantly associated with the extent of surgical margin of excision (Table 22.4).
Assessment of the Regional Lymph Node Metastasis and Lymph Node Dissection
1 mm in depth, the risk is increased if ulceration or mitosis is present.Principle: The risk of melanoma metastasis to the regional lymph nodes is directly proportional to the depth of invasion (>1 mm deep), tumor ulceration, and mitosis. For invasive melanoma less than or equal to
Regional lymph node metastasis: Reflects aggressive tumor biology and depth of dermal invasion that if left untreated pose serious risk of spread to adjacent lymph nodes or to systemic organs.
Historically, complete excision of primary cutaneous melanoma is followed with elective, therapeutic, or delayed lymph node dissection from the respective basin.
■Elective lymph node dissection: Although clinically not palpable, all the lymph nodes are dissected from the respective basin because of concerns of melanoma metastasis.
■Therapeutic lymph node dissection: Is performed if the regional lymph nodes are enlarged and clinically palpable (suspected lymph node metastasis).
■Delayed lymph node dissection: Is performed when initially nonpalpable regional lymph nodes become palpable over a follow-up period (delayed metastasis).
Lymph node dissection is recommended only if regional lymph node metastasis is present and is avoided if lack of lymph node metastasis could be predicted by a reliable test.
Sentinel Node Biopsy
■Sentinel lymph node biopsy as a tool to detect regional lymph node metastasis.
■Characteristics of a sentinel lymph node:
•First lymph node in the basin at greatest risk of metastasis.
•Easily accessible and identified by lymphoscintigraphy.
•Pathologic evaluation helps to detect occult melanoma lymph node metastasis.
■Surgical approach to obtain a sentinel lymph node: lymphoscintigraphy
•Preoperative lymphoscintigraphy uses vital blue dye injected around cutaneous melanoma that provides a road map of the lymph node basin. Intraoperative lymphoscintigraphy uses radio colloid injection around the primary tumor, and a handheld device detects the radioactivity from the involved lymph node. The combination of vital blue dye and technetium-labeled sulfur colloid helps the surgeon navigate the identity of sentinel lymph node in the respective nodal basin for metastasis in 94% of cases.
■Implications of sentinel lymph node biopsy:
•Complete lymph node dissection is recommended only if sentinel lymph node is positive.
•A negative sentinel lymph node saves the patient morbidity of this procedure.
•Sentinel lymph node biopsy–guided information about the extent of lymph node metastasis helps in prognostication of primary melanoma and reduces the risk of local recurrence. Its impact on overall survival is not clear.
•In one study, immediate lymph node dissection after positive microscopic metastasis in the sentinel lymph node conferred survival advantage in a subset analysis.
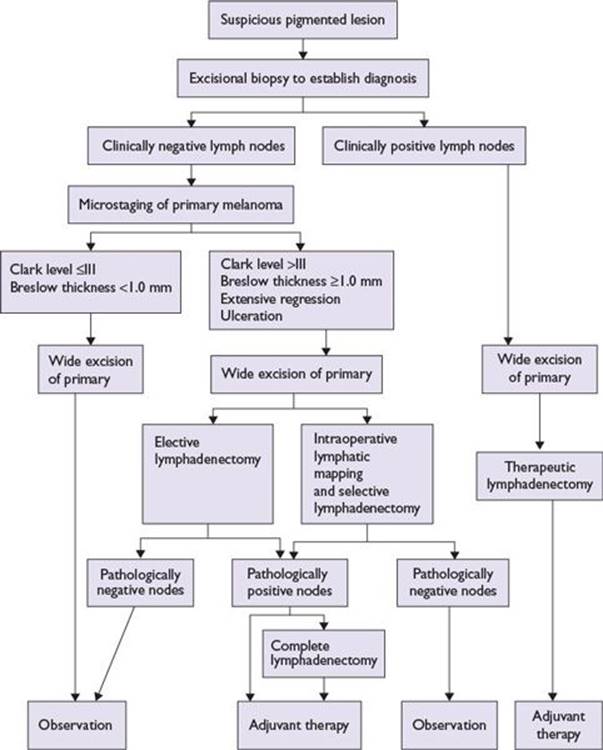
FIGURE 22.4 Algorithm for melanoma management.

Adjuvant Treatment of Melanoma in Patients at Risk of Recurrence after Surgery
Interferon alpha (IFN-α) Treatment Principle: There is a high rate of relapse of cutaneous melanoma (35% to 75%) among patients with AJCC melanoma stages IIB, IIC, and III after primary surgical treatment. Based on antiproliferative and immunomodulatory effects, prolonged use of IFN-α (high dose, low dose, intermediate dose for variable periods of time) has been extensively studied in large prospective randomized clinical trials as an adjuvant to primary surgery to assess benefit of reducing recurrences and improving survival (Table 22.5).
■IFN-α treatment conferred consistent relapse and disease-free survival benefit.
■Impact upon overall survival has been variable and less consistent.
■High-dose IFN-α is superior when compared to low-dose.
•One month of high-dose intravenous IFN-α induction-only therapy is not sufficient and must be followed by the maintenance phase to confer any benefit.
•Pegylated form of IFN-α (slow release) given subcutaneously once weekly for up to 5 years conferred relapse-free survival advantage without overall survival benefit.
•The predominant toxicity of high-dose IFN-α severe flu-like symptoms, liver toxicity, and depression that adversely affect quality of life and may compromise the intended benefit of interferon therapy.
•The decision to use interferon in the adjuvant setting of cutaneous melanoma treatment should be based on the perceived relative merits of disease control, quality of life, and financial cost.
■Biochemotherapy as an adjuvant treatment of cutaneous melanoma after primary surgery did not confer survival advantage in one randomized phase 3 clinical trial.
■Ongoing clinical trials are evaluating the role of ipilimumab, an anti-CTLA antibody, and vemurafenib, an agent that targets BRAF mutation as adjuvant treatment of melanoma after primary surgery. Both of these agents have recently been approved for use in patients with metastatic melanoma.

Role of Radiation Therapy
■Pain relief of melanoma metastasis to the musculoskeletal region
■As an adjuvant treatment after the regional lymph node dissection:
•In melanoma of the head and neck region
•When lymph node metastases are bulky and/or involve four or more lymph nodes or exhibit extracapsular spread
•Local recurrence of melanoma in a previously dissected lymph node basin
■After surgical resection of desmoplastic melanoma with neurotropism
■Brain metastasis of melanoma
Radiation therapy of brain metastasis of melanoma includes
■Whole-brain radiation if multiple and large size brain metastases are present
■Stereotactic brain radiation is preferred if small-sized or isolated or fewer (two to three) brain metastases are present
ResuLts of the studies by Skibber et al. and others have suggested that external radiation to the brain after surgical resection of the solitary brain metastasis from malignant melanoma has survival benefits.
Isolated Limb Perfusion or Infusion as a Treatment of Melanoma
Principle: To deliver maximally tolerated chemotherapy doses in patients with locally advanced and metastatic melanoma to a regionally confined tumor area such as a limb while limiting systemic toxicity.
Isolated limb perfusion (ILP): Involves hyperthermia and oxygenation of the circulation that potentiate the tumoricidal effects of the chemotherapeutic agents that include melphalan (L-PAM), thiotepa, mechlorethamine with or without tumor necrosis factor (TNF-α), and IFNγ. This procedure provides palliative benefit to patients with in-transit metastasis of melanoma otherwise difficult to resect surgically or at high risk of recurrence after surgery (Table 22.6).
Isolated limb infusion: Is a simplified and minimally invasive procedure developed at the Sydney Melanoma Unit (SMU) intended to obtain the benefits of ILP without major disadvantages. It is a low-flow ILP procedure performed via percutaneous catheters without oxygenation.
Both the procedures provide local tumor control and pain relief with improvement of quality of life usually without impact on overall survival.

Management of Patients with Metastatic Melanoma
The management options for a patient with metastatic melanoma have expanded following the recent FDA approvals of two novel agents:
■Ipilimumab is a monoclonal antibody to CTLA-4 antigen on T lymphocytes that regulate T lymphocyte activation after an encounter with melanoma antigen.
■Vemurafenib is a targeted therapy that selectively blocks mutated BRAF in melanoma patients harboring BRAF mutation.
Both of these agents have shown overall survival benefits to patients with metastatic melanoma and have unique toxicity profiles (see below).
■High-dose interleukin-2 (IL-2) (see below).
■Surgical resection of an accessible isolated metastatic lesion has a curative potential in about 25% of patients, while those who are not candidates for complete surgical resection of a metastatic lesion will require systemic treatment.
■Chemotherapy as a single agent, in combination, or with biologic agents (biochemotherapy) in patients with metastatic melanoma.
■Experimental therapies such as adaptive cell therapy and newer targeted therapies still remain promising options for selected patients with metastatic melanoma.
Chemotherapy of Metastatic Melanoma: Single-Agent Chemotherapy
■Dacarbazine is the only FDA-approved chemotherapeutic agent for melanoma treatment that has a response rate of about 10% to 20% without overall survival benefit.
■Temozolomide is a synthetic analog of dacarbazine that is orally bioavailable, crosses the blood–brain barrier, has comparable efficacy, and has a reduced toxicity profile.
■In a recent phase 3 trial, nab-paclitaxel (abraxane) showed a statistically significant improvement in progression-free survival and a trend toward improved overall survival as compared to dacarbazine.
Combination Chemotherapy Regimens of Metastatic Melanoma (Table 22.7)
■M.D. Anderson regimen: cisplatin, vinblastine, dacarbazine (CVD)
■Dartmouth regimen: cisplatin, carmustine, dacarbazine, and tamoxifen (CBDT)

A phase 3 multicenter randomized clinical trial of dacarbazine alone versus the Dartmouth regimen in patients with metastatic melanoma showed higher response rates of 25% to 30% and increased toxicity with Dartmouth regimen without significant survival benefit.
Biologic Agents in the Treatment of Metastatic Melanoma: IFN-α
IFN-α is the first recombinant cytokine investigated in phase 1 and 2 clinical trials of patients with metastatic melanoma based on its antiproliferative and immunomodulatory effects.
■Initial studies showed response rates of about 15% in patients with metastatic melanoma.
■One-third of these responses were complete and durable.
■Responses could be observed up to 6 months after the therapy was initiated.
■Small volume disease and uninterrupted use resulted in pronounced responses.
Falkson et al. reported the outcome of patients with metastatic melanoma treated with either dacarbazine alone or a combination of dacarbazine and IFN-α-2b. The results indicated a response rate and median survival of 20% and 9.6 months, respectively, with dacarbazine alone compared to a response rate and median survival of 53% and 17.6 months, respectively, in patients receiving both these agents. These results could not be reproduced in subsequent randomized phase 3 studies.
Essentially, IFN-α is rarely used as primary therapy for metastatic melanoma.
IL-2 Based Therapy
■IL-2 is a T-cell growth factor produced primarily by T-helper cells upon antigen binding.
■Interacts with IL-2 receptors expressed on activated cytotoxic T lymphocytes (CTLs) and causes:
•Increased production of other interleukins, IFN-γ, and TNF-α
•Proliferation and differentiation of both B and T lymphocytes and cytotoxic cells
■Antitumor effects are mediated by its ability to stimulate proliferation of natural killer cells (NK cells), lymphokine-activated killer cells (LAKs), and CTLs.
The U.S. Food and Drug Administration (FDA) approved high-dose IL-2 for treatment of metastatic melanoma at 600,000 international units (The FDA approved the high dose IL-2 at 600,000 international units per kg) per kilogram body weight administered as a bolus over 15 minutes every 8 hours for a maximum of 14 doses on days 1 to 5 and 15 to 19. Imaging studies are repeated after two such courses to evaluate efficacy.
In patients responding to high-dose IL-2, the treatment is continued unless the patient has significant side effects or the physician decides to stop treatment for safety reasons.
■The overall response rate is about 16%, which includes a complete response rate of 6%.
■Responses are observed in all disease sites.
■Durable responses are achieved in those who achieved complete responses.
■Good baseline performance and chemo-naive status are predictive of response.
■A toxicity profile results from capillary leak syndrome that is dependent on dose, route, and duration of administration. Common toxicities are as follows:
•High fever and fluid retention
•Gastrointestinal system side effects (i.e., nausea, vomiting, and diarrhea)
•Cardiovascular system side effects (i.e., hypotension or arrhythmias)
•Pulmonary side effects (i.e., hypoxemia and pleural effusions)
•Renal side effects (i.e., azotemia and renal failure)
•Central nervous system side effects (i.e., confusion and delirium)
■Patients with active comorbidities involving heart, lung, kidney, and liver disease or those with untreated brain metastasis with vasogenic edema are at high risk of life-threatening complications and mortality related to capillary leak caused by high-dose IL-2 treatment emphasizing rigorous patient selection for treatment with this agent.
■High-dose IL-2 should only be administered by health care teams experienced in its use.
■Lower doses of IL-2 administered either subcutaneously or as a continuous intravenous infusion at 9 to 18 million international units/m2/day for 4 to 5 days have been studied in patients not eligible for high-dose IL-2 treatment. Although total response rates as high as 20% have been reported, complete responses appear to be lower than those with high-dose IL-2.

Combining Chemotherapy and Biologic Agents in Metastatic Melanoma Biochemotherapy: Rationale
■Preclinical studies suggest that combining chemotherapeutic and biologic agents (biochemotherapy) may confer additive or synergistic effects against melanoma.
■Chemotherapeutic and biologic agents have different mechanisms of antimelanoma effects.
■There are no overlapping toxicity and cross-resistance.
Biochemotherapy: Built upon CVD regimen of chemotherapy plus continuous intravenous infusion of moderate dose IL-2 and –IFN-α administered subcutaneously showed high response rates and durable survival of between 10% and 20% in selected patients (Table 22.8).
■The toxicity associated with biochemotherapy regimen and lack of reproducibility of survival benefit among investigators has dampened interest in its universal use.
■A recent meta-analysis of 18 clinical trials and a phase 3 randomized clinical trial comparing biochemotherapy to CVD chemotherapy in patients with stage IV melanoma confirmed high response rates (40% to 50%) and increased toxicity with biochemotherapy without overall survival advantage.
Principles of Immune-Based Therapy of Melanoma
■Melanoma is considered to be one of the best models of an immunogenic tumor attracting T lymphocyte infiltration at both the primary and metastatic sites representing the immune footprints.
■A number of well-defined melanoma antigens have been identified both at a protein and gene level that evoke a cellular immune-based antimelanoma response (Table 22.9).
■Antigen-specific CD8+ CTLs lead the antimelanoma response with critical help from CD4+ helper T cells and antigen-presenting cells (APCs).
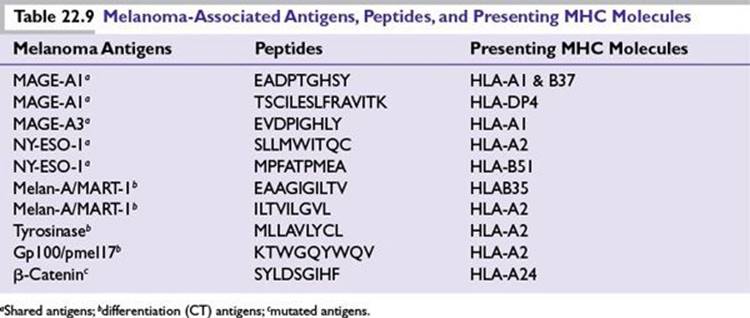
Activation of CTLs against melanoma requires two signals:
■Signal 1 (CD8+ CTL priming): The melanoma antigen (peptide) is presented either by the tumor cell or by the APCs at their MHC class I or MHC class II molecules, respectively, to the T-cell receptor of CD8+ CTL or CD4+ T cells.
■Signal 2 (CD8+ CTL activation and proliferation): The antigen-primed CD8+ CTLs express CD28 molecules on their surface for engagement with costimulatory molecules B7.1 (CD80) and B7.2 (CD86) on the APCs resulting in CD8+ CTL activation and proliferation.
Activated CD8+ CTLs kill the tumor cells directly and by the elaboration of secreted cytokines such as TNF-α, IFNγ, granulocyte-macrophage colony stimulating factor (GMCSF), and IL-2 all helping shape the tumor microenvironment.
Immune-Based Treatment Strategies of Metastatic Melanoma: Two Approaches
■Specific immunity: Evoked by subcutaneous or intradermal administration of one or more (monovalent or polyvalent) melanoma antigens as a tumor vaccine either directly or after being pulsed on to monocyte-derived APCs (dendritic cell vaccine). Adjuvants are intended to enhance the immune response and are either premixed with vaccine or preapplied to the skin at the site of the vaccine.
■Nonspecific immunity: Administration of biologic agents such as IL-2 and/or –IFN-α enhances antitumor immunity by activation and expansion of preexisting circulating melanoma antigen-specific CD8+ CTL precursors as well as NK cells (LAKs).
Tumor vaccines generate antimelanoma immune response. Based on our ability to successfully elicit melanoma-specific T-cell immune responses in mice and armed with a variety of tumor vaccines, a large number of phase 1, 2, and 3 clinical trials were completed. Although a proof of concept was demonstrated, generation of melanoma-specific T-cell activity did not consistently correlate with meaningful patient responses.
Failure to Generate Effective Antimelanoma Immunity in Patients: Immune Regulation Laboratory research showed that activated CD8+ CTL response to a tumor antigen (lysate or a vaccine) is highly regulated or held in check by a number of processes so as to prevent uncontrolled inflammation and body injury. These regulatory processes are due to factors intrinsic or extrinsic to the CD8+ CTLs.
■Intrinsic mechanisms that inhibit activated CD8+ CTLs:
•CTL antigen 4: Activated T lymphocytes are downregulated by their own expression of an inhibitory molecule called CTL antigen 4. This molecule has higher affinity and successfully competes with CD28 for binding to costimulatory molecules CD80 and CD86 on APCs inhibiting its activity.
•PD-1: Tumor expression of PD-1 ligand evokes CTL expression of PD-1 molecules. Interaction between these molecules leads to exhaustion and premature programmed cell death of activated CD8+ CTLs abrogating its activity.
■Extrinsic mechanisms that regulate activated CD8+ CTLs:
•Regulatory T cells: CD8+ CTL responses are regulated by naturally occurring CD4+CD24+ T cells (nTreg) and those derived from CD4+ T cells that acquire inhibitory properties upon interaction with tumor antigens to negate CTL activity (induced Treg). The regulation of CD8+ CTLs by CD4 T cells is mediated by expression of a transcription factor foxP3.
•Tumor factors: Include tumor secretion of inhibitory cytokines and molecules that negatively influence antimelanoma T-cell responses.
•Downregulation of MHC class I molecules on both tumor cells and the APCs compromising effective antigen presentation to the T cells.
New FDA-Approved Immune-Based Therapy of Metastatic Melanoma Recognition that CTLA-4 antigen is the key inhibitory molecule expressed by antigen-activated CD8+ CTLs orchestrating downregulation of antitumor immunity led to successful targeting of this molecule for blockade to regain CD8+ CTL activity.
Ipilimumab is an IGG1 monoclonal antibody designed to block inhibitory CTLA-4 antigen on activated CD8+ CTLs that fulfilled its promise by effectively blocking the intended target. This strategy showed promising antitumor activity in preclinical studies in mice bearing melanoma leading to successful human studies.
Two large randomized phase 3 clinical trials reported survival benefit of anti-CTLA-4 antibody ipilimumab in patients with previously treated or treatment naïve unresectable stage III and IV melanoma when compared with a glycoprotein 100 (gp100) peptide vaccine (Hodi et al. 2010) or dacarbazine (Robert et al.), respectively. In March 2011, this agent was approved by the U.S. FDA for the treatment of metastatic melanoma administered as an infusion at 3 mg/kg given every 3 weeks for four doses.
■Important facts about ipilimumab treatment of patients with metastatic melanoma:
•The objective response is between 10% and 16% with a disease control rate (the proportion of patients with a partial or complete response or stable disease) of about 25% to 35%.
•Responses are seen in previously untreated or treated patients including those with high-risk visceral metastasis and elevated serum levels of lactate dehydrogenase.
•The onset of clinical benefit is slow and mediated by antigen-specific tumor infiltrating CD8+ CTLs consistent with the proposed mechanism.
•Responses are sometimes preceded by transient tumor progression followed by continued tumor shrinkage beyond week 24 and lasting beyond 2 years.
•In order to capture response patterns unique to immune therapy, immune-related response criteria (IRRC) are defined.
•Reinduction therapy with ipilimumab at the time of disease progression can result in further benefit in a significant proportion of patients (reinduction is currently not FDA approved).
•The effect on overall survival is independent of age, sex, baseline serum lactate dehydrogenase levels, metastasis stage, and previous treatment with IL-2 therapy.
•The treatment is associated with autoimmune side effects due to cross-reactivity of antigen-specific CD8+ CTLs with normal tissues that often share these antigens. Such manifestations are referred to as immune-related adverse effects (irAEs).
•irAEs may occur in up to 60% of patients, but high grades (3 and 4) occur in up to 10% to 15% cases.
•The most common irAEs involve skin, gastrointestinal tract, liver, and endocrine organs although any organ can be affected requiring careful patient evaluation.
•Clinical presentations include skin rashes of varying grades, nausea, abdominal pain, and diarrhea, or in severe cases colitis symptoms such as abdominal distension, blood in stools, and symptoms of intestinal obstruction or perforation.
•Early diagnosis and prompt institution of immune-suppressive therapy with steroids or other agents are key to managing irAEs with slow taper over 3 to 4 weeks.
•Responses and improved outcomes are sometimes more likely to occur in those who develop irAEs. Use of steroids to treat irAEs did not seem to decrease antitumor effects.
•Majority of the irAEs usually occur about 3 to 4 weeks after the first ipilimumab infusion although in some cases it may occur earlier or later than this period. Skin manifestations of irAEs usually occur earlier and are first to appear followed by symptoms of toxicity in other organ system such as colon (colitis), liver (hepatitis) and endocrine organs (endocrinopathy) in that order (although any organ can be affected). The median time to resolution of severe irAEs of grade 2, 3, or 4 after initiation of immune-suppressive therapy is about 6.3 weeks.
•Ipilimumab can safely be administered along with chemotherapeutic agents, vaccines, or biologic agents.
Targeted Therapy of Melanoma
■Targeted therapy of melanoma is based upon a precise understanding of the functional cellular genetic machinery generating critical signaling pathways for cellular growth signals from outside of the cells to the nucleus leading to transcription of key genes critical for cellular homeostasis controlling proliferation, differentiation, and cell death.
■The mitogen-activated protein (MAP) kinase pathway that consists of Ras/Raf/MEK/ERK signaling is a key signal cascade driving cell-cycle proliferation, differentiation, and survival.
■Mutations of the genes serving this pathway promote uncontrolled cell proliferation and increased survival leading to malignant progression of cancer.
■B-Raf is a serine/threonine kinase that occupies a central place in the MAP kinase pathway, mutations of which serve as a valuable therapeutic target for melanoma.
■Activating mutations in B-Raf (BRAF) were first described to occur in about 40% to 60% of cutaneous melanoma in 2002.
■About 90% of mutations in BRAF result in the substitution of glutamic acid for valine at codon 600 (BRAF V600E). Other BRAF mutations include V600K and V600D/V600R variants.
■Vemurafenib is a first-in-class, oral small molecule kinase inhibitor that selectively targets cells harboring BRAF mutations and recently been shown to have high response rates (50%), benefiting about 80% patients and improving survival compared to dacarbazine (Chapman et al. NEJM).
■Vemurafenib is now FDA approved for patients with metastatic or unresectable melanoma in the dose of 960 mg administered orally twice a day until disease progression or intolerance.
■Important facts about BRAF kinase inhibitor treatment of metastatic melanoma:
•The survival benefit of vemurafenib was observed in each prespecified subgroup according to age, sex, performance status, tumor stage, serum levels of lactate dehydrogenase, and geographic region.
•Unique toxicities include the development of squamous cell carcinomas in approximately 25% of patients along with photosensitivity and muscle pains. Other side effects include arthralgia, pruritis, fatigue, alopecia, diarrhea, and nausea.
•About 38% of patients taking vemurafenib required dose modifications due to drug toxicity.
•Acquired drug resistance to BRAF inhibitor agent frequently leads to treatment failures and is linked to upregulation of platelet-derived growth factor receptor beta (PDGFRβ) or mutated NRAS gene.
•A second selective BRAF inhibitor dabrafenib has shown survival benefit in patients with metastatic melanoma carrying BRAF mutations. Of significance, the clinical benefit was extended to melanoma patients with brain metastasis of melanoma.
•MEK is another therapeutic target in the MAPK pathway downstream of BRAF. Trametinib is an oral selective MEK inhibitor that improved progression-free survival and overall survival in patients with metastatic melanoma harboring BRAF mutation in a phase 3 randomized clinical trial.
•Of interest, combining a MEK inhibitor (downstream of B-RAF in the MAP kinase pathway) with a B-RAF inhibitor may improve efficacy and at the same time reduce skin toxicity as reported in a recent randomized clinical trial.
Uveal Choroidal Melanoma
Uveal choroidal melanoma is the most common primary malignancy of the eye.
■Estimated incidence in the United States is six to seven cases per 1 million people.
■Depth and diameter determine the treatment indication and prognosis (Table 22.10).
■Benign choroidal nevi are up to 5 mm and 1 mm in diameter and depth, respectively.
■Monosomy of chromosome 3 is a common cytogenetic abnormality and confers poor disease-free survival and high risk of death from melanoma.
■Other cytogenetic abnormalities involve chromosomes 1, 6, and 8.
■The most common site of metastasis is the liver, although in later stages the tumor can spread to other sites such as the lungs, bones, and skin.
Management of Uveal Choroidal Melanoma
■Local ablative treatment such as brachytherapy (iodine-125 plaque therapy), photoradiation, cryotherapy, and ultrasonic hyperthermia.
■Surgical treatments that include local resection, or enucleation of the eye.
■Systemic chemotherapy or biologic therapy is ineffective in metastatic uveal melanoma.
■Experimental therapies for liver metastasis include in situ ablative therapies such as radiofrequency ablation and hyperthermic isolated perfusion using melphalan.

A recent randomized trial evaluated the use of liver chemosaturation with melphalan using a specialized approach of isolating the liver using a system of catheters (PHP, percutaneous hepatic perfusion) in patients with melanoma (mostly uveal) metastasis to the liver. The trial showed high response rates and improved liver-specific progression-free survival.
Follow-up of patients with uveal choroidal melanoma after local treatment includes close surveillance for liver metastasis with liver function tests and imaging studies of the liver that include sonography every 6 months in the first 5 years for early diagnosis of liver metastasis.
Indications for Enucleation of the Eye
■Tumor growing in a blind eye
■Melanoma involving more than half of the iris
■Tumor involving the anterior chamber of the eye or extraocular extension
■Failure of previous local therapy
NONMELANOMA SKIN CANCER
There are two major types of nonmelanoma skin cancers: basal cell carcinoma (BCC) and squamous cell carcinoma. Together they account for nearly 1 million cases in the United States per year. The immune system plays an important role in the pathogenesis of nonmelanoma skin cancers, as demonstrated clinically by their increased incidence in patients with immune-suppressed states, such as the aging population and transplant recipients. Histologically, the regressing nonmelanoma skin cancers show infiltration of the tumor by activated T cells and cytokines such as IFN-α, TNF-β, and IL-2.
Basal Cell Carcinoma
■BCCs are keratinocyte tumors most commonly diagnosed in people of European ancestry.
■Exposure to ultraviolet rays is the most important etiologic factor. Other causative factors include exposure to ionizing radiation and arsenic.
■BCC is the commonest cancer in the US white population over 50 years of age, accounting for 75% of 1 million new cases of nonmelanoma skin cancers.
■Usual location of BCC is the skin of the head and neck region (sun-exposed area).
■BCC is highly cured by surgery on most of the occasions and despite its high rate of occurrence, the death rate is extraordinarily low.
■When locally advanced or metastatic (rare occasions), local invasion can lead to tissue destruction that makes surgical treatment very difficult and outcomes very poor.
Clinical Presentations of BCC
■Typical presentation of BCC is a shiny pink translucent papule with telangiectasia.
■Nodular variant of BCC presents with central depression and rolled margins that may bleed from trauma and include pigmented types with brown to black pigment.
■Sclerosing or morphea-type BCC is yellowish, infiltrative tumor with indistinct borders often remaining undiagnosed for a long time. Management includes Mohs surgery.
■Hyperkeratotic-type carcinoma (less common) usually involves the head and neck area as multicentric ulcer and scar tissues or giant exophytic type or cystic type presenting as a blue–gray nodule on the face or as a sessile growth on the lower trunk.
BCC as a Heritable Disorder
■A rare familial presentation of BCC is called basal cell nevus syndrome (BCNS) also known as Gorlin syndrome characterized by high incidence of BCCs and medulloblastomas.
■The autosomal dominant inheritance of this syndrome is due to uncontrolled activation of the Hedgehog (Hh) signaling pathway.
■The genetic abnormality underlying this condition is linked to mutation of a gene called patched 1 (PTCH1) identified as a tumor suppressor gene and mapped to human chromosome 9q22.
■The mutations of genes PTCH1 and TP53 critical to BCC carcinogenesis are believed to be produced by exposure to UV radiation, elucidating the essential role of UV exposure in the causation of BCC.
Hedgehog Signaling Pathway and Targeted Therapy of BCC
■Hh signaling is a pivotal abnormality in all the BCCs resulting in uncontrolled proliferation of the basal cells and carcinogenesis.
■The Hh pathway is activated after binding of Hh ligand to the PATCHED 1 protein (PTCH1)—a tumor suppressor gene present on target cells.
■In the absence of excessive Hh ligand, the tumor-suppressive gene PTCH1 inhibits a downstream protein called smoothened (SMO) and prevents its translocation into the cilium.
■Binding of the Hh ligand to PTCH1 inhibits its protective activity of inhibiting SMO allowing uninhibited SMO to translocate to the primary cilium.
■Downstream effects of SMO activity lead to increased transcription factors GLI1 and GLI2, both of which result in transcription of gene important in proliferation and cell survival.
■Activity of SMO also leads to increasing transcription of Hh ligands GLI1 and GLI2 as well as decreasing PTCH1 expression constitutively activating the Hh signaling.
Approximately 90% of sporadic BCCs have at least one allele of PTCH1 mutated, while about 10% of BCCs have mutations in the downstream SMO protein that makes SMO resistant to inhibition by PTCH1. Targeted therapy of BCC is directed toward identifying agents that inhibit Hh signaling.
Plant alkaloid cyclopamin is the first well-studied Hh inhibitor (HHI) that when applied locally caused regression of BCC. Cyclopamin is a competitive inhibitor of SMO signaling, binding directly to the protein PTCH1 or SMO.
■Vismodegib is a first-in-class, small molecule inhibitor of SMO that is FDA approved for metastatic or locally advanced BCC at a dose of 150 mg given orally daily until disease progression or intolerable toxicity.
■A phase 1 study of vismodegib involving 33 patients with advanced BCC showed a 58% confirmed response rate and a median duration of response of 12.8 months.
■In a phase 2 study containing 33 patients with metastatic BCC and 63 patients with locally advanced basal cell carcinoma, vismodegib produced a response rate of 30% and 43%, respectively, with the median duration of response being 7.6 months.
■Common toxicity of vismodegib includes alopecia, dysgeusia (taste disturbance), muscle spasms, fatigue, and weight loss. Serious adverse events were reported in 25% of patients resulting in seven deaths.
Squamous Cell Carcinoma
■Usually found as single or multiple lesions in elderly white men with sun-damaged skin.
■Common sites include back of the hand, forearm, face, and neck.
■Presents as a firm, indurated, expanding nodule, often at the site of actinic keratosis.
■The nodule may be ulcerated, and regional lymph nodes may be enlarged.
Squamous Cell Carcinoma of a Mucocutaneous Site
■Elderly men with chronic history of smoking, alcohol use, or chewing tobacco or betel nut.
■Common sites include mouth and lower lip.
■Lesions usually start as erosion or a nodule that ulcerates.
■Other sites of origin include sole of the foot (verrucous form) and male genitalia related to human papillomavirus in underlying condylomata of Buschke-Lowenstein tumor.
Diagnosis of Nonmelanoma Skin Cancer
Detailed clinical history should include duration of the lesion, symptoms such as pain or itching, and recent changes of the surface in addition to the following:
■Chronic sun exposure and recreational and occupational history
■Radiation and arsenic exposure, chronic ulcer/burn scar, or osteomyelitis
■Ethnic background and type of skin
A complete skin examination includes
■Examination of scalp, ears, palms, soles, interdigital areas, and mucous membranes
■Evaluation of the extent of sun damage to skin (i.e., solar elastosis, scaling, erythema, telangiectasia, and solar lentigines)
■Assessment of the locoregional lymph nodes and distant metastases
An excisional or incisional tumor biopsy in small or large tumor, respectively, is obtained for histologic diagnosis. A shave biopsy with a scalpel may be used in noduloulcerative, cystic, or superficial type.
Treatment of Nonmelanoma Skin Cancer
Complete surgical resection with negative margins of at least 4 to 6 mm is recommended with lymph node resection if enlarged. Plastic surgery may be needed to close the defects produced by excision of the tumor.
Mohs Surgery
Mohs surgery allows excision of the tumor until the negative margins are achieved. It includes micrographic surgery that is guided by examination of a frozen section to ascertain complete resection.
Imiquimod is an FDA-approved agent for treatment of superficial BCC when used in cream form. The drug works via toll-like receptor agonistic activity and causes stimulation of innate and adaptive immune systems. Common side effects include local skin rashes, burning sensation, erythema, edema, induration, erosion, and pruritus.
Radiation Therapy
X-rays delivered at a total dose of 2,000 to 3,000 cGy penetrate up to 2 to 5 mm, where most of the basal cell and squamous cell carcinomas infiltrate. The total dose is divided into multiple smaller doses, usually over 3 to 4 weeks, to reduce side effects.
MERKEL CELL CARCINOMA
Merkel cell carcinoma occurs due to the neoplastic proliferation of the Merkel cells located in the basal layer of the epidermis and hair follicles. These cells, which originate from the neural crest, are a member of the amine precursor uptake and decarboxylation cell system (APUD). Merkel cells are important for tactile sensations in lower animals and they function as a mechanoreceptor in humans.
Characteristics of Merkel Cell Tumors
■Occur in the elderly population with chronically sun-damaged areas of skin.
■Common sites include head and neck skin; less common sites are extremities and genitals.
■Present as 0.5 to 1 cm intracutaneous, firm, bluish-purple, nontender nodule.
■Histologically, a small round cell tumor containing neurosecretory cytoplasmic granules that may look similar to small cell carcinoma, melanoma, Ewing sarcoma, and lymphoma.
■Tumor cells stain positive for neuron-specific enolase and anticytokeratin antibody CAM 5.2.
■Recent identification of polyomaviral DNA integration in Merkel tumor cells indicates implication of polyomavirus in the pathogenesis of this tumor. Higher incidence of this tumor in the aging population may have clinical relevance to the aging immune system.
■Early spread occurs to locoregional lymph nodes and hematogenously to the distant sites.
Management of Merkel Cell Tumors
Management of Merkel cell tumors includes complete primary surgical excision with lymph node assessment by the sentinel lymph node procedure and lymph node dissection if necessary, as in cutaneous melanoma. Adjuvant radiation treatment is recommended in patients at high risk of local recurrence due to incomplete resection or larger tumor size (2 cm or more).
A metastatic Merkel cell tumor is managed with systemic chemotherapy. Effective chemotherapeutic agents include cisplatin, etoposide, adriamycin, cyclophosphamide, vincristine, and irinotecan. Although the tumor is responsive to chemotherapy, high recurrence rates lead to uniformly poor outcomes.
RARE TUMORS ARISING FROM THE SKIN
Rarely, skin appendageal tumors arise in the following locations:
■Hair follicle
■Arrector pili muscle
■Apocrine sweat gland
■Sebaceous gland
Most of these tumors are benign, and carcinomas are rare. The treatment principle of carcinomas includes complete surgical excision and lymph node assessment as in melanoma.
Dermatofibrosarcoma Protuberans
A rare fibrohistiocytic tumor arising in the skin and subcutaneous tissue of intermediate malignant potential demonstrating slow growth and consistent cytogenetic abnormality t(17;22) in more than 90% of patients, with the following characteristics:
■Common tumor sites include trunk and extremities.
■Frequent local recurrences occur after surgical resection.
■Low propensity to distant metastasis.
The translocation t(17;22) between chromosomes 17 and 22 places platelet derived growth factor -β (PDGF-β) under the control of COL1A1, resulting in upregulation, expression, and activation of tyrosine kinase PDGFβ.
Recent reports suggest high response rates to imatinib mesylate. This potent and specific inhibitor of PDGFRβ is effectively used in neoadjuvant settings and in patients with recurrent disease.
REVIEW QUESTIONS
1.You are seeing a 44-year-old patient with newly diagnosed cutaneous melanoma of the upper extremity. The biopsy of the tumor shows the tumor to be 0.8 mm deep, Clark level III, ulceration absent, mitosis 0/mm2. A chest x-ray is negative for metastasis and the serum level of lactate dehydrogenase is normal. Which of the following treatment recommendations is correct?
A.Wide resection of the tumor with wide negative margins
B.Wide resection of the tumor with negative margins followed by sentinel lymph node biopsy
C.High-dose interferon treatment
D.No further treatment
2.The independent prognostic factor(s) for melanoma is/are
A.Clark level
B.Tumor infiltrating lymphocytes, regression, and solar elastosis
C.Depth of invasion in the dermis, presence of ulceration, mitosis, and older age
D.Perineural invasion and vascular invasion
3.A 67-year-old Caucasian male is referred to you with a diagnosis of metastatic melanoma to the lung. He was found to have a 1.5 cm nodule on an annual chest x-ray by his primary care provider who confirmed it to be a metastasis from previously resected cutaneous melanoma of the upper back. The patient who has mild diabetes mellitus and well-controlled hypertension is otherwise asymptomatic. A whole-body PET study showed a PET avid lesion in the right upper lobe measuring about 1.5 cm ×1.5 cm with no other abnormalities. A brain MRI is negative for metastasis and a peripheral blood showed normal level of serum lactate dehydrogenase. What is the best option for treatment in this patient?
A.Discuss with him treatment with high-dose IL-2.
B.Discuss with him treatment with biochemotherapy.
C.Arrange a repeat chest CT scan within 3 months.
D.Refer him to a thoracic surgeon for resection of the lung metastasis.
4.A 35-year-old Caucasian woman is diagnosed with metastatic melanoma to the liver from a previously resected cutaneous melanoma of the right lower chest wall 3 years ago. The patient has lost about 20 pounds of weight in the last 4 weeks and is increasingly short of breath. On examination, the patient sits on a wheel chair with pain in the right upper abdomen. Clinical examination reveals an ill-looking individual with mild anemia and an enlarged liver of 7 cm below the costal margins. Whole-body CT scan showed multiple enhancing lesions in both the lobes of liver with right lower pleural effusion. The serum level of lactate dehydrogenase is 450 units/L (upper limit of normal is 175 units/L). A brain MRI is negative for metastasis, while the tumor tested for mutational analysis confirmed the presence of BRAF mutation V600E. What is the best treatment option for this patient?
A.High-dose IL-2
B.Intravenous administration of dacarbazine
C.Ipilimumab treatment
D.Start oral doses of vemurafenib at 960 mg twice a day
5.Which of the following skin cancers is associated with polyoma virus?
A.BCC
B.Squamous cell carcinoma
C.Merkel cell tumor
D.Cutaneous melanoma
Suggested Readings
1.Balch CM, Gershenwald JE, Soong S-J, et al. Final version of 2009 AJCC melanoma staging and classification. J Clin Oncol. 2009;27:6199-6206.
2.Breslow A. Thickness, cross-sectional areas and depth of invasion in the prognosis of cutaneous melanoma. Ann Surg. 1970;172:902.
3.Chapman PB, Hauschild A, Robert C, et al. Improve survival with Vemurafenib in melanoma with BRAF V600E mutation. N Engl J Med. 2011;364:2507-2516.
4.Clark WH Jr., From L, Bernadino EA, et al. The histogenesis and biologic behavior of primary human malignant melanomas of skin. Cancer Res. 1969;29:705.
5.Davies H, Bignell GR, Cox C, et al. Mutations of the BRAF gene in human cancer. Nature. 2002;417:949-954.
6.Feng H, Shuda M, Chang Y, Moore PS. Clonal integration of a polyomavirus in human Merkel cell carcinoma. Science. 2008;319:1096-1100.
7.Ghussen F, Nage IK, Groth W, et al. Hyperthermic perfusion with chemotherapy and melanoma of the extremities. World J Surg. 1989;13:598.
8.Hodi FS, O’Day SJ, McDermott DF, et al. Improved survival with ipilimumab in patients with metastatic melanoma. N Engl J Med. 2010;363:711-723.
9.Robert C, Thomas L, Bondarenko I, et al. Ipilimumab plus dacarbazine for previously untreated metastatic melanoma. N Engl J Med. 2011; 364:2517-2526.
10.Leachman SA, Carucci J, Kohlmann W, et al. Selection criteria for genetic assessment of patients with familial melanoma. J Am Acad Dermatol. 2009;61:677.el-1-14.
11.Legha SS, Ring S, Bedikian A, et al. Treatment of metastatic melanoma with combined chemotherapy containing cisplatin, vinblastine, dacarbazine (CVD) and biotherapy using interleukin 2 and interferon alpha. Ann Oncol. 1996;7:827-835.
12.Low JA, de Sauvage FJ. Clinical experience with Hedgehog pathway inhibitors. J Clin Oncol. 2010;28:5321-5326.
13.Marquette A, Bagot M, Bensussan A, Dumaz N. Recent discoveries in the genetics of melanoma and their therapeutic implications. Arch Immunol Ther Exp. 2007;55:363-372.
14.Morton DL, Thompson JF, Cochran AJ, et al. Sentinel-node biopsy or nodal observation in melanoma. N Engl J Med. 2006;355:1307-1317.
15.Mukherji B, Chakraborty NG, Sporn JR, et al. Induction of peptide antigen reactive cytolytic T cells following immunization with MAGE-1 peptide pulsed autologous antigen presenting cells. Proc Natl Acad Sci U S A. 1995;92:8078-8082.
16.O’Day SJ, Hamid O, Urba WJ. Targeting cytotoxic T-lymphocyte antigen-4 (CTLA-4): a novel strategy for the treatment of melanoma and other malignancies. Cancer. 2007;110:2614-2627.
17.Rosenberg SA, Yang JC, Topalian SL, et al. Treatment of 283 consecutive patients with metastatic melanoma or renal cell cancer using high-dose bolus interleukin 2. JAMA. 1994;271:907-913.
18.Takahira T, Oda Y, Tamiya S, et al. Detection of COL1A1-PDGFB fusion transcripts and PDGFB/PDGFRB mRNA expression in dermatofibrosarcoma protuberans. Mod Pathol. 2007;20:668-675.
19.Santa Cruz DJ, Hurt MA. Neoplasms of skin [chapter 2] In: Sternberg SS. Diagnostic surgical pathology 2nd ed. Vol.1. New York: Raven Press; 1994: 57-102.
20.Weber J. Review: anti-CTLA-4 antibody ipilimumab: case studies of clinical response and immune-related adverse events. Oncologist. 2007;12:864-872.
21.Wolchok JD, Hoos A, O’Day S, et al. Guidelines for the evaluation of immune therapy activity in solid tumors: immune-related response criteria. Clin Cancer Res. 2009;15(23):7412-7420.